Abstract
Enzyme therapies are attracting significant attention as thrombolytic drugs during the current scenario owing to their great affinity, specificity, catalytic activity, and stability. Among various sources, the application of microbial-derived thrombolytic and fibrinolytic enzymes to prevent and treat vascular occlusion is promising due to their advantageous cost–benefit ratio and large-scale production. Thrombotic complications such as stroke, myocardial infarction, pulmonary embolism, deep venous thrombosis, and peripheral occlusive diseases resulting from blood vessel blockage are the major cause of poor prognosis and mortality. Given the ability of microbial thrombolytic enzymes to dissolve blood clots and prevent any adverse effects, their use as a potential thrombolytic therapy has attracted great interest. A better understanding of the hemostasis and fibrinolytic system may aid in improving the efficacy and safety of this treatment approach over classical thrombolytic agents. Here, we concisely discuss the physiological mechanism of thrombus formation, thrombo-, and fibrinolysis, thrombolytic and fibrinolytic agents isolated from bacteria, fungi, and algae along with their mode of action and the potential application of microbial enzymes in thrombosis therapy.
Keywords: fibrinogen, fibrin, thrombolytic therapy, microbial enzymes, thrombosis, hemostasis
1. Introduction
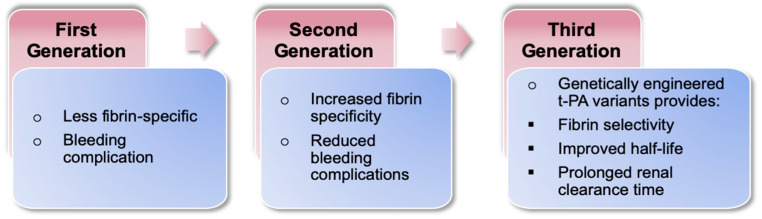
Dysregulation of the intrinsic balance between the coagulation cascade and the fibrin- and thrombolytic pathways can lead to severe outcomes such as myocardial infarction, stroke, and massive pulmonary embolism caused by fibrin accumulation in the blood vessel walls, resulting in the formation of hemostatic plug or a clot [1]. Currently, the tissue plasminogen activator (tPA) or plasminogen activator (PA) is an indispensable clinical tool in thrombolytic therapy [2]. They are categorized into first, second, and third generation (Figure 1) [3]. PA are serine proteases that form active plasmin from inactive plasminogen and mediate fibrin lysis through two modes of action: (i) direct—channels direct action on plasminogen to catalyze its activation; and (ii) indirect—forms a 1:1 stoichiometric complex with plasminogen or plasmin followed by activation of the circulating plasminogen molecule. Despite their clinical effectiveness, however, these agents have several undesirable side effects, including immunogenicity, gastrointestinal bleeding, intracranial hemorrhage, and major systemic hemorrhage [4,5]. Consequently, there is an urgent need for approaches exploring new fibrinolytic agents with better specificity and efficacy.
Figure 1.
Generational classification and features of thrombolytic drugs.
There are several known sources of fibrinolytic enzymes, including plants, animals, and microbes. Among them, microbial-derived fibrinolytic enzymes have gained particular interest because they possess the following advantages: (i) cost-effective production, (ii) fewer to no side effects [6], (iii) broad biochemical diversity, (iv) mass-culture feasibility, and (v) allow genetic manipulation. Hence, various microorganisms have been used for the isolation of fibrinolytic enzymes, including bacteria, fungi, and algae.
The purpose of this review is to provide an overview of the microbial-derived enzymes with thrombolytic and fibrinolytic potential and summarize the existing literature on the microbes proposed to produce enzymes with thrombolytic and fibrinolytic activities.
1.1. Mechanism of Thrombus Formation
Hemostasis is defined as the physiological process of cessation of blood loss by clot formation at the site of an injury. Fibrin is a major component of blood clots, and is formed from fibrinogen (large soluble plasma glycoprotein) via proteolysis by thrombin. The accumulation of fibrin clots results in thrombus formation. A thrombus is a blood clot formed within the blood vessel [7]. Under physiological conditions, a proteolytic enzyme—plasmin—hydrolyses the fibrin clot to prevent thrombosis in blood vessels. During vascular pathophysiology or damage to the vascular system, the process of clot hydrolysis is disrupted, which may result in thrombosis [8].
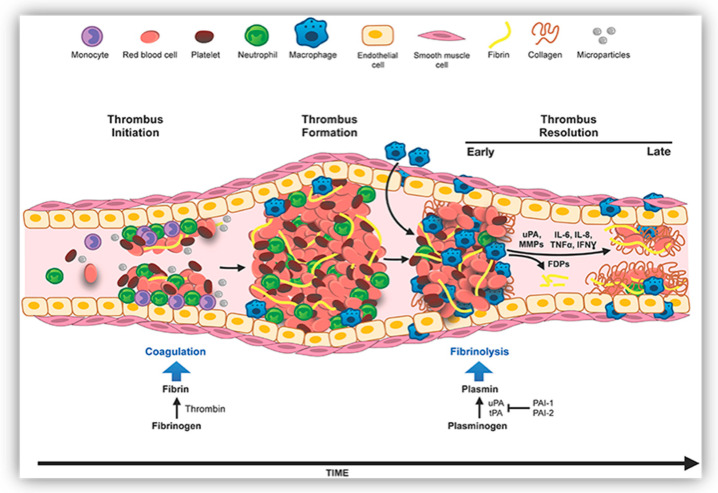
Hemostasis and thrombosis are intricate, multifactorial processes. Platelets, in conjugation with endothelial cells and coagulation proteins, are the crucial mediator of vascular hemostasis and thrombosis. Disruption to any of these processes could result in atherosclerotic plaque formation [9,10,11] (Figure 2, leading to variety of thrombotic diseases including CVDs (cardiovascular diseases) [12,13], abdominal aortic aneurysms (AAAs) [14,15], pulmonary embolism (PE) [16], and stroke [17,18].
Figure 2.
Innate immune cells in deep vein thrombosis (DVT). Adapted with permission from Mukhopadhyay et al., copyright © 2019 [11].
The process of hemostasis is divided into two stages: (1) primary (involves rapid platelet activation) and (2) secondary (requires additional coagulation pathways to form polymeric fibrin) processes (Table 1).
Table 1.
|
Primary Hemostasis (Platelet Plug Formation) |
Secondary Hemostasis (Coagulation Cascade to Form Fibrin Network) |
|---|---|
|
|
|
|
|
|
1.2. Fibrinolysis and Thrombolysis
Fibrinolysis is the breakdown of fibrin in the blood clot, and thrombolysis can be simply defined as the process of thrombus dissolution.
Fibrin is a primary protein component of a blood clot that is formed from fibrinogen (340 kDa glycoprotein) by thrombin-mediated proteolysis and elimination of N-terminal fibrinopeptides from the Aα and Bβ chains. A serine protease, plasmin, is an activated form of inert plasma precursor, plasminogen (PLG). PLG is a 791-AA-long glycoprotein that circulates in blood plasma as an inactive zymogen. The plasmin is a crucial enzyme in the dissolution of fibrin clots. There exist two major glycoforms of human plasminogen, namely, type I (consists of two molecules of glycosylation) and type II (consists of one molecule of glycosylation) [8,22]. The binding of circulating PLG to the cell surface or blood clot acquires an open conformation, which aids in the formation of catalytically active plasmin by the Arg561-Val562 bond cleavage via a direct PLG activator [23]. Direct PA includes tissue plasminogen activator (tPA), streptokinase and urokinase or their variants [3,24]. Plasmin formation leads to fibrin lysis, giving soluble fibrin degradation products (FDPs) [22,25]. Therefore, the process of fibrinolysis involves two phases: (i) PLG activation on the surface of the fibrin clot to forms plasmin, which dissolves fibrin—i.e., a physiological process; and (ii) amplification of plasmin-induced clot breakdown by exposing the additional binding sites of degraded fibrin—i.e., pharmacologically mediated processes such as fibrinolytic and thrombolytics.
2. Microbes in Thrombolytic Therapy
Many microbial-derived thrombolytic enzymes have been discovered and characterized. In general, the strains that produce thrombolytic enzymes are screened and isolated by using sterile skimmed milk agar with subsequent incubation of culture at 37 °C for 24 h; protease-positive isolates form a hydrolytic zone, which is screened by fibrin plate assay [26,27,28,29]. The fibrin plate assay is a precise and sensitive method to quantitate the extent of fibrin breakdown. Briefly, a fibrin clot covering the bottom of a petri dish is treated with the fibrinolytically active solutions, and then the relative amounts of fibrin (converted substrates) are measured by determining the areas of the lysed zones. These enzymes are produced via solid and liquid state fermentation in a medium enriched with carbon sources (such as cellulose, dextran, dextrose, fructose, starch, maltose, ribose, sucrose, galactose, and trehalose) and nitrogen sources (including organic-based (yeast extract, peptone, gelatin, urea, casein, tryptone, and soya meal) and inorganic salts (ammonium chloride, sodium dihydrogen phosphate, ammonium sulfate, ferrous sulfate, potassium nitrate, calcium chloride, ferrous sulfate, and disodium hydrogen phosphate)). Inhibitors such as ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid (EGTA), phenylmethylsulfonyl fluoride (PMSF), and ethylenediaminetetraacetic acid (EDTA); and metal ions (Cu2+, Co2+, Ca2+, Fe3+, Fe2+, K+, Mg2+, Mn2+, Na+, and Zn2+) are supplemented in the culture medium to further enhance the production process of thrombolytic enzymes [30,31]. One of the physiological advantages of microbial-derived thrombolytic enzymes is their stability at a wide range of pH values (5–11) and temperatures (30–70 °C). In addition, molecular cloning approaches abet the characterization of genes encoding target-specific modes of action with minimal side-effects, facilitating the potent application of thrombolytic enzymes in warfare for blood clotting disorders, discussed elsewhere in greater detail [32,33].
To overcome the associated drawbacks of currently available thrombolytic agents, various preliminary evaluation approaches are used to test the pharmacokinetics and dynamics of potential drug candidates of microbial origin with respect to their pharmacological cogency and clinical relevance. Briefly, in vitro and in vivo assays to assess the clot lysis properties of these agents are performed either by using fibrin as a direct substrate or by the production of a fibrin clot using thrombin.
The in vitro assays include: (i) Antiaggregatory activity—measures reduction or inhibition of platelet aggregation [34]; (ii) Anticoagulant and thrombolytic activity—measures ability to prevent blood clot formation and dissolution of an existing blood clot [35,36]; (iii) Clot formation and lysis assay (CloFAL)—periodic quantitative measurement of clot lysis, aid in the clinical evaluation of net hemostatic balance, and bleeding and clotting disorders [37]; (iv) Euglobulin clot lysis assay—rapid and sensitive method of measuring fibrinolysis within the euglobulin fraction [38]; and (v) Thromboelastography (TEG)—the ability to measure the viscoelastic properties of the whole blood clot [39].
The in vivo assays include: (i) Carrageenan-induced thrombosis model—helps assessment of clinically relevant anti-thrombus and thrombolytic agents by demonstrating disappearance of wine-colored thrombus [40]; (ii) D—Dimer test—fibrin derivatives containing cross-linked D—dimer (XDP) domains help monitor fibrinolysis [41]; (iii) Ferric chloride-induced thrombosis model—aids in assessing anti-platelet and anticoagulant drugs [42]; and (iv) Rat groin flap model—an important tool in comparative analysis of various anticoagulants and vasomotor drugs [43]. In addition to promising a better understanding of the role of the drug candidate in alleviating disease pathophysiology and determining its therapeutic potential, the combination of in vitro and in vivo assays also provides propitious translational application of microbial-derived novel thrombolytic agents.
2.1. Promising Microbial Producers of Thrombolytic Enzymes
The past decade has witnessed a surge in the discovery of thrombolytic agents from numerous microbes, and this has kindled the development and characterization of microbial-derived agents with thrombolytic activity and minimal or no side effects. The known microbial producers of thrombolytic enzymes include bacteria, fungi, and algae.
