Abstract
Objective
Severe acute respiratory syndrome coronavirus 2 (SARS-COV2) was first detected in Wuhan, China in December, 2019. The emerging virus causes a respiratory illness, that can trigger a cytokine storm in the body.
Method
Cytokine storm in patient’s body is associated with severe forms of disease. It is one of the main complications of coronavirus disease-2019 (COVID-19), in which immune cells play a major role. Studies have shown immune cells in the tumor environment can be effective to induce resistance to chemotherapy in cancer patients.
Result
Therefore, considering the role of immune cells to induce cytokine storm in COVID-19 patients, and their role to cause resistance to chemotherapy, they are effective on disease progression and creation of severe form of disease.
Conclusion
By examining the signaling pathways and inducing resistance to chemotherapy in tumor cells and the cells affect them, it is possible to prevent the occurrence of severe forms of the disease in cancer patients with COVID-19; it is applicable using target therapy and other subsequent treatment strategies.
Keywords: Cytokine storm, Chemotherapy resistance, Pathogenesis, COVID-19
Introduction
In late December 2019, an outbreak of pneumonia associated with a novel coronavirus, coronavirus disease-2019 (COVID-19), was first reported in Wuhan, Hubei Province, China; it quickly spread in all the countries around the world. The virus targets human respiratory system and has a relatively high mortality rate. Fever, cough, musculoskeletal and gastrointestinal symptoms and fatigue are the common symptoms of virus infection [1, 2].
Accumulating evidence indicates cytokine storm syndrome may be present in the severely ill COVID-19 patients; it is a sign of immune dysregulation [3]. It has been shown to be associated with poor outcomes in COVID-19 patients. Cytokine storm is characterized by secretion of interleukins, interferons, chemokines, tumor-necrosis factors (TNF) and several other inflammatory mediators [4]. Plasma concentration of multiple inflammatory cytokines, such as granulocyte–macrophage colony-stimulating factor (GM-CSF), interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), IL-2, 7, 10, and granulocyte colony-stimulating factor (G-CSF) increases following Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection.
In addition, it is demonstrated that peripheral inflammatory monocytes and pathogenic T cells might induce cytokine storms in severe COVID-19 patients [5, 6]. Various conditions including malignancies, sepsis, macrophage activation syndrome (MAS), systemic juvenile idiopathic arthritis (SJIA), cytokine release syndrome (CRS) and acute respiratory distress syndrome (ARDS) are generated by cytokine storm [7].
A higher risk of severe stages of COVID-19 infection is reported in actively treated cancer patients. It is also shown, that unlike the events in severely ill COVID-19 patients, cancer development is usually associated with immune responses dampen; it is portrayed via the increased secretion of anti-inflammatory cytokines, repressed induction of pro-inflammatory mediators and enhanced population of immunosuppressive leukocytes [8]. According to these reports, the cytokine storm threatens life in COVID-19 patients, but it can be a therapeutic option in cancer patients. In this study, the pathophysiology of cytokine storm has been studied in response to COVID-19 infection in malignancies.
Cytokine release: protective versus pathogenesis
Cytokine storm occurs with a sudden increase in the level of pro-inflammatory cytokines such as IL-1, IL-6, and TNF-α. These cytokines are among the most important pro-inflammatory ones; they considered as the innate immunity, and are produced by cells such as macrophages and inflamed endothelial cells. Increased level of these cytokines in blood leads to the summoning of immune cells to the inflamed and infected sites, which in turn leads to tissue damage, organ failure and eventually death [9, 10].
Some cytokines have the protective role, while others have the pathological role. Cytokine storm has also been reported in COVID-19 patients; serological assessments of plasma indicates increased levels of IL-1b, IL-6, IL-10 and TNF-α in the cited patients. Studies have also shown, that increase in these cytokines leads to disease progression and worsening patients' clinical conditions, which ultimately leads to survival reduction [11, 12]. Despite various studies, the molecular mechanisms of these cytokines (IL-1, IL-6 and TNF-α) in relation to the pathogenesis of malignancy in COVID-19 patients have not been fully identified yet.
IL-6
IL-6 is a pleiotropic cytokine, that has a wide range of biological activities, including: immune regulation, hematopoiesis, inflammation, effects on metabolic, proliferative and regenerative processes. It is produced by various cells in the tumor microenvironment (TME), and plays an important role in the expansion and differentiation of tumor cells [13].
IL-6 binds to the IL-6R (IL-6 Receptor) on the surface of target cell to form a heterohexameric complex, consisting of two molecules IL-6 and IL-6R, together with the IL-6 receptor subunit (gp130). In the downstream, the signaling pathway of this cytokine is activated by the JAK/STAT pathway. This pathway is also involved in a process called Epithelial Mesenchymal Transition (EMT), by activating a series of transcription factors.
Snail, Zeb1, JUNB and Twist1 are among the most important copying factors in the EMT induction pathway (EMT-TF) [14]. Snail transcription factor via Smad1/AKT/GSK3β signaling pathway and increased expression of Nanog molecule can induce EMT in cell. Regarding other effective factors in EMT, the Zeb1 factor also applies a positive feedback to HAS2 molecule; also, JUNB factor can lead to EMT process by activating the TGF-β molecule, and using its related signaling pathway [15–17].
Finally, Twist1 factor can induce EMT process in cells by reducing the H2AX expression, and subsequently by increasing the expression of VCAN, THBS1, TGFB2, ITGB4, SERPINE genes, and reducing the expression of CDH1, CTNNB1, OCLN genes [18]. EMT status plays an important role in resistance to chemotherapy, so the JAK/STAT signaling pathway through this condition can lead to resistance to chemotherapy in patients with malignancy.
On the other hand, the heterohexameric complex IL-6/IL-6R/gp130 can induce PI3K/AKT and MAPK/ERK signaling pathways in its downstream. These pathways also lead to the EMT status by applying Snail and Slug transcription factors, and increasing the expression of E-Cadherin and N-cadherin molecules; by inducing this process in cells, they lead to resistance to chemotherapy in people with malignancy [19]. Studies show an increase in IL-6 levels in cancer patients with COVID-19, indicating a cytokine storm [20, 21]. It is hypothesized that, due to cytokine storms, elevated IL-6 levels in COVID-19 cancer patients and using the mentioned pathways in them, the phenomenon of resistance to chemotherapy drug occurs; it is accompanied by therapeutic effect decrement, increased mortality and survival reduction in patients.
IL-1
The IL-1 family includes IL-1α, IL-1β, and IL-1 Receptor Antagonist (IL1-Ra). IL-1α/β binds to the IL-1 type one receptor at the surface of the target cell. Recent studies have shown, that IL-1 and its signaling pathways play an important role in tumorigenesis and cancer progression [22]. IL-1β can induce EMT process in cells by two ways. In one of these pathways, IL-1β combines with IL-1R1 and β-Catenin to form the IL-1β/IL-1R1/Beta-Catenin; on the other hand, IL-1β increases the expression of Twist1 transcription factor by activating the PI3K/AKT signaling pathway; which plays an important role in the EMT process. It can subsequently induce the phenomenon of chemo resistance.
Also, Twist1 transcription factor produced in the downstream of the above pathway can lead to estrogen receptor methylation (ER1). By causing this epigenetic change, it reduces ERα receptor expression and causes resistance to Tamoxifam. The ER1 is the target of Tamoxifam drug [23].
On the other hand, IL-1 activates the NF-Kβ transcription factor in its downstream signaling. Studies have shown, that NF-Kβ levels are inversely related to the PTEN molecule level in a person with malignancy and chemo resistance; it means, that NF-Kβ is high and PTEN is low in these people. IL-1 increases miR-21 transcription by activating the NF-Kβ transcription factor in people with malignancy. This microRNA inhibits cellular apoptosis by suppressing the expression of PTEN molecule.
Subsequently, Caspase 3 can lead to chemo resistance induction in cited individuals [24]. In COVID-19 patients, IL-1 increases following cytokine storm. Considering the role of IL-1 to induce chemotherapy resistance in cancer patients with COVID-19, it is hypothesized the survival rate reduces and the treatment process is challenged.
