Abstract
Objective:
To evaluate the role of procalcitonin (PCT) results in antibiotic decisions for COVID-19 patients at hospital presentation.
Design, setting, and participants:
Multicenter retrospective observational study of patients ≥18 years hospitalized due to COVID-19 at the Johns Hopkins Health system. Patients who were transferred from another facility with >24 hours stay and patients who died within 48 hours of hospitalization were excluded.
Methods:
Elevated PCT values were determined based on each hospital’s definition. Antibiotic therapy and PCT results were evaluated for patients with no evidence of bacterial community-acquired pneumonia (bCAP) and patients with confirmed, probable, or possible bCAP. The added value of PCT testing to clinical criteria in detecting bCAP was evaluated using receiving operating curve characteristics (ROC).
Results:
Of 962 patients, 611 (64%) received a PCT test. ROC curves for clinical criteria and clinical criteria plus PCT test were similar (at 0.5 ng/mL and 0.25 ng/mL). By bCAP group, median initial PCT values were 0.58 ng/mL (interquartile range [IQR], 0.24–1.14), 0.23 ng/mL (IQR, 0.1–0.63), and 0.15 ng/mL (IQR, 0.09–0.35) for proven/probable, possible, and no bCAP groups, respectively. Among patients without bCAP, an elevated PCT level was associated with 1.8 additional days of CAP therapy (95% CI, 1.01–2.75; P < .01) compared to patients with a negative PCT result after adjusting for potential confounders. Duration of CAP therapy was similar between patients without a PCT test ordered and a low PCT level for no bCAP and possible bCAP groups.
Conclusions:
PCT results may be abnormal in COVID-19 patients without bCAP and may result in receipt of unnecessary antibiotics.
Procalcitonin (PCT) is produced in response to cytokines (interleukin [IL]-1β, tumor necrosis factor [TNF]-α, and IL-6) released during bacterial infections.1 PCT testing has emerged as a potential tool to optimize antibiotic decisions in patients with sepsis and lower respiratory tract infections, with mixed results in real-world studies.2–6
The pathogenesis of coronavirus disease 2019 (COVID-19) remains under study; however, overproduction of proinflammatory cytokines including IL-6 which induce procalcitonin (PCT) production may play a role.7–9 A recent observational study of hospitalized COVID-19 patients showed that the PCT level was elevated in 21% of patients without laboratory evidence of bacterial community-acquired pneumonia (bCAP).10 To better understand whether clinicians and antibiotic stewardship programs (ASP) can rely on PCT testing to make antibiotic decisions in hospitalized COVID-19 patients, we evaluated PCT results in COVID-19 patients with and without evidence of bCAP. We explored the value of adding initial admission PCT testing to clinical criteria alone to identify bCAP and the impact of admission PCT testing on subsequent antibiotic use (frequency and duration) among patients without a PCT test ordered and those with initially high and low PCT results.
Methods
Study population and setting
Patients aged ≥18 years admitted to the Johns Hopkins Health System (JHHS) with a positive molecular SARS-CoV-2 test between March 1, 2020, and May 30, 2020, were included in the study, and we excluded patients with admission to a non-JHHS hospital for >24 hours before transfer, SARS-CoV-2 detected on asymptomatic screening, and death or discharge within 2 days of hospitalization. Additional exclusion criteria applied to calculate the duration of CAP therapy were death within 5 days of hospitalization or nonrespiratory bacterial coinfection. JHHS includes 5 acute-care hospitals in the Baltimore–Washington, DC, region, all with antibiotic stewardship programs (ASPs). Hospital A is a 1,056-bed tertiary-care teaching hospital without access to PCT testing; hospital B is a 350-bed teaching hospital with a disease severity-based PCT algorithm4; and hospitals C, D, and E, are 264-bed, 222-bed, and 318-bed, community hospitals, respectively, that use PCT test results based on the interpretation guidance provided by the test manufacturer (Supplementary Table 1 online). We included hospital A because we wanted to evaluate antibiotic use in patients with and without PCT tests, and most patients in hospitals B, C, D and E receive PCT testing. The study was approved by the Johns Hopkins Institutional Review Board.
