Abstract
Background
TP53 status based on TP53 signature, a gene expression profile to determine the presence or absence of TP53 mutation, is an independent prognostic factor of breast cancer. The purpose of this study was to develop a simple diagnostic system for TP53 signature status.
Methods
We developed a multiplex reverse transcription–polymerase chain reaction system to determine TP53 status. Based on this system, prospectively collected 189 patients with stage I and II breast cancer were determined to have TP53 mutant signature or TP53 wild-type signature. The prognostic significance of the TP53 signature by the diagnostic system was analyzed.
Results
The diagnostic accuracy of TP53 status and reproducibility of this diagnosis system was confirmed. Using the diagnostic system, 89 patients were classified as TP53 mutant signature and the remaining 100 cases were classified as TP53 wild-type signature. Recurrence-free survival (RFS) among patients with TP53 mutant signature was significantly shorter than that among those with TP53 wild-type signature. On univariate and multivariate analyses, the TP53 signature status was an independent predictor of RFS. RFS among patients with TP53 mutant signature was significantly shorter than that among those with TP53 wild-type signature in a cohort of estrogen receptor-positive breast cancer. Although a difference was not significant, no recurrent cases was observed in TP53 wild-type signature group in triple negative breast cancer.
Conclusion
This simple and precise diagnostic system to determine TP53 signature status may help in prognostic assessment, therapeutic decision-making, and treatment optimization in patients with breast cancer.
Supplementary Information
The online version contains supplementary material available at 10.1007/s12282-021-01250-z.
Keywords: TP53 signature, Breast cancer, Prognostic factor, Diagnostic system
Introduction
The plethora of comprehensive gene expression analyses in the context of breast cancer has gradually helped unravel the molecular biology of breast cancer. In addition, a large number of gene expression profiles that predict prognosis, recurrence, and therapeutic response to anticancer drugs and endocrine therapies has been reported [1]. Representative gene expression profiles, such as Onocotype DX [2–4], Mammaprint [5, 6], and Prosigna [7, 8], have already been approved by US Food and Drug Administration.
Tumor suppressor gene TP53 is the most frequently mutated gene in human cancers, and the patients with TP53 mutations are known to have poor clinical outcomes [9]. Several large-scale meta-analyses have shown TP53 mutation to be an independent predictor of poor prognosis for breast cancer [10, 11]. Furthermore, TP53 status is a predictive factor for chemotherapy [12, 13].
We had earlier found a gene expression signature (TP53 signature) that correlates with presence or absence of TP53 mutation [14]. The TP53 status determined using the TP53 signature was a prognostic factor independent of other known clinicopathological prognostic factors. Also, the TP53 status determined using gene expression signature was a superior predictor of prognosis compared with that determined using immunohistochemical examination and direct DNA sequencing. Similar results were earlier reported by Miller et al. [15].
The purpose of this study was to develop a simple diagnostic system for TP53 signature using multiplex reverse transcription–polymerase chain reaction (RT–PCR), to test its diagnostic precision and prognostic predictability in a prospective cohort and to examine the clinical significance of TP53 signature among breast cancer subtypes.
Patients and methods
Patients and tumor tissues
This study was approved by the Ethics Committee at the Tohoku University Hospital (TU), Hoshi General Hospital (HG), and Miyagi Cancer Center (MCC). The TU cohort, which was used in our previous study [14], was used for the development of the TP53 signature diagnosis system. Validation cohort is a breast cancer case series from HG and MCC prospectively from September, 2007 to October, 2013 [16]. None of the cases received chemotherapy or endocrine therapy preoperatively. Written informed consent for the study was obtained from all patients. A part of the surgical specimen of breast cancer was stored as fresh frozen (FF) tissue and/or formalin-fixed paraffin embedded (FFPE) tissue. Among patients enrolled in this study, we selected curatively resected patients with stage I–II breast cancer. Patients with ductal carcinoma in situ, those with unknown histology or those with squamous cell carcinoma were excluded from the analysis. The validation cohort was used to assess the prognostic ability of the TP53 signature diagnosis system.
Clinicopathological characteristics
Clinicopathological characteristics data (pathological tumor size, pathological lymph node status, pathological stage, ER, PgR, HER2, Grade, Ki-67, adjuvant chemotherapy and adjuvant endocrine therapy) were obtained from medical records. For cases for which Ki-67 data were not available, immunohistological staining for Ki-67 was performed at the Department of Pathology, Tohoku University Hospital, using the MIB-1 antibody (Dako, Carpinteria, CA, USA).
RNA extraction
The glass slide specimen with 10-µm thick sections of FF and FFPE tissue blocks were prepared. In reference to the HE stained specimen, tumor cells were collected from FF tissue or FFPE tissue by macrodissection technique. Total RNA was extracted from FF tissue or FFPE tissue with use of RNeasy mini kit (Qiagen, Valencia, CA, USA) or RNeasy FFPE kit (Qiagen, Valencia, CA, USA), respectively.
