ABSTRACT
Background
Boosting NAD+ via supplementation with niacin equivalents has been proposed as a potential modality capable of promoting healthy aging and negating age-dependent declines of skeletal muscle mass and function.
Objectives
We investigated the efficacy of NAD+-precursor supplementation (tryptophan, nicotinic acid, and nicotinamide) on skeletal muscle mitochondrial function in physically compromised older adults.
Methods
A randomized, double-blind, controlled trial was conducted in 14 (female/male: 4/10) community-dwelling, older adults with impaired physical function [age, 72.9 ± 4.0 years; BMI, 25.2 ± 2.3 kg/m2]. Participants were supplemented with 207.5 mg niacin equivalents/day [intervention (INT)] and a control product (CON) that did not contain niacin equivalents, each for 32 days. The primary outcomes tested were mitochondrial oxidative capacity and exercise efficiency, analyzed by means of paired Student's t-tests. Secondary outcomes, such as NAD+ concentrations, were analyzed accordingly.
Results
Following supplementation, skeletal muscle NAD+ concentrations [7.5 ± 1.9 compared with 7.9 ± 1.6 AU, respectively] in INT compared with CON conditions were not significantly different compared to the control condition, whereas skeletal muscle methyl-nicotinamide levels were significantly higher under NAD+-precursor supplementation [INT, 0.098 ± 0.063 compared with CON, 0.025 ± 0.014; P = 0.001], suggesting an increased NAD+ metabolism. Conversely, neither ADP-stimulated [INT, 82.1 ± 19.0 compared with CON, 84.0 ± 19.2; P = 0.716] nor maximally uncoupled mitochondrial respiration [INT, 103.4 ± 30.7 compared with CON, 108.7 ± 33.4; P = 0.495] improved under NAD+-precursor supplementation, nor did net exercise efficiency during the submaximal cycling test [INT, 20.2 ± 2.77 compared with CON, 20.8 ± 2.88; P = 0.342].
Conclusions
Our findings are consistent with previous findings on NAD+ efficacy in humans, and we show in community-dwelling, older adults with impaired physical function that NAD+-precursor supplementation through L-tryptophan, nicotinic acid, and nicotinamide does not improve mitochondrial or skeletal muscle function. This study was registered at clinicaltrials.gov as NCT03310034.
Keywords: NAD+-precursors, skeletal muscle, mitochondrial function, older adults, metabolism, muscle health
Introduction
Aging has been defined as the time-dependent decline of function (1), with 1 of the most striking features of the aging process being the progressive loss of skeletal muscle mass (sarcopenia) and, in turn, the decline of skeletal muscle physical function (2). The loss of skeletal muscle mass and function leads to mobility impairments, an increased risk of falls, physical frailty, and metabolic impairments (3). Hence, a compromised physical function increases the risk of dependency in activities of daily living and care needs. With society confronted with an ever-aging population, a large effort of research has focused on identifying strategies to promote healthy aging.
Interestingly, the loss of skeletal muscle function is paralleled by an age-dependent decline in mitochondrial function (1), an established hallmark of aging that is considered to be a driving force behind skeletal muscle aging (4–6). Therefore, promoting the mitochondrial metabolism may be a promising strategy to negate the metabolic and functional disturbances seen in skeletal muscle during aging (7).
One pathway that has received much attention in this context is the NAD+–sirtuin (SIRT) axis (8–11). The SIRT enzyme family is comprised of NAD+-dependent deacetylases involved in the regulation of mitochondrial metabolism (12), which act as sensors to the bioavailability of NAD+ (13). The coenzyme NAD+ and its reduced form NAD(H) are critical to the cellular redox potential and are the predominant electron donors in the electron transport chain. NAD+, however, is the rate-limiting substrate in SIRT activity, and NAD+ bioavailability has been shown to decline with increasing age in mice (4, 10) and humans (14, 15). The human body derives NAD+ from various dietary sources, as it can be synthesized de novo from the essential amino-acid L-tryptophan (L-Trp) or from niacin compounds such as nicotinic acid (NA) through the Preiss-Handler pathway, or salvaged from nicotinamide (NAM) and nicotinamide riboside (NR) (16). From a dietary perspective, NR is a trace element, whereas L-Trp, NA, and NAM are more abundantly available in our diets (17).
Preclinical work in murine models of aging has demonstrated the efficacy of supplementing with NAD+ precursors, such as NR and nicotinamide mononucleotide (NMN), to negate the effects of aging induced by declining NAD+ levels (4, 18), promote longevity (18), and improve energy metabolism, body weight, and mitochondrial oxidative metabolism (11). Following suit, several human intervention studies have focused on the use of the NAD+ precursor NR to improve cardiovascular health in middle-aged to older adults (19), to combat metabolic dysfunction in middle-aged individuals with obesity (20, 21), and to improve skeletal muscle metabolism in older adults (22). Although these studies provide some evidence on the efficacy of supplementation with NAD+ precursors in older adults to increase NAD+ levels in blood and skeletal muscle through NR supplementation, evidence stemming from studies involving older adults with compromised physical function and from studies focusing on skeletal muscle function are still lacking.
Furthermore, other than through supplementation with high doses of NR or NA, there is only limited evidence discerning NAD+-precursor supplementation at lower doses. Dietary supplements bearing a greater resemblance to increased dietary intake may also have the potency to increase NAD+ bioavailability in humans (23, 24). Moreover, increasing NAD+ synthesis from L-Trp by inhibiting the α-amino-β-carboxymuconate-ε-semialdehyde decarboxylase (ACMSD) enzyme also leads to activation of SIRT1 and its downstream health benefits (25). Here, we aim to elucidate the efficacy of NAD+-precursor supplementation through a novel combination of L-Trp, NA, and NAM on skeletal muscle mitochondrial function in community-dwelling, older adults with impaired physical function.
Methods
Participants
Through advertisements, 14 physically compromised, older adults (65 years and older; BMI, 20–30 kg/m2) were recruited from the general population in the vicinity of Maastricht. The study was conducted in accordance with the principles of the declaration of Helsinki and approved by the Ethics Committee of the Maastricht University Medical Center. All participants provided written informed consent, and the study was registered at clinicaltrials.gov as NCT03310034.
All participants (4 females, 10 males) were Caucasian, did not smoke, and were generally healthy as predetermined by a medical questionnaire and physical examination by a physician, with only 7 participants using prescription medication, including anti-hypertensives, statins, and proton pump inhibitors (summarized in Table 1). Impaired physical function of the participants was defined as a walking distance of 465 meters or less as assessed during a 6-minute walk test (6MWT). Thus, during a clocked 6-minute session, participants were instructed to walk as far as possible without running, over a 20-meter flat surface track. The cutoff used to define impaired physical function was based on the median 6MWT walking distance (C.P.G.M. de Groot, Dept. Of Agrotechnology and Food Sciences, Human Nutrition & Health, Wageningen University and Research, personal communication 2016) of the pre-frail subgroup of the NU-AGE (European Project on Nutrition in Elderly People) cohort (26). Habitual physical activity was assessed and analyzed by Baecke's Habitual Physical Activity Questionnaire (27). Exclusion from participation was primarily based on the following: excessive alcohol and/or drug abuse, physical activity > 1 hour/week, inability to complete the required measurements, type 2 diabetes mellitus (T2DM), significant allergies or intolerances concerning the study products, and medication use known to hamper the participant's safety during the study procedures.
TABLE 1.
Baseline participant characteristics at screening
| Participant characteristics | |
| Sex, female/male | 4/10 |
| Age, years | 72.9 ± 4.0 |
| Height, cm | 170 ± 6 |
| Weight, kg | 72.9 ± 7.1 |
| BMI, kg/m2 | 25.2 ± 2.3 |
| Systolic blood pressure, mmHg | 144 ± 12 |
| Diastolic blood pressure, mmHg | 84 ± 6 |
| Heart rate, beats per minute | 63 ± 10 |
| 6-minute walking distance, m | 393 ± 53 |
| Maximal aerobic capacity, L/min | 2.31 ± 0.66 |
| Maximal cycling resistance, Watt | 176 ± 63 |
| Blood parameters | |
| Hemoglobin, mmol/L | 9.1 ± 0.6 |
| Creatinine, μmol/L | 88 ± 18 |
| eGFR CKD-EPI | 71.6 ± 10.1 |
| Alkaline phosphatase, U/L | 74 ± 22 |
| γGT, U/L | 23 ± 9 |
| AST, U/L | 23 ± 5 |
| ALT, U/L | 21 ± 5 |
| Bilirubin, μmol/L | 11.5 ± 5.0 |
| Cholesterol, mmol/L | 5.1 ± 0.7 |
| HDL cholesterol, mmol/L | 1.7 ± 0.5 |
| LDL cholesterol, mmol/L | 2.9 ± 0.6 |
| Triglycerides, mmol/L | 1.03 ± 0.32 |
| Free fatty acids, mmol/L | 0.47 ± 0.11 |
| Hb1Ac, % | 5.4 ± 0.3 |
| Glucose, mmol/L | 5.1 ± 0.6 |
| Baecke Physical Activity Index | 7.26 ± 1.34 |
| Work Index | 1.62 ± 0.98 |
| Sport Index | 2.25 ± 0.49 |
| Leisure Index | 3.39 ± 0.41 |
| Medication used, n | |
| Statin | 1 |
| Proton pump inhibitor | 1 |
| Thrombocyte aggregation inhibitor | 1 |
| Calcium and vitamin D | 2 |
| Thyromimetic | 1 |
| Anti-hypertensive drugs | 3 |
| Alpha blocker | 1 |
| NSAID | 1 |
| Benzodiazepine | 1 |
Data are presented as means ±SDs, n = 14. Abbreviations: ALT, alanine transaminase; AST, aspartate transaminase; eGRF CKD-EPI, estimated glomerular filtration rate chronic kidney disease epidemiology collaboration; Hb1Ac, glycated hemoglobin; NSAID, nonsteroidal anti-inflammatory drug; γGT, gamma-glutamyltransferase.
