Abstract
COVID-19 pandemic led to a worldwide increase of hospitalizations for interstitial pneumonia with thrombosis complications, endothelial injury and multiorgan disease. Common CT findings include lung bilateral infiltrates, bilateral ground-glass opacities and/or consolidation whilst no current laboratory parameter consents rapidly evaluation of COVID-19 risk and disease severity. In the present work we investigated the association of sFLT-1 and CA 15.3 with endothelial damage and pulmonary fibrosis. Serum sFlt-1 has been associated with endothelial injury and sepsis severity, CA 15.3 seems an alternative marker for KL-6 for fibrotic lung diseases and pulmonary interstitial damage. We analysed 262 SARS-CoV-2 patients with differing levels of clinical severity; we found an association of serum sFlt-1 (ROC AUC 0.902, decision threshold > 90.3 pg/mL, p < 0.001 Sens. 83.9% and Spec. 86.7%) with presence, extent and severity of the disease. Moreover, CA 15.3 appeared significantly increased in COVID-19 severe lung fibrosis (ICU vs NON-ICU patients 42.6 ± 3.3 vs 25.7 ± 1.5 U/mL, p < 0.0001) and was associated with lung damage severity grade (ROC AUC 0.958, decision threshold > 24.8 U/mL, p < 0.0001, Sens. 88.4% and Spec. 91.8%). In conclusion, serum levels of sFlt-1 and CA 15.3 appeared useful tools for categorizing COVID-19 clinical stage and may represent a valid aid for clinicians to better personalise treatment.
Subject terms: Biomarkers, Diseases, Health care, Medical research
Introduction
The novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), responsible for the coronavirus disease 2019 (COVID-19) pandemic, has led to a rapid and extensive worldwide increase in hospitalisations for interstitial pneumonia, with several complications such as thrombosis, endothelial injury and multiorgan disease.
SARS-CoV-2 infection targets nasal, bronchial, epithelial cells and pneumocytes which express the angiotensin-converting enzyme (ACE2) receptor1 for the viral spike (S) protein. Inflammatory signalling molecules are released by infected cells and alveolar macrophages; T lymphocytes, monocytes and neutrophils are recruited on the site of infection and cytokines release enhances inflammatory response. Diffuse alveolar interstitial thickening and interstitial mononuclear inflammatory infiltrates and edema develop, and typical ground-glass opacities appear on CT imaging. Autoptic analysis of lung tissue showed marked fibrotic lung parenchyma remodelling, fibroblast proliferation and airspace obliteration2.
Endotheliitis and presence of viral elements inside endothelial cells has been shown in postmortem analysis of different organs of Covid-19 patients3. SARS-CoV-2 infects capillary endothelial cells due to the expression of ACE2 receptor also by endothelial cells4. Covid-19 endotheliitis has been suggested as an explanation of the systemic impaired microcirculatory function with consequent pro-coagulant state5 and clinical sequelae in SARS-CoV-2 infected patients3.
Common laboratory abnormalities of individuals with COVID-19 include lymphopenia, elevated inflammatory markers (e.g., C-reactive protein, ferritin, tumor necrosis factor-α, IL-1, IL-6), and abnormal coagulation parameters (e.g., elevated D-dimer, prolonged prothrombin time, thrombocytopenia, low fibrinogen)6. Increase of Red Cell Distribution Width (RDW) has been associated with disease severity in hospitalised adults with SARS-CoV-2 infection, previously published RDW thresholds of ≥ 14.5% (RDW-CV) or ≥ 47 fL (RDW-SD) have been associated to increased risk of death7–9.
However, no specific laboratory marker has been so far established for COVID-19 and for evaluation of the risk of COVID-19 pulmonary fibrosis and endothelial dysfunction.
In the present study we analysed blood samples from 262 patients who had tested positive for SARS-CoV-2 infection at different time from diagnosis and with differing clinical severity status and we utilised sFlt-1 and CA 15.3 as circulating markers of endothelial damage and pulmonary fibrosis, respectively. Serum sFlt-1 being the soluble form of Vascular Endothelial Growth Factor receptor (sVEGFR) which binds and antagonizes Vascular Endothelial Growth Factor (VEGF) and Placental Growth Factor (PlGF) signal10 and it has been demonstrated to promote endothelial dysfunction, notably during preeclampsia11. According to our previous results, sFlt-1 is as a biomarker of endothelial injury associated to bacterial sepsis and sepsis severity12 and previous evidence showed it increased in COVID-1913,14. The CA 15-3 is as an alternative marker for KL-6 (serum Krebs von den Lungen-6) in fibrotic lung diseases which increases in pulmonary interstitial damage, fibroblast activity and progression to fibrosis of the lung15,16 and might be of great interest when studying the effects of COVID-19.
Materials and methods
Patients
The present study has been conducted on 262 patients hospitalised in the COVID Division of the Vito Fazzi Hospital of Lecce (Italy), 227 survived and 35 died during the hospitalisation; 101 not hospitalised healthy individuals were used as controls (Table 1).
Table 1.
