Abstract
Background
Recent studies, predominantly in Western populations, suggest that both weight loss and weight gain are associated with an increased mortality risk in old age. However, evidence of this association in older Asian populations remains sparse. This study aimed to examine the association between weight change and all-cause mortality in a nationally representative sample of community-dwelling older Japanese people.
Methods
Data were obtained from the National Survey of the Japanese Elderly, which included 4869 adults aged ≥60 years. Participants were followed for up to 30 years. We considered 3 indicators of weight change according to the follow-up interval: short-term (3 years), medium-term (6–7 years), and long-term (12–13 years). Weight change was classified as loss ≥ 5%, loss 2.5%–4.9%, stable (±2.4%), gain 2.5%–4.9%, and gain ≥ 5%. Cox proportional hazards models were used to calculate the relative mortality risk of each weight change category.
Results
Weight loss ≥ 5% for all intervals was associated with higher mortality than stable weight and the effects were largely similar across all 3 intervals (hazard ratio [95% confidence interval]: 1.36 [1.22–1.51] for short-term, 1.36 [1.22–1.51] for medium-term, and 1.31 [1.11–1.54] for long-term). A similar pattern of results was observed among the young–old and old–old, and among men and women. The effect of weight loss on higher mortality was greater among those with a lower body mass index at baseline.
Conclusions
These findings could inform clinical and public health approaches to body-weight management aimed at improving the health and survival of older adults, particularly in Asian populations.
Keywords: Epidemiology, Japan, Longevity, Obesity
Weight change over the course of multiple years (approximately 2–13 years) is a good predictor of mortality risk in middle and old age (1–15) and has been shown to predict mortality better than a single weight assessment, particularly in old age (16). However, previous findings on the association between weight change (including both weight loss and weight gain) and mortality have been inconsistent. Moreover, the effect of weight change on mortality can be modified by several factors such as health status, intentionality to change body weight, and baseline body mass index (BMI) (17).
To date, there have been 2 meta-analyses on the association between weight change and all-cause mortality in later life (18,19). Both studies highlight the potential adverse effect of weight change, particularly weight loss, on all-cause mortality in later life. For example, a meta-analysis by Alharbi et al reported that weight loss and weight gain were associated with a 59% and 10% increase in mortality risk, respectively (18).
In addition, the other meta-analysis by Cheng et al concluded that there was no difference in the effect of weight change on mortality between Western and Asian nations (19). However, they included only 2 studies from Asian countries (4,8). Alharbi et al included only 4 studies from Asian nations (4,5,10,14). This is problematic because the distribution of BMI in Western societies is different from that in Asian countries (20). Moreover, another previous study using data from the Multiethnic Cohort Study showed heterogeneity in the impact of weight change on mortality across race/ethnicity (13). Thus, further investigations among older people in Asian nations are warranted to examine whether the association between weight change and mortality in Asian populations differs from that observed in Western populations.
The existing research can be improved in at least 2 ways. First, previous studies defined weight change measured at just 2 points in time, with some studies using a relatively short interval such as 2 or 3 years, and some using a longer interval such as 12 or 13 years. Because the length of the interval over which weight change is assessed varies across different studies, it is difficult to evaluate whether the effect of weight change on mortality risk differs according to the interval. An evaluation of the influence of weight change on mortality according to the interval of change could provide notable implications for body-weight management in later life. Second, most earlier studies, particularly those from Asian countries, followed up the participants for mortality for less than 10 years. A longer mortality follow-up interval would likely capture a cumulative effect of weight change on mortality and encompass greater fluctuations in macrosocial and economic circumstances at the individual level in later life.
To address these gaps, we used up to 30 years of longitudinal data from a nationally representative sample of community-dwelling Japanese people aged ≥60 years, and aimed to examine the association between weight change, including both loss and gain, and all-cause mortality during short- (3 years), medium- (6–7 years), and long-term (12–13 years) intervals. Based on prior research, we hypothesized that weight change during these 3 intervals would be associated with mortality risk.
Materials and Methods
Study Population
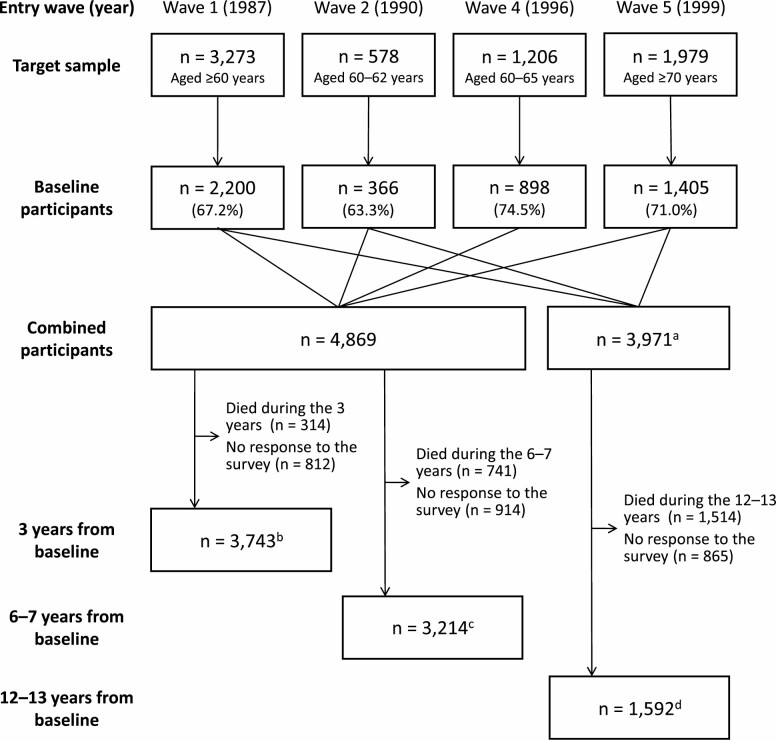
We obtained data from the National Survey of the Japanese Elderly. The National Survey of the Japanese Elderly originally began in 1987 with a cohort of 2200 people aged ≥60 years (Wave 1). Subsequently, data collection was conducted in 1990, 1993, 1996, 1999, 2002, 2006, 2012, and 2017. The new sample was supplemented in 1990 (Wave 2; 366 people aged 60–62 years), 1996 (Wave 4; 898 people aged 60–65 years), 1999 (Wave 5; 1405 people aged ≥70 years), and 2012 (Wave 8; 1324 people aged 60–92 years). Response rates for each wave ranged from 67% to 93%. The National Survey of the Japanese Elderly data were collected through face-to-face home visits by trained interviewers using a structured questionnaire that was consistent over time. The current study used data from people who participated in the National Survey of the Japanese Elderly in 1987, 1990, 1996, and 1999 (n = 4869). We excluded proxy interviews because body weight was not included in the questionnaire for proxy interviews. The number of participants who completed both the baseline and follow-up surveys was 3743 for the short-term (3 years from baseline), 3214 for the medium-term (6–7 years from baseline), and 1592 for the long-term (12–13 years from baseline). Figure 1 shows a flow diagram of study participation, follow-up, and attrition. The study protocol was approved by the Institutional Review Board of the Tokyo Metropolitan Institute of Gerontology and the University of Michigan.
