Abstract
Ischemia-reperfusion (I/R) injury often occurred in some pathologies and surgeries. I/R injury not only harmed to physiological functions of corresponding organ and tissue but also induced multiple tissue or organ dysfunctions (even these in distant locations). Although the reperfusion of blood attenuated I/R injury to a certain degree, the risk of secondary damages was difficult to be controlled and it even caused failures of these tissues and organs. Lipoic acid (LA), as an endogenous active substance and a functional agent in food, owns better safety and effects in our body (e.g., enhancing antioxidant activity, improving cognition and dementia, controlling weight, and preventing multiple sclerosis, diabetes complication, and cancer). The literature searching was conducted in PubMed, Embase, Cochrane Library, Web of Science, and SCOPUS from inception to 20 May 2021. It had showed that endogenous LA was exhausted in the process of I/R, which further aggravated I/R injury. Thus, supplements with LA timely (especially pretreatments) may be the prospective way to prevent I/R injury. Recently, studies had demonstrated that LA supplements significantly attenuated I/R injuries of many organs, though clinic investigations were short at present. Hence, it was urgent to summarize these progresses about the effects of LA on different I/R organs as well as the potential mechanisms, which would enlighten further investigations and prepare for clinic applications in the future.
1. Introduction
Ischemia refers to decreased blood perfusions or total occlusions in tissues or organs. It causes deficiencies of oxygen and other nutrients and massive accumulations of metabolic wastes. Ischemia may be caused by many reasons, such as occlusion (e.g., thrombi), trauma, organ transplantation, and atherosclerosis. Ischemia can quickly result in tissue necrosis if there are no timely perfusions [1]. Early reperfusion is the preferred intervention to prevent the pathological process. However, the reperfusion after ischemia often caused inflammations and oxidative damages via oxidative stress due to increased levels of reactive oxygen/nitrogen species [2]. Enzymes (e.g., sodium-hydrogen exchanger pumps), molecules (e.g., DNA), cell damages (even deaths), and tissue dysfunctions ensued [3]. As a result, I/R injury caused further damages of many tissues (e.g., liver, heart, lung, and kidney). Still, the detailed mechanism of I/R injury was not fully clarified despite the above potential reasons. The short treatment window period affected the effects of reperfusion therapy [4], and developing new drugs became especially important [5]. Therefore, it was urgent to develop new agents with better effects and lower side effects to prevent I/R injury. Recently, many foodborne active substances had been developed for the prevention of I/R injury (e.g., coenzyme Q10 [6], resveratrol [7], and n-3 polyunsaturated fatty acids [8]). Due to these substances (especially those also existing in the body) owning the property of low security risks, it provided a prospective research area for developing more substances to prevent I/R injury.
2. Brief of Lipoic Acid
2.1. The Source, Biosynthesis, and Metabolism of LA
LA (also called as alpha-lipoic acid, thioctic acid, 6,8-dithiooctanoic acid, etc.), as an endogenous active molecule, was firstly isolated in 1951 by Reed et al. [9]. Afterwards, LA was confirmed as a vital cofactor of several enzymes (the pyruvate dehydrogenase complex, the α-ketoglutarate dehydrogenase complex, the branched-chain α-ketoacid dehydrogenase complex, the 2-oxoadipate dehydrogenase complex, and the glycine cleavage system) necessary for aerobic metabolism in mitochondria and the synthesis of nucleic acids [10]. Its natural molecule of (R)-(+)-enantiomer is mostly present in the form of combining with lysine residues of proteins in a wide variety of foods of plants (e.g., spinach and broccoli) and animals (e.g., kidney, heart, and liver) [11]. Free LA is mainly from chemical synthesis, which produced two enantiomers (R)-(+)-LA and (S)-(-)-LA. The former is the preferred form for the purpose of nutrition and therapy [12]. Na+-dependent multivitamin transporter is responsible for LA uptake in the gastrointestinal track and its transportation into tissues from blood [13]. Then, LA plays its biological roles.
Endogenous LA is made from octanoic acid in mitochondria of eukaryotes. The octanoyl moiety of octanoyl-acyl carrier protein formed in the process of fatty acid synthesis is first transferred to the lysine of the H protein of the glycine cleavage system by octanoyltransferase, and then, two hydrogens (octanoyl positions 6 and 8) are replaced with sulfur groups, forming LA which can be reduced to dihydrolipoic acid (DHLA, the more bioactive form of LA) by the LA synthetase. Then, dihydrolipoyl moiety is transferred to lysine residues on the E2 components of the α-ketoacid dehydrogenase multienzyme complexes by the dihydrolipoamide dehydrogenase [10]. For free LA, the major metabolic fate is β-oxidation [14]. Combined form of LA in vivo can be metabolized as diversified forms: the conjugation of unmodified LA to glycine, tetranorlipoic acid, bisnorlipoate, and β-hydroxy-bisnorlipoate and the sulfoxide and S-methylation of the sulfide from its sulfur atoms in mammals [10, 14].
2.2. Biological Functions of LA
In physiological condition, endogenous LA can meet the need of fundamental functions mentioned above, with rare deficiency cases in human [15]. Moreover, many studies had shown that dietary supplements (in the free form of LA from chemical synthesis) can provide more additional effects, for example, owning antioxidant activities (through restoring reduced glutathione/glutathione [16]; disulfide regenerating vitamin C and vitamin E [17]; chelating iron, copper, manganese, and zinc [18]; inhibiting nuclear factor-κB (NF-κB) [19]; and activating nuclear factor erythroid 2-related factor 2(Nrf-2)/antioxidase [20]), improving cognition and dementia [21], controlling weight [22], and the treatment and prevention of other diseases (e.g., multiple sclerosis [23], diabetes complication [24], and cancer [25]) (Figure 1). There are more signaling pathways that LA acted on (e.g., insulin receptor substrate 1 [26], protein kinase C δ (PKCδ) [26], extracellular-regulated kinase 1/2 (Erk1/2) [27], phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt), and mitogen-activated protein kinase p38 (p38 MAPK) [28]. Meanwhile, LA can exert effects on almost every organ in the body (e.g., heart, kidney, and intestine) [29]. The elevated plasma LA levels after LA supplements may be responsible for these effects [30]. However, it seems impossible for exogenous LA to utterly replace the endogenous synthesis because it cannot be incorporated into LA-dependent enzymes, which is necessary for normal growth and development [31]. Still, LA had been approved for treating diabetic neuropathies by prescription in Germany [17]. Even, it is also available as an over-the-counter nutritional supplement in the United States. It indicated that free LA may own biological functions almost utterly consistent with the combined form of LA. Studies showed that it was safe for LA at the dosage of 600 mg/kg via a single intravenous administration without serious side effects [32, 33], exception of fewer exceptions in pregnancy [31] and lactation [34], together with certain risks of high doses in children [35] and the aging animal model [36]. And, there was a report showing that oral administration of 1,800 mg for 6 months had not any significant adverse side effects [37]. Therefore, it was attracting more researchers to explore potential biological functions of LA.
Figure 1.
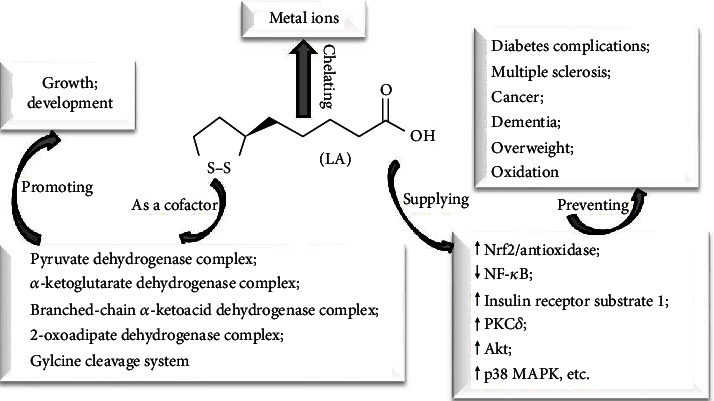
The biological functions of LA and potential mechanisms. Akt: protein kinase B; LA: lipoic acid; p38 MAPK: mitogen-activated protein kinase p38; Nrf2: nuclear factor erythroid 2-related factor 2; PKC: protein kinase C.
Many studies using LA to prevent I/R injury had been carried out in recent ten years though scientists had noticed these effects twenty-five years ago [38]. Increasing studies showed that supplements with free LA can play the vital role in attenuating I/R injuries of different organs and tissues. Meanwhile, more mechanisms apart from LA antioxidation involved in these processes were updated rapidly. Therefore, it was necessary to timely review these data in order to provide insights for further mechanism explorations and clinic investigations.
3. The Effects of LA on I/R Injuries of Different Organs
3.1. The Protective Role of LA in Cardiac I/R Injury
Myocardial ischemia is caused by reduced coronary blood flow accompanied with electric, functional, metabolic, and structural abnormities [39]. Restoring blood supply is the first choice of intervention, or lack of oxygen and energy supplies would cause myocardial atrophies, apoptosis, and necrosis [40]. Although the reperfusion reduced infarct size and necrosis, it also brought about additional injuries to the myocardium (myocardial I/R injury). The molecular mechanism of myocardial I/R injury mainly lay in a series of pathological processes: Ca2+ oscillations between cytosol and mitochondria, opening of the mitochondrial permeability transition pore, the production of reactive oxygen species, and the effects of transferring pH correction between adjacent myocytes due to acidosis [41]. In addition, prothrombogenic factors, endothelial cell activations, and inflammatory responses also contributed to the final damage of myocytes (even death) [42]. The myocardial I/R process was also considered as the main pathological manifestation of coronary artery disease [43]. Therefore, exploring cardioprotective bioactive agents was needed for attenuating myocardial I/R injury. In fact, the role of LA in myocardial I/R injury had been showed in an early study. The study [44] found that myocardial I/R can induce the loss of native LA in α-ketoglutarate dehydrogenase and the decline in α-ketoglutarate dehydrogenase activity. It was considered as the result of lipid peroxides reacting with LA residues on the E2 subunit of α-ketoglutarate dehydrogenase under free radicals produced in myocardial I/R process [45]. Therefore, supplements with LA may be a potential choice for attenuating myocardial I/R injury. Recently, studies demonstrated that LA supplements attenuated myocardial I/R injury in vivo and in vitro (Table 1). Main mechanisms included reducing oxidative stress via activating the PI3K/Akt/Nrf2 pathway and restoring alpha-tocopherol, inhibiting autophagy, restoring PKCε-dependent aldehyde dehydrogenase 2 (ALDH2) activities, and activating potassium ATP-sensitive channels.
Table 1.
