Abstract
Background
Recent evidence has shown that the pathogenesis of ischaemic stroke associated with atrial fibrillation (AF) is complex and involves other factors in addition to arrhythmias. The purpose of this study was to investigate the relationship among AF, CHA2DS2-VASc score and ischaemic stroke in patients with coronary artery disease (CAD) in Hebei, China.
Methods
A total of 2,335 patients with CAD from September 2016 to May 2019 at the Second Hospital of Hebei Medical University were included (mean age 62.73 ± 10.35 years, range 26–92 years; 41.58% female). This was a cross-sectional study, and participants were divided into non-stroke (n = 1997) and ischaemic stroke groups (n = 338). Propensity score matching (PSM) was performed to match ischaemic stroke patients with non-stroke patients in a 1:4 ratio. The relationship among AF, the CHA2DS2-VASc score and ischaemic stroke was evaluated using univariable generalized linear models for different sex, age, body mass index (BMI), CAD and CHA2DS2-VASc score subgroups. Univariable and multivariable generalized linear models were used to evaluate the relationship between AF and ischaemic stroke in the different models.
Results
Compared with that in the non-stroke group, the prevalence of AF (8.81% vs. 14.20%, P = 0.002) in the ischaemic stroke group was higher. The proportion of patients with ischaemic stroke was significantly different between the AF group and the non-AF group (28.74% vs. 19.04%, P = 0.003). An increasing CHA2DS2-VASc score was associated with a gradual increase in the prevalence of AF (P for trend < 0.001). Subgroup analysis showed that the trend towards increased stroke risk in the AF group was consistent across the various subgroups. The multivariable analysis demonstrated that AF was not associated with ischaemic stroke compared with the absence of AF (OR = 1.55, 95% CI 0.94–2.56, P = 0.087).
Conclusion
In our cross-sectional study, after adjustment for confounding factors, there was no association between AF and ischaemic stroke. The increased risk of ischaemic stroke associated with AF was attenuated by atherosclerotic factors. Our study supports the current view that enhanced control of modifiable cardiovascular risk factors in patients with AF is essential.
Keywords: Atrial fibrillation, CHA2DS2-VASc score, Coronary artery disease, Ischaemic stroke
Background
Atrial fibrillation (AF) is a common arrhythmia in clinical practice [1–3]. According to the data of the Global Burden of Disease Study 2017, the age-standardized AF prevalence, incidence, and mortality rates were 0.48%, 0.04% and 0.004%, respectively [4]. In a national cross-sectional study including 31,230 participants from all 31 provinces in China, the prevalence of AF in Chinese adults over 35 years of age was 0.71% [5]. A study in 81,103 male coal miners in Hebei Province found that the prevalence of AF was 0.49%. The mean CHADS2 score in AF patients was 0.73. Participants with AF had a significantly higher prevalence of ischaemic stroke than those without AF (7.82% vs. 2.50%, P = 0.001) [6]. Although evidence in recent decades has indicated a strong association between AF and ischaemic stroke [7, 8], a gap remains in our understanding of the mechanism underlying this association. The current view is that AF causes blood clotting and thrombus detachment, resulting in cerebral embolism [8]. This mechanism has long been considered the leading cause of ischaemic stroke [9]. However, recent studies have shown that the pathogenesis of ischaemic stroke associated with AF is more complex than previously thought and involves other factors in addition to arrhythmias [10]. Coronary artery disease (CAD) and ischaemic stroke have similar risk factors [11], such as hypertension and advanced age. The purpose of this study was to explore the relationship among AF, CHA2DS2-VASc score and ischaemic stroke in patients with coronary heart disease and to further understand the role of AF in ischaemic stroke in patients with CAD. The results will have far-reaching significance for future treatment strategies.
Materials and methods
Study population
This study was a cross-sectional study conducted according to the Strengthening the Reporting of Observational studies in Epidemiology guidelines. A total of 5583 patients were admitted to the first department of the Cardiovascular Department of the Second Hospital of Hebei Medical University for cardiovascular reasons from September 2016 to May 2019.
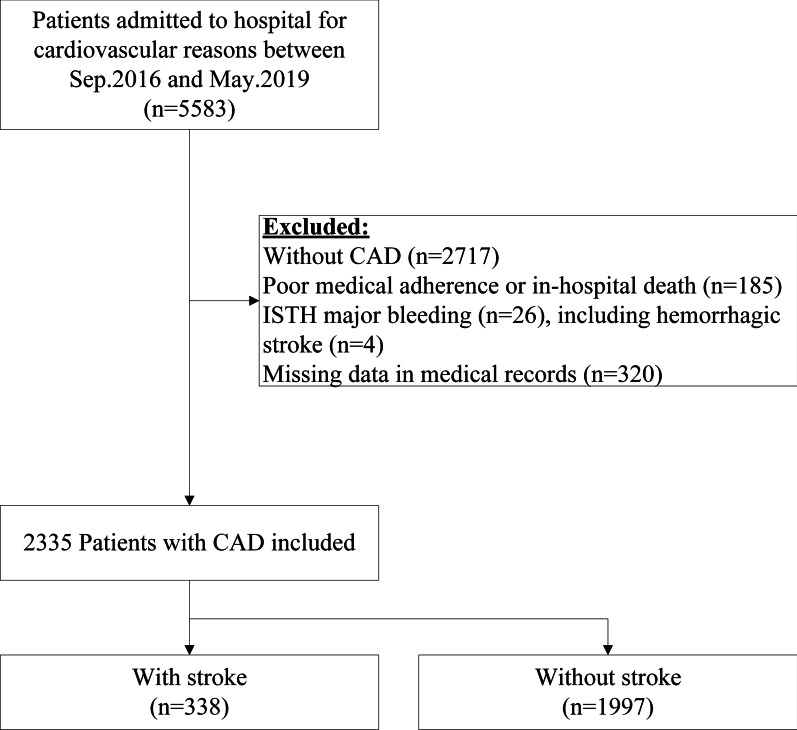
Patient demographic information, medical histories and laboratory assessments were collected and recorded. As shown in Fig. 1, the exclusion criteria were no CAD (n = 2717), poor medical adherence or in-hospital death (n = 185), major bleeding according to the International Society on Thrombosis and Haemostasis (ISTH) (n = 26), or missing medical record data (n = 320). Finally, 2335 patients (mean age 62.73 ± 10.35 years, range 26–92 years; 41.58% female) were included in this analysis.
