Abstract
Introduction
Atopic dermatitis (AD) is a systemic inflammatory condition that may increase the risk of cardiovascular disease (CVD); however, ongoing debate exists surrounding its direct association. We aimed to elucidate whether AD contributes to a higher incidence of CVD and major adverse cardiovascular events (MACE) in adult patients with AD, independent of metabolic disorders.
Methods
We retrospectively analyzed a large US-based population of patients with AD (≥ 18 years of age). Logistic regression estimated the risk of CVD and MACE in adult patients with AD, independent of metabolic disorders (including diabetes, hypertension, and obesity).
Results
The odds ratio (OR; 95% confidence interval [CI]) for patients without metabolic disorders was 1.25 (1.13, 1.39) for CVD and 1.22 (1.01, 1.47) for MACE. The OR (95% CI) for AD patients with metabolic disorders was 1.09 (1.07, 1.12) for CVD and 1.14 (1.09, 1.18) for MACE. This trend was even more pronounced after long-term follow-up (≥ 3 years). Lifestyle and health behavioral factors of the subjects were not available in the dataset. The lack of control for these factors could potentially confound our results.
Conclusions
Atopic dermatitis may contribute to the risk of developing CVD and MACE in adults, independent of metabolic disorders.
Keywords: Atopic dermatitis, Cardiovascular disease, Metabolic disorders, Retrospective analysis
Key Summary Points
| Why carry out this study? |
| The link between atopic dermatitis (AD) and cardiovascular disease (CVD) remains unclear |
| We retrospectively analyzed a large US-based population of patients with AD to assess whether AD is associated with CVD |
| What was learned from this study? |
| Patients with AD appeared to be at greater risk of CVD than non-AD patients, independent of concomitant metabolic disorders, and this trend was more pronounced in patients with longer (≥ 3 years) follow-up |
| Effective treatment of AD may reduce future risk of CVD; this needs to be confirmed with prospective longitudinal studies |
Introduction
Atopic dermatitis (AD) is a chronic, relapsing, inflammatory skin disease that is clinically characterized by intense pruritus, xerosis, and eczematous lesions; it has a prevalence of about 2–10% in adults in the USA [1–4]. AD is associated with immune dysregulation and skin barrier dysfunction, and increasing evidence supports AD as a systemic inflammatory disease rather than just a cutaneous disease [5].
For patients with rheumatoid arthritis and psoriasis, the inflammatory nature of their disease has been suggested to contribute to an increased risk of developing cardiovascular disease (CVD) [6–8]. While the underlying mechanisms of the inflammatory component of AD appear to differ from those of psoriasis [5], AD has also been associated with a blood signature of increased inflammatory and cardiovascular (CV) risk proteins, including C-reactive protein [5, 9]. This systemic inflammatory component of AD, which contributes to the “inflammatory skin march,” may also be involved in the development and increased prevalence of comorbidities such as CVD and metabolic syndrome [10]; in a recent retrospective study of 195 adults and children with AD, 29% also had CVD and 9% had metabolic syndrome. Patient age has been found to correlate positively with both comorbidities, and a significant positive correlation has also been found between metabolic syndrome and CVD [11].
Adults with AD were found to have a larger number of CV risk factors than those without AD in a study of 34,525 adults in the USA aged 18–85 years, identified from the 2012 US National Health Interview Survey (NHIS), confirming results of a previous 2010 NHIS study of 27,157 adults in the USA [12]. Adults with AD also appear to have an increased risk of coronary artery disease, myocardial infarction (MI), congestive heart failure, angina, stroke, and peripheral vascular disease (PVD) compared with controls, based on results from a multivariate analysis adjusting for baseline characteristics of data from the 2005–2006 National Health and Nutrition Examination Survey (NHANES; n = 4970), 2010 NHIS, and 2012 NHIS [12]; these data were also supported by several other studies that evaluated CV risk factors in patients with AD [13–17]. Regardless of this observed increase in CVD events in patients with AD, it is yet to be elucidated whether the systemic inflammatory nature of the disease itself is responsible, or whether it is merely due to the existence of comorbidities such as metabolic disorders and associated CV risk factors.
In this study, our goal was to assess major adverse cardiovascular events (MACE) and CVD events in patients with and without metabolic disorders, within a large cohort of adult patients with AD in the USA.
