Objective:
Does extracorporeal membrane oxygenation (ECMO) improve outcomes in ECMO-eligible patients with COVID-19 respiratory failure compared to maximum ventilation alone (MVA)?
Summary Background Data:
ECMO is beneficial in severe cases of respiratory failure when mechanical ventilation is inadequate. Outcomes for ECMO-eligible COVID-19 patients on MVA have not been reported. Consequently, a direct comparison between COVID-19 patients on ECMO and those on MVA has not been established.
Methods:
A total of 3406 COVID-19 patients treated at two major medical centers in Chicago were studied. One hundred ninety-five required maximum ventilatory support, and met ECMO eligibility criteria. Eighty ECMO patients were propensity matched to an equal number of MVA patients using detailed demographic, physiological, and comorbidity data. Primary outcome was survival and disposition at discharge.
Results:
Seventy-one percent of patients were decannulated from ECMO. Mechanical ventilation was discontinued in 75% ECMO and 16% MVA patients. Twenty-five percent of patients in the ECMO arm expired, 21% while on ECMO, compared with 74% in the MVA cohort. Mortality was significantly lower across all age and BMI groups in the ECMO arm. Sixty-eight percent ECMO and 26% MVA patients were discharged from the hospital. Fewer ECMO patients required long-term rehabilitation. Major complications such as septic shock, ventilator associated pneumonia, inotropic requirements, acute liver and kidney injuries are less frequent among ECMO patients.
Conclusions:
ECMO-eligible patients with severe COVID-19 respiratory failure demonstrate a 3-fold improvement in survival with ECMO. They are also in a better physical state at discharge and have lower overall complication rates. As such, strong consideration should be given for ECMO when mechanical ventilatory support alone becomes insufficient in treating COVID-19 respiratory failure.
Keywords: coronavirus disease 2019 (COVID-19), extracorporeal membrane oxygenation (ECMO), mechanical ventilation, propensity score match analysis, severe respiratory failure
Infection with the SARS-CoV-2 virus, Coronavirus Disease 2019 (COVID-19), has claimed nearly 4 million lives worldwide since December 2019.1 Although majority of those affected do not develop serious symptoms, some experience respiratory distress requiring mechanical ventilation.2 In severe cases, even ventilatory support fails. These patients develop refractory hypoxemia, acute respiratory distress syndrome (ARDS) with severe cytokine storm, and eventual multisystem organ failure.3
Veno-venous extracorporeal membrane oxygenation (ECMO) is a technique that has been shown to reduce mortality in severe cases of respiratory failure such as in Middle East Respiratory Syndrome (MERS) coronavirus infections.4–6 However, the need for prolonged ventilation, sedation, and immobility may limit the long-term benefits of this technique. In the field of lung transplantation, attempts at discontinuing mechanical ventilation while on ECMO are showing promising results.7,8 However, this novel approach has not been widely applied outside the realm of this subspecialty.
Recent articles on ECMO for severe COVID-19 respiratory failure have reported 15% to 38% mortality.9–11 However, the outcomes for ECMO-eligible COVID-19 patients on maximum ventilation alone (MVA) due to severe respiratory failure have not been described. As a result, a direct comparison between COVID-19 patients on ECMO and those on MVA has not been established. The goal of this study was to compare the two approaches using propensity match analysis to determine the impact of ECMO and MVA on survival, patient disposition at discharge, and complications in severe COVID-19 respiratory failure.
METHODS
Patient Selection
Data was collected retrospectively from 3406 COVID-19 patients treated at the Advocate Christ Medical Center and Rush University Health System (IRB No. AHC-7426-C5000376 and ORA-20040401, respectively) in Chicago, IL, between March 1st, 2020 and June 9th, 2021. Waiver of the need for consent was obtained. The Data were extracted from the official institutional databases for all COVID-19 patients treated at these facilities, and de-identified to protect patient privacy. COVID-19 infection was confirmed using PCR-based assays.
The following criteria were used to determine ECMO candidacy: patients ≤70 years’ old, and suffering from severe hypoxia or hypercarbia despite maximum ventilatory support similar to what was described by the EOLIA trial group [ie, if arterial blood gas (ABG) partial pressure of oxygen (PaO2) to the fraction of inspired oxygen (FiO2) ratio (P/F ratio) is ≤50 mm Hg for >3 hours, ≤80 mm Hg for >6 hours, or if pH is ≤7.25 with a partial pressure of carbon dioxide (PaCO2) ≥60 mm Hg for >6 hours].5 Maximized ventilator settings constituted of a minimal FiO2 of ≥0.8, positive end expiratory pressure (PEEP) ≥10 cm H2O, and tidal volumes of 6 mL/kg predicted body weight, while keeping a plateau pressure ≤32 cm H2O.