2.1.1. Bacteria
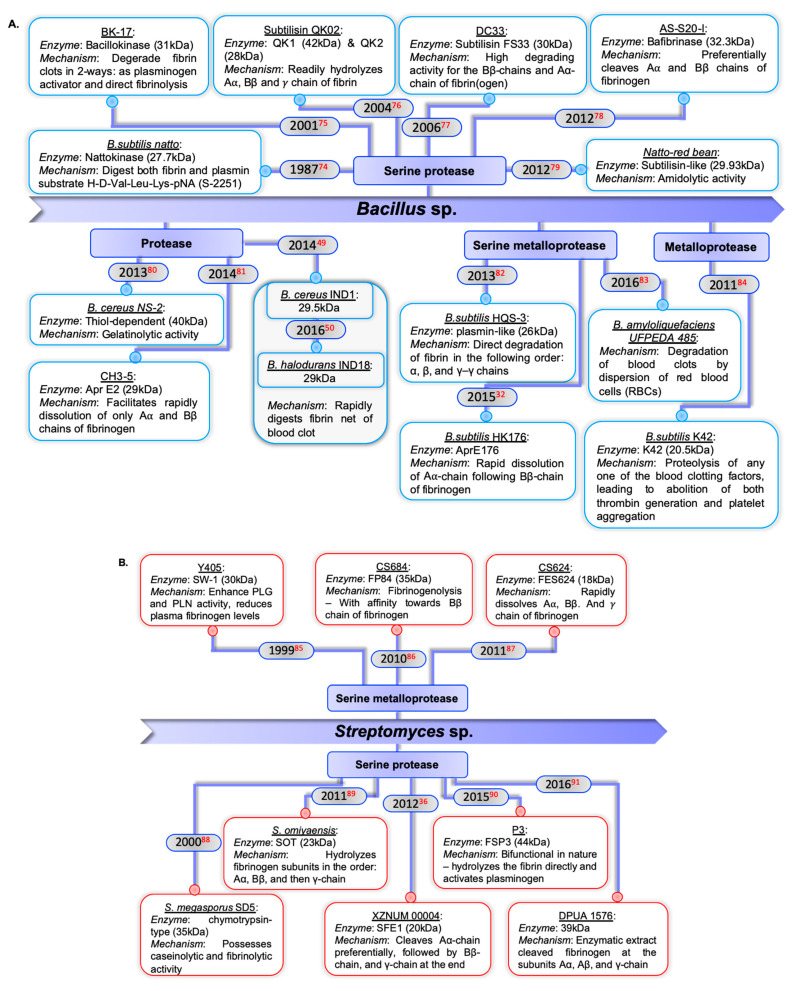
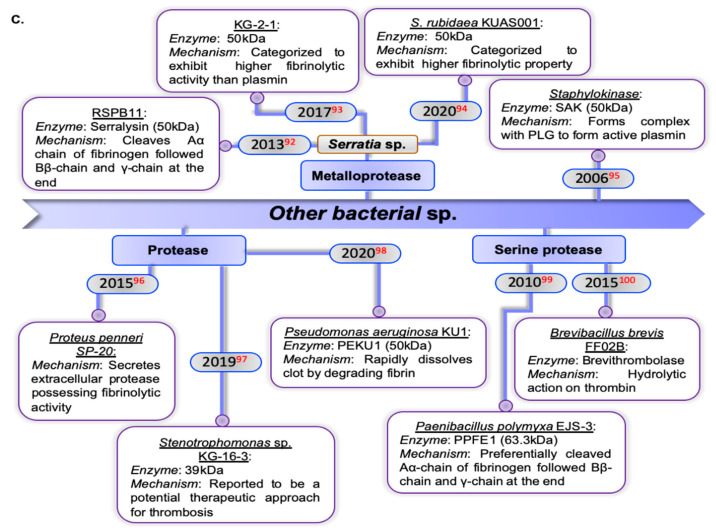
Bacteria are the first-line sources because bacterial proteins are suitable for oral administration and facilitate large-scale production. (a) Bacillus sp. are among the most preferred sources; various strains have been reported to have fibrinolytic activity (Figure 3A). In addition, several other strains with fibrinolytic properties have been reported, although their mode of action is yet to be elucidated, such as Bacillus sp. DJ–2 [44], B. subtilis A26-derived subtilisin BSF1 and BAF1 obtained from B. amyloliquefaciens An6 [45,46], enzyme URAK produced by B. cereus NK1 [47], B. cereus GD 55-derived protease [48], B. cereus IND1 [49] and B. halodurans IND18 [50] are the sources of proteolytic enzymes that exhibit both thrombolytic activity and PLG activation properties, while a fibrinolytic protease with absolute clot dissolution ability in a short span of time (within 4 h) in in vitro conditions was obtained from Bacillus sp. IND12 [51], B. pseudomycoides strain MA02 [52], and B. cereus RSA1 [53]; (b) Streptomyces sp. are the largest fibrinolytic enzyme-producing genus. Figure 3B summarizes the fibrinolytic agents derived from different strains of Streptomyces sp.; (c) other bacterial sp. that have been reported to produce fibrinolytic enzymes are summarized in Figure 3C. Additionally, a proteolytic enzyme of ~50 kDa serrapeptase (SP) or serralysin derived from enterobacterium Serratia E–15 [54] was noted to have fibrinolytic potential [55] along with the ability to distinguish and dissolve only dead and damaged tissue without harming the living tissue [55], Treponema denticola was used to produce thrombolytic enzymes [56], proteases of 44 kDa and 64 kDa obtained from Shewanella sp. IND20 and Psuedoalteromonas sp. IND11, respectively, showed direct clot lysis activity as well as PLG activation ability [57,58], Paenibacillus sp. IND8 [59], and Stenotrophomonas maltophilia Gd2 were shown to possess robust fibrinolytic activity [30].
Figure 3.
Bacterial-derived fibrinolytic enzymes and their mode of action. (A) Bacillus sp. [32,49,50,74,75,76,77,78,79,80,81,82,83,84]; (B) Streptomyces sp. [36,85,86,87,88,89,90,91]; (C) other bacterial species [92,93,94,95,96,97,98,99,100].
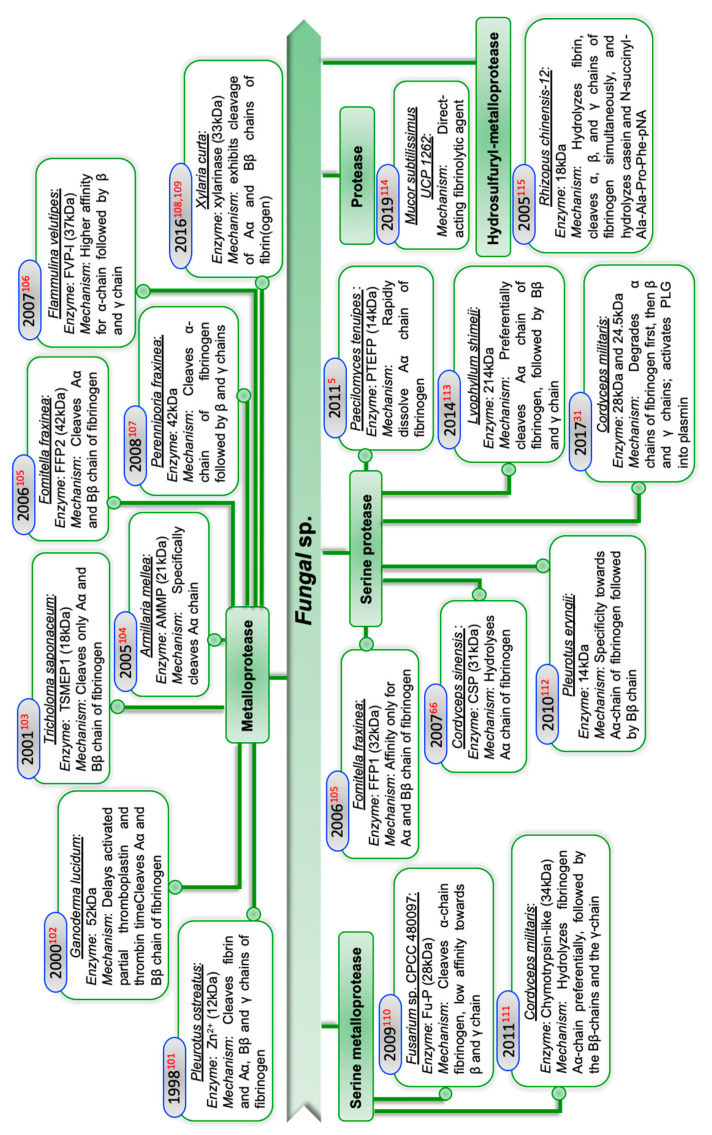
2.1.2. Fungi
The enzymatic properties of fungal sp. are a novel and largely unexplored facet, and there exists exiguous literature on the fibrinolytic potential of these microorganisms. Fungi are considered a suitable source of thrombolytic enzymes because of their ability to grow on solid substrates like agro-industrial waste residues. Different fibrinolytic agents obtained from fungal species and their mechanism of action are outlined in Figure 4. Furthermore, Cochliobolus lunatus and Penicillium chrysogenum H9 were documented as fibrinolytic enzyme producers [60,61], metalloprotease with plasmin-like activity was produced by Fusarium pallidoroseum [62], purified Aspergillus ochraceus 513 was reported to possess both fibrinolytic and anticoagulant potential [63], Oidiodendron flavum is another fibrinolytic producer [64], serine protease isolated from Fusarium BLB were proposed to have fibrinolytic ability [65], 27.3 kDa fibrinolytic enzyme, CMase, is a metalloprotease produced from Cordyceps militaris [66,67], metalloprotease of 18.2 kDa obtained from a medicinal mushroom, Schizophyllum commune, was reported to have fibrin lysis activity [68], Aspergillus oryzae KSK-3-derived serine protease of 30 kDa exhibit thrombolytic potential [69], serine proteases from Bionectria sp. can be the potential treatment for thrombotic diseases [70], alkaline protease and metalloprotease obtained from Aspergillus strain KH 17 and Aspergillus brasiliensis AUMC 9735, respectively, were proposed to have robust fibrinolytic potential [71,72], and protease isolated from Mucor subtilissimus UCP 1262 via two different approaches—solid-state fermentation and aqueous two-phase system—were reported as a potential promising agent for the prevention and therapy against thrombosis [73].
Figure 4.
Fungal-derived enzymes with fibrinolytic potential and their mechanism of action [5,31,66,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115].
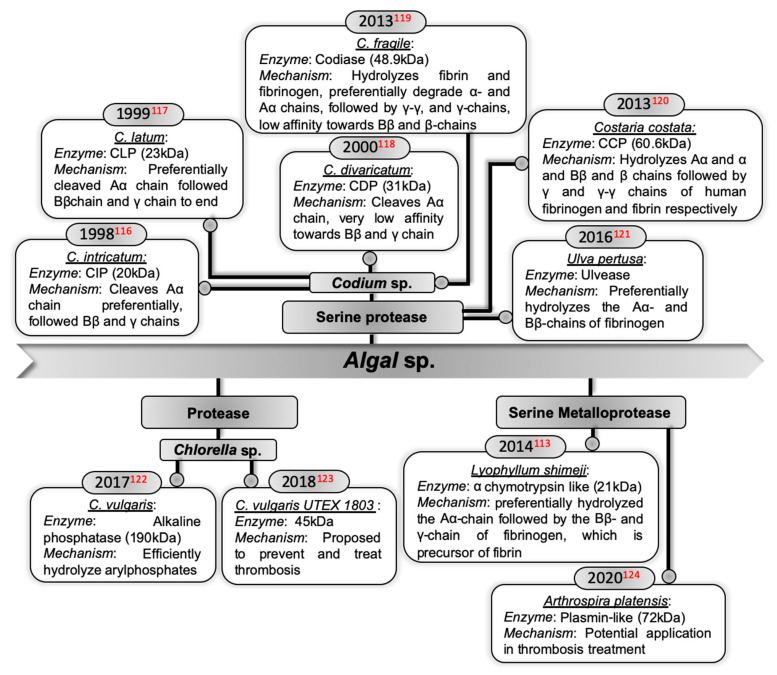
2.1.3. Algae
Algae are excellent sources of various biologically active agents with pleiotropic effects and are rich sources of food, feed, and energy. Few studies have evaluated the potential applications of algal-derived enzymes in fibrinolytic therapies (summarized in Figure 5), thus introducing a new era of bioprospecting of enzymes from algae for their role in thrombolytic activity.
Figure 5.
Fibrinolytic potential of algal-derived enzymes [113,116,117,118,119,120,121,122,123,124].
3. Microbial-Derived Drugs in Thrombolytic Therapy
3.1. Streptokinase
Streptokinase (SK) is the most commonly used first-generation thrombolytic agent. It is a 47 kDa protein and comprises 414 amino acid (AA) residues and exhibits its maximum activity at pH ~7.5 [125]. SK is isolated and derived from several strains of β-hemolytic Streptococci of Lancefield group A, C, and G [126,127]. The Streptococcus equisimilis GCS strain H46A and group C have been chiefly used for SK production [128]. SK comprises three structural domains: α, β, and γ at the 1–146, 147–290, and 291–414 AA positions catenated by two flexible coil regions. SK has a half-life of ~30 min [129,130]. The thrombolytic potential of SK was first acknowledged by Johnson and Tillett in 1952, where they observed that artificially induced intravascular clots within the ear vein of rabbits via sodium morrhuate were successfully dissolved after intravenous infusion of SK [131].
Moreover, the large-scale purification of human plasminogen achieved by Kline [132] allowed the direct application of plasmin as a clot buster. However, the major solicitude associated with direct use of plasmin or SK-mediated plasminogen activation may result in indiscriminate, systemic plasmin generation and substantial diminution of circulating PLG and α-2-antiplasmin [133,134], which can lead to a significant reduction in blood clotting ability, and consequently, a serious risk of hemorrhage. Hence, limiting the application of plasminogen activators as a therapeutic agent rather than circulating plasmin. Correspondingly, Tillette and Sherry reported accompanying adverse effects of SK, which causes pyogenic reactions with symptoms like arthralgia, occasionally nausea, and headache, etc., ergo, limiting the administration of multiple doses of SK [135]. Nevertheless, continued efforts have been made to achieve reduced pyrogenic reaction of SK by using various approaches such as structural and chemical modifications, liposomal entrapment or encapsulation, and domain fusion (Table 2); the recombinant SK (rSK) thus obtained possessed less antigenicity compared to wild-type SK [136,137]. Cloning the SK gene in non-pathogenic microbes permits the production of fortified rSK that eliminates the risk of infection by potentially pathogenic Streptococci [138].
Table 2.
Summary of strategies used for SK modifications.
| Modifications | Type | Examples | Mode of Action | References |
|---|---|---|---|---|
| Structural | Deletion | SK60-386 and SK143-386 | Fibrin-specific activity, lower immunogenicity | [151] |
| Substitution | Lys59 and Lys386 for glutamine | Increases half-life | [152] | |
| Chemical | PEGylation | cysteine-specific thiol-mediated | Increases half-life and stability | [125] |
| Acylation | Human plasminogen-bacterial streptokinase complex | Enhances specificity | [153] | |
| Delivery system | Liposomal entrapment or encapsulation | PEG or chitosan nanoparticles, platelet directed liposomes | Increases half-life and stability, reduced immunogenicity, improved clot penetration properties | [154,155,156] |
| Domain fusion | Chimeric and conjugated protein | Fusion with epidermal growth factor 4, 5, and 6 domains of human thrombomodulin | Reduced risk of re-occlusion and hemorrhage | [157] |
3.1.1. Action Mechanism
SK functions by hijacking the fibrinolysis cascade, where it stoichiometrically binds to plasminogen, inducing a conformational change in plasminogen to form an enzymatically active streptokinase–plasminogen (SK-PLG) complex [139,140]. SK-PLG complex is a highly specific protease that further cleaves other circulating PLG molecules and is capable of converting them to active serine protease, plasmin, that can degrade the fibrin clot via its specific lysine binding site [141,142]. There are several functional regions across SK domains, including: (i) α domain—the Asp41–His48 region between 1-59 AA residues regulates binding to PLG [140,143]. It additionally contains single residues (V19F, V35E, and S44K) for substitution that are crucial for SK-plasmin complex activation [142,144]; (ii) β domain—Lys 256, 257, and Val158–Arg219 region aid in PLG recognition, processing, and SK-plasmin complex formation [145]; and (iii) γ domain—a coiled region (Leu314-Ala342) that plays an essential role in stabilization of SK-micro plasmin complex for PLG activation. SK was the first Food and Drug Administration (FDA)-approved PLG activator for thrombosis therapy.