Methods
Data Source and Study Design
Data were derived from the Truven Health MarketScan® database between 1 January 2008, and 31 December 2014 (observation period). The first recorded diagnosis of AD during the observation period was the index date and the 12 months prior to the index date served as the baseline period. For patients in the control cohort, the index date was selected at random from within the study observation period, and a 12-month ‘lookback’ was done to ensure eligibility. This study was approved by the Sanofi and Regeneron Alliance internal protocol review committee.
Study Population
The study population comprised two cohorts: an AD cohort of adults (≥ 18 years of age) who had ≥ 2 AD diagnosis codes (International Classification of Diseases, Ninth Revision [ICD-9] code 691.8; https://www.cdc.gov/nchs/icd/icd9.htm) during the observation period, and a non-AD control cohort of adults without a diagnosis of AD. Patients with AD were matched to non-AD controls based on age, sex, index date, and duration of follow-up. For inclusion in the study, patients were required to have been enrolled continuously during the baseline period (i.e., the 12 months immediately prior to the index date). Patients with CVD or MACE during the 12-month baseline period were excluded from the analysis.
Outcomes
Outcomes were the first post-index diagnosis of CVD or MACE in patients with AD and non-AD controls. CVD was defined as a diagnosis of cerebrovascular disease (ICD-9 codes: 362.34, 430, 431, 432, 433, 434, 435, 436, 437, 438), MI (ICD-9 codes: 410, 412), coronary heart disease (ICD-9 codes: 440.2, 440.3, 440.4, 440.9, 443.9), congestive heart failure (ICD-9 codes: 398.91, 402.01, 402.11, 402.91, 404.01, 404.03, 404.11, 404.13, 404.91, 404.93, 425.4, 425.5, 425.7, 425.8, 425.9, 428), peripheral artery disease (ICD-9 codes: 4402, 4403, 4404, 4409, 4439), atherosclerosis (ICD-9 code: 440), stroke (ICD-9 codes: 433.x1, 434.x1), or PVD (ICD-9 codes: 0930, 4373, 440, 441, 4431, 4432, 4438, 4439, 4471, 5571, 5579, V434).
MACE included diagnoses of MI (ICD-9 codes: 410.xx, 410.x1), stroke (ICD-9 codes: 433.x1, 434.x1), angina with hospitalization (ICD-9 codes: 411.1, 411.81), and coronary revascularization procedure (ICD-9 codes: 36.0x, 36.1x, 36.2x, 36.3x, 00.66; Current Procedural Terminology codes: 33510–33529, 33531–33536, 92982, 92984, 92995, 92996, 92980, 92981; Healthcare Common Procedure Coding System codes: S2205–S2209, G0290, G0291).
To further understand potential clinically relevant factors that contribute to CV risk, patients with AD and non-AD controls were stratified by the presence of metabolic disorders pre-index and/or during follow-up, consisting of diabetes, hypoglycemia, hyperlipidemia, calcium metabolism disorders, hypertension, obesity, and tobacco use.
A sub-population of patients with ≥ 3-year follow-up was also evaluated to determine the long-term risk of CVD and MACE in AD patients with or without metabolic disorders.
Statistical Analyses
Logistic regression was used to estimate the odds ratio (OR) and 95% confidence interval (CI) to describe the risk of CVD and MACE associated with AD in the analysis of the entire population.
Results
Baseline Characteristics
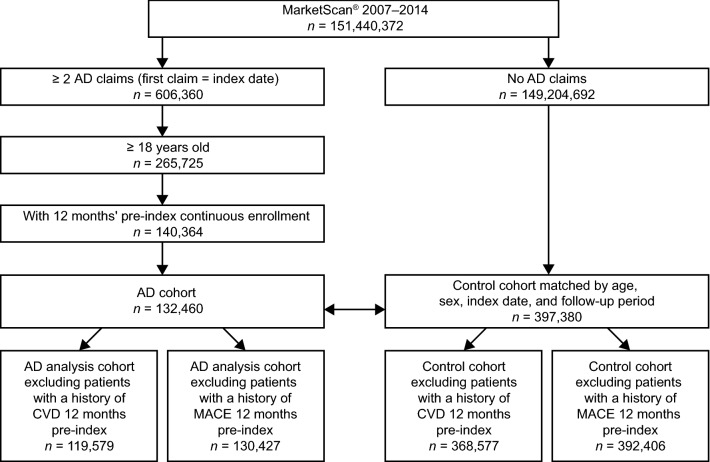
Of the 151,440,372 patients identified in the Truven Health MarketScan® database during the observation period, 132,460 patients met the inclusion criteria of ≥ 2 claims for AD, ≥ 18 years of age, and 12-month pre-index continuous enrollment period. Of the 149,204,692 patients with no AD claims, 397,380 patients were matched as non-AD controls by age, sex, index date, and follow-up (Fig. 1).
