Abstract
Considering the global impact of the coronavirus disease 2019 (COVID-19) pandemic, generating suitable experimental models is imperative. For pre-clinical studies, researchers require animal models displaying pathological features similar to those observed in patients; therefore, establishing animal models for COVID-19 is crucial. The golden Syrian hamster model mimics conditions observed in humans with mild severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. However, a golden Syrian hamster model of severe infection has not been reported. J2N-k hamsters are utilized as a cardiomyopathy model; therefore, we used cardiomyopathic J2N-k hamsters showing conditions similar to those of severe COVID-19 complicated with cardiovascular diseases, as patients with cardiovascular diseases exhibit a higher risk of morbidity and mortality due to COVID-19 than patients without cardiovascular diseases. Unlike that in golden Syrian hamsters, SARS-CoV-2 infection was lethal in J2N-k hamsters, with a median lethal dose of 104.75 plaque-forming units for the S clade of SARS-CoV-2 (A, GenBank: MW466791.1). High viral titers and viral genomes were detected in the lungs of J2N-k and golden Syrian hamster models harvested 3 days after infection. Pathological features of SARS-CoV-2-associated lung injury were observed in both models. The J2N-k hamster model can aid in developing vaccines or therapeutics against COVID-19.
Abbreviations: SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; COVID-19, coronavirus disease 2019; CVD, cardiovascular disease; dpi, days post-infection; PFU, plaque-forming unit; LD50, median lethal dose; TCID50, median tissue culture infectious dose
Keywords: SARS-CoV-2, Golden Syrian hamster, J2N-k hamster, COVID-19
The golden Syrian hamster model was established in 2005 for studying severe acute respiratory syndrome (SARS) (Roberts et al., 2005). Recently, this model was deemed suitable for studying SARS-coronavirus 2 (SARS-CoV-2; family Coronaviridae, order Nidovirales, subgenus Sarbecovirus), the etiological agent for the ongoing coronavirus disease 2019 (COVID-19) pandemic (Chan et al., 2020; Sia et al., 2020). The human and hamster angiotensin-converting enzyme 2 (ACE2) sequences share a high similarity (82.8 %) (Chan et al., 2020), and hamsters infected with SARS-CoV-2 exhibit loss in body weight similar to that observed in patients with SARS-CoV-2 infection. So far, lethal outcomes have not been observed in the golden Syrian hamster model following SARS-CoV-2 infection. Thus, this model effectively mimics the conditions of patients experiencing mild COVID-19 symptoms. However, lethal outcomes have been reported in dwarf hamster (Trimpert et al., 2020), STAT-knockout hamster (Boudewijns et al., 2020), and human ACE2-transgenic mouse models (Winkler et al., 2020) with SARS-CoV-2 infection.
Higher risk of morbidity and mortality (10 %–15 %) due to SARS-CoV-2 infection has been reported in patients with cardiovascular diseases (CVDs) than in those without cardiovascular diseases (˜5%) (Clerkin et al., 2020; Zheng et al., 2020). Therefore, an animal model for severe SARS-CoV-2 infection complicated with CVDs is urgently required. As J2N-k hamsters have previously been utilized as a cardiomyopathic animal model harboring abnormalities in the ADP/ATP carrier protein and a defective delta-sarcoglycan encoding gene (Kato et al., 1993; Mitsuhashi et al., 2003), we investigated the susceptibility of the J2N-k hamster model to SARS-CoV-2 infection in this study and compared it with that of the golden Syrian hamster model. All animal experiments were approved by the Institutional Animal Care and Use Committee (approval no. KCDC-088-20-2A) and were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals of Korea National Institute of Health. All experiments with infectious SARS-CoV-2 were performed in biosafety level 3 and animal biosafety level 3 facilities.
Seven-week-old male golden Syrian hamsters and J2N-k hamsters were purchased from Japan SLC, Inc. (Shizuoka, Japan). They were anesthetized with tiletamine/zolazepam combined with xylazine injected intraperitoneally before infection. Subsequently, serially 10-fold diluted SARS-CoV-2 was administered intranasally (from 106 to 103 plaque-forming units (PFU)/100 μL) to the hamsters (golden Syrian (n = 6) and J2N-k (n = 4) per group). The infected hamsters were monitored daily for weight loss and mortality for 14 days. The median lethal dose (LD50) was determined by assessing the number of dead and live hamsters on day 14, following methods previously described (Ramakrishnan, 2016).