3.1.2. A Potential Therapeutic Tool
Several clinical trials have been designed to determine the therapeutic potential and safety of SK. These trials concluded the increased survival rate in patients receiving SK during the early onset of myocardial infarction (AMI) [146,147,148]. Ruegsegger and colleagues were the first to prove SK-mediated intracoronary clot dissolution [149]. A subsequent study investigated the protective role of adjuvant intracoronary SK (ICSK) on late-phase infarct size and left ventricle volumes and functions in the setting of primary percutaneous coronary intervention (PCI). This study reported that immediate administration of a low dose of ICSK not only limits the long-term infarct size but also preserves left ventricle volumes and functions [150]. SK is, therefore, the drug of choice in thrombolytic therapy, especially in developing countries due to its reduced cost.
3.2. Staphylokinase (SAK)
SAK is a third-generation plasminogen activator obtained from Staphylococcus aureus GH38 which exerts its anti-thrombin activity by converting passive plasminogen to active plasmin. SAK is a 136 AA monomer of 15.5 kDa and comprises two equal-sized domains with flexible dumbbell shapes [95,158]. Structurally, it has been reported that AA at the 26th position (methionine-26) is useful for PLG activation by SAK. Notably, the functional activity is lost if this AA is replaced with arginine or valine, whereas little or no effect on functional activity was observed by replacing it with leucine or cysteine [159]. SAK was precipitated: (i) initially from the supernatant fluid of cultures at pH around 3.3 with 10 mM HCL; and also (ii) at 75% saturation of (NH4)2SO4 (ammonium sulfate) [160,161]. The half-life of SAK is ~6 min [162]. Lewis and Sweet evaluated the in vitro fibrinolytic properties of SAK [163,164]. Correspondingly, Lewis and Kanae observed in vivo thrombolytic activity of SAK in dogs [165].
3.2.1. Action Mechanism
Fibrin degradation via SAK involves the following two steps: (i) formation of the SAK–PLG complex—this hydrolyzes a peptide bond between lysine 10 and 11 of SAK, which triggers peptide bond lysis between arginine 561 and valine 562 of PLG; (ii) resulting in the initial conversion of PLG to plasmin. SAK then binds to synthesized plasmin directly to catalyze the PLG conversion to plasmin [165]. α2-antiplasmin impedes the SAK-PLG or plasmin complex formation in the absence of fibrin [166,167]. However, the lysine binding domain of the complex is occupied in the presence of fibrin, preventing its inhibition by α2-antiplasmin [158,168]. This suggests the fibrin-specific nature of SAK, and that it can therefore be a potentially potent thrombolytic therapy [158].
3.2.2. A Potential Therapeutic Tool
THR-174 is the optimized form of the SAK sequence variant produced by ThromboGenics NV, Iselin, NJ, USA documented to have augmented efficacy and safety profiles in the pre-clinical trials (2012. Official Website. http://www.thrombogenics.com—Retrieved online on 30 January 2021).
Furthermore, the 3-D structure of the protein facilitated the design of PEG (polyethylene glycol) attachment sites, prolonging plasma half-life and reducing antigenicity [169,170,171,172,173,174]. A recent study reported that N-terminal lipid modification of SAK fosters its activity, stability, and translocation across the blood–brain barrier (BBB), and therefore, holds great promise in treating diseases like stroke [175].
Studies have shown a key role of activated platelets in thrombosis, secondary clot formation, and blood vessel re-blocking. Following thrombolytic therapy, platelet aggregation promotes secondary clot formation. The coagulation cascade is activated by the ensuing clot lysis; in addition, a large amount of thrombin release elicits the platelets activation and aggregation. The activated platelets consequently inhibit fibrin lysis by tPA (tissue plasminogen activator) via the release of type I plasminogen activator inhibitor in the blood circulation, causing vessel re-blockage [176]. Interestingly, RGD (arginine, lysine, and aspartic acid tripeptide)–SAK complex bind to GPIIb/IIIa (glycoprotein membrane receptor) at the platelet surface, which averts the binding of fibrinogen to this receptor, thereby precludes the activated platelets accumulation [168]. Moreover, several thrombin inhibitors combined with recombinant SAK (rSAK) are known to decrease secondary clot formation [177,178,179,180,181,182].
3.3. Nattokinase (NK)
A serine protease derived from natto, a Japanese fermented food, was first extracted by Sumi and college [78] and was termed nattokinase. It is a 275-AA-long single polypeptide chain of 27.7 kDa. Nattokinase possesses the inherent ability to boost the endogenous mechanisms to degrade the blood clots and is achieved via following different ways: (i) oral administration—in vivo studies noted reduced euglobulin clot lysis time (ECLT), prolonged partial thromboplastin time (PATT), and averted platelet aggregation, therefore considered to be a direct-acting fibrinolytic enzyme [183,184]; (ii) intraduodenal administration—existing evidence support the transport of NK across the intestinal tract where it hydrolyzes the plasma fibrinogen [185]; (iii) efficacy—promotes fibrinolysis by degrading plasminogen activator inhibitor-1 (PAI-1) and aiding in plasmin formation by escalating the production of PLG activator [183,186]; and (iv) affinity—lower specificity and more affinity towards fibrinogen and cross-linked fibrin, respectively [187]. In summary, use of NK has several advantages, including efficacy in clot lysis and restoration of arterial blood flow in contrast with plasmin and elastase [188].
Clinical Trials
NK was shown to lower the plasma levels of fibrinogen FVII, and FVIII in two separate groups of patients undergoing dialysis and with cardiovascular risk factors without influencing blood lipids [189], enhanced fibrinolysis and anticoagulant activity [190], lower vWF levels [191], and is under Phase II trial to test its therapeutic ability against atherothrombosis [192].
3.4. Reteplase (Recombinant Plasminogen Activator, r-PA)
Reteplase or r-PA (39 kDa) is produced in E. coli K12 as insoluble inclusion bodies and is an unglycosylated single-chain deletion variant of tPA. Comprised of 1–3 and 176–527 AA of tPA, it therefore only contains the catalytic protease domains and kringle 2, and is deficient in the finger, kringle 1, and epidermal growth factor of t-PA, as signified by the deletion of the Val4-Glu [193,194]. Since the kringle 1 domain of r-PA is crucial for its rapid renal clearance, loss of kringle 1 domain aids in extending the half-life (~from 4 to 15 min) of r-PA [195,196]. Notably, the absence of the fibrin-binding finger domain in r-PA decreases (~5-fold) its binding affinity towards fibrin [194,197]. Interestingly, patients with acute myocardial infarction (AMI) administered with a double bolus regimen (10 + 10 M after 30 min) of r-PA showed robust thrombolysis activity [198,199]. Additionally, in The Global Use of Strategies to Open Occluded Coronary Arteries (GUSTO III) trial, hemorrhagic stroke frequency and mortality post-30 days were comparable in r-PA and alteplase [200]. r-PA is commercially available as Retavase® (Centocor, Inc., Malvern, PA, USA) and Rapilysin® (Roche) for AMI therapy.
4. Use of Waste Biomass/by-Products for Thrombolytic/Fibrinolytic Enzyme Production
There is a constant search for novel and safer fibrinolytic enzymes all over the world owing to the short life cycle and allergic reactions caused by the tPA and certain fibrinolytic agents [201]. The microbial fibrinolytic enzymes are quite economical, and have been studied in insects [202], marine organisms [203], and fermented foods [187,204]. Solid-state fermentation (SSF) is an efficient method for enzyme production and metabolite bioconversion. Agro-industrial waste, fishery waste, and wastewater can be used for the production of fibrinolytic enzymes. A wide variety of agro-wastes, such as groundnut husk [205], green gram husk [206], copra waste [207], deproteinized acid cheese whey and wheat bran [208], and cake of Jatropha curcas seed [201] have been effectively used for the production of enzymes. The solid substrate provides essential nutrients for both the growth of microbes and the production of enzymes. Various studies have shown that B. subtilis can produce a variety of fibrinolytic enzymes [209,210]. Vijayaraghavan and group [50] extracted fibrinolytic enzymes from Bacillus halodurans using agro-wastes. They observed wheat bran to be an efficient substrate for the production of fibrinolytic enzymes which could degrade fibrin clot thus behaving as an effective thrombolytic agent. Wu et al. [211] developed a cost-effective method to optimize the parameters of fermentation for the production of fibrinolytic enzymes by Bacillus subtilis WR350. Their results revealed that sucrose can be used as a low-cost substrate for the production of the fibrinolytic enzyme in a 100-L fermenter by B. subtilis WR350. Fish wastes also act as good substrates for the production of fibrinolytic enzymes due to their high nutrient content [212]. Biji et al. [201] produced fibrinolytic enzymes by Bacillus cereus IND5 using cow dung and cuttlefish waste in SSF. The purified enzyme obtained had a specific activity of 364.5 U/g proteins, the molecular weight of 47 kDa, stability at pH 8.0, high activity at 50 °C and was shown to possess fibrinolytic properties. Thus, the mixture of cuttlefish waste and cow dung had great applications as solid substrates for the production of fibrinolytic enzymes. Hence, use of waste biomass not only yields cost-effective and efficient fibrinolytic enzymes, but also reduces the chances of environmental pollution.
5. Conclusions and Future Perspectives
Vascular occlusion remains a major cause of morbidity and mortality worldwide. Although numerous thrombolytic agents have been identified and characterized from diverse sources, promising scientific data available from in vitro and in vivo studies have failed to translate into clinical trials successfully. Therefore, continuous efforts are needed in the search for more efficacious, safer, and cost-effective thrombolytic drugs. Microbial-derived thrombolytic agents represent a step towards a potent approach in the prevention and treatment of vascular diseases such as CVDs, stroke, transient ischemic attack (TIA), PE, AAAs, venous thromboembolism (VTE), etc. Several thrombolytic enzymes have been reported to be isolated from microbial sources with therapeutic application in vascular diseases and have been shown to possess the following advantages over currently available treatment strategies: (i) extended plasma half-life, (ii) increased fibrin specificity, (iii) high therapeutic index, (iv) lower allergic response, and (v) reduced risk of bleeding complications. Therefore, they promise efficacious translational potential. Both PLG activators and plasmin-like enzymes have been reported to exhibit these advantages. Thus, thrombolytic and fibrinolytic enzymes isolated from microbial sources would spur novel therapeutic strategies for advancing the prospects of these microbial-derived enzyme complexes in the therapeutic armamentarium of drugs.
Acknowledgments
V.K.T. and V.K.G. would like to research and development facilities provided by SRUC, UK.
