Abstract
Intraoperative ultrasound (IOUS) is a valuable adjunctive tool that can provide real-time diagnostic information in surgery that has the potential to alter patient management and decrease complications. Lesion localization, characterization and staging can be performed, as well as surveying for additional lesions and metastatic disease. IOUS is commonly used in the liver for hepatic metastatic disease and hepatocellular carcinoma, in the pancreas for neuroendocrine tumors, and in the kidney for renal cell carcinoma. IOUS allows real-time evaluation of vascular patency and perfusion in organ transplantation and allows for early intervention for anastomotic complications. It can also be used to guide intraoperative procedures such as biopsy, fiducial placement, radiation, or ablation. A variety of adjuncts including microbubble contrast and elastography may provide additional information at IOUS. It is important for the radiologist to be familiar with the available equipment, common clinical indications, technique, relevant anatomy and intraoperative imaging appearance to optimize performance of this valuable imaging modality.
Introduction
Ultrasonography is an ideal intraoperative imaging tool as it is portable, provides real-time information, does not use ionizing radiation, and requires no patient preparation. Intraoperative ultrasound (IOUS) allows for placement of the transducer directly on the surface of the organ being evaluated allowing for much higher resolution imaging not degraded by overlying air, bone or soft tissue.1,2 IOUS was first performed in 1961 by Schlegel et al3 for detection of kidney stones, and was subsequently used by Knight and Newell in 1963 for detection of common bile duct stones.1,4 Diagnostic use of IOUS has expanded to a variety of surgical applications, and can be used to accurately localize pathology, guide extent of surgical resection, improve tumor staging and influence surgical decision making.2 Multiple studies have shown that IOUS impacts choice of surgical strategy ranging from 2.7 to 49% of the time depending on the clinical scenario.1 In the 1980s and early 1990s, intraoperative ultrasound went beyond use solely for diagnostic purposes when groups began using it to guide and monitor intraoperative procedures including cryosurgery or thermal ablation.5,6
The utility and application of IOUS has continued to evolve since that time as pre-operative imaging and percutaneous techniques have improved. However, it remains a valuable tool for a spectrum of operative interventions. Currently, IOUS is commonly used for localization of known lesions and assessment of planned surgical margin, as well as detection and characterization of small lesions, particularly in the liver, kidney and pancreas. IOUS with color and pulsed Doppler is commonly used for assessment of vascular structures and blood flow/perfusion, particularly of the graft in transplant. There is growing use of IOUS to guide intraoperative interventions, ranging from biopsy to fiducial placement to ablation, and it has an important role in increasingly less-invasive surgical approaches including laparoscopy, endocavitary, and robotic assisted surgery.7–10 New ultrasound adjunctive tools including ultrasound microbubble contrast and ultrasound elastography have widened the scope of potential information that can be provided intraoperatively. The purpose of this review is to summarize the utility and common indications for IOUS, discuss preparation and technique to optimize IOUS, and review emerging tools that may add value at IOUS.
Technique
Although the principles behind IOUS are very similar to those used with routine transabdominal or endocavitary sonography, there are a variety of additional preparatory steps, logistical considerations, and selection of suitable equipment to promote a successful trip to the operating room. From a workflow standpoint, it is important to schedule IOUS studies in advance, as this will require a radiologist, sonographer, and ultrasound machine to leave the department and travel to the OR for a variable period of time. In preparation for the OR, thoughtful selection of probes and review of the existing imaging is crucial for successful sonography.
Several scan approaches may be available depending on the indication, clinical question, and operative access. In general, intraoperative probes are often smaller and designed to gain access into tight anatomical regions during surgery (Figure 1). They are configured as end-fire or side-fire probes that are usually multifrequency (5–9 MHz). Laparoscopic probes are designed to fit through different sized laparoscopic ports, with a long shaft to enable reach from the port to the organ in question, a flexible or steerable end, and a small contact surface (30 × 5 mm). Increasingly, minimally invasive laparoscopic or robotic surgical approaches are being utilized, which require either a laparoscopic or drop-in transducer that are usually configured with linear or convex linear arrays (5–10 MHz).2,7,9,10 If an open approach is being used, a low profile side-fire T- or I-shaped convex array 5–10 MHz transducer with a small contact surface (5 × 50 mm) may be helpful. The side-fire T shaped transducer can fit against the palm and between the index and middle fingers and enable scanning along small spaces like the liver dome.
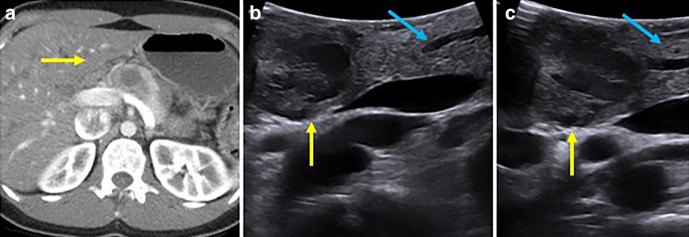
Figure 1.
Sampling of intraoperative probes including a laparoscopic probe (a, b) with long shaft, small face (inset), flexible end and directional controls (arrow, b), small low profile T probe (c), and robotic drop in probe (d) with curved face and ridge for grasping with laparoscopic tools (arrow, inset).
Because these probes may be different from what is normally used in sonography daily practice and the radiologist may be performing the scanning, practicing with the probes prior to the OR, perhaps on a phantom, or accompanying a more experienced staff to the OR to watch their technique may be helpful.7,9 Occasionally, small endocavitary or small endoluminal probes such as those used for transvaginal exams can be useful for deep structures like the pancreas.2 Prior to arrival in the OR, it is critical to know the type of operative access, the clinical information needed by the surgeon, and the available equipment at your institution and relative strengths of different machines and transducers. For example, when evaluating transplant grafts, selecting a machine with robust color and pulsed Doppler capabilities is often most important for the clinical question. When leaving the radiology department for the OR, the sonographer and radiologist should communicate and gather the optimal machine, several potential transducers, and the needed materials to sterilely drape or use each transducer if the approach requires it.
In addition to selecting the right equipment, it is important for the radiologist to carefully review the pre-procedure imaging with the clinical question in mind. Anatomy is often distorted by mobilization or retraction and may be confusing in the sometimes daunting OR environment. Once sterile, it can be challenging to look back at pre-procedure imaging. When evaluating pre-procedure imaging, the radiologist should note the location of the surgical target and any indeterminate lesions and their relationship to easily identifiable landmarks, which can include vascular structures or adjacent benign lesions such as cysts. The proximity of lesions to vulnerable or critical structures and relevant vascular anatomy should also be noted.1 Discussion with the surgeon prior to arrival in the OR can be helpful, particularly if there might be a need for intraoperative interventions like ablation depending on the ultrasound findings. Intraoperative interventions may require additional equipment and staff as well as specific expertise, so knowing and scheduling this in advance is helpful.