TP53 signature diagnosis system
Genome Lab GeXP Genetic Analysis System (Beckman Coulter, Brea, CA) was used to obtain gene expression profile. To obtain the TP53 signature gene set for GeXP, genes for which the average signal value in the raw data exceeded 1000 in the previous microarray data of the TU cohort [14] and which had less homolog genes were selected. Based on these criteria, 23 genes were chosen among TP53 signature genes. Three genes were added to this gene set as internal control; as a result, a TP53 signature gene set that comprised of 26 genes was established (Supplemental Table 1). Primers for reverse transcription (RT) and for PCR were designed using Genome Lab eXpress Designer GeXP Software (Beckman Coulter, Brea, CA). The multiplex reaction was optimized as per the manual and optimal primer concentrations determined. RT and PCR were performed with GenomeLab GeXP Start Kit (Beckman Coulter, Brea, CA) in accordance to the manual. The quantity of input RNA was 1 μg for FFPE tissues and 50 ng for the FF tissues.
TP53 signature score
TP53 status was determined by TP53 signature score, which is the ratio of the sum of expression levels of 16 genes that were upregulated in tumors with TP53 mutation to the sum of expression values of 7 genes downregulated in tumors withTP53 mutation. The cutoff level for TP53 signature score was determined by Receiver Operating Characteristic curve (ROC) analysis based on the TP53 signature status by microarray of TU cohort [14]. When TP53 signature score of a certain sample was greater than 1.11, the sample was labeled as TP53 mutant signature.
Outcomes
The primary end point of the study was recurrence-free survival (RFS), which was defined as the period from the date of surgery for breast cancer to the date on which tumor recurrence. Overall survival (OS) was defined as the period from the date of surgery for breast cancer to the date of death. Breast cancer-specific survival (BCSS) was defined as the period from the date of surgery for breast cancer to the date of death by breast cancer.
Statistical analysis
All statistical analyses were performed using JMP Pro 14.3.0 (SAS Institute Japan Co., Ltd., Tokyo, Japan). Baseline characteristics of patients (except age) were assessed by chi-squared test. Kruskal–Wallis test was used for statistical analysis of age. Survival curves were made with Kaplan–Mayer method, and between-group differences assessed with log-rank test. Univariate and multivariate analyses (Cox proportional hazard model) were conducted to assess the association between clinicopathological factors and the TP53 status for RFS. P value under 0.05 was considered indicative of a statistically significant difference. This study is registered in UMIN-CTR (http://www.umin.ac.jp/ctr/) (000005172).
Results
Patients for analysis
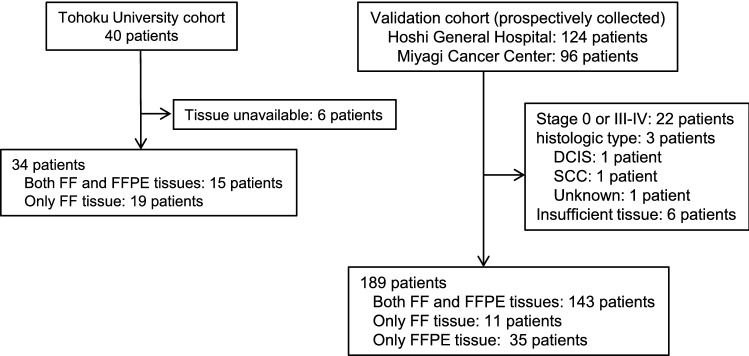
The TU cohort comprises 40 patients, 34 of whom were included in this analysis. The validation cohort comprised 220 patients who had undergone surgery between October, 2013 and September, 2007. Out of the 220 patients, 31 patients were excluded based on the exclusion criteria (Fig. 1). The remaining 189 patients were included in the analysis. Median duration of observation period was 8.06 years (range 0.91–10.18 years).
Fig. 1.
The details of the cohorts. The Tohoku University cohort comprises 40 patients, 34 of whom were included in the analysis. The validation cohort comprised 220 patients who underwent surgery. Out of the 220 patients, 31 were excluded based on the exclusion criteria (stage and histological type) or due to inadequate specimens. The remaining189 patients were included in the analysis. FF fresh frozen, FFPE formalin-fixed paraffin-embedded
Cutoff value of TP53 signature score
RNAs extracted from 34 samples of the TU cohort were available for analysis. TP53 signature of these 34 patients was examined with multiplex PCR method. From the result of ROC analysis, the cutoff value for TP53 signature score was set at 1.11 (Area under the curve: AUC = 0.993) (Table 1).
Table 1.