Author Contributions
Conceptualization, D.D., Z.U. and M.S.; writing—original draft preparation, D.D.; writing—review and editing, Z.U., M.S., J.W.N., G.M., G.C., V.K.T.; supervision, V.K.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Study did not report any data.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Weisel J.W., Litvinov R.I. Visualizing thrombosis to improve thrombus resolution. Res. Pract. Thromb. Haemost. 2021;5:38–50. doi: 10.1002/rth2.12469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mackman N. Triggers, targets and treatments for thrombosis. Nature. 2008;451:914–918. doi: 10.1038/nature06797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Adivitiya, Khasa Y.P. The evolution of recombinant thrombolytics: Current status and future directions. Bioengineered. 2017;8:331–358. doi: 10.1080/21655979.2016.1229718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Choi B.-S., Sapkota K., Choi J.-H., Shin C.-H., Kim S., Kim S.-J. Herinase: A Novel Bi-functional Fibrinolytic Protease from the Monkey Head Mushroom, Hericium erinaceum. Appl. Biochem. Biotechnol. 2013;170:609–622. doi: 10.1007/s12010-013-0206-2. [DOI] [PubMed] [Google Scholar]
- 5.Kim H.C., Choi B.-S., Sapkota K., Kim S., Lee H.J., Yoo J.C., Kim S.-J. Purification and characterization of a novel, highly potent fibrinolytic enzyme from Paecilomyces tenuipes. Process. Biochem. 2011;46:1545–1553. doi: 10.1016/j.procbio.2011.04.005. [DOI] [Google Scholar]
- 6.Rajaselvam J., Benit N., Alotaibi S.S., Rathi M.A., Srigopalram S., Biji G.D., Vijayaraghavan P. In vitro fibrinolytic activity of an enzyme purified from Bacillus amyloliquefaciens strain KJ10 isolated from soybean paste. Saudi J. Biol. Sci. 2021;28:4117–4123. doi: 10.1016/j.sjbs.2021.04.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Key N.S., Makris M., Lillicrap D., editors. Practical Hemostasis and Thrombosis. John Wiley & Sons, Ltd.; Chichester, UK: 2017. [Google Scholar]
- 8.Weisel J.W., Litvinov R.I. Fibrin Formation, Structure and Properties. Subcell. Biochem. 2017;82:405–456. doi: 10.1007/978-3-319-49674-0_13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Asada Y., Yamashita A., Sato Y., Hatakeyama K. Pathophysiology of atherothrombosis: Mechanisms of thrombus formation on disrupted atherosclerotic plaques. Pathol. Int. 2020;70:309–322. doi: 10.1111/pin.12921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Asada Y., Yamashita A., Sato Y., Hatakeyama K. Thrombus Formation and Propagation in the Onset of Cardiovascular Events. J. Atheroscler. Thromb. 2018;25:653–664. doi: 10.5551/jat.RV17022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mukhopadhyay S., Johnson T.A., Duru N., Buzza M.S., Pawar N.R., Sarkar R., Antalis T.M. Fibrinolysis and Inflammation in Venous Thrombus Resolution. Front. Immunol. 2019;10:1348. doi: 10.3389/fimmu.2019.01348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tatli E., Ozcelik F., Aktoz M. Plasma fibrinogen level may predict critical coronary artery stenosis in young adults with myocardial infarction. Cardiol. J. 2009;16:317–320. [PubMed] [Google Scholar]
- 13.Liu J., Zhang Y., Lavie C.J., Tabung F.K., Xu J., Hu Q., He L., Zhang Y. Associations of C-reactive protein and fibrinogen with mortality from all-causes, cardiovascular disease and cancer among U.S. adults. Prev. Med. 2020;139:106044. doi: 10.1016/j.ypmed.2020.106044. [DOI] [PubMed] [Google Scholar]
- 14.Parry D.J., Al-Barjas H.S., Chappell L., Rashid T., Ariens R., Scott D.J.A. Haemostatic and fibrinolytic factors in men with a small abdominal aortic aneurysm. Br. J. Surg. 2009;96:870–877. doi: 10.1002/bjs.6632. [DOI] [PubMed] [Google Scholar]
- 15.Kapetanios D., Karkos C.D., Pliatsios I., Mitka M., Giagtzidis I.T., Konstantinidis K., Papazoglou K.O. Association Between Perioperative Fibrinogen Levels and the Midterm Outcome in Patients Undergoing Elective Endovascular Repair of Abdominal Aortic Aneurysms. Ann. Vasc. Surg. 2019;56:202–208. doi: 10.1016/j.avsg.2018.09.021. [DOI] [PubMed] [Google Scholar]
- 16.Klovaite J., Nordestgaard B.G., Tybjærg-Hansen A., Benn M. Elevated Fibrinogen Levels Are Associated with Risk of Pulmonary Embolism, but Not with Deep Venous Thrombosis. Am. J. Respir. Crit. Care Med. 2013;187:286–293. doi: 10.1164/rccm.201207-1232OC. [DOI] [PubMed] [Google Scholar]
- 17.Danesh J., Lewington S., Thompson S.G., Lowe G.D.O., Collins R., Kostis J.B., Wilson A.C., Folsom A.R., Wu K., Benderly M., et al. Plasma fibrinogen level and the risk of major cardiovascular diseases and nonvascular mortality: An individual participant meta-analysis. J. Am. Med. Assoc. 2005;294:1799–1809. doi: 10.1001/jama.294.14.1799. [DOI] [PubMed] [Google Scholar]
- 18.Chelluboina B., Vemuganti R. Chronic kidney disease in the pathogenesis of acute ischemic stroke. J. Cereb. Blood Flow Metab. 2019;39:1893–1905. doi: 10.1177/0271678X19866733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chang J.C. Hemostasis based on a novel ‘two-path unifying theory’ and classification of hemostatic disorders. Blood Coagul. Fibrinolysis. 2018;29:573–584. doi: 10.1097/MBC.0000000000000765. [DOI] [PubMed] [Google Scholar]
- 20.D’Alessandro E., Posma J., Spronk H., Cate H.T. Tissue factor (:Factor VIIa) in the heart and vasculature: More than an envelope. Thromb. Res. 2018;168:130–137. doi: 10.1016/j.thromres.2018.06.020. [DOI] [PubMed] [Google Scholar]
- 21.Kapoor S., Opneja A., Nayak L. The role of neutrophils in thrombosis. Thromb. Res. 2018;170:87–96. doi: 10.1016/j.thromres.2018.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cesarman-Maus G.C., Hajjar K.A. Molecular mechanisms of fibrinolysis. Br. J. Haematol. 2005;129:307–321. doi: 10.1111/j.1365-2141.2005.05444.x. [DOI] [PubMed] [Google Scholar]
- 23.Mican J., Toul M., Bednar D., Damborsky J. Structural Biology and Protein Engineering of Thrombolytics. Comput. Struct. Biotechnol. J. 2019;17:917–938. doi: 10.1016/j.csbj.2019.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Longstaff C., Thelwell C. Understanding the enzymology of fibrinolysis and improving thrombolytic therapy. FEBS Lett. 2005;579:3303–3309. doi: 10.1016/j.febslet.2005.03.058. [DOI] [PubMed] [Google Scholar]
- 25.Chapin J.C., Hajjar K.A. Fibrinolysis and the control of blood coagulation. Blood Rev. 2015;29:17–24. doi: 10.1016/j.blre.2014.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yogesh D., Halami P.M. A fibrin degrading serine metallo protease of Bacillus circu lans with α-chain specificity. Food Biosci. 2015;11:72–78. doi: 10.1016/j.fbio.2015.04.007. [DOI] [Google Scholar]
- 27.Mahajan P.M., Nayak S., Lele S.S. Fibrinolytic enzyme from newly isolated marine bacterium Bacillus subtilis ICTF-1: Media optimization, purification and characterization. J. Biosci. Bioeng. 2012;113:307–314. doi: 10.1016/j.jbiosc.2011.10.023. [DOI] [PubMed] [Google Scholar]
- 28.Majumdar S., Sarmah B., Gogoi D., Banerjee S., Ghosh S.S., Banerjee S., Chattopadhyay P., Mukherjee A.K. Characteriza-tion, mechanism of anticoagulant action, and assessment of therapeutic potential of a fibrinolytic serine protease (Brevithrombolase) purified from Brevibacillusbrevis strain FF02B. Biochimie. 2014;103:50–60. doi: 10.1016/j.biochi.2014.04.002. [DOI] [PubMed] [Google Scholar]
- 29.Raj A., Khess N., Pujari N., Bhattacharya S., Das A., Rajan S.S. Enhancement of protease production by Pseudomonas aeru-ginosa isolated from dairy effluent sludge and determination of its fibrinolytic potential. Asian Pac. J. Trop. Biomed. 2012;2:S1845–S1851. doi: 10.1016/S2221-1691(12)60506-1. [DOI] [Google Scholar]
- 30.Khursade P.S., Galande S.H., Krishna P.S., Prakasham R. Stenotrophomonas maltophilia Gd2: A potential and novel isolate for fibrinolytic enzyme production. Saudi J. Biol. Sci. 2019;26:1567–1575. doi: 10.1016/j.sjbs.2018.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu X., Kopparapu N.-K., Li Y., Deng Y., Zheng X. Biochemical characterization of a novel fibrinolytic enzyme from Cordyceps militaris. Int. J. Biol. Macromol. 2017;94:793–801. doi: 10.1016/j.ijbiomac.2016.09.048. [DOI] [PubMed] [Google Scholar]
- 32.Jeong S.-J., Heo K., Park J.Y., Lee K.W., Park J.-Y., Joo S.H., Kim J.H. Characterization of AprE176, a fibrinolytic enzyme from Bacillus subtilis HK176. J. Microbiol. Biotechnol. 2015;25:89–97. doi: 10.4014/jmb.1409.09087. [DOI] [PubMed] [Google Scholar]
- 33.Leedagger S.-Y., Yudagger S.-N., Choi H.-J., Kim K.-Y., Kim S.-H., Choi Y.-L., Kim C.-M., Ahn S.-C. Cloning and characterization of a thermostable and alkaline fibrinolytic enzyme from a soil metagenome. Afr. J. Biotechnol. 2013;12:6389–6399. doi: 10.5897/ajb2013.12148. [DOI] [Google Scholar]
- 34.Han F., Yao W., Yang X., Liu X., Gao X. Experimental study on anticoagulant and antiplatelet aggregation activity of a chemically sulfated marine polysaccharide YCP. Int. J. Biol. Macromol. 2005;36:201–207. doi: 10.1016/j.ijbiomac.2005.06.003. [DOI] [PubMed] [Google Scholar]
- 35.Narasimhan M.K., Chandrasekaran M., Rajesh M. Fibrinolytic enzyme production by newly isolated Bacillus cereus SRM-001 with enhanced in-vitro blood clot lysis potential. J. Gen. Appl. Microbiol. 2015;61:157–164. doi: 10.2323/jgam.61.157. [DOI] [PubMed] [Google Scholar]
- 36.Ju X., Cao X., Sun Y., Wang Z., Cao C., Liu J., Jiang J. Purification and characterization of a fibrinolytic enzyme from Streptomyces sp. XZNUM 00004. World J. Microbiol. Biotechnol. 2012;28:2479–2486. doi: 10.1007/s11274-012-1055-9. [DOI] [PubMed] [Google Scholar]
- 37.Chitlur M., Simpson M. Role of global assays in thrombosis and thrombophilia. In: Goldenberg N., Manco-Johnson M., editors. Pediatric Thrombotic Disorders. Cambridge University Press; Cambridge, UK: 2015. pp. 142–157. [DOI] [Google Scholar]
- 38.Nordby E., Arnesen H., Andersen P., Godal H.C. The euglobulin clot lysis time, a rapid and sensitive method for the as-say of fibrinolytic activity after venous stasis. Scand. J. Haematol. 1981;25:407–411. doi: 10.1111/j.1600-0609.1981.tb01421.x. [DOI] [PubMed] [Google Scholar]
- 39.Artang R., Galloway G., Nielsen J.D. Monitoring Novel Anticoagulants Dabigatran, Rivaroxaban and Apixaban Using Thrombelastography. J. Am. Coll. Cardiol. 2014;63:A439. doi: 10.1016/S0735-1097(14)60439-3. [DOI] [Google Scholar]
- 40.Simkhada J.R., Cho S.S., Mander P., Choi Y.H., Yoo J.C. Purification, biochemical properties and antithrombotic effect of a novel Streptomyces enzyme on carrageenan-induced mice tail thrombosis model. Thromb. Res. 2012;129:176–182. doi: 10.1016/j.thromres.2011.09.014. [DOI] [PubMed] [Google Scholar]
- 41.Olson J.D., Cunningham M.T., Higgins R.A., Eby C.S., Brandt J.T. D-dimer: Simple test, tough problems. Arch. Pathol. Lab. Med. 2013;137:1030–1038. doi: 10.5858/arpa.2012-0296-CP. [DOI] [PubMed] [Google Scholar]
- 42.Eckly A., Hechler B., Freund M., Zerr M., Cazenave J.-P., Lanza F., Mangin P.H., Gachet C. Mechanisms underlying FeCl3-induced arterial thrombosis. J. Thromb. Haemost. 2011;9:779–789. doi: 10.1111/j.1538-7836.2011.04218.x. [DOI] [PubMed] [Google Scholar]
- 43.Wallmichrath J., Baumeister R.G., Gottschalk O., Giunta R.E., Frick A. The free groin flap in the rat: A model for improving microsurgical skills and for microvascular perfusion studies. J. Plast. Surg. Hand Surg. 2014;48:191–196. doi: 10.3109/2000656X.2013.852100. [DOI] [PubMed] [Google Scholar]
- 44.Choi N.S., Yoo K.H., Hahm J.H., Yoon K.S., Chang K.T., Hyun B.H., Maeng P.J., Kim S.H. Purification and characteriza-tion of a new peptidase, bacillopeptidase DJ-2, having fibrinolytic activity: Produces by Bacillus sp. DJ-2 from Doen-Jang. J. Microbiol. Biotechnol. 2005;15:72–79. [Google Scholar]
