Abstract
Purpose of Review
To review the effects of early-life, preconception, and prior-generation exposures on reproductive health in women.
Recent Findings
Women’s early-life factors can affect reproductive health by contributing to health status or exposure level on entering pregnancy. Alternately, they can have permanent effects, regardless of later-life experience. Nutrition, social class, parental smoking, other adverse childhood experiences, environmental pollutants, infectious agents, and racism and discrimination all affect reproductive health, even if experienced in childhood or in utero. Possible transgenerational effects are now being investigated through three- or more-generation studies. These effects occur with mechanisms that may include direct exposure, behavioral, endocrine, inflammatory, and epigenetic pathways.
Summary
Pregnancy is increasingly understood in a life course perspective, but rigorously testing hypotheses on early-life effects is still difficult. In order to improve the health outcomes of all women, we need to expand our toolkit of methods and theory.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40471-021-00279-0.
Keywords: Pregnancy, Transgenerational, Early-life, Life course, Birth outcomes, Preconception
Introduction
Traditionally research on pregnancy has been focused on the 9 months prior to birth. However, there is increasing recognition of the effects of preconception, early-life, and transgenerational exposures on reproductive outcomes. Such ideas have been developed from perspectives that range from sociological and life-course [1] to next-generation and technological (such as epigenetic analysis of DNA methylation [2]), and are incorporated into the concept of the exposome, addressing the totality of particularly environmental exposures throughout the life course [3]. Preconception can be construed narrowly as the period when pregnancy is planned, or broadly, as any point in the life span prior to pregnancy, while “early-life” usually indicates a focus on childhood and adolescence [4]. Exposures from both time periods affect pregnancy and infant health (e.g., folic acid supplementation [5] or adverse childhood experiences [6]). Adverse health behaviors, whether smoking, sedentary lifestyle, or poor diet, rarely start during pregnancy. They are usually in place well before pregnancy, so that pregnancy begins under less optimal conditions and potentially leads to chronic diseases for both mother and child [7]. In addition, social class, stress, and trauma, whenever experienced, affect both biology and behavior, with consequences for pregnancy health, fecundability, and fertility decisions [8]. Developmental Origins of Health and Disease theory and research, while primarily focused on cardiometabolic health, also have been applied to reproductive outcomes [9].
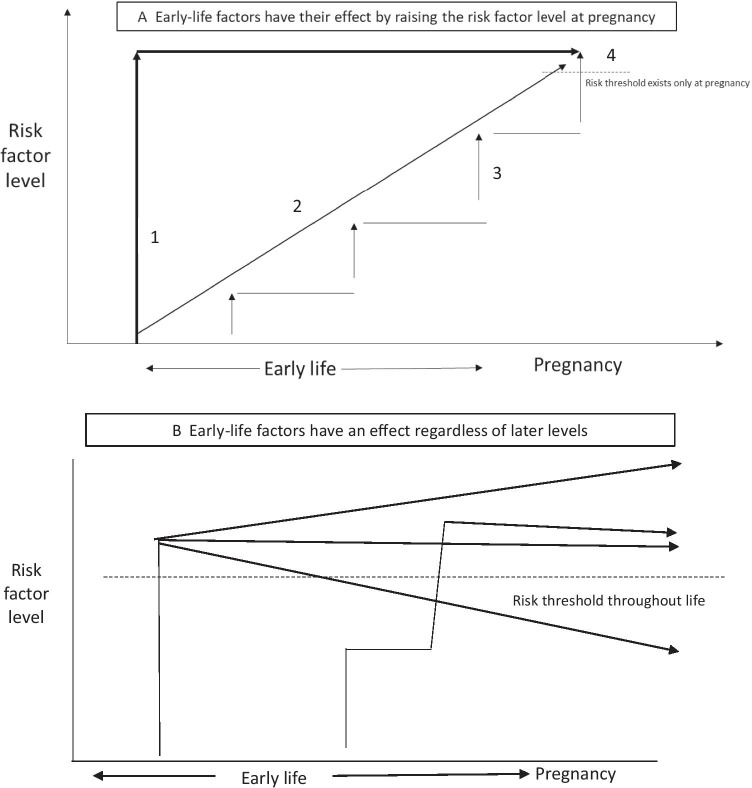
Early-life exposures (whether in childhood, adolescence, or a previous generation) set a trajectory of risk. In some cases, this trajectory indicates an association between the early-life risk factor and risk factor levels during pregnancy (or at conception attempt) (Fig. 1a). Knowledge about early-life exposures is therefore important for understanding the course of disease, identifying women who are at high risk, and determining interventions that will have the greatest impact. However, measuring early-life risk factors does not provide additional predictive power beyond what is measured during pregnancy; the threshold for risk is met only when the pregnancy happens. For example, adolescent smoking might predict reproductive outcomes, but only because it is associated with smoking during pregnancy. If a woman quit smoking prior to pregnancy, the early-life smoking would not predict outcomes. In other cases, early-life exposures have independent effects on reproductive and pregnancy outcomes (Fig. 1b), and predict and cause these outcomes. For these, an early-life exposure has a permanent effect, regardless of later exposures: early-life smoking predicts reproductive outcomes, regardless of smoking during pregnancy. The exposures considered in this review may be of either type, but we will focus on possible mechanisms for independent, permanent effects of early-life exposures (Table 1).
Fig. 1.
Schematic for possible effect trajectories of early-life exposures on pregnancy and reproduction. a Early-life risk factors stay at a high level throughout the preconception period (trajectory 1), gradually rise (trajectory 2), or accumulate (trajectory 3). However, the important risk level only occurs at pregnancy (4). If we measured only at time 4, we would have the same degree of predictive power. b Early-life risk factors have an independent effect, regardless of their levels at the time of pregnancy. Once a risk threshold is crossed, whether levels of the risk factor stay constant, increase, or decrease, the early-life risk factor is still predictive
Table 1.
Mechanisms of early-life effects on reproductive outcomes
| Pathway | Example exposure | Example mediator | Example outcomes |
|---|---|---|---|
| Direct effects | Syphilis | Transplacental infection | Stillbirth, miscarriage [80] |
| Pb | Release from bone during pregnancy | Hypertension during pregnancy [81] | |
| Structural | Vitamin D deficiency | Pelvic deformities | Obstructed labor and stillbirth |
| Chlamydia | Tubal damage | Infertility | |
| Health behaviors | Adverse childhood experiences | Smoking | Low birthweight [82] |
| Low neighborhood SES | Gestational weight gain | Preterm birth [83] | |
| Endocrine | Sexual abuse | HPA axis activation and insulin resistance | Gestational diabetes [84] |
| Parental smoking | Anti-Mullerian hormone[85] | Fertility | |
| Inflammatory | Social class | C-reactive protein, IL-6 [86] | Intra-amniotic infection and preterm delivery [87] |
| Maternal infection | Pro-inflammatory cytokines | ovarian function and development [88] | |
| Epigenetic | Warzone stress | Methylation of NR3C1 gene promoter[89] | Birthweight [90] |
| Maternal overnutrition and obesity | Methylation of aryl-hydrocarbon receptor repressor | Birthweight and gestational age [91] |
SES, socioeconomic status; HPA, hypothalamic–pituitary–adrenal
Early-Life Influences on Women’s Reproductive Health
Nutrition
Childhood nutrition has long been recognized for its far-reaching effects on adult health. At the most basic level, sufficient calories are necessary for menarche and normal gynecological development to occur [10], and this can extend to lifelong influences; for example, low early-life crop yields in preindustrial societies [11] and early-life exposure to the Dutch Hunger Winter were associated with reduced fertility [12]. Other early deficiencies also cause long-term problems, e.g., rickets in early life leading to pelvic deformities inconsistent with carrying a pregnancy to term [13]. Other micronutrient deficiencies, including iron, thiamine, and vitamin B12, have long-term physiological sequelae that can be permanent [14]. Women with a history of eating disorders are at higher risk of miscarriage and low birthweight (LBW) [15]. High-calorie and low-nutrient density diets lead to obesity in childhood and later, associated with infertility, gestational diabetes, and complicated labor [16].
Social Class
Early-life social class predicts reproductive health. Economic hardship during the mother’s childhood is associated with negative outcomes in neonates, including lower birthweight, longer hospital stays, and higher rates of neonatal intensive care unit admissions [17]. Maternal socioeconomic decline from childhood has been associated with infant mortality and preterm birth (PTB) [18, 19], while lifetime improved SES has been associated with reduced risk of small-for-gestational-age (SGA) and PTB [20, 21]. Recent work has extended this idea from individual SES to the wider community; for instance, neighborhood SES [22] modified age-related patterns of LBW and SGA [23], while inequality and polarization in childhood neighborhood was associated with greater likelihood of PTB in adulthood [24].