Figure 1.
Flow diagram of study participants of the National Survey of Japanese Elderly. a: We excluded the cohort surveyed in 1996 (Wave 4; n = 898), because they had no corresponding survey to calculate long-term weight change. b: The number of participants who responded to both the baseline and 3-year follow-up surveys. c: The number of participants who responded to both the baseline and 6- or 7-year follow-up surveys. d: The number of participants who responded to both the baseline and 12- or 13-year follow-up surveys.
Mortality and Follow-up
All-cause mortality was recorded until December 31, 2017. Dates of death or dropout of the participants from the study were collected by reviewing residence certificates with the permission of the local municipalities where the participants lived, or by reports from the families of the participants. Overall, 3302 of 4869 participants (67.8%) died during the follow-up period. Those who had refused the survey after the baseline (ie, dropout from the study) were censored because we were unable to ascertain their death date (8.1%; n = 397); the proportion of women was higher and they were younger compared to those who were not censored (68.3% vs 54.2%; 67.0 years vs 70.1 years). The follow-up period was up to 30 years (15.5 years per participant on average).
Weight Change
We used 3 weight change indicators according to the interval over which the change was assessed: short-term (3 years), medium-term (6–7 years), and long-term (12–13 years). The percentage of weight change between the baseline and follow-up surveys, based on the baseline body weight, was calculated. Because this study included 4 cohorts of participants (ie, entry years of 1987, 1990, 1996, and 1999), each cohort had different corresponding years to calculate short-term weight change (ie, 1987–1990, 1990–1993, 1996–1999, and 1999–2002), medium-term weight change (ie, 1987–1993, 1990–1996, 1996–2002, and 1999–2006), and long-term weight change (ie, 1987–1999, 1990–2002, and 1999–2012; we excluded the cohort surveyed in 1996 because they had no corresponding survey). Body weight was self-reported from 1987 to 2006 and actually measured in 2012. We classified the percentage of weight change into 5 categories: loss ≥ 5%, loss 2.5%–4.9%, stable (±2.4%), gain 2.5%–4.9%, and gain ≥ 5%.
Covariates
Age, sex, marital status (married or not married), current working status (working or not working), education (years of schooling), annual household income, health behaviors, history of chronic diseases at baseline, and entry wave (Wave 1, 2, 4, or 5) were incorporated as covariates in the present study. Annual household income was classified into 5 categories: low (<1.2 million yen), middle–low (1.2–2.9 million yen), middle (3.0–4.9 million yen), middle–high (5.0–9.9 million yen), or high (≥10.0 million yen). The average annual income of aged households was approximately 3.3 million yen in 2017 (21). Health behaviors included the number of cigarettes smoked per day (0, 1–19, or ≥20), the number of drinking days per month (0, 1–15, 16–25, or ≥26), and the frequency of exercise (often, sometimes, rarely, or none). Chronic diseases, including cancer, heart disease, stroke, lung disease, liver disease, and kidney disease, were reported by the respondents. In addition, baseline BMI, calculated from self-reported height and weight (weight/height2 [kg/m2]), was used as a covariate to account for possible variations due to the starting body size of each participant.
Statistical Analyses
Cox proportional hazards models were used to calculate the relative mortality risk of each weight change category, with the “stable weight” category as the reference. All covariates were controlled for in the models. To control for possible effects of precipitated weight loss before death on baseline BMI, we undertook a subgroup analysis excluding people whose death occurred within a year after the follow-up survey. Furthermore, to rule out the confounding effects of smoking, physical activity, and chronic diseases on the relationship between weight change and mortality, we conducted parallel analyses of selected subgroups: noncurrent smokers; physically active people (those who responded “often/sometimes” to the item regarding the frequency of exercise); and those without a history of cancer, heart disease, stroke, lung disease, liver disease, or kidney disease. Moreover, we performed stratified analyses by baseline age (aged 60–74 and ≥75 years) and sex. Finally, because the association between weight change and mortality could vary according to BMI, we further divided the weight change categories according to baseline BMI (underweight [<18.5 kg/m2], normal weight [18.5–24.9 kg/m2], and overweight/obese [≥25.0 kg/m2]; a total of 15 categories were generated), and set “stable at normal weight” as the reference. The results were expressed as hazard ratios (HRs) with 95% confidence intervals (CIs). The analyses were performed using IBM SPSS 23 (IBM Corp., Armonk, NY).
In addition to the relative mortality risk of each weight change category, we fit a smoothing spline curve of the adjusted HR for mortality by weight change to examine the association using weight change as a continuous variable. This analysis was performed with Stata 14 (StataCorp, College Station, TX).
To minimize the potential bias associated with item nonresponse, multiple imputation was undertaken using an inclusive strategy with not only variables included in our analytical models but also auxiliary variables not included in our analysis (22). Examples of auxiliary variables included social support, depressive symptoms, and follow-up measures of the predictors in the Cox proportional hazards models, which were measured at the baseline. In particular, 3 complete data sets were imputed using the NORM software (23), and analyses were run on each of these 3 data sets with parameter estimates derived by averaging across 3 imputations and by adjusting for their variance. The imputation procedure was performed for the surviving participants temporally by removing participants after death. The descriptive statistics and Cox proportional hazards models were conducted using the imputed data sets.