- 45.Agrebi R., Haddar A., Hmidet N., Jellouli K., Manni L., Nasri M. BSF1 fibrinolytic enzyme from a marine bacterium Bacillus subtilis A26: Purification, biochemical and molecular characterization. Process. Biochem. 2009;44:1252–1259. doi: 10.1016/j.procbio.2009.06.024. [DOI] [Google Scholar]
- 46.Agrebi R., Hmidet N., Hajji M., Ktari N., Haddar A., Fakhfakh-Zouari N., Nasri M. Fibrinolytic Serine Protease Isolation from Bacillus amyloliquefaciens An6 Grown on Mirabilis jalapa Tuber Powders. Appl. Biochem. Biotechnol. 2010;162:75–88. doi: 10.1007/s12010-009-8800-z. [DOI] [PubMed] [Google Scholar]
- 47.Deepak V., Ilangovan S., Sampathkumar M.V., Victoria M.J., Pasha S.P.B.S., Pandian S.R.K., Gurunathan S. Medium optimization and immobilization of purified fibrinolytic URAK from Bacillus cereus NK1 on PHB nanoparticles. Enzym. Microb. Technol. 2010;47:297–304. doi: 10.1016/j.enzmictec.2010.07.004. [DOI] [Google Scholar]
- 48.Venkata Naga Raju E., Divakar G. Bacillus Cereus GD 55 Strain Improvement by Physical and Chemical Mutagenesis for Enhanced Production of Fibrinolytic Protease. Int. J. Pharma Sci. Res. 2013;4:81–93. [Google Scholar]
- 49.Vijayaraghavan P., Vincent S.G.P. Statistical Optimization of Fibrinolytic Enzyme Production Using Agroresidues by Bacillus cereus IND1 and Its Thrombolytic ActivityIn Vitro. BioMed Res. Int. 2014;2014:725064. doi: 10.1155/2014/725064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Vijayaraghavan P., Vincent S.G.P., Arasu M.V., Al-Dhabi N.A. Bioconversion of agro-industrial wastes for the production of fibrinolytic enzyme from Bacillus halodurans IND18: Purification and biochemical characterization. Electron. J. Biotechnol. 2016;20:1–8. doi: 10.1016/j.ejbt.2016.01.002. [DOI] [Google Scholar]
- 51.Vijayaraghavan P., Rajendran P., Vincent S.G.P., Arun A., Al-Dhabi N.A., Arasu M.V., Kwon O.Y., Kim Y.O. Novel Sequential Screening and Enhanced Production of Fibrinolytic Enzyme by Bacillus sp. IND12 Using Response Surface Methodology in Solid-State Fermentation. BioMed Res. Int. 2017;2017:3909657. doi: 10.1155/2017/3909657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Chandramohan M., Yee C.Y., Beatrice P.H.K., Ponnaiah P., Narendrakumar G., Samrot A.V. Production, characterization and optimization of fibrinolytic protease from Bacillus pseudomycoides strain MA02 isolated from poultry slaughter house soils. Biocatal. Agric. Biotechnol. 2019;22:101371. doi: 10.1016/j.bcab.2019.101371. [DOI] [Google Scholar]
- 53.Sharma C., Salem G.E.M., Sharma N., Gautam P., Singh R. Thrombolytic Potential of Novel Thiol-Dependent Fibrinolytic Protease from Bacillus cereus RSA1. Biomolecules. 2019;10:3. doi: 10.3390/biom10010003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Miyata K., Maejima K., Tomoda K., Isono M. Serratia Protease Part I. Purification and General Properties of the Enzyme. Agric. Biol. Chem. 1970;34:310–318. doi: 10.1271/bbb1961.34.310. [DOI] [Google Scholar]
- 55.Bhagat S., Agarwal M., Roy V. Serratiopeptidase: A systematic review of the existing evidence. Int. J. Surg. 2013;11:209–217. doi: 10.1016/j.ijsu.2013.01.010. [DOI] [PubMed] [Google Scholar]
- 56.Rosen G., Naor R., Kutner S., Sela M.N. Characterization of fibrinolytic activities of Treponema denticola. Infect. Immun. 1994;62:1749–1754. doi: 10.1128/iai.62.5.1749-1754.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Vijayaraghavan P., Raj S.F., Vi S.G.P. Purification and Characterization of Fibrinolytic Enzyme from Pseudoalteromonas sp., IND11 and its in vitro Activity on Blood Clot. Int. J. Biol. Chem. 2014;9:11–20. doi: 10.3923/ijbc.2015.11.20. [DOI] [Google Scholar]
- 58.Vijayaraghavan P., Vincent S.P. A low cost fermentation medium for potential fibrinolytic enzyme production by a newly isolated marine bacterium, Shewanella sp. IND20. Biotechnol. Rep. 2015;7:135–142. doi: 10.1016/j.btre.2015.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Vijayaraghavan P., Vincent S.G.P., Arasu M.V. Purification, Characterization of a Novel Fibrinolytic Enzyme from Paenibacillus sp. IND8, and its in Vitro Thrombolytic Activity. South Indian J. Biol. Sci. 2016;2:434. doi: 10.22205/sijbs/2016/v2/i4/103450. [DOI] [Google Scholar]
- 60.Abdel-Fattah A.F., Ismail A.-M.S. Purification and some properties of pure Cochliobolus lunatus fibrinolytic Enzyme. Biotechnol. Bioeng. 1984;26:407–411. doi: 10.1002/bit.260260502. [DOI] [PubMed] [Google Scholar]
- 61.El-Aassar S., El-Badry H., Abdel-Fattah A. The biosynthesis of proteases with fibrinolytic activity in immobilized cultures of Penicillium chrysogenum H9. Appl. Microbiol. Biotechnol. 1990;33:26–30. doi: 10.1007/BF00170564. [DOI] [PubMed] [Google Scholar]
- 62.El-Aassar S.A. Production and properties of fibrinolytic enzyme in solid state cultures of Fusarium pallidoroseum. Biotechnol. Lett. 1995;17:943–948. doi: 10.1007/BF00127431. [DOI] [Google Scholar]
- 63.Batomunkueva B.P., Egorov N.S. Isolation, Purification, and Resolution of the Extracellular Proteinase Complex of Aspergillus ochraceus 513 with Fibrinolytic and Anticoagulant Activities. Microbiology. 2001;70:519–522. doi: 10.1023/A:1012343718772. [DOI] [PubMed] [Google Scholar]
- 64.Tharwat N.A.T. Purification and Biochemical Characterization of Fibrinolytic Enzyme Produced by Thermophilic Fungus Oidiodendron flavum. Biotechnology. 2006;5:160–165. doi: 10.3923/biotech.2006.160.165. [DOI] [Google Scholar]
- 65.Ueda M., Kubo T., Miyatake K., Nakamura T. Purification and characterization of fibrinolytic alkaline protease from Fusarium sp. BLB. Appl. Microbiol. Biotechnol. 2007;74:331–338. doi: 10.1007/s00253-006-0621-1. [DOI] [PubMed] [Google Scholar]
- 66.Li H.-P., Hu Z., Yuan J.-L., Fan H.-D., Chen W., Wang S.-J., Zheng S.-S., Zheng Z.-L., Zou G.-L. A novel extracellular protease with fibrinolytic activity from the culture supernatant of Cordyceps sinensis: Purification and characterization. Phytother. Res. 2007;21:1234–1241. doi: 10.1002/ptr.2246. [DOI] [PubMed] [Google Scholar]
- 67.Cui L., Dong M.S., Chen X.H., Jiang M., Lv X., Yan G. A novel fibrinolytic enzyme from Cordyceps militaris, a Chinese traditional medicinal mushroom. World J. Microbiol. Biotechnol. 2008;24:483–489. doi: 10.1007/s11274-007-9497-1. [DOI] [Google Scholar]
- 68.Lu C.-L., Chen S.-N. Purification and characterization of a novel fibrinolytic protease from Schizophyllum commune. J. Food Drug Anal. 2010;18:4. doi: 10.38212/2224-6614.2283. [DOI] [Google Scholar]
- 69.Shirasaka N., Naitou M., Okamura K., Fukuta Y., Terashita T., Kusuda M. Purification and characterization of a fibrinolytic protease from Aspergillus oryzae KSK-3. Mycoscience. 2012;53:354–364. doi: 10.1007/S10267-011-0179-3. [DOI] [Google Scholar]
- 70.Rovati J.I., Delgado O.D., Figueroa L.I.C., Fariña J.I. A novel source of fibrinolytic activity: Bionectria sp., an unconventional enzyme-producing fungus isolated from Las Yungas rainforest (Tucumán, Argentina) World J. Microbiol. Biotechnol. 2009;26:55–62. doi: 10.1007/s11274-009-0142-z. [DOI] [Google Scholar]
- 71.Palanivel P., Ashokkumar L., Balagurunathan R. Production, purification and fibrinolytic characterization of alkaline protease from extremophilic soil fungi. Int. J. Pharma Bio Sci. 2013;4:101–110. [Google Scholar]
- 72.Kotb E., Helal G.E.-D.A., Edries F.M. Screening for fibrinolytic filamentous fungi and enzymatic properties of the most potent producer, Aspergillus brasiliensis AUMC 9735. Biologia. 2015;70:1565–1574. doi: 10.1515/biolog-2015-0192. [DOI] [Google Scholar]
- 73.Nascimento T.P., Sales A., Porto C.S., Brandão R.M.P., Campos-Takaki G.M., Teixeira J., Porto T., Porto A.L.F., Converti A. Purification of a fibrinolytic protease from Mucor subtilissimus UCP 1262 by aqueous two-phase systems (PEG/sulfate) J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2016;1025:16–24. doi: 10.1016/j.jchromb.2016.04.046. [DOI] [PubMed] [Google Scholar]
- 74.Sumi H., Hamada H., Tsushima H., Mihara H., Muraki H. A novel fibrinolytic enzyme (nattokinase) in the vegetable cheese Natto; a typical and popular soybean food in the Japanese diet. Experientia. 1987;43:1110–1111. doi: 10.1007/BF01956052. [DOI] [PubMed] [Google Scholar]
- 75.Jeong Y.K., Park J.U., Baek H., Park S.H., Kong I.S., Kim D.W., Joo W.H. Purification and biochemical characterization of a fibrinolytic enzyme from Bacillus subtilis BK-17. World J. Microbiol. Biotechnol. 2001;17:89–92. doi: 10.1023/A:1016685411809. [DOI] [Google Scholar]
- 76.Ko J.H., Yan J.P., Zhu L., Qi Y.P. Identification of two novel fibrinolytic enzymes from Bacillus subtilis QK02. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2004;137:65–74. doi: 10.1016/j.cca.2003.11.008. [DOI] [PubMed] [Google Scholar]
- 77.Wang C.T., Ji B.P., Li B., Nout R., Li P.L., Ji H., Chen L.F. Purification and characterization of a fibrinolytic enzyme of Bacillus subtilis DC33, isolated from Chinese traditional Douchi. J. Ind. Microbiol. Biotechnol. 2006;33:750–758. doi: 10.1007/s10295-006-0111-6. [DOI] [PubMed] [Google Scholar]
- 78.Mukherjee A.K., Rai S.K., Thakur R., Chattopadhyay P., Kar S.K. Bafibrinase: A non-toxic, non-hemorrhagic, direct-acting fibrinolytic serine protease from Bacillus sp. strain AS-S20-I exhibits in vivo anticoagulant activity and thrombolytic potency. Biochimie. 2012;94:1300–1308. doi: 10.1016/j.biochi.2012.02.027. [DOI] [PubMed] [Google Scholar]
- 79.Chang C.-T., Wang P.-M., Hung Y.-F., Chung Y.-C. Purification and biochemical properties of a fibrinolytic enzyme from Bacillus subtilis-fermented red bean. Food Chem. 2012;133:1611–1617. doi: 10.1016/j.foodchem.2012.02.061. [DOI] [Google Scholar]
- 80.Bajaj B.K., Sharma N., Singh S. Enhanced production of fibrinolytic protease from Bacillus cereus NS-2 using cotton seed cake as nitrogen source. Biocatal. Agric. Biotechnol. 2013;2:204–209. doi: 10.1016/j.bcab.2013.04.003. [DOI] [Google Scholar]
- 81.Jeong S.-J., Cho K.M., Lee C.K., Kim G.M., Shin J.-H., Kim J.S., Kim J.H. Overexpression of aprE2, a Fibrinolytic Enzyme Gene from Bacillus subtilis CH3-5, in Escherichia coli and the Properties of AprE2. J. Microbiol. Biotechnol. 2014;24:969–978. doi: 10.4014/jmb.1401.01034. [DOI] [PubMed] [Google Scholar]
- 82.Huang S., Pan S., Chen G., Huang S., Zhang Z., Li Y., Liang Z. Biochemical characteristics of a fibrinolytic enzyme purified from a marine bacterium, Bacillus subtilis HQS-3. Int. J. Biol. Macromol. 2013;62:124–130. doi: 10.1016/j.ijbiomac.2013.08.048. [DOI] [PubMed] [Google Scholar]
- 83.De Souza F.A.S.D., Sales A.E., Silva P.E.C.E., Bezerra R.P., Silva G.M.D.M.E., De Araújo J.M., Takaki G.M.D.C., Porto T.S., Teixeira J.A.C., Porto A.L.F. Optimization of production, biochemical characterization and in vitro evaluation of the therapeutic potential of fibrinolytic enzymes from a new Bacillus amyloliquefaciens. Macromol. Res. 2016;24:587–595. doi: 10.1007/s13233-016-4089-2. [DOI] [Google Scholar]
- 84.Hassanein W.A., Kotb E., Awny N.M., El-Zawahry Y.A. Fibrinolysis and anticoagulant potential of a metallo protease produced by Bacillus subtilis K42. J. Biosci. 2011;36:773–779. doi: 10.1007/s12038-011-9151-9. [DOI] [PubMed] [Google Scholar]
- 85.Zhao T., Xiong J., Chen W., Xu A., Du Zhu D., Liu J. Purification and Characterization of a Novel Fibrinolytic Enzyme from Cipangopaludina Cahayensis. Iran. J. Biotechnol. 2021;19:121–127. doi: 10.30498/IJB.2021.2805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Simkhada J.R., Mander P., Cho S.S., Yoo J.C. A novel fibrinolytic protease from Streptomyces sp. CS684. Process. Biochem. 2010;45:88–93. doi: 10.1016/j.procbio.2009.08.010. [DOI] [Google Scholar]
- 87.Mander P., Cho S.S., Simkhada J.R., Choi Y.H., Yoo J.C. A low molecular weight chymotrypsin-like novel fibrinolytic enzyme from Streptomyces sp. CS624. Process. Biochem. 2011;46:1449–1455. doi: 10.1016/j.procbio.2011.03.016. [DOI] [Google Scholar]
