Abstract
Right-to-left (RTL) interatrial shunt (IAS) may complicate select cases of COVID-19 pneumonia. We describe the use of serial imaging to monitor shunt in critically ill patients. A 52-year-old man presented with COVID-19 pneumonia. Hypoxia worsened despite maximal medical therapy and non-invasive ventilation. On day 8, saline microbubble contrast-enhanced transthoracic echocardiography revealed a patent foramen ovale (PFO) with RTLIAS. Invasive ventilation was initiated the next day. The course was complicated by intermittent severe desaturation without worsening aeration or haemodynamic instability, so PFO closure was considered. However, on day 12, saline microbubble contrast-enhanced transoesophageal echocardiography excluded RTLIAS. The patient was extubated on day 27 and discharged home 12 days later. Thus, RTLIAS may be dynamic and changes can be detected and monitored by serial imaging. Bedside echocardiography with saline microbubble contrast, a simple, minimally invasive bedside test, may be useful in the management of patients with severe hypoxia.
Keywords: adult intensive care, mechanical ventilation, pneumonia (respiratory medicine), COVID-19
Background
There is significant heterogeneity in the respiratory phenotype of COVID-19.1 2 The pathogenesis is complex and is still being elucidated. It differs from other causes of hypoxic respiratory failure in that ongoing viral activity within already damaged tissues can cause a spectrum of anatomically and temporally distinct pathologies.3 These include tracheobronchitis, diffuse alveolar damage and vascular injuries.3
Thus, COVID-19 involves, to a variable extent, loss of aeration, dysregulation of pulmonary vascular tone and microvascular thrombosis.3 4 Pulmonary embolism is also common.3 4 These thrombi should increase dead space. However, a series of critically ill patients with acute respiratory distress syndrome induced by COVID-19 had a significant right-to-left (RTL) shunt despite relatively normal lung compliance.1 Indeed, it has been demonstrated that both intrapulmonary shunt and acute RTL interatrial shunt (IAS) can contribute to hypoxia in COVID-19.5 6
The sequelae of COVID-19 can induce or exacerbate pulmonary hypertension and thereby precipitate RTLIAS.6–8 The standard approach to the management of hypoxia may also worsen RTLIAS.6–9 However, no previous reports have described the use of serial imaging to monitor changes in RTLIAS. We describe the use of saline microbubble contrast-enhanced echocardiography to demonstrate that, when present in patients with COVID-19, IAS may be dynamic.
Case presentation
A 52-year-old man with no medical history presented with fever, cough and breathlessness due to COVID-19. His oxygenation gradually deteriorated despite treatment with antibiotics, dexamethasone and tocilizumab (table 1). Eight days after admission, he developed acute severe hypoxia (oxygen saturation (SpO2) 85%; arterial oxygen pressure 54 mm Hg) despite high-flow nasal oxygen (fractional inspired oxygen (FiO2) 0.7, 60 L/min). Oxygenation initially improved with awake prone positioning.
Table 1.
