Abstract
The adenovirus type 5 early region 1A gene (E1A) has previously been known as an immortalization oncogene because E1A is required for transforming oncogenes, such as ras and E1B, to transform cells in primary cultures. However, E1A has also been shown to downregulate the overexpression of the Her-2/neu oncogene, resulting in suppression of transformation and tumorigenesis induced by that oncogene. In addition, E1A is able to promote apoptosis induced by anticancer drugs, irradiation, and serum deprivation. Many tyrosine kinases, such as the epidermal growth factor receptor, Her-2/Neu, Src, and Axl, are known to play a role in oncogenic signals in transformed cells. To study the mechanism underlying the E1A-mediated tumor-suppressing function, we exploited a modified tyrosine kinase profile assay (D. Robinson, F. Lee, T. Pretlow, and H.-J. Kung, Proc. Natl. Acad. Sci. USA 93:5958–5962, 1996) to identify potential tyrosine kinases regulated by E1A. Reverse transcription (RT)-PCR products were synthesized with two degenerate primers derived from the conserved motifs of various tyrosine kinases. A tyrosine kinase downregulated by E1A was identified by analyzing the AluI-digested RT-PCR products. We isolated the DNA fragment of interest and found that E1A negatively regulated the expression of the transforming receptor tyrosine kinase Axl at the transcriptional level. To study whether downregulation of the Axl receptor is involved in E1A-mediated growth suppression, we transfected axl cDNA into E1A-expressing cells (ip1-E1A) to establish cells that overexpressed Axl. The Axl ligand Gas6 triggered a greater mitogenic effect in these ip1-E1A-Axl cells than in ip1-E1A control cells and protected the Axl-expressing cells from serum deprivation-induced apoptosis. These results indicate that downregulation of the Axl receptor by E1A is involved in E1A-mediated growth suppression and E1A-induced apoptosis and thereby contributes to E1A’s antitumor activities.
The adenovirus type 5 early region 1A gene (E1A), the first viral gene expressed in a cell after adenovirus infection, encodes two major proteins, 243R (12S) and 289R (13S), that are produced by alternative splicing (40). The primary function of these two proteins is to activate viral promoters of early genes, including E1B, E2A, E3, and E4, during a permissive viral infection by modifying the host cell transcriptional apparatus, thereby resulting in host cell immortalization or transformation (2). Cellular genes that are transcriptionally activated by the E1A proteins include those encoding β-tubulin (42), heat shock proteins (22), c-Fos (10, 37), c-Jun (10), JunB (10), and c-Myc (37). E1A can also repress several viral and cellular genes at the transcriptional level, such as the simian virus 40 enhancer (3, 50), the polyomavirus enhancer (3, 49), the immunoglobulin heavy-chain gene (21), the cytochrome P450 gene (41), the insulin gene (43), and the Her-2/neu gene (57).
Adenovirus type 5 E1A is known to be an immortalization oncogene because of the fact that some transforming oncogenes, such as ras and E1B, although able to induce transformation of immortalized rodent cell lines, require E1A to transform cells in primary cultures. However, adenovirus type 5 E1A alone cannot transform established cell lines (5, 23, 36). Furthermore, there exist many established cell lines that are immortalized but nontumorigenic. As a matter of fact, we have previously shown that E1A could be a tumor suppressor for Her-2/neu-transformed cells by transcriptional repression of the oncogene (52, 57). In addition to downregulating Her-2/Neu expression, E1A has multiple antitumor effects on tumor cells that do not overexpress Her-2/Neu, including reversion of transformation (12, 15, 16, 28, 55), inhibition of metastasis (17, 28, 53, 54, 56), and induction of apoptosis (9, 11, 33, 45). To understand the detailed mechanisms underlying E1A’s antitumor activities, we examined the E1A-regulated tyrosine kinases and identified Axl, whose expression was suppressed by E1A.
The Axl tyrosine kinase (p140) is the prototype of a family of transmembrane receptors called UFO that include Sky and Eyk. These receptors have a unique extracellular composition of immunoglobin-like and fibronectin type III-like domains (13, 30, 47). Similar domain architectures have been found in adhesion molecules of the cadherin and immunoglobulin superfamily and in receptor tyrosine phosphatases such as PTPμ and PTPκ, suggesting that Axl family members may mediate cell adhesion and intracellular signaling. Increased expression of the Axl receptor can transform NIH 3T3 cells and renders the transformed cells highly tumorigenic in nude mice (30).
All members of the UFO family can transform NIH 3T3 cells. When transfected, the axl gene transforms NIH 3T3 cells by overexpression (30). Since no genetic rearrangements or mutations have been found, its transforming ability is most probably a consequence of normal receptor overexpression. The Axl ligand Gas6 is a vitamin K-dependent growth-potentiating factor (26, 44, 48). The ligand contains a γ-carboxyglutamic acid-rich domain and four epidermal growth factor-like repeats. It is widely secreted by most tissues, particularly those of the lung, intestine, and vascular endothelium (26). Under serum-starved conditions, Gas6 has a mitogenic effect on growth-arrested cells (1, 19, 20). Gas6-Axl signaling also seems to play a critical role in modulating cellular responses to adhesion; in one study, Gas6 stimulated the binding of Axl-expressing monoblast U937 cells to phosphatidylserine, a phospholipid marker used by macrophages to identify dying cells (29). Gas6 also functions as a novel chemoattractant that induces Axl-mediated migration of vascular smooth-muscle cells, suggesting that the Gas6-Axl interaction may enhance cell migration in conditions involving vascular damage (14). In addition, the Axl receptor has been found to be highly expressed in metastatic colon carcinoma (8), melanoma (32), and sarcoma (51) cells, implying that this receptor is involved in cancer invasion and metastasis.
The published data, taken together, suggest that E1A is associated with a tumor suppressor function in human tumor cells and that the Axl receptor behaves as an oncoprotein when it is overexpressed. To study the mechanism underlying E1A’s tumor-suppressing activities, we focused on E1A-regulated tyrosine kinases. In the current study, we found that E1A can downregulate the expression of the Axl receptor and that the Gas6-Axl interaction can counteract E1A-mediated growth inhibition and proapoptotic activity, suggesting that downregulation of Axl by E1A may contribute to E1A-mediated antitumor activities.
MATERIALS AND METHODS
Cell lines and cell cultures.
SKOV3.ip1 (abbreviated ip1) is a subline of the SKOV3 ovarian cancer cell line. 2774 C-10 (abbreviated 2774) is a human ovarian cancer cell line. The E1A transfectants were designated ip1-E1A and 2774-E1A. The transfectants of the E1A frameshift mutant were designated ip1-efs and 2774-efs. Cells were grown in Dulbecco’s modified Eagle’s medium plus F12 medium (1:1; GIBCO-Bethesda Research Laboratories [BRL]) supplemented with 10% fetal calf serum in a humidified atmosphere of 5% CO2 at 37°C. ip1-efs, ip1-E1A, 2774-efs, and 2774-E1A cells were maintained in medium containing neomycin at 500 μg/ml (Boehringer Mannheim).
Tyrosine kinase display assay.
