Abstract
Background and Purpose
CD40 and CD40 ligand (CD40L) are costimulatory molecules of the tumor necrosis factor receptor superfamily and well known for their involvement in inflammatory diseases: atherosclerotic mouse models with disrupted CD40 signalling develop lesions of reduced size with a more stable plaque profile. This study investigated the potential of plasma and intraplaque levels of CD40 and CD40L as markers for cardiovascular disease (CVD) in humans and their association with plaque stability.
Methods
Soluble CD40 and CD40L (sCD40L) were measured in plasma in 1,437 subjects from The SUrrogate markers for Micro- and Macro-vascular hard endpoints for Innovative diabetes Tools (SUMMIT) cohort. Intra-plaque levels of sCD40 and sCD40L were measured in atherosclerotic plaque homogenates from 199 subjects of the Carotid Plaque Imaging Project (CPIP) cohort.
Results
Both plasma sCD40 and sCD40L levels were elevated in individuals with prevalent stroke, while sCD40 levels also were higher in individuals with a prior acute myocardial infarction. Plasma levels of sCD40 correlated with carotid intima-media thickness and total carotid plaque area and were associated with risk of cardiovascular events over a 3-year follow-up period. Intra-plaque levels of sCD40 and sCD40L were associated with plaque components characteristic for plaque vulnerability and extracellular matrix remodelling.
Conclusions
Higher plasma sCD40 and sCD40L levels are associated with prevalent CVD. Plasma sCD40 levels also correlate with the severity of carotid atherosclerosis and predict future cardiovascular events, while intra-plaque levels correlate with a vulnerable plaque phenotype. Our findings thus demonstrate that elevated levels of sCD40 and sCD40L are markers of CVD.
Keywords: CD40, CD40 ligand, Atherosclerosis, Carotid arteries, Inflammation
Introduction
Cardiovascular disease (CVD) is the leading cause of mortality worldwide, accounting for 17.9 million deaths each year [1]. The main underlying cause is atherosclerosis. Compromised stability of atherosclerotic plaques results in thrombus formation, and, consequently, stroke or myocardial infarction (MI) depending on the affected territory. Characteristics of a classically rupture-prone or vulnerable plaque include a thin fibrous cap, abundance of lipids, a prominent necrotic core and infiltration by immune cells, such as macrophages and T cells [2], while eroded lesions are less inflamed and rich in proliferating smooth muscle cells (SMCs), proteoglycans and hyaluronan, and neovascularization [3]. Ideally, ameliorating atherosclerosis involves two steps: (1) finding a reliable method to identify individuals with vulnerable plaques to start treatment in time to prevent ruptures—and subsequently symptoms, and (2) identifying key players driving destabilization in rupture-prone plaques. Addressing both steps, this study not only evaluates the CD40/CD40L signalling dyad as a potential CVD marker but also explores its biological association with a vulnerable plaque phenotype.
CD40 and CD40 ligand (CD40L) are costimulatory molecules and members of the tumor necrosis factor (receptor) superfamily (TNF(R)SF) [4]. CD40 is expressed on a variety of immune and non-immune cells, including B cells, monocytes/macrophages, dendritic cells, SMCs and endothelial cells. CD40L is mainly expressed by activated T cells and platelets, but expression has also been described on B cells, as well as on endothelial, epithelial, and SMCs [5] CD40 signalling is propagated via adaptor proteins—the TNF receptor-associated factors [6] —and modulates a multitude of immunological responses [5]. Atherosclerotic mouse models (apolipoprotein E [ApoE-/-], low-density lipoprotein receptor [LDLr-/-]) with disrupted CD40 signalling develop lesions of reduced size with a more stable plaque profile, an effect achieved by CD40/CD40L knock out models, as well as by small molecule inhibitors and antibodies inhibiting the CD40 signalling pathway [6-9].
While not much is known about the specific actions of CD40 in human CVD, an association for CD40L in CVD has been suggested. Increased levels of platelet CD40L have been reported in patients with hypercholesterolemia [10] and plasma levels of soluble CD40L (sCD40L) were increased in patients with acute coronary syndromes [11]. Furthermore, clinical evidence for involvement of sCD40L in CVD include association with cardiovascular (CV) events in coronary artery disease [11], recurrent cardiac events and future events in healthy women [12], carotid intima-media thickness (IMT) and hypertension [13], and via correlations with other biomarkers of inflammation [14]. A number of studies, however, have found no associations between plasma sCD40L and CVD [15,16] or long-term CV (or all-cause) mortality [17]. Further clarification is thus essential to determine the value of sCD40L as a marker of CVD, and to investigate whether sCD40 may be a useful complement or replacement biomarker.
To address the potential of sCD40 and sCD40L as markers of CVD, we have analysed plasma and carotid artery plaque levels of CD40/CD40L for associations with CV events, as well as carotid IMT and plaque vulnerability markers in two large human cohorts.
Methods
Extended methods are provided in the Supplementary Methods.
Study cohorts and samples
Plasma samples were included from 1,438 subjects (Figure 1) of the SUrrogate markers for Micro- and Macro-vascular hard endpoints for Innovative diabetes Tools (SUMMIT) study [18] population, recruited between December 2010 and April 2013 from existing population cohorts and hospital registries at the university hospitals in Malmö (Sweden), Pisa (Italy), Dundee and Exeter (UK).
Figure 1.