Fig. 1.
Patient selection flowchart
The study population was divided into 20 subgroups according to sex, age, body mass index (BMI), type of CAD, and CHA2DS2-VASc2 score ≥ 2 (male) or ≥ 3 (female).
CAD was defined as significant narrowing (≥ 50%) of the coronary lumen detected either invasively or noninvasively; it was further defined as one or more of the following types: stable coronary artery disease (SCAD), unstable angina (UA), non-ST-elevation myocardial infarction (NSTEMI), ST-elevation myocardial infarction (STEMI), previous myocardial infarction (previous MI), and ischaemic cardiomyopathy. Specific diagnoses were performed on the basis of standard reference guidelines.
In this study, ischaemic stroke was defined as a history of ischaemic stroke or new-onset ischaemic stroke during hospitalization.
AF was classified into paroxysmal, persistent, long-standing persistent and permanent AF, which were determined according to the 2014 American College of Cardiology (ACC)/American Heart Association (AHA)/Heart Rhythm Society (HRS) Task Force on Practice Guidelines and the 2016 European Society of Cardiology (ESC) guidelines [12, 13]. In this study, persistent, long-standing persistent, and permanent AF were grouped and collectively referred to as non-paroxysmal AF.
The CHA2DS2-VASc score is a clinical prediction tool commonly used to identify the risk of ischaemic stroke in patients with nonvalvular AF; it is calculated by assigning 2 points for ischaemic stroke or age ≥ 75 years and 1 point for each additional component, including heart failure, hypertension, vascular disease, age 65–74 years, female sex and diabetes. The CHA2DS2-VASc score was calculated for each patient at discharge.
We defined diabetes mellitus (DM) as a casual plasma glucose concentration ≥ 200 mg/dL, or fasting plasma glucose concentration ≥ 126 mg/dL, or 2-h plasma glucose concentration ≥ 200 mg/dL according to a 75 g oral glucose tolerance test or drug or insulin use for DM. Hypertension was defined as systolic blood pressure ≥ 140 mmHg, or diastolic blood pressure ≥ 90 mmHg or antihypertensive medication use. Dyslipidaemia was defined as low-density lipoprotein cholesterol (LDL-C) ≥ 140 mg/dL, or high-density lipoprotein cholesterol (HDL-C) < 40 mg/dL, or triglycerides > 150 mg/dL. Heart failure included past or current signs and symptoms of heart failure with both low (< 40%) and preserved ejection fractions (EFs) (≥ 40%) and/or other clinical evidence of cardiac dysfunction. Chronic kidney disease (CKD) was defined as an estimated glomerular filtration rate < 60 mL/min per 1.73 m2. Left atrial diameter (LA) and EF were measured by transthoracic echocardiography or transoesophageal echocardiography. Pulse wave velocity (PWV) was defined as the velocity of pulse wave propagation between two fixed points in the arterial system. The ankle-brachial index (ABI) was calculated as the ratio between the ankle artery pressure and the brachial artery pressure. Both PWV and ABI were measured by a specially trained nurse using PWV/ABI instruments (form PWV/ABI, BP-203RPE; Omron-Colin, Japan).
Statistical analysis
The investigators performed the data analysis using Empower (R) (www.empowerstats.com, X&Y solutions, Inc. Boston, Massachusetts, USA) and R 3.6.3 (http://www.R-project.org). The Shapiro–Wilk test was used to examine whether continuous variables were normally distributed. Continuous variables in this study that did not conform to a normal distribution are presented as medians and interquartile ranges (IQRs) and were compared using the Mann–Whitney test. Categorical variables are presented as counts and composition ratios and were compared using the chi-square test or Fisher’s exact test as appropriate.
Given the inherent differences between patients with and without ischaemic stroke, we calculated a propensity score for the following variables: sex, age, and BMI. The propensity score was estimated by a logistic regression model. The matching process was based on the nearest-neighbour matching method without replacement within a calliper of 0.01; participants were 1:4 matched, which yielded a stroke group and a matched non-stroke group.
To control for potential covariates, we used multivariable generalized linear regression models. The multivariable analysis was adjusted for age, hypertension, aortic atherosclerosis, AF, ABI, CKD, PWV, LA, STEMI, heart failure, previous MI, ischaemic cardiomyopathy, smoking, alcohol consumption, and DM. These variables were selected on the basis of statistical significance (P < 0.1) in the univariable analysis or previous reports or because they are empirically associated with stroke. Variables with multicollinearity were excluded. The Cochran–Mantel–Haenszel test was used in the analysis to calculate the P value for trend. The likelihood ratio test was performed after adjustment for subgroup factors. A two-tailed P value < 0.05 was considered statistically significant in this study.
Results
Patient characteristics
A total of 2335 patients with CAD, including 1997 patients without ischaemic stroke (85.52%) and 338 patients with ischaemic stroke (14.48%), were enrolled in this study. Three (0.89%) of the ischaemic stroke patients had acute stroke, while the remaining 335 (99.11%) had prior stroke. The matched cross-sectional analysis included 1,685 patients (1348 patients without ischaemic stroke and 337 patients with ischaemic stroke). The patients’ baseline characteristics before and after PSM are shown in Table 1.