Total RNA was isolated with the TRIzol reagent (GIBCO-BRL), and the tyrosine kinase display was carried out by a modified form of the method of Robinson et al. (34, 35). Reverse transcription (RT)-PCR was performed with degenerate primers derived from conserved motifs in the activation loop of the catalytic domains of various tyrosine kinases as follows: primer 1 (sense primer), 5′-AARRTTDCNGAYTTYGG encoding the amino acid sequence K[V/I][S/C/G]DFG; primer 2 (antisense primer), 5′-RHAIGMCCAIACRTC encoding the amino acid sequence DVW[S/A][F/Y]. The mixed bases were defined as follows: N = A + C + T + G, D = A + T + G, H = A + T + C, R = A + G, Y = C + T, M = A + C, and I = deoxyinosine. Single-stranded cDNA was synthesized with a kit provided by GIBCO-BRL. Primer 1 was labeled with [γ-32P]ATP (Amersham Life Science) and T4 polynucleotide kinase (New England Biolabs) before PCR (Taq DNA polymerase; Fisher Biotech). The RT-PCR products were analyzed by gel electrophoresis with an 8% polyacrylamide gel. DNA was stained with ethidium bromide at 1 μg/ml. The ∼150-bp bands were excised from the polyacrylamide gel. DNA was eluted, precipitated, and dissolved in TE buffer (10 mM Tris, 1 mM EDTA, pH 8.0). Equal amounts of radioactive DNA (105 cpm) of each cell type were digested with various restriction enzymes recognizing four bases, resolved with a 6% DNA sequencing gel, and then subjected to autoradiography.
Cloning of the band of interest revealed by the tyrosine kinase display assay.
In the tyrosine kinase display assay, we found that the E1A proteins downregulated a tyrosine kinase whose catalytic domain sequence possessed the AluI site. This tyrosine kinase was tentatively named TK-Alu I. Because the TK-Alu I band shown in Fig. 1B had been digested by AluI, we could not PCR amplify the TK-Alu I DNA by using the two primers described previously, because the annealing sequence of the TK-Alu I for primer 2 had been separated from the radioactive sequence shown in Fig. 1B by the AluI digestion. To clone TK-Alu I, we established a tyrosine kinase cDNA library from the RT-PCR products of ip1 cells with the TA cloning strategy. At first, all of the RT-PCR products of the 150-bp DNA fragments were ligated into the TA cloning vector PCR2.1 (Invitrogen Inc.) to make a cDNA library of tyrosine kinases. The library was amplified in Escherichia coli DH10B. Then, we performed a large-scale preparation of the AluI-digested RT-PCR product. The TK-Alu I band was excised, and the DNA was eluted from the sequencing gel. A TK-Alu I probe was made by synthesizing single-stranded DNA by PCR in a mixture containing primer 1, [α-32P]dCTP, and the eluted TK-Alu I DNA. The radiolabeled probe was used to screen the tyrosine kinase cDNA library described above, the positive clones were picked out, and the plasmid DNA was purified and subjected to PCR as described for the tyrosine kinase display assay. RT-PCR and plasmid-PCR products were digested with AluI and then analyzed by sequencing gel electrophoresis to determine whether the selected clones contained the TK-Alu I insert. Finally, the truly positive TK-Alu I clone was sequenced and that sequence was identified by comparison with the GenBank database sequences of the National Center for Biotechnology Information by using the BLAST algorithm.
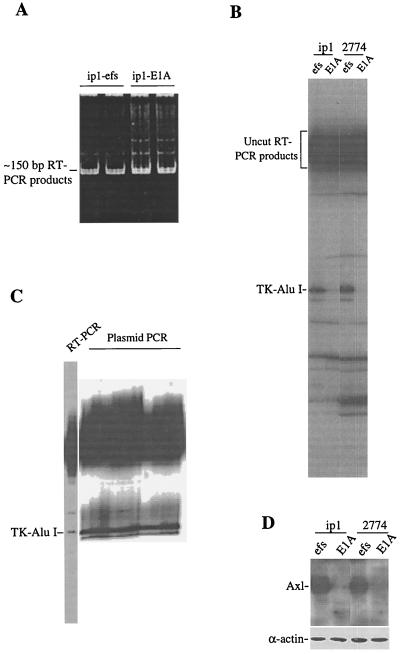
FIG. 1.
(A) The 150-bp RT-PCR products representing the activation loop of the catalytic domains of various tyrosine kinases. RNA was isolated by using the TRIzol reagent. Single-stranded cDNA was synthesized with a GIBCO-BRL kit. Primer 1 was labeled with [γ-32P]ATP and T4 polynucleotide kinase before PCR. The RT-PCR products were analyzed by 8% PAGE. DNA was stained with ethidium bromide at 1 μg/ml. SKOV3.ip1 is a subline of the SKOV3 ovarian cancer cell line; ip1-efs is ip1 cells transfected with an E1A frameshift mutant construct. (B) Tyrosine kinase display assay showing downregulation of a tyrosine kinase, designated TK-Alu I, in E1A-transfected cells. The RT-PCR products shown in panel A were excised and eluted from the gel. The eluted DNAs of equal radioactivity were digested with the restriction enzyme AluI and then fractionated with a 6% DNA sequencing gel. 2774 is the ovarian cancer cell line 2774-C10. (C) Tyrosine kinase display assay of bacterial clones obtained from the tyrosine kinase cDNA library screening. The TK-Alu I probe was PCR labeled and used to screen a cDNA library of tyrosine kinases. The positive clones were picked out, and the plasmid DNA was purified and subjected to PCR as described for panel A. The products of both RT-PCR from ip1-efs cells and plasmid PCR from bacterial clones were digested with AluI and then resolved with a 6% DNA sequencing gel. The TK-Alu I band was seen in all of the plasmid PCR clones. (D) Immunoblotting analysis showing decreased level of the Axl receptor protein in E1A-expressing ip1 and 2774 cells. Western blot analysis was performed as described in Materials and Methods, and the Axl receptor was detected by treating the transblot with an anti-Axl antibody. As a gel loading control, the same blot was reprobed with an anti-α-actin antibody.
Nuclear run-on assay.
The nuclear run-on assay was performed as described previously (27). Briefly, cells were washed with phosphate-buffered saline (PBS) and lysed in ice-cold buffer (10 mM Tris [pH 7.4], 10 mM NaCl, 3 mM MgCl2, 0.5% Nonidet P-40). The nuclei were then pelleted by centrifugation and suspended in buffer A (50 mM Tris [pH 8.3], 5 mM MgCl2, 0.1 mM EDTA, 40% glycerol). The nascent RNA chains were elongated by mixing the nuclear suspension with an equal volume of the reaction buffer (10 mM Tris [pH 8.0]; 5 mM MgCl2; 0.3 M KCl; 5 mM dithiothreitol; 1 mM each ATP, CTP, and GTP; 0.1 mCi [α-32P]UTP). After incubation at 30°C for 30 min, the 32P-labeled RNA was purified with TRIzol (GIBCO-BRL). Samples (5 μg) of plasmids, pcDNA3, pcDNA3-Axl, and pcDNA3–glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were denatured and immobilized on Hybond N membranes. The membranes were prehybridized in a solution of 50% formamide, 5× SSPE (1× SSPE is 0.18 M NaCl, 10 mM NaH2PO4, and 1 mM EDTA), 2× Denhardt’s reagent, 0.5% sodium dodecyl sulfate (SDS), and salmon sperm DNA at 100 μg/ml for 2 h at 42°C. Equal amounts of radioactivity (1.0 × 106 cpm) from the test samples were hybridized with the immobilized DNA at 42°C for 24 h. The membranes were then washed for 60 min with 2× SSC (1× SSC is 0.15 M NaCl plus 0.015 M sodium citrate)–0.1% SDS at 55°C and then for 30 min with 0.2× SSC–0.1% SDS at 55°C and finally exposed to X-ray film at −70°C.
Establishment of stable cell lines.
The axl cDNA was cloned into a pCEP4 vector carrying the hygromycin phosphotransferase gene (hph). Expression of the axl and hph genes was driven by the cytomegalovirus promoter and the thymidine kinase promoter, respectively. The axl gene transfection was carried out by using DOTAP liposomes (24). Briefly, 6 μg of plasmid pCEP4-Axl along with the DOTAP liposome mixture was transfected to 1.0 × 106 ip-E1A cells cultured in a 3-cm-diameter tissue culture dish. Approximately 4 h after transfection, the cells were washed with PBS, cultured in fresh medium for 24 h, and then split 1:20. The cells were then grown in a selection medium containing hygromycin B (Boehringer Mannheim) at 20 μg/ml and neomycin at 500 μg/ml for 3 to 5 weeks, after which individual hygromycin-resistant clones were picked out and expanded to mass culture.
Immunoblotting.
Immunoblotting analyses were performed as described elsewhere (46). Briefly, cell lysates were resolved by SDS-polyacrylamide gel electrophoresis (PAGE) and then transferred to nitrocellulose membranes. The membranes were treated separately with an anti-Axl (Santa Cruz Biotech), an anti-E1A protein (Oncogene Research), or an anti-α-actin (Amersham Life Science) antibody and then incubated with peroxidase-conjugated secondary antibodies and detected by the enhanced-chemiluminescence method (Amersham Life Science). For the analysis of Gas6 expression, supernatants of cultures were subjected first to SDS-PAGE and then to immunoblotting with an anti-Gas6 polyclonal antibody (Santa Cruz Biotech).
[3H]thymidine incorporation assay.
Cells were trypsinized, washed twice with PBS, and then seeded onto 96-well plates at 2,000/well. To the wells was added 10 μCi of [3H]thymidine (Amersham Life Science) at specific times. The cells were then incubated at 37°C for 8 h and then processed by a cell harvester (Cambridge Technology Inc.). Radioactivity was determined with a liquid scintillation counter.
Analysis of [3H]thymidine incorporation after the gas6 cDNA transfection.
The medium used to do the gas6 cDNA transfection and other studies related to Gas6 contained 4 μM menadione sodium bisulfite, a vitamin K analog (Sigma). The gas6 cDNA, whose expression was driven by the cytomegalovirus promoter, was cloned in a pcDNA3 vector. Cells (1.0 × 106) were plated in a 3-cm-diameter tissue culture dish and cultured in drug-free medium for 24 h, and then 4 μg of the vector pcDNA3 or plasmid pcDNA3-Gas6 was transfected into ip1-efs, ip1-E1A-pCEP4, and three independent clones of ip1-E1A-Axl cells by using DOTAP liposomes. Cells were exposed to the DNA-liposome composites for 8 h, washed twice with PBS, and cultured in serum-supplemented medium for 24 h. Cell were then trypsinized, washed twice with PBS, and then seeded onto 96-well plates at 2,000/well. At 24, 48, and 72 h thereafter, cells were subjected to [3H]thymidine incorporation. Alternatively, 24 h after being seeded onto 96-well plates, cells were serum deprived for another 24 h and then subjected to [3H]thymidine incorporation.
Analysis of the phosphorylation state of the Axl receptor.
Cells for this assay were lysed in a buffer containing 20 mM Tris [pH 7.2], 150 mM NaCl, 50 mM NaF, 2 mM Na3VO4, 1 mM phenylmethylsulfonyl fluoride, leupeptin at 25 μg/ml, and aprotinin at 25 μg/ml. The lysates were cleared by centrifugation and immunoprecipitated with 5 μg of anti-Axl polyclonal antibody (Santa Cruz), followed by 20 μl of protein A-agarose (Boehringer Mannheim). The immunocomplexes were washed with lysis buffer, resolved by SDS–10% PAGE, transferred to a nitrocellulose membrane, and then detected with an antiphosphotyrosine antibody (Oncogene Research).
Analysis of apoptosis by flow cytometry.
For analysis of apoptosis by flow cytometry, 1.0 × 106 cells were transfected with gas6 cDNA by using the LPD1 liposome (24). At 24 h after transfection, cells were serum starved for 48 h, harvested by trypsinization, washed twice with PBS, and then fixed in 75% ethanol at 4°C overnight. The fixed cells were washed twice with PBS and suspended in 1 ml of PBS containing 0.5% Tween 20, to which were added 10 μg of RNase and 10 μg of propidium iodide. The propidium iodide-stained cells were analyzed with a FACScan flow cytometer (Becton Dickinson). Apoptotic cells were determined by the percentage of cells in the sub-G1 region.
RESULTS
Identification of the tyrosine kinase Axl that was downregulated by the E1A proteins.
To identify the E1A-regulated tyrosine kinases, we compared the tyrosine kinase expression profiles of cancer cell lines with those of their E1A transfectants by using a novel tyrosine kinase display assay (34), a modification of the tyrosine kinase profile approach (35). Briefly, the entire collection of expressed tyrosine kinases was generated by RT-PCR using degenerate primers corresponding to the highly conserved DFG and DVW motifs. This would yield a relatively homogeneous band of approximately 150 bp for virtually all tyrosine kinases. Different kinases would then be differentiated by digestion with restriction enzymes, based on the characteristic sizes predicted from the GenBank data. To help visualize the restriction products, the 5′ end of the sense primer was radiolabeled with [γ-32P]ATP and the digested products were resolved in a DNA sequencing gel and then autoradiographed. An example of the polyacrylamide gel-resolved and ethidium bromide-stained total RT-PCR products in the range of 150 bp is shown in Fig. 1A. Restriction enzyme digestion of the RT-PCR products would reduce the lengths of certain radiolabeled DNA fragments, which would migrate faster than uncut fragments in a DNA sequencing gel. A tyrosine kinase cDNA fragment, tentatively named TK-AluI, that had been digested with AluI displayed a differential expression pattern in E1A-transfected cells compared with the parental controls (Fig. 1B). This band exhibits a very low radiointensity in both the E1A-expressing ip1 and 2774 ovarian cancer cells compared with the control cells. To confirm that the lower expression of TK-Alu I was due to the expression of E1A, we subjected the breast cancer cell line MDA-MB231 and its E1A stable transfectant (231-E1A) to the same analysis and found downregulation of Axl in 231-E1A cells (data not shown). Thus, it is likely that the mRNA level of the TK-Alu I gene was negatively regulated by E1A. The size of the AluI-digested band predicts that it may be the tyrosine kinase Axl.
To confirm that the TK-Alu I gene encodes Axl but not any fortuitous RT-PCR product, we set out to clone this product. A radioactive probe was prepared by PCR using the TK-Alu I DNA as a template, and that probe was used to screen a tyrosine kinase cDNA library established by ligating the RT-PCR products synthesized from ip1 cells to the TA cloning vector PCR2.1. As expected, the positive clones showed the same TK-Alu I band upon tyrosine kinase display (Fig. 1C). DNA sequencing revealed that TK-Alu I indeed represents the Axl kinase. Once we identified the product of the TK-Alu I gene as Axl, we analyzed Axl protein levels in cancer cell lines and their E1A transfectants. We found that expression of the Axl protein was repressed in E1A transfectants (Fig. 1D). Thus, both protein and mRNA expression findings support the inhibition of Axl expression by E1A.
Repression of axl gene expression by E1A at the transcriptional level.
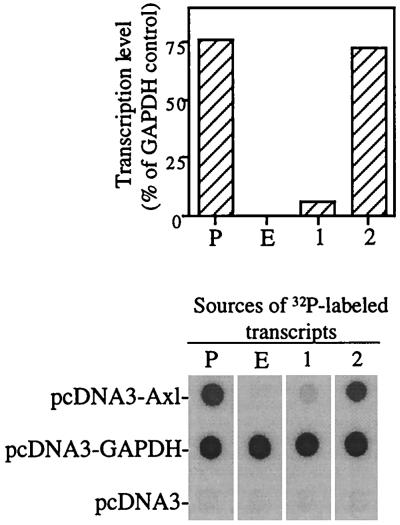
The foregoing results suggest that E1A can negatively regulate the expression of both the axl transcripts and the Axl protein. We next used a nuclear run-on assay to determine whether E1A represses transcription of the axl gene. The radiolabeled transcripts derived from the nuclei of ip1-efs cells hybridized strongly with the axl cDNA, but the radiolabeled transcripts derived from ip1-E1A cells gave no hybridization signal (Fig. 2). Hybridization of the same radiolabeled transcripts to GAPDH cDNA, a housekeeping gene, produced equal signals in the ip1-efs and ip1-E1A cells. These results indicate that ip1-E1A cells produced little or no Axl mRNA. To determine whether this phenomenon was caused by transcriptional repression of the axl gene or by posttranscriptional degradation of axl mRNA in ip1-E1A cells, we elongated mRNA from ip1-efs cells and then added a nuclear suspension of ip1-E1A cells to the products of in vitro transcription. The hybridization result (Fig. 2, lane 2) was the same as that seen in the lane representing ip1-efs. Thus, the nuclear suspension of ip1-E1A cells did not contain factors that accelerated degradation of the axl mRNA. In vitro transcriptional repression was also observed in a mixture of the nuclear suspensions of ip1 cells and ip1-E1A cells (Fig. 2, lane 1), suggesting that some factor in ip1-E1A cells is able to act in trans to suppress axl transcription in ip1-efs cells. Thus, we conclude that the rate of axl gene transcription was greatly inhibited in ip1-E1A cells compared with that in the control ip1-efs cells.
FIG. 2.
Nuclear run-on assay showing transcriptional repression of the axl gene in ip1-E1A cells. 32P-labeled transcripts were prepared from isolated nuclei by in vitro transcription with [α-32P]UTP. Equal amounts of labeled RNA (106 cpm) were hybridized to Hybond N membranes containing the immobilized plasmids pcDNA3-Axl, pcDNA3-GAPDH, and pcDNA3 at 5 μg per blot. P, ip1-efs cells; E, ip1-E1A cells. Lanes: 1, membrane strip hybridized with the in vitro transcription product derived from an equal amount mixture of the nuclear suspensions of ip1-efs and ip1-E1A cells; 2, membrane strip hybridized with the in vitro transcription product derived from the nuclear suspension of ip1-efs cells, to which was added an equal amount of nuclear suspension of ip1-E1A cells after mRNA elongation was completed.
Establishment of Axl-overexpressing ip1-E1A cells.
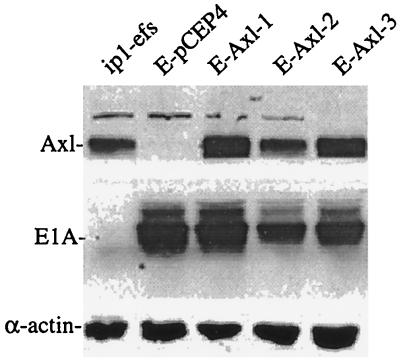
ip1-E1A cells grow more slowly than do the parental ip1 cells (58). To study whether downregulation of the Axl receptor by E1A might be involved in the E1A-mediated reduction of the growth rate of ip1-E1A cells, we established an ip1-E1A cell line that overexpresses Axl (ip1-E1A-Axl). The axl cDNA was cloned into the expression vector pCEP4 that carries the hygromycin phosphotransferase gene. The stable cell lines were established by transfection of the pCEP4-Axl plasmid into ip1-E1A cells and selection with hygromycin B. These ip1-E1A-Axl clones overexpressed the Axl receptor; the expression level in clone 2 was comparable to that of the ip1-efs cells (Fig. 3). To verify that E1A was still present in the stable clones, the same blot was reprobed with an anti-E1A antibody. Expression of α-actin was used as a gel loading control.
FIG. 3.
Immunoblot showing re-expression of the Axl receptor in axl-transfected ip1-E1A cells. axl cDNA was cloned into the pCEP4 vector, and the DNA was transfected as described in Materials and Methods. Three clones of ip1-E1A-Axl (E-Axl-1, E-Axl-2, and E-Axl-3) showed increased expression of Axl compared to that of ip1-E1A-pCEP4 (E-pCEP4) control cells that were transfected with the pCEP4 vector. An equal amount of cell lysate for each cell type was resolved by SDS–10% PAGE. After electrophoresis, proteins were transferred to a nitrocellulose membrane and then probed with anti-Axl, anti-E1A, and anti-α-actin antibodies.
Promotion of mitogenesis by the Gas6-Axl interaction in ip1-E1A-Axl cells.
To determine whether ip1-E1A-Axl cells might recover after growth rate reduction by E1A, we measured mitogenesis by determining [3H]thymidine incorporation. Re-expression of the Axl receptor in ip1-E1A cells (ip1-E1A-Axl) had no significant effect on mitogenesis, compared to ip1-E1A-pCEP4 control cells that were transfected with the vector plasmid (Fig. 4A). Since previous reports had shown that activation of Axl is Gas6 dependent (1, 19, 20), we questioned whether the Axl receptor expressed in ip1-E1A-Axl cells requires the ligand Gas6 for activation.
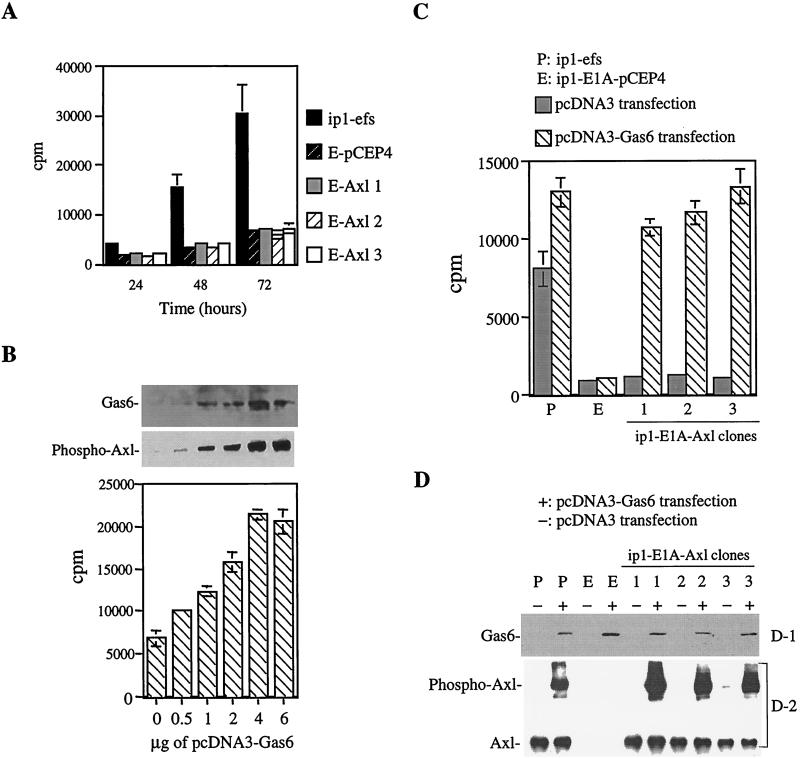
FIG. 4.
Mitogenesis in ip1-efs, ip1-E1A, and ip1-E1A-Axl cells. (A) [3H]thymidine incorporation showing no significant difference in mitogenesis between ip1-E1A-pCEP4 (E-pCEP4) cells and ip1-E1A-Axl clones (E-Axl-1, -2, and -3). Cells were seeded onto 96-well plates, [3H]thymidine was added at 24, 48, and 72 h, and cells were incubated at 37°C for 8 h and then processed by a cell harvester. Each bar is the average of four replicates. (B) Dose-dependent activation of the Axl receptor in ip1-E1A-Axl-2 cells by gas6 cDNA transfection. Various concentrations of pcDNA3-Gas6 plasmid DNA were transfected into 106 ip1-E1A-Axl-2 cells as described in Materials and Methods; 4 μg of plasmid pcDNA3-Gas6 was sufficient to cause maximum phosphorylation of the Axl receptor and plateau incorporation of [3H]thymidine. Phosphorylation of the Axl receptor was analyzed by immunoprecipitation with an anti-Axl antibody, followed by Western blotting with an antiphosphotyrosine antibody. Gas6 expression was determined by immunoblotting of culture supernatants. All of the assays were performed 72 h after transfection and under serum-supplemented conditions. (C) The effect of Gas6 on serum-deprived ip1-E1A-Axl cells. Cells (106) were transfected with 4 μg of pcDNA3-Gas6 or vector pcDNA3 as described for panel B and seeded onto a 96-well plate at 2,000 cells/well 24 h after transfection. After 24 h of culture in serum-supplemented medium to allow the cells to attach to the wells, the cells were serum deprived for another 24 h and then subjected to [3H]thymidine incorporation. Each bar is the average of triplicate samples. (D-1) Immunoblot showing Gas6 expression in pcDNA3-Gas6-transfected cells. Western blot analysis was performed with supernatants of cell cultures as described for panel B. (D-2) Increased phosphorylation of the Axl receptor in Axl-expressing cells transfected with gas6 cDNA. Cells were transfected with pcDNA3-Gas6 or vector plasmid pcDNA3 as described for panel C. At 24 h after transfection, cells were serum deprived for 24 h and then lysed, immunoprecipitated with anti-Axl antibody, and subjected to Western blotting with an antiphosphotyrosine antibody. As a control, the same blot was reprobed with an anti-Axl antibody.
To determine whether Gas6 could promote mitogenesis in ip1-E1A-Axl clones, cells were transfected with either vector pcDNA3 or plasmid pcDNA3-Gas6 and [3H]thymidine incorporation was performed in the presence of serum at specific times thereafter. Gas6 produced a dose-dependent activation of the Axl receptor in ip1-E1A-Axl cells (Fig. 4B) with transfection of 4 μg of plasmid pcDNA-Gas6 to 106 ip1-E1A-Axl-2 cells giving the maximum phosphorylation of the Axl receptor and a plateau incorporation of [3H]thymidine. Gas6 expression in culture supernatants was confirmed by Western blotting. These results indicate that the transfection assay we established can produce enough secreted Gas6 to induce Axl-dependent mitogenesis.
Using the above-described assay, we found that transiently expressed Gas6 had an increased mitogenic effect on ip1-E1A-Axl clones in the absence of serum compared with the same clones without Gas6 stimulation (Fig. 4C). Expression of Gas6 is shown in Fig. 4D-1. This protein had a lesser impact on mitogenesis in ip1-efs cells (1.6-fold increase) than on that in ip1-E1A-Axl cells (10-fold increase). For ip1-E1A-Axl-2 cells, Gas6 induced a 10-fold increase in mitogenesis in the absence of serum (Fig. 4C) but only a 3-fold increase in the presence of serum (4-μg lane in Fig. 4B), suggesting that serum contains other stimulating factors that cover the effect of Gas6.
To confirm the effect of the Gas6-Axl interaction, we analyzed the intrinsic phosphorylation of the Axl receptor in the presence or absence of Gas6 stimulation. Cells were transfected with either the pcDNA3-Gas6 plasmid or the control vector, serum deprived for 24 h, immunoprecipitated with an anti-Axl antibody, and Western blotted with an antiphosphotyrosine antibody. The increased tyrosine phosphorylation of the Axl receptor in the Gas6-stimulated ip1-E1A-Axl cells (Fig. 4D-2) correlates well with the enhanced mitogenic effect in ip1-E1A-Axl cells (Fig. 4C). The same blot was reprobed with an anti-Axl antibody as a control.
Protection of E1A-transfected cells from serum deprivation-induced apoptosis by the Gas6-Axl interaction.
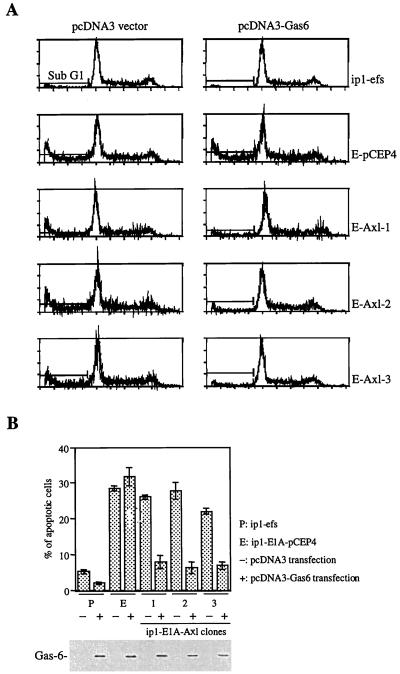
E1A-transfected cells are more sensitive to serum deprivation-induced apoptosis than are parental cells (11, 33). We found that Gas6 can induce mitogenesis in ip1-E1A-Axl cells in the absence of serum (Fig. 4C). To examine whether the Gas6-Axl interaction could also protect ip1-E1A-Axl cells from apoptosis triggered by serum deprivation, we transfected cells with plasmid pcDNA3-Gas6, deprived those cells of serum for 48 h, and then processed them for fluorescence-activated cell sorter (FACS) analysis. Fewer Gas6-stimulated ip1-E1A-Axl cells than unstimulated cells were in the sub-G1 region (Fig. 5A and B). Gas6 expression is shown in the lower part of Fig. 5B. The ip1-E1A-pCEP4 cells were sensitive to serum deprivation despite the presence of Gas6; the ip1-efs cells were less sensitive to serum withdrawal under the same conditions. These results indicate that Gas6 can protect E1A transfectants from serum deprivation-induced apoptosis if Axl is re-expressed.
FIG. 5.
Protection of Axl-expressing cells from serum deprivation-induced apoptosis by Gas6. (A and B) Decreased apoptosis in ip1-E1A-Axl cells by transiently expressed Gas6. Cells were transfected with gas6 cDNA or control plasmid pcDNA3 as described for Fig. 4C. At 24 h after transfection, cells were serum deprived for 48 h and then subjected to FACS analysis. The apoptotic cells were determined by measuring the percentage of cells in the sub-G1 region (A). Each bar in panel B is the average of triplicate samples. Expression of Gas6 in cells transfected with gas6 cDNA is shown at the bottom of panel B. Supernatants from cells used for FACS analysis were subjected to SDS–10% PAGE. Western blot analysis was performed with an anti-Gas6 polyclonal antibody. Abbreviations for cell types are the same as those in Fig. 3 and 4C and D.
DISCUSSION
E1A has been shown to be associated with multiple antitumor activities, including transcriptional repression of the Her-2/neu gene (52, 57), suppression of transformation (12, 15, 16, 28, 55), inhibition of metastasis (17, 28, 53–55), and induction of apoptosis (9, 11, 33, 45). To further understand E1A-mediated tumor suppression, we focused on the tyrosine kinases that are regulated by E1A because tyrosine kinases usually play a pivotal role in the signal pathways that cause cellular transformation. In the current study, we used the tyrosine kinase display and nuclear run-on assays to confirm that the expression of the transforming receptor tyrosine kinase Axl is transcriptionally suppressed by E1A. Our experimental results indicate that the Gas6-Axl interaction counteracts E1A-mediated cell growth suppression and proapoptotic activity.
Multiple molecular mechanisms may account for the tumor- and metastasis-suppressing functions of E1A in different cancer cell types. The tumor-suppressing function has been explained, at least in part, as resulting from the induction of apoptosis through p53-dependent or p53-independent mechanisms (9, 45). E1A may also suppress tumor growth by modulating the response of tumor cells to immune cells, since this protein can sensitize transfected cells to the cytotoxic effects of tumor necrosis factor (TNF) (6, 38) and make target cells susceptible to NK cells and activated macrophages (7). In addition, E1A can abrogate NF-κB activation, resulting in susceptibility of cells to apoptotic stimuli such as TNF and γ irradiation (38, 39). As for the metastasis-suppressing function of E1A, one known mechanism is the transcriptional repression of various proteases that are important for tumor invasion and metastasis, including type IV collagenase (17, 18), interstitial collagenase, urokinase (17), and stromelysin (25, 31). In summary, E1A suppresses tumor growth and metastasis through cumulative changes in cellular gene expression, and many unknown mechanisms remain to be discovered.
Gas6 induced serum-starved NIH 3T3 cells to enter the cell cycle (19, 20); the signaling was shown to be transmitted through the stimulation of Axl tyrosine kinase with subsequent activation of mitogen-activated protein kinase and phosphatidylinositol 3-kinase (19, 20). Adding Gas6 to serum-starved NIH 3T3 cells also prevented cell death induced by complete serum removal and TNF addition, indicating that Gas6 acts as a survival factor for growth-arrested cells (1, 19, 20). In a study of fibroblasts from axl knockout mice, the absence of the Axl receptor resulted in higher levels of serum deprivation-induced apoptosis that could not be rescued by the addition of Gas6 (1). Instead of studying normal fibroblasts, we established an ovarian cancer model in which parental ip1 cells, unlike NIH 3T3 cells, were not sensitive to serum deprivation. In this model, downregulation of the Axl receptor by E1A rendered the E1A transfectant ip1-E1A susceptible to serum deprivation-induced apoptosis that was prevented by the Gas6-Axl interaction. In short, abrogation of Gas6-Axl signaling by E1A is involved in E1A-mediated suppression of cell growth and susceptibility to serum deprivation.
The mildly increased rate of [3H]thymidine incorporation in Gas6-stimulated ip1-efs cells (Fig. 4C) is consistent with previous reports that Gas6 is a weaker mitogen than basic fibroblast growth factor and serum (1, 19). In the mitogenesis and apoptosis studies (Fig. 4C and 5), we found that ip1-efs cells were less responsive to Gas6 stimulation than were ip1-E1A-Axl cells, although both expressed Axl. Conversely, the mitogenic effect and resistance to serum withdrawal-induced apoptosis in ip1-E1A-Axl cells were dependent on Gas6 stimulation (Fig. 4C and 5), implicating the role of downregulation of Axl in E1A-mediated tumor suppression and E1A’s other activities that render ip1-E1A-Axl cells more dependent than ip1-efs cells on Gas6 during serum depletion. One of these E1A activities may be downregulation of the receptor tyrosine kinase Her-2/Neu (52, 55, 57). Re-expression of Axl did not change the repressed expression of Her-2/Neu in ip1-E1A cells (data not shown). Overexpression of Her-2/Neu in ip1-efs cells (58) may be the reason why ip1-efs cells were less dependent on Gas6 stimulation (Fig. 4C) and more resistant to serum withdrawal-induced apoptosis (Fig. 5) than were ip1-E1A-Axl cells.
Expression of the activated axl gene in NIH 3T3 (AF6295) cells can result in cellular transformation (30). However, re-expression of the Axl receptor in ip1-E1A cells (ip1-E1A-Axl) had no significant effect on mitogenesis as analyzed by [3H]thymidine incorporation, compared to ip1-E1A-pCEP4 control cells that were transfected with the vector plasmid (Fig. 4A). This might be because the level of the Axl receptor in ip1-E1A-Axl clones was not as high as that in the AF6295 cells described by O’Bryan et al. (30). The expression of Axl in AF6295 cells is at least 10-fold more abundant than that in ip1-efs cells (data not shown). The mitogenesis study indicates that the receptor expressed in ip1-E1A-Axl cells requires the Axl ligand Gas6 for activation (Fig. 4B and D-2). Although Gas6 was able to stimulate mitogenesis in ip1-E1A-Axl cells, endogenous Gas6 expression could not be detected in the supernatants of parental ip1 cells or ip1-E1A cells (Fig. 4D-1 and 5B). This finding suggests that repression of the axl gene by E1A is not the main mechanism by which growth is suppressed in cultured ip1 cells. However, Gas6 secretion is ubiquitous in the human body (26), so Axl-mediated survival and mitogenic effects may be more important in whole organisms than in cultured cells. It should be mentioned that Axl is overexpressed in approximately 25% of primary breast cancers (4). Given that a tumor cell line can secrete detectable amounts of Gas6 protein, Gas6-Axl signaling may play a supportive role in preventing apoptosis induced by serum deprivation or TNF. On the other hand, Gas6 also can act as a chemoattractant and is involved in cell migration (14); several metastatic cell types have increased expression of the Axl receptor (7, 32, 50). Thus, suppression of the Axl receptor by E1A may partly explain E1A’s metastasis-suppressing effect.
In summary, the known characteristics of the Axl receptor and E1A are consistent with our previous findings that E1A functions as a tumor suppressor. Further investigations are required to determine whether negative regulation of Axl by E1A is also involved in the decreased tumorigenicity and decreased metastatic potential observed in ip1-E1A cells. However, the present findings identify a second transforming receptor tyrosine kinase, Axl, that is repressed by E1A.
ACKNOWLEDGMENTS
This work was supported by NIH grants R01-CA58880 and R01-CA77858 (to M.-C.H.).
REFERENCES
- 1.Bellosta P, Zhang Q, Goff S P, Basilico C. Signaling through the ARK tyrosine kinase receptor protects from apoptosis in the absence of growth stimulation. Oncogene. 1997;15:2387–2397. doi: 10.1038/sj.onc.1201419. [DOI] [PubMed] [Google Scholar]
- 2.Berk A J. Adenovirus promoters and E1A transactivation. Annu Rev Genet. 1986;20:45–79. doi: 10.1146/annurev.ge.20.120186.000401. [DOI] [PubMed] [Google Scholar]
- 3.Borrelli E, Hen R, Chambon P. Adenovirus-2 E1A products repress enhancer-induced stimulation of transcription. Nature. 1984;312:608–612. doi: 10.1038/312608a0. [DOI] [PubMed] [Google Scholar]
- 4.Burcher A, Attar E C, McCloskey P, Fridell Y-C, Liu E T. Determinants for transformation induced by the Axl receptor tyrosine kinase. Oncogene. 1998;16:3177–3187. doi: 10.1038/sj.onc.1201865. [DOI] [PubMed] [Google Scholar]
- 5.Byrd P J, Grand R J A, Gallimore P H. Differential transformation of primary human embryo retinal cells by adenovirus E1A regions and combinations of E1A + ras. Oncogene. 1988;2:477–484. [PubMed] [Google Scholar]
- 6.Chen M-J, Holskin B, Strickler J, Gorniak J, Clark M A, Johnson P J, Mitcho M, Shalloway D. Induction by E1A oncogene expression of cellular susceptibility to lysis by TNF. Nature. 1987;330:581–584. doi: 10.1038/330581a0. [DOI] [PubMed] [Google Scholar]
- 7.Cook J L, May D L, Wilson B A, Holskin B, Chen M-J, Shalloway D, Walker T A. Role of tumor necrosis factor-α in E1A oncogene-induced susceptibility of neoplastic cells to lysis by natural killer cells and activated macrophages. J Immunol. 1989;142:4527–4534. [PubMed] [Google Scholar]
- 8.Craven R J, Xu L, Weiner T M, Fridell Y-W, Dent G A, Srivastava S, Varnum B, Liu E T, Cance W G. Receptor tyrosine kinases expressed in metastatic colon cancer. Int J Cancer. 1995;60:791–797. doi: 10.1002/ijc.2910600611. [DOI] [PubMed] [Google Scholar]
- 9.Debbas M, White E. Wild-type p53 mediates apoptosis by E1A, which is inhibited by E1B. Genes Dev. 1993;7:546–554. doi: 10.1101/gad.7.4.546. [DOI] [PubMed] [Google Scholar]
- 10.DeGroot R, Foulkes N, Mulder M, Kruijer W, Sassone-Corsi P. Positive regulation of jun/Ap-1 by E1A. Mol Cell Biol. 1991;11:192–201. doi: 10.1128/mcb.11.1.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Deng J, Xia W, Hung M-C. Adenovirus 5 E1A-mediated tumor suppression associated with E1A-mediated apoptosis in vivo. Oncogene. 1998;17:2167–2175. doi: 10.1038/sj.onc.1202148. [DOI] [PubMed] [Google Scholar]
- 12.Douglas J L, Gopalakrishnan S, Quinlan M P. Modulation of transformation of primary epithelial cells by the second exon of the Ad5 E1A 12S gene. Oncogene. 1991;6:2093–2103. [PubMed] [Google Scholar]
- 13.Fantl W J, Johnson D E, Williams L T. Signaling by receptor tyrosine kinases. Annu Rev Biochem. 1993;62:453–481. doi: 10.1146/annurev.bi.62.070193.002321. [DOI] [PubMed] [Google Scholar]
- 14.Fridell Y-W C, Villa J, Jr, Attar E C, Liu E T. Gas6 induces Axl-mediated chemotaxis of vascular smooth muscle cells. J Biol Chem. 1998;273:7123–7126. doi: 10.1074/jbc.273.12.7123. [DOI] [PubMed] [Google Scholar]
- 15.Frisch S M. Antioncogenic effect of adenovirus E1A in human tumor cells. Proc Natl Acad Sci USA. 1991;88:9077–9081. doi: 10.1073/pnas.88.20.9077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Frisch S M, Dolter K E. Adenovirus E1A-mediated tumor suppression by a c-erbB2/neu-independent mechanism. Cancer Res. 1995;55:5551–5555. [PubMed] [Google Scholar]
- 17.Frisch S M, Reich R, Collier I E, Genrich L T, Martin G, Goldberg G I. Adenovirus E1A represses protease gene expression and inhibits metastasis of human tumor cells. Oncogene. 1990;5:75–83. [PubMed] [Google Scholar]
- 18.Garbisa S, Pozzatti R, Muschel R J, Saffiotti U, Ballin M, Goldfarb R H, Khoury G, Liotta L A. Secretion of type IV collagenolytic protease and metastatic phenotype: induction by transfection with c-Ha-ras but not c-Ha-ras plus Ad2-E1a. Cancer Res. 1987;47:1523–1528. [PubMed] [Google Scholar]
- 19.Goruppi S, Ruaro E, Schneider C. Gas6, the ligand of Axl tyrosine kinase receptor, has mitogenic and survival activities for serum starved NIH3T3 fibroblasts. Oncogene. 1996;12:471–480. [PubMed] [Google Scholar]
- 20.Goruppi S, Ruaro E, Varnum B, Schneider C. Requirement of phosphatidylinositol 3-kinase-dependent pathway and Src for Gas6-Axl mitogenic and survival activities in NIH 3T3 fibroblasts. Mol Cell Biol. 1997;17:4442–4453. doi: 10.1128/mcb.17.8.4442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hen R, Borrelli E, Chambon P. Repression of the immunoglobulin heavy chain enhancer by the adenovirus-2 E1A products. Science. 1985;230:1391–1394. doi: 10.1126/science.2999984. [DOI] [PubMed] [Google Scholar]
- 22.Kao H-T, Nevins J R. Transcriptional activation and subsequent control of the human heat shock gene during adenoviral infection. Mol Cell Biol. 1984;4:2792–2810. doi: 10.1128/mcb.3.11.2058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Land H, Parada L F, Weinberg R A. Tumorigenic conversion of primary embryo fibroblasts requires at least two cooperating oncogenes. Nature. 1983;304:596–602. doi: 10.1038/304596a0. [DOI] [PubMed] [Google Scholar]
- 24.Li S, Rizzo M A, Bhattacharya S, Huang L. Characterization of cationic lipid-protamine-DNA (LPD) complexes for intravenous gene delivery. Gene Ther. 1998;5:930–937. doi: 10.1038/sj.gt.3300683. [DOI] [PubMed] [Google Scholar]
- 25.Linder S, Popowicz P, Svensson C, Marshall H, Bondesson M, Akusjarvi G. Enhanced invasive properties of rat embryo fibroblasts transformed by adenovirus E1A mutants with deletions in the carboxyl-terminal exon. Oncogene. 1992;7:439–443. [PubMed] [Google Scholar]
- 26.Manfioletti G, Brancolini C, Avanzi G, Schneider C. The protein encoded by a growth arrest-specific gene (gas6) is a new member of the vitamin K-dependent proteins related to protein S, a negative coregulator in the blood coagulation cascade. Mol Cell Biol. 1993;13:4976–4985. doi: 10.1128/mcb.13.8.4976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Miyazawa K, Mori A, Miyata H, Akahane M, Ajisawa Y, Okudaira H. Regulation of interleukin-1b-induced interleukin-6 gene expression in human fibroblast-synoviocytes by p38 mitogen-activated protein kinase. J Biol Chem. 1998;273:24832–24838. doi: 10.1074/jbc.273.38.24832. [DOI] [PubMed] [Google Scholar]
- 27a.Montell C, Courtois G, Eng C, Berk A J. Complete transformation by adenovirus 2 requires both E1A proteins. Cell. 1984;36:951–961. doi: 10.1016/0092-8674(84)90045-x. [DOI] [PubMed] [Google Scholar]
- 28.Mymryk J S. Tumor suppressive properties of the adenovirus 5 E1A oncogene. Oncogene. 1996;13:1581–1589. [PubMed] [Google Scholar]
- 29.Nakano T, Ishimoto Y, Kishino J, Umeda M, Inoue K, Nagata K, Ohashi K, Mizuno K, Arita H. Cell adhesion to phosphatidylserine mediated by a product of growth arrest-specific gene 6. J Biol Chem. 1997;272:29411–29414. doi: 10.1074/jbc.272.47.29411. [DOI] [PubMed] [Google Scholar]
- 30.O’Bryan J P, Frye R A, Cogswell P C, Neubauer A, Kitch B, Prokop C, Espinosa III R, LeBeau M M, Earp H S, Liu E T. axl, a transforming gene isolated from primary human myeloid leukemia cells, encodes a novel receptor tyrosine kinase. Mol Cell Biol. 1991;11:5016–5031. doi: 10.1128/mcb.11.10.5016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Offringa R, Smits A M, Houweling A, Bos J L, van der Eb A J. Similar effects of adenovirus E1A and glucocorticoid hormones on the expression of the metalloprotease stromelysin. Nucleic Acids Res. 1988;16:10973–10984. doi: 10.1093/nar/16.23.10973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Quong R Y Y, Bickford S T, Ing Y L, Terman B, Herlyn M, Lassam N J. Protein kinases in normal and transformed melanocytes. Melanoma Res. 1994;4:313–319. doi: 10.1097/00008390-199410000-00008. [DOI] [PubMed] [Google Scholar]
- 33.Rao L, Debbas M, Sabbatini P, Hockenbery D, Korsmeyer S, White E. The adenovirus E1A proteins induce apoptosis, which is inhibited by the E1B 19-kDa and Bcl-2 proteins. Proc Natl Acad Sci USA. 1992;89:7742–7746. doi: 10.1073/pnas.89.16.7742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Robinson D, Chen H-C, Yustein J T, He F, Lin W-C, Hayman M J, Kung H-J. Tyrosine kinase expression profiles of chicken erythro-progenitor cells and oncogene-transformed erythroblasts. J Biomed Sci. 1998;5:93–100. doi: 10.1007/BF02258362. [DOI] [PubMed] [Google Scholar]
- 35.Robinson D, Le F, Pretlow T, Kung H-J. A tyrosine kinase profile of prostate carcinoma. Proc Natl Acad Sci USA. 1996;93:5958–5962. doi: 10.1073/pnas.93.12.5958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ruley H E. Adenovirus early region 1A enables viral and cellular transforming genes to transform primary cells in culture. Nature. 1983;304:602–606. doi: 10.1038/304602a0. [DOI] [PubMed] [Google Scholar]
- 37.Sassone-Corsi P, Borrelli E. Promoter transactivation of protooncogenes c-fos and c-myc, but not c-Ha-ras, by products of adenovirus early region 1A. Proc Natl Acad Sci USA. 1987;84:6430–6433. doi: 10.1073/pnas.84.18.6430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shao, R., M. C.-T. Hu, B. P. Zhou, S.-Y. Lin, P. J. Chiao, R. H. von Lindern, B. Spohn, and M.-C. Hung. E1A sensitizes cells to tumor necrosis factor-induced apoptosis through inhibition of IκB kinases and nuclear factor κB activities. J. Biol. Chem., in press. [DOI] [PubMed]
- 39.Shao R, Karunagaran D, Zhou B P, Li K, Lo S-S, Deng J, Chiao P, Hung M-C. Inhibition of nuclear factor-κB activity is involved in E1A-mediated sensitization of radiation-induced apoptosis. J Biol Chem. 1997;272:32739–32742. doi: 10.1074/jbc.272.52.32739. [DOI] [PubMed] [Google Scholar]
- 40.Shenk T. Adenoviridae: the viruses and their replication. In: Fields B N, Knipe D M, Howley P M, editors. Virology. Philadelphia, Pa: Lippincott-Raven Publishers; 1996. pp. 2111–2148. [Google Scholar]
- 41.Sogawa K, Handa H, Fujisawa-Sehara A, Hiromasa T, Yamane M, Fujii-Kuriyama Y. Repression of cytochrome P450c gene expression by cotransfection with adenovirus E1A DNA. Eur J Biochem. 1989;181:539–544. doi: 10.1111/j.1432-1033.1989.tb14757.x. [DOI] [PubMed] [Google Scholar]
- 42.Stein R, Ziff E. Hela cell β-tubulin gene transcription is stimulated by adenovirus 5 in parallel with viral early genes in an E1A-dependent mechanism. Mol Cell Biol. 1984;4:2792–2801. doi: 10.1128/mcb.4.12.2792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Stein R, Ziff E B. Repression of insulin gene expression by adenovirus type 5 E1A proteins. Mol Cell Biol. 1987;7:1164–1170. doi: 10.1128/mcb.7.3.1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Stitt T N, Conn G, Gore M, Lai C, Bruno J, Radziejewski C, Mattsson K, Fisher J, Gies D R, Jones P F, Masiakowski P, Ryan T E, Tobkes N J, Chen D H, DiStefano P S, Long G L, Basillico C, Goldfarb M P, Lemke G, Glass D J, Yancopoulos G D. The anticoagulation factor protein S and its relative, GAS6, are ligands for the Tyro 3/Axl family of receptor tyrosine kinase. Cell. 1995;80:661–670. doi: 10.1016/0092-8674(95)90520-0. [DOI] [PubMed] [Google Scholar]
- 45.Teodoro J G, Shore G C, Branton P E. Adenovirus E1A proteins induce apoptosis by both p53-dependent and p53-independent mechanisms. Oncogene. 1995;11:467–474. [PubMed] [Google Scholar]
- 46.Towbin H, Staehelin T, Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA. 1979;76:4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.van der Geer P, Hunter T, Lindberg R A. Receptor protein-tyrosine kinases and their signal transduction pathways. Annu Rev Cell Biol. 1994;10:251–337. doi: 10.1146/annurev.cb.10.110194.001343. [DOI] [PubMed] [Google Scholar]
- 48.Varnum B C, Young C, Elliott G, Garcia A, Bartley T D, Fridell Y-W, Hunt R W, Trail G, Clogston C, Toso R J, Yanaglhara D, Bennett L, Sylber M, Merewether L A, Tseng A, Escobar E, Liu E T, Yamane H K. Axl receptor tyrosine kinase stimulated by the vitamin K-dependent protein encoded by growth-arrest-specific gene 6. Nature. 1995;373:623–626. doi: 10.1038/373623a0. [DOI] [PubMed] [Google Scholar]
- 49.Velich A, Kern F G, Basilico C, Ziff E B. Adenovirus E1A proteins repress expression from polyomavirus early and late promoters. Mol Cell Biol. 1987;7:1164–1170. doi: 10.1128/mcb.6.11.4019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Velich A, Ziff E B. Adenovirus E1A proteins repress transcription from the SV40 early promoter. Cell. 1985;40:705–716. doi: 10.1016/0092-8674(85)90219-3. [DOI] [PubMed] [Google Scholar]
- 51.Weiner T M, Liu E T, Craven R J, Cance W G. Expression of growth factor receptors, the focal adhesion kinase, and other tyrosine kinases in human soft tissue tumors. Ann Surg Oncol. 1994;1:18–27. doi: 10.1007/BF02303537. [DOI] [PubMed] [Google Scholar]
- 52.Yan D-H, Chang L-S, Hung M-C. Repressed expression of the HER-2/c-erbB-2 proto-oncogene by the adenovirus E1A gene products. Oncogene. 1991;6:343–345. [PubMed] [Google Scholar]
- 53.Yu D, Hamada J-I, Zhang H, Nicolson G L, Hung M-C. Mechanisms of neu oncogene induced metastasis and abrogation of metastatic properties by the adenovirus 5 E1A gene products. Oncogene. 1992;7:2263–2270. [PubMed] [Google Scholar]
- 54.Yu D, Martin A, Xia W, Sorgi F, Huang L, Hung M-C. Liposome-mediated in vivo E1A gene transfer suppressed dissemination of ovarian cancer cells that overexpress Her-2/neu. Oncogene. 1995;11:1383–1388. [PubMed] [Google Scholar]
- 55.Yu D, Scorsone K, Hung M-C. Adenovirus type 5 E1A gene products act as transformation suppressors of the neu oncogene. Mol Cell Biol. 1991;11:1745–1750. doi: 10.1128/mcb.11.3.1745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yu D, Shi D, Scanlon M, Hung M-C. Reexpression of neu-encoded oncoprotein counteracts the tumor-suppressing but not the metastasis-suppressing function of E1A. Cancer Res. 1993;53:5784–5790. [PubMed] [Google Scholar]
- 57.Yu D, Suen T-C, Yan D-H, Chang L S, Hung M-C. Transcriptional repression of the neu protooncogene by the adenovirus 5 E1A products. Proc Natl Acad Sci USA. 1990;87:4499–4503. doi: 10.1073/pnas.87.12.4499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yu D, Wolf J K, Scanlon M, Price J E, Hung M-C. Enhanced c-erbB-2/neu expression in human ovarian cancer cells correlates with more severe malignancy that can be suppressed by E1A. Cancer Res. 1993;53:891–898. [PubMed] [Google Scholar]