TP53 status diagnosed by TP53 signature score, microarray and Sanger sequence in the Tohoku University cohort
| Sample | TP53 signature score | TP53 status by TP53 signature score | TP53 status by microarray |
|---|---|---|---|
| BR047 | 0.1707 | Wild | Wild |
| BR038 | 0.3383 | Wild | Wild |
| BR019 | 0.4267 | Wild | Wild |
| BR044 | 0.5178 | Wild | Wild |
| BR033 | 0.5206 | Wild | Wild |
| BR050 | 0.5406 | Wild | Wild |
| BR034 | 0.5696 | Wild | Wild |
| BR045 | 0.5910 | Wild | Wild |
| BR063 | 0.6151 | Wild | Wild |
| BR016 | 0.7157 | Wild | Wild |
| BR024 | 0.7668 | Wild | Wild |
| BR052 | 0.7820 | Wild | Wild |
| BR036 | 0.8019 | Wild | Wild |
| BR027 | 0.8796 | Wild | Wild |
| BR048 | 0.9536 | Wild | Wild |
| BR043 | 1.0681 | Wild | Wild |
| BR058 | 1.1003 | Wild | Wild |
| BR064 | 1.2907 | Mutant | Mutant |
| BR040 | 1.4288 | Mutant | Mutant |
| BR013 | 1.4350 | Mutant | Wild |
| BR020 | 1.4504 | Mutant | Mutant |
| BR035 | 1.4809 | Mutant | Mutant |
| BR026 | 1.6511 | Mutant | Mutant |
| BR046 | 1.6968 | Mutant | Mutant |
| BR017 | 1.7842 | Mutant | Mutant |
| BR010 | 1.9654 | Mutant | Mutant |
| BR001 | 2.1603 | Mutant | Mutant |
| BR005 | 2.1959 | Mutant | Mutant |
| BR021 | 2.3457 | Mutant | Mutant |
| BR022 | 2.3744 | Mutant | Mutant |
| BR053 | 2.4841 | Mutant | Mutant |
| BR011 | 2.5209 | Mutant | Mutant |
| BR041 | 3.6260 | Mutant | Mutant |
| BR009 | 4.0595 | Mutant | Mutant |
TP53 signature score for 189 patients in the validation cohort
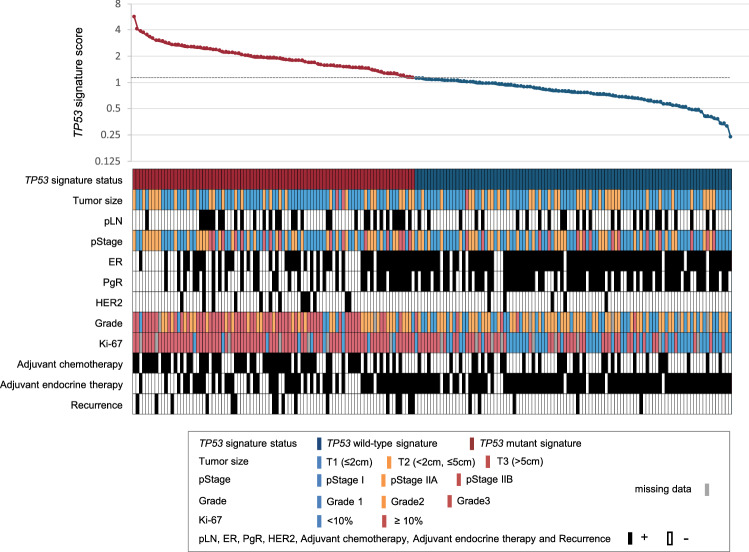
TP53 signature scores of 189 cases of the validation cohort were calculated. With use of a cutoff value of 1.11, 89 patients were classified as TP53 mutant signature, and the remaining 100 cases were classified as TP53 wild-type signature. Patient characteristics disaggregated by TP53 status is shown in Table 2 and Fig. 2. A significant difference was observed between the two different TP53 signatures with respect to ER, PgR, HER2, tumor grade, histological type, Ki-67, postoperative adjuvant chemotherapy, and postoperative adjuvant endocrine therapy.
Table 2.
