Abstract
A disintegrin and metalloproteinases (ADAMs) family are proteolytic transmembrane proteases that modulate diverse cell functions and coordinate intercellular communication. ADAMs are responsible for regulating cell proliferation, differentiation, migration, and organ morphogenesis in kidney development. Abnormally activated ADAMs drive inflammation and fibrosis in response to kidney diseases such as acute kidney injury, diabetic kidney disease, polycystic kidney disease, and chronic allograft nephropathy. ADAM10 and ADAM17, known as the most characterized members of ADAMs, are extensively investigated in kidney diseases. Notably, ADAM proteases have the potential to be targets for developing novel treatment approaches in kidney diseases.
Keywords: A disintegrin and metalloproteinases, Nephrogenesis, Inflammation, Fibrosis
1. Introduction
A disintegrin and metalloproteases (ADAMs) family belong to type I transmembrane and secreted metalloendopeptidases proteins, which mediate the cleaving of the extracellular domain (ectodomain) of membrane proteins. The ADAMs regulate several proteins, including growth factors, cytokines, receptors, and adhesion molecules, which play essential roles in numerous biological processes such as cell migration, cell adhesion, proteolysis and signal transduction. To date, ADAMs have been found in various mammalian genomes, e.g. 37 ADAMs in rats, 34 ADAMs in mice, and 21 ADAMs in humans. ADAMs are fundamental for normal development and morphogenesis, including sperm-egg interaction, embryonic development and differentiation, cell fate determination, and diverse aspects of immunity. Dysregulation of ADAMs is associated with multiple human pathologies such as cardiovascular and neurodegenerative diseases, cancer, asthma, inflammation, and kidney diseases, and thus provides potential therapeutic targets for these diseases.
ADAMs regulate and coordinate cellular signaling in response to changes in renal physiology and pathology. In this review, we focus on the physiological roles of ADAMs in renal embryological development and summarize the pathological roles of ADAMs in kidney diseases. We also discuss the potential and challenges in targeting ADAMs to prevent and treat kidney diseases.
2. ADAMs structure, regulation, and function
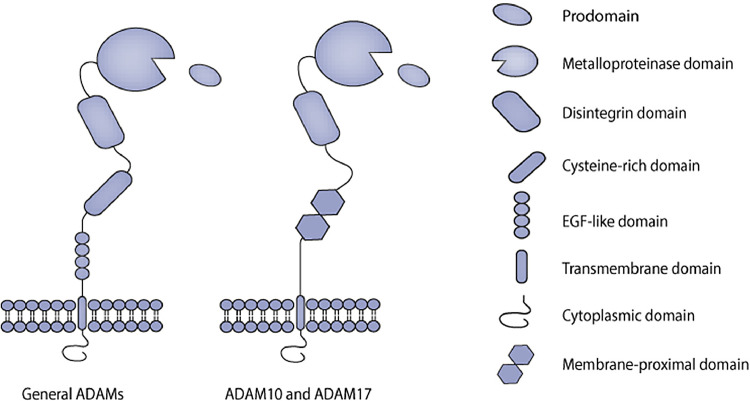
The typical structure of ADAMs consists of a prodomain, a metalloproteinase domain, a disintegrin domain, a cysteine-rich domain, an epidermal growth factor (EGF)-like domain, followed by a transmembrane region and a cytoplasmic tail (Fig. 1). The cysteine-rich domain and the EGF-like domain are replaced by the membrane-proximal domain in ADAM10 and ADAM17, which are considered atypical family members of ADAMs [1], [2], [3]. Structurally, the prodomain is followed by the metalloprotease domain, and their interaction is essential for the ADAMs proteolytic activity [2, 4]. To some degree, the disintegrin domain can selectively bind to integrins to mediate cell adhesion, cell migration, and cell-cell interactions. The cysteine-rich domain contains a hyper-variable region (HVR), that contributes to ADAMs function by recognizing their substrates and regulating the catalytic activity. This region may be involved in modulating subcellular localization and protein-protein interactions [5]. The different composition of the cytoplasmic tails is crucial for their varying functions in metalloprotease activity, intracellular signaling, and subcellular localization.
Fig. 1.
The structure of ADAM proteins. The typical multidomain of general ADAMs consists of a prodomain, a metalloproteinase domain, a disintegrin domain, a cysteine-rich domain, an epidermal growth factor (EGF)-like domain, followed by a transmembrane region and a cytoplasmic tail. While the cysteine-rich domain and the EGF-like domain are replaced by the membrane-proximal domain in ADAM10 and ADAM17.
The extracellular and intracellular pathways of ADAM activation are intricate, as regulation often occurs through conformational changes, activators or inhibitors. After biosynthesis in the endoplasmic reticulum and maturation in the Golgi compartment, ADAMs secrete as inactive zymogens. The cytoplasmic tail controls the retention of properly folded ADAMs in the Golgi compartment by an unidentified mechanism, which impedes the release of ADAMs activity at the cell membrane [6, 7]. The prodomain inhibits ADAMs activity via maintaining the metalloproteinase domain in an inactive conformation, and the prodomain cleavage by furin-type proteases, and PC7 releases the catalytical activity in a process occurring when transiting to the cell surface [1,4,8]. In addition to the effects of conformational changes on ADAMs activities, ADAMs activities also suppress by endogenous tissue inhibitors of metalloproteinases (TIMPs), which have a specific inhibitory effect on various ADAMs family members.
ADAM-mediated cleavage of ectodomain shedding is a process of releasing the extracellular domain by proteolytic cleavage. The diverse substrates of ADAMs include growth factors, cytokines, chemokines, adhesion molecules, proteins of the extracellular matrix, and receptors and ligands of signaling pathways, and thus functions of ADAMs ectodomain shedding play a prominent role not only in cell proliferation, differentiation, and adhesion but also in signal transduction. Moreover, ADAMs-mediated ectodomain shedding has been identified in exosomes, potentially contributing to intercellular communication [9], [10], [11]. In addition to their roles in ectodomain shedding, ADAMs also have the ability to modulate intracellular signal transduction. The ADAMs play a major role in transmembrane protein shedding, an initiating step for regulated intramembrane proteolysis (RIP) that releases intracellular domains and regulates intracellular signaling events. Notch signaling and amyloid-precursor-protein processing are typical examples of RIP.
In the past few years, ADAM10 and ADAM17 were the most studied family members. Several researchers have confirmed the roles of ADAM10 and ADAM17 in the renal physiological and pathological processes (Table 1). ADAM10 is expressed in renal tubular cells and is involved in several kidney diseases [12]. While ADAM17 is weakly expressed in proximal tubules, glomerular endothelium, and mesangium, its expression is markedly induced in interstitial fibrosis and tubular atrophy [13, 14]. The importance of ADAM-mediated signaling in the kidney has been recognized and highlighted. Next, we review recent insights into the molecular cell biology of ADAMs in the context of normal and abnormal kidneys, and discuss the precise functions of ADAM proteins in nephrogenesis, kidney cellular effects and kidney diseases separately.
Table 1.