On arrival in the OR, evaluation of the approach and appropriate transducer can be confirmed with the surgeon. If scanning directly on the surgical field, sterile preparation of the probe is needed, which requires cooperation between the radiologist and the sonographer or other non-sterile OR staff. Sterile condom sheaths are the most common approach to prepare the transducer for sterile use. Specially designed sheaths are available for dedicated intraoperative transducers which must be long enough to cover the length of the supply cord and should fit snugly to avoid artifacts.11 Once the radiologist is scrubbed, gowned and gloved according to institutional protocols, the radiologist can prepare the probe for use by placing the condom sheath over the probe in sterile fashion. It is important to make sure to utilize the appropriate cover and gel for the transducer being used. Gel should be generously applied to the inside of the probe cover, then the sonographer can carefully place the probe in the cover. As the radiologist holds the covered end of the probe, the sonographer can carefully pull the sheath over the length of the cord, touching only the open end of the sheath. Once the probe is in the cover, air between the transducer and the cover interface should be eliminated. The cover can be tightly secured with the provided clip or rubber band (supplemental online video). The laparoscopic probe cover may have a longer sleeve and very slippery lubricant; some probes can be autoclaved. A robotic or drop-in probe may not require a sleeve or cover, as these can often be autoclaved between uses, and can be steered from the robot without donning sterile gown and gloves. These probes are highly versatile, but some education about how to use the robot from the surgeon or time in the robot simulation lab can be very helpful.
When scanning, it can be helpful to add fluid such as sterile saline to the resection cavity or the surface of the organ being scanned. This can improve contact between the probe and the organ surface and can facilitate smooth movement of the probe along the surface. In addition, it can be helpful for imaging lesions in the near field.11 It is useful to turn down the OR lights the probe has been placed on the field. If possible, the radiologist should stand on the side of the patient opposite the ultrasound machine or have images projected on the larger surgical monitors so they can be easily viewed without turning or twisting. When scanning, light pressure should be used to maintain contact between the probe and the organ. The actual anatomy scanned and method of scanning will be influenced by the purpose of the exam. If there is a specific clinical question (e.g. presence of vascular flow in a graft), doing a tailored exam to evaluate the vasculature is sufficient. If a small probe is being used to perform a systematic survey to identify lesions (e.g. metastatic survey), consecutive slightly overlapping vertical/sagittal sweeps along the organ being evaluated can be used to try to cover as much of the organ in question as possible. For example, when scanning the liver, it can be helpful to start at the lateral margin of the left lobe and move to the right, covering as much of the liver as possible. Make sure that expected lesions are identified based on vascular structures and benign lesions identified as landmarks. It is helpful to have the sonographer at the ultrasound console to optimize parameters, to activate color and spectral Doppler when needed (e.g. to properly identify vascular landmarks and assess flow) and to periodically save images for documentation. When complete, carefully remove the probe from the surgical field and remove soiled probe covers while still gowned and gloved.
Clinical applications
Liver
IOUS is frequently used during planned liver resection, most commonly with metastatic colorectal cancer or hepatocellular carcinoma (HCC),12–14 or during liver transplantation. During hepatic resection, either for primary tumor resection or metastasectomy, it is used for localization of lesions with respect to the planned surgical margin, to evaluate for and characterize additional lesions that may be present, assess vascular occlusion or invasion, and occasionally to guide intraoperative interventions. Two main approaches are commonly used, either open or laparoscopic. Since scanning is almost always performed directly on the liver surface, very high resolution images can be obtained. With current transducer resolution, lesions larger than 2 mm can be identified, with sensitivity reported in the 90–95% range.15,16 Therefore, IOUS may improve detection, characterization, localization and local staging of hepatic tumors (Figure 2).13 In addition, IOUS is considered to be the best method for evaluating the relationship between the liver lesions and nearby vascular structures and bile ducts.11 Typically, a 1–2 cm margin is required for adequate lesion resection. For an open approach, the liver may be partially mobilized and a side-firing T-probe may work best, fit into the palm and stabilized with the index and middle fingers. As above, determining whether a directed localization or general survey is needed can be helpful. Scanning can begin in the left lobe and progress to the right in overlapping strokes. Visualizing the high dome may be challenging, and scanning through another lobe or from the inferior surface of the liver is sometimes necessary.11 If a laparoscopic approach is required, the radiologist should choose the available port with the best access and angle to the liver, and take a similarly systematic approach with careful interrogation of lesions when identified. Metastatic lesions are often targetoid and may have an ill-defined hypoechoic rim (Figure 2). Mucinous tumors may occasionally contain fine calcifications and appear slightly hyperechoic (Figure 2). Metastatic lesions that are similar in size and arise from a single primary tumor typically appear similar on ultrasound.11 It is also important to evaluate the hepatic vasculature for bland or tumor thrombus, particularly with HCC, and to evaluate for lymph nodes in the porta hepatis.
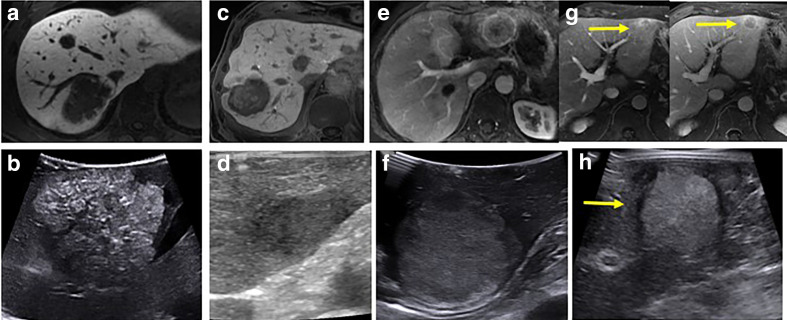
Figure 2.