Timeline correlating arterial blood gases and respiratory support with select investigations and treatment
| Day | 0 | 1 | 2 | 2 | 3 | 3 | 8 | 8 | 9 | 10 | 11 | 11 | 12 | 27 | 33 | 39 |
| Time | 10:00 | 01:10 | 10:20 | 14:10 | 08:25 | 20:00 | 12:50 | 21:20 | 15:50 | 04:40 | 04:10 | 16:30 | 03:40 | 12:40 | 17:10 | 10:00 |
| Intervention | COVID-19 PCR positive | 07:00 steroid started | Day 4 15:00 tocilizumab given |
15:00 TTE PFO and RTLIAS anticoagulated |
13:00 tracheal intubation |
13:00 TOE no RTLIAS |
Tracheal extubation |
Home | ||||||||
| Lymphocytes, ×109/L | 0.67 | 0.92 | 0.79 | 0.53 | 0.65 | 0.73 | 2.45 | |||||||||
| CRP, mg/L | 76 | 70 | 39 | 7 | 17 | 27 | ||||||||||
| Ferritin, µg/L | 1847 | 2925 | 1979 | 785 | 892 | |||||||||||
| D-dimer, mg/L | 0.54 | 0.55 | 0.74 | 9.65 | 1.95 | 1.12 | ||||||||||
| LDH, U/L | 791 | 901 | 1167 | 726 | ||||||||||||
| SpO2, % | 94 | 91 | 93 | 90 | 92 | 91 | 84 | 94 | 91 | 98 | 98 | 92 | 95 | 92 | 95 | 95 |
| RR, breaths/min | 24 | 29 | 29 | 30 | 36 | 35 | 29 | 28 | 24 | 32 | 30 | 30 | 30 | 32 | 22 | 21 |
| Position | Sitting | Sitting | Sitting | Sitting | Sitting | Prone | Sitting | Prone | Supine | Prone | Supine | Supine | Prone | Sitting | Sitting | |
|
Respiratory
support |
NC | FM | NRB FM | NRB FM | HFNC | HFNC | Helmet BiPAP |
ETT VC+ |
ETT VC AC |
ETT VC AC |
ETT VC AC |
ETT VC AC |
HFNC | NC | ||
| FiO2 | 0.21 | 0.35 | 0.7 | 1.0 | 1.0 | 0.6 | 0.5 | 0.5 | 0.4 | 0.5 | 0.21 | |||||
| Flow, L/min | 1 | 10 | 12 | 15 | 40 | 60 | 60 | 60 | 60 | 50 | 50 | 50 | 50 | 1.5 | ||
| PEEP, cmH20 | 10 | 14 | 14 | 12 | 12 | 10 | ||||||||||
| Peak AP, cmH20 | 16 | 35 | 34 | 38 | 37 | 35 | ||||||||||
| VT, mL | 450 | 325 | 441 | 437 | 438 | |||||||||||
| Compliance, mL/cmH20* | 21.4 | 16.3 | 17.0 | 17.5 | 17.5 | |||||||||||
| pH | 7.46 | 7.45 | 7.46 | 7.49 | 7.45 | 7.28 | 7.24 | 7.37 | 7.491 | 7.51 | 7.5 | 7.49 | ||||
| PaCO2, mm Hg | 35 | 37 | 35 | 31 | 35 | 49 | 66 | 51 | 39.8 | 37 | 41 | 36 | ||||
| BE, mmol/L | 1.1 | 1.3 | 1.2 | 0.6 | 0.2 | −4.2 | −1.2 | 2.6 | 6 | 5.6 | 7.6 | 3.6 | ||||
| PaO2, mm Hg | 52 | 57 | 62 | 43 | 67 | 71 | 97 | 71 | 57.6 | 68 | 78 | 66 | ||||
| PaO2/FiO2 | 51 | 67 | 71 | 162 | 142 | 115 | 171 | 155 |
Timeline correlating arterial blood gases and respiratory support with select investigations and treatment. Although SpO2 intermittently fell below 80%, each incident triggered an assessment by a physician and a series of interventions. So, the arterial blood gas analyses presented reflect the patient’s oxygenation after adjustment of respiratory support. For example, on day 9, oxygenation did not improve significantly after endotracheal ventilation and initiation of mechanical ventilation so the patient was positioned prone. The arterial blood gas analyses, performed at 04:40 on day 10 and 03:40 on day 12, reflect the improvement in oxygenation after several hours of prone positioning and reduction of PEEP.
*Dynamic compliance was calculated.
VC AC, volume control-assist control; Peak AP, peak airway pressure; BE, base excess; BiPAP, bilevel positive airway pressure; CRP, C reactive protein; ETT, endotracheal tube; FiO2, fractional inspired oxygen; FM, face mask; HFNC, high-flow nasal cannula; LDH, lactate dehydrogenase; NC, nasal cannula; NRB, non-rebreathe; PaCO2, arterial carbon dioxide pressure; PaO2, arterial oxygen pressure; PEEP, positive end expiratory pressure; PFO, patent foramen ovale; RR, respiratory rate; RTLIAS, right-to-left interatrial shunt; SpO2, oxygen saturation; TOE, transoesophageal echocardiography; TTE, transthoracic echocardiography; VC+, volume control plus; VT, tidal volume.