The following were absolute contraindications to ECMO placement: patients with cardiac arrest without return of spontaneous circulation, lactate ≥14 mMol/L or pH ≤6.9, in multi-system organ failure involving three or more organ systems, projected life expectancy ≤5 years before SARS-CoV-2 infection, known devastating neurological injury, recent hemorrhagic stroke or any known major bleeding diathesis, known DNR/DNI status, those who refuse to receive blood transfusions, those with permanent immobility, known active malignancy, and finally, patients with severe, symptomatic chronic organ failure, such as cirrhosis, end-stage renal disease on dialysis, end-stage cardiomyopathy, or those with severe chronic lung disease requiring home oxygen therapy, among others.
ECMO was initiated using a single access, dual-stage right atrium to pulmonary artery cannula. Please see supplemental material for details on the cannulation technique. Patients were extubated and mobilized while on ECMO.
All patients were followed for up to 150 days of hospitalization post-intubation.
Outcome Measures
The primary outcome was survival and disposition at discharge. Times spent in various treatment phases were determined, as well as types and frequencies of complications.
Statistical Analysis
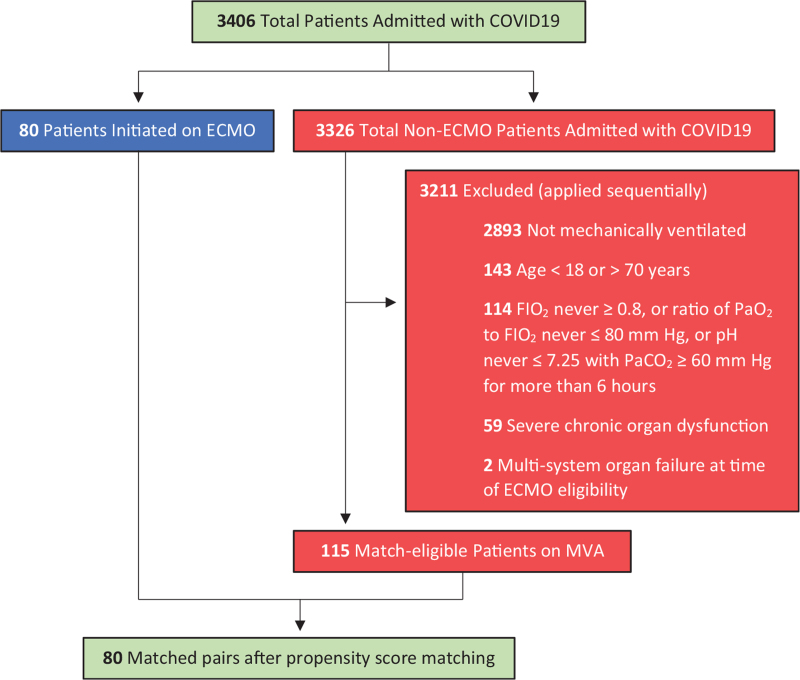
For the 3326 non-ECMO patients, match eligibility was determined based on the following criteria applied sequentially: patients required mechanical ventilation, were between 18 and 70 years’ old, required FIO2 ≥0.8 with arterial blood gas recording P/F ratio ≤80 mm Hg, or pH ≤7.25 with PaCO2 ≥60 mm Hg for >6 hours. Also, a diagnosis of any severe chronic organ dysfunction, or multi-system organ failure at the time of ECMO eligibility resulted in exclusion from matching (Fig. 1).
FIGURE 1.
Algorithm to identify matching-eligible patients.
Propensity score match analysis was carried out using a logistic regression model as described by Noah et al.12 Detailed demographic, physiological, and comorbidity data were utilized for matching as outlined in Table 1.
TABLE 1.