Abuse and Other Adverse Childhood Experiences
Women who experience physical abuse in childhood are at increased risk of PTB and LBW [25]. Sexual abuse during childhood has a particularly strong association with pregnancy complications, including increased hospitalization, premature contractions, cervical insufficiency, and PTB [26]. Abuse is also strongly associated with mental health issues, which raise the risk for several poor outcomes, both directly and via health behaviors, substance use, and partner violence during the pregnancy [27]. Experiencing out-of-home care (foster or residential care) as a child is associated with increased risk of PTB [28]. More generally, adverse childhood experiences (ACEs), such as abuse, parental substance use, or neglect, are linked to adverse birth outcomes [29, 30], unintended pregnancy [31], and risky reproductive health behaviors, including early sexual debut and prenatal substance use [32–34]. It should be noted that positive experiences in earlier life may also have long-term effects; women with greater athletic opportunities in adolescence (due to Title IX) gave birth to babies with higher Apgar scores and who were less likely to be LBW [35].
Parental Smoking
Tobacco smoke is composed of hundreds of toxins, many of which affect reproduction. Exposure to parental smoking in childhood is associated with stillbirth, spontaneous abortion, and ectopic pregnancy, and reduced success in fertility treatment [36, 37], even among women who never smoked themselves. In utero exposure to maternal smoking has been associated with reduced fertility [38] and miscarriage [39], though effects are small and inconsistent [40].
Environmental Pollutants
Many environmental pollutants are retained in the body. Lipophilic chemicals accumulate in fat tissues and heavy metals are taken up in bones. Such toxicants may be released during pregnancy even if exposure occurred many years previously; this has been clearly demonstrated with studies of lead (Pb) isotopes [41]. Arsenic, to which several worldwide populations are highly exposed, is associated with alterations in hormonal levels and may cause anemia, exacerbated during pregnancy [42]. Preconception and prenatal exposure to endocrine disruptors can impact a woman’s reproductive health, altering hormonal signaling in reproductive organs by, for instance, antiestrogenic effects. The focus of this research has generally been on organogenesis and neonatal periods, and then again during the reproductive years [43]; for instance, prenatal exposure to dioxins and polychlorinated biphenyls (PCBs) has been associated with longer time to pregnancy [44]. Few studies have directly linked childhood and adolescent exposures to later-life pregnancy outcomes. Young women living in the Chernobyl region had a higher risk of miscarriage, premature birth, and menstrual disturbances approximately 20 years after the nuclear disaster there, although in that case there were long-lasting and ongoing exposures to radiation and other pollutants [45].
Infections
HIV and Other Sexually Transmitted Infections
Proper treatment of chronic infections is crucial in management of infectious sequelae impacting reproductive health. Although research on non-congenitally inherited early-life exposure to sexually transmitted diseases is extremely limited, infections occurring years prior to child-bearing can significantly impact fecundity. Women living with HIV are at increased odds of developing coinfection with other sexually transmitted infections including human papillomavirus, genital herpes, syphilis, trichomonas, gonorrhea, and chlamydia, which may progress to pelvic inflammatory disease, with deleterious effects on reproductive health and fecundity [46, 47]. Women with HIV are more likely to experience anovulation and amenorrhea [48]. Preconception treatment with antiretroviral therapy increases the risk of both PTB and LBW, compared to women treated with antiretroviral therapy after conceiving [49]. Other long-term infectious diseases, such as tuberculosis, also carry reproductive sequelae, including infertility, miscarriage, and ectopic pregnancy [50].
COVID-19
At this point, nothing can be said conclusively about the long-term effects of COVID-19 on reproductive health. Several changes in reproductive physiology observed with COVID-19 have effects on later fertility and pregnancy, but it will be many years before we can truly assess the effects of early-life exposure to COVID-19. Patients infected with COVID-19 have been found to have a prolonged menstrual cycle, but these changes did not extend beyond a few months after resolution of infection [51]. Alterations of Angiotensin-Converting Enzyme (ACE-2) have led to cases of orchitis and infertility in males with COVID-19 infection, and researchers have hypothesized that gonadotropin-dependent expression of ovarian ACE-2 could affect female gametogenesis including ovarian function, oocyte quality, and later pregnancy outcomes [46], although initial studies of hormone concentrations are reassuring [51]. Pro-inflammatory ACE-2 can lead to damage of endometrial epithelial cells, which may affect early embryo implantation [52, 53], and ACE-2 effects on angiotensin-II could also lead to dysfunctional uterine bleeding secondary to hyperplastic endometrium. Parental treatment of COVID-19-induced pneumonia, including antiviral therapy, has been hypothesized to influence later development of the fetus, leading to a recommendation that contraception be encouraged for a time after active infection [54], although current evidence suggests no association between medical treatments and birth defects or preterm delivery [55]. Beyond possible effects of the virus and related treatments, the social, economic, educational, and emotional toll of the pandemic may be substantial and lifelong or transgenerational. The 1918 influenza pandemic led to reduced income and health for at least two generations [56].
Racism and Other Forms of Discrimination
Structural racism and discrimination produce a situation where the abovementioned risk factors are more likely to be seen in populations of color and disadvantaged populations. The high prevalence of these early-life risk factors is likely a major contributor to disparities in reproductive outcomes. Studies of lifetime experiences of interpersonal racism or ethnic discrimination have generally found associations with adverse birth outcomes [57] or with potential mediators of such associations, such as depressive symptoms or psychological distress [58]. Experiences of stigma and discrimination among sexual minority women contribute to worse reproductive health [59].
Mechanisms
These early-life risk factors could lead to poorer reproductive and pregnancy outcomes through several pathways (Table 1). Certain exposures (infectious agents or environmental pollutants) are retained in the body, so that the early-life exposure is still present during pregnancy even when there was no recent exposure. Others have direct effects on the structure of the reproductive system. Some exposures, particularly social ones, may lead to adverse health behaviors. Many types of exposures produce long-term hormonal or inflammatory changes [60]. For instance, micronutrient deficiencies cause subtle changes such as cortisol levels and rhythms [14]; adverse childhood experiences have also been shown to alter hypothalamic–pituitary–adrenal axis function [61, 62]; in utero exposure to maternal smoking changes ovarian developmental signaling [63]; and prenatal exposure to perfluoroalkyl substances has been associated with anti-Müllerian hormone levels, associated with ovarian reserve [64]. Increasingly, there is interest in possible epigenetic changes that persist across the life course, carry into pregnancy, and impact reproduction [65], although evidence for causal effects is still limited.
Multigenerational Effects
Developmental origins of health and disease (DOHaD) research have made us aware that early life can encompass the period in utero, or even prior to conception, and studies are now beginning to address transgenerational effects. Such studies provide powerful evidence for life experiences affecting health and disease across the life course and generations, and how “life circumstances, health, and disease are linked at a molecular scale” [66]. PubMed, EMBASE, and CINAHL were searched for papers with keywords related to transgenerational effects (transgenerational, multigenerational, three-generation) and reproductive health (birthweight, preterm, gestational age, fertility, miscarriage; see supplementary material for details). Several three- or more-generation studies exist addressing reproductive health (Table 2). Perhaps because the original focus among DOHaD studies was birthweight (and likely because it is easily measured and reported), this remains the primary reproductive outcome studied. However, a few studies have also addressed infant mortality, preterm birth, birth defects, or hypertensive disorders (Table 2). Results from the multigenerational studies are inconclusive. Generally, grandparents’ exposures were related to grandchildren’s birth outcomes, but often that was mediated by known risk factors in the intermediate generation, such as maternal BMI, smoking, or birthweight.
Table 2.
