Abstract
Advancements in both material science and bionanotechnology are transforming the health care sector. To this end, nanoparticles are increasingly used to improve diagnosis, monitoring, and therapy. Huge research is being carried out to improve the design, efficiency, and performance of these nanoparticles. Nanoparticles are also considered as a major area of research and development to meet the essential requirements for use in nanomedicine where safety, compatibility, biodegradability, biodistribution, stability, and effectiveness are requirements towards the desired application. In this regard, lipids have been used in pharmaceuticals and medical formulations for a long time. The present work focuses on the use of lipid nanostructures to combat brain tumors. In addition, this review summarizes the literature pertaining to solid lipid nanoparticles (SLN) and nanostructured lipid carriers (LNC), methods of preparation and characterization, developments achieved to overcome blood brain barrier (BBB), and modifications used to increase their effectiveness.
Keywords: Lipid nanoparticles, Blood brain barrier (BBB), Solid lipid nanoparticles (SLN), Lipid nanocarriers (LNC), Glioblastoma
Lipid nanoparticles; Blood brain barrier (BBB); Solid lipid nanoparticles (SLN); Lipid nanocarriers (LNC); Glioblastoma
1. Introduction
Nanomedicine is the use of nanoparticles and application of nanotechnology in medicine. This implies the use of nanomaterials for the diagnosis, monitoring, control, prevention, and treatment of diseases [1]. It could be considered a huge step in both the advancement and personalization of therapy since it has the potential to overcome numerous problems of conventional medicine such as solubility, targeting, and drug release [2]. Whether it achieved its full potential is still a matter of dispute, but there is no doubt that many advancements have been accomplished [3, 4]. Nanomedicine had a huge effect in numerous diseases such as cardiovascular [5], cancer targeting [6], diagnosis and therapy [7], human immunodeficiency virus (HIV) [8], and Alzheimer's disease [9]. However, cancer has been the most affected by the advancements made in nanomedicine and nanobiotechnology. Along this line, cancer continues to be a global concern, despite the advent of technological and pharmaceutical improvements over the past few decades. Conventional strategies to combat cancer include the use of anticancer drugs (chemotherapy), surgery, and radiotherapy. Both surgery and radiotherapy are most effective in early stage tumors, but are insufficient for stage III and IV tumors, and the use of chemotherapy becomes essential [10]. In this respect, drug delivery (either passive or targeted) is becoming very promising in reducing the side effects and increasing the therapeutic index of chemotherapy.
Brain tumor is the 10th leading cause of death of all cancer patients worldwide, and patients still suffer from the very poor prognosis and therapy. The median survival time for this disease is about 14 months after diagnosis [11]. There are several factors that render brain tumor therapies ineffective; these include the blood brain barrier (BBB), which limits penetration of drugs to brain, and the limitations of surgery in removing tumors due to its location in the central nervous system (CNS) without affecting the quality of life of survivors [12]. For this, nanomedicine can be a game changer. In addition, it has the potential of personalization of therapy and overcoming the BBB to achieve better delivery and distribution of anticancer drugs [13].
Numerous nanoparticles, including liposomes, polymeric nanoparticles, micelles, dendrimers, and gold nanoparticles among others, have been investigated for their drug delivery efficiency as anticancer drugs. In this review, the use of lipid nanostructures for the treatment of brain cancer will be discussed. In this context, lipids are amphiphilic biomolecules that are, generally insoluble in water, and have been used in pharmaceutical formulations in emulsions, lotions, and ointments among others. They are nontoxic, biocompatible, and biodegradable, which are the main requirements for the design of nanoparticles for biomedical applications. In addition, functionalization of these nanoparticles is a relatively easy process, converting them to smart delivery systems [14].
In a similar fashion, solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs) are mainly composed of biocompatible natural substances and/or a combination of natural lipids. These nanoparticles undergo –in general-safe biodegradation, which depends on the lipids used, have minimum influence on the extracellular/intracellular environment due to the similarity between these nanoparticles and cell membrane (both chemically and structurally), and provide good controlled release of numerous encapsulated biologically active compounds for long periods of time [15, 16, 17]. They have a small average size that enables them to simply flow in the blood avoiding macrophages uptake. As other lipid or polymeric nanoparticles, SLNs and NLCs can be modified with several targeting ligands such as peptides, aptamers, antibodies, or even small targeting molecules [18]. These nanoparticles were considered ideal candidates to deliver mRNA for covid-19 vaccines due to the thorough optimization process SLN and NLC went through for delivering nucleic acids [19]. Moreover, SLNs and NLCs are considered the best candidates for targeting CNS diseases due to their high biocompatibility and low immunogenicity [16]. Based on the preceding discussion, and due to the wide range of therapeutic potentials of SLNs and NLCs in cancer treatment, this review highlights the current knowledge dealing with the therapeutic capacity of these particles. For this purpose, we obtained the most recent relevant references pertaining to these topics from different known databases such as, Google Scholar, Science Direct, MEDLINE (PubMed), Scopus, Scientific Information Database (SID), and SciFinder. We hope this work will be a valuable addition to the field and will be a great help for researchers and health professionals.
2. Methodology
To accomplish this task, a literature search in known databases including Science Direct, Google Scholar, MEDLINE (PubMed), Scopus, Scientific Information Database (SID), and SciFinder was conducted. Search was carried out using the keywords “solid lipid nanoparticles” and “lipid nanocarriers” pairing with cancer, brain cancer, therapeutics, and drug delivery system. In our search, we did not impose language restrictions; however, articles with full text in English were included for further analysis. Titles and abstracts of published articles recognized in the search were examined following inclusion and exclusion criteria. Within this context, inclusion criteria involved: (a) In vitro, in vivo or ex vivo experimental and clinical studies on solid lipid nanoparticles and lipid nanocarriers in cancer and brain cancer treatment, (b) Studies related to synthesis and properties of solid lipid nanoparticles and lipid nanocarriers, and (c) Studies with or without proposed mechanism for anticancer use of solid lipid nanoparticles and lipid nanocarriers. On the other hand, exclusion criteria included (a) Duplicated data, titles and/or abstracts that did not meet the inclusion criteria, (b) Unpublished data and overlapped evidence, and (c) The literature focusing on other effects of solid lipid nanoparticles and lipid nanocarriers. A large number of references pertaining to the nanoparticles were found in the aforementioned databases, and the overall findings are discussed below.
3. Solid lipid nanocarriers
3.1. Introduction
Cancer, a global public health concern, is characterized by the abnormal cell growth that can infiltrate other parts of the body and destroy them [20]. Cancer traditional treatments, which involve surgery [21], radiotherapy [22], and chemotherapy [23, 24, 25], are aggressive and have various adverse effects [22, 24, 26]. Moreover, cancer cells may develop resistance against most cytotoxic drugs during or shortly after therapy [27, 28]. Recently, a new approach was applied to support and improve traditional treatments of tumors by utilizing nano-sized-carriers to overcome the side effects [29, 30], enhance the low therapeutic efficacy, and show acceptable controlled release of the drug load [31]. Additionally, fabrication of nanocarriers with targeting moieties is possible, making them selectively bind to specific receptors overexpressed in cancer cells [32, 33, 34]. The composition of these nanomaterials, which are made of either synthetic or natural substances, determines their properties [35]. Nanocarriers are classified into inorganic [36], polymer-based [37], and lipid-based nanocarriers [38]. Use of lipid-based nanocarriers is a promising strategy to manage various types of brain tumors [39, 40, 41]. In this section of the review, various types of nanostructure lipids that generally target brain diseases and are responsible for the delivery of chemotherapeutics to the brain tumors are discussed.
3.2. Types of lipid-based nanocarriers
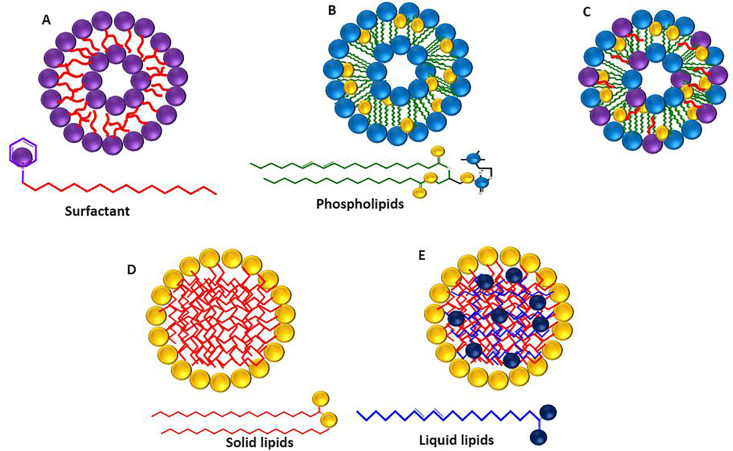
Based on their physical and chemical properties and the process of their production, lipid-based nanocarriers are categorized into niosomes [42], transferosomes, liposomes [43], solid lipid nanoparticles (SLNs), and nanostructured lipid carriers (NLCs) [16] (Figure 1). Lipid nanocarriers possess a brain-targeting ability that improves the drugs selectivity and therapeutic efficacy [44]. Furthermore, these carriers protect sensitive drugs from enzymatic degradation and enhance the bioavailability of drugs by increasing their diffusion through biological membranes [45]. On the other hand, solid lipid nanoparticles and nanostructured lipid carriers are most useful for treating central nervous system (CNS) diseases [46, 47].
Figure 1.
Lipid-based nanocarriers A) Niosomes B) Liposomes C) Transferosomes D) SLNs E) NLCs.
3.2.1. Solid lipid nanoparticles (SLNs)
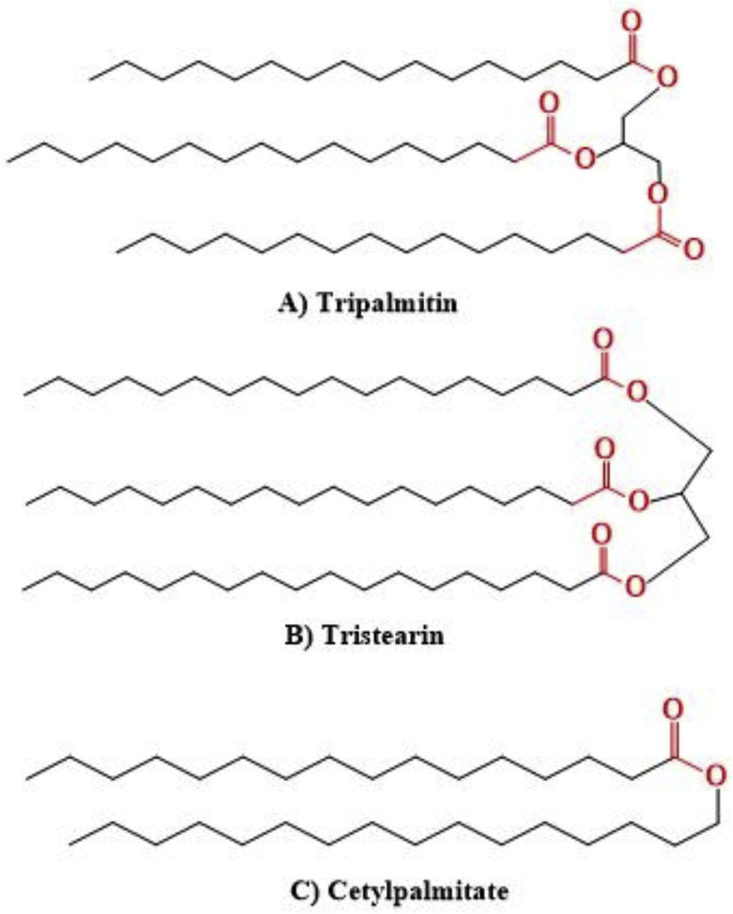
SLNs were first described in 1990 and introduced to overcome several disadvantages of some colloidal carriers including polymeric nanoparticles, liposomes, and nano emulsions [15, 48]. These colloidal nanocarriers suffer setbacks of toxicity, instability, and low loading capacity [49, 50, 51]. SLNs consist of a mixture of one or more solid lipid with bioactive compounds entrapped within the lipid matrix and stabilized by a surfactant or polymer. They have average diameters between 50 and 100 nm, and are solid at both room and body temperatures [52]. Several characteristics make SLNs interesting for drug delivery systems; they are prepared from biocompatible and biodegradable lipids that make them safe and feasible for large-scale production at low cost [53]. SLNs show good physical stability and improved bioavailability of hydrophobic drugs [54]. In addition, they protect the incorporated drug from degradation and enable controlled drug release (sustained or fast), while their preparation does not require toxic organic solvents [54, 55]. Furthermore, grafting specific surfaces with antibody receptors, proteins, and peptides enables them to actively target the infected location [49]. In this respect, three forms of solid lipids are used in the production process, purified triglycerides (e.g., tripalmitin, tristearin (Figure 2a and b)), complex glyceride mixtures (e.g., Witepsol® W35), and waxes (e.g., Cetyl palmitate (Figure 2c)), Carnauba wax, and Beeswax) [15, 56].
Figure 2.
Chemical structures of solid lipids used in SLNs formulations.
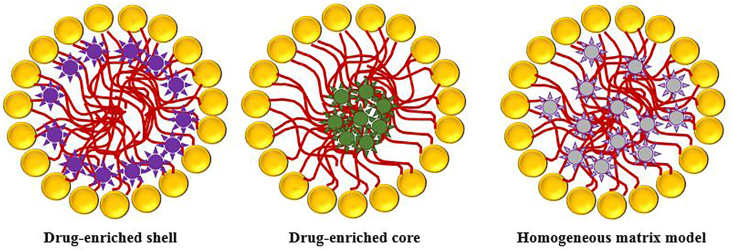
Bioactive molecules are incorporated into SLNs based on three basic models: drug-enriched shell, drug-enriched core, and homogeneous matrix model as shown in Figure 3 [54,57]. SLNs transfer from an α form (α: less ordered polymorph having hexagonal chain packing) and β′ form (β′: an orthorhombic perpendicular chain packing) to β form (β: triclinic parallel chain packing) [58, 59]. This transition to a more ordered form phase is responsible for reducing the loading capacity and enhancing drug expulsion (α < β′ < β) [59]. Therefore, the presence of a high degree of order reduces those defects in the crystal lattice, leading to drug expulsion and decreases the loading capacity [60]. On the other hand, a less ordered solid lipid matrix is necessary to have a higher loading capacity of the incorporated drugs [48]. As a result, the lipid matrix consisting of similar molecules such as tristearin is replaced by more complex lipids (mono, di-, triglycerides, with different chain lengths) to enhance the loading capacity and reduce drug expulsion via increasing imperfections. Besides drug expulsion and limited loading capacity, SLNs suffer from the high water content of dispersion (more than 70%). Hence, nanostructured lipid carriers (NLCs) have been developed to solve the drawbacks associated with SLNs [52, 61].
Figure 3.
Models of bioactive molecules incorporation into SLNs.
3.2.2. Nanostructured lipid carriers (NLCs)
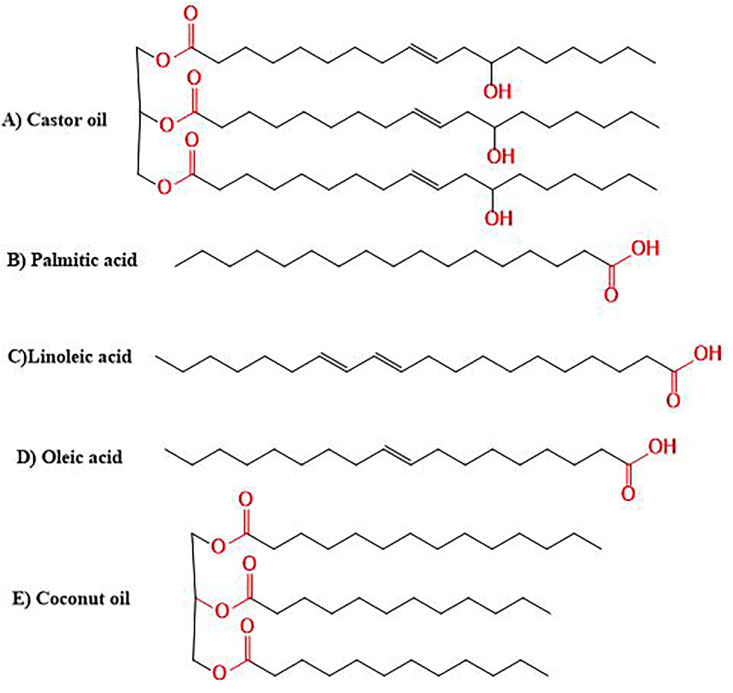
NLCs are the second generation of SLNs and are formulated by mixing liquid lipids with solid lipids [57]. The most commonly used liquid lipids are: castor oil, oleic acid, palmitic acid, olive oil, linoleic acid, and coconut oil (Figure 4). The ratio of liquid lipid to solid lipid ranges from 30: 70% to 0.10: 99.9%, and the particles are stabilized by adding 0.5–5% surfactant solutions [48, 62]. Although NLCs contain liquid lipids, they are still solid at room and body temperatures. Development of these nanosized compartments supports overcoming many of the limitations associated with SLNs [63, 64]. In this regard, NLCs enhance the loading capacity of drugs, control release properties, improve encapsulation efficiency, and extend chemical stability [65]. Furthermore, adding liquid lipids to the lipid matrix prevents drug expulsion, hence increases the drug incorporation capacity compared to SLNs [57]. Additionally, more imperfections are created in the crystal structure to generate more space for drug inclusion [66].
Figure 4.
Chemical structures of liquid lipids used in NLCs formulations.