Characteristics of ECMO and MVA Patients Before and After Propensity Score Matching
| MVA Patients | |||||
| Match Categories | ECMO Patients | Pre-Match | P | Post-Match | P |
| Age, y | 49.0 (1.1) [51.0] {22–67} | 56.5 (0.9) [59.0] {22–70} | 0.001 | 52.1 (1.0) [53.5] {22–64} | 0.06 |
| Male, % | 76.3 | 68.7 | 0.25 | 72.5 | 0.59 |
| BMI, kg/m2 | 34.1 (0.8) [33.7] | 34.5 (0.9) [32.8] | 0.69 | 35.3 (1.2) [32.8] | 0.42 |
| Obesity∗, % | 71.3 | 69.6 | 0.8 | 72.5 | 0.86 |
| Ventilator†, days | 3.9 (0.4) | 4.1 (0.5) | 0.72 | 4.0 (0.5) | 0.79 |
| FIO2 | 0.97 (0.01) | 0.94 (0.01) | 0.04 | 0.95 (0.01) | 0.26 |
| PEEP, cm H2O | 15.6 (0.4) | 14.6 (0.3) | 0.06 | 14.7 (0.4) | 0.19 |
| ABG values | |||||
| pH | 7.25 (0.01) | 7.24 (0.01) | 0.84 | 7.24 (0.01) | 0.64 |
| PaCO2, mm Hg | 68.2 (2.3) | 64.2 (1.9) | 0.18 | 66.5 (2.4) | 0.63 |
| PaO2, mm Hg | 62.3 (1.9) | 68.1 (1.3) | 0.01 | 66.9 (1.4) | 0.04 |
| PaO2/FIO2, mm Hg | 65.0 (2.2) | 73.7 (1.8) | 0.002 | 71.3 (1.9) | 0.03 |
| Pronation and/or Chemical Paralysis, % | 93.8 | 98.3 | 0.1 | 97.5 | 0.25 |
Mean (SEM), [median], {range}.
Obesity is defined by a BMI ≥30 kg/m2.
Ventilator days refer to number of days intubated at time of ECMO eligibility.
ABG, arterial blood gas; BMI, body mass index; ECMO, extracorporeal membrane oxygenation; FIO2, fraction of inspired oxygen; MVA, maximum ventilation alone; PaCO2, partial pressure of carbon dioxide; PaO2, partial pressure of oxygen.
Data are expressed as mean ± standard error of the mean (SEM), median, and range. Relative risks or ratios (RR) between ECMO and MVA patients with 95% confidence intervals (CIs) are also indicated where appropriate. Data were analyzed by 2-tailed Student t test for continuous variable and Chi-Square test for categorical variables with a P value significance level of <0.05. The 2020 Excel for Office 365 (Microsoft) with the added XLMiner Analysis ToolPak was used for data analysis.
RESULTS
Study Population and Patient Characteristics
A total of 3406 patients with COVID-19 infection were admitted (Fig. 1). A total of 3326 were not supported on ECMO, of whom 3,211 were excluded based on the match eligibility criteria. One hundred ninety-five patients qualified for ECMO. Eighty were supported on ECMO, and 115 on MVA, of whom 80 were selected for comparison with the ECMO cohort.
ECMO was performed in patients between the ages of 22 and 67 years [mean (SE), 49.0 (1.1) years] (Table 1). MVA patients ranged from 22 to 64 years [52.1 (1.0) years]. The difference was not statistically significant (P = 0.06). Greater than 70% of patients in each group were male (P = 0.59). Average body mass index (BMI) was also similar [34.1 (0.8) kg/m2 for ECMO vs 35.3 (1.2) kg/m2 for MVA, P = 0.42]. All patients had reached maximum ventilatory support at the time of ECMO eligibility. The average duration of intubation before ECMO initiation was 3.9 (0.4) days, compared with 4.0 (0.5) days for the MVA cohort at time of ECMO eligibility (P = 0.79). Following propensity score matching, arterial blood gas values were also similar in the 2 groups except for PaO2 and P/F ratio, which were both slightly higher in the MVA arm. Pronation and/or chemical paralysis was undertaken in 94% ECMO and 98% MVA patients (P = 0.25).