The effects of LA on myocardial I/R injury.
| Species | I/R models | Treatments | Effects and mechanisms | Ref |
|---|---|---|---|---|
| Wistar rats | Left coronary artery I/R (15 min/30 min) | LA (10−7 M) from 15 min before ischemia to the end of reperfusion | Protecting cardiac I/R arrhythmias via its antioxidation and the activation of potassium ATP-sensitive channels | [49] |
| Sprague-Dawley rats | Isolated cardiac I/R (60 min/30 min) | LA (1, 5, 10∗10−8 M) for 10 min before ischemia | Reducing cardiac dysfunction and apoptosis via restoring PKCε/ALDH2 activity | [50] |
| Sprague-Dawley rats | Isolated cardiac I/R (30 min/30 min) | LA (0.5 μM) at 15 min before ischemia and during reperfusion | Improving cardiac hemodynamic function and damage via inhibiting mitochondrial O·2- formation (by DHLA) | [51] |
| Wistar rats | Isolated cardiac I/R (30 min/30 min) | LA (50 mg/kg∗day, i.p for 7 d) before surgery | Increasing coronary flow via scavenging free radicals | [52] |
| Rat H9c2 cardiomyocytes | Hypoxia/reoxygenation (4 h/3 h) | LA (300 μM) at 12 h before H/R | Protecting H/R injuries of cardiomyocytes by inhibiting autophagy | [53] |
| Sprague-Dawley rats | Left anterior descending coronary artery I/R (30 min/3 h) | LA (5, 10, 15, 25, or 50 mg/kg) via tail vein injection before I/R | Attenuating cardiomyocyte necrosis, apoptosis, and inflammation via activating PI3K/Akt/Nrf2 nuclear translocation/HO-1 | [54] |
| Wistar rats | Hypoxia/reoxygenation (45 min/60 min) in isolated hearts | LA (0.3 mM) in the hypoxic period, at 15 min before reoxygenation | Accelerating the recovery of the aortic flow via increasing ATP synthesis and phosphocreatine contents | [55, 56] |
| Wistar diabetic rats | Global no-flow I/R (30 min/30 min) in isolated hearts | LA (100 mg/kg i.p., 5 times/w, for 8 w) before perfusion | Reducing serious reperfusion dysrhythmias | [57] |
| Sprague-Dawley rats | Aortic cannula I/R (40 min/20 min) | LA (1.65 g/kg∗d) pretreatment for 6 weeks | Decreasing lipid peroxidation products and the loss of alpha-tocopherol | [58] |
Although the beneficial effects of LA on attenuating myocardial I/R injury are partly attributed to increased levels of other antioxidative agents (e.g., vitamins E and C) [46], sometimes, it was still essential for combining LA and these agents in preventing myocardial I/R injury because the combination would overcome the potential defects of applying LA alone in specific conditions (e.g., increased lipid peroxide levels in the heart of an aging person [36]). A study [47] showed that cardiac performances of aging rats after I/R were notably improved and lipid peroxidation in the heart was remarkably decreased after the supplement with vitamin E and LA. In addition, there was an enhancing effect for DHLA on vitamin E attenuating myocardial I/R injury [48]. The study also found that this interaction was independent on their concentrations. However, whether there were other potential mechanisms still needed further investigations.
3.2. The Protective Role of LA on Renal I/R Injury
Renal I/R injury usually occurred under some medical conditions (e.g., shock, sepsis, hypoperfusion, and renal transplantation) [59, 60]. These conditions caused direct damages to tubules, blood vessels, and glomeruli. Meanwhile, oxidative stress, inflammation, and mitochondrial dysfunction together contributed to acute kidney injury, which led to the abrupt decline of kidney function and increased mortality [61]. It was very vital to improve blood supplies and reduce pathological damages in the process of renal I/R. However, there were unmet medicines by the present day. It was critical to develop safe and effective strategies before renal I/R lesions. Encouragingly, some studies had shown that LA pretreatments at shorter time before I/R can attenuate renal I/R injury whenever in both lateral and bilateral animal models (Table 2). Renal function, histological lesions, and molecular indexes (e.g., aquaporins, sodium transporters, creatinine, and urea) were restored to varying degrees. The mechanisms involved included the elevation of antioxidative abilities and the reduction of inflammation and necrosis through regulating some signaling pathways (e.g., maintaining the normal status of arginine vasopressin/cAMP, nitric oxide/cGMP, and endothelin (ET) systems; depressing ET-1; increasing matrix metalloproteinases-2 and -9 activities; and decreasing metalloproteinases-1 and -2 levels). Based on these studies, the pretreatment with LA for a long time before renal I/R may have better effect and may be considered in further studies. And clinic investigations should also be carried out for prospective applications in patients that suffered from renal I/R injury.
Table 2.
The effects of LA on renal I/R injury.
| Species | I/R models | Treatments | Effects and mechanisms | Ref |
|---|---|---|---|---|
| Albino rats | Both renal I/R (1 h/2 h) | LA (100 mg/kg, i.p) at 2 days before I/R | Attenuating histopathological injury via reducing oxidative damage | [62] |
| Wistar albino rats | Right nephrectomy and left renal I/R (45 min/24 h) | LA (100 mg/kg, i.p) or saline twice, at 30 min before ischemia | Inhibiting neutrophil infiltration and inflammation generation, balancing the oxidant-antioxidant status | [63] |
| Sprague-Dawley rats | Bilateral renal I/R (45 min/24 h) | Pretreatment with LA (50 mg/kg i.p) for 2 weeks | Decreasing levels of creatinine and urea together with oxidative stress and inflammation; elevating GSH levels and activities of antioxidant enzymes | [64] |
| Albino Wistar rats | Right nephrectomy and the left renal I/R (1 h/24 h) | LA (100 mg/kg, i.p) 30 min before I/R | Reducing degradation of extracellular matrix and oxidative stress via increasing matrix metalloproteinases-2 and -9 activities and decreasing metalloproteinases-1 and -2 levels | [65] |
| Sprague-Dawley rats | Bilateral renal I/R (40 min/2 d) | LA (80 mg/kg i.p) at 48 and 24 h before ischemia and at 6 and 24 h after reperfusion | Protecting renal function (e.g., aquaporins and sodium transporters) via normalizing activities of local arginine vasopressin/cAMP, nitric oxide/cGMP, and ET systems | [66] |
| Sprague-Dawley rats | Right nephrectomy and left renal I/R (45 min/24 h) | LA (10 or 100 mg/kg, i.p) at 24 and 1 h before ischemia | Attenuating the deterioration of renal function; repressing tubular necrosis, proteinaceous casts in tubuli, and medullary congestion through suppressing ET-1 | [67] |
3.3. The Protective Role of LA on Hepatic I/R Injury
Liver ischemia is secondary to some pathologies (e.g., hemorrhagic shock and resuscitation [68]) and surgeries (e.g., liver resections [69] and liver transplantation [70]). Although, to some extent, the liver is tolerant of ischemia injury in the initial phase, the productions of xanthine oxidase and NADPH oxidase in liver cells drive more serious damages in the second phase of reperfusion [71]. The two-stage hepatic I/R injury was still the vital cause of liver surgical failure (e.g., hepatic resection and liver transplantation) and finally increased liver morbidity and mortality [72]. Reactive oxygen species and activation of inflammatory played the key role in the pathology of hepatic I/R injury [73]. It had previously demonstrated that LA can be used for curing liver-related diseases (e.g., hepatic cirrhosis, hepatomegaly in hepatic intoxication, alcohol clearance, and increased glucose disposal in the liver) [34]. Recent studies showed that LA can attenuate hepatic I/R injury in clinical and animal trials (Table 3). In two cases of human liver transplantation and resection, LA pretreatments (intravenous injections of 600 mg) remarkably reduced manifestations of postreperfusion syndrome via blocking oxidative stress and apoptosis. In animal models, LA treatments also alleviated hepatic I/R injuries in functions and pathological constructs. The mechanism included scavenging radicals, increasing antioxidant enzyme activities, inhibiting inflammatory reaction via activating the PI3-kinase/Akt pathway, inhibiting NF-κB P65 and macrophage inflammatory protein-2 (MIP-2), and preventing hepatocyte deaths caused by apoptosis and necrosis. Therefore, LA was a prospective agent for preventing hepatic I/R injury in further clinical applications.
Table 3.
The effects of LA on hepatic I/R injury.
| Subjects | I/R | Treatments | Effects and mechanisms | Ref |
|---|---|---|---|---|
| Human | Hepatic I/R (2 h/2 h); blood samples (before; 1, 2, 3, 7, and 30 d after liver transplantation) | LA (600 mg) to the donor portal vein before ischemia and another LA (600 mg) at 15 min before reperfusion | Protecting against oxidative stress and reducing the appearance of postreperfusion syndrome | [74] |
| Human | Hepatic I/R (45 min /60 min) in resection | LA (600 mg, i.v) at 15 min before transection | Reducing I/R injury via attenuating apoptosis | [75] |
| Wistar albino rats | Hepatic I/R (45 min/60 min) | LA (100 mg/kg, i.p.) before ischemia and immediately before reperfusion period | Alleviating the I/R-induced liver injury in the hepatic structure and function via antioxidant properties | [76] |
| Wistar rats | Hepatic I/R (60 min/(1, 3, 6, and 12 h) | Injection with LA (25 mg) into the caudal vein at 15 min before reperfusion and 60 min after ischemia | Scavenging oxygen and nitrogen free radicals, increasing GSH levels, reducing inflammatory reaction mediated by NF-κB P65 and MIP-2 | [77] |
| Sprague-Dawley rats | Hepatic I/R (60 min/45,90 min) | LA (10 or 50 μM) at 20 min before ischemia | Attenuating I/R injury via activating the PI3-kinase/Akt pathway | [78] |
| Brown Norway rats | Hepatic selective lobe I/R (10 min/10 min), then sustained I/R (90 min/4 h) | LA (500 μmol, i.v) at 15 min before ischemia | Improving tolerance to ischemia via downregulating proapoptotic gene | [79] |
| Brown Norway rats | Hepatic I/R (90 min/1 h) | LA (120 mg) via the inferior vena cava at 15 min in advance | Attenuation of I/R by reducing necrosis and apoptosis | [80] |
| Brown Norway rats | Hepatic I/R (90 min/1 and 4 h) | LA (1.5 ml, 5,000 mol, i.v) at 15 min prior to ischemia | Attenuating I/R via reducing cell deaths from necrosis and apoptosis | [81] |
3.4. The Protective Role of LA on Cerebral I/R Injury
Cerebral ischemia, resulting from pathologies of brain blood vessels (occlusion or rupture) and cardiac dysfunctions [82], caused brain damages (e.g., neuronal deaths of adults [83] and hypoxic-ischemic encephalopathy of infants [84]). The morbidity and mortality of cerebral ischemia can be controlled to a certain degree by the timely reperfusion. However, cerebral reperfusion further aggregated cerebral injuries via inducing inflammation, oxidative stress, and the breakdown of the blood-brain barrier [85, 86]. Therefore, it is vital to explore more strategies to protect cerebral functions and pathological developments in cerebral I/R. Because LA can cross the blood-brain barrier easily without any serious side effects [87], it was considered an effective candidate to attenuate numerous neurodegenerative disorders via its antioxidative action [88]. It had previously been proven that LA pretreatment significantly protected cerebral focal and forebrain ischemia [89]. Further, studies in vivo and in vitro had shown that LA treatments restored neurological structures and functions in cerebral I/R via attenuating oxidative damage mediated by the Nrf2/heme oxygenase-1 (HO-1) pathway and reduced cerebral infarct volumes, apoptosis, and inflammation reaction via inhibiting the Toll-like receptor 4 (TLR4)/myeloid differentiation primary response 88 (MyD88)/NF-κB pathway and Akt/mammalian target of rapamycin (mTOR) (Table 4).
Table 4.