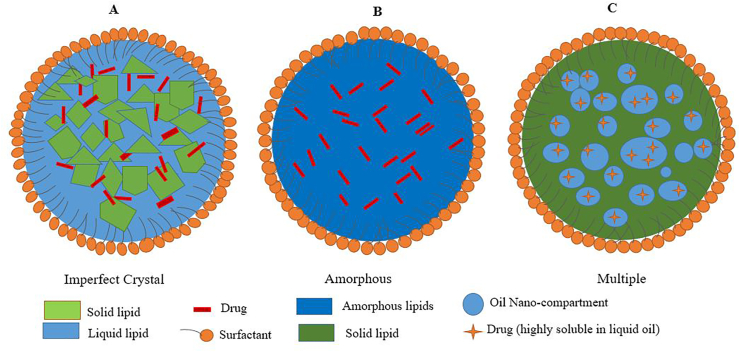
NLCs are convenient for different routes of administration: Ocular [67], topical [68], oral, and parenteral [69]. They are classified into three types [66] as shown in Figure 5: (1) Type I, the imperfect: This type is characterized by imperfection in crystal formation (Figure 5a) where a fraction of the solid lipid is replaced by liquid lipids to increase the ratio of imperfections in the lipid matrix's lattice, leading to more free space for drug accommodation. Therefore, the loading capacity increases, and the incorporated drug will not be expelled outside the core formulation [70]. (2) Type II, the amorphous: In this type, the drug remains embedded in a solid amorphous (formless) matrix (Figure 5b). Liquid lipids such as hydroxyoctacosanyl hydroxystearate and isopropyl myristate are mixed to obtain α-polymorph (less ordered polymorph) instead of the imperfect crystal. In general, amorphous type NLCs are more favored than the imperfect NLCs because they are more stable and have a higher loading capacity [71]. (3) Type III, multiple oil-in-fat-in-water (O/F/W): This type is derived from water-in-oil-in-water (w/o/w) emulsions and compromises an oil-in-fat-in-water dispersion (Figure 5c). This type is usually prepared by mixing solid lipids with large amounts of liquid lipids using hot homogenization technique [48]. Then, oily nano-compartments appear after cooling due to phase separation of the excess liquid lipid. This type of NLCs is so attractive, specifically for drugs with higher solubility in liquid lipids than in solid lipids, and gives high loading capacity [72]. Additionally, the external lipid matrix surrounding the nano-compartments performs as a barrier that prevents drug leakage and allows the controlled release [38].
Figure 5.
Different polymorphic types of NLCs A) Imperfect B)Amorphous C) Multiple.
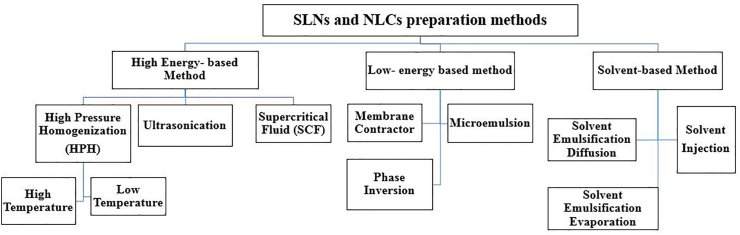
3.3. Formulation of SLNs and NLCs
Preparation methods of NLCs and SLNs can be divided into three types. High energy based-methods, low energy-based methods, and organic solvent-based methods as shown in Figure 6 [73].
Figure 6.
General classification of SLNs and NLCs preparation methods.
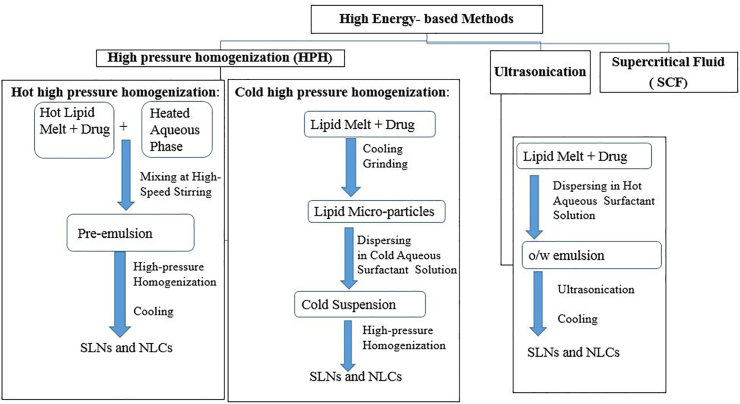
3.3.1. High energy-based methods
High energy-based methods involve high-pressure homogenization, ultrasound, and supercritical fluid (SCF) as depicted in Figure 7 [74]. The high-pressure homogenization technique, developed by Muller and Lucks [75], depends on reducing the size of particles through high-pressure. This approach can be performed at high temperature, called hot high-pressure homogenization, or at low temperature, called cold high-pressure homogenization [76]. In this method, the bioactive compound is dissolved, dispersed, or solubilized in the lipid that is melted at approximately 5–10 °C above its melting point. In a high-pressure process, the drug-containing lipid melt is solubilized under stirring in a hot aqueous surfactant solution of the exact temperature of lipid melt by high-speed stirring [77]. The obtained pre-emulsion is passed through a homogenizer and cooled to room temperature, the standard production conditions are 500 bar and two or three homogenization cycles [78]. In the cold homogenization technique, the active compound-containing melt lipid is cooled and solidified. After that, the lipid matrix is quickly grounded under liquid nitrogen to produce lipid microparticles. The lipid microparticles are then dispersed in a cold surfactant solution by stirring to obtain a micro-suspension. This micro-suspension is passed through a high-pressure homogenizer at low temperature or at room temperature to break down to the solid lipid nanoparticles. The standard production conditions are 500 bar and five homogenization cycles [79]. The produced particles by a cold process have larger diameters and higher polydispersity indices. The cold homogenization technique is recommended to encapsulate hydrophilic and extreme temperature-sensitive compounds to protect its partition from the liquid lipid phase to the water phase. On the other hand, the high-pressure homogenization is the most preferred preparation method because of its simplicity and low cost of production [79].
Figure 7.
Classification of high energy-based methods.
The ultrasound method contains an initial stage that is similar to high-pressure homogenization where the active compound is dissolved, dispersed, or solubilized in the lipid melted 5–10 °C above its melting point [57]. The aqueous surfactant solution, preheated at the same temperature, is added to the drug-lipid melt and dispersed by a high shear mixer to obtain coarsely hot oil in-water emulsion. The emulsion is ultrasonicated by a probe sonicator until the desired size is reached, and then allowed to reach room temperature. This preparation method could be a health risk because the formulations are susceptible to metallic contamination from the probe [80, 81]. On the other hand, the supercritical fluid (SCF) combines the best properties between liquids and gases such as high density, high solvation power, high diffusivity, and viscosity. These properties make SCF, especially supercritical carbon dioxide (SCF–CO2), feasible for formulating SLNs [82]. In this respect, there are three types of SCF technologies used in SLNs formulations. These include supercritical fluid extraction of emulsion solvent (SCEE) [83], particles from gas saturated solution (PGSS) [84], and the gas-assisted melting atomization (GAMA) [85].
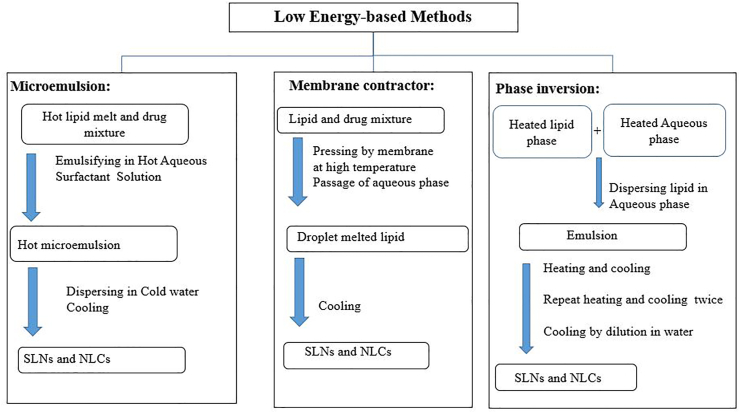
3.3.2. Low energy-based methods
Low energy based methods include microemulsion [81], membrane contractor [86], and phase inversion as described in Figure 8 [87]. In the microemulsion method, a melted lipid and a preheated bioactive molecule dissolved in oil are mixed in an optimized ratio. Then the mixture is added to an aqueous surfactant solution at the same temperature to obtain a transparent dispersion subjected to mechanical stirring at the same temperature to allow the formation of the microemulsion [88]. This warm microemulsion is added to cold water (2–10 °C) under mild stirring, causing the lipid phase to precipitate as fine particles. This method is simple, economical, and flexible to formulate NLCs incorporated with polar and/or non-polar drugs [89]. The main drawback of the microemulsion method is the need to use a large amount of surfactant. In addition, the formulation needs to be concentrated by lyophilization or ultrafiltration [90].
Figure 8.
Classification of low energy-based methods.
In the membrane contractor method, a melted lipid matrix is pressed through membrane pores. The aqueous phase spreads inside the membrane module and sweeps away the droplets forming at the pore outlets, and the formulation is then cooled to room temperature [91]. Advantages of this technique are high scale production and simplicity. Moreover, particle sizes are easily adjusted by manipulating various parameters such as lipid phase pressure, cross-flow velocity, membrane pore size, and aqueous phase and lipid phase temperatures. However, membrane clogging is the major drawback of the low energy approach [86].
In the phase inversion method, a phase inversion occurs due to temperature variation. Typically, three heating and cooling cycles (85–60–85 °C) are involved in achieving a consecutive phase reversal from an O/W emulsion to a W/O emulsion until finally reaching an O/W emulsion [92]. In each inversion step, sizes of drops are reduced, thus permitting the collection of nanoparticles (SLNs or NLCs) in the last step by cold water. The phase inversion process has received considerable attention due to achieving the particles' reduced size without the need to use organic solvents or consume a large amount of energy, as in the case of the ultrasound method and high-pressure homogenization [87].
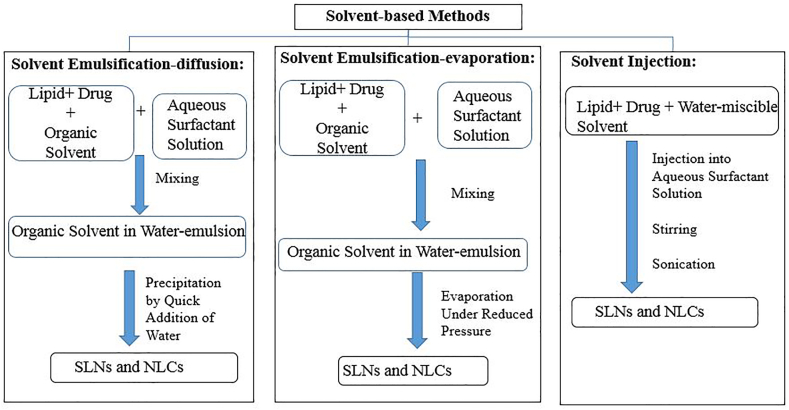
3.3.3. Organic solvent based methods
The organic solvent based methods include solvent emulsification-evaporation, solvent emulsification-diffusion [93], and solvent injection as shown in Figure 9 [94]. In the solvent emulsification-evaporation method, a bioactive molecule and lipid mixture are dissolved in a water-immiscible organic solvent such as chloroform and cyclohexane. The prepared solution is emulsified in an aqueous phase by a high shear homogenizer or ultrasonic probe followed by organic solvent evaporation under reduced pressure [95]. In the solvent emulsification-diffusion, water-immiscible solvents are replaced by partially water-miscible solvents. A partially water-miscible solvent (e.g., ethyl formate and benzyl alcohol) is previously saturated with water to ensure initial thermodynamic equilibrium with water [96]. The lipid is dissolved in the water-saturated solvent, emulsified with solvent-saturated aqueous surfactant solution at elevated temperature. The dispersed phase is diluted by excess water under continuous stirring to produce solid nanoparticles [97]. On the other hand, the solvent injection method relies on dissolving the lipid in a water-miscible solvent such as isopropanol, ethanol, and acetone. The formed solution is injected into an aqueous phase under stirring with the aid of a syringe needle. Then upon contact with an aqueous phase, the lipid precipitates into nanoparticles. The main advantages of the solvent injection method are avoidance of heating and low energy input [97, 98].
Figure 9.
Organic solvent-based methods.
3.4. Characterization of SLNs and NLCs
3.4.1. Particle size
Determination of the mean particle size and polydispersity index (PDI) is an exceptionally vital part of assessing solubility, physical stability, biological performance, and release rate. In general, diameters of lipid-based nanocarriers extend from 10 nm to 1000 nm [99]. Only, nanocarriers within the range of 50–300 nm should be used with chemotherapeutic agents and the central nervous system. This range permits crossing of the blood-brain barrier (BBB) and provides enhanced uptake in cells [100]. Diameters of lipid-based nanocarriers are controlled by adjusting numerous factors such as properties of lipids, types of surfactants, manufacturing process, and the processing temperature. In this respect, adding a high surfactant/lipid ratio contributes to reducing the sizes, whereas adding low surfactant concentrations causes growth in the diameters [101, 102, 103].
Numerous instruments are designed to measure diameters of lipid-based nanocarriers in a submicron scale, with high accuracy and repeatability. These instruments are mainly non-destructive technique based on the interaction between a laser beam and a dispersion, and the indirect calculation of the particle size from their light scattering effect. Dynamic light scattering (DLS), also known as photon correlation spectroscopy (PCS), measures the variance of the intensity of the scattered light caused by particle movement (diffusion) and is useful to measure diameters ranging from a few nanometers to 3 μm. However, laser diffraction (LD), also known as static light scattering, is recommended to measure particles with diameters greater than 3 μm in the SLN and NLC formulations [104, 105]. LD is related to the diffraction angle on the particle radius (Fraunhofer spectra); smaller particles cause intense scattering at high angles compared with the larger ones [81]. It is always preferable to simultaneously utilize both PCS and LD methods if the particles are not spherical [106].
Poly dispersity index (PDI) indicates the size distribution of all nanoparticles including SLNs and NLCs. PCS usually measures PDI by analyzing the scattered light intensity as a function of the angle between the incident and scattered beams depending on a suitable light scattering theory. Formulations with PDI values within the range 0.0–0.5 are described as homogeneous and monodispersed (narrow size distribution). However, those with PDI values more than 0.5 are described as heterogeneous and polydispersed (broad distribution). In most cases, PDI values less than 0.3 are considered the optimum for SLNs and NLCs [107].
3.4.2. Surface charge
Zeta potential (ζ-potential) is a measure of the effective electric charge on the nanoparticle's surface. The magnitude of the zeta potential provides information about particle stability. Nanoparticles with high zeta potential values exhibit high stability because of a larger electrostatic repulsion between particles surfaces [108]. Zeta potential depends on various factors such as the nature of particles surfaces, the particles environment such as ionic strength, pH, and the nature and concentration of the electrolyte present in the solution [45, 109]. Zeta potential values greater than ±30 mV show great repulsion between charged particles, which guarantees minimum aggregation or flocculation; the stabilization is electrostatic. Sometimes, steric stabilizers are combined with electrostatic stabilizer to obtain steric and electrostatic stabilization. In such cases, zeta potential with minimum values of ±20 mV is acceptable and sufficient [110]. The positively charged particles are helpful in many cases because they tend to stick to biological surfaces (since these are negatively-charged surfaces). Analytical instruments used to determine the zeta potential depend on electrophoretic/electroacoustic mobility. For many commercial pieces of equipment, zeta potential and DLS are coupled in a single piece of equipment. Therefore, the particle size and particle potential can be determined by the same instrument [110].
3.4.3. Morphology and shape
Both PCS and LD calculate particle size of the nanoparticles from light scattering. Advanced microscopy techniques are usually used to gain more detailed information regarding the structure, size, and surface morphology of the nanoparticles [45]. Advanced microscopy techniques include scanning electron microscopy (SEM), transmission electron microscopy (TEM), and atomic force microscopy (AFM). These techniques help determining the particle size, surface topography, morphology, state of aggregation, and even the internal structure of the lipid-based nanocarriers [111, 112]. SEM provides details about the surface characteristics and the three-dimensional morphology of the particles whereas AFM helps in analyzing the three-dimensional morphology of the particles. Since lipid-based nanocarriers consist of chemical elements with few atomic numbers, it requires per-treatment of the samples to increase the contrast before measurements using high vacuum. In this respect, SEM requires drying of samples (mostly by freeze-drying) and coating them with a thin metallic layer (e. g: gold, gold/palladium) to permit conduction of electrons [113, 114].
TEM provides two-dimensional images of the internal structure of the lipid-based nanocarriers. In general, samples are negatively stained with uranyl acetate or phosphotungstic acid to contrast the particle [114]. By comparison, TEM is more beneficial than SEM due to its higher resolution power (0.4 nm for TEM and 3–4 nm for SEM) and the absence of exposure to the vacuum, which protects particles from structural change [114]. On the other hand, AFM technique depends on the force occurring between a surface and a probing tip resulting in a spatial resolution of up to 0.01 nm for imaging. AFM is characterized by the simplicity of sample preparation, performing analysis without vacuum, and samples do not require drying or coating. Therefore, nanocarriers are measured in the hydrated form to prevent any morphological distortions [115].
3.4.4. Crystallinity and lipid modification
In NLCs and SLNs formulation, the crystal structure and the polymorphic forms of lipid components significantly affect the efficiency of encapsulation and release profile of bioactive compounds [116]. In principle, as the thermodynamic stability and lipid packing decrease, the bioactive encapsulation increases according to the following sequence: β-modification (a perfect crystal of triclinic parallel packing with few imperfections) < β′-modification (orthorhombic perpendicular) < α-modification (hexagonal packing) < supercooled melt. In this regards, crystallization and modification changes can be delayed due to the presences of emulsifier and the small size of particles. Differential scanning calorimetry (DSC) and X-ray diffraction are frequently used to investigate the crystalline patterns and polymorphic behavior of the constituents of NLCs and SLNs formulations [52, 116]. DSC is a thermo-analytical technique that provides useful information about lipid melting and crystallization behavior. DSC measures various thermal transitions associated with physicochemical transformation within nanoparticles when heated or cooled in a controlled manner. Many details about polymorphism, crystal structure, eutectic systems, and glass transition temperatures can also be obtained [117, 118, 119]. On the other hand, X-ray diffractions can identify polymorphism status of nanoparticles including NLCs and SLNs. Wide angle diffraction may elucidate the crystalline order of NLCs [115]. Moreover, X-ray scattering enables determining the length of the long and short spacing of the lipid lattice [120].