Investigations
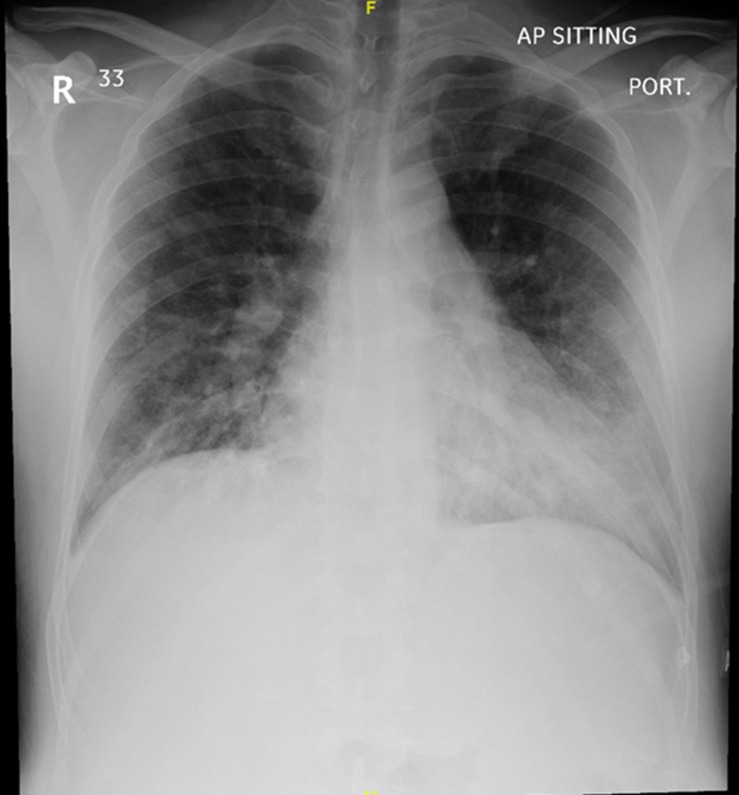
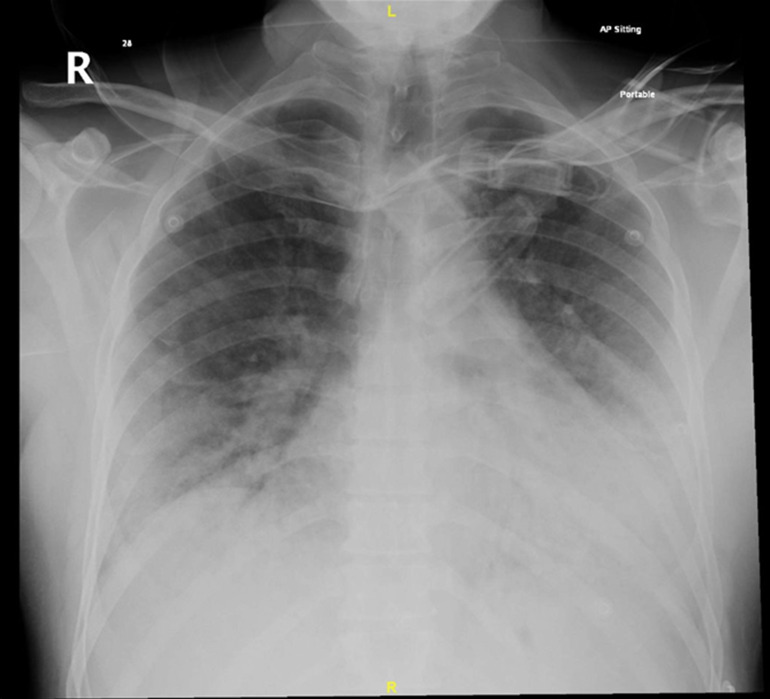
Table 1 correlates the patients’ inflammatory markers, blood gases and treatment over the course of his illness. The chest X-ray on admission (figure 1) revealed bilateral patchy interstitial oedema, and COVID-19 PCR was positive. On day 8, the infiltrates on the chest X-ray had worsened (figure 2) and lung ultrasound revealed bilateral patchy B-pattern. However, the degree of aeration loss shown by imaging was not thought to be sufficient to explain the severity of the hypoxaemia, so additional diagnoses were considered.