Table 1.
Comparison of baseline characteristics
| Before PSM | After PSM | |||||
|---|---|---|---|---|---|---|
| Without stroke (n = 1997) | With stroke (n = 338) | P value | Without stroke (n = 1348) | With stroke (n = 337) | P value | |
| Sex | 0.771 | 0.980 | ||||
| Female | 828 (41.46%) | 143 (42.31%) | 573 (42.51%) | 143 (42.43%) | ||
| Male | 1169 (58.54%) | 195 (57.69%) | 775 (57.49%) | 194 (57.57%) | ||
| Age (years) | 63.00 (55.00–69.00) | 65.00 (60.00–72.00) | < 0.001* | 64.00 (58.00–70.00) | 65.00 (60.00–72.00) | < 0.001* |
| BMI (kg/m2) | 25.39 (23.44–27.68) | 25.50 (23.23–27.68) | 0.929 | 25.39 (23.29–27.68) | 25.50 (23.23–27.68) | 0.941 |
| CAD type | ||||||
| SCAD | 128 (6.41%) | 36 (10.65%) | 0.005* | 86 (6.38%) | 36 (10.68%) | 0.006* |
| UA | 1471 (73.66%) | 229 (67.75%) | 0.024* | 989 (73.37%) | 229 (67.95%) | 0.047* |
| NSTEMI | 286 (14.32%) | 41 (12.13%) | 0.283 | 201 (14.91%) | 41 (12.17%) | 0.199 |
| STEMI | 79 (3.96%) | 23 (6.80%) | 0.018* | 51 (3.78%) | 22 (6.53%) | 0.027* |
| Previous MI | 205 (10.27%) | 49 (14.50%) | 0.021* | 146 (10.83%) | 49 (14.54%) | 0.057 |
| Ischaemic cardiomyopathy | 34 (1.70%) | 11 (3.25%) | 0.082 | 23 (1.71%) | 11 (3.26%) | 0.069 |
| Heart failure | 312 (15.62%) | 72 (21.30%) | 0.009* | 228 (16.91%) | 72 (21.36%) | 0.056 |
| Hypertension | 1307 (65.45%) | 262 (77.51%) | < 0.001* | 899 (66.69%) | 262 (77.74%) | < 0.001* |
| Dyslipidaemia | 605 (30.30%) | 95 (28.11%) | 0.417 | 414 (30.71%) | 94 (27.89%) | 0.313 |
| Total cholesterol (mmol/L) | 3.99 (3.40–4.67) | 3.94 (3.32–4.76) | 0.544 | 4.00 (3.37–4.68) | 3.94 (3.32–4.76) | 0.478 |
| LDL-C (mmol/L) | 2.51 (1.92–3.11) | 2.45 (1.91–3.15) | 0.615 | 2.51 (1.90–3.11) | 2.45 (1.91–3.15) | 0.567 |
| Valvular heart disease | 43 (2.15%) | 8 (2.37%) | 0.840 | 32 (2.37%) | 8 (2.37%) | 1 |
| AF | 176 (8.81%) | 48 (14.20%) | 0.002* | 119 (8.83%) | 48 (14.24%) | 0.003* |
| AF type | < 0.001* | 0.003* | ||||
| No | 1821 (91.19%) | 290 (85.80%) | 1229 (91.17%) | 289 (85.76%) | ||
| paroxysmal AF | 106 (5.31%) | 22 (6.51%) | 69 (5.12%) | 22 (6.53%) | ||
| Non-paroxysmal AF | 70 (3.51%) | 26 (7.69%) | 50 (3.71%) | 26 (7.72%) | ||
| DM | 551 (27.59%) | 102 (30.18%) | 0.327 | 378 (28.04%) | 102 (30.27%) | 0.418 |
| COPD | 40 (2.00%) | 7 (2.07%) | 0.836 | 30 (2.23%) | 7 (2.08%) | 0.868 |
| CKD | 39 (1.95%) | 13 (3.85%) | 0.043* | 23 (1.71%) | 13 (3.86%) | 0.015* |
| Creatine (μmol/L) | 69.00 (59.00–80.95) | 72.00 (61.30–86.00) | 0.001* | 70.00 (59.00–82.00) | 72.00 (61.30–86.00) | 0.011* |
| Fatty liver | 39 (1.95%) | 5 (1.48%) | 0.670 | 30 (2.23%) | 5 (1.48%) | 0.393 |
| Smoking | 409 (20.48%) | 57 (16.86%) | 0.124 | 261 (19.36%) | 56 (16.62%) | 0.249 |
| Alcohol consumption | 347 (17.38%) | 47 (13.91%) | 0.115 | 224 (16.62%) | 46 (13.65%) | 0.184 |
| Aortic atherosclerosis | 529 (26.81%) | 146 (43.84%) | < 0.001* | 386 (28.94%) | 146 (43.98%) | < 0.001* |
| LA (mm) | 34.00 (32.00–37.00) | 35.00 (32.00–37.00) | 0.073 | 34.00 (32.00–37.00) | 35.00 (32.00–37.00) | 0.024* |
| EF (%) | 61.74 (60.61–62.89) | 61.70 (60.64–62.77) | 0.574 | 61.74 (60.61–62.86) | 61.70 (60.64–62.77) | 0.73 |
| PWV (cm/s) | 1556.50 (1367.50–1798.50) | 1623.00 (1418.00–1906.50) | < 0.001* | 1575.25 (1388.00–1814.38) | 1625.00 (1418.00–1908.62) | 0.022* |
| ABI | 1.09 (1.02–1.156) | 1.08 (0.97–1.16) | 0.057 | 1.08 (1.01–1.16) | 1.08 (0.97–1.16) | 0.005* |
| CHA2DS2-VASc score | 3.00 (2.00–4.00) | 4.00 (3.00–5.00) | < 0.001* | 3.00 (2.00–4.00) | 4.00 (3.00–5.00) | < 0.001* |
| Propensity score | 0.16 ± 0.04 | 0.14 ± 0.04 | < 0.001* | 0.16 ± 0.04 | 0.15 ± 0.04 | < 0.001* |
*Indicates p < 0.05
