Abstract
Inflammatory bowel disease (IBD) is a multisystemic disease with a wide range of extraintestinal manifestations in both ulcerative colitis and Crohn’s disease, while increasing evidence supports the interaction between gut and central nervous system, described as “gut-brain axis”. According to epidemiological studies, it seems that patients with IBD present more frequently with impaired mental status compared to the general population, leading to diagnostic and management problems in this group of patients. The association between IBD and mental disorders, such as dementia and autism spectrum disorders, has not been fully clarified; genetic factors and the gut-brain axis seem to be involved. The purpose of this review is to present and analyze the epidemiological data about this issue, describe the possible pathogenetic mechanisms and discuss some considerations about the management of patients with IBD and impaired mental status.
Keywords: Inflammatory bowel disease, Autism, Dementia, Psychiatric diseases
Introduction
Inflammatory bowel disease (IBD) is a chronic idiopathic unknown etiology disease of gastrointestinal tract, comprising two main forms: Crohn’s disease (CD) and ulcerative colitis (UC). Both disorders may also affect other organ systems as well [1]. IBD has been associated with various neurological manifestations, such as cerebrovascular disease, demyelinating central nervous system disorder and peripheral neuropathy, indicating a gut-brain interaction in IBD [2]. In recent decade, many studies have documented a possible association between IBD and mental disorders, such as dementia, autism spectrum disorders (ASDs) and psychiatric diseases [3]. Diagnostic and Statistical Manual of Mental Disorders, 5th edition, characterizes as mental disorder, a clinical significant disturbance in an individual’s cognition, emotion regulation or behavior that reflects a dysfunction in the psychological, biological or developmental processes underlying mental functioning, resulting usually in significant distress or disability in social, occupational or other important activities [4]. It is worth noting that the estimated prevalence of mental disorders among adults in the United States is approximately 20% [5], while the worldwide prevalence of autism spectrum disorders is about 1% [6].
In this narrative review, we intend to analyze the available literature regarding IBD in patients with mental disorders and discuss issues of epidemiology, pathophysiology and management in order to raise awareness and present the clinical aspects about this subgroup of IBD patients.
Epidemiology
In patients with IBD, several studies have described an increased frequency of various mental disorders, ranging from mild depression to severe form of schizophrenia and serious dementia, while the prevalence of mental disorders depends on the study (Table 1) [7-21].
Table 1. Mental Disorders Associated With Inflammatory Bowel Disease.
Psychiatric diseases
Many studies have described that IBD patients often suffer from anxiety and depression that may influence the course of disease through the gut-brain axis [22]. A recent systematic review demonstrated that the prevalence of anxiety and depression is higher in patients with IBD (anxiety 19.1% versus 9.6% and depression 21.2% versus 13.4%), while the rates seem to be higher when the disease is active [7]. Risk factors of depression among patients with IBD seem to be age, severe disease, flares, disable or unemployed status, socioeconomic deprivation [23] and coronavirus disease 2019 (COVID-19) lockdown [24]. Furthermore, several studies have demonstrated that the rate of suicidal ideation seems to be increased in IBD patients and is approximately 6% [25].
Regarding the association between bipolar disorder and IBD, the data are conflicting. A population-based IBD cohort study found lower rates of bipolar disorder in IBD patients than in control subjects in the United States and New Zeeland [26]. However, a population-based cross-sectional study from Taiwan demonstrated that patients with IBD were more likely to have bipolar disorder (adjusted odds ratio (OR): 2.1; 95% confidence interval (CI): 1.30 - 3.38) [8], while a national cohort study from Denmark found higher risk of bipolar disorder only in patients with CD [9]. In addition, a Canadian cohort study demonstrated a higher incidence rate ratio of schizophrenia in IBD patients compared to matched-control individuals to age and sex, but this was not statistically significant (1.64; 95% CI: 0.95 - 2.84) [10]. Also, a general population-based study from United Kingdom demonstrated no evidence of an increased likelihood of schizophrenia in patients with CD (OR: 0.74; 95% CI: 0.44 - 1.3) and UC (OR: 0.71; 95% CI: 0.44 - 1.1) in comparison with the general population [27].
Some case reports and case series have suggested a possible association between eating disorders, such as bulimia nervosa and anorexia, and IBD; however, the evidence is scarce and most studies are required in order to support this causal association [28].
ASDs
According to the Diagnostic and Statistical Manual of Mental Disorders, 5th edition, ASDs are characterized by deficits in social communications and the presence of restricted interests and repetitive behaviors, including four separate disorders: autistic disorder, Asperger’s syndrome, childhood disintegrative disorder and the catch-all diagnosis of pervasive developmental disorder not otherwise specified [29]. Many studies have demonstrated that children with ASD suffer frequently from gastrointestinal symptoms, such as diarrhea, constipation, abdominal pain or gaseousness, mainly children with ASD and increased problem behaviors, such as social withdrawal, sleep problems and self-injuries [30, 31]. According to a meta-analysis, children with ASD have three times higher risk of gastrointestinal symptoms compared to children without ASD [32].