Outcomes
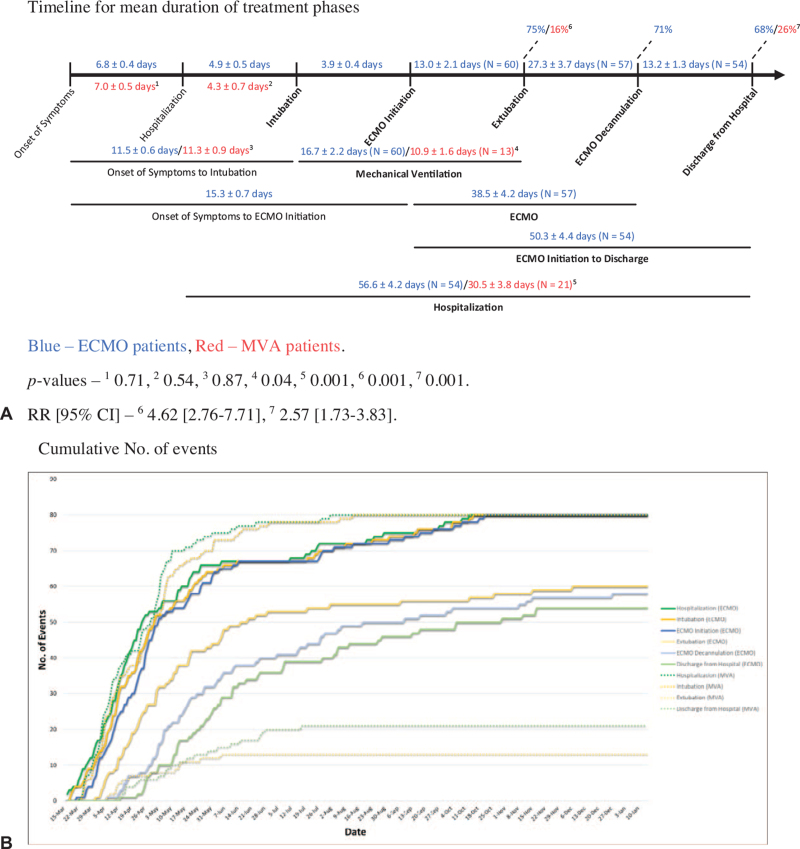
Patients supported on ECMO spent an average of 4.9 (0.5) days in the hospital before requiring intubation, whereas MVA patients were intubated at 4.3 (0.7) days (P = 0.54) (Fig. 2A). Onset of symptoms to intubation between the ECMO and MVA cohorts were 11.5 (0.6) and 11.3 (0.9) days, respectively (P = 0.87). The average time from intubation to ECMO initiation was 3.9 (0.4) days. Total duration of mechanical ventilation for extubated patients was 16.7 (2.2) days for the ECMO cohort, and 10.9 (1.6) days for the MVA group (P = 0.04). However, although 60 ECMO patients (75%) were successfully weaned off the ventilator, only 13 MVA patients (16%) achieved the same [RR (95% CI), 4.62 (2.76–7.71), P = 0.001]. Fifty-seven patients (71%) have been decannulated from ECMO yielding an average ECMO run time of 38.5 (4.2) days. The total time of hospitalization for those discharged were 56.6 (4.2) and 30.5 (3.8) days for the ECMO and MVA groups, respectively (P = 0.001).
FIGURE 2.
Timeline of events. A, Timeline for mean duration of treatment phases. B, Cumulative No. of events.
Overall, 20 patients (25%) in the ECMO group expired, 17 (21%) while on ECMO, compared to 59 patients (74%) in the MVA group [RR (95% CI), 0.34 (0.23–0.51), P = 0.001] (Table 2). Eighty-nine of the 115 match-eligible MVA patients (77%) did not survive.
TABLE 2.
Outcomes (Adverse Events)
| Categories | Subcategories | ECMO patients | MVA patients | RR (95% CI) | P |
| Neurologic | Stroke | 7 (8.8) | 6 (7.5) | 1.17 (0.41–3.32) | 0.77 |
| Cardiovascular | Inotropic requirement | 1 (1.3) | 29 (36.3) | 0.03 (0.005–0.25) | 0.001 |
| Significant bleeding∗ | 10 (12.5) | 2 (2.5) | 5.0 (1.13–22.1) | 0.02 | |
| Septic Shock† | 14 (17.5) | 44 (55.0) | 0.32 (0.19–0.53) | 0.001 | |
| Pulmonary | Tracheostomy | 10 (12.5) | 12 (15.0) | 0.83 (0.38–1.82) | 0.65 |
| Infectious | VAP | 19 (23.8) | 33 (41.3) | 0.58 (0.36–0.92) | 0.02 |
| Bacteremia | 27 (33.8) | 21 (26.3) | 1.29 (0.8–2.08) | 0.3 | |
| Renal | AKI requiring RRT | 18 (22.5) | 36 (45.0) | 0.5 (0.31–0.8) | 0.003 |
| Hepatic | Acute liver injury‡ | 6 (7.5) | 20 (25.0) | 0.3 (0.13–0.71) | 0.003 |
| ECMO | Revisions | 3 (3.8) | NA | NA | NA |
| cannulation Site bleeding | 1 (1.3) | NA | NA | NA | |
| Mortality | Overall | 20 (25.0) | 59 (73.8) | 0.34 (0.23–0.51) | 0.001 |
| On ECMO | 17 (21.3) | NA | NA | NA |
No. (%), (95% confidence interval).