Fig. 1.
Flowchart of study cohort selection. Patients were selected from the MarketScan® database from 2007 to 2014 as part of the AD cohort if they had at least two AD claims, or as part of the non-AD cohort if they had no AD claims. Patients with AD were matched with non-AD controls by age, sex, index date, and follow-up period. Patients with a known history of CVD or MACE were excluded from the study. AD atopic dermatitis, CVD cardiovascular disease, MACE major adverse cardiovascular events
Demographic characteristics for patients in the AD cohort and the non-AD control cohort are provided in Table 1. In both cohorts, the mean (standard deviation) age was 47.2 (17.5) years and 62.9% were female (Table 1). The majority of patients in the AD cohort (84.5%) and in the control cohort (83.9%) were enrolled in commercial or Medicare insurance plans. There was a slightly higher prevalence of metabolic disorders in the AD cohort than in the control cohort, including a slightly higher frequency of tobacco use in AD patients than in controls. The geographical distribution across the USA was similar between both cohorts.
Table 1.
Demographic characteristics of atopic dermatitis and matched control cohorts
| Demographic characteristics | AD cohort (n = 132,460) | Control cohort (n = 397,380) |
|---|---|---|
| Female, % | 62.9 | 62.9 |
| Mean (SD) age, years | 47.2 (17.5) | 47.2 (17.5) |
| Commercial/Medicare, % | 84.5 | 83.9 |
| Metabolic disorders at baseline, % | ||
| Diabetes | 10.4 | 9.4 |
| Hypoglycemia | 0.9 | 0.6 |
| Hyperlipidemia | 27.5 | 20.2 |
| Calcium metabolism disorder | 0.6 | 0.4 |
| Hypertension | 28.8 | 22.9 |
| Obesity | 5.3 | 3.3 |
| Tobacco | 3.4 | 2.5 |
| Geographical distribution according to region of the USA, % | ||
| Northeast | 18.3 | 14.5 |
| North Central | 20.1 | 26.0 |
| South | 35.2 | 37.8 |
| West | 25.2 | 20.2 |
AD atopic dermatitis, SD standard deviation
Overall Risk of CVD
Following the exclusion of patients with a history of CVD events during the 12 months immediately prior to the index date, 119,579 and 368,577 patients remained in the AD cohort and control cohort, respectively (Fig. 1). Mean follow-up for AD patients with CVD was 706.0 days compared with 763.4 days for non-AD controls. The overall incidence of CVD during follow-up was 8.4% and 6.3% for the AD cohort and control cohort, respectively, with an OR (95% CI) of 1.38 (1.34, 1.41) (Table 2).
Table 2.