Figure 1.
Chest X-ray on admission. Chest X-ray demonstrating bilateral patchy alveolar infiltrate.
Figure 2.
Chest X-ray on day 8. Chest X-ray demonstrating worsening bilateral patchy alveolar infiltrate in comparison with the chest X-ray performed on admission (figure 1).
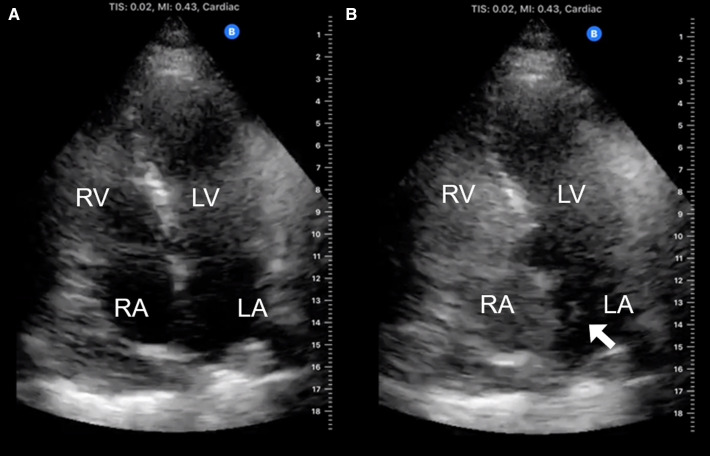
The D-dimer was raised (9.65 mg/L), but clinical instability initially precluded CT. However, bedside ultrasound excluded deep vein thrombosis and acute cor pulmonale. Sputum cultures were negative. The patient’s ECG was unremarkable and although his heart appeared normal on transthoracic echocardiography (TTE), fixed bowing of the interatrial septum to the left (video 1) suggested that the right atrial (RA) pressure was greater than the left atrial (LA) pressure. Saline microbubble contrast-enhanced TTE also demonstrated a patent foramen ovale (PFO) with RTLIAS (figure 3 and video 1).
Video 1.
Figure 3.
Transthoracic echocardiography (TTE) demonstrating right-to-left interatrial shunt in a patient receiving high-flow nasal oxygen. (A) Labelled 2D apical four-chamber TTE view of the heart. (B) Labelled 2D apical four-chamber TTE view of the heart after intravenous injection of saline microbubble contrast demonstrating grade 2 shunt (5–25 bubbles in LA). Bubbles appeared in the LA within three cardiac cycles of opacification of the RA demonstrating the presence of a right-to left interatrial shunt. LA, left atrium; LV, left ventricle; RA, right atrium; RV, right ventricle.
Thereafter, it was noted that, despite unchanged aeration and haemodynamic conditions, the SpO2 intermittently fell below 80%. Each of these episodes triggered an assessment by a physician and a series of interventions (described below). So, the arterial blood gas analyses presented in table 1 reflect the oxygenation after adjustment of respiratory support. For example, on day 9, the patient was positioned prone when oxygenation did not improve significantly after endotracheal ventilation and initiation of mechanical ventilation. The arterial blood gas analyses, performed at 04:40 on day 10 and 03:40 on day 12, reflect the improvement in oxygenation after several hours of prone positioning and reduction of positive end expiratory pressure (PEEP).