The effects of LA on cerebral I/R injury.
| Subjects | I/R | Treatments | Effects and mechanisms | Ref |
|---|---|---|---|---|
| Sprague-Dawley rats | Left external carotid artery permanently occluded and left internal carotid artery I/R (90 min/24 h) | LA (50, 70, and 100 mg/kg, i.h) at 2 h before I/R | Attenuating I/R injury via reducing oxidative stress and caspase-dependent apoptosis | [94] |
| Sprague-Dawley rats | Middle cerebral artery selective I/R (30 s/30 s) before sustained I/R (2 h/24 h) | LA (50 mg/kg i.p) for 30 min before I/R | Exhibiting neuroprotection by attenuating neuroinflammation via inhibiting the TLR4/MyD88/NF-κB pathway | [95] |
| Sprague-Dawley rats | Left middle cerebral artery I/R (2 h/1, 3, 7, 14, 21, 28, 35, 42, 49, and 56 d) | LA injection (20 mg/kg) via the left external jugular vein immediately after I/R | Exhibiting neurorestorative effects via insulin receptor activation, anti-inflammatory and antioxidant actions | [96] |
| Sprague-Dawley rats | Ligating external carotid arteries and the internal carotid artery I/R (2 h/24 h) | LA (10, 20,40, and 80 mg/kg) via the left external jugular vein after reperfusion | Promoting functional recovery via attenuating oxidative damage mediated by the Nrf2/HO-1 pathway | [97] |
| Sprague-Dawley rats | Right middle cerebral artery I/R (30 min/5.5 h) | LA (0.05, 0.5, 5.0, or 50 mg/kg, i.v) at 30 min | LA as a neuroprotectant via increasing protein expression of superoxide dismutase 2 | [98] |
| Sprague-Dawley rats | Permanent distal middle cerebral artery occlusion-bilateral common carotid artery I/R (30 min/48 h) | LA (40 mg/kg, i.p) at 30 min after ischemia | Protective effects by LA via reducing phosphate mTOR | [99] |
| Albino rats | Bilateral femoral arteries I/R (3 h/1d) | LA (100 mg/kg, i.p) for 7 d after I/R | Ameliorating neuronal damages, maintaining the normal structure, surface area, and the number of light neurons in the pyramidal layer | [100] |
| Mouse primary brain endothelial cell and bEnd.3 cell | Oxygen glucose deprivation/reperfusion (6 h/4 h) | LA (1 mM) pre- and posttreatment for ischemia | Protecting against oxygen glucose deprivation/reperfusion injury via promoting the Akt/mTOR pathway | [101] |
| Sprague-Dawley rats | Bilateral carotid artery I/R (30 min/24 h) | LA (25 mg/kg, i.v) pretreatments | Improving survival and protecting the rat brain | [102] |
| Gerbils | Forebrain I/R (5 min/5 d) | LA (20 mg/kg∗d, i.p) for 7 d | Improving locomotor abilities and damages of the CA1 hippocampal pyramidal cells via antioxidant actions | [103] |
| C57blk mice | Internal carotid artery I/R (45 min/24 h) | LA (100 mg/kg, i.h)at 1.5 h before ischemia | Reducing stroke infarct volume via antioxidant actions | [104] |
LA also produced synergistic effects against cerebral I/R injuries when combined with other agents. For instance, the pretreatment with LA (20 mg/kg) and vitamin E (50 mg/kg) can reduce neurological deficits and reactive gliosis and promote neuronal remodelings in cerebral I/R rats [90]. Another study showed that the pretreatment of LA for 2 h in a lower dose (50 μM) together with other thiol-stimulating agents (e.g., ambroxol and enalapril) can significantly increase neuronal survivals in oxygen glucose deprivation/reperfusion of ex vivo rat organotypic hippocampal slice cultures [91], despite no effects while it being used alone. Further, the cotreatment with LA and other agents reduced not only the effective dosage of LA but also that of other agents. Therefore, it would help reduce the potential toxicities of these agents when it was used at a noneffective low dose together with them. Two previous studies [92, 93] had shown that LA administrations at a very low dose (0.005 mg/kg) can significantly enhance the effects of two agents (apocynin and resveratrol) at very low doses (0.05 mg/kg and 0.002 mg/kg) prior to middle cerebral I/R. In contrast, the effective dosages of apocynin and resveratrol were 200- and 1000-fold decreases in the amount to achieve alike protective effects in cerebral I/R injuries while they were used alone. Therefore, the combination of LA with the two agents would significantly reduce occurrences of their potential side effects (e.g., possible prooxidant effects for apocynin and renal toxicity for resveratrol) at high dosages. Thus, to explore more combination of LA and other agents and corresponding mechanisms would be meaningful for clinic applications.
3.5. The Protective Role of LA on Spinal I/R Injury
Spinal I/R injury is usually induced by the cardiovascular surgery (e.g., the decompression of a previous compressed region of the spinal cord due to thoracoabdominal aortic interventions [109]). Spinal I/R injury further induces nerve injuries and damages of sensation and movement (even paraplegia) [110]. Oxidative stress, inflammation, and excitotoxicity were considered playing vital roles in spinal I/R injury [111]. In previous studies, some therapeutic measures had been used to cure spinal I/R injury (e.g., the preconditionings of hyperbaric oxygen [112] and remote ischemia [113], pharmacologic administrations such as progesterone [114], and the application of pluripotent stem cells [115]), but the effects of treatments were yet unsatisfied. Therefore, it was necessary to explore new treatments or agents for attenuating spinal I/R injury. LA, owning strong antioxidative abilities, had been used for curing spinal I/R injuries in animal models (Table 5). The studies showed that LA pretreatments can attenuate spinal neurological injuries via reducing oxidative damages and inflammation. The effect was related to the time point of exposing to LA in advance (e.g., LA pretreatments exhibiting poorer effects on spinal I/R injuries at 5 and 10 min before ischemia compared with these at longer time points from Table 5). However, further studies and clinical investigations were needed for potential applications of LA in attenuating spinal I/R injury.
Table 5.
The effects of LA on spinal I/R injury.
| Species | I/R models | Treatments | Effects and mechanisms | Ref |
|---|---|---|---|---|
| New Zealand rabbits | Abdominal aorta I/R (30 min/48 h) | LA (100 mg/kg, i.p) at 5 min before I/R | Decreasing spinal I/R injury via reducing plasma and spinal levels of nitric oxide, glutathione, malondialdehyde, and advanced oxidation protein products | [105] |
| New Zealand rabbits | Abdominal aorta I/R (30 min/48 h) | LA (100 mg/kg, i.p) at 20 min before ischemia | Decreasing neuronal degeneration, axonal damage, and microglial and astrocytic infiltration via oxidative stress | [106] |
| Wistar rats | Abdominal aorta I/R (30 min/3 d) | LA (100 mg/kg, i.h) at 3 days before ischemia | Reducing neurologic injury by maintaining the oxidant/antioxidant balance | [107] |
| New Zealand rabbits | Abdominal aorta I/R (20 min/2 h) | LA (100 mg/kg) at 10 min before ischemia | Exhibiting antioxidant efficacy with no effect on oxidative stress | [108] |
3.6. The Protective Role of LA on Intestinal I/R Injury
Compared with other organs, the intestine is more sensitive to I/R injury [116]. Intestinal I/R injury occurred in many cases such as trauma, strangulated intestinal obstruction, acute mesenteric ischemia, thrombosis of the mesenteric artery, shock, incarcerated hernia, hemorrhagic, and surgical procedures (e.g., small intestinal transplantation, cardiopulmonary bypass, or abdominal aortic aneurysm) [117–120]. The high mortality [121] of intestinal I/R injury still occurred due to inflammation and oxidative stress though many intervention efforts had been made. It is essential to constantly search for more prevention strategies. Several studies showed that either early or simultaneous treatments of LA attenuated intestinal I/R injury. The difference between the two kinds of treatments was just that the simultaneous treatment of LA together with ischemia occurrence produced less protective effects on intestinal I/R injuries [122] compared to better protective effects exhibited by both LA preconditioning [123] and the subsequent continuous administration [124]. However, the defect of the former treatment can be offset through its combination with other agents (e.g., anti-inflammatory drug: ebselen). The mechanism of LA attenuating intestinal I/R injury in these studies can be attributed to its abilities of antagonizing oxidative damages and these anti-inflammatory effects of ebselen. Therefore, combined effects of LA and other drugs possibly provided an alternative treatment strategy for intestinal I/R injury.
3.7. The Protective Role of LA on Limb I/R Injury
Limb ischemia occurred in the lower extremities secondary to atherosclerotic vascular stenosis and other pathologies (e.g., aneurysms with intraluminal thrombus) [125]. Although there are lower incidences and longer reperfusion window periods [126], untimely reperfusion treatments yet induce irreversible tissue damages from skin numbness and paralysis of the musculature to limb loss and even death [127]. Treatment options had shifted from surgeries to endovascular therapies, which made preconditioning with active agents more vital for preventing limb I/R injury. Studies had shown that LA treatment can attenuate functional damages in the lower extremities of some pathologies (e.g., diabetic foot syndrome [128] and the polyneuropathy of patients with hemoblastosis [129]), indicating possible preventive and protective efforts of LA on limb I/R injury. A study [130] showed that LA treatment (24 mg/kg, i.p) after 4 hours of ischemia and at the onset of 24-hour reperfusion decreased the levels of muscle tumor necrosis factor-alpha and oxidative stress indexes in muscles of hind limb I/R model rats. Another study [55] showed that LA treatment (8.3 mM) along with 30 min reperfusion after 4 h hind limb ischemia (isolated rat hind limbs) also recovered the function (contractile and flexibility). The protective effect of LA on limb I/R injury was of inconsistent efficacies, depending on lasting time of the ischemia. For instance, intraperitoneal (i.p) pretreatments of LA (100 mg/kg daily for 3 days) improved distal sensory conductions of the sciatic-tibial nerves in the right hind limb artery I/R (3 h/7 d) model rats [131]. However, the ischemic nervous fiber degeneration was not attenuated by LA if ischemia time was prolonged to 5 h. It indicated that the period of ischemia may be the key factor, which aggravated I/R injury and made the injury hard to be reversed. However, whether long-term preconditionings with LA can attenuate these I/R injuries experiencing long-term ischemia needed further studies. LA treatment also attenuated the injuries of other tissues caused by limb I/R. A recent study [100] showed that limb I/R induced the degenerative injury of rat hippocampal CA3 neurons (e.g., the reduction of light neuron number, surface areas of neurons, and the thickness of the CA3 layer together with increased capillary lesions), and an injection of LA (100 mg/kg, i.p) for 7 d significantly ameliorated these damages. The result indicated the potential application of LA in both nervous system and peripheral tissues simultaneously induced by limb I/R.
3.8. The Protective Role of LA on I/R Injury of the Gonad
Gonadal ischemia is usually the emergency occurring in males (e.g., testis ischemia) or females (e.g., ovarian ischemia). Torsion is the main reason inducing ischemia in the process. For instance, testis ischemia is frequently caused by the rotation of spermatic cord and the involved patients mostly come from adolescents under 30 years old [132]. Ovarian ischemia is also caused by the torsion, which results from ovarian diseases, surgery, traumas, or pregnancy [133]. Due to acute pains in gonadal ischemia, the two kinds of I/R of gonad needed emergency treatments of surgical detorsion to restore blood perfusion to the gonad. If the reperfusion was not carried out timely, further dysfunctions of the gonad occurred, for example, apoptosis, testicular atrophy, and disturbances of spermatogenesis in males [134] and ovary edema, bleeding, and necrosis in females [135]. Even the successful surgical intervention cannot utterly restore the gonadal dysfunctions resulting from oxidative stress and inflammatory response after reperfusions [136, 137]. Therefore, it must be considered to develop other interventions of drugs and bioactive agents. Recently, several studies had been reported about protective efforts of LA treatments on I/R injury of the gonad. First, LA treatment can attenuate testicular I/R injury in males. The pretreatment with LA injections (12 mg/kg, i.p at 21, 9, and 1 h before ischemia) reduced testicular I/R injury through decreasing lipid peroxidation and increasing total levels of antioxidant power in rat blood plasma and testis [138]. Another research [139] found that LA administration (100 mg/kg, i.p) at 30 min before ischemia also produced beneficial effects in protecting testicular I/R injury, which decreased apoptosis and oxidative damage, and increased antioxidative abilities were also responsible for the effects.
Meanwhile, LA treatment also reduced ovarian I/R injury in females. Cosar et al. [140] found that injections with LA (36 mg/kg/d, i.p) in 21, 9, and 1 hour before ischemia can prevent rat ovarian I/R injuries (3 h/3 h) through reducing oxidative damage (downregulated malondialdehyde, xanthine oxidase, and nitric oxide; upregulated superoxide dismutase). By contrast, high dosage of LA pretreatment (100 mg/kg) in adnexa I/R injury ameliorated ovary dysfunctions (e.g., restored numbers of primordial follicles and primary follicles) based on attenuating total oxidant status and oxidative stress index [141]. However, bilateral ovarian damages may be differently affected by LA treatment while a lateral ovarian I/R occurred. LA treatment only attenuated the lateral ovarian damage undergoing I/R. The contralateral ovarian injury induced by increased sympathetic activity secondary to the I/R procedure was not restored [142]. Therefore, synergistic therapy in applying LA to prevent gonadal I/R injury may be considered to reduce the contralateral ovarian injury second to gonadal I/R.