Tumor necrosis factor-alpha (TNF-α)
TNF can be involved in activating, inducing function and differentiating the immune regulatory cells, such as Myeloid-derived Suppressor cell and the Regulatory T cell through the use of TNF-R2. It also plays an impressive role in cancer cells differentiation and tumorigenesis induction.
It also activates the Snail transcription factor through the AKT/GSK3β/Snail signaling pathway, which is involved in EMT process; subsequently, the chemo resistance phenomenon is induced in tumor cells [25].TNF can also induce immune check point molecules expression, such as PDL1 and CD73 in cancer cells. The expression of these molecules is also effective to induce chemo resistance [26]. Thus, NF-Kβ transcription factor increases PDL1 expression, and consequently increases PDL1 expression on the surface of tumor cells by binding to its receptor; PD1 on the surface of Cytotoxic T lymphocyte (CTL) reduces the inhibitory function of CTL on tumor cells.
Through the ERK and mTOR signaling pathway, it can induce proliferation, tumor cells survival increment, and resistance to chemotherapy drugs. So, blocking the PDL1 signaling pathway using monoclonal antibodies is effective to improve treatment process with chemotherapy drugs in people with malignancy [27].
Downstream of the MAPK signaling pathway, the C-jun/AP-1 transcription factor also increases the CD73 molecule expression. CD73 can also increase the number of tumor cells through HGF/Met/ERK1 pathway. Subsequently, increased expression of CD73 through A2B Adenosine Receptor, and increased expression of Multi Drug Resistance Associated Protein 1 (MDR P1) molecule can lead to chemo resistance [28].
Some studies have shown, that despite the tumorigenesis role of TNF molecule, it can also induce apoptosis by binding to TNFR1 on the surface of tumor endothelial cells; it also plays an important role in the process of chemo sensitivity. By increasing the expression of TNF-related apoptosis inducing ligand (TRAIL) and TRAIL receptors, such as DR4 and DR5 on the surface of cancer cells, chemotherapy drugs increase Caspase8; subsequently, it induces apoptosis. Therefore, it can play a positive role in the process of treatment with chemotherapy drugs [29–31].
TNF can also act as an antitumor molecule, by activating the CTLs and tumor-infiltrating macrophages and dendritic cells (DCs), and thereby lead to chemo sensitivity [32]. It is elevated in COVID-19 infected individuals following a cytokine storm. However, due to its dual role in inducing chemotherapy resistance and creating a positive effect on chemotherapy, more studies are needed to decide about its effect on the process of chemotherapy in cited patients.
Aberrant function of innate immune cells
Monocyte and macrophage
Monocytes are components of mononuclear phagocytic system, that play an important role in regulating immune responses. These cells can also be effective to induce cytokine storm by releasing pro-inflammatory cytokines; they can induce cytokine storm in COVID-19 patients, too [5]. Monocytes are also impressive in generating chemo resistance in cancer patients.
Surrounding tumor cells are a number of cells, including Tumor Associated Macrophages (TAMs) and Bone-Marrow derived macrophages (BM-DMs). TAMs play an important role in tumor cells growth and inducing resistance to chemotherapy drugs. BM-DMs infiltrate tumor cells through DAMP, CCL2, and CSF-1 molecules; they are also called M1-like macrophages. Tumor cells secrete cytokines, such as IL4, IL10, IL13, and lactic acid in association with CD4 + (Th2) T cells, leading to M1 to M2 macrophage polarization [33, 34].
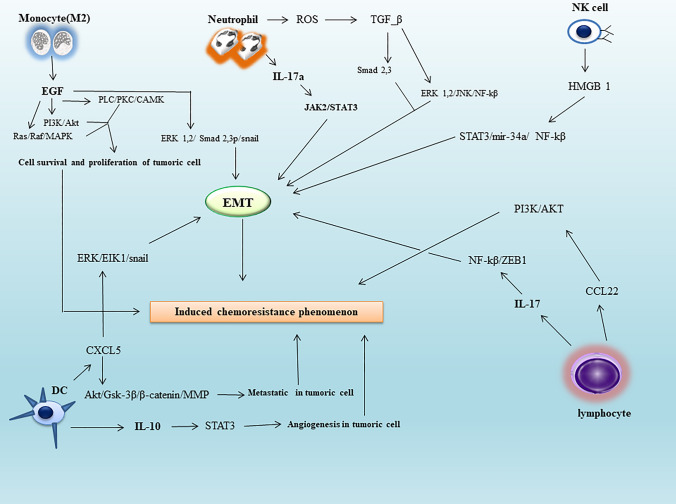
M2 macrophages can also play an important role in proliferation, angiogenesis, and EMT induction in tumor cells by secreting factors such as EGF, VEGF, and PDGF; it subsequently leads to their resistance to chemotherapy drugs [35, 36]. Secreted EGF by M2 macrophages induces RAS/RAF/MAPK, PI3K/AKT and PLC/PKC/CAMK signaling in the downstream of EGF/EGFR signal transmission pathway in the tumor cells, and via binding to the EGFR receptor on the surface of cited cells; it subsequently can be effective to create survival and proliferation of malignant cells through the mentioned signaling pathways [37] (Fig. 1).
Fig. 1.
The role of cytokine-induced immune cells in resistance to chemotherapy drugs in cancer cells. Cancer cells via EMT, cell survival, angiogenesis, proliferation, and cytokine storm-induced metastatic power can inhibit chemotherapy drugs. It leads to the chemo resistance phenomenon in people with cancer. Therefore, timely detection of signaling pathways during the cytokine storm can minimize the occurrence of chemo resistance in people with COVID-19. Abbreviation: TME tumor microenvironment, EGF epidermal growth factor, PKC protein kinase C, PI3K phosphatidylinositol-4,5-bisphosphate 3-kinase, PLC phospholipase C, MAPK mitogen-activated protein kinase, ERK extracellular signal-regulated kinase, ROS reactive oxygen species, TGF-β transforming growth factor-beta, JAK just another kinase, STAT signal transducer and activator of transcription, NF-Kβ nuclear factor kappa light chain enhancer of activated B cells, HMGB1 high mobility group box 1, miR microRNA, EMT epithelial–mesenchymal transition, MMP matrix metalloproteinase
In addition, EGF can induce EMT in the tumor cells via ERK1/2-phospho-Smad2/3-Snail signaling pathway [38, 39] (Fig. 1). VEGF can also lead to angiogenesis in TME by binding to its receptor called VEGFR; it acts via the PI3K/AKT signaling pathway. Subsequently by TGF-β/Smad and TGF-β/Snail/Slug signaling pathways, EMT is induced in tumor cells; EMT process plays an effective role in the resistance of tumor cells to chemotherapy drugs [40, 41].
On the other hand, PDGF secreted by M2 macrophages can also lead to angiogenesis by utilizing the TGF-β signaling pathway. It also induces EMT in tumor cells by Notch1/Twist1 pathway, and finally induces chemotherapy resistance [40, 42]. In general, EGF, VEGF and PDGF can induce EMT, and chemotherapy resistance in cancer cells through the TGF-β/Smad pathway. Therefore, this common pathway can be used to create appropriate therapies to reduce the effect of chemotherapy resistance [43–45]. Considering the role of monocyte to induce cytokine storm in COVID-19 patients, and also its role to induce chemo resistance in cancer patients, it seems they can worsen patient’s condition.
Therefore, activation of monocytes can be prevented by providing the appropriate cell therapies; it consequently reduces the induction of cytokine storm and chemotherapy resistance in COVID-19 cancer patients, and will improve the process of treatment.
Neutrophils
Neutrophils are important cells in defense against microorganisms; they are one of the essential cells in the innate immune responses. In addition, neutrophils in people with COVID-19 may be a part of process to induce cytokine storm. After calling neutrophils to inflamed alveolar cells and exposure to SARS-COV2, they can induce cytokine storm through the formation of Neutrophil Extra Cellular Trap (NET) network [46].
Studies have also shown, that neutrophils are involved in chemotherapy resistance induction in people with cancer. Neutrophils called Tumor Associated Neutrophil (TAN) can secrete IL-17 and release Reactive Nitrogen Species (RNS) and Reactive Oxygen Species (ROS) in TME; they also form a NET network to induce EMT in tumor cells. These processes lead to cancer cells progression and chemotherapy resistance induction [47].