BMI body mass index, CAD coronary artery disease, SCAD stable coronary artery disease, UA unstable angina, NSTEMI non-ST-elevation myocardial infarction, STEMI ST-elevation myocardial infarction, previous MI previous myocardial infarction, AF atrial fibrillation, DM diabetes mellitus, COPD chronic obstructive pulmonary disease, CKD chronic kidney disease, LDL-C low-density lipoprotein cholesterol, LA left atrial diameter, EF ejection fraction, PWV pulse wave velocity, ABI ankle-brachial index
Before PSM, compared with the non-stroke group, the ischaemic stroke group was older (63.00 vs. 65.00, P < 0.001), and the proportions of SCAD (6.41% vs. 10.65%, P = 0.005), STEMI (3.96% vs. 6.80%, P = 0.018), and previous MI (10.27% vs. 14.50%, P = 0.021) were higher. The proportion of UA (73.66% vs. 67.75%, P = 0.024) was lower, and the proportions of heart failure (15.62% vs. 21.30%, P = 0.009), hypertension (65.45% vs. 77.51%, P < 0.001), AF (8.81% vs. 14.20%, P = 0.002), CKD (1.95% vs. 3.85%, P = 0.043), and aortic atherosclerosis (26.81% vs. 84%, P < 0.001) were higher. The creatinine level, PWV and CHA2DS2-VASc score were also higher. These differences were statistically significant.
After PSM, compared with those in the non-stroke group, the proportions of STEMI, UA, hypertension, AF, and aortic atherosclerosis were higher in the ischaemic stroke group. The serum creatinine level and CHA2DS2-VASc score were also higher. These differences were statistically significant.
A comparison of the baseline data of patients with and without AF after PSM is shown in Table 2. Compared with the non-AF group, the AF group was older (63.50 vs. 69.00, P < 0.001); had higher prevalence rates of SCAD (4.61% vs. 31.14%, P < 0.001), previous MI (10.94% vs. 17.37%, P = 0.014), ischaemic cardiomyopathy (1.58% vs. 5.99%, P < 0.001), and UA (74.57% vs. 51.50%); had lower prevalence rates of STEMI (4.68% vs. 1.20%, P = 0.036), heart failure (13.90% vs. 53.29%, P < 0.001), valvular heart disease (1.91% vs. 6.59%, P < 0.001), CKD (1.84% vs. 4.79%, P = 0.012), smoking (19.50% vs. 12.57%, P = 0.03), and aortic atherosclerosis (30.53% vs. 44.58%); had a higher serum creatinine level; had a larger LA; and had a higher PWV and CHA2DS2-VASc score. Furthermore, the ABI and EF were lower. These differences were statistically significant. The proportion of patients with ischaemic stroke was significantly different between the two groups (19.04% vs. 28.74%, P = 0.003).
Table 2.
Comparison of baseline characteristics after PSM with and without AF
| Without (n = 1518) | With AF (n = 167) | P value | |
|---|---|---|---|
| Sex | 0.505 | ||
| Female | 641 (42.23%) | 75 (44.91%) | |
| Male | 877 (57.77%) | 92 (55.09%) | |
| Age (years) | 63.50 (58.00–70.00) | 69.00 (63.00–77.00) | < 0.001* |
| BMI (kg/m2) | 25.39 (23.23–27.68) | 25.69 (23.44–27.68) | 0.872 |
| CAD type | |||
| SCAD | 70 (4.61%) | 52 (31.14%) | < 0.001* |
| UA | 1132 (74.57%) | 86 (51.50%) | < 0.001* |
| NSTEMI | 225 (14.82%) | 17 (10.18%) | 0.104 |
| STEMI | 71 (4.68%) | 2 (1.20%) | 0.036* |
| Previous MI | 166 (10.94%) | 29 (17.37%) | 0.014* |
| Ischaemic cardiomyopathy | 24 (1.58%) | 10 (5.99%) | < 0.001* |
| Heart failure | 211 (13.90%) | 89 (53.29%) | < 0.001* |
| Hypertension | 1041 (68.58%) | 120 (71.86%) | 0.385 |
| Dyslipidaemia | 456 (30.04%) | 52 (31.14%) | 0.769 |
| Total cholesterol (mmol/L) | 4.00 (3.39–4.70) | 3.83 (3.28–4.70) | 0.229 |
| LDL-C (mmol/L) | 2.52 (1.91–3.12) | 2.32 (1.88–3.09) | 0.301 |
| Valvular heart disease | 29 (1.91%) | 11 (6.59%) | < 0.001* |
| DM | 430 (28.33%) | 50 (29.94%) | 0.661 |
| COPD | 35 (2.31%) | 2 (1.20%) | 0.354 |
| CKD | 28 (1.84%) | 8 (4.79%) | 0.012* |
| Creatine (μmol/L) | 69.00 (59.00–81.00) | 79.85 (68.00–94.75) | < 0.001* |
| Fatty liver | 33 (2.17%) | 2 (1.20%) | 0.401 |