Characteristics of SARS-CoV-2 patients and controls.
| COVID-19 patients | Control patients | |||
|---|---|---|---|---|
| All | Survivors | Dead | ||
| N Age (years) | 262 66.6 2.1 | 227 68.9 1.4 | 35 72.1 4.4 | 101 60 1.1 |
| Males Age (years) | 155 62 1.1 | 130 62.8 1.9 | 27 68.9 4.9 | 47 59.1 1.4 |
| Females Age (years) | 107 72.1 1.4 | 97 74.5 2.0 | 8 80.7 9.3 | 54 60.0 1.6 |
| Disease severity in hospitalized patients: | ||||
| Dyspnea | 41 (18%) | 25 (71.4%) | ||
| Respiratory Rate (> 30 breaths/min) | 34 (14.9%) | 19 (54%) | ||
| Blood Oxygen Saturation (< 93%) | 47 (20.7%) | 23 (65.7%) | ||
| PaO2:FiO2 (< 300 mm Hg) | 53 (23.3%) | 29 (82.8%) | ||
| Lung Field Infiltrate (> 50%) | 100 (44.1%) | 26 (72.3%) | ||
Serum samples were collected in the Clinical Pathology and Microbiology Laboratory of the Vito Fazzi Hospital of Lecce (Italy); selected patients included individuals positive for SARS-CoV-2 infection (without bacterial co-infection) and healthy individuals undergoing routine clinical investigation as control patients. All leftover serum specimens were anonymised and stored at − 80 °C until further use. Sampling of COVID-19 patients was performed over a time frame of 0–28 days during hospitalisation, from first oro-nasopharyngeal swab positive for SARS-CoV-2 RNA.
The diagnostic criteria for COVID-19 followed the interim or 7th edition guideline of The National Health Commission17 which mainly included epidemiological history, clinical symptom, thoracic CT examination, confirmed presence of SARS-CoV-2 RNA by real-time reverse transcription polymerase chain reaction (RT-PCR) of oro-nasopharyngeal swab.
The severity of the illness was based on the presence of dyspnea, respiratory rate of 30 or more breaths per minutes, blood oxygen saturation of 93% or less, a ratio of PaO2:FiO2 of less than 300 mm Hg, an infiltrate in > 50% of the lung fields. Indicator of severe disease were marked tachypnea, hypoxemia, abnormal lung findings (Table 1) and abnormal laboratory results listed in Table 218,19.
Table 2.
Hematochemical parameters in survived and deceased COVID-19 patients in relation to sFLT-1 quartiles (I–IV).
| I | II | III | IV | ||
|---|---|---|---|---|---|
| 91–99.7 | 99.8–117 | 117.1–142 | > 142.1 | ||
| Lymphocytes (103/µL) | Survivors | 1.6 ± 0.1 | 1.2 ± 0.1 | 1.2 ± 0.1 | 1.1 ± 0.1 |
| Dead | – | 0.3 ± 0.1 | 0.5 ± 0.1 | 0.9 ± 0.1 | |
| Neutrophils (103/µL) | Survivors | 4.8 ± 0.4 | 5.8 ± 0.5 | 7.5 ± 0.9 | 9.4 ± 1.2 |
| Dead | – | 12.9 ± 3.5 | 9.7 ± 3.9 | 9.5 ± 0.9 | |
| RDW-SD (fL) | Survivors | 45.3 ± 1.1 | 44.6 ± 1.2 | 45.8 ± 0.9 | 45.3 ± 1.1 |
| Dead | – | 50.0 ± 2.2 | 45.5 ± 2.3 | 45.4 ± 1.7 | |
| D-Dimer (ng/mL FEU) | Survivors | 3997.6 ± 1142.9 | 1389.6 ± 183.4 | 7306.2 ± 2575.0 | 1863.6 ± 397.8 |
| Dead | – | 1783.0 ± 753.1 | 34,956.0 ± 16,886.8 | 2028.7 ± 465.1 | |
| CRP (mg/L) | Survivors | 34.5 ± 7.2 | 51.9 ± 9.7 | 84.4 ± 13.8 | 118.9 ± 19.1 |
| Dead | – | 117.7 ± 61.2 | 193.6 ± 50.4 | 110.9 35.9 | |
| LDH (U/L) | Survivors | 198.2 ± 15.0 | 234.2 ± 25.5 | 265.2 ± 20.2 | 313.8 ± 31.9 |
| Dead | – | 276.0 ± 75.2 | 341.0 ± 39.7 | 401.2 ± 40.5 | |
| Ferritin (ng/mL) | Survivors | 569.7 ± 59.5 | 793.2 ± 79.0 | 1012.4 ± 126.8 | 1362.9 ± 216.8 |
| Dead | – | 1697.0 ± 1224.7 | 1927.8 ± 738.8 | 1643.1 ± 606.0 | |
| PCT (ng/mL) | Survivors | 0.1 ± 0.05 | 1.5 ± 1.1 | 0.7 ± 0.3 | 1.6 ± 0.7 |
| Dead | – | 1.5 ± 0.5 | 0.5 ± 0.2 | 1.9 ± 0.8 |
Real-time PCR assay for SARS-CoV-2 RNA detection
Oro-nasopharyngeal swabs in UTM were fully automated processed for extraction, amplification and result analysis with ELITe InGenius (ELITechGroup, Torino, Italy) according to manufacturer’s directions. Reverse transcription of SARS-CoV-2 RNA and Real-Time PCR assay were performed in one-step process. From March to June 2020 analysed targets of SARS-CoV-2 were RdRp, E and N genes (GeneFinder COVID-19 Plus RealAmp Kit, OSANG Healthcare); from July 2020 analysed targets were RdRp and ORF-8 genes (SARS-CoV-2 ELITe MGB Kit).
Hematological and biochemical investigation
Complete blood count was performed on Sysmex XN-3100 Automatic Hematology Analyzer (Sysmex, Kobe, Japan) based on the fluorescence flow cytometry and hydrodynamically focused impedance.
Serum levels of biochemicals parameters analysed (C-reactive protein, CRP, ferritin, lactate dehydrogenase, LDH, Interleukine-6, IL-6) were performed on the Cobas® 8000 analyser according to manufacturer’s directions (Roche Diagnostic GmbH, Germany).