TAN cells secreted IL17a in the TME binds to the IL17R receptor on the surface of tumor cells; IL17a can induce EMT in the tumor cells using JAK2/STAT3 signaling pathway in the downstream of this receptor [48]. Produced ROS by TAN cells lead to the activation of extracellular matrix-associated TGF-β latent complex. By activating the latent TGF-β complex on tumor cells surface, and then its binding to TβRI and TβRII receptors on cell surface, it activates and phosphorylates the Smad 2,3 molecules in the downstream of these receptors, which can induce EMT process in tumor cells (Fig. 1).
Also in another path called non Smad signaling pathway, which causes TGF-β binding to its receptors in the downstream, using molecules such as ERK1,2/JNK/NF-Kβ can start EMT process in tumor cells (Fig. 1). On the other hand, the activity of NOXs increases through the NF-Kβ pathway; consequently, this molecule can also produce ROS from tumor cells and leads to a synergistic effect on ROS produced by TANs. Thereby, it increases EMT induction in tumor cells [49].
RNS secreted by TANs can also lead to phosphorylation and activation of the CREB molecule by Protein Kinase A in tumor cells; this molecule can also induce EMT through these pathways [50]. In tumor cells, the CREB molecule activates an lncRNA, called HULC; this lncRNA can induce angiogenesis and EMT by suppressing miR-107 and miR-200a-3P. miR-107 suppression increases the efficiency of E2F1/SPHK1 molecules, and finally these molecules lead to angiogenesis in the tumor cells.
Suppression of miR-200a-3P also increases ZEB1 transcription factor activity; in the next step, EMT process occurs in the tumor cells. On the other hand, HULC lncRNA can induce proliferation in the tumor cells in cooperation with P18 molecule [51]. As a result, by inducing the processes of angiogenesis, EMT and tumor cells proliferation increment, chemo resistance also occurs in these cells. In addition, the formed NET network in the TME medium can activate the EMT process in tumor cells via TGF-β signaling pathway [52].
Considering the role of neutrophils to induce chemotherapy resistance and cytokine storm creation, it is hypothesized that, their function can worsen the COVID-19 cancer patient’s condition. In other words, neutrophil count increment in these patients is a sign of low survival; it also complexes the treatment process.
Dendritic cells
Dendritic cell (DC) is a professional antigen presenting cell (APC), that plays a key role in relationship between innate and acquired immune responses. DCs can also be effective in inflammation induction. Of course, their role in inflammation is effective on T-helper cells polarization. In COVID-19 patients, DCs are also indirectly involved in cytokine storm induction; they affect T helper cells [53].
Studies have shown, that DCs play an important role to induce chemo resistance in patients with malignancy. In tumor medium, DCs called Tumor-Associated Dendritic Cell (TADCs) are present, which are effective in angiogenesis, metastasis and EMT induction in tumor cells; in this way, they can cause resistance to chemotherapy drugs [54]. They are also impressive in the progression of cancer cells by secreting CCL5 chemokine, and inducing MALT1/Snail signaling pathway in tumor cells [55]. DCs can secrete IL-10; it can also induce angiogenesis in TME, by inducing IL10/STAT signaling pathway [56] (Fig. 1).
In addition, these cells can secrete CXCL5 chemokine in TME medium. By CXCL5binding to CXCR2 receptor on the surface of cancer cells, CXCL5 can induce two important signaling pathways; they are effective to induce EMT and metastasis in tumor cells. Thus, in ERK/Elk1/Snail pathway, the EMT process occurs in cancer cells, and in another pathway called AKT/GSK3β/β-Catenin/MMP, metastasis occurs in cancer cells [57] (Fig. 1).
CXCL1e is another chemokine secreted by DCs in TME; it plays an important role in the metastasis and EMT induction in tumor cells [54]. It induces the NF-Kβ/SOX4 and ERK/MMP2,9 signaling pathways by binding to its receptor on the surface of tumor cells; mentioned pathways play vital roles in tumor cell metastasis, migration and invasion [58, 59]. Also, by connecting CXCL chemokine to CXCR4, AKT signaling pathway is activated in the downstream of which is the fork head box class (FOX) molecule.
The FOX molecule can activate the Wnt signaling pathway due to T-cell Factor (TCF)/Beta-Catenin/FOX complex; it leads to EMT induction in cancer cells [60]. In TME and under acidic conditions, some cancer cells secrete TGF-β2 molecules; they bind to the TGFβR1, leading to TGF-β/Smad 2,3 pathway induction. Downstream of this pathway, the enzyme diacylglycerol O-acyltransferase (DGAT) is activated, which leads to the accumulation of lipid particles in the DCs called Lipid Droplet (LD); it prevents DCs activation and migration to the lymph nodes to induce TCD8 + function, and remove present tumor cells in TME. It inhibits tumor cells suppression by immune system [61].
Therefore, by suppressing the DCs in TME, they perform their role as the antigen presenters to produce TCD8+ cells. TCD8+ can remove tumor cells and secretes chemokines and other soluble substances, which give the ability to induce EMT and metastatic power to tumor cells. DCs induce resistance to chemotherapy in TME due to TCD8+ effects.
Considering the cited roles of DCs, they will worsen the condition of cancer patients. Therefore, using appropriate cell therapies a useful treatment strategy can be presented in COVID-19 patients suffering malignancy, to overcome the phenomenon of chemotherapy resistance.
NK cells (natural killer cell)
NK cells are in the primary line of defense against viral pathogens. They can also induce cytokine storm in COVID-19 infected patients. NK cells also play a vital role to induce resistance to chemotherapy in cancer patients. They lead to tumor cells progression through the High Mobility Group Box1 (HMGB1) molecule, which in turn induces chemo resistance [62, 63]. HMGB1 secretion induces STAT3/miR-34a/NF-Kβ signaling pathway in tumor cells; subsequently this pathway leads to angiogenesis and EMT process in tumor cells [64] (Fig. 1). In another pathway induced by HMGB1 in tumor cells, it can play a decisive role in tumor cell metastasis by interacting with the Brahma-Related Gene 1 (BRG1) molecule, and activating AKT signaling pathway [65, 66].
NK cells secrete TGF-β and macrophage migration inhibitory factor (MIF) in the tumor environment; they are also effective to induce EMT in tumor cells. TGF-β induces Snail 1,2, Id1 and Runx 2 transcription factors activity, via the Smad 2,3/Smad 4 molecule in the downstream of own signaling pathway; they are also important to set up EMT process in tumor cells. Finally, TGF-beta can induce EMT in tumor cells [67]. TGF-β can also induce migration and invasion in the tumor cells by activating the EGFR receptor through Smad3/ERK/SP1 signaling pathway; cited processes lead to chemotherapy resistance through tumor cells aggression [68, 69].
The secreted MIF molecule can induce Akt signaling pathway in tumor cells by binding to the CXCR4. It induces EMT in the tumor cells, using EMT associated transcription factors (EMT-TF). Also, EMT-TF can induce angiogenesis in TME by decreasing E-cadherin expression, and increasing N-cadherin and Vimentin expression. Angiogenesis and EMT play an effective role to induce resistance to chemotherapy drugs [70]. According to the cited facts, it seems NK cells will progress disease and increase mortality in patients.
Aberrant function of adaptive immune cells
Lymphocytes are the main executive cells in adaptive immune responses. They are involved in cytokine storm induction in COVID-19 patients, and can also induce resistance to chemotherapy drugs in tumor cells. In the tumor medium, T lymphocytes secrete cytokines such as IL-17 and IL-13; the second one causes resistance to chemotherapy drugs [71]. These cytokines induce Akt signaling pathway in tumor cells. This signaling pathway using Snail transcription factor can induce EMT process in tumor cells; it also leads to VEGF production through IL-6 upregulation and subsequent application of JAK2/STAT3 signaling pathway in the downstream of IL-6 (Akt dependent IL-6/JAK2/STAT3). VEGF plays an important role in inducing angiogenesis in TME [72].
IL-17 is also impressive in migration and invasion of tumor cells through NF-Kβ/ZEB1 signaling pathway; it also induces EMT process [73] (Fig. 1). Therefore, by inducing the EMT process and angiogenesis in TME medium, chemotherapy resistance occurs. IL-13 can also induce EMT and chemotherapy resistance via STAT6 signaling pathway [74].