Substrates of ADAMs in the kidneys
| ADAMs | Substrates | Roles in kidney development and disease | References |
|---|---|---|---|
| ADAM10 | Notch | Involved in metanephric development; determined the cell fate of the collecting ducts; promoted podocyte and mesangial cell maturation; aggravated renal fibrosis | [16, [19], [20], [21], 60] |
| CXCL16 | Promoted mesangial cells proliferation and migration; mediated inflammation factors production and podocyte migration; recruited of T cells to the kidney | [41, 45, 46] | |
| KIM-1 | Modulated the phagocytic process | [57] | |
| E-cadherin | Involved in kidney fibrosis | [59] | |
| Meprin A | Induced inflammation and matrix remodulation | [56] | |
| ADAM17 | EGFR ligands (HB-EGF and TGFα) | Promoted proinflammatory factors upregulation and inflammatory cell infiltration; promoted profibrotic factors release and induced interstitial fibrosis; induced glomerular matrix accumulation leading to glomerulosclerosis | [31], [32], [33], [34], [35] |
| ACE2 | Induced albuminuria; mediated the shedding of TCEs | [74] | |
| Notch | Involved in glomerular development | [30] | |
| CXCL16 | Promoted mesangial cells proliferation and migration; mediated inflammation factors production and podocyte migration | [41, 45] | |
| Nox4 | Enhanced oxidative stress; induced matrix accumulation | [32] | |
| TNFα | Promoted inflammation and fibrosis | [33] | |
| EPCR | Promoted glomerular endothelium damage | [68] |
3. ADAMs in nephrogenesis
ADAMs are essential proteases involved in embryo development via controlling cell proliferation, cell differentiation, cell migration and organ morphogenesis. Diverse phenotypes have been observed in ADAM knockout mice due to the broad expression of ADAMs in mammalian tissues, but only ADAM10, ADAM17, and ADAM19 null mice have defects in embryo development [15]. In this review, we focus on extensive information on the ADAM family during kidney development, and briefly summarize the roles of the two well-studied members, ADAM10 and ADAM17.
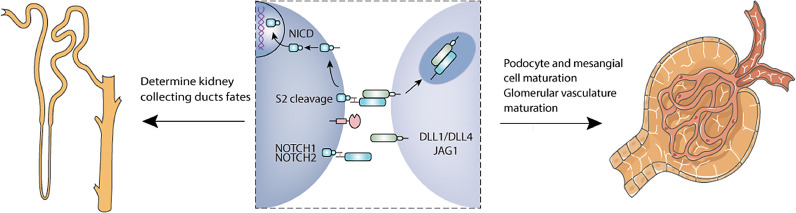
ADAM10 is best known for its function in ligand-dependent Notch S2 cleavage, which is responsible for mediating the Notch pathway. Notch signaling has multiple effects throughout the development of diverse tissues and cell types. In kidney development, Notch signaling controls proximal tubular epithelial cell fate as well as collecting duct cell composition. High expression of Notch2 is observed in the developing ureteral bud (UB) and ADAM10 is also highly expressed in the late UB, indicating that ADAM10 is involved in metanephric development [16]. Moreover, slit genes are expressed in the metanephric mesenchyme, while Robo1 is also found in the UB. Thus, ADAM10 may participate in regulating Slit-Robo signaling during nephrogenesis [17, 18]. It showed that ADAM10 deficiency in UB reduced principal cells/intercalated cells ratios in the collecting duct with reduction of Notch activity. ADAM10 deficiency mice present with polyuria and hydronephrosis, suggesting that these mice have defects in urinary concentration. Taken together, ADAM10 plays an indispensable role in determining the cell fate of collecting ducts, which is partially necessary for the normal development of renal collecting ducts [19]. During development, glomerular endothelial cells interact with mesangial cells and podocytes to generate the mature glomerular vascular structure. Normal Notch signaling is crucial for podocyte and mesangial cell maturation, which is essential for proper glomerular development [20, 21]. Notch1 deficiency, as well as inactivation of ADAM10 in endothelial cells, result in similar glomerular defects that are rescued by overexpression of Notch signaling in ADAM10 deficiency mice. These findings indicate that ADAM10-dependent Notch signaling plays a central role in developing glomerulus [22]. Furthermore, Farber's group reported that ADAM10/Notch signaling promoted the maturation of the glomerular vasculature [23]. In addition, increased expression of ADAM10 is detected in differentiated podocytes [24]. Therefore, ADAM10 contributes to diverse aspects of renal development (Fig. 2).
Fig. 2.
Roles of ADAM10 signaling in kidney development. The Notch receptor is activated by ligand binding after ADAM10 mediated the S2 site cleavage of Notch ligands and second gamma-secretase proteolytic cleavage at the S3 site. During nephrogenesis, ADAM10/Notch signaling is partially necessary for the normal development of renal collecting ducts. And normal Notch signaling is crucial for podocyte and mesangial cell maturation, which is essential for proper glomerular development. In addition, ADAM10/Notch signaling promotes the maturation of the glomerular vasculature.
ADAM17 participates in cell proliferation, differentiation and migration as a pleiotropic regulator of organ development, and these effects are mainly associated with EGFR signaling. ADAM17-deficient mice and EGFR-signaling defective mice show similar developmental defects, which may be explained by decreased levels of EGFR ligands and inadequate EGFR activation [25]. The EGFR pathway also plays a crucial role in kidney development through contributing to the induction of metanephric structures. Several EGF family members, such as EGF, TGFα, and HB-EGF, are expressed during metanephric development and may contribute to nephron development through EGFR activation. In vitro studies demonstrated that EGFR ligands EGF and TGFα stimulated embryonic kidney cell growth and proliferation [26]. The deletion of EGFR ligands EREG, AREG and TGFα impair the process of UB morphogenesis in vitro cultures of rat metanephros [27]. In human nephrogenesis, immunoreactivity to HB-EGF can be detected in the UB from the E14.5 day and persisted throughout embryogenesis, whereas immunoreactive EGF and TGFα are detected in all metanephric structures from the 7th week and decrease during the process of nephrons differentiation [28, 29]. Thus, role of ADAM17-mediated EGFR signaling during kidney development deserve further investigation. Interestingly, ADAM17 also participates in generating the active form of Notch, which is important in glomerular development [30]. Conditional knockout mice in the kidney could further elucidate the contribution of ADAM17 during nephrogenesis.
4. Cellular effects of ADAMs in the kidney
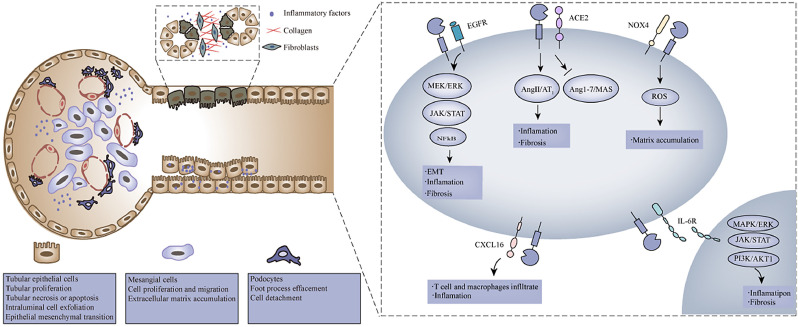
Roles of ADAM17 in the regulation of tubular epithelial cells, mesangial cells and podocytes are summarized in Fig. 3, and we also briefly discuss the cellular effects of ADAM10.
Fig. 3.
Pathways of ADAM17 in the regulation of tubular epithelial cells, mesangial cells and podocytes. In response to kidney injury, ADAM17 activates the epidermal growth factor receptor pathway by inducing the MEK/ERK signaling, the JAK/STAT signaling and the NFκB signaling, which induces proinflammatory factors upregulation, inflammatory cell infiltration, and profibrotic factors release. The increased ADAM17-mediated ACE2 shedding exacerbates the imbalance of RAS, which increases inflammation and fibrosis in a looping feedback manner. And ADAM17 is involved in shedding membrane-bound IL-6 receptor and activating the IL-6 trans-signaling, which mediates renal inflammation and fibrosis by inducing the MAPK/ERK signaling, the JAK/STAT signaling, and the PI3K/AKT1 signaling. Moreover, ADAM17 mediates the release of CXCL16, which induces T cells and macrophages infiltration. In addition, the shedding of NADPH oxidase 4 (Nox4) mediating ROS upregulation and matrix accumulation known to be implicated by ADAM17.
4.1. Tubular epithelial cells
Renal tubules, the major component of the kidney, are vulnerable to several deleterious factors such as hypoxia, toxins, proteinuria, etc. In response to injury, tubular epithelial cells (TECs) start several repair mechanisms. Maladaptive repair mechanisms lead to TECs interacting with various bioactive molecules to drive interstitial inflammation and fibrosis, and the critical transition point from acute kidney injury (AKI) to chronic kidney disease (CKD).