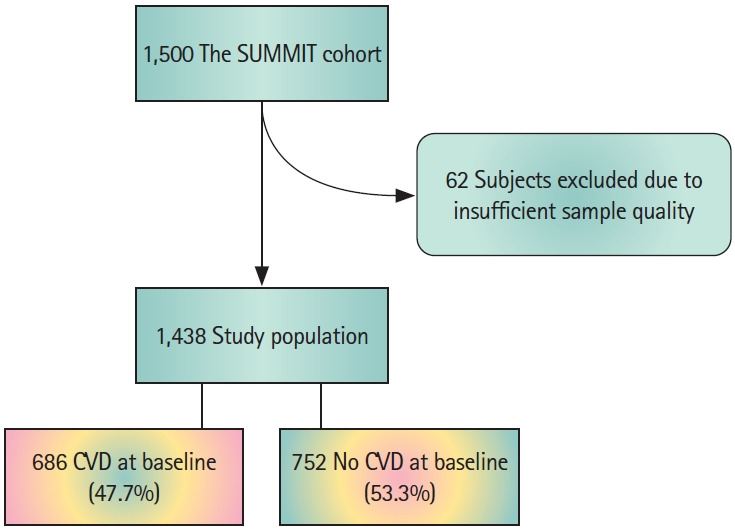
Flow chart showing the study population of the The SUrrogate markers for Micro- and Macro-vascular hard endpoints for Innovative diabetes Tools (SUMMIT) cohort, including excluded subjects and subjects with/without cardiovascular disease (CVD).
Carotid plaques (n=198) were collected from patients during carotid endarterectomies at the Vascular Department of Skane University Hospital (Malmö, Sweden) between 2005 and 2010 (The Carotid Plaque Imaging Project [CPIP]) [19]. Immediately during surgery, removed plaques were snap-frozen in liquid nitrogen. Histological analyses were performed on portions (1 mm thick) cut from the most stenotic plaque region and embedded in optimal cutting medium (Sakura Finetek Europe BV, Tokyo, Japan). Plaque homogenates were prepared from the rest of the plaques and used for all other plaque component quantifications. Clinical characteristics of the included patients are summarized in Supplementary Table 1. The studies were approved by the local ethical review boards and were carried out in accordance with the principles of the Declaration of Helsinki. There was no overlap between subjects of the two study cohorts.
Statistics
Shapiro-Wilk and D’Agostino-Pearson omnibus K2 tests were used to assess Gaussian distribution. Variables found to be normally distributed are shown as mean with standard deviation while non-normally distributed variables are shown as median with interquartile range (IQR). For analysis of plasma, plaque histology sections and homogenates, the Mann-Whitney U test was used to compare groups and Spearman’s rank correlation was used for continuous variables. For comparisons between categorical variables, the chi-square test was used. A logistic regression model was used to test for associations with CV events and mortality in the SUMMIT cohort. The model was adjusted for age, diabetes and prevalent ischemic stroke, acute myocardial infarction (AMI), and hypertension as covariates. A P-value of <0.05 was considered statistically significant. Statistical analysis was performed using SPSS version 26.0 (IBM Co., Armonk, NY, USA) and GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA, www. graphpad.com). Adjustments for multiple comparisons were done when appropriate using the Holm-Šídák test.
Results
Plasma sCD40 levels are associated with stroke and acute myocardial infarction
To assess associations between sCD40, sCD40L and CVD, we analysed the plasma from 1,438 subjects from the SUMMIT cohort using Proximity Extension Assay (PEA). Comparatively high sCD40 levels were associated with prevalent ischemic stroke (P=3.0×10–5), MI (P=0.016), and hypertension (P=0.00021) (Table 1 and Supplementary Tables 2 and 3). The association with stroke remained after adjusting for age, diabetes, hypertension, and MI (odds ratio [OR], 1.5; 95% confidence interval [CI], 1.084 to 1.959; P=0.013) and the association with MI after adjusting for age, diabetes, hypertension, and stroke (OR, 1.2; 95% CI, 1.006 to 1.393; P=0.042).
Table 1.
Spearman correlations between the circulating levels of sCD40 and sCD40L (arbitrary unit, SUMMIT cohort) and carotid plaque burden
| Vascular ultrasound* | CD40 (n=1,438 at inclusion; n=798 at follow-up) |
CD40L (n=1,438 at inclusion; n=798 at follow-up) |
||||
|---|---|---|---|---|---|---|
| r | P | P, adjusted† | r | P | P, adjusted | |
| Total plaque area‡ | 0.355 | <1×10–16 | <1×10–16 | 0.409 | <1×10–16 | <1×10–16 |
| Maximum plaque area (mm2) | 0.322 | <1×10–16 | <1×10–16 | 0.340 | <1×10–16 | <1×10–16 |
| No. of plaques, right carotid artery | 0.354 | <1×10–16 | <1×10–16 | 0.152 | 2.0×10–8 | 1.0×10–7 |
| No. of plaques, left carotid artery | 0.398 | <1×10–16 | <1×10–16 | 0.142 | 1.9×10–7 | 7.6×10–7 |
| Plaque length (mm) | 0.316 | <1×10–16 | <1×10–16 | 0.286 | 1.3×10–15 | 9.3×10–15 |
| Plaque height (mm) | 0.223 | 5.9×10–10 | 1.2×10–9 | 0.164 | 0.000006 | 0.000018 |
| Lumen diameter reduction (%) | 0.129 | 0.00041 | 0.00041 | 0.224 | 5.2×10–10 | 3.1×10–9 |
| Pulse wave velocity | 0.247 | 8.0×10–8 | <1×10–16 | 0.057 | 0.052 | 0.052 |
sCD40, soluble CD40; sCD40L, soluble CD40 ligand; SUMMIT, The SUrrogate markers for Micro- and Macro-vascular hard endpoints for Innovative diabetes Tools.