Recently, four case-control study populations have documented higher prevalence of IBD among patients with ASD than that of their respective controls [11]. Furthermore, a population-based study of more than 48,000 children with ASD demonstrated a higher prevalence of CD and UC in children with ASD compared to controls (0.18% vs. 0.11%). Also, this study demonstrated that children with ASD and IBD received more frequently second-tier biological agents, such as adalimumab and certolizumab, compared to pediatric IBD population without ASD. These findings may have two possible explanations. Children with ASD and IBD may suffer from more severe IBD or IBD refractory to first-line biological agents, such as infliximab. The second explanation is that children with ASD due to their behavior present less compliance and adherence to oral and intravenous treatment, making subcutaneous treatment more feasible [12]. In addition, a retrospective study documented an increased prevalence of IBD in hospitalized patients with ASD compared to the overall hospital population in four hospitals in Boston (0.83% vs. 0.54%) [13].
Dementia
Dementia is a heterogeneous syndrome that may be caused by many different neurological diseases. The major cause of dementia is neurodegenerative disorders, such as Alzheimer disease, Lewy bodies dementia and Parkinson’s disease, while the estimated global prevalence is approximately 7% of individuals 65 years or older [33]. A recent population-based cohort study demonstrated an overall higher incidence of dementia among patients with IBD (5.5% vs. 1.4% among controls). In addition, dementia was diagnosed at an earlier age among patients with IBD (76.24 years vs. 83.45 years) and the risk ratio of developing dementia among patients with IBD was 2.54 (95% CI: 1.91 - 3.37). Furthermore, the risk for dementia was associated with the chronicity of IBD diagnosis and dementia risk seems to accelerate over time correlating with the chronicity of IBD diagnosis, while no significant differences in the risk for dementia were demonstrated between patients with UC and CD [14].
Concerning Parkinson disease, dementia occurs in 30% of patients with Parkinson’s disease [15], while several epidemiological studies have demonstrated an association between Parkinson’s disease and IBD [34]. A recent meta-analysis found an overall increased risk of Parkinson’s disease development compared to control individuals (risk ratio (RR): 1.41; 95% CI: 1.19 - 1.66). Patients with CD had a 28% increased risk of Parkinson’s disease (RR: 1.28; 95% CI: 1.08 - 1.52) and UC patients had a 30% increased risk of Parkinson’s disease (RR: 1.30; 95% CI: 1.15 - 1.47) [16]. In addition, many studies have demonstrated that IBD treatment seems to decrease the subsequent risk of Parkinson’s disease development [35]. Instead, patients with Parkinson’s disease seem to have no higher risk for either CD or UC [36].
Genetic syndromes with impaired intellectual ability
Several case reports have described IBD development in patients with genetic syndromes that cause intellectual disability, such as Down syndrome [17], Turner syndrome [18] and Williams syndrome [19]; however, data are very limited and no association has been documented.
Pathogenesis
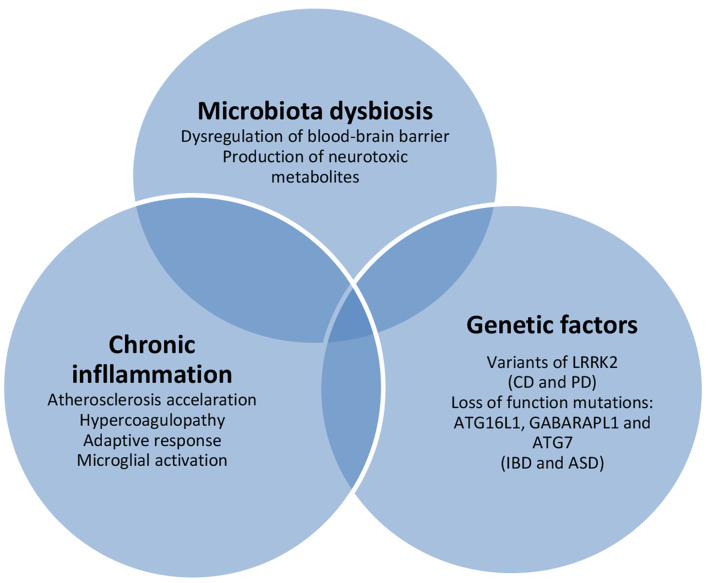
Although the pathogenetic mechanisms between IBD and mental disorders have not been fully elucidated, genetic factors and the gut-brain interaction seem to contribute to the elevated prevalence of mental disorders in patients with IBD (Fig. 1).
Figure 1.
Inflammatory bowel disease-associated pathogenetic mechanisms of mental disorders. CD: Crohn’s disease; PD: Parkinson’s disease; IBD: inflammatory bowel disease; ASD: autism spectrum disorder.
Psychiatric diseases
Several inflammatory diseases, including IBD, have been associated with anxiety and depression development. Clinical studies have demonstrated that cytokine release may affect neurobehavioral functions and cause negative effects on emotional and memory effects [37, 38]. Also, animal models have shown that inflammation induction may cause hypothalamic-pituitary-adrenal axis dysregulation and changes in blood-brain barrier integrity. The disruption of blood-brain barrier leads to increased inflammatory mediators in central neural system (CNS), affecting the neurotransmitter metabolism, in brain regions associated with emotional regulation, such as the nucleus accumbens, amygdale and hippocampus [39]. Furthermore, some authors have suggested that depression in inflammatory conditions is a form or a consequence of sickness behavior and it is an adaptive response that enhances recovery by conserving energy to combat acute inflammation [40].