Results
The characteristics of the study participants are shown in Table 1. At baseline, the mean age was 69.9 years (standard deviation [SD] = 7.2), and 55.3% of the sample was female. The mean body weight was 53.6 kg (SD = 9.3), and the mean BMI was 22.3 kg/m2 (SD = 3.2). The distribution of BMI was 10.7% for underweight (<18.5 kg/m2), 71.4% for normal weight (18.5–24.9 kg/m2), 16.4% for overweight (25.0–29.9 kg/m2), and 1.4% for obesity (≥30.0 kg/m2). Moreover, the average BMI by age and sex was 22.1 kg/m2 (SD = 2.9) for males, 22.5 kg/m2 (SD = 3.1) for females, 22.5 kg/m2 (SD = 3.1) for those aged 60–74 years, and 21.5 kg/m2 (SD = 3.4) for those aged ≥75 years (not shown in Table 1). Participants who completed the follow-up surveys were more likely to be younger and female than the overall participants who completed the baseline survey (n = 4869). They were also more likely to report healthy behaviors and fewer chronic diseases. The descriptive statistics from the nonimputed data set (including the proportion of missing values in each variable) are shown in Supplementary Material 1.
Table 1.
Participants’ Characteristics
| Baseline Enrollment (n = 4869) | Completed 3-Year Follow-up (n = 3743) | Completed 6-/7-Year Follow-up (n = 3214) | Completed 12-/13-Year Follow-up (n = 1592) | ||
|---|---|---|---|---|---|
| Age (years) | 69.9 (7.2) | 69.0 (6.7) | 68.2 (6.3) | 67.7 (5.6) | |
| Female | 55.3 | 56.5 | 56.8 | 59.8 | |
| Married | 67.1 | 68.8 | 69.9 | 70.0 | |
| Current working | 31.6 | 34.0 | 35.8 | 33.7 | |
| Education (years) | 9.2 (2.8) | 9.2 (2.6) | 9.3 (2.6) | 9.0 (2.5) | |
| Annual household income | Low (<1.2 million yen) | 10.4 | 9.3 | 8.9 | 9.3 |
| Middle-low (1.2–2.9 million yen) | 29.0 | 28.6 | 28.9 | 29.3 | |
| Middle (3.0–4.9 million yen) | 30.5 | 30.4 | 30.7 | 31.6 | |
| Middle–high (5.0–9.9 million yen) | 22.4 | 23.8 | 23.3 | 22.3 | |
| High (≥10.0 million yen) | 7.6 | 7.9 | 8.0 | 7.5 | |
| Average number of cigarettes smoked per day | 0 | 75.5 | 76.1 | 75.5 | 77.6 |
| 1–19 | 12.4 | 11.9 | 12.0 | 11.6 | |
| ≥20 | 12.1 | 12.1 | 12.5 | 10.8 | |
| Average number of drinking days per month | 0 | 59.7 | 59.1 | 58.4 | 60.1 |
| 1–15 | 14.2 | 14.5 | 15.1 | 15.2 | |
| 16–25 | 4.9 | 5.2 | 5.2 | 5.3 | |
| ≥26 | 21.2 | 21.2 | 21.3 | 19.3 | |
| Frequency of exercise | Often/sometimes | 48.0 | 49.8 | 49.4 | 51.2 |
| Rarely/none | 52.0 | 50.2 | 50.6 | 48.8 | |
| Body weight (kg) | 53.6 (9.3) | 53.8 (9.1) | 54.0 (9.1) | 53.9 (8.7) | |
| Body mass index (kg/m2) | 22.3 (3.2) | 22.3 (3.1) | 22.4 (3.1) | 22.5 (3.0) | |
| Underweight (<18.5 kg/m2) | 10.7 | 9.8 | 9.2 | 8.2 | |
| Normal weight (18.5–24.9 kg/m2) | 71.4 | 72.1 | 71.9 | 72.0 | |
| Overweight (25.0–29.9 kg/m2) | 16.4 | 16.7 | 17.7 | 18.8 | |
| Obesity (≥30.0 kg/m2) | 1.4 | 1.4 | 1.2 | 0.9 | |
| Cancer | 1.3 | 1.3 | 1.0 | 0.9 | |
| Heart disease | 11.0 | 10.3 | 9.5 | 8.4 | |
| Stroke | 2.9 | 2.6 | 1.8 | 1.4 | |
| Lung disease | 4.9 | 4.3 | 4.4 | 3.0 | |
| Liver disease | 3.6 | 3.3 | 3.2 | 2.6 | |
| Kidney disease | 2.2 | 2.1 | 1.8 | 1.4 | |
| Entry wave | Wave 1 (year of 1987) | 45.2 | 44.6 | 47.7 | 63.1 |
| Wave 2 (year of 1990) | 7.5 | 8.3 | 8.8 | 14.6 | |
| Wave 4 (year of 1996) | 18.4 | 20.1 | 21.3 | - | |
| Wave 5 (year of 1999) | 28.9 | 27.0 | 22.2 | 22.3 |
Note: Values are presented as means (SDs) or percentages.
Table 2 illustrates the association between weight change and all-cause mortality. This table shows 3 types of analysis using different weight change variables according to the interval (ie, short-term, medium-term, and long-term). A total of 43.5% of the participants had a stable body weight during the short-term interval, and 34.4% and 23.1% had a stable body weight during the medium- and long-term intervals, respectively. We ran Cox proportional hazards models after confirming the proportional hazard assumption using a log–log plot. In terms of short-term weight change, both weight loss and gain ≥ 5% were associated with higher mortality compared with the stable weight category in Model 1 (unadjusted model; HR [95% CI] = 1.54 [1.39–1.70] for loss ≥ 5% and 1.26 [1.12–1.42] for gain ≥ 5%). These associations were attenuated, although they remained significant after adjusting for age and sex in Model 2. In the fully adjusted model (Model 3), the significant association between loss ≥ 5% and mortality was unchanged (1.36 [1.22–1.51]), although the association of gain ≥ 5% with mortality became marginally significant (1.12 [0.99–1.26]). This was also the case for medium-term weight change; loss ≥ 5% was associated with higher mortality (1.36 [1.22–1.51] in Model 3), but the other categories had no significant association with mortality. Regarding long-term weight change, loss ≥ 5% also increased mortality risk, although gain 2.5%–4.9% had a marginally significant association with lower mortality (1.31 [1.11–1.54] and 0.77 [0.58–1.01] in Model 3, respectively).
Table 2.