The study was conducted between November 2017 and February 2020. The study was hampered and curtailed prematurely during the coronavirus disease 2019 outbreak in the Netherlands, in accordance with the imposed governmental restrictions in early 2020. Ultimately, 13 participants completed the study protocol, with the fourteenth participant completing only the first research period and wash-out of the study protocol.
Study design
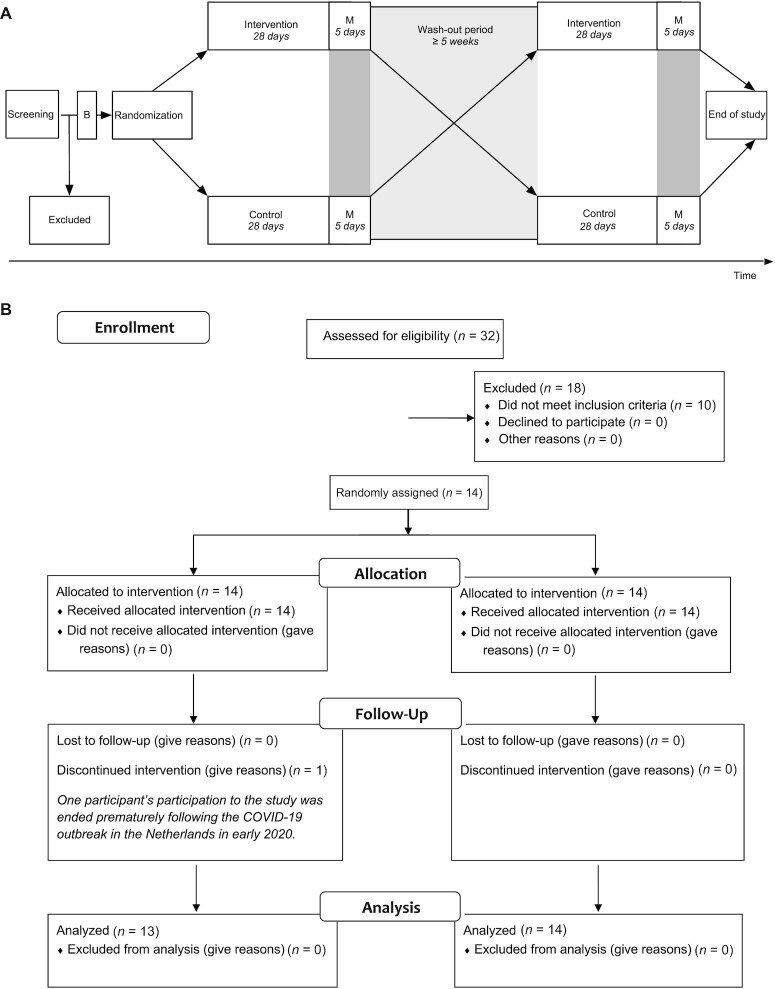
We conducted a double-blind, randomized, controlled, cross-over trial in 13 physically compromised, older adults. Primary outcome parameters were ex and in vivo mitochondrial functions and resting and submaximal exercise energy expenditures. Secondary outcome parameters were glucose tolerance, ectopic fat content, acetyl-L-carnitine levels, and physical function. All other outcome measures were exploratory. Each study period consisted of 32 days of supplementation with either the intervention product (INT) or control (CON), separated by at least 5 weeks of wash-out. We chose this intervention duration based on previous studies in our lab with other nutritional supplements, such as resveratrol, that showed effects on metabolic outcome parameters (28). During the entire study, participants were instructed to maintain their habitual physical activity levels and dietary choices, and no dietary restrictions were imposed. An overview of the study design is presented in Figure 1.
FIGURE 1.
(A) Study timeline and (B) study protocol depicting a CONSORT flow diagram of participant inclusion in the study. Abbreviations: B, baseline measurements preceding randomization; COVID-19, coronavirus disease 2019; M, end of study period measurements on days 29, 31, and 33.
Supplementation protocol
The intervention product (INT) was composed of a whey protein powder as a source of tryptophan, to which NA and NAM were added. In total, the equivalent of 207.5 mg/day niacin equivalents (NE) of NAD+ precursors was supplemented through L-Trp [210 mg/day at a 60:1 conversion rate (29, 30)], NA (4 mg/day), and NAM (200 mg/day). The control product (CON) was composed of an amino acid powder mixture resembling whey protein, but lacking L-Trp, NA, and NAM. Participants received the dietary supplements in a single daily serving, to be ingested during breakfast after dissolving the powder in 200 mL of orange juice. Production and packaging of the supplements were done by Danone Research and approved by the Toxicology and Safety department of Danone Research.
Measurements
Baseline testing
At baseline, participants underwent a maximal cycling test to determine their maximal aerobic capacity and maximal power output on an electronically braked cycle ergometer (Lode Excalibur), as described previously (31). O2 consumption and CO2 production were measured using indirect calorimetry (Omnical, IDEE), and the respiratory exchange ratio (RER) was monitored as a verification of the participant's maximal effort.
Day 1: start of supplementation
Following the baseline cycling test, participants were randomly allocated to the intervention or control arm, and supplementation was initiated. Participants then reported to our facility on Days 1 and 15 to measure blood pressure, to provide a venous blood sample, and to fill out the RAND-36 Health Survey 1.0 questionnaire (32, 33). The venous blood sample was analyzed for standard clinical lab values in the Maastricht Hospital. The participants were randomized in blocks of 4 using the website Randomizer.org for randomization by an independent researcher.
Blood pressure was measured in a sitting position on the nondominant arm with an oscillometric cuff (Omron M6 Comfort, OMRON Healthcare Europe B.V.) in triplicate, from which the mean arterial pressure (MAP) was estimated from the systolic blood pressure (SBP) and diastolic blood pressure (DBP) mean values of the measurement, calculated as MAP = [(2/3 x DBP) + (1/3 x SBP)].
Day 29: hepatic lipid content
On day 29, participants reported to our facility in a fasted state at 07:00 to undergo proton magnetic resonance spectroscopy (1H-MRS) for the quantification of hepatic lipid content [intrahepatic lipid (IHL)] with a 3T (Tesla)whole-body MR system (Achieva 3Tx; Philips Healthcare), as previously described (34). To prevent motion artifacts, participants were asked to breathe in the rhythm of the spectroscopic measurement. Subsequently, the water signal dominating the proton spectra was suppressed using frequency-selective pre-pulses, and the spectra was fitted to quantify the lipid peak. The lipid/water ratio was used as a parameter of intrahepatic lipid content, as previously reported (34).
Next, a venous blood sample was acquired, the RAND-36 Health Survey 1.0 questionnaire was repeated, and the short physical performance battery (SPPB) (35) was performed to assess physical function of the lower extremities. Lastly, blood pressure was measured as described above.
Day 31: muscle volume
The participants underwent another MRI session at 17:00 on Day 31 to determine the muscle volume of the upper leg. Henceforth, the participant was positioned in the 3T MRI scanner (Achieva 3Tx; Philips Healthcare) in the supine position, feet first. T2-weighted MRI measurements were acquired covering from the patella to the art. coxae, allowing muscle volume to be determined using 3D segmentation.
Day 33: submaximal cycling test and skeletal muscle biopsy
On the last day of the study period (Day 33), participants reported to the research facility in the fasted state (from 22:00 the evening before). Body composition was measured by means of air displacement plethysmography (BodPod, COSMED). Subsequently, indirect calorimetry through a ventilated hood system was used to determine the basal metabolic rate (BMR) and RER over a period of 30 minutes.
Next, a skeletal muscle biopsy was obtained from the m. vastus lateralis according to the Bergström method (36) with a side-cutting needle under local anesthesia using 1.0% lidocaine (10 mg/mL) without epinephrine, from which a portion was kept in a preservation medium and freshly used to prepare permeabilized muscle fibers according to Hoeks et al. (37). Subsequently, ex vivo high-resolution respirometry was performed using a 2-chamber Oxygraph (OROBOROS Instruments) using a multiple substrate/inhibitor titration protocol to extensively characterize the mitochondrial capacity of skeletal muscle tissue, as previously reported (38). Measurements were performed in quadruplicate, and the integrity of the outer mitochondrial membrane was assessed by the addition of cytochrome C (CytC) upon maximal coupled respiration (State 3) and was deemed intact if the response to the CytC addition remained under 10%; hence, 16 out of 100 individual measurements were excluded.
Following the muscle biopsy, participants were submitted to a submaximal exercise session of 1 hour on an electrically braked cycling ergometer (Lode Excalibur) at 50% of their power output achieved in the maximal cycling test at baseline. Indirect calorimetry was measured during the second and fourth quarters of the 1-hour session.
Calculations
From the indirect calorimetry data, energy expenditure and substrate oxidation rates were calculated using Weir's formula under the assumption of negligible protein oxidation (39). In addition, gross exercise efficiency (GEE) was calculated as (Wext/Etot) and net exercise efficiency (NEE) was calculated as [Wext/(Etot - Erest)] (40) from the submaximal exercise indirect calorimetry data.
Biochemical analyses
Plasma metabolites
Using EDTA plasma, glucose (Horiba), insulin (Crystal Chem), total free fatty acids (FFA; Wako), and triglycerides (TG; Sigma), concentrations were determined in samples collected on Day 1 and 29 by means of enzymatic quantification with the ABX Pentra C400 (Horiba). All time points of 1 participant were measured in the same run, with insulin and FFA levels measured in duplicates.
Acylcarnitine analysis
We used 2–4 mg of freeze-dried muscle tissue to analyze skeletal muscle acylcarnitines, as previously described (41). Quantitative determination of the propylated acylcarnitines in the medium was performed using tandem MS in an Acquity UPLC System (Waters) coupled to a Quattro Premier XE Tandem Quadrupole Mass Spectrometer (Waters). The internal standards d3-0, d3-3, d3-6, d3-8, d3-10, and d3-16 carnitine were purchased from Dr. Herman J. ten Brink (Vrije Universiteit Medical Hospital).