On the other hand, it should be mentioned that psychological factors may affect the course of IBD and several studies have described the role of psychological factors on the course of IBD, demonstrating that psychological stress may cause disease relapse or exacerbation [41]. Stress may induce changes in gastrointestinal function, such as increased intestinal motility and permeability, and activation mucosal mast cells, from which the course of IBD may be influenced [42, 43].
ASDs
Genetic and immunological factors are included to the possible explanations of the epidemiological association between IBD and ASD. A study demonstrated that two losses of function mutations previously implicated in IBD on the autophagy pathway (ATG16L1 and GABARAPL1) may play a role in ASD development, while one ASD-associated loss of function mutation (ATG7) may contribute to IBD [44]. Furthermore, identification of an overlap gene expression in gastrointestinal tissue in pediatric patients with IBD and in children with ASD has been described [45]. In addition, gut microbiome may participate in association between IBD and ASD, as individuals with ASD seem to have an altered gut microbiota [46] and microbiota dysbiosis may contribute to IBD development [47]. Further studies are required due to the lack of data about this topic.
Dementia
The pathogenic mechanism of the increased risk of dementia in patients with IBD may be multifactorial and involves chronic inflammation, atherosclerosis acceleration and hypercoagulopathy. In particular, Alzheimer’s disease is the most frequent cause of dementia and the major pathogenetic mechanisms include the development of amyloid β plaques and neurofibrillary tangles [48]. Several studies have suggested that systemic chronic inflammation may accelerate the progression of Alzheimer’s disease via alterations of the blood-brain barrier and the microglial activation [49], indicating that IBD as a chronic inflammatory condition may contribute to Alzheimer’s disease progression.
Regarding Parkinson’s disease, mutations of leucine-rich repeat kinase 2 (LRRK-2) seem to cause disrupted autophagy and participate in Parkinson’s disease development [50], as a recent study identified shared genetic variants of LRRK-2 between Parkinson’s disease and CD [51]. Furthermore, gut-brain axis may contribute to Parkinson’s disease development in patients with IBD, as animal studies have suggested that alterations of gut microbiota may trigger Parkinson’s disease [52], while a recent study demonstrated that gastrointestinal infections are associated with a higher risk of Parkinson’s disease [53].
In addition, IBD has been associated with a higher risk of thromboembolic events and stroke development due to thrombocytosis, coagulation abnormalities, hyperlipidemia and hyperhomocysteinemia [20], thus contributing to the development of vascular dementia. It is worth mentioning that vascular dementia seems to cause approximately 15% of dementia cases [21].
Furthermore, dysbiosis of the gut microbiome seems to be present in patients with IBD [54] and many studies have suggested that dysregulation of the gut microbiota may play a role in the progression of dementia through alterations of the gut barrier, production of microbial neurotoxic metabolites in systemic circulation and increased inflammation [55, 56].
Management
Treatment and management of patients with IBD and mental illness poses many serious problems and is often a challenge due to difficulties in compliance, communication problems and non-cooperation in the physical examination of this group of patients (Table 2). We suggest that collaboration between gastroenterologists, psychiatrists and patient’s family members may be beneficial and helpful in treatment compliance and monitoring of these patients. For the aforementioned reasons, diagnosis of IBD in patients with mental disorders may be difficult, while endoscopy may be a complex process or even not feasible, especially in patients with severe dementia or major psychiatric disorders. Deep sedation may be mandatory during endoscopy, while adequated bowel preparation for colonoscopy remains a significant issue. In addition, the choice of the appropriate treatment should take into account several aspects. The use of intravenous biological agents requires inpatient treatment, while azathioprine use requires close monitoring due to frequent adverse events, mainly within the first weeks. Furthermore, we would recommend avoiding the frequent use of corticosteroids in patients with psychiatric disorders. Many studies have demonstrated corticosteroid-induced psychiatric side effects, including disturbances of cognition, mood, sleep and behavior through implication of the hypothalamic-pituitary-adrenal axis. The estimated prevalence of psychiatric side effects ranges from 1% to 10%, while dosage and duration of corticosteroids administration are directly related to the frequency of adverse effects [57, 58]. In a recent prospective study, the rate of corticosteroid-induced mood changes in IBD patients after 2 weeks of prednisone 40 mg daily was 49.1% [59]. In the last decade, second generation of topical active corticosteroids, such as budesonide and beclomethasone, are often used in UC and CD treatment and have been associated with high efficacy, presenting fewer adverse events through their low systemic bioavailability and approximately 90% first-pass metabolism in liver [60]. On the other hand, according to available data, treatment with biological agents does not seem to be associated with an increased risk of anxiety, depression, suicidality and psychosis [61]. Thus, the use of subcutaneous biological agents, such as adalimumab, golimumab and ustekinumab, in patients with mental illness may be a safe, effective and feasible treatment.