To measure the viral load in the tissues of SARS-CoV-2-infected hamsters, the lung or tracheal tissues were weighed and homogenized in 1 mL of DPBS using bead-beating homogenizer. After centrifugation at 13,000 rpm for 20 min, the clarified supernatant was harvested. Aliquots of the supernatant were incubated with confluent Vero-E6 cells in 96-well plates for the median tissue culture infectious dose (TCID50) assay. Briefly, 10-fold serial dilutions of each sample were cultured for 3 days. The plates were observed for cytopathic effects. Viral titer was calculated using the Reed and Munch endpoint method and expressed as TCID50/mL of tissue. To assess SARS-CoV-2 viral genomes in the tissues, RNA extracted from the homogenized tissues (Maxwell® RSC Viral Total Nucleic Acid Purification Kit, Promega) was quantified by determining the amount of RNA copies using the RdRp-gene RT-PCR (PowerChek™ 2019-nCoV Real-time PCR Kit; Kogen Biotech, Seoul, Republic of Korea) and quantifying RdRp-gene transcripts.
Intranasal administration of 106 PFU/100 μL SARS-CoV-2 in 7-week-old male golden Syrian hamsters resulted in disease symptoms similar to those reported previously (Fig. 1 A) (Chan et al., 2020; Sia et al., 2020). A 20 % decline in body weight was noted, and recovery was observed 7 days post-infection (dpi). However, 7-week-old male J2N-k hamsters infected with 106 PFU SARS-CoV-2 exhibited 25.7 % weight loss. Additionally, lethality was observed in all infected J2N-k hamsters at 9 dpi (Fig. 1). The LD50 was calculated as 104.75 PFU for the S clade of SARS-CoV-2. In contrast, no lethality was detected in the six golden Syrian hamsters infected with SARS-CoV-2.
Fig. 1.
Changes in body weight and survival rate of golden Syrian and J2N-k hamster models. Hamsters (golden Syrian (n = 6) and J2N-k (n = 4) per group) used in the experiments were anesthetized via intraperitoneal injection of tiletamine/zolazepam and xylazine combination for the intranasal infection of SARS-CoV-2. Next, the hamsters were inoculated with a 100 μL solution containing 106 PFU of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) isolate (BetaCoV/Korea/KCDC03/2020) or 100 μL of phosphate-buffered saline (control) via the intranasal route. Body weight (A) and survival rate (B) were monitored daily for 14 days. Significant differences (p < 0.0001) were observed between the uninfected (control) and infected groups through the Mann–Whitney U test PFU, plaque-forming unit.
High viral titers and viral genomes were detected in the lungs of both animal models sacrificed 3 dpi but not after 7 dpi, as evidenced by the TCID50 assay and real-time RT-PCR assay, respectively (Fig. 2 A). However, comparable viral titers and viral genomes were detected in the lung tissues when both models were administered with different viral doses (Fig. 2B). Additionally, the lungs of both models infected with SARS-CoV-2 exhibited severe chronic active pneumonia in the pulmonary parenchyma. Furthermore, their lungs showed hyperplastic type II alveolar epithelial cells with infiltration of inflammatory cells and neutrophils (Fig. 2C).
Fig. 2.
Viral load and histopathology of golden Syrian and J2N-k hamsters infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Three golden Syrian hamsters and two J2N-k hamsters were sacrificed at 3 dpi, and an equal number of hamsters was sacrificed at 7 dpi. (A) Infectious viral load (log10TCID50/mL) and viral genome copies (log10 Genome copies/mL) detected in the lungs of both hamster models infected with 106 PFU SARS-CoV-2 at 3- and 7-days post-infection. (B) log10 TCID50/mL and log10 Genome copies/mL detected in the lungs of hamsters infected with 105 or 106 PFU of SARS-CoV-2 at 3 days post-infection. The dotted line represents the detection limit. (C) Diffuse and severe active pneumonia (inflammation) observed in the pulmonary parenchyma in both hamster models. The scale bars in the lower left regions of ×40- and ×200-magnified images are 250 μm and 50 μm, respectively. The ×200-magnified images show that the alveolar walls were markedly thickened by hyperplastic type II alveolar epithelial cells, with infiltration of inflammatory cells and neutrophils. PFU, plaque-forming unit; dpi, days post-infection; a, alveolus; b, bronchiole; v, blood vessel; PN, pulmonary parenchyma, PI, post-infection.