Plasmatic levels of PCT (procalcitonin) were determined by Enzyme-Linked Fluorescence Assay (ELFA) with VIDAS® B.R.A.H.M.S. PCT™ system (Biomerieux, Marcy-l’Etoile, France) according to manufacturer recommendations and expressed in ng/mL.
D-dimer was quantified by particle-enhanced immunoturbidimetric assay for the quantitative determination of cross-linked fibrin degradation products according to manufacturer recommendations and expressed in ng/mL FEU (Siemens Healthcare GmbH, Erlangen, Germany).
sFlt-1 and CA 15.3 assay in serum
Serum levels of sFlt-1 and/or CA15.3 were both determined by Electro-Chemi-Luminescence Immuno Assay (ECLIA) fully automated on the immunoanalyzer Elecsys 2010®, Cobas 8000, according to manufacturer recommendations (Roche Diagnostic GmbH, Germany). sFlt-1 was expressed in pg/mL and CA 15.3 was expressed in U/mL; all analysed sera were collected and stored at − 80 °C until assay.
Evaluation of thoracic CT images
Computed Tomography (CT) imaging is the diagnostic tool with the highest sensitivity for COVID-19. In our study, chest CT images of patients were inspected and classified according to a scoring system based on the assessment of grade of pulmonary fibrosis, ground-glass opacities and consolidation extension, and pleural effusion. Lung fibrosis has been categorized as absent (A) or moderate (M), severe (S) or critic (SS) grade, based on the type of lesions and extent of the affected area. The types of lesions were ground-glass opacities, linear opacities, interlobular septal thickening, reticulation, honeycombing and traction bronchiectasis. The extent of each type of lesion was scored based on the number of lung segments affected.
Statistics
Results are reported as means with standard deviation and interquartiles ranges. The Student’s t test was used for comparison between patients and controls; statistically significant differences were established according to p value (< 0.05). Receiver Operating Curve (ROC) and the Area Under the Curve (AUC) were used to establish cut-off and decision rules for serum markers associated to COVID-19. Statistical analysis was performed by MedCalc v19.9.1 statistical software.
Ethics approval and informed consent
All procedures undertaken in this study were in accordance with the ethical standards of the institutional and national research committee as well as the Declaration of Helsinki, its later amendments and other comparable ethical standards. Our study was performed during COVID-19 pandemic on patients and material was collected for diagnostic purposes during patients hospitalisation (secondary use). All leftover serum specimens were anonymised and stored at − 80 °C until further use; patient’s names and IDs were deleted, and researchers analysed only renumbered anonymous data after association to clinical characteristics. In accordance with the Institutional Review Board of the Vito Fazzi Hospital, where the approved study has been conducted, during COVID-19 pandemic the need for informed consent from the patients was waived due to the urgent need for research insight in the context of this rapidly evolving disease.
Ethical compliance with human/animal study
All procedures described in the study have been actuated according to ethical principles for medical research involving human subject stated in the Declaration of Helsinki.
Results
We investigated and analysed the data regarding serum level of sFlt-1, previously described as a serum marker of endothelial dysfunction during bacterial sepsis and CA 15.3, analog of KL-6, for pulmonary fibrosis, in 262 patients affected by SARS-CoV-2 infection (Table 1).
Serum sFlt-1 showed an association with SARS-CoV-2 infection status with a decisive threshold value for positive patients > 90.3 pg/mL, as resulting from the receiver operating characteristic (ROC) curve (AUC 0.902 with p < 0.001, Sensitivity 83.9%, Specificity 86.7%; Fig. 1).
Figure 1.
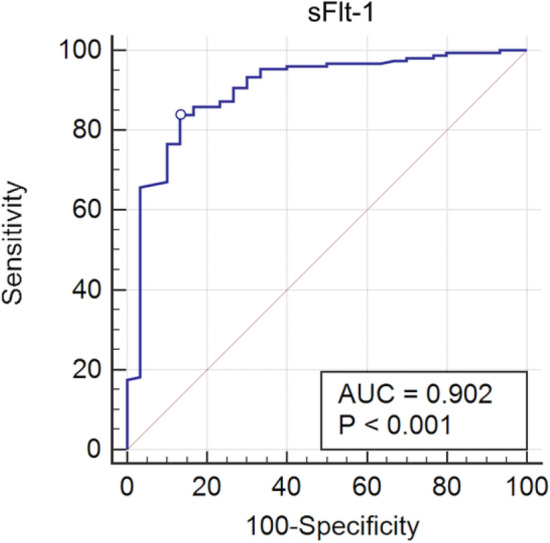
Receiver operating characteristic (ROC) analysis of sFlt-1 serum levels between SARS-CoV-2 infected and non-infected patients; the circle indicates the cut-off point (90.3 pg/mL) with 83.89% Sensitivity and 86.67% Specificity for positive patients. AUC denotes area under the curve.
Stratifying patients according to the time that passed from the beginning of the disease (first positive swab) and disease outcome, sFlt-1appeared associated with the extent and severity of the disease. A significant increase has been found starting from the third day of hospitalisation in patients with the poorest outcomes (122.4 ± 6.9 vs 155.6 ± 10.6 pg/mL survived vs died, p = 0.01), especially, after second week of the disease (up to an average value of 199.4 ± 32.4 pg/mL, at second week, and 199.0 ± 12.0 pg/mL, at third week) in patients with poorest outcome (Fig. 2). In control patients we observed a concentration of 78.9 ± 2.5 pg/mL of sFlt-1.
