Abstract
With the recent urbanization and globalization, the adult obesity rate has been increasing, which was paralleled with a dramatic surge in the incidence and prevalence of nonalcoholic fatty liver disease (NAFLD). NAFLD poses a growing threat to human health as it represents the most common cause of chronic liver disease in developed countries. It encompasses a wide spectrum of conditions starting from a build-up of fat in hepatocytes (steatosis), to developing inflammation (steatohepatitis), and reaching up to cirrhosis. It is also associated with higher rates of cardiovascular mortalities. Therefore, proper timely treatment is essential and weight loss remains the cornerstone in the treatment of obesity-related liver diseases. When diet, exercise, and lifestyle changes are not successful, the current recommendation for weight loss includes antiobesity medications and bariatric endoscopic and surgical interventions. These interventions have shown to result in significant weight loss and improve liver steatosis and fibrosis. In the current literature review, we highlight the expected outcomes and side effects of the currently existing options to have a weight-centric NAFLD approach.
Keywords: nonalcoholic fatty liver disease, nonalcoholic steatohepatitis, obesity, weight loss, bariatric surgery
Obesity is defined as the excessive accumulation of fat in the adipose tissue throughout the body. It represents a complex and multifactorial disease with multiple adverse effects on human health. 1 Obesity is ranked as the second most common preventable risk factor for overall death, after smoking. 2 When comparing the all-cause mortality between patients with normal body weight and those with obesity, the hazard ratio for severe obesity was 1.29. 3 Due to the significant rise in its incidence and prevalence, obesity is now regarded as “the single greatest threat to public health for the United States of America (USA).” 1 The most recent statistics from the Center for Disease Control and Prevention (CDC) reveal an increase in obesity prevalence within the past 18 years, from 30.5% in 1999 to 2000 to 42.4% in 2017 to 2018, 4 5 and according to the direst projections, 50% of the United States population will have obesity by 2030. 6 This, by no doubt, would translate into a remarkable rise in obesity-related comorbidities which include diabetes mellitus (DM), hypertension, cardiovascular diseases, and nonalcoholic fatty liver disease (NAFLD), among others. 7 Consequently, evidence has shown that obesity increases the risk of developing liver diseases, particularly NAFLD (relative risk [RR] 4.6 [95% confidence interval or CI 2.5–11.0]), cirrhosis (RR 4.1 [95% CI 1.4–11.4]) and hepatocellular carcinoma (RR 1.89 [95% CI 1.5–2.4]). 7 It also plays a significant role in determining the prognosis of other liver diseases, like viral hepatitis, as it diminishes the response to treatment by hindering the fibrosis improvement. 8
Nonalcoholic Liver Disease
The hepatic manifestation of metabolic syndrome presents in the form of NAFLD, which is the leading cause of chronic liver disease in the United States. 9 Based on multiple epidemiological studies, the prevalence rate of NAFLD in United States and North America is 21 to 24.7%, 10 while the annual incidence is 2.2 to 3.2%. 11 This prevalence is even higher among people with obesity (up to 90%) 12 and type 2 DM (T2DM) (59.67%). 13 It encompasses a wide clinical and histological spectrum ranging from mild hepatic steatosis to the more severe forms of nonalcoholic steatohepatitis (NASH), leading to hepatic fibrosis, cirrhosis, and hepatocellular carcinoma. The exclusion of other liver disease causes, such as excessive alcohol intake, is essential for diagnosis. 14 NAFLD pathogenesis follows a series of liver insults, known as the “multi-hit hypothesis.” 14 In genetically predisposed subjects, insulin resistance marks the beginning of the chain, activating B-oxidation and adipose tissue lipolysis, which results in free radical injury to the hepatic cells. It promotes the production of proinflammatory cytokines with a worsening cycle of inflammation, necrosis, and fibrosis. 14
The diagnosis of NAFLD necessitates the presence of proof for the excessive accumulation of fatty acids in the hepatocytes, by either imaging or direct visualization under microscopy. 15 However, some “noninvasive” scoring systems have been developed. 15 The National Institutes of Health's NASH Committee 16 formulated a NAFLD severity score called NAFLD Activity Score (NAS). The NAS is a numerical grading system, calculated according to the severity level of each of three defining histological features. These include: (1) severity of steatosis (0–3), (2) hepatocellular ballooning (0–2), and (3) lobular inflammation (0–3). 16 The final score represents the sum of each element, ranging from 0 to 8. A reduction in the NAS marks a refinement in the histological features toward the resolution. 16 Another noninvasive scoring system for liver fibrosis is the nonalcoholic fatty liver disease fibrosis score (NFS), which utilizes routinely measured clinical and biochemical laboratory findings. 17 However, the low sensitivity and specificity of NFS in diagnosing NAFLD fibrosis necessitates further diagnostic modalities. Imaging tests are considered acceptable forms of noninvasive testing with relatively higher detection accuracy. Imaging modalities, such as ultrasound-based transient elastography (USTE) and magnetic resonance (MR) elastography, can detect the level of liver stiffness. 18 19 A meta-analysis of seven studies conducted to measure USTE accuracy revealed the presence of a direct relationship between the NAFLD stage on biopsy and the USTE sensitivity and specificity levels. 18 On the other hand, MR elastography can be used to both diagnose and grade NAFLD. 19 According to another meta-analysis, the sensitivity and specificity of MRE reach up to 82.3 and 86.9%, respectively. 20 Despite the impracticality and invasiveness, puncture liver biopsy remains the gold standard diagnostic test for NAFLD, with the highest sensitivity and specificity amongst all. 21
With the increased detection rate of NAFLD, management practices are focused on preventing the development of steatohepatitis (NASH) and hepatic fibrosis and reducing the risk of any NAFLD-associated cardiovascular comorbidities. According to a population-based cohort study, 22 NAFLD acts as an independent risk factor for cardiovascular mortality, mandating further cardiovascular risk control in these patients. 22 Interestingly, Ampuero et al 23 also followed metabolically healthy patients with NAFLD for 15 years and observed that those with a significant level of hepatic fibrosis were at a higher risk of developing metabolic conditions such as T2DM (HR = 2.95, p = 0.019) and hypertension (HR = 2.39, p = 0.028). They had four-times higher incidence rate than those with mild fibrosis. 23 Therefore, early initiation of NAFLD treatment reduces the risk of worsening hepatic inflammation with better overall health outcomes. Given the fact that obesity and metabolic syndrome are regarded as the main key players in the development of NAFLD, efforts toward weight reduction and improving the metabolic profile highlight the most beneficial interventions and remain the cornerstone of NAFLD treatment. 24 The suggested therapeutic approach begins with lifestyle modification (diet and exercise), and if sufficient and sustained weight loss is not achieved, second-level therapies (e.g., antiobesity medications, bariatric endoscopic and surgical procedures) may be used to enhance the overall effect. 25 Strong evidence suggested that a 5 to 10% loss of the initial body weight led to a recovery in the histological findings of NAFLD. 26 In this review study, we aim to shed light on the feasibility and sustainability of various obesity management approaches and their consequence on related liver diseases.
Management of Obesity in Patients with NAFLD
Lifestyle Modifications (Cornerstone of Treatment)
The cornerstone of NAFLD management is weight loss. 27 This requires adopting an energy restrictive lifestyle that includes a decrease in oral caloric intake and an increase in the body's energy expenditure. According to multiple randomized-controlled trials (RCTs), 27 28 29 30 31 32 33 34 weight loss in NAFLD patients led to an improvement in hepatic triglyceride contents (determined using an MR) and in NAS (determined by liver biopsy) with an accompanied reduction in cardiovascular risk factors such as insulin resistance and the level of serum lipid. 27 28 29 30 31 32 33 34 According to Musso et al, 35 a weight loss of ≥ 7% led to significant positive outcomes in histological findings and cardiometabolic profile. 35 Patients with biopsy-proven steatohepatitis (NASH) should aim for a higher percentage of weight loss than patients with steatosis (NAFLD); it is recommended that in NASH the weight loss goal should be close to 10%. 35
Current guidelines endorse that to achieve weight loss, an energy deficit is essential. Limiting dietary energy intake below that required for energy balance can be achieved through a reduction of daily calories to 1,200 to 1,500 for women, and 1,500 to 1,800 for men (kilocalorie levels are usually adjusted for the individual's body weight and physical activity levels); or estimation of individual daily energy requirements and prescription of an energy deficit of 500 or 750 kcal/d. 25 As a proof, Vilar-Gomez et al conducted a prospective study over 52 weeks on 293 subjects. These subjects were encouraged to adopt a healthy lifestyle to reduce their body weight. 36 By the end of the study, 30% had lost ≥ 5% of their weight. While comparing body weight loss with improvement in NASH-related histological parameters, weight loss was found to be an independent factor (Odds ratio = 1.1–2.0; p < 0.01). Twenty-five per cent of these subjects achieved NASH resolution, 47% had a reduction in NAS score, and 19% had complete fibrosis regression. 36 Despite the ongoing controversies, the macronutrient composition of the diet does not influence the total body weight loss (TBWL) in large population studies, signifying the low-calorie and diet-adherence remain the key factors for successful weight loss. 37 Meanwhile, the main obstacle in lifestyle modifications is putting the patient in a negative energy balance to achieve an ideal weight and sustain it. Thus, finding an alternative therapeutic approach independent of weight loss will be favored.