MRI and corresponding IOUS images of a spectrum of liver lesions. Mucinous CRC metastatic lesion on hepatobiliary phase MRI (a) demonstrates fine calcifications and a hyperechoic appearance at IOUS (b), while a different 88-year-old male with multifocal CRC on hepatobiliary phase MRI (c) demonstrates a lobulated ill-defined hypoechoic lesion at IOUS (d). A heterogeneously enhancing lesion on arterial phase MRI imaging (e) found to be metastatic NET in a 57-year-old female was hyperechoic at IOUS (f). A left lobe lesion that demonstrated arterial enhancement with portal venous washout (arrows, (g) in a 67-year-old male without known liver disease was found to be hepatocellular carcinoma at biopsy. This appeared targetoid with a hypoechoic rim at IOUS (h). CRC, colorectal cancer; IOUS, intraoperative ultrasound; NET, neuroendocrine tumor.
Multiple studies have looked at the impact of IOUS on surgical decision-making. Even with state-of-the-art pre-operative CT and MR imaging, several studies have shown that IOUS still impacts surgical strategy in 12–30% of patients undergoing colorectal cancer liver metastasectomy.12,17 IOUS is also useful in staging primary hepatic neoplasms such as HCC and may identify intravascular growth of tumor or tumor thrombus that was equivocal on preprocedure imaging.18
New adjunctive techniques such as use of microbubble contrast or elastography have been utilized with IOUS of the liver. Contrast-enhanced ultrasound (CEUS) may increase sensitivity and specificity in detecting lesions, and may provide additional information for characterization of lesions.14,19–21 Treated lesions may be particularly challenging to identify, and CEUS may be helpful in this setting. CEUS can also be helpful for the evaluation of liver vasculature. Some unique workflow challenges may exist in the OR, and use of CEUS may require advance planning and cooperation with the anesthesia team. Similarly, intraoperative elastography may be helpful for characterization of liver lesions based on differences in stiffness,22,23 or identification of challenging lesions, providing “visual palpation” of non-palpable or barely visible lesions.24 Elastography is also an additional way to evaluate intraoperative interventions such as ablation. 3D printed models may be a useful adjunct to IOUS in complex liver resections.25
Echogenic shadowing due to gas at the resection margin can be seen on IOUS. Cautery used to score the liver or packing material around the liver can also produce acoustic shadowing. It is important to be aware of these artifacts in the intraoperative setting.11,26
Intraoperative biopsy, fiducial placement, or ablation
Despite use of diagnostic IOUS and adjunctive techniques, a lesion may not always be definitively characterized and may require intraoperative biopsy to determine the surgical plan. If the lesion is superficial, wedge resection can be performed. If not, IOUS-guided biopsy can be used to sample indeterminate lesions in real time and has high diagnostic yield and favorable safety profile. However, these biopsies can be technically challenging due to the location of the incision, the small face of the transducer and a more difficult angle of approach. If the appropriate angle cannot be obtained through the incision, a percutaneous entry site away from the incision can be used to obtain a more desirable trajectory (Figure 3). Biopsy specimens can be sent for rapid frozen section to obtain more timely results. In addition to biopsy, occasionally requests are made for fiducial placement for localization for future radiation therapy and these can be placed in a similar manner intraoperatively.
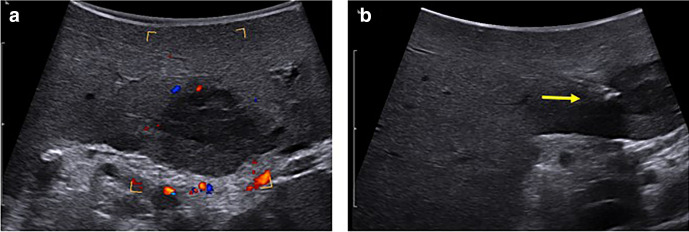
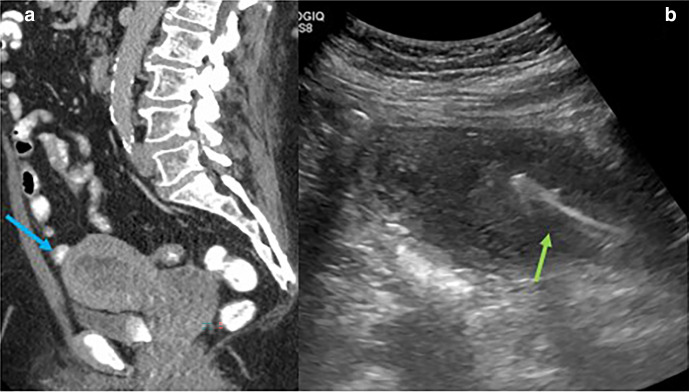
Figure 3.
48-year-old male with pancreas cancer undergoing Whipple with IOUS found to have indeterminate liver lesion (a) underwent intraoperative biopsy which demonstrated metastatic disease (b, arrow, needle). It was challenging to obtain the desired angle of approach through the surgical incision, so a percutaneous puncture site away from the incision was selected for optimal targeting. IOUS, intraoperative ultrasound.
Due to advances in adjunctive techniques, many ablation cases can be performed percutaneously or sequentially with surgery.27,28 Occasionally, it can be performed intraoperatively to extend the limits of hepatic surgery.6,29,30 For example, ablation plus surgery may allow local tumor control in patients with technically unresectable disease (Figure 4). Like biopsy, it can be technically challenging but may allow access to locations that may be difficult to access percutaneously, or may allow for both resection and ablation to be done under a single general anesthetic. Ablation can be used to treat lesion(s), to fortify a margin that is close, or to devascularize a non-anatomic resection plane. The acoustic shadowing caused by thermal ablation (gas with heat based modalities, ETOH; iceball with cryoablation) may produce a blind spot on the deep side of the ablation zone and may require scanning from an alternate approach for visualization.
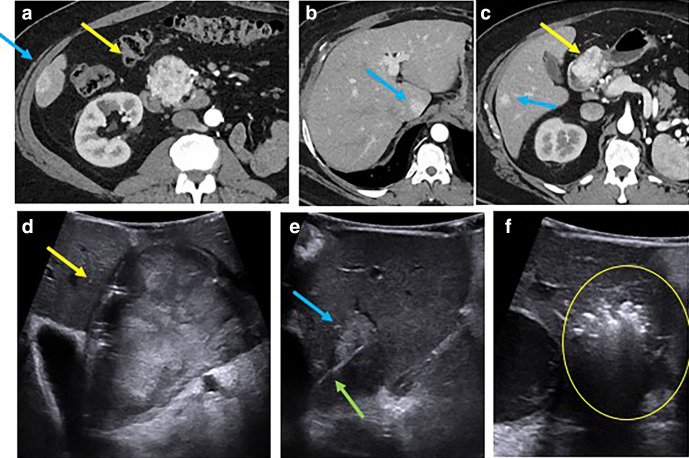
Figure 4.