Measured at study inclusion;
P-value adjusted for multiple comparisons;
Sum of all plaque areas/patient.
Similar to sCD40, higher plasma sCD40L levels were associated with prevalent stroke (P=0.020). However, sCD40L levels were not associated with hypertension or prevalent AMI, and associations did not remain after adjusting for the aforementioned confounders (stroke: OR, 1.1; 95% CI, 0.976 to 1.304; P=0.103; and MI: OR, 1.0; 95% CI, 0.968 to 1.125; P=0.270).
Plasma sCD40 levels are associated with carotid plaque burden and arterial stiffness
Plaque burden in the carotid artery was assessed via ultrasound imaging at study inclusion and we searched for possible associations between sCD40 and sCD40L and plaque burden. A positive Spearman’s rank correlation was found between both plasma sCD40 and sCD40L levels and total plaque area (r=0.355, P<1×10–16 and r=0.409, P<1×10–16, respectively) (Table 2). The association between sCD40 and sCD40L and carotid plaque burden was also reflected by positive correlations with overall number of plaques, measurements of plaque length, height and area, as well as with lumen diameter reduction (Table 2).
Table 2.
Spearman correlations between the circulating levels of sCD40 and sCD40L (arbitrary unit, SUMMIT cohort) and IMT in the CCA and the carotid bulb region assessed by vascular ultrasound (measured at study inclusion)
| IMT measurements | CD40 (n=1,438 at inclusion; n=798 at follow-up) |
CD40L (n=1,438 at inclusion; n=798 at follow-up) |
||||
|---|---|---|---|---|---|---|
| r | P | P, adjusted* | r | P | P, adjusted | |
| Right | ||||||
| Mean IMT in CCA | 0.139 | 2.7×10–7 | 1.1×10–6 | 0.053 | 0.048 | 0.256 |
| Max IMT in CCA | 0.122 | 0.000006 | 0.000018 | 0.012 | 0.65 | 0.903 |
| Mean IMT in bulb | 0.234 | 1.4×10–16 | 6.6×10–16 | 0.081 | 0.005 | 0.039 |
| Max IMT in bulb | 0.268 | <1×10–16 | <1×10–16 | 0.077 | 0.008 | 0.055 |
| Left | ||||||
| Mean IMT in CCA | 0.088 | 0.001 | 0.0020 | 0.042 | 0.12 | 0.472 |
| Max IMT in CCA | 0.073 | 0.007 | 0.0070 | 0.000 | 0.99 | 0.990 |
| Mean IMT in bulb | 0.203 | 9.1×10–13 | 4.5×10–12 | 0.017 | 0.54 | 0.903 |
| Max IMT in bulb | 0.240 | <1×10–16 | <1×10–16 | 0.038 | 0.19 | 0.570 |
sCD40, soluble CD40; sCD40L, soluble CD40 ligand; SUMMIT, The SUrrogate markers for Micro- and Macro-vascular hard endpoints for Innovative diabetes Tools; IMT, intima-media thickness; CCA, common carotid artery.
P-value adjusted for multiple comparisons.
Arterial stiffness—a well-known factor associated with atherosclerosis progression [20]—was studied by pulse wave velocity at study inclusion. Plasma levels of sCD40, but not sCD40L, correlated with carotid arterial stiffness (r=0.247, P<1×10–16) (Table 2). Furthermore, sCD40 plasma levels measured at study inclusion showed a positive correlation with both mean and maximum IMT in the right and left common carotid artery (CCA), as well as in in the plaque bulb regions (Table 3). Association was also found between plasma sCD40L and mean IMT in the bulb region of the right carotid artery (Table 3).
Table 3.
Logistic regression analysis of association between the circulating levels of sCD40 (sCD40, SUMMIT cohort) and cardiovascular events (adjusted for confounding factors*) (n=1,438)
| Variable | Confidence interval | Odds ratio | P |
|---|---|---|---|
| sCD40 | 1.042–1.625 | 1.3 | 0.020 |
| Age | 0.996–1.042 | 1.0 | 0.109 |
| Diabetes | 0.759–1.681 | 1.1 | 0.549 |
| Hypertension | 0.596–1.319 | 0.9 | 0.553 |
| Acute myocardial infarction | 1.178–2.467 | 1.7 | 0.005 |
| Stroke | 1.058–3.507 | 1.9 | 0.032 |
sCD40, soluble CD40; SUMMIT, The SUrrogate markers for Micro- and Macro-vascular hard endpoints for Innovative diabetes Tools.
Odds ratios were adjusted for age (years) and prevalent diabetes, hypertension, prior acute myocardial infarction, and stroke (yes vs. no).
Plasma sCD40L levels are associated with carotid artery plaque progression and sCD40 levels with future cardiovascular events
To elucidate whether plasma sCD40 and sCD40L levels predicted carotid atherosclerosis progress, in addition to correlating with plaque burden at study inclusion, we compared the initial IMT measurements calculated via ultrasound imaging with IMT measurements taken after 3 years. Plasma sCD40L, but not sCD40, levels were associated with change—specifically plaque growth—in maximum IMT in the CCA and mean and maximum IMT in the bulb region of the plaques (Supplementary Table 4).