- 88.Chitte R., Dey S. Potent fibrinolytic enzyme from a thermophilic Streptomyces megasporus strain SD5. Lett. Appl. Microbiol. 2000;31:405–410. doi: 10.1046/j.1365-2672.2000.00831.x. [DOI] [PubMed] [Google Scholar]
- 89.Uesugi Y., Usuki H., Iwabuchi M., Hatanaka T. Highly potent fibrinolytic serine protease from Streptomyces. Enzym. Microb. Technol. 2011;48:7–12. doi: 10.1016/j.enzmictec.2010.08.003. [DOI] [PubMed] [Google Scholar]
- 90.Cheng G., He L., Sun Z., Cui Z., Du Y., Kong Y. Purification and Biochemical Characterization of a Novel Fibrinolytic Enzyme from Streptomyces sp. P3. J. Microbiol. Biotechnol. 2015;25:1449–1459. doi: 10.4014/jmb.1503.03015. [DOI] [PubMed] [Google Scholar]
- 91.Silva G.M.D.M.E., Bezerra R.P., Teixeira J., Silva F.O., Correia J.M., Porto T., Lima-Filho J.L., Porto A.L.F. Sleção, produção e caracterização bioquímica de uma nova enzima fibrinolítica produzida por Streptomyces sp. (Streptomyce taceae) isolada de liquens da Amazônia. Acta Amaz. 2016;46:323–332. doi: 10.1590/1809-4392201600022. [DOI] [Google Scholar]
- 92.Bhargavi P.L., Prakasham R. A fibrinolytic, alkaline and thermostable metalloprotease from the newly isolated Serratia sp RSPB11. Int. J. Biol. Macromol. 2013;61:479–486. doi: 10.1016/j.ijbiomac.2013.07.009. [DOI] [PubMed] [Google Scholar]
- 93.Taneja K., Bajaj B.K., Kumar S., Dilbaghi N. Production, purification and characterization of fibrinolytic enzyme from Serratia sp. KG-2-1 using optimized media. 3 Biotech. 2017;7:184. doi: 10.1007/s13205-017-0808-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Anusree M., Swapna K., Aguilar C., Sabu A. Optimization of process parameters for the enhanced production of fibrinolytic enzyme by a newly isolated marine bacterium. Bioresour. Technol. Rep. 2020;11:100436. doi: 10.1016/j.biteb.2020.100436. [DOI] [Google Scholar]
- 95.Bokarewa M., Jin T., Tarkowski A. Staphylococcus aureus: Staphylokinase. Int. J. Biochem. Cell Biol. 2006;38:504–509. doi: 10.1016/j.biocel.2005.07.005. [DOI] [PubMed] [Google Scholar]
- 96.Jhample S.B., Bhagwat P., Dandge P. Statistical media optimization for enhanced production of fibrinolytic enzyme from newly isolated Proteus penneri SP-20. Biocatal. Agric. Biotechnol. 2015;4:370–379. doi: 10.1016/j.bcab.2015.05.006. [DOI] [Google Scholar]
- 97.Taneja K., Taneja K., Bajaj B.K., Bajaj B.K., Kumar S., Kumar S., Dilbaghi N., Dilbaghi N., Taneja K., Taneja K., et al. Process optimization for production and purification of novel fibrinolytic enzyme from Stenotrophomonas sp. KG-16-3. Biocatal. Biotransform. 2018;37:124–138. doi: 10.1080/10242422.2018.1504925. [DOI] [Google Scholar]
- 98.Kumar S.S., Haridas M., AbdulHameed S. A novel fibrinolytic enzyme from marine Pseudomonas aeruginosa KU1 and its rapid in vivo thrombolysis with little haemolysis. Int. J. Biol. Macromol. 2020;162:470–479. doi: 10.1016/j.ijbiomac.2020.06.178. [DOI] [PubMed] [Google Scholar]
- 99.Lu F., Lu Z., Bie X., Yao Z., Wang Y., Lu Y., Guo Y. Purification and characterization of a novel anticoagulant and fibrinolytic enzyme produced by endophytic bacterium Paenibacillus polymyxa EJS-3. Thromb. Res. 2010;126:e349–e355. doi: 10.1016/j.thromres.2010.08.003. [DOI] [PubMed] [Google Scholar]
- 100.Majumdar S., Chattopadhyay P., Mukherjee A.K. In Vivo Anticoagulant and Thrombolytic Activities of a Fibrinolytic Serine Protease (Brevithrombolase) with the k-Carrageenan-Induced Rat Tail Thrombosis Model. Clin. Appl. Thromb. 2015;22:594–598. doi: 10.1177/1076029615569567. [DOI] [PubMed] [Google Scholar]
- 101.Choi H.-S., Shin H.-H. Purification and Partial Characterization of a Fibrinolytic Protease in Pleurotus ostreatus. Mycologia. 1998;90:674. doi: 10.2307/3761226. [DOI] [Google Scholar]
- 102.Choi H.-S., Sa Y.-S. Fibrinolytic and Antithrombotic Protease from Ganoderma lucidum. Mycologia. 2000;92:545. doi: 10.2307/3761514. [DOI] [Google Scholar]
- 103.Kim J.-H., Kim Y.S. Characterization of a Metalloenzyme from a Wild Mushroom, Tricholoma saponaceum. Biosci. Biotechnol. Biochem. 2001;65:356–362. doi: 10.1271/bbb.65.356. [DOI] [PubMed] [Google Scholar]
- 104.Lee S.-Y., Kim J.-S., Kim J.-E., Sapkota K., Shen M.-H., Kim S., Chun H.-S., Yoo J.-C., Choi H.-S., Kim M.-K., et al. Purification and characterization of fibrinolytic enzyme from cultured mycelia of Armillaria mellea. Protein Expr. Purif. 2005;43:10–17. doi: 10.1016/j.pep.2005.05.004. [DOI] [PubMed] [Google Scholar]
- 105.Lee J.S., Baik H.S., Park S.S. Purification and characterization of two novel fibrinolytic proteases from mushroom, Fomitella fraxinea. J. Microbiol. Biotechnol. 2006;16:264–271. [Google Scholar]
- 106.Park S.-E., Li M.-H., Kim J.-S., Sapkota K., Kim J.-E., Choi B.-S., Yoon Y.-H., Lee J.-C., Lee H.-H., Kim C.-S., et al. Purification and Characterization of a Fibrinolytic Protease from a Culture Supernatant of Flammulina velutipes Mycelia. Biosci. Biotechnol. Biochem. 2007;71:2214–2222. doi: 10.1271/bbb.70193. [DOI] [PubMed] [Google Scholar]
- 107.Kim J.-S., Kim J.-E., Choi B.-S., Park S.-E., Sapkota K., Kim S., Lee H.-H., Kim C.-S., Park Y., Kim M.-K., et al. Purification and characterization of fibrinolytic metalloprotease from Perenniporia fraxinea mycelia. Mycol. Res. 2008;112:990–998. doi: 10.1016/j.mycres.2008.01.029. [DOI] [PubMed] [Google Scholar]
- 108.Meshram V., Saxena S., Paul K. Xylarinase: A novel clot busting enzyme from an endophytic fungus Xylaria curta. J. Enzym. Inhib. Med. Chem. 2016;31:1502–1511. doi: 10.3109/14756366.2016.1151013. [DOI] [PubMed] [Google Scholar]
- 109.Meshram V., Saxena S., Paul K., Gupta M., Kapoor N. Production, Purification and Characterisation of a Potential Fibrinolytic Protease from Endophytic Xylaria curta by Solid Substrate Fermentation. Appl. Biochem. Biotechnol. 2016;181:1496–1512. doi: 10.1007/s12010-016-2298-y. [DOI] [PubMed] [Google Scholar]
- 110.Wu B., Wu L., Chen D., Yang Z., Luo M. Purification and characterization of a novel fibrinolytic protease from Fusarium sp. CPCC 480097. J. Ind. Microbiol. Biotechnol. 2009;36:451–459. doi: 10.1007/s10295-008-0516-5. [DOI] [PubMed] [Google Scholar]
- 111.Choi D., Cha W.-S., Park N., Kim H.-W., Lee J.H., Park J.S., Park S.-S. Purification and characterization of a novel fibrinolytic enzyme from fruiting bodies of Korean Cordyceps militaris. Bioresour. Technol. 2011;102:3279–3285. doi: 10.1016/j.biortech.2010.10.002. [DOI] [PubMed] [Google Scholar]
- 112.Cha W.-S., Park S.-S., Kim S.-J., Choi D. Biochemical and enzymatic properties of a fibrinolytic enzyme from Pleurotus eryngii cultivated under solid-state conditions using corn cob. Bioresour. Technol. 2010;101:6475–6481. doi: 10.1016/j.biortech.2010.02.048. [DOI] [PubMed] [Google Scholar]
- 113.Moon S.-M., Kim J.-S., Kim H.-J., Choi M.S., Park B.R., Kim S.-G., Ahn H., Chun H.S., Shin Y.K., Kim J.-J., et al. Purification and characterization of a novel fibrinolytic α chymotrypsin like serine metalloprotease from the edible mushroom, Lyophyllum shimeji. J. Biosci. Bioeng. 2013;117:544–550. doi: 10.1016/j.jbiosc.2013.10.019. [DOI] [PubMed] [Google Scholar]
- 114.da Silva M.M., Rocha T.A., de Moura D.F., Chagas C.A., Júnior F.C.A.D.A., Santos N.P.D.S., Sobral R.V.D.S., Nascimento J.M.D., Leite A.C.L., Pastrana L., et al. Effect of acute exposure in swiss mice (Mus musculus) to a fibrinolytic protease produced by Mucor subtilissimus UCP 1262: An histomorphometric, genotoxic and cytological approach. Regul. Toxicol. Pharmacol. 2019;103:282–291. doi: 10.1016/j.yrtph.2019.02.009. [DOI] [PubMed] [Google Scholar]
- 115.Liu X.-L., Du L.-X., Lu F.-P., Zheng X.-Q., Xiao J. Purification and characterization of a novel fibrinolytic enzyme from Rhizopus chinensis-12. Appl. Microbiol. Biotechnol. 2004;67:209–214. doi: 10.1007/s00253-004-1846-5. [DOI] [PubMed] [Google Scholar]
- 116.Matsubara K., Sumi H., Hori K., Miyazawa K. Purification and Characterization of Two Fibrinolytic Enzymes from a Marine Green Alga, Codium intricatum. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 1998;119:177–181. doi: 10.1016/S0305-0491(97)00303-9. [DOI] [PubMed] [Google Scholar]
- 117.Matsubara K., Hori K., Matsuura Y., Miyazawa K. A fibrinolytic enzyme from a marine green alga, Codium latum. Phytochemistry. 1999;52:993–999. doi: 10.1016/S0031-9422(99)00356-8. [DOI] [PubMed] [Google Scholar]
- 118.Matsubara K., Hori K., Matsuura Y., Miyazawa K. Purification and characterization of a fibrinolytic enzyme and identification of fibrinogen clotting enzyme in a marine green alga, Codium divaricatum. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2000;125:137–143. doi: 10.1016/S0305-0491(99)00161-3. [DOI] [PubMed] [Google Scholar]
- 119.Choi J.-H., Sapkota K., Park S.-E., Kim S., Kim S.-J. Thrombolytic, anticoagulant and antiplatelet activities of codiase, a bi-functional fibrinolytic enzyme from Codium fragile. Biochimie. 2013;95:1266–1277. doi: 10.1016/j.biochi.2013.01.023. [DOI] [PubMed] [Google Scholar]
- 120.Kim D.-W., Sapkota K., Choi J.-H., Kim Y.-S., Kim S., Kim S.-J. Direct acting anti-thrombotic serine protease from brown seaweed Costaria costata. Process. Biochem. 2012;48:340–350. doi: 10.1016/j.procbio.2012.12.012. [DOI] [Google Scholar]
- 121.Kang S.-R., Choi J.-H., Kim D.-W., Park S.-E., Sapkota K., Kim S., Kim S.-J. A bifunctional protease from green alga Ulva pertusa with anticoagulant properties: Partial purification and characterization. Environ. Boil. Fishes. 2015;28:599–607. doi: 10.1007/s10811-015-0550-4. [DOI] [Google Scholar]
- 122.da Costa e Silva P.E., de Souza F.A.S.D., de Barros R.C., Marques D.A.V., Porto A.L.F., Bezerra R.P. Enhanced Production of Fibrinolytic Protease from Microalgae Chlorella Vulgaris using Glycerol and Corn Steep Liquor as Nutrient. Ann. Microbiol. Res. 2017;1:9–19. doi: 10.36959/958/564. [DOI] [Google Scholar]
- 123.Silva P., de Barros R.C., Albuquerque W., Brandão R.M.P., Bezerra R.P., Porto A.L.F. In vitro thrombolytic activity of a purified fibrinolytic enzyme from Chlorella vulgaris. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2018;1092:524–529. doi: 10.1016/j.jchromb.2018.04.040. [DOI] [PubMed] [Google Scholar]
- 124.de Barros P.D.S., e Silva P.E.C., Nascimento T.P., Costa R.M.P.B., Bezerra R.P., Porto A.L.F. Fibrinolytic enzyme from Arthrospira platensis cultivated in medium culture supplemented with corn steep liquor. Int. J. Biol. Macromol. 2020;164:3446–3453. doi: 10.1016/j.ijbiomac.2020.08.217. [DOI] [PubMed] [Google Scholar]
- 125.Banerjee A., Chisti Y., Banerjee U. Streptokinase—a clinically useful thrombolytic agent. Biotechnol. Adv. 2004;22:287–307. doi: 10.1016/j.biotechadv.2003.09.004. [DOI] [PubMed] [Google Scholar]
- 126.Babu V., Devi C.S. In vitro thrombolytic activity of purified streptokinase extracted from Streptococcus equinus VIT_VB2 isolated from bovine milk. J. Thromb. Thrombolysis. 2014;39:71–78. doi: 10.1007/s11239-014-1093-2. [DOI] [PubMed] [Google Scholar]
- 127.Yadav S., Datt M., Singh B., Sahni G. Role of the 88–97 loop in plasminogen activation by streptokinase probed through site-specific mutagenesis. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2008;1784:1310–1318. doi: 10.1016/j.bbapap.2008.05.013. [DOI] [PubMed] [Google Scholar]
- 128.Vadla P.K., Tummuru M., Kumar D. Purification and characterization of recombinant Streptokinase expressed in E.coli from Streptococcus equisimilis with N-terminal methionine. J. Teknol. Lab. 2019;8:8–17. doi: 10.29238/teknolabjournal.v8i1.153. [DOI] [Google Scholar]
- 129.Anderson J.L. Development and evaluation of anisoylated plasminogen streptokinase activator complex (APSAC) as a second generation thrombolytic agent. J. Am. Coll. Cardiol. 1987;10:22B–27B. doi: 10.1016/S0735-1097(87)80424-2. [DOI] [PubMed] [Google Scholar]