55-year-old male with metastatic RCC to the pancreas (a, c yellow arrow) and liver (a–c, blue arrows). IOUS image from open approach shows the mass in the pancreas (d, yellow arrow) and hyperechoic caudate liver lesion (e, blue arrow). Patient underwent pancreaticoduodenectomy, wedge resection of hepatic metastatic in segment 4b (not shown) and 6 and intraoperative microwave ablation of hepatic lesions in segment 4a (not shown), caudate, and segments 6/7 for clearance of all metastatic disease. IOUS image demonstrates advancement of a microwave ablation applicator (e, green arrow) into the caudate liver lesion with subsequent ablation and gas cloud with posterior acoustic shadowing encompassing the lesion (f, circle). Due to the small transducer face, it is difficult to lay the needle out along its full length. After this treatment, the patient remains disease free 1 year later. IOUS, intraoperative ultrasound; RCC, renal cellcarcinoma
These intraoperative procedures are most commonly applied in the liver, but can be similarly applied to other sites such as the kidney.
Kidney
There is a continued strong push toward nephron-sparing surgery in the kidney, with improved surgical techniques and approaches to partial nephrectomy and increased utilization of locoregional therapies like thermal ablation.26 These nephron-sparing procedures utilize imaging to more precisely define the margins and extent of tumors. IOUS is one imaging tool commonly used for this task, particularly important for partially or completely endophytic lesions.8,10 Precise tumor delineation allows optimal sparing of adjacent parenchyma while maintaining adequate oncologic margin, and IOUS is extremely helpful to define the size and extension of tumor into adjacent structures.31 Sensitivity and specificity for renal sinus or vascular invasion is very high, approaching 100%.31 For endophytic renal lesions, robotic partial nephrectomies that used IOUS showed lower estimated blood loss, shorter warm ischemia times, fewer complications, and improved oncologic outcomes (Figure 5).8 IOUS can also be used to survey for additional unexpected lesions or to characterize additional indeterminate lesions, which can alter the operative plan or technique, as described with the liver. In one series of patients undergoing open partial nephrectomy, 10% of patients had additional findings at IOUS not seen on pre-operative imaging that altered surgical management in the majority of cases (71%).32 The operative approach to partial nephrectomy is now commonly laparoscopic or robotic which can be technically challenging and may require practice.10 As discussed in the technique section, a robotic approach may utilize a drop-in probe that can be controlled via the robot and may not require sterile technique. Some advance practice with the robotic tools or in the robotic simulation lab can be helpful for preparation.10 As with the liver, irrigating the surface of the kidney with sterile saline and keeping the transducer perpendicular to the kidney surface with good contact is helpful for optimal visualization. CEUS may occasionally be helpful in evaluation of the adjacent vasculature, particularly when assessing for bland or tumor thrombus.10
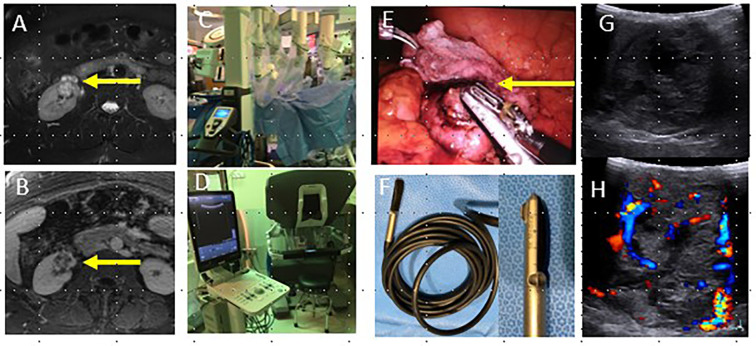
Figure 5.
42-year-old female with complex cystic lesion of the right kidney (arrows, a, b) identified on pre-operative T2W (a) and T1W with contrast (b). Patient underwent robotic partial nephrectomy (c), which utilizes the drop in probe and grasper tool to apply the probe to the lesion (arrow, d) and an ultrasound machine connected to the robot control panel (e). Intraoperative ultrasound images (f) demonstrate a predominantly solid, vascular mass and delineated the proximity of the mass to the renal hilum and major vessels in real time. Surgical pathology demonstrated clear cell RCC. RCC, renalcell carcinoma
Laparoscopic ablation can also be performed for renal lesions that are challenging to approach percutaneously, although improvements in adjunctive techniques such as hydrodissection and pyeloperfusion have allowed for more aggressive patient selection for percutaneous approaches.33
Pancreas
The pancreas is a deep structure and can often be imaged with the T-probe. However, other effective probe selection include a small hockey stick probe, the laparoscopic probe, or a transvaginal probe if maintaining adequate organ contact is difficult. Sterile saline can be useful to aid in scanning, and direct scanning of the gland from the head and uncinate process to the tail can be performed. The pancreas generally appears homogeneously hyperechoic and the duct is a hypoechoic tubular structure at the center of the gland. Cystic or solid hypoechoic tumors are readily identified.26 IOUS in the pancreas is most commonly used for localization of small tumors, particularly neuroendocrine tumors such as insulinoma, for localization of non-palpable lesions and evaluating proximity to the pancreatic duct (Figure 6).34,35 This can again impact the surgical approach, where both enucleation or partial pancreatectomy may be options. It can also be used to assess the adequacy of the margin after enucleation, and to assess for the presence of additional occult lesions as can be seen with hereditary syndromes like multiple endocrine neoplasia (MEN)36,37 and can be used to survey the liver for metastatic disease where relevant.38 IOUS can be used to biopsy pancreatic or indeterminate liver lesions if indicated. IOUS also is valuable in the setting of pancreatitis-related surgery, particularly for localizing and determining the size and appearance of the pancreatic duct, localizing collections and evaluating the vasculature.38–40
Figure 6.