As sCD40L levels correlated with carotid plaque progression, we then investigated the potential for plasma sCD40 and sCD40L levels to predict future CV events using logistic regression. Interestingly, the level of sCD40, but not sCD40L, in plasma was associated with the occurrence of fatal or non-fatal CV events during the 3 years follow-up period (OR, 1.3; 95% CI, 1.042 to 1.625) after adjusting for age, diabetes, hypertension as well as prevalent AMI and stroke at baseline (Table 3). Neither sCD40 nor sCD40L was associated with mortality (CV or all-cause). Considering sCD40 (with confounders age, diabetes, hypertension as well as prevalent MI and stroke at baseline) in a multiple logistic regression, the area under the curve achieved was 0.629 (95% CI, 0.582 to 0.677), with sensitivities and specificities for varying cut-offs of the predicted values (10% to 90% quantiles) presented in Supplementary Table 5.
CD40 and CD40L have similar expression patterns in human carotid plaques
To further explore the presence of CD40 and CD40L in atherosclerosis we examined their expression pattern in carotid endarterectomy plaques from the CPIP cohort (Figure 2). Immunofluorescence staining was performed on sections obtained from the most stenotic plaque region. CD40 and CD40L expression was observed in several plaque parts, in particular in the shoulder and cap regions. Immunoreactivity was also found—though far less prominent—in the base of the plaque.
Figure 2.
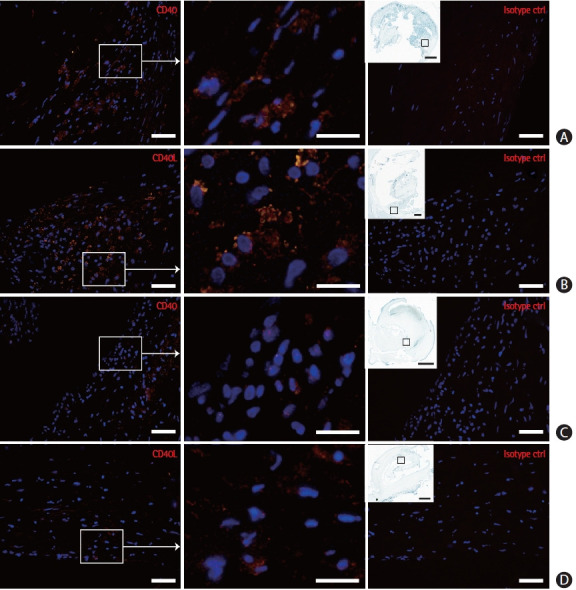
CD40 and CD40 ligand (CD40L) localization in endarterectomy plaques. CD40 and CD40L immunoreactivity was found in particular in the shoulder (A, B) and cap (C, D) regions. Squares in insets mark enlarged plaque regions. Scale bars are 50 μm (far left and right image columns), 20 μm (middle column), and 1 mm (insets, far left column) (n=22 plaques).
sCD40 is associated with a vulnerable plaque phenotype
To further explore the presence of CD40 and CD40L in atherosclerosis we examined their expression in homogenates from 198 carotid endarterectomy plaques of the CPIP cohort (here comprised of both the soluble and cell associated isoforms). Disrupted CD40 signalling in a murine atherosclerosis model results in increased stabilisation characterised by reductions in lipid core size and inflammatory cell presence accompanied by increased collagen content [6-9]. We therefore investigated whether intra-plaque levels of these molecules are similarly associated with a vulnerable plaque profile in human lesions. Indeed, we found sCD40 plaque levels measured in plaque homogenates of the CPIP cohort to correlate with plaque components collectively indicating a vulnerable plaque phenotype (Table 4). Accordingly, there was a correlation between sCD40 and lipid content (visualized on tissue sections by Oil Red O, r=0.199, P=0.012; and oxidized low density lipoprotein [oxLDL] measured in plaque homogenate, r=0.236, P=0.004), whereas a negative correlation was found with calcium (von Kossa, r= –0.208, P=0.012). The level of sCD40L in plaque showed an overall association with a vulnerable plaque phenotype, albeit weaker than that for sCD40 levels (Table 4). sCD40L levels correlated with the plaque content of lipids (oxLDL measured in plaque homogenate, r=0.259, P=0.0037), while there was an inverse correlation with plaque levels of calcium (von Kossa, r=–0.268, P=0.00034). Finally, for further plaque profile evaluation a vulnerability index was calculated, which was also found to correlate with both sCD40 (r=0.178, P=0.029) and sCD40L (r=0.180, P=0.041) plaque levels.
Table 4.