Associations Between Weight Change and All-Cause Mortality
| Number of Participants | People Who Died Before December 31, 2017 | Incidence Rate | Model 1: Unadjusted | Model 2: Age- and Sex-Adjusted | Model 3: Fully Adjusted* | |||
|---|---|---|---|---|---|---|---|---|
| n | % | n | % | HR (95% CI) | HR (95% CI) | HR (95% CI) | ||
| Short-term weight change | ||||||||
| Loss ≥ 5% | 795 | 21.2 | 637 | 80.2 | 0.052 | 1.54 (1.39–1.71) | 1.38 (1.24–1.53) | 1.36 (1.22–1.51) |
| Loss 2.5%–4.9% | 486 | 13.0 | 352 | 72.4 | 0.041 | 1.09 (0.96–1.22) | 1.00 (0.88–1.13) | 1.02 (0.90–1.15) |
| Stable (±2.4%) | 1629 | 43.5 | 1100 | 67.5 | 0.038 | 1.00 | 1.00 | 1.00 |
| Gain 2.5%–4.9% | 345 | 9.2 | 229 | 66.2 | 0.037 | 0.98 (0.85–1.13) | 0.96 (0.83–1.11) | 0.98 (0.85–1.13) |
| Gain ≥ 5% | 488 | 13.0 | 372 | 76.3 | 0.045 | 1.26 (1.12–1.42) | 1.19 (1.06–1.34) | 1.12 (0.99–1.26) |
| Medium-term weight change | ||||||||
| Loss ≥ 5% | 985 | 30.6 | 749 | 76.1 | 0.045 | 1.60 (1.44–1.77) | 1.37 (1.23–1.52) | 1.36 (1.22–1.51) |
| Loss 2.5%–4.9% | 369 | 11.5 | 245 | 66.4 | 0.035 | 1.15 (1.00–1.33) | 1.02 (0.88–1.18) | 1.04 (0.90–1.21) |
| Stable (±2.4%) | 1106 | 34.4 | 716 | 64.7 | 0.033 | 1.00 | 1.00 | 1.00 |
| Gain 2.5%–4.9% | 266 | 8.3 | 157 | 59.0 | 0.030 | 0.92 (0.84–1.01) | 0.89 (0.75–1.06) | 0.90 (0.75–1.07) |
| Gain ≥ 5% | 488 | 15.2 | 342 | 70.1 | 0.038 | 1.25 (1.09–1.42) | 1.14 (1.00–1.29) | 1.06 (0.93–1.21) |
| Long-term weight change | ||||||||
| Loss ≥ 5% | 735 | 46.2 | 522 | 71.0 | 0.034 | 1.48 (1.27–1.73) | 1.27 (1.08–1.48) | 1.31 (1.11–1.54) |
| Loss 2.5%–4.9% | 154 | 9.7 | 100 | 64.8 | 0.030 | 1.08 (0.85–1.36) | 1.01 (0.80–1.28) | 1.02 (0.81–1.30) |
| Stable (±2.4%) | 368 | 23.1 | 243 | 66.1 | 0.029 | 1.00 | 1.00 | 1.00 |
| Gain 2.5%–4.9% | 106 | 6.7 | 59 | 55.2 | 0.024 | 0.77 (0.57–1.02) | 0.78 (0.58–1.04) | 0.77 (0.58–1.01) |
| Gain ≥ 5% | 228 | 14.3 | 155 | 68.1 | 0.030 | 1.08 (0.88–1.32) | 1.01 (0.83–1.24) | 0.96 (0.78–1.19) |
Notes: CI = confidence interval; HR = hazard ratio.
*Adjusted for age, sex, marital status, working status, education, household income, number of smoking cigarettes, number of drinking days, frequency of exercise, cancer, heart disease, stroke, lung disease, liver disease, kidney disease and body mass index at baseline, and entry wave.
To check the dose–response relationship between continuous weight change and mortality risk, we plotted the spline curves, as seen in Supplementary Material 2. The results from these analyses confirmed dose–response relationships of weight change (particularly weight loss) during short-term, medium-term, and long-term intervals with mortality. This relationship was most pronounced in the link between weight loss and mortality: greater weight loss was associated with higher mortality risk.
Subgroup analyses excluding participants who died within a year after the follow-up survey, current smokers, physically inactive people, and those with a history of chronic diseases are shown in Supplementary Material 3. The analysis for physically active people slightly increased the HRs for short-term and medium-term weight loss ≥ 5% and decreased the HR for long-term weight gain 2.5%–4.9%. The other analyses did not substantially alter the estimation of mortality risk.
Table 3 presents the results of the analyses stratified according to age and sex. The association of weight loss ≥ 5% with higher mortality was observed in both age groups (except for long-term weight change among people aged ≥75 years), and in both sexes.
Table 3.