Table 2. Management of Patients With Inflammatory Bowel Disease and Mental Disorder.
| Collaboration between gastroenterologists, neurologists, psychiatrists and patient’s family members |
| Deep sedation during endoscopy |
| Subcutaneous biological agents might be preferred due to better compliance |
| Avoid corticosteroid treatment |
| Second-generation corticosteroids could be preferred |
| Physical activity |
| Avoid alcohol and narcotics use |
Regarding the impact of thiopurine on clinical course of mental disorders in IBD patients, there are conflicting data. Thiopurines seem to inhibit the Ras-related C3 botulinum substrate (Rac 1), which has been implicated in Alzheimer’s disease development via increased amyloid precursor protein [62]. A recent national cohort study from United States described that IBD patients receiving thiopurine have a lower risk of Alzheimer’s disease compared with IBD patients without thiopurine exposed, demonstrating an adjusted hazard ratio (HR) per additional year of thiopurine exposure 0.917 (95% CI: 0.851 - 0.989) for comorbidities and medications [63]. On the other hand, a retrospective cohort study demonstrated that IBD patients exposed to thiopurine less than 1 year had a 67% higher risk of depression (HR: 1.667; 95% CI: 1.501 - 1.805) compared to IBD patients without thiopurine exposure [64]. However, prospective studies and further researches into the effect of thiopurines in IBD patients on neuro-psychiatric disorders are required.
As mentioned above, patients with IBD appear to have a higher risk of dementia, which is prevalent in the elderly. It is estimated that 10-30% of patients with IBD are > 60 years old. Elderly patients may have less complicated clinical course; however, they have higher or similar rates of surgery [65]. In addition, elderly patients tent to have more frequent comorbidities, such as heart disease and diabetes, which may affect the management and clinical course of disease. IBD patients with diabetes may have an increased risk of flare and IBD-related hospitalization [66], while anti-TNF agents are contraindicated in patients with severe heart failure [67]. Consequently, an individualized care of IBD patients with dementia is required.
Furthermore, it is worth mentioning that increased clinicians’ awareness for the development of dementia and psychiatric disorders in patients with IBD is warranted to facilitate an early diagnosis and prompt the appropriate treatment initiation. In addition, because of high prevalence of irritable bowel syndrome (IBS) in patients with psychiatric disorders and ASDs [68], clinicians should distinguish IBD from IBS. IBS is a functional disorder, characterized by chronic abdominal pain and altered bowel habits. Patients with IBS report at least once weekly abdominal pain in association with a change in stool form, frequency and/or abdominal pain related to defecation, while IBS diagnosis requires exclusion of disorders, such as IBD and celiac disease [69]. Likewise, symptoms indicating IBD, such as bloody stools, malabsorption, chronic diarrhea and iron deficiency, in patients with mental disorders should be evaluated in detail and endoscopy should be considered.
Lifestyle modifications may have a significant role in IBD and mental disorders. Patients with IBD should be encouraged to exercise because physical activity may improve psychological well-being, reducing anxiety and depression, as well as having a preventative or mitigated role against Alzheimer’s disease [70], Parkinson’s disease [71] and ASD [72]. Also, IBD patients should avoid alcohol consumption and narcotics use that have been correlated with comorbid mental illnesses and worsening of IBD course [73].
Conclusions
The prevalence of disorders with impaired mental status, such as psychiatric diseases, ASDs and dementia seems to be higher in patients with IBD compared to the general population. The pathogenetic mechanisms may be multifactorial, including alteration of gut microbiota, genetic factors and chronic inflammation. The diagnosis of IBD and management of this group of IBD patients is often challenging due to poor compliance and a close collaboration between a team of gastroenterologists, neurologists, psychiatrists and family members of the patient is required.
Acknowledgments
None to declare.
Financial Disclosure
No funding has been received.
Conflict of Interest
All authors declare no conflict of interest.