Clinicopathological characteristics disaggregated by TP53 status
| Total | Mutant signature | Wild-type signature | P* | ||||
|---|---|---|---|---|---|---|---|
| No. of patients | % | No. of patients | % | No. of patients | % | ||
| Samples | 189 | 100 | 89 | 47 | 100 | 53 | |
| Age, years (median) | 29–98 (58.0) | 29–83 (59.0) | 26–98 (56.0) | 0.077 | |||
| pStage | 0.55 | ||||||
| I | 95 | 50 | 41 | 46 | 54 | 54 | |
| IIA | 65 | 34 | 33 | 37 | 32 | 32 | |
| IIB | 29 | 15 | 15 | 17 | 14 | 14 | |
| ER | < 0.0001 | ||||||
| + | 138 | 73 | 49 | 55 | 89 | 89 | |
| − | 51 | 27 | 40 | 45 | 11 | 11 | |
| PgR | < 0.0001 | ||||||
| + | 101 | 54 | 34 | 38 | 67 | 67 | |
| − | 88 | 46 | 55 | 62 | 33 | 33 | |
| HER2 | 0.025 | ||||||
| + | 18 | 10 | 13 | 15 | 5 | 5 | |
| − | 171 | 90 | 76 | 85 | 95 | 95 | |
| Pathological tumor size, cm | 0.78 | ||||||
| ≤ 2 | 125 | 66 | 59 | 66 | 66 | 66 | |
| > 2, ≤ 5 | 61 | 32 | 28 | 31 | 33 | 33 | |
| > 5 | 3 | 2 | 2 | 2 | 1 | 1 | |
| pLN | 0.19 | ||||||
| + | 57 | 30 | 31 | 35 | 26 | 26 | |
| − | 132 | 70 | 58 | 65 | 74 | 74 | |
| Grade | < 0.0001 | ||||||
| 1 | 46 | 25 | 8 | 9 | 38 | 40 | |
| 2 | 82 | 45 | 29 | 33 | 53 | 55 | |
| 3 | 55 | 30 | 50 | 57 | 5 | 5 | |
| NA | 6 | 2 | 4 | ||||
| Histology | 0.043 | ||||||
| Invasive ductal carcinoma | 179 | 95 | 85 | 96 | 94 | 94 | |
| Invasive lobular carcinoma | 3 | 2 | 0 | 0 | 3 | 3 | |
| Medulary carcinoma | 3 | 2 | 3 | 3 | 0 | 0 | |
| Mucinous | 3 | 2 | 0 | 0 | 3 | 3 | |
| Undiff. carcinoma | 1 | 1 | 1 | 1 | 0 | 0 | |
| Ki-67 | < 0.0001 | ||||||
| < 10 | 55 | 32 | 9 | 11 | 46 | 51 | |
| ≥ 10 | 118 | 68 | 37 | 89 | 45 | 49 | |
| NA | 16 | 7 | 9 | ||||
| Adjuvant chemotherapy | < 0.0001 | ||||||
| + | 86 | 46 | 54 | 61 | 32 | 32 | |
| − | 103 | 54 | 35 | 39 | 68 | 68 | |
| Adjuvant endocrine therapy | < 0.0001 | ||||||
| + | 137 | 72 | 49 | 55 | 88 | 88 | |
| − | 52 | 28 | 40 | 45 | 12 | 12 | |
P* Chi-square test was used for statistical analysis of patients' characteristics except for age. Kruskal–Wallis test was used for statistical analysis of patients' age
pStage pathological stage, ER estrogen receptor, PgR progesterone receptor, HER2 human epidermal growth factor receptor type 2, pLN pathological lymph node, NA not available
Fig. 2.
TP53 signature score and clinicopathological characteristics. The upper graph shows the TP53 signature score of each case in the validation cohort. Red and blue represents TP53 mutant signature and TP53 wild-type signature, respectively. The lower figure shows the clinicopathological characteristics of the corresponding cases in the upper graph. Legend of colors is shown in the figure. pStage, pathological stage; pLN, pathological lymph node; ER, estrogen receptor; PgR, progesterone receptor; HER2, human epidermal growth factor receptor type 2
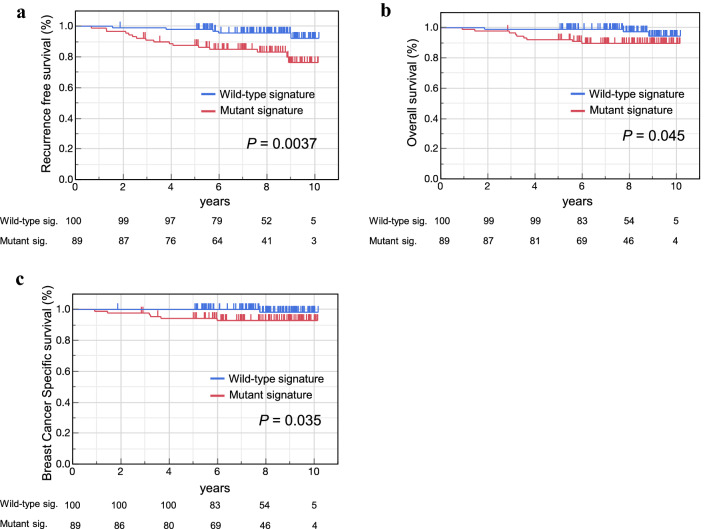
Recurrence-free survival, overall survival and breast cancer-specific survival by TP53 signature status
The TP53 mutant signature group showed significantly poorer RFS than that shown by the TP53 wild-type signature group (Fig. 3a). In OS and BCSS, the TP53 mutant signature group showed significantly worse than TP53 wild-type signature group (Fig. 3b, c).