Some of the recently published studies have highlighted the potential plausible effect of the macronutrient composition of the Mediterranean Diet (MedDiet) in resolving NAFLD, independent of accompanying weight loss. 38 The MedDiet is a well-known dietary pattern in the Mediterranean countries and is mainly characterized by a high intake of plant-based and fiber-rich foods, like vegetables, fruits, whole grains and nuts, along with a moderate intake of protein-rich lean meats like poultry and fish. 39 Also, it is rich in monounsaturated fatty acids mainly from olive and olive oil and low in red meat consumption. 39 A promising crossover study was conducted by Ryan et al 40 to examine the effect of MedDiet, when compared with low fat-high carbohydrate diet (LF-HCD), on NAFLD regression. After randomizing and matching both groups, all potential confounders such as physical activity, abdominal obesity, mean serum glucose, and insulin sensitivity were excluded. Despite the lack of significant difference in weight loss between the two dietary groups ( p = 0.1), the changes in NAFLD and insulin sensitivity were significant. The mean reduction of liver fat was 39 ± 4% in the MedDiet group when compared with only 7 ± 2% in the LF-HCD ( p = 0.03). Moreover, circulating insulin concentration declined significantly in MedDiet ( p = 0.003), suggesting an enhancement in insulin sensitivity. 40 Further, an interesting study with a larger cohort of participants ( n = 94) is currently being conducted aiming to examine the sustainability of these metabolic changes after 12 months follow-up. 38
Along with the prescription for a reduced-calorie diet, a comprehensive lifestyle intervention program should advocate of increasing aerobic physical activity for ≥ 150 min per week (such as brisk walking for ≥ 30 min/d most days of the week), and a goal of > 10,000 steps per day. Higher levels of physical activity, approximately 200 to 300 minutes per week, are recommended to maintain weight loss or minimize weight regain for long term (> 1 year). 41 Diet and physical activity recommendations can be in combination with a hospital/university or commercial behavioral programs; these are usually comprehensive lifestyle interventions that provide structured behavior strategies to facilitate adherence to diet and activity recommendations. These strategies include regular self-monitoring of food intake, body weight, physical activity, and food cravings. These same behaviors are recommended to maintain lost weight, with the addition of frequent (i.e., weekly or more frequent) monitoring of body weight. 42
Intense lifestyle interventions vary in outcomes and on average, most weight loss attempts with diet and exercise result in 30% of the patients losing more than 5% TBWL in 6 to 12 months. 43 The reasons for the variability of response are attributed to many causes from diet adherence to metabolic adaptations that thwart weight loss attempts. 44 45 46 Thus, it is essential to escalate obesity care via utilizing second-level therapies to achieve significant weight loss and to prevent weight regain ( Fig. 1 ). 25 In this white paper endorsed by nine academic societies, 25 the recommendation is to complement the cornerstone therapy of diet and exercise with antiobesity medications, bariatric endoscopy, and surgery to all the patients with obesity or obesity-related comorbidities who do not achieve successful weight loss with lifestyle alone.
Fig. 1.
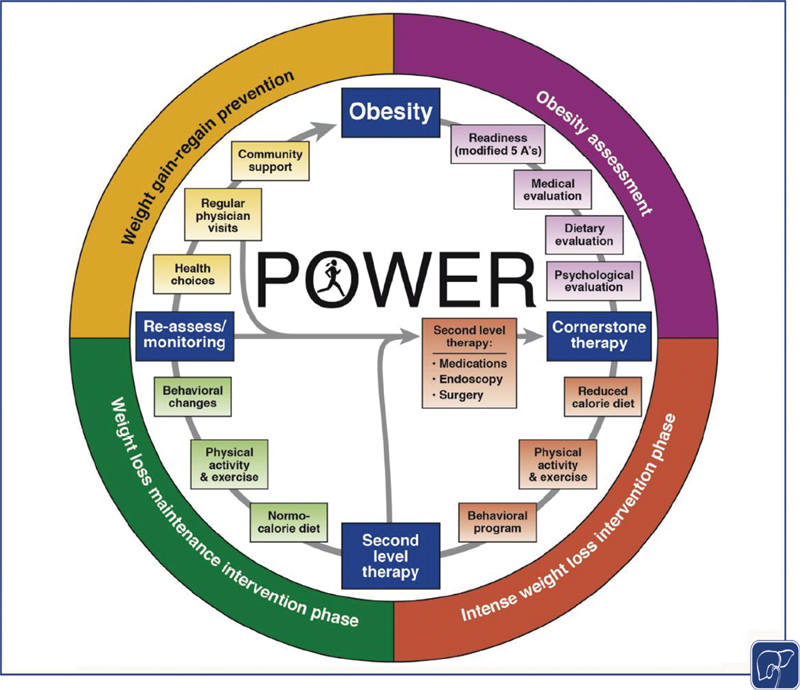
Practice Guide on Obesity and Weight Management, Education, and Resources (POWER) for representing the continuous management of obesity. Introducing four main stages of obesity care and the need to introduce second-level therapies. 25
Antiobesity Medications
Antiobesity medications (AOMs) approved by the Food and Drug Administration (FDA) for weight loss might be considered as the next line of treatment for obesity or obesity-related liver diseases. The FDA adopts a figure of at least 5% difference in mean weight loss of medication, compared with placebo, to be approved for the long-term use in patients with a body mass index (BMI) > 30 kg/m 2 , or those with a BMI > 27 kg/m 2 and have one or more obesity-related comorbidities such as T2DM, hypertension, and dyslipidemia. 47 The current AOMs approved for long-term use include phentermine/topiramate, orlistat, liraglutide, and bupropion/naltrexone reviewed in detail elsewhere. 25 These medications achieved 6 to 11% TBWL over 12 months in conjunction with lifestyle modifications. 47 In theory, all the AOMs should result in NAFLD improvement based purely on their weight loss effect, however, only liraglutide and orlistat have been formally studied in liver disease ( Table 1 ).
Table 1. Comparison between different weight-reducing medications based on %TBWL, NAS, and NFS improvement and the incidence rate of adverse events.
| Medications | Mechanism of action | Mean %TBWL (medication vs. placebo) | NAS improvement | NFS improvement | Adverse event |
|---|---|---|---|---|---|
| Liraglutide 3.0 mg 51 | GLP-1 analog inhibits food intake by inducing satiety; 51 | −8.0 vs. −2.6% after 56 wk. 51 | − 1.3 change in total NAS score and 74% had NAS improvement. 39% had complete resolution. |
− 0.2 change in fibrosis ( p = 0.11). 51 | Nausea: 39.3% Diarrhea: 20.9% |
| Semaglutide0.4 mg 52 | Long-acting GLP-1 analog inhibits food intake by inducing satiety. 52 | −13 vs. −1% after 72 wk. 52 | − 59 vs. −17% after 72 wk. 52 | 43% had an improvement in fibrosis stage in the 0.4-mg group and in 33% in the placebo group ( p = 0.48). 52 | Nausea: 42 vs. 11% Constipation: 22 vs. 12% Decreased appetite: 22 vs. 5% Vomiting: 15 vs. 2% Abdominal pain: 7 vs. 4%. 52 |
| Orlistat | Reversible pancreatic lipase inhibitor, decreasing the hydrolysis of intestinal fat, therefore, absorption. 56 | −8.3 vs. −6% after 36 wk. | NA | − 0.14 change in fibrosis. 119 | Fecal incontinence: 26.6%. Flatus with discharge: 23.9%. Fecal urgency: 22.1%. 120 |
| Phentermine/Topiramate ER |
Phentermine
is a noradrenergic drug that was speculated to suppress the appetite by acting centrally in the nervous system.
Topiramate is a fructose derivative and acts as a neurostabilizer by modifying the sodium and calcium voltage-gated channels, which increases energy expenditure and decreases food intake by suppressing appetite and enhancing satiety. |
−10.9 vs. −1.6% after 56 wk. 67 | NA | NA | Constipation: 16.3%. Paresthesia: 13.7%. Dry mouth: 13.7%. 120 |
| Bupropion/naltrexone SR |
Bupropion
inhibits the reuptake of dopamine and NE and stimulates POMC release in the hypothalamus; it acts as a precursor for α-MSH and β-endorphin. The α-MSH subunit has an appetite suppressing effect when activating MC4R leading to reduced food intake and therefore weight loss.
Naltrexone is an opioid receptor antagonist acting mainly on μ-type opioid receptors which include μ-opioid receptors on POMC; therefore, it diminishes the appetite by blocking the autoinhibitory effect of β-endorphins on the POMC neurons. |
−8.1 vs. −1.8% after 56 wk. 73 | Improvement in liver enzymes was reported. 121 | Improvement in FIB-4 fibrosis score. 121 | Nausea: 29.8%. Headache: 13.8%. Constipation: 15.7%. 73 |
Abbreviations: NAS, nonalcoholic fatty liver disease activity score; NE, norepinephrine; POMC, proopiomelanocortin; TBWL, total body weight loss.
GLP-1 Analogs
GLP-1 is an endogenously produced, gut-derived incretin hormone. It augments insulin secretion by β-cells and reduces glucagon release. It also reduces gastric emptying and food intake. GLP-1 analogs, e.g., Liraglutide, have been initially FDA-approved in 2005 for the treatment of T2DM. 48 In 2015, a large double-blinded RCT ( n = 3731) was conducted to study the efficacy of 3.0 mg liraglutide on weight loss, compared with placebo. The liraglutide group achieved a mean weight loss of 8.4 ± 7.3 kg compared with 2.8 ± 6.5 kg only ( p < 0.001) in the placebo group. 63.2% of the candidates in the liraglutide group lost more than 5% of their body weight as compared with 27.1% in the placebo group ( p < 0.001). 49 Following multiple similar RCTs, liraglutide was approved by the FDA in December 2014 for weight loss. 50 In addition, another 48-week double-blinded RCT called “LEAN” trial 51 was conducted to objectively measure the effect of 3.0 mg liraglutide on NASH. The study showed that 39% (nine out of 23) patients on liraglutide versus 9% (two out of 22) patients on placebo had complete resolution of NASH without worsening of fibrosis based on the end-of-treatment liver biopsy and histological findings. 51 Additionally, 19 (83%) of liraglutide group versus 10 (45%) had improvement in steatosis with a significant difference between the groups ( p = 0.009). The level of improvement in fibrosis was not significant ( p = 0.46); however, the worsening in fibrosis was more prominent ( p = 0.04) in the placebo group (36%) compared with the liraglutide (9%). 51 GLP-1 analogs demonstrated potency in enhancing insulin sensitivity and reducing body weight; thus, improving the overall outcome in patients with NAFLD and NASH.
Recently, in a double-blinded RCT conducted over 72 weeks, a similar long-acting GLP-1 receptor agonist, 0.4 mg of semaglutide, showed a significant reduction in mean body weight of 13% in the treatment groups versus 1% in placebo. The improvement of NASH was 59% of patients in the treatment group versus 17% in the placebo group ( p < 0.001). 52 However, the amelioration in the fibrosis stage was not significant between both groups as it was 43% in the treatment group and 33% in the placebo group ( p = 0.48). 52 These findings promote liraglutide—or other GLP-1 analogs—as an attractive medication(s) to consider in patients with NASH.