CXCL13 is a T lymphocytes secreted chemokines in TME; it is also effective to induce resistance to chemotherapy drugs [75]. It can induce two MEK/ERK and Rac-GEF signaling pathways by binding to the CXCR5 on the surface of tumor cells, which result in resistance to chemotherapy drugs. MEK/ERK also leads to proliferation of tumor cells, and Rac-GEF signaling pathway is effective in cancer cells migration.
In addition to the two signaling pathways, CXCL13 binding of T lymphocytes to CXCR5 on the surface of tumor cells via PI3K/Akt signaling pathway can be effective to increase the survival of tumor cells; it is also important in cancer progression and increasing drug resistance [76].
B lymphocytes in TME medium are also effective in chemotherapy resistance creation. Thus, secretion of CCL22 chemokine from B lymphocytes can lead to tumor cells resistance to chemotherapy, through CCL22/PI3K/Akt signaling pathway [77] (Fig. 1). B lymphocytes are also important in the phenomenon of drug resistance through CXCR4/CXCL12 signaling pathway [78]. By CXCL12 binding to CXCR4, JAK2/STAT3 signaling pathway is generated in their downstream, which results in STAT3 phosphorylation by the JAK2 enzyme.
Phosphorylated STAT3 molecule can lead to BCL-XL, BCL2 and Cyclin D1 molecules transcription in tumor cells; they inhibit apoptosis and induce proliferation by cell cycle advancing; the product of which is the development of resistance to chemotherapy drugs [79]. It is hypothesized, that lymphocytes may worsen the clinical condition in COVID-19 cancer patients.
In addition to immune cells, other cells may be also important in inducing cytokine storms in patients with COVID-19; one of the most important of these cells is fibroblasts [80]. They can be effective in promoting cancer and resistance to chemotherapy by inducing cytokine storm in the cited population. IL-8 and the CXCL8 chemokine secreted by active fibroblasts bind to CXCR2 on the surface of cancer cells. In the next step, Prostaglandin J2 molecule can activate Snail, Slug, and ZEB1 transcription factors in the downstream of CXCR2 signaling pathway.
Cited transcription factors by binding to the E-box of the E-cadherin molecule, lead to downregulation of this molecule; they can induce EMT in cancer cells. Since EMT can play an important role in inducing the chemo resistance phenomenon in cancer cells, the consequence of the above signaling pathway is chemo resistance development in cancer patients with COVID-19. Also, the ZEB1 transcription factor, which is activated in downstream of this signaling pathway, can inhibit apoptosis induction of chemotherapy drugs by binding to the P53 molecule. Finally, P53 degradation leads to drug resistance in cancer patients with COVID-19 [81, 82].
Conclusion
Considering the role of immune cells and pro-inflammatory cytokines in the occurrence of cytokine storm and subsequent induction of resistance to chemotherapy (Tables 1 and 2), it is important to identify the signaling pathways. The cytokine storm is effective in the development of severe form of the disease in cancer patients infected with SARS-COV2 virus; by identifying these pathways and providing appropriate treatment strategies the incidence of disease can be reduced in cancer people with COVID-19.
Table1.
Summary of some cytokines involved in the chemotherapy resistance
| Cytokine | Type of disease | Mechanism | Potential drug | Refs. |
|---|---|---|---|---|
| IL-6 | ovarian cancer | IL-6 induced chemo resistance is dependent on the PYK2 phosphorylation | Carboplatin | [83] |
| IL-6 | NSCLC | IL-6 in the STAT3 pathway leads to the TIMP molecule expression, and thus can induce drug resistance | Gemcitabine | [84] |
| IL-1 | Pancreatic Cancer | Tumor-Stroma IL-1β-IRAK4/NF-Kβ pathway induces chemo resistance in the PDAC patient | Paclitaxel | [85, 86] |
| IL-1 | AML | IL1β/Cox-2/PGE2/β-Catenin/ARC pathway induces drug resistance in acute myeloid leukemia (AML) | Cytarabine | [87] |
| TNF-α | Breast cancer | TNF-α/NTF-ERK-GST-π axis and TNF-α/NTF-NF-κB- mediated anti-apoptotic functions are required for TNF-α-induced DOX-resistance | 6-Mercaptopurine | [88] |
| TNF-α | Breast cancer | The MEK5/Erk5 pathway through the SNAIL2 and ZEB1 molecules expression can induce EMT, and consequently drug resistance | Docetaxel | [89, 90] |
| TGF-β | Colorectal cancer | Lack of SMAD4 molecule in the downstream of TGF-β signaling can induce drug resistance by activating PI3K/Akt/CDC2/Survivin pathway | Vincristine | [91, 92] |
| TGF-β | NSCLC | Simultaneous over activation of Wnt/β-catenin and TGFβ signaling by miR-128-3p confers chemo resistance | Cisplatin | [93] |
IL-6 interleukin-6, NSCLC non-small cell lung carcinoma, TIMP-1 tissue inhibitor of metalloproteinase-1, STAT3 signal transducer and activator of transcription 3, IL-1 interlukin-1, IRAK4 interleukin-1 receptor associated kinase 4, NF-Kβ nuclear factor-kβ, PDAC pancreatic ductal adenocarcinoma, AML acute myeloid leukemia, ARC apoptosis repressor with caspase recruitment domain, COX-2 cyclooxygenase-2, PGE2 prostaglandin 2, TNF-α tumor necrosis factor alpha, NTF N-terminal fragment, ERK extracellular signal-regulated kinase, GST-π glutathione S-transferase-π, DOX doxorubicin, MEK mitogen-activated protein kinase kinase, ZEB1 δ-EF1, TGF-β transforming growth factor beta, PI3K phosphoinositide 3-kinase, Akt serine threonine kinase, CDC2 cell-division cycle 2, WNT wingless-related integration site, SMAD4 SMAD family member 4, EMT epithelial-mesenchymal transition, miR MicroRNA
Table 2.
Some of chemotherapy drugs involved in cytokines expression
| Chemotherapy | Cytokine | Mechanism | Refs. |
|---|---|---|---|
| Daunorubicin | IL-3 | IL-3 can induce ERK pathway. ERK/Mcl-1 pathway can inhibit BCL2 protein activity, and lead to drug resistance | [94] |
| Cyclophosphamide | IL-6 | IL6 can induce drug resistance via JAK/STAT3 and NF-kappaB IL6-dependent pathway | [95] |
| Taxotere | IL-8 | IL-8 can induce drug resistance and cancer progression, through PLC-PKC, PI3K-AKT, Rho-GTPase family, FAK/Src and MAPK cascade signals and NF-κB pathway | [96] |
| Methyl methane sulfonate | TGF-β | TGF-β can induce drug resistance through miR21 expression and subsequent suppression of MSH2 mRNA production | [97] |
| 5-Fluorouracil | OSM* | OSMRs/JAK1/STAT3 axis contributes to the resistance to the targeted drugs in cancer cells | [95, 98] |
| Methotrexate | IL-17 | IL-17 leads to the production of KEAP1-Nrf2 and NF-KB transcription factors, through PI3K/AKT/mTOR pathway; it can induce drug resistance by expressing the BCRP molecule | [99, 100] |
| Etoposide | IL-11 | IL-11 induces resistance to chemotherapy drugs, through gp130/JAK/STAT3/Bcl2 pathway | [101] |
| Oxaliplatin | CXCL1 | Drug resistance is induced via CXC-Chemokine/NF-κB signaling pathway | [102] |
Abbreviation: IL interleukin, ERK extracellular signal-regulated kinase, Mcl-1 myeloid cell leukemia 1, Bax Bcl-2-associated X protein, JAK Janus kinase, STAT signal transducer and activator of transcription 3, NF-Kβ nuclear factor-kβ, PLC phospholipase C, PKC protein kinase C, PI3K phosphoinositide 3-kinase, Akt serine threonine kinase, FAK focal adhesion kinase, Src short for sarcoma, MAPK mitogen-activated protein kinase, TGF-β transforming growth factor beta, miR MicroRNA, MSH2 MutS homolog 2, OSM oncostatin-M (* IL-6 family cytokine), OSMR OSM receptor, mTOR mammalian target of rapamycin, BCRP breast cancer resistance protein, KEAP1 Kelch-like ECH-associated protein 1, Nrf2 nuclear factor erythroid 2-related factor 2, gp glycoprotein, BCL-2 B-cell lymphoma 2, CXCL C-X-C motif chemokine ligand
It is important to note, when the patient is healthy after the end of cytokine storm, it will not have any effects on cancer treatment process. Therefore, only at the beginning of the cytokine storm and before its end, signaling pathways induction can have a negative effect on the treatment response in cancer patients, and also affect survival rate of these patients. So, early detection of cytokine storm in cancer patients with COVID-19 is also important to maximize the effectiveness of target therapy.