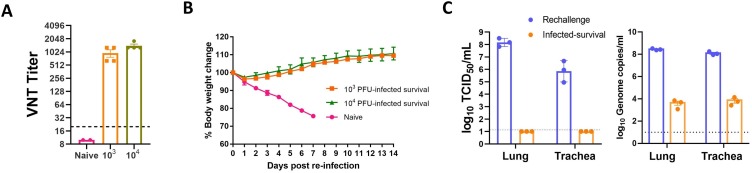
Next, the possibility of using the J2N-k hamster model for developing vaccines or therapeutic drugs against SARS-CoV-2 was considered. Seven J2N-k hamsters that survived SARS-CoV-2 infection (103 and 104 PFU) were assessed for resistance to viral re-infection. The exposed J2N-k hamsters tested positive for the presence of neutralizing antibodies prior to re-infection (Fig. 3 A). Seven J2N-k hamsters were re-infected with 106 PFU of the homologous virus on day 25 after the primary infection (Fig. 3B). The body weight and the survival rate were monitored daily, and the lung and trachea tissues were collected 3 dpi for virus titration. Six naïve J2N-k hamsters showed the presence of infection and succumbed, thus proving that survival was associated with body weight loss, as predicted. In contrast, all previously infected hamsters survived re-infection without any significant weight loss. Additionally, the surviving hamsters presented with markedly lower viral titers and viral genomes (unpaired t-test, with p-value < 0.05) in their lung and trachea tissues homogenates 3 dpi than the naïve hamsters (Fig. 3C). In conclusion, pre-exposure to SARS-CoV-2 elicited neutralizing antibodies that helped in protecting the J2N-k hamsters against lethal SARS-CoV-2 re-infection. Therefore, J2N-k hamsters are a potential model for testing vaccines and antiviral drugs to treat SARS-CoV-2 infection.
Fig. 3.
Re-infection of J2N-k hamsters with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). (A) Virus neutralizing test (VNT) titers in infected-survived hamsters evaluated at 2 days before re-infection. Bars indicate the neutralizing titers in naïve (pink, n = 6), 103 PFU virus-infected (orange, n = 4), and 104 PFU virus-infected (green, n = 3) J2N-k hamsters, respectively. (B) Seven infected-survived hamsters were re-infected with 106 PFU of the homologous virus on day 25 after the primary infection. Body weights of hamsters were monitored after re-infection for 14 days. (C) Infectious viral load (log10 TCID50/mL) and viral genome copies (log10 Genome copies/mL) detected in the lungs and trachea of hamsters re-challenged with SARS-CoV-2. A selected number of hamsters from the re-challenged group (n = 3) and from the infected-survival group, which included animals that had survived infection with 103 PFU (n = 2) and 104 PFU (n = 1) of SARS-CoV-2, were sacrificed at 3 dpi. The infected-survival group is indicated with orange bars and dots. The dotted line represents the detection limit. Blue bars and dots indicate the virus titers and viral genomes, respectively, of the previously uninfected hamsters infected with 106 PFU of SARS-CoV-2. The SARS-CoV-2 live virus titers in the lungs and trachea were not detected in the surviving hamsters re-infected with SARS-CoV-2. The remaining hamsters were maintained alive to monitor the changes in body weight until 14 dpi. All three challenged naïve hamsters were found dead at 6 or 7 dpi. PFU, plaque-forming unit.
Although this study had a few limitations, including a low number of hamsters per group, not having used female J2N-k hamsters, and performing viral titration only using samples from the lungs and trachea, the results suggested that the J2N-k hamster model could successfully simulate severe COVID-19 infection in patients with CVDs. In addition, male hamsters have been reported to be more susceptible to SARS-CoV-2 infection than females (Dhakal et al., 2021; Yuan et al., 2021). It is assumed that male J2N-k hamsters would be more suitable as SARS-CoV-2 models owing to their higher susceptibility to SARS-CoV-2 than females. Notably, other hamster models are also useful for studying SARS-CoV-2. For instance, an STAT2-knockout hamster model lacking STAT2-dependent interferon response may be used for systemic immune-compromised COVID-19 studies (Boudewijns et al., 2020). Additionally, the susceptibility of Chinese hamsters to SARS-CoV-2 is comparable to that of normal golden Syrian hamsters (Bertzbach et al., 2021). Dwarf hamsters can be utilized as models for severe COVID-19; however, factors contributing to the pathology of severe SARS-CoV-2 infection in this model remain unclear (Trimpert et al., 2020). Several strategies, including repurposing of drugs, and use of monoclonal therapeutic antibodies, anti-inflammatory agents, and vaccines have been used to treat patients with severe COVID-19 or to prevent SARS-CoV-2 infection. Therapeutic and immunization strategies for COVID-19 patients with CVDs are crucial, considering that mortality due to COVID-19 is significantly attributed to CVDs (Yi et al., 2021). To evaluate the potential efficacy of cardiovascular or antiviral drugs in these patients, the theoretical basis of pre-clinical animal models should be established before clinical treatment. This study may help guide the preclinical evaluation of treatment strategies and the effects of vaccination for severe COVID-19 in patients with CVDs. Additionally, the J2N-k hamster model could be used in studies pertaining to CVDs and SARS-CoV-2 infection and viral transmission.