Orlistat (Xenical)
Gastric and pancreatic lipases are secreted into the alimentary tracts and play important roles in the digestion of dietary fat. These enzymes hydrolyze triglycerides into absorbable free fatty acid chains and a monoglyceride. Orlistat acts locally by reversibly inhibiting gastric and pancreatic lipases through forming covalent bonds with the serine residues of the lipase active sites rendering them inactive. The recommended dosage of orlistat (120 mg capsule TID) reduces dietary fat absorption by around 30% and improves insulin resistance. 53 According to a systematic review, 54 orlistat is effective for improving serum lipid profiles, including total cholesterol, LDL, and HDL cholesterols, in patients with obesity and with/without diabetes. 54 Multiple studies 55 56 57 58 have investigated the effect of orlistat use and dietary modifications on NAFLD, and all of them reported improvement in the liver enzymes (aspartate aminotransferase [ALT] and alanine aminotransferase [AST]) and liver fat content based on liver histopathology. 55 56 57 58 Patients who used orlistat for at least 24 weeks had improvement of their hepatic profile (biopsy and enzymes) with 5 to 10% weight loss, in all five studies. Interestingly, an RCT conducted over 24 weeks, assessed the efficacy of orlistat in the treatment of NAFLD on 52 subjects. Both groups had a similar amount of significant weight loss with no differences between groups ( p = 0.5). However, an almost twofold reduction in the serum levels of ALT and AST occurred in the orlistat group. In addition, reversal of fatty liver by ultrasound was observed in the orlistat group with no significant difference in the degree of steatosis and fibrosis between both groups. 59
On the contrary, Harrison et al, 56 conducted a 36-week double-blinded RCT to assess the efficacy 120 mg TID orlistat to treat NASH on 41 patients. All participants were instructed to have their level of physical activities without recommending a specific type of exercise. The outcomes of this study were: (1) an overall mean weight loss of 8.3% ( p < 0.001) and (2) reduction in serum ALT from a mean of 108 ± 95 IU/L to 53 ± 41 IU/L ( p < 0.001) and AST from 64 ± 39 IU/L to 36 ± 17 IU/L ( p < 0.001) when compared with baseline. However, no statistically significant difference in weight loss was detected between both groups ( p = 0.85). Harrison et al concluded that orlistat improvement in NAFLD is weight loss dependent. 56
Phentermine/Topiramate Extended Release (Qsymia)
Phentermine is a noradrenergic drug that was speculated to suppress appetite by acting centrally in the nervous system to enhance norepinephrine (NE) release and reuptake. It has been approved in the United States to treat obesity since 1959 with the therapeutic dosage ranging from 15 to 37.5 mg per day. 60 It does not have any serotonergic or dopaminergic activities, therefore, it possesses a lesser risk for addiction as compared with amphetamines. 61 On the other hand, Topiramate is a fructose derivative which acts as a neurostabilizer by modifying the sodium and calcium voltage-gated channels. It has been used as an antiepileptic in 1996 and for migraine prophylaxis in 2004. 62 Topiramate was found to induce weight loss as a side effect. Further studies were performed to evaluate the efficacy of these medications in reducing body weight. 63 The proposed theory behind this effect was the increase in energy expenditure, promoting satiation, and suppressing appetite. 64
Phentermine/topiramate extended release is a combination that has been approved by the FDA in 2012 for weight loss. 65 These two medications combined reduce appetite and enhance satiation. 66 Two large RCTs, the EQUIP and the CONQUER trials were conducted to study the efficacy of phentermine/topiramate extended release on weight loss with large sample sizes of 1,267 and 2,487, respectively. 65 According to the EQUIP study, 25.5, 13.0, and 5.9% for the placebo group participants were able to achieve a weight reduction of 5, 10, and 15%, respectively. However, 59.1, 27.7, and 12.4% of the candidates who received 3.75/23 mg of phentermine/topiramate extended release attain these targets. A higher dosage of 15/92 mg was associated with larger figures up to 83.5, 67.7, and 48.1%, respectively (between groups, p < 0.001). 67 Currently, there are no available studies in PUBMED that investigate the direct efficacy of phentermine/topiramate extended release on NAFLD or NASH. However, indirect effects on the course of NAFLD are rather speculated, given their efficacy in weight loss. Further RCT studies are required to objectively establish the relationship between phentermine/topiramate extended release and NAFLD.
Bupropion/Naltrexone Sustained Release (Contrave)
Bupropion/Naltrexone sustained release is a combination of two drugs, Bupropion and Naltrexone. Bupropion is a centrally acting drug that inhibits the reuptake of dopamine and NE, which will stimulate the proopiomelanocortin (POMC) release in the arcuate nucleus of the hypothalamus; it acts as a precursor of the two subunits, α-melanocyte-stimulating hormone (α-MSH) and β-endorphin (opioid). 68 The α-MSH subunit has an appetite suppressing effect when activating melanocortin-4 receptors (MC4R) leading to reduced food intake and therefore weight loss. However, this weight loss does not exceed 5% of the total body weight even on long-term use. 69 The suggested limitation in the efficacy of weight loss is due to the autoinhibitory feedback on μ-opioid receptors of the POMC neurons by the action of the β-endorphins subunit. Primarily, bupropion has been used to treat patients with depression or for smoking cessation with a daily dosage of 300 mg. 69 70 In contrast, naltrexone is an opioid receptor antagonist acting mainly on μ-type opioid receptors. It is primarily used to treat the dependence on alcohol and opioids. As a side effect, it diminishes the appetite by blocking the autoinhibitory effect of β-endorphins on the POMC neurons. Thus, the unsuppressed action of α-MSH on the MC4R results in weight loss. 69
In September 2014, bupropion/naltrexone sustained release was approved by the FDA for weight loss in adult patients with obesity (BMI ≥ 30 kg/m 2 ) with/without comorbidities or in overweight patients (BMI ≥ 27 kg/m 2 ) with weight-related comorbidities. 71 72 Contrave Obesity Research I (COR-I) multicentric randomized trial was performed on 1,682 participants to study the efficacy of bupropion/naltrexone sustained release on weight loss over a period of 56 weeks. In patients taking bupropion (360 mg) naltrexone (32 mg) sustained release daily had an overall −8.1% change in their body weight upon the completion of the study when compared with the placebo group ( p < 0.0001). 73 A study 74 of 781 subjects to measure the effect of the medication on liver enzymes revealed the presence of a positive linear correlation between the level of weight loss and reduction in ALT ( r = 0.396, p < 0.001). 74 These results suggest the presence of positive outcomes when using bupropion/naltrexone sustained release to treat obesity and obesity-related liver diseases (NAFLD). Bajaj et al 75 confirmed these findings in a post-hoc analysis of four RCTs, demonstrating a significant improvement in the ALT and fibrosis index ( p < 0.0001). 75 However, bupropion/naltrexone sustained release has not been studied prospectively in biopsy-proven NAFLD or NASH and such studies are needed.
Procedural Interventions
As previously stated, lifestyle changes still mark the backbone of nonalcoholic steatosis and hepatitis management, mainly targeting a sustained long-term weight loss. The threshold for obtaining a meaningful improvement in the liver profile is set as 7 to 10% TBWL. However, only 30% of patients on lifestyle changes accomplish this goal, 43 which they rarely maintain for an adequate duration to allow full recovery of the liver. 76 Consequently, more advanced invasive approaches are needed, especially in those with initial failure of NAFLD amelioration. Bariatric endoscopic and surgical procedures have significantly higher levels of sustainable weight loss when compared with lifestyle modifications only. 77 Preliminary data shows promising results in reversing hepatic steatosis, steatohepatitis, and regression of fibrosis in 30% of patients. 78
Bariatric Endoscopic Interventions
Recently, endoscopic bariatric therapy (EBT) has emerged as an alternative to the more invasive and traditional bariatric surgeries. It is performed using a flexible gastrointestinal endoscopic tube inserted into the oral opening reaching the upper gastrointestinal tract. 79 EBT proves to be efficient in reducing body weight and serves as a safe alternative for those failing lifestyle and pharmacological measures especially with mild-to-moderate obesity (BMI range 30–40 kg/m 2 ) without comorbidities. 80 EBTs are reviewed in detail elsewhere. 79 Theoretically, EBTs might be one of the safest, least invasive, and most efficient procedures to treat patients with NAFLD ( Table 2 ). In this section, we will briefly cover the most useful and validated studies available in the literature on the effect of these procedures on NAFLD.
Table 2. Comparison between different weight-reducing bariatric endoscopic interventions based on %TBWL, NAS, and NFS improvement and the incidence rate of adverse events.
| Endoscopic intervention | Procedure | Mean %TBWL | NAS improvement | NFS improvement | Adverse Event (%) |
|---|---|---|---|---|---|
| Endoscopic intragastric balloon | Introducing an empty balloon into the stomach through an upper endoscopy. Then, the balloon is inflated with air or saline to reduce the stomach volume. This will result in early satiety and weight loss. 85 | −11.7 ± 7.7% after 6 mo. 66 | 90% had −3 points improvement in their NAS. 66 | − 1.17 stages improvement in 15% of participants in NFS. 66 | Abdominal pain (33.7%) Nausea (29%) GERD (18.3%) Erosion (12%) Early removal (7.5%) Others (ulceration, migration and small bowel obstruction) (3.7%). 89 |
| Endoscopic sleeve gastroplasty | A minimally invasive bariatric procedure that is done through endoscopically inserting a suturing device to remodel the greater curvature of the stomach. 67 | −14.9% after 6 mo. 69 | −4.0 points per year using Hepatic steatosis index (HSI). 70 |
20% had significant improvement in NFS (from F3-F4 or indeterminate to F0-F2).
70
−0.3 points annually following the procedure. 70 |
Overall incidence of adverse events is 2.2%. Pain and nausea requiring hospitalization (1.08%). Upper GI bleeding (0.56%). Perigastric leak or collection (0.48%). Pulmonary embolism (0.06%). Pneumoperitoneum (0.06%). |
Abbreviations: NAS, nonalcoholic fatty liver disease activity score; NFS, nonalcoholic fatty liver disease fibrosis score; TBWL, total body weight loss.