3.9. The Protective Role of LA on Retinal I/R Injury
Retinal ischemia often occurred in some pathologies (e.g., glaucoma, diabetic retinopathy, ophthalmic artery, and some central retinal vein occlusion) and further developed into visual impairment and blindness [143]. Restored blood supplement after ischemia further induced injuries of retinal cells via producing reactive oxygen species. Inflammatory reactions and the damage [144] of retinal neurons [145] were considered as the key factors in structural and functional dysfunctions of the retina. Previous studies had shown that LA can play a protective role on retina pathologies. LA administration had been confirmed to protect the complications of retinal lesions in some diseases (e.g., glaucoma [146] and diabetes [147]). LA may be applied to attenuate retinal I/R injury. A study [148] showed that the injection with LA treatment (100 mg/kg∗day, i.p. for 11 days) rectified rat retinal I/R injuries (e.g., reducing amplitudes of the electroretinogram a and b waves, Thy-1(CD90) immunoreactivities, and retinal ganglion cell-specific mRNA levels and increasing bFGF and CNTF mRNA levels) via reducing nitric oxide synthase. In vitro, LA supplement (100 mM) in the cultural medium ameliorated the loss of GABA neurons in isolated rat retinas undergoinganoxic/reoxygenation. However, the mechanism was unclarified. Even in a long-term lead exposure model, the administration of LA (70 mg/kg, i.p) 24 hour prior to retinal I/R (2 h/5d) produced the anti-inflammatory effects via elevating levels of glial-derived neurotrophic factor (GDNF) and ciliary neurotrophic factor (CNTF) and reducing levels of glial fibrillary acidic protein (GFAP) [149]. Therefore, LA was a potential agent for preventing retinal I/R injury in future.
4. The Dose of LA in Attenuating I/R Injury and Potential Side Effects
The dosage of LA based on present data is of importance. The effects of LA on I/R injuries of different organs were mostly obtained from animal models. At present, the side effects were mainly affected by the dosage and the period of treatments. Firstly, for animal tests, the studies were mainly conducted in a short time prior to ischemia, in which even high doses of LA (up to 2000 mg/kg∗d) treatments nearly caused no acute toxicity except for some subhealthy signs in behavior (sedation, piloerection, hunched posture, and eye closure) [150]. However, a long-term treatment (more than 1 month) with middle doses of LA (e.g., 61.9 mg mg/kg∗d in females) caused single-cell necrosis of hepatocytes. Compared with animals, the dosages for human were relatively lower.For humans, a safe dose of LA as the health ingredient used in food supplements was recommended as a daily intake of 150-200 mg/d in Denmark [151], through the intakes of 300 to 1800 mg/d to improve symptoms of corresponding diseases had been applied. However, allergic reactions in the skin (e.g., hives and itchiness) and gastrointestinal symptoms (e.g., stomach ache, nausea, and diarrhea) often occurred in long-term applications [152–154]. Although there were no reports about the side effects of LA attenuating I/R injury at present and the dosages in present studies were relatively low, it should be considered the potential side effects of LA applying to prevent I/R injury. It was because of increasing risks of LA side effects in certain conditions (as discussed above in pregnancy [31], lactation [34], children [35], and the aging animal model [36]).Therefore, dosages of LA should be carefully explored for health maintenance and interventional therapies combined with different lengths of treatment time and the possible side effects.
5. Conclusion
Ischemia is caused by shortness of blood supplements, and the direct result is derangements of metabolic function (e.g., the decrease of ATP production) and dysfunctions of some gene expressions, such as the genes related to inflammatory response and genes producing free radicals due to hypoxia in mitochondria. Although reperfusion is always considered able to reduce further damages, I/R injury is unavoidable. Researchers constantly developed new interventions to overcome potential damages caused by reperfusion alone. LA, as a functional agent of food sources and an endogenous active substance, also owned additional biological functions and the roles for some diseases (improving cognition and dementia, controlling weight, and preventing multiple sclerosis, diabetes complications, and cancers) while supplying as a health component or the prescription. In the I/R process, the exhaustion of LA indicated its potential protective roles. Recently, studies had shown that supplements with LA can significantly attenuate I/R injuries in multiple organs (nervous system, heart, kidney, liver, intestine, gonad gland, retina, and limb). Apart from its antioxidative actions, other mechanisms also involved in protecting I/R injury (Figure 2) include the activation of potassium ATP-sensitive channels; the restoration of PKCε-dependent ALDH2 activity/apoptosis inhibiting; autophagy inhibition; the activation of PI3K/Akt/Nrf2 nuclear translocation/HO-1; increases of ATP synthesis and phosphocreatine contents; the reduction of inflammatory reaction via maintaining the normal status of arginine vasopressin/cAMP, nitric oxide/cGMP, and ET systems; increasing matrix metalloproteinases-2, and -9 activities and decreasing metalloproteinase-1 and -2 levels; inhibiting TLR4/MyD88/NF-κB P65 and MIP-2 mRNA; downregulating proapoptotic genes; reducing phosphate mTOR via Akt; elevating levels of GDNF and CNTF; and lowering levels of GFAP. Therefore, LA may be a prospective agent for preventing I/R injuries of multiple organs. Interestingly, the majority of these data was based on studies of pretreatments with LA. Therefore, fortifying the contents of lipoic acid in foods by chemical synthesis or supplying it from foods before I/R may provide a potential way to attenuate I/R injuries compared with the difficulty to prevent I/R injuries in treatments after I/R. In this respect, clinical investigations including the relationship between LA intake and the risk of I/R injuries as well as the data about LA intervention strategy as the health agent in human were very limited and may become a further research topic.
Figure 2.
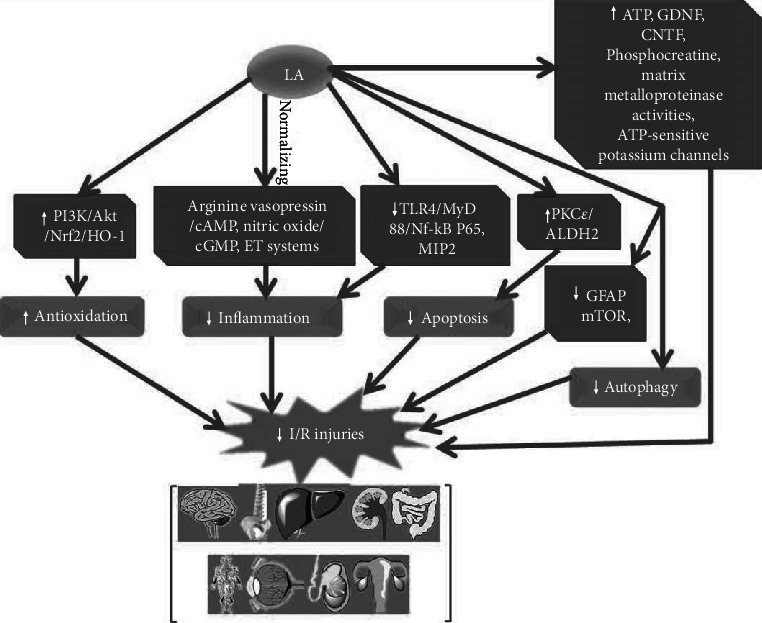
The mechanism of LA attenuating I/R injuries in the brain, spine, heart, liver, kidney, intestine, gonad, retina, and limb. Akt: protein kinase B; ALDH2: aldehyde dehydrogenase 2; CNTF: ciliary neurotrophic factor; ET: endothelin; GDNF: glial-derived neurotrophic factor; GFAP: reducing levels of glial fibrillary acidic protein; HO-1: heme oxygenase-1; LA: lipoic acid; MIP-2: macrophage inflammatory protein-2; mTOR: mammalian target of rapamycin; MyD88: myeloid differentiation primary response 88; Nrf2: nuclear factor erythroid 2-related factor 2; PI3K: phosphatidylinositol 3-kinase; PKC: protein kinase C; TLR4: Toll-like receptor 4.
Besides, animal tests and human applications indicated potential side effects of LA. Thus, it was also an important concern about how to balance its protective actions and the potential side effects in I/R injury (with no reports). In conclusion, LA, as an endogenous and foodborne active agent, can attenuate I/R injuries of many organs, which widened the range of it as functional nutritional factors. The reasonable choices of administration time, dosages, and duration together with potential side effects should be considered in future investigations.
Acknowledgments
This research was supported by the Henan Philosophy and Social Science Planning Project, China (grant no. 2019BJY006).
Abbreviations
- Akt:
Protein kinase B
- ALDH2:
Aldehyde dehydrogenase 2
- CNTF:
Ciliary neurotrophic factor
- DHLA:
Dihydrolipoic acid
- Erk1/2:
Extracellular-regulated kinase 1/2
- ET:
Endothelin
- GDNF:
Glial-derived neurotrophic factor
- GFAP:
Reducing levels of glial fibrillary acidic protein
- HO-1:
Heme oxygenase-1
- LA:
Lipoic acid
- MIP-2:
Macrophage inflammatory protein-2
- mTOR:
Mammalian target of rapamycin
- MyD88:
Myeloid differentiation primary response 88
- Nrf2:
Nuclear factor erythroid 2-related factor 2
- p38 MAPK:
Mitogen-activated protein kinase p38
- PI3K:
Phosphatidylinositol 3-kinase
- PKC:
Protein kinase C
- TLR4:
Toll-like receptor 4.
Contributor Information
Zhenxing Xie, Email: pet213@163.com.
Chaoran Chen, Email: kfccr@henu.edu.cn.
Conflicts of Interest
The authors declare that they have no conflict of interest.
Authors' Contributions
Zhang Wunong, Jia Shang, and Zhenxing Xie collected the data and composed the manuscript. Yueming Ding, Yiming Zhang, and Chaoran Chen helped a lot in designing the graphic abstracts. All authors have reviewed and agreed to the publication of the manuscript. Yiming Zhang and Wunong Zhang contributed (co-first authors) equally to this work as Yueming Ding.