Significant bleeding is defined by ≥5 U of PRBC transfusion within a 12-hour period.
Septic shock is defined as requiring ≥2 vasopressors.
Acute liver injury is defined as AST and ALT > 5× upper limits of normal.
AKI indicates acute kidney injury; NA, not applicable; RRT, renal replacement therapy; VAP, ventilator associated pneumonia.
Of the 20 ECMO deaths, 3 patients (15%) succumbed to stroke, and the rest to overwhelming sepsis and multi-system organ failure. Three patients expired after ECMO decannulation from nonrespiratory causes. For the 59 MVA deaths, 29 (49%) resulted from refractory respiratory failure, 27 (46%) from the development of overwhelming sepsis and multisystem organ failure, and 3 patients (5%) died of stroke.
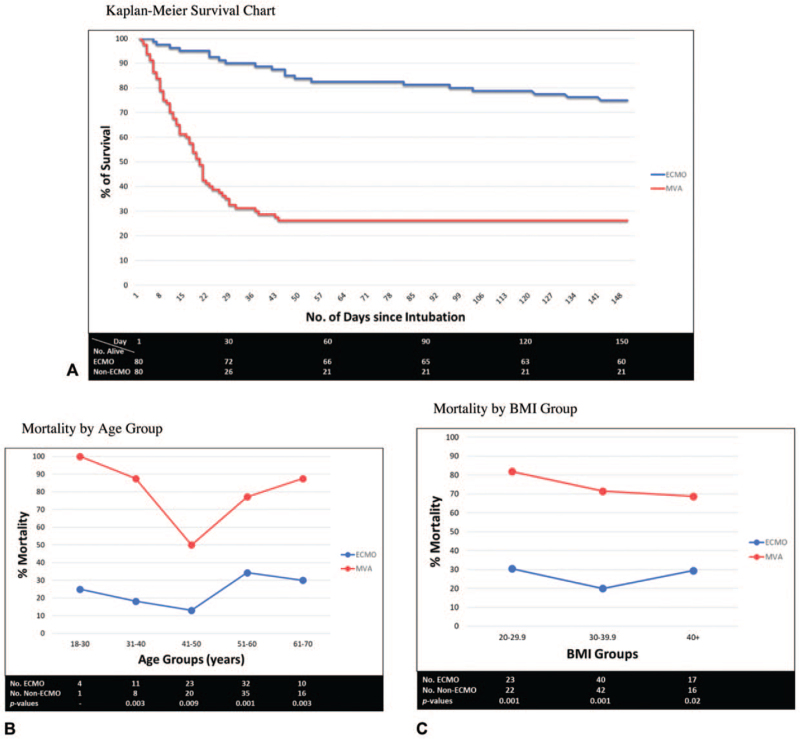
More than 90% of the MVA mortality occurred within the first four weeks of intubation (Fig. 3A). It was more protracted for those on ECMO. Mortality was significantly lower across all age and BMI groups in the ECMO arm (Fig. 3B and 3C). Patients in their 40 s displayed the highest survival rates in both groups.
FIGURE 3.
Survival Analysis. A, Kaplan-Meier Survival Chart. B, Mortality by Age Group. C, Mortality by BMI Group.