Figure 2.
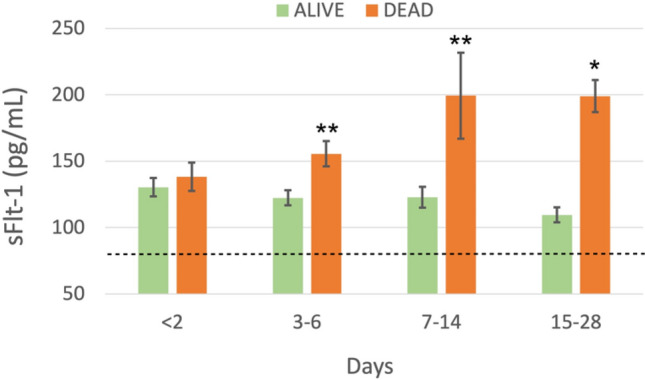
Longitudinal analysis of serum levels of sFlt-1 during hospitalisation of SARS-CoV-2 patients, divided in Alive and Dead according to the outcome of the disease. Patients were monitored for four weeks (less than 2 days, 3–6 days, 7–14 days, 15–28 days from first positive detection of SARS-CoV-2 RNA). The dotted line indicates sFlt-1 level observed in Controls (78.9 ± 2.5 pg/mL); asterisks indicate statistically significant difference: *p < 0.05; **p < 0.01.
IL-6, circulating marker associated with extent and severity of the disease, showed a significant increase from the third day of disease in patients with poorest outcome (171.1 ± 68.7 vs 979.4 ± 440.8 pg/mL survived vs died, p = 0.005), especially at the second week of disease, and afterwards, (300.5 ± 103.6 vs 1514.2 ± 464.2 pg/mL, p < 0.0001).
Values of IL-6 showed an increasing trend in higher sFlt-1 quartiles (Table 2). Moreover, laboratory parameters assayed in COVID-19 patients (CRP, ferritin, LDH, D-dimer, lymphocytes and neutrophils count, RDW-SD, PCT) showed a trend of association with the sFlt-1 interquartile values (Table 2). As reported in Table 2, for poor outcome patients, severe lymphopenia, neutrophilia, increase of RDW-SD were observed for second quartile values of sFlt-1 (99.8–117 pg/mL). RDW-SD (fL) evaluation was used for RDW assessment since it is an independent statistical index, not affected by level of MCV, as the coefficient of variation of red blood cell distribution (RDW-CV)8. Severe increase of serum level of CRP, LDH, ferritin and D-dimer were measured in association to third quartile values of sFlt-1 (117.1–142 pg/mL). Patients who survived the virus showed analogous increase of several biochemical parameters (CRP, LDH, ferritin) for fourth quartile values of sFlt-1 (> 142.1 pg/mL).
We further investigated the possible use of CA 15.3 as marker of pulmonary interstitial damage and progression to fibrosis of the lung in SARS-CoV-2 infection; we found a significant increase of CA 15.3 in SARS-CoV-2 infected patients and a correlation with the severity of clinical conditions. CA 15.3 serum levels of patients hospitalised in Intensive Care ward presented a significant 40% increase compared to patients who were hospitalised in different ward (42.6 ± 3.3 vs 25.7 ± 1.5U/mL, p < 0.0001; control patients 14.5 ± 0.3U/mL; Fig. 3). We analysed thoracic CT imaging of the same patients and they presented a severe grade of lung ground-glass opacities, signs of interstitial damage and presence of fibrosis. Although distinguishing between UIP (Usual Interstitial Pneumonia), NSIP (Non-specific Interstitial Pneumonia) and chronic fibrotic hypersensitivity pneumonitis is often difficult due to significant overlap of the radiological features and often requires bronchoalveolar lavage (BAL) examination and/or surgical lung biopsy20, in our patients the integrated clinical features and High-Resolution Computed Tomography (HRCT) lesions were in accordance with a prevalent NSIP pattern. Some HRTC findings in our patients may be present in Acute Fibrinous and Organising Pneumonia (AFOP), a histologic entity of lung injury with the hallmark features of intra-alveolar fibrin deposits associated with pneumonia, type II pneumocyte hyperplasia and a patchy lymphohistiocytic proliferations21. Although our patients were not submitted to open lung biopsy for the histological diagnosis of their diffuse parenchymal lung disorder, AFOP has been confidently excluded based on the thorough clinical history and extended laboratory diagnostic workup, the shorter duration of the disease, the absence of underlying hematologic disorders and other possible etiologic and risk factors to whom AFOP has been associated to date and the rarity of this entity (Gomes et al. reported just 13 cases of AFOP in a tertiary referral hospital over a period of 14 years)22. Correlation of the CT severity grade with the serum level of CA 15.3 of all COVID-19 patients revealed a noteworthy association, the ROC curve analysis exhibited 0.958 AUC value (p < 0.0001, Sensitivity 88.4%, Specificity 91.8%) and > 24.8 U/mL cut-off value, which significantly differentiated severe/critic pulmonary interstitial damage in SARS-CoV-2 infection (Fig. 4). Conversely, the absence of pulmonary fibrosis in COVID-19 was associated with a CA 15.3 serum level < 13.9 U/mL (p < 0.0001, AUC 0.822, Sensitivity 82.4% and Specificity 83.3%).
Figure 3.