Upon kidney injury, ADAM17 is upregulated and activated in TECs, inducing inflammation and proliferation. ADAM17 releases EGFR ligands TGFα and HB-EGF to activate EFGR signaling, resulting in proinflammatory factors upregulation and inflammatory cells infiltration. A study in cultured TECs showed aldosterone-induced proinflammatory gene upregulation and proinflammatory factors overexpression via ADAM17/EGFR activation. Blockade of the ADAM17/EGFR pathway in these cells had anti-inflammatory effects in response to aldosterone [31]. Ford et al reported that the increased oxidative stress in high glucose-promoted TECs injury was prevented by ADAM17 inhibition, suggesting that ADAM17 is an important mediator of renal tubular inflammation [32]. These findings highlight ADAM17 as an important effector of inflammation upon TECs injury. Moreover, activation of ADAM17 pathway induces TECs proliferation and epithelial-to-mesenchymal transition (EMT), that is characterized by increased expression of fibronectin and cellular collagen [33], [34], [35]. The ADAM17-mediated sustained activation of EGFR signaling interacts with profibrotic factors release and induces interstitial fibrosis. These findings point to a complex regulatory network between ADAM17 and TECs in inflammation and fibrosis.
4.2. Mesangial cells
Renal mesangial cells, the major constituents of the glomerular mesangium, are critically involved in various glomerular injuries. In response to injury, mesangial cells proliferated and were activated to produce mesangial matrix components, such as collagen Ⅳ, fibronectin and laminin, which contribute to excess extracellular matrix (ECM). Moreover, the activation of mesangial cells also induces the secretion of various inflammatory cytokines, chemokines and adhesion molecules, promoting the accumulation of inflammatory cells and the process of renal fibrosis.
ADAM17 is activated in response to injury, leading to mesangial cell proliferation and ECM accumulation. As mentioned above, ADAM17 participates in the shedding of EGFR ligands to activate EGFR and induces downstream ERK phosphorylation. The initiation of downstream signaling drives cell proliferation, migration, and apoptosis [36, 37]. Moreover, by engaging the EGFR pathway, ADAM17 induces glomerular matrix accumulation leading to glomerulosclerosis and interstitial fibrosis. Studies in mesangial cells have shown that in high glucose conditions, ADAM17 is activated and regulates profibrotic TGFα in accumulation of matrix proteins [38, 39]. These effects are abrogated by TAPI2, an ADAM17 inhibitor, via blocking glomerular collagen accumulation [40]. Additionally, ADAM10 and ADAM17 mediate the release of CXCL16 in mesangial cells, promoting cell proliferation and migration. Notably, both ADAM10 and ADAM17 appear to participate in the recruitment of immune cells into the glomerulus to drive the inflammatory process [41]. In addition, ADAM15 participates in the reorganization of the mesangial matrix and the migration of mesangial cells in diseases [42]. These findings indicate that ADAMs are centrally involved in the response of mesangial cells following injury.
4.3. Podocytes
Podocytes play vital roles in maintaining the glomerular filtration barrier and thus are the major target of injury in various glomerular diseases. Under injury, podocytes undergo morphological changes, including foot process effacement, irregular shape, and cytoplasmic droplets, which alter the construction of glomerular basement membrane leading to proteinuria. Furthermore, podocytes generate reactive oxygen species and increase the expression of TGFβ and chemokine receptors to activate inflammation-associated signals, which in turn promotes podocyte injury [43].
The increased expressions of ADAM10 and ADAM17 constitute the main driver of podocyte injury in glomerular diseases. Sustained podocyte injury leads to ADAM17/EGFR signal activation, which decreases podocyte permeability and induces podocyte regeneration. Dey et al showed that ADAM17-dependent EGFR activation changed podocyte permeability via promoting protein zonula occludens-1 rearrangement, whereas these effects were attenuated by EGFR inhibition and ADAM17 down-regulation [44]. In cultured human podocytes, inhibition of ADAM17 reduces constitutive and phorbol ester-induced shedding of the EGFR ligand TGFα as well as cellular proliferation [13]. Moreover, ADAM10 and ADAM17 are involved in the release of CXCL16 from podocytes, which plays an important role in mediating inflammatory factors and podocytes migration. CXCL16 is a scavenger receptor, and its release mediates the uptake of oxLDL to induce reactive oxygen species and fibronectin in podocytes [45]. Another study revealed that ADAM10-mediated CXCL16 release modulated the actin cytoskeleton, as well as promoted podocyte migration [46]. Notably, ADAM10 has been identified as the major regulator for Notch signaling, which takes part in the development of glomerular disease. Several studies have suggested that Notch activation in podocytes contributed to foot process effacement and subsequently albuminuria and glomerulosclerosis [47]. The complex regulatory network between ADAM10 and Notch signaling in podocyte injury still needs to be elucidated.
5. ADAMs in kidney diseases
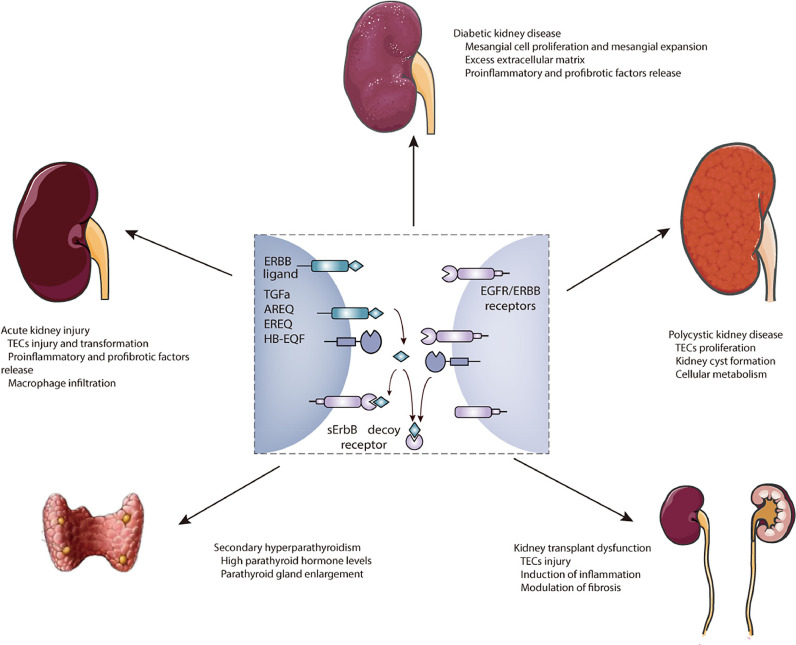
ADAMs signaling is fundamental for modulating cellular processes during kidney development, whereas upregulation and activation of ADAMs are involved in kidney diseases. In this review, we focus on the prominent roles of ADAM10 and ADAM17 with ubiquitous expression in kidney diseases. Notably, ADAM17/EGFR signaling is not only involved in the initiation of AKI and its progression to CKD, but also is of importance in other kidney diseases as shown in Fig. 4.
Fig. 4.
Roles of ADAM17 signaling in kidney diseases. ADAM17 cleaves different membrane-anchored ERBB ligands to release soluble ligands and activate EGFR signaling pathways. Moreover, ADAM17 is involved in the shedding of ERBB receptors to reduce functional ERBB signaling. Damaging effects of ADAM17 have been established in acute kidney injury, diabetic kidney disease, polycystic kidney disease, kidney transplant dysfunction, and secondary hyperparathyroidism.