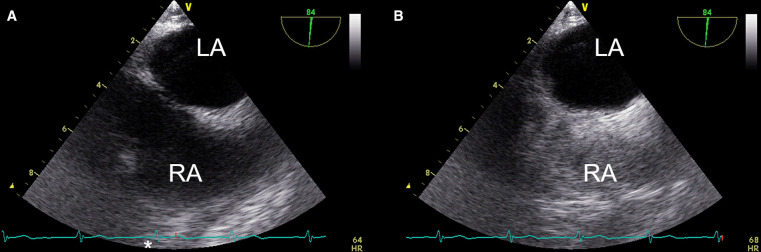
Percutaneous closure of the PFO was considered. However, on day 12, saline microbubble contrast-enhanced transoesophageal echocardiography (TOE), performed while the patient was supine, demonstrated absence of RTLIAS (figure 4 and video 2). Bowing of the interatrial septum to the right with mid-systolic buckling (video 2) suggested that the pulmonary capillary wedge pressure (PCWP) and the pressure gradient between the LA and the RA had normalised. On day 20, CT pulmonary angiography (CTPA) demonstrated consolidation, diffuse ground-glass opacities and small pleural effusions but excluded pulmonary emboli.
Figure 4.
Transoesophageal echocardiography (TOE) excluding right-to-left interatrial shunt. TOE demonstrating right-to-left interatrial shunt in a patient receiving high-flow nasal oxygen. (A) Labelled 2D mid-oesophageal bicaval TOE view of the heart. (B) Labelled 2D mid-oesophageal bicaval TOE view of the heart after intravenous injection of saline microbubble contrast demonstrating grade 2 shunt (5–25 bubbles in LA). Bubbles appeared in the LA within three cardiac cycles of opacification of the RA demonstrating the presence of a right-to left interatrial shunt. LA, left atrium; RA, right atrium.
Video 2.
Differential diagnosis
Although the patient was diagnosed with COVID-19 pneumonia, he deteriorated suddenly 8 days after admission. The aetiology of the hypoxaemia was thought to be multifactorial. While COVID-19 had caused significant lung injury, the severity of the changes on chest X-ray (figure 2) and lung ultrasound was not thought to be sufficient to explain the severity of the patient’s hypoxaemia (table 1). Pulmonary emboli, mucus plugging and superimposed bacterial infection were considered. However, CTPA and respiratory cultures were subsequently negative.
Oxygenation improved with prone positioning but not with dorsal recumbency. So, diagnostic criteria for platypnoea–orthodeoxia were not fulfilled. Regardless, the presence of relevant RTLIAS using saline microbubble contrast-enhanced echocardiography on day 8 suggested that this shunt contributed to the patient’s severe hypoxaemia. The subsequent observation that this RTLIAS had resolved on day 12, paralleling an improvement in oxygenation and normalisation of the transatrial pressure gradient, demonstrates that RTLIAS, when present, may be dynamic. Thus, intermittent reversal of IAS may have contributed to the episodic, sudden, severe desaturation.
Treatment
In view of the sudden severe deterioration on day 8, the dose of enoxaparin was increased to provide therapeutic anticoagulation, and antibiotic therapy was escalated to include meropenem and vancomycin. The next day, the ratio of SpO2/FiO2 to respiratory rate (ROX) index fell (3.04).
Improvements in oxygenation with incentive spirometry, chest physiotherapy and awake prone positioning were only sustained for a few hours and the patient began to fatigue. Thus, on day 9, when a trial of non-invasive ventilation via a helmet interface failed, invasive mechanical ventilation was initiated. This initially seemed to improve oxygenation, but the FiO2 could not be reduced below 1.0 and sudden, severe deoxygenation still occurred intermittently. Prone positioning allowed some reduction of supplemental oxygen and ventilatory support. However, these improvements in oxygenation were not sustained when the patient was turned supine. So several ‘doses’ of prone positioning were administered.