Spearman correlations of CD40 and CD40L (A.U., CPIP cohort) measured in plaque homogenate versus plaque elements
| Variable | CD40 (n=198) |
CD40L (n=198) |
||||
|---|---|---|---|---|---|---|
| r | P | P, adjusted* | r | P | P, adjusted | |
| Histology (area %) | ||||||
| Smooth muscle α-actin | –0.070 | 0.33 | 0.33 | –0.142 | 0.048 | 0.094 |
| Calcium (van Kossa) | –0.208 | 0.004 | 0.016 | –0.268 | 0.00017 | 0.00068 |
| Oil Red O | 0.199 | 0.005 | 0.016 | 0.043 | 0.55 | 0.55 |
| Vulnerability index† | 0.178 | 0.014 | 0.029 | 0.180 | 0.014 | 0.041 |
| Cytokines (pg/g) | ||||||
| CXCL1 | 0.228 | 0.002 | 0.012 | 0.268 | 0.002 | 0.008 |
| IL-6 | 0.305 | 0.00032 | 0.0022 | 0.235 | 0.001 | 0.007 |
| IL-12p70 | 0.211 | 0.004 | 0.020 | 0.230 | 0.002 | 0.008 |
| Tumor necrosis factor α | 0.391 | 3.4×10–8 | 3.1×10–7 | 0.290 | 6.9×10–5 | 0.0006 |
| CCL5 | 0.021 | 0.772 | 0.772 | 0.200 | 0.007 | 0.014 |
| CCL2 | 0.175 | 0.017 | 0.034 | 0.186 | 0.001 | 0.007 |
| CCL4 | 0.202 | 0.006 | 0.020 | 0.186 | 0.011 | 0.014 |
| PDGF-AA/AB/BB | 0.268 | 0.0002 | 0.0018 | 0.243 | 0.001 | 0.007 |
| Plaque components (mg/g) | ||||||
| oxLDL (μU/g) | 0.236 | 0.001 | 0.004 | 0.259 | 0.00041 | 0.0037 |
CD40L, CD40 ligand; A.U., arbitrary unit; CPIP, Carotid Plaque Imaging Project; CXCL1, C-X-C motif chemokine ligand 1; IL, interleukin; CCL, chemokine (C-C motif) ligand; PDGF, platelet-derived growth factor; oxLDL, oxidized low density lipoprotein.
P-value adjusted for multiple comparisons;
Calculated as (CD68++glycophorin A++Oil Red O+ area%) / (smooth muscle α-actin++collagen++von Kossa+area%).
CD40 ligation has been previously well-described to induce pro-inflammatory cytokine and chemokines, including interleukin (IL)-1β, IL-6, IL-8, IL-12, TNF-α, and chemokine (C-C motif) ligand 5 (CCL5) in vitro [21-23]. Concordantly, sCD40 levels correlated with both IL-12p70 and CCL2 measured in plaque homogenate, as well as with several other inflammatory cytokines and chemokines, including IL-6, TNF-α, CCL4, and C-X-C motif chemokine ligand 1 (CXCL1) (Table 4). Correlation was also found with platelet-derived growth factor (PDGF). Similarly, sCD40L levels correlated with the inflammatory cytokines and chemokines IL-12p70, CCL2, IL-6, TNF-α, CCL4, CCL5, CXCL1 as well as with PDGF (Table 4).
sCD40 plaque levels are associated with features of extracellular matrix turnover
The reported increase in fibrous cap thickness and collagen content in plaques from CD40-deficient mice has been proposed to result from the accompanying decreased plaque expression and activity of matrix metalloproteinase (MMP)-2 and -9 together with increased tissue inhibitor of metalloproteinase (TIMP)-1 expression, and has thus prompted the suggestion that CD40 signalling destabilizes plaques by promoting extracellular matrix (ECM) degradation and, as a result, fibrous cap thinning [6]. Therefore, in this study we searched for similar associations in human samples. Plaque levels of CD40 correlated with plaque content of MMP-1, -2, -9, and -10, while sCD40L levels correlated with only MMP-10 (Supplementary Table 6) Furthermore, both sCD40 and sCD40L levels correlated with TIMP-1 and TIMP-2. Thus, sCD40 (and CD40L) were indeed associated with plaque ECM turnover. Of note, correlations were also found between both sCD40 and sCD40L levels and content of intact elastin and collagen (type I–V) measured in plaque homogenate.
Plaque sCD40 and sCD40L levels are not associated with pre-operative cerebrovascular symptoms
As sCD40 and sCD40L plasma levels were higher in subjects who had suffered a stroke in the SUMMIT cohort, we next analysed whether sCD40 and/or sCD40L levels are similarly elevated in the plaques from individuals after a cerebrovascular event (median time between symptom and surgery was 14 days [IQR, 7 to 22]; the CPIP cohort). Levels of CD40 and CD40L measured in plaque strongly correlate with each other (r=0.613, P<0.0001) but neither sCD40, nor sCD40L levels correlated with the time between symptoms exhibited by the patients and the time of endarterectomy (r=0.04, P=0.688 and r=0.063, P=0.523, respectively). Also, plaque levels of sCD40 and sCD40L were similar in plaques from asymptomatic and symptomatic patients (n=102 and n=96, respectively).
Discussion
With this study, we set out to assess the value of sCD40 as a possible marker of CVD and investigate the biological associations that support a role for CD40 in atherogenesis. Our findings that sCD40 levels in plasma are associated with carotid plaque burden, while levels in carotid plaques are associated with a vulnerable plaque phenotype, strengthens the potential for sCD40 as marker for and mediator of CVD.
In contrast to the paucity of data in the literature for sCD40 as a marker for CVD, sCD40L has been previously widely linked to CVD via increased levels in patients with hypercholesterolemia [10] and acute coronary syndromes [11,24]. Upregulated platelet surface expression of CD40L was also reported to correlate with worse clinical outcome after stroke [25] and plasma sCD40L levels were higher in patients with carotid artery lesions that have intra-plaque lipid as detected by high-resolution magnetic resonance imaging [26]. Furthermore, Balla et al. [27] identified serum sCD40L levels as a risk factor for increased IMT in subjects with white-coat hypertension. Our current results are in line with these studies.