Study design and study objectives
We conducted a retrospective descriptive study of PCT results among symptomatic adult patients hospitalized with COVID-19 who were adjudicated to have proven, probable, possible, or no bCAP at hospital presentation (primary objective). Secondary objectives included the added value of PCT testing to clinical criteria in identifying bCAP cases and antibiotic use in the context of different PCT results and evidence of bCAP.
Definitions
A low PCT level was defined based on each hospital’s definition to best capture the treating clinician’s interpretation for making antibiotic decisions at the different hospitals: <0.25 ng/mL outside the intensive care unit (ICU) and <0.5 ng/mL in the ICU for hospital B, and <0.25 ng/mL for hospitals C, D, and E. Abnormal PCT level refers to a PCT result not considered low by the hospital.
bCAP was defined by a multidisciplinary team including antimicrobial stewardship, hospital epidemiology and infection control, general infectious disease, transplant/oncology infectious disease, pulmonary/critical care, and infectious disease pharmacy experts based on consensus definitions as previously described.11 Briefly, definitions of proven, probable, and possible bCAP were developed based on clinical, laboratory, radiographic, and microbiologic criteria detected within the first 48 hours of hospitalization. PCT results were not part of the definition. Proven bCAP was adjudicated if both clinical and microbiologic criteria were met. Clinical criteria were met when all of the following were present: fever (T > 38°C) or hypothermia (T < 36°C), white cell count ≥12,000/mm3, purulent sputum (at least moderate PMNs on sputum Gram stain), need for supplemental O2, and imaging consistent with bCAP). Microbiological criteria included a positive respiratory culture with a respiratory pathogen (eg, not enterococcus or Candida) and/or a positive Streptococcus or Legionella bacterial urinary antigen. Probable bCAP was adjudicated when cases met clinical but not microbiologic bacteria and clinical improvement occurred for those on antibiotics within 48–72 hours. Possible bCAP cases included those not meeting all clinical criteria. Charts were randomly assigned to committee members. The committee met 4 times to ensure uniform application of the definitions and to clarify any uncertainties or discrepancies that arose.
Statistical analysis
Categorical and continuous variables were compared using χ2 and Wilcoxon rank-sum tests, respectively. We compared receiver operating characteristics (ROC) curves of the joint logistic regression of elevated PCT level (both ≥0.25 ng/mL and ≥0.5 ng/mL cutoffs) plus clinical criteria to ROC curves of clinical criteria only. Duration of bCAP therapy (calendar days) included both inpatient antibiotics and antibiotics prescribed upon discharge. We used inverse probability of treatment weighting (IPTW) to compare duration of bCAP therapy between patients with no PCT test ordered and those with a negative PCT result. Baseline characteristics were considered balanced if standardized difference values were <10%, covariates that remained unbalanced were included in a linear regression model. Covariates included in the IPTW were selected based on clinical relevance and significant differences on univariate analysis12 and included sex, immunocompromise, ICU admission, transfer to hospital A, admitting hospital, and World Health Organization (WHO) COVID-19 disease severity scale.13 A 2-sided P value <.05 was considered statistically significant for all tests. Statistical analysis was completed using STATA version 16.0 software (StataCorp, College Station, TX).
Results
Cohort characteristics
In total, 962 adult patients hospitalized due to COVID-19 met inclusion criteria (Table 1). The median age of the cohort was 62; 47% were women; 21% had severe COVID-19 per WHO classification; 19% were residents at a long-term care facility (LTCF); and 12% were admitted to the intensive care unit (ICU). Only 11 patients (1%) met proven/probable bCAP criteria, while 48% met possible bCAP criteria (limited evidence of bCAP).
Table 1.