Stratified Analyses of the Association Between Weight Change and All-Cause Mortality According to Age and Sex
| 60–74 Years* | ≥75 Years* | Male† | Female† | |||||
|---|---|---|---|---|---|---|---|---|
| % | HR (95% CI) | % | HR (95% CI) | % | HR (95% CI) | % | HR (95% CI) | |
| Short-term weight change | ||||||||
| Loss ≥ 5% | 19.0 | 1.33 (1.18–1.51) | 29.1 | 1.37 (1.14–1.65) | 14.2 | 1.41 (1.21–1.65) | 23.0 | 1.32 (1.15–1.52) |
| Loss 2.5%–4.9% | 13.4 | 1.06 (0.93–1.22) | 11.5 | 0.88 (0.68–1.15) | 9.8 | 1.03 (0.86–1.23) | 12.9 | 1.02 (0.86–1.21) |
| Stable (±2.4%) | 45.8 | 1.00 | 35.6 | 1.00 | 45.1 | 1.00 | 42.3 | 1.00 |
| Gain 2.5%–4.9% | 9.4 | 1.03 (0.87–1.21) | 8.6 | 0.82 (0.60–1.11) | 10.0 | 1.11 (0.91–1.35) | 8.6 | 0.88 (0.72–1.09) |
| Gain ≥ 5% | 12.4 | 1.08 (0.93–1.25) | 15.2 | 1.19 (0.94–1.51) | 12.8 | 1.11 (0.92–1.33) | 13.2 | 1.14 (0.96–1.36) |
| Medium-term weight change | ||||||||
| Loss ≥ 5% | 28.3 | 1.37 (1.22–1.54) | 41.6 | 1.28 (1.00–1.65) | 28.1 | 1.37 (1.17–1.60) | 32.5 | 1.38 (1.19–1.60) |
| Loss 2.5%–4.9% | 11.7 | 1.09 (0.92–1.28) | 10.3 | 0.92 (0.64–1.32) | 11.3 | 1.02 (0.82–1.26) | 11.7 | 1.12 (0.92–1.38) |
| Stable (±2.4%) | 36.8 | 1.00 | 23.3 | 1.00 | 35.8 | 1.00 | 33.4 | 1.00 |
| Gain 2.5%–4.9% | 8.3 | 0.83 (0.75–0.92) | 8.0 | 1.10 (0.75–1.60) | 8.4 | 0.91 (0.70–1.18) | 8.1 | 0.92 (0.72–1.17) |
| Gain ≥ 5% | 14.8 | 1.06 (0.91–1.23) | 16.9 | 1.03 (0.75–1.40) | 16.4 | 0.98 (0.82–1.18) | 14.3 | 1.18 (0.97–1.42) |
| Long-term weight change | ||||||||
| Loss ≥ 5% | 43.9 | 1.33 (1.12–1.58) | 61.0 | 1.30 (0.73–2.31) | 41.5 | 1.25 (1.00–1.59) | 49.3 | 1.42 (1.14–1.76) |
| Loss 2.5%–4.9% | 10.1 | 0.97 (0.76–1.25) | 7.1 | 2.03 (0.85–4.87) | 11.6 | 1.13 (0.81–1.58) | 8.4 | 0.95 (0.67–1.36) |
| Stable (±2.4%) | 24.3 | 1.00 | 15.2 | 1.00 | 23.0 | 1.00 | 23.2 | 1.00 |
| Gain 2.5%–4.9% | 7.1 | 0.79 (0.58–1.07) | 3.8 | 0.61 (0.16–2.36) | 8.2 | 0.84 (0.57–1.23) | 5.7 | 0.72 (0.46–1.14) |
| Gain ≥ 5% | 14.5 | 1.00 (0.80–1.25) | 12.9 | 0.63 (0.25–1.64) | 15.7 | 1.00 (0.74–1.35) | 13.3 | 0.97 (0.72–1.31) |
Notes: CI = confidence interval; HR = hazard ratio.
*Adjusted for age, sex, marital status, working status, education, household income, number of smoking cigarettes, number of drinking days, frequency of exercise, cancer, heart disease, stroke, lung disease, liver disease, kidney disease and body mass index at baseline, and entry wave.
†Adjusted for age, marital status, working status, education, household income, number of smoking cigarettes, number of drinking days, frequency of exercise, cancer, heart disease, stroke, lung disease, liver disease, kidney disease and body mass index at baseline, and entry wave.
Finally, the results on the association between weight change and mortality risk by baseline BMI are shown in Table 4. An association between weight loss (particularly loss ≥ 5%) and mortality was generally found for all baseline BMI categories, and its effect was stronger for lower BMIs. In addition, mortality risk was increased for weight gain ≥ 5% during the short- and medium-term among people who were overweight/obese.
Table 4.
Association Between Weight Change and All-Cause Mortality According to Baseline Body Mass Index Level
| Short-term Weight Change | Medium-term Weight Change | Long-term Weight Change | |||||
|---|---|---|---|---|---|---|---|
| % | HR (95% CI) | % | HR (95% CI) | % | HR (95% CI) | ||
| Underweight (BMI < 18.5 kg/m2) | Loss ≥ 5% | 1.6 | 1.91 (1.44–2.52) | 2.5 | 1.44 (1.12–1.87) | 3.2 | 1.92 (1.34–2.73) |
| Loss 2.5%–4.9% | 1.1 | 1.40 (0.98–2.01) | 0.9 | 1.94 (1.27–2.95) | 0.8 | 2.10 (1.13–3.90) | |
| Stable (±2.4%) | 3.9 | 1.21 (0.99–1.49) | 2.7 | 1.32 (1.01–1.71) | 1.3 | 1.15 (0.68–1.95) | |
| Gain 2.5%–4.9% | 1.0 | 1.03 (0.66–1.60) | 0.8 | 1.25 (0.81–1.93) | 0.6 | 1.66 (0.81–3.40) | |
| Gain ≥ 5% | 2.1 | 1.20 (0.89–1.61) | 2.3 | 1.23 (0.92–1.65) | 2.2 | 1.02 (0.66–1.56) | |
| Normal weight (BMI 18.5–24.9 kg/m2) | Loss ≥ 5% | 14.6 | 1.32 (1.17–1.49) | 20.6 | 1.40 (1.24–1.59) | 31.7 | 1.32 (1.10–1.58) |
| Loss 2.5%–4.9% | 8.6 | 1.03 (0.89–1.19) | 8.3 | 1.07 (0.90–1.27) | 7.2 | 0.96 (0.73–1.27) | |
| Stable (±2.4%) | 32.5 | 1.00 | 25.8 | 1.00 | 18.0 | 1.00 | |
| Gain 2.5%–4.9% | 6.7 | 0.99 (0.83–1.17) | 5.9 | 0.88 (0.71–1.08) | 5.0 | 0.73 (0.52–1.02) | |
| Gain ≥ 5% | 9.9 | 1.12 (0.98–1.29) | 11.5 | 1.06 (0.91–1.24) | 10.1 | 0.97 (0.76–1.24) | |
| Overweight/obesity (BMI ≥ 25.0 kg/m2) | Loss ≥ 5% | 5.0 | 1.24 (1.03–1.49) | 7.5 | 1.25 (1.04–1.49) | 11.2 | 1.04 (0.82–1.33) |
| Loss 2.5%–4.9% | 3.3 | 0.83 (0.66–1.05) | 2.4 | 0.79 (0.58–1.08) | 1.6 | 0.86 (0.48–1.55) | |
| Stable (±2.4%) | 7.2 | 0.92 (0.78–1.10) | 6.0 | 1.02 (0.83–1.25) | 3.8 | 0.94 (0.65–1.36) | |
| Gain 2.5%–4.9% | 1.5 | 0.96 (0.68–1.33) | 1.6 | 0.84 (0.57–1.24) | 1.0 | 0.69 (0.32–1.48) | |
| Gain ≥ 5% | 1.0 | 1.58 (1.08–2.30) | 1.4 | 1.39 (1.00–2.00) | 2.0 | 1.08 (0.69–1.70) | |
Notes: BMI = body mass index; CI = confidence interval; HR = hazard ratio. Adjusted for age, sex, marital status, working status, education, household income, number of smoking cigarettes, number of drinking days, frequency of exercise, cancer, heart disease, stroke, lung disease, liver disease, and kidney disease at baseline, and entry wave.