Author Contributions
DKC and KHK conceived designed and critically revised the article for important intellectual content. FSF, GK, MS, and EA contributed to the data collection and literature research. FSF and AHK analyzed the data and contributed substantially in the manuscript preparations. All authors approved the final version of the article to be published.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
References
- 1.Vavricka SR, Schoepfer A, Scharl M, Lakatos PL, Navarini A, Rogler G. Extraintestinal manifestations of inflammatory bowel disease. Inflamm Bowel Dis. 2015;21(8):1982–1992. doi: 10.1097/MIB.0000000000000392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Moris G. Inflammatory bowel disease: an increased risk factor for neurologic complications. World J Gastroenterol. 2014;20(5):1228–1237. doi: 10.3748/wjg.v20.i5.1228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nowakowski J, Chrobak AA, Dudek D. Psychiatric illnesses in inflammatory bowel diseases - psychiatric comorbidity and biological underpinnings. Psychiatr Pol. 2016;50(6):1157–1166. doi: 10.12740/PP/62382. [DOI] [PubMed] [Google Scholar]
- 4.Telles-Correia D. Mental disorder: Are we moving away from distress and disability? J Eval Clin Pract. 2018;24(5):973–977. doi: 10.1111/jep.12871. [DOI] [PubMed] [Google Scholar]
- 5.McGinty EE, Daumit GL. Integrating mental health and addiction treatment into general medical care: the role of policy. Psychiatr Serv. 2020;71(11):1163–1169. doi: 10.1176/appi.ps.202000183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lai MC, Lombardo MV, Baron-Cohen S. Autism. Lancet. 2014;383(9920):896–910. doi: 10.1016/S0140-6736(13)61539-1. [DOI] [PubMed] [Google Scholar]
- 7.Mikocka-Walus A, Knowles SR, Keefer L, Graff L. Controversies revisited: a systematic review of the comorbidity of depression and anxiety with inflammatory bowel diseases. Inflamm Bowel Dis. 2016;22(3):752–762. doi: 10.1097/MIB.0000000000000620. [DOI] [PubMed] [Google Scholar]
- 8.Kao LT, Lin HC, Lee HC. Inflammatory bowel disease and bipolar disorder: A population-based cross-sectional study. J Affect Disord. 2019;247:120–124. doi: 10.1016/j.jad.2019.01.014. [DOI] [PubMed] [Google Scholar]
- 9.Eaton WW, Pedersen MG, Nielsen PR, Mortensen PB. Autoimmune diseases, bipolar disorder, and non-affective psychosis. Bipolar Disord. 2010;12(6):638–646. doi: 10.1111/j.1399-5618.2010.00853.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bernstein CN, Hitchon CA, Walld R, Bolton JM, Sareen J, Walker JR, Graff LA. et al. Increased burden of psychiatric disorders in inflammatory bowel disease. Inflamm Bowel Dis. 2019;25(2):360–368. doi: 10.1093/ibd/izy235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Doshi-Velez F, Avillach P, Palmer N, Bousvaros A, Ge Y, Fox K, Steinberg G. et al. Prevalence of inflammatory bowel disease among patients with autism spectrum disorders. Inflamm Bowel Dis. 2015;21(10):2281–2288. doi: 10.1097/MIB.0000000000000502. [DOI] [PubMed] [Google Scholar]
- 12.Lee M, Krishnamurthy J, Susi A, Sullivan C, Gorman GH, Hisle-Gorman E, Erdie-Lalena CR. et al. Association of autism spectrum disorders and inflammatory bowel disease. J Autism Dev Disord. 2018;48(5):1523–1529. doi: 10.1007/s10803-017-3409-5. [DOI] [PubMed] [Google Scholar]
- 13.Kohane IS, McMurry A, Weber G, MacFadden D, Rappaport L, Kunkel L, Bickel J. et al. The co-morbidity burden of children and young adults with autism spectrum disorders. PLoS One. 2012;7(4):e33224. doi: 10.1371/journal.pone.0033224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang B, Wang HE, Bai YM, Tsai SJ, Su TP, Chen TJ, Wang YP. et al. Inflammatory bowel disease is associated with higher dementia risk: a nationwide longitudinal study. Gut. 2021;70(1):85–91. doi: 10.1136/gutjnl-2020-320789. [DOI] [PubMed] [Google Scholar]
- 15.Hanagasi HA, Tufekcioglu Z, Emre M. Dementia in Parkinson's disease. J Neurol Sci. 2017;374:26–31. doi: 10.1016/j.jns.2017.01.012. [DOI] [PubMed] [Google Scholar]
- 16.Zhu F, Li C, Gong J, Zhu W, Gu L, Li N. The risk of Parkinson's disease in inflammatory bowel disease: A systematic review and meta-analysis. Dig Liver Dis. 2019;51(1):38–42. doi: 10.1016/j.dld.2018.09.017. [DOI] [PubMed] [Google Scholar]
- 17.Souto-Rodriguez R, Barreiro-de-Acosta M, Dominguez-Munoz JE. Down's syndrome and inflammatory bowel disease: is there a real link? Rev Esp Enferm Dig. 2014;106(3):220–222. [PubMed] [Google Scholar]