Three- or more generation studies of transgenerational exposures and reproductive or pregnancy outcomes
| Study or author name; Country, state, or region | Primary outcome | F0 exposure(s) | Study design/data source | Multigenerational results* |
|---|---|---|---|---|
|
1958 National Child Development Study [92] England, Scotland, Wales |
Birthweight | Smoking during pregnancy (up to or after the fourth month) | Cohort enrolled at birth. Grandmothers reported on smoking during pregnancy. Maternal report on offspring numbers and birthweight | Grandmother’s smoking associated with reduced birthweight via maternal smoking. If mother did not smoke, adjusted association between smoking and birthweight was positive |
|
3G Multigenerational Cohort of Nova Scotian women [93] Nova Scotia, Canada |
Birthweight, gestational age, neonatal complications | Sociodemographics, weight, smoking | Population database linkage | |
|
Aberdeen children of the 1950s [94] Scotland, UK |
Birthweight | Occupation | Interview and medical record | Those in the highest quintile of SES (F0) had higher birthweight for mother (F1) and children (F2) |
|
Agius [73] Malta |
Birthweight | BMI and metabolic syndrome | Linkage of clinical datasets | No associations between F0 metabolic syndrome and F2 birthweight |
| ALSPAC (Avon Longitudinal Study of Parents and Children), Avon, UK [95] | Birthweight | Smoking, diabetes | Parents reported during their pregnancies on grandparents’ smoking and diabetes status; children were followed prospectively |
Grandchildren of maternal grandparents with type 2 diabetes were more likely to be in the top tertile of birth weight than grandchildren of non-diabetics Inverted U-shaped association between birth weight of grandchildren and diabetes in paternal grandmothers |
|
Andrasfay [70] Florida, USA |
Birthweight, LBW | Race/ethnicity and foreign/domestic birth | Linked vital statistics | Hispanic foreign-born F0 associated with less LBW in F2; black foreign-born F0 LBW advantage much reduced |
|
Bogalusa Heart Study and Bogalusa Daughters [96, 97] Louisiana, USA |
Birthweight, gestational age | Cardiometabolic health | Cohort follow-up and F1 interview/linkage to birth certificates for F2 | F0 higher glucose levels associated with higher F2 birthweight; F0 triglycerides and LDL associated with lower F2 birthweight |
|
Children of 1997 [98] Hong Kong |
Birthweight | Education | Birth cohort with report on grandparental information | F0 education not associated with birthweight |
|
Consortium of Health Outcomes Research in Transitioning Societies Cebu, Philippines |
Birthweight | Nutritional status | Birth cohort originally enrolled mothers and infants; children followed up for outcomes of their children | |
|
Danish Perinatal Study [101] Denmark |
PTB, SGA | Smoking, BMI | Birth cohort with linkage and follow-up of children | |
|
DES follow-up study |
Birth defects, menstrual cycle characteristics, fertility, miscarriage, preterm birth, neonatal death | DES in utero | Combination and follow-up of 4 cohorts; DES exposure verified by medical record | Overall birth defects were elevated but possibly reporting bias; higher risk of irregular menses and amenorrhea; preterm delivery |
|
Dutch Famine Birth Cohort Study [104, 105] Netherlands |
Infant mortality, birthweight | Famine | Follow-up of birth cohort | Excess F2 perinatal mortality if F0 exposed to famine in the third trimester. F2 birthweight lower if F0 exposed, due primarily to effects on F1 birthweight |
|
Early Life Exposures in Mexico to ENvironmental Toxicants (ELEMENT) [106] Mexico City |
Birthweight, head circumference | Lead, endocrine disruptors, pesticides | Follow-up of pregnancy/birth cohort | |
|
Giuntella [107] California and Florida, USA |
Birthweight | Assimilation/endogamy | Linked birth certificates (siblings as well as direct line) | F2 children of F1 intermarried Hispanic women are more likely to be LBW |
|
Add Health [108] USA |
Birthweight | Education | Maternal report | Grandmaternal education associated with higher grandchild birthweight |
|
Illawarra Born [109] Illawarra-Shoalhaven, Australia |
Birthweight, length, head circumference, gestational age | Mental health, personality, quality of life, alcohol use | Pregnant women recruited along with grandmothers and children | |
|
Illinois Transgenerational Study [110–112, 113 and others] USA |
Birthweight |
Immigration generation Place of birth |
Links across generations; limited to existing data on birth certificate | Upward mobility associated with reduced risk of PTB among those not born LBW |
|
Isle of Wight birth cohort [114] UK |
Birthweight | BMI and smoking | Birth cohort and medical records | F0 BMI and smoking associated with F2 birthweight via maternal birthweight, smoking, and BMI |
|
Jerusalem Perinatal Family Follow-Up Study [115] Israel |
Birthweight | Demographics, socioeconomic status, smoking | Follow-up of a birth cohort | |
|
Lee [116] South Korea |
Birthweight, gestational age | Kwangju uprising | Vital statistics | Grandmother exposure during pregnancy (particularly second trimester) associated with lower birthweight and gestational age |
|
Lifecourse Influences on Fetal Environment [20, 117] Michigan, USA |
PTB, SGA, LBW | Socioeconomic position, health, BMI, neighborhood | Birth cohort with additional interviewing of F0 mothers | Improved F1 education relative to F0 associated with reduced risk of SGA |
|
Lifelines NEXT [118] Netherlands |
Gestational age, birthweight, birth length, birth defects | Anthropometry, blood pressure, pulmonary function, neuropsychiatric health, family, work, physical activity, smoking | F0 recruitment of adults (aged 25 to 50) through general practitioners; recruitment of their children and grandchildren through F0 | |
|
Life Span Study Atomic Bomb Survivor Cohort [119] Japan |
Birthweight, birth defects, infant mortality | Atom bomb | Cohort follow-up | |
|
Lifeways Cross-generational Study [120, 121] Ireland |
BMI at birth Height at birth |
Height and BMI | Prospective study of children 2001–2013. Height and BMI collected at 0, 5, and 9 years of age. Retrospective on adults |
Offspring height correlated with both parents at birth Adherence to Healthy Eating Index in maternal grandparents associated with lower likelihood of LBW and greater likelihood of macrosomia |
|
Mater-University of Queensland Study of Pregnancy [122] Queensland, Australia |
Pregnancy outcomes | Mental illness, physical activity, BMI, substance use, poverty | Birth cohort with added F2 generation | |
|
Michigan Bone Health and Metabolism Study [123] Michigan, USA |
Birthweight | Smoking | Study of female adult children of participants community health study; reported on grandmother’s and children’s health | Birthweight was higher in F2 grandchildren whose grandmother (F0) and mother (F1) both smoked during pregnancy if F0 was born between 1929 and 1945; not found for earlier births |
|
Nurses’ Health Study 2, Growing Up Today Study (GUTS), Nurses’ Mothers Study [124] USA |
Birthweight | Smoking | Children and mothers of NHSII women were recruited. Information on grandpaternal smoking was provided by mothers | Birthweight and child BMI were higher in those whose grandmothers smoked. No association between grandparental smoking and adolescent BMI |
|
Naess [69] Norway |
Birthweight | Death certificates for ICD: breast cancer, chronic obstructive pulmonary disease, cardiovascular causes, coronary heart disease, stroke, diabetes, lung cancer | Identified children (F2) births from 1967–2009 and linked parents (Maternal and Paternal) (F1) and grandparents (Maternal and Paternal) (F0); | Grandparents who died of cardiovascular causes associated with lower likelihood of higher birthweight; much of this was due to maternal smoking in pregnancy. U-shaped associations were seen with grandchild birthweight and maternal grandmother mortality from diabetes; inverse associations for all other grandparents |
|
Nebraska Mother Index [125] USA |
Birthweight, gestational age, delivery type | Linked vital statistics | ||
|
New Jersey [126] USA |
PTB | Education, marital status | Linked vital statistics | Grandmother’s education predicted PTB for grandchildren |
|
Norwegian Mother and Child Birth Cohort (MoBa) [127] Norway |
Birthweight, gestational age, birth defects | Health, medication, smoking | Primarily a birth cohort with child follow-up, but linked to other registries for some analyses | |
|
Pathways to Adulthood Baltimore, USA |
Birthweight | Smoking, health, BMI, income | Follow-up of the Collaborative Perinatal Project birth cohort; includes interviews and medical records | Higher F0 income/needs ratio associated with higher birthweight. Higher F0 education associated with higher birthweight only if F1 education was lower. Effects of F1 smoking moderated by F0 smoking |
|
Qian [130] Taiwan |
Birthweight for gestational age | Education, town-level educational status | Linked vital statistics | |
| Respiratory Health in Northern Europe, Spain, and Australia (RHINESSA) [131] | PTB | Respiratory health, place of residence, exposure to allergens, smoking | Population-based study of adults; grandparents and children recruited through participants | |
|
Song [132] China |
Infant mortality | Famine (Chinese Great Leap Forward) | 2001 National Family Planning and Reproductive Health Survey (NFPRHS) and the 1982 Chinese Population Census |
In regions of low famine severity, mothers’ prenatal famine exposure significantly reduced children’s infant mortality; in regions of high famine severity, prenatal exposure associated with higher infant mortality |
|
Seattle Social Development Project (SSDP) and the SSDP Intergenerational Project [133] Washington, USA |
Birthweight | Socioeconomic disadvantage | Longitudinal study of mothers followed in the Seattle School Development Project starting at age 10 and followed until age 27 | Low grandparental SES in maternal childhood associated with birthweight. Abuse in maternal childhood was associated with birthweight, mediated through maternal substance abuse as an adolescent and/or prenatally |
|
South Carolina Multigenerational Linked Birth Dataset [72] USA |
Birthweight | Education, social mobility | Linked birth certificates | |
|
Uppsala Birth Cohort Multigenerational Study [67] Sweden |
Birthweight Birth length Gestational age |
Education, marital status, occupational class |
Birth cohort study F0 born 1915–1929 linked to grandchildren who were born after 1972 |
Shared environment had a small but significant effect on birthweight and birth length |
|
US National Longitudinal Study of Youth [74] USA (nationally representative) |
Birthweight, controlled for gestational age |
Education (self-reported) Nonmarital childbearing |
Mother is index subject. Grandmother and mother interviewed at baseline, information collected about child from mother or directly from child at follow-ups. Birthweight measured on grandchildren of original study participants | |
|
Utah Population Database [134, 135] USA |
Fertility, birth defects | Age, chemotherapy exposure | Linked birth, death, cancer registry | No association between grandmaternal age and F2 trisomy 21 |
|
Vägerö [136] Sweden |
Birthweight, PTB | Childhood trauma: parental (F0) death during F1 childhood | Linked information from several Swedish national registers, including the Population and Housing Census 1960, Cause of Death Register 1961–2002, the Swedish Register of Education 1990 and the Swedish Medical Birth Register (MBR) 1973–2002 | Grandparental death during parental childhood predicted lower birthweight and PTB in generation 3 |
|
Virginia Intergenerational Linked Birth File [137] USA |
Birthweight | Linked vital statistics | ||
|
Wallace [138] New York, USA |
LBW | Housing destruction | Birth records and vital statistics | 1970–1980 percent population change associated with socioeconomic factors that were associated with low birthweight in 2008 |
|
Washington State Intergenerational Cohort [139] USA |
LBW, PTB | Education, residence in a deprived area, smoking | Linked vital statistics |
*Generations are referred to as F0 (grandparent), F1 (parent), F2 (child) for consistency, regardless of how they were referred to in the paper. Last column blank for studies that present a 3-generation study design but for which we did not find papers on effects of F0 exposures on F2 outcomes
LBW, low birthweight; PTB, preterm birth; SES, socioeconomic status; DES, diethystilbestrol
Conducting a multigenerational study is difficult. Randomized trials are impossible and prospective cohort studies are nearly as hard. The Uppsala study is the closest to a prospective design, identifying F0 births (first-generation, grandparents) at a single hospital (1915–1929), then linking to F1 (children) and F2 (grandchildren) [67]. Few other studies started with a multigenerational design, and many include both prospective and retrospective components. Several studies began as birth cohorts, enrolling either pregnant women or children at birth and following the children until they had their own children. Others are linkages of existing data sources (e.g., Swedish and Norwegian medical registries [68, 69], vital statistics data [70–72], or medical records [73]). Some focus on a defined exposure (e.g., famine, traumatic experience, diethylstilbestrol (DES), the atomic bomb); others were developed as studies of social and economic development in youth, and expanded to include biological measures [74]. For some, the focus is on change in risk factors across generations, particularly those with a focus on social or neighborhood exposures [18, 19]. All the study designs share issues with completeness of measurement across generations and informative missing data [75]. Interpretation of these studies is additionally complex in that it is generally easier to follow the maternal line, but many epigenetic and transgenerational effects in both humans and animals have been shown to be paternal [76]. Perhaps because of these difficulties, many studies that have collected information on three generations appear to have published primarily on two-generation associations.