Cohort Characteristics
| Characteristic | Total (N = 962) |
PCT Not Done (N = 351) |
Low PCT (N = 370) |
P Valuea |
Elevated PCT (N = 241) |
P Valueb |
|---|---|---|---|---|---|---|
| Age, median y (IQR) | 62 (49–74) | 57 (44–70) | 62 (49–75) | <.01 | 68 (55–82) | <.01 |
| Sex, female, no. (%) | 448 (47) | 179 (51) | 182 (49) | .62 | 87 (36) | <.01 |
| Race, no. (%) | <.01 | .88 | ||||
| White | 299 (31) | 93 (26) | 138 (37) | 68 (28) | ||
| Black | 337 (35) | 144 (41) | 95 (28) | 98 (29) | ||
| Other | 326 (34) | 114 (32) | 137 (37) | 75 (31) | ||
| Latino/Hispanic ethnicity, no. (%) | 269 (28) | 99 (28) | 112(30) | .54 | 58 (24) | .27 |
| Long-term care resident, no. (%) | 181 (19) | 42 (12) | 73 (20) | <.01 | 66 (27) | <.01 |
| Charlson comorbidity index, median (IQR) | 0 (0–1) | 0 (0–1) | 0 (0–0) | <.01 | 0 (0–1) | .57 |
| Intensive care unit admission, no. (%) | 113 (12) | 26 (7) | 33 (9) | .45 | 54 (22) | <.01 |
| WHO disease severity, no. (%)c | .05 | <.01 | ||||
| Mild | 757 (79) | 282 (80) | 317 (86) | 158 (66) | ||
| Severe | 205 (21) | 69 (20) | 53 (14) | 83 (34) | ||
| Immuncompromising condition, no. (%)d | 53 (6) | 31 (9) | 16(4) | .01 | 6 (11) | <.01 |
| Hospital A | 241 (25) | 241 (100) | … | NA | … | NA |
| Hospital B | 157 (16) | 18 (5) | 100 (27) | 39 (16) | ||
| Hospital C | 222 (23) | 14 (4) | 122 (33) | 86 (36) | ||
| Hospital D | 257 (27) | 56 (16) | 106 (29) | 95 (39) | ||
| Hospital E | 85 (9) | 22 (6) | 42 (11) | 21 (9) | ||
| C-reactive protein maximum value (mg/dL) ≤48 h, median (IQR) | 13.5 (5.9–34.6) | 10.1 (4.6–21) | 12.4 (5.3–27.5) | .04 | 24.8 (11.4–137) | <.01 |
| Temperature ≥38oC, no. (%) | 543 (56) | 196 (56) | 193 (52) | .32 | 154 (64) | .05 |
| Purulent sputum, no. (%) | 34 (5) | 16 (6) | 7 (2) | .04 | 11 (5) | .74 |
| Peripheral WBC ≥12,000, no. (%) | 1,140 (15) | 43 (12) | 13 (6) | .04 | 84 (21) | <.01 |
| Any consolidative opacity, no. (%) | 169 (18) | 75 (21) | 59 (16) | .06 | 35 (15) | .03 |
| bCAP adjudication, no. (%) | .92 | <.01 | ||||
| Proven/probable bCAP | 11 (1) | 2 (1) | 3 (1) | 6 (2) | ||
| Possible bCAP | 457 (48) | 159 (45) | 164 (44) | 134 (56) | ||
| No bCAP coinfection | 494 (51) | 190 (54) | 203 (55) | 101 (42) |
Note. IQR, interquartile range; WBC, white blood cell count; bCAP, bacterial community acquired pneumonia; WHO, World Health Organization.
P values for comparisons between PCT negative and no PCT groups.
P values for comparison between PCT positive to no PCT group.
The WHO scale is an 8-point ordinal severity scale, mild includes WHO score of 3 (not on oxygen) or 4 (on nasal cannula or facemask oxygen) while severe disease includes WHO score 5 (high-flow nasal cannula or noninvasive positive pressure ventilation), 6 (intubation and mechanical ventilation), and 7 (intubated; mechanical ventilation; and other signs of organ failure, including use of extracorporeal membrane oxygen, hemodialysis, or vasopressors).
Immunocompromising condition includes HIV/AIDS, receipt of biologics, prednisone ≥ 20mg daily for ≥ 2 weeks, chemotherapy within 6 months, and solid-organ or hematopoietic stem-cell transplant.
Of 962 patients, 611 (64%) had PCT testing. The median initial PCT values were 0.58 ng/mL (interquartile range [IQR], 0.24–1.14) in the proven/probable bCAP group, 0.23 ng/mL (IQR, 0.1–0.63) in the possible bCAP group, and 0.15 ng/mL (IQR, 0.09–0.35) in the no bCAP group (P < .01 for both proven/probable and possible bCAP vs no bCAP). The proportions of patients with a PCT ≥0.5 ng/mL by bCAP group were as follows: 56% in patients with proven/probable bCAP, 31% in those with possible bCAP, and 20% in those without bCAP. With a PCT cutoff of ≥0.25 ng/mL, these results were 67% in patients with proven/probable bCAP, 46% in those with possible bCAP, and 33% in those without bCAP.