References
- 1.Gourgoutis G., Das G. Gastrointestinal manifestations of cocaine addiction. International Journal of Clinical Pharmacology and Therapeutics . 1994;32(3):136–141. [PubMed] [Google Scholar]
- 2.Carden D. L., Granger D. N. Pathophysiology of ischaemia-reperfusion injury. The Journal of Pathology . 2000;190(3):255–266. doi: 10.1002/(SICI)1096-9896(200002)190:3<255::AID-PATH526>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 3.Wu M. Y., Yiang G. T., Liao W. T., et al. Current mechanistic concepts in ischemia and reperfusion injury. Cellular Physiology and Biochemistry . 2018;46(4):1650–1667. doi: 10.1159/000489241. [DOI] [PubMed] [Google Scholar]
- 4.Bhaskar S., Stanwell P., Cordato D., Attia J., Levi C. Reperfusion therapy in acute ischemic stroke: dawn of a new era? BMC Neurology . 2018;18(1):p. 8. doi: 10.1186/s12883-017-1007-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hsu C. Y., Ahmed S. H., Lees K. R. The therapeutic time window--theoretical and practical considerations. Journal of Stroke and Cerebrovascular Diseases . 2000;9(6, Part 2):24–31. doi: 10.1053/jscd.2000.19318. [DOI] [PubMed] [Google Scholar]
- 6.Liang S., Ping Z., Ge J. Coenzyme Q10 regulates antioxidative stress and autophagy in acute myocardial ischemia-reperfusion injury. Oxidative Medicine and Cellular Longevity . 2017;2017:12. doi: 10.1155/2017/9863181.9863181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liao Z. P., Liu D., Tang L., et al. Long-term oral resveratrol intake provides nutritional preconditioning against myocardial ischemia/reperfusion injury: involvement of VDAC1 downregulation. Molecular Nutrition & Food Research . 2015;59(3):454–464. doi: 10.1002/mnfr.201400730. [DOI] [PubMed] [Google Scholar]
- 8.Zúñiga J., Cancino M., Medina F., et al. N-3 PUFA supplementation triggers PPAR-α activation and PPAR-α/NF-κB interaction: anti-inflammatory implications in liver ischemia-reperfusion injury. Plos One . 2011;6(12, article e28502) doi: 10.1371/journal.pone.0028502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Reed L. J., DeBusk B. G., Gunsalus I. C., Hornberger C. S. Crystalline alpha-lipoic acid; a catalytic agent associated with pyruvate dehydrogenase. Science . 1951;114(2952):93–94. doi: 10.1126/science.114.2952.93. [DOI] [PubMed] [Google Scholar]
- 10.LPI. Lipoic acid . Corvallis: Micronutrient Information Center, Oregon State University; 2019. [Google Scholar]
- 11.Lodge J. K., Youn H. D., Handelman G. J., et al. Natural sources of lipic acid: determination of lipoyllysine released from protease-digested tissues by high performance liquid chromatography incorporating electrochemical detection. Journal of Applied Nutrition . 1997;49(1&2):3–11. [Google Scholar]
- 12.Carlson D. Y., Fischer K. L., Ulrich S. J. Lipoic Acid: Energy Production, Antioxidant Activity and Health Effects . Packer & Patel; 2008. Ch. 10: an evaluation of the stability and pharmacokinetics of R-lipoic acid and R-dihydrolipoic acid dosage forms in plasma from healthy human subjects; pp. 235–270. [Google Scholar]
- 13.Prasad P. D., Wang H., Kekuda R., et al. Cloning and Functional Expression of a cDNA Encoding a Mammalian Sodium- dependent Vitamin Transporter Mediating the Uptake of Pantothenate, Biotin, and Lipoate∗. Journal of Biological Chemistry . 1998;273(13):7501–7506. doi: 10.1074/jbc.273.13.7501. [DOI] [PubMed] [Google Scholar]
- 14.Schupke H., Hempel R., Peter G., et al. New metabolic pathways of alpha-lipoic acid. Drug Metabolism and Disposition . 2001;29(6):855–862. [PubMed] [Google Scholar]
- 15.Kohlmeier L. Nutrient Metabolism . Elsevier; 2003. Lipoate; pp. 526–532. [DOI] [Google Scholar]
- 16.Bitar M. S., Ayed A. K., Abdel-Halim S. M., Isenovic E. R., al-Mulla F. Inflammation and apoptosis in aortic tissues of aged type II diabetes: Amelioration with α-lipoic acid through phosphatidylinositol 3-kinase/Akt- dependent mechanism. Life Sciences . 2010;86(23-24):844–853. doi: 10.1016/j.lfs.2010.03.019. [DOI] [PubMed] [Google Scholar]
- 17.Biewenga G. P., Haenen G. R., Bast A. The pharmacology of the antioxidant lipoic acid. General Pharmacology . 1997;29(3):315–331. doi: 10.1016/S0306-3623(96)00474-0. [DOI] [PubMed] [Google Scholar]
- 18.Valko M., Morris H., Cronin M. T. Metals, toxicity and oxidative stress. Current Medicinal Chemistry . 2005;12(10):1161–1208. doi: 10.2174/0929867053764635. [DOI] [PubMed] [Google Scholar]
- 19.Hofmann M. A., Schiekofer S., Kanitz M., et al. Insufficient glycemic control increases nuclear factor-kappa B binding activity in peripheral blood mononuclear cells isolated from patients with type 1 diabetes. Diabetes Care . 1998;21(8):1310–1316. doi: 10.2337/diacare.21.8.1310. [DOI] [PubMed] [Google Scholar]
- 20.Van Nguyen T., Piao C. H., Fan Y. J., et al. Anti-allergic rhinitis activity of α-lipoic acid via balancing Th17/Treg expression and enhancing Nrf2/HO-1 pathway signaling. Scientific Reports . 2020;10(1, article 12528) doi: 10.1038/s41598-020-69234-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Liu J. The effects and mechanisms of mitochondrial nutrient alpha-lipoic acid on improving age-associated mitochondrial and cognitive dysfunction: an overview. Neurochemical Research . 2008;33(1):194–203. doi: 10.1007/s11064-007-9403-0. [DOI] [PubMed] [Google Scholar]
- 22.Escoté X., Félix-Soriano E., Gayoso L., et al. Effects of EPA and lipoic acid supplementation on circulating FGF21 and the fatty acid profile in overweight/obese women following a hypocaloric diet. Food & Function . 2018;9(5):3028–3036. doi: 10.1039/C8FO00355F. [DOI] [PubMed] [Google Scholar]
- 23.Salinthone S., Yadav V., Bourdette D. N., Carr D. Lipoic acid: a novel therapeutic approach for multiple sclerosis and other chronic inflammatory diseases of the CNS. Endocrine, Metabolic & Immune Disorders - Drug Targets . 2008;8(2):132–142. doi: 10.2174/187153008784534303. [DOI] [PubMed] [Google Scholar]
- 24.Rochette L., Ghibu S., Muresan A., Vergely C. Alpha-lipoic acid: molecular mechanisms and therapeutic potential in diabetes. Canadian Journal of Physiology and Pharmacology . 2015;93(12):1021–1027. doi: 10.1139/cjpp-2014-0353. [DOI] [PubMed] [Google Scholar]
- 25.Farhat D., Lincet H. Lipoic acid a multi-level molecular inhibitor of tumorigenesis. Biochimica et Biophysica Acta - Reviews on Cancer . 2020;1873(1, article 188317) doi: 10.1016/j.bbcan.2019.188317. [DOI] [PubMed] [Google Scholar]
- 26.Sen C. K., Sashwati R., Packer L. Fas mediated apoptosis of human Jurkat T-cells: intracellular events and potentiation by redox-active α-lipoic acid. Cell Death & Differentiation . 1999;6(5):481–491. doi: 10.1038/sj.cdd.4400514. [DOI] [PubMed] [Google Scholar]
- 27.Shi S. S., Day R. M., Halpner A. D., Blumberg J. B., Suzuki Y. J. Homocysteine and alpha-lipoic acid regulate p44/42 MAP kinase phosphorylation in NIH/3T3 cells. Antioxidants & Redox Signaling . 1999;1(1):123–128. doi: 10.1089/ars.1999.1.1-123. [DOI] [PubMed] [Google Scholar]
- 28.Konrad D., Somwar R., Sweeney G., et al. The antihyperglycemic drug alpha-lipoic acid stimulates glucose uptake via both GLUT4 translocation and GLUT4 activation - Potential role of p38 mitogen-activated protein kinase in GLUT4 activation. Diabetes . 2001;50(6):1464–1471. doi: 10.2337/diabetes.50.6.1464. [DOI] [PubMed] [Google Scholar]
- 29.Jia J., Gong X., Zhao Y., et al. Autophagy Enhancing Contributes to the Organ Protective Effect of Alpha-Lipoic Acid in Septic Rats. Frontiers in Immunology . 2019;10, article 1491 doi: 10.3389/fimmu.2019.01491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yadav V., Marracci G. H., Munar M. Y., et al. Pharmacokinetic study of lipoic acid in multiple sclerosis: comparing mice and human pharmacokinetic parameters. Multiple Sclerosis Journal . 2010;16(4):387–397. doi: 10.1177/1352458509359722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hiltunen J. K., Autio K. J., Schonauer M. S., Kursu V. A. S., Dieckmann C. L., Kastaniotis A. J. Mitochondrial fatty acid synthesis and respiration. Biochimica et Biophysica Acta . 2010;1797(6-7):p. 78. doi: 10.1016/j.bbabio.2010.04.237. [DOI] [PubMed] [Google Scholar]
- 32.Ziegler D., Hanefeld M., Ruhnau K. J., et al. Treatment of symptomatic diabetic peripheral neuropathy with the anti-oxidant alpha-lipoic acid. A 3-week multicentre randomized controlled trial (ALADIN Study) Diabetologia . 1995;38(12):1425–1433. doi: 10.1007/BF00400603. [DOI] [PubMed] [Google Scholar]
- 33.Gorąca A., Huk-Kolega H., Piechota A., Kleniewska P., Ciejka E., Skibska B. Lipoic acid - biological activity and therapeutic potential. Pharmacological Reports . 2011;63(4):849–858. doi: 10.1016/S1734-1140(11)70600-4. [DOI] [PubMed] [Google Scholar]
- 34.Bustamante J., Lodge J. K., Marcocci L., Tritschler H. J., Packer L., Rihn B. H. α-Lipoic Acid in Liver Metabolism and Disease. Free Radical Biology and Medicine . 1998;24(6):1023–1039. doi: 10.1016/S0891-5849(97)00371-7. [DOI] [PubMed] [Google Scholar]
- 35.Hadzik B., Grass H., Mayatepek E., Daldrup T., Hoehn T. Fatal non-accidental alpha-lipoic acid intoxication in an adolescent girl. Klinische Pädiatrie . 2014;226(5):292–294. doi: 10.1055/s-0034-1372622. [DOI] [PubMed] [Google Scholar]
- 36.Çakatay U., Kayalı R., Sivas A., Tekeli F. Prooxidant activities of alpha-lipoic acid on oxidative protein damage in the aging rat heart muscle. Archives of Gerontology and Geriatrics . 2005;40(3):231–240. doi: 10.1016/j.archger.2004.09.001. [DOI] [PubMed] [Google Scholar]
- 37.Ziegler D., Hanefeld M., Ruhnau K. J., et al. Treatment of symptomatic diabetic polyneuropathy with the antioxidant alpha-lipoic acid: a 7-month multicenter randomized controlled trial (ALADIN III Study). ALADIN III Study Group. Alpha-Lipoic Acid in Diabetic Neuropathy. Diabetes Care . 1999;22(8):1296–1301. doi: 10.2337/diacare.22.8.1296. [DOI] [PubMed] [Google Scholar]
- 38.Packer L., Witt E. H., Tritschler H. J. Alpha-Lipoic acid as a biological antioxidant. Free Radical Biology and Medicine . 1995;19(2):227–250. doi: 10.1016/0891-5849(95)00017-R. [DOI] [PubMed] [Google Scholar]
- 39.Heusch G. Myocardial ischemia: lack of coronary blood flow or myocardial oxygen supply/demand imbalance? Circulation Research . 2016;119(2):194–196. doi: 10.1161/CIRCRESAHA.116.308925. [DOI] [PubMed] [Google Scholar]