Enzymatic cyclic assay for quantitative NAD determination
We homogenized 1–5 mg of freeze-dried muscle tissue in 250 μL of 2M HClO4 using a TissueLyser II (Qiagen) bead mill for 5 minutes at 30 pulses per second. After centrifugation at 16,000 g, 100 μL of the acidic supernatant was neutralized by addition of 150 μL 2M/0.6M KOH/MOPS (KOH = potassium hydroxide/MOPS = 3-(N-morpholino)propanesulfonic acid) and centrifuged again to remove precipitated salts. NAD+ content was determined using an enzymatic spectrophotometric cycling assay based on the coupled reaction of malate and alcohol dehydrogenases, as previously described (42). Enzymes used for NAD+ determination (alcohol dehydrogenase from yeast, malate dehydrogenase from pig heart, and glutamate oxaloacetic transaminase from pig liver) were purchased from Sigma.
Metabolomics procedure
We used 2–4 mg of freeze-dried muscle tissue for a metabolomics analysis, as previously described (20). Briefly, the metabolite analysis was carried out in an Aquity UPLC system (Waters) coupled to an Impact II Ultra-High Resolution Qq-Time-Of-Flight MS (Bruker). Chromatographic separation of the compounds was achieved using a SeQuant ZIC-cHILIC column (PEEK 100 × 2.1 mm; 3 μm particle size; Merck) at 30°C. The LC method consisted of a gradient running at 0.25 mL/min from 100% mobile phase B (9:1 acetonitrile: water with 5 mM ammonium acetate pH 6.8) to 100% mobile phase A (1:9 acetonitrile: water with 5 mM ammonium acetate pH 6.8) in 28 minutes, followed by a pre-equilibration step at 100% B of 5 minutes. MS data were acquired both in negative and positive ionization modes in full scan mode over the range of m/z 50–1200.
Statistical analyses
The variation in the primary outcome mitochondrial state 3 respiration on a combination of malate, octanoyl-carnitine, glutamate, and succinate (σ = ∼8 pmol* mg wet tissue–1*min–1) was established previously during pre- and postintervention high-resolution respirometry (43). We have previously shown that the difference in mitochondrial function between healthy and insulin resistance volunteers is ∼ 20%. To detect an ∼10% increase in mitochondrial function (μ = ∼6.0 pmol* mg wet tissue–1*min–1)—which we believe is physiologically relevant in this population and which is achievable with a short-term nutritional intervention as previously demonstrated (28)—14 participants must be included with a power (π) of 80% and a 2-sided significance level (α) of 0.05.
The statistical analysis was performed in SPSS 26 for Mac OS (IBM SPSS Statistics) and graphs were made in Prism 8.4.2 for Windows (GraphPad Software, Inc.). Descriptive statistics were reported for all data parameters as means ± SDs, unless otherwise stated. Normality of the data distributions was evaluated with the Kolmogorov-Smirnov normality test and deemed significant at a P value < 0.05. The paired Student's t-test was used to compare the results of the intervention to the control, or the equivalent Wilcoxon signed-rank test was used if normality was not met. In the event of missing values, the values were not replaced and the participant in question did not contribute to the analysis of that outcome; hence, all analyses encompass 13 data points, unless stated otherwise. No carry-over or order effect was detected. In addition, Pearson correlation coefficients were computed. Bonferroni corrections were imposed to correct for multiple testing of single outcome measures. Moreover, due to multiplicity of the primary outcomes, the type 1 error was corrected for using a Bonferroni correction as (alpha/n). Statistical significance was achieved at a P value < 0.05.
Results
NAD+-precursor supplementation increased methyl-nicotinamide but not NAD+ bioavailability in skeletal muscle
Compliance to the supplement use was estimated by the fraction of sachets returned compared to the number of sachets provided to the participants. For the supplementation with precursors (INT), 100% of all sachets were consumed, compared to 99% (range, 97%–100%) in the control condition (CON). Overall, the products were well tolerated, with only 1 participant reporting cold sores (herpes labialis), which resolved without intervention. Subject characteristics at the start of the study are summarized in Table 1. No carry-over or order effect was detected for any of the primary outcome parameters.
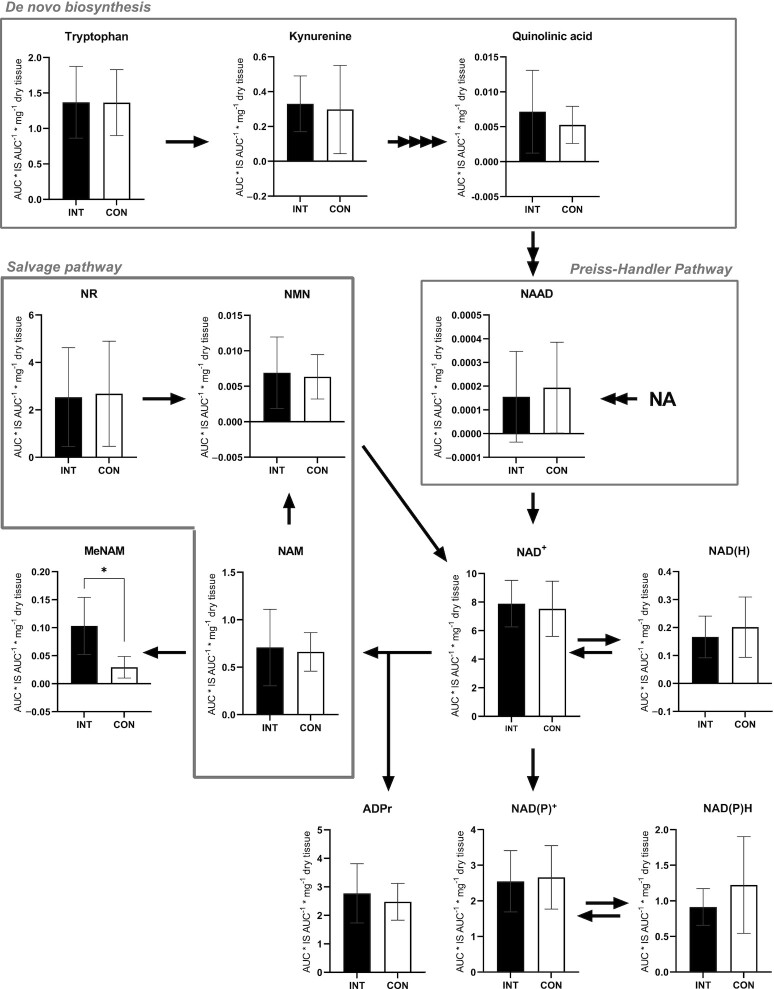
In order to determine the efficacy of the NAD+-precursor supplementation, the concentrations of NAD+ and several metabolites of the NAD+ synthesis pathways were measured in skeletal muscle tissue, obtained from the m. vastus lateralis. These analyses showed that NAD+ levels were similar between INT and CON (P = 0.123; Figure 2).
FIGURE 2.
NAD+ metabolome in skeletal muscle tissue obtained from older adults with compromised physical function administered supplemental niacin equivalents and placebo, each for 32 days. The y-axis defines the AUC (AUC* IS AUC–1) per mg of dry tissue used in the MS analysis. Data are displayed as means ± SDs, n = 11. *P < 0.01. Abbreviations: ADPr, adenosine diphosphate riboside; AUC, area under curve; CON, control; INT, NAD intervention; IS, internal standard; MeNAM, methyl-nicotinamide; NA, nicotinic acid; NAAD, nicotinic acid adenine dinucleotide; NAM, nicotinamide; NMN, nicotinamide mononucleotide; NR, nicotinamide riboside.
From the de novo biosynthesis pathway, L-Trp, kynurenine, and quinolinic acid (QA) did not differ between INT and CON (P ≥ 0.05; n = 11; Figure 2). Synthesis of NAD+ from NA through the Preiss-Handler pathway leads to synthesis of nicotinic acid adenine dinucleotide (NAAD), which was not different between INT and CON (P = 0.262; n = 11; Figure 2). NAD+ can also be salvaged from NMN, NAM, and NR, but no differences between INT and CON were observed in these precursors either (P ≥ 0.05; n = 11; Figure 2). Consumption of NAD yields NAM and ADP riboside (ADPr), with no differences observed in ADPr levels between INT and CON (P = 0.413; n = 11; Figure 2). Conversely, the concentration of the primary breakdown product of NAM, methyl-NAM (MeNAM), which is also commonly described as a marker for increased NAD turnover (22), was measured and revealed significantly higher MeNAM concentrations in INT compared to CON (P = 0.001; Figure 2). Alternatively, NAD+ can be phosphorylated into NAD(P)+, which did not differ between conditions (P = 0.833; n = 11; Figure 2). The respective reduced forms, NAD(H) and NAD(P)H, also did not differ between conditions (P = 0.389 and P = 0.365 respectively; n = 11; Figure 2).
Skeletal muscle acylcarnitine levels were unaffected by NAD+-precursor supplementation
We previously showed that NR supplementation in humans altered acylcarnitine levels in skeletal muscle (20). Therefore, acylcarnitine levels were determined in the skeletal muscle tissue and species were categorized in subgroups as being free (C0), independent (C2), short-chain (C3–C5), medium-chain (C6–C12), and long-chain (C13–C20) acylcarnitines. Ultimately, no differences were observed in the concentrations of acylcarnitine subgroups between INT and CON (P ≥ 0.05; Figure 3A–D; n = 11).
FIGURE 3.