- 18.Hyodo H, Tomita Y, Hirai K, Hirakawa H, Ueno S, Ishiguro H. Turner syndrome with ulcerative colitis. Clin Pediatr Endocrinol. 2009;18(4):101–105. doi: 10.1297/cpe.18.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gilbert-Barness E, Fox T, Morrow G, Luquette M, Pomerance HH. Williams syndrome associated with Crohn disease, multiple infections, and chronic granulomatous disease. Fetal Pediatr Pathol. 2004;23(1):29–37. doi: 10.1080/15227950490423016. [DOI] [PubMed] [Google Scholar]
- 20.Katsanos AH, Kosmidou M, Giannopoulos S, Katsanos KH, Tsivgoulis G, Kyritsis AP, Tsianos EV. Cerebral arterial infarction in inflammatory bowel diseases. Eur J Intern Med. 2014;25(1):37–44. doi: 10.1016/j.ejim.2013.08.702. [DOI] [PubMed] [Google Scholar]
- 21.O'Brien JT, Thomas A. Vascular dementia. Lancet. 2015;386(10004):1698–1706. doi: 10.1016/S0140-6736(15)00463-8. [DOI] [PubMed] [Google Scholar]
- 22.Sajadinejad MS, Asgari K, Molavi H, Kalantari M, Adibi P. Psychological issues in inflammatory bowel disease: an overview. Gastroenterol Res Pract. 2012;2012:106502. doi: 10.1155/2012/106502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nahon S, Lahmek P, Durance C, Olympie A, Lesgourgues B, Colombel JF, Gendre JP. Risk factors of anxiety and depression in inflammatory bowel disease. Inflamm Bowel Dis. 2012;18(11):2086–2091. doi: 10.1002/ibd.22888. [DOI] [PubMed] [Google Scholar]
- 24.Sempere L, Bernabeu P, Cameo J, Gutierrez A, Laveda R, Garcia MF, Aguas M. et al. Evolution of the emotional impact in patients with early inflammatory bowel disease during and after Covid-19 lockdown. Gastroenterol Hepatol. 2021 doi: 10.1016/j.gastrohep.2021.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Santa A, Szanto KJ, Sarlos P, Miheller P, Farkas K, Molnar T. Letter: suicide risk among adult inflammatory bowel disease patients. Aliment Pharmacol Ther. 2020;51(11):1213–1214. doi: 10.1111/apt.15708. [DOI] [PubMed] [Google Scholar]
- 26.Walker JR, Ediger JP, Graff LA, Greenfeld JM, Clara I, Lix L, Rawsthorne P. et al. The Manitoba IBD cohort study: a population-based study of the prevalence of lifetime and 12-month anxiety and mood disorders. Am J Gastroenterol. 2008;103(8):1989–1997. doi: 10.1111/j.1572-0241.2008.01980.x. [DOI] [PubMed] [Google Scholar]
- 27.West J, Logan RF, Hubbard RB, Card TR. Risk of schizophrenia in people with coeliac disease, ulcerative colitis and Crohn's disease: a general population-based study. Aliment Pharmacol Ther. 2006;23(1):71–74. doi: 10.1111/j.1365-2036.2006.02720.x. [DOI] [PubMed] [Google Scholar]
- 28.Ilzarbe L, Fabrega M, Quintero R, Bastidas A, Pintor L, Garcia-Campayo J, Gomollon F. et al. Inflammatory bowel disease and eating disorders: a systematized review of comorbidity. J Psychosom Res. 2017;102:47–53. doi: 10.1016/j.jpsychores.2017.09.006. [DOI] [PubMed] [Google Scholar]
- 29.Hodges H, Fealko C, Soares N. Autism spectrum disorder: definition, epidemiology, causes, and clinical evaluation. Transl Pediatr. 2020;9(Suppl 1):S55–S65. doi: 10.21037/tp.2019.09.09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chaidez V, Hansen RL, Hertz-Picciotto I. Gastrointestinal problems in children with autism, developmental delays or typical development. J Autism Dev Disord. 2014;44(5):1117–1127. doi: 10.1007/s10803-013-1973-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Restrepo B, Angkustsiri K, Taylor SL, Rogers SJ, Cabral J, Heath B, Hechtman A. et al. Developmental-behavioral profiles in children with autism spectrum disorder and co-occurring gastrointestinal symptoms. Autism Res. 2020;13(10):1778–1789. doi: 10.1002/aur.2354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.McElhanon BO, McCracken C, Karpen S, Sharp WG. Gastrointestinal symptoms in autism spectrum disorder: a meta-analysis. Pediatrics. 2014;133(5):872–883. doi: 10.1542/peds.2013-3995. [DOI] [PubMed] [Google Scholar]
- 33.Gale SA, Acar D, Daffner KR. Dementia. Am J Med. 2018;131(10):1161–1169. doi: 10.1016/j.amjmed.2018.01.022. [DOI] [PubMed] [Google Scholar]
- 34.Brudek T. Inflammatory bowel diseases and Parkinson's disease. J Parkinsons Dis. 2019;9(s2):S331–S344. doi: 10.3233/JPD-191729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lee HS, Lobbestael E, Vermeire S, Sabino J, Cleynen I. Inflammatory bowel disease and Parkinson's disease: common pathophysiological links. Gut. 2021;70(2):408–417. doi: 10.1136/gutjnl-2020-322429. [DOI] [PubMed] [Google Scholar]
- 36.Camacho-Soto A, Gross A, Searles Nielsen S, Dey N, Racette BA. Inflammatory bowel disease and risk of Parkinson's disease in Medicare beneficiaries. Parkinsonism Relat Disord. 2018;50:23–28. doi: 10.1016/j.parkreldis.2018.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Reichenberg A, Yirmiya R, Schuld A, Kraus T, Haack M, Morag A, Pollmacher T. Cytokine-associated emotional and cognitive disturbances in humans. Arch Gen Psychiatry. 2001;58(5):445–452. doi: 10.1001/archpsyc.58.5.445. [DOI] [PubMed] [Google Scholar]