Acknowledgements
We wish to thank all our colleagues in Iranian Blood Transfusion Organization.
Author contributions
M.A has conceived the manuscript and revised it. M.H, M.M, M.S, M.B, S.A wrote the manuscript.
Data availability
This is a review study, and it is not an original. Data availability is corresponding author responsibility.
Declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Mohammad Heydarian, Email: reza.heidarian.1382@gmail.com.
Mohammadjavad Mohammadtaghizadeh, Email: dr.taghizadeh87@gmail.com.
Mahboobeh Shojaei, Email: mahboobehshojaei22@yahoo.com.
Marziyeh Babazadeh, Email: marziyehbabazadehbio@gmail.com.
Sadegh Abbasian, Email: abbasian_sadegh@yahoo.com.
Mehran Amrovani, Email: e.amrovani@gmail.com.
References
- 1.Carfì A, Bernabei R, Landi F. Persistent symptoms in patients after acute COVID-19. JAMA. 2020;324(6):603–605. doi: 10.1001/jama.2020.12603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hamidian Jahromi A, Mazloom S, Ballard D (2020) What the European and American health care systems can learn from China COVID-19 epidemic; action planning using purpose designed medical telecommunication, courier services, home-based quarantine, and COVID-19 walk-in centers. Immunopathol Persa 6(2):e17. 10.34172/ipp.2020.17. By failing to prepare, you are preparing to fail. 2020.
- 3.Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ. COVID-19: consider cytokine storm syndromes and immunosuppression. The Lancet. 2020;395(10229):1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sinha P, Matthay MA, Calfee CS. Is a “cytokine storm” relevant to COVID-19? JAMA Intern Med. 2020;180(9):1152–1154. doi: 10.1001/jamainternmed.2020.3313. [DOI] [PubMed] [Google Scholar]
- 5.Guo C, Li B, Ma H, Wang X, Cai P, Yu Q, et al. Single-cell analysis of two severe COVID-19 patients reveals a monocyte-associated and tocilizumab-responding cytokine storm. Nat Commun. 2020;11(1):1–11. doi: 10.1038/s41467-020-17834-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Daminov B, Abdullaev S, Sharapov O, Igamberdieva R. Epidemiological, clinical and morphological aspects of kidney damage in COVID-19. J Renal Injury Prev. 2021;10(3):e17. [Google Scholar]
- 7.Henderson LA, Canna SW, Schulert GS, Volpi S, Lee PY, Kernan KF, et al. On the alert for cytokine storm: immunopathology in COVID-19. Arthritis Rheumatol. 2020;72(7):1059–1063. doi: 10.1002/art.41285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Xia Y, Jin R, Zhao J, Li W, Shen H. Risk of COVID-19 for patients with cancer. Lancet Oncol. 2020;21(4):e180. doi: 10.1016/S1470-2045(20)30150-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ragab D, Salah Eldin H, Taeimah M, Khattab R, Salem R. The COVID-19 cytokine storm; what we know so far. Front Immunol. 2020;11:1446. doi: 10.3389/fimmu.2020.01446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Roohaninasab M, Sadeghzadeh-Bazargan A, Goodarzi A. Effects of laser therapy on periorbital hyperpigmentation: a systematic review on current studies. Lasers Med Sci. 2021 doi: 10.1007/s10103-020-03241-6. [DOI] [PubMed] [Google Scholar]
- 11.Coomes EA, Haghbayan H. Interleukin-6 in COVID-19: a systematic review and meta-analysis. Rev Med Virol. 2020;30(6):1–9. doi: 10.1002/rmv.2141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Khademi M, Roohaninasab M, Goodarzi A, Seirafianpour F, Dodangeh M, Khademi A. The healing effects of facial BOTOX injection on symptoms of depression alongside its effects on beauty preservation. J Cosmet Dermatol. 2021;20(5):1411–1415. doi: 10.1111/jocd.13990. [DOI] [PubMed] [Google Scholar]
- 13.Bromberg J, Wang TC. Inflammation and cancer: IL-6 and STAT3 complete the link. Cancer Cell. 2009;15(2):79–80. doi: 10.1016/j.ccr.2009.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jin W. Role of JAK/STAT3 signaling in the regulation of metastasis, the transition of cancer stem cells, and chemoresistance of cancer by epithelial–mesenchymal transition. Cells. 2020;9(1):217. doi: 10.3390/cells9010217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Preca B-T, Bajdak K, Mock K, Lehmann W, Sundararajan V, Bronsert P, et al. A novel ZEB1/HAS2 positive feedback loop promotes EMT in breast cancer. Oncotarget. 2017;8(7):11530. doi: 10.18632/oncotarget.14563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Velden JLVD, Alcorn JF, Guala AS, Badura EC, Janssen-Heininger YM. c-Jun N-terminal kinase 1 promotes transforming growth factor–β1–induced epithelial-to-mesenchymal transition via control of linker phosphorylation and transcriptional activity of Smad3. Am J Respir Cell Mol Biol. 2011;44(4):571–581. doi: 10.1165/rcmb.2009-0282OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Seddigh-Shamsi M, Mahali SN, Mozdourian M, Allahyari A, Saeedian N, Emadzade M, et al. Investigation of hematological parameters related to the severity of COVID-19 disease in Mashhad, Iran. Immunopathol Persa. 2020;7(2):e19-e. [Google Scholar]
- 18.Weyemi U, Redon CE, Sethi TK, Burrell AS, Jailwala P, Kasoji M, et al. Twist1 and Slug mediate H2AX-regulated epithelial-mesenchymal transition in breast cells. Cell Cycle. 2016;15(18):2398–2404. doi: 10.1080/15384101.2016.1198864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lin X, Zhang H, Dai J, Zhang W, Zhang J, Xue G, et al. TFF3 contributes to epithelial-mesenchymal transition (EMT) in papillary thyroid carcinoma cells via the MAPK/ERK signaling pathway. J Cancer. 2018;9(23):4430. doi: 10.7150/jca.24361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chaidos A, Katsarou A, Mustafa C, Milojkovic D, Karadimitris A. Interleukin 6-blockade treatment for severe COVID-19 in two patients with multiple myeloma. Br J Haematol. 2020;190(1):e9–e11. doi: 10.1111/bjh.16787. [DOI] [PubMed] [Google Scholar]
- 21.Sadeghzadeh-Bazargan A, Ghassemi M, Goodarzi A, Roohaninasab M, Najar Nobari N, Behrangi E. Systematic review of low-dose isotretinoin for treatment of acne vulgaris: focus on indication, dosage, regimen, efficacy, safety, satisfaction, and follow up, based on clinical studies. Dermatol Ther. 2021;34(1):e14438. doi: 10.1111/dth.14438. [DOI] [PubMed] [Google Scholar]
- 22.Apte RN, Dotan S, Elkabets M, White MR, Reich E, Carmi Y, et al. The involvement of IL-1 in tumorigenesis, tumor invasiveness, metastasis and tumor-host interactions. Cancer Metastasis Rev. 2006;25(3):387–408. doi: 10.1007/s10555-006-9004-4. [DOI] [PubMed] [Google Scholar]
- 23.Jiménez-Garduño AM, Mendoza-Rodríguez MG, Urrutia-Cabrera D, Domínguez-Robles MC, Pérez-Yépez EA, Ayala-Sumuano JT, et al. IL-1β induced methylation of the estrogen receptor ERα gene correlates with EMT and chemoresistance in breast cancer cells. Biochem Biophys Res Commun. 2017;490(3):780–785. doi: 10.1016/j.bbrc.2017.06.117. [DOI] [PubMed] [Google Scholar]
- 24.Zhang K, Chen J, Chen D, Huang J, Feng B, Han S, et al. Aurora-A promotes chemoresistance in hepatocelluar carcinoma by targeting NF-kappaB/microRNA-21/PTEN signaling pathway. Oncotarget. 2014;5(24):12916. doi: 10.18632/oncotarget.2682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhang Z, Xu J, Liu B, Chen F, Li J, Liu Y, et al. Ponicidin inhibits pro-inflammatory cytokine TNF-α-induced epithelial–mesenchymal transition and metastasis of colorectal cancer cells via suppressing the AKT/GSK-3β/Snail pathway. Inflammopharmacology. 2019;27(3):627–638. doi: 10.1007/s10787-018-0534-5. [DOI] [PubMed] [Google Scholar]