5.1. Acute kidney injury
AKI is characterized by a rapid increase of serum creatinine, manifests with oliguria or anuria, and presents renal functional and structural changes. Several studies have confirmed that activation of ADAM17 contributes to the development of AKI, suggesting that ADAM17-mediated shedding disrupts cellular homeostasis and promotes tissue injury [33, 48, 49]. In the progression of AKI, ADAM17 overexpression drives tubular epithelial cell injury, mainly by activation of EGFR signaling to induce inflammation and proliferation. The sustained activation of ADAM17/EGFR signaling after AKI upregulates the synthesis and release of pro-inflammatory and pro-fibrotic factors leading to macrophage infiltration as well as renal fibrosis [35, 50]. Moreover, ADAM17 induces IL-6 trans-signaling by shedding of the membrane-bound IL-6 receptor [51], which has been shown to drive crescentic glomerulonephritis [52, 53] and lupus nephritis [54]. On the other hand, ADAM17 induces the shedding of KIM-1. The shedding of constitutive and induced KIM-1 by ADAM17 reduces the apoptotic capacity of TECs and restricts the recovery of the kidney [55]. Additionally, ADAM10 may be important in renal tubule injury. Activated ADAM10 mainly expresses in TECs in experimental models of AKI. ADAM10 is involved in the release and redistribution of Meprin A, which is responsible for inflammation and ECM remodeling in AKI [56]. ADAM10 also takes part in KIM-1 ectodomain release, and ADAM10-induced KIM-1 shedding is essential for the modulation of phagocytic clearance of apoptotic cells after AKI [57]. Collectively, ADAM17 and ADAM10 are overexpressed and activated in renal tubular cells and play a damaging role in AKI.
5.2. Chronic kidney disease
ADAMs not only participate in the initiation of AKI but also are critical for the progression of CKD. Higher ADAM17 expression has been observed in kidneys of CKD patients, co-localizing with TGFα in fibrotic regions, highlighting the importance of the proinflammatory and profibrotic roles of ADAM17 in the progression of CKD [13]. Moreover, circulating ADAM17 activity is markedly increased with CKD progression, and it has been identified as an independent risk factor for renal outcomes [58]. In the unilateral ureteral obstruction (UUO) animal model, the activation of ADAM17 is relevant to the kidney fibrotic response to various tubular injuries [33, 34]. In addition, the upregulation of ADAM10 has been observed in both CKD patients and UUO mice, which aggravates kidney damage resulting in increased fibrotic factors and EMT of tubular epithelia [59]. Moreover, Li et al have shown that ADAM10 expression increased during the progression of IgA nephropathy and that the continuous activation of ADAM10, in turn, promoted kidney interstitial fibrosis and renal dysfunction [60]. Importantly, our recent research found that ADAM19 upregulated in the acute-chronic kidney model, along with increased expression of ADAM19 in the kidney of IgA nephrology patients with severe classes [61]. Upregulation of ADAM19 directly induced fibrotic genes, CCL2, and macrophage infiltration, while depletion of macrophages could ameliorate the fibrotic effects of ADAM19 [61]. Moreover, upregulation of ADAM19 induced accumulation of the Notch1 intracellular domain, whereas the Notch1 pathway antagonist could reduce CCL2 level and macrophage infiltration [61]. These findings provide new insights into the expression of ADAM19 during kidney diseases and offer potential mechanisms of ADAM19 underlying renal fibrosis.
Secondary hyperparathyroidism (SHPT) is a common complication in CKD patients, characterized by mineral and skeletal abnormalities, and predisposition to vascular stiffness and calcification. Parathyroid hyperplasia is in part ascribed to EGFR activation, and inhibition of EGFR activation should effectively attenuate the progression of SHPT. Notably, in patients with SHPT, ADAM17 levels are elevated in the parathyroid tissues, exacerbating EGFR-driven nodular hyperplasia. Arcidiacono et al have found that suppression of ADAM17 expression attenuated parathyroid gland enlargement and decreased parathyroid hormone levels in SHPT rats [62]. Importantly, 1,25-dihydroxy vitamin D inhibition of ADAM17 not only ameliorates the progression of SHPT but also attenuates ADAM17/TGFα-driven systemic inflammation [63].
CKD represents a well-established risk factor for cardiovascular (CV) events. The rate of CV events is high with kidney disease progression, and the broad spectrum of CV events in this population is crucial for CKD prognoses. ADAMs are involved in CV disease development and progression, for example, ADAM17 is associated with an increased risk of CV death in patients with coronary artery diseases [64]. It deserves more attention to the role of ADAMs in the progression of renal function and CV events in CKD patients. Circulating ADAM activity from 2570 CKD patients has been assessed in the NEFRONA study, which indicates that ADAM activity is independently associated with CV events in CKD patients [58]. Moreover, the high level of ADAM17 is correlated with high level of fibroblast growth factor 23, which is an important indicator of oxidative stress and CV risk in CKD patients [65]. Hence, the possible pathogenic link between these conditions is represented by the enhanced production of ADAM17-released TGFβ, which acts as a mediator in the cross-talk kidney and CV disease. Similarities have also been found among the matrix metalloproteinases family as discussed in recent reviews [66, 67]. All of these important pieces of evidence have confirmed that metalloproteinases contribute to reinforcing the risk oriented from CKD to CV diseases.
5.3. Diabetic kidney disease
Diabetic kidney disease (DKD) is characterized by persistent albuminuria and progressive decline in kidney function, accompanied by the accumulation of ECM and fibrosis. Growing evidence has shown that ADAM10, ADAM17 and ADAM19 are induced explicitly in both glomeruli and tubules in DKD patients [68, 69]. Alan et al found that the overexpression of serum ADAM10 was remarkably associated with advanced glycation end products, which has been recognized as important contributors to diabetic complications [70]. Moreover, upregulation of urinary ADAM17 is observed in diabetic patients with albuminuria and is accompanied by elevated urinary angiotensin converting enzyme 2 (ACE2), which may indicate a potential role of ADAM17-mediated ACE2 in DKD pathogenesis [71].
In line with these, hyperglycemia in type 1 diabetic mice upregulates renal ADAM17 expression and induces ADAM17 activation, which enhances oxidative stress and extracellular matrix accumulation. Of note, mice treated with TMI-005, an ADAM17 inhibitor, are protected against renal damage by decreasing type Ⅳ collagen, Nox4, and NADPH oxidase activity [32]. More importantly, ADAM17 knockout mice have been used in protecting renal pro-inflammatory and pro-fibrotic injury caused by type 1 diabetes mellitus, which shows that specific endothelial ADAM17 deletion prevents renal fibrosis and inflammation, and specific proximal tubular ADAM17 deletion protects from pro-fibrotic events, podocyte loss, and attenuates the renal RAS [72]. In db/db mice, ADAM17 is upregulated and co-localized with ACE2 in renal tubules. One study reported that activated ADAM17 impaired kidney function by inducing ACE2 activity, and treatment with rosiglitazone ameliorated the hyperglycemia and restored the ADAM17, thereby attenuating AGE-induced kidney injury in db/db mice [73]. Another study reported that in db/db mice, exercise training alone or combining with metformin protected against albuminuria by preventing ADAM17-mediated renal ACE2 shedding [74]. ADAM17 is significantly increased in kidneys of streptozotocin (STZ)-induced diabetic rats, while ADAM17 inhibition could ameliorate renal inflammation [75]. Furthermore, Src-dependent EGFR transactivation in STZ-induced diabetic nephropathy leads to podocyte depletion and ECM accumulation, whereas ADAM17 inhibition abrogates EGFR phosphorylation, underscoring the interaction between ADAM17 and EGFR signaling in DKD [40]. The deficiency of TIMP3, an endogenous ADAM17 inhibitor, aggravated membrane thickness and mesangial expansion in diabetic mice, contributing to oxidative stress and autophagy through FoxO1/STAT1 interplay [76]. Besides, podocyte deletion of ADAM17 attenuates STZ‐induced ECM accumulation, glomerular damage and urinary albumin, which indicates that targeting ADAM17 signaling may be therapeutic for DKD [77].