- 130.Nguyen T.H.T. High-level expression, purification and properties of a fully active even glycosylated staphylokinase variant SakfC from Staphylococcus aureus QT08 in Pichia pastoris. Afr. J. Microbiol. Res. 2012;6:2129–2136. doi: 10.5897/ajmr11.1453. [DOI] [Google Scholar]
- 131.Johnson A.J., Tillett W.S. The Lysis in Rabbits of Intravascular Blood Clots by The Streptococcal Fibrinolytic System (Streptokinase) J. Exp. Med. 1952;95:449–464. doi: 10.1084/jem.95.5.449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Kline D.L. The Purification and Crystallization of Plasminogen (Profibrinolysin) J. Biol. Chem. 1953;204:949–955. doi: 10.1016/S0021-9258(18)66098-3. [DOI] [PubMed] [Google Scholar]
- 133.Alkjaersig N., Fletcher A.P., Sherry S. The Mechanism of Clot Dissolution by Plasmin. J. Clin. Investig. 1959;38:1086–1095. doi: 10.1172/JCI103885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Fletcher A.P., Sherry S., Alkjaersig N., Smyrniotis F.E., Jick S. The maintenance of a sustained thrombolytic state in man. II. clinical observations on patients with myocardial infarction and other thromboembolic disorders. J. Clin. Investig. 1959;38:1111–1119. doi: 10.1172/JCI103887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Tillett W.S., Sherry S. The effect in patients of streptococcal fibrinolysin (streptokinase) and streptococcal desoxyribonuclease on fibrinous, purulent, and sanguinous pleural exudations 1. J. Clin. Investig. 1949;28:173–190. doi: 10.1172/JCI102046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Collen D., Moreau H., Stockx L., Vanderschueren S. Recombinant Staphylokinase Variants with Altered Immunoreactivity.. II: Thrombolytic properties and antibody induction. Circulation. 1996;94:207–216. doi: 10.1161/01.CIR.94.2.207. [DOI] [PubMed] [Google Scholar]
- 137.Roohvand F. Streptokinase for treatment of thrombotic disorders: The end? Or the end of the beginning? Iran. Biomed. J. 2018;22:140–141. [PMC free article] [PubMed] [Google Scholar]
- 138.Malke H., Ferretti J.J. Streptokinase: Cloning, expression, and excretion by Escherichia coli. Proc. Natl. Acad. Sci. USA. 1984;81:3557–3561. doi: 10.1073/pnas.81.11.3557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Nolan M., Bouldin S.D., Bock P.E. Full Time Course Kinetics of the Streptokinase-Plasminogen Activation Pathway. J. Biol. Chem. 2013;288:29482–29493. doi: 10.1074/jbc.M113.477935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Verhamme I.M., Bock P.E. Rapid Binding of Plasminogen to Streptokinase in a Catalytic Complex Reveals a Three-step Mechanism. J. Biol. Chem. 2014;289:28006–28018. doi: 10.1074/jbc.M114.589077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Castellino F.J. Recent advances in the chemistry of the fibrinolytic system. Chem. Rev. 1981;81:431–446. doi: 10.1021/cr00045a001. [DOI] [Google Scholar]
- 142.Kazemi F., Arab S.S., Mohajel N., Keramati M., Niknam N., Aslani M.M., Roohvand F. Computational simulations assessment of mutations impact on streptokinase (SK) from a group G streptococci with enhanced activity—Insights into the functional roles of structural dynamics flexibility of SK and stabilization of SK–μplasmin catalytic complex. J. Biomol. Struct. Dyn. 2018;37:1944–1955. doi: 10.1080/07391102.2018.1472668. [DOI] [PubMed] [Google Scholar]
- 143.Sazonova I.Y., Robinson B.R., Gladysheva I.P., Castellino F.J., Reed G.L. α Domain Deletion Converts Streptokinase into a Fibrin-dependent Plasminogen Activator through Mechanisms Akin to Staphylokinase and Tissue Plasminogen Activator. J. Biol. Chem. 2004;279:24994–25001. doi: 10.1074/jbc.M400253200. [DOI] [PubMed] [Google Scholar]
- 144.Aneja R., Datt M., Yadav S., Sahni G. Multiple Exosites Distributed across the Three Domains of Streptokinase Co-Operate to Generate High Catalytic Rates in the Streptokinase–Plasmin Activator Complex. Biochemistry. 2013;52:8957–8968. doi: 10.1021/bi400142s. [DOI] [PubMed] [Google Scholar]
- 145.Tharp A.C., Laha M., Panizzi P., Thompson M., Fuentes-Prior P., Bock P.E. Plasminogen Substrate Recognition by the Streptokinase-Plasminogen Catalytic Complex Is Facilitated by Arg253, Lys256, and Lys257 in the Streptokinase β-Domain and Kringle 5 of the Substrate. J. Biol. Chem. 2009;284:19511–19521. doi: 10.1074/jbc.M109.005512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Gruppo Italiano per lo Studio Effectiveness of intravenous thrombolytic treatment in acute myocardial infarction. Lancet. 1986;327:397–402. doi: 10.1016/s0140-6736(86)92368-8. [DOI] [PubMed] [Google Scholar]
- 147.ISIS-2 (Second International Study of Infarct Survival) Collaborative Group Randomized trial of intravenous streptokinase, oral aspirin, both, or neither among 17,187 cases of suspected acute myocardial infarction: ISIS-2. J. Am. Coll. Cardiol. 1988;12:A3–A13. doi: 10.1016/0735-1097(88)92635-6. [DOI] [PubMed] [Google Scholar]
- 148.Koren G., Weiss A.T., Hasin Y., Appelbaum D., Welber S., Rozenman Y., Lotan C., Mosseri M., Sapoznikov D., Luria M.H., et al. Prevention of Myocardial Damage in Acute Myocardial Ischemia by Early Treatment with Intravenous Streptokinase. N. Engl. J. Med. 1985;313:1384–1389. doi: 10.1056/NEJM198511283132204. [DOI] [PubMed] [Google Scholar]
- 149.Ruegsegger P., Nydick I., Hutter R.C., Freiman A.H., Bang N.U., Cliffton E.E., Ladue J.S. Fibrinolytic (Plasmin) Therapy of Experimental Coronary Thrombi with Alteration of the Evolution of Myocardial Infarction. Circulation. 1959;19:7–13. doi: 10.1161/01.CIR.19.1.7. [DOI] [PubMed] [Google Scholar]
- 150.Sezer M., Çimen A., Aslanger E., Elitok A., Umman B., Buğra Z., Yormaz E., Türkmen C., Adalet I., Nişanci Y., et al. Effect of Intracoronary Streptokinase Administered Immediately After Primary Percutaneous Coronary Intervention on Long-Term Left Ventricular Infarct Size, Volumes, and Function. J. Am. Coll. Cardiol. 2009;54:1065–1071. doi: 10.1016/j.jacc.2009.04.083. [DOI] [PubMed] [Google Scholar]
- 151.Arabi R., Roohvand F., Norouzian D., Sardari S., Aghasadeghi M.R., Khanahmad H., Memarnejadian A., Motevalli F. A Comparative Study on the Activity and Antigenicity of Truncated and Full-length Forms of Streptokinase. Pol. J. Microbiol. 2011;60:243–251. doi: 10.33073/pjm-2011-034. [DOI] [PubMed] [Google Scholar]
- 152.Adivitiya, Babbal, Mohanty S., Khasa Y.P. Engineering of deglycosylated and plasmin resistant variants of recombinant streptokinase in Pichia pastoris. Appl. Microbiol. Biotechnol. 2018;102:10561–10577. doi: 10.1007/s00253-018-9402-x. [DOI] [PubMed] [Google Scholar]
- 153.Ali M.R., Salim Hossain M., Islam M.A., Saiful Islam Arman M., Sarwar Raju G., Dasgupta P., Noshin T.F. Aspect of Thrombolytic Therapy: A Review. Sci. World J. 2014;2014:586510. doi: 10.1155/2014/586510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Vaidya B., Agrawal G., Vyas S.P. Platelets directed liposomes for the delivery of streptokinase: Development and characterization. Eur. J. Pharm. Sci. 2011;44:589–594. doi: 10.1016/j.ejps.2011.10.004. [DOI] [PubMed] [Google Scholar]
- 155.Baharifar H., Khoobi M., Bidgoli S.A., Amani A. Preparation of PEG-grafted chitosan/streptokinase nanoparticles to improve biological half-life and reduce immunogenicity of the enzyme. Int. J. Biol. Macromol. 2019;143:181–189. doi: 10.1016/j.ijbiomac.2019.11.157. [DOI] [PubMed] [Google Scholar]
- 156.Hasanpour A., Esmaeili F., Hosseini H., Amani A. Use of mPEG-PLGA nanoparticles to improve bioactivity and hemocompatibility of streptokinase: In-vitro and in-vivo studies. Mater. Sci. Eng. C Mater. Biol. Appl. 2020;118:111427. doi: 10.1016/j.msec.2020.111427. [DOI] [PubMed] [Google Scholar]
- 157.Maheshwari N., Kantipudi S., Maheshwari A., Arora K., Vandana, Kwatra N., Sahni G. Amino-Terminal Fusion of Epidermal Growth Factor 4,5,6 Domains of Human Thrombomodulin on Streptokinase Confers Anti-Reocclusion Characteristics along with Plasmin-Mediated Clot Specificity. PLoS ONE. 2016;11:e0150315. doi: 10.1371/journal.pone.0150315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Nedaeinia R., Faraji H., Javanmard S.H., Ferns G.A., Ghayour-Mobarhan M., Goli M., Mashkani B., Nedaeinia M., Haghighi M.H.H., Ranjbar M. Bacterial staphylokinase as a promising third-generation drug in the treatment for vascular occlusion. Mol. Biol. Rep. 2020;47:819–841. doi: 10.1007/s11033-019-05167-x. [DOI] [PubMed] [Google Scholar]
- 159.Schlott B., Hartmann M., Gührs K.-H., Birch-Hirschfeld E., Gase A., Vettermann S., Collen D., Lijnen H.R. Functional properties of recombinant staphylokinase variants obtained by site-specific mutagenesis of methionine-26. Biochim. Biophys. Acta (BBA)-Protein Struct. Mol. Enzym. 1994;1204:235–242. doi: 10.1016/0167-4838(94)90013-2. [DOI] [PubMed] [Google Scholar]
- 160.Davidson F.M. The activation of plasminogen by staphylokinase: Comparison with streptokinase. Biochem. J. 1960;76:56–61. doi: 10.1042/bj0760056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Glanville K.L.A. A simple method of purifying staphylokinase. Biochem. J. 1963;88:11–14. doi: 10.1042/bj0880011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Ross A.M. New plasminogen activators: A clinical review. Clin. Cardiol. 1999;22:165–171. doi: 10.1002/clc.4960220303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Bodey G.P., Stewart D. In Vitro Studies of Tobramycin. Antimicrob. Agents Chemother. 1972;2:109–113. doi: 10.1128/AAC.2.3.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Lewis J.H., Kerber C.W., Wilson J.H. Effects of fibrinolytic agents and heparin on intravascular clot lysis. Am. J. Physiol. 1964;207:1044–1048. doi: 10.1152/ajplegacy.1964.207.5.1044. [DOI] [PubMed] [Google Scholar]
- 165.Lewis J.H., Shirakawa M. Effects of fibrinolytic agents and heparin on blood coagulation in dogs. Am. J. Physiol. Content. 1964;207:1049–1052. doi: 10.1152/ajplegacy.1964.207.5.1049. [DOI] [PubMed] [Google Scholar]
- 166.Sakai M., Watanuki M., Matsuo O. Mechanism of fibrin-specific fibrinolysis by staphylokinase: Participation of α2-plasmin inhibitor. Biochem. Biophys. Res. Commun. 1989;162:830–837. doi: 10.1016/0006-291X(89)92385-1. [DOI] [PubMed] [Google Scholar]
- 167.Lijnen H.R., Van Hoef B., Matsuo O., Collen D. On the molecular interactions between plasminogen-staphylokinase, α2-antiplasmin and fibrin. Biochim. Biophys. Acta (BBA)-Protein Struct. Mol. Enzym. 1992;1118:144–148. doi: 10.1016/0167-4838(92)90142-Z. [DOI] [PubMed] [Google Scholar]
- 168.Pulicherla K.K., Kumar A., Gadupudi G.S., Kotra S.R., Rao K.R.S.S. In VitroCharacterization of a Multifunctional Staphylokinase Variant with Reduced Reocclusion, Produced from Salt InducibleE. coliGJ1158. BioMed Res. Int. 2013;2013:297305. doi: 10.1155/2013/297305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Ram K.S., Jb P., Kumar A., Rao K.S., Pulicherla K. Staphylokinase: A boon in medical sciences. Mintage J. Pharm. Med. Sci. 2013;2:28–34. [Google Scholar]
- 170.Laroche Y., Heymans S., Capaert S., De Cock F., Demarsin E., Collen D. Recombinant staphylokinase variants with reduced antigenicity due to elimination of B-lymphocyte epitopes. Blood. 2000;96:1425–1432. doi: 10.1182/blood.V96.4.1425. [DOI] [PubMed] [Google Scholar]
- 171.Szemraj J., Walkowiak B., Kawecka I., Janiszewska G., Buczko W., Bartkowiak J., Chabielska E. A new recombinant thrombolytic and antithrombotic agent with higher fibrin affinity—A staphylokinase variant. I. In vitro study. J. Thromb. Haemost. 2005;3:2156–2165. doi: 10.1111/j.1538-7836.2005.01480.x. [DOI] [PubMed] [Google Scholar]