| Smoking | 296 (19.50%) | 21 (12.57%) | 0.03* |
| Alcohol consumption | 249 (16.40%) | 21 (12.57%) | 0.201 |
| Aortic atherosclerosis | 458 (30.53%) | 74 (44.58%) | < 0.001* |
| LA (mm) | 34.00 (32.00–36.00) | 38.00 (35.00–42.50) | < 0.001* |
| EF (%) | 61.74 (60.64–62.89) | 60.78 (55.48–62.05) | 0.001* |
| PWV (cm/s) | 1565.00 (1387.75–1810.75) | 1734.75 (1505.88–2149.62) | < 0.001* |
| ABI | 1.08 (1.00–1.16) | 1.06 (0.94–1.14) | 0.004* |
| CHA2DS2-VASc score | 3.00 (2.00–4.00) | 4.00 (3.00–6.00) | < 0.001* |
| Stroke | 289 (19.04%) | 48 (28.74%) | 0.003* |
*Indicates p < 0.05
BMI body mass index, CAD coronary artery disease, SCAD stable coronary artery disease, UA unstable angina, NSTEMI non-ST-elevation myocardial infarction, STEMI ST-elevation myocardial infarction, previous MI previous myocardial infarction, AF atrial fibrillation, DM diabetes mellitus, COPD chronic obstructive pulmonary diseases, CKD chronic kidney disease, LDL-C low-density lipoprotein cholesterol, LA left atrial diameter, EF ejection fraction, PWV pulse wave velocity, ABI ankle-brachial index
Subgroup analysis
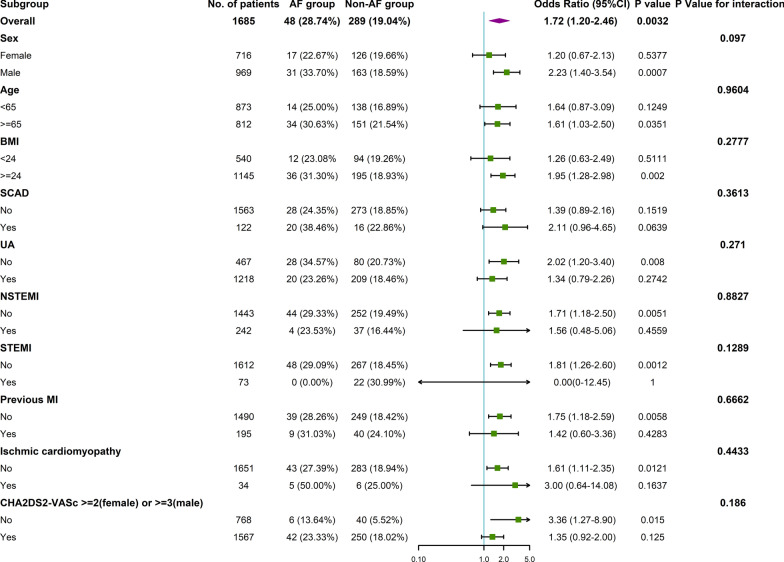
Subgroup analysis according to baseline characteristics between the AF group and the non-AF group was performed (Fig. 2). A trend towards an increased risk of stroke in the AF group was observed in the following subgroups: male, age ≥ 65 years, BMI ≥ 24 kg/m2, non-UA, non-NSTEMI, non-STEMI, non-previous MI, non-ischaemic cardiomyopathy and CHA2DS2-VASc < 2(male) or < 3(female) subgroups. Interestingly, AF seemed to have no association with stroke in the CHA2DS2-VASc ≥ 2 (female) or ≥ 3 (male) subgroup (OR = 1.35, 95% CI 0.92–2.00, P = 0.125, P for interaction = 0.186). However, the subgroup analyses did not indicate any significant interactions between ischaemic stroke and the stratification variables.
Fig. 2.
Association between AF and ischaemic stroke in prespecified and exploratory subgroups after PSM. P for interaction is based on the likelihood ratio test
Association between AF and ischaemic stroke
The univariable analysis of the relationship between AF and ischaemic stroke after PSM (Table 3) showed that AF was associated with ischaemic stroke (OR = 1.71, 95% CI 1.20–2.46, P = 0.003). However, the multivariable analysis showed that AF was not associated with ischaemic stroke compared with no AF (OR = 1.55, 95% CI 0.94–2.56, P = 0.087). The multivariable analysis was adjusted for age, hypertension, aortic atherosclerosis, AF, ABI, CKD, PWV, LA, STEMI, heart failure, previous MI, ischaemic cardiomyopathy, smoking, alcohol consumption, and DM.
Table 3.
Univariable and multivariable analysis of the association between AF and ischaemic stroke after PSM
| Variable | Univariable OR (95% CI) P value |
Multivariable OR (95% CI) P value |
|---|---|---|
| Without AF | 1.00 | 1.00 |
| With AF |
1.71 (1.20–2.46) 0.003* |
1.55 (0.94–2.56) 0.087 |
*Indicates p < 0.05
The multivariable analysis was adjusted for age, hypertension, aortic atherosclerosis, AF, ABI, CKD, PWV, LA, STEMI, heart failure, previous MI, ischaemic cardiomyopathy, smoking, alcohol consumption, and DM
Association between AF burden and ischaemic stroke
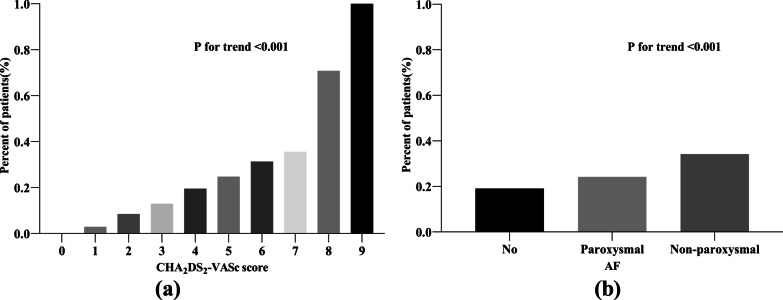
The univariable analysis of the relationship between the AF burden and ischaemic stroke after PSM (Table 4) showed that compared with paroxysmal AF, non-paroxysmal AF did not increase the prevalence of ischaemic stroke (OR = 1.63, 95% CI 0.83–3.20, P = 0.155). The multivariable analysis produced similar results (OR = 1.02, 95% CI 0.32–3.24, P = 0.973). However, as shown in Fig. 3b, an increasing AF burden was associated with a continuous increase in the prevalence of ischaemic stroke (P for trend < 0.001).