Fig. 3.
RFS, OS and BCSS by TP53 signature status. RFS (a), OS (b), and BCSS (c) by TP53 status based on the TP53 signature score in the validation cohort were constructed using the Kaplan–Meier method. The differences were compared using the log-rank test. The short vertical line on the curve represent censored. RFS recurrence-free survival, OS Overall survival, BCSS breast cancer-specific survival
Univariate and multivariate analyses to identify factors associated with RFS
On univariate analysis, tumor stage, lymph node and TP53 status by signature were significantly associated with RFS (Table 3). On multivariate analysis, only the TP53 status by signature showed a significant association with RFS. Our results indicate that TP53 signature based on multiplex RT–PCR was an independent predictor of RFS.
Table 3.
Results of uni- and multivariate analysis (Cox proportional hazard model) showing correlation of RFS with clinicopathological factors in patients with breast cancer
| Variable | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | P | HR | 95% CI | P | |
| pStage (vs. Stage I) | 2.60 | 1.01–6.69 | 0.05 | 1.72 | 0.48–6.10 | 0.40 |
| pLN (vs. negative) | 2.55 | 1.08–6.01 | 0.03 | 1.65 | 0.52–5.17 | 0.39 |
| Pathological tumor size (vs. T1) | 1.27 | 0.53–3.07 | 0.59 | |||
| Grade (vs. 1–2) | 1.14 | 0.46–2.82 | 0.78 | |||
| ER (vs. positive) | 1.33 | 0.54–3.30 | 0.54 | |||
| PR (vs. positive) | 1.54 | 0.65–3.67 | 0.32 | |||
| HER2 (vs. negative) | 0.46 | 0.06–3.39 | 0.44 | |||
| Ki-67 (vs. < 10%) | 4.15 | 0.95–18.1 | 0.06 | |||
| Adjuvant chemotherapy (vs. non-therapy) | 1.47 | 0.62–3.51 | 0.38 | |||
| Adjuvant endocrinetherapy (vs. non-therapy) | 0.97 | 0.38–2.50 | 0.95 | |||
| TP53 status by signature (vs. wild-type) | 3.96 | 1.45–10.8 | 0.01 | 3.73 | 1.36–10.20 | 0.01 |
pStage pathological stage, pLN pathological lymph node, ER estrogen receptor, PgR progesterone receptor, HER2 human epidermal growth factor receptor type 2, HR hazard ratio, CI confidence interval
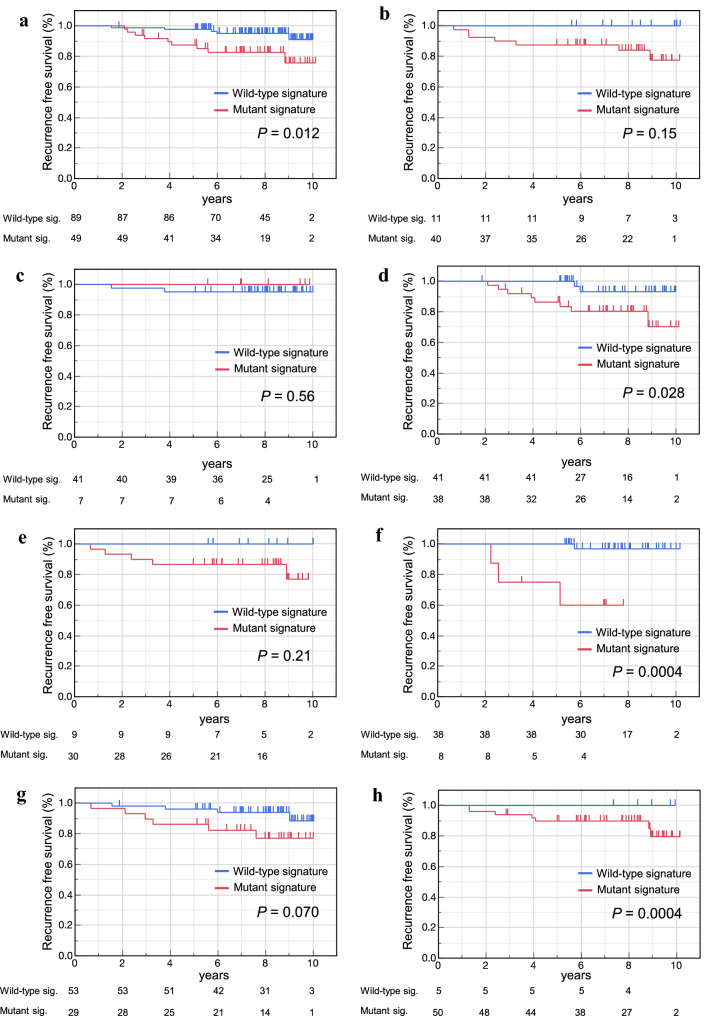
RFS by TP53 signature status in subtypes of breast cancer
In ER positive subtype, RFS of TP53 wild-type signature was significantly better than that of TP53 mutant signature (P = 0.012) (Fig. 4a). Although a significant difference between TP53 signature status was not shown in ER negative subtype, luminal A like group (ER positive and Ki-67 < 10%) and triple negative breast cancer (TNBC) group, no recurrent cases was observed in TP53 wild-type signature group (Fig. 4b, c, e). On the other hand, a significant difference was observed between TP53 signature status in luminal B like subtype (ER positive and Ki-67 ≥ 10%) (Fig. 4d). A survival analysis in HER2 positive subtype did not be carried out because of lack of patients. In grade 1 and 3, RFS of TP53 mutant signature group was significantly worse than that of wild-type group (Fig. 4f, h). Although the significant difference could not be observed, RFS of TP53 mutant signature group showed worse trend than that of wild-type group in grade 2 (Fig. 4g).