- 40.Murphy E., Steenbergen C. Mechanisms underlying acute protection from cardiac ischemia-reperfusion injury. Physiological Reviews . 2008;88(2):581–609. doi: 10.1152/physrev.00024.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Garcia-Dorado D., Rodríguez-Sinovas A., Ruiz-Meana M., Inserte J. Proteccion contra el daño miocardico por isquemia-reperfusion en la practica clinica. Revista Española de Cardiología (English Edition) . 2014;67(5):394–404. doi: 10.1016/j.rec.2014.01.010. [DOI] [PubMed] [Google Scholar]
- 42.Mokhtari-Zaer A., Marefati N., Atkin S. L., Butler A. E., Sahebkar A. The protective role of curcumin in myocardial ischemia-reperfusion injury. Journal of Cellular Physiology . 2019;234(1):214–222. doi: 10.1002/jcp.26848. [DOI] [PubMed] [Google Scholar]
- 43.Heusch G., Libby P., Gersh B., et al. Cardiovascular remodelling in coronary artery disease and heart failure. The Lancet . 2014;383(9932):1933–1943. doi: 10.1016/S0140-6736(14)60107-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sadek H. A., Humphries K. M., Szweda P. A., Szweda L. I. Selective inactivation of redox-sensitive mitochondrial enzymes during cardiac reperfusion. Archives of Biochemistry and Biophysics . 2002;406(2):222–228. doi: 10.1016/S0003-9861(02)00446-0. [DOI] [PubMed] [Google Scholar]
- 45.Humphries K. M., Yoo Y., Szweda L. I. Inhibition of NADH-linked mitochondrial respiration by 4-hydroxy-2-nonenal. Biochemistry . 1998;37(2):552–557. doi: 10.1021/bi971958i. [DOI] [PubMed] [Google Scholar]
- 46.Ko K. M., Yiu H. Y. Schisandrin B modulates the ischemia-reperfusion induced changes in non-enzymatic antioxidant levels in isolated-perfused rat hearts. Molecular and Cellular Biochemistry . 2001;220(1/2):141–147. doi: 10.1023/A:1010979404447. [DOI] [PubMed] [Google Scholar]
- 47.Coombes J. S., Powers S. K., Hamilton K. L., et al. Improved cardiac performance after ischemia in aged rats supplemented with vitamin E and α-lipoic acid. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology . 2000;279(6):R2149–R2155. doi: 10.1152/ajpregu.2000.279.6.R2149. [DOI] [PubMed] [Google Scholar]
- 48.Haramaki N., Packer L., Assadnazari H., Zimmer G. Cardiac Recovery during Postischemic Reperfusion Is Improved by Combination of Vitamin E with Dihydrolipoic Acid. Biochemical and Biophysical Research Communications . 1993;196(3):1101–1107. doi: 10.1006/bbrc.1993.2364. [DOI] [PubMed] [Google Scholar]
- 49.Dudek M., Knutelska J., Bednarski M., et al. Alpha lipoic acid protects the heart against myocardial post ischemia- reperfusion arrhythmias _via_ KATP channel activation in isolated rat hearts. Pharmacological Reports . 2014;66(3):499–504. doi: 10.1016/j.pharep.2013.11.001. [DOI] [PubMed] [Google Scholar]
- 50.He L., Liu B., Dai Z., et al. Alpha lipoic acid protects heart against myocardial ischemia-reperfusion injury through a mechanism involving aldehyde dehydrogenase 2 activation. European Journal of Pharmacology . 2012;678(1-3):32–38. doi: 10.1016/j.ejphar.2011.12.042. [DOI] [PubMed] [Google Scholar]
- 51.Schonheit K., Gille L., Nohl H. Effect of α-lipoic acid and dihydrolipoic acid on ischemia/reperfusion injury of the heart and heart mitochondria. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease . 1995;1271(2-3):335–342. doi: 10.1016/0925-4439(95)00052-6. [DOI] [PubMed] [Google Scholar]
- 52.Ghibu S., Lauzier B., Delemasure S., et al. Antioxidant properties of alpha-lipoic acid: effects on red blood membrane permeability and adaptation of isolated rat heart to reversible ischemia. Mol Cell Biochem . 2009;320(1-2):141–148. doi: 10.1007/s11010-008-9916-0. [DOI] [PubMed] [Google Scholar]
- 53.Cao X., Chen A., Yang P., et al. Alpha-lipoic acid protects cardiomyocytes against hypoxia/reoxygenation injury by inhibiting autophagy. Biochemical and Biophysical Research Communications . 2013;441(4):935–940. doi: 10.1016/j.bbrc.2013.10.166. [DOI] [PubMed] [Google Scholar]
- 54.Deng C., Sun Z. C., Tong G., et al. α-Lipoic acid reduces infarct size and preserves cardiac function in rat myocardial ischemia/reperfusion injury through activation of PI3K/Akt/Nrf2 pathway. Plos One . 2013;8(3, article e58371) doi: 10.1371/journal.pone.0058371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Freisleben H. J. Lipoic acid reduces ischemia-reperfusion injury in animal models. Toxicology . 2000;148(2-3):159–171. doi: 10.1016/S0300-483X(00)00207-9. [DOI] [PubMed] [Google Scholar]
- 56.Freisleben H. J. Lipoate ameliorates ischemia-reperfusion in animal models. Clin Hemorheol Microcirc . 2000;23(2-4):219–224. [PubMed] [Google Scholar]
- 57.Broskova Z., Kyselova Z., Knezl V. Ischemia-reperfusion injury of the isolated diabetic rat heart: effect of the antioxidant stobadine. General physiology and biophysics . 2013;32(2):285–292. doi: 10.4149/gpb_2013028. [DOI] [PubMed] [Google Scholar]
- 58.Serbinova E., Reznick S. K., Packer L. Thioctic acid protects against ischemia-reperfusion injury in the isolated perfused Langendorff heart. Free Radical Research Communications . 1992;17(1):49–58. doi: 10.3109/10715769209061088. [DOI] [PubMed] [Google Scholar]
- 59.Lameire N. H., Bagga A., Cruz D., et al. Acute kidney injury: an increasing global concern. The Lancet . 2013;382(9887):170–179. doi: 10.1016/S0140-6736(13)60647-9. [DOI] [PubMed] [Google Scholar]
- 60.Zhao H., Alam A., Soo A. P., George A. J. T., Ma D. Ischemia-reperfusion injury reduces long term renal graft survival: mechanism and beyond. EBioMedicine . 2018;28(2):31–42. doi: 10.1016/j.ebiom.2018.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bonventre J. V., Yang L. Cellular pathophysiology of ischemic acute kidney injury. Journal of Clinical Investigation . 2011;121(11):4210–4221. doi: 10.1172/JCI45161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Farag M. M., Ahmed S. M., Elhadidy W. F., Rashad R. M. Superior protective effects of febuxostat plus alpha-lipoic acid on renal ischemia/reperfusion-induced hepatorenal injury in rats. Saudi Journal of Kidney Diseases and Transplantation . 2019;30(6):1364–1374. doi: 10.4103/1319-2442.275480. [DOI] [PubMed] [Google Scholar]
- 63.Şehirli Ö., Şener E., Çetinel S.¸., Yüksel M., Gedik N., Şener G. α-LIPOIC acid protects against renal ischaemia-reperfusion injury in rats. Clinical and Experimental Pharmacology and Physiology . 2008;35(3):249–255. doi: 10.1111/j.1440-1681.2007.04810.x. [DOI] [PubMed] [Google Scholar]
- 64.Ahmadvand H., Mahdavifard S. Protective effect of thioctic acid on renal ischemia-reperfusion injury in rat. International Journal of Preventive Medicine . 2019;10(1):p. 176. doi: 10.4103/ijpvm.IJPVM_396_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Cavdar Z., Ozbal S., Celik A., et al. The effects of alpha-lipoic acid on MMP-2 and MMP-9 activities in a rat renal ischemia and re-perfusion model. Biotechnic & Histochemistry . 2014;89(4):304–314. doi: 10.3109/10520295.2013.847498. [DOI] [PubMed] [Google Scholar]
- 66.Bae E. H., Lee K. S., Lee J., et al. Effects of α-lipoic acid on ischemia-reperfusion-induced renal dysfunction in rats. American Journal of Physiology-Renal Physiology . 2008;294(1):F272–F280. doi: 10.1152/ajprenal.00352.2007. [DOI] [PubMed] [Google Scholar]
- 67.Takaoka M., Ohkita M., Kobayashi Y., Yuba M., Matsumura Y. Protective effect of alpha-lipoic acid against ischaemic acute renal failure in rats. Clinical and Experimental Pharmacology & Physiology . 2002;29(3):189–194. doi: 10.1046/j.1440-1681.2002.03624.x. [DOI] [PubMed] [Google Scholar]
- 68.Douzinas E. E., Livaditi O., Tasoulis M. K., et al. Nitrosative and oxidative stresses contribute to post-ischemic liver injury following severe hemorrhagic shock: the role of hypoxemic resuscitation. Plos One . 2012;7(3, article e32968) doi: 10.1371/journal.pone.0032968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Arkadopoulos N., Defterevos G., Nastos C., et al. Development of a porcine model of post-hepatectomy liver failure. Journal of Surgical Research . 2011;170(2):e233–e242. doi: 10.1016/j.jss.2011.06.006. [DOI] [PubMed] [Google Scholar]
- 70.Jaeschke H. Molecular mechanisms of hepatic ischemia-reperfusion injury and preconditioning. American Journal of Physiology-Gastrointestinal and Liver Physiology . 2003;284(1):G15–G26. doi: 10.1152/ajpgi.00342.2002. [DOI] [PubMed] [Google Scholar]
- 71.Nastos C., Kalimeris K., Papoutsidakis N., et al. Global consequences of liver ischemia/reperfusion injury. Oxidative Medicine and Cellular Longevity . 2014;2014:13. doi: 10.1155/2014/906965.906965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Jiménez-Castro M. B., Cornide-Petronio M. E., Gracia-Sancho J., Peralta C. Inflammasome-mediated inflammation in liver ischemia-reperfusion injury. Cells . 2019;8(10):p. 1131. doi: 10.3390/cells8101131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Hirao H., Dery K. J., Kageyama S., Nakamura K., Kupiec-Weglinski J. W. Heme Oxygenase-1 in liver transplant ischemia-reperfusion injury: From bench- to-bedside. Free Radical Biology and Medicine . 2020;157:75–82. doi: 10.1016/j.freeradbiomed.2020.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Casciato P., Ambrosi N., Caro F. α-lipoic acid reduces postreperfusion syndrome in human liver transplantation - a pilot study. Transplant International . 2018;31(12):1357–1368. doi: 10.1111/tri.13314. [DOI] [PubMed] [Google Scholar]
- 75.Dunschede F., Erbes K., Kircher A. Reduction of ischemia reperfusion injury after liver resection and hepatic inflow occlusion by α-lipoic acid in humans. World Journal of Gastroenterology . 2006;12(42):6812–6817. doi: 10.3748/wjg.v12.i42.6812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Dulundu E., Ozel Y., Topaloglu U., et al. Alpha-lipoic acid protects against hepatic ischemia-reperfusion injury in rats. Pharmacology . 2007;79(3):163–170. doi: 10.1159/000098953. [DOI] [PubMed] [Google Scholar]
- 77.REN Y., WANG L. H., DENG F. S., LI J. S., JIANG L. Protective effect and mechanism of α-Lipoic acid on partial hepatic ischemia-reperfusion injury in adult male rats. Physiological Research . 2019;68(5):739–745. doi: 10.33549/physiolres.934095. [DOI] [PubMed] [Google Scholar]
- 78.Müller C., Dünschede F., Koch E., Vollmar A. M., Kiemer A. K. α-Lipoic acid preconditioning reduces ischemia-reperfusion injury of the rat liver via the PI3-kinase/Akt pathway. American Journal of Physiology. Gastrointestinal and Liver Physiology . 2003;285(4):G769–G778. doi: 10.1152/ajpgi.00009.2003. [DOI] [PubMed] [Google Scholar]
