Abstract
Group B Streptococcus (GBS) or Streptococcus agalactiae is an uncommon causative agent of urinary tract infection (UTI). We present a series of seven cases of UTI due to GBS from a tertiary care hospital of Eastern India, highlighting its emerging role in a hitherto less commonly described clinical entity.
Keywords: Group B Streptococcus , Streptococcus agalactiae, Urinary tract infection
An increased awareness is needed for accurate identification of urinary tract infection due to Group B Streptococcus (GBS). All catalase‐negative, bile‐esculin negative, gram‐positive cocci obtained on cystine lactose electrolyte deficient medium should be sub‐cultured to a blood agar medium to examine for specific hemolytic property enabling identification of GBS.

1. INTRODUCTION
Urinary tract infection (UTI) is one of the most common reasons of healthcare consultations in adults, accounting for nearly 8 million office and 1.7 million emergency department visits, resulting in 350,000 hospitalizations per year along with considerable associate costs.1, 2 The condition is more prevalent in females with nearly 50%–60% adult women having at least one episode of UTI in their lifetime.1, 2 Common etiological agents include the uropathogenic Escherichia coli, followed by Klebsiella species and Enterococcus faecalis or Proteus mirabilis in majority of patient cohorts studied.1, 3, 4 Streptococcus agalactiae or Lancefield group B Streptococcus (GBS), a gram‐positive ß‐hemolytic chain‐forming coccus, is an uncommon causative agent estimated to cause approximately 1%–2% of all monomicrobial UTIs.1, 5 However, among elderly populations with UTI, GBS may be involved in as many as 39% of nursing home residents over 70 years of age.5, 6 UTI caused by GBS are also common in pregnant, diabetic, and immunocompromised individuals, as well as those with pre‐existing urologic abnormalities, wherein there is a higher risk of ascending pyelonephritis with potential to progress to bacteremia and/or urosepsis.5, 7, 8, 9 At times, other severe complications like massive abdominopelvic abscess10 and acute paraspinal myositis11 have also been described in diabetic patients secondary to cystitis due to GBS.
An extensive and rigorous Pubmed literature search for “Group B Streptococcus and India,” “S. agalactiae and India,” “beta‐hemolytic streptococci and India,” and “urinary tract infections and India” revealed a mention of UTI due to GBS only on a few instances, that is, of a single case in an adult diabetic male patient12 and in pregnant women.8, 13 Here, we aim to share our experience on cases of UTI due to GBS at our institute for academic interest and to increase awareness regarding this uncommon uropathogen.
2. METHODS
We report a series of seven patients with culture‐proven UTI due to GBS, who presented to the outpatient department of our institute, a tertiary care referral center in Eastern India, with various symptoms of cystitis that included dysuria, increased urinary frequency, urgency, fever >38℃, flank pain, and/or lumbar tenderness. The available medical records/charts and microbiology requisition forms of these patients were reviewed for relevant clinical details and results of microbiological investigations.
3. RESULTS AND DISCUSSION
All the 7 patients with GBS UTI were sexually active, married non‐pregnant adult females in the reproductive age group, with a median age of 26 years (range 19–52 years) (Table 1). The duration of symptoms ranged from 7 days to one month, with two patients (28.8%) giving a history of intermittent recurrence of symptoms. Three patients (42.8%) had associated co‐morbidities comprising of diabetes (two patients) and uterine prolapse (one patient).
TABLE 1.
Demographic, clinical, and microbiological features of urinary tract infection due to group B Streptococcus
| Characteristics | Case 1 | Case 2 | Case 3 | Case 4 | Case 5 | Case 6 | Case 7 |
|---|---|---|---|---|---|---|---|
| Age (in years) | 23 | 48 | 19 | 40 | 25 | 26 | 52 |
| Sex | Female | Female | Female | Female | Female | Female | Female |
| Presenting complaints | Fever, dysuria | Burning micturition with fever and pain at urethral region | Burning micturition frequency | Burning micturition frequency, urgency | Burning micturition frequency | Burning micturition frequency | Burning micturition, flank pain |
| Duration of present symptoms | One month | 7 days | 15 days | 20 days | 7 days | 10 days | 8 days |
| History of recurrence of symptoms | No | 4–5 times in last 2 years | No | 1–2 times in last 6 months | No | No | No |
| Pregnancy status | Non‐pregnant | Non‐pregnant | Non‐pregnant | Non‐pregnant | Non‐pregnant | Non‐pregnant | Non‐pregnant |
| Associated co‐morbidities | ‐ | Diabetes | ‐ | Uterine prolapsea | ‐ | ‐ | Diabetes |
| Antimicrobial susceptibility | |||||||
| Penicillin (10 U) | S | S | S | S | S | S | S |
| Ampicillin (10 µg) | S | S | S | S | S | S | S |
| Ceftriaxone (30 µg) | S | S | S | S | S | S | S |
| Levofloxacin (5 µg) | S | R | R | S | R | R | S |
| Chloramphenicol (30 µg) | S | S | S | S | S | S | S |
| Tetracycline (30 µg) | R | R | S | S | S | S | R |
| Erythromycin (15 µg) | S | S | S | S | S | S | S |
| Clindamycin (2 µg) | S | S | S | S | S | S | S |
| Vancomycin (30 µg) | S | S | S | S | S | S | S |
| Linezolid (30 µg) | S | S | S | S | S | S | S |
| Treatment advised | Oral ampicillin | Oral ampicillin | Oral ampicillin | Oral ampicillin | Oral ampicillin | Oral ampicillin | Oral ampicillin |
| Follow‐up | Lost to follow‐up | No recurrence | No recurrence | Lost to follow‐up | No recurrence | Lost to follow‐up | No recurrence |
Abbreviations: R, resistant; S, susceptible.