Fifty-four ECMO patients (68%) have been discharged alive, as opposed to 21 MVA patients (26%) [RR (95% CI), 2.57 (1.73–3.83), P = 0.001] (Fig. 2A). As of June 9th, 6 ECMO patients (8%) remained hospitalized with none in the MVA cohort. Of the 54 ECMO patients discharged thus far, 27 (50%) went home, 23 (43%) went to a short-term rehabilitation center (STR), and 4 (7%) to a long-term rehabilitation center (LTR) (Table 3). One patient received a double lung transplant before discharge. Of the 21 MVA discharges, 7 (33%) went home, 6 (29%) went to an STR, and 8 (38%) to an LTR. Overall, of the ECMO patients discharged, 50 (93%) were sent home or to an STR compared to 13 (62%) in the MVA cohort [RR (95% CI), 1.5 (1.06–2.11), P = 0.001]. All STR patients are now home. Twenty-four ECMO (44%) and 13 MVA patients (62%) required ≥2 L/min of supplemental oxygen at discharge [RR (95% CI), 0.72 (0.46–1.12), P = 0.17].
TABLE 3.
Discharges
| Discharges | ECMO Patients | MVA Patients | RR (95% CI) | P |
| Overall | 54 (67.5) | 21 (26.3) | 2.57 (1.73–3.83) | 0.001 |
| Home | 27 (50.0) | 7 (33.3) | 1.5 (0.77–2.91) | 0.19 |
| STR center | 23 (42.6) | 6 (28.6) | 1.49 (0.71–3.14) | 0.26 |
| Home or STR | 50 (92.6) | 13 (61.9) | 1.5 (1.06–2.11) | 0.001 |
| LTR center | 4 (7.4) | 8 (38.1) | 0.19 (0.07–0.58) | 0.001 |
| Supplemental oxygen | 24 (44.4) | 13 (61.9) | 0.72 (0.46–1.12) | 0.17 |
| Tracheostomy | 3 (5.6) | 8 (38.1) | 0.15 (0.04–0.5) | 0.001 |
No. (%), (95% confidence interval).
Cumulative number of events is illustrated by dates in Figure 2B. Approximately 75% of the ECMO and MVA patients were hospitalized within the first 2 months of the study coinciding with the first wave of COVID-19 cases in the state.1
Complication rates were generally lower for ECMO patients (Table 2). Septic shock was noted in 18% ECMO and 55% MVA patients [RR (95% CI), 0.32 (0.19–0.53), P = 0.001]. Inotropic requirements were 1% and 36% in ECMO and MVA patients, respectively [RR (95% CI), 0.03 (0.005–0.25), P = 0.001]. Incidence of ventilator associated pneumonia was also lower in the ECMO group (24% vs 41%) [RR (95% CI), 0.58 (0.36–0.92), P = 0.02]. Acute kidney injury requiring renal replacement therapy was noted in 23% ECMO and 45% MVA patients [RR (95% CI), 0.5 (0.31–0.8), P = 0.003]. Acute liver injury was seen in 8% and 25% of ECMO and MVA patients, respectively [RR (95% CI), 0.3 (0.13–0.71), P = 0.003]. Stroke and bacteremia rates were similar in both groups (P = 0.77 and 0.3, respectively). Significant bleeding requiring more than five units of blood transfusion within a 12-hour period was seen in 13% ECMO and 3% MVA patients [RR (95% CI), 5.0 (1.13–22.1), P = 0.02]. Overall, 13% ECMO and 15% MVA patients underwent a tracheostomy [RR (95% CI), 0.83 (0.38–1.82), P = 0.65]. However, of those discharged, 6% had received a tracheostomy in the ECMO group as opposed to 38% among the MVA cohort [RR (95% CI), 0.15 (0.04–0.5), P = 0.001] (Table 3).
Three patients (4%) required ECMO cannula revisions due to dislodgement or fractures, and 1 patient (1%) experienced significant bleeding from the cannula insertion site (Table 2).
DISCUSSION
In this study, we present the first propensity matched outcomes of ECMO and MVA in patients with severe COVID-19 respiratory failure.
Patient Matching and Eligibility
We have looked at >3400 patients with COVID-19 hospitalized at Advocate Christ Medical Center and Rush University Health System with >500 requiring ventilatory support. 195 were ECMO-eligible with 80 undergoing ECMO, and 115 supported on MVA. Those not mechanically ventilated, out of age range, or not having failed maximum ventilatory support were excluded from matching. Similarly, patients with severe chronic organ dysfunction, or in multisystem organ failure at the time of ECMO eligibility were also excluded. Using propensity match analysis, 80 MVA patients were identified as suitable matches for the 80 ECMO patients. Majority, 75%, in each group were admitted to the same hospitals around the same time frame (ie, within the first 2 months of the study), further limiting the variability between the groups.