Pre-operative CT (a) in a 23-year-old female demonstrates a mass in the pancreatic head (yellow arrow). EUS biopsy showed solid pseudopapillary neoplasm. At IOUS (b, c), note the small cystic spaces with the lesion identifiable at high resolution but not well seen from a percutaneous approach. The pancreatic duct (blue arrows) is also localized leading up to the mass to aid in operative approach. EUS, enhancedultrasound; IOUS, intraoperative ultrasound
Intraoperative radiation therapy (IORT) is an emerging therapy for marginally or borderline resectable pancreas cancer. IOUS may be helpful in targeting and assessing resectability.41
Solid organ transplants
IOUS is commonly used in transplant for assessment of graft vasculature and perfusion. IOUS to evaluate graft vascular anastomoses and flow may allow for early intervention and prevention of later or more severe complications. IOUS is often combined with serial post-procedure Doppler ultrasound. CEUS may have some utility in trouble shooting or better visualizing vessels (Figure 7). When evaluating a transplant, it is critical to choose a machine and probe with adequate Doppler capabilities. This IOUS evaluation of transplant is performed with a spectrum of graft types, most commonly including liver, kidney and pancreas.42 Small studies have shown a sensitivity of near 100% and specificity up to 89% in assessing inflow and outflow vessel patency in liver transplantation, which can lead to improved graft survival.43–45 In liver transplant, the hepatic artery and portal veins can be interrogated in the porta hepatis but should also include evaluation of the intrahepatic branches, particularly for the hepatic artery. Spectral Doppler findings of hepatic artery inflow compromise include an intrahepatic tardus parvus waveform (slow upstroke and low-resistance flow, RI <60%) and high velocity jet at the anastomosis or area of narrowing (>200 cm s−1). Undetectable intrahepatic arterial flow raises concern for hepatic artery thrombus. On grayscale, echogenic thrombus can be seen with loss of color Doppler signal. It is critical to identify arterial or venous thrombosis immediately before operative closure.46 Similar findings can be seen in other types of grafts where both arterial and venous structures should be interrogated (Figure 8). Intra- and immediate post-operative ultrasound is of high utility in these patients. At times, the arterial structures may appear patent intraoperatively, then appear abnormal after abdominal closure, sometimes due to kinking in small or pediatric patients where the liver is slightly large for the recipient. In these cases, packing the incision open initially can be helpful. Identification of these changes on ultrasound can be helpful in suggesting this etiology.
Figure 7.
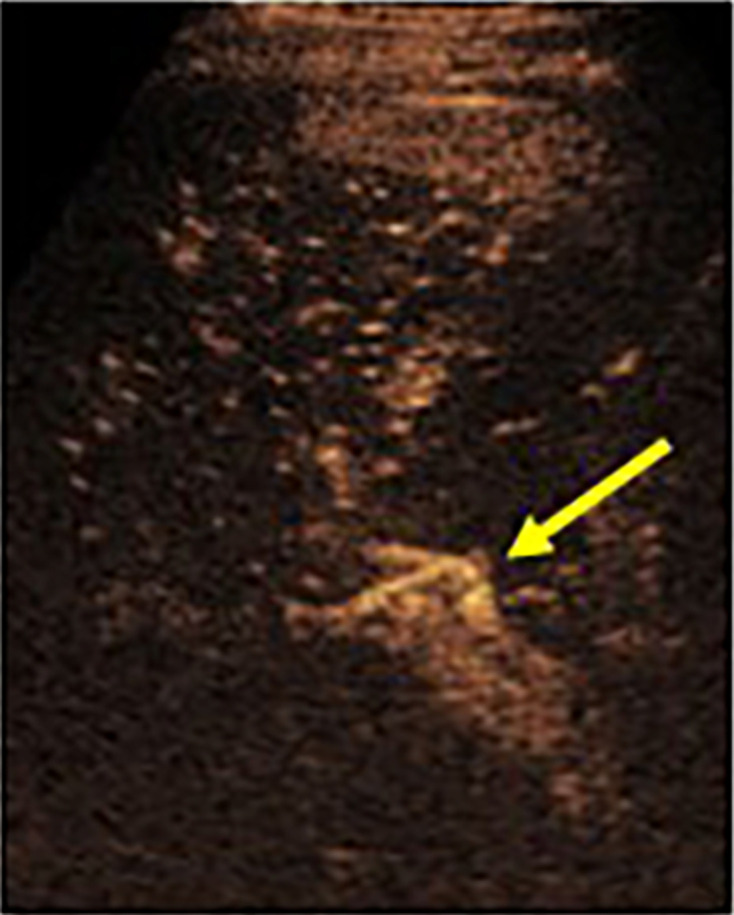
CEUS in a living donor split liver transplant patient demonstrating a patent hepatic artery (arrow) in the porta that was challenging to see on grayscale ultrasound. The artery was challenging to separate from the adjacent portal vein and CEUS allowed for real-time separation. CEUS, contrast-enhanced ultrasound.
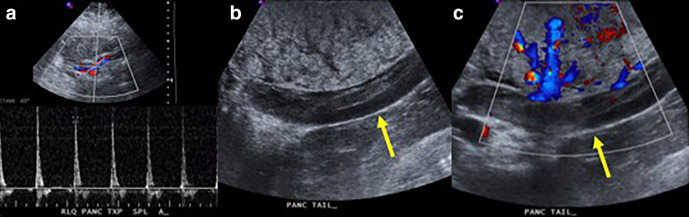
Figure 8.
Ultrasound images obtained immediately post-pancreas transplant (a) demonstrate markedly elevated resistive indices and reversal of diastolic flow in the pancreatic graft. Based on these findings, the patient was taken back to the operating room and extensive graft venous thrombosis was identified on grayscale (b) and color Doppler (c) IOUS images (arrows). Vein graft thrombosis is the most common cause of pancreatic graft loss. IOUS, intraoperative ultrasound
In living donor split liver transplantation specifically, IOUS may also be used to help identify an avascular resection plane in the donor and to document flow once the graft is in the recipient.11,47
GYN applications
As the trend toward more laparoscopic gynecologic surgeries continues, the role and utility of IOUS in the procedures is being explored. Laparoscopic myomectomy is one place where IOUS has shown promise for identifying smaller, nonpalpable lesions and determining optimal hysterotomy incision site.48 Similarly, dilation and curettage procedures performed with ultrasound guidance had significantly lower overall complication rate, reduced intraoperative blood loss, decreased procedure time, decreased post-operative bleeding and faster recovery (Figure 9).49 Ultrasound guidance can also be used to map pelvic anatomy prior to and during complex GYN procedures, to characterize adnexal masses at laparoscopy, to serve as an adjunct to hysteroscopy and to guide embryo transfer.50 In a recent small series of patients with deep infiltrating endometriosis affecting bowel, IOUS was used to assess the length and depth of penetration of disease for surgical decision making. IOUS showed improved sensitivity for muscularis propria invasion and improved accuracy for quantifying lesion size compared to pre-operative ultrasound imaging.51 Transabdominal ultrasound can be used to during tandem placement for uterine brachytherapy in order to guide placement, optimize position in the endometrial cavity and prevent complications like uterine perforation.
Figure 9.