5.4. Polycystic kidney disease
Polycystic kidney disease (PKD) is characterized by gradually growing renal cysts, resulting in progressive fibrocystic renal disease and renal dysfunction. As PKD progresses, changes in cell proliferation, apoptosis and adhesion, as well as increased ECM and cellular metabolism play critical roles in the aggravation of kidney injury. ADAM17/EGFR signaling may also be important in the development of PKD. Excessive expression and activation of ADAM17 have been identified in PKD cells, while ADAM17-regulated EFGR shedding induces EGFR/MAPK/ERK pathway activation and probably promotes TECs proliferation. Moreover, changes in cellular metabolism and glucose consumption are accompanied by renal cyst formation and expansion, and ADAM17 inhibition participates in blocking these effects [78]. In addition, PKD1 mutation alters cell polarity and adhesion via intensifying E-cadherin shedding, which is modulated by increased ADAM10. Specific inhibition of ADAM10 serves as a strategy to ameliorate cystogenesis [79]. Presently, some important advances in the treatment of PKD have focused on halting the progression of kidney cysts and attenuating the decline of kidney function, whereas no newer studies regarding podocytes have published. Although, ADAMs represent promising therapeutic targets to attenuate kidney cyst progression, the future management of PKD will probably involve multidrug therapy to target distinct molecular pathways that influence cyst fluid secretion, cell proliferation, inflammation, and fibrosis.
5.5. Kidney transplant dysfunction
Dysregulated ADAMs have also been recognized in allograft nephropathy, which as important contributors to inflammation, immune response, and fibrosis. A study observed that massive ADAM10 expression was together with infiltrating T cells in kidney transplant patients with acute interstitial rejection, suggesting that ADAM10 may regulate immune response and inflammation during acute allograft rejection [12]. ADAM17 signaling involved in the induction of inflammation during allograft nephropathy has also been assessed. ADAM17 is expressed and activated in TECs of transplanted kidneys, and the activation of ADAM17 promotes the shedding of tumor necrosis factor receptors [80]. Other studies further demonstrated that ADAM17 expression was upregulated in chronic renal allograft rejection, which may be responsible for modulating inflammation and fibrosis [14, 81]. In addition, enhanced ADAM19 expression was detected in glomerular sclerotic lesions, renal tubule and inflammatory CD4+ cells in chronic allograft nephropathy, as well as in patients with the acute rejection of kidney allografts [82].
6. Therapeutic outlook
ADAM family is emerging as a novel therapeutic target in patients with kidney diseases and associated complications. Importantly, roles of ADAMs in human kidney disease will be validated after specific therapies targeting its pathways. To date, small molecule inhibitors modulating metalloproteinases focuses mainly on downregulating the proteolytic activity of ADAMs and matrix metalloproteinases. The first generation of small molecule inhibitors targets the zinc ion of metalloproteinases, which shows poor selectivity and several side effects [83]. Thus, specifically targeted small molecules are under development and clinical trials, such as GI254023X with high selectivity for ADAM10 [84]. On the other hand, protein therapeutics, based on antigen-antibody and TIMP-ADAMs interaction, offer superior potential for selectivity and stability. Although TIMPs control the activity of ADAMs in space and time precisely, ADAMs-independent interactions of TIMPs make them controversial protein scaffolds for treating diseases. Therefore, understanding ADAMs-independent function of TIMPs is critical to developing the next generation of TIMP scaffolds as efficient therapeutics. Moreover, natural antibodies, targeting the surface of the catalytic domain, are emerging with low immunogenicity and toxicity. Besides, further understanding of the particular substrates of ADAMs in kidney diseases could aid the development of highly targeted treatments. Since molecular characterization of ADAMs and regulators vary in different tissues, understanding ADAMs homeostasis throughout the body is required to harness its full potential for therapeutic modulation.
7. Outstanding questions
ADAMs are important regulators in kidney embryogenesis and pathological progression. In kidney embryo development, ADAMs participate in cell proliferation, differentiation and migration, mainly via ADAM10-mediated Notch signaling and ADAM17-mediated EGFR signaling. Upon kidney injury, ADAMs are re-expressed and activated in proximal tubules, glomerular mesangium and podocytes, which contributes to inflammation and fibrosis. The field continues to advance, both with the recognition of ADAMs substrate in kidney diseases and a growing understanding of the mechanisms that underpin the associations between the ADAM family and kidney diseases.
Although the damaging effects of ADAMs in kidney injury have been established, the cellular effects of ADAMs are highly diverse in different kidney diseases, and the precise signaling due to ADAMs activation remains largely elusive. Therefore, there are still several important questions needed to be further addressed. On the one hand, the foremost question will be which substrates of ADAMs determine certain kidney pathological process, and whether aspects of kidney injury induced by ADAMs are due to particular effectors remains unknown. On the other hand, understanding how ADAMs activity is regulated in different cells in an environment-dependent manner is crucial for unraveling insights into its roles in kidney pathology. Importantly, clinical translation of experimental data to the human system remains challenging. The targets of ADAMs might regulate cellular processes beyond the kidney and their pharmacological targeting might result in unexpected side effects. Hence, considering the tissue specificity and degree of ADAMs modulation will be essential to maximize its protective effect. Despite the obvious difficulties, based on the ubiquity and importance of ADAMs in physiology and pathology, knowledge gained from the kidney may advance our understanding of potential applications of ADAMs family and provide novel translational insights into other organs.
8. Search strategy and selection criteria
Data for this Review were identified by searches of MEDLINE, Current Contents, PubMed, and references from relevant articles using the search terms “kidney”, “ADAM”, and “nephrogenesis”. Abstracts and reports from meetings were included only when they related directly to previously published work. Only articles published in English between 1980 and 2021 were included.
Contributors
Huanhuan Zhu - Writing - original draft; Junni Wang - Writing - review & editing; Wanyun Nie - Writing - review & editing; Ines Armando - Writing - review & editing; Fei Han - Funding acquisition, Conceptualization, Supervision, Writing - review & editing; All authors read and approved the final version of the manuscript.
Declaration of Competing Interest
The authors have declared that no competing interest exists.
Acknowledgements
This study was supported by the funds from National Natural Science Foundation of China (81770674) and Primary Research and Development Plan of Zhejiang Province (2020C03034) to Fei Han. The funding organization did not have a role in the design, interpretation, or writing of this review article.