Risk of cardiovascular disease and and major adverse cardiovascular events in patients with AD
| Risk of CVD and MACE in study populations | CVD | MACE | ||||
|---|---|---|---|---|---|---|
| AD cohort | Control cohort | OR (95% CI) | AD cohort | Control cohort | OR (95% CI) | |
| Incident, overall population, n | 130,427 | 392,406 | 1.38 (1.34, 1.41) | 130,427 | 392,406 | 1.40 (1.34, 1.45) |
| % with event | 8.4% | 6.3% | 2.6% | 1.9% | ||
| Patients with metabolic disorders, n | 79,199 | 191,699 | 1.09 (1.07, 1.12) | 89,657 | 214,137 | 1.14 (1.09, 1.18) |
| % with event | 12.1% | 11.2% | 3.7% | 3.3% | ||
| Patients without metabolic disorders, n | 51,228 | 200,707 | 1.25 (1.13, 1.39) | 40,770 | 178,269 | 1.22 (1.01, 1.47) |
| % with event | 1.2% | 0.9% | 0.3% | 0.3% | ||
| Long-term (≥ 3 years) follow-up population | ||||||
| Overall population, n | 29,900 | 108,058 | 1.76 (1.69, 1.82) | 29,900 | 108,508 | 1.68 (1.58, 1.78) |
| % with event | 17.0% | 10.4% | 5.2% | 3.1% | ||
| Patients with metabolic disorders, n | 22,195 | 61,421 | 1.33 (1.28, 1.38) | 24,573 | 67,320 | 1.30 (1.23, 1.39) |
| % with event | 20.5% | 16.3% | 6.2% | 4.8% | ||
| Patients without metabolic disorders, n | 7705 | 46,637 | 1.41 (1.15, 1.71) | 5327 | 41,188 | 1.46 (1.01, 2.10) |
| % with event | 2.2% | 1.4% | 0.7% | 0.5% | ||
AD atopic dermatitis, CI confidence interval, CVD cardiovascular disease, MACE major adverse cardiovascular events, OR odds ratio
Overall Risk of MACE
Following the exclusion of patients with a history of MACE events in the 12 months immediately prior to the index date, 130,427 and 392,406 patients remained in the AD cohort and control cohort, respectively (Fig. 1). Mean follow-up for AD patients with MACE was 706.1 days compared with 763.6 days for non-AD controls. The overall incidence of MACE during follow-up was 2.6% and 1.9% for the AD cohort and control cohort, respectively, with an OR (95% CI) of 1.40 (1.34, 1.45) (Table 2).
Risk of CVD and MACE in Patients With and Without Metabolic Disorders
In general, patients with metabolic disorders, in both the AD cohort and control cohort, had a higher incidence of CVD and MACE than patients without metabolic disorders (Table 2). However, the ORs estimating the risk of CVD and MACE in AD patients versus non-AD controls were higher in patients without metabolic disorders than in those with metabolic disorders (Table 2).
Risk of CVD and MACE in Patients With Long-Term Follow-Up
Long-term follow-up (≥ 3 years) was available for 25.0% and 29.3% of patients in the AD cohort and control cohort, respectively. Mean follow-up for AD patients with CVD and MACE was 1598.4 and 1624.6 days, respectively. Mean follow-up for non-AD controls with CVD and MACE was 1597.5 and 1623.4 days, respectively. Overall, the estimated OR (95% CI) indicated that patients with AD were 1.76 (1.69, 1.82) and 1.68 (1.58, 1.78) -fold more likely to develop CVD and MACE, respectively, than non-AD controls (Table 2).
The increased risk of CVD and MACE in patients without metabolic disorders compared with those with metabolic disorders was even more pronounced in the long-term (≥ 3 years) follow-up. The OR (95% CI) for the AD cohort and control cohort without metabolic disorders was 1.41 (1.15, 1.71) for CVD and 1.46 (1.01, 2.10) for MACE, whereas the OR (95% CI) for the AD cohort and control cohort with metabolic disorders was 1.33 (1.28, 1.38) for CVD and 1.30 (1.23, 1.39) for MACE (Table 2).
Discussion
Our retrospective study based on data from the Truven Health MarketScan® database found that, in a predominantly insured US patient population, adult patients with AD are at an increased risk of developing CVD and MACE compared with non-AD controls, a trend that became more pronounced in the longer-term (≥ 3 years) follow-up. Indeed, patients with AD were found to have a 1.38- and 1.40-fold greater risk of developing CVD and MACE, respectively, than non-AD controls. Furthermore, at ≥ 3 years of follow-up, patients with AD were found to have a 1.76- and 1.68-fold greater risk for developing CVD and MACE, respectively, than non-AD controls.
It remains unclear whether the inflammatory nature of AD contributes to the higher risk of CVD in patients with AD or whether it is merely due to the existence of comorbidities, such as metabolic disorders. For example, some studies have attributed the increase in MI and stroke among patients with AD to the increased prevalence of metabolic disorders and associated lifestyle factors, rather than to AD itself [14, 18]. Plausibly, this debate arises as metabolic disorders and the associated sedentary lifestyle, cigarette smoking, and alcohol consumption are known CV risk factors that are prevalent in the adult AD population [12, 19].