68-year-old female with heterogeneous endometrial thickening (arrow, a, sagittal CT) and abnormal uterine bleeding. Intraoperative ultrasound guidance for dilation, curettage and endometrial sampling was performed from an anterior percutaneous transabdominal approach. Note the curette within the endometrial cavity (b, arrow). IOUS helps ensure a robust sample but prevents pushing the curette through the uterine fundus. Surgical pathology demonstrated endometrial cancer and sufficient material was obtained for additional pathologic and genetic testing. IOUS, intraoperative ultrasound
Additional applications: there are a spectrum of potential applications for IOUS outside of the abdomen/pelvis, e.g. in ENT or neurosurgery. For resection of brain tumors, ultrasound may be helpful for tumor localization, assessment of adequacy of resection, and evaluation of tumor vascularity and position relative to vascular structures.52 IOUS can be used to aid in parathyroid localization or to guide transoral biopsy of head and neck lesions not accessible percutaneously (e.g. retropharyngeal lesions) using endocavitary probe.53 These areas are emerging areas of IOUS utilization that may see future growth.
Conclusion
Despite improvements in pre-operative imaging, IOUS remains a valuable adjunctive tool that can provide real-time diagnostic information in surgery that may alter patient management and decrease complications. Lesion localization, characterization, and staging can be performed, as well as surveying for additional lesions and metastatic disease. It allows real-time evaluation of vascular patency and perfusion in transplant and allows for early intervention on anastomotic complications. It can also be used to guide intraoperative procedures such as biopsy, fiducial placement or ablation. It is important for the radiologist to be familiar with the available equipment, clinical indication/question, technique, relevant anatomy and intraoperative imaging appearance to optimize performance of this valuable imaging modality.
Footnotes
Disclosure: MGL: Prior grant funding, Philips, Ethicon, LMG: None, ND: None, PJP: Advisor: Bracco and Zebra; Shareholder: SHINE, Elucent, Cellectar, DHK: Shareholder for Cellectar, shareholder for Elucent, TJZ: Neuwave/Ethicon- Consultant, research funding; Histosonics- Consultant, shareholder, research funding.
Contributor Information
Meghan G Lubner, Email: mlubner@uwhealth.org.
Lori Mankowski Gettle, Email: LMankowskiGettle@uwhealth.org.
David H Kim, Email: Dkim@uwhealth.org.
Timothy J Ziemlewicz, Email: TZiemlewicz@uwhealth.org.
Nirvikar Dahiya, Email: Dahiya.Nirvikar@mayo.edu.
Perry Pickhardt, Email: ppickhardt2@uwhealth.org.
REFERENCES
- 1.Luck AJ, Maddern GJ. Intraoperative abdominal ultrasonography. Br J Surg 1999; 86: 5–16. doi: 10.1046/j.1365-2168.1999.00990.x [DOI] [PubMed] [Google Scholar]
- 2.Silas AM, Kruskal JB, Kane RA. Intraoperative ultrasound. Radiol Clin North Am 2001; 39: 429–48. doi: 10.1016/S0033-8389(05)70290-6 [DOI] [PubMed] [Google Scholar]
- 3.Schlegel JU, Diggdon P, Cuellar J. The use of ultrasound for localizing renal calculi. J Urol 1961; 86: 367–9. doi: 10.1016/S0022-5347(17)65180-2 [DOI] [PubMed] [Google Scholar]
- 4.Knight PR, Newell JA. Operative use of ultrasonics in cholelithiasis. Lancet 1963; 1: 1023–5. doi: 10.1016/S0140-6736(63)92427-9 [DOI] [PubMed] [Google Scholar]
- 5.Onik G, Kane R, Steele G, McDermott W, Khettry U, Cady B, et al. Society of gastrointestinal radiologists Roscoe E. Miller award. monitoring hepatic cryosurgery with sonography. AJR Am J Roentgenol 1986; 147: 665–9. doi: 10.2214/ajr.147.4.665 [DOI] [PubMed] [Google Scholar]
- 6.Lee FT, Mahvi DM, Chosy SG, Onik GM, Wong WS, Littrup PJ, et al. Hepatic cryosurgery with intraoperative us guidance. Radiology 1997; 202: 624–32. doi: 10.1148/radiology.202.3.9051005 [DOI] [PubMed] [Google Scholar]
- 7.Ganguli S, Kruskal JB, Brennan DD, Kane RA. Intraoperative laparoscopic ultrasound. Radiol Clin North Am 2006; 44: 925–35. doi: 10.1016/j.rcl.2006.10.020 [DOI] [PubMed] [Google Scholar]
- 8.Sun Y, Wang W, Zhang Q, Zhao X, Xu L, Guo H. Intraoperative ultrasound: technique and clinical experience in robotic-assisted renal partial nephrectomy for endophytic renal tumors. Int Urol Nephrol 2020;01 Oct 2020. doi: 10.1007/s11255-020-02664-y [DOI] [PubMed] [Google Scholar]
- 9.Bezzi M, Merlino P, Orsi F, Di Nardo R, Silecchia G, Basso N, et al. Laparoscopic sonography during abdominal laparoscopic surgery: technique and imaging findings. AJR Am J Roentgenol 1995; 165: 1193–8. doi: 10.2214/ajr.165.5.7572501 [DOI] [PubMed] [Google Scholar]
- 10.Di Cosmo G, Verzotti E, Silvestri T, Lissiani A, Knez R, Pavan N, et al. Intraoperative ultrasound in robot-assisted partial nephrectomy: state of the art. Arch Ital Urol Androl 2018; 90: 195–8. doi: 10.4081/aiua.2018.3.195 [DOI] [PubMed] [Google Scholar]
- 11.Kruskal JB, Kane RA. Intraoperative us of the liver: techniques and clinical applications. Radiographics 2006; 26: 1067–84. doi: 10.1148/rg.264055120 [DOI] [PubMed] [Google Scholar]
- 12.Damian C-D, Rednic N, Munteanu D, Cazacu M. The role of intraoperative ultrasound for the assessment of the focal liver lesions in patients with colorectal cancer. Med Ultrason 2014; 16: 114–8. doi: 10.11152/mu.201.3.2066.162.cdd1nr2 [DOI] [PubMed] [Google Scholar]