- 79.Duenschede F., Erbes K., Riegler N. Protective effects of ischemic preconditioning and application of lipoic acid prior to 90 min of hepatic ischemia in a rat model. World Journal of Gastroenterology . 2007;13(27):3692–3698. doi: 10.3748/wjg.v13.i27.3692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Duenschede F., Erbes K., Kircher A., et al. Protection from hepatic ischemia/reperfusion injury and improvement of liver regeneration by ??-LIPOIC acid. Shock . 2007;27(6):644–651. doi: 10.1097/01.shk.0000248582.25647.ee. [DOI] [PubMed] [Google Scholar]
- 81.Duenschede F., Westermann S., Riegler N., et al. Different protection mechanisms after pretreatment with glycine or α-Lipoic acid in a rat model of warm hepatic ischemia. European Surgical Research . 2006;38(6):503–512. doi: 10.1159/000096061. [DOI] [PubMed] [Google Scholar]
- 82.Sanderson T. H., Reynolds C. A., Kumar R., Przyklenk K., Hüttemann M. Molecular mechanisms of ischemia-reperfusion injury in brain: pivotal role of the mitochondrial membrane potential in reactive oxygen species generation. Molecular Neurobiology . 2013;47(1):9–23. doi: 10.1007/s12035-012-8344-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Jenkins L. W., Povlishock J. T., Lewelt W., Miller J. D., Becker D. P. The role of postischemic recirculation in the development of ischemic neuronal injury following complete cerebral ischemia. Acta Neuropathologica . 1981;55(3):205–220. doi: 10.1007/BF00691320. [DOI] [PubMed] [Google Scholar]
- 84.Sie L. T., van der Knaap M. S., Oosting J., de Vries L. S., Lafeber H. N., Valk J. MR patterns of hypoxic-ischemic brain damage after prenatal, perinatal or postnatal asphyxia. Neuropediatrics . 2000;31(3):128–136. doi: 10.1055/s-2000-7496. [DOI] [PubMed] [Google Scholar]
- 85.Kleikers P. W. M., Wingler K., Hermans J. J. R., et al. NADPH oxidases as a source of oxidative stress and molecular target in ischemia/reperfusion injury. Journal of Molecular Medicine . 2012;90(12):1391–1406. doi: 10.1007/s00109-012-0963-3. [DOI] [PubMed] [Google Scholar]
- 86.Liao S., Apaijai N., Chattipakorn N., Chattipakorn S. C. The possible roles of necroptosis during cerebral ischemia and ischemia / reperfusion injury. Archives of Biochemistry and Biophysics . 2020;695(11) doi: 10.1016/j.abb.2020.108629. [DOI] [PubMed] [Google Scholar]
- 87.Malinska D., Winiarska K. Lipoic acid: characteristics and therapeutic application. Postepy higieny i medycyny doswiadczalnej . 2005;59:535–543. [PubMed] [Google Scholar]
- 88.Packer L., Tritschler H. J., Wessel K. Neuroprotection by the Metabolic Antioxidant _α_ -Lipoic Acid. Free Radical Biology and Medicine . 1997;22(1-2):359–378. doi: 10.1016/S0891-5849(96)00269-9. [DOI] [PubMed] [Google Scholar]
- 89.Shmonin A. A., Mel'nikova E. V., Churilova I. V., Vlasov T. D. Neuroprotective effects of lipoic acid and superoxide dismutase in the rat model of cerebral ischemia. Rossiiskii fiziologicheskii zhurnal imeni IM Sechenova . 2011;97(1):65–71. [PubMed] [Google Scholar]
- 90.Gonzalez-Perez O., Gonzalez-Castañeda R. E., Huerta M., et al. Beneficial effects of α-lipoic acid plus vitamin E on neurological deficit, reactive gliosis and neuronal remodeling in the penumbra of the ischemic rat brain. Neuroscience Letters . 2002;321(1-2):100–104. doi: 10.1016/S0304-3940(02)00056-3. [DOI] [PubMed] [Google Scholar]
- 91.Röhnert P., Schröder U. H., Ziabreva I., Täger M., Reymann K. G., Striggow F. Insufficient endogenous redox buffer capacity may underlie neuronal vulnerability to cerebral ischemia and reperfusion. Journal of Neuroscience Research . 2012;90(1):193–202. doi: 10.1002/jnr.22754. [DOI] [PubMed] [Google Scholar]
- 92.Connell B. J., Saleh T. M. Co-administration of apocynin with lipoic acid enhances neuroprotection in a rat model of ischemia/reperfusion. Neuroscience Letters . 2012;507(1):43–46. doi: 10.1016/j.neulet.2011.11.047. [DOI] [PubMed] [Google Scholar]
- 93.Saleh M. C., Connell B. J., Rajagopal D., et al. Co-administration of resveratrol and lipoic acid, or their synthetic combination, enhances neuroprotection in a rat model of ischemia/reperfusion. Plos One . 2014;9(1, article e87865) doi: 10.1371/journal.pone.0087865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Deng H., Zuo X., Zhang J., et al. α-lipoic acid protects against cerebral ischemia/reperfusion-induced injury in rats. Molecular Medicine Reports . 2015;11(5):3659–3665. doi: 10.3892/mmr.2015.3170. [DOI] [PubMed] [Google Scholar]
- 95.Zhang J., Xiao F., Zhang L., et al. Alpha-lipoic acid preconditioning and ischaemic postconditioning synergistically protect rats from cerebral injury induced by ischemia and reperfusion partly via inhibition TLR4/MyD88/ NF-κB signaling pathway. Cellular Physiology and Biochemistry . 2018;51(3):1448–1460. doi: 10.1159/000495593. [DOI] [PubMed] [Google Scholar]
- 96.Choi K. H., Park M. S., Kim H. S., et al. Alpha-lipoic acid treatment is neurorestorative and promotes functional recovery after stroke in rats. Molecular Brain . 2015;8(1) doi: 10.1186/s13041-015-0101-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Lv C., Maharjan S., Wang Q., et al. α-Lipoic acid promotes neurological recovery after ischemic stroke by activating the Nrf2/HO-1 pathway to attenuate oxidative damage. Cellular Physiology and Biochemistry . 2017;43(3):1273–1287. doi: 10.1159/000481840. [DOI] [PubMed] [Google Scholar]
- 98.Connell B. J., Saleh M., Khan B. V., Saleh T. M. Lipoic acid protects against reperfusion injury in the early stages of cerebral ischemia. Brain Research . 2011;1375(2):128–136. doi: 10.1016/j.brainres.2010.12.045. [DOI] [PubMed] [Google Scholar]
- 99.Gao X. J., Chen W., Li J. Q., et al. The protective effect of alpha-lipoic acid against brain ischemia and reperfusion injury via mTOR signaling pathway in rats. Neuroscience Letters . 2018;671(4):108–113. doi: 10.1016/j.neulet.2018.02.012. [DOI] [PubMed] [Google Scholar]
- 100.Hussein O. A., Abdel-Hafez A. M. M., Abd El Kareim A. Rat hippocampal CA3 neuronal injury induced by limb ischemia/reperfusion: a possible restorative effect of alpha lipoic acid. Ultrastructural Pathology . 2018;42(2):133–154. doi: 10.1080/01913123.2018.1427165. [DOI] [PubMed] [Google Scholar]
- 101.Xie R., Li X. M., Ling Y., et al. Alpha-lipoic acid pre- and post-treatments provide protection against _in vitro_ ischemia-reperfusion injury in cerebral endothelial cells via Akt/mTOR signaling. Brain Research . 2012;1482(10):81–90. doi: 10.1016/j.brainres.2012.09.009. [DOI] [PubMed] [Google Scholar]
- 102.Panigrahi M., Sadguna Y., Shivakumar B. R., et al. α-Lipoic acid protects against reperfusion injury following cerebral ischemia in rats. Brain Research . 1996;717(1-2):184–188. doi: 10.1016/0006-8993(96)00009-1. [DOI] [PubMed] [Google Scholar]
- 103.Cao X., Phillis J. W. The free radical scavenger, α-Lipoic acid, protects against cerebral ischemia-reperfusion injury in gerbils. Free Radical Research . 1995;23(4):365–370. doi: 10.3109/10715769509065257. [DOI] [PubMed] [Google Scholar]
- 104.Clark W. M., Rinker L. G., Lessov N. S., Lowery S. L., Cipolla M. J. Efficacy of antioxidant therapies in transient focal ischemia in mice. Stroke . 2001;32(4):1000–1004. doi: 10.1161/01.STR.32.4.1000. [DOI] [PubMed] [Google Scholar]
- 105.Emmez H., Yildirim Z., Kale A., et al. Anti-apoptotic and neuroprotective effects of alpha-lipoic acid on spinal cord ischemia-reperfusion injury in rabbits. Acta Neurochirurgica . 2010;152(9):1591–1601. doi: 10.1007/s00701-010-0703-9. [DOI] [PubMed] [Google Scholar]
- 106.Kumbasar U., Demirci H., Emmez G. Protection from spinal cord ischemia-reperfusion damage with alpha-lipoic acid preconditioning in an animal model. The Turkish Journal of Thoracic and Cardiovascular Surgery . 2018;26(1):138–145. doi: 10.5606/tgkdc.dergisi.2018.14432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Shaafi S., Afrooz M. R., Hajipour B., Dadadshi A., Hosseinian M. M., Khodadadi A. Anti-oxidative effect of lipoic acid in spinal cord ischemia/reperfusion. Medical Principles and Practice . 2011;20(1):19–22. doi: 10.1159/000319772. [DOI] [PubMed] [Google Scholar]
- 108.Boyaci M. G., Eser O., Kocogullari C. U., Karavelioglu E., Tokyol C., Can Y. Neuroprotective effect of alpha-lipoic acid and methylprednisolone on the spinal cord ischemia/reperfusion injury in rabbits. British Journal of Neurosurgery . 2015;29(1):46–51. doi: 10.3109/02688697.2014.954986. [DOI] [PubMed] [Google Scholar]
- 109.Coselli J. S., LeMaire S. A., Conklin L. D., Köksoy C., Schmittling Z. C. Morbidity and mortality after extent II thoracoabdominal aortic aneurysm repair. The Annals of Thoracic Surgery . 2002;73(4):1107–1116. doi: 10.1016/S0003-4975(02)03370-2. [DOI] [PubMed] [Google Scholar]
- 110.Miranda V., Sousa J., Mansilha A. Spinal cord injury in endovascular thoracoabdominal aortic aneurysm repair: prevalence, risk factors and preventive strategies. International Angiology . 2018;37(2):112–126. doi: 10.23736/S0392-9590.18.03960-3. [DOI] [PubMed] [Google Scholar]
- 111.Kardes O., Civi S., Tufan K., Oyar E. O., Omeroglu S., Aykol S. Effects of atorvastatin on experimental spinal cord ischemia-reperfusion injury in rabbits. Turkish Neurosurgery . 2016;27(4):594–602. doi: 10.5137/1019-5149.JTN.16627-15.2. [DOI] [PubMed] [Google Scholar]
- 112.Hentia C., Rizzato A., Camporesi E., et al. An overview of protective strategies against ischemia/reperfusion injury: the role of hyperbaric oxygen preconditioning. Brain and Behavior . 2018;8(5, article e00959) doi: 10.1002/brb3.959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Karimipour M., Farjah G. H., Molazadeh F., Khadem Ansari M. H., Pourheidar B. Protective effect of contralateral, ipsilateral, and bilateral remote ischemic preconditioning on spinal cord ischemia reperfusion injury in rats. Turkish Neurosurgery . 2019;29(6):933–939. doi: 10.5137/1019-5149.JTN.26237-19.3. [DOI] [PubMed] [Google Scholar]
- 114.Faheem H., Mansour A., Elkordy A., et al. Neuroprotective effects of minocycline and progesterone on white matter injury after focal cerebral ischemia. Journal of Clinical Neuroscience . 2019;64(6):206–213. doi: 10.1016/j.jocn.2019.04.012. [DOI] [PubMed] [Google Scholar]
- 115.Jia X. F., Kowalski R. G., Sciubba D. M., Geocadin R. G. Critical care of traumatic spinal cord injury. Journal of Intensive Care Medicine . 2013;28(1):12–23. doi: 10.1177/0885066611403270. [DOI] [PubMed] [Google Scholar]