Matching was accomplished using several patient characteristics that ultimately yielded similar age, sex, BMI, and arterial blood gas values in both groups. All patients were maximized on the ventilator and intubated for the same duration before ECMO initiation or eligibility. In fact, the MVA group had a slightly higher average PaO2 and P/F ratio than its ECMO counterpart. Nearly all patients were proned and/or chemically paralyzed.
Outcomes
As of June 9th, 75% of the ECMO patients had been taken off mechanical ventilation compared to 16% in the MVA group. Seventy-one percent in the ECMO arm had been successfully decannulated from ECMO, with 68% discharged alive from the hospital. Only 26% of the MVA patients had left the hospital. Although the overall time of hospitalization appears to be greater in the ECMO cohort, 90% of the MVA deaths occurred within a month of intubation.
Mortality in the ECMO arm was 25% (21% while on ECMO), and in the MVA group, 74%. Three patients expired after ECMO decannulation from nonrespiratory causes. Mortality was significantly lower across all age and BMI groups in the ECMO cohort. Interestingly, patients in their 40s had the lowest mortality among both groups. The reason for this is unclear, and warrants further investigation.
ECMO patients were primarily discharged home, and those who were sent to a short-term rehabilitation center are now home. Thus, >90% of the discharged ECMO patients are now home. For MVA patients, majority were discharged to a long-term rehabilitation center. Only 6% of the ECMO discharges had a tracheostomy at discharge compared to 38% in the MVA group. A greater percentage of MVA patients were also discharged on supplemental oxygen.
Overall, complication rates were lower in the ECMO cohort. Specifically, septic shock, ventilator associated pneumonia, inotropic requirements, acute liver and kidney injuries were less frequent among ECMO patients. Although stroke and bacteremia rates were similar in both groups, ECMO patients had greater incidence of bleeding.
Many reports have now shown that COVID-19 patients are prone to developing deep vein thrombosis, pulmonary emboli, and strokes.13,14 The infection produces a hypercoagulable state as evidenced by elevation in d-dimer levels.9,15 Hypercoagulability is also detrimental to the ECMO circuit as clots can hamper flows and damage the oxygenator.16 As such, systemic anticoagulation was started immediately after ECMO initiation. However, as anticoagulation carried an increased risk of hemorrhage, significant bleeding requiring transfusion therapy was noted more frequently in the ECMO cohort. Most of the bleeding was gastrointestinal in origin, and typically controlled by withholding anticoagulation for short periods of time. Our transfusion rate, however, was similar to that of the EOLIA trial group's.5
Cannulation Technique, Extubation, and Patient Mobilization on Ecmo
A single access, dual-stage right atrium to pulmonary artery cannula was utilized for all ECMO patients. The advantages of this technique have been described previously by Mustafa et al.9 Numerous studies have demonstrated that COVID-19 infections, ARDS, and obesity, independently promote right ventricle (RV) failure.17–21 One of the main advantages of our cannulation technique is the addition of right heart support. Furthermore, there is less mixing of deoxygenated blood as the cannula's inflow and outflow openings are separated by the RV. The single access through the neck also facilitates patient mobility once off the ventilator. Finally, we have experienced minimal cannula-associated complications or revisions.
Prolonged mechanical ventilation with the need for high-dose sedatives and patient immobility are associated with various complications.22 Extubating patients while on ECMO mitigates these issues and also averts ventilator-related airway trauma. Our experienced teams of ICU nurses, physical and respiratory therapists, as well as perfusionists led the mobilization efforts, and allowed the patients to actively participate in their recovery. At discharge, ECMO patients were in good physical condition with only a few requiring long-term rehabilitation.
Study Limitations
Although the results of this study demonstrate significant benefits of ECMO over MVA, it nonetheless remains a retrospective analysis conducted in two major medical centers. As such, a future multicenter study may allow for a greater generalizability of our findings.
CONCLUSIONS
In this propensity match analysis of patients with severe COVID-19 respiratory failure, ECMO leads to a significant three-fold improvement in survival compared to maximum ventilator support alone. This survival benefit is consistent across all age and BMI groups. ECMO patients are also in better physical state at discharge with majority now home and fewer requiring long-term rehabilitation. Major complications rates are also significantly lower in the ECMO cohort. This is the first direct comparison between these 2 groups of patients treated in the same hospitals, over the same time frame, and with similar baseline characteristics.