Of the 611 patients with an initial PCT test, 199 (33%) had a repeated PCT test within the first 4 days of hospitalization (median, 23 hours; IQR, 5–48). A discrepant repeated PCT result occurred in 34 (17%) of 199 cases: a higher to lower PCT value in 9 cases and a lower to higher PCT value in 25 cases.
Usefulness of initial procalcitonin in detection of bacterial CAP in COVID-19 patients
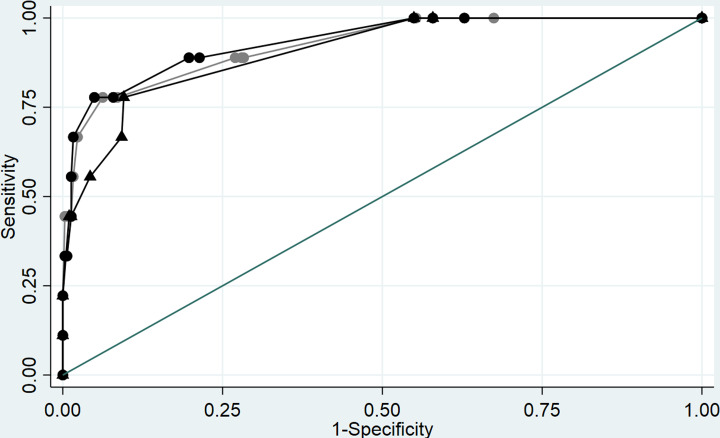
We evaluated whether the initial PCT result added value to clinical criteria (eg, vital signs, laboratory, and radiographic data) in identifying bCAP in COVID-19 patients. We compared the ROC curve of clinical criteria plus PCT testing with clinical criteria alone determined by chart review. ROC curves were similar using both ≥0.25 ng/mL and 0.5 ng/mL PCT as cutoffs (Fig. 1, Table 2). The AUC for clinical criteria alone was 0.90 (95% confidence interval [CI], 0.81–0.98), whereas the AUC for clinical criteria plus PCT at ≥0.5 ng/mL was 0.93 (95% CI, 0.85–1.00; P = .35) and was 0.92 (95% CI, 0.83–1.00; P = .39) at ≥0.25 ng/mL.
Fig. 1.
Area under the curve (AUC) for clinical criteria plus PCT and clinical criteria only to predict proven/probable bacterial community-acquired pneumonia. PCT cutoff, 0.25 ng/mL, Black circles: clinical criteria plus PCT cutoff 0.25 ng/mL. Gray circles: clinical criteria plus PCT cutoff 0.25 ng/mL. Rectangles: clinical criteria only.
Table 2.
Results of the Receiver Operating Characteristic (ROC) Curve Analysis of Clinical Variables With and Without Procalcitonin (PCT) to Predict Proven/Probable Bacterial Community-Acquired Pneumonia
| Clinical Variable | Proven/Probable Coinfection, AUC (95% CI) | ||||
|---|---|---|---|---|---|
| Alone | Plus PCT (0.5 ng/mL) |
P Valuea |
Plus PCT (0.25 ng/mL) |
P Valuea |
|
| Individual clinical criteria | |||||
| Fever | 0.66 (0.54–0.79) | 0.73 (0.57–0.90) | .76 | 0.71 (0.54–0.89) | .80 |
| Leukocytosis | 0.70 (0.54–0.85) | 0.81 (0.65–0.97) | .54 | 0.78 (0.60–0.96) | .42 |
| Purulent sputum | 0.63 (0.49–0.76) | 0.74 (0.57–0.92) | .56 | 0.75 (0.58–0.92) | .62 |
| Consolidative opacity (unilateral or bilateral) | 0.65 (0.50–0.80) | 0.73 (0.54–0.91) | .71 | 0.74 (0.56–0.91) | .78 |
| PCT 0.5 ng/mL PCT 0.25 ng/mL |
0.67 (0.50–0.84) 0.66 (0.50–0.83) |
… | … | … | … |
| Combined clinical criteriab | |||||
| Fever, leukocytosis, purulent sputum | 0.88 (0.80–0.97) | 0.92 (0.84–1.00) | .21 | 0.91 (0.82–1.00) | .35 |
| Fever, leukocytosis, purulent sputum, any consolidative opacity | 0.90 (0.81–0.98) | 0.93 (0.85–1.00) | .23 | 0.92 (0.83–1.00) | .39 |
Note. AUC, area under the curve; CI, confidence interval.