Fig. 4.
RFS by TP53 signature status in subtypes of breast cancer and in grade. RFS stratified by TP53 status based on the TP53 signature score in patients with ER + (a), ER(−) (b), Luminal A like (c), Luminal B Like (d), TNBC (e), Grade 1 (f), Grade 2 (g) and Grade 3 (h) were constructed using the Kaplan–Meier method. The differences were compared using the log-rank test. The short vertical line on the curve represent censored. ER estrogen receptor, TNBC triple negative breast cancer
Discussion
The TP53 mutation has long been known as an independent predictor of poor prognosis among patients with breast cancer [10, 11]. To develop a reliable diagnostic kit, we created the gene expression signature that could diagnose the TP53 gene status using microarray analysis [14]. Uji et al. reported that the TP53 status determined by gene expression signature was a superior predictor of prognosis than TP53 status determined on direct DNA sequencing (including the classical Sanger sequencing and the NGS method) [17]. Today, although the TP53 gene mutation can be analyzed in detail by the cancer genome profiling test, the TP53 signature is considered to have an advantage in terms of prognosis prediction for breast cancer. Lehmann et al. verified the prognostic predictability of 351 reported gene expression profiles on a meta-analysis based on 31 breast cancer cohorts [18]. They found TP53 signature was a robust prognostic factor, and was better than well-known gene expression profiles such as OnctypeDX and Mammaprint. Furthermore, Lehmann et al. verified that TP53 signature was a predictor of therapeutic response in their meta-analysis [18]. Similarly, Oshima et al. reported that signature could predict response to preoperative chemotherapy [19]. As described above, TP53 signature is confirmed to be both an independent prognostic factor and an independent predictor for response to chemotherapy.
In this report, a simple and easy multiplex RT–PCR diagnostic system for TP53 signature was developed and the rate of agreement of TP53 status by TP53 signature score and the TP53 status by microarray was enough high (97.1%) (Table 1).
In the validation cohort, a significant difference was observed between the two TP53 signatures with respect to ER, PgR, HER2, histological grade, Ki-67 histological type, adjuvant chemotherapy and adjuvant endocrine therapy (Table 2). These results do not contradict those reported from previous studies [14, 15, 20, 21].
The TP53 mutant signature based on the TP53 signature score was associated with significantly poor RFS, OS and BCSS as compared to that associated with the TP53 wild-type signature. On univariate and multivariate analysis, TP53 signature was significantly associated with PFS independent of other clinicopathological factors. These results indicate that the TP53 status diagnosed by this diagnostic system was an independent prognostic factor in patients with breast cancer for whom curative resection (stage I–II) is performed.
In this report, we showed for the first time that there was clinical significance among breast cancer subtypes and grades. In the ER positive, especially in Luminal B like subgroup, Grade1 and 3 subgroup, it was clearly seen that the prognosis was closely associated with the TP53 status. In ER negative group, Luminal A like subtype and TNBC, the significant difference was not observed between TP53 signature status. But, because there was no recurrence in TP53 wild-type signature group, it can be said that TP53 signature had clinical significance in these subtypes.
There are some limitations of the interpretation of this study. First, the sample size was relatively small, and the recurrence events were few so far. We are going to follow up recurrent events sequentially. Second, uniform treatment intervention was not carried out for the study cohort because it is an observational, prospective study. We are currently conducting a large scare retrospective-prospective study to confirm the clinical significance of TP53 signature using several prospective studies conducted in Japan.
In conclusion, we developed a relatively simple multiplex RT–PCR diagnostic system to determine the TP53 signature. Its diagnostic accuracy and prognostic value were verified in a prospective cohort. And we showed the clinical significance of TP53 signature among breast cancer subtypes. This simple and precise diagnostic system may help in prognostic assessment, therapeutic decision-making, and treatment optimization in patients with breast cancer.