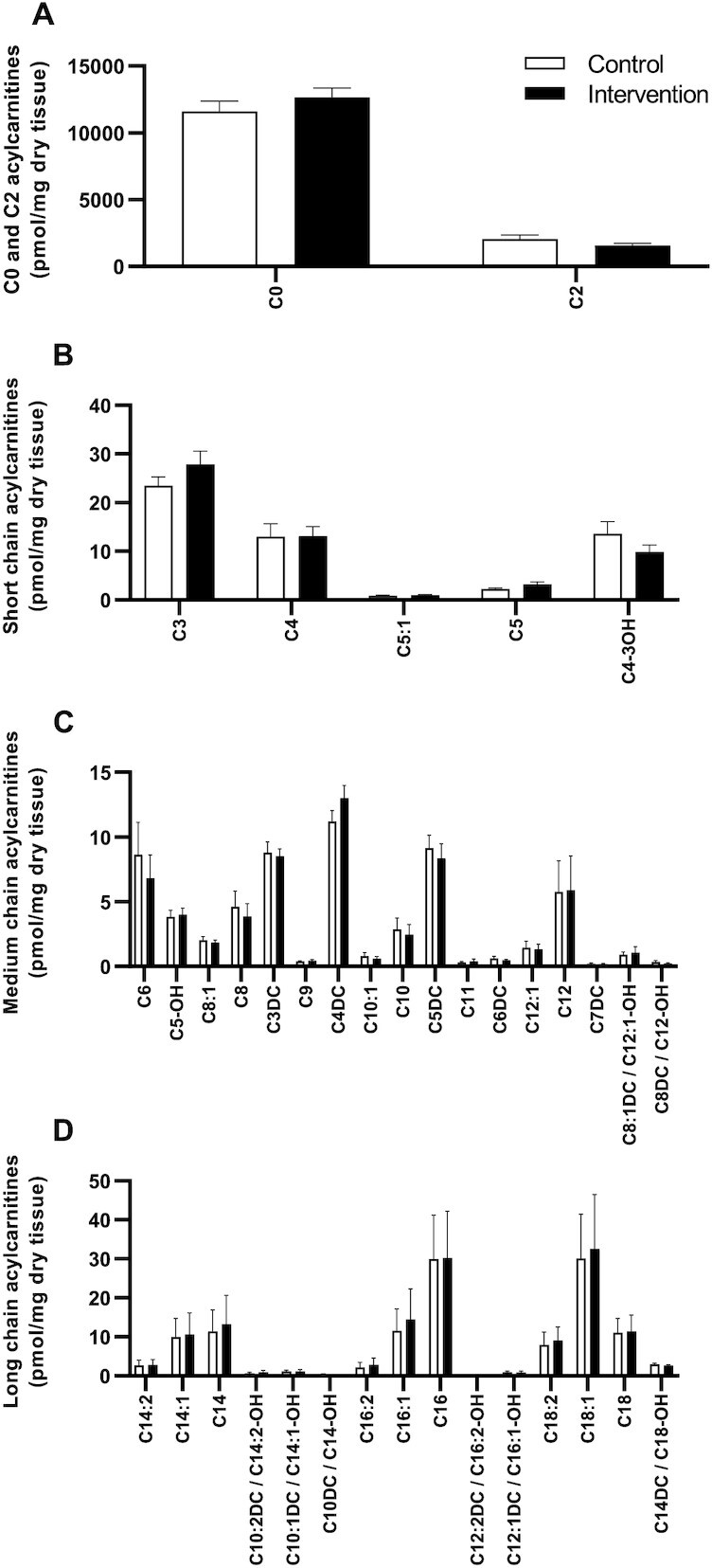
Skeletal muscle acylcarnitine levels in older adults with compromised physical function administered supplemental niacin equivalents and placebo. (A) Acylcarnitine levels in skeletal muscle for free (C0) and independent species (C2). Acylcarnitine levels for (B) short chain, (C) medium chain, and (D) long chain species (LC). Data are displayed as means ± SDs, n = 11.
Body composition was not affected by NAD+-precursor supplementation
Body weight did not differ between INT (73.0 kg; 95% CI: 68.6 kg, 77.5 kg) and CON (72.9 kg; 95% CI: 68.6 kg,77.2 kg; P = 0.812). Also, body composition was unaffected by NAD+-precursor supplementation, as fat percentages were not different for INT and CON [34.2% (95% CI: 29.3%, 39.2%) and 33.3% (95% CI: 27.6%, 38.9%), respectively; P = 0.121].
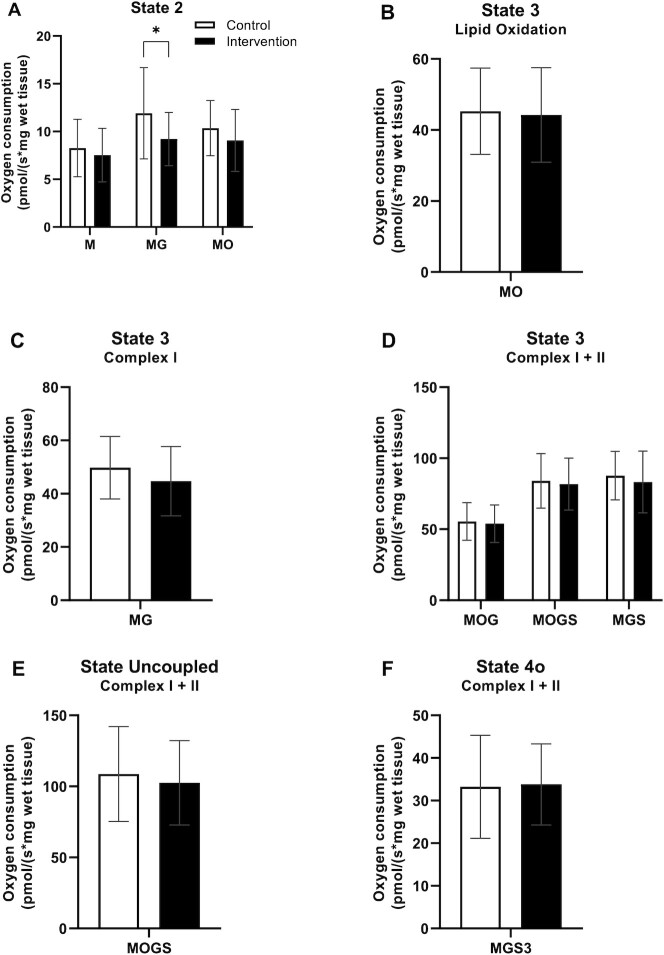
NAD+-precursor supplementation did not improve overall ex vivo mitochondrial respiration capacity
To determine skeletal muscle mitochondrial capacity, we performed ex vivo high-resolution respirometry on permeabilized muscle fibers obtained from skeletal muscle biopsies. In the presence of substrates alone (State 2, Figure 4A), oxygen flux was similar in INT and CON, although a slight but significant difference of malate + glutamate (MG; P = 0.010) was observed between INT and CON. Also, the assessment of ADP-stimulated (State 3) respiration fueled by complex I-linked substrates (malate and octanoyl-carnitine; Figure 4B, P = 0.882) did not reveal any differences between INT and CON. If anything, state 3 respiration upon MG tended to be lower in INT compared with CON (P = 0.054; Figure 4C). State 3 respiration upon parallel electron input into complexes I and II showed no differences between INT and CON with the substrate combinations malate + octanoyl-carnitine + glutamate (P = 0.753), malate + octanoyl-carnitine + glutamate + succinate (P = 0.716), or malate + glutamate + succinate (P = 0.388; Figure 4D). Maximal uncoupled respiration (State 3U), a reflection of the maximal capacity of the electron transport chain, did not differ between INT and CON (P = 0.495; Figure 4E). Lastly, mitochondrial proton leak (State 4o) was assessed through the addition of oligomycin and did not differ between INT and CON (P = 0.888; Figure 4F). CytC responses were, on average, 1.32% for INT and 0.73% for CON, and did not differ between conditions (P = 0.368).
FIGURE 4.
States of mitochondrial respiration of skeletal muscle tissue obtained from older adults with compromised physical function administered supplemental niacin equivalents and placebo, each for 32 days. (A) State 2 respiration. (B) State 3 respiration under ADP stimulation with lipid substrates. (C) State 3 respiration with substrates focusing on Complex I. (D) State 3 respiration with parallel substrates focus on Complex I and II. (E) Uncoupled respiration under FCCP addition. (F) State 4o respiration uncoupled from ATP synthase. Values are represented as means ± SDs, n = 13. *P < 0.05. Abbreviations: FCCP, carbonylcyanide-4-(trifluoromethoxy)-phenylhydrazone; G, glutamate; M, malate; O, octanoyl-carnitine; S, succinate.
NAD+-precursor supplementation did not affect plasma substrate concentrations
Plasma samples obtained on Day 29 of each research period were analyzed for the concentrations of glucose, insulin, FFA, and TG. Glucose concentrations did not differ for INT (5.87 mmol/L; 95% CI: 5.57, 6.16 mmol/L) and CON (5.76 mmol/L; 95% CI: 5.52, 6.01 mmol/L; P = 0.223). Correspondingly, insulin levels were not different between INT (9.41 mU/L; 95% CI: 4.18, 14.7 mU/L) and CON (9.12 mU/L; 95% CI: 5.40, 12.8 mU/L; P = 0.807). Concentrations of FFA showed no differences between INT (496 μmol/L; 95% CI: 397, 595 μmol/L) and CON (512 μmol/L; 95% CI: 416, 609 μmol/L; P = 0.812]. Lastly, plasma TG levels were not different between INT (0.830 mmol/L; IQR, 0.675–1.03 mmol/L) and CON (0.990 mmol/L; IQR, 0.610–1.43 mmol/L; P = 0.266).
Basal metabolic rate and exercise efficiency were not altered by NAD+-precursor supplementation
Basal metabolic rates and fasting RERs were similar between INT and CON (P = 0.824 and P = 0.297, respectively; Figure 5A and B). As a result, neither carbohydrate nor lipid oxidation in the fasted state was different between INT and CON [P = 0.588 (Figure 5C) and P = 0.787 (Figure 5D), respectively].
FIGURE 5.