P values correspond to comparisons between clinical criteria and clinical criteria plus PCT at 2 different cutoffs.
Fever, ≥ 38°C; leukocytosis = WBC count > 12,000 cells/mm3.
Frequency of CAP therapy for hospitalized COVID-19 patients
CAP antibiotics were administered to of 207 (86%) of 241 patients with an initial high PCT level, to 246 (66%) of 370 patients with an initially low PCT level, and to 234 (66%) of 351patients without a PCT test. The frequency of CAP antibiotic use in the bCAP group is summarized in Table 3. Specifically, among patients without bCAP, 106 (56%) of 190 patients without a PCT test received CAP therapy, 116 (57%) of 203 patients with a low PCT result received CAP therapy, and 79 (78%) of 101 patients with an elevated PCT level (P < .01) received CAP therapy.
Table 3.
Frequency of CAP Antibiotic Therapy in Patients Hospitalized With COVID-19 by Procalcitonin (PCT) Results in Patients Without Bacterial Community-Acquired Pneumonia (bCAP) and Patients With Possible bCAP
| Patients Without Bacterial Community-Acquired Pneumonia (N = 494) | ||||
| No PCT (N = 190), No. (%) |
Low PCT (N = 203), No. (%) |
P Valuea | Elevated PCT (N = 101), No. (%) |
P Valueb |
| 106 (56) | 116 (57) | .43 | 79 (78) | <.01 |
| Patients with possible bacterial community-acquired pneumonia N = 457 (%) | ||||
| No PCT (N = 159), No. (%) |
Low PCT (N = 164), No. (%) |
P Valuea | Elevated PCT (N = 134), No. (%) |
P Valueb |
| 126 (79) | 127 (77) | .69 | 122 (91) | <.01 |
P value of the comparison between no PCT and low PCT groups.
P value of the comparison between no PCT or low PCT and elevated PCT groups.
Duration of CAP therapy in hospitalized COVID-19 patients
The median duration of CAP antibiotics was 1.0 day in both the no PCT test group and the low PCT group (IQR, 0–3.9 and IQR, 0–2.9, respectively; P = .26), and the median duration of CAP antibiotics was 4 days (IQR, 1–5) in the high PCT group: P < .01 for both no PCT versus high PCT level and low PCT level versus high PCT level. The durations of CAP therapy in patients without bCAP and possible bCAP are summarized in Table 4. Among patients without bCAP (n = 435), the median duration of therapy was 1 day both in patients without a PCT test ordered and in those with a low PCT level (IQR, 0–2.0 and IQR, 0–3.4, respectively; P = .43). The median duration of therapy was 3.5 days (IQR, 0–4.9) in patients with a high PCT level (P < .01 for high PCT level vs low level or no PCT test).
Table 4.
Median Duration of CAP Antibiotic Therapy in Days by Procalcitonin (PCT) Results in Patients Hospitalized With COVID-19 and No Evidence of Bacterial Community-Acquired Pneumonia (bCAP) and Possible bCAP
| Patients Without Bacterial Community-Acquired Pneumonia (N = 435), No. (IQR) | ||||
| No PCT (N = 171), No. (%) |
Low PCT (N = 184), No. (%) |
P Valuea | Elevated PCT (N = 80), No. (%) |
P Valueb |
| 1 (0–2) | 1 (0–3.4) | .43 | 3.5 (0–4.9) | <.01 |
| Patients With Possible Bacterial Community-Acquired Pneumonia (N = 398), No. (IQR) | ||||
| No PCT (N = 135), No. (%) |
Low PCT (N = 151), No. (%) |
P Valuea | Elevated PCT (N = 112), No. (%) |
P Valueb |
| 1.6 (0–4.6) | 1.9 (0.9–4.5) | .30 | 4.7 (3–6.6) | <.01 |
Note. IQR, interquartile range.