Supplementary Information
Below is the link to the electronic supplementary material.
Abbreviations
- BCSS
Breast cancer-specific survival
- ER
Estrogen receptor
- FEC
5-FU/epirubicin/cyclophosphamide
- FF
Fresh frozen
- FFPE
Formalin-fixed paraffin embedded
- HE
Hematoxylin–Eosin
- HER2
Human epidermal growth factor receptor type 2
- OS
Overall survival
- PCR
Polymerase chain reaction
- RFS
Recurrence-free survival
- PgR
Progesterone receptor
- RT
Reverse transcription
- TNBC
Triple negative breast cancer
Author contributions
Conceptualization: ST, CI; Methodology: ST, CI; Formal analysis and investigation: ST, TF and FF; Writing—original draft preparation: ST; Writing—review and editing: CI, TF, TN, YK, FF, TI, NO, Funding acquisition: ST, CI; Resources: TN, YK, TI, NO.
Funding
This work was supported by the Grants-in-Aid from the JSPS KAKENHI (24701000) and the Research funds for preventive medicine from Miyagi Health Care Association (no grant number). We thank patients and medical and technical staff, particularly Ms. Eri Yokota and Ms. Hiromi Nakano.
Declarations
Conflict of interest
Dr. Takahashi reports personal fees from Taiho, Chugai, Asahikasei, Bayer, Japan blood products organization, Medicon, Termo, Sanofi, Nippon-kayaku, Takeda, Yakult, grants and personal fees from Merckbiopharma, grants from Ono, outside the submitted work; In addition, Dr. Takahashi has a patent JP4370409B2 issued. Dr. Ishida reports grants from Taiho, Eisai and Kyowa-Kirin, grants and personal fees from Chugai, personal fees from Pfizer and Astra Zeneca, outside the submitted work. Prof. Ishioka reports grants and personal fees from Novartis, Daiichi Sankyo, Bayer, Bristol-Myers Squibb, Nippon-Kayaku, Hitachi, personal fees from Taiho, Ono, Merckbiopharma, AsahiKasei, Sanofi, Takeda, Eisai, Eli Lilly, Mundipharma, Teijin, Chugai, Konica Minolta, Pfizer, Mochida, grants from Riken Genesis, MSD, Linical, outside the submitted work; In addition, Prof. Ishioka has a patent JP4370409B2 issued. All the remaining authors have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Shin Takahashi, Email: shin.takahashi.e7@tohoku.ac.jp.
Takafumi Fukui, Email: takafumi.fukui@falco.co.jp.
Tadashi Nomizu, Email: nomizu@hoshipital.jp.
Yoichiro Kakugawa, Email: kakugawayo1@gmail.com.
Fumisyoshi Fujishima, Email: ffujishima@patholo2.med.tohoku.ac.jp.
Takanori Ishida, Email: takanori@med.tohoku.ac.jp.
Noriaki Ohuchi, Email: noriaki-ohuchi@med.tohoku.ac.jp.
Chikashi Ishioka, Email: chikashi@tohoku.ac.jp.
References
- 1.Reis-Filho JS, Pusztai L. Gene expression profiling in breast cancer: classification, prognostication, and prediction. Lancet. 2011;378(9805):1812–1823. doi: 10.1016/S0140-6736(11)61539-0. [DOI] [PubMed] [Google Scholar]
- 2.Sparano JA, Gray RJ, Makower DF, Pritchard KI, Albain KS, Hayes DF, et al. Prospective validation of a 21-gene expression assay in breast cancer. N Engl J Med. 2015;373(21):2005–2014. doi: 10.1056/NEJMoa1510764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Albain KS, Barlow WE, Shak S, Hortobagyi GN, Livingston RB, Yeh IT, et al. Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: a retrospective analysis of a randomised trial. Lancet Oncol. 2010;11(1):55–65. doi: 10.1016/S1470-2045(09)70314-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Paik S, Tang G, Shak S, Kim C, Baker J, Kim W, et al. Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J Clin Oncol. 2006;24(23):3726–3734. doi: 10.1200/JCO.2005.04.7985. [DOI] [PubMed] [Google Scholar]
- 5.Cardoso F, van’t Veer LJ, Bogaerts J, Slaets L, Viale G, Delaloge S, et al. 70-gene signature as an aid to treatment decisions in early-stage breast cancer. N Engl J Med. 2016;375(8):717–729. doi: 10.1056/NEJMoa1602253. [DOI] [PubMed] [Google Scholar]
- 6.van de Vijver MJ, He YD, van’t Veer LJ, Dai H, Hart AA, Voskuil DW, et al. A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med. 2002;347(25):1999–2009. doi: 10.1056/NEJMoa021967. [DOI] [PubMed] [Google Scholar]