Much less is known about the possible role of sCD40 as a marker for CVD. Though sCD40 appears to be actively produced under physiological conditions (as suggested by comparatively large amounts of sCD40 found in urine of healthy individuals) [28], elevated plasma levels of sCD40 has nonetheless been described in pathologies such as systemic lupus erythematosus [29], slow coronary flow phenomenon [30] and early Alzheimer’s disease [31]. In the SUMMIT cohort, we found higher plasma sCD40 levels in individuals that had suffered a prior ischemic stroke or MI or were hypertensive. Plasma sCD40 levels also correlated with carotid plaque burden and were associated with future CV events over a follow-up period of 3 years. We therefore propose sCD40 as a potential marker for the presence and severity of CVD.
Efficient targeting of inflammatory processes can successfully stabilize vulnerable plaques in mice [8,9] but this approach is not yet utilized in human disease beyond clinical trials [32] due to detrimental side effects. In atherosclerotic mouse models disrupted CD40 signalling—after blockade with antagonistic antibodies, small molecule inhibitors and in CD40- and CD40L-deficient mice—consistently results in plaques of reduced size and, importantly, a stabilized phenotype characterized by reduced lipid core content, macrophage and T-lymphocyte content accompanied by increased fibrous cap thickness, and relative collagen and SMC cell content [6-9]. Previous studies exploring CD40 signalling in relation to severity of inflammatory diseases in humans have mainly focused on systemic levels of the signalling dyad, in particular levels of sCD40L and CD40L. This is the first study to also investigate associations between carotid intra-plaque CD40/CD40L content and plaque phenotype and disease progression. We found intra-plaque sCD40 (and to a certain extent also sCD40L) levels to be associated with a plaque profile rich in lipids, MMPs and pro-inflammatory cytokines and chemokines: destabilising elements corresponding to those affected by blocking of the CD40/CD40L axis in mice [6-9]. This link between human disease and mouse model strengthens the merit of translating therapeutic targeting of CD40 from the academic setting one step closer to the clinical application.
Neither CD40 nor CD40L was associated with time between cerebrovascular symptoms and endarterectomy, suggesting they are not merely upregulated as part of the general healing response that occurs after plaque rupture. However, though sCD40 levels were associated with a less stable carotid plaque phenotype, plaque levels were nonetheless similar in plaques from symptomatic and asymptomatic patients. This inconsistency may, at least in part, be explained by the accompanying association with components of the ECM turnover machinery, leading to a net effect in CD40-rich plaques, where enough ECM synthesis of stable plaque elements balances out the more vulnerable elements, resulting in a plaque phenotype still stable enough not to rupture/erode and cause cerebrovascular symptoms. This in turn may indicate a local protective element in CD40-CD40L signalling, in addition to its well-described actions promoting the inflammatory process. We hypothesize that this may demonstrate the contrasting results of CD40 and CD40L interactions with (and in) different cell types of varying functions, such as macrophages, T cells, and SMCs in a model where CD40-signalling simultaneously for example promotes inflammation via macrophages and ECM synthesis via SMCs, even if the phenotypes of these cells have also been widely discussed [33]. The therapeutic angle described by Seijkens et al. [9] of targeting CD40 signalling blockade specifically to macrophages (via incorporation into reconstituted high-density lipoproteins nanoparticles) therefore also emerges as an especially relevant approach to achieve plaque stabilisation.
Also interesting from a therapeutic standpoint is the study by Fernandez et al. [34], in which single cell RNA sequencing was compared in carotid plaques from patients who had or had not experienced cerebrovascular symptoms. Unexpectedly, the most upregulated pathways in T cells and macrophages from plaques from symptomatic patients were those of repair, while pro-inflammatory pathways were elevated in plaques from asymptomatic patients. Repair pathways are thus deduced to be induced by plaque rupture, and therefore represent a less striking presence in plaques whose integrity is more intact. Consequently, inflammatory activity weighs heavier in plaques from asymptomatic than from symptomatic individuals. This notion confirms the significance of our report that sCD40 is associated with vulnerable plaque components such as lipid content and pro-inflammatory cytokines, as well as with overall plaque burden, regardless of whether symptoms have manifested or not. Furthermore, the great success in ameliorating size and vulnerability of murine lesions (that are, per definition, pre-rupture) by blocking CD40 signalling affirms the value of targeting inflammatory responses in non-ruptured lesions.
As limitations, it must be noted that correlations do not indicate causality, and we cannot rule out that the reported associations may also result from an upregulation of sCD40 and sCD40L stimulated by an upstream signalling cascade or events. It is also important to bear in mind that in the plaque homogenates derived from the CPIP cohort it is not possible to distinguish between soluble and cell-bound CD40 and CD40L. The analysis thus shows the net effect of both isoforms, even if the effects of the individual isoforms may differ. Moreover, though it is impossible to be certain of the exact type of ischemic stroke, as subjects with atrial fibrillation were excluded in both cohorts, and—in the CPIP cohort—no signs of lacunar infarctions were found (assessed by brain computed tomography), the probability of the underlying mechanisms to be atrial fibrillation and small vessel disease rather than atherosclerosis can be considered low. Unfortunately, the presence of cardiac valvular disease is impossible to exclude as not all patients underwent echocardiography. Finally, as we in both cohorts measured sCD40L in plasma to evaluate its potential as a non-cell associated marker, platelet expression of CD40L—previously reported e.g., by Cha et al. [35] to be higher in subjects with atherosclerotic ischemic stroke than with asymptomatic carotid stenosis—has not been taken into account.