The association between AD and metabolic disorders remains unclear owing to conflicting reports. Whereas the authors of one study claim that it seems unlikely the association is causal [20], those of another study suggest that the disturbance in lipid metabolism observed in metabolic disorders may lead to skin defects that contribute to AD [21]. Nevertheless, patients with AD continue to have metabolic disorders more often than those without AD [22].
To better understand the contribution of metabolic disorders to the CVD and MACE risk seen in patients with AD, we further stratified patients by presence of metabolic disorders. Consistent with previous findings, we observed that patients with metabolic disorders had a greater incidence of CVD and MACE than those without metabolic disorders [19]. Interestingly, however, the estimated risk of developing CVD and MACE was higher for AD patients without metabolic disorders than for those with metabolic disorders. This trend was greatly pronounced in the long-term (≥ 3 years) follow-up, at which the risk of developing CVD and MACE was estimated to be 1.41- and 1.46-fold greater, respectively, for AD patients without metabolic disorders than for non-AD controls without metabolic disorders. These results suggest that AD may in fact contribute to the risk of developing CVD, independent of metabolic disorders.
Previous studies show conflicting results as to the link between AD and CVD. For example, the authors of a population-based Danish study that analyzed 2527 patients (aged ≥ 15 years) with severe AD concluded that the risk of CVD in patients with severe AD (when adjusted for CV risk factors such as socioeconomic status, smoking, comorbidities, and medication use) was similar to that of the general population [18]. By contrast, 4814 patients with AD were selected in a larger population-based Danish study that found AD patients were 1.74-fold more likely to develop MI than non-AD controls, when adjusted for CV risk factors [17]. Furthermore, a larger population-based study with a cohort of 387,439 adult (≥ 18 years) patients with AD carried out in the UK found that patients with severe and active AD were at greater risk of CVD than patients without AD [15]. Specifically, the authors of this study reported that patients with severe AD had a 20% increase in the risk of stroke; 40–50% increase in the risk of MI, unstable angina, atrial fibrillation, and CV death; and 70% increase in the risk of heart failure.
Through our approach of analyzing the risk of CVD and MACE in a large cohort of patients with AD and non-AD controls by the presence of metabolic disorders, we were able to shed some light on the ongoing debate around the direct association between the inflammatory nature of AD and CVD. We found that, although AD patients with metabolic disorders had a higher incidence of CVD and MACE, AD patients without metabolic disorders (including obesity and tobacco use) showed a higher risk of CVD and MACE than non-AD controls. Therefore, the results of this large, real-world, retrospective cohort study suggest that the systemic inflammatory nature of AD, rather than underlying metabolic disorders, may be linked directly to the increased risk of CVD and MACE in patients with AD, similar to available evidence for rheumatoid arthritis and psoriasis [6, 7].
Our study has some limitations that need to be considered when interpreting the results. Owing to the nature of the data source (i.e., a large retrospective database), outcome misclassification is likely because patients were identified through administrative claims data for reimbursement purposes as opposed to medical records. We could have missed some events or mistakenly identified some patients as having had the event. But these misclassifications are most likely to be nondifferential, which would not have impact on our conclusion. We were also not able to control for lifestyle and health behavior factors of the subjects, thus leading to confounding of our results. Additionally, the generalizability of our findings may be limited because the MarketScan® database contains data from patients who were enrolled in commercial or Medicare insurance plans paid for by employers, thereby excluding patients without insurance or other forms of insurance. Finally, exclusion of some patients diagnosed with AD may have occurred as ≥ 2 ICD-9 codes were used to improve the accuracy of identifying the AD cohort. Using more than one occurrence of an ICD-9 code to identify AD has been shown to improve specificity [23], ensuring that our cohort of AD patients were more likely to be ‘true’ AD patients.
Despite the above limitations, our study—owing to the large sample size, long-term follow-up, and assessment of outcomes with or without metabolic disorders—provides valuable evidence to support a potential direct link between the inflammatory nature of AD and the increased risk of CVD in patients with AD. Further studies to confirm this association and to identify factors to mitigate the risk (i.e., whether effective AD treatments may reduce this risk) are warranted.
Conclusions
Adults with AD appear to be at greater risk of CVD and MACE than adults without AD. The risk associated with AD was increased even when accounting for the presence of metabolic disorders, and was especially evident among patients with a longer duration of follow-up. Additional studies are warranted to confirm these associations and explore whether effective control of AD helps reduce future risk of CVD and MACE.