References
- 1.Edwards DR, Handsley MM, Pennington CJ. The ADAM metalloproteinases. Mol Aspects Med. 2008;29:258–289. doi: 10.1016/j.mam.2008.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Primakoff P, Myles DG. The ADAM gene family: surface proteins with adhesion and protease activity. Trends Genet. 2000;16:83–87. doi: 10.1016/s0168-9525(99)01926-5. [DOI] [PubMed] [Google Scholar]
- 3.Moss ML, Lambert MH. Shedding of membrane proteins by ADAM family proteases. Essays Biochem. 2002;38:141–153. doi: 10.1042/bse0380141. [DOI] [PubMed] [Google Scholar]
- 4.Endres K, Anders A, Kojro E, Gilbert S, Fahrenholz F, Postina R. Tumor necrosis factor-alpha converting enzyme is processed by proprotein-convertases to its mature form which is degraded upon phorbol ester stimulation. Eur J Biochem. 2003;270:2386–2393. doi: 10.1046/j.1432-1033.2003.03606.x. [DOI] [PubMed] [Google Scholar]
- 5.Takeda S, Igarashi T, Mori H, Araki S. Crystal structures of VAP1 reveal ADAMs’ MDC domain architecture and its unique C-shaped scaffold. EMBO J. 2006;25:2388–2396. doi: 10.1038/sj.emboj.7601131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Prox J, Willenbrock M, Weber S, Lehmann T, Schmidt-Arras D, Schwanbeck R. Tetraspanin15 regulates cellular trafficking and activity of the ectodomain sheddase ADAM10. Cell Mol Life Sci. 2012;69:2919–2932. doi: 10.1007/s00018-012-0960-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Saraceno C, Marcello E, Di Marino D, Borroni B, Claeysen S, Perroy J. SAP97-mediated ADAM10 trafficking from Golgi outposts depends on PKC phosphorylation. Cell Death Dis. 2014;5:e1547. doi: 10.1038/cddis.2014.492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Seals DF, Courtneidge SA. The ADAMs family of metalloproteases: multidomain proteins with multiple functions. Genes Dev. 2003;17 doi: 10.1101/gad.1039703. [DOI] [PubMed] [Google Scholar]
- 9.Jones JC, Rustagi S, Dempsey PJ. ADAM proteases and gastrointestinal function. Annu Rev Physiol. 2016;78:243–276. doi: 10.1146/annurev-physiol-021014-071720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shimoda M, Principe S, Jackson HW, Luga V, Fang H, Molyneux SD. Loss of the Timp gene family is sufficient for the acquisition of the CAF-like cell state. Nat Cell Biol. 2014;16:889–901. doi: 10.1038/ncb3021. [DOI] [PubMed] [Google Scholar]
- 11.Stoeck A, Keller S, Riedle S, Sanderson MP, Runz S, Le Naour F. A role for exosomes in the constitutive and stimulus-induced ectodomain cleavage of L1 and CD44. Biochem J. 2006;393:609–618. doi: 10.1042/BJ20051013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Schramme A, Abdel-Bakky MS, Gutwein P, Obermüller N, Baer PC, Hauser IA. Characterization of CXCL16 and ADAM10 in the normal and transplanted kidney. Kidney Int. 2008;74:328–338. doi: 10.1038/ki.2008.181. [DOI] [PubMed] [Google Scholar]
- 13.Melenhorst WB, Visser L, Timmer A, van den Heuvel MC, Stegeman CA, van Goor H. ADAM17 upregulation in human renal disease: a role in modulating TGF-alpha availability? Am J Physiol Renal Physiol. 2009;297 doi: 10.1152/ajprenal.90610.2008. F781-F90. [DOI] [PubMed] [Google Scholar]
- 14.Mulder GM, Melenhorst WBWH, Celie JWAM, Kloosterhuis NJ, Hillebrands J-L, Ploeg RJ. ADAM17 up-regulation in renal transplant dysfunction and non-transplant-related renal fibrosis. Nephrol Dial Transplant. 2012;27:2114–2122. doi: 10.1093/ndt/gfr583. [DOI] [PubMed] [Google Scholar]
- 15.Takeda S. ADAM and ADAMTS family proteins and snake venom metalloproteinases: a structural overview. Toxins (Basel) 2016;8 doi: 10.3390/toxins8050155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Stuart RO, Bush KT, Nigam SK. Changes in gene expression patterns in the ureteric bud and metanephric mesenchyme in models of kidney development. Kidney Int. 2003;64:1997–2008. doi: 10.1046/j.1523-1755.2003.00383.x. [DOI] [PubMed] [Google Scholar]
- 17.Brose K, Bland KS, Wang KH, Arnott D, Henzel W, Goodman CS. Slit proteins bind Robo receptors and have an evolutionarily conserved role in repulsive axon guidance. Cell. 1999;96:795–806. doi: 10.1016/s0092-8674(00)80590-5. [DOI] [PubMed] [Google Scholar]
- 18.Piper M, Georgas K, Yamada T, Little M. Expression of the vertebrate Slit gene family and their putative receptors, the Robo genes, in the developing murine kidney. Mech Dev. 2000;94:213–217. doi: 10.1016/s0925-4773(00)00313-0. [DOI] [PubMed] [Google Scholar]
- 19.Guo Q, Wang Y, Tripathi P, Manda KR, Mukherjee M, Chaklader M. Adam10 mediates the choice between principal cells and intercalated cells in the kidney. J Am Soc Nephrol. 2015;26:149–159. doi: 10.1681/ASN.2013070764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Boyle SC, Liu Z, Kopan R. Notch signaling is required for the formation of mesangial cells from a stromal mesenchyme precursor during kidney development. Development. 2014;141:346–354. doi: 10.1242/dev.100271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cheng H-T, Kopan R. The role of Notch signaling in specification of podocyte and proximal tubules within the developing mouse kidney. Kidney Int. 2005;68:1951–1952. doi: 10.1111/j.1523-1755.2005.00627.x. [DOI] [PubMed] [Google Scholar]
- 22.Alabi RO, Glomski K, Haxaire C, Weskamp G, Monette S, Blobel CP. ADAM10-dependent signaling through Notch1 and Notch4 controls development of organ-specific vascular beds. Circ Res. 2016;119:519–531. doi: 10.1161/CIRCRESAHA.115.307738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Farber G, Hurtado R, Loh S, Monette S, Mtui J, Kopan R. Glomerular endothelial cell maturation depends on ADAM10, a key regulator of Notch signaling. Angiogenesis. 2018;21:335–347. doi: 10.1007/s10456-018-9599-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gutwein P, Schramme A, Abdel-Bakky MS, Doberstein K, Hauser IA, Ludwig A. ADAM10 is expressed in human podocytes and found in urinary vesicles of patients with glomerular kidney diseases. J Biomed Sci. 2010;17:3. doi: 10.1186/1423-0127-17-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Capone C, Dabertrand F, Baron-Menguy C, Chalaris A, Ghezali L, Domenga-Denier V. Mechanistic insights into a TIMP3-sensitive pathway constitutively engaged in the regulation of cerebral hemodynamics. Elife. 2016;5 doi: 10.7554/eLife.17536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sakurai H, Tsukamoto T, Kjelsberg CA, Cantley LG, Nigam SK. EGF receptor ligands are a large fraction of in vitro branching morphogens secreted by embryonic kidney. Am J Physiol. 1997;273 doi: 10.1152/ajprenal.1997.273.3.F463. F463-F72. [DOI] [PubMed] [Google Scholar]
- 27.Rogers SA, Ryan G, Hammerman MR. Metanephric transforming growth factor-alpha is required for renal organogenesis in vitro. Am J Physiol. 1992;262 doi: 10.1152/ajprenal.1992.262.4.F533. F533-F9. [DOI] [PubMed] [Google Scholar]
- 28.Carev D, Saraga M, Saraga-Babic M. Expression of intermediate filaments, EGF and TGF-alpha in early human kidney development. J Mol Histol. 2008;39:227–235. doi: 10.1007/s10735-007-9157-7. [DOI] [PubMed] [Google Scholar]
- 29.Takemura T, Hino S, Kuwajima H, Yanagida H, Okada M, Nagata M. Induction of collecting duct morphogenesis in vitro by heparin-binding epidermal growth factor-like growth factor. Am Soc Nephrol. 2001;12:964–972. doi: 10.1681/ASN.V125964. [DOI] [PubMed] [Google Scholar]