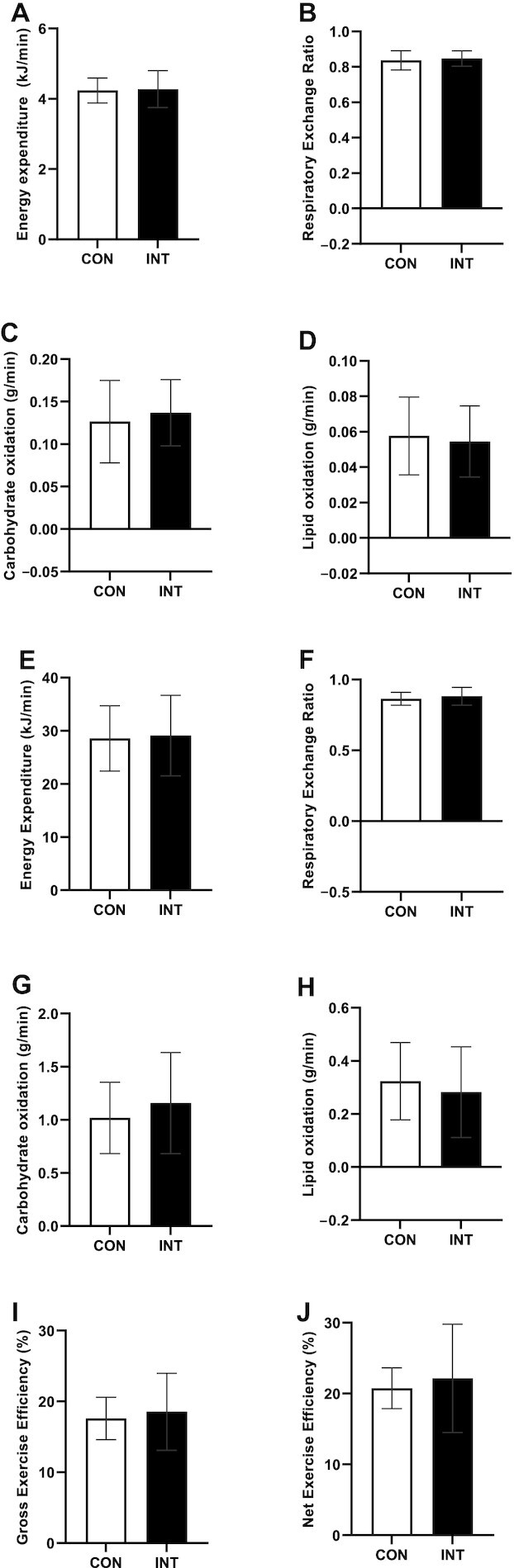
Whole-body indirect calorimetry of older adults with compromised physical function administered supplemental niacin equivalents and placebo. (A) Basal metabolic rate and (B) respiratory exchange ratio, with corresponding (C) carbohydrate oxidation rate and (D) lipid oxidation rate. (E) Submaximal exercise energy expenditure with corresponding (F) respiratory exchange ratio with corresponding (G) carbohydrate and (H) lipid oxidation rates. (I) Gross and (J) net exercise efficiency from submaximal cycling test. Data are represented as means ± SDs, n = 13. Abbreviations: CO, control; INT, NAD intervention.
Besides assessments in resting conditions, substrate utilization was also determined during a submaximal cycling test. All participants completed the first 30 minutes of the cycling test; however, 6 out of 14 participants could not proceed with the cycling test at the set resistance and either decided to cease testing (n = 2) or required the resistance to be reduced (n = 4) in order to complete the remaining 30 minutes of the test. Therefore, we only report parameters measured in the first half-hour of the cycling test.
Besides substrate utilization, GEE and NEE were also evaluated during the submaximal cycling test. The total energy expenditure during cycling did not differ between INT and CON (P = 0.103; Figure 5E), nor did cycling RER, carbohydrate oxidation, or lipid oxidation rates (P = 0.366, 0.186, and 0.466, respectively; Figure 5F–H). GEE did not differ between conditions (P = 0.419; Figure 5I). NEE (i.e., GEE corrected for the BMR) was also not different between conditions (P = 0.636; Figure 5J).
Intrahepatic lipid content and muscle volume were not affected by NAD+-precursor supplementation
Intrahepatic lipid content was measured noninvasively by 1H-MRS and did not differ between INT (2.8%; 0.8%–4.8%) and CON (2.7%; 1.1%–4.3%; P = 0.624). Muscle volume of the m. vastus lateralis, assessed by MRI, was not different for INT (1700 cm3; 1440–1960 cm3) compared to CON (1710 cm3; 1450–1980 cm3; n = 11; P = 0.450).
NAD+-precursor supplementation did not affect blood pressure
SBP and DBP were measured throughout each study period to evaluate the effects of NAD+-precursor supplementation on these parameters. A summary of the data is presented in Table 2. No differences were observed between INT and CON in SBP on Day 1 (P = 0.821), Day 15 (P = 0.741), or Day 29 (n = 12; P = 0.877). DBP did not differ between conditions on Day 1 (P = 0.551), Day 15 (P = 0.733), or Day 29 (n = 12; P = 0.433). Lastly, MAP was estimated from the acquired SBP and DBP values and was not different for INT and CON on Day 1 (P = 0.653), Day 15 (P = 0.976), or Day 29 (n = 12; P = 0.738).
TABLE 2.
Blood pressure in older adults with compromised physical function administered supplemental niacin equivalents and placebo, each for 32 days
| Intervention group | Control group | P value | |
|---|---|---|---|
| SBP, mmHg | |||
| Day 1 | 140 (131–148) | 141 (131–151) | 0.821 |
| Day 15 | 140 (131–149) | 139 (129–148) | 0.741 |
| Day 29 | 136 (128–145) | 137 (130–143) | 0.877 |
| DBP, mmHg | |||
| Day 1 | 82.9 (77.6–88.2) | 84.3 (80.0–88.6) | 0.551 |
| Day 15 | 82.9 (78.1–87.8) | 83.8 (78.5–89.0) | 0.733 |
| Day 29 | 81.1 (75.1–87.0) | 79.4 (74.8–84.1) | 0.433 |
| MAP, mmHg | |||
| Day 1 | 102 (97–107) | 103 (97–109) | 0.653 |
| Day 15 | 102 (97–107) | 102 (96–108) | 0.976 |
| Day 29 | 99.4 (93.5–105) | 98.5 (94.0–103) | 0.738 |
Values are presented as means (95% CIs), n = 13 or 12 (Day 29).
Abbreviations: DBP, diastolic blood pressure; MAP, mean arterial pressure; SBP, systolic blood pressure.
SPPB performance did not improve under NAD+-precursor supplementation
The SPPB was used as a clinical measure of physical functioning. Scores for the balance test, the walking speed, and the chair-rise test were similar between INT and CON (P > 0.999, P = 0.500, and P = 0.489, respectively). The overall score on the SPPB was also not different between INT (11.0; IQR, 10.00–11.5) and CON (11.0; IQR, 9.50–12.0; P > 0.999).
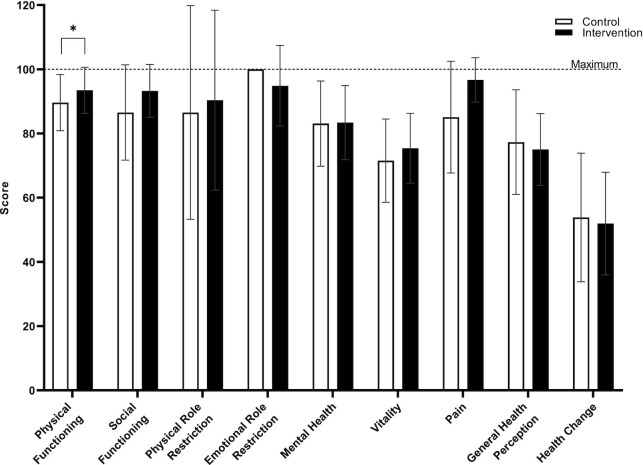
Perceived physical functioning was positively influenced by NAD+-precursor supplementation
The RAND-36 Health Survey 1.0 questionnaire was used as a measure of perceived health status. Although measured physical functioning was unaffected by NAD+-precursor supplementation, significantly higher scores were observed for the perceived physical functioning domain in INT (92.0; IQR, 90.0–100) as compared to CON (90.0; IQR, 86.3–93.8; n = 12; P = 0.031; Figure 6). However, the other 8 out of 9 domains of the RAND-36 did not show any differences between INT and CON on Day 29 (P ≥ 0.05; Figure 6).
FIGURE 6.
RAND-36 Health Survey 1.0 questionnaire of older adults with compromised physical function administered supplemental niacin equivalents and placebo over 8 domains on Day 29. All domains can be scored between 0 and 100, with higher scores being better. Data are represented as means ± SDs, n = 12, *P < 0.05.
Discussion
Mitochondrial dysfunction is an established hallmark of aging and, together with declining NAD+ levels, these factors are thought to drive the age-dependent decline in skeletal muscle physical function. Here, we hypothesized that supplementation with the NAD+ precursors L-Trp, NA, and NAM, in a dose that could be achieved through increased dietary intake, would improve mitochondrial oxidative capacity in community-dwelling, older adults with impaired physical function. We tested this hypothesis by conducting a double-blind, randomized, controlled, cross-over trial with detailed metabolic phenotyping. We show that supplementing with L-Trp, NA, and NAM does elevate MeNAM concentrations in skeletal muscle tissue but, in discordance with our hypothesis, we found no improvement in the mitochondrial respiration capacity in older adults with an impaired physical function.
In the current study, we did not observe an improvement in the mitochondrial respiratory capacity in skeletal muscle under NAD+-precursor supplementation compared to the control condition. We have previously shown that the NAD+ precursor acipimox was able to improve the mitochondrial function in T2DM patients (44). However, the lack of an effect of NAD+-precursor supplementation in the current study is in accordance with similar recent observations with NR supplementation in individuals with obesity (45, 46) and in older adults (22), and extends this observation to physically compromised older adults. Along the same lines, SPPB performance, energy expenditure, substrate utilization, and exercise efficiency were all unaffected by NAD+-precursor supplementation. These findings are in agreement with those of Martens et al. (19), who investigated the effects of NR supplementation in healthy, lean, middle-aged to older adults on parameters of physical function such as resting energy expenditure, maximum aerobic capacity, and body composition, and found no differences between NR and placebo. In accordance, Elhassan et al. (22) showed that 3 weeks of NR supplementation does not increase handgrip strength in older adults. In contrast to our previous finding that NR supplementation resulted in a minor but significant reduction in fat mass in middle-aged humans with obesity (45), we did not find any effect of NAD+-precursor supplementation on body composition in the current study. Also, the muscle volume was similar in both conditions. Interestingly, although no improvements in physical function were detected upon NAD+-precursor supplementation, perceived physical function was improved upon NAD+. Possibly, subtle changes in physical function did occur that were not detected by our outcome parameters. However, given the small number of volunteers in our study and the fact that this conclusion was based on 1 out of 9 domains of the RAND-36 survey, one should be cautious in interpreting this result. Future studies should determine both perceived compared with actual physical function in (NAD+) supplementation studies.