Acknowledgements
Funding
This study, and the journal’s Rapid Service Fee, were funded by Sanofi and Regeneron Pharmaceuticals, Inc.
Medical Writing and/or Editorial Assistance
Medical writing support was provided by Saba Choudhary, PhD, and Chloe Malloy, MSc, of Prime Global, London, UK, according to Good Publication Practice guidelines and was funded by Sanofi and Regeneron Pharmaceuticals, Inc.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Jashin J. Wu, Caroline Amand, Daniel J. No, Puneet Mahajan, Abhijit Gadkari, Eric Ghorayeb, Mandeep Kaur, Andrew Korotzer and Laurent Eckert all made substantial contributions to study design, and to acquisition, analysis, and interpretation of study data. Jashin J. Wu, Caroline Amand, Daniel J. No, Puneet Mahajan, Abhijit Gadkari, Eric Ghorayeb, Mandeep Kaur, Andrew Korotzer, and Laurent Eckert all contributed substantially to drafting and revising the manuscript, approve the final version for publication, and agree to be accountable for the contents of the final publication.
Disclosures
Jashin J. Wu is or has been an investigator, consultant, or speaker for AbbVie, Almirall, Amgen, Arcutis, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant, Dr. Reddy's Laboratories, Eli Lilly, Galderma, Janssen, LEO Pharma, Novartis, Regeneron, Sanofi Genzyme, Sun Pharmaceutical, UCB, and Valeant Pharmaceuticals North America LLC. Caroline Amand and Laurent Eckert are employees of and stockholders in Sanofi. Daniel J. No is an employee of Kaiser Permanente Los Angeles Medical Center. Andrew Korotzer is an employee of and stockholder in Regeneron Pharmaceuticals, Inc. Puneet Mahajan, Eric Ghorayeb, and Mandeep Kaur were former employees of and stockholders in Sanofi at the time of study. Puneet Mahajan is a current employee of Amgen, Eric Ghorayeb is a current employee of Janssen, and Mandeep Kaur is a current employee of Arena Pharmaceuticals. Abhijit Gadkari was a former employee of and stockholder in Regeneron Pharmaceuticals, Inc. at the time of study and is a current employee of Boehringer Ingelheim.
Compliance with Ethics Guidelines
This study was approved by the Sanofi and Regeneron Alliance internal protocol review committee.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
- 1.Barbarot S, Auziere S, Gadkari A, et al. Epidemiology of atopic dermatitis in adults: results from an international survey. Allergy. 2018;73(6):1284–1293. doi: 10.1111/all.13401. [DOI] [PubMed] [Google Scholar]
- 2.Gelbard CM, Hebert AA. New and emerging trends in the treatment of atopic dermatitis. Patient Prefer Adherence. 2008;2:387–392. [PMC free article] [PubMed] [Google Scholar]
- 3.Sidbury R, Davis DM, Cohen DE, et al. Guidelines of care for the management of atopic dermatitis: section 3. Management and treatment with phototherapy and systemic agents. J Am Acad Dermatol. 2014;71(2):327–349. doi: 10.1016/j.jaad.2014.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Silverberg JI, Hanifin JM. Adult eczema prevalence and associations with asthma and other health and demographic factors: a US population-based study. J Allergy Clin Immunol. 2013;132(5):1132–1138. doi: 10.1016/j.jaci.2013.08.031. [DOI] [PubMed] [Google Scholar]
- 5.Brunner PM, Silverberg JI, Guttman-Yassky E, et al. Increasing comorbidities suggest that atopic dermatitis is a systemic disorder. J Invest Dermatol. 2017;137(1):18–25. doi: 10.1016/j.jid.2016.08.022. [DOI] [PubMed] [Google Scholar]
- 6.Wu JJ. Contemporary management of moderate to severe plaque psoriasis. Am J Manag Care. 2017;23(21 Suppl):S403–S416. [PubMed] [Google Scholar]
- 7.Ingram JR. Atopic eczema and cardiovascular disease. BMJ. 2018;361:k2064. doi: 10.1136/bmj.k2064. [DOI] [PubMed] [Google Scholar]