- 30.Niranjan T, Bielesz B, Gruenwald A, Ponda MP, Kopp JB, Thomas DB. The Notch pathway in podocytes plays a role in the development of glomerular disease. Nat Med. 2008;14:290–298. doi: 10.1038/nm1731. [DOI] [PubMed] [Google Scholar]
- 31.Morgado-Pascual JL, Rayego-Mateos S, Valdivielso JM, Ortiz A, Egido J, Ruiz-Ortega M. Paricalcitol inhibits aldosterone-induced proinflammatory factors by modulating epidermal growth factor receptor pathway in cultured tubular epithelial cells. Biomed Res Int. 2015;2015 doi: 10.1155/2015/783538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ford BM, Eid AA, Göőz M, Barnes JL, Gorin YC, Abboud HE. ADAM17 mediates Nox4 expression and NADPH oxidase activity in the kidney cortex of OVE26 mice. Am J Physiol Renal Physiol. 2013;305 doi: 10.1152/ajprenal.00522.2012. F323-F32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kefaloyianni E, Muthu ML, Kaeppler J, Sun X, Sabbisetti V, Chalaris A. ADAM17 substrate release in proximal tubule drives kidney fibrosis. JCI Insight. 2016;1 doi: 10.1172/jci.insight.87023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kefaloyianni E, Keerthi Raja MR, Schumacher J, Muthu ML, Krishnadoss V, Waikar SS. Proximal tubule-derived amphiregulin amplifies and integrates profibrotic EGF receptor signals in kidney fibrosis. J Am Soc Nephrol. 2019;30:2370–2383. doi: 10.1681/ASN.2019030321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhuang S, Kinsey GR, Rasbach K, Schnellmann RG. Heparin-binding epidermal growth factor and Src family kinases in proliferation of renal epithelial cells. Am J Physiol Renal Physiol. 2008;294 doi: 10.1152/ajprenal.00473.2007. F459-F68. [DOI] [PubMed] [Google Scholar]
- 36.Göőz M, Göőz P, Luttrell LM, Raymond JR. 5-HT2A receptor induces ERK phosphorylation and proliferation through ADAM-17 tumor necrosis factor-alpha-converting enzyme (TACE) activation and heparin-bound epidermal growth factor-like growth factor (HB-EGF) shedding in mesangial cells. J Biol Chem. 2006;281:21004–21012. doi: 10.1074/jbc.M512096200. [DOI] [PubMed] [Google Scholar]
- 37.Akool E-S, Gauer S, Osman B, Doller A, Schulz S, Geiger H. Cyclosporin A and tacrolimus induce renal Erk1/2 pathway via ROS-induced and metalloproteinase-dependent EGF-receptor signaling. Biochem Pharmacol. 2012;83:286–295. doi: 10.1016/j.bcp.2011.11.001. [DOI] [PubMed] [Google Scholar]
- 38.Li R, Wang T, Walia K, Gao B, Krepinsky JC. Regulation of profibrotic responses by ADAM17 activation in high glucose requires its C-terminus and FAK. J Cell Sci. 2018:131. doi: 10.1242/jcs.208629. [DOI] [PubMed] [Google Scholar]
- 39.Uttarwar L, Peng F, Wu D, Kumar S, Gao B, Ingram AJ. HB-EGF release mediates glucose-induced activation of the epidermal growth factor receptor in mesangial cells. Am J Physiol Renal Physiol. 2011;300 doi: 10.1152/ajprenal.00436.2010. F921-F31. [DOI] [PubMed] [Google Scholar]
- 40.Taniguchi K, Xia L, Goldberg HJ, Lee KWK, Shah A, Stavar L. Inhibition of Src kinase blocks high glucose-induced EGFR transactivation and collagen synthesis in mesangial cells and prevents diabetic nephropathy in mice. Diabetes. 2013;62:3874–3886. doi: 10.2337/db12-1010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Schramme A, Abdel-Bakky MS, Kämpfer-Kolb N, Pfeilschifter J, Gutwein P. The role of CXCL16 and its processing metalloproteinases ADAM10 and ADAM17 in the proliferation and migration of human mesangial cells. Biochem Biophys Res Commun. 2008;370:311–316. doi: 10.1016/j.bbrc.2008.03.088. [DOI] [PubMed] [Google Scholar]
- 42.Martin J, Eynstone LV, Davies M, Williams JD, Steadman R. The role of ADAM 15 in glomerular mesangial cell migration. J Biol Chem. 2002;277:33683–33689. doi: 10.1074/jbc.M200988200. [DOI] [PubMed] [Google Scholar]
- 43.Nagata M. Podocyte injury and its consequences. Kidney Int. 2016;89:1221–1230. doi: 10.1016/j.kint.2016.01.012. [DOI] [PubMed] [Google Scholar]
- 44.Dey M, Baldys A, Sumter DB, Göoz P, Luttrell LM, Raymond JR. Bradykinin decreases podocyte permeability through ADAM17-dependent epidermal growth factor receptor activation and zonula occludens-1 rearrangement. J Pharmacol Exp Ther. 2010;334:775–783. doi: 10.1124/jpet.110.168054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gutwein P, Abdel-Bakky MS, Schramme A, Doberstein K, Kämpfer-Kolb N, Amann K. CXCL16 is expressed in podocytes and acts as a scavenger receptor for oxidized low-density lipoprotein. Am J Pathol. 2009;174:2061–2072. doi: 10.2353/ajpath.2009.080960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chen Y, Wang Z, Li Q, Yu L, Zhu Y, Wang J. oxLDL promotes podocyte migration by regulating CXCL16, ADAM10 and ACTN4. Mol Med Rep. 2020;22:1976–1984. doi: 10.3892/mmr.2020.11292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Niranjan T, Murea M, Susztak K. The pathogenic role of Notch activation in podocytes. Nephron Exp Nephrol. 2009;111 doi: 10.1159/000209207. e73-e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chueh T-I, Zheng C-M, Hou Y-C, Lu K-C. Novel evidence of acute kidney injury in COVID-19. J Clin Med. 2020;9 doi: 10.3390/jcm9113547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kato T, Hagiyama M, Ito A. Renal ADAM10 and 17: Their physiological and medical meanings. Front Cell Dev Biol. 2018;6:153. doi: 10.3389/fcell.2018.00153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Waheed F, Dan Q, Amoozadeh Y, Zhang Y, Tanimura S, Speight P. Central role of the exchange factor GEF-H1 in TNF-α-induced sequential activation of Rac, ADAM17/TACE, and RhoA in tubular epithelial cells. Mol Biol Cell. 2013;24:1068–1082. doi: 10.1091/mbc.E12-09-0661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Schumacher N, Rose-John S. ADAM17 activity and IL-6 trans-signaling in inflammation and cancer. Cancers (Basel) 2019:11. doi: 10.3390/cancers11111736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Braun GS, Nagayama Y, Maruta Y, Heymann F, van Roeyen CR, Klinkhammer BM. IL-6 trans-signaling drives murine crescentic GN. J Am Soc Nephrol. 2016;27:132–142. doi: 10.1681/ASN.2014111147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Luig M, Kluger MA, Goerke B, Meyer M, Nosko A, Yan I. Inflammation-induced IL-6 functions as a natural brake on macrophages and limits GN. J Am Soc Nephrol. 2015;26:1597–1607. doi: 10.1681/ASN.2014060620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Tsantikos E, Maxwell MJ, Putoczki T, Ernst M, Rose-John S, Tarlinton DM. Interleukin-6 trans-signaling exacerbates inflammation and renal pathology in lupus-prone mice. Arthritis Rheum. 2013;65:2691–2702. doi: 10.1002/art.38061. [DOI] [PubMed] [Google Scholar]
- 55.Gandhi R, Yi J, Ha J, Shi H, Ismail O, Nathoo S. Accelerated receptor shedding inhibits kidney injury molecule-1 (KIM-1)-mediated efferocytosis. Am J Physiol Renal Physiol. 2014;307 doi: 10.1152/ajprenal.00638.2013. F205-F21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Herzog C, Haun RS, Ludwig A, Shah SV, Kaushal GP. ADAM10 is the major sheddase responsible for the release of membrane-associated meprin A. J Biol Chem. 2014;289:13308–13322. doi: 10.1074/jbc.M114.559088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sriranganathan S, Tutunea-Fatan E, Abbasi A, Gunaratnam L. Mapping and functional characterization of murine kidney injury molecule-1 proteolytic cleavage site. Mol Cell Biochem. 2021;476:1093–1108. doi: 10.1007/s11010-020-03975-5. [DOI] [PubMed] [Google Scholar]