- 7.Gnant M, Filipits M, Greil R, Stoeger H, Rudas M, Bago-Horvath Z, et al. Predicting distant recurrence in receptor-positive breast cancer patients with limited clinicopathological risk: using the PAM50 Risk of Recurrence score in 1478 postmenopausal patients of the ABCSG-8 trial treated with adjuvant endocrine therapy alone. Ann Oncol. 2014;25(2):339–345. doi: 10.1093/annonc/mdt494. [DOI] [PubMed] [Google Scholar]
- 8.Parker JS, Mullins M, Cheang MC, Leung S, Voduc D, Vickery T, et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol. 2009;27(8):1160–1167. doi: 10.1200/JCO.2008.18.1370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Soussi T, Beroud C. Assessing TP53 status in human tumours to evaluate clinical outcome. Nat Rev Cancer. 2001;1(3):233–240. doi: 10.1038/35106009. [DOI] [PubMed] [Google Scholar]
- 10.Olivier M, Langerod A, Carrieri P, Bergh J, Klaar S, Eyfjord J, et al. The clinical value of somatic TP53 gene mutations in 1,794 patients with breast cancer. Clin Cancer Res. 2006;12(4):1157–1167. doi: 10.1158/1078-0432.CCR-05-1029. [DOI] [PubMed] [Google Scholar]
- 11.Borresen-Dale AL. TP53 and breast cancer. Hum Mutat. 2003;21(3):292–300. doi: 10.1002/humu.10174. [DOI] [PubMed] [Google Scholar]
- 12.Geisler S, Lonning PE, Aas T, Johnsen H, Fluge O, Haugen DF, et al. Influence of TP53 gene alterations and c-erbB-2 expression on the response to treatment with doxorubicin in locally advanced breast cancer. Cancer Res. 2001;61(6):2505–2512. [PubMed] [Google Scholar]
- 13.Bertheau P, Plassa F, Espie M, Turpin E, de Roquancourt A, Marty M, et al. Effect of mutated TP53 on response of advanced breast cancers to high-dose chemotherapy. Lancet (London, England) 2002;360(9336):852–854. doi: 10.1016/S0140-6736(02)09969-5. [DOI] [PubMed] [Google Scholar]
- 14.Takahashi S, Moriya T, Ishida T, Shibata H, Sasano H, Ohuchi N, et al. Prediction of breast cancer prognosis by gene expression profile of TP53 status. Cancer Sci. 2008;99(2):324–332. doi: 10.1111/j.1349-7006.2007.00691.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Miller LD, Smeds J, George J, Vega VB, Vergara L, Ploner A, et al. An expression signature for p53 status in human breast cancer predicts mutation status, transcriptional effects, and patient survival. Proc Natl Acad Sci USA. 2005;102(38):13550–13555. doi: 10.1073/pnas.0506230102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yamaguchi S, Takahashi S, Mogushi K, Izumi Y, Nozaki Y, Nomizu T, et al. Molecular and clinical features of the TP53 signature gene expression profile in early-stage breast cancer. Oncotarget. 2018;9(18):14193–14206. doi: 10.18632/oncotarget.24447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Uji K, Naoi Y, Kagara N, Shimoda M, Shimomura A, Maruyama N, et al. Significance of TP53 mutations determined by next-generation "deep" sequencing in prognosis of estrogen receptor-positive breast cancer. Cancer Lett. 2014;342(1):19–26. doi: 10.1016/j.canlet.2013.08.028. [DOI] [PubMed] [Google Scholar]
- 18.Lehmann BD, Ding Y, Viox DJ, Jiang M, Zheng Y, Liao W, et al. Evaluation of public cancer datasets and signatures identifies TP53 mutant signatures with robust prognostic and predictive value. BMC Cancer. 2015;15:179. doi: 10.1186/s12885-015-1102-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Oshima K, Naoi Y, Kishi K, Nakamura Y, Iwamoto T, Shimazu K, et al. Gene expression signature of TP53 but not its mutation status predicts response to sequential paclitaxel and 5-FU/epirubicin/cyclophosphamide in human breast cancer. Cancer Lett. 2011;307(2):149–157. doi: 10.1016/j.canlet.2011.03.027. [DOI] [PubMed] [Google Scholar]
- 20.Marchetti A, Buttitta F, Pellegrini S, Campani D, Diella F, Cecchetti D, et al. p53 mutations and histological type of invasive breast carcinoma. Cancer Res. 1993;53(19):4665–4669. [PubMed] [Google Scholar]
- 21.Dobes P, Podhorec J, Coufal O, Jureckova A, Petrakova K, Vojtesek B, et al. Influence of mutation type on prognostic and predictive values of TP53 status in primary breast cancer patients. Oncol Rep. 2014;32(4):1695–1702. doi: 10.3892/or.2014.3346. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.