Conclusions
In the present study we report for the first time elevated plasma sCD40 levels in individuals with a prior ischemic stroke or MI, as well as an association with arterial stiffness, carotid plaque burden, and IMT, and, finally, an association with increased risk of future CV events (Figure 3). Plasma levels of sCD40L are confirmed to not only be elevated in subjects after stroke, but also to correlate with future plaque progression in the form of IMT increase. We also present a novel association between intra-plaque levels of CD40 and CD40L and a vulnerable plaque phenotype.
Figure 3.
Graphic representation of plasma soluble CD40 (sCD40) in cardiovascular disease. Higher plasma sCD40 levels are associated with prevalent cardiovascular disease, severity of carotid atherosclerosis and predict future cardiovascular events (CV events). MI, myocardial infarction.
Acknowledgments
This work was supported by funding from the Innovative Medicines Initiative (the SUMMIT consortium), IMI-2008/115006, Swedish Research Council, Swedish Heart and Lung Foundation, Swedish Society for Medical Research, Swedish Society of Medicine, Emil and Wera Cornell foundation, Hjelt foundation, ALF Grants Region Skane, Bundy Academy, Diabetes foundation, Diabetes Research & Wellness Foundation, SUS foundations and funds, Stroke foundation, the Royal Physiographic Society in Lund, The Swedish Heart and Lung Association, The Swedish Stroke Association, Albert Påhlsson’s foundation and Swedish Foundation for Strategic Research (Dnr IRC15-0067). Angela C. Shore is supported by the UK National Institute of Health Research (NIHR) Exeter Clinical Research Facility. The views in this manuscript are those of the authors and not necessarily those of the NIHR or the UK Department of Health.
The authors wish to thank Mihaela Nitulescu, Ana Persson, Lena Sundius, and Fong To for expert technical assistance.
Footnotes
Disclosure
The authors have no financial conflicts of interest.
Supplementary materials
Supplementary materials related to this article can be found online at https://doi.org/10.5853/jos.2021.00178.
Patient demographics in the CPIP and SUMMIT study cohorts
Spearman correlations between sCD40 and sCD40L in plasma (arbitrary unit) and the subjects’ general clinical characteristics (SUMMIT cohort)
Median with IQR values of sCD40 and sCD40L in plasma (A.U., SUMMIT cohort) when comparing between two groups (MannWhitney U test)
Spearman correlations between the circulating levels of sCD40 and sCD40L (A.U., SUMMIT cohort) and change in IMT in the CCA and the carotid bulb region assessed by vascular ultrasound (at the 3-year follow-up point compared to study inclusion)
Sensitivities, specificities and their 95% CI at various cut-offs of predicted values from LR in predicting atherosclerotic vascular events
Spearman correlations of CD40 and CD40L (A.U., CPIP cohort) measured in plaque homogenate versus plaque elements
References
- 1.World Health Organization . Cardiovascular Disease Fact Sheet. Geneva, CH: World Health Organization; 2017. [Google Scholar]
- 2.Sakakura K, Nakano M, Otsuka F, Ladich E, Kolodgie FD, Virmani R. Pathophysiology of atherosclerosis plaque progression. Heart Lung Circ. 2013;22:399–411. doi: 10.1016/j.hlc.2013.03.001. [DOI] [PubMed] [Google Scholar]
- 3.Hansson GK, Libby P, Tabas I. Inflammation and plaque vulnerability. J Intern Med. 2015;278:483–493. doi: 10.1111/joim.12406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Noelle RJ, Ledbetter JA, Aruffo A. CD40 and its ligand, an essential ligand-receptor pair for thymus-dependent B-cell activation. Immunol Today. 1992;13:431–433. doi: 10.1016/0167-5699(92)90068-I. [DOI] [PubMed] [Google Scholar]
- 5.Jobling K, Ng WF. CD40 as a therapeutic target in Sjögren’s syndrome. Expert Rev Clin Immunol. 2018;14:535–537. doi: 10.1080/1744666X.2018.1485492. [DOI] [PubMed] [Google Scholar]
- 6.Lutgens E, Lievens D, Beckers L, Wijnands E, Soehnlein O, Zernecke A, et al. Deficient CD40-TRAF6 signaling in leukocytes prevents atherosclerosis by skewing the immune response toward an antiinflammatory profile. J Exp Med. 2010;207:391–404. doi: 10.1084/jem.20091293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lutgens E, Gorelik L, Daemen MJ, de Muinck ED, Grewal IS, Koteliansky VE, et al. Requirement for CD154 in the progression of atherosclerosis. Nat Med. 1999;5:1313–1316. doi: 10.1038/15271. [DOI] [PubMed] [Google Scholar]