Endoscopic Intragastric Balloon
Intragastric balloon (IGB) is a space-occupying endoscopic procedure that results in weight loss through reducing the preprandial sense of hunger, increasing the intrameal satiation, and delaying gastric emptying to enhance postprandial satiety, thus, endorsing weight loss. 81 The balloons are inserted in the stomach, and then filled with either air or a normal saline solution. Usually, normal saline-containing balloons provide better outcomes as they produce both, volume and weight effects inside the stomach, which will further affect the satiety and satiation in those patients. 82 On August 5, 2015, FDA approved the use of BioEnterics Intragastric Balloon (Orbera) for weight loss. 83 84 Recently, in March 2021, Apollo Endosurgery received a Breakthrough Device Designation from the U.S. FDA for the use of Orbera in treating patients with BMI between 30 and 40 kg/m 2 and noncirrhotic NASH with liver fibrosis. 85
Bazerbachi et al 86 evaluated the effect of the single fluid-filled IGB on the metabolic and histologic features of NASH in 21 NAFLD patients. Following 6 months of IGB, the mean initial TBWL was 11.7%. 86 Eighteen of 20 patients (90%) had significant improvement in their NAS with a median of 3 points reduction. Interestingly, liver fibrosis was improved by 1.17 stages in 15% of participants, while MR elastography revealed an improvement of 1.5 stages in 50% of patients. Their overall conclusion is that 50% of NAFLD patients achieved the FDA-approved level of impactful amelioration of NASH and regression in liver fibrosis. 86 These positive outcomes reveal a promising long-standing intervention for patients with mild-to-moderate obesity. A thorough meta-analysis 87 included 10 published studies on the impact of Orbera IGBs on NAFLD resolution, revealed a mean reduction in the participants' body weight of 11.9 kg, with a significant ( p = 0.03) histological improvement in NAS with a mean score of 2 ± 0.75 versus 4 ± 2.25 in the control sham group, after 6 months of follow-up. 87 Also, Chandan et al 88 conducted a systematic review to evaluate the efficacy of Orbera IGB on NAFLD. The review included nine studies and shows that 79.2% of patients had improvement in steatosis while 83.5% improved in NAS score. All these promising data suggest a great future for the use of IGB in treating patients with NASH and NAFLD. While other IGBs may be beneficial for these conditions, further studies are needed. The effect of the other IGBs in weight loss is reviewed elsewhere. 79
IGBs are like any other procedural interventions, carry their own risks of complications and adverse events. According to a pooled study conducted by Abu Dayyeh et al 89 of a manual review for 67 studies with 8,500 implantations, the most common adverse events of IGBs were abdominal pain (33.7%), nausea (29%), GERD (18.3%), erosion (12%), early removal (7.5%), and others (ulceration, migration, and small bowel obstruction) (3.7%). 89
Endoscopic Sleeve Gastroplasty
Endoscopic sleeve gastroplasty (ESG) is an incisionless, minimally invasive bariatric endoscopy procedure that is done through inserting a suturing device attached to the endoscope to remodel the greater curvature. 90 This reduces the gastric capacity and alters digestive physiology by inducing satiation. 91 The efficacy of this procedure in achieving substantial weight loss has been well-studied. 92 A systematic review and a meta-analysis of eight observational studies with 1,859 participants revealed a pooled %TBWL of 14.9, 16.4, 16.8, and 20%, after 6, 12, 18, and 24 months, respectively. 93 Therefore, ESG has a strong potential to improve NAFLD and steatohepatitis. Hajifathalian et al 94 also studied liver steatosis on 118 patients who underwent ESG and showed a mean weight loss of 14.6 and 15.5% in 6 months and 2 years, respectively. Surprisingly, 83% of patients had a TBWL of ≥ 7% at 2 years postoperatively and found a substantial regression in NAS scores ( p = 0.034) with 20% of patients improving the risk for liver fibrosis from F3-F4 to F0-F2 ( p = 0.02). 94 Several clinical trials are on-going to study the effect of ESG on NAFLD and NASH.
Regarding the most common adverse events following ESG, a thorough and comprehensive meta-analysis conducted by Hedjoudje et al 95 showed promising findings. In nine large sample studies, the pooled incidence of severe post-ESG adverse event rate was 2.2%. The incidence of pain and nausea required hospitalization (1.08%), upper GI bleeding (0.56%), perigastric leak or collection (0.48%), pulmonary embolism and pneumoperitoneum (0.06%). These results reinforce the safety of ESG in minimizing adverse events while achieving a significant weight loss along with noticeable improvement in obesity-related liver diseases. 95
Endoscopic Duodenal Mucosal Resurfacing
Duodenum has been considered a targeted organ for weight loss. Favorable metabolic outcomes have been noticed when altering, excluding, or bypassing the duodenal exposure to intraluminal nutrients. 96 Some human and animal studies have suggested that the duodenal surface plays a pivotal role in glucose hemostasis. 97 98 Endoscopic duodenal mucosal resurfacing (DMR) is considered a minimally invasive procedure. The duodenal mucosa is separated from the submucosal layers by injecting normal saline, followed by circumferential hypothermal ablation of the mucosal layer. 99 A direct relationship was observed between the length of the ablated duodenal segment and the improvement in metabolic profile. 100 To further study the efficacy of DMR, de Oliveira et al 101 conducted a meta-analysis embracing various metabolic variables (HbA1C, fasting plasma glucose, weight loss, liver enzymes, and hepatic steatosis). After 3 months of DMR, HbA1C levels have significantly dropped by 1.72% in 52 patients ( p = 0.02). Whereas for weight loss, the mean weight reduction is 3.1 kg ( p < 0.001). In terms of hepatic improvement, liver enzymes (ALT) and MRI-evident hepatic steatosis were significantly reduced ( p < 0.001). 101 These findings support the effectiveness of the DMR procedure in NAFLD resolution. Despite the lack of 5 to 7% mean of TBWL, hepatic improvement was still evident. This might suggest an alternative mechanism in which the duodenal mucosa and secreted incretin hormones play a role in the development of NAFLD. A study with a longer follow-up duration is required to evaluate the long-term metabolic effects of DMR procedures. Besides, this procedure should be compared with other endoscopic procedures to further validate its efficacy and study its feasibility in treating NAFLD. A further ongoing clinical trial is being conducted to study the safety and effectiveness of DMR in the treatment of T2DM. 102
Bariatric Surgical Intervention
The effect of bariatric and metabolic surgeries in accomplishing sustainably significant weight loss and improvement in obesity-related comorbidities, including T2DM, HTN, and NAFLD, among others, has been well-studied. The popularity of these procedures has increased and reached up to 252,000 bariatric procedures performed in the United States in 2018. 103 These procedures are indicated for severe obesity, patients with a BMI of ≥ 40 kg/m 2 or a BMI of ≥ 35 kg/m 2 associated with obesity-related comorbidities. 104 Bariatric procedures have proven to enhance body metabolism independent of weight loss, therefore, leading to an improvement in blood glycemic and pressure control. 105 As NAFLD has been closely related to obesity and insulin resistance, further studies including the direct influence of these procedures on NAFLD patients who failed other therapeutic modalities may include satisfactory results ( Table 3 ). In 2020, a long-term prospective study published by Lassailly et al 106 revealed that bariatric surgery-induced resolution in 84% of patients with NASH without worsening of fibrosis ( p < 0.001) and the regression of fibrosis occurred in 70% of patients ( p < 0.001) at 5-years follow-up. 106 This novel study uncovers some hidden benefits of bariatric procedures in reducing liver-related mortalities. 106 However, the safety and risks of these procedures should be weighed against the benefits in improving NASH levels before considering them as first-line therapy. Additionally, further broad cohort studies to evaluate survival rates following bariatric procedure should be considered. 106
Table 3. Comparison between different weight-reducing bariatric surgical interventions based on %TBWL, NAS, and NFS improvement and the incidence rate of adverse events.
| Surgical intervention | Procedure | Mean %TBWL | NAS improvement | NFS improvement | Adverse event (%) |
|---|---|---|---|---|---|
| Sleeve gastrectomy | A restrictive bariatric procedure that involves resection of two-thirds of the stomach's greater curvature and gastric fundus creating a long tubular gastric conduit running along the lesser curvature. 76 | −31.7% after 6 mo. 84 | −2.3 NAS score improvement. 81 | Significant reduction in NFS of −0.7. 81 | Nutritional deficiencies (7.4–30.5%). Nondysplastic Barrett's esophagus (15.16%) Choledocholithiasis (5.15%) Cholecystectomy (3%). 87 |
| Roux-en-Y gastric bypass | A reconstructive procedure performed by connecting a limb of the small intestine to a small gastric pouch forming a shape of “ Y .” Bypassing a portion of the large stomach pouch and proximal small intestine reduces the amount of nutrients and calories being absorbed. 77 | −34.6% after 6 mo. 84 |
Improvement or complete resolution of steatosis, steatohepatitis by 91 and 60%, respectively.
78
−2.8 NAS score improvement. 79 |
31% improvement in NFS
78
−1.0 reduction in NFS score. 79 |
Cholelithiasis (32–42%) Nutritional deficiencies (30–35%) Dumping syndrome (13%) Gastro-gastric fistula (1–2%) Small bowel obstruction (1.5–5%). 88 |
Abbreviations: NAS, nonalcoholic fatty liver disease activity score; NFS, nonalcoholic fatty liver disease fibrosis score; TBWL, total body weight loss.
Sleeve Gastrectomy
Sleeve gastrectomy (SG) is a restrictive bariatric procedure that involves a resection of two-thirds of the stomach's greater curvature and gastric fundus creating a long tubular gastric conduit running along the lesser curvature. 107 This results in increased satiation and a reduction in appetite by diminishing the gastric capacity, therefore, facilitating body weight loss. In 2018, SG continued to be the most common bariatric procedure performed in the United States, almost encompassing 61%. 103 Its significant effect on weight loss has been extensively studied in multiple meta-analyses, 78 108 109 and the expected %TWL is 23.4% over a year. 110 Moreover, the ameliorating effect on NAFLD and liver profile overall has been studied. A systematic review and a meta-analysis conducted by Baldwin et al 108 compared Roux-en-Y Gastric Bypass (RYGB) and SG, with their impacts on liver histology, NAS, and NFS. The patients who underwent SG experienced a significant reduction in NAS of −2.3 ( p < 0.00001) without any differences when compared with RYGB. Regarding NFS reduction, both procedures resulted in statistically significant difference ( p < 0.00001) with a mean postoperative NFS reduction of −0.9, when comparing each type of procedure individually, LSG resulted in a mean reduction of −0.7 ( p = 0.07) while RYGB had −1.0 reduction. 108 According to a prospective study conducted on 94 patients with obesity by Salman et al, 111 SG resulted in a statistically significant weight loss with a marked reduction of BMI from 44.54 ± 5.45 kg/m 2 to 34.23 ± 2.66 kg/m 2 ( p < 0.001) in 12 months after the surgery. Additionally, the NAS score significantly decreased from 5.2 ± 1.96 to 2.63 ± 1.55 in 1 year ( p < 0.001). 111 These findings support the utility of SG as part of a treatment regimen for obesity complicated with NAFLD.