It is important to note that ECMO does not directly contribute to the recovery of lung function. Rather, it provides cardiopulmonary support while the lungs continue to heal. We recognize that ECMO requires specialized resources that are not readily available in every medical center. However, when available, strong consideration should be given for ECMO when mechanical ventilatory support alone becomes insufficient in treating COVID-19 respiratory failure.
The authors thank all our brave medical staff who selflessly took care of the patients.
Supplementary Material
REFERENCES
- 1. The Center for Systems Science and Engineering (CSSE) at Johns Hopkins University. Coronavirus COVID-19 global cases. Available at: https://coronavirus.jhu.edu. Accessed June 9th, 2021. [Google Scholar]
- 2.Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA 2020; 323:2052–2059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bhatraju PK, Ghassemieh BJ, Nichols M, et al. Covid-19 in critically ill patients in the Seattle region—case series. N Engl J Med 2020; 382:2012–2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brodie D, Bacchetta M. Extracorporeal membrane oxygenation for ARDS in adults. N Engl J Med 2011; 365:1905–1914. [DOI] [PubMed] [Google Scholar]
- 5.Combes A, Hajage D, Capellier G, et al. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. N Engl J Med 2018; 378:1965–1975. [DOI] [PubMed] [Google Scholar]
- 6.Alshahrani MS, Sindi A, Alshamsi F, et al. Extracorporeal membrane oxygenation for severe Middle East respiratory syndrome coronavirus. Ann Intensive Care 2018; 8:3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Abrams D, Garan AR, Brodie D. Awake and fully mobile patients on cardiac extracorporeal life support. Ann Cardiothorac Surg 2019; 8:44–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lehr CJ, Zaas DW, Cheifetz IM, et al. Ambulatory extracorporeal membrane oxygenation as a bridge to lung transplantation: walking while waiting. Chest 2015; 147:1213–1218. [DOI] [PubMed] [Google Scholar]
- 9.Mustafa AK, Alexander PJ, Joshi DJ, et al. Extracorporeal membrane oxygenation for patients with COVID-19 in severe respiratory failure. JAMA Surg 2020; 155:990–992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Barbaro RP, MacLaren G, Boonstra PS, et al. Extracorporeal membrane oxygenation support in COVID-19: an international cohort study of the Extracorporeal Life Support Organization registry. Lancet 2020; 396:1071–1078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schmidt M, Hajage D, Lebreton G, et al. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome associated with COVID-19: a retrospective cohort study. Lancet Respir Med 2020; 8:1121–1131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Noah MA, Peek GJ, Finney SJ, et al. Referral to an extracorporeal membrane oxygenation center and mortality among patients with severe 2009 influenza A(H1N1). JAMA 2011; 306:1659–1668. [DOI] [PubMed] [Google Scholar]
- 13.Bikdeli B, Madhavan MV, Jimenez D, et al. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up: JACC State-of-the-art review. J Am Coll Cardiol 2020; 75:2950–2973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Oxley TJ, Mocco J, Majidi S, et al. Large-vessel stroke as a presenting feature of Covid-19 in the young. N Engl J Med 2020; 382:e60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020; 395:1054–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lubnow M, Philipp A, Foltan M, et al. Technical complications during veno-venous extracorporeal membrane oxygenation and their relevance predicting a system-exchange--retrospective analysis of 265 cases. PLoS One 2014; 9:e112316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Creel-Bulos C, Hockstein M, Amin N, et al. Acute cor pulmonale in critically ill patients with Covid-19. N Engl J Med 2020; 382:e70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li Y, Li H, Zhu S, et al. Prognostic value of right ventricular longitudinal strain in patients with COVID-19. JACC Cardiovasc Imaging 2020; 13:2287–2299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with Coronavirus Disease 2019 pneumonia in Wuhan, China. JAMA Intern Med 2020; 180:934–943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chahal H, McClelland RL, Tandri H, et al. Obesity and right ventricular structure and function: the MESA-Right Ventricle Study. Chest 2012; 141:388–395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zochios V, Parhar K, Tunnicliffe W, et al. The right ventricle in ARDS. Chest 2017; 152:181–193. [DOI] [PubMed] [Google Scholar]
- 22.Hill AD, Fowler RA, Burns KE, et al. Long-term outcomes and health care utilization after prolonged mechanical ventilation. Ann Am Thorac Soc 2017; 14:355–362. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.