Conclusions
Pregnancy is increasingly understood in a life course perspective, but rigorous methods for researching this are still difficult. Retrospective studies are subject to recall bias and selective fertility [77]. Prospective life course studies require a long-term investment of time and money, and information relevant to hypotheses of most interest decades or generations later may not have been measured initially. Even when data are collected at regular time points, statistical methods may not be sufficient for determining critical periods, while measurement error may not allow determining independent effects [78]. Parental — particularly paternal — information may be missing, especially for important confounders. These concerns are magnified in multigenerational studies. Technological developments in both statistical methods [75] and epigenetics [79] will be needed to improve our understanding of patterns of disease and of mechanisms. However, given the lack of efficacy for many interventions implemented during pregnancy, improving health in this crucial period may not be possible without improving health throughout life.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
We thank Elaine Hicks for assistance in conducting the literature review.
Funding
This project was supported by a Collaborative Research Network (CORNET) award in health disparities research from Tulane University, the University of Tennessee Health Sciences Center, and University of Mississippi Medical Center. It was also supported in part by a planning grant for preconception research from the Louisiana Clinical and Translational Science Center and the National Institute of General Medical Sciences of the National Institutes of Health (U54GM104940), which funds the Louisiana Clinical and Translational Science Center. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Footnotes
This article is part of the Topical Collection on Reproductive and Perinatal Epidemiology
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Lu MC. Improving maternal and child health across the life course: where do we go from here? Matern Child Health J. 2014;18(2):339–343. doi: 10.1007/s10995-013-1400-0. [DOI] [PubMed] [Google Scholar]
- 2.Bar-Sadeh B, Rudnizky S, Pnueli L, Bentley GR, Stöger R, Kaplan A, et al. Unravelling the role of epigenetics in reproductive adaptations to early-life environment. Nat Rev Endocrinol. 2020;16(9):519–533. doi: 10.1038/s41574-020-0370-8. [DOI] [PubMed] [Google Scholar]
- 3.Buck Louis GM, Yeung E, Sundaram R, Laughon SK, Zhang C. The exposome–exciting opportunities for discoveries in reproductive and perinatal epidemiology. Paediatr Perinat Epidemiol. 2013;27(3):229–236. doi: 10.1111/ppe.12040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fleming TP, Watkins AJ, Velazquez MA, Mathers JC, Prentice AM, Stephenson J, et al. Origins of lifetime health around the time of conception: causes and consequences. Lancet. 2018;391(10132):1842–1852. doi: 10.1016/S0140-6736(18)30312-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Toivonen KI, Lacroix E, Flynn M, Ronksley PE, Oinonen KA, Metcalfe A, et al. Folic acid supplementation during the preconception period: a systematic review and meta-analysis. Prev Med. 2018;114:1–17. doi: 10.1016/j.ypmed.2018.05.023. [DOI] [PubMed] [Google Scholar]
- 6.Racine N, Plamondon A, Madigan S, McDonald S, Tough S. Maternal adverse childhood experiences and infant development. Pediatrics. 2018;141(4):e20172495. doi: 10.1542/peds.2017-2495. [DOI] [PubMed] [Google Scholar]
- 7.Stephenson J, Heslehurst N, Hall J, Schoenaker D, Hutchinson J, Cade JE, et al. Before the beginning: nutrition and lifestyle in the preconception period and its importance for future health. Lancet. 2018;391(10132):1830–1841. doi: 10.1016/S0140-6736(18)30311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Flaviano M, Harville EW. Adverse Childhood Experiences on Reproductive Plans and Adolescent Pregnancy in the Gulf Resilience on Women’s Health Cohort. Int J Environ Res Public Health. 2020;18(1):165. doi: 10.3390/ijerph18010165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bridge-Comer PE, Vickers MH, Reynolds CM. Preclinical models of altered early life nutrition and development of reproductive disorders in female offspring. Adv Exp Med Biol. 2019;1134:59–87. doi: 10.1007/978-3-030-12668-1_4. [DOI] [PubMed] [Google Scholar]
- 10.Chan KA, Tsoulis MW, Sloboda DM. Early-life nutritional effects on the female reproductive system. J Endocrinol. 2015;224(2):R45–62. doi: 10.1530/JOE-14-0469. [DOI] [PubMed] [Google Scholar]
- 11.Hayward AD, Rickard IJ, Lummaa V. Influence of early-life nutrition on mortality and reproductive success during a subsequent famine in a preindustrial population. Proc Natl Acad Sci U S A. 2013;110(34):13886–13891. doi: 10.1073/pnas.1301817110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Elias SG, van Noord PA, Peeters PH, den Tonkelaar I, Grobbee DE. Childhood exposure to the 1944–1945 Dutch famine and subsequent female reproductive function. Hum Reprod. 2005;20(9):2483–2488. doi: 10.1093/humrep/dei090. [DOI] [PubMed] [Google Scholar]
- 13.Thoms H. The diagnosis of rachitic pelves by the x-ray. Am J Obstet Gynecol. 1927;14(1):45–48. [Google Scholar]
- 14.Felt BT, Peirano P, Algarín C, Chamorro R, Sir T, Kaciroti N, et al. Long-term neuroendocrine effects of iron-deficiency anemia in infancy. Pediatr Res. 2012;71(6):707–712. doi: 10.1038/pr.2012.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Micali N, Simonoff E, Treasure J. Risk of major adverse perinatal outcomes in women with eating disorders. Br J Psychiatry. 2007;190:255–259. doi: 10.1192/bjp.bp.106.020768. [DOI] [PubMed] [Google Scholar]
- 16.Thornburg LL. Antepartum obstetrical complications associated with obesity. Semin Perinatol. 2011;35(6):317–323. doi: 10.1053/j.semperi.2011.05.015. [DOI] [PubMed] [Google Scholar]
- 17.Miller GE, Culhane J, Grobman W, Simhan H, Williamson DE, Adam EK, et al. Mothers' childhood hardship forecasts adverse pregnancy outcomes: role of inflammatory, lifestyle, and psychosocial pathways. Brain Behav Immun. 2017;65:11–19. doi: 10.1016/j.bbi.2017.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Collins JW, Jr, Colgan J, Rankin KM, Desisto C. Affluent-born white mother's descending neighborhood income and infant mortality: a population-based study. Matern Child Health J. 2018;22(10):1484–1491. doi: 10.1007/s10995-018-2544-8. [DOI] [PubMed] [Google Scholar]
- 19.Collins JW, Rankin KM, David RJ. Downward economic mobility and preterm birth: an exploratory study of Chicago-born upper class White mothers. Matern Child Health J. 2015;19(7):1601–1607. doi: 10.1007/s10995-015-1670-9. [DOI] [PubMed] [Google Scholar]
- 20.Osypuk TL, Slaughter-Acey JC, Kehm RD, Misra DP. Life-course social mobility and reduced risk of adverse birth outcomes. Am J Prev Med. 2016;51(6):975–982. doi: 10.1016/j.amepre.2016.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Collins JW, Jr, Rankin KM, David RJ. African American women's lifetime upward economic mobility and preterm birth: the effect of fetal programming. Am J Public Health. 2011;101(4):714–719. doi: 10.2105/AJPH.2010.195024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ncube CN, Enquobahrie DA, Burke JG, Ye F, Marx J, Albert SM. Transgenerational transmission of preterm birth risk: the role of race and generational socio-economic neighborhood context. Matern Child Health J. 2017;21(8):1616–1626. doi: 10.1007/s10995-016-2251-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Love C, David RJ, Rankin KM, Collins JW., Jr Exploring weathering: effects of lifelong economic environment and maternal age on low birth weight, small for gestational age, and preterm birth in African-American and white women. Am J Epidemiol. 2010;172(2):127–134. doi: 10.1093/aje/kwq109. [DOI] [PubMed] [Google Scholar]