First degree utero‐vaginal descent with cystocele and rectocele.
Culture of clean‐catch midstream urine specimens of all the seven patients (two patients, Case 1 and Case 2, being repeat positive for specimens obtained 2–3 days apart) after overnight incubation, yielded pure growth of GBS observed as tiny, yellow, opaque colonies with a count of ≥104 colony‐forming units per milliliter (ml) of sample on cystine lactose electrolyte deficient (CLED) agar plate (Figure 1A). Gram stain of the colonies revealed gram‐positive cocci in chains with negative reactions for catalase and bile esculin. The organisms were beta‐hemolytic, bacitracin resistant, Christie‐Atkins Munch‐Petersen (CAMP) test positive on sheep blood agar plate (Figure 1B and 1C) and were able to agglutinate with GBS‐specific antisera (HiStrep Latex Test Kit; HiMedia). Further confirmation of the isolates was achieved by the VITEK‐ 2 automated identification system (bioMérieux) using gram‐positive (GP) identification cards with 98%–99% probability. Antimicrobial susceptibility testing performed by the modified Kirby Bauer disk diffusion method as per Clinical and Laboratory Standards Institute14 guidelines revealed that all the seven (100%) GBS isolates were uniformly susceptible to penicillin, ampicillin, ceftriaxone, chloramphenicol, erythromycin, clindamycin, vancomycin, and linezolid, while 4 (57.1%) and 3 (42.8%) isolates were resistant to levofloxacin and tetracycline, respectively (Figure 1D) (Table 1). Susceptibility testing to daptomycin performed by Etest (HiMedia, Mumbai, India) showed all isolates to be susceptible to daptomycin (minimum inhibitory concentration range 0.19 −0.38 µg/ml).14 All the patients were advised oral ampicillin for five days. Repeat urine cultures after 7–14 days of therapy in four patients were sterile indicating resolution of infection, three patients were lost to follow‐up.
FIGURE 1.
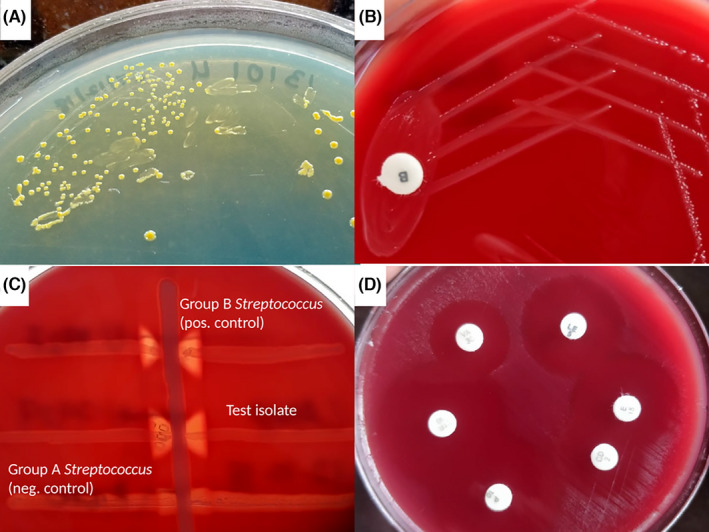
Group B Streptococcus isolate showing (A) tiny, yellow, opaque colonies on CLED agar, (B) bacitracin resistant beta‐hemolytic colonies on sheep blood agar, (C) positive CAMP test, and (D) antimicrobial sensitivity testing indicating susceptibility to various antibiotics by disc‐diffusion test
Commonly present as asymptomatic colonizers or inhabitants of lower gastrointestinal and female reproductive tracts, GBS has been recognized as a leading cause of sepsis and meningitis in newborns with infection acquired by the newborn in utero or during passage through the colonized birth canal.15, 16 It is also a recognized pathogen in post‐partum and pregnant females responsible for a broad range of infections such as chorioamnionitis, endometritis, and urinary tract infection.8, 15 However, the real burden of GBS as a urinary pathogen in other population groups, such as non‐pregnant adult females and adult males has largely remained unexplored and undefined.