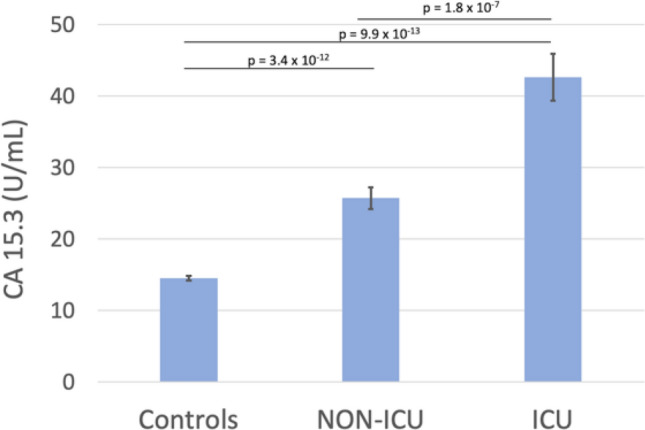
CA 15.3 serum levels in SARS-CoV-2 infected patients, divided according to their hospitalisation in Intensive Care Unit (ICU) or other in different wards (NON-ICU), and control patients.
Figure 4.
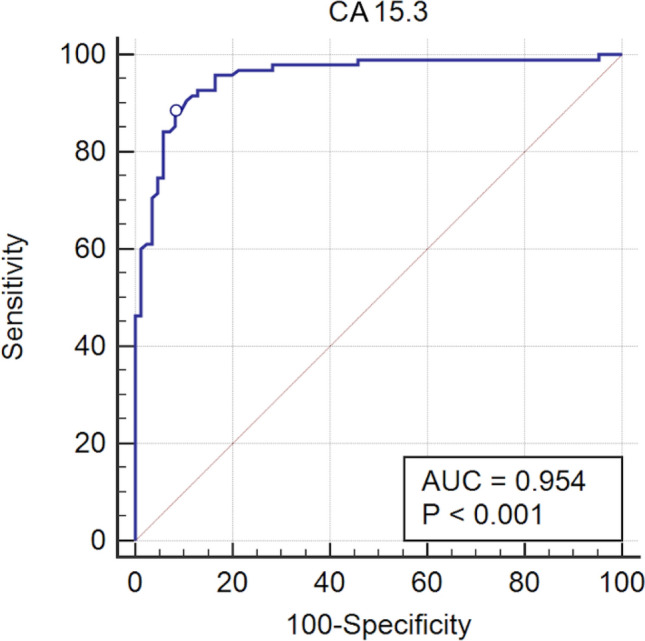
Receiver operating characteristic (ROC) analysis of sFlt-1 serum levels between severe and non-severe SARS-CoV-2 infected patients; the circle indicates the cut-off point (24.8 U/mL) with 88.4% Sensitivity and 91.8% Specificity for positive patients. AUC denotes area under the curve.
By dividing patients according to the lung fibrosis severity grade CT imaging (i.e., absent, moderate, severe or critic grade of pulmonary ground-glass opacities) a statistically significant increase of serum levels of CA 15.3 was observed in SS and in S patients compared to M patients (i.e., SS, 110.0 ± 6.9 U/mL; S, 43.2 ± 2.2 U/mL; M, 18.4 ± 0.7U/mL and A, 11.6 1.5 U/mL ± 1.5; p < 0.0001 in all comparisons; Fig. 5).
Figure 5.
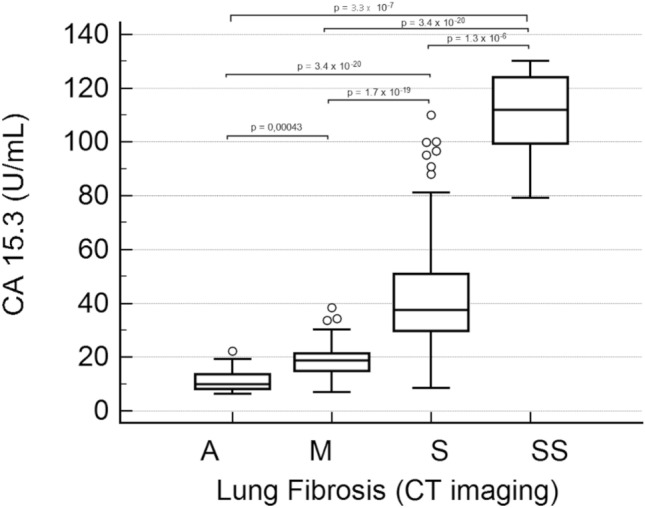
CA 15.3 serum levels in SARS-CoV-2 infected patients according to lung fibrosis and damage severity grade obtained by CT imaging. A, absence of lung fibrosis; M, moderate grade; S, severe grade; SS; critic grade. Boxes represent first and third quartiles (25 to 75 percentile), bands within boxes represent the median value, whiskers display ranges of 1.5 interquartile ranges from the end of the box, outside values are displayed as separate points.
Discussion
We found a significant increase of sFlt-1, a marker of endothelial dysfunction previously associated with bacterial sepsis12, in SARS-CoV-2 infected patients studied from March 2020 to February 2021 during COVID-19 pandemic. Decision threshold value for positive patients was > 90.3 pg/mL (Fig. 1), in accordance with previous results (95 pg/mL) found in respiratory failure of COVID-19 patients13,23 and with the finding of high sFlt-1 circulating levels in severe disease14. In the present study, patients who died during the second or third week of disease, showed significant increase of sFlt-1 serum levels compared to surviving patients at the same week of observation (Fig. 2). In presence of bacterial sepsis previous evidence showed that sFlt-1 values over 200 pg/mL12. Libby et al.24 defined COVID-19 as an endothelial disease; impaired endothelial barrier function in COVID-19 appears to contribute to protein and fluid accumulation in the alveolar space and impaired oxygenation of the blood. Endothelial dysfunction and consequent uneven balance of prothrombotic/antithrombotic action contribute to thrombosis in situ observed in the pulmonary vasculature of COVID patients25 and predispose them to thrombosis events in all arterial beds within the microvasculature, including that of the coronary, kidneys and cerebral circulation. Venous thrombosis and pulmonary embolism commonly complicate COVID-19 and are related to unbalanced endothelial functions26. In the present study sFlt-1 higher quartiles were associated with higher concentrations of IL-6 and the worsening of other hematochemical parameters including D-dimer, LDH, Ferritin, CRP, lymphopenia, neutrophilia, RDW-SD, especially when evaluated in severe disease compared to mild or moderate disease (survived patients) (Table2).