- 13.Joo I. The role of intraoperative ultrasonography in the diagnosis and management of focal hepatic lesions. Ultrasonography 2015; 34: 246–57. doi: 10.14366/usg.15014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rojas Llimpe FL, Di Fabio F, Ercolani G, Giampalma E, Cappelli A, Serra C, et al. Imaging in resectable colorectal liver metastasis patients with or without preoperative chemotherapy: results of the PROMETEO-01 study. Br J Cancer 2014; 111: 667–73. doi: 10.1038/bjc.2014.351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Guimarães CM, Correia MM, Baldisserotto M, de Queiroz Aires EP, Coelho JF. Intraoperative ultrasonography of the liver in patients with abdominal tumors: a new approach. J Ultrasound Med 2004; 23: 1549–55. doi: 10.7863/jum.2004.23.12.1549 [DOI] [PubMed] [Google Scholar]
- 16.Wagnetz U, Atri M, Massey C, Wei AC, Metser U. Intraoperative ultrasound of the liver in primary and secondary hepatic malignancies: comparison with preoperative 1.5-T MRI and 64-MDCT. AJR Am J Roentgenol 2011; 196: 562–8. doi: 10.2214/AJR.10.4729 [DOI] [PubMed] [Google Scholar]
- 17.Sietses C, Meijerink MR, Meijer S, van den Tol MP. The impact of intraoperative ultrasonography on the surgical treatment of patients with colorectal liver metastases. Surg Endosc 2010; 24: 1917–22. doi: 10.1007/s00464-009-0874-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Conlon R, Jacobs M, Dasgupta D, Lodge JPA. The value of intraoperative ultrasound during hepatic resection compared with improved preoperative magnetic resonance imaging. Eur J Ultrasound 2003; 16: 211–6. doi: 10.1016/S0929-8266(02)00075-7 [DOI] [PubMed] [Google Scholar]
- 19.Fioole B, de Haas RJ, Wicherts DA, Elias SG, Scheffers JM, van Hillegersberg R, et al. Additional value of contrast enhanced intraoperative ultrasound for colorectal liver metastases. Eur J Radiol 2008; 67: 169–76. doi: 10.1016/j.ejrad.2007.03.017 [DOI] [PubMed] [Google Scholar]
- 20.Torzilli G. Contrast-Enhanced intraoperative ultrasonography in surgery for liver tumors. Eur J Radiol 2004; 51 Suppl: S25–9. doi: 10.1016/j.ejrad.2004.03.025 [DOI] [PubMed] [Google Scholar]
- 21.Torzilli G, Olivari N, Moroni E, Del Fabbro D, Gambetti A, Leoni P, et al. Contrast-Enhanced intraoperative ultrasonography in surgery for hepatocellular carcinoma in cirrhosis. Liver Transpl 2004; 10(2 Suppl 1): S34–8. doi: 10.1002/lt.20050 [DOI] [PubMed] [Google Scholar]
- 22.Kato K, Sugimoto H, Kanazumi N, Nomoto S, Takeda S, Nakao A. Intra-Operative application of real-time tissue elastography for the diagnosis of liver tumours. Liver Int 2008; 28: 1264–71. doi: 10.1111/j.1478-3231.2008.01701.x [DOI] [PubMed] [Google Scholar]
- 23.Omichi K, Inoue Y, Hasegawa K, Sakamoto Y, Okinaga H, Aoki T, et al. Differential diagnosis of liver tumours using intraoperative real-time tissue elastography. Br J Surg 2015; 102: 246–53. doi: 10.1002/bjs.9728 [DOI] [PubMed] [Google Scholar]
- 24.Inoue Y, Takahashi M, Arita J, Aoki T, Hasegawa K, Beck Y, et al. Intra-operative freehand real-time elastography for small focal liver lesions: "visual palpation" for non-palpable tumors. Surgery 2010; 148: 1000–11. doi: 10.1016/j.surg.2010.02.009 [DOI] [PubMed] [Google Scholar]
- 25.Witowski J, Budzyński A, Grochowska A, Ballard DH, Major P, Rubinkiewicz M, et al. Decision-Making based on 3D printed models in laparoscopic liver resections with intraoperative ultrasound: a prospective observational study. Eur Radiol 2020; 30: 1306–12. doi: 10.1007/s00330-019-06511-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Marcal LP, Patnana M, Bhosale P, Bedi DG. Intraoperative abdominal ultrasound in oncologic imaging. World J Radiol 2013; 5: 51–60. doi: 10.4329/wjr.v5.i3.51 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Okuno M, Kawaguchi Y, De Bellis M, Vega EA, Huang SY, Ahrar K, et al. A new sequential treatment strategy for multiple colorectal liver metastases: planned incomplete resection and postoperative completion ablation for intentionally-untreated tumors under guidance of cross-sectional imaging. Eur J Surg Oncol 2021; 47: 311–6. doi: 10.1016/j.ejso.2020.08.018 [DOI] [PubMed] [Google Scholar]
- 28.Winkelmann MT, Archid R, Gohla G, Hefferman G, Kübler J, Weiss J, et al. Mri-Guided percutaneous thermoablation in combination with hepatic resection as parenchyma-sparing approach in patients with primary and secondary hepatic malignancies: single center long-term experience. Cancer Imaging 2020; 20: 37. doi: 10.1186/s40644-020-00316-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Huang Y, Song J, Zheng J, Jiang L, Yan L, Yang J, et al. Comparison of hepatic resection combined with intraoperative radiofrequency ablation, or hepatic resection alone, for hepatocellular carcinoma patients with multifocal tumors meeting the University of California San Francisco (UCSF) criteria: a propensity score-matched analysis. Ann Surg Oncol 2020; 27: 2334–45. doi: 10.1245/s10434-020-08231-0 [DOI] [PubMed] [Google Scholar]
- 30.Lee SM, Ko HK, Shin JH, Kim J-H, Chu HH. Combination of intraoperative radiofrequency ablation and surgical resection for treatment of cholangiocarcinoma: feasibility and long-term survival. Diagn Interv Radiol 2020; 26: 45–52. doi: 10.5152/dir.2019.18552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Secil M, Elibol C, Aslan G, Kefi A, Obuz F, Tuna B, et al. Role of intraoperative us in the decision for radical or partial nephrectomy. Radiology 2011; 258: 283–90. doi: 10.1148/radiol.10100859 [DOI] [PubMed] [Google Scholar]