- 26.Reinhardt J, Landsberg J, Schmid-Burgk JL, Ramis BB, Bald T, Glodde N, et al. MAPK signaling and inflammation link melanoma phenotype switching to induction of CD73 during immunotherapy. Can Res. 2017;77(17):4697–4709. doi: 10.1158/0008-5472.CAN-17-0395. [DOI] [PubMed] [Google Scholar]
- 27.Black M, Barsoum IB, Truesdell P, Cotechini T, Macdonald-Goodfellow SK, Petroff M, et al. Activation of the PD-1/PD-L1 immune checkpoint confers tumor cell chemoresistance associated with increased metastasis. Oncotarget. 2016;7(9):10557. doi: 10.18632/oncotarget.7235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Peng H, Xue R, Ju Z, Qiu J, Wang J, Yan W, et al. Cancer-associated fibroblasts enhance the chemoresistance of CD73+ hepatocellular carcinoma cancer cells via HGF-Met-ERK1/2 pathway. Ann Transl Med. 2020;8(14):856. doi: 10.21037/atm-20-1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang X, Lin Y. Tumor necrosis factor and cancer, buddies or foes? 1. Acta Pharmacol Sin. 2008;29(11):1275–1288. doi: 10.1111/j.1745-7254.2008.00889.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hasanpour Dehkordi A, Keikhaei B, Bahadoram M, Razmjoo S, Barahman M (2020) Keep the corners; impact of chemotherapy on renal function. J Nephropathol 9(1)
- 31.Darakhshandeh A, Momenzadeh M. Cachexia and anorexia in cancer; a systematic review. Immunopathol Persa. 2021 doi: 10.34172/ipp.2021.20. [DOI] [Google Scholar]
- 32.Wohlleber D, Kashkar H, Gärtner K, Frings MK, Odenthal M, Hegenbarth S, et al. TNF-induced target cell killing by CTL activated through cross-presentation. Cell Rep. 2012;2(3):478–487. doi: 10.1016/j.celrep.2012.08.001. [DOI] [PubMed] [Google Scholar]
- 33.Ireland LV, Mielgo A. Macrophages and fibroblasts, key players in cancer chemoresistance. Front Cell Dev Biol. 2018;6:131. doi: 10.3389/fcell.2018.00131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nokhostin F, Dargahi MalAmir M, Tutunchi S, Rezaeeyan H. Evaluation of prognostic/diagnostic value of Hematological markers in the detection of inflammation in coronavirus disease: a review study. J Adv Med Biomed Res. 2020;28(128):171–174. [Google Scholar]
- 35.Singh S, Chakrabarti R. Consequences of EMT-driven changes in the immune microenvironment of breast cancer and therapeutic response of cancer cells. J Clin Med. 2019;8(5):642. doi: 10.3390/jcm8050642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pouladzadeh M, Safdarian M, Choghakabodi PM, Amini F, Sokooti A. Validation of red cell distribution width as a COVID-19 severity screening tool. Future Sci OA. 2021 doi: 10.2144/fsoa-2020-0199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Choudhary KS, Rohatgi N, Halldorsson S, Briem E, Gudjonsson T, Gudmundsson S, et al. EGFR signal-network reconstruction demonstrates metabolic crosstalk in EMT. PLoS Comput Biol. 2016;12(6):e1004924. doi: 10.1371/journal.pcbi.1004924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kim J, Kong J, Chang H, Kim H, Kim A. EGF induces epithelial-mesenchymal transition through phospho-Smad2/3-Snail signaling pathway in breast cancer cells. Oncotarget. 2016;7(51):85021. doi: 10.18632/oncotarget.13116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pouladzadeh M, Safdarian M, Eshghi P, Abolghasemi H, Sheibani B, Choghakabodi PM, et al. A randomized clinical trial evaluating the immunomodulatory effect of convalescent plasma on COVID-19-related cytokine storm. Intern Emerg Med. 2021 doi: 10.1007/s11739-021-02734-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nantajit D, Lin D, Li JJ. The network of epithelial–mesenchymal transition: potential new targets for tumor resistance. J Cancer Res Clin Oncol. 2015;141(10):1697–1713. doi: 10.1007/s00432-014-1840-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zadeh FJ, Akbari T, Samimi A, Davari N, Rezaeeyan H. The role of molecular mechanism of Ten-Eleven Translocation2 (TET2) family proteins in pathogenesis of cardiovascular diseases (CVDs) Mol Biol Rep. 2020;47(7):5503–5509. doi: 10.1007/s11033-020-05602-4. [DOI] [PubMed] [Google Scholar]
- 42.Chen J, Yuan W, Wu L, Tang Q, Xia Q, Ji J, et al. PDGF-D promotes cell growth, aggressiveness, angiogenesis and EMT transformation of colorectal cancer by activation of Notch1/Twist1 pathway. Oncotarget. 2017;8(6):9961. doi: 10.18632/oncotarget.14283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Grande M, Franzen A, Karlsson J-O, Ericson LE, Heldin N-E, Nilsson M. Transforming growth factor-β and epidermal growth factor synergistically stimulate epithelial to mesenchymal transition (EMT) through a MEK-dependent mechanism in primary cultured pig thyrocytes. J Cell Sci. 2002;115(22):4227–4236. doi: 10.1242/jcs.00091. [DOI] [PubMed] [Google Scholar]
- 44.Janatmakan F, Nesioonpour S, Zadeh FJ, Teimouri A, Vaziri M. Comparing the effect of clonidine and dexmedetomidine on intraoperative bleeding in spine surgery. Anesthesiol Pain Med. 2019;9(1):e83967. doi: 10.5812/aapm.83967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rajaei E, Shahbazian N, Rezaeeyan H, Mohammadi AK, Hesam S, Zayeri ZD. The effect of lupus disease on the pregnant women and embryos: a retrospective study from 2010 to 2014. Clin Rheumatol. 2019;38(11):3211–3215. doi: 10.1007/s10067-019-04682-3. [DOI] [PubMed] [Google Scholar]
- 46.Zuo Y, Zuo M, Yalavarthi S, Gockman K, Madison JA, Shi H, et al. Neutrophil extracellular traps and thrombosis in COVID-19. J Thromb Thrombolysis. 2021;51(2):446–453. doi: 10.1007/s11239-020-02324-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wu L, Saxena S, Singh RK. Neutrophils in the tumor microenvironment. Tumor Microenviron. 2020 doi: 10.1007/978-3-030-35723-8_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang J, Chen X, Shen D, Ge D, Chen J, Pei J, et al. A long noncoding RNA NR_045363 controls cardiomyocyte proliferation and cardiac repair. J Mol Cell Cardiol. 2019;127:105–114. doi: 10.1016/j.yjmcc.2018.12.005. [DOI] [PubMed] [Google Scholar]
- 49.Murillo MM, Carmona-Cuenca I, Del Castillo G, Ortiz C, Roncero C, Sánchez A, et al. Activation of NADPH oxidase by transforming growth factor-β in hepatocytes mediates up-regulation of epidermal growth factor receptor ligands through a nuclear factor-κB-dependent mechanism. Biochem J. 2007;405(2):251–259. doi: 10.1042/BJ20061846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Barlow CA, Kitiphongspattana K, Siddiqui N, Roe MW, Mossman BT, Lounsbury KM. Protein kinase A-mediated CREB phosphorylation is an oxidant-induced survival pathway in alveolar type II cells. Apoptosis. 2008;13(5):681–692. doi: 10.1007/s10495-008-0203-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ma Z, Huang H, Xu Y, He X, Wang J, Hui B, et al. Current advances of long non-coding RNA highly upregulated in liver cancer in human tumors. Onco Targets Ther. 2017;10:4711. doi: 10.2147/OTT.S136915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bozzini S, Pandolfi L, Vertui V, Frangipane V, Violatto M, De Luigi A, et al. Induction of epithelial-mesenchymal transition (EMT) by neutrophil extracellular traps (NETs) as possible molecular mechanism in CLAD. J Heart Lung Transpl. 2021;40(4):S151. [Google Scholar]