Blood pressure has also been reported to be susceptible to NAD+-precursor supplementation in humans (19). However, we did not observe changes in SBP, DBP, or MAP under NAD+-precursor supplementation. This is in agreement with our previous NR supplementation study in healthy humans with obesity, which revealed no improvements in SBP, DBP, or MAP when performing 36-hour blood pressure measurements (45). These data, however, conflict with previous findings from Martens et al. (19) showing improvements in SBP and DBP in healthy middle-aged to older adults following 6 weeks of NR supplementation, which may be explained by discrepancies in the methodologies used to measure blood pressure, such as the higher dose used and duration of the supplementation.
Several studies have demonstrated the ability of NAD+-precursor supplementation to reduce IHL content in mice (47–49). Moreover, Dollerup et al. (21) reported a tendency for IHL content to decline in humans with obesity following 12 weeks of NR supplementation. Although this observation did not reach statistical significance, the majority of the NR-supplemented group (13 out of 20 participants) had an IHL content greater than 5% prior to supplementation, and 9 out of these 13 individuals had a reduced IHL content after supplementation. Here, we did not observe a beneficial effect of NAD+-precursor supplementation on IHL content. This is possibly due to the already low IHL content in our cohort, with only 2 participants displaying IHL contents greater than 5%, which is representative of nonalcoholic fatty liver disease (50), thereby limiting the potential for a reduction in IHL content.
It was previously shown that aging is associated with reduced intracellular NAD+ levels in skeletal muscle in humans (14, 15). It is now generally accepted that the NAD+ metabolism is a main pathway in the regulation of mitochondrial function in skeletal muscle and would be a target for intervention to stimulate mitochondrial function and thereby prevent age-related declines in physical muscle function. In this light, Pirinen et al. (51) have shown that a titration protocol of 250 mg/day to 1000 mg/day of niacin over 4 weeks is able to rescue mitochondrial respiration in adult-onset mitochondrial myopathy patients, demonstrating the potential and efficacy of NAD+-precursor supplementation in humans with preexisting conditions. Notably, the window of opportunity for NAD+-precursor supplementation in this population with niacin deficiency by far exceeds the possibilities in populations who are not niacin deficient. Here, we studied a group of community-dwelling, older adults with an impaired physical function and, in measuring the NAD+ levels in skeletal muscle tissue, we found that NAD+ concentrations in skeletal muscle remained unaffected upon supplementation. We supplemented with 3 separate NAD+ precursors—L-Trp, NA, and NAM—aiming to take advantage of their respective pathways to contribute to the overall NAD+ pool, as illustrated in Figure 2. It has previously been shown that NAM supplementation to cells and mice leads to an extended lifespan and healthspan, respectively (52, 53). Furthermore, NAM supplementation has been shown to improve the glucose metabolism in a rodent model of obesity and T2DM (54). Recently, de novo biosynthesis of NAD+ from L-Trp has received more attention as a viable dietary source of NAD+ (25). Katsyuba et al. (25) demonstrated that when the ACMSD enzyme is inhibited in the liver and kidney, the conversion of L-Trp to NAD+ has the potential to induce SIRT1 activation and, thereby, improve mitochondrial function. Here, we did not observe an increase in metabolites belonging to the kynurenine pathway from L-Trp to QA and subsequently to NAAD, a suggested sensitive biomarker of NAD+-precursor supplementation efficacy (55). It is possible that the majority of the L-Trp ingested was consumed by the ACSMD enzyme instead of allowing for spontaneous cyclization of ACSM into QA, further enzymatic conversion to NAAD, and ultimately NAD+. However, this remains speculative, as this can only be investigated by acquiring liver tissue, which we did not do in this study. Therefore, more human studies are needed to examine the effects of L-Trp and NAM, given at higher doses, on metabolic health outcomes.
Moreover, we opted to use a low dose of 4 mg of NA daily in order to limit the occurrence of flushing due to its vasoactive properties (56). NAAD is the point of convergence for L-Trp and NA metabolism towards NAD+ synthesis. However, NAAD concentrations did not increase in skeletal muscle, suggesting either an insufficient dose or the preference of skeletal muscle tissue for another NAD+ precursor. Additionally, Liu et al. (57) showed through isotope-tracer methodology that L-Trp, NA, and NAM are the predominant physiological circulatory NAD+ precursors. Labeling of L-Trp and NA showed that these precursors are converted to NAD+ in the liver and subsequently excreted into the circulation as NAM, and that the NAM exchange from circulation to skeletal muscle tissue is slow, in addition to skeletal muscle tissue exhibiting a low turnover rate of NAD+ (57). This may explain why we did not observe an increase in NAM, or any other metabolite of the NAD+ precursors used in our supplementation protocol, in skeletal muscle tissue in this study. Herein, our results contrast with observations of increasing NAAD in skeletal muscle previously made by us and others when supplementing with high doses of NR in individuals with obesity (45) and aged individuals (22), suggesting that NAAD may be a sensitive yet specific biomarker for NR supplementation efficacy.
The concentrations of NAD+ in skeletal muscle tissue observed in both conditions of the current study appear to be comparable to concentrations found previously by us in a group of healthy, middle-aged individuals with obesity supplemented with 1000 mg/day of NR, who demonstrated unresponsive NAD+ levels to NR supplementation compared to placebo (20). This raises the question of whether NAD+ levels per se are determinants of efficacy of NAD+-precursor supplementation or whether fluxes of NAD+ are more reliable measures of efficacy. We found that the NAD+ concentrations in skeletal muscle tissue were unresponsive to supplementation under differing supplementation strategies in 2 different populations [the present study and Remie et al. (20)] and observed increased levels of MeNAM in both studies. This observation is also in line with the observations of Elhassan et al. (22) in NR-supplemented older adults, showing increased MeNAM levels in skeletal muscle, although admittedly unchanged NAD+ concentrations. From the above, one could speculate that the concentration of NAD+ in skeletal muscle tissue is likely to be near the homeostatic maximum in humans in the absence of disease or niacin deficiency, as illustrated by the study of Pirinen et al. (51). This has also been proposed in the “second hit” theory by Elhassan et al. (22), in which NAD+ levels may only decline if chronological age is struck by a second trigger leading to metabolic stress, such as physical inactivity or (chronic) disease. In accordance with this reasoning, we previously showed that supplementation with the synthetic NA analogue acipimox increased the skeletal muscle mitochondrial respiration capacity in individuals with T2DM, who are characterized by a relatively low mitochondrial capacity compared to BMI-matched healthy controls (44). From the above, it could be concluded that there is potentially little room for improvement in skeletal muscle NAD+ levels in healthy individuals through supplementation with NAD+ precursors and, as a consequence, little or no effect on downstream targets such as mitochondrial function, such as was observed in the current study.
We acknowledge that this study is susceptible to limitations. The participants may represent the “cream of the crop” in terms of older adults with an impaired physical function. As per the 6MWT distances recorded during the screening, the average walking speed in this cohort was 1.09 m/s, fitting of a healthy older (58) and prefrail population (59). Although the individual 6MWT performances met our inclusion criterium, performance on the maximal cycling test yielded a larger variation in overall maximum cycling performance, leading to a more heterogeneous cohort than expected. Walking performance is a good predictor of physical function and frailty, comorbidity, and death (58), but it is hard to distinguish whether walking performance is limited by muscle function or may be limited by other health problems; for example, a decreased walking performance does not necessarily have to translate to a reduced cycling performance, as joint issues could be the limiting factor and may be less prominent during cycling compared to walking. Secondly, the selected dose of 207.5 mg/day NE, although 12–15 times the RDA for vitamin B-3, may be insufficient to elevate NAD+ levels in humans. The RDA of niacin for adult humans is set at 16 mg NE for males and 14 mg for females (17). However, no guidelines exist for older adults specifically. To date, only a small number of studies have looked into the NAD+ boosting ability of lower doses of NAD+ precursors in humans (23, 24), and found that doses of 100 mg/day, 250 mg/day, and 300 mg/day NE are able to increase plasma NAD+ metabolite levels over baseline, such as levels of NAM and MeNAM. However, these studies did not investigate intracellular levels in target tissues, such as skeletal muscle. Here, we have supplemented older adults with 207.5 mg NE per day, a level of supplementation emulating an increased dietary intake, and show that this is insufficient to increase levels of NAD+ and its metabolites in skeletal muscle tissue but does increase skeletal muscle MeNAM levels, suggesting an increase in the skeletal muscle NAD+ metabolism. Further, the duration of the intervention was set at 32 days, which may be relatively short for health benefits to surface. However, we have shown that 30 days of resveratrol supplementation in individuals with obesity improves the mitochondrial respiration capacity (28), supporting the notion that short-term interventions can positively affect the human metabolism. Lastly, we cannot exclude that the sample size of the study was too small, although we have previously shown with a similar cross-over design and the same methodology that differences upon nutritional supplementation can be observed with a similar sample size. Also, the chance of making a type I error due to testing of multiple study outcomes cannot be excluded.
In summary, we investigated the efficacy of NAD+ precursor supplementation on the mitochondrial function, aiming to combat the age-dependent declines in skeletal muscle function. We show in community-dwelling, older adults with impaired physical function that L-Trp, NA, and NAM did increase MeNAM levels in skeletal muscle tissue but did not improve the mitochondrial function and skeletal muscle function. Notwithstanding, further research is merited to elucidate the full potential of NAD+-precursor supplementation in specific groups with more prominent metabolic dysfunction, such as those with (morbid) obesity and T2DM. Future studies must also tailor their interventions towards the tissues targeted, optimizing delivery and taking the fate of the chosen NAD+ precursor into account.
Acknowledgments
The authors’ responsibilities were as follows – NJC, LG, JdV, RH, JG, BH, JH, PS: designed research; NJC, LG, CEF, YMHB: performed research; NJC, EM-K, YMHB, VBS-H, BH, RZ-P, RHH, JH, PS: analyzed samples and data; NJC, JH, PS: wrote the manuscript; PS: is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis; and all authors: critically reviewed the manuscript and read and approved the final manuscript.