- 24.Shrimali BP, Pearl M, Karasek D, Reid C, Abrams B, Mujahid M. Neighborhood privilege, preterm delivery, and related racial/ethnic disparities: an intergenerational application of the index of concentration at the extremes. Am J Epidemiol. 2020;189(5):412–421. doi: 10.1093/aje/kwz279. [DOI] [PubMed] [Google Scholar]
- 25.Nesari M, Olson JK, Vandermeer B, Slater L, Olson DM. Does a maternal history of abuse before pregnancy affect pregnancy outcomes? A systematic review with meta-analysis. BMC Pregnancy Childbirth. 2018;18(1):404. doi: 10.1186/s12884-018-2030-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leeners B, Stiller R, Block E, Görres G, Rath W. Pregnancy complications in women with childhood sexual abuse experiences. J Psychosom Res. 2010;69(5):503–510. doi: 10.1016/j.jpsychores.2010.04.017. [DOI] [PubMed] [Google Scholar]
- 27.Leeners B, Rath W, Block E, Görres G, Tschudin S. Risk factors for unfavorable pregnancy outcome in women with adverse childhood experiences. J Perinat Med. 2014;42(2):171–178. doi: 10.1515/jpm-2013-0003. [DOI] [PubMed] [Google Scholar]
- 28.Liu C, Vinnerljung B, Östberg V, Gauffin K, Juarez S, Cnattingius S, et al. Out-of-home care and subsequent preterm delivery: an intergenerational cohort study. Pediatrics. 2018;142(2):e20172729. doi: 10.1542/peds.2017-2729. [DOI] [PubMed] [Google Scholar]
- 29.Christiaens I, Hegadoren K, Olson DM. Adverse childhood experiences are associated with spontaneous preterm birth: a case-control study. BMC Med. 2015;13:124. doi: 10.1186/s12916-015-0353-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Madigan S, Wade M, Plamondon A, Maguire JL, Jenkins JM. Maternal adverse childhood experience and infant health: biomedical and psychosocial risks as intermediary mechanisms. J Pediatr. 2017;187:282–9.e1. doi: 10.1016/j.jpeds.2017.04.052. [DOI] [PubMed] [Google Scholar]
- 31.Miller E, Levenson R, Herrera L, Kurek L, Stofflet M, Marin L. Exposure to partner, family, and community violence: gang-affiliated Latina women and risk of unintended pregnancy. J Urban Health. 2012;89(1):74–86. doi: 10.1007/s11524-011-9631-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Brown MJ, Masho SW, Perera RA, Mezuk B, Cohen SA. Sex and sexual orientation disparities in adverse childhood experiences and early age at sexual debut in the United States: results from a nationally representative sample. Child Abuse Negl. 2015;46:89–102. doi: 10.1016/j.chiabu.2015.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pear VA, Petito LC, Abrams B. The role of maternal adverse childhood experiences and race in intergenerational high-risk smoking behaviors. Nicotine Tob Res. 2017;19(5):623–630. doi: 10.1093/ntr/ntw295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Frankenberger DJ, Clements-Nolle K, Yang W. The association between adverse childhood experiences and alcohol use during pregnancy in a representative sample of adult women. Womens Health Issues. 2015;25(6):688–695. doi: 10.1016/j.whi.2015.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Schulkind L. Getting a sporting chance: Title IX and the intergenerational transmission of health. Health Econ. 2017;26(12):1583–1600. doi: 10.1002/hec.3453. [DOI] [PubMed] [Google Scholar]
- 36.Hyland A, Piazza KM, Hovey KM, Ockene JK, Andrews CA, Rivard C, et al. Associations of lifetime active and passive smoking with spontaneous abortion, stillbirth and tubal ectopic pregnancy: a cross-sectional analysis of historical data from the Women's Health Initiative. Tob Control. 2015;24(4):328–335. doi: 10.1136/tobaccocontrol-2013-051458. [DOI] [PubMed] [Google Scholar]
- 37.Meeker JD, Missmer SA, Vitonis AF, Cramer DW, Hauser R. Risk of spontaneous abortion in women with childhood exposure to parental cigarette smoke. Am J Epidemiol. 2007;166(5):571–575. doi: 10.1093/aje/kwm128. [DOI] [PubMed] [Google Scholar]
- 38.Ye X, Skjaerven R, Basso O, Baird DD, Eggesbo M, CupulUicab LA, et al. In utero exposure to tobacco smoke and subsequent reduced fertility in females. Hum Reprod. 2010;25(11):2901–2906. doi: 10.1093/humrep/deq235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tweed S, Bhattacharya S, Fowler PA. Effects of maternal smoking on offspring reproductive outcomes: an intergenerational study in the North East of Scotland. Hum Reprod Open. 2017;2017(2):hox006. doi: 10.1093/hropen/hox006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hawkins Bressler L, Bernardi LA, De Chavez PJ, Baird DD, Carnethon MR, Marsh EE. Alcohol, cigarette smoking, and ovarian reserve in reproductive-age African-American women. Am J Obstet Gynecol. 2016;215(6):758.e1–.e9. doi: 10.1016/j.ajog.2016.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gulson BL, Mizon KJ, Korsch MJ, Palmer JM, Donnelly JB. Mobilization of lead from human bone tissue during pregnancy and lactation–a summary of long-term research. Sci Total Environ. 2003;303(1–2):79–104. doi: 10.1016/s0048-9697(02)00355-8. [DOI] [PubMed] [Google Scholar]
- 42.Vahter M. Effects of arsenic on maternal and fetal health. Annu Rev Nutr. 2009;29:381–399. doi: 10.1146/annurev-nutr-080508-141102. [DOI] [PubMed] [Google Scholar]
- 43.Giudice LC. Environmental toxicants: hidden players on the reproductive stage. Fertil Steril. 2016;106(4):791–794. doi: 10.1016/j.fertnstert.2016.08.019. [DOI] [PubMed] [Google Scholar]
- 44.Gennings C, Carrico C, Factor-Litvak P, Krigbaum N, Cirillo PM, Cohn BA. A cohort study evaluation of maternal PCB exposure related to time to pregnancy in daughters. Environ Health. 2013;12(1):66. doi: 10.1186/1476-069X-12-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kundiev YI, Chernyuk VI, Karakashyan AN, Martynovskaya TY. Chornobyl and reproductive health of a female rural population (an epidemiological study) Probl Radiat Med Radiobiol. 2013;18:102–118. [PubMed] [Google Scholar]
- 46.Segars J, Katler Q, McQueen DB, Kotlyar A, Glenn T, Knight Z, et al. Prior and novel coronaviruses, coronavirus disease 2019 (COVID-19), and human reproduction: what is known? Fertil Steril. 2020;113(6):1140–1149. doi: 10.1016/j.fertnstert.2020.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kushnir VA, Lewis W. Human immunodeficiency virus/acquired immunodeficiency syndrome and infertility: emerging problems in the era of highly active antiretrovirals. Fertil Steril. 2011;96(3):546–553. doi: 10.1016/j.fertnstert.2011.05.094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cejtin HE, Kalinowski A, Bacchetti P, Taylor RN, Watts DH, Kim S, et al. Effects of human immunodeficiency virus on protracted amenorrhea and ovarian dysfunction. Obstet Gynecol. 2006;108(6):1423–1431. doi: 10.1097/01.AOG.0000245442.29969.5c. [DOI] [PubMed] [Google Scholar]
- 49.Uthman OA, Nachega JB, Anderson J, Kanters S, Mills EJ, Renaud F, et al. Timing of initiation of antiretroviral therapy and adverse pregnancy outcomes: a systematic review and meta-analysis. Lancet HIV. 2017;4(1):e21–e30. doi: 10.1016/S2352-3018(16)30195-3. [DOI] [PubMed] [Google Scholar]
- 50.Grace GA, Devaleenal DB, Natrajan M. Genital tuberculosis in females. Indian J Med Res. 2017;145(4):425–436. doi: 10.4103/ijmr.IJMR_1550_15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Li K, Chen G, Hou H, Liao Q, Chen J, Bai H, et al. Analysis of sex hormones and menstruation in COVID-19 women of child-bearing age. Reprod Biomed Online. 2021;42(1):260–267. doi: 10.1016/j.rbmo.2020.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sharma I, Kumari P, Sharma A, Saha SC. SARS-CoV-2 and the reproductive system: known and the unknown..!! Middle East Fertil Soc J. 2021;26(1):1. doi: 10.1186/s43043-020-00046-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Herr D, Bekes I, Wulff C. Local Renin-Angiotensin system in the reproductive system. Front Endocrinol (Lausanne) 2013;4:150. doi: 10.3389/fendo.2013.00150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Li R, Yin T, Fang F, Li Q, Chen J, Wang Y, et al. Potential risks of SARS-CoV-2 infection on reproductive health. Reprod Biomed Online. 2020;41(1):89–95. doi: 10.1016/j.rbmo.2020.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rizzi S, Wensink MJ, Lindahl-Jacobsen R, Tian L, Lu Y, Eisenberg ML. Risk of pre-term births and major birth defects resulting from paternal intake of COVID-19 medications prior to conception. BMC Res Notes. 2020;13(1):509. doi: 10.1186/s13104-020-05358-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cook CJ, Fletcher JM, Forgues A. Multigenerational effects of early-life health shocks. Demography. 2019;56(5):1855–1874. doi: 10.1007/s13524-019-00804-3. [DOI] [PubMed] [Google Scholar]