Roux-en-Y Gastric Bypass
RYGB is a surgical intervention with a combined restrictive and malabsorptive effects. It is considered to be a gastrointestinal reconstructive procedure performed by connecting a limb of the small intestine to a small gastric pouch forming a shape of the letter “Y.” 112 Bypassing a portion of the large stomach pouch and proximal small intestine reduces the amount of nutrients and calories being absorbed, therefore, creating a caloric deficit that will lead to weight loss. 112 RYGB represents the second most commonly performed bariatric procedure in the United States for the year 2018, after SG comprising 17% (42,945/252,000). 103 The expected %TBWL is 30.9% over a year. 110 Multiple meta-analyses have been performed to understand its effect on NAFLD. 78 108 Fakhry et al 78 conducted a meta-analysis on 21 RCT studies (12/21 were for RYGB) of 2,374 patients investigating the impact of RYGB on NAFLD and comparing it to other types of procedures. They noticed that a higher number of RYGB patients had a regression in NAFLD compared with other surgeries. Precisely, the pooled portion of RYGB candidates who had improvement or complete resolution of steatosis, steatohepatitis, and fibrosis were 91, 60, and 31%, respectively. 78 Another previously mentioned meta-analysis, Baldwin et al, 108 revealed that in only seven out of 20 studies had reported the NAS outcome pre- and postoperatively. Following RYGB surgery, a significant reduction in NAS and NFS of −2.8 ( p < 0.00001) and −1.0 ( p < 0.00001) was accomplished, respectively. However, the head-to-head comparison in NAS between RYGB and SG yielded an insignificant difference, while favoring RYGB results. Overall, their analysis concluded that both RYGB and SG had a positive impact on the liver profile, with failed superiority between the two types of procedures. 108
Acute Liver Decompensation following Bariatric Surgery
As the main indication of bariatric procedures is severe obesity, with or without comorbidities, the coincidence of NAFLD in these patients should be considered. In fact, this indicates the presence of various degrees of liver impairment in those undergoing surgery, warranting prompt, and cautious preoperative surveillance. Surgery is contraindicated in specific liver conditions such as acute liver failure, viral and alcoholic hepatitis. 113 Regarding the prevalence of NAFLD in patients scheduled for bariatric surgery, a prospective cross-sectional study 114 conducted on 184 patients revealed that 84% of them had a degree of NAFLD (22% steatosis; 30.8% mild steatohepatitis; and 32% moderate–severe steatohepatitis). Praveenraj et al 115 demonstrated almost similar findings when studying 134 patients, 65.7% had NAFLD (26.1% steatosis; 33.6% steatohepatitis; and 31.3% hepatic fibrosis). 115 It is noticeable that most patients in both studies have an advanced level of the disease, possessing a higher risk for liver-related mortality. Following bariatric surgery, there is evidence that NAFLD might worsen in up to 12% of patients and might get to fibrosis. 116
Furthermore, patients undergoing malabsorptive-dominant bariatric surgeries are at risk of developing acute hepatic impairment. 117 Consequently, rapidly progressive liver damage might arise following bariatric surgery as a result of drastic weight loss and subsequent protein malnourishment prompting excessive lipolysis, which increases liver metabolism of long-chain fatty acids from visceral adipose tissue. 118 Therefore, thorough preoperative evaluation, weighing the risks (short- and long-term complications) and benefits (weight reduction and comorbidity resolution) in each patient individually, along with proper counseling on the lifelong follow-up commitment are vital steps before considering such invasive surgeries.
Conclusion
NAFLD is the most common chronic liver disease in the developed world, and it is associated with several metabolic comorbidities. Prompt intervention upon diagnosis is crucial to reduce the progression of the disease into an advanced stage. As weight loss is the mainstay in treating NAFLD, this opens the door for a wide variety of possible therapeutic approaches that can be tailored based on the patient's characteristics. This includes diet, exercise, antiobesity medications, and endoscopic and surgical bariatric procedures ( Fig. 2 ). Medications and bariatric interventions provide more sustainable weight loss, which has been evident to have some improvement in the NAFLD activity score and fibrosis. The exact efficacy of these interventions needs to be further evaluated in standardized and well-designed randomized-controlled clinical trials.
Fig. 2.
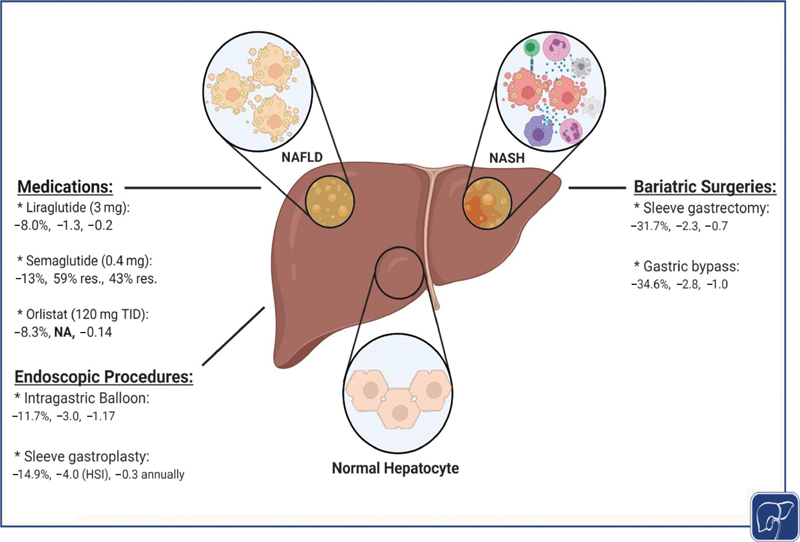
An illustration demonstrating the effect of various weight loss interventions on %TBWL, NAS, and NFS improvements, respectively. “res.” indicates complete resolution of NASH and fibrosis, sequentially. NASH, nonalcoholic steatohepatitis; NFS, nonalcoholic fatty liver disease fibrosis score; TBWL, total body weight loss.
Main Concepts and Learning Points
Most of the available interventions that lead to ≥ 5 to 10% weight loss result in substantial improvement in nonalcoholic fatty liver disease activity (NAFLD) scores (NAS) and NAFLD fibrosis scores (NFS) and even lead to complete NAFLD resolution.
Antiobesity medications, especially liraglutide, semaglutide, and orlistat, improve NAS and NFS scores.
Bariatric endoscopy and surgery achieved the highest and most sustainable weight loss and resulted in significant improvement in NAS and fibrosis scores.
Bariatric endoscopy and surgery provide higher odds for NAFLD resolution.
Funding Statement
Funding A.A. is supported by NIH (NIH K23-DK114460, C-Sig P30DK84567), ANMS Career Development Award, Mayo Clinic Center for Individualized Medicine—Gerstner Career Development Award and Magnus Trust.
Footnotes
Conflicts of Interest A.A. is a stockholder in Gila Therapeutics, and Phenomix Sciences; he serves as a consultant for Rhythm Pharmaceuticals, General Mills.