RDW reflects the variation of red blood cell (RBC) volume and it implicates an increased rate of RBC destruction, dysfunctional erythropoiesis and/or shortened RBC lifespan which has been suggested like a compensatory up-regulation due to excessive blood hypoxemia7 or, as suggested by others, like a consequence of counterregulatory changes of RBC production and turnover due to increased production and turnover of leukocytes or platelets, such as would occur in inflammation9. Our results showed a significant increase of RDW-SD in deceased COVID-19 patients compared to those who survived (47.8 fL vs 45.4, p = 0.042); highest values for RDW-SD were found in deceased patients with II quartile sFlt-1 levels (Table 2), as observed for leukocytes values (lymphocytes and neutrophils, Table 2). Inflammatory status negatively affects endothelium leading to its dysfunction and, in turn, to impaired angiogenesis and coagulation25,27.
In lung tissue from non-survivors with COVID-19, as with influenza, Ackermann et al. showed that microvascular injuries (endothelialitis) and pathologic inflammation induced a significant higher level of intussusceptive angiogenesis with tissue over-expression of pro-angiogenic factors25. According to the authors, vascular angiogenesis distinguished the pulmonary pathobiology of COVID-19 from that of equally severe influenza virus infection, moreover the increased degree of intussusceptive angiogenesis appeared significantly correlated with increasing duration of hospitalization. Implication of sFlt-1 in angiogenesis or its competition with membrane bound Flt-1 for binding VEGF and potential role of decoy molecule in the angiogenic process appears an interesting topic for next investigation. In the present study it could not be assessed since patients were not submitted to open lung biopsy for the histological evaluation of angiogenesis.
High serum levels of the inflammatory cytokine IL-6 are associated to the SARS-CoV-2 infection and severity28–30. IL-1a and IL-1b, IL-6, and TNF-α, and other components of the inflammatory cascade contribute to host defence against infections although an excessive synthesis determines the cytokines storm, a severe acute systemic inflammatory response which confers increased risks of vascular hyperpermeability, multiorgan failure, and eventually death31.
In this context, the potential use of sFlt-1 as a marker of endothelial dysfunction opens the possibility of new diagnostic and treatment strategy of COVID-19 patients.
Lung findings of patients who died with COVID-19 showed that the main pathological feature is an early-phase or intermediate-phase diffuse alveolar damage, marked fibrotic lung parenchymal remodeling, characterized by fibroblast proliferation, airspace obliteration, and micro-honeycombing, in addition to the presence of fibrin thrombi in small arterial vessels32. In the present study we correlated the disease severity and lung fibrosis of COVID-19 patients with circulating levels of CA 15.3.
The CA 15.3 has been previously described as alternative marker for KL-6 fibrotic lung disease33, a mucin-like glycoprotein mainly expressed on the type II alveolar epithelial cell surface34 and as marker of pulmonary fibrosis35,36.
Kl-6 is recognized as a prognostic biomarker of Interstitial Lung Disease (ILDs), predicting response to antifibrotic therapies33,37,38, has been correlated with risk of mortality in Acute Respiratory Distress Syndrome (ARDS)39 and reported as indicator of alveolar epithelial cell damage induced by mechanical ventilatory support39. High peripheral levels of this protein have also been reported in Legionella, Pneumocystis jivorecii infections40,41 and measles‐associated pneumonia42, as well as in viral pneumonia43.
Recent evidence indicated serum KL-6 as a biomarker of severe COVID-19, being elevated in severe patients admitted to the Intensive Care Unit (ICU) requiring intubation and mechanical ventilation for diffuse interstitial pneumonia44.
Similarly, Deng et al.45 described KL-6 as sensitive and specific biomarker for distinguishing mild and severe/critical patients correlated to computed tomography lung lesions areas and predicting the prognosis of lung injury of discharged patients.
Kruit et al.16 reported that KL-6 is identical to the target molecule to which antibodies collectively known as CA 15.3 have been developed, obtaining equivalent results for the assay of the two molecules in patients with fibrotic lung disease.
In the present study we observed a very significant association of CA 15.3 with the presence of lung ground-glass opacities and pulmonary lesions typical of thoracic CT imaging of COVID-19 patients with a cut-off value > 24.8 U/mL able to discriminate COVID-19 patients with severe lung fibrosis (Fig. 4).
We found serum levels of CA 15.3 slightly, although significantly, increased in patients with moderate grade of lung fibrosis compared to controls (18.4 ± 0.7 U/mL vs 14.5 ± 2.5 U/mL in M patients vs Controls, p < 0.001); no increase was found in COVID-19 patients with no lung fibrosis. On the other hand, severe and critic lung fibrosis were characterized with a marked and significant increase of CA 15.3 (up to 43.2 ± 2.2 and 110.0 ± 6.9 U/mL average value respectively, Fig. 5). ICU hospitalised patients analysed in the present study had circulating levels of CA 15.3 significantly higher than NON-ICU hospitalised patients (42.6 ± 3.3 vs 25.7 ± 1.5 U/mL, p < 0.0001, Fig. 3). Although CA 15.3 circulating levels tend to increase during hospitalisation, there is no significant difference between poor and good outcome patients at the same week of observation in the time course of the disease. The opposite behaviour of analysed markers of inflammatory and endothelial damage, IL-6 and sFlt-1, further evidences the significant role of inflammation and endothelial damage in determining the severity of COVID-19.