- 116.Sasaki M., Joh T. Oxidative stress and ischemia-reperfusion injury in gastrointestinal tract and antioxidant, protective agents. Journal of Clinical Biochemistry and Nutrition . 2007;40(1):1–12. doi: 10.3164/jcbn.40.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Matsuda A., Yang W. L., Jacob A., et al. FK866, a visfatin inhibitor, protects against acute lung injury after intestinal ischemia-reperfusion in mice via NF-κB pathway. Annals of Surgery . 2014;259(5):1007–1017. doi: 10.1097/SLA.0000000000000329. [DOI] [PubMed] [Google Scholar]
- 118.Mallick I. H., Yang W. X., Winslet M. C., Seifalian A. M. REVIEW: Ischemia-reperfusion injury of the intestine and protective strategies against injury. Digestive Diseases and Sciences . 2004;49(9):1359–1377. doi: 10.1023/B:DDAS.0000042232.98927.91. [DOI] [PubMed] [Google Scholar]
- 119.Yuan Y., Guo H., Zhang Y. Protective effects of L-carnitine on intestinal ischemia/reperfusion injury in a rat model. Journal of Clinical Medical Research . 2011;3(2):78–84. doi: 10.4021/jocmr540w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Wang B., Huang Q., Zhang W., Li N., Li J. Lactobacillus plantarum prevents bacterial translocation in rats following ischemia and reperfusion injury. Digestive Diseases and Sciences . 2011;56(11):3187–3194. doi: 10.1007/s10620-011-1747-2. [DOI] [PubMed] [Google Scholar]
- 121.Slone E. A., Fleming S. D. Membrane lipid interactions in intestinal ischemia/reperfusion-induced Injury. Clinical Immunology . 2014;153(1):228–240. doi: 10.1016/j.clim.2014.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Guven A., Tunc T., Topal T., et al. α-Lipoic acid and ebselen prevent ischemia/reperfusion injury in the rat intestine. Surgery Today . 2008;38(11):1029–1035. doi: 10.1007/s00595-007-3752-9. [DOI] [PubMed] [Google Scholar]
- 123.Tunc T., Öter S., Güven A., et al. Protective effect of sulfhydryl-containing antioxidants against ischemia/reperfusion injury of prepubertal rat intestine. Journal of Gastroenterology and Hepatology . 2009;24(4):681–687. doi: 10.1111/j.1440-1746.2008.05673.x. [DOI] [PubMed] [Google Scholar]
- 124.Roldan E. J., Kerzberg E. M., Boveris A. Kinetics of chemoluminiscence of rat intestine during ischemia and reperfusion. Medicina . 1989;49(4):336–340. [PubMed] [Google Scholar]
- 125.Lukasiewicz A. Treatment of acute lower limb ischaemia. VASA . 2016;45(3):213–221. doi: 10.1024/0301-1526/a000527. [DOI] [PubMed] [Google Scholar]
- 126.Khan S., Hawkins B. M. Acute limb ischemia interventions. Interventional Cardiology Clinics . 2020;9(2):221–228. doi: 10.1016/j.iccl.2019.12.002. [DOI] [PubMed] [Google Scholar]
- 127.Simon F., Oberhuber A., Floros N., et al. Acute limb Ischemia—Much more than just a lack of oxygen. International Journal of Molecular Sciences . 2018;19(2):p. 374. doi: 10.3390/ijms19020374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Volchegorskii I. A., Alekseev M. N., Volchegorskaia M. I., Rassokhina L. M. Effect of alpha-lipoic acid and mexidol on neuro- and the affective status in patients at early stages of diabetic foot syndrome. Klinicheskaia meditsina . 2008;86(10):52–59. [PubMed] [Google Scholar]
- 129.Piddubna O. O., Lytvynenko N. V., Krivchun A. M., Taryanyk K. A. The effect of Α-LIPOIC acid on the functional state of peripheral nerve fibers in polyneuropathy of the lower extremities in patients with chronic hemoblastosis. Wiadomości Lekarskie . 2020;73(9):1990–1994. doi: 10.36740/WLek202009217. [DOI] [PubMed] [Google Scholar]
- 130.Aydin A., Yildirim A. M. Effects of alpha lipoic acid on ishemia/reperfusion injury in rat hindlimb ischemia model. Ulusal Travma ve Acil Cerrahi Dergisi . 2016;22(6):509–515. doi: 10.5505/tjtes.2016.00258. [DOI] [PubMed] [Google Scholar]
- 131.Mitsui Y., Schmelzer J. D., Zollman P. J., Mitsui M., Tritschler H. J., Low P. A. Alpha-lipoic acid provides neuroprotection from ischemia-reperfusion injury of peripheral nerve. Journal of the Neurological Sciences . 1999;163(1):11–16. doi: 10.1016/S0022-510X(99)00017-9. [DOI] [PubMed] [Google Scholar]
- 132.Altaffer L. F., 3rd Testicular torsion in men. The Journal of Urology . 1980;123(1):37–38. doi: 10.1016/S0022-5347(17)55769-9. [DOI] [PubMed] [Google Scholar]
- 133.Martin C. M. K., Magee K. ovarian torsion in a 20-year-old patient. Canadian Journal of Emergency Medicine . 2006;8(2):126–129. doi: 10.1017/s1481803500013622. [DOI] [PubMed] [Google Scholar]
- 134.Turner T. T., Bang H. J., Lysiak J. L. The molecular pathology of experimental testicular torsion suggests adjunct therapy to surgical repair. The Journal of Urology . 2004;172, 6 Part 2:2574–2578. doi: 10.1097/01.ju.0000144203.30718.19. [DOI] [PubMed] [Google Scholar]
- 135.Yildirim N., Simsek D., Kose S., et al. The protective effect of Gingko biloba in a rat model of ovarian ischemia/reperfusion injury: improvement in histological and biochemical parameters. Advances in Clinical and Experimental Medicine . 2018;27(5):591–597. doi: 10.17219/acem/68896. [DOI] [PubMed] [Google Scholar]
- 136.Krarup T. The testes after torsion. British Journal of Urology . 1978;50(1):43–46. doi: 10.1111/j.1464-410X.1978.tb02764.x. [DOI] [PubMed] [Google Scholar]
- 137.Refaie M. M. M., El-Hussieny M. Protective effect of pioglitazone on ovarian ischemia reperfusion injury of female rats via modulation of peroxisome proliferator activated receptor gamma and heme-oxygenase 1. International Immunopharmacology . 2018;62(9):7–14. doi: 10.1016/j.intimp.2018.06.037. [DOI] [PubMed] [Google Scholar]
- 138.Guimarães S. B., Santos J. M. V., Aragão A. A., de Sandes Kimura O., Barbosa P. H. U., de Vasconcelos P. R. L. Protective effect of α-lipoic acid in experimental spermatic cord torsion. Nutrition . 2007;23(1):76–80. doi: 10.1016/j.nut.2006.09.005. [DOI] [PubMed] [Google Scholar]
- 139.Ozbal S., Ergur B. U., Erbil G., Tekmen I., Bagrıyanık A., Cavdar Z. The effects of alpha-lipoic acid against testicular ischemia-reperfusion injury in rats. ScientificWorld Journal . 2012;2012, article 489248:1–8. doi: 10.1100/2012/489248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Cosar E., Sahin F. K., Koken G., Toy H., Basarali K., Buyukbas S. The protective effect of α-lipoic acid in experimental ovarian ischaemia-reperfusion injury. Australian and New Zealand Journal of Obstetrics and Gynaecology . 2007;47(6):499–503. doi: 10.1111/j.1479-828X.2007.00787.x. [DOI] [PubMed] [Google Scholar]
- 141.Dokuyucu R., Karateke A., Gokce H., et al. Antioxidant effect of erdosteine and lipoic acid in ovarian ischemia- reperfusion injury. European Journal of Obstetrics & Gynecology and Reproductive Biology . 2014;183:23–27. doi: 10.1016/j.ejogrb.2014.10.018. [DOI] [PubMed] [Google Scholar]
- 142.Tuncer A. A., Bozkurt M. F., Koken T., Dogan N., Pektaş M. K., Embleton D. B. The protective effects of alpha-lipoic acid and coenzyme Q10 combination on ovarian ischemia-reperfusion injury: an experimental study. Advances in Medicine . 2016;2016:8. doi: 10.1155/2016/3415046.3415046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Osborne N. N., Casson R. J., Wood J. P. M., Chidlow G., Graham M., Melena J. Retinal ischemia: mechanisms of damage and potential therapeutic strategies. Progress in Retinal and Eye Research . 2004;23(1):91–147. doi: 10.1016/j.preteyeres.2003.12.001. [DOI] [PubMed] [Google Scholar]
- 144.Cao X., Li W., Liu Y., Huang H., Ye C.-H. The anti-inflammatory effects of CXCR5 in the mice retina following ischemia-reperfusion injury. BioMed Research International . 2019;2019:10. doi: 10.1155/2019/3487607.3487607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Nakahara T., Hoshino M., Hoshino S., Mori A., Sakamoto K., Ishii K. Structural and functional changes in retinal vasculature induced by retinal ischemia-reperfusion in rats. Experimental Eye Research . 2015;135:134–145. doi: 10.1016/j.exer.2015.02.020. [DOI] [PubMed] [Google Scholar]
- 146.Inman D. M., Lambert W. S., Calkins D. J., Horner P. J. α-Lipoic acid antioxidant treatment limits glaucoma-related retinal ganglion cell death and dysfunction. Plos One . 2013;8(6, article e65389) doi: 10.1371/journal.pone.0065389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Kim Y. S., Kim M., Choi M. Y., et al. Alpha-lipoic acid reduces retinal cell death in diabetic mice. Biochemical and Biophysical Research Communications . 2018;503(3):1307–1314. doi: 10.1016/j.bbrc.2018.07.041. [DOI] [PubMed] [Google Scholar]
- 148.Chidlow G., Schmidt K. G., Wood J. P. M., Melena J., Osborne N. N. _α_ -lipoic acid protects the retina against ischemia-reperfusion. Neuropharmacology . 2002;43(6):1015–1025. doi: 10.1016/S0028-3908(02)00129-6. [DOI] [PubMed] [Google Scholar]
- 149.Modgil S., Bammidi S., Banik A., Sharma V. L., Anand A. Alpha lipoic acid attenuates the long-term effects of lead exposure in retinal ischemic injury mouse model. Environmental Science and Pollution Research . 2019;26(15):15548–15558. doi: 10.1007/s11356-019-04700-6. [DOI] [PubMed] [Google Scholar]
- 150.Cowled P., Fitridge R. Pathophysiology of reperfusion injury. In: Fitridge R., Thompson M., editors. Mechanisms of Vascular Disease: A Reference Book for Vascular Specialists . Adelaide (AU): Technical University of Denmark National Food Institute; 2011. [Google Scholar]
- 151. https://ec.europa.eu/food/sites/food/files/safety/docs/labelling-nutrition_vitamins-minerals_add_alpha-lipoic-acid_nfi-da.pdf .
- 152.Ryeal Hahm J., Kim B. J., Kim K. W. Clinical experience with thioctacid (thioctic acid) in the treatment of distal symmetric polyneuropathy in Korean diabetic patients. Journal of Diabetes and its Complications . 2004;18(2):79–85. doi: 10.1016/S1056-8727(03)00033-3. [DOI] [PubMed] [Google Scholar]
- 153.Han T. T., Bai J. F., Liu W., Hu Y. THERAPY OF ENDOCRINE DISEASE: A systematic review and meta-analysis of α-lipoic acid in the treatment of diabetic peripheral neuropathy. European Journal of Endocrinology . 2012;167(4):465–471. doi: 10.1530/EJE-12-0555. [DOI] [PubMed] [Google Scholar]
- 154.Porasuphatana S., Suddee S., Nartnampong A., Konsil J., Harnwong B., Santaweesuk A. Glycemic and oxidative status of patients with type 2 diabetes mellitus following oral administration of alpha-lipoic acid: a randomized double-blinded placebo-controlled study. Asia Pacific Journal of Clinical Nutrition . 2012;21(1):12–21. [PubMed] [Google Scholar]