- 57.Thayer Z, Bécares L, Atatoa CP. Maternal experiences of ethnic discrimination and subsequent birth outcomes in Aotearoa New Zealand. BMC Public Health. 2019;19(1):1271. doi: 10.1186/s12889-019-7598-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Alhusen JL, Bower KM, Epstein E, Sharps P. Racial discrimination and adverse birth outcomes: an integrative review. J Midwifery Womens Health. 2016;61(6):707–720. doi: 10.1111/jmwh.12490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Higgins JA, Carpenter E, Everett BG, Greene MZ, Haider S, Hendrick CE. Sexual minority women and contraceptive use: complex pathways between sexual orientation and health outcomes. Am J Public Health. 2019;109(12):1680–1686. doi: 10.2105/AJPH.2019.305211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.McDade TW, Borja JB, Largado F, Adair LS, Kuzawa CW. Adiposity and chronic inflammation in young women predict inflammation during normal pregnancy in the philippines. J Nutr. 2016;146(2):353–357. doi: 10.3945/jn.115.224279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bublitz MH, Parade S, Stroud LR. The effects of childhood sexual abuse on cortisol trajectories in pregnancy are moderated by current family functioning. Biol Psychol. 2014;103:152–157. doi: 10.1016/j.biopsycho.2014.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Schreier HM, Enlow MB, Ritz T, Gennings C, Wright RJ. Childhood abuse is associated with increased hair cortisol levels among urban pregnant women. J Epidemiol Community Health. 2015;69(12):1169–1174. doi: 10.1136/jech-2015-205541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Fowler PA, Childs AJ, Courant F, MacKenzie A, Rhind SM, Antignac JP, et al. In utero exposure to cigarette smoke dysregulates human fetal ovarian developmental signalling. Hum Reprod. 2014;29(7):1471–1489. doi: 10.1093/humrep/deu117. [DOI] [PubMed] [Google Scholar]
- 64.Donley GM, Taylor E, Jeddy Z, Namulanda G, Hartman TJ. Association between in utero perfluoroalkyl substance exposure and anti-Müllerian hormone levels in adolescent females in a British cohort. Environ Res. 2019;177:108585. doi: 10.1016/j.envres.2019.108585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Steegers-Theunissen RP, Twigt J, Pestinger V, Sinclair KD. The periconceptional period, reproduction and long-term health of offspring: the importance of one-carbon metabolism. Hum Reprod Update. 2013;19(6):640–655. doi: 10.1093/humupd/dmt041. [DOI] [PubMed] [Google Scholar]
- 66.Penkler M, Hanson M, Biesma R, Müller R. DOHaD in science and society: emergent opportunities and novel responsibilities. J Dev Orig Health Dis. 2019;10(3):268–273. doi: 10.1017/S2040174418000892. [DOI] [PubMed] [Google Scholar]
- 67.De Stavola BL, Leon DA, Koupil I. Intergenerational correlations in size at birth and the contribution of environmental factors: the Uppsala Birth Cohort Multigenerational Study, Sweden, 1915–2002. Am J Epidemiol. 2011;174(1):52–62. doi: 10.1093/aje/kwr032. [DOI] [PubMed] [Google Scholar]
- 68.Vagero D, Pinger PR, Aronsson V, van den Berg GJ. Paternal grandfather's access to food predicts all-cause and cancer mortality in grandsons. Nat Commun. 2018;9(1):5124. doi: 10.1038/s41467-018-07617-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Naess O, Stoltenberg C, Hoff DA, Nystad W, Magnus P, Tverdal A, et al. Cardiovascular mortality in relation to birth weight of children and grandchildren in 500,000 Norwegian families. Eur Heart J. 2013;34(44):3427–3436. doi: 10.1093/eurheartj/ehs298. [DOI] [PubMed] [Google Scholar]
- 70.Andrasfay T, Goldman N. Intergenerational change in birthweight: effects of foreign-born status and race/ethnicity. Epidemiology. 2020;31(5):649–658. doi: 10.1097/EDE.0000000000001217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Giuntella O. Why does the health of Mexican immigrants deteriorate? New evidence from linked birth records. J Health Econ. 2017;54:1–16. doi: 10.1016/j.jhealeco.2017.03.002. [DOI] [PubMed] [Google Scholar]
- 72.Fleischer NL, Abshire C, Margerison CE, Nitcheva D, Smith MG. The South Carolina multigenerational linked birth dataset: developing social mobility measures across generations to understand racial/ethnic disparities in adverse birth outcomes in the US South. Matern Child Health J. 2019;23(6):787–801. doi: 10.1007/s10995-018-02695-z. [DOI] [PubMed] [Google Scholar]
- 73.Agius R, Savona-Ventura C, Vassallo J. Transgenerational metabolic determinants of fetal birth weight. Exp Clin Endocrinol Diabetes. 2013;121(7):431–435. doi: 10.1055/s-0033-1345121. [DOI] [PubMed] [Google Scholar]
- 74.Kane JB. An integrative model of inter- and intragenerational preconception processes influencing birthweight in the United States. J Health Soc Behav. 2015;56(2):246–261. doi: 10.1177/0022146515582043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.McGee G, Perkins NJ, Mumford SL, Kioumourtzoglou MA, Weisskopf MG, Schildcrout JS, et al. Methodological issues in population-based studies of multigenerational associations. Am J Epidemiol. 2020;189(12):1600–1609. doi: 10.1093/aje/kwaa125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Zambrano E, Martinez-Samayoa PM, Bautista CJ, Deas M, Guillen L, Rodriguez-Gonzalez GL, et al. Sex differences in transgenerational alterations of growth and metabolism in progeny (F2) of female offspring (F1) of rats fed a low protein diet during pregnancy and lactation. J Physiol. 2005;566(Pt 1):225–236. doi: 10.1113/jphysiol.2005.086462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.McGee G, Weisskopf MG, Kioumourtzoglou MA, Coull BA, Haneuse S. Informatively empty clusters with application to multigenerational studies. Biostatistics. 2020;21(4):775–789. doi: 10.1093/biostatistics/kxz005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hallqvist J, Lynch J, Bartley M, Lang T, Blane D. Can we disentangle life course processes of accumulation, critical period and social mobility? An analysis of disadvantaged socio-economic positions and myocardial infarction in the Stockholm Heart Epidemiology Program. Soc Sci Med. 2004;58(8):1555–1562. doi: 10.1016/S0277-9536(03)00344-7. [DOI] [PubMed] [Google Scholar]
- 79.Relton CL, Davey SG. Two-step epigenetic Mendelian randomization: a strategy for establishing the causal role of epigenetic processes in pathways to disease. Int J Epidemiol. 2012;41(1):161–176. doi: 10.1093/ije/dyr233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Peeling RW, Mabey D, Kamb ML, Chen XS, Radolf JD, Benzaken AS. Syphilis. Nat Rev Dis Primers. 2017;3:17073. doi: 10.1038/nrdp.2017.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Rothenberg SJ, Kondrashov V, Manalo M, Jiang J, Cuellar R, Garcia M, et al. Increases in hypertension and blood pressure during pregnancy with increased bone lead levels. Am J Epidemiol. 2002;156(12):1079–1087. doi: 10.1093/aje/kwf163. [DOI] [PubMed] [Google Scholar]