Data statement
Angiotensin-converting enzyme 2 (ACE2) amino acid sequences of golden Syrian and J2N-k (originating from Bio14.6 golden hamster strain) hamsters (Mesocricetus auratus XP_005074266.1) and humans (NP_001358344.1) were used in the study. J2N-k hamsters were inbred with Bio14.6 cardiomyopathic hamsters (Bio Breeders Inc., Watertown, MA, USA) and Golden Syrian hamsters and provided by Japan SLC, Inc. (Shizuoka, Japan). According to the policy of Japan SPL, Inc., only male J2N-k hamsters can be distributed.
Authorship statement
All persons who meet authorship criteria are listed as authors, and all authors certify that they have participated sufficiently in the work to take public responsibility for the content, including participation in the concept, design, analysis, writing, or revision of the manuscript. Furthermore, each author certifies that this material or similar material has not been and will not be submitted to or published in any other publication before its appearance in the Journal of Virological Methods.
Authorship contributions
Concention and design of study: Hansaem Lee, Joo-Yeon Lee
Acquisition of data: Hansaem Lee, Tae-Young Lee, Pyeonghwa Jeon, Nayoung Kim
Resources: Jun-Won Kim, Jeong-Sun Yang
Analysis and interpretation of data: Hansaem Lee, Tae-Young Lee, Jeong-Sun Yang, Kyung-Chang Kim, Joo-Yeon Lee
Statistical analysis: Nayoung Kim
Drafting the manuscript: Hansaem Lee, Tae-Young Lee
Revising the manuscript: Hansaem Lee, Tae-Young Lee, Joo-Yeon Lee
Funding acquisition: Kyung-Chang Kim, Joo-Yeon Lee
Funding
This study was funded by the National Institute of Health, Korea Centers for Diseases Control and Prevention [grant numbers 2020-NI-039-00, 4861-312-210-11].
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Bertzbach L.D., Vladimirova D., Dietert K., Abdelgawad A., Gruber A.D., Osterrieder N., Trimpert J. SARS-CoV-2 infection of Chinese hamsters (Cricetulus griseus) reproduces COVID-19 pneumonia in a well-established small animal model. Transbound. Emerg. Dis. 2021;68:1075–1079. doi: 10.1111/tbed.13837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boudewijns R., Thibaut H.J., Kaptein S.J.F., Li R., Vergote V., Seldeslachts L., Van Weyenbergh J., De Keyzer C., Bervoets L., Sharma S., Liesenborghs L., Ma J., Jansen S., Van Looveren D., Vercruysse T., Wang X., Jochmans D., Martens E., Roose K., De Vlieger D., Schepens B., Van Buyten T., Jacobs S., Liu Y., Martí-Carreras J., Vanmechelen B., Wawina-Bokalanga T., Delang L., Rocha-Pereira J., Coelmont L., Chiu W., Leyssen P., Heylen E., Schols D., Wang L., Close L., Matthijnssens J., Van Ranst M., Compernolle V., Schramm G., Van Laere K., Saelens X., Callewaert N., Opdenakker G., Maes P., Weynand B., Cawthorne C., Vande Velde G., Wang Z., Neyts J., Dallmeier K. STAT2 signaling restricts viral dissemination but drives severe pneumonia in SARS-CoV-2 infected hamsters. Nat. Commun. 2020;11:5838. doi: 10.1038/s41467-020-19684-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan J.F.W., Zhang A.J., Yuan S., Poon V.K.M., Chan C.C.C., Lee A.C.Y., Chan W.M., Fan Z., Tsoi H.W., Wen L., Liang R., Cao J., Chen Y., Tang K., Luo C., Cai J.P., Kok K.H., Chu H., Chan K.H., Sridhar S., Chen Z., Chen H., To K.K.W., Yuen K.Y. Simulation of the clinical and pathological manifestations of coronavirus disease 2019 (COVID-19) in a golden Syrian hamster model: implications for disease pathogenesis and transmissibility. Clin. Infect. Dis. 2020;71:2428–2446. doi: 10.1093/cid/ciaa325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clerkin K.J., Fried J.A., Raikhelkar J., Sayer G., Griffin J.M., Masoumi A., Jain S.S., Burkhoff D., Kumaraiah D., Rabbani L., Schwartz A., Uriel N. COVID-19 and cardiovascular disease. Circulation. 2020;141:1648–1655. doi: 10.1161/CIRCULATIONAHA.120.046941. [DOI] [PubMed] [Google Scholar]