In conclusion, serum level of sFlt-1 and Ca 15.3 can help categorise clinical phase of COVID-19 patients. From our result, sFlt-1 might identify hospitalised patients with a severe clinical condition related to endothelial dysfunction and consequent clinical sequalae, which could help clinicians to better personalise treatment; CA 15.3 appears a promising biomarker for severity of pulmonary involvement in COVID-19. Further studies on expanded number of patients and extended clinical evaluation would help to better understand clinical utility of these biomarkers.
Acknowledgements
We thank Prof. Nigel Paul Wilson for English language editing and revision.
Author contributions
M.G. and G.L. conceptualized and designed the study, wrote and edited the manuscript. S.S., R.A.L. and F.S. collected and analyzed the data; C.C. provided patients characteristics. All authors approved the final article as submitted and agree to be accountable for all aspects of the work.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Hoffmann M, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Grillo F, Barisione E, Ball L, Mastracci L, Fiocca R. Lung fibrosis: An undervalued finding in COVID-19 pathological series. Lancet Infect. Dis. 2021;4:e72. doi: 10.1016/S1473-3099(20)30582-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Varga Z, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ferrario CM, et al. Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation. 2005;111:2605–2610. doi: 10.1161/CIRCULATIONAHA.104.510461. [DOI] [PubMed] [Google Scholar]
- 5.Bonetti PO, Lerman LO, Lerman A. Endothelial dysfunction—a marker of atherosclerotic risk. Arterioscl. Throm. Vas. 2003;23:168–175. doi: 10.1161/01.ATV.0000051384.43104.FC. [DOI] [PubMed] [Google Scholar]
- 6.Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): A review. JAMA. 2020;324:782–793. doi: 10.1001/jama.2020.12839. [DOI] [PubMed] [Google Scholar]
- 7.Karampitsakos T, et al. Increased red cell distribution width is associated with disease severity in hospitalized adults with SARS-CoV-2 infection: An observational multicentric study. Front. Med. 2020;7:616292. doi: 10.3389/fmed.2020.616292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pouladzadeh M, et al. Validation of red cell distribution width as a COVID-19 severity screening tool. Future Sci OA. 2021;7(7):FSO712. doi: 10.2144/fsoa-2020-0199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Foy BH, et al. Association of red blood cell distribution width with mortality risk in hospitalized adults with SARS-CoV-2 infection. JAMA Netw. Open. 2020;3(9):e2022058. doi: 10.1001/jamanetworkopen.2020.22058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cindrova-Davies T, Sanders DA, Burton GJ, Charnock-Jones DS. Soluble FLT1 sensitizes endothelial cells to inflammatory cytokines by antagonizing VEGF receptor-mediated signaling. Cardiovasc. Res. 2011;89:6719. doi: 10.1093/cvr/cvq346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maynard SE, et al. Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J. Clin. Invest. 2003;111:649–658. doi: 10.1172/JCI17189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Greco M, Palumbo C, Sicuro F, Lobreglio G. Soluble Fms-like tyrosine kinase-1 is a marker of endothelial dysfunction during sepsis. J. Clin. Med. Res. 2018;10:700–706. doi: 10.14740/jocmr3505w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Eguiburu-Jaime JL, et al. Soluble fms-like tyrosine kinase-1: A potential early predictor of respiratory failure in COVID-19 patients. Clin. Chem. Lab. Med. 2021 doi: 10.1515/cclm-2021-0126. [DOI] [PubMed] [Google Scholar]
- 14.Dupont V, et al. Excess soluble fms-like tyrosine kinase 1 correlates with endothelial dysfunction and organ failure in critically ill COVID-19 patients. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ricci A, et al. Serum CA 15–3 is increased in pulmonary fibrosis. Sarcoidosis Vasc. Diffuse Lung. Dis. 2009;26:54–63. [PubMed] [Google Scholar]
- 16.Kruit A, et al. CA 15–3 as an alternative marker for KL-6 in fibrotic lung diseases. Sarcoidosis Vasc. Diffuse Lung. Dis. 2010;27:138–146. [PubMed] [Google Scholar]
- 17.The guideline of diagnosis and treatment on novel coronavirus pneumonia. https://www.nhc.gov.cn/yzygj/s7652m/202003/a31191442e29474b98bfed5579d5af95.shtml/Accessed 3 Mar 2020 (2020).