- 58.Palau V, Riera M, Duran X, Valdivielso JM, Betriu A, Fernández E. Circulating ADAMs are associated with renal and cardiovascular outcomes in chronic kidney disease patients. Nephrol Dial Transpl. 2020;35:130–138. doi: 10.1093/ndt/gfy240. [DOI] [PubMed] [Google Scholar]
- 59.Hou L, Du Y, Zhao C, Wu Y. PAX2 may induce ADAM10 expression in renal tubular epithelial cells and contribute to epithelial-to-mesenchymal transition. Int Urol Nephrol. 2018;50:1729–1741. doi: 10.1007/s11255-018-1956-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Li B, Zhu C, Dong L, Qin J, Xiang W, Davidson AJ. ADAM10 mediates ectopic proximal tubule development and renal fibrosis through Notch signalling. J Pathol. 2020;252:274–289. doi: 10.1002/path.5517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wang J, Nie W, Xie X, Bai M, Ma Y, Jin L. MicroRNA-874-3p/ADAM (A Disintegrin and Metalloprotease) 19 mediates macrophage activation and renal fibrosis after acute kidney injury. Hypertension. 2021;77:1613–1626. doi: 10.1161/HYPERTENSIONAHA.120.16900. [DOI] [PubMed] [Google Scholar]
- 62.Arcidiacono MV, Yang J, Fernandez E, Dusso A. The induction of C/EBPβ contributes to vitamin D inhibition of ADAM17 expression and parathyroid hyperplasia in kidney disease. Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association - European Renal Association. 2015; 30: 423-33. [DOI] [PMC free article] [PubMed]
- 63.Dusso A, Arcidiacono MV, Yang J, Tokumoto M. Vitamin D inhibition of TACE and prevention of renal osteodystrophy and cardiovascular mortality. J Steroid Biochem Mol Biol. 2010;121:193–198. doi: 10.1016/j.jsbmb.2010.03.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Morange PE, Tregouet DA, Godefroy T, Saut N, Bickel C, Rupprecht HJ. Polymorphisms of the tumor necrosis factor-alpha (TNF) and the TNF-alpha converting enzyme (TACE/ADAM17) genes in relation to cardiovascular mortality: the AtheroGene study. J Mol Med. 2008;86:1153–1161. doi: 10.1007/s00109-008-0375-6. [DOI] [PubMed] [Google Scholar]
- 65.Perna AF, Pizza A, Di Nunzio A, Bellantone R, Raffaelli M, Cicchella T. ADAM17, a New Player in the Pathogenesis of Chronic Kidney Disease-Mineral and Bone Disorder. J Ren Nutr. 2017;27:453–457. doi: 10.1053/j.jrn.2017.05.007. [DOI] [PubMed] [Google Scholar]
- 66.Provenzano M, Andreucci M, Garofalo C, Faga T, Michael A, Ielapi N. The association of matrix metalloproteinases with chronic kidney disease and peripheral vascular disease: a light at the end of the tunnel? Biomolecules. 2020:10. doi: 10.3390/biom10010154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Andreucci M, Provenzano M, Faga T, Michael A, Patella G, Mastroroberto P. Aortic aneurysms, chronic kidney disease and metalloproteinases. Biomolecules. 2021:11. doi: 10.3390/biom11020194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lattenist L, Ochodnický P, Ahdi M, Claessen N, Leemans JC, Satchell SC. Renal endothelial protein C receptor expression and shedding during diabetic nephropathy. J Thromb Haemost. 2016;14:1171–1182. doi: 10.1111/jth.13315. [DOI] [PubMed] [Google Scholar]
- 69.Melenhorst WBWH, van den Heuvel MC, Timmer A, Huitema S, Bulthuis M, Timens W. ADAM19 expression in human nephrogenesis and renal disease: associations with clinical and structural deterioration. Kidney Int. 2006;70:1269–1278. doi: 10.1038/sj.ki.5001753. [DOI] [PubMed] [Google Scholar]
- 70.Lee ACH, Lam JKY, Shiu SWM, Wong Y, Betteridge DJ, Tan KCB. Serum level of soluble receptor for advanced glycation end products is associated with a disintegrin and metalloproteinase 10 in Type 1 diabetes. PLoS One. 2015;10 doi: 10.1371/journal.pone.0137330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gutta S, Grobe N, Kumbaji M, Osman H, Saklayen M, Li G. Increased urinary angiotensin converting enzyme 2 and neprilysin in patients with type 2 diabetes. Am J Physiol Renal Physiol. 2018;315 doi: 10.1152/ajprenal.00565.2017. F263-F74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Palau V, Nugraha B, Benito D, Pascual J, Emmert MY, Hoerstrup SP. Both specific endothelial and proximal tubular adam17 deletion protect against diabetic nephropathy. Int J Mol Sci. 2021:22. doi: 10.3390/ijms22115520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Chodavarapu H, Grobe N, Somineni HK, Salem ESB, Madhu M, Elased KM. Rosiglitazone treatment of type 2 diabetic db/db mice attenuates urinary albumin and angiotensin converting enzyme 2 excretion. PLoS One. 2013;8:e62833. doi: 10.1371/journal.pone.0062833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Somineni HK, Boivin GP, Elased KM. Daily exercise training protects against albuminuria and angiotensin converting enzyme 2 shedding in db/db diabetic mice. J Endocrinol. 2014;221:235–251. doi: 10.1530/JOE-13-0532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Liu S, Ye L, Tao J, Ge C, Huang L, Yu J. Total flavones of Abelmoschus manihot improve diabetic nephropathy by inhibiting the iRhom2/TACE signalling pathway activity in rats. Pharm Biol. 2017;56 doi: 10.1080/13880209.2017.1412467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Fiorentino L, Cavalera M, Menini S, Marchetti V, Mavilio M, Fabrizi M. Loss of TIMP3 underlies diabetic nephropathy via FoxO1/STAT1 interplay. EMBO Mol Med. 2013;5:441–455. doi: 10.1002/emmm.201201475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Casagrande V, Iuliani G, Menini S, Pugliese G, Federici M, Menghini R. Restoration of renal TIMP3 levels via genetics and pharmacological approach prevents experimental diabetic nephropathy. Clin Transl Med. 2021;11:e305. doi: 10.1002/ctm2.305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Beck Gooz M, Maldonado EN, Dang Y, Amria MY, Higashiyama S, Abboud HE. ADAM17 promotes proliferation of collecting duct kidney epithelial cells through ERK activation and increased glycolysis in polycystic kidney disease. Am J Physiol Renal Physiol. 2014;307 doi: 10.1152/ajprenal.00218.2014. F551-F9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Xu JX, Lu T-S, Li S, Wu Y, Ding L, Denker BM. Polycystin-1 and Gα12 regulate the cleavage of E-cadherin in kidney epithelial cells. Physiol Genomics. 2015;47:24–32. doi: 10.1152/physiolgenomics.00090.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wang J, Li Z, Al-Lamki R, Wang J, Zuo N, Bradley JR. The role of tumor necrosis factor-α converting enzyme in renal transplant rejection. Am J Nephrol. 2010;32:362–368. doi: 10.1159/000320467. [DOI] [PubMed] [Google Scholar]
- 81.Berthier CC, Lods N, Joosten SA, van Kooten C, Leppert D, Lindberg RLP. Differential regulation of metzincins in experimental chronic renal allograft rejection: potential markers and novel therapeutic targets. Kidney Int. 2006;69:358–368. doi: 10.1038/sj.ki.5000049. [DOI] [PubMed] [Google Scholar]
- 82.Melenhorst WBWH, van den Heuvel MC, Stegeman CA, van der Leij J, Huitema S, van den Berg A. Upregulation of ADAM19 in chronic allograft nephropathy. Am J Transpl. 2006;6:1673–1681. doi: 10.1111/j.1600-6143.2006.01384.x. [DOI] [PubMed] [Google Scholar]
- 83.Raeeszadeh-Sarmazdeh M, Do LD, Hritz BG. Metalloproteinases and their inhibitors: potential for the development of new therapeutics. Cells. 2020;9 doi: 10.3390/cells9051313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hoettecke N, Ludwig A, Foro S, Schmidt B. Improved synthesis of ADAM10 inhibitor GI254023X. Neurodegener Dis. 2010;7:232–238. doi: 10.1159/000267865. [DOI] [PubMed] [Google Scholar]