Notes
This work was supported by the Top Institute Food and Nutrition (TIFN) research program on Mitochondrial Health under grant number 16NH01.
Author disclosures: The authors report no conflicts of interest.
Abbreviations used: 1H-MRS, 1H-magnetic resonance spectroscopy; 6MWT, 6-minute walk test; ACMSD, α-amino-β-carboxymuconate-ε-semialdehyde decarboxylase; ADPr, ADP riboside; BMR, basal metabolic rate; CytC, cytochrome C; DBP, diastolic blood pressure; FFA, free fatty acids; GEE, gross exercise efficiency; IHL, intrahepatic lipid; INT, intervention; L-Trp, L-tryptophan; MAP, mean arterial pressure; MeNAM, methyl-nicotinamide; MG, malate + glutamate; NA, nicotinic acid; NAAD, nicotinic acid adenine dinucleotide; NAM, nicotinamide; NE, niacin equivalent; NEE, net exercise efficiency; NMN, nicotinamide mononucleotide; NR, nicotinamide riboside; QA, quinolinic acid; RER, respiratory exchange ratio; SBP, systolic blood pressure; SIRT, sirtuin; SPPB, short physical performance battery; T2DM, type 2 diabetes mellitus; TG, triglycerides.
Contributor Information
N J Connell, Department of Nutrition and Movement Sciences, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University, Maastricht, The Netherlands.
L Grevendonk, Department of Nutrition and Movement Sciences, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University, Maastricht, The Netherlands.
C E Fealy, Department of Nutrition and Movement Sciences, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University, Maastricht, The Netherlands.
E Moonen-Kornips, Department of Nutrition and Movement Sciences, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University, Maastricht, The Netherlands.
Y M H Bruls, Department of Radiology and Nuclear Medicine, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University Medical Center+, Maastricht, The Netherlands.
V B Schrauwen-Hinderling, Department of Nutrition and Movement Sciences, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University, Maastricht, The Netherlands; Department of Radiology and Nuclear Medicine, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University Medical Center+, Maastricht, The Netherlands.
J de Vogel, Danone Nutricia Research, Utrecht, The Netherlands.
R Hageman, Danone Nutricia Research, Utrecht, The Netherlands.
J Geurts, Friesland Campina, Amersfoort, The Netherlands.
R Zapata-Perez, Laboratory Genetic Metabolic Diseases, Amsterdam University Medical Center, University of Amsterdam, Amsterdam Gastroenterology, Endocrinology and Metabolism, Amsterdam Cardiovascular Sciences, Amsterdam, The Netherlands.
R H Houtkooper, Laboratory Genetic Metabolic Diseases, Amsterdam University Medical Center, University of Amsterdam, Amsterdam Gastroenterology, Endocrinology and Metabolism, Amsterdam Cardiovascular Sciences, Amsterdam, The Netherlands.
B Havekes, Department of Nutrition and Movement Sciences, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University, Maastricht, The Netherlands; Department of Internal Medicine, Division of Endocrinology, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University Medical Center+, Maastricht, The Netherlands.
J Hoeks, Department of Nutrition and Movement Sciences, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University, Maastricht, The Netherlands.
P Schrauwen, Department of Nutrition and Movement Sciences, NUTRIM School of Nutrition and Translational Research in Metabolism, Maastricht University, Maastricht, The Netherlands.
References
- 1.Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153(6):1194–217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Roubenoff R. Sarcopenia and its implications for the elderly. Eur J Clin Nutr. 2000;54(S3):S40–7. [DOI] [PubMed] [Google Scholar]
- 3.Crescenzo R, Bianco F, Mazzoli A, Giacco A, Liverini G, Iossa S. Skeletal muscle mitochondrial energetic efficiency and aging. Int J Mol Sci. 2015;16(12):10674–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gomes AP, Price NL, Ling AJ, Moslehi JJ, Montgomery MK, Rajman L, White JP, Teodoro JS, Wrann CD, Hubbard BPet al. Declining NAD(+) induces a pseudohypoxic state disrupting nuclear-mitochondrial communication during aging. Cell. 2013;155(7):1624–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Conley KE, Jubrias SA, Esselman PC. Oxidative capacity and ageing in human muscle. J Physiol. 2000;526(1):203–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kim Y, Triolo M, Hood DA. Impact of aging and exercise on mitochondrial quality control in skeletal muscle. Oxid Med Cell Longev. 2017;2017:3165396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Andreux PA, Houtkooper RH, Auwerx J. Pharmacological approaches to restore mitochondrial function. Nat Rev Drug Discovery. 2013;12(6):465–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Camacho-Pereira J, Tarrago MG, Chini CCS, Nin V, Escande C, Warner GM, Puranik AS, Schoon RA, Reid JM, Galina Aet al. CD38 dictates age-related NAD decline and mitochondrial dysfunction through an SIRT3-dependent mechanism. Cell Metab. 2016;23(6):1127–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhu XH, Lu M, Lee BY, Ugurbil K, Chen W. In vivo NAD assay reveals the intracellular NAD contents and redox state in healthy human brain and their age dependences. Proc Natl Acad Sci. 2015;112(9):2876–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yoshino J, Mills KF, Yoon MJ, Imai S. Nicotinamide mononucleotide, a key NAD(+) intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice. Cell Metab. 2011;14(4):528–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mills KF, Yoshida S, Stein LR, Grozio A, Kubota S, Sasaki Y, Redpath P, Migaud ME, Apte RS, Uchida Ket al. Long-term administration of nicotinamide mononucleotide mitigates age-associated physiological decline in mice. Cell Metab. 2016;24(6):795–806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Houtkooper RH, Pirinen E, Auwerx J. Sirtuins as regulators of metabolism and healthspan. Nat Rev Mol Cell Biol. 2012;13(4):225–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Canto C, Auwerx J. Targeting sirtuin 1 to improve metabolism: All you need is NAD(+)?. Pharmacol Rev. 2012;64(1):166–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.de Guia RM, Agerholm M, Nielsen TS, Consitt LA, Sogaard D, Helge JW, Larsen S, Brandauer J, Houmard JA, Treebak JT. Aerobic and resistance exercise training reverses age-dependent decline in NAD(+) salvage capacity in human skeletal muscle. Physiol Rep. 2019;7(12):e14139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Massudi H, Grant R, Braidy N, Guest J, Farnsworth B, Guillemin GJ. Age-associated changes in oxidative stress and NAD+ metabolism in human tissue. PLoS One. 2012;7(7):e42357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mouchiroud L, Houtkooper RH, Auwerx J. NAD(+) metabolism: A therapeutic target for age-related metabolic disease. Crit Rev Biochem Mol Biol. 2013;48(4):397–408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bogan KL, Brenner C. Nicotinic acid, nicotinamide, and nicotinamide riboside: A molecular evaluation of NAD+ precursor vitamins in human nutrition. Annu Rev Nutr. 2008;28:115–30. [DOI] [PubMed] [Google Scholar]
- 18.Zhang H, Ryu D, Wu Y, Gariani K, Wang X, Luan P, D'Amico D, Ropelle ER, Lutolf MP, Aebersold Ret al. NAD(+) repletion improves mitochondrial and stem cell function and enhances life span in mice. Science. 2016;352(6292):1436–43. [DOI] [PubMed] [Google Scholar]
- 19.Martens CR, Denman BA, Mazzo MR, Armstrong ML, Reisdorph N, McQueen MB, Chonchol M, Seals DR. Chronic nicotinamide riboside supplementation is well-tolerated and elevates NAD(+) in healthy middle-aged and older adults. Nat Commun. 2018;9(1):1286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Remie CME, Roumans KHM, Moonen MPB, Connell NJ, Havekes B, Mevenkamp J, Lindeboom L, de Wit VHW, van de Weijer T, Aarts Set al. Nicotinamide riboside supplementation alters body composition and skeletal muscle acetylcarnitine concentrations in healthy obese humans. Am J Clin Nutr. 2020;112:413–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dollerup OL, Christensen B, Svart M, Schmidt MS, Sulek K, Ringgaard S, Stodkilde-Jorgensen H, Moller N, Brenner C, Treebak JTet al. A randomized placebo-controlled clinical trial of nicotinamide riboside in obese men: Safety, insulin-sensitivity, and lipid-mobilizing effects. Am J Clin Nutr. 2018;108(2):343–53. [DOI] [PubMed] [Google Scholar]
- 22.Elhassan YS, Kluckova K, Fletcher RS, Schmidt MS, Garten A, Doig CL, Cartwright DM, Oakey L, Burley CV, Jenkinson Net al. Nicotinamide riboside augments the aged human skeletal muscle NAD(+) metabolome and induces transcriptomic and anti-inflammatory signatures. Cell Rep. 2019;28(7):1717–1728.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Irie J, Inagaki E, Fujita M, Nakaya H, Mitsuishi M, Yamaguchi S, Yamashita K, Shigaki S, Ono T, Yukioka Het al. Effect of oral administration of nicotinamide mononucleotide on clinical parameters and nicotinamide metabolite levels in healthy Japanese men. Endocr J. 2020;67(2):153–60. [DOI] [PubMed] [Google Scholar]