- 18.Berlin DA, Gulick RM, Martinez FJ. Severe Covid-19. N. Engl. J. Med. 2020;383:2451–2460. doi: 10.1056/NEJMcp2009575. [DOI] [PubMed] [Google Scholar]
- 19.Gandhi RT, Lynch JB, Del Rio C. Mild or Moderate Covid-19. N. Engl. J. Med. 2020;383:1757–1766. doi: 10.1056/NEJMcp2009249. [DOI] [PubMed] [Google Scholar]
- 20.Vogelmeier C. IPF or NSIP? That is the question. Eur. Respir. J. 2003;22:191–192. doi: 10.1183/09031936.03.00037003. [DOI] [PubMed] [Google Scholar]
- 21.Kim JY, et al. Acute fibrinous and organizing pneumonia: Imaging features, pathologic correlation, and brief literature review. Radiol. Case Rep. 2018;13:867–870. doi: 10.1016/j.radcr.2018.04.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gomes R, et al. Acute fibrinous and organizing pneumonia. A report of 13 cases in a tertiary university hospital. Medicine. 2016;95:4073. doi: 10.1097/MD.0000000000004073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Giardini V, et al. Increased sFLT-1/PlGF ratio in COVID-19: A novel link to angiotensin II-mediated endothelial dysfunction. Am. J. Hematol. 2020;95:E188–E191. doi: 10.1002/ajh.25882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Libby P, Lüscher T. COVID-19 is, in the end, an endothelial disease. Eur. Heart. J. 2020;41:3038–3044. doi: 10.1093/eurheartj/ehaa623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ackermann M, et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N. Engl. J. Med. 2020;383:120–128. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wichmann D, et al. Autopsy findings and venous thromboembolism in patients with COVID-19: A prospective cohort study. Ann. Intern. Med. 2020;173:268–277. doi: 10.7326/M20-2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bernard I, et al. Endothelium infection and dysregulation by SARS-CoV-2: Evidence and caveats in COVID-19. Viruses. 2020;13(1):29. doi: 10.3390/v13010029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liu Z, et al. Dynamic interleukin-6 level changes as a prognostic indicator in patients with COVID-19. Front. Pharmacol. 2020;11:1093. doi: 10.3389/fphar.2020.01093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cai Q, et al. COVID-19 in a designated infectious diseases hospital outside Hubei Province, China. Allergy. 2020;75:1742–1752. doi: 10.1111/all.14309. [DOI] [PubMed] [Google Scholar]
- 30.Chen G, et al. Clinical and immunological features of severe and moderate coronavirus disease. J. Clin. Invest. 2020;130:2620–2629. doi: 10.1172/JCI137244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fajgenbaum DC, June CH. Cytokine Storm. N. Engl. J. Med. 2020;383:2255–2273. doi: 10.1056/NEJMra2026131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Carsana L, et al. Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: A two-centre descriptive study. Lancet Infect. Dis. 2020;20:1135–1140. doi: 10.1016/S1473-3099(20)30434-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ma H, Lu J, Song Y, Wang H, Yin S. The value of serum Krebs von den lungen-6 as a diagnostic marker in connective tissue disease associated with interstitial lung disease. BMC Pulm. Med. 2020;20:6. doi: 10.1186/s12890-019-1043-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Inoue Y, et al. Pulmonary epithelial cell injury and alveolar-capillary permeability in berylliosis. Am. J. Respir. Crit. Care Med. 1997;156:109–115. doi: 10.1164/ajrccm.156.1.9612043. [DOI] [PubMed] [Google Scholar]
- 35.Ricci A, et al. Serum CA 15–3 is increased in pulmonary fibrosis. Sarcoidosis Vasc. Diffuse Lung Dis. 2009;26:54–63. [PubMed] [Google Scholar]
- 36.Miyoshi S, et al. Comparative evaluation of serum markers in pulmonary sarcoidosis. Chest. 2010;137:1391–1397. doi: 10.1378/chest.09-1975. [DOI] [PubMed] [Google Scholar]
- 37.D'Alessandro M, et al. Serum KL-6 levels in pulmonary Langerhans' cell histiocytosis. Eur. J. Clin. Invest. 2020;20:e13242. doi: 10.1111/eci.13242. [DOI] [PubMed] [Google Scholar]
- 38.D'Alessandro M, et al. BAL biomarkers' panel for differential diagnosis of interstitial lung diseases. Clin. Exp. Med. 2020;20:207–216. doi: 10.1007/s10238-020-00608-5. [DOI] [PubMed] [Google Scholar]
- 39.Sato H, et al. KL-6 levels are elevated in plasma from patients with acute respiratory distress syndrome. Eur. Respir. J. 2004;23:142–145. doi: 10.1183/09031936.03.00070303. [DOI] [PubMed] [Google Scholar]
- 40.Nakamura H, et al. Clinical utility of serum beta-D-glucan and KL-6 levels in Pneumocystis jirovecii pneumonia. Intern. Med. 2009;48:195–220. doi: 10.2169/internalmedicine.48.1680. [DOI] [PubMed] [Google Scholar]
- 41.Urabe N, et al. Serial change in serum biomarkers during treatment of non-HIV Pneumocystis pneumonia. J. Infect. Chemother. 2019;25:936–942. doi: 10.1016/j.jiac.2019.05.007. [DOI] [PubMed] [Google Scholar]
- 42.Arai Y, et al. Clinical significance of the serum surfactant protein D and KL-6 levels in patients with measles complicated by interstitial pneumonia. Eur. J. Pediatr. 2001;160:425–429. doi: 10.1007/s004310100763. [DOI] [PubMed] [Google Scholar]
- 43.Kawasaki Y, et al. Serum KL-6 levels as a biomarker of lung injury in respiratory syncytial virus bronchiolitis. J. Med. Virol. 2009;81:2104–2108. doi: 10.1002/jmv.21634. [DOI] [PubMed] [Google Scholar]
- 44.D'Alessandro M, et al. Serum KL-6 concentrations as a novel biomarker of severe COVID-19. J. Med. Virol. 2020;92:2216–2220. doi: 10.1002/jmv.26087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Deng K, et al. Prognostic roles of KL-6 in disease severity and lung injury in COVID-19 patients: A longitudinal retrospective analysis. J. Med. Virol. 2021;93:2505–2512. doi: 10.1002/jmv.26793. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.