Table 4.
Univariable and multivariable analysis of the association between AF burden and ischaemic stroke after PSM
| Variable | Univariable OR (95% CI) P value |
Multivariable OR (95% CI) P value |
|---|---|---|
| Paroxysmal AF | 1.00 | 1.00 |
| Non-paroxysmal AF |
1.63 (0.83–3.20) 0.155 |
1.02 (0.32–3.24) 0.973 |
*Indicates p < 0.05
The multivariable analysis was adjusted for age, hypertension, aortic atherosclerosis, AF, ABI, CKD, PWV, LA, STEMI, heart failure, previous MI, ischaemic cardiomyopathy, smoking, alcohol consumption, and DM
Fig. 3.
a Association between the CHA2DS2-VASc score and AF. b Association between AF type and ischaemic stroke
Relationship among CHA2DS2-VASc score and ischaemic stroke
An increasing CHA2DS2-VASc score was associated with a gradual increase in the prevalence of AF (P for trend < 0.001, Fig. 3a).
Discussion
AF and ischaemic stroke have common risk factors
The results of this study suggest that age, heart failure, hypertension, CKD, creatinine level, aortic atherosclerosis, PWV, and the CHA2DS2-VASc score are associated with ischaemic stroke and AF.
Previous studies have shown that age, male sex, hypertension, DM, valvular heart disease, heart failure, CAD, CKD, inflammatory disease, obstructive sleep apnoea syndrome and smoking are common risk factors for AF and ischaemic stroke [12, 14, 15]. Potential risk factors, such as obesity and metabolic syndrome, contribute to the development of AF and atrial cardiomyopathy through a variety of mechanisms [16]. The Framingham Heart Study [17] reported that the lifetime risk of AF in those with an optimal risk profile was approximately 20%, which was lower than that in those with a high-risk profile (38.4%). The UK Biobank study [16] confirmed that there were significant differences in AF risk among the ideal lifestyle group, the general lifestyle group, and the poor lifestyle group, regardless of genetic risk. As more common risk factors were considered, the association between AF and ischaemic stroke was reduced, suggesting confounding.
Possible mechanism of ischaemic stroke in patients with CAD complicated with AF
The univariable analysis showed that in patients with CAD, AF was associated with ischaemic stroke; however, the association between AF and ischaemic stroke disappeared in the multivariable analysis, which included adjustment for other risk factors. In the subgroup analysis, AF appeared to have no association in the CHA2DS2-VASc ≥ 2 (female) or ≥ 3 (male) subgroup. It is speculated that the risk of ischaemic stroke does not increase after the CHA2DS2-VASc score reaches a certain threshold in patients with CAD. Furthermore, comparing a group with CAD plus AF to a group with AF without CAD would be important for determining whether atherosclerotic factors play important roles in ischaemic stroke. A report of the Western Denmark Heart Registry [18] demonstrated that CAD was an independent risk factor for a composite thromboembolic outcome in addition to the risk factors already included in the CHA2DS2-VASc score. As a result, CAD diagnosed by coronary angiography has been added as a vascular disease criterion in the CHA2DS2-VASc score [19]. Our outcome is contrary to that of a meta-analysis by Santarpia et al., which showed that oral anticoagulant use after successful radiofrequency cardioversion of AF did not reduce the prevalence of embolic events [20]. A possible explanation might be as follows: both AF and atherosclerosis are independent risk factors for stroke, and the hazard ratios of these two risk factors for stroke vary among individuals. Antithrombotic therapy after successful radiofrequency ablation should be determined based on the patient's baseline risk of thromboembolism and bleeding. Large-scale randomized controlled trials are needed to determine the best anticoagulant therapy regimen for patients with AF following successful radiofrequency ablation to achieve the greatest clinical net benefit.
Association between AF burden and ischaemic stroke
In this study, both the univariable and multivariable analyses showed that non-paroxysmal AF does not increase the prevalence of ischaemic stroke compared with paroxysmal AF. However, the trend results showed that with increasing AF burden, the prevalence of ischaemic stroke increased continuously. These inconsistent findings suggest that compared with patients with paroxysmal AF, patients with non-paroxysmal AF do not appear to have an increased risk of ischaemic stroke. Our study did not provide sufficient evidence that the AF burden is positively associated with ischaemic stroke.
Although many studies have found a dose–response relationship between AF load and ischaemic stroke [21–23], the results are inconsistent. A recent KP-Rhythm study identified a higher risk of stroke in the high-load group (> 11%, approximately 2.5 h/24 h) [23]. A study including 384 patients with cardiovascular implantable electronic device-detected AF not receiving oral anticoagulation suggested that the duration of AF was not associated with the risk of stroke, whereas the CHA2DS2-VASc score was [24]. There is no standardized AF burden threshold to predict a significantly increased risk of thrombosis. The current guidelines [12, 13] generally consider that if the duration of AF is less than 48 h, there is no need to perform transoesophageal echocardiography. Regardless of the CHA2DS2-VASc score and the method used to restore the sinus rhythm, cardioversion can be performed without anticoagulation. However, whether the 48-h threshold applies to all patients with AF is debatable [25]. In addition, a brief episode of subclinical AF doubled the risk of ischaemic stroke in elderly patients [26]. while there was no significant increase in the risk of ischaemic stroke in young, healthy patients with AF [27]. Additionally, atrial remodelling in animal models of AF occurred at least one week after continuous rapid pacing [28]. Therefore, any atrial changes caused by AF are unlikely to explain the association between a short (6-min) AF episode and an increased risk of ischaemic stroke [26]. Overall, these conflicting data are insufficient to establish a clear dose–response relationship between AF burden and the risk of ischaemic stroke.
Study strength and limitations
The novelty of this study is the finding that among patients with CAD, who tend to have a higher degree of atherosclerosis than patients with only AF, the risk of ischemic stroke associated with AF was determined by atherosclerotic factors.
There are several limitations to our research. First, it was a cross-sectional study, and ischaemic stroke, the CHA2DS2-VASc score, and AF were measured at the same time. In addition, several variables remained imbalanced after PSM. Therefore, we also performed multivariable analysis to minimize bias.
Conclusion
In our cross-sectional study, after adjustment for confounding risk factors, the association between AF and ischaemic stroke in patients with CAD disappeared. In the subgroup analysis, AF appeared to have no association with stroke in the CHA2DS2-VASc ≥ 2 (female) or ≥ 3 (male) subgroup. The increased risk of ischemic stroke associated with AF was determined by atherosclerotic factors. Our study supports the current view that enhanced control of modifiable cardiovascular risk factors in patients with AF is essential.
Acknowledgements
We would like to acknowledge author services from Springer Nature for its linguistic assistance during the preparation of this manuscript.
Abbreviations
- BMI
body mass index
- CAD
coronary artery disease
- SCAD
stable coronary artery disease
- UA
unstable angina
- NSTEMI
non-ST-elevation myocardial infarction
- STEMI
ST-elevation myocardial infarction
- previous MI
previous myocardial infarction
- AF
atrial fibrillation
- DM
diabetes mellitus
- COPD
chronic obstructive pulmonary disease
- CKD
chronic kidney disease
- LDL-C
low-density lipoprotein cholesterol
- LA
left atrial diameter
- EF
ejection fraction
- PWV
pulse wave velocity
- ABI
ankle-brachial index
Authors' contributions
BQS, DML, and WC conceived and designed the study. BQS, DML, QW, XG, QH, GQG and XRQ collected the data. BQS performed the statistical analysis and interpreted the data. BQS wrote the first draft of the paper; DML and WC revised the article. This manuscript was read and approved by all credited authors. All authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author upon reasonable request.
Declarations
Ethics approval and consent to participate
The study was conducted in line with the principles of the Helsinki Declaration. Ethical approval (No: 2019-P060) was granted by the Research Ethics Committee of the Second Hospital of Hebei University in 2019. The present study retrospectively analysed anonymized data; thus, no informed consent was required.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Rahman F, Kwan GF, Benjamin EJ. Global epidemiology of atrial fibrillation. Nat Rev Cardiol. 2014;11(11):639–654. doi: 10.1038/nrcardio.2014.118. [DOI] [PubMed] [Google Scholar]
- 2.Zimetbaum P. Atrial fibrillation. Ann Intern Med. 2017;166(5):ITC33–ITC48. doi: 10.7326/AITC201703070. [DOI] [PubMed] [Google Scholar]
- 3.Bai Y, Wang YL, Shantsila A, Lip GYH. The global burden of atrial fibrillation and stroke: a systematic review of the clinical epidemiology of atrial fibrillation in Asia. Chest. 2017;152(4):810–820. doi: 10.1016/j.chest.2017.03.048. [DOI] [PubMed] [Google Scholar]
- 4.Dai H, Zhang Q, Much AA, Maor E, Segev A, Beinart R, Adawi S, Lu Y, Bragazzi NL, Wu J. Global, regional, and national prevalence, incidence, mortality, and risk factors for atrial fibrillation, 1990–2017: results from the Global Burden of Disease Study 2017. Eur Heart J Qual Care Clin Outcomes. 2020 doi: 10.1093/ehjqcco/qcaa061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang Z, Chen Z, Wang X, Zhang L, Li S, Tian Y, et al. The disease burden of atrial fibrillation in china from a national cross-sectional survey. Am J Cardiol. 2018;122(5):793–798. doi: 10.1016/j.amjcard.2018.05.015. [DOI] [PubMed] [Google Scholar]
- 6.Yu K, Xing A, Wang D, Qi S, Wang G, Chen R, et al. Prevalence and relative risk factors of atrial fibrillation in male coal miners in North China. Int J Cardiol. 2014;174(1):223–224. doi: 10.1016/j.ijcard.2014.04.002. [DOI] [PubMed] [Google Scholar]
- 7.Wolf PA, Abbott RD, Kannel WB. Atrial fibrillation as an independent risk factor for stroke: the Framingham Study. Stroke. 1991;22(8):983–988. doi: 10.1161/01.STR.22.8.983. [DOI] [PubMed] [Google Scholar]
- 8.Wolf PA, Dawber TR, Thomas HE, Jr, Kannel WB. Epidemiologic assessment of chronic atrial fibrillation and risk of stroke: the Framingham study. Neurology. 1978;28(10):973–977. doi: 10.1212/WNL.28.10.973. [DOI] [PubMed] [Google Scholar]
- 9.Caplan LR. Atrial fibrillation, past and future: from a stroke non-entity to an over-targeted cause. Cerebrovasc Dis. 2018;45(3–4):149–153. doi: 10.1159/000488063. [DOI] [PubMed] [Google Scholar]