- 53.Meftahi GH, Jangravi Z, Sahraei H, Bahari Z. The possible pathophysiology mechanism of cytokine storm in elderly adults with COVID-19 infection: the contribution of “inflame-aging”. Inflamm Res. 2020;69(9):825–839. doi: 10.1007/s00011-020-01372-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hsu Y-L, Chen Y-J, Chang W-A, Jian S-F, Fan H-L, Wang J-Y, et al. Interaction between tumor-associated dendritic cells and colon cancer cells contributes to tumor progression via CXCL1. Int J Mol Sci. 2018;19(8):2427. doi: 10.3390/ijms19082427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kan JY, Wu DC, Yu FJ, Wu CY, Ho YW, Chiu YJ, et al. Chemokine (C-C motif) ligand 5 is involved in tumor-associated dendritic cell-mediated colon cancer progression through non-coding RNA MALAT-1. J Cell Physiol. 2015;230(8):1883–1894. doi: 10.1002/jcp.24918. [DOI] [PubMed] [Google Scholar]
- 56.Nakamura R, Sene A, Santeford A, Gdoura A, Kubota S, Zapata N, et al. IL10-driven STAT3 signalling in senescent macrophages promotes pathological eye angiogenesis. Nat Commun. 2015;6(1):1–14. doi: 10.1038/ncomms8847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zhao J, Ou B, Han D, Wang P, Zong Y, Zhu C, et al. Tumor-derived CXCL5 promotes human colorectal cancer metastasis through activation of the ERK/Elk-1/Snail and AKT/GSK3β/β-catenin pathways. Mol Cancer. 2017;16(1):1–15. doi: 10.1186/s12943-017-0629-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wang N, Liu W, Zheng Y, Wang S, Yang B, Li M, et al. CXCL1 derived from tumor-associated macrophages promotes breast cancer metastasis via activating NF-κB/SOX4 signaling. Cell Death Dis. 2018;9(9):1–18. doi: 10.1038/s41419-018-0876-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yang C, Yu H, Chen R, Tao K, Jian L, Peng M, et al. CXCL1 stimulates migration and invasion in ER-negative breast cancer cells via activation of the ERK/MMP2/9 signaling axis. Int J Oncol. 2019;55(3):684–696. doi: 10.3892/ijo.2019.4840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zheng X, Lin J, Wu H, Mo Z, Lian Y, Wang P, et al. Forkhead box (FOX) G1 promotes hepatocellular carcinoma epithelial-Mesenchymal transition by activating Wnt signal through forming T-cell factor-4/Beta-catenin/FOXG1 complex. J Exp Clin Cancer Res. 2019;38(1):1–15. doi: 10.1186/s13046-019-1433-3. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 61.Trempolec N, Degavre C, Doix B, Brusa D, Corbet C, Feron O. Acidosis-induced TGF-β2 production promotes lipid droplet formation in dendritic cells and alters their potential to support anti-mesothelioma T cell response. Cancers. 2020;12(5):1284. doi: 10.3390/cancers12051284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kang R, Zhang Q, Zeh HJ, Lotze MT, Tang D. HMGB1 in cancer: good, bad, or both? Clin Cancer Res. 2013;19(15):4046–4057. doi: 10.1158/1078-0432.CCR-13-0495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hajsadeghi S, Pazoki M, Pakbaz M, Zeraatian S, Zaeim MA. Aortic valve cusp aneurysm as a result of blood culture-negative infective endocarditis, interesting echocardiographic and surgical images. Echocardiography. 2020;37(3):469–471. doi: 10.1111/echo.14609. [DOI] [PubMed] [Google Scholar]
- 64.Zhang Y, Ren H, Li J, Xue R, Liu H, Zhu Z, et al. Elevated HMGB1 expression induced by hepatitis B virus X protein promotes epithelial-mesenchymal transition and angiogenesis through STAT3/miR-34a/NF-κB in primary liver cancer. Am J Cancer Res. 2021;11(2):479. [PMC free article] [PubMed] [Google Scholar]
- 65.Lv DJ, Song XL, Huang B, Yu Y-Z, Shu FP, Wang C, et al. HMGB1 promotes prostate cancer development and metastasis by interacting with Brahma-related gene 1 and activating the Akt signaling pathway. Theranostics. 2019;9(18):5166. doi: 10.7150/thno.33972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Hajsadeghi S, Amirfarhangi A, Pakbaz M, Pazoki M, Tanha K. Postinfarction intramyocardial dissection, an interesting case report and systematic review. Echocardiography. 2020;37(1):124–131. doi: 10.1111/echo.14565. [DOI] [PubMed] [Google Scholar]
- 67.Sancisi V, Gandolfi G, Ragazzi M, Nicoli D, Tamagnini I, Piana S, et al. Cadherin 6 is a new RUNX2 target in TGF-β signalling pathway. PLoS ONE. 2013;8(9):e75489. doi: 10.1371/journal.pone.0075489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zhao Y, Ma J, Fan Y, Wang Z, Tian R, Ji W, et al. TGF-β transactivates EGFR and facilitates breast cancer migration and invasion through canonical Smad3 and ERK/Sp1 signaling pathways. Mol Oncol. 2018;12(3):305–321. doi: 10.1002/1878-0261.12162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hajsadeghi S, Pazoki M, Meimand SE, Pouresmaeeli M, Zeraatian S. More is better, not always true: misdiagnosis tamponade in postoperative patient due to thick hemostatic tissue. Echocardiography. 2021;38(6):1074–1076. doi: 10.1111/echo.15061. [DOI] [PubMed] [Google Scholar]
- 70.Guo X, Xu S, Gao X, Wang J, Xue H, Chen Z, et al. Macrophage migration inhibitory factor promotes vasculogenic mimicry formation induced by hypoxia via CXCR4/AKT/EMT pathway in human glioblastoma cells. Oncotarget. 2017;8(46):80358. doi: 10.18632/oncotarget.18673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Salazar Y, Zheng X, Brunn D, Raifer H, Picard F, Zhang Y, et al. Microenvironmental Th9 and Th17 lymphocytes induce metastatic spreading in lung cancer. J Clin Investig. 2020;130(7):3560–3575. doi: 10.1172/JCI124037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Gu F-M, Li Q-L, Gao Q, Jiang J-H, Zhu K, Huang X-Y, et al. IL-17 induces AKT-dependent IL-6/JAK2/STAT3 activation and tumor progression in hepatocellular carcinoma. Mol Cancer. 2011;10(1):1–13. doi: 10.1186/1476-4598-10-150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Gu K, Li M-M, Shen J, Liu F, Cao J-Y, Jin S, et al. Interleukin-17-induced EMT promotes lung cancer cell migration and invasion via NF-κB/ZEB1 signal pathway. Am J Cancer Res. 2015;5(3):1169. [PMC free article] [PubMed] [Google Scholar]
- 74.Cao H, Zhang J, Liu H, Wan L, Zhang H, Huang Q, et al. IL-13/STAT6 signaling plays a critical role in the epithelial-mesenchymal transition of colorectal cancer cells. Oncotarget. 2016;7(38):61183. doi: 10.18632/oncotarget.11282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kobayashi S, Murata K, Shibuya H, Morita M, Ishikawa M, Furu M, et al. A distinct human CD4+ T cell subset that secretes CXCL13 in rheumatoid synovium. Arthritis Rheum. 2013;65(12):3063–3072. doi: 10.1002/art.38173. [DOI] [PubMed] [Google Scholar]