- 82.Harville EW, Boynton-Jarrett R, Power C, Hypponen E. Childhood hardship, maternal smoking, and birth outcomes: a prospective cohort study. Arch Pediatr Adolesc Med. 2010;164(6):533–539. doi: 10.1001/archpediatrics.2010.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Clayborne ZM, Giesbrecht GF, Bell RC, Tomfohr-Madsen LM. Relations between neighbourhood socioeconomic status and birth outcomes are mediated by maternal weight. Soc Sci Med. 2017;175:143–151. doi: 10.1016/j.socscimed.2016.12.041. [DOI] [PubMed] [Google Scholar]
- 84.Mason SM, Tobias DK, Clark CJ, Zhang C, Hu FB, Rich-Edwards JW. Abuse in childhood or adolescence and gestational diabetes: a retrospective cohort study. Am J Prev Med. 2016;50(4):436–444. doi: 10.1016/j.amepre.2015.08.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Fraser A, McNally W, Sattar N, Anderson EL, Lashen H, Fleming R, et al. Prenatal exposures and anti-Mullerian hormone in female adolescents: the Avon Longitudinal Study of Parents and Children. Am J Epidemiol. 2013;178(9):1414–1423. doi: 10.1093/aje/kwt137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pedersen JM, Mortensen EL, Christensen DS, Rozing M, Brunsgaard H, Meincke RH, et al. Prenatal and early postnatal stress and later life inflammation. Psychoneuroendocrinology. 2018;88:158–166. doi: 10.1016/j.psyneuen.2017.12.014. [DOI] [PubMed] [Google Scholar]
- 87.Park H, Park KH, Kim YM, Kook SY, Jeon SJ, Yoo HN. Plasma inflammatory and immune proteins as predictors of intra-amniotic infection and spontaneous preterm delivery in women with preterm labor: a retrospective study. BMC Pregnancy Childbirth. 2018;18(1):146. doi: 10.1186/s12884-018-1780-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Sominsky L, Fuller EA, Hodgson DM. Factors in early-life programming of reproductive fitness. Neuroendocrinology. 2015;102(3):216–225. doi: 10.1159/000431378. [DOI] [PubMed] [Google Scholar]
- 89.Yehuda R, Lehrner A. Intergenerational transmission of trauma effects: putative role of epigenetic mechanisms. World Psychiatry. 2018;17(3):243–257. doi: 10.1002/wps.20568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Cunliffe VT. The epigenetic impacts of social stress: how does social adversity become biologically embedded? Epigenomics. 2016;8(12):1653–1669. doi: 10.2217/epi-2016-0075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Burris HH, Baccarelli AA, Byun HM, Cantoral A, Just AC, Pantic I, et al. Offspring DNA methylation of the aryl-hydrocarbon receptor repressor gene is associated with maternal BMI, gestational age, and birth weight. Epigenetics. 2015;10(10):913–921. doi: 10.1080/15592294.2015.1078963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Hyppönen E, Smith GD, Power C. Effects of grandmothers' smoking in pregnancy on birth weight: intergenerational cohort study. BMJ. 2003;327(7420):898. doi: 10.1136/bmj.327.7420.898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Brown MM, Woolcott CG, Dodds L, Ashley-Martin J, Allen VM, Fahey J, et al. The 3G Multigenerational Cohort of Nova Scotian women and their mothers and offspring. Paediatr Perinat Epidemiol. 2020;34(2):214–221. doi: 10.1111/ppe.12647. [DOI] [PubMed] [Google Scholar]
- 94.Morton SM, De Stavola BL, Leon DA. Intergenerational determinants of offspring size at birth: a life course and graphical analysis using the Aberdeen Children of the 1950s Study (ACONF) Int J Epidemiol. 2014;43(3):749–759. doi: 10.1093/ije/dyu028. [DOI] [PubMed] [Google Scholar]
- 95.McCarron P, Davey Smith G, Hattersley AT. Type 2 diabetes in grandparents and birth weight in offspring and grandchildren in the ALSPAC study. J Epidemiol Community Health. 2004;58(6):517–522. doi: 10.1136/jech.2003.007989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Harville EW, Jacobs MB, Qi L, Chen W, Bazzano LA. Multigenerational cardiometabolic risk as a predictor of birth outcomes: the Bogalusa Heart Study. J Pediatr. 2017;181:154–62.e1. doi: 10.1016/j.jpeds.2016.10.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Harville EW, Apolzan JW, Bazzano LA. Maternal Pre-pregnancy cardiovascular risk factors and offspring and grandoffspring health: Bogalusa daughters. Int J Environ Res Public Health. 2018;16(1):15. doi: 10.3390/ijerph16010015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Kwok MK, Leung GM, Lam TH, Leung SS, Schooling CM. Grandparental education, parental education and child height: evidence from Hong Kong's "Children of 1997" birth cohort. Ann Epidemiol. 2013;23(8):475–484. doi: 10.1016/j.annepidem.2013.05.016. [DOI] [PubMed] [Google Scholar]
- 99.Kuzawa CW, Tallman PS, Adair LS, Lee N, McDade TW. Inflammatory profiles in the non-pregnant state predict offspring birth weight at Cebu: evidence for inter-generational effects of low grade inflammation. Ann Hum Biol. 2012;39(4):267–274. doi: 10.3109/03014460.2012.692810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Borja JB. The impact of early nutrition on health: key findings from the Cebu Longitudinal Health and Nutrition Survey (CLHNS) Malays J Nutr. 2013;19(1):1–8. [PubMed] [Google Scholar]
- 101.Klebanoff MA, Mednick BR, Schulsinger C, Secher NJ, Teasdale TW, Baker RL, et al. Second generation follow-up of the Danish perinatal study women: study design and factors affecting response. Paediatr Perinat Epidemiol. 1993;7(1):9–22. doi: 10.1111/j.1365-3016.1993.tb00596.x. [DOI] [PubMed] [Google Scholar]
- 102.Titus-Ernstoff L, Troisi R, Hatch EE, Palmer JR, Hyer M, Kaufman R, et al. Birth defects in the sons and daughters of women who were exposed in utero to diethylstilbestrol (DES) Int J Androl. 2010;33(2):377–384. doi: 10.1111/j.1365-2605.2009.01010.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Titus L, Hatch EE, Drake KM, Parker SE, Hyer M, Palmer JR, et al. Reproductive and hormone-related outcomes in women whose mothers were exposed in utero to diethylstilbestrol (DES): a report from the US National Cancer Institute DES Third Generation Study. Reprod Toxicol. 2019;84:32–38. doi: 10.1016/j.reprotox.2018.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Lumey LH, Stein AD. In utero exposure to famine and subsequent fertility: The Dutch Famine Birth Cohort Study. Am J Public Health. 1997;87(12):1962–1966. doi: 10.2105/ajph.87.12.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lumey LH, Stein AD, Ravelli AC. Timing of prenatal starvation in women and birth weight in their first and second born offspring: the Dutch Famine Birth Cohort study. Eur J Obstet Gynecol Reprod Biol. 1995;61(1):23–30. doi: 10.1016/0028-2243(95)02149-m. [DOI] [PubMed] [Google Scholar]
- 106.Perng W, Tamayo-Ortiz M, Tang L, Sanchez BN, Cantoral A, Meeker JD, et al. Early Life Exposure in Mexico to ENvironmental Toxicants (ELEMENT) Project. BMJ Open. 2019;9(8):e030427. doi: 10.1136/bmjopen-2019-030427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Giuntella O. Assimilation and health: evidence from linked birth records of second- and third-generation Hispanics. Demography. 2016;53(6):1979–2004. doi: 10.1007/s13524-016-0522-6. [DOI] [PubMed] [Google Scholar]
- 108.Huang JY, Gavin AR, Richardson TS, Rowhani-Rahbar A, Siscovick DS, Enquobahrie DA. Are early-life socioeconomic conditions directly related to birth outcomes? Grandmaternal education, grandchild birth weight, and associated bias analyses. Am J Epidemiol. 2015;182(7):568–578. doi: 10.1093/aje/kwv148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Townsend ML, Kelly MA, Pickard JA, Larkin TA, Flood VM, Caputi P, et al. Illawarra Born cross-generational health study: feasibility of a multi-generational birth cohort study. Pilot Feasibility Stud. 2019;5:32. doi: 10.1186/s40814-019-0418-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Collins JW, Rankin KM, David RJ. Low birth weight across generations: the effect of economic environment. Matern Child Health J. 2011;15(4):438–445. doi: 10.1007/s10995-010-0603-x. [DOI] [PubMed] [Google Scholar]
- 111.Collins JW, Jr, David RJ, Mendivil NA, Wu SY. Intergenerational birth weights among the direct female descendants of US-born and Mexican-born Mexican-American women in Illinois: an exploratory study. Ethn Dis. 2006;16(1):166–171. [PubMed] [Google Scholar]