Reports on the incidence of genitourinary infections due to GBS appears to have increased in the past decade.5, 17, 18, 19, 20 In a study quantifying its role as a cause of surgical site and non‐invasive infections at all ages, it was found that the prevalence of GBS in community and hospital UTI isolates was 1.61% (95% confidence interval 1.13–2.30%) and 0.73% (95% confidence interval 0.43–1.23%), respectively.17 In a retrospective study on UTIs diagnosed in a single center in Mexico through 10 years, GBS constituted 2.1% (18 of 859) of the urinary isolates.18 A recent review of the overall burden of S. agalactiae UTI demonstrated approximately 160,000 cases in the United States annually underscoring the importance of these infections as a major public‐health concern.5 In another study, GBS were isolated most frequently from the urinary tract (52%) followed by the skin (36.2%) in non‐pregnant adult patients.19 In fact, the incidence of neonatal disease appears to have decreased due to improvements in screening and prophylaxis,21, 22 and the changing spectrum of the disease in adults has been noted.10, 23, 24, 25 In a study, over 60% of cases of invasive GBS disease in the United States of America (USA) occurred in adults and most were unrelated to pregnancy.23 In Northern Taiwan, out of 120 episodes of invasive GBS disease recorded at a tertiary care teaching hospital from January 1998 to June 2009, 58.3% was found in the elderly (age ≥65), 36.1% in non‐pregnant women and young adults (age 18–64), and 5.9% in the neonates (0–90 days).24 A more recent study also observed that invasive GBS incidence among non‐pregnant adults increased significantly from 8.1 cases per 100 000 population in 2008 to 10.9 in 2016.25 In the current study, all the patients were non‐pregnant adult females ranging in age from 19 to 52 years. A similar observation has been reported by Magliano et al, where S. agalactiae was shown to occur most frequently in women aged between 15 and 59 years presenting with community‐acquired UTI.1
As regards the risk factors, GBS infection in adults is often seen to be associated with diabetes or obesity, which are considered as risk factors for disease due to this organism.5, 7, 19, 25 However, diabetes was not identified as a risk factor for GBS UTI in other studies.20 Similarly, in the current series, diabetes as an associated co‐morbidity was observed in only two patients out of seven (28.6%) with GBS UTI. Thus, in our study, majority of the patients (4 out of 7; 57.1%) were apparently immune‐competent and did not have any associated co‐morbidities. Another point of note is that, though in our study, all the specimens with GBS UTI yielded significant colony counts of ≥104/ml of urine, previous studies have observed that a low colony count does not preclude the diagnosis of GBS UTI or prevent future intrapartum colonization.26, 27 Thus, even a low urinary colony count of GBS in a symptomatic patient should not be ignored as contaminants; rather a diagnosis of true cystitis due to GBS should be kept in mind in such cases.
Limited reports are available on antimicrobial resistance profile of GBS from UTIs. In a study investigating the serotype distribution and antimicrobial resistance of GBS strains isolated from urine in China, the resistant rates measured for tetracycline, erythromycin, clindamycin, and fluoroquinolones were 74.1, 63.0, 44.4, and 48.1%, respectively.28 Similarly, a study in USA also found a high rate of resistance to tetracycline (80.4%), erythromycin (39.5%), and clindamycin (26.4%) in urinary GBS isolates.20 In contrast, none of the isolates in the current study demonstrated resistance to erythromycin or clindamycin, though tetracycline resistance was observed in 42.8% isolates. Also a high rate of resistance (57.1%) to fluoroquinolone (levofloxacin) was observed in our study isolates. However, similar to these studies,20, 28 the isolates in the current series were uniformly sensitive to penicillin, ceftriaxone, vancomycin, linezolid, and chloramphenicol.
We believe the present series of cases will lead to increased awareness for laboratory detection and diagnosis of GBS in cases of UTI due to gram‐positive, catalase‐negative cocci. The present series of cases further delineates the potential role of GBS in causing UTI even in apparently immune‐competent non‐pregnant adult females with propensity to persist without appropriate treatment. Since the persistent presence of GBS in the genitourinary tract may pose a significant threat of transmission to the neonate in case of future pregnancy, UTI due to GBS needs to be diagnosed and treated early, especially in women of reproductive age group. More studies should be conducted in the future to obtain accurate estimates of GBS prevalence in UTI by more specific means such as by using selective media in routine patient specimens and further sub‐culturing of any catalase‐negative, bile‐esculin negative, gram‐positive cocci obtained on CLED agar medium to a blood agar medium to examine for specific hemolytic property enabling accurate identification.
CONFLICTS OF INTEREST
None.
AUTHOR CONTRIBUTIONS
SM contributed in study conception and design; acquisition, analysis, and interpretation of data, literature search, and gave the final approval for the manuscript to be published. GP involved in acquisition, analysis and interpretation of data, performed the literature search, and drafted the initial version of the manuscript. SR and RKS involved in acquisition, analysis and interpretation of data, literature search, and critical review of the manuscript. RRM: was the treating physician, contributed to the acquisition, analysis, and interpretation of data and performed critical review of the work for important intellectual content.
ETHICAL APPROVAL
Ethics approval has been obtained with review exemption from the Institutional Ethics Committee of our Institute, that is, the Institutional Ethics Committee of All India Institute of Medical Sciences, Bhubaneswar, Odisha, India. Reference number – T/IM‐NF/Micro/19/58 dated 11.11.2019.
CONSENT
The authors have confirmed during submission that patient consent has been signed and collected in accordance with the journal's patient consent policy. Published with written consent of the patient.
ACKNOWLEDGMENT
We acknowledge the technical support provided by Ms. Alakananda Mohapatra for this work.
Mohanty S, Purohit G, Rath S, Seth RK, Mohanty RR. Urinary tract infection due to Group B Streptococcus: A case series from Eastern India. Clin Case Rep. 2021;9:e04885. 10.1002/ccr3.4885
Funding information
None
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available within the article.
REFERENCES
- 1.Magliano E, Grazioli V, Deflorio L, et al. Gender and age‐dependent etiology of community‐acquired urinary tract infections. Scientific World J. 2012;2012:349597. 10.1100/2012/349597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Medina M, Castillo‐Pino E. An introduction to the epidemiology and burden of urinary tract infections. Ther Adv Urol. 2019;11:1756287219832172. 10.1177/1756287219832172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mohanty S, Kapil A, Das BK, Dhawan B. Antimicrobial resistance profile of nosocomial uropathogens in a tertiary care hospital. Indian J Med Sci. 2003;57(4):148‐154. [PubMed] [Google Scholar]
- 4.Ganesh R, Shrestha D, Bhattachan B, Rai G. Epidemiology of urinary tract infection and antimicrobial resistance in a pediatric hospital in Nepal. BMC Infect Dis. 2019;19(1):420. 10.1186/s12879-019-3997-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kline KA, Lewis AL. Gram‐positive uropathogens, polymicrobial urinary tract infection, and the emerging microbiota of the urinary tract. Microbiology Spectrum. 2016;4(2). 10.1128/microbiolspec.UTI-0012-2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Beyer I, Mergam A, Benoit F, Theunissen C, Pepersack T. Management of urinary tract infections in the elderly. Z Gerontol Geriatr. 2001;34(2):153‐157. [DOI] [PubMed] [Google Scholar]
- 7.Edwards MS, Baker CJ. Group B streptococcal infections in elderly adults. Clin Infect Dis. 2005;41(6):839‐847. [DOI] [PubMed] [Google Scholar]
- 8.Mathai E, Thomas RJ, Chandy S, Mathai M, Bergstrom S. Antimicrobials for the treatment of urinary tract infection in pregnancy: practices in southern India. Pharmacoepidemiol Drug Saf. 2004;13(9):645‐652. [DOI] [PubMed] [Google Scholar]
- 9.Trivalle C, Martin E, Martel P, Jacque B, Menard JF, Lemeland JF. Group B streptococcal bacteraemia in the elderly. J Med Microbiol. 1998;47(7):649‐652. [DOI] [PubMed] [Google Scholar]
- 10.Ulett KB, Shuemaker JH, Benjamin WH Jr, Tan CK, Ulett GC. Group B streptococcus cystitis presenting in a diabetic patient with a massive abdominopelvic abscess: a case report. J Med Case Rep. 2012;6:237. 10.1186/1752-1947-6-237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Unnikrishnana D, Delacruz MA, Saha A, Daniels R. Case of acute paraspinal pyomyositis in an elderly diabetic secondary to spread from urinary tract infection. BMJ Case Rep. 2018;pii:bcr‐2018‐225739. 10.1136/bcr-2018-225739 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Swain B, Rakshit A, Sahu KK, Sahoo N, Otta S. Group B Streptococcus: an unusual cause for urinary tract infection. J Clin Diag Res. 2017;11:DL05‐DL06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Goel N, Wattal C, Gujral K, Dhaduk N, Mansukhani C, Garg P. Group B Streptococcus in Indian pregnant women: Its prevalence and risk factors. Indian J Med Microbiol. 2020;38(3&4):357‐361. [DOI] [PubMed] [Google Scholar]
- 14.CLSI Clinical and Laboratory Standards Institute . (2019). Performance Standards for Antimicrobial Susceptibility Testing; 29th ed. CLSI Document M100 Wayne, PA: Clinical and Laboratory Standards Institute.