References
- 1.Kyle T K, Dhurandhar E J, Allison D B. Regarding obesity as a disease: evolving policies and their implications. Endocrinol Metab Clin North Am. 2016;45(03):511–520. doi: 10.1016/j.ecl.2016.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Holly J MP, Biernacka K, Perks C M. Systemic metabolism, its regulators, and cancer: past mistakes and future potential. Front Endocrinol (Lausanne) 2019;10:65. doi: 10.3389/fendo.2019.00065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Flegal K M, Kit B K, Orpana H, Graubard B I. Association of all-cause mortality with overweight and obesity using standard body mass index categories: a systematic review and meta-analysis. JAMA. 2013;309(01):71–82. doi: 10.1001/jama.2012.113905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Adult Obesity Facts. CDCPublished 2020. Accessed April 7, 2021 at:https://www.cdc.gov/obesity/data/adult.html
- 5.Center of Disease ControlAccessed April 5, 2021 at:http://www.cdc.gov/media/pressrel/2008/r080624.htm
- 6.Wang Y, Beydoun M A, Liang L, Caballero B, Kumanyika S K. Will all Americans become overweight or obese? Estimating the progression and cost of the US obesity epidemic. Obesity (Silver Spring) 2008;16(10):2323–2330. doi: 10.1038/oby.2008.351. [DOI] [PubMed] [Google Scholar]
- 7.Camilleri M, Malhi H, Acosta A. Gastrointestinal complications of obesity. Gastroenterology. 2017;152(07):1656–1670. doi: 10.1053/j.gastro.2016.12.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Seto W K. Chronic hepatitis B and metabolic risk factors: a call for rigorous longitudinal studies. World J Gastroenterol. 2019;25(03):282–286. doi: 10.3748/wjg.v25.i3.282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Adams L A, Angulo P, Lindor K D. Nonalcoholic fatty liver disease. CMAJ. 2005;172(07):899–905. doi: 10.1503/cmaj.045232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mitra S, De A, Chowdhury A. Epidemiology of non-alcoholic and alcoholic fatty liver diseases. Transl Gastroenterol Hepatol. 2020;5:16. doi: 10.21037/tgh.2019.09.08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kanwal F, Kramer J R, Duan Z, Yu X, White D, El-Serag H B. Trends in the burden of nonalcoholic fatty liver disease in a United States cohort of veterans. Clin Gastroenterol Hepatol. 2016;14(02):301–8.e1, 2. doi: 10.1016/j.cgh.2015.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Divella R, Mazzocca A, Daniele A, Sabbà C, Paradiso A. Obesity, nonalcoholic fatty liver disease and adipocytokines network in promotion of cancer. Int J Biol Sci. 2019;15(03):610–616. doi: 10.7150/ijbs.29599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dai W, Ye L, Liu A. Prevalence of nonalcoholic fatty liver disease in patients with type 2 diabetes mellitus: a meta-analysis. Medicine (Baltimore) 2017;96(39):e8179. doi: 10.1097/MD.0000000000008179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Buzzetti E, Pinzani M, Tsochatzis E A. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD) Metabolism. 2016;65(08):1038–1048. doi: 10.1016/j.metabol.2015.12.012. [DOI] [PubMed] [Google Scholar]
- 15.Dumitrascu D L, Neuman M G. Non-alcoholic fatty liver disease: an update on diagnosis. Clujul Med. 2018;91(02):147–150. doi: 10.15386/cjmed-993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.NASH Clinical Research Network (CRN) . Brunt E M, Kleiner D E, Wilson L A, Belt P, Neuschwander-Tetri B A, Network N CR. Nonalcoholic fatty liver disease (NAFLD) activity score and the histopathologic diagnosis in NAFLD: distinct clinicopathologic meanings. Hepatology. 2011;53(03):810–820. doi: 10.1002/hep.24127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Angulo P, Hui J M, Marchesini G. The NAFLD fibrosis score: a noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology. 2007;45(04):846–854. doi: 10.1002/hep.21496. [DOI] [PubMed] [Google Scholar]
- 18.Hashemi S-A, Alavian S-M, Gholami-Fesharaki M. Assessment of transient elastography (FibroScan) for diagnosis of fibrosis in non-alcoholic fatty liver disease: a systematic review and meta-analysis. Caspian J Intern Med. 2016;7(04):242–252. [PMC free article] [PubMed] [Google Scholar]
- 19.Loomba R, Cui J, Wolfson T. Novel 3D magnetic resonance elastography for the noninvasive diagnosis of advanced fibrosis in NAFLD: a prospective study. Am J Gastroenterol. 2016;111(07):986–994. doi: 10.1038/ajg.2016.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang X M, Zhang X J, Ma L. Diagnostic performance of magnetic resonance technology in detecting steatosis or fibrosis in patients with nonalcoholic fatty liver disease: a meta-analysis. Medicine (Baltimore) 2018;97(21):e10605. doi: 10.1097/MD.0000000000010605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nalbantoglu I L, Brunt E M. Role of liver biopsy in nonalcoholic fatty liver disease. World J Gastroenterol. 2014;20(27):9026–9037. doi: 10.3748/wjg.v20.i27.9026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dunn W, Xu R, Wingard D L. Suspected nonalcoholic fatty liver disease and mortality risk in a population-based cohort study. Am J Gastroenterol. 2008;103(09):2263–2271. doi: 10.1111/j.1572-0241.2008.02034.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.HEPAmet Registry . Ampuero J, Aller R, Gallego-Durán R. Significant fibrosis predicts new-onset diabetes mellitus and arterial hypertension in patients with NASH. J Hepatol. 2020;73(01):17–25. doi: 10.1016/j.jhep.2020.02.028. [DOI] [PubMed] [Google Scholar]
- 24.Ganguli S, DeLeeuw P, Satapathy S K. A review of current and upcoming treatment modalities in non-alcoholic fatty liver disease and non-alcoholic steatohepatitis. Hepat Med. 2019;11:159–178. doi: 10.2147/HMER.S188991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Acosta A, Streett S, Kroh M D. White Paper AGA: POWER—practice guide on obesity and weight management, education, and resources. Clin Gastroenterol Hepatol. 2017;15(05):631–6.49E12. doi: 10.1016/j.cgh.2016.10.023. [DOI] [PubMed] [Google Scholar]
- 26.Hannah W N, Jr, Harrison S A. Effect of weight loss, diet, exercise, and bariatric surgery on nonalcoholic fatty liver disease. Clin Liver Dis. 2016;20(02):339–350. doi: 10.1016/j.cld.2015.10.008. [DOI] [PubMed] [Google Scholar]
- 27.St George A, Bauman A, Johnston A, Farrell G, Chey T, George J. Effect of a lifestyle intervention in patients with abnormal liver enzymes and metabolic risk factors. J Gastroenterol Hepatol. 2009;24(03):399–407. doi: 10.1111/j.1440-1746.2008.05694.x. [DOI] [PubMed] [Google Scholar]
- 28.Gepner Y, Shelef I, Komy O. The beneficial effects of Mediterranean diet over low-fat diet may be mediated by decreasing hepatic fat content. J Hepatol. 2019;71(02):379–388. doi: 10.1016/j.jhep.2019.04.013. [DOI] [PubMed] [Google Scholar]
- 29.Sun W-H, Song M-Q, Jiang C-Q. Lifestyle intervention in non-alcoholic fatty liver disease in Chengyang District, Qingdao, China. World J Hepatol. 2012;4(07):224–230. doi: 10.4254/wjh.v4.i7.224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Promrat K, Kleiner D E, Niemeier H M. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology. 2010;51(01):121–129. doi: 10.1002/hep.23276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Eckard C, Cole R, Lockwood J. Prospective histopathologic evaluation of lifestyle modification in nonalcoholic fatty liver disease: a randomized trial. Therap Adv Gastroenterol. 2013;6(04):249–259. doi: 10.1177/1756283X13484078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ueno T, Sugawara H, Sujaku K. Therapeutic effects of restricted diet and exercise in obese patients with fatty liver. J Hepatol. 1997;27(01):103–107. doi: 10.1016/s0168-8278(97)80287-5. [DOI] [PubMed] [Google Scholar]
- 33.Yasutake K, Nakamuta M, Shima Y. Nutritional investigation of non-obese patients with non-alcoholic fatty liver disease: the significance of dietary cholesterol. Scand J Gastroenterol. 2009;44(04):471–477. doi: 10.1080/00365520802588133. [DOI] [PubMed] [Google Scholar]
- 34.Assy N, Nasser G, Kamayse I. Soft drink consumption linked with fatty liver in the absence of traditional risk factors. Can J Gastroenterol. 2008;22(10):811–816. doi: 10.1155/2008/810961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Musso G, Cassader M, Rosina F, Gambino R. Impact of current treatments on liver disease, glucose metabolism and cardiovascular risk in non-alcoholic fatty liver disease (NAFLD): a systematic review and meta-analysis of randomised trials. Diabetologia. 2012;55(04):885–904. doi: 10.1007/s00125-011-2446-4. [DOI] [PubMed] [Google Scholar]
- 36.Vilar-Gomez E, Martinez-Perez Y, Calzadilla-Bertot L.Weight loss through lifestyle modification significantly reduces features of nonalcoholic steatohepatitis Gastroenterology 201514902367–7.8E6., quiz e14–e15 [DOI] [PubMed] [Google Scholar]
- 37.Sacks F M, Bray G A, Carey V J. Comparison of weight-loss diets with different compositions of fat, protein, and carbohydrates. N Engl J Med. 2009;360(09):859–873. doi: 10.1056/NEJMoa0804748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Papamiltiadous E S, Roberts S K, Nicoll A J. A randomised controlled trial of a Mediterranean Dietary Intervention for Adults with Non Alcoholic Fatty Liver Disease (MEDINA): study protocol. BMC Gastroenterol. 2016;16:14. doi: 10.1186/s12876-016-0426-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Eslamparast T, Tandon P, Raman M. Dietary composition independent of weight loss in the management of non-alcoholic fatty liver disease. Nutrients. 2017;9(08):E800. doi: 10.3390/nu9080800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ryan M C, Itsiopoulos C, Thodis T. The Mediterranean diet improves hepatic steatosis and insulin sensitivity in individuals with non-alcoholic fatty liver disease. J Hepatol. 2013;59(01):138–143. doi: 10.1016/j.jhep.2013.02.012. [DOI] [PubMed] [Google Scholar]