- 8.Lutgens E, Cleutjens KB, Heeneman S, Koteliansky VE, Burkly LC, Daemen MJ. Both early and delayed anti-CD40L antibody treatment induces a stable plaque phenotype. Proc Natl Acad Sci U S A. 2000;97:7464–7469. doi: 10.1073/pnas.97.13.7464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Seijkens TTP, van Tiel CM, Kusters PJH, Atzler D, Soehnlein O, Zarzycka B, et al. Targeting CD40-induced TRAF6 signaling in macrophages reduces atherosclerosis. J Am Coll Cardiol. 2018;71:527–542. doi: 10.1016/j.jacc.2017.11.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Garlichs CD, John S, Schmeisser A, Eskafi S, Stumpf C, Karl M, et al. Upregulation of CD40 and CD40 ligand (CD154) in patients with moderate hypercholesterolemia. Circulation. 2001;104:2395–2400. doi: 10.1161/hc4501.099312. [DOI] [PubMed] [Google Scholar]
- 11.Heeschen C, Dimmeler S, Hamm CW, van den Brand MJ, Boersma E, Zeiher AM, et al. Soluble CD40 ligand in acute coronary syndromes. N Engl J Med. 2003;348:1104–1111. doi: 10.1056/NEJMoa022600. [DOI] [PubMed] [Google Scholar]
- 12.Schönbeck U, Varo N, Libby P, Buring J, Ridker PM. Soluble CD40L and cardiovascular risk in women. Circulation. 2001;104:2266–2268. doi: 10.1161/hc4401.099447. [DOI] [PubMed] [Google Scholar]
- 13.Huang YQ, Jie LI, Chen JY, Tang ST, Huang C, Feng YQ. The relationship between soluble CD40 ligand level and atherosclerosis in white-coat hypertension. J Hum Hypertens. 2017;32:40–45. doi: 10.1038/s41371-017-0016-z. [DOI] [PubMed] [Google Scholar]
- 14.Lobbes MB, Lutgens E, Heeneman S, Cleutjens KB, Kooi ME, van Engelshoven JM, et al. Is there more than C-reactive protein and fibrinogen? The prognostic value of soluble CD40 ligand, interleukin-6 and oxidized low-density lipoprotein with respect to coronary and cerebral vascular disease. Atherosclerosis. 2006;187:18–25. doi: 10.1016/j.atherosclerosis.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 15.de Lemos JA, Zirlik A, Schönbeck U, Varo N, Murphy SA, Khera A, et al. Associations between soluble CD40 ligand, atherosclerosis risk factors, and subclinical atherosclerosis: results from the Dallas Heart Study. Arterioscler Thromb Vasc Biol. 2005;25:2192–2196. doi: 10.1161/01.ATV.0000182904.08513.60. [DOI] [PubMed] [Google Scholar]
- 16.Verma S, Wang CH, Li SH, Lonn E, Charbonneau F, Title LM, et al. The relationship between soluble CD40 ligand levels and Framingham coronary heart disease risk score in healthy volunteers. Atherosclerosis. 2005;182:361–365. doi: 10.1016/j.atherosclerosis.2005.02.019. [DOI] [PubMed] [Google Scholar]
- 17.Gergei I, Kälsch T, Scharnagl H, Kleber ME, Zirlik A, März W, et al. Association of soluble CD40L with short-term and long-term cardiovascular and all-cause mortality. The Ludwigshafen Risk and Cardiovascular Health (LURIC) study. Atherosclerosis. 2019;291:127–131. doi: 10.1016/j.atherosclerosis.2019.09.004. [DOI] [PubMed] [Google Scholar]
- 18.Shore AC, Colhoun HM, Natali A, Palombo C, Östling G, Aizawa K, et al. Measures of atherosclerotic burden are associated with clinically manifest cardiovascular disease in type 2 diabetes: a European cross-sectional study. J Intern Med. 2015;278:291–302. doi: 10.1111/joim.12359. [DOI] [PubMed] [Google Scholar]
- 19.Asciutto G, Edsfeldt A, Dias NV, Nilsson J, Prehn C, Adamski J, et al. Treatment with beta-blockers is associated with lower levels of Lp-PLA2 and suPAR in carotid plaques. Cardiovasc Pathol. 2013;22:438–443. doi: 10.1016/j.carpath.2013.04.005. [DOI] [PubMed] [Google Scholar]
- 20.Kim HL, Kim SH. Pulse wave velocity in atherosclerosis. Front Cardiovasc Med. 2019;6:41. doi: 10.3389/fcvm.2019.00041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Companjen AR, van der Wel LI, Boon L, Prens EP, Laman JD. CD40 ligation-induced cytokine production in human skin explants is partly mediated via IL-1. Int Immunol. 2002;14:669–676. doi: 10.1093/intimm/dxf033. [DOI] [PubMed] [Google Scholar]
- 22.Basok A, Shnaider A, Man L, Chaimovitz C, Douvdevani A. CD40 is expressed on human peritoneal mesothelial cells and upregulates the production of interleukin-15 and RANTES. J Am Soc Nephrol. 2001;12:695–702. doi: 10.1681/ASN.V124695. [DOI] [PubMed] [Google Scholar]
- 23.Buhtoiarov IN, Lum H, Berke G, Paulnock DM, Sondel PM, Rakhmilevich AL. CD40 ligation activates murine macrophages via an IFN-gamma-dependent mechanism resulting in tumor cell destruction in vitro. J Immunol. 2005;174:6013–6022. doi: 10.4049/jimmunol.174.10.6013. [DOI] [PubMed] [Google Scholar]
- 24.Fouad HH, Al-Dera H, Bakhoum SW, Rashed LA, Sayed RH, Rateb MA, et al. Levels of sCD40 ligand in chronic and acute coronary syndromