Abstract
Purpose of Review
To explore the immunological underpinnings of psychosis in the COVID-19 patients.
Recent Findings
COVID-19 pandemic led to a surge in psychiatric morbidities, including psychosis. Various putative biological and psychosocial changes have been implicated in COVID-19-related psychosis. COVID-19 is a proinflammatory state. Alterations in immunological processes both as a direct consequence of infection or secondary to the hyperimmune response heuristically explain the etiopathogenesis of psychosis in the affected individual. The uses of immunosuppressant and immunomodulatory drugs may be the other moderators of a psychotic presentation in COVID-19 patients. Evidence to substantiate this hypothesis is still lacking however, which further studies should address. Because of its management implications, a better understanding of the involved immunological mechanisms becomes extremely important.
Summary
Evidence suggests a putative role of immunological alterations in the pathogenesis of COVID-19-related psychosis. The immunological abnormalities are primarily attributed to the pathophysiology of COVID-19 infection, medications used, and stress.
Keywords: COVID-19, Psychosis, Immunological changes, Schizophrenia, Inflammation
Introduction
The COVID-19 pandemic has significantly affected almost all the countries globally. The world has witnessed several peaks in the number of cases and mortality in the form of waves. As per the World Health Organization (WHO) available data, the USA, India, Brazil, several European countries, and South American countries faced the most devastating aspect of the COVID-19 pandemic [1]. COVID-19 attributes significant morbidity and mortality globally with higher mortality rates in hospitalized patients, particularly those in intensive care units [2]. A systematic review and meta-analysis of the studies that measured the immediate and long-term mental health sequel of the severe acute respiratory syndrome (SARS) in the past reported that anxiety disorders, depressive disorders, and post-traumatic stress disorder are common immediate as well as long-term complications associated with SARS [3]. Recently, emerging shreds of evidence also support the findings of previous researches on SARS.
The impact of COVID-19 on mental health is an enormous and growing body of research that suggests that a significant number of people affected with COVID-19, people at risk, healthcare workers, and the general population at large develop mental health sequels due to COVID-19 [4–7]. A recent study retrospectively analyzed the electronic databases of patients with COVID-19 diagnosis in a sample of 236,379 patients and found that nearly one-third of the patients (33.62%) had a psychiatric or neurological diagnosis at 6 months follow-up period [8••]. Patients with COVID-19 diagnosis hospitalized to an intensive care unit had a psychiatric or neurological diagnosis, as high as 46.42% at 6 months follow-up [8••].
The impact of COVID-19 on the brain of the patients is significant. The SARS-CoV-2 infection produces brain inflammation mediated by specific immunologic mechanisms, which in turn impairs normal brain function [9]. The disrupted neurological function may be responsible for developing various neurological and psychiatric disorders, including cognitive impairment [9].
Over a period of 6 months follow-up after COVID-19 diagnosis, 17.4% of people diagnosed with anxiety disorder, whereas 1.40% people had diagnosable psychotic disorder; but, when it goes to the patients hospitalized in intensive care units, the diagnosis of anxiety disorder and psychosis at 6 months follow-up becomes 19.15% and 2.77%, respectively [8••].
COVID-19 Pathophysiology
It is crucial to understand the pathophysiology of COVID-19 because it causes multi-system involvement, including the central nervous system. In addition, various immunological changes that occur during the course of the disease can cause several neurological abnormalities and psychological issues.
COVID-19 is caused by the SARS-CoV-2 virus. SARS-CoV-2 belongs to the Coronaviridae family, members of which have led to significant outbreaks like Severe Acute Respiratory Syndrome Coronavirus-1 (SARS-CoV-1) in 2002 and Middle East respiratory syndrome Coronavirus (MERS-CoV) in 2012. Fortunately, they were contained before taking the shape of a pandemic. SAR-CoV-2 was thought to have a zoonotic transmission associated with the seafood market in Wuhan, China, in the initial part of the outbreak. Later it was realized that human-to-human transmission played a major role in the spreading of the disease. The mode of transmission is respiratory droplets or aerosol generated by sneezing and coughing of an infected person. Fomites also can be a source of transmission. Asymptomatic disease occurs in a significant fraction of the affected people, like in many other viral infections [10]. There are many false negative (in RT-PCR) cases that are asymptomatic but have close contact history. Many people remain asymptomatic or mildly symptomatic and untested [10]. Thus, the immune system plays a significant role.
The dysregulation of both innate and adaptive immune systems is responsible largely for the ultimate patient outcome. Cytokine storm is a major event in the later part of the illness that has caused many mortalities [11•]. Uncontrolled and excessive activation of cytokine-secreting cells leads to a poor prognosis in these patients. In addition, lymphocytopenia occurs in such cases. The study has demonstrated that a higher viral load of SARS-CoV-2 is negatively correlated with lymphocyte count. There is, however, a marked increase in neutrophil count and C-reactive protein (CRP) in patients with a high load of viruses in them. Overwhelming immune response results in the progression of severity [12]. Neurodegenerative changes have been seen to occur in patients of COVID-19 patients, and survivors have been speculated to develop psychiatric illnesses in the long run [13].
The virus has four structural proteins—the spike protein (S), membrane protein (M), envelop protein (E), and nucleocapsid (N) protein. Spike (S protein)–like structures on the virus’ surface act as the viral ligand for Angiotensin Convertase Enzyme-2 (ACE-2) receptors present in different parts like respiratory epithelium, lung alveolar cells, and GI epithelium. SARS-CoV-2 also uses transmembrane protease serine 2 (TMPRSS2) as a co-receptor. S protein has 2 subunits, S1 and S2. S1 is responsible for binding to the ACE-2 receptor and S2 causes fusion of viral and host cellular membranes. On binding to the ACE-2, S protein undergoes activation via a two-step protease cleavage: the first one for priming at the S1/S2 cleavage site (brought about by TMPRSS-2) and the second cleavage for activation at a position adjacent to a fusion peptide within the S2 subunit. The conformational change in S2 results in the fusion of membranes [10, 14, 15•].
Following the entry into the pulmonary epithelium, the viral replication occurs. The viral RNA, which is single-stranded positive sense, converts itself into negative-sense RNA, with the help of RNA-dependent RNA polymerase (RdRP) enzyme and helicase enzyme, then translates into viral proteins. This coincides with the formation of viral genomic RNA by replication. After translation, these viral structural proteins are localized to the Golgi bodies and endoplasmic reticulum, from where they reach the cell surface, budding occurs. New virions assembled along with total genomic RNA are thus released from the cell and ready to infect other nearby host cells. Apart from the four structural proteins, various non-structural proteins are also formed, which are responsible for various functions of the virus. Proteins like NSP-1 (non-structural protein-1) and ORF-8 (Open Reading Frame-8) have been seen to be responsible for downregulating the immune system and letting the virus evade it [16, 17] Viral proteins can inhibit antiviral response by blocking RIG I and IRF3/7 pathway on different levels, causing inefficient production of type 1 interferons and thus impaired antiviral response. In addition, they can increase NF-kB activation, proinflammatory cytokine production, and necroptosis. All these events can lead to increased hyper-inflammation, increased cellular death, and cytokine storm.
COVID-19 and Immunological Changes
Immunological changes are one of the hallmarks of COVID-19 [18]. A study on COVID-19 patients had revealed that the function of T cells and natural killer (NK) cells is critically altered in severely ill patients [18]. It is found that the ratio between interleukin-2 (IL-2) and interferon-gamma (IFN-γ) is one of the sensitive indicators of outcome and disease progression [18]. However, current evidence showed a high concentration of interleukin-6 (IL-6) related to severe illness [11•]. Postmortem examination lung tissue study has shown excessive infiltration of proinflammatory cells, mainly macrophages and T-helper 17 cells [11•]. Flow cytometric analyses have shown an increase in the percentage of CD4+ naïve T cells and decreased memory helper T cells in peripheral blood. The percentage of cytotoxic T cells and regulatory T (Treg) cells is lower [10].
Specific or adaptive immune system response includes virus-specific neutralizing antibodies. IgM antibodies develop and reach a peak within about 7–10 days of illness in the acute phase. This is subsequent to the development of IgG antibodies, which protect from the virus for a longer time. This is peculiar to SARS-CoV-2 infection [10]. Also, IgM and IgG levels were seen to be higher in severely ill patients [10]. In addition, IgA antibodies can provide mucosal immunity. CRP, lactate dehydrogenase (LDH), procalcitonin, ferritin, erythrocyte sedimentation rate (ESR), D-dimer, etc. have also been used as predictors for progressive severity. All these factors can have an impact in the long run on the brain and cause neuropsychiatric manifestations [19•]. They also contribute to disseminated intravascular coagulation (DIC), thrombocytopenia, and decreased fibrinogen in patients leading to thromboembolic phenomenon and bleeding manifestations, which can have neurological manifestations in the survivors.
Drugs like tocilizumab, corticosteroids, antimalarials (hydroxychloroquine), and ivermectin have been tried at different stages of illness (different severity level) to find any beneficial outcome in COVID-19 patients. These are immunomodulatory drugs and have adverse effects like producing psychiatric symptoms. These drugs have been implicated in inducing psychiatric manifestations when used in other illnesses [20]. For example, steroid can produce mood symptoms; psychosis-like symptoms may be induced by hydroxychloroquine.
COVID-19 and Psychosis
The association of psychosis with viral pandemics is well known. During the influenza pandemic of the early 1900s, there was an emergence of more number of cases of psychosis [21••].
Evidences support the association of SARS-CoV-2 infection with inflammatory changes in the brain and possible new-onset psychosis [6]. Psychosis in COVID-19 can be explained by various mechanisms [22]:
Immune and inflammatory changes occurring in the brain in COVID-19 patients may be responsible for the development of psychosis.
Patients with COVID-19 with relatively severe symptoms often receive steroids, and steroid is implicated in the development of psychosis.
Several patients with COVID-19 also receive treatments like chloroquine and hydroxychloroquine, which are known to induce psychotic symptoms like hallucinations and delusions [21••].
The circumstantial changes happening during COVID-19 are stressful, and it is well known that stress is a precipitating factor for psychosis, and it may flare up the psychotic symptoms in patients with pre-existing psychotic disorders.
There have been reports that found the association of psychosis (new-onset) with elevated C-reactive protein, serum ferritin, lactate dehydrogenase (LDH), D-dimer, and derangements of white blood cells [21••, 23–25]. In addition, it has been reported that autoimmune changes in the brain due to the development of novel neuronal autoantibody are associated with psychosis which is refractory to antipsychotic treatment [26].
The clinical presentation of psychosis in COVID-19 patients is often characterized by delusions along with features of confusion [23]. A study from China reported that COVID-19 patients with new-onset mental illness report delusion, insomnia, and aggressive symptoms more frequently than non-COVID-19 patients with new-onset mental illness [25]. The psychotic symptoms in the COVID-19 patient may persist for a longer time, and the symptoms may present despite normal neuroimaging findings, electroencephalogram (EEG), and cerebrospinal fluid examination [27]. The development of psychosis in COVID-19 patients may occur without a family history of psychosis [28–31]. COVID-19 patients with new-onset psychosis may respond well to antipsychotic treatment with rapid resolution of their psychotic symptoms [32]. The onset of psychotic symptoms may occur in a later age without any prior history of psychosis [30, 31]. It has been seen that delirium has prominent psychotic features, which is often misdiagnosed as psychosis [33••]. The brain changes that occur in COVID-19 patients may result in a delirious state. Therefore, the clinician needs to be cautious while making a diagnosis of psychosis in COVID-19 patients.
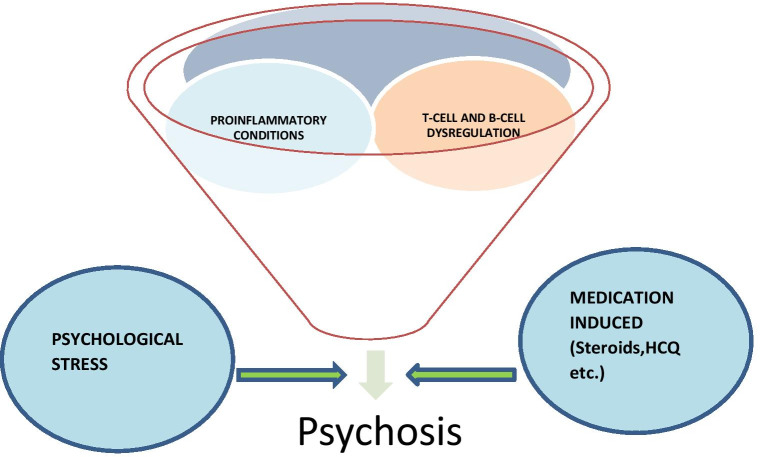
There is robust evidence regarding the association of immune-biological factors in the development of psychosis in COVID-19 patients (Fig. 1), alternate explanations like the role of socio-cultural factors and stress associated with COVID-19 pandemic in the development of psychosis [29, 34].
Fig. 1.
Factors attributing to psychosis in COVID-19 affected individuals
Immunological Links Associated with Psychosis
Inflammatory response associated with COVID-19 virus infection is thought to be implicated with neuropsychiatric symptoms through various immunological mechanisms [35, 36]. COVID-19 is a proinflammatory state, and several inflammatory cytokines like IL-6, IL-8, IL-10, tumor necrosis factor-alpha, IL-2R, and CRP are elevated in a patient suffering from COVID-19 infection [37]. These inflammatory markers are the predictors of the impending cytokine storm and reflect the disease severity.
Now, we have ample evidence suggesting COVID-19 as a proinflammatory condition, but how does this state paved a fertile base for a neuropsychiatric illness like psychosis? The answer lies in the exhaustive literature highlighting the immunological basis of psychosis [38••, 39–41]. Several studies done so far link the numerous psychiatric symptoms, including psychosis, with an underlying immunological process. Hematological [42••, 43] and CSF findings [44] in patients with psychosis have shown increased levels of inflammatory markers. For instance, the post-mortem brain studies have revealed diverse immunological and inflammatory abnormalities in the patients suffering from schizophrenia [42••, 43]. Also, studies have demonstrated an increase in proinflammatory state in the same patients. The explanatory model is the “vulnerability stress inflammation model,” which explains an underlying predisposition associated with the state and a stressful state that could trigger a psychotic episode [41].
The major finding of the several studies shows that the major inflammatory abnormalities are:
Microglial activation and proliferation in dorsolateral prefrontal, anterior cingulate, superior temporal cortices regions of the brain, areas of functional relevance to psychosis
Upregulation of proinflammatory cytokines and matrix metalloproteinases
Other findings include increased “CSF: serum albumin ratio” (studied in patients with drug-resistant schizophrenia) which reflects the blood-CSF barrier hyperpermeability; elevated neopterin and elevated cytokines levels; and increased intrathecal synthesis of IgM, IgG, or IgA [45]. This along with microglial activation and proliferation and increased blood-brain permeability and disruption of blood-CSF barrier leads to trans-endothelial inflammatory cell migration and increased interaction between the brain’s innate immunity and peripheral adaptive immunity.
Recent studies have postulated the underlying immunological process in the development of psychosis and have suggested an entity named autoimmune psychosis and have also attempted to given an etiological subclassification for the same [38••] (Table 1).
Table 1.
Etiological subclassification of autoimmune psychosis
| Psychosis associated with classical systemic inflammatory and autoimmune disorders | Neuropsychiatric systemic lupus erythematosus, neuroinflammatory disorders, autoimmune encephalitis associated with antibodies targeting intracellular antigens |
| Psychosis associated with autoantibodies targeting well-characterized synaptic and neuronal cell surface proteins | NMDAR antibodies, AMPAR antibodies, GABAßR antibodies, VGKC-protein complex antibodies, CASPR2 antibodies |
| Seronegative but probable autoimmune encephalitis (SPAP) |
Unknown autoantibodies Innate- or T-cell autoimmunity? |
CNS autoimmune and inflammatory disorders like systemic lupus erythematosus, autoimmune vasculitides, multiple sclerosis, and sarcoidosis are also implicated in psychosis. It has been demonstrated that many of the autoimmune illnesses are associated with alleles in the MHC region (located on chromosome 6). HLA association has emerged as a consistent genome-wide association study (GWAS) finding in schizophrenia. Apart from the HLA association, studies have also focused on the associations with genes expressed in B-lymphocyte lineages (CD19 and CD20). GWAS has postulated the following major processes based on the findings: T-cell activation; B-cell activation; role of transforming growth factor-β signalling. Animal studies have also demonstrated C4 variations regulating the microglia-mediated synaptic pruning in schizophrenia [45].
Studies have also shown the immunological link with the development of postpartum psychosis. The proinflammatory state during pregnancy changes the levels of various cytokines, chemokines, and growth factors. There is an increase in the levels of transforming growth factor-beta (TGF-b), and IL-4, IL-5, IL-10, and a decrease in IL-2, interferon-gamma (INF-g), and tumor necrosis factor-a (TNF-a)[41]. Also, there are peculiar changes in the maternal immune cells with increased activity of regulatory T cells, macrophages, granulocytes, and natural killer cells.
Apart from studies in the patients with postpartum psychosis, increased in number of B-cell have also been studied in acute un-medicated psychosis. Steiner et al. in the flow cytometry studies have found higher CD19+ lymphocyte populations in patients with acute psychosis who were in an un-medicated group than healthy controls [46]. A similar study found that after 6 weeks of antipsychotic treatment, there were decreased CD19+ lymphocyte populations. Miller et al. in a meta-analysis have found an increased CD4:CD8 ratio and reduced CD3+ lymphocyte levels in patients versus controls. These findings were reversed after antipsychotic medication. A previous study by Maino et al. also found the elevated CD19+ counts and reduced CD3+ counts in un-medicated patients which decreased to within normal level after antipsychotic treatment [47]. Thus, all these studies suggest strong immunological links associated with psychosis.
Implications
Thus far, we have discussed the direct and indirect ways in which COVID-19 may lead to the advent of psychosis. It is clear that the neural inflammation and acute systemic inflammatory states can lead to the development of psychosis. COVID-19 infection being a proinflammatory state may have a possible role in developing psychosis in affected individuals. To our knowledge, no existing studies delineate the individual contributions of risk states, including the various biological and psychosocial alterations during the pandemic. Thus, the potential immune system–mediated psychosis described here concerning COVID-19 infection warrants further investigation. It may have diagnostic and management implications. It will lead the healthcare providers to make treatment decisions such as whether to manage both the disorders independently or simply manage the COVID-19 inflammation, eventually resolving co-occurring psychosis.
Furthermore, the role of immunomodulators and cytokine inhibitors in the management will become clearer. Similarly, answers to the complex enquiries related to glucocorticoid use, a causal factor for psychosis, in COVID-19 management are needed. Besides, research investigating the association of clinical characteristics and outcomes of psychosis with COVID-19 inflammatory markers in the brain and periphery will help in establishing the causal relationship.
Declarations
Conflict of Interest
The authors declare no competing interests.
Research Involving Human Participants and/or Animals
None.
Informed Consent
Not applicable.
Footnotes
This article is part of the Topical Collection on Psychosis
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Presentation at a meeting: Nil
Contributor Information
Sarvodaya Tripathy, Email: sarvo_trip@yahoo.co.in.
Nitika Singh, Email: nitikasingh57@gmail.com.
Amit Singh, Email: amitsingh0612@gmail.com.
Sujita Kumar Kar, Email: skkar1981@yahoo.com.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
- 1.World Health Organization. Coronavirus disease (COVID-19) pandemic [Internet]. 2021 [cited 2021 Jun 10]. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019. Accessed 10 Jun 2021.
- 2.Abate SM, Checkol YA, Mantefardo B. Global prevalence and determinants of mortality among patients with COVID-19: a systematic review and meta-analysis. Ann Med Surg 2012. 2021;64:102204. doi: 10.1016/j.amsu.2021.102204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chau SWH, Wong OWH, Ramakrishnan R, Chan SSM, Wong EKY, Li PYT, et al. History for some or lesson for all? A systematic review and meta-analysis on the immediate and long-term mental health impact of the 2002–2003 Severe Acute Respiratory Syndrome (SARS) outbreak. BMC Public Health. 2021;21:670. doi: 10.1186/s12889-021-10701-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Aranda Z, Rodríguez-Cuevas FG. COVID-19 and global mental health. Lancet Psychiatry. Elsevier. 2021;8:457. doi: 10.1016/S2215-0366(21)00124-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kar SK, Oyetunji TP, Prakash AJ, Ogunmola OA, Tripathy S, Lawal MM, et al. Mental health research in the lower-middle-income countries of Africa and Asia during the COVID-19 pandemic: a scoping review. Neurol Psychiatry Brain Res. 2020;38:54–64. doi: 10.1016/j.npbr.2020.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Psychiatry TL. COVID-19 and mental health. Lancet Psychiatry. Elsevier. 2021;8:87. doi: 10.1016/S2215-0366(21)00005-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Taquet M, Luciano S, Geddes JR, Harrison PJ. Bidirectional associations between COVID-19 and psychiatric disorder: retrospective cohort studies of 62 354 COVID-19 cases in the USA. Lancet Psychiatry. Elsevier. 2021;8:130–40. doi: 10.1016/S2215-0366(20)30462-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Taquet M, Geddes JR, Husain M, Luciano S, Harrison PJ. 6-month neurological and psychiatric outcomes in 236 379 survivors of COVID-19: a retrospective cohort study using electronic health records. Lancet Psychiatry. Elsevier. 2021;8:416–27. doi: 10.1016/S2215-0366(21)00084-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Alnefeesi Y, Siegel A, Lui LMW, Teopiz KM, Ho RCM, Lee Y, et al. Impact of SARS-CoV-2 infection on cognitive function: a systematic review. Front Psychiatry. 2020;11:621773. doi: 10.3389/fpsyt.2020.621773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Azkur AK, Akdis M, Azkur D, Sokolowska M, van de Veen W, Brüggen M-C, et al. Immune response to SARS-CoV-2 and mechanisms of immunopathological changes in COVID-19. Allergy. 2020;75:1564–81. doi: 10.1111/all.14364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tang Y, Liu J, Zhang D, Xu Z, Ji J, Wen C. Cytokine storm in COVID-19: the current evidence and treatment strategies. Front Immunol. 2020;11:1708. doi: 10.3389/fimmu.2020.01708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen W, Xiao Q, Fang Z, Lv X, Yao M, Deng M. Correlation analysis between the viral load and the progression of COVID-19. Comput Math Methods Med. 2021;2021:9926249. doi: 10.1155/2021/9926249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Savage M. Coronavirus: the possible long-term mental health impacts [Internet]. [cited 2021 Jul 2]. Available from: https://www.bbc.com/worklife/article/20201021-coronavirus-the-possible-long-term-mental-health-impacts
- 14.Parasher A. COVID-19: current understanding of its pathophysiology, clinical presentation and treatment. Postgrad Med J. The Fellowship of Postgraduate Medicine. 2021;97:312–20. doi: 10.1136/postgradmedj-2020-138577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yuki K, Fujiogi M, Koutsogiannaki S. COVID-19 pathophysiology: a review. Clin Immunol Orlando Fla. 2020;215:108427. doi: 10.1016/j.clim.2020.108427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thoms M, Buschauer R, Ameismeier M, Koepke L, Denk T, Hirschenberger M, et al. Structural basis for translational shutdown and immune evasion by the Nsp1 protein of SARS-CoV-2. Science. American Association for the Advancement of Science. 2020;369:1249–55. doi: 10.1126/science.abc8665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Park MD. Immune evasion via SARS-CoV-2 ORF8 protein? Nat Rev Immunol. 2020;20:408–408. doi: 10.1038/s41577-020-0360-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kalicińska E, Szymczak D, Zińczuk A, Adamik B, Smiechowicz J, Skalec T, et al. Immunosuppression as a hallmark of critical COVID-19: prospective study. Cells. 2021;10:1293. doi: 10.3390/cells10061293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kar SK, Verma P, Verma SK, Saxena SK. Mental illnesses among COVID-19 patients: possible immunological underpinnings. Asian J Psychiatry. 2020;53:102376. doi: 10.1016/j.ajp.2020.102376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bascones-Martinez A, Mattila R, Gomez-Font R, Meurman JH. Immunomodulatory drugs: oral and systemic adverse effects. Med Oral Patol Oral Cir Bucal. 2014;19:e24–31. doi: 10.4317/medoral.19087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nakamura ZM, Nash RP, Laughon SL, Rosenstein DL. Neuropsychiatric complications of COVID-19. Curr Psychiatry Rep. 2021;23:25. doi: 10.1007/s11920-021-01237-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ferrando SJ, Klepacz L, Lynch S, Tavakkoli M, Dornbush R, Baharani R, et al. COVID-19 psychosis: a potential new neuropsychiatric condition triggered by novel coronavirus infection and the inflammatory response? Psychosomatics. 2020;61:551–5. doi: 10.1016/j.psym.2020.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Parra A, Juanes A, Losada CP, Álvarez-Sesmero S, Santana VD, Martí I, et al. Psychotic symptoms in COVID-19 patients. A retrospective descriptive study. Psychiatry Res. 2020;291:113254. doi: 10.1016/j.psychres.2020.113254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Paterson RW, Brown RL, Benjamin L, Nortley R, Wiethoff S, Bharucha T, et al. The emerging spectrum of COVID-19 neurology: clinical, radiological and laboratory findings. Brain J Neurol. 2020;143:3104–20. doi: 10.1093/brain/awaa240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Xie Q, Fan F, Fan X-P, Wang X-J, Chen M-J, Zhong B-L, et al. COVID-19 patients managed in psychiatric inpatient settings due to first-episode mental disorders in Wuhan, China: clinical characteristics, treatments, outcomes, and our experiences. Transl Psychiatry. 2020;10:1–11. doi: 10.1038/s41398-019-0665-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.McAlpine LS, Lifland B, Check JR, Angarita GA, Ngo TT, Pleasure SJ, et al. Remission of subacute psychosis in a COVID-19 patient with an antineuronal autoantibody after treatment with intravenous immunoglobulin. Biol Psychiatry. 2021;90(4):e23–6. 10.1016/j.biopsych.2021.03.033. [DOI] [PMC free article] [PubMed] [Retracted]
- 27.Lim ST, Janaway B, Costello H, Trip A, Price G. Persistent psychotic symptoms following COVID-19 infection. BJPsych Open. 2020;6(5):e105. 10.1192/bjo.2020.76. [DOI] [PMC free article] [PubMed]
- 28.Noone R, Cabassa JA, Gardner L, Schwartz B, Alpert JE, Gabbay V. Letter to the editor: New onset psychosis and mania following COVID-19 infection. J Psychiatr Res. 2020;130:177–9. doi: 10.1016/j.jpsychires.2020.07.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chandra PS, Shiva L, Nagendrappa S, Ganjekar S, Thippeswamy H. COVID 19 related psychosis as an interface of fears, socio-cultural issues and vulnerability- case report of two women from India. Psychiatry Res. 2020;290:113136. doi: 10.1016/j.psychres.2020.113136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Parker C, Slan A, Shalev D, Critchfield A. Abrupt Late-onset psychosis as a presentation of coronavirus 2019 disease (COVID-19): a longitudinal case report. J Psychiatr Pract. 2021;27:131–6. doi: 10.1097/PRA.0000000000000533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Smith CM, Komisar JR, Mourad A, Kincaid BR. COVID-19-associated brief psychotic disorder. BMJ Case Rep CP. BMJ Specialist Journals. 2020;13:e236940. doi: 10.1136/bcr-2020-236940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Alba L, Coll C, Sáez S, Alonso L, Pérez H, Palma S, et al. New-onset psychosis: a case report of brief psychosis related to COVID-19 infection. Psychiatry Res. 2021;301:113975. doi: 10.1016/j.psychres.2021.113975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Watson CJ, Thomas RH, Solomon T, Michael BD, Nicholson TR, Pollak TA. COVID-19 and psychosis risk: real or delusional concern? Neurosci Lett. 2021;741:135491. doi: 10.1016/j.neulet.2020.135491. [DOI] [PubMed] [Google Scholar]
- 34.Valdés-Florido MJ, López-Díaz Á, Palermo-Zeballos FJ, Martínez-Molina I, Martín-Gil VE, Crespo-Facorro B, et al. Reactive psychoses in the context of the COVID-19 pandemic: clinical perspectives from a case series. Rev Psiquiatr Salud Ment Engl Ed. 2020;13:90–4. doi: 10.1016/j.rpsm.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pfefferbaum B, North CS. Mental health and the Covid-19 pandemic. N Engl J Med. Mass Medical Soc; 2020;383(6):510–2. [DOI] [PubMed]
- 36.Troyer EA, Kohn JN, Hong S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain Behav Immunol. 2020;87:34–9. [DOI] [PMC free article] [PubMed]
- 37.Meftahi GH, Jangravi Z, Sahraei H, Bahari Z. The possible pathophysiology mechanism of cytokine storm in elderly adults with COVID-19 infection: the contribution of “inflame-aging”. Inflamm Res. Springer. 2020;69:825–39. doi: 10.1007/s00011-020-01372-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Al-Diwani AA, Pollak TA, Irani SR, Lennox BR. Psychosis: an autoimmune disease? Immunology. Wiley Online Library. 2017;152:388–401. doi: 10.1111/imm.12795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jeppesen R, Benros ME. Autoimmune diseases and psychotic disorders. Front Psychiatry. Frontiers. 2019;10:131. doi: 10.3389/fpsyt.2019.00131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jones AL, Mowry BJ, Pender MP, Greer JM. Immune dysregulation and self-reactivity in schizophrenia: do some cases of schizophrenia have an autoimmune basis? Immunol Cell Biol. Wiley Online Library. 2005;83:9–17. doi: 10.1111/j.1440-1711.2005.01305.x. [DOI] [PubMed] [Google Scholar]
- 41.Maguire J. Hormonal and immunological factors in postpartum psychosis. In Biomark of Postpartum Psychiatric Disorders. Academic Press. Elsevier; 2020. p. 159–79. 10.1016/C2017-0-03209-3.
- 42.Miller BJ, Buckley P, Seabolt W, Mellor A, Kirkpatrick B. Meta-analysis of cytokine alterations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry. Elsevier. 2011;70:663–71. doi: 10.1016/j.biopsych.2011.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Goldsmith DR, Rapaport MH, Miller BJ. A meta-analysis of blood cytokine network alterations in psychiatric patients: comparisons between schizophrenia, bipolar disorder and depression. Mol Psychiatry. Nature Publishing Group. 2016;21:1696–709. doi: 10.1038/mp.2016.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bechter K, Reiber H, Herzog S, Fuchs D, Tumani H, Maxeiner HG. Cerebrospinal fluid analysis in affective and schizophrenic spectrum disorders: identifying subgroups with immune responses and blood–CSF barrier dysfunction. J Psychiatr Res. Elsevier. 2010;44:321–30. doi: 10.1016/j.jpsychires.2009.08.008. [DOI] [PubMed] [Google Scholar]
- 45.Consortium SWG of the PG Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511:421–7. doi: 10.1038/nature13595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Steiner J, Jacobs R, Panteli B, Brauner M, Schiltz K, Bahn S, et al. Acute schizophrenia is accompanied by reduced T cell and increased B cell immunity. Eur Arch Psychiatry Clin Neurosci. Springer. 2010;260:509–18. doi: 10.1007/s00406-010-0098-x. [DOI] [PubMed] [Google Scholar]
- 47.Maino K, Gruber R, Riedel M, Seitz N, Schwarz M, Müller N. T-and B-lymphocytes in patients with schizophrenia in acute psychotic episode and the course of the treatment. Psychiatry Res. Elsevier. 2007;152:173–80. doi: 10.1016/j.psychres.2006.06.004. [DOI] [PubMed] [Google Scholar]