- 41.Riebe D, Franklin B A, Thompson P D. Updating ACSM's recommendations for exercise preparticipation health screening. Med Sci Sports Exerc. 2015;47(11):2473–2479. doi: 10.1249/MSS.0000000000000664. [DOI] [PubMed] [Google Scholar]
- 42.American College of Cardiology/American Heart Association Task Force on Practice Guidelines Obesity Society Jensen M D, Ryan D H, Apovian C M.2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society J Am Coll Cardiol 201463(25 Pt B):2985–3023. [DOI] [PubMed] [Google Scholar]
- 43.Heymsfield S B, Wadden T A. Mechanisms, pathophysiology, and management of obesity. N Engl J Med. 2017;376(15):1492. doi: 10.1056/NEJMc1701944. [DOI] [PubMed] [Google Scholar]
- 44.Heymsfield S B, Wadden T A. Mechanisms, pathophysiology, and management of obesity. N Engl J Med. 2017;376(03):254–266. doi: 10.1056/NEJMra1514009. [DOI] [PubMed] [Google Scholar]
- 45.Ochner C N, Tsai A G, Kushner R F, Wadden T A. Treating obesity seriously: when recommendations for lifestyle change confront biological adaptations. Lancet Diabetes Endocrinol. 2015;3(04):232–234. doi: 10.1016/S2213-8587(15)00009-1. [DOI] [PubMed] [Google Scholar]
- 46.Hall K D. Metabolic adaptations to weight loss. Obesity (Silver Spring) 2018;26(05):790–791. doi: 10.1002/oby.22189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Daneschvar H L, Aronson M D, Smetana G W. FDA-approved anti-obesity drugs in the United States. Am J Med. 2016;129(08):8790–8.79E8. doi: 10.1016/j.amjmed.2016.02.009. [DOI] [PubMed] [Google Scholar]
- 48.Gryskiewicz K A, Coleman C I. Exenatide. Formulary. 2005;40(03):86. [Google Scholar]
- 49.SCALE Obesity and Prediabetes NN8022-1839 Study Group . Pi-Sunyer X, Astrup A, Fujioka K. A randomized, controlled trial of 3.0 mg of liraglutide in weight management. N Engl J Med. 2015;373(01):11–22. doi: 10.1056/NEJMoa1411892. [DOI] [PubMed] [Google Scholar]
- 50.Iepsen E W, Torekov S S, Holst J J. Liraglutide for type 2 diabetes and obesity: a 2015 update. Expert Rev Cardiovasc Ther. 2015;13(07):753–767. doi: 10.1586/14779072.2015.1054810. [DOI] [PubMed] [Google Scholar]
- 51.LEAN trial team Armstrong M J, Gaunt P, Aithal G P.Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study Lancet 2016387(10019):679–690. [DOI] [PubMed] [Google Scholar]
- 52.Newsome P N, Buchholtz K, Cusi K. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med. 2021;384(12):1113–1124. doi: 10.1056/NEJMoa2028395. [DOI] [PubMed] [Google Scholar]
- 53.Guerciolini R. Mode of action of orlistat. Int J Obes Relat Metab Disord. 1997;21 03:S12–S23. [PubMed] [Google Scholar]
- 54.Hutton B, Fergusson D. Changes in body weight and serum lipid profile in obese patients treated with orlistat in addition to a hypocaloric diet: a systematic review of randomized clinical trials. Am J Clin Nutr. 2004;80(06):1461–1468. doi: 10.1093/ajcn/80.6.1461. [DOI] [PubMed] [Google Scholar]
- 55.Assy N, Hussein O, Abassi Z. Weight loss induced by orlistat reverses fatty infiltration and improves hepatic fibrosis in obese patients with non-alcoholic steatohepatitis. Gut. 2007;56(03):443–444. doi: 10.1136/gut.2006.106021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Harrison S A, Fecht W, Brunt E M, Neuschwander-Tetri B A. Orlistat for overweight subjects with nonalcoholic steatohepatitis: a randomized, prospective trial. Hepatology. 2009;49(01):80–86. doi: 10.1002/hep.22575. [DOI] [PubMed] [Google Scholar]
- 57.Harrison S A, Fincke C, Helinski D, Torgerson S, Hayashi P. A pilot study of orlistat treatment in obese, non-alcoholic steatohepatitis patients. Aliment Pharmacol Ther. 2004;20(06):623–628. doi: 10.1111/j.1365-2036.2004.02153.x. [DOI] [PubMed] [Google Scholar]
- 58.Kelley D E, Kuller L H, McKolanis T M, Harper P, Mancino J, Kalhan S. Effects of moderate weight loss and orlistat on insulin resistance, regional adiposity, and fatty acids in type 2 diabetes. Diabetes Care. 2004;27(01):33–40. doi: 10.2337/diacare.27.1.33. [DOI] [PubMed] [Google Scholar]
- 59.Zelber-Sagi S, Kessler A, Brazowsky E. A double-blind randomized placebo-controlled trial of orlistat for the treatment of nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol. 2006;4(05):639–644. doi: 10.1016/j.cgh.2006.02.004. [DOI] [PubMed] [Google Scholar]
- 60.Greydanus D E, Bricker L A, Feucht C.Pharmacotherapy for obese adolescents Pediatr Clin North Am 20115801139–153., xi. [DOI] [PubMed] [Google Scholar]
- 61.Rothman R B, Baumann M H. Appetite suppressants, cardiac valve disease and combination pharmacotherapy. Am J Ther. 2009;16(04):354–364. doi: 10.1097/MJT.0b013e31817fde95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Cosentino G, Conrad A O, Uwaifo G I. Phentermine and topiramate for the management of obesity: a review. Drug Des Devel Ther. 2011;7:267–278. doi: 10.2147/DDDT.S31443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Verrotti A, Scaparrotta A, Agostinelli S, Di Pillo S, Chiarelli F, Grosso S. Topiramate-induced weight loss: a review. Epilepsy Res. 2011;95(03):189–199. doi: 10.1016/j.eplepsyres.2011.05.014. [DOI] [PubMed] [Google Scholar]
- 64.Astrup A, Caterson I, Zelissen P. Topiramate: long-term maintenance of weight loss induced by a low-calorie diet in obese subjects. Obes Res. 2004;12(10):1658–1669. doi: 10.1038/oby.2004.206. [DOI] [PubMed] [Google Scholar]
- 65.Lonneman D J, Jr, Rey J A, McKee B D. Phentermine/Topiramate extended-release capsules (qsymia) for weight loss. P&T. 2013;38(08):446–452. [PMC free article] [PubMed] [Google Scholar]
- 66.Acosta A, Camilleri M, Shin A. Quantitative gastrointestinal and psychological traits associated with obesity and response to weight-loss therapy. Gastroenterology. 2015;148(03):537–5.46E6. doi: 10.1053/j.gastro.2014.11.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Allison D B, Gadde K M, Garvey W T. Controlled-release phentermine/topiramate in severely obese adults: a randomized controlled trial (EQUIP) Obesity (Silver Spring) 2012;20(02):330–342. doi: 10.1038/oby.2011.330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gadde K M, Xiong G L. Bupropion for weight reduction. Expert Rev Neurother. 2007;7(01):17–24. doi: 10.1586/14737175.7.1.17. [DOI] [PubMed] [Google Scholar]
- 69.Greenway F L, Whitehouse M J, Guttadauria M. Rational design of a combination medication for the treatment of obesity. Obesity (Silver Spring) 2009;17(01):30–39. doi: 10.1038/oby.2008.461. [DOI] [PubMed] [Google Scholar]
- 70.Christou G A, Kiortsis D N. The efficacy and safety of the naltrexone/bupropion combination for the treatment of obesity: an update. Hormones (Athens) 2015;14(03):370–375. doi: 10.14310/horm.2002.1600. [DOI] [PubMed] [Google Scholar]
- 71.Sherman M M, Ungureanu S, Rey J A. Naltrexone/Bupropion ER (Contrave): newly approved treatment option for chronic weight management in obese adults. P&T. 2016;41(03):164–172. [PMC free article] [PubMed] [Google Scholar]
- 72.FDA Food and Drug Administration FDA approves weight-management drug Contrave 2014. Published 2014. Accessed April 7, 2021 at:www.fda.gov/newsevents/newsroom/pressannouncements/ucm413896.htm
- 73.COR-I Study Group Greenway F L, Fujioka K, Plodkowski R A.Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial Lancet 2010376(9741):595–605. [DOI] [PubMed] [Google Scholar]
- 74.Chalasani . Allison Winokur A H, Dybala C, Lam H, Chen S, Naga P. Naltrexone/bupropion extended-release 32 mg/360 mg significantly improves liver enzymes in obese/overweight individuals with elevated liver enzymes. Hepatology. 2015;62:1268A. [Google Scholar]
- 75.Bajaj H S, Burrows M, Blavignac J. Extended-release naltrexone/bupropion and liver health: pooled, post hoc analysis from four randomized controlled trials. Diabetes Obes Metab. 2021;23(03):861–865. doi: 10.1111/dom.14284. [DOI] [PubMed] [Google Scholar]
- 76.Abu Dayyeh B K, Bazerbachi F, Graupera I, Cardenas A. Endoscopic bariatric and metabolic therapies for non-alcoholic fatty liver disease. J Hepatol. 2019;71(06):1246–1248. doi: 10.1016/j.jhep.2019.07.026. [DOI] [PubMed] [Google Scholar]
- 77.Martins C, Strømmen M, Stavne O A, Nossum R, Mårvik R, Kulseng B. Bariatric surgery versus lifestyle interventions for morbid obesity—changes in body weight, risk factors and comorbidities at 1 year. Obes Surg. 2011;21(07):841–849. doi: 10.1007/s11695-010-0131-1. [DOI] [PubMed] [Google Scholar]
- 78.Fakhry T K, Mhaskar R, Schwitalla T, Muradova E, Gonzalvo J P, Murr M M. Bariatric surgery improves nonalcoholic fatty liver disease: a contemporary systematic review and meta-analysis. Surg Obes Relat Dis. 2019;15(03):502–511. doi: 10.1016/j.soard.2018.12.002. [DOI] [PubMed] [Google Scholar]
- 79.Pajot G, Calderon G, Acosta A. Endoscopic treatments for obesity. Curr Treat Options Gastroenterol. 2017;15(04):660–675. doi: 10.1007/s11938-017-0158-7. [DOI] [PubMed] [Google Scholar]
- 80.American Society for Metabolic and Bariatric Surgery Clinical Issues Committee . Ali M R, Moustarah F, Kim J J. American Society for Metabolic and Bariatric Surgery position statement on intragastric balloon therapy endorsed by the Society of American Gastrointestinal and Endoscopic Surgeons. Surg Obes Relat Dis. 2016;12(03):462–467. doi: 10.1016/j.soard.2015.12.026. [DOI] [PubMed] [Google Scholar]
- 81.Gaur S, Levy S, Mathus-Vliegen L, Chuttani R. Balancing risk and reward: a critical review of the intragastric balloon for weight loss. Gastrointest Endosc. 2015;81(06):1330–1336. doi: 10.1016/j.gie.2015.01.054. [DOI] [PubMed] [Google Scholar]
- 82.Bazerbachi F, Haffar S, Sawas T. Fluid-filled versus gas-filled intragastric balloons as obesity interventions: a network meta-analysis of randomized trials. Obes Surg. 2018;28(09):2617–2625. doi: 10.1007/s11695-018-3227-7. [DOI] [PubMed] [Google Scholar]