Discussion
Previous studies have indicated mixed results regarding the association between weight change and mortality in later life. Further, the evidence on this association among older people in Asian nations has been sparse. The present study used a nationally representative sample of community-dwelling older Japanese people followed for up to 30 years and examined the association between weight change and all-cause mortality with an improved methodology.
We found that weight loss of ≥5% at all intervals (ie, short-term, medium-term, and long-term) was associated with an increased risk of all-cause mortality. This was also observed in the subgroup analyses by age and sex. This finding is consistent with previous studies, including not only a meta-analysis (18,19) but also several reports on older Asian populations (4,5,8,10,14). It has been reported that weight loss increases the mortality risk associated with cancer, heart disease, and cerebrovascular disease among Japanese adults aged 60–69 years (8). In addition, because decreasing body weight in old age is frequently accompanied by muscle and/or bone loss (24–26), this may result in a reduced ability to maintain a proper energy balance, thereby sharing immunological and neuroendocrine features with disease-associated wasting syndromes (27). Indeed, it has been reported that weight loss is associated with an elevated risk of respiratory death (11). It has also been indicated that weight loss is associated with a decline in both activities of daily living and in living-space mobility (28), which could increase the mortality risk among older people (29).
Increased mortality risk associated with weight loss ≥ 5% was similar across the short-, medium-, and long-term intervals. Weight loss ≥ 5% during short-/medium-term and long-term intervals increased the risk of dying by 36% and 31%, respectively, compared to maintaining a stable weight. We did not perform a statistical test for this difference, although the slightly lower HR of long-term weight loss might be due to selection bias. Some people died or dropped out of the survey due to health problems, and those who completed the 12-/13-year follow-up survey would be relatively healthier people. In fact, they tended to have healthy behaviors and fewer chronic diseases (Table 1). Despite this limitation, we demonstrated that the change in the risk of death associated with weight loss did not greatly differ according to the interval.
The current study also showed that the harmful effect of weight gain was generally weaker than that of weight loss. Previous studies showed mixed results on the significance of the association between weight gain and mortality in old age (18,19), which implies that its effect differs based on the characteristics of the participants and the weight change and follow-up periods. However, these studies consistently reported a weaker influence of weight gain on mortality than weight loss (18,19). In later life, weight loss increased mortality rates associated with cancer, cardiovascular disease, stroke, and respiratory disease, and weight gain increased the risk of death associated with cardiovascular disease (8,11). Therefore, weight gain is expected to have a smaller range of effects on mortality than weight loss.
Our study also showed that long-term weight gain, in the range of 2.5%–4.9%, possibly decreased mortality risk. Numerous previous studies reported findings consistent with an “obesity paradox” in old age: older people with a low BMI had a higher mortality risk, and those in the high–normal, overweight, or obesity range had the lowest mortality risk (30). This might be due to better energy and nutritional reserves among people with a slightly higher BMI, an explanation supported by findings showing that malnutrition is known to be a predictor of all-cause mortality (31,32). Weight gain in old age is predominantly due to increasing fat mass rather than increasing lean body mass (25). Therefore, a slight weight gain over a relatively long period could be protective against mortality. In fact, a small weight gain was found to decrease the risk of respiratory death among middle-aged and older people (11).
The association between weight loss ≥ 5% and all-cause mortality was the strongest among underweight people and the weakest among overweight/obese people. This trend was found regardless of the duration of the weight change interval and is consistent with previous observations in Western nations showing that weight loss among people who are already underweight is associated with a higher increase in mortality risk compared to normal-weight or overweight/obese people (33–35). Also consistent with previous studies (5,12,14), weight gain ≥ 5% in the overweight/obese category was associated with higher mortality, and its influence was greater than that of weight loss, particularly during the short- and medium-term intervals. This implies that clinical and public health management guarding against weight gain is important, especially among people with a BMI ≥ 25.0 kg/m2.
This study could not distinguish between intentional and unintentional weight changes. However, a meta-analysis of observational studies reported that, even among studies where the cause of weight loss was unspecified or unknown, weight loss increased the risk of all-cause mortality (17), which supports our results. In addition, prior studies have shown that intentional weight loss decreased all-cause mortality among obese people (ie, BMI ≥ 30.0 kg/m2) (17,36), but increased the mortality rate among overweight but not obese individuals (ie, BMI 25.0–29.9 kg/m2) (17). Therefore, intentional weight loss seems to be beneficial only for people with obesity. With regard to unintentional weight change, prior research has shown that unintentional weight loss increased the risk of all-cause mortality among both healthy and unhealthy individuals (with obesity-related risk factors) (17). Our Japanese sample included only 1% obese individuals, and a national survey also reported a similar proportion of older people with obesity (3.3% among people aged ≥65 years) (37). Therefore, our finding that weight loss increased the mortality risk seems to be reasonable. Nonetheless, differentiating between these 2 types of weight change in the future could contribute to a better understanding of the factors underlying the association between weight change and mortality.
This study has several major strengths. First, it compared the influence of weight change across 3 different intervals on mortality. Previous studies used only single set intervals to capture weight change. Second, we used a longer follow-up of morality than prior studies, which helped us to understand the long-term effects of weight change on mortality. Third, this Japanese study advances our understanding of Asian nations, which still remain understudied in terms of the relationship between weight change and mortality in old age. Beyond this, our findings should help with assessing the generalizability of findings from Western countries by comparing the association between weight change and mortality in Western and non-Western countries.
Several limitations should also be noted. First, body weight was self-reported in all waves except the 2012 survey, when it was objectively measured. It is known that people tend to underreport their weight (38–40), and that heavier people tend to underreport their weight more than thinner people (40). However, if the underreporting of weight is consistent over time, the analysis should yield valid estimates of changes in body weight, and the potential for bias associated with self-reporting of weight and height might be mitigated. Second, as mentioned above, we did not distinguish between intentional and unintentional weight changes. As the effect of intentional/unintentional weight change can vary by individuals’ body weight and health condition (17), we adjusted for baseline health status to partially account for unintentional weight loss due to underlying and possible yet-undiagnosed diseases in the analysis. Third, we did not consider the fluctuation of body weight during the observation intervals. Weight fluctuation has been reported as a mortality risk (15). Finally, this study did not consider body composition. A previous study demonstrated that weight change was closely associated with changes in body composition (41). Future research should investigate the same question using indicators of body composition to more fully understand the mechanisms linking weight changes over different time ranges to mortality risk.