- 172.Moreadith R.W., Collen D. Clinical development of PEGylated recombinant staphylokinase (PEG–Sak) for bolus thrombolytic treatment of patients with acute myocardial infarction. Adv. Drug Deliv. Rev. 2003;55:1337–1345. doi: 10.1016/S0169-409X(03)00113-3. [DOI] [PubMed] [Google Scholar]
- 173.Moons L., Vanlinthout I., Roelants I., Moreadith R., Collen D., Rapold H.J. Toxicology Studies with Recombinant Staphylokinase and with SY 161-P5, a Polyethylene Glycol-Derivatized Cysteine-Substitution Mutant. Toxicol. Pathol. 2001;29:285–291. doi: 10.1080/019262301316905237. [DOI] [PubMed] [Google Scholar]
- 174.Liu J., Wang Z., He J., Wang G., Zhang R., Zhao B. Effect of site-specific PEGylation on the fibrinolytic activity, immunogenicity, and pharmacokinetics of staphylokinase. Acta Biochim. Biophys. Sin. 2014;46:782–791. doi: 10.1093/abbs/gmu068. [DOI] [PubMed] [Google Scholar]
- 175.Mannully S.T., Shanthi C., Pulicherla K.K. Lipid modification of staphylokinase and its implications on stability and activity. Int. J. Biol. Macromol. 2018;121:1037–1045. doi: 10.1016/j.ijbiomac.2018.10.134. [DOI] [PubMed] [Google Scholar]
- 176.Kowalski M., Brown G., Bieniasz M., Oszajca K., Chabielska E., Pietras T., Szemraj Z., Makandjou-Ola E., Bartkowiak J., Szemraj J. Cloning and expression of a new recombinant thrombolytic and anthithrombotic agent—A staphylokinase variant. Acta Biochim. Pol. 2008;56:41–53. doi: 10.18388/abp.2009_2515. [DOI] [PubMed] [Google Scholar]
- 177.Cappello M., Li S., Chen X., Li C.-B., Harrison L., Narashimhan S., Beard C.B., Aksoy S. Tsetse thrombin inhibitor: Bloodmeal-induced expression of an anticoagulant in salivary glands and gut tissue of Glossina morsitans. Proc. Natl. Acad. Sci. USA. 1998;95:14290–14295. doi: 10.1073/pnas.95.24.14290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Wang M., Wang Y., Wang J., Zou M., Liu S., Xu T., Cai X., Wu C., Wang J., Xu D. Construction and characterization of a novel staphylokinase variant with thrombin-inhibitory activity. Biotechnol. Lett. 2009;31:1923–1927. doi: 10.1007/s10529-009-0094-2. [DOI] [PubMed] [Google Scholar]
- 179.Skrzypczak-Jankun E., Carperos V.E., Ravichandran K., Tulinsky A., Westbrook M., Maraganore J.M. Structure of the hirugen and hirulog 1 complexes of α-thrombin. J. Mol. Biol. 1991;221:1379–1393. doi: 10.1016/0022-2836(91)90939-4. [DOI] [PubMed] [Google Scholar]
- 180.van Zyl W.B., Pretorius G.H., Hartmann M., Kotzé H.F. Production of a Recombinant Antithrombotic and Fibrinolytic Protein, PLATSAK, in Escherichia coli. Thromb. Res. 1997;88:419–426. doi: 10.1016/S0049-3848(97)00277-6. [DOI] [PubMed] [Google Scholar]
- 181.Icke C., Schlott B., Ohlenschläger O., Hartmann M., Gührs K.-H., Glusa E. Fusion Proteins with Anticoagulant and Fibrinolytic Properties: Functional Studies and Structural Considerations. Mol. Pharmacol. 2002;62:203–209. doi: 10.1124/mol.62.2.203. [DOI] [PubMed] [Google Scholar]
- 182.Kotra S.R., Peravali J.B., Yanamadala S., Kumar A., Rao K.R.S.S.S., Pulicherla K.K. Large scale production of soluble re-combinant staphylokinase variant from cold shock expression system using IPTG inducible E. coli BL21(DE3) Int. J. Bio-Sci. Bio-Technol. 2013;5:107–116. [Google Scholar]
- 183.Sumi H., Hamada H., Nakanishi K., Hiratani H. Enhancement of the Fibrinolytic Activity in Plasma by Oral Administration of Nattokinases. Acta Haematol. 1990;84:139–143. doi: 10.1159/000205051. [DOI] [PubMed] [Google Scholar]
- 184.Park K.-J., Kang J.-I., Kim T.-S., Yeo I.-H. The Antithrombotic and Fibrinolytic Effect of Natto in Hypercholesterolemia Rats. Prev. Nutr. Food Sci. 2012;17:78–82. doi: 10.3746/pnf.2012.17.1.078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Fujita M., Hong K., Ito Y., Misawa S., Takeuchi N., Kariya K., Nishimuro S. Transport of Nattokinase across the Rat Intestinal Tract. Biol. Pharm. Bull. 1995;18:1194–1196. doi: 10.1248/bpb.18.1194. [DOI] [PubMed] [Google Scholar]
- 186.Urano T., Ihara H., Umemura K., Suzuki Y., Oike M., Akita S., Tsukamoto Y., Suzuki I., Takada A. The Profibrinolytic Enzyme Subtilisin NAT Purified from Bacillus subtilis Cleaves and Inactivates Plasminogen Activator Inhibitor Type 1. J. Biol. Chem. 2001;276:24690–24696. doi: 10.1074/jbc.M101751200. [DOI] [PubMed] [Google Scholar]
- 187.Fujita M., Ito Y., Hong K., Nishimuro S. Characterization of nattokinase-degraded products from human fibrinogen or cross-linked fibrin. Fibrinolysis. 1995;9:157–164. doi: 10.1016/S0268-9499(95)80005-0. [DOI] [Google Scholar]
- 188.Fujita M., Hong K., Ito Y., Fujii R., Kariya K., Nishimuro S. Thrombolytic Effect of Nattokinase on a Chemically Induced Thrombosis Model in Rat. Biol. Pharm. Bull. 1995;18:1387–1391. doi: 10.1248/bpb.18.1387. [DOI] [PubMed] [Google Scholar]
- 189.Hsia C.-H., Shen M.-C., Lin J.-S., Wen Y.-K., Hwang K.-L., Cham T.-M., Yang N.-C. Nattokinase decreases plasma levels of fibrinogen, factor VII, and factor VIII in human subjects. Nutr. Res. 2009;29:190–196. doi: 10.1016/j.nutres.2009.01.009. [DOI] [PubMed] [Google Scholar]
- 190.Kurosawa Y., Nirengi S., Homma T., Esaki K., Ohta M., Clark J.F., Hamaoka T. A single-dose of oral nattokinase potentiates thrombolysis and anti-coagulation profiles. Sci. Rep. 2015;5:11601. doi: 10.1038/srep11601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Jensen G.S., Lenninger M., Ero M.P., Benson K.F. Consumption of nattokinase is associated with reduced blood pressure and von Willebrand factor, a cardiovascular risk marker: Results from a randomized, double-blind, placebo-controlled, multicenter North American clinical trial. Integr. Blood Press. Control. 2016;9:95–104. doi: 10.2147/IBPC.S99553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Weng Y., Yao J., Sparks S., Wang K.Y. Nattokinase: An Oral Antithrombotic Agent for the Prevention of Cardiovascular Disease. Int. J. Mol. Sci. 2017;18:523. doi: 10.3390/ijms18030523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Domsalla A., Görick C., Melzig M.F. Proteolytic activity in latex of the genus Euphorbia—A chemotaxonomic marker? Pharmazie. 2010;65:227–230. doi: 10.1691/ph.2010.9709. [DOI] [PubMed] [Google Scholar]
- 194.Kohnert U., Rudolph R., Verheijen J.H., Jacoline E., Weening-Verhoeff D., Stern A., Opitz U., Martin U., Lill H., Prinz H., et al. Biochemical properties of the kringle 2 and protease domains are maintained in the refolded t-PA deletion variant BM 06.022. Protein Eng. Des. Sel. 1992;5:93–100. doi: 10.1093/protein/5.1.93. [DOI] [PubMed] [Google Scholar]
- 195.Martin U., Von Möllendorff E., Akpan W., Kientsch-Engel R., Kaufmann B., Neugebauer G. Pharmacokinetic and Hemostatic Properties of the Recombinant Plasminogen Activator BM 06.022 in Healthy Volunteers. Thromb. Haemost. 1991;66:569–574. doi: 10.1055/s-0038-1646461. [DOI] [PubMed] [Google Scholar]
- 196.Flemmig M., Melzig M.F. Serine-proteases as plasminogen activators in terms of fibrinolysis. J. Pharm. Pharmacol. 2012;64:1025–1039. doi: 10.1111/j.2042-7158.2012.01457.x. [DOI] [PubMed] [Google Scholar]
- 197.Martin U., Von Möllendorff E., Akpan W., Kientsch-Engel R., Kaufmann B., Neugebauer G. Dose-ranging study of the novel recombinant plasminogen activator BM 06.022 in healthy volunteers. Clin. Pharmacol. Ther. 1991;50:429–436. doi: 10.1038/clpt.1991.160. [DOI] [PubMed] [Google Scholar]
- 198.Smalling R.W., Bode C., Kalbfleisch J., Sen S., Limbourg P., Forycki F., Habib G., Feldman R., Hohnloser S., Seals A. More Rapid, Complete, and Stable Coronary Thrombolysis With Bolus Administration of Reteplase Compared With Alteplase Infusion in Acute Myocardial Infarction. Circulation. 1995;91:2725–2732. doi: 10.1161/01.CIR.91.11.2725. [DOI] [PubMed] [Google Scholar]
- 199.Bode C., Smalling R.W., Berg G., Burnett C., Lorch G., Kalbfleisch J.M., Chernoff R., Christie L.G., Feldman R.L., Seals A.A., et al. Randomized Comparison of Coronary Thrombolysis Achieved With Double-Bolus Reteplase (Recombinant Plasminogen Activator) and Front-Loaded, Accelerated Alteplase (Recombinant Tissue Plasminogen Activator) in Patients With Acute Myocardial Infarction. Circulation. 1996;94:891–898. doi: 10.1161/01.CIR.94.5.891. [DOI] [PubMed] [Google Scholar]
- 200.GUST-III Investigators A comparison of reteplase with alteplase for acute myocardial infarction. The Global Use of Strategies to Open Occluded Coronary Arteries (GUSTO III) Investigators. N. Engl. J. Med. 1997;337:1118–1123. doi: 10.1056/NEJM199710163371603. [DOI] [PubMed] [Google Scholar]
- 201.Biji G.D., Arun A., Muthulakshmi E., Vijayaraghavan P., Arasu M.V., Al-Dhabi N.A. Bio-prospecting of cuttle fish waste and cow dung for the production of fibrinolytic enzyme from Bacillus cereus IND5 in solid state fermentation. 3 Biotech. 2016;6:231. doi: 10.1007/s13205-016-0553-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Ahn M.Y., Hahn B.S., Ryu K.S., Kim J.W., Kim I., Kim Y.S. Purification and characterization of a serine protease with fibri nolytic activity from the dung beetles, Catharsius molossus. Thromb. Res. 2003;112:339–347. doi: 10.1016/j.thromres.2004.01.005. [DOI] [PubMed] [Google Scholar]
- 203.Sumi H., Nakajima N., Mihara H. Fibrinolysis relating substances in marine creatures. Comp. Biochem. Physiol. Part B Comp. Biochem. 1992;102:163–167. doi: 10.1016/0305-0491(92)90290-8. [DOI] [PubMed] [Google Scholar]
- 204.Sugimoto S., Fujii T., Morimiya T., Johdo O., Nakamura T. The Fibrinolytic Activity of a Novel Protease Derived from a Tempeh Producing Fungus, Fusarium sp. BLB. Biosci. Biotechnol. Biochem. 2007;71:2184–2189. doi: 10.1271/bbb.70153. [DOI] [PubMed] [Google Scholar]
- 205.Salihu A., Sallau A.B., Adamu A., Kudu F.A., Tajo M.M., Bala T.F., Yashim W.D. Utilization of Groundnut Husk as a Solid Substrate for Cellulase Production by Aspergillus niger Using Response Surface Methodology. Waste Biomass-Valorization. 2013;5:585–593. doi: 10.1007/s12649-013-9268-1. [DOI] [Google Scholar]
- 206.Prakasham R.S., Rao C.S., Sarma P.N. Green gram husk—an inexpensive substrate for alkaline protease production by Bacillus sp. in solid-state fermentation. Bioresour Technol. 2006;97:1449–1454. doi: 10.1016/j.biortech.2005.07.015. [DOI] [PubMed] [Google Scholar]
- 207.Dilipkumar M., Rajasimman M., Rajamohan N. Enhanced inulinase production by Streptomyces sp. in solid state fermenta-tion through statistical designs. 3 Biotech. 2013;3:509–515. doi: 10.1007/s13205-012-0112-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Raol G.G., Raol B.V., Prajapati V.S., Bhavsar N.H. Utilization of agro-industrial waste for β-galactosidase production under solid state fermentation using halotolerant Aspergillus tubingensis GR1 isolate. 3 Biotech. 2015;5:411–421. doi: 10.1007/s13205-014-0236-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Chang C.T., Fan M.H., Kuo F.C., Sung H.Y. Potent fibrinolytic enzyme from a mutant of Bacillus subtilis IMR-NK1. J. Agric. Food Chem. 2000;48:3210–3216. doi: 10.1021/jf000020k. [DOI] [PubMed] [Google Scholar]
- 210.Yao Z., Kim J.H. Gene Cloning, Expression, and Properties of a Fibrinolytic Enzyme Secreted by Bacillus pumilus BS15 Isolated from Gul (Oyster) Jeotgal. Biotechnol. Bioprocess Eng. 2018;23:293–301. doi: 10.1007/s12257-018-0029-7. [DOI] [Google Scholar]
- 211.Wu R., Chen G., Pan S., Zeng J., Liang Z. Cost-effective fibrinolytic enzyme production by Bacillus subtilis WR350 using medium supplemented with corn steep powder and sucrose. Sci. Rep. 2019;9:6824. doi: 10.1038/s41598-019-43371-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Ghaly A., Vegneshwaran vasudevan R., Brooks M., Budge S., Dave D. Fish Processing Wastes as a Potential Source of Proteins, Amino Acids and Oils: A Critical Review. J. Microb. Biochem. Technol. 2013;2:107–129. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Study did not report any data.