- 83.Kim S H, Chun H J, Choi H S, Kim E S, Keum B, Jeen Y T. Current status of intragastric balloon for obesity treatment. World J Gastroenterol. 2016;22(24):5495–5504. doi: 10.3748/wjg.v22.i24.5495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.ORBERA™ Intragastric Balloon System—P140008Published. Accessed April 6, 2021 at:http://wayback.archive-it.org/7993/20170111141139/http:/www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/DeviceApprovalsandClearances/Recently-ApprovedDevices/ucm457416.htm2015
- 85.NASH News March 2021. —Recognizing NASH patient advocacy. Accessed April 5, 2021 at:https://www.globalliver.org/news/2021/nash-news-march
- 86.Bazerbachi F, Vargas E J, Rizk M. Intragastric balloon placement induces significant metabolic and histologic improvement in patients with nonalcoholic steatohepatitis. Clin Gastroenterol Hepatol. 2020;19(01):146–154. doi: 10.1016/j.cgh.2020.04.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Popov V B, Thompson C C, Kumar N, Ciarleglio M M, Deng Y, Laine L. Effect of intragastric balloons on liver enzymes: a systematic review and meta-analysis. Dig Dis Sci. 2016;61(09):2477–2487. doi: 10.1007/s10620-016-4178-2. [DOI] [PubMed] [Google Scholar]
- 88.Chandan S, Mohan B P, Khan S R. Efficacy and safety of intragastric balloon (IGB) in non-alcoholic fatty liver disease (NAFLD): a Comprehensive review and meta-analysis. Obes Surg. 2021;31(03):1271–1279. doi: 10.1007/s11695-020-05084-0. [DOI] [PubMed] [Google Scholar]
- 89.ASGE Bariatric Endoscopy Task Force ; ASGE Technology Committee . Abu Dayyeh B K, Edmundowicz S A, Jonnalagadda S. Endoscopic bariatric therapies. Gastrointest Endosc. 2015;81(05):1073–1086. doi: 10.1016/j.gie.2015.02.023. [DOI] [PubMed] [Google Scholar]
- 90.Lopez-Nava G, Galvão M P, Bautista-Castaño I, Jimenez-Baños A, Fernandez-Corbelle J P. Endoscopic sleeve gastroplasty: how I do it? Obes Surg. 2015;25(08):1534–1538. doi: 10.1007/s11695-015-1714-7. [DOI] [PubMed] [Google Scholar]
- 91.Abu Dayyeh B K, Acosta A, Camilleri M. Endoscopic sleeve gastroplasty alters gastric physiology and induces loss of body weight in obese individuals. Clin Gastroenterol Hepatol. 2017;15(01):37–430. doi: 10.1016/j.cgh.2015.12.030. [DOI] [PubMed] [Google Scholar]
- 92.Sharaiha R Z, Kumta N A, Saumoy M. Endoscopic sleeve gastroplasty significantly reduces body mass index and metabolic complications in obese patients. Clin Gastroenterol Hepatol. 2017;15(04):504–510. doi: 10.1016/j.cgh.2016.12.012. [DOI] [PubMed] [Google Scholar]
- 93.Singh S, Hourneaux de Moura D T, Khan A, Bilal M, Ryan M B, Thompson C C. Safety and efficacy of endoscopic sleeve gastroplasty worldwide for treatment of obesity: a systematic review and meta-analysis. Surg Obes Relat Dis. 2020;16(02):340–351. doi: 10.1016/j.soard.2019.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Hajifathalian K, Mehta A, Ang B. Improvement in insulin resistance and estimated hepatic steatosis and fibrosis after endoscopic sleeve gastroplasty. Gastrointest Endosc. 2021;93(05):1110–1118. doi: 10.1016/j.gie.2020.08.023. [DOI] [PubMed] [Google Scholar]
- 95.Hedjoudje A, Abu Dayyeh B K, Cheskin L J. Efficacy and safety of endoscopic sleeve gastroplasty: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2020;18(05):1043–1.053E7. doi: 10.1016/j.cgh.2019.08.022. [DOI] [PubMed] [Google Scholar]
- 96.van Baar A CG, Holleman F, Crenier L. Endoscopic duodenal mucosal resurfacing for the treatment of type 2 diabetes mellitus: one year results from the first international, open-label, prospective, multicentre study. Gut. 2020;69(02):295–303. doi: 10.1136/gutjnl-2019-318349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Theodorakis M J, Carlson O, Michopoulos S. Human duodenal enteroendocrine cells: source of both incretin peptides, GLP-1 and GIP. Am J Physiol Endocrinol Metab. 2006;290(03):E550–E559. doi: 10.1152/ajpendo.00326.2004. [DOI] [PubMed] [Google Scholar]
- 98.Verdam F J, Greve J W, Roosta S. Small intestinal alterations in severely obese hyperglycemic subjects. J Clin Endocrinol Metab. 2011;96(02):E379–E383. doi: 10.1210/jc.2010-1333. [DOI] [PubMed] [Google Scholar]
- 99.de Moura E GH, Ponte-Neto A M, Tsakmaki A, Aiello V D, Bewick G A, Brunaldi V O. Histologic assessment of the intestinal wall following duodenal mucosal resurfacing (DMR): a new procedure for the treatment of insulin-resistant metabolic disease. Endosc Int Open. 2019;7(05):E685–E690. doi: 10.1055/a-0862-0263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Rajagopalan H, Cherrington A D, Thompson C C. Endoscopic duodenal mucosal resurfacing for the treatment of type 2 diabetes: 6-month interim analysis from the first-in-human proof-of-concept study. Diabetes Care. 2016;39(12):2254–2261. doi: 10.2337/dc16-0383. [DOI] [PubMed] [Google Scholar]
- 101.de Oliveira G HP, de Moura D TH, Funari M P. Metabolic effects of endoscopic duodenal mucosal resurfacing: a systematic review and meta-analysis. Obes Surg. 2021;31(03):1304–1312. doi: 10.1007/s11695-020-05170-3. [DOI] [PubMed] [Google Scholar]
- 102.Safety & effectiveness of duodenal mucosal resurfacing (DMR) using the Revita™ system in treatment of type 2 diabetesAccessed April 7, 2021 at:https://clinicaltrials.gov/ct2/show/NCT03653091
- 103.English W J, DeMaria E J, Hutter M M. American Society for Metabolic and Bariatric Surgery 2018 estimate of metabolic and bariatric procedures performed in the United States. Surg Obes Relat Dis. 2020;16(04):457–463. doi: 10.1016/j.soard.2019.12.022. [DOI] [PubMed] [Google Scholar]
- 104.Buchwald H, Avidor Y, Braunwald E. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292(14):1724–1737. doi: 10.1001/jama.292.14.1724. [DOI] [PubMed] [Google Scholar]
- 105.Wolfe B M, Kvach E, Eckel R H. Treatment of obesity: weight loss and bariatric surgery. Circ Res. 2016;118(11):1844–1855. doi: 10.1161/CIRCRESAHA.116.307591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Lassailly G, Caiazzo R, Ntandja-Wandji L C. Bariatric surgery provides long-term resolution of nonalcoholic steatohepatitis and regression of fibrosis. Gastroenterology. 2020;159(04):1290–1.301E8. doi: 10.1053/j.gastro.2020.06.006. [DOI] [PubMed] [Google Scholar]
- 107.Benaiges D, Más-Lorenzo A, Goday A. Laparoscopic sleeve gastrectomy: more than a restrictive bariatric surgery procedure? World J Gastroenterol. 2015;21(41):11804–11814. doi: 10.3748/wjg.v21.i41.11804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Baldwin D, Chennakesavalu M, Gangemi A. Systematic review and meta-analysis of Roux-en-Y gastric bypass against laparoscopic sleeve gastrectomy for amelioration of NAFLD using four criteria. Surg Obes Relat Dis. 2019;15(12):2123–2130. doi: 10.1016/j.soard.2019.09.060. [DOI] [PubMed] [Google Scholar]
- 109.Shoar S, Saber A A. Long-term and midterm outcomes of laparoscopic sleeve gastrectomy versus Roux-en-Y gastric bypass: a systematic review and meta-analysis of comparative studies. Surg Obes Relat Dis. 2017;13(02):170–180. doi: 10.1016/j.soard.2016.08.011. [DOI] [PubMed] [Google Scholar]
- 110.Maciejewski M L, Arterburn D E, Van Scoyoc L. Bariatric surgery and long-term durability of weight loss. JAMA Surg. 2016;151(11):1046–1055. doi: 10.1001/jamasurg.2016.2317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Salman M A, Salman A A, Abdelsalam A. Laparoscopic sleeve gastrectomy on the horizon as a promising treatment modality for NAFLD. Obes Surg. 2020;30(01):87–95. doi: 10.1007/s11695-019-04118-6. [DOI] [PubMed] [Google Scholar]
- 112.Björklund P, Laurenius A, Een E, Olbers T, Lönroth H, Fändriks L. Is the Roux limb a determinant for meal size after gastric bypass surgery? Obes Surg. 2010;20(10):1408–1414. doi: 10.1007/s11695-010-0192-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Friedman L S.Surgery in the patient with liver disease Trans Am Clin Climatol Assoc 2010121192–204., discussion 205 [PMC free article] [PubMed] [Google Scholar]
- 114.Morita S, Neto DdeS, Morita F HA, Morita N K, Lobo S MA. Prevalence of non-alcoholic fatty liver disease and steatohepatitis risk factors in patients undergoing bariatric surgery. Obes Surg. 2015;25(12):2335–2343. doi: 10.1007/s11695-015-1696-5. [DOI] [PubMed] [Google Scholar]
- 115.Praveenraj P, Gomes R M, Kumar S. Prevalence and predictors of non-alcoholic fatty liver disease in morbidly obese south Indian patients undergoing bariatric surgery. Obes Surg. 2015;25(11):2078–2087. doi: 10.1007/s11695-015-1655-1. [DOI] [PubMed] [Google Scholar]
- 116.Lee Y, Doumouras A G, Yu J. Complete resolution of nonalcoholic fatty liver disease after bariatric surgery: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2019;17(06):1040–1.06E14. doi: 10.1016/j.cgh.2018.10.017. [DOI] [PubMed] [Google Scholar]
- 117.Mahawar K K, Parmar C, Graham Y. Monitoring of liver function tests after Roux-en-Y gastric bypass: an examination of evidence base. Obes Surg. 2016;26(10):2516–2522. doi: 10.1007/s11695-016-2280-3. [DOI] [PubMed] [Google Scholar]
- 118.Mocanu I, Carvalhana S, Costa P, Palma R. Liver failure after bariatric surgery–clinical case and literature review. J Gastroenterol Metabol. 2018;1:104. [Google Scholar]
- 119.Wang H, Wang L, Cheng Y, Xia Z, Liao Y, Cao J. Efficacy of orlistat in non-alcoholic fatty liver disease: a systematic review and meta-analysis. Biomed Rep. 2018;9(01):90–96. doi: 10.3892/br.2018.1100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Plodkowski R A, McGarvey M E, Reisinger-Kindle K. Obesity management: clinical review and update of the pharmacologic treatment options. Fed Pract. 2016;33(01):6–16. [PMC free article] [PubMed] [Google Scholar]
- 121.Harpreet S, Bajaj M B, Blavignac J. Extended-release naltrexone/bupropion and liver health: pooled, post-hoc analysis from 4 RCTs. Diabetes Obes Metab. 2021;23(03):861–865. doi: 10.1111/dom.14284. [DOI] [PubMed] [Google Scholar]