In conclusion, this study using up to 30 years of longitudinal data from a nationally representative sample of community-dwelling older Japanese people found that weight loss >5% increased the all-cause mortality risk in later life, and provided additional evidence that the effect did not substantially differ based on the interval. These trends were consistently found across subgroups divided according to age and sex and after excluding potential sources of bias such as early death, smoking, physical inactivity, and baseline chronic diseases. Furthermore, the effect of weight loss on mortality was stronger in lower BMI groups than in higher BMI groups. The results highlight the importance of clinical and public health approaches to managing body weight to improve health and survival in later life, particularly in Asian populations.
Funding
This work was supported by the National Institute on Aging (R01 AG031109), the National Institutes of Health (P60 AG024824), and JSPS KAKENHI (23243062 and 17H01555).
Conflict of Interest
None declared.
Supplementary Material
References
- 1. Amador LF, Al Snih S, Markides KS, Goodwin JS. Weight change and mortality among older Mexican Americans. Aging Clin Exp Res. 2006;18:196–204. doi: 10.1007/BF03324649 [DOI] [PubMed] [Google Scholar]
- 2. Dahl AK, Fauth EB, Ernsth-Bravell M, Hassing LB, Ram N, Gerstof D. Body mass index, change in body mass index, and survival in old and very old persons. J Am Geriatr Soc. 2013;61:512–518. doi: 10.1111/jgs.12158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Dey DK, Rothenberg E, Sundh V, Bosaeus I, Steen B. Body mass index, weight change and mortality in the elderly. A 15 y longitudinal population study of 70 y olds. Eur J Clin Nutr. 2001;55:482–492. doi: 10.1038/sj.ejcn.1601208 [DOI] [PubMed] [Google Scholar]
- 4. Ho SC, Woo J, Sham A. Risk factor change in older persons, a perspective from Hong Kong: weight change and mortality. J Gerontol. 1994;49:M269–M272. doi: 10.1093/geronj/49.6.m269 [DOI] [PubMed] [Google Scholar]
- 5. Lee WJ, Peng LN, Loh CH, Chen LK. Effect of body weight, waist circumference and their changes on mortality: a 10-year population-based study. J Nutr Health Aging. 2018;22:959–964. doi: 10.1007/s12603-018-1042-4 [DOI] [PubMed] [Google Scholar]
- 6. Li JB, Luo S, Wong MCS, et al. Longitudinal associations between BMI change and the risks of colorectal cancer incidence, cancer-relate and all-cause mortality among 81,388 older adults: BMI change and the risks of colorectal cancer incidence and mortality. BMC Cancer. 2019;19:1082. doi: 10.1186/s12885-019-6299-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Maru S, van der Schouw YT, Gimbrère CH, Grobbee DE, Peeters PH. Body mass index and short-term weight change in relation to mortality in Dutch women after age 50 y. Am J Clin Nutr. 2004;80:231–236. doi: 10.1093/ajcn/80.1.231 [DOI] [PubMed] [Google Scholar]
- 8. Nanri A, Mizoue T, Takahashi Y, Noda M, Inoue M, Tsugane S; Japan Public Health Center-based Prospective Study Group . Weight change and all-cause, cancer and cardiovascular disease mortality in Japanese men and women: the Japan Public Health Center-Based Prospective Study. Int J Obes (Lond). 2010;34:348–356. doi: 10.1038/ijo.2009.234 [DOI] [PubMed] [Google Scholar]
- 9. Newman AB, Yanez D, Harris T, Duxbury A, Enright PL, Fried LP; Cardiovascular Study Research Group . Weight change in old age and its association with mortality. J Am Geriatr Soc. 2001;49:1309–1318. doi: 10.1046/j.1532-5415.2001.49258.x [DOI] [PubMed] [Google Scholar]
- 10. Nishida MM, Okura M, Ogita M, Aoyama T, Tsuboyama T, Arai H. Two-year weight loss but not body mass index predicts mortality and disability in an older Japanese community-dwelling population. J Am Med Dir Assoc. 2019;20:1654.e11–1654.e18. doi: 10.1016/j.jamda.2019.04.029 [DOI] [PubMed] [Google Scholar]
- 11. Pan XF, Yuan JM, Koh WP, Pan A. Weight change in relation to mortality in middle-aged and elderly Chinese: the Singapore Chinese Health Study. Int J Obes (Lond). 2019;43:1590–1600. doi: 10.1038/s41366-018-0259-y [DOI] [PubMed] [Google Scholar]
- 12. Park S, Pi S, Hwang J, Kang JH, Kwon JW. Effects of initial body mass index and weight change on all-cause mortality: a 10-year Cohort Study in Korea. Asia Pac J Public Health. 2018;30:217–226. doi: 10.1177/1010539518756981 [DOI] [PubMed] [Google Scholar]
- 13. Park SY, Wilkens LR, Maskarinec G, Haiman CA, Kolonel LN, Marchand LL. Weight change in older adults and mortality: the Multiethnic Cohort Study. Int J Obes (Lond). 2018;42:205–212. doi: 10.1038/ijo.2017.188 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Son KY, Kwon H, Park JH, et al. Association between weight change over a period of 4 years and mortality in Korean older adults. Geriatr Gerontol Int. 2020;20:474–481. doi: 10.1111/ggi.13908 [DOI] [PubMed] [Google Scholar]
- 15. Wannamethee SG, Shaper AG, Walker M. Weight change, weight fluctuation, and mortality. Arch Intern Med. 2002;162:2575–2580. doi: 10.1001/archinte.162.22.2575 [DOI] [PubMed] [Google Scholar]
- 16. Somes GW, Kritchevsky SB, Shorr RI, Pahor M, Applegate WB. Body mass index, weight change, and death in older adults: the systolic hypertension in the elderly program. Am J Epidemiol. 2002;156:132–138. doi: 10.1093/aje/kwf019 [DOI] [PubMed] [Google Scholar]