- 10.Kamel H, Okin PM, Elkind MS, Iadecola C. Atrial fibrillation and mechanisms of stroke: time for a new model. Stroke. 2016;47(3):895–900. doi: 10.1161/STROKEAHA.115.012004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Soler EP, Ruiz VC. Epidemiology and risk factors of cerebral ischemia and ischemic heart diseases: similarities and differences. Curr Cardiol Rev. 2010;6(3):138–149. doi: 10.2174/157340310791658785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kirchhof P, Benussi S, Kotecha D, Ahlsson A, Atar D, Casadei B, et al. 2016 ESC guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J. 2016;37(38):2893–2962. doi: 10.1093/eurheartj/ehw210. [DOI] [PubMed] [Google Scholar]
- 13.January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC, Jr, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Heart Rhythm Society. Circulation. 2014;130(23):e199–267. doi: 10.1161/CIR.0000000000000041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hankey GJ. Stroke. Lancet. 2017;389(10069):641–654. doi: 10.1016/S0140-6736(16)30962-X. [DOI] [PubMed] [Google Scholar]
- 15.Homan EA, Reyes MV, Hickey KT, Morrow JP. Clinical overview of obesity and diabetes mellitus as risk factors for atrial fibrillation and sudden cardiac death. Front Physiol. 2018;9:1847. doi: 10.3389/fphys.2018.01847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Said MA, Verweij N, van der Harst P. Associations of combined genetic and lifestyle risks with incident cardiovascular disease and diabetes in the UK biobank study. JAMA Cardiol. 2018;3(8):693–702. doi: 10.1001/jamacardio.2018.1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Staerk L, Wang B, Preis SR, Larson MG, Lubitz SA, Ellinor PT, et al. Lifetime risk of atrial fibrillation according to optimal, borderline, or elevated levels of risk factors: cohort study based on longitudinal data from the Framingham Heart Study. BMJ. 2018;361:k1453. doi: 10.1136/bmj.k1453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Steensig K, Olesen KKW, Thim T, Nielsen JC, Jensen SE, Jensen LO, et al. Should the presence or extent of coronary artery disease be quantified in the CHA2DS2-VASc score in atrial fibrillation? A report from the Western Denmark heart registry. Thromb Haemost. 2018;118(12):2162–2170. doi: 10.1055/s-0038-1675401. [DOI] [PubMed] [Google Scholar]
- 19.Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomstrom-Lundqvist C, et al. 2020 ESC guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association of Cardio-Thoracic Surgery (EACTS) Eur Heart J. 2020;42:373–498. doi: 10.1093/eurheartj/ehaa612. [DOI] [PubMed] [Google Scholar]
- 20.Santarpia G, De Rosa S, Sabatino J, Curcio A, Indolfi C. Should we maintain anticoagulation after successful radiofrequency catheter ablation of atrial fibrillation? The need for a randomized study. Front Cardiovasc Med. 2017;4:85. doi: 10.3389/fcvm.2017.00085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vanassche T, Lauw MN, Eikelboom JW, Healey JS, Hart RG, Alings M, et al. Risk of ischaemic stroke according to pattern of atrial fibrillation: analysis of 6563 aspirin-treated patients in ACTIVE-A and AVERROES. Eur Heart J. 2015;36(5):281–287. doi: 10.1093/eurheartj/ehu307. [DOI] [PubMed] [Google Scholar]
- 22.Nuotio I, Hartikainen JE, Gronberg T, Biancari F, Airaksinen KE. Time to cardioversion for acute atrial fibrillation and thromboembolic complications. JAMA. 2014;312(6):647–649. doi: 10.1001/jama.2014.3824. [DOI] [PubMed] [Google Scholar]
- 23.Go AS, Reynolds K, Yang J, Gupta N, Lenane J, Sung SH, et al. Association of burden of atrial fibrillation with risk of ischemic stroke in adults with paroxysmal atrial fibrillation: the KP-RHYTHM study. JAMA Cardiol. 2018;3(7):601–608. doi: 10.1001/jamacardio.2018.1176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Al-Gibbawi M, Ayinde HO, Bhatia NK, El-Chami MF, Westerman SB, Leon AR, et al. Relationship between device-detected burden and duration of atrial fibrillation and risk of ischemic stroke. Heart Rhythm. 2021;18(3):338–346. doi: 10.1016/j.hrthm.2020.10.017. [DOI] [PubMed] [Google Scholar]
- 25.Rankin AJ, Rankin SH. Cardioverting acute atrial fibrillation and the risk of thromboembolism: not all patients are created equal. Clin Med (Lond) 2017;17(5):419–423. doi: 10.7861/clinmedicine.17-5-419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Healey JS, Connolly SJ, Gold MR, Israel CW, Van Gelder IC, Capucci A, et al. Subclinical atrial fibrillation and the risk of stroke. N Engl J Med. 2012;366(2):120–129. doi: 10.1056/NEJMoa1105575. [DOI] [PubMed] [Google Scholar]
- 27.Chao TF, Liu CJ, Chen SJ, Wang KL, Lin YJ, Chang SL, et al. Atrial fibrillation and the risk of ischemic stroke: does it still matter in patients with a CHA2DS2-VASc score of 0 or 1? Stroke. 2012;43(10):2551–2555. doi: 10.1161/STROKEAHA.112.667865. [DOI] [PubMed] [Google Scholar]
- 28.De Jong AM, Maass AH, Oberdorf-Maass SU, Van Veldhuisen DJ, Van Gilst WH, Van Gelder IC. Mechanisms of atrial structural changes caused by stretch occurring before and during early atrial fibrillation. Cardiovasc Res. 2011;89(4):754–765. doi: 10.1093/cvr/cvq357. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author upon reasonable request.