- 38.Grigoleit JS, Kullmann JS, Wolf OT, Hammes F, Wegner A, Jablonowski S, Engler H. et al. Dose-dependent effects of endotoxin on neurobehavioral functions in humans. PLoS One. 2011;6(12):e28330. doi: 10.1371/journal.pone.0028330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Abautret-Daly A, Dempsey E, Parra-Blanco A, Medina C, Harkin A. Gut-brain actions underlying comorbid anxiety and depression associated with inflammatory bowel disease. Acta Neuropsychiatr. 2018;30(5):275–296. doi: 10.1017/neu.2017.3. [DOI] [PubMed] [Google Scholar]
- 40.Maes M, Berk M, Goehler L, Song C, Anderson G, Galecki P, Leonard B. Depression and sickness behavior are Janus-faced responses to shared inflammatory pathways. BMC Med. 2012;10:66. doi: 10.1186/1741-7015-10-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Maunder RG, Levenstein S. The role of stress in the development and clinical course of inflammatory bowel disease: epidemiological evidence. Curr Mol Med. 2008;8(4):247–252. doi: 10.2174/156652408784533832. [DOI] [PubMed] [Google Scholar]
- 42.Hollander D. Inflammatory bowel diseases and brain-gut axis. J Physiol Pharmacol. 2003;54(Suppl 4):183–190. [PubMed] [Google Scholar]
- 43.Theoharides TC, Cochrane DE. Critical role of mast cells in inflammatory diseases and the effect of acute stress. J Neuroimmunol. 2004;146(1-2):1–12. doi: 10.1016/j.jneuroim.2003.10.041. [DOI] [PubMed] [Google Scholar]
- 44.Somekh J, Peleg M, Eran A, Koren I, Feiglin A, Demishtein A, Shiloh R. et al. A model-driven methodology for exploring complex disease comorbidities applied to autism spectrum disorder and inflammatory bowel disease. J Biomed Inform. 2016;63:366–378. doi: 10.1016/j.jbi.2016.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Walker SJ, Fortunato J, Gonzalez LG, Krigsman A. Identification of unique gene expression profile in children with regressive autism spectrum disorder (ASD) and ileocolitis. PLoS One. 2013;8(3):e58058. doi: 10.1371/journal.pone.0058058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Vuong HE, Hsiao EY. Emerging roles for the gut microbiome in autism spectrum disorder. Biol Psychiatry. 2017;81(5):411–423. doi: 10.1016/j.biopsych.2016.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Qi Y, Wu HM, Yang Z, Zhou YF, Jin L, Yang MF, Wang FY. New insights into the role of oral microbiota dysbiosis in the pathogenesis of inflammatory bowel disease. Dig Dis Sci. 2021 doi: 10.1007/s10620-021-06837-2. [DOI] [PubMed] [Google Scholar]
- 48.Tiwari S, Atluri V, Kaushik A, Yndart A, Nair M. Alzheimer's disease: pathogenesis, diagnostics, and therapeutics. Int J Nanomedicine. 2019;14:5541–5554. doi: 10.2147/IJN.S200490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Calsolaro V, Edison P. Neuroinflammation in Alzheimer's disease: Current evidence and future directions. Alzheimers Dement. 2016;12(6):719–732. doi: 10.1016/j.jalz.2016.02.010. [DOI] [PubMed] [Google Scholar]
- 50.Alessi DR, Sammler E. LRRK2 kinase in Parkinson's disease. Science. 2018;360(6384):36–37. doi: 10.1126/science.aar5683. [DOI] [PubMed] [Google Scholar]
- 51.Hui KY, Fernandez-Hernandez H, Hu J, Schaffner A, Pankratz N, Hsu NY, Chuang LS. et al. Functional variants in the LRRK2 gene confer shared effects on risk for Crohn's disease and Parkinson's disease. Sci Transl Med. 2018;10(423):eaai7795. doi: 10.1126/scitranslmed.aai7795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Matheoud D, Cannon T, Voisin A, Penttinen AM, Ramet L, Fahmy AM, Ducrot C. et al. Intestinal infection triggers Parkinson's disease-like symptoms in Pink1(-/-) mice. Nature. 2019;571(7766):565–569. doi: 10.1038/s41586-019-1405-y. [DOI] [PubMed] [Google Scholar]
- 53.Nerius M, Doblhammer G, Tamguney G. GI infections are associated with an increased risk of Parkinson's disease. Gut. 2020;69(6):1154–1156. doi: 10.1136/gutjnl-2019-318822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ni J, Wu GD, Albenberg L, Tomov VT. Gut microbiota and IBD: causation or correlation? Nat Rev Gastroenterol Hepatol. 2017;14(10):573–584. doi: 10.1038/nrgastro.2017.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Alkasir R, Li J, Li X, Jin M, Zhu B. Human gut microbiota: the links with dementia development. Protein Cell. 2017;8(2):90–102. doi: 10.1007/s13238-016-0338-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Saji N, Murotani K, Hisada T, Kunihiro T, Tsuduki T, Sugimoto T, Kimura A. et al. Relationship between dementia and gut microbiome-associated metabolites: a cross-sectional study in Japan. Sci Rep. 2020;10(1):8088. doi: 10.1038/s41598-020-65196-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Warrington TP, Bostwick JM. Psychiatric adverse effects of corticosteroids. Mayo Clin Proc. 2006;81(10):1361–1367. doi: 10.4065/81.10.1361. [DOI] [PubMed] [Google Scholar]