- 17. Harrington M, Gibson S, Cottrell RC. A review and meta-analysis of the effect of weight loss on all-cause mortality risk. Nutr Res Rev. 2009;22:93–108. doi: 10.1017/S0954422409990035 [DOI] [PubMed] [Google Scholar]
- 18. Alharbi TA, Paudel S, Gasevic D, Ryan J, Freak-Poli R, Owen AJ. The association of weight change and all-cause mortality in older adults: a systematic review and meta-analysis. Age Ageing. 2020. doi: 10.1093/ageing/afaa231 [DOI] [PubMed] [Google Scholar]
- 19. Cheng FW, Gao X, Jensen GL. Weight change and all-cause mortality in older adults: a meta-analysis. J Nutr Gerontol Geriatr. 2015;34:343–368. doi: 10.1080/21551197.2015.1090362 [DOI] [PubMed] [Google Scholar]
- 20. Sassi F. Obesity and the Economics of Prevention: Fit Not Fat. Paris: OECD Publishing; 2010. [Google Scholar]
- 21. Cabinet Office. Annual Report on the Aged Society 2020. Tokyo: Nikkei Printing; 2020. [Google Scholar]
- 22. Enders CK. Applied Missing Data Analysis. New York: Guilford Press; 2010. [Google Scholar]
- 23. Schafer JL. Analysis of Incomplete Multivariate Data. London: Chapman & Hall; 1997. [Google Scholar]
- 24. Abe T, Sakamaki M, Yasuda T, et al. Age-related, site-specific muscle loss in 1507 Japanese men and women aged 20 to 95 years. J Sports Sci Med. 2011;10:145–150. [PMC free article] [PubMed] [Google Scholar]
- 25. Newman AB, Lee JS, Visser M, et al. Weight change and the conservation of lean mass in old age: the Health, Aging and Body Composition Study. Am J Clin Nutr. 2005;82:872–878. doi: 10.1093/ajcn/82.4.872 [DOI] [PubMed] [Google Scholar]
- 26. Visser M. Epidemiology of muscle mass loss with age. In: Cruz-Jentoft AJ, Morley JE, eds. Sarcopenia. Oxford: John Wiley & Sons; 2012:1–7. [Google Scholar]
- 27. Schwartz MW, Seeley RJ. Seminars in medicine of the Beth Israel Deaconess Medical Center. Neuroendocrine responses to starvation and weight loss. N Engl J Med. 1997;336:1802–1811. doi: 10.1056/NEJM199706193362507 [DOI] [PubMed] [Google Scholar]
- 28. Ritchie CS, Locher JL, Roth DL, McVie T, Sawyer P, Allman R. Unintentional weight loss predicts decline in activities of daily living function and life-space mobility over 4 years among community-dwelling older adults. J Gerontol A Biol Sci Med Sci 2008;63:67–75. doi: 10.1093/gerona/63.1.67 [DOI] [PubMed] [Google Scholar]
- 29. Mackey DC, Lui LY, Cawthon PM, Ensrud K, Yaffe K, Cummings SR. Life-space mobility and mortality in older women: prospective results from the Study of Osteoporotic Fractures. J Am Geriatr Soc. 2016;64:2226–2234. doi: 10.1111/jgs.14474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Oreopoulos A, Kalantar-Zadeh K, Sharma AM, Fonarow GC. The obesity paradox in the elderly: potential mechanisms and clinical implications. Clin Geriatr Med. 2009;25:643–659. doi: 10.1016/j.cger.2009.07.005 [DOI] [PubMed] [Google Scholar]
- 31. Correia MI, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235–239. doi: 10.1016/s0261-5614(02)00215-7 [DOI] [PubMed] [Google Scholar]
- 32. Sahyoun NR, Jacques PF, Dallal G, Russell RM. Use of albumin as a predictor of mortality in community dwelling and institutionalized elderly populations. J Clin Epidemiol. 1996;49:981–988. doi: 10.1016/0895-4356(96)00135-7 [DOI] [PubMed] [Google Scholar]
- 33. Corrada MM, Kawas CH, Mozaffar F, Paganini-Hill A. Association of body mass index and weight change with all-cause mortality in the elderly. Am J Epidemiol. 2006;163:938–949. doi: 10.1093/aje/kwj114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Myrskylä M, Chang VW. Weight change, initial BMI, and mortality among middle- and older-aged adults. Epidemiology. 2009;20:840–848. doi: 10.1097/EDE.0b013e3181b5f520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Nilsson PM, Nilsson JA, Hedblad B, Berglund G, Lindgärde F. The enigma of increased non-cancer mortality after weight loss in healthy men who are overweight or obese. J Intern Med. 2002;252:70–78. doi: 10.1046/j.1365-2796.2002.01010.x [DOI] [PubMed] [Google Scholar]
- 36. Kritchevsky SB, Beavers KM, Miller ME, et al. Intentional weight loss and all-cause mortality: a meta-analysis of randomized clinical trials. PLoS One. 2015;10:e0121993. doi: 10.1371/journal.pone.0121993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Ministry of Health, Labour and Welfare. The National Health and Nutrition Survey in Japan, 2018. Tokyo: Ministry of Health, Labour and Welfare; 2020. [Google Scholar]
- 38. Gunnell D, Berney L, Holland P, et al. How accurately are height, weight and leg length reported by the elderly, and how closely are they related to measurements recorded in childhood? Int J Epidemiol. 2000;29:456–464. doi: 10.1093/intjepid/29.3.456 [DOI] [PubMed] [Google Scholar]
- 39. Nakamura K, Hoshino Y, Kodama K, Yamamoto M. Reliability of self-reported body height and weight of adult Japanese women. J Biosoc Sci. 1999;31:555–558. doi: 10.1017/s0021932099005556 [DOI] [PubMed] [Google Scholar]
- 40. Nawaz H, Chan W, Abdulrahman M, Larson D, Katz DL. Self-reported weight and height: implications for obesity research. Am J Prev Med. 2001;20:294–298. doi: 10.1016/s0749-3797(01)00293-8 [DOI] [PubMed] [Google Scholar]
- 41. Kyle UG, Zhang FF, Morabia A, Pichard C. Longitudinal study of body composition changes associated with weight change and physical activity. Nutrition. 2006;22:1103–1111. doi: 10.1016/j.nut.2006.08.003 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.