P value of the comparison between noPCT and low PCT groups.
P value of the comparison between no PCT or low PCT groups and elevated PCT groups.
Among those 398 patients with possible bCAP, the median duration of therapy was 1.6 days (IQR, 0–4.6) in those without a PCT test,1.9 days (IQR, 0.9–4.5) for those with a low PCT level (P = .30), and 4.7 days (IQR, 3–6.6) for patients with a high PCT level (P < .01 for positive PCT vs negative). Duration of CAP therapy remained similar after adjusting for potential confounders (Supplementary Table 2 online) between those patients without a PCT test ordered and those with a low PCT level (−0.36 days; 95% CI, −1.21 to 0.47; P = .39). We detected similar findings in a subgroup analysis of the no bCAP and the possible bCAP groups (−0.06 days; 95% CI, −1.06 to 0.93; P = 0.89 and −1.03; 95% CI, −2.4 to 0.40; P = .15, respectively). Among the 264 patients with no bCAP, those with a high PCT level received 1.8 additional days (95% CI, 1.01–2.75; P < .01) of CAP therapy compared to those with a low PCT level after adjusting for potential confounders including patient characteristics and hospital (Supplementary Table 3 online). Among 263 patients with possible bCAP, the adjusted duration of bCAP therapy was 2.5 days longer among those with an elevated PCT level than those with a low PCT level (95% CI, 1.63–3.28; P < .01) (Supplementary Table 4 online). Findings remained unchanged after excluding hospital A (Supplementary Table 5 online).
Discussion
In this multicenter observational study of 962 adult individuals hospitalized with COVID-19, an initially high PCT level did not provide additional value to clinical criteria in predicting bacterial CAP coinfection and, when low, PCT level did not impact duration of CAP antibiotics compared to patients who did not undergo PCT testing. Furthermore, use of PCT testing was associated with unnecessary antibiotic use among patients without bCAP.
The use of PCT testing to determine the need to start antibiotics in COVID-19 patients is confounded by the finding that SARS-CoV-2 infection may produce cytokines that induce PCT production.8,9 Although 56% of patients in our cohort with proven/probable bCAP had PCT values ≥ 0.5 ng/mL, 20% of patients with no evidence of bCAP also had a high PCT (or 37% using ≥0.25 ng/mL cutoff). This finding is in agreement with other studies showing high PCT levels in 10%–35% of patients with COVID-19,14,15 including COVID-19 patients without evidence of bCAP.10 Among patients without bCAP, an abnormal PCT led to a median of 1.8 more days of bCAP therapy compared to patients with a negative PCT result. Furthermore, an elevated PCT level did not increase the accuracy of clinical criteria in distinguishing bacterial from viral pneumonia, which is consistent with previously reported data in non–COVID-19 patients.16
Duration of CAP antibiotics in this cohort was short and was similar between patients without PCT testing ordered and those with negative PCT test results after adjusting for admitting hospital (ie, each hospital has a different ASP and/or PCT use), transfer to hospital A (which accepted transfers from system hospitals), and baseline comorbidities and illness severity. In hospitals that routinely use PCT testing, a low PCT value may be useful as an additional confirmatory element to stop or not start antibiotics in COVID-19 patients; however, ASPs should council against using an elevated PCT level as a major criterion to start antibiotics in these patients.
This study has several limitations. Most patients who did not receive a PCT test were admitted to hospital A and differences in ASP or other variables that could impact antibiotic use were not measured; however, we included hospital as an independent variable in our analysis to address for differences in antibiotic practices specific to each hospital. We did not assess the impact of PCT level in clinical outcomes because the focus of the study was to evaluate the value of PCT testing in assisting antibiotic decisions.
In summary, PCT tests are frequently positive in COVID-19 patients presenting to the hospital without evidence of bCAP, which limits their use in making decisions to start or continue antibiotics. Additional antibiotic stewardship efforts may be needed to reduce unnecessary exposure of antibiotics in patients with abnormal PCT results.