The RTLIAS via the PFO was thought to be exacerbating the hypoxaemia caused by COVID-19 pneumonia. So, percutaneous closure of the PFO was considered. However, pending the decision, the oxygenation became more stable (table 1; 3 days later SpO2 95%, FiO2 0.4, PEEP 10 cmH20, peak airway pressure 35 cmH20). Although the RTLIAS may have been dynamic, the TOE performed on day 12 demonstrated that it had then resolved. Thus, as the patient’s oxygenation was improving, a multidisciplinary team including an internist, cardiologist, neurologist and intensivist agreed that current guidelines did not support closure of the patient’s PFO.
Outcome and follow-up
The patient gradually improved and ventilatory support was weaned. On day 20, anticoagulation was reduced to a prophylactic dose after the CTPA excluded pulmonary emboli. The patient was extubated 18 days after tracheal intubation (ie, day 27). Although rehabilitation was required, the patient was ultimately discharged home 39 days after his initial presentation.
Discussion
Intrapulmonary shunt and acute RTLIAS are known to contribute to hypoxia in COVID-19.5 6 However, no previous reports have described the use of serial imaging to guide management based on changes in shunt. The present case illustrates that when present, RTLIAS may be dynamic. It can improve as COVID-19 resolves.
In the present case, oxygenation improved with prone positioning but not dorsal recumbency. While diagnostic criteria for platypnoea–orthodeoxia syndrome (POS) were not fulfilled, prone positioning may reduce RTLIAS.10 Yet, although microbubble contrast-enhanced transcranial Doppler ultrasound demonstrated that a large RTLIAS decreased significantly on proning,10 the precise mechanism for this remains uncertain. In the prone position, the dependent heart rests on the sternum. This change in the position of the heart relative to the vena cavae may divert blood flow away from the PFO.10 Improvement in lung recruitment, hypoxia, pulmonary vascular resistance and right heart function on proning11 may also be relevant.
Precise measurement of atrial pressures and the contributions of intrapulmonary shunt (IPS) and extrapulmonary shunt to the total RTL shunt require left and right heart catheterisation. However, this is invasive and impractical for screening. Saline microbubble contrast-enhanced echocardiography can characterise and monitor RTLIAS, while analysis of interatrial septum motion can predict transatrial pressure gradients and PCWPs (figures 3 and 4; videos 1 and 2).12 13
Acute RTLIAS can cause profound hypoxia refractory to oxygen therapy, POS and paradoxical embolism. The pathogenesis of RTLIAS is complex and requires14–16:
An anatomical component (eg, interatrial defect).
A functional component that transiently increases RA pressures or preferentially directs blood flow through the anatomical component.
With a prevalence of 20%–30% in the general population, PFO is the most common interatrial defect.14–16 A patient with an asymptomatic interatrial defect may develop RTLIAS with a secondary cardiac or pulmonary insult. Several complications of COVID-19 can induce or exacerbate pulmonary hypertension and could therefore precipitate RTLIAS. Thus, acute RTLIAS could occur in millions of people with COVID-19 worldwide. However, few reports describe the clinical course of COVID-19 in patients with a PFO.6–8 Indeed, the contribution of PFO, RTLIAS and POS to hypoxia is often unrecognised.9 15 16 In a prospective study of 108 mechanically ventilated patients, the overall prevalence of PFO with acute RTLIAS was 27%,17 but was significantly higher in patients with PEEP over 9 cmH2O (45%) or plateau pressure over 26 cmH2O (46%).17 Patients with RTLIAS are ventilated longer, receive more adjuncts to treat refractory hypoxia and have longer admissions in intensive care units.7–9 16
However, no data from randomised controlled clinical trials are available to guide the management of RTLIAS.7 8 16 Indeed, in some situations, the standard approach to the management of refractory hypoxia, which aims to reduce IPS, can exacerbate RTLIAS and may worsen hypoxia.7 8 16 17 Pragmatic, physiology-guided treatment of hypoxia in patients with RTLIAS should aim to reduce total shunt by balancing the effects of any interventions on both IPS and RTLIAS. Using serial echocardiography to monitor the effect of interventions on RTLIAS could support decision-making in this context as refractory hypoxia with persistent RTLIAS may necessitate closure of the PFO.