- 24.Conze D, Brenner C, Kruger CL. Safety and metabolism of long-term administration of NIAGEN (nicotinamide riboside chloride) in a randomized, double-blind, placebo-controlled clinical trial of healthy overweight adults. Sci Rep. 2019;9(1):9772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Katsyuba E, Mottis A, Zietak M, De Franco F, van der Velpen V, Gariani K, Ryu D, Cialabrini L, Matilainen O, Liscio Pet al. De novo NAD(+) synthesis enhances mitochondrial function and improves health. Nature. 2018;563(7731):354–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Grootswagers P, Mensink M, Berendsen AAM, Deen CPJ, Kema IP, Bakker SJL, Santoro A, Franceschi C, Meunier N, Malpuech-Brugere Cet al. Vitamin B-6 intake is related to physical performance in European older adults: Results of the New Dietary Strategies Addressing the Specific Needs of the Elderly Population for Healthy Aging in Europe (NU-AGE) study. Am J Clin Nutr. 2021;113:781–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Baecke JA, Burema J, Frijters JE. A short questionnaire for the measurement of habitual physical activity in epidemiological studies. Am J Clin Nutr. 1982;36(5):936–42. [DOI] [PubMed] [Google Scholar]
- 28.de Ligt M, Bruls YMH, Hansen J, Habets MF, Havekes B, Nascimento EBM, Moonen-Kornips E, Schaart G, Schrauwen-Hinderling VB, van Marken Lichtenbelt Wet al. Resveratrol improves ex vivo mitochondrial function but does not affect insulin sensitivity or brown adipose tissue in first degree relatives of patients with type 2 diabetes. Mol Metab. 2018;12:39–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. . Dietary reference intakes for thiamin, riboflavin, niacin, vitamin B6, folate, vitamin B12, pantothenic acid, biotin, and choline. Washington, DC: 1998. 10.17226/6015 [DOI] [PubMed] [Google Scholar]
- 30.Fukuwatari T, Shibata K. Nutritional aspect of tryptophan metabolism. Int J Tryptophan Res. 2013;6(Suppl 1):3–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kuipers H, Verstappen FT, Keizer HA, Geurten P, van Kranenburg G. Variability of aerobic performance in the laboratory and its physiologic correlates. Int J Sports Med. 1985;6(4):197–201. [DOI] [PubMed] [Google Scholar]
- 32.VanderZee KI, Sanderman R, Heyink JW, de Haes H. Psychometric qualities of the RAND 36-Item Health Survey 1.0: A multidimensional measure of general health status. Int J Behav Med. 1996;3(2):104–22. [DOI] [PubMed] [Google Scholar]
- 33.VanderZee KI, Sanderman R. Het meten van de algemene gezondheidstoestand met de RAND-36, een handleiding. UMGC /Rijksuniversiteit Groningen, Research Institute SHARE:. 2012; 2nd edition. Available from: https://www.researchgate.net/profile/Robbert-Sanderman/publication/283996758_Het_meten_van_de_algemene_gezondheidstoestand_met_de_RAND-36/links/5761102008aeeada5bc310e0/Het-meten-van-de-algemene-gezondheidstoestand-met-de-RAND-36.pdf. [Google Scholar]
- 34.Lindeboom L, Nabuurs CI, Hesselink MK, Wildberger JE, Schrauwen P, Schrauwen-Hinderling VB. Proton magnetic resonance spectroscopy reveals increased hepatic lipid content after a single high-fat meal with no additional modulation by added protein. Am J Clin Nutr. 2015;101(1):65–71. [DOI] [PubMed] [Google Scholar]
- 35.Guralnik JM, Simonsick EM, Ferrucci L, Glynn RJ, Berkman LF, Blazer DG, Scherr PA, Wallace RB. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49(2):M85–94. [DOI] [PubMed] [Google Scholar]
- 36.Bergstrom J. Percutaneous needle biopsy of skeletal muscle in physiological and clinical research. Scand J Clin Lab Invest. 1975;35(7):609–16. [PubMed] [Google Scholar]
- 37.Hoeks J, van Herpen NA, Mensink M, Moonen-Kornips E, van Beurden D, Hesselink MK, Schrauwen P. Prolonged fasting identifies skeletal muscle mitochondrial dysfunction as consequence rather than cause of human insulin resistance. Diabetes. 2010;59(9):2117–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Phielix E, Meex R, Ouwens DM, Sparks L, Hoeks J, Schaart G, Moonen-Kornips E, Hesselink MK, Schrauwen P. High oxidative capacity due to chronic exercise training attenuates lipid-induced insulin resistance. Diabetes. 2012;61(10):2472–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Weir JB. New methods for calculating metabolic rate with special reference to protein metabolism. J Physiol. 1949;109(1–2):1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Matomaki P, Linnamo V, Kyrolainen H. A comparison of methodological approaches to measuring cycling mechanical efficiency. Sports Med Open. 2019;5(1):23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.van Vlies N, Tian L, Overmars H, Bootsma AH, Kulik W, Wanders RJ, Wood PA, Vaz FM. Characterization of carnitine and fatty acid metabolism in the long-chain acyl-CoA dehydrogenase-deficient mouse. Biochem J. 2005;387(Pt 1):185–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kato T, Berger SJ, Carter JA, Lowry OH. An enzymatic cycling method for nicotinamide-adenine dinucleotide with malic and alcohol dehydrogenases. Anal Biochem. 1973;53(1):86–97. [DOI] [PubMed] [Google Scholar]
- 43.Timmers S, Konings E, Bilet L, Houtkooper RH, van de Weijer T, Goossens GH, Hoeks J, van der Krieken S, Ryu D, Kersten Set al. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab. 2011;14:612–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.van de Weijer T, Phielix E, Bilet L, Williams EG, Ropelle ER, Bierwagen A, Livingstone R, Nowotny P, Sparks LM, Paglialunga Set al. Evidence for a direct effect of the NAD+ precursor acipimox on muscle mitochondrial function in humans. Diabetes. 2015;64(4):1193–201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Remie CM, Roumans KH, Moonen MP, Connell NJ, Havekes B, Mevenkamp J, Lindeboom L, de Wit VH, van de Weijer T, Aarts SA. Nicotinamide riboside supplementation alters body composition and skeletal muscle acetylcarnitine concentrations in healthy obese humans. Am J Clin Nutr. 2020;112:413–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dollerup OL, Chubanava S, Agerholm M, Sondergard SD, Altintas A, Moller AB, Hoyer KF, Ringgaard S, Stodkilde-Jorgensen H, Lavery GGet al. Nicotinamide riboside does not alter mitochondrial respiration, content or morphology in skeletal muscle from obese and insulin-resistant men. J Physiol. 2020;598(4):731–54. [DOI] [PubMed] [Google Scholar]
- 47.Zhou CC, Yang X, Hua X, Liu J, Fan MB, Li GQ, Song J, Xu TY, Li ZY, Guan YFet al. Hepatic NAD(+) deficiency as a therapeutic target for non-alcoholic fatty liver disease in ageing. Br J Pharmacol. 2016;173(15):2352–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gariani K, Menzies KJ, Ryu D, Wegner CJ, Wang X, Ropelle ER, Moullan N, Zhang H, Perino A, Lemos Vet al. Eliciting the mitochondrial unfolded protein response by nicotinamide adenine dinucleotide repletion reverses fatty liver disease in mice. Hepatology. 2016;63(4):1190–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Trammell SA, Weidemann BJ, Chadda A, Yorek MS, Holmes A, Coppey LJ, Obrosov A, Kardon RH, Yorek MA, Brenner C. Nicotinamide riboside opposes type 2 diabetes and neuropathy in mice. Sci Rep. 2016;6:26933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gaggini M, Morelli M, Buzzigoli E, DeFronzo RA, Bugianesi E, Gastaldelli A. Non-alcoholic fatty liver disease (NAFLD) and its connection with insulin resistance, dyslipidemia, atherosclerosis and coronary heart disease. Nutrients. 2013;5(5):1544–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Pirinen E, Auranen M, Khan NA, Brilhante V, Urho N, Pessia A, Hakkarainen A, Kuula J, Heinonen U, Schmidt MSet al. Niacin cures systemic NAD(+) deficiency and improves muscle performance in adult-onset mitochondrial myopathy. Cell Metab. 2020;31(6):1078–1090.e5. [DOI] [PubMed] [Google Scholar]
- 52.Lim CS, Potts M, Helm RF. Nicotinamide extends the replicative life span of primary human cells. Mech Ageing Dev. 2006;127(6):511–4. [DOI] [PubMed] [Google Scholar]
- 53.Mitchell SJ, Bernier M, Aon MA, Cortassa S, Kim EY, Fang EF, Palacios HH, Ali A, Navas-Enamorado I, Di Francesco Aet al. Nicotinamide improves aspects of healthspan, but not lifespan, in mice. Cell Metab. 2018;27(3):667–676.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yang SJ, Choi JM, Kim L, Park SE, Rhee EJ, Lee WY, Oh KW, Park SW, Park CY. Nicotinamide improves glucose metabolism and affects the hepatic NAD-sirtuin pathway in a rodent model of obesity and type 2 diabetes. J Nutr Biochem. 2014;25(1):66–72. [DOI] [PubMed] [Google Scholar]
- 55.Trammell SA, Schmidt MS, Weidemann BJ, Redpath P, Jaksch F, Dellinger RW, Li Z, Abel ED, Migaud ME, Brenner C. Nicotinamide riboside is uniquely and orally bioavailable in mice and humans. Nat Commun. 2016;7:12948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Benyo Z, Gille A, Bennett CL, Clausen BE, Offermanns S. Nicotinic acid-induced flushing is mediated by activation of epidermal langerhans cells. Mol Pharmacol. 2006;70(6):1844–9. [DOI] [PubMed] [Google Scholar]
- 57.Liu L, Su X, Quinn WJ 3rd,, Hui S, Krukenberg K, Frederick DW, Redpath P, Zhan L, Chellappa K, White Eet al. Quantitative analysis of NAD synthesis-breakdown fluxes. Cell Metab. 2018;27(5):1067–1080.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Abellan van Kan G, Rolland Y, Andrieu S, Bauer J, Beauchet O, Bonnefoy M, Cesari M, Donini LM, Gillette Guyonnet S, Inzitari Met al. Gait speed at usual pace as a predictor of adverse outcomes in community-dwelling older people an International Academy on Nutrition and Aging (IANA) Task Force. J Nutr Health Aging. 2009;13(10):881–9. [DOI] [PubMed] [Google Scholar]
- 59.Tompkins BA, DiFede DL, Khan A, Landin AM, Schulman IH, Pujol MV, Heldman AW, Miki R, Goldschmidt-Clermont PJ, Goldstein BJet al. Allogeneic mesenchymal stem cells ameliorate aging frailty: A phase II randomized, double-blind, placebo-controlled clinical trial. J Gerontol A Biol Sci Med Sci. 2017;72(11):1513–22. [DOI] [PMC free article] [PubMed] [Google Scholar]