3.4.5. Drug loading (DL) and encapsulation efficiency (EE)
Drug loading (DL) of SLNs or NLCs is expressed as the ratio of an encapsulated bioactive compound to the quantity of lipid phase or lipid nanoparticles in SLN and NLC formulation multiplied by 100 (equation 1); high DL is helpful in minimizing the cost of production [121]. Similarly, encapsulation efficiency (EE) is defined as the ratio of an encapsulated bioactive compound in nanoparticles to the original amount of bioactive compound incorporated into the lipid phase of NLCs and SLNs formulation multiplied by 100 (equation 2) [122]. After quantification of the encapsulated portion or free portion of the bioactive compound, the EE can be measured. The free portion of the bioactive compound may dissolve in an aqueous solution or crystalizes as a precipitate in micron size. Then, the free portion of the bioactive compound is collected from the aqueous solution by extraction with an organic solvent, or separated from nanoparticles by microfiltration, ultrafiltration, (ultra-) centrifugation, and size exclusion chromatography, and quantified by sensitive techniques such as HPLC and UV spectroscopy [123, 124, 125].
| (1) |
| (2) |
Similarly, magnetic resonance techniques such as nuclear magnetic resonance (NMR) and electron spin resonance (ESR) are also helpful in the study of dynamic phenomena in SLNs and NLCs formulations. NMR is based on various nuclei relaxation times (which depends on the local magnetic field experienced by the nucleus under study) in the liquid and semisolid/solid-state [79]. NMR can be used to determine both the size and the qualitative nature of nanoparticles. NMR chemical shifts provide information on the physicochemical status of components within the nanoparticle. Moreover, NMR can detect supercooled melts in lipids and investigate the reduced mobility of molecules due to the lipid protons' broad and weak amplitude signal [126, 127]. In the ESR characterization, a paramagnetic probe is required to evaluate NLCs and SLNs formulation. Other techniques such as infrared and Raman spectroscopy can also be used to investigate structural features of NLCs and SLNs formulations [63].
4. Brain tumors and the role of blood brain barrier (BBB)
Brain tumors result from the unusual growth of cells inside the brain. Brain tumors can be either cancerous and non-cancerous [128]. Cancerous tumors are, in turn categorized as primary and secondary. Primary tumors begin inside the brain whereas metastatic (secondary) tumors extend from tumors found outside the brain [129, 130]. The major types of primary tumors are glioblastoma (glioma) which is the most aggressive kind of cancer originated inside the brain. On the other hand, meningioma and lymphoma are non-cancerous primary tumors [128]. Glioma is usually characterized according to its origin, astrocytes or oligodendrocytes or from both former origins. Glioma was graded from 2 to 4 according to its aggressiveness [131]. Common treatments of brain tumors include surgery, chemotherapy, and radiotherapy [132]. These traditional treatments are life-threatening and cannot completely cure some invasive types of cancerous tumors such as glioma [133]. The major challenge in brain cancer treatments, and even any CNS diseases, is the blood brain barrier (BBB). This barrier limits the delivery of therapeutic drugs into CNS. In order for drugs to be delivered, they must be functionalized with special targeting moieties or/and they should be carried by an appropriate delivery system [134]. Within this contest, numerous continuously improved nanostructures carriers such as lipid nanostructures are able to penetrate the BBB and deliver the drug into the targeted affected area in the brain [40].
4.1. Blood brain barrier (BBB)
Blood brain pressure is a term used to describe the distinctive properties of the microvasculature of the CNS. CNS blood vessels tightly regulate the movement of ions, molecules, and cells between the blood and the brain [135]. BBB contains three barriers sequentially arranged from the blood to the brain: the glycocalyx, endothelium, and extravascular compartment [136]. Specialized proteins such as cadherins, occludin, and claudin, hold together the endothelial cells to create tight junction (TJs) regions where adjacent endothelial cells are physically kept together. This makes it incredibly difficult for passive transcellular transport of small hydrophilic molecules to permeate the necessary nutrients into the brain while serving as a barrier to the passage of undesired substances from plasma [137].
Most large molecules and more than 98% of small biomolecules that target CNS are unable to cross BBB, and are thus phased out and cleared in the initial screening [138]. In order to successfully cross the BBB, drugs should have a low molecular weight (400 Da) with high lipid solubility [139]. Different approaches were used to enable drugs transport across the BBB. For example, lipid-soluble molecules as anesthetics and heroin can diffuse passively into the brain [140], whereas polar molecules may be transported across the brain endothelial cell membrane by means of facilitated transporters or secondary active transporters as L-DOPA drugs [141, 142, 143]. Efflux transporters represent a significant transport mechanism at the BBB. For example, nitrogen and oxygen-rich lipid-soluble molecules use this mechanism to cross the BBB [144]. Receptor-mediated transcytosis (RMT) is a method that utilizes the vesicular transport system of the endothelial cells to take substrates across the brain side of the barrier [145].
4.2. Nanoparticles and the BBB
Biocompatible nanoparticles are promising drug carriers to fit the BBB transport and cause drug release in the brain. This delivery system uses a wide variety of nanoscale drug delivery platforms such as lipid- and polymer-based nanoparticles (NPs) that guarantee a controlled and improved release of their load into site of action [146]. Main recent efforts focused on increasing the ability of NPs to effectively target the therapeutic site, thus minimizing the doses of drugs released at undesired sites. In this regard, multiple pathways were involved in the transport of brain drugs into the BBB through surfactant-coated nanoparticles (NPs) [147]. SLNs and NLCs have unique advantages as drug carriers. However, the mechanism behind translocating these lipid nanoparticles into the brain remains unclear. They may follow a simple passive diffusion or open the tight junctions between endothelial cells in the brain microvasculature and create a paracellular pathway for their translocation [148].
4.3. Brain tumors
Brain tumors include a wide range of neoplasms that can be primary or metastatic. The primary brain tumors are known as gliomas. The metastatic ones arise from nearby malignancies and then develop within the brain [130, 149]. In this respect, the World Health Organization (WHO recognizes two major types of brain tumors), gliomas and nongliomas:
4.3.1. Gliomas
Glioma is the most common CNS neoplasm that originates from glial cells. Gliomas are very diffusely infiltrative tumors that affect the surrounding brain tissue [150]. Gliomas are classified into astrocytoma, oligodendroglioma, and ependymoma [151]. Astrocytoma is a tumor that originates in astrocytes and in the cerebrum and the cerebellum. Astrocytoma makes up around 50% of all critical brain tumors [152]. Oligodendroglioma is a tumor that develops in oligodendrocytes and makes up approximately 4% of primary brain tumors [153]. Ependymoma is a tumor that affects ependymal cells [154]. On the other hand, glioblastoma is the most malignant astrocytomas type while pilocytic astrocytoma is the least malignant brain tumor [150]. Research findings indicated that this type of brain cancer may resist treatment because it contains stem cells that are responsible for driving the formation of blood vessels (angiogenesis), spread of the tumor (metastasis), and resistance to treatments [155].
4.3.2. Non-gliomas
Non-gliomas are tumors that do not originate from glial cells [131]. The most common examples of non-gliomas include meningiomas and medulloblastomas. Meningioma tumors are regularly formed from arachnoid cells. These cells are responsible for absorption of the cerebrospinal fluid (CSF). Meningioma is responsible for 13–30% of all tumors growing inside the skull [156]. Medulloblastoma is the most common type of cancerous brain tumor in children. This tumor occurs exclusively in the posterior fossa and has the potential for leptomeningeal spread [157]. Brain cancer treatment is still one of the major challenges in oncology. The most commonly used therapies for brain tumors include the use of surgical approaches and/or radiotherapy, which not only pose a grave danger to the patient, but are also unable to treat more effectively aggressive glioma type [158, 159].
4.4. CNS drug delivery
Chemotherapeutics that targets brain tumors are still insufficient in achieving the desired effects in brain cancer treatment. Most drugs have low aqueous solubility, are unable to cross the BBB, and have low potency in the CNS, leading to high toxicity and major side effects [160]. Therefore, development of drug delivery models could be promising for brain cancer treatment. Within this context, SLNs and NLCs have been utilized for the treatment of various diseases affecting CNS and have become the major drug nanocarriers for brain tumor managements [44]. SLNs have succeeded in overcoming numerous limitations reported with other conventional polymeric or lipid nanocarriers such as high toxicity and insufficient stability. Furthermore, an improved encapsulation efficiency could be obtained during the nanoparticle mixing, recrystallization and preparation steps [161]. Adding liquid lipids to the SLN formula leads to the formation of NLCs, which have an imperfect crystal structure with more internal space that provides higher loading capacity. Moreover, SLNs and NLCs functionalization with a wide range of ligands enables them to deliver treatments specifically to the target tissue [15].
4.5. Blood–brain–tumor barrier (BBTB)
Brain tumors reduce the stiffness and selectivity of the BBB giving a new vasculature form, known as the blood-brain-tumor barrier (BBTB). BBTB is more leaky with a high discharge of blood products due to the unorganized and heterogeneous structure of BBTB based on the enhanced permeability and retention (EPR) effect of most cancerous tissues [162]. In BBTB, a new shaped capillaries are structurally produced and are more permeable than even normal peripheral capillaries [162]. BBTB contributes to the delivery of nutrients and oxygen to the tumor, and enables glioma cell migration to other parts of the brain. Although BBTB is considered leaky in glioblastomas but its more closely similar to the normal BBB, and prevents efficient passage of cancer therapeutics including small molecules and antibodies [163]. Both BBB and BBTB barriers constitute a major challenge in brain tumor management by limiting the delivery of enough quantities of potentially effective anticancer agents [164]. Transport of anticancer drugs by nanocarriers may be achieved by taking advantage of this leaky BBTB associated with human brain cancers by the selective passive diffusion of large nanoparticles compared to free drugs [165].
5. Lipid nanostructures for brain cancer targeting
Published research showed that drug nanocarriers and their surface modification exhibit higher bio-distribution and improved bioavailability of drugs with a positive effect on BBB uptake [79]. Since SLNs and NLCs are mainly composed of biocompatible natural substances and/or a combination of natural lipids, they undergo safer biodegradation with limited effects on the extracellular/intracellular environment, and provide good controlled release of numerous encapsulated biologically active compounds for extended periods of time [158]. At the same time they protect loaded drugs from degradation due to their higher stability than other lipid-based nanoparticles [15, 16, 17, 166]. They additionally have a small average size that enables them to simply flow in the blood avoiding uptake of macrophages. As other lipid or polymeric nanoparticles, SLNs and NLCs can be modified with several targeting ligands such as peptides, aptamers, antibodies, or even small targeting molecules. Moreover, SLNs and NLCs are considered the best candidates for targeting CNS diseases due to their high biocompatibility and low immunogenicity [167, 168].
5.1. SLNs and NLCs targeting brain
SLNs and NLCs generate smart drug carrier systems. They are composed of a solid lipophilic core encapsulating the hydrophobic drug in a stable lipid matrix. Their small size (10–200 nm) allows them to cross the endothelial cells of the BBB and penetrate through the endocytosis mechanism [169, 170]. Passive targeting of CNS is based on non-modified surface nanoparticles that increase the drug concentration gradient across the BBB, whereas active targeting of the CNS is based on functionalization of the nanocarriers' surface with targeting ligands [171, 172]. In this respect, the enhanced solubility of the hydrophobic drug in the biocompatible liquid lipids is a key factor for NLCs development and has superior formulation properties over SLNs [79]. Both NLCs and SLNs are stable at room temperature. However, NLCs were more potent in achieving the preferred drug release, and showed enhanced stability compared to SLNs [58].
SLN and NLCs have been investigated for brain targeting of water sparingly soluble and lipophilic drugs, without the need for organic solvents. In this case, numerous drugs that target neurodegenerative diseases belong to class II (decreased water solubility and high permeability) drugs [172, 173]. For example, NLCs were used to enhance the bioavailability of curcumin (CRM) and increase its localization in the brain cells to cure brain tumors. For this, CRM was encapsulated into NLC and into NLC coated with polysorbate 80 to examine the effect of the surfactant on the brain targeting ability of NLC taken by intravenous injection. Both in vivo and ex vivo studies confirmed a more brain deposition of surfactant-coated NLC with respect to bare NLC [112]. Moreover, CRM-loaded NLC showed an enhanced BBB penetration of CRM with increased accumulation in CNS of mice compared to free CRM [174, 175]. Similarly, baicalin-loaded NLC also exhibited a higher baicalin accumulation in the brain compared to baicalin dispersions [176].
A brain targeting nanosystem based on OX-26 antibody attachment on PEGylated cationic SLNs loaded with baicalin was prepared, and results showed a baicalin entrapment efficiency of 83.03 ± 0.01%. In a similar fashion, OX-26 conjugation on PEGylated SLNs enhanced the uptake of baicalin through the BBB by eleven folds on the brain of male Wistar rats compared to free baicalin [177]. Recently, pralidoxime chloride loaded SLNs were formulated to investigate their ability to target and cross BBB. In vivo studies showed a high BBB penetration with the benefit of moderating toxicity side effects of free paraoxon [178]. Similarly, in vivo studies indicated that olanzapine-loaded SLN may significantly increase drug bioavailability up to 23-fold over the pure drug suspension, due to the high ability of SLN to cross the BBB [170]. Furthermore, NLC greatly improved apomorphine brain targeting as compared to an aqueous solution of the drug [175]. Vyas and coworkers investigated the transport of efavirenz (EFV, a non-nucleoside reverse transcriptase 8 inhibitor) via SLN across BBB using phenylalanine (PA) as a targeting amino acid. A controlled release profile for more than 24 h was observed in an in vitro study. Moreover, transcytosis studies verified the ability of PA-SLN to cross BBB 10-fold higher compared to EFV [179].
Lazaroid (LAZ), a potent inhibitor of lipid peroxidation, which exhibits an in vitro anti-proliferative activity, was loaded into NLC (LAZ-NLCs) to enhance LAZ brain exposure. The optimal LAZ-NLCs have a size of 172.3 ± 3.54 nm, a charge of −4.54 ± 0.87 mV, and an entrapment efficiency of 85.01 ± 2.60%. Results indicated that the LAZ exposure in brain was doubled with a decreased lipid content by half for the NLCs group compared to the free LAZ [180]. NLCs containing idebenone (IDE-NLCs), an antioxidant and essential component of the electron transport chain, were developed for brain delivery. IDE-NLCs reached 90.68 ± 2.90% encapsulation efficiency with almost three-fold enhancement in brain concentration compared to the drug in aqueous dispersions [181]. In a recent study, temazepam (TZP), a gamma-aminobutyric acid (GABA) modulator, was loaded into NLCs to enhance the bioavailability and brain targeting for insomnia treatment; a 75.2 ± 0.1% entrapment efficiency was achieved with a sustained drug release profile. Besides, the NLC formulation demonstrated a distinctive targeting affinity to rat brain over free TZP [182].
5.2. SLNs and NLCs targeting brain tumors
SLNs and NLCs have been used for the treatment of several brain diseases but mostly for brain cancer due to their attractive properties. Various studies introduced SLNs and NLCs as efficient nanocarriers with extraordinary properties to enhance the therapeutic efficacy of anticancer agents used to treat brain tumors. Moreover, SLNs and NLCs help anticancer agents to penetrate easily through the BBB and show a high accumulation in brain tumors [169]. SLNs and NLCs are favorable lipid nanocarriers that might effectively target a brain tumor [183]. For example, cationic solid lipid nanoparticles (CA-SLNs) have been synthesized to encapsulate carmustine (CRN), and further decorated with an anti-EGFR receptor for targeting malignant glioblastoma cells. In this regard, the number of cationic surfactants used affect CRN entrapment efficiency. Results showed efficient delivery and anti-proliferative effects against U87 cell line. Moreover, an enhanced cell viability with low expression of tumor necrosis factor (TNF-α) was observed when tested on human brain microvascular endothelial cells [184].
Kuo and coworkers investigated the effect of several types of solid lipids to encapsulate a variety of antitumor agents including doxorubicin (DOX), tamoxifen (TAM), and etoposide (ETP) functionalized with different ligands to target brain tumors using HBMEC, human U87 malignant glioma, and human astrocytes cell lines. Based on the type of lipids used, drug encapsulation efficiency varied from 17 to 95% with size ranging from 80 to 280 nm, and the surface charge was either negative or positive depending on the targeting ligand. In addition, results demonstrated that all types of tested SLNs were safe, had anti-proliferative effects, and could penetrate the BBB [85, 96, 99, 184, 185, 186, 187, 188, 189]. Furthermore, in vitro and in vivo studies on C6 glioma cell line and C6 glioma xenograft tumor, respectively, were conducted using the anticancer drug edelfosine (EDF) encapsulated in SLNs composed from Compritol® or Precirol®. Results showed that these SLNs accumulate in the brain tissue resulting in an anti-proliferative effect and a major decline in the tumor growth [190].