Regardless, it is probably best to avoid positive pressure ventilation, initially; if possible, administer oxygen and optimise positioning. If invasive mechanical ventilation is required, supplemental oxygen, plateau pressure and PEEP should be frequently titrated against SpO2, arterial blood gases and haemodynamics so that the lowest airway pressures can be applied. This may be facilitated by measures to improve respiratory system compliance (eg, neuromuscular blockade and prone positioning).10
Fundamentally, definitive treatment of RTLIAS requires closure of the anatomical substrate. This may be achieved surgically or percutaneously.14 15 Treatment of chronic hypoxia and platypnoea–orthodeoxia is a well-recognised indication for PFO closure.14 18 19 When failure to wean invasive ventilatory support is due to RTLIAS, PFO closure may facilitate liberation from mechanical ventilation.20 However, secondary prevention of recurrent stroke after a PFO-related stroke is the only indication, currently supported by high-quality data from randomised trials.21 Further studies are required to define the criteria for closure of PFO in the context of acute respiratory failure.
In select patients, closure of the substrate for RTLIAS could markedly improve hypoxia, reducing the need for invasive ventilation and the incidence of paradoxical embolisation. However, while successful closure of the interatrial defect is often reported,14–16 acute cor pulmonale can develop if pulmonary hypertension is worsened by closing the RTL shunt.14–16 Furthermore, RTLIAS may improve as the functional trigger resolves, so closure is not always required.16
Learning points.
Consider saline microbubble contrast-enhanced echocardiography to screen for interatrial shunt in patients with severe or refractory hypoxaemia.
Early diagnosis of right-to-left interatrial shunt may avoid unnecessary mechanical ventilation.
Avoid interventions that may precipitate or exacerbate right-to-left shunt in patients with an anatomical substrate for interatrial shunt.
Interatrial shunt can be detected, characterised and monitored using saline microbubble contrast-enhanced echocardiography.
Acute right-to-left interatrial shunt may resolve as the precipitant (ie, COVID-19) improves, so closure of a patent foramen ovale is not always required.
Footnotes
Contributors: RR, AH and NM were involved in the management of the case. They were also involved in the conceptualisation, data collection for, and preparation of the manuscript, as well as editing and approval of the final manuscript for publication. GV was involved in conceptualisation and preparation of the manuscript, as well as in editing and approval of the final manuscript for publication.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Obtained.
References
- 1.Gattinoni L, Coppola S, Cressoni M, et al. COVID-19 Does not lead to a "typical" acute respiratory distress syndrome. Am J Respir Crit Care Med 2020;201:1299–300. 10.1164/rccm.202003-0817LE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gattinoni L, Chiumello D, Caironi P, et al. COVID-19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med 2020;46:1099–102. 10.1007/s00134-020-06033-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Borczuk AC, Salvatore SP, Seshan SV, et al. COVID-19 pulmonary pathology: a multi-institutional autopsy cohort from Italy and New York City. Mod Pathol 2020;33:2156–68. 10.1038/s41379-020-00661-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Al-Ani F, Chehade S, Lazo-Langner A. Thrombosis risk associated with COVID-19 infection. A scoping review. Thromb Res 2020;192:152–60. 10.1016/j.thromres.2020.05.039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Masi P, Bagate F, d'Humières T, et al. Is hypoxemia explained by intracardiac or intrapulmonary shunt in COVID-19-related acute respiratory distress syndrome? Ann Intensive Care 2020;10:108. 10.1186/s13613-020-00726-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rajendram R, Hussain A, Mahmood N, et al. Feasibility of using a handheld ultrasound device to detect and characterize shunt and deep vein thrombosis in patients with COVID-19: an observational study. Ultrasound J 2020;12:49. 10.1186/s13089-020-00197-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rajendram R, Kharal GA, Mahmood N, et al. Identifying phenotypes of COVID-19, defining their pathogenesis, and targeting treatments could improve outcomes. Respir Physiol Neurobiol 2020;280:103477. 10.1016/j.resp.2020.103477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rajendram R, Kharal GA, Mahmood N, et al. Rethinking the respiratory paradigm of COVID-19: a 'hole' in the argument. Intensive Care Med 2020;46:1496–7. 10.1007/s00134-020-06102-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mekontso Dessap A, Boissier F, Leon R, et al. Prevalence and prognosis of shunting across patent foramen ovale during acute respiratory distress syndrome. Crit Care Med 2010;38:1786–92. 10.1097/CCM.0b013e3181eaa9c8 [DOI] [PubMed] [Google Scholar]
- 10.Legras A, Dequin PF, Hazouard E, et al. Right-to-left interatrial shunt in ARDS: dramatic improvement in prone position. Intensive Care Med 1999;25:412–4. 10.1007/s001340050868 [DOI] [PubMed] [Google Scholar]
- 11.Jozwiak M, Teboul J-L, Anguel N, et al. Beneficial hemodynamic effects of prone positioning in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med 2013;188:1428–33. 10.1164/rccm.201303-0593OC [DOI] [PubMed] [Google Scholar]
- 12.Ellis K, Kanter IE, King DL, et al. The atrial septal sign: a neglected aid in the interpretation of angiocardiograms. Am J Roentgenol Radium Ther Nucl Med 1970;109:37–50. [PubMed] [Google Scholar]
- 13.Royse CF, Royse AG, Soeding PF, et al. Shape and movement of the interatrial septum predicts change in pulmonary capillary wedge pressure. Ann Thorac Cardiovasc Surg 2001;7:79–83. [PubMed] [Google Scholar]
- 14.Agrawal A, Palkar A, Talwar A. The multiple dimensions of platypnea-orthodeoxia syndrome: a review. Respir Med 2017;129:31–8. 10.1016/j.rmed.2017.05.016 [DOI] [PubMed] [Google Scholar]
- 15.Marples IL, Heap MJ, Suvarna SK, et al. Acute right-to-left inter-atrial shunt; an important cause of profound hypoxia. Br J Anaesth 2000;85:921–5. 10.1093/bja/85.6.921 [DOI] [PubMed] [Google Scholar]
- 16.Gallagher G, Joseph A, Rajendram R. Platypnea-orthodeoxia: patent foramen ovale unmasked by pulmonary emboli. Indian J Respir Care 2018;7:50–2. [Google Scholar]
- 17.Vavlitou A, Minas G, Zannetos S, et al. Hemodynamic and respiratory factors that influence the opening of patent foramen ovale in mechanically ventilated patients. Hippokratia 2016;20:209–13. [PMC free article] [PubMed] [Google Scholar]
- 18.De Cuyper C, Pauwels T, Derom E, et al. Percutaneous closure of PFO in patients with reduced oxygen saturation at rest and during exercise: short- and long-term results. J Interv Cardiol 2020;2020:1–8. 10.1155/2020/9813038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mojadidi MK, Gevorgyan R, Noureddin N, et al. The effect of patent foramen ovale closure in patients with platypnea-orthodeoxia syndrome. Catheter Cardiovasc Interv 2015;86:701–7. 10.1002/ccd.25953 [DOI] [PubMed] [Google Scholar]
- 20.Salazar C, Majano RA. Acute hypoxic and refractory respiratory failure induced by an underlying PFO: an unusual case of Platypnea orthodeoxia and transient complication after transcatheter closure. Case Rep Crit Care 2017;2017:1–4. 10.1155/2017/4397163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Saver JL, Carroll JD, Thaler DE, et al. Long-term outcomes of patent foramen ovale closure or medical therapy after stroke. N Engl J Med 2017;377:1022–32. 10.1056/NEJMoa1610057 [DOI] [PubMed] [Google Scholar]