- Dhakal S., Ruiz-Bedoya C.A., Zhou R., Creisher P.S., Villano J.S., Littlefield K., Ruelas C.J., Marinho P., Jedlicka A.E., Ordonez A.A., Bahr M., Majewska N., Betenbaugh M.J., Flavahan K., Mueller A.R.L., Looney M.M., Quijada D., Mota F., Beck S.E., Brockhurst J., Braxton A.M., Castell N., Stover M., D’Alessio F.R., Metcalf Pate K.A., Karakousis P.C., Mankowski J.L., Pekosz A., Jain S.K., Klein S.L., Johns Hopkins COVID-19 Hamster Study Group Sex differences in lung imaging and SARS-CoV-2 antibody responses in a COVID-19 golden Syrian Hamster model. mBio. 2021;12 doi: 10.1128/mBio.00974-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato M., Yang J., Iwai T., Tanamura A., Arino T., Kawashima O., Takeda N. Abnormalities of ADP/ATP carrier protein in J-2-N cardiomyopathic hamsters. Mol. Cell. Biochem. 1993;119:89–94. doi: 10.1007/bf00926858. [DOI] [PubMed] [Google Scholar]
- Mitsuhashi S., Saito N., Watano K., Igarashi K., Tagami S., Shima H., Kikuchi K. Defect of delta-sarcoglycan gene is responsible for development of dilated cardiomyopathy of a novel hamster strain, J2N-k: calcineurin/PP2B activity in the heart of J2N-k Hamster. J. Biochem. 2003;134:269–276. doi: 10.1093/jb/mvg140. [DOI] [PubMed] [Google Scholar]
- Ramakrishnan M.A. Determination of 50% endpoint titer using a simple formula. World J. Virol. 2016;5:85–86. doi: 10.5501/wjv.v5.i2.85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts A., Vogel L., Guarner J., Hayes N., Murphy B., Zaki S., Subbarao K. Severe acute respiratory syndrome coronavirus infection of golden Syrian hamsters. J. Virol. 2005;79:503–511. doi: 10.1128/jvi.79.1.503-511.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sia S.F., Yan L.M., Chin A.W.H., Fung K., Choy K.T., Wong A.Y.L., Kaewpreedee P., Perera R.A.P.M., Poon L.L.M., Nicholls J.M., Peiris M., Yen H.L. Pathogenesis and transmission of SARS-CoV-2 in golden Syrian hamsters. Nature. 2020;583:834–838. doi: 10.1038/s41586-020-2342-2345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trimpert J., Vladimirova D., Dietert K., Abdelgawad A., Kunec D., Dökel S., Voss A., Gruber A.D., Bertzbach L.D., Osterrieder N. The Roborovski dwarf hamster is a highly susceptible model for a rapid and fatal course of SARS-CoV-2 infection. Cell Rep. 2020;33 doi: 10.1016/j.celrep.2020.108488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler E.S., Bailey A.L., Kafai N.M., Nair S., McCune B.T., Yu J., Fox J.M., Chen R.E., Earnest J.T., Keeler S.P., Ritter J.H., Kang L.-I., Dort S., Robichaud A., Head R., Holtzman M.J., Diamond M.S. SARS-CoV-2 infection of human ACE2-transgenic mice causes severe lung inflammation and impaired function. Nat. Immunol. 2020;21:1327–1335. doi: 10.1038/s41590-020-0778-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yi Y., Xu Y., Jiang H., Wang J. Cardiovascular disease and COVID-19: insight from cases with heart failure. Front. Cardiovasc. Med. 2021;8:163. doi: 10.3389/fcvm.2021.629958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan L., Zhu H., Zhou M., Ma J., Chen R., Chen Y., Chen L., Wu K., Cai M., Hong J., Li L., Liu C., Yu H., Zhang Y., Wang J., Zhang T., Ge S., Zhang J., Yuan Q., Chen Y., Tang Q., Chen H., Cheng T., Guan Y., Xia N. Gender associates with both susceptibility to infection and pathogenesis of SARS-CoV-2 in Syrian hamster. Sig. Transduct. Target. Ther. 2021;6:136. doi: 10.1038/s41392-021-00552-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Y.Y., Ma Y.T., Zhang J.Y., Xie X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020;17:259–260. doi: 10.1038/s41569-020-0360-5. [DOI] [PMC free article] [PubMed] [Google Scholar]