Cytarabine (CRB), a hydrophilic drug used to target meningeal leukemia, was encapsulated in SLNs, and offered a size of 90 nm with 50% entrapment efficiency. CRB-SLNs showed in vitro sustained release over 72 h and a higher dose-dependent cytotoxicity compared to free drug against EL-4 cell line [191]. Similarly, small interfering RNAs (siRNAs) were incorporated into SLNs, to deliver them and to enhance their therapeutic index. These SLNs were made from low-density lipoprotein (LDL) conjugated with a PEGylated c-tyrosine-protein kinase (c-Met). Results indicated a decrease in cell proliferation, down-regulation of the expression levels of c-Met, and a tumor suppression when these SLNs were tested in vitro and in vivo on U87MG cell line and U87MG xenograft tumor. Results showed the feasibility of using siRNA-PEG/SLN complexes as a potential carrier of therapeutic siRNAs for the treatment of glioblastoma in the clinic [192]. Studies dealing with the effectiveness of NLC loaded with ferulic acid against U87MG glioblastoma cancer cell lines, and with loaded NLCs decorated with didodecyldimethylammonium bromide to compare the anticancer effectiveness with uncoated NLC were conducted. Results showed the effectiveness of the NLC compared to uncoated free drug [193].
Rhodamine 123 (R123), a florescent substance, was utilized to evaluate the cellular uptake of cetyl palmitate (CP) SLNs in various gliomas (U373, U251, U87 and A172) cell lines compared to macrophages (THP1) cell line. Experiments results showed a higher uptake of SLNs of 200 nm and –20 mV surface charge into gliomas compared to THP1 cell line [194]. The same SLN encapsulated with camptothecin (CPT) indicated similar cellular uptake results when tested on porcine brain capillary endothelial cells (BCEC) compared to macrophages (RAW-264.7). Furthermore, CPT-loaded SLNs demonstrated a decrease in BCEC cell viability compared to free CPT [195] Similarly, a modified CP SLNs with 1,2-dimyristoyl-sn-glycero-3-phosphocholine (DMPC) were prepared to improve the in vitro release of CPT. This formula led to a higher and a prolonged concentration of the loaded CPT in the brain over the free drug [210]. Moreover, CP-based NLCs were formulated with different fatty amines to encapsulate etoposide, and modified with an anti-transferrin receptor (anti-Trf) to target myelogenous leukemia. The etoposide-loaded NLCs conjugated with Trf exhibited higher cellular uptake and enhanced anti-proliferative effect against K562 cell line compared to the unconjugated formula [196].
Interestingly, new formulas of lipid nanocapsules (LNC) were prepared and functionalized with L1 peptide to load locked nucleic acid (LNA) for silencing the oncogenic miR21. LNC showed a reduction of miR21 expression against U87 malignant glioma (U87-MG) cell line [197].
LNCs again confirmed their ability to deliver drugs across the BBB and enhance their sustained release. For example, CRM-loaded LNCs (CRM-LNC) exerted higher cytotoxicity when tested against U251MG cell line. Besides, in vivo studies showed a reduction in tumor size and malignancy with prolonged animal survival in rats bearing C6 gliomas compared to free CRM [198]. A 90% loading efficiency of CRM was achieved in NLCs (CRM-NLCs) with a size of 150 nm and –20 mV. Furthermore, CRM-NLCs enhanced cytotoxicity towards the U373MG cell line with a higher accumulation in the brain compared to free CRM [108]. Similar studies indicated that CRM-NLCs showed 75% lower IC50 than free CRM, and prolonged CRM blood half-life by 6.4-folds. CRM-NLCs also displayed an enhanced-tumor growth inhibition ratio up to 82.3% in mice bearing A172 xenografts [104].
Indomethacin was also encapsulated in LNCs (IndOH-LNCs). This new formula exhibited high selectivity toward C6 and U138-MG glioma cell lines compared to human astrocytes through reduced cell viability by promoting apoptosis of glioblastoma cells [199]. Additionally, a higher concentration of resveratrol (RVR) in the brain was achieved when the drug was loaded inside the non-targeting-SLNs as compared to free RVR. This result proves the intrinsic ability of SLNs passive targeting into the brain [171]. In a similar fashion, doxorubicin (DOX) and paclitaxel (PTX) were also loaded inside positively charged SLNs to improve drugs spread through an in vitro BBB model. The positively charged stearyl amine and glycol chitosan based SLNs showed higher permeation through an hCMEC/D3 cell monolayer compared to the free DOX. In addition, an increase in toxicity towards glioblastoma cells was obtained from the PTX-loaded SLNs [200, 201]. Research findings revealed that the ability of bevacizumab (BVZ) to cross the hCMEC/D3 monolayer of cells increased by 100–200-fold when BVZ-SLNs was used compared to free BVZ [202]. Findings also indicated that in U87MG cells, the cytotoxic effectiveness of PTX-loaded NLCs and functionalized with Trf is superior as compared to unconjugated NLCs [106]. In this regard, Emami et al. (2018) developed a transferrin-conjugated NLC encapsulated artemisinin (ART) (TF-ART-NLCs) for brain delivery of artemisinin (ART). The cytotoxic activity of TF-ART-NLCs against U-87MG brain cancer cell line was much higher compared to non-targeted NLCs and free drug [103].
A novel NLCs of temozolomide (TMZ), a short half-life alkylating agent with good ability to penetrate BBB, along with DNA or functionalized by an arginine-glycine-aspartic acid (RGD) peptide were reported by Chen and coworkers [109] and Song and colleagues [105], respectively, with a size of 120–180 nm, +23 to +28 mV, and 83–85% encapsulation efficiency. These NLCs demonstrated potent anti-tumor activity against U87MG cells [105, 109]. Other TMZ-NLCs were prepared to enhance brain targeting via nasal route administration. TMZ-NLCs showed a nanometer size range with high drug loading and prolonged drug release, where a double brain targeting enhancement ratio was obtained compared to free TMZ [110]. NLCs were also co-encapsulated with TMZ and vincristine (VCR) and then co-functionalized with lactoferrin- and RGD for targeting glioblastoma multiforms (GBM). This combination targeting therapy was stable with high drug encapsulation efficiency and showed sustained-release, high cytotoxicity, and synergy effects with increased drug accumulation in the tumor tissue [203].
The therapeutic efficacy of SLNs and NLCs loaded with TMZ and VCR were also evaluated. Results revealed that co-delivery of the two encapsulated drugs was better than the use of TMZ or VCR alone in glioma in vitro and in vivo studies [86]. Costa and colleagues studied the delivery of anti-miR21 for treatment of GBM using NLCS coated with chlorotoxin (CTX), a toxin which binds preferentially to glioma cells; results showed a high drug encapsulation efficiency with a better accumulation within brain tumors compared to the free anti-miR-21 [204]. Researchers formulated TMZ and CUR co-loaded NLCs to enhance the synergistic anticancer efficacy of combined drugs. These researchers demonstrated that the co-loaded NLCs exhibit improved inhibitory effects on glioma cells compared to single drug loaded NLCs, and can accumulate in the brain and tumor sites effectively and achieve a major synergistic anticancer effect in vivo [205]. A comparative study to evaluate the effectiveness of lipid nanostructures (SLNs and NLCs) against glioblastoma with polymeric nanoparticles (PNPs) was performed, where nanoparticles were loaded with TMZ. In vitro and in vivo studies emphasized that TMZ loaded-NLCs have the best antitumor activity than other preparations with higher glioma inhibition over drug loaded-PNPs [95]. Table 1 provides a detailed summary of SLNs and NLCs targeting brain tumors.
Table 1.
A detailed summary of SLNs and NLCs targeting brain tumors.
| Sr. | Type of carrier | Solid lipid | Liquid lipid | Emulsifiers/stabilizers/surfactants | Preparation method | Therapeutic molecule | Targeting ligand | Bioassay | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Cationic SLN | Cacao butter or Compritol 888 ATO Hexadecyl trimethylammonium bromide |
- | Sodium dodecylsulfate (SDS) | Microemulsion | Carmustine | anti-EGFR or serotonergic 1B receptor antagonist | U87MG cells | [185, 187] |
| 2 | Cationic SLN | Cacao butter and cardiolipin or tripalmitin | Hexadecyl trimethylammonium bromide | Sodium dodecylsulfate (SDS) | Microemulsion | Doxorubicin | Anti-EGFR, or aprotinin, | U87MG cells | [99, 185] |
| 3 | Cationic SLN | Cacao butter and cardiolipin tripalmitin | Hexadecyl trimethylammonium bromide | Sodium dodecylsulfate (SDS) | Microemulsion | Etoposide | melanotransferrin antibody | U87MG cells | [206] |
| 4 | Cationic SLN | low-density lipoprotein (LDL) PEGylated c- tyrosine-protein kinase (c-Met) | Cholesteryl oleate and glyceryl trioleate | - | Emulsification-solvent evaporation | siRNAs | - | U87MG cell lines and U87MG xenograft tumor | [192] |
| 5 | Cationic SLN | Glyceryl tripalmitate glyceryl tristearate |
- | Tween 80 | High pressure homogenization method | pemetrexed and miR-21 | - | U87MG cells | [207] |
| 6 | SLN | cacao butter and tripalmitin Behenic acid |
- | - | Microemulsion | Carmustine | Tamoxifen and lactoferrin | U87MG cells | [96] |
| 7 | SLN | palmitic acid, stearic acid 1,3-ditetradecanoyloxypropan-2-yl tetradecanoate |
- | - | Emulsification-solvent evaporation | Etoposide | p-aminophenyl-α-D-manno-pyranoside and folic acid or 8314MAb | U87MG cells | [208] |
| 8 | SLN | Compritol® or Precirol | - | Tween 80 | Hot homogenization method | Edelfosine | _ | C6 glioma cell line and C6 glioma xenograft tumor | [190] |
| 9 | SLN | Stearic acid | Oleic acid | Polysorbate 80 | Emulsification–ultrasonication | Cytarabine | _ | Meningeal leukemic EL-4 cell line | [191] |
| 10 | SLN | Cetyl palmitate | - | Polysorbate 60 and 80 | High shear homogenisation and ultrasonication techniques. | Rhodamine 123 | - | (U373, U251, U87 and A172) cell lines | [194] |
| 11 | SLN | Cetyl palmitate, dynasan 114, Witepsol E85 | - | Polysorbate 20, 40, 60 and 80 | Hot high pressure homogenisation | Camptothecin | - | BCEC cell | [195] |
| 12 | SLN | Cetyl palmitate1,2- dimyristoyl-sn-glycero-3-phosphocholine (DMPC) | - | Polysorbate 60, 80 | Hot high pressure homogenisation | Camptothecin | - | Human glioma cell lines (A172, U251, U373, 153, U87) | [209] |
| 13 | SLN | Resveratrol and Compritol 888 ATO | - | Tween 80 polyvinyl alcohol | Solvent evaporation with high speed homogenization and ultrasonication | Resveratrol | glyceryl behenate | C6 glioma cells | [175] |
| 14 | SLN | Sodium stearate, sodium palmitate arachidic acid, ehenic acid | Sodium dioctyl sulfosuccinate and hydrochloric acid | - | Fatty acid coacervation technique | Doxorubicin | - | hCMEC/D3 cell monolayer | [204] |
| 15 | SLN | Stearylamine and glycol chitosan sodium Behenate | - | Tween_80, sodium dodecyl sulfate, Triton X-100 | Fatty acid coacervation technique | Paclitaxel | - | hCMEC/D3 cells monolayer and GBM cell lines | [205] |
| 16 | SLN | Stearic acid (SA) and palmitic acid (PA) | - | polyvinyl alcohol | fatty acid coacervation technique | Bevacizumab | - | hCMEC/D3 monolayer of cells | [95] |
| 17 | SLN | Soya Lecithin Stearic acid Glyceryl monostearate |
- | Tween 80 | Emulsification and solvent evaporation method | Docetaxel | p-Aminophenyl-a-Dmannopyranoside | U-87 MG cells and Swiss albino mice | [83] |
| 18 | SLN | Soya Lecithin Stearic acid Glyceryl monostearate |
- | Tween 80 | Emulsification and solvent evaporation method | Docetaxel | Lactoferin | U-87 MG cells and Swiss albino mice | [84] |
| 19 | SLN | Monostearate Distearate Tristearate |
- | Poloxamer 188 | Solvent evaporation and hot homogenisation technique | Asiatic acid | - | U87 MG cells | [210] |
| 20 | SLN | Stearic acid Compritol 888 ATO Cardiolipin Cholesteryl hemisuccinate |
- | Tween 80 | Emulsification and solvent evaporation method | Etoposide | Surface 83-14 monoclonal antibody and antiepithelial growth factor receptor | U87MG cells and HBMECs | [85] |
| 21 | SLN | Glyceryl monostearate Glycerol tristearate Sodium cholate |
- | Poloxamer188 | Emulsification–ultrasonication | β-Elemene | - | U87MG cells C6 glioma cells GL261 glioma |
[87] |
| 22 | SLN | Cetyl palmitate, Propylene glycol |
- | Cremephor RH 40, Peceol |
Melt-emulsification technique | Small interfering RNAs | Cyclic peptide: iRGD | U87MG cells GL261 glioma mice bearing malignant glioma |
[90] |
| 23 | SLN | Glycerol monostearate | - | Poloxamer-188 | Solvent injection and ultrasonication method | Vincristine | Red blood cell membrane NGR peptide (CYGGRGNG) T7 peptide (CHAIYPRH) |
C6 glioma cells ICR mice (male and female) |
[94] |
| 24 | SLN | Cetyl palmitate | - | Tween 80 | Solvent evaporation and hot homogenisation technique | Kiteplatin/Pt (IV)-prodrugs: SMF 111 SMF 196 SMF 200 SMF 144 |
16: 0 PEG-2-PE | U87MG cells hCMEC/D3 |
[97] |
| 25 | NLC | Cetyl palmitate isopropyl myristate isopropyl palmitate |
Glyceryl oleate | Isoceteth-20 ceteth-20 oleth-20 |
PIT method | Ferulic acid | didodecyldimethylammonium bromide | U87MG glioblastoma cancer cells line | [193] |
| 26 | NLC | Cetylpalmitate, Octyldodecanol Soy lecithin |
Different fatty amines tearylamine (SA), dodecylamine (DA) and spermine (SP) |
Poloxamer 188 | Emulsion-solvent evaporation method followed by probe sonication | Etoposide | Anti-transferrin | Myelogenous leukemia K562 cell line | [196] |
| 27 | NLC | Precirol soya lecithin | Capmul | Tween 80 | Hot high pressure homogenization | Curcumin | - | U373MG cell line | [111] |
| 28 | NLC | Tripalmitin | Oleic acid | Polysorbate 80 | Hot high pressure homogenization | Curcumin | - | mice bearing A172 xenografts | [104] |
| 29 | NLC | Cholesterol, triolein, soy lecithin, transferrin | Stearylamine, | Poloxamer 188 |
solvent evaporation method | Paclitaxel | anti-transferrin | U87MG cells | [106] |
| 30 | NLC | Compritol stearylamine, soy lecithin | oleic acid | Tween 80 | Solvent evaporation method | Artemisinin | anti-transferrin | U-87MG brain cancer cell line | [103] |
| 31 | NLC | Compritol 888 ATO soya lecithin |
Polyoxyl castor oil | Tween-80 and DDAB | Solvent diffusion method | Temozolomide | DNA | U87MG cells | [109] |
| 32 | NLC | NH2-PEG-DSPE Compritol 888 |
stearic acid | DDAB 888 ATO |
Solvent diffusion method | Temozolomide | arginine-glycine-aspartic acid (RGD) | U87MG cells | [105] |
| 33 | NLC | Gelucire Transcuto |
Vit E | Tween 80 | High-pressure homogenization followed by ultrasonication |
Temozolomide | - | Kinetic model | [110] |
| 34 | NLC | Compritol 888 ATO stearic acid, soybean phosphatidylcholine | Polyoxyl castor oil | DDAB | Solvent diffusion method | Temozolomide Vincristine | Lactoferrin and RGD | Glioblastoma multiforme (GBM) | [203] |
| 35 | NLC and SLNs | Soya lecithin | Stearic acid | DDAB 888 ATO, Cremophor |
solvent displacement technique solvent diffusion method |
Temozolomide Vincristine | - | U87MG cells | [86] |
| 36 | NLC | DSPC C16 Ceramide PEG2000 DSPE-PEG-Maleimide |
- | DODAP | postinsertion method | Chlorotoxin | anti-miR21 | Glioblastoma (GBM) | [204] |
| 37 | NLC | Glyceryl monostearate | medium chain triglycerides | Poloxamer 188 | Microemulsion method | TemozolomideCurcumin | - | C6 glioma cell line | [205] |
| 38 | NLC | Labrasol® Lauroglycol 90 Kolliphor® HS15 ynasan®114 |
Four liquid lipids | - | Hot homogenisation technique | Docetaxel | - | U87MG | [107] |
| 39 | NLC | Cetyl palmitate Soy phospholipid Labrasol® Transcutol® HP | Stearylamine | Tween 80 | High shear homogenization followed by an ultrasonication | Atorvastatin Curcumin | Hyaluronic acid conjugates that contain the cRGDfK and H7k(R2)2 peptides, and folic acid | U87MG cells mice bearing malignant glioma | [108] |
| 40 | NLC | Phosphatidyl choline glycerol trioleate and Precirol ATO 5 | Cholesterol oleate | Polysorbate 80 | Solvent evaporation technique | Curcumin | - | RAW 264.7, b. End3, Sprague-Dawley rats and ICR mice | [112] |
| 41 | LNC | triglycerides: Labrafac® WL 1349 | - | Solutol lecithins | Phase inversion microemulsion process | Locked nucleic acid (LNA) | L1 peptide | U87 malignant glioma (U87-MG) cell line | [197] |
| 42 | LNC | poly(e-caprolactone), sorbitan | Monoestearate grape seed oil |
Polysorbate 80 | Interfacial deposition | Curcumin | - | U251MG cell lineC6 gliomas | [198] |
| 43 | LNC | Poly(ε-caprolactone) sorbitan monostearate | Capric/caprylic triglycerides | Polysorbate 80 | Interfacial deposition | Indomethacin | - | C6 and U138-MG glioma cell lines | [199] |
5.3. Toxicity and biocompatibility
Since most of the components involved in SLNs and NLCs were approved for use in pharmaceutical formulations, these lipid nanoformulations are considered safe drug nanocarriers. Similar to other nanoparticles, toxicity of SLNs and NLCs may arise from the used linkers for targeting ligand attachment. In this regard, toxicity assessment of SLNs and/or NLCs involves investigating all components making the nanoparticles' backbone for their biological compatibility and safety [211].
In vitro studies involving SLNs in prostate cancer cell lines showed insignificant cytotoxicity with 90% cell viability of up to 3 mg/mL of dispersions [212]. However, precautions must be taken when using stabilizing or targeting agents such as cationic cetyltrimethylammonium bromide (CTAB) or different types of surfactants in the preparation of solid nanoparticles [213]. Within this context, CTAB-coated SLNs displayed decreased toxicity at concentrations up to 1 mg/mL with certain risks that CTAB promotes the release of calcium from neutrophil and induces their destruction [214, 215]. Intravenous injection of mice with SLNs caused neuroinflammation and neurovascular injury due to incorporation of cationic ingredients into SLNs [214, 216]. Moreover, CTAB-coated SLNs showed signs of oxidative stress activation in HepG2 liver cancer cells [217], which remarkably decreased with incorporation of an antioxidant active constituents into SLNs formulation [218, 219]. Similarly, polysorbate 80 and poloxamer 188 surfactants displayed acceptable biocompatibility and low toxicity as compared to cationic surfactants [220]. Furthermore, NLCs made from a combination of glycerol monostearate and polysorbate 80 exhibited a minor hemolytic effect, even at lipid doses of 1 mg/mL [221]. Research findings indicated that the major side effect of lipid-based nanoparticles is related to the loaded chemotherapeutic agents and not the nanocarrier itself [211]. In general, solid lipid nanoparticles are safe after oral, ocular, and dermal administration [222, 223].
On the other hand, stability enhancement of SLNs and NLCs was achieved by adding polyethylene glycol (PEG), which is appropriate to eliminate inflammatory reaction [64, 215]. Physical instability of NLC in terms of aggregation during storage is a major concern. In this respect, two methods can be used to enhance physical stability of the NLC; water elimination from the nanoparticle by freeze-drying and adding a preservative to the formulation [70].
6. Conclusions and future perspectives
Brain cancer is one of the common types of human cancers, which is known by being a fatal and non-curable disease with poor outcomes. There are various brain tumor treatment options, including surgery, radiation therapy, chemotherapy, and immunotherapy. However, these therapies do not have a high survival rate due to drug resistance and the high protection advantage by the blood brain barrier (BBB). BBB is characterized by a very low permeability for biomolecules required to regulate metabolism of the brain due to the presence of tight junctions, efflux transporters, and low enzymatic activity. Although these properties protect the CNS from the bloodstream components, they also limit most effective drugs reaching the CNS, thereby raising challenges of finding drugs and drug delivery systems that can cross the BBB. Nanomedicine can provide fertile grounds for the development of passive and active drug delivery systems that can reach the brain. In nanomedicine, different types of organic and inorganic nanoparticles such as lipid, polymeric, micelles, and metallic nanoparticles could be used for diagnosis, and the delivery of anticancer agents to the brain. In this context, lipid nanoparticles are the most successful drug delivery systems, and are in clinical use due to properties including biocompatibility, biodegradability, low toxicity, and acceptable pharmacokinetics and pharmacodynamics properties. Indeed, several studies provided proof-of-concept for the ability of lipid nanocarriers to cross the BBB in vivo, thereby confirming the important value of nanomedicine as a treatment strategy for brain cancer.
Despite the potent applications of lipid nanocarriers to reach the brain through crossing the BBB, the strategies and protocols require careful optimization to reach an optimal therapeutic index. First, materials used in formulating lipid nanoparticles should be selected based on the high biocompatibility and low toxicity; thereby not harming normal tissues and the integrity of BBB. Second, lipid nanocarriers should be stable for enough time in the bloodstream to achieve the required task properly. Third, functionalizing the surface of lipid nanoparticles to cross the BBB and reach the brain should be investigated to limit accumulation of the lipid nanoparticles in normal tissues and/or the BBB. Fourth, choosing proper animal models is crucial in pre-clinical trials. In summary, a quality-by-design (QbD) approach to investigate the efficacy of lipid and any type of nanocarriers can address all-important requirements for proper therapeutic outcomes.
Declarations
Author contribution statement
All authors listed have significantly contributed to the development and the writing of this article.
Funding statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data availability statement
No data was used for the research described in the article.
Declaration of interests statement
The authors declare no conflict of interest.
Additional information
No additional information is available for this paper.
Contributor Information
Fadwa Odeh, Email: f.odeh@ju.edu.jo.
Mohammad S. Mubarak, Email: mmubarak@ju.edu.jo.
References
- 1.Tinkle S., McNeil S.E., Mühlebach S., Bawa R., Borchard G., Barenholz Y.C. Nanomedicines: addressing the scientific and regulatory gap. Ann. N. Y. Acad. Sci. 2014;1313:35–56. doi: 10.1111/nyas.12403. [DOI] [PubMed] [Google Scholar]
- 2.Sarmento B. Have nanomedicines progressed as much as we’d hoped for in drug discovery and development? Expet Opin. Drug Discov. 2019;14(8):723–725. doi: 10.1080/17460441.2019.1621286. [DOI] [PubMed] [Google Scholar]
- 3.Krukemeyer M.G., Krenn, Hübner F., Wagner W., Resch R. History and possible uses of nanomedicine based on nanoparticles and nanotechnological progress. J. Nanomed. Nanotechnol. 2015;6:1–7. [Google Scholar]
- 4.Decuzzi P., Peer D., Mascolo D.D., Palange A.L., Manghnani P.N., Moghimi S.M. Roadmap on nanomedicine. Nanotechnology. 2020;32(1) doi: 10.1088/1361-6528/abaadb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cicha I., Chauvierre C., Texier I., Cabella C., Metselaar J.M., Szebeni J. From design to the clinic: practical guidelines for translating cardiovascular nanomedicine. Cardiovasc. Res. 2018;114(13):1714–1727. doi: 10.1093/cvr/cvy219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fu X., Shi Y., Qi T., Qiu S., Huang Y., Zhao X. Precise design strategies of nanomedicine for improving cancer therapeutic efficacy using subcellular targeting. Signal Transduct. Target. Ther. 2020;5(1):262. doi: 10.1038/s41392-020-00342-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bhise K., Sau S., Alsaab H., Kashaw S.K., Tekade R.K., Iyer A.K. Nanomedicine for cancer diagnosis and therapy: advancement, success and structure-activity relationship. Ther. Deliv. 2017;8(11):1003–1018. doi: 10.4155/tde-2017-0062. [DOI] [PubMed] [Google Scholar]
- 8.Macchione M.A., Aristizabal Bedoya D., Figueroa F.N., Muñoz-Fernández M., Strumia M.C. Nanosystems applied to HIV infection: prevention and treatments. Int. J. Mol. Sci. 2020;21(22):8647. doi: 10.3390/ijms21228647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Binda A., Murano C., Rivolta I. Innovative therapies and nanomedicine applications for the treatment of alzheimer's disease: a state-of-the-art (2017–2020) Int. J. Nanomed. 2020;15:6113–6135. doi: 10.2147/IJN.S231480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim K., Khang D. Past, present, and future of anticancer nanomedicine. Int. J. Nanomed. 2020;15:5719–5743. doi: 10.2147/IJN.S254774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Desland F.A., Hormigo A. The CNS and the brain tumor microenvironment: implications for glioblastoma immunotherapy. Int. J. Mol. Sci. 2020;21(19):7358. doi: 10.3390/ijms21197358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sharma H.S., Muresanu D.F., Castellani R.J., Nozari A., Lafuente J.V., Tian Z.R. In: International Review of Neurobiology. Bryukhovetskiy I., editor. Academic Press; 2020. Chapter One - pathophysiology of blood-brain barrier in brain tumor. Novel therapeutic advances using nanomedicine; pp. 1–66. [DOI] [PubMed] [Google Scholar]
- 13.Khaitan D., Reddy P.L., Ningaraj N. Targeting brain tumors with nanomedicines: overcoming blood brain barrier challenges. Curr. Clin. Pharmacol. 2018;13(2):110–119. doi: 10.2174/1574884713666180412150153. [DOI] [PubMed] [Google Scholar]
- 14.Scioli Montoto S., Muraca G., Ruiz M.E. Solid lipid nanoparticles for drug delivery: pharmacological and biopharmaceutical aspects. Front. Mole. Biosci. 2020;7 doi: 10.3389/fmolb.2020.587997. 587997–587997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Naseri N., Valizadeh H., Zakeri-Milani P. Solid lipid nanoparticles and nanostructured lipid carriers: structure, preparation and application. Adv. Pharmaceut. Bull. 2015;5(3):305–313. doi: 10.15171/apb.2015.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Contini C., Schneemilch M., Gaisford S., Quirke N. Nanoparticle–membrane interactions. J. Exp. Nanosci. 2018;13(1):62–81. [Google Scholar]
- 17.Panariti A., Miserocchi G., Rivolta I. The effect of nanoparticle uptake on cellular behavior: disrupting or enabling functions? Nanotechnol. Sci. Appl. 2012;5:87–100. doi: 10.2147/NSA.S25515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ghasemiyeh P., Mohammadi-Samani S. Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: applications, advantages and disadvantages. Res. Pharm. Sci. 2018;13(4):288–303. doi: 10.4103/1735-5362.235156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Thi T., Suys E., Lee J., Nguyen D., Park K., Truong N. Lipid-based nanoparticles in the clinic and clinical trials: from cancer nanomedicine to COVID-19 vaccines. Vaccines (Basel) 2021;9(4):359. doi: 10.3390/vaccines9040359. Apr 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Siegel R.L., Miller K.D., Jemal A. Cancer statistics, 2019. CA A Cancer J. Clin. 2019;69(1):7–34. doi: 10.3322/caac.21551. [DOI] [PubMed] [Google Scholar]
- 21.Safaee M.M., Lyon R., Barbaro N.M., Chou D., Mummaneni P.V., Weinstein P.R. Neurological outcomes and surgical complications in 221 spinal nerve sheath tumors, Journal of Neurosurgery. Spine. 2017;26(1):103–111. doi: 10.3171/2016.5.SPINE15974. [DOI] [PubMed] [Google Scholar]
- 22.Parashar B., Arora S., Wernicke A.G. Radiation therapy for early stage lung cancer. Semin. Intervent. Radiol. 2013;30(2):185–190. doi: 10.1055/s-0033-1342960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Arruebo M., Vilaboa N., Sáez-Gutierrez B., Lambea J., Tres A., Valladares M. Assessment of the evolution of cancer treatment therapies. Cancers. 2011;3(3):3279–3330. doi: 10.3390/cancers3033279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cavanagh T.T., Holle L.M. Potential new gene therapy option with sitimagene ceradenovec for newly diagnosed patients with glioblastoma multiforme. Canc. Biol. Ther. 2014;15(3):263–265. doi: 10.4161/cbt.27326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lindley C., McCune J.S., Thomason T.E., Lauder D., Sauls A., Adkins S. Perception of chemotherapy side effects cancer versus noncancer patients. Canc. Pract. 1999;7(2):59–65. doi: 10.1046/j.1523-5394.1999.07205.x. [DOI] [PubMed] [Google Scholar]
- 26.Prise K.M., Schettino G., Folkard M., Held K.D. New insights on cell death from radiation exposure. Lancet Oncol. 2005;6(7):520–528. doi: 10.1016/S1470-2045(05)70246-1. [DOI] [PubMed] [Google Scholar]
- 27.Gillet J.P., Gottesman M.M. Mechanisms of multidrug resistance in cancer. Methods Mol. Biol. 2010;596:47–76. doi: 10.1007/978-1-60761-416-6_4. [DOI] [PubMed] [Google Scholar]
- 28.Alfarouk K.O., Stock C.-M., Taylor S., Walsh M., Muddathir A.K., Verduzco D. Resistance to cancer chemotherapy: failure in drug response from ADME to P-gp. Canc. Cell Int. 2015;15(1):71. doi: 10.1186/s12935-015-0221-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mfouo Tynga I., Abrahamse H. Nano-mediated photodynamic therapy for cancer: enhancement of cancer specificity and therapeutic effects. Nanomaterials. 2018;8(11):923. doi: 10.3390/nano8110923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Alshaer W., Zraikat M., Amer A., Nsairat H., Lafi Z., Alqudah D.A. Encapsulation of echinomycin in cyclodextrin inclusion complexes into liposomes: in vitro anti-proliferative and anti-invasive activity in glioblastoma. RSC Adv. 2019;9(53):30976–30988. doi: 10.1039/c9ra05636j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Prasad M., Lambe U.P., Brar B., Shah I., J M., Ranjan K. Nanotherapeutics: an insight into healthcare and multi-dimensional applications in medical sector of the modern world. Biomed. Pharmacother. 2018;97:1521–1537. doi: 10.1016/j.biopha.2017.11.026. [DOI] [PubMed] [Google Scholar]
- 32.Nsairat H., Mahmoud I.S., Odeh F., Abuarqoub D., Al-Azzawi H., Zaza R. Grafting of anti-nucleolin aptamer into preformed and remotely loaded liposomes through aptamer-cholesterol post-insertion. RSC Adv. 2020;10(59):36219–36229. doi: 10.1039/d0ra07325c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wilhelm S., Tavares A.J., Dai Q., Ohta S., Audet J., Dvorak H.F. Analysis of nanoparticle delivery to tumours. Nat. Rev. Mater. 2016;1(5):16014. [Google Scholar]
- 34.Dvir T., Bauer M., Schroeder A., Tsui J.H., Anderson D.G., Langer R. Nanoparticles targeting the infarcted heart. Nano Lett. 2011;11(10):4411–4414. doi: 10.1021/nl2025882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jeevanandam J., Barhoum A., Chan Y.S., Dufresne A., Danquah M.K. Review on nanoparticles and nanostructured materials: history, sources, toxicity and regulations. Beilstein J. Nanotechnol. 2018;9:1050–1074. doi: 10.3762/bjnano.9.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shi Z., Zhou Y., Fan T., Lin Y., Zhang H., Mei L. Inorganic nano-carriers based smart drug delivery systems for tumor therapy. Smart Mater. Med. 2020;1:32–47. [Google Scholar]
- 37.Yan G., Li A., Zhang A., Sun Y., Liu J. Polymer-based nanocarriers for Co-delivery and combination of diverse therapies against cancers. Nanomaterials. 2018;8(2):85. doi: 10.3390/nano8020085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Puri A., Loomis K., Smith B., Lee J.-H., Yavlovich A., Heldman E. Lipid-based nanoparticles as pharmaceutical drug carriers: from concepts to clinic. Crit. Rev. Ther. Drug Carrier Syst. 2009;26(6):523–580. doi: 10.1615/critrevtherdrugcarriersyst.v26.i6.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.García-Pinel B., Porras-Alcalá C., Ortega-Rodríguez A., Sarabia F., Prados J., Melguizo C. Lipid-based nanoparticles: application and recent advances in cancer treatment. Nanomaterials. 2019;9(4):638. doi: 10.3390/nano9040638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Masserini M. Nanoparticles for brain drug delivery. ISRN Biochem. 2013;2013 doi: 10.1155/2013/238428. 238428–238428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jnaidi R., Almeida A.J., Gonçalves L.M. Solid lipid nanoparticles and nanostructured lipid carriers as smart drug delivery systems in the treatment of glioblastoma multiforme. Pharmaceutics. 2020;12(9):860. doi: 10.3390/pharmaceutics12090860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Arumugam K., Borawake P.D., Shinde J.V., Chavan R.S. Niosomes: a novel carrier drug delivery system. J. Drug Deliv. Ther. 2021;11(1):162–170. [Google Scholar]
- 43.Sapkota R., Dash A.K. Liposomes and transferosomes: a breakthrough in topical and transdermal delivery. Ther. Deliv. 2021;12(2):145–158. doi: 10.4155/tde-2020-0122. [DOI] [PubMed] [Google Scholar]
- 44.Ezzati Nazhad Dolatabadi J., Omidi Y. Solid lipid–based nanocarriers as efficient targeted drug and gene delivery systems. Trac. Trends Anal. Chem. 2016;77:100–108. [Google Scholar]
- 45.Feng L., Mumper R.J. A critical review of lipid-based nanoparticles for taxane delivery. Canc. Lett. 2013;334(2):157–175. doi: 10.1016/j.canlet.2012.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mehnert W., Mäder K. Solid lipid nanoparticles: production, characterization and applications. Adv. Drug Deliv. Rev. 2001;47(2):165–196. doi: 10.1016/s0169-409x(01)00105-3. [DOI] [PubMed] [Google Scholar]
- 47.Wissing S.A., Kayser O., Müller R.H. Solid lipid nanoparticles for parenteral drug delivery. Adv. Drug Deliv. Rev. 2004;56(9):1257–1272. doi: 10.1016/j.addr.2003.12.002. [DOI] [PubMed] [Google Scholar]
- 48.Mukherjee S., Ray S., Thakur R.S. Solid lipid nanoparticles: a modern formulation approach in drug delivery system. Indian J. Pharmaceut. Sci. 2009;71(4):349–358. doi: 10.4103/0250-474X.57282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Din F.U., Aman W., Ullah I., Qureshi O.S., Mustapha O., Shafique S. Effective use of nanocarriers as drug delivery systems for the treatment of selected tumors. Int. J. Nanomed. 2017;12:7291–7309. doi: 10.2147/IJN.S146315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Andonova V., Peneva P. Current Pharmaceutical Design; 2017. Characterization Methods for Solid Lipid Nanoparticles (SLN) and Nanostructured Lipid Carriers (NLC) [DOI] [PubMed] [Google Scholar]
- 51.Odeh F., Nsairat H., Alshaer W., Alsotari S., Buqaien R., Ismail S. Remote loading of curcumin-in-modified β-cyclodextrins into liposomes using a transmembrane pH gradient. RSC Adv. 2019;9(64):37148–37161. doi: 10.1039/c9ra07560g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Müller R.H., Mäder K., Gohla S. Solid lipid nanoparticles (SLN) for controlled drug delivery – a review of the state of the art. Eur. J. Pharm. Biopharm. 2000;50(1):161–177. doi: 10.1016/s0939-6411(00)00087-4. [DOI] [PubMed] [Google Scholar]
- 53.Duan Y., Dhar A., Patel C., Khimani M., Neogi S., Sharma P. A brief review on solid lipid nanoparticles: part and parcel of contemporary drug delivery systems. RSC Adv. 2020;10(45):26777–26791. doi: 10.1039/d0ra03491f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Uner M., Yener G. Importance of solid lipid nanoparticles (SLN) in various administration routes and future perspectives. Int. J. Nanomed. 2007;2(3):289–300. [PMC free article] [PubMed] [Google Scholar]
- 55.Patra J.K., Das G., Fraceto L.F., Campos E.V.R., Rodriguez-Torres M.D.P., Acosta-Torres L.S. Nano based drug delivery systems: recent developments and future prospects. J. Nanobiotechnol. 2018;16(1) doi: 10.1186/s12951-018-0392-8. 71–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Duong V.-A., Nguyen T.-T.-L., Maeng H.-J. Preparation of solid lipid nanoparticles and nanostructured lipid carriers for drug delivery and the effects of preparation parameters of solvent injection method. Molecules. 2020;25(20):4781. doi: 10.3390/molecules25204781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Das S., Chaudhury A. Recent advances in lipid nanoparticle formulations with solid matrix for oral drug delivery. AAPS PharmSciTech. 2011;12(1):62–76. doi: 10.1208/s12249-010-9563-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Das S., Ng W.K., Tan R.B. Are nanostructured lipid carriers (NLCs) better than solid lipid nanoparticles (SLNs): development, characterizations and comparative evaluations of clotrimazole-loaded SLNs and NLCs? Eur. J. Pharmaceut. Sci. 2012;47(1):139–151. doi: 10.1016/j.ejps.2012.05.010. [DOI] [PubMed] [Google Scholar]
- 59.Jenning V., Thünemann A.F., Gohla S.H. Characterisation of a novel solid lipid nanoparticle carrier system based on binary mixtures of liquid and solid lipids. Int. J. Pharm. 2000;199(2):167–177. doi: 10.1016/s0378-5173(00)00378-1. [DOI] [PubMed] [Google Scholar]
- 60.Tan S.W., Billa N., Roberts C.R., Burley J.C. Surfactant effects on the physical characteristics of Amphotericin B-containing nanostructured lipid carriers. Colloid. Surface. Physicochem. Eng. Aspect. 2010;372(1):73–79. [Google Scholar]
- 61.Manjunath K., Reddy J.S., Venkateswarlu V. Solid lipid nanoparticles as drug delivery systems. Methods Find. Exp. Clin. Pharmacol. 2005;27(2):127–144. doi: 10.1358/mf.2005.27.2.876286. [DOI] [PubMed] [Google Scholar]
- 62.Cirri M., Maestrini L., Maestrelli F., Mennini N., Mura P., Ghelardini C. Design, characterization and in vivo evaluation of nanostructured lipid carriers (NLC) as a new drug delivery system for hydrochlorothiazide oral administration in pediatric therapy. Drug Deliv. 2018;25(1):1910–1921. doi: 10.1080/10717544.2018.1529209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Chauhan I., Yasir M., Verma M., Singh A.P. Nanostructured lipid carriers: a groundbreaking approach for transdermal drug delivery. Adv. Pharmaceut. Bull. 2020;10(2):150–165. doi: 10.34172/apb.2020.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Haider M., Abdin S.M., Kamal L., Orive G. Nanostructured lipid carriers for delivery of chemotherapeutics: a review. Pharmaceutics. 2020;12(3):288. doi: 10.3390/pharmaceutics12030288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Poonia N., Kharb R., Lather V., Pandita D. Nanostructured lipid carriers: versatile oral delivery vehicle. Fut. Sci. OA. 2016;2(3) doi: 10.4155/fsoa-2016-0030. FSO135–FSO135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Pardeshi C., Rajput P., Belgamwar V., Tekade A., Patil G., Chaudhary K. Solid lipid based nanocarriers: an overview. Acta Pharm. 2012;62(4):433–472. doi: 10.2478/v10007-012-0040-z. [DOI] [PubMed] [Google Scholar]
- 67.Sharif Makhmal Zadeh B., Niro H., Rahim F., Esfahani G. Ocular delivery system for propranolol hydrochloride based on nanostructured lipid carrier. Sci. Pharm. 2018;86(2):16. doi: 10.3390/scipharm86020016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Müller R.H., Radtke M., Wissing S.A. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv. Drug Deliv. Rev. 2002;54(Suppl 1):S131–155. doi: 10.1016/s0169-409x(02)00118-7. [DOI] [PubMed] [Google Scholar]
- 69.Gorantla S., Rapalli V.K., Waghule T., Singh P.P., Dubey S.K., Saha R.N. Nanocarriers for ocular drug delivery: current status and translational opportunity. RSC Adv. 2020;10(46):27835–27855. doi: 10.1039/d0ra04971a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Khosa A., Reddi S., Saha R.N. Nanostructured lipid carriers for site-specific drug delivery. Biomed. Pharmacother. 2018;103:598–613. doi: 10.1016/j.biopha.2018.04.055. [DOI] [PubMed] [Google Scholar]
- 71.Souto E.B., Baldim I., Oliveira W.P., Rao R., Yadav N., Gama F.M. SLN and NLC for topical, dermal, and transdermal drug delivery. Expet Opin. Drug Deliv. 2020;17(3):357–377. doi: 10.1080/17425247.2020.1727883. [DOI] [PubMed] [Google Scholar]
- 72.Mishra V., Bansal K.K., Verma A., Yadav N., Thakur S., Sudhakar K. Solid lipid nanoparticles: emerging colloidal nano drug delivery systems. Pharmaceutics. 2018;10(4):191. doi: 10.3390/pharmaceutics10040191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Bunjes H., Koch M.H. Saturated phospholipids promote crystallization but slow down polymorphic transitions in triglyceride nanoparticles. J. Contr. Release. 2005;107(2):229–243. doi: 10.1016/j.jconrel.2005.06.004. [DOI] [PubMed] [Google Scholar]
- 74.Müller R.H., Petersen R.D., Hommoss A., Pardeike J. Nanostructured lipid carriers (NLC) in cosmetic dermal products. Adv. Drug Deliv. Rev. 2007;59(6):522–530. doi: 10.1016/j.addr.2007.04.012. [DOI] [PubMed] [Google Scholar]
- 75.Müller R.H., Shegokar R., Keck C.M. 20 years of lipid nanoparticles (SLN and NLC): present state of development and industrial applications. Curr. Drug Discov. Technol. 2011;8(3):207–227. doi: 10.2174/157016311796799062. [DOI] [PubMed] [Google Scholar]
- 76.Kasongo K.W., Müller R.H., Walker R.B. The use of hot and cold high pressure homogenization to enhance the loading capacity and encapsulation efficiency of nanostructured lipid carriers for the hydrophilic antiretroviral drug, didanosine for potential administration to paediatric patients. Curr. Drug Discov. Technol. 2012;17(3):353–362. doi: 10.3109/10837450.2010.542163. [DOI] [PubMed] [Google Scholar]
- 77.Nobari Azar F.A., Pezeshki A., Ghanbarzadeh B., Hamishehkar H., Mohammadi M. Nanostructured lipid carriers: promising delivery systems for encapsulation of food ingredients. J. Agri. Food Res. 2020;2:100084. [Google Scholar]
- 78.Guo T., Zhang Y., Zhao J., Zhu C., Feng N. Nanostructured lipid carriers for percutaneous administration of alkaloids isolated from Aconitum sinomontanum. J. Nanobiotechnol. 2015;13(1):47. doi: 10.1186/s12951-015-0107-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Salvi V.R., Pawar P. Nanostructured lipid carriers (NLC) system: a novel drug targeting carrier. J. Drug Deliv. Sci. Technol. 2019;51:255–267. [Google Scholar]
- 80.Suarez S., Almutairi A., Christman K.L. Micro- and nanoparticles for treating cardiovascular disease. Biomater. Sci. 2015;3(4):564–580. doi: 10.1039/C4BM00441H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wohlfart S., Gelperina S., Kreuter J. Transport of drugs across the blood–brain barrier by nanoparticles. J. Contr. Release. 2012;161(2):264–273. doi: 10.1016/j.jconrel.2011.08.017. [DOI] [PubMed] [Google Scholar]
- 82.Sanjula B., Shah F.M., Javed A., Alka A. Effect of poloxamer 188 on lymphatic uptake of carvedilol-loaded solid lipid nanoparticles for bioavailability enhancement. J. Drug Target. 2009;1(3):249–256. doi: 10.1080/10611860902718672. [DOI] [PubMed] [Google Scholar]
- 83.Singh I., Swami R., Jeengar M.K., Khan W., Sistla R. p-Aminophenyl-α-D-mannopyranoside engineered lipidic nanoparticles for effective delivery of docetaxel to brain. Chem. Phys. Lipids. 2015;188:1–9. doi: 10.1016/j.chemphyslip.2015.03.003. [DOI] [PubMed] [Google Scholar]
- 84.Singh I., Swami R., Pooja D., Jeengar M.K., Khan W., Sistla R. Lactoferrin bioconjugated solid lipid nanoparticles: a new drug delivery system for potential brain targeting. J. Drug Target. 2016;24(3):212–223. doi: 10.3109/1061186X.2015.1068320. [DOI] [PubMed] [Google Scholar]
- 85.Kuo Y.C., Lee C.H. Dual targeting of solid lipid nanoparticles grafted with 83–14 MAb and anti-EGF receptor for malignant brain tumor therapy. Life Sci. 2016;146:222–231. doi: 10.1016/j.lfs.2016.01.025. [DOI] [PubMed] [Google Scholar]
- 86.Wu M., Fan Y., Lv S., Xiao B., Ye M., Zhu X. Vincristine and temozolomide combined chemotherapy for the treatment of glioma: a comparison of solid lipid nanoparticles and nanostructured lipid carriers for dual drugs delivery. Drug Deliv. 2016;23(8):2720–2725. doi: 10.3109/10717544.2015.1058434. [DOI] [PubMed] [Google Scholar]
- 87.He H., Yao J., Zhang Y., Chen Y., Wang K., Lee R.J. Solid lipid nanoparticles as a drug delivery system to across the blood-brain barrier. Biochem. Biophys. Res. Commun. 2019;519(2):385–390. doi: 10.1016/j.bbrc.2019.09.017. [DOI] [PubMed] [Google Scholar]
- 88.Mao S.R., Wang Y.Z., Ji H.Y., Bi D.Z. [Preparation of solid lipid nanoparticles by microemulsion technique] Yao Xue Xue Bao. 2003;38(8):624–626. [PubMed] [Google Scholar]
- 89.Tartaro G., Mateos H., Schirone D., Angelico R., Palazzo G. Microemulsion microstructure(s): a tutorial review. Nanomaterials. 2020;10:1657. doi: 10.3390/nano10091657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Erel-Akbaba G., Carvalho L.A., Tian T., Zinter M., Akbaba H., Obeid P.J. Radiation-Induced targeted nanoparticle-based gene delivery for brain tumor therapy. ACS Nano. 2019;13(4):4028–4040. doi: 10.1021/acsnano.8b08177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Vladisavljević G.T. Integrated Membrane Systems and Processes. 2016. Integrated membrane processes for the preparation of emulsions, particles and bubbles; pp. 79–140. [Google Scholar]
- 92.Rao J., McClements D.J. Stabilization of phase inversion temperature nanoemulsions by surfactant displacement. J. Agric. Food Chem. 2010;58(11):7059–7066. doi: 10.1021/jf100990r. [DOI] [PubMed] [Google Scholar]
- 93.Suarez S., Almutairi A., Christman K.L. Micro- and nanoparticles for treating cardiovascular disease. Biomater. Sci. 2015;3(4):564–580. doi: 10.1039/C4BM00441H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Fu S., Liang M., Wang Y., Cui L., Gao C., Chu X. Dual-modified novel biomimetic nanocarriers improve targeting and therapeutic efficacy in glioma. ACS Appl. Mater. Interfaces. 2019;11(2):1841–1854. doi: 10.1021/acsami.8b18664. [DOI] [PubMed] [Google Scholar]
- 95.Qu J., Zhang L., Chen Z., Mao G., Gao Z., Lai X. Nanostructured lipid carriers, solid lipid nanoparticles, and polymeric nanoparticles: which kind of drug delivery system is better for glioblastoma chemotherapy? Drug Deliv. 2016;23(9):3408–3416. doi: 10.1080/10717544.2016.1189465. [DOI] [PubMed] [Google Scholar]
- 96.Kuo Y.C., Cheng S.J. Brain targeted delivery of carmustine using solid lipid nanoparticles modified with tamoxifen and lactoferrin for antitumor proliferation. Int. J. Pharm. 2016;499(1–2):10–19. doi: 10.1016/j.ijpharm.2015.12.054. [DOI] [PubMed] [Google Scholar]
- 97.Arduino I., Depalo N., Re F., Dal Magro R., Panniello A., Margiotta N. PEGylated solid lipid nanoparticles for brain delivery of lipophilic kiteplatin Pt(IV) prodrugs: an in vitro study. Int. J. Pharm. 2020;583:119351. doi: 10.1016/j.ijpharm.2020.119351. [DOI] [PubMed] [Google Scholar]
- 98.Fu S., Liang M., Wang Y., Cui L., Gao C., Chu X. Dual-modified novel biomimetic nanocarriers improve targeting and therapeutic efficacy in glioma. ACS Appl. Mater. Interfaces. 2019;11(2):1841–1854. doi: 10.1021/acsami.8b18664. [DOI] [PubMed] [Google Scholar]
- 99.Kuo Y.-C., Lee I.H. Delivery of doxorubicin to glioblastoma multiforme in vitro using solid lipid nanoparticles with surface aprotinin and melanotransferrin antibody for enhanced chemotherapy. J. Taiwan. Instit. Chem. Eng. 2016;61:32–45. [Google Scholar]
- 100.Shidhaye S.S., Vaidya R., Sutar S., Patwardhan A., Kadam V.J. Solid lipid nanoparticles and nanostructured lipid carriers-innovative generations of solid lipid carriers. Curr. Drug Deliv. 2008;5(4):324–331. doi: 10.2174/156720108785915087. [DOI] [PubMed] [Google Scholar]
- 101.Mo Z., Ban J., Zhang Y., Du Y., Wen Y., Huang X. Nanostructured lipid carriers-based thermosensitive eye drops for enhanced, sustained delivery of dexamethasone. Nanomedicine (Lond) 2018;13(11):1239–1253. doi: 10.2217/nnm-2017-0318. [DOI] [PubMed] [Google Scholar]
- 102.Selvamuthukumar S., Velmurugan R. Nanostructured lipid carriers: a potential drug carrier for cancer chemotherapy. Lipids Health Dis. 2012;11:159. doi: 10.1186/1476-511X-11-159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Emami J., Yousefian H., Sadeghi H. Targeted nanostructured lipid carrier for brain delivery of artemisinin: design, preparation, characterization, optimization and cell toxicity. J. Pharm. Pharmaceut. Sci. 2018;21(1s):225s–241s. doi: 10.18433/jpps30117. [DOI] [PubMed] [Google Scholar]
- 104.Chen Y., Pan L., Jiang M., Li D., Jin L. Nanostructured lipid carriers enhance the bioavailability and brain cancer inhibitory efficacy of curcumin both in vitro and in vivo. Drug Deliv. 2016;23(4):1383–1392. doi: 10.3109/10717544.2015.1049719. [DOI] [PubMed] [Google Scholar]
- 105.Song S., Mao G., Du J., Zhu X. Novel RGD containing, temozolomide-loading nanostructured lipid carriers for glioblastoma multiforme chemotherapy. Drug Deliv. 2016;23(4):1404–1408. doi: 10.3109/10717544.2015.1064186. [DOI] [PubMed] [Google Scholar]
- 106.Emami J., Rezazadeh M., Sadeghi H., Khadivar K. Development and optimization of transferrin-conjugated nanostructured lipid carriers for brain delivery of paclitaxel using Box-Behnken design. Pharmaceut. Dev. Technol. 2017;22(3):370–382. doi: 10.1080/10837450.2016.1189933. [DOI] [PubMed] [Google Scholar]
- 107.Zwain T., Alder J.E., Sabagh B., Shaw A., Burrow A.J., Singh K.K. Tailoring functional nanostructured lipid carriers for glioblastoma treatment with enhanced permeability through in-vitro 3D BBB/BBTB models. Mater. Sci. Eng. C. 2021;121:111774. doi: 10.1016/j.msec.2020.111774. [DOI] [PubMed] [Google Scholar]
- 108.Basso J., Mendes M., Silva J., Sereno J., Cova T., Oliveira R. Peptide-lipid nanoconstructs act site-specifically towards glioblastoma growth impairment. Eur. J. Pharm. Biopharm. 2020;155:177–189. doi: 10.1016/j.ejpb.2020.08.015. [DOI] [PubMed] [Google Scholar]
- 109.Chen Z., Lai X., Song S., Zhu X., Zhu J. Nanostructured lipid carriers based temozolomide and gene co-encapsulated nanomedicine for gliomatosis cerebri combination therapy. Drug Deliv. 2016;23(4):1369–1373. doi: 10.3109/10717544.2015.1038857. [DOI] [PubMed] [Google Scholar]
- 110.Khan A., Imam S.S., Aqil M., Ahad A., Sultana Y., Ali A. Brain targeting of temozolomide via the intranasal route using lipid-based nanoparticles: brain pharmacokinetic and scintigraphic analyses. Mol. Pharmacol. 2016;13(11):3773–3782. doi: 10.1021/acs.molpharmaceut.6b00586. [DOI] [PubMed] [Google Scholar]
- 111.Madane R.G., Mahajan H.S. Curcumin-loaded nanostructured lipid carriers (NLCs) for nasal administration: design, characterization, and in vivo study. Drug Deliv. 2016;23(4):1326–1334. doi: 10.3109/10717544.2014.975382. [DOI] [PubMed] [Google Scholar]
- 112.Meng F., Asghar S., Xu Y., Wang J., Jin X., Wang Z. Design and evaluation of lipoprotein resembling curcumin-encapsulated protein-free nanostructured lipid carrier for brain targeting. Int. J. Pharm. 2016;506(1):46–56. doi: 10.1016/j.ijpharm.2016.04.033. [DOI] [PubMed] [Google Scholar]
- 113.Gordillo-Galeano A., Mora-Huertas C.E. Solid lipid nanoparticles and nanostructured lipid carriers: a review emphasizing on particle structure and drug release. Eur. J. Pharm. Biopharm. 2018;133:285–308. doi: 10.1016/j.ejpb.2018.10.017. [DOI] [PubMed] [Google Scholar]
- 114.Silva A.C., González-Mira E., García M.L., Egea M.A., Fonseca J., Silva R. Preparation, characterization and biocompatibility studies on risperidone-loaded solid lipid nanoparticles (SLN): high pressure homogenization versus ultrasound. Colloids Surf. B Biointerfaces. 2011;86(1):158–165. doi: 10.1016/j.colsurfb.2011.03.035. [DOI] [PubMed] [Google Scholar]
- 115.Chattopadhyay P., Shekunov B.Y., Yim D., Cipolla D., Boyd B., Farr S. Production of solid lipid nanoparticle suspensions using supercritical fluid extraction of emulsions (SFEE) for pulmonary delivery using the AERx system. Adv. Drug Deliv. Rev. 2007;59(6):444–453. doi: 10.1016/j.addr.2007.04.010. [DOI] [PubMed] [Google Scholar]
- 116.Iqbal M.A., Md S., Sahni J.K., Baboota S., Dang S., Ali J. Nanostructured lipid carriers system: recent advances in drug delivery. J. Drug Target. 2012;20(10):813–830. doi: 10.3109/1061186X.2012.716845. [DOI] [PubMed] [Google Scholar]
- 117.Carbone C., Cupri S., Leonardi A., Puglisi G., Pignatello R. Lipid-based nanocarriers for drug delivery and targeting: a patent survey of methods of production and characterization. Pharm. Patent Anal. 2013;2(5):665–677. doi: 10.4155/ppa.13.43. [DOI] [PubMed] [Google Scholar]
- 118.Koshy O., Subramanian L., Thomas S. Thermal and Rheological Measurement Techniques for Nanomaterials Characterization. Elsevier; 2017. Differential scanning calorimetry in nanoscience and nanotechnology; pp. 109–122. [Google Scholar]
- 119.Byrn S.R., Zografi G., Chen X. Solid State Properties of Pharmaceutical Materials. Willy online Library; 2017. Differential scanning calorimetry and thermogravimetric analysis. Capter 10. [Google Scholar]
- 120.Almeida A.J., Souto E. Solid lipid nanoparticles as a drug delivery system for peptides and proteins. Adv. Drug Deliv. Rev. 2007;59(6):478–490. doi: 10.1016/j.addr.2007.04.007. [DOI] [PubMed] [Google Scholar]
- 121.Salmaso S., Elvassore N., Bertucco A., Caliceti P. Production of solid lipid submicron particles for protein delivery using a novel supercritical gas-assisted melting atomization process. J. Pharmaceut. Sci. 2009;98(2):640–650. doi: 10.1002/jps.21434. [DOI] [PubMed] [Google Scholar]
- 122.Yoon G., Park J.W., Yoon I.-S. Solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs): recent advances in drug delivery. J. Pharm. Invest. 2013;43(5):353–362. [Google Scholar]
- 123.Charcosset C., El-Harati A., Fessi H. Preparation of solid lipid nanoparticles using a membrane contactor. J. Contr. Release. 2005;108(1):112–120. doi: 10.1016/j.jconrel.2005.07.023. [DOI] [PubMed] [Google Scholar]
- 124.Izquierdo P., Esquena J., Tadros T.F., Dederen J.C., Feng J., Garcia-Celma M.J. Phase behavior and nano-emulsion formation by the phase inversion temperature method. Langmuir. 2004;20(16):6594–6598. doi: 10.1021/la049566h. [DOI] [PubMed] [Google Scholar]
- 125.Ma Q.H., Xia Q., Lu Y.Y., Hao X.Z., Gu N., Lin X.F. Preparation of tea polyphenols-loaded solid lipid nanoparticles based on the phase behaviors of hot microemulsions. Solid State Phenom. 2007;121–123:705–708. [Google Scholar]
- 126.Zimmermann E., Souto E.B., Muller R.H. Physicochemical investigations on the structure of drug-free and drug-loaded solid lipid nanoparticles (SLN) by means of DSC and 1H NMR. Pharmazie. 2005;60:508–513. [PubMed] [Google Scholar]
- 127.Mukherjee S., Ray S., Thakur R.S. Solid lipid nanoparticles: a modern formulation approach in drug delivery system. Indian J. Pharmaceut. Sci. 2009;71(4):349–358. doi: 10.4103/0250-474X.57282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Board P.A.T.E. PDQ Cancer Information Summaries. National Cancer Institute (US); 2020. Adult central nervous system tumors treatment (PDQ®) [Internet] [Google Scholar]
- 129.Grisold W., Grisold A. Cancer around the brain. Neuro-Oncol. Pract. 2014;1(1):13–21. doi: 10.1093/nop/npt002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Schiffer D. Springer; VI: 2006. Brain Tumor Pathology: Current Diagnostic Hotspots and Pitfalls; p. 272. [Google Scholar]
- 131.Zong H., Verhaak R.G.W., Canoll P. The cellular origin for malignant glioma and prospects for clinical advancements. Expert Rev. Mol. Diagn. 2012;12(4):383–394. doi: 10.1586/erm.12.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Minniti G., Filippi A.R., Osti M.F., Ricardi U. Radiation therapy for older patients with brain tumors. Radiat. Oncol. 2017;12(1) doi: 10.1186/s13014-017-0841-9. 101–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Koo Y.-E.L., Reddy G.R., Bhojani M., Schneider R., Philbert M.A., Rehemtulla A. Brain cancer diagnosis and therapy with nanoplatforms. Adv. Drug Deliv. Rev. 2006;58(14):1556–1577. doi: 10.1016/j.addr.2006.09.012. [DOI] [PubMed] [Google Scholar]
- 134.Upadhyay R.K. Drug delivery systems, CNS protection, and the blood brain barrier. BioMed Res. Int. 2014;2014 doi: 10.1155/2014/869269. 869269–869269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Daneman R., Prat A. The blood-brain barrier. Cold Spring Harbor Perspect. Biol. 2015;7(1) doi: 10.1101/cshperspect.a020412. a020412–a020412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Kutuzov N., Flyvbjerg H., Lauritzen M. Contributions of the glycocalyx, endothelium, and extravascular compartment to the blood–brain barrier. Proc. Natl. Acad. Sci. Unit. States Am. 2018;115(40):E9429–E9438. doi: 10.1073/pnas.1802155115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Abbott N.J., Rönnbäck L., Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 2006;7(1):41–53. doi: 10.1038/nrn1824. [DOI] [PubMed] [Google Scholar]
- 138.Aviral J., Sanjay K.J. Drug targeting to the brain - a review. Curr. Nanosci. 2011;7(1):21–36. [Google Scholar]
- 139.Pardridge W.M. The blood-brain barrier: bottleneck in brain drug development. NeuroRx. 2005;2(1):3–14. doi: 10.1602/neurorx.2.1.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Tosi G., Bortot B., Ruozi B., Dolcetta D., Vandelli M.A., Forni F. Potential use of polymeric nanoparticles for drug delivery across the blood-brain barrier. Curr. Med. Chem. 2013;20(17):2212–2225. doi: 10.2174/0929867311320170006. [DOI] [PubMed] [Google Scholar]
- 141.Lucy S.-C., Lauren M.S., Brandon J.T., Thomas P.D., Patrick T.R. Transporters at CNS barrier sites: obstacles or opportunities for drug delivery? Curr. Pharmaceut. Des. 2014;20(10):1422–1449. doi: 10.2174/13816128113199990463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.David J.B., Charles C.P., Maurizio S. Lysosomal storage diseases and the blood-brain barrier. Curr. Pharmaceut. Des. 2008;14(16):1566–1580. doi: 10.2174/138161208784705504. [DOI] [PubMed] [Google Scholar]
- 143.Abbott N.J., Patabendige A.A.K., Dolman D.E.M., Yusof S.R., Begley D.J. Structure and function of the blood–brain barrier. Neurobiol. Dis. 2010;37(1):13–25. doi: 10.1016/j.nbd.2009.07.030. [DOI] [PubMed] [Google Scholar]
- 144.David J.B. ABC transporters and the blood-brain barrier. Curr. Pharmaceut. Des. 2004;10(12):1295–1312. doi: 10.2174/1381612043384844. [DOI] [PubMed] [Google Scholar]
- 145.Preston J.E., Joan Abbott N., Begley D.J. In: Advances in Pharmacology. Davis T.P., editor. Academic Press; 2014. Chapter five - transcytosis of macromolecules at the blood–brain barrier; pp. 147–163. [DOI] [PubMed] [Google Scholar]
- 146.Barua S., Mitragotri S. Challenges associated with penetration of nanoparticles across cell and tissue barriers: a review of current status and future prospects. Nano Today. 2014;9(2):223–243. doi: 10.1016/j.nantod.2014.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Tapeinos C., Battaglini M., Ciofani G. Advances in the design of solid lipid nanoparticles and nanostructured lipid carriers for targeting brain diseases. J. Contr. Release. 2017;264:306–332. doi: 10.1016/j.jconrel.2017.08.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Gastaldi L., Battaglia L., Peira E., Chirio D., Muntoni E., Solazzi I. Solid lipid nanoparticles as vehicles of drugs to the brain: current state of the art. Eur. J. Pharm. Biopharm. 2014;87(3):433–444. doi: 10.1016/j.ejpb.2014.05.004. [DOI] [PubMed] [Google Scholar]
- 149.Laquintana V., Trapani A., Denora N., Wang F., Gallo J.M., Trapani G. New strategies to deliver anticancer drugs to brain tumors. Expet Opin. Drug Deliv. 2009;6(10):1017–1032. doi: 10.1517/17425240903167942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.F. B. Mesfin and M. A. Al-Dhahir, Gliomas, in StatPearls. 2020, © 2020, StatPearls Publishing LLC.: Treasure Island FL.
- 151.Huang J., Gholami B., Agar N.Y.R., Norton I., Haddad W.M., Tannenbaum A.R. Annual International Conference of the IEEE Engineering in Medicine and Biology Society IEEE Engineering in Medicine and Biology Society Annual International Conference 2011. 2011. Classification of astrocytomas and oligodendrogliomas from mass spectrometry data using sparse kernel machines; pp. 7965–7968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Rajesh Y., Pal I., Banik P., Chakraborty S., Borkar S.A., Dey G. Insights into molecular therapy of glioma: current challenges and next generation blueprint. Acta Pharmacol. Sin. 2017;38(5):591–613. doi: 10.1038/aps.2016.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Blakeley J., Grossman S. Anaplastic oligodendroglioma. Curr. Treat. Options Neurol. 2008;10(4):295–307. doi: 10.1007/s11940-008-0032-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Qian X., Goumnerova L.C., De Girolami U., Cibas E.S. Cerebrospinal fluid cytology in patients with ependymoma. Canc. Cytopathol. 2008;114(5):307–314. doi: 10.1002/cncr.23799. [DOI] [PubMed] [Google Scholar]
- 155.Hambardzumyan D., Bergers G. Glioblastoma: defining tumor niches. Trends Canc. 2015;1(4):252–265. doi: 10.1016/j.trecan.2015.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Nanda A., Vannemreddy P. Recurrence and outcome in skull base meningiomas: do they differ from other intracranial meningiomas? Skull Base : Off. J. N. Am. Skull Base Soc. 2008;18(4):243–252. doi: 10.1055/s-2007-1016956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Millard N.E., De Braganca K.C. Medulloblastoma. J. Child Neurol. 2016;31(12):1341–1353. doi: 10.1177/0883073815600866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Rampling R., James A., Papanastassiou V. The present and future management of malignant brain tumours: surgery, radiotherapy, chemotherapy. J. Neurol. Neurosurg. Psychiatr. 2004;75(suppl 2):ii24. doi: 10.1136/jnnp.2004.040535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Shapiro W.R. Current therapy for brain tumors: back to the future. Arch. Neurol. 1999;56(4):429–432. doi: 10.1001/archneur.56.4.429. [DOI] [PubMed] [Google Scholar]
- 160.Upadhyay R.K. Drug delivery systems, CNS protection, and the blood brain barrier. BioMed Res. Int. 2014;2014:869269. doi: 10.1155/2014/869269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Müller R.H., Radtke M., Wissing S.A. Nanostructured lipid matrices for improved microencapsulation of drugs. Int. J. Pharm. 2002;242(1–2):121–128. doi: 10.1016/s0378-5173(02)00180-1. [DOI] [PubMed] [Google Scholar]
- 162.Arvanitis C.D., Ferraro G.B., Jain R.K. The blood–brain barrier and blood–tumour barrier in brain tumours and metastases. Nat. Rev. Canc. 2020;20(1):26–41. doi: 10.1038/s41568-019-0205-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.van Tellingen O., Yetkin-Arik B., de Gooijer M.C., Wesseling P., Wurdinger T., de Vries H.E. Overcoming the blood–brain tumor barrier for effective glioblastoma treatment. Drug Resist. Updates. 2015;19:1–12. doi: 10.1016/j.drup.2015.02.002. [DOI] [PubMed] [Google Scholar]
- 164.Juillerat-Jeanneret L. The targeted delivery of cancer drugs across the blood–brain barrier: chemical modifications of drugs or drug-nanoparticles? Drug Discov. Today. 2008;13(23):1099–1106. doi: 10.1016/j.drudis.2008.09.005. [DOI] [PubMed] [Google Scholar]
- 165.Rabinow B.E. Nanosuspensions in drug delivery. Nat. Rev. Drug Discov. 2004;3(9):785–796. doi: 10.1038/nrd1494. [DOI] [PubMed] [Google Scholar]
- 166.Li F., Wang Y., Liu Z., Lin X., He H., Tang X. Formulation and characterization of bufadienolides-loaded nanostructured lipid carriers. Drug Dev. Ind. Pharm. 2010;36(5):508–517. doi: 10.3109/03639040903264397. [DOI] [PubMed] [Google Scholar]
- 167.Craparo E.F., Bondì M.L., Pitarresi G., Cavallaro G. Nanoparticulate systems for drug delivery and targeting to the central nervous system. CNS Neurosci. Ther. 2011;17(6):670–677. doi: 10.1111/j.1755-5949.2010.00199.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Alam T., Pandit J., Vohora D., Aqil M., Ali A., Sultana Y. Optimization of nanostructured lipid carriers of lamotrigine for brain delivery: in vitro characterization and in vivo efficacy in epilepsy. Expet Opin. Drug Deliv. 2015;12(2):181–194. doi: 10.1517/17425247.2014.945416. [DOI] [PubMed] [Google Scholar]
- 169.Haque S., Md S., Alam M.I., Sahni J.K., Ali J., Baboota S. Nanostructure-based drug delivery systems for brain targeting. Drug Dev. Ind. Pharm. 2012;38(4):387–411. doi: 10.3109/03639045.2011.608191. [DOI] [PubMed] [Google Scholar]
- 170.Natarajan J., Baskaran M., Humtsoe L.C., Vadivelan R., Justin A. Enhanced brain targeting efficacy of Olanzapine through solid lipid nanoparticles. Artif. Cell. Nanomed. Biotechnol. 2017;45(2):364–371. doi: 10.3109/21691401.2016.1160402. [DOI] [PubMed] [Google Scholar]
- 171.Jose S., Anju S.S., Cinu T.A., Aleykutty N.A., Thomas S., Souto E.B. In vivo pharmacokinetics and biodistribution of resveratrol-loaded solid lipid nanoparticles for brain delivery. Int. J. Pharm. 2014;474(1–2):6–13. doi: 10.1016/j.ijpharm.2014.08.003. [DOI] [PubMed] [Google Scholar]
- 172.Teixeira M.C., Carbone C., Souto E.B. Beyond liposomes: recent advances on lipid based nanostructures for poorly soluble/poorly permeable drug delivery. Prog. Lipid Res. 2017;68:1–11. doi: 10.1016/j.plipres.2017.07.001. [DOI] [PubMed] [Google Scholar]
- 173.Esposito E., Drechsler M., Mariani P., Carducci F., Servadio M., Melancia F. Lipid nanoparticles for administration of poorly water soluble neuroactive drugs. Biomed. Microdevices. 2017;19(3):44. doi: 10.1007/s10544-017-0188-x. [DOI] [PubMed] [Google Scholar]
- 174.Gabathuler R. Approaches to transport therapeutic drugs across the blood-brain barrier to treat brain diseases. Neurobiol. Dis. 2010;37(1):48–57. doi: 10.1016/j.nbd.2009.07.028. [DOI] [PubMed] [Google Scholar]
- 175.Hsu S.H., Wen C.J., Al-Suwayeh S.A., Chang H.W., Yen T.C., Fang J.Y. Physicochemical characterization and in vivo bioluminescence imaging of nanostructured lipid carriers for targeting the brain: apomorphine as a model drug. Nanotechnology. 2010;21(40):405101. doi: 10.1088/0957-4484/21/40/405101. [DOI] [PubMed] [Google Scholar]
- 176.Tsai M.J., Wu P.C., Huang Y.B., Chang J.S., Lin C.L., Tsai Y.H. Baicalein loaded in tocol nanostructured lipid carriers (tocol NLCs) for enhanced stability and brain targeting. Int. J. Pharm. 2012;423(2):461–470. doi: 10.1016/j.ijpharm.2011.12.009. [DOI] [PubMed] [Google Scholar]
- 177.Liu Z., Zhao H., Shu L., Zhang Y., Okeke C., Zhang L. Preparation and evaluation of Baicalin-loaded cationic solid lipid nanoparticles conjugated with OX26 for improved delivery across the BBB. Drug Dev. Ind. Pharm. 2015;41(3):353–361. doi: 10.3109/03639045.2013.861478. [DOI] [PubMed] [Google Scholar]
- 178.Pashirova T.N., Zueva I.V., Petrov K.A., Babaev V.M., Lukashenko S.S., Rizvanov I.K. Nanoparticle-delivered 2-PAM for rat brain protection against paraoxon central toxicity. ACS Appl. Mater. Interfaces. 2017;9(20):16922–16932. doi: 10.1021/acsami.7b04163. [DOI] [PubMed] [Google Scholar]
- 179.Vyas A., Jain A., Hurkat P., Jain A., Jain S.K. Targeting of AIDS related encephalopathy using phenylalanine anchored lipidic nanocarrier. Colloids Surf. B Biointerfaces. 2015;131:155–161. doi: 10.1016/j.colsurfb.2015.04.049. [DOI] [PubMed] [Google Scholar]
- 180.Gadgil P., Shah J., Chow D.S.L. Enhanced brain delivery with lower hepatic exposure of lazaroid loaded nanostructured lipid carriers developed using a design of experiment approach. Int. J. Pharm. 2018;544(1):265–277. doi: 10.1016/j.ijpharm.2018.04.046. [DOI] [PubMed] [Google Scholar]
- 181.Salunkhe S.S., Bhatia N.M., Bhatia M.S. Implications of formulation design on lipid -based nanostructured carrier system for drug delivery to brain. Drug Deliv. 2016;23(4):1306–1316. doi: 10.3109/10717544.2014.943337. [DOI] [PubMed] [Google Scholar]
- 182.E Eleraky N., M Omar M., A Mahmoud H., Abou-Taleb H.A. Nanostructured lipid carriers to mediate brain delivery of temazepam: design and in vivo study. Pharmaceutics. 2020;12(5):451. doi: 10.3390/pharmaceutics12050451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Anand A., Sugumaran A., Narayanasamy D. Brain targeted delivery of anticancer drugs: prospective approach using solid lipid nanoparticles. IET Nanobiotechnol. 2019;13(4):353–362. doi: 10.1049/iet-nbt.2018.5322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Kuo Y.C., Liang C.T. Inhibition of human brain malignant glioblastoma cells using carmustine-loaded catanionic solid lipid nanoparticles with surface anti-epithelial growth factor receptor. Biomaterials. 2011;32(12):3340–3350. doi: 10.1016/j.biomaterials.2011.01.048. [DOI] [PubMed] [Google Scholar]
- 185.Kuo Y.C., Liang C.T. Catanionic solid lipid nanoparticles carrying doxorubicin for inhibiting te growth of U87MG cells. Colloids Surf. B Biointerfaces. 2011;85(2):131–137. doi: 10.1016/j.colsurfb.2011.02.011. [DOI] [PubMed] [Google Scholar]
- 186.Kuo Y.-C., Lee C.-H. Inhibition against growth of glioblastoma multiforme in vitro using etoposide-loaded solid lipid nanoparticles with aminophenyl-D-manno-pyranoside and folic acid. J. Pharmaceut. Sci. 2015;104(5):1804–1814. doi: 10.1002/jps.24388. [DOI] [PubMed] [Google Scholar]
- 187.Kuo Y.-C., Wang C.-C. Carmustine-loaded catanionic solid lipid nanoparticles with serotonergic 1B receptor subtype antagonist for in vitro targeted delivery to inhibit brain cancer growth. J. Taiwan. Instit. Chem. Eng. 2015;46:1–14. [Google Scholar]
- 188.Kuo Y.-C., Chao I.-W. Conjugation of melanotransferrin antibody on solid lipid nanoparticles for mediating brain cancer malignancy. Biotechnol. Prog. 2016;32(2):480–490. doi: 10.1002/btpr.2214. [DOI] [PubMed] [Google Scholar]
- 189.Kuo Y.C., Wang I.H. Enhanced delivery of etoposide across the blood-brain barrier to restrain brain tumor growth using melanotransferrin antibody- and tamoxifen-conjugated solid lipid nanoparticles. J. Drug Target. 2016;24(7):645–654. doi: 10.3109/1061186X.2015.1132223. [DOI] [PubMed] [Google Scholar]
- 190.Estella-Hermoso de Mendoza A., Préat V., Mollinedo F., Blanco-Prieto M.J. In vitro and in vivo efficacy of edelfosine-loaded lipid nanoparticles against glioma. J. Contr. Release. 2011;156(3):421–426. doi: 10.1016/j.jconrel.2011.07.030. [DOI] [PubMed] [Google Scholar]
- 191.Sharma P., Dube B., Sawant K. Development and evaluation of nanostructured lipid carriers of cytarabine for treatment of meningeal leukemia. J. Nanosci. Nanotechnol. 2011;11(8):6676–6682. doi: 10.1166/jnn.2011.4235. [DOI] [PubMed] [Google Scholar]
- 192.Jin J., Bae K.H., Yang H., Lee S.J., Kim H., Kim Y. In vivo specific delivery of c-Met siRNA to glioblastoma using cationic solid lipid nanoparticles. Bioconjugate Chem. 2011;22(12):2568–2572. doi: 10.1021/bc200406n. [DOI] [PubMed] [Google Scholar]
- 193.Carbone C., Campisi A., Musumeci T., Raciti G., Bonfanti R., Puglisi G. FA-loaded lipid drug delivery systems: preparation, characterization and biological studies. Eur. J. Pharmaceut. Sci. 2014;52:12–20. doi: 10.1016/j.ejps.2013.10.003. [DOI] [PubMed] [Google Scholar]
- 194.Martins S., Costa-Lima S., Carneiro T., Cordeiro-da-Silva A., Souto E.B., Ferreira D.C. Solid lipid nanoparticles as intracellular drug transporters: an investigation of the uptake mechanism and pathway. Int. J. Pharm. 2012;430(1–2):216–227. doi: 10.1016/j.ijpharm.2012.03.032. [DOI] [PubMed] [Google Scholar]
- 195.Martins S., Tho I., Reimold I., Fricker G., Souto E., Ferreira D. Brain delivery of camptothecin by means of solid lipid nanoparticles: formulation design, in vitro and in vivo studies. Int. J. Pharm. 2012;439(1–2):49–62. doi: 10.1016/j.ijpharm.2012.09.054. [DOI] [PubMed] [Google Scholar]
- 196.Khajavinia A., Varshosaz J., Dehkordi A.J. Targeting etoposide to acute myelogenous leukaemia cells using nanostructured lipid carriers coated with transferrin. Nanotechnology. 2012;23(40):405101. doi: 10.1088/0957-4484/23/40/405101. [DOI] [PubMed] [Google Scholar]
- 197.Griveau A., Bejaud J., Anthiya S., Avril S., Autret D., Garcion E. Silencing of miR-21 by locked nucleic acid-lipid nanocapsule complexes sensitize human glioblastoma cells to radiation-induced cell death. Int. J. Pharm. 2013;454(2):765–774. doi: 10.1016/j.ijpharm.2013.05.049. [DOI] [PubMed] [Google Scholar]
- 198.Zanotto-Filho A., Coradini K., Braganhol E., Schröder R., de Oliveira C.M., Simões-Pires A. Curcumin-loaded lipid-core nanocapsules as a strategy to improve pharmacological efficacy of curcumin in glioma treatment. Eur. J. Pharm. Biopharm. 2013;83(2):156–167. doi: 10.1016/j.ejpb.2012.10.019. [DOI] [PubMed] [Google Scholar]
- 199.Bernardi A., Frozza R.L., Hoppe J.B., Salbego C., Pohlmann A.R., Battastini A.M.O. The antiproliferative effect of indomethacin-loaded lipid-core nanocapsules in glioma cells is mediated by cell cycle regulation, differentiation, and the inhibition of survival pathways. Int. J. Nanomed. 2013;(8):711–728. doi: 10.2147/IJN.S40284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Battaglia L., Gallarate M., Peira E., Chirio D., Muntoni E., Biasibetti E. Solid lipid nanoparticles for potential doxorubicin delivery in glioblastoma treatment: preliminary in vitro studies. J. Pharmaceut. Sci. 2014;103(7):2157–2165. doi: 10.1002/jps.24002. [DOI] [PubMed] [Google Scholar]
- 201.Chirio D., Gallarate M., Peira E., Battaglia L., Muntoni E., Riganti C. Positive-charged solid lipid nanoparticles as paclitaxel drug delivery system in glioblastoma treatment. Eur. J. Pharm. Biopharm. 2014;88(3):746–758. doi: 10.1016/j.ejpb.2014.10.017. [DOI] [PubMed] [Google Scholar]
- 202.Battaglia L., Gallarate M., Peira E., Chirio D., Solazzi I., Giordano S.M. Bevacizumab loaded solid lipid nanoparticles prepared by the coacervation technique: preliminary in vitro studies. Nanotechnology. 2015;26(25):255102. doi: 10.1088/0957-4484/26/25/255102. [DOI] [PubMed] [Google Scholar]
- 203.Zhang J., Xiao X., Zhu J., Gao Z., Lai X., Zhu X. Lactoferrin- and RGD-comodified, temozolomide and vincristine-coloaded nanostructured lipid carriers for gliomatosis cerebri combination therapy. Int. J. Nanomed. 2018;13:3039–3051. doi: 10.2147/IJN.S161163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Costa P.M., Cardoso A.L., Custódia C., Cunha P., Pereira de Almeida L., Pedroso de Lima M.C. MiRNA-21 silencing mediated by tumor-targeted nanoparticles combined with sunitinib: a new multimodal gene therapy approach for glioblastoma. J. Contr. Release. 2015;207:31–39. doi: 10.1016/j.jconrel.2015.04.002. [DOI] [PubMed] [Google Scholar]
- 205.Xu M., Li G., Zhang H., Chen X., Li Y., Yao Q. Sequential delivery of dual drugs with nanostructured lipid carriers for improving synergistic tumor treatment effect. Drug Deliv. 2020;27(1):983–995. doi: 10.1080/10717544.2020.1785581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Kuo Y.C., Chao I.W. Conjugation of melanotransferrin antibody on solid lipid nanoparticles for mediating brain cancer malignancy. Biotechnol. Prog. 2016;32(2):480–490. doi: 10.1002/btpr.2214. [DOI] [PubMed] [Google Scholar]
- 207.Küçüktürkmen B., Bozkır A. Development and characterization of cationic solid lipid nanoparticles for co-delivery of pemetrexed and miR-21 antisense oligonucleotide to glioblastoma cells. Drug Dev. Ind. Pharm. 2018;44(2):306–315. doi: 10.1080/03639045.2017.1391835. [DOI] [PubMed] [Google Scholar]
- 208.Kuo Y.C., Lee C.H. Inhibition against growth of glioblastoma multiforme in vitro using etoposide-loaded solid lipid nanoparticles with p-aminophenyl-α-D-manno-pyranoside and folic acid. J. Pharmaceut. Sci. 2015;104(5):1804–1814. doi: 10.1002/jps.24388. [DOI] [PubMed] [Google Scholar]
- 209.Martins S.M., Sarmento B., Nunes C., Lúcio M., Reis S., Ferreira D.C. Brain targeting effect of camptothecin-loaded solid lipid nanoparticles in rat after intravenous administration. Eur. J. Pharm. Biopharm. 2013;85(3 Pt A):488–502. doi: 10.1016/j.ejpb.2013.08.011. [DOI] [PubMed] [Google Scholar]
- 210.Garanti T., Stasik A., Burrow A.J., Alhnan M.A., Wan K.W. Anti-glioma activity and the mechanism of cellular uptake of asiatic acid-loaded solid lipid nanoparticles. Int. J. Pharm. 2016;500(1–2):305–315. doi: 10.1016/j.ijpharm.2016.01.018. [DOI] [PubMed] [Google Scholar]
- 211.Doktorovová S., Kovačević A.B., Garcia M.L., Souto E.B. Preclinical safety of solid lipid nanoparticles and nanostructured lipid carriers: current evidence from in vitro and in vivo evaluation. Eur. J. Pharm. Biopharm. 2016;108:235–252. doi: 10.1016/j.ejpb.2016.08.001. [DOI] [PubMed] [Google Scholar]
- 212.Akanda M.H., Rai R., Slipper I.J., Chowdhry B.Z., Lamprou D., Getti G. Delivery of retinoic acid to LNCap human prostate cancer cells using solid lipid nanoparticles. Int. J. Pharm. 2015;493(1):161–171. doi: 10.1016/j.ijpharm.2015.07.042. [DOI] [PubMed] [Google Scholar]
- 213.Almeida H., Lobão P., Frigerio C., Fonseca J., Silva R., Sousa Lobo J.M. Preparation, characterization and biocompatibility studies of thermoresponsive eyedrops based on the combination of nanostructured lipid carriers (NLC) and the polymer Pluronic F-127 for controlled delivery of ibuprofen. Pharmaceut. Dev. Technol. 2017;22(3):336–349. doi: 10.3109/10837450.2015.1125922. [DOI] [PubMed] [Google Scholar]
- 214.Hwang T.-L., Aljuffali I.A., Hung C.-F., Chen C.-H., Fang J.-Y. The impact of cationic solid lipid nanoparticles on human neutrophil activation and formation of neutrophil extracellular traps (NETs) Chem. Biol. Interact. 2015;235:106–114. doi: 10.1016/j.cbi.2015.04.011. [DOI] [PubMed] [Google Scholar]
- 215.Hwang T.L., Aljuffali I.A., Lin C.F., Chang Y.T., Fang J.Y. Cationic additives in nanosystems activate cytotoxicity and inflammatory response of human neutrophils: lipid nanoparticles versus polymeric nanoparticles. Int. J. Nanomed. 2015;10:371–385. doi: 10.2147/IJN.S73017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Kasongo W.A., Pardeike J., Müller R.H., Walker R.B. Selection and characterization of suitable lipid excipients for use in the manufacture of didanosine-loaded solid lipid nanoparticles and nanostructured lipid carriers. J. Pharmaceut. Sci. 2011;100(12):5185–5196. doi: 10.1002/jps.22711. [DOI] [PubMed] [Google Scholar]
- 217.Doktorovova S., Souto E.B., Silva A.M. Nanotoxicology applied to solid lipid nanoparticles and nanostructured lipid carriers - a systematic review of in vitro data. Eur. J. Pharm. Biopharm. 2014;87(1):1–18. doi: 10.1016/j.ejpb.2014.02.005. [DOI] [PubMed] [Google Scholar]
- 218.Kyadarkunte A.Y., Patole M.S., Pokharkar V.B. Cellular interactions and photoprotective effects of idebenone-loaded nanostructured lipid carriers stabilized using PEG-free surfactant. Int. J. Pharm. 2015;479(1):77–87. doi: 10.1016/j.ijpharm.2014.12.044. [DOI] [PubMed] [Google Scholar]
- 219.El-Far M., Salah N., Essam A., El-Azim A.A., Karam M., El-Sherbiny I.M. Potential anticancer activity and mechanism of action of nanoformulated curcumin in experimental Ehrlich ascites carcinoma-bearing animals. Nanomedicine. 2019;14(5):553–573. doi: 10.2217/nnm-2018-0298. [DOI] [PubMed] [Google Scholar]
- 220.Müller R.H., Rühl D., Runge S., Schulze-Forster K., Mehnert W. Cytotoxicity of solid lipid nanoparticles as a function of the lipid matrix and the surfactant. Pharmaceut. Res. 1997;14(4):458–462. doi: 10.1023/a:1012043315093. [DOI] [PubMed] [Google Scholar]
- 221.Lakkadwala S., Nguyen S., Lawrence J., Nauli S.M., Nesamony J. Physico-chemical characterisation, cytotoxic activity, and biocompatibility studies of tamoxifen-loaded solid lipid nanoparticles prepared via a temperature-modulated solidification method. J. Microencapsul. 2014;31(6):590–599. doi: 10.3109/02652048.2014.898707. [DOI] [PubMed] [Google Scholar]
- 222.Rao M.P., Manjunath K., Bhagawati S.T., Thippeswamy B.S. Bixin loaded solid lipid nanoparticles for enhanced hepatoprotection-preparation, characterisation and in vivo evaluation. Int. J. Pharm. 2014;473(1–2):485–492. doi: 10.1016/j.ijpharm.2014.07.027. [DOI] [PubMed] [Google Scholar]
- 223.del Pozo-Rodríguez A., Delgado D., Gascón A.R., Solinís M. Lipid nanoparticles as drug/gene delivery systems to the retina. J. Ocul. Pharmacol. Therapeut. 2013;29(2):173–188. doi: 10.1089/jop.2012.0128. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data was used for the research described in the article.