- 32.Bhosale PR, Wei W, Ernst RD, Bathala TK, Reading RM, Wood CG, et al. Intraoperative sonography during open partial nephrectomy for renal cell cancer: does it alter surgical management? AJR Am J Roentgenol 2014; 203: 822–7. doi: 10.2214/AJR.13.12254 [DOI] [PubMed] [Google Scholar]
- 33.Hinshaw JL, Shadid AM, Nakada SY, Hedican SP, Winter TC, Lee FT. Comparison of percutaneous and laparoscopic cryoablation for the treatment of solid renal masses. AJR Am J Roentgenol 2008; 191: 1159–68. doi: 10.2214/AJR.07.3706 [DOI] [PubMed] [Google Scholar]
- 34.Gorman B, Charboneau JW, James EM, Reading CC, Galiber AK, Grant CS, et al. Benign pancreatic insulinoma: preoperative and intraoperative sonographic localization. AJR Am J Roentgenol 1986; 147: 929–34. doi: 10.2214/ajr.147.5.929 [DOI] [PubMed] [Google Scholar]
- 35.Grover AC, Skarulis M, Alexander HR, Pingpank JF, Javor ED, Chang R, et al. A prospective evaluation of laparoscopic exploration with intraoperative ultrasound as a technique for localizing sporadic insulinomas. Surgery 2005; 138: 1003–8. doi: 10.1016/j.surg.2005.09.017 [DOI] [PubMed] [Google Scholar]
- 36.Addeo P, Julliard O, Imperiale A, Goichot B, Bachellier P. Middle-segment preserving pancreatectomy for multifocal neuroendocrine pancreatic tumors. Surg Oncol 2020; 35: 466–7. doi: 10.1016/j.suronc.2020.10.004 [DOI] [PubMed] [Google Scholar]
- 37.Zhao Y-pei, Zhan H-xiang, Zhang T-ping, Cong L, Dai M-hua, Liao Q, et al. Surgical management of patients with insulinomas: result of 292 cases in a single institution. J Surg Oncol 2011; 103: 169–74. doi: 10.1002/jso.21773 [DOI] [PubMed] [Google Scholar]
- 38.Sun MRM, Brennan DD, Kruskal JB, Kane RA. Intraoperative ultrasonography of the pancreas. Radiographics 2010; 30: 1935–53. doi: 10.1148/rg.307105051 [DOI] [PubMed] [Google Scholar]
- 39.Ní Mhuircheartaigh JM, Sun MRM, Callery MP, Siewert B, Vollmer CM, Kane RA. Pancreatic surgery: a multidisciplinary assessment of the value of intraoperative us. Radiology 2013; 266: 945–55. doi: 10.1148/radiol.12120201 [DOI] [PubMed] [Google Scholar]
- 40.Smith SJ, Vogelzang RL, Donovan J, Atlas SW, Gore RM, Neiman HL. Intraoperative sonography of the pancreas. American Journal of Roentgenology 1985; 144: 557–62. doi: 10.2214/ajr.144.3.557 [DOI] [PubMed] [Google Scholar]
- 41.Jin L, Shi N, Ruan S, Hou B, Zou Y, Zou X, et al. The role of intraoperative radiation therapy in resectable pancreatic cancer: a systematic review and meta-analysis. Radiat Oncol 2020; 15: 76. doi: 10.1186/s13014-020-01511-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Thalhammer C, Aschwanden M, Bilecen D, Staub D, Guerke L, Schaub S, et al. Intraoperative color duplex ultrasound during renal transplantation. Ultraschall in Med 2008; 29: 652–6. doi: 10.1055/s-2008-1027441 [DOI] [PubMed] [Google Scholar]
- 43.Waldman DL, Lee DE, Bronsther O, Orloff MS. Use of intraoperative ultrasonography during hepatic transplantation. J Ultrasound Med 1998; 17: 1–6. doi: 10.7863/jum.1998.17.1.1 [DOI] [PubMed] [Google Scholar]
- 44.Cheng YF, Huang TL, Chen CL, Lee TY, Chen TY, Chen YS. Intraoperative Doppler ultrasound in liver transplantation. Clin Transplant 1998; 12: 292–9. [PubMed] [Google Scholar]
- 45.Sadej P, Feld RI, Frank A. Transplant renal vein thrombosis: role of preoperative and intraoperative Doppler sonography. American Journal of Kidney Diseases 2009; 54: 1167–70. doi: 10.1053/j.ajkd.2009.06.036 [DOI] [PubMed] [Google Scholar]
- 46.LH G, Fang H, FH L, Li P, Zhu CX, Zhu JJ. Prediction of early hepatic artery thrombosis by intraoperative color Doppler ultrasound in pediatric segmental liver transplantation. Clin Transplant 2012; 26: 571–6. [DOI] [PubMed] [Google Scholar]
- 47.Hecht EM, Kambadakone A, Griesemer AD, Fowler KJ, Wang ZJ, Heimbach JK. Living donor liver transplantation: overview, imaging technique, and diagnostic considerations. AJR Am J Roentgenol 2019;: 1–11. [DOI] [PubMed] [Google Scholar]
- 48.Lin PC, Thyer A, Soules MR. Intraoperative ultrasound during a laparoscopic myomectomy. Fertil Steril 2004; 81: 1671–4. doi: 10.1016/j.fertnstert.2003.10.049 [DOI] [PubMed] [Google Scholar]
- 49.Acharya G, Morgan H, Paramanantham L, Fernando R. A randomized controlled trial comparing surgical termination of pregnancy with and without continuous ultrasound guidance. Eur J Obstet Gynecol Reprod Biol 2004; 114: 69–74. doi: 10.1016/j.ejogrb.2003.09.042 [DOI] [PubMed] [Google Scholar]
- 50.Criniti A, Lin PC. Applications of intraoperative ultrasound in gynecological surgery. Curr Opin Obstet Gynecol 2005; 17: 339–42. doi: 10.1097/01.gco.0000175349.10684.e8 [DOI] [PubMed] [Google Scholar]
- 51.Puppo A, Olearo E, Gattolin A, Rimonda R, Novelli A, Ceccaroni M. Intraoperative ultrasound for bowel deep infiltrating endometriosis. J Ultrasound Med 2020; 2020. doi: 10.1002/jum.15511 [DOI] [PubMed] [Google Scholar]
- 52.Sastry R, Bi WL, Pieper S, Frisken S, Kapur T, Wells W, WL B, Wells W 3rd, et al. Applications of ultrasound in the resection of brain tumors. J Neuroimaging 2017; 27: 5–15. doi: 10.1111/jon.12382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Fornage BD, Edeiken BS, Clayman GL. Use of transoral sonography with an endocavitary transducer in diagnosis, fine-needle aspiration biopsy, and intraoperative localization of retropharyngeal masses. AJR Am of Roentgenol 2014; 202: W481–6. doi: 10.2214/AJR.13.11398 [DOI] [PubMed] [Google Scholar]