Acknowledgments
The data utilized for this publication were obtained from the Johns Hopkins COVID-19 Precision Medicine Center of Excellence, officially designated by the Precision Medicine Analytics Platform (PMAP) leadership and based on the contribution of many patients and clinicians. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Supplementary material
For supplementary material accompanying this paper visit http://dx.doi.org/10.1017/ice.2021.175.
click here to view supplementary material
Financial support
S.K. is supported by a training grant from the National Institute of Allergy and Infectious Diseases (grant no. T32-A1007291).
Conflicts of interest
All authors report no conflicts of interest relevant to this article.
References
- 1.Melendi GA, Laham FR, Monsalvo AC, et al. Cytokine profiles in the respiratory tract during primary infection with human metapneumovirus, respiratory syncytial virus, or influenza virus in infants. Pediatrics 2007;120:e410–e415. [DOI] [PubMed] [Google Scholar]
- 2.Gilbert DN.Procalcitonin as a biomarker in respiratory tract infection. Clin Infect Dis 2011;52 suppl4:S346–S350. [DOI] [PubMed] [Google Scholar]
- 3.Schuetz P, Christ-Crain M, Muller B.Biomarkers to improve diagnostic and prognostic accuracy in systemic infections. Curr Opin Crit Care 2007;13:578–585. [DOI] [PubMed] [Google Scholar]
- 4.Townsend J, Adams V, Galiatsatos P, et al. Procalcitonin-guided antibiotic therapy reduces antibiotic use for lower respiratory tract infections in a United States medical center: results of a clinical trial. Open Forum Infect Dis 2018;5:ofy327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huang DT, Yealy DM, Angus DC, the Pro ACTI. Procalcitonin-guided antibiotic use. N Engl J Med 2018;379:1973. [DOI] [PubMed] [Google Scholar]
- 6.Cole JL.Experience during the first year of procalcitonin implementation: a precautionary tale for smaller facilities. Infect Control Hosp Epidemiol 2018;39:1142–1143. [DOI] [PubMed] [Google Scholar]
- 7.Giamarellos-Bourboulis EJ, Netea MG, Rovina N, et al. Complex immune dysregulation in COVID-19 patients with severe respiratory failure. Cell Host Microbe 2020;27:992–1000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ye Q, Wang B, Mao J.The pathogenesis and treatment of the ‘cytokine storm’ in COVID-19. J Infect 2020;80:607–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pirofski LA, Casadevall A.Pathogenesis of COVID-19 from the perspective of the damage-response framework. mBio 2020;11. doi: 10.1128/mBio.01175-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vaughn VM, Gandhi T, Petty LA, et al. Empiric antibacterial therapy and community-onset bacterial coinfection in patients hospitalized with COVID-19: a multihospital cohort study. Clin Infect Dis 2020. doi: 10.1093/cid/ciaa1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Karaba SM, Jones G, Helsel T, et al. Prevalence of coinfection at the time of hospital admission in COVID-19 patients, a multicenter study. Open Forum Infect Dis 2020. doi: 10.1093/ofid/ofaa578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Amoah J, Stuart EA, Cosgrove SE, et al. Comparing propensity score methods versus traditional regression analysis for the evaluation of observational data: a case study evaluating the treatment of gram-negative bloodstream infections. Clin Infect Dis 2020;71:e497–e505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Garibaldi BT, Fiksel J, Muschelli J, et al. Patient trajectories among persons hospitalized for COVID-19: a cohort study. Ann Intern Med 2021;174:33–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang JJ, Dong X, Cao YY, et al. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy 2020;75:1730–1741. [DOI] [PubMed] [Google Scholar]
- 15.Lippi G, Plebani M.Laboratory abnormalities in patients with COVID-2019 infection. Clin Chem Lab Med 2020;58:1131–1134. [DOI] [PubMed] [Google Scholar]
- 16.Kamat IS, Ramachandran V, Eswaran H, Guffey D, Musher DM.Procalcitonin to distinguish viral from bacterial pneumonia: a systematic review and meta-analysis. Clin Infect Dis 2019;70:538–542. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
For supplementary material accompanying this paper visit http://dx.doi.org/10.1017/ice.2021.175.
click here to view supplementary material