- 76.Kazanietz MG, Durando M, Cooke M. CXCL13 and its receptor CXCR5 in cancer: inflammation, immune response, and beyond. Front Endocrinol. 2019;10:471. doi: 10.3389/fendo.2019.00471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Wei C, Yang C, Wang S, Shi D, Zhang C, Lin X, et al. M2 macrophages confer resistance to 5-fluorouracil in colorectal cancer through the activation of CCL22/PI3K/AKT signaling. Onco Targets Ther. 2019;12:3051. doi: 10.2147/OTT.S198126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Shen M, Wang J, Ren X. New insights into tumor-infiltrating B lymphocytes in breast cancer: clinical impacts and regulatory mechanisms. Front Immunol. 2018;9:470. doi: 10.3389/fimmu.2018.00470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wang M, Lin T, Wang Y, Gao S, Yang Z, Hong X, et al. CXCL12 suppresses cisplatin-induced apoptosis through activation of JAK2/STAT3 signaling in human non-small-cell lung cancer cells. Onco Targets Ther. 2017;10:3215. doi: 10.2147/OTT.S133055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Rowaiye AB, Okpalefe OA, Adejoke OO, Ogidigo JO, Oladipo OH, Ogu AC, et al. Attenuating the effects of novel COVID-19 (SARS-CoV-2) infection-induced cytokine storm and the implications. J Inflamm Res. 2021;14:1487. doi: 10.2147/JIR.S301784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Choi J, Suh J-Y, Kim D-H, Na H-K, Surh Y-J. 15-Deoxy-Δ12, 14-prostaglandin J2 induces epithelial-to-mesenchymal transition in human breast cancer cells and promotes fibroblast activation. J Cancer Prev. 2020;25(3):152. doi: 10.15430/JCP.2020.25.3.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Jiang J, Wang K, Chen Y, Chen H, Nice EC, Huang C. Redox regulation in tumor cell epithelial–mesenchymal transition: Molecular basis and therapeutic strategy. Signal Transduct Target Ther. 2017;2(1):1–12. doi: 10.1038/sigtrans.2017.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Pasquier J, Gosset M, Geyl C, Hoarau-Véchot J, Chevrot A, Pocard M, et al. CCL2/CCL5 secreted by the stroma induce IL-6/PYK2 dependent chemoresistance in ovarian cancer. Mol Cancer. 2018;17(1):1–14. doi: 10.1186/s12943-018-0787-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Xiao W, Wang L, Howard J, Kolhe R, Rojiani AM, Rojiani MV. TIMP-1-mediated chemoresistance via induction of IL-6 in NSCLC. Cancers. 2019;11(8):1184. doi: 10.3390/cancers11081184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Zhang D, Li L, Jiang H, Li Q, Wang-Gillam A, Yu J, et al. Tumor–stroma IL1β-IRAK4 feedforward circuitry drives tumor fibrosis, chemoresistance, and poor prognosis in pancreatic cancer. Can Res. 2018;78(7):1700–1712. doi: 10.1158/0008-5472.CAN-17-1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Voloshin T, Alishekevitz D, Kaneti L, Miller V, Isakov E, Kaplanov I, et al. Blocking IL1β pathway following paclitaxel chemotherapy slightly inhibits primary tumor growth but promotes spontaneous metastasis. Mol Cancer Ther. 2015;14(6):1385–1394. doi: 10.1158/1535-7163.MCT-14-0969. [DOI] [PubMed] [Google Scholar]
- 87.Carter BZ, Mak PY, Wang X, Tao W, Ruvolo V, Mak D, et al. An ARC-regulated IL1β/Cox-2/PGE2/β-Catenin/ARC circuit controls leukemia-microenvironment interactions and confers drug resistance in AML. Can Res. 2019;79(6):1165–1177. doi: 10.1158/0008-5472.CAN-18-0921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Huang H-Y, Chang H-F, Tsai M-J, Chen J-S, Wang M-J. 6-Mercaptopurine attenuates tumor necrosis factor-α production in microglia through Nur77-mediated transrepression and PI3K/Akt/mTOR signaling-mediated translational regulation. J Neuroinflammation. 2016;13(1):1–20. doi: 10.1186/s12974-016-0543-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Zhou C, Nitschke AM, Xiong W, Zhang Q, Tang Y, Bloch M, et al. Proteomic analysis of tumor necrosis factor-α resistant human breast cancer cells reveals a MEK5/Erk5-mediated epithelial-mesenchymal transition phenotype. Breast Cancer Res. 2008;10(6):1–14. doi: 10.1186/bcr2210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Edwardson DW, Boudreau J, Mapletoft J, Lanner C, Kovala AT, Parissenti AM. Inflammatory cytokine production in tumor cells upon chemotherapy drug exposure or upon selection for drug resistance. PLoS ONE. 2017;12(9):e0183662. doi: 10.1371/journal.pone.0183662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Itatani Y, Kawada K, Sakai Y. Transforming growth factor-β signaling pathway in colorectal cancer and its tumor microenvironment. Int J Mol Sci. 2019;20(23):5822. doi: 10.3390/ijms20235822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Zhou L, Tang J, Hu F, Liao Y, Li R, Zhou Y, et al. Effects of different levels of TGF-β expression and tumor cell necrosis rates in osteosarcoma on the chemotherapy resistance of osteosarcoma. J Bone Oncol. 2020;23:100299. doi: 10.1016/j.jbo.2020.100299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Cai J, Fang L, Huang Y, Li R, Xu X, Hu Z, et al. Simultaneous overactivation of Wnt/β-catenin and TGFβ signalling by miR-128-3p confers chemoresistance-associated metastasis in NSCLC. Nat Commun. 2017;8(1):1–19. doi: 10.1038/ncomms15870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.McCubrey JA, Steelman LS, Franklin RA, Abrams SL, Chappell WH, Wong EW, et al. Targeting the RAF/MEK/ERK, PI3K/AKT and p53 pathways in hematopoietic drug resistance. Adv Enzyme Regul. 2007;47:64. doi: 10.1016/j.advenzreg.2006.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Chen W, Qin Y, Liu S. Cytokines, breast cancer stem cells (BCSCs) and chemoresistance. Clin Transl Med. 2018;7(1):1–13. doi: 10.1186/s40169-018-0205-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Li JS, Zhao P, Yang LP, Li Y, Tian YG, Li SY, et al. Integrating 3-omics data analyze rat lung tissue of COPD states and medical intervention by delineation of molecular and pathway alterations. Biosci Rep. 2017;37:BSR20170042. doi: 10.1042/BSR20170042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Li Y, Rong G, Kang H. Taxotere-induced elevated expression of IL8 in carcinoma-associated fibroblasts of breast invasive ductal cancer. Oncol Lett. 2017;13(3):1856–1860. doi: 10.3892/ol.2017.5612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Shien K, Papadimitrakopoulou VA, Ruder D, Behrens C, Shen L, Kalhor N, et al. JAK1/STAT3 activation through a proinflammatory cytokine pathway leads to resistance to molecularly targeted therapy in non–small cell lung cancer. Mol Cancer Ther. 2017;16(10):2234–2245. doi: 10.1158/1535-7163.MCT-17-0148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Dong C, Wu J, Chen Y, Nie J, Chen C. Activation of PI3K/AKT/mTOR pathway causes drug resistance in breast cancer. Front Pharmacol. 2021;12:143. doi: 10.3389/fphar.2021.628690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Lainetti PDF, Leis-Filho AF, Laufer-Amorim R, Battazza A, Fonseca-Alves CE. Mechanisms of resistance to chemotherapy in breast cancer and possible targets in drug delivery systems. Pharmaceutics. 2020;12(12):1193. doi: 10.3390/pharmaceutics12121193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Ma J, Song X, Xu X, Mou Y. Cancer-associated fibroblasts promote the chemo-resistance in gastric cancer through secreting IL-11 targeting JAK/STAT3/Bcl2 pathway. Cancer Res Treatm. 2019;51(1):194. doi: 10.4143/crt.2018.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.de Porras VR, Bystrup S, Martínez-Cardús A, Pluvinet R, Sumoy L, Howells L, et al. Curcumin mediates oxaliplatin-acquired resistance reversion in colorectal cancer cell lines through modulation of CXC-Chemokine/NF-κB signalling pathway. Sci Rep. 2016;6(1):1–17. doi: 10.1038/srep24675. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
This is a review study, and it is not an original. Data availability is corresponding author responsibility.