- 15.Winn HN. Group B Streptococcus infection in pregnancy. Clin Perinatol. 2007;34(3):387‐392. [DOI] [PubMed] [Google Scholar]
- 16.Melin P. Neonatal group B streptococcal disease: from pathogenesis to preventive strategies. Clin Microbiol Infect. 2011;17(9):1294‐1303. [DOI] [PubMed] [Google Scholar]
- 17.Collin SM, Shetty N, Guy R, et al. Group B Streptococcus in surgical site and non‐invasive bacterial infections worldwide: A systematic review and meta‐analysis. Int J Infect Dis. 2019;83:116‐129. [DOI] [PubMed] [Google Scholar]
- 18.Lagunas‐ Rangel FA. Antimicrobial susceptibility profiles of bacteria causing urinary tract infections in Mexico: Single‐centre experience with 10 years of results. J Glob Antimicrob Resist. 2018;14:90‐94. [DOI] [PubMed] [Google Scholar]
- 19.Toumi A, Ferjani A, Ben Abdallah H, Boukadida J. Streptococcus agalactiae in nonpregnant adults. Tunis Med. 2006;84(3):161‐164. [PubMed] [Google Scholar]
- 20.Ulett KB, Benjamin WH Jr, Zhuo F, et al. Diversity of group B streptococcus serotypes causing urinary tract infection in adults. J Clin Microbiol. 2009;47(7):2055‐2060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Van Dyke MK, Phares CR, Lynfield R, et al. Evaluation of universal antenatal screening for group B streptococcus. N Engl J Med. 2009;360(25):2626‐2636. [DOI] [PubMed] [Google Scholar]
- 22.Schrag SJ, Zywicki S, Farley MM, et al. Group B streptococcal disease in the era of intrapartum antibiotic prophylaxis. N Engl J Med. 2000;342(1):15‐20. [DOI] [PubMed] [Google Scholar]
- 23.Phares CR, Lynfield R, Farley MM, et al. Epidemiology of invasive group B streptococcal disease in the United States, 1999–2005. JAMA. 2008;299(17):2056‐2065. [DOI] [PubMed] [Google Scholar]
- 24.Wong SS, Tsui K, Liu QD, et al. Serotypes, surface proteins, and clinical syndromes of invasive Group B Streptococcal infections in northern Taiwan, 1998–2009. J Microbiol Immunol Infect. 2011;44(1):8‐14. [DOI] [PubMed] [Google Scholar]
- 25.Francois Watkins LK, McGee L, Schrag SJ, et al. Epidemiology of Invasive Group B Streptococcal Infections Among Nonpregnant Adults in the United States, 2008–2016. JAMA Intern Med. 2019;179(4):479‐488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tan CK, Ulett KB, Steele M, Benjamin WH Jr, Ulett GC. Prognostic value of semi‐quantitative bacteruria counts in the diagnosis of group B streptococcus urinary tract infection: a 4‐year retrospective study in adult patients. BMC Infect Dis. 2012;12:273. 10.1186/1471-2334-12-273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pérez‐Moreno MO, Picóo‐Plana E, Grande‐Armas J, Centelles‐Serrano MJ, Arasa‐Subero M, Ochoa NC. led by MO Pérez‐Moreno MOTSGG. Group B streptococcal bacteriuria during pregnancy as a risk factor for maternal intrapartum colonization: a prospective cohort study. J Med Microbiol. 2017;66(4):454‐460. [DOI] [PubMed] [Google Scholar]
- 28.Guo Y, Deng X, Liang Y, Zhang L, Zhao GP, Zhou Y. The draft genomes and investigation of serotype distribution, antimicrobial resistance of group B Streptococcus strains isolated from urine in Suzhou, China. Ann Clin Microbiol Antimicrob. 2018;17(1):28. 10.1186/s12941-018-0280-y [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available within the article.