- 112.Collins JW, Jr, Wu SY, David RJ. Differing intergenerational birth weights among the descendants of US-born and foreign-born Whites and African Americans in Illinois. Am J Epidemiol. 2002;155(3):210–216. doi: 10.1093/aje/155.3.210. [DOI] [PubMed] [Google Scholar]
- 113.Coutinho R, David RJ, Collins JW., Jr Relation of parental birth weights to infant birth weight among African Americans and whites in Illinois: a transgenerational study. Am J Epidemiol. 1997;146(10):804–809. doi: 10.1093/oxfordjournals.aje.a009197. [DOI] [PubMed] [Google Scholar]
- 114.Shen Y, Zhang H, Jiang Y, Mzayek F, Arshad H, Karmaus W. Maternal birth weight and BMI mediate the transgenerational effect of grandmaternal BMI on grandchild's birth weight. Obesity. 2020;28(3):647–654. doi: 10.1002/oby.22680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Hochner H, Friedlander Y, Calderon-Margalit R, Meiner V, Sagy Y, Avgil-Tsadok M, et al. Associations of maternal prepregnancy body mass index and gestational weight gain with adult offspring cardiometabolic risk factors: the Jerusalem Perinatal Family Follow-up Study. Circulation. 2012;125(11):1381–1389. doi: 10.1161/CIRCULATIONAHA.111.070060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Lee C. Intergenerational health consequences of in utero exposure to maternal stress: evidence from the 1980 Kwangju uprising. Soc Sci Med. 2014;119:284–291. doi: 10.1016/j.socscimed.2014.07.001. [DOI] [PubMed] [Google Scholar]
- 117.Straughen JK, Caldwell CH, Osypuk TL, Helmkamp L, Misra DP. Direct and proxy recall of childhood socio-economic position and health. Paediatr Perinat Epidemiol. 2013;27(3):294–302. doi: 10.1111/ppe.12045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Warmink-Perdijk WDB, Peters LL, Tigchelaar EF, Dekens JAM, Jankipersadsing SA, Zhernakova A, et al. Lifelines NEXT: a prospective birth cohort adding the next generation to the three-generation Lifelines cohort study. Eur J Epidemiol. 2020;35(2):157–168. doi: 10.1007/s10654-020-00614-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Ozasa K, Cullings HM, Ohishi W, Hida A, Grant EJ. Epidemiological studies of atomic bomb radiation at the Radiation Effects Research Foundation. Int J Radiat Biol. 2019;95(7):879–891. doi: 10.1080/09553002.2019.1569778. [DOI] [PubMed] [Google Scholar]
- 120.Kelly GE, Murrin C, Viljoen K, O'Brien J, Kelleher C. Body mass index is associated with the maternal lines but height is heritable across family lines in the Lifeways Cross-Generation Cohort Study. BMJ Open. 2014;4(12):e005732. doi: 10.1136/bmjopen-2014-005732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Navarro P, Mehegan J, Murrin CM, Kelleher CC, Phillips CM. Adherence to the Healthy Eating Index-2015 across generations is associated with birth outcomes and weight status at age 5 in the Lifeways Cross-Generation Cohort Study. Nutrients. 2019;11(4):928. doi: 10.3390/nu11040928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Najman JM, Alati R, Bor W, Clavarino A, Mamun A, McGrath JJ, et al. Cohort profile update: the Mater-University of Queensland Study of Pregnancy (MUSP) Int J Epidemiol. 2015;44(1):78-f. doi: 10.1093/ije/dyu234. [DOI] [PubMed] [Google Scholar]
- 123.Rillamas-Sun E, Harlow SD, Randolph JF., Jr Grandmothers' smoking in pregnancy and grandchildren's birth weight: comparisons by grandmother birth cohort. Matern Child Health J. 2014;18(7):1691–1698. doi: 10.1007/s10995-013-1411-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Chavarro JE, Rich-Edwards JW, Gaskins AJ, Farland LV, Terry KL, Zhang C, et al. Contributions of the Nurses' Health Studies to reproductive health research. Am J Public Health. 2016;106(9):1669–1676. doi: 10.2105/AJPH.2016.303350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Liu D, Lin G, Su D, Alexender JM, Sun X, Qu M. Intergenerational associations of adverse birth outcomes: a surveillance report. Prev Med Rep. 2020;20:101226. doi: 10.1016/j.pmedr.2020.101226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Jain NJ, Faiz AS, Ohman-Strickland PA, Smulian JC, Rhoads GG. Educational attainment of grandmothers and preterm birth in grandchildren. Matern Child Health J. 2020;25(2):293–301. doi: 10.1007/s10995-020-03021-2. [DOI] [PubMed] [Google Scholar]
- 127.Magnus P, Birke C, Vejrup K, Haugan A, Alsaker E, Daltveit AK, et al. Cohort profile update: the Norwegian Mother and Child Cohort Study (MoBa) Int J Epidemiol. 2016;45(2):382–388. doi: 10.1093/ije/dyw029. [DOI] [PubMed] [Google Scholar]
- 128.Astone NM, Misra D, Lynch C. The effect of maternal socio-economic status throughout the lifespan on infant birthweight. Paediatr Perinat Epidemiol. 2007;21(4):310–318. doi: 10.1111/j.1365-3016.2007.00821.x. [DOI] [PubMed] [Google Scholar]
- 129.Misra DP, Astone N, Lynch CD. Maternal smoking and birth weight: interaction with parity and mother's own in utero exposure to smoking. Epidemiology. 2005;16(3):288–293. doi: 10.1097/01.ede.0000158198.59544.cf. [DOI] [PubMed] [Google Scholar]
- 130.Qian M, Chou SY, Deily ME, Liu JT. Gender inequality, economic growth, and the intergenerational transmission of adverse health consequences at birth. Soc Sci Med. 1982;2018(200):174–181. doi: 10.1016/j.socscimed.2018.01.020. [DOI] [PubMed] [Google Scholar]
- 131.Timm S, Svanes C, Frydenberg M, Sigsgaard T, Holm M, Janson C, et al. Does parental farm upbringing influence the risk of asthma in offspring? A three-generation study. Int J Epidemiol. 2020;49(6):1874–1882. doi: 10.1093/ije/dyaa091. [DOI] [PubMed] [Google Scholar]
- 132.Song S. Identifying the intergenerational effects of the 1959–1961 Chinese Great Leap Forward Famine on infant mortality. Econ Hum Biol. 2013;11(4):474–487. doi: 10.1016/j.ehb.2013.08.001. [DOI] [PubMed] [Google Scholar]
- 133.Gavin AR, Hill KG, Hawkins JD, Maas C. The role of maternal early-life and later-life risk factors on offspring low birth weight: findings from a three-generational study. J Adolesc Health. 2011;49(2):166–171. doi: 10.1016/j.jadohealth.2010.11.246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Patel B, Meeks H, Wan Y, Johnstone EB, Glenn M, Smith KR, et al. Transgenerational effects of chemotherapy: both male and female children born to women exposed to chemotherapy have fewer children. Cancer Epidemiol. 2018;56:1–5. doi: 10.1016/j.canep.2018.07.001. [DOI] [PubMed] [Google Scholar]
- 135.Schliep KC, Feldkamp ML, Hanson HA, Hollingshaus M, Fraser A, Smith KR, et al. Are paternal or grandmaternal age associated with higher probability of trisomy 21 in offspring? A population-based, matched case-control study, 1995–2015. Paediatr Perinat Epidemiol. 2021;35(3):281–291. doi: 10.1111/ppe.12737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Vågerö D, Rajaleid K. Does childhood trauma influence offspring's birth characteristics? Int J Epidemiol. 2017;46(1):219–229. doi: 10.1093/ije/dyw048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Chapman DA, Gray G. Developing a maternally linked birth dataset to study the generational recurrence of low birthweight in Virginia. Matern Child Health J. 2014;18(2):488–496. doi: 10.1007/s10995-013-1277-y. [DOI] [PubMed] [Google Scholar]
- 138.Wallace D. Discriminatory mass de-housing and low-weight births: scales of geography, time, and level. J Urban Health. 2011;88(3):454–468. doi: 10.1007/s11524-011-9581-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Emanuel I, Leisenring W, Williams MA, Kimpo C, Estee S, O'Brien W, et al. The Washington State Intergenerational Study of Birth Outcomes: methodology and some comparisons of maternal birthweight and infant birthweight and gestation in four ethnic groups. Paediatr Perinat Epidemiol. 1999;13(3):352–369. doi: 10.1046/j.1365-3016.1999.00184.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.