- 8.Crowson CS, Liao KP, Davis JM, 3rd, et al. Rheumatoid arthritis and cardiovascular disease. Am Heart J. 2013;166(4):622–628. doi: 10.1016/j.ahj.2013.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Vekaria AS, Brunner PM, Aleisa AI, et al. Moderate-to-severe atopic dermatitis patients show increases in serum C-reactive protein levels, correlating with skin disease activity. F1000Res. 2017;6:1712. doi: 10.12688/f1000research.12422.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Furue M, Kadono T. "Inflammatory skin march" in atopic dermatitis and psoriasis. Inflamm Res. 2017;66(10):833–842. doi: 10.1007/s00011-017-1065-z. [DOI] [PubMed] [Google Scholar]
- 11.Bekić S, Martinek V, Talapko J, Majnarić L, Vasilj Mihaljević M, Škrlec I. Atopic dermatitis and comorbidity. Healthcare (Basel) 2020;8(2):70. doi: 10.3390/healthcare8020070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Silverberg JI, Greenland P. Eczema and cardiovascular risk factors in 2 US adult population studies. J Allergy Clin Immunol. 2015;135(3):721–728. doi: 10.1016/j.jaci.2014.11.023. [DOI] [PubMed] [Google Scholar]
- 13.Hjuler KF, Bottcher M, Vestergaard C, et al. Increased prevalence of coronary artery disease in severe psoriasis and severe atopic dermatitis. Am J Med. 2015;128(12):1325–1334. doi: 10.1016/j.amjmed.2015.05.041. [DOI] [PubMed] [Google Scholar]
- 14.Standl M, Tesch F, Baurecht H, et al. Association of atopic dermatitis with cardiovascular risk factors and diseases. J Invest Dermatol. 2017;137(5):1074–1081. doi: 10.1016/j.jid.2016.11.031. [DOI] [PubMed] [Google Scholar]
- 15.Silverwood RJ, Forbes HJ, Abuabara K, et al. Severe and predominantly active atopic eczema in adulthood and long term risk of cardiovascular disease: population based cohort study. BMJ. 2018;361:k1786. doi: 10.1136/bmj.k1786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Su VY, Chen TJ, Yeh CM, et al. Atopic dermatitis and risk of ischemic stroke: a nationwide population-based study. Ann Med. 2014;46(2):84–89. doi: 10.3109/07853890.2013.870018. [DOI] [PubMed] [Google Scholar]
- 17.Riis JL, Vestergaard C, Hjuler KF, et al. Hospital-diagnosed atopic dermatitis and long-term risk of myocardial infarction: a population-based follow-up study. BMJ Open. 2016;6(11):e011870. doi: 10.1136/bmjopen-2016-011870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Andersen YM, Egeberg A, Gislason GH, Hansen PR, Skov L, Thyssen JP. Risk of myocardial infarction, ischemic stroke, and cardiovascular death in patients with atopic dermatitis. J Allergy Clin Immunol. 2016;138(1):310–312. doi: 10.1016/j.jaci.2016.01.015. [DOI] [PubMed] [Google Scholar]
- 19.Szuszkiewicz-Garcia MM, Davidson JA. Cardiovascular disease in diabetes mellitus: risk factors and medical therapy. Endocrinol Metab Clin North Am. 2014;43(1):25–40. doi: 10.1016/j.ecl.2013.09.001. [DOI] [PubMed] [Google Scholar]
- 20.Ali Z, Ulrik CS, Agner T, Thomsen SF. Association between atopic dermatitis and the metabolic syndrome: a systematic review. Dermatology. 2018;234(3–4):79–85. doi: 10.1159/000491593. [DOI] [PubMed] [Google Scholar]
- 21.Wollina U. Atopic dermatitis and the metabolic syndrome. Clin Dermatol. 2018;36(1):62–66. doi: 10.1016/j.clindermatol.2017.09.010. [DOI] [PubMed] [Google Scholar]
- 22.Lee JH, Jung HM, Han KD, et al. Association between metabolic syndrome and atopic dermatitis in Korean adults. Acta Derm Venereol. 2017;97(1):77–80. doi: 10.2340/00015555-2441. [DOI] [PubMed] [Google Scholar]
- 23.Hsu DY, Dalal P, Sable KA, et al. Validation of international classification of disease ninth revision codes for atopic dermatitis. Allergy. 2017;72(7):1091–1095. doi: 10.1111/all.13113. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.