- 58.Kenna HA, Poon AW, de los Angeles CP, Koran LM. Psychiatric complications of treatment with corticosteroids: review with case report. Psychiatry Clin Neurosci. 2011;65(6):549–560. doi: 10.1111/j.1440-1819.2011.02260.x. [DOI] [PubMed] [Google Scholar]
- 59.Ou G, Bressler B, Galorport C, Lam E, Ko HH, Enns R, Telford J. et al. Rate of corticosteroid-induced mood changes in patients with inflammatory bowel disease: a prospective study. J Can Assoc Gastroenterol. 2018;1(3):99–106. doi: 10.1093/jcag/gwy023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Salice M, Rizzello F, Calabrese C, Privitera Hrustemovic H, Gionchetti P. Budesonide MMX: efficacy and safety profile in the treatment of ulcerative colitis. Expert Rev Gastroenterol Hepatol. 2019;13(7):607–613. doi: 10.1080/17474124.2019.1621745. [DOI] [PubMed] [Google Scholar]
- 61.Jain A, Marrie RA, Shafer LA, Graff LA, Patten SB, El-Gabalawy R, Sareen J. et al. Incidence of adverse psychiatric events during treatment of inflammatory bowel disease with biologic therapies: a systematic review. Crohns Colitis 360. 2020;2(1):otz053. doi: 10.1093/crocol/otz053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang PL, Niidome T, Akaike A, Kihara T, Sugimoto H. Rac1 inhibition negatively regulates transcriptional activity of the amyloid precursor protein gene. J Neurosci Res. 2009;87(9):2105–2114. doi: 10.1002/jnr.22039. [DOI] [PubMed] [Google Scholar]
- 63.Sutton SS, Magagnoli J, Cummings T, Hardin JW. Association between thiopurine medication exposure and Alzheimer's disease among a cohort of patients with inflammatory bowel disease. Alzheimers Dement (N Y) 2019;5:809–813. doi: 10.1016/j.trci.2019.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sutton SS, Magagnoli J, Cummings T, Hardin JW, Love BL. Association between thiopurine exposure and depression in patients with inflammatory bowel disease and rheumatoid arthritis. J Psychopharmacol. 2020;34(10):1163–1167. doi: 10.1177/0269881120908898. [DOI] [PubMed] [Google Scholar]
- 65.Ananthakrishnan AN, Shi HY, Tang W, Law CC, Sung JJ, Chan FK, Ng SC. Systematic review and meta-analysis: phenotype and clinical outcomes of older-onset inflammatory bowel disease. J Crohns Colitis. 2016;10(10):1224–1236. doi: 10.1093/ecco-jcc/jjw054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kumar A, Teslova T, Taub E, Miller JD, Lukin DJ. Comorbid diabetes in inflammatory bowel disease predicts adverse disease-related outcomes and infectious complications. Dig Dis Sci. 2021;66(6):2005–2013. doi: 10.1007/s10620-020-06439-4. [DOI] [PubMed] [Google Scholar]
- 67.Ananthakrishnan AN, Donaldson T, Lasch K, Yajnik V. Management of inflammatory bowel disease in the elderly patient: challenges and opportunities. Inflamm Bowel Dis. 2017;23(6):882–893. doi: 10.1097/MIB.0000000000001099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Fadgyas-Stanculete M, Buga AM, Popa-Wagner A, Dumitrascu DL. The relationship between irritable bowel syndrome and psychiatric disorders: from molecular changes to clinical manifestations. J Mol Psychiatry. 2014;2(1):4. doi: 10.1186/2049-9256-2-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lacy BE, Pimentel M, Brenner DM, Chey WD, Keefer LA, Long MD, Moshiree B. ACG clinical guideline: management of irritable bowel syndrome. Am J Gastroenterol. 2021;116(1):17–44. doi: 10.14309/ajg.0000000000001036. [DOI] [PubMed] [Google Scholar]
- 70.Gronek P, Balko S, Gronek J, Zajac A, Maszczyk A, Celka R, Doberska A. et al. Physical activity and Alzheimer's disease: a narrative review. Aging Dis. 2019;10(6):1282–1292. doi: 10.14336/AD.2019.0226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Fayyaz M, Jaffery SS, Anwar F, Zil-E-Ali A, Anjum I. The effect of physical activity in Parkinson's disease: a mini-review. Cureus. 2018 doi: 10.7759/cureus.2995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Huang J, Du C, Liu J, Tan G. Meta-analysis on intervention effects of physical activities on children and adolescents with autism. Int J Environ Res Public Health. 2020;17(6):1950. doi: 10.3390/ijerph17061950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mantzouranis G, Fafliora E, Saridi M, Tatsioni A, Glanztounis G, Albani E, Katsanos KH. et al. Alcohol and narcotics use in inflammatory bowel disease. Ann Gastroenterol. 2018;31(6):649–658. doi: 10.20524/aog.2018.0302. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The authors declare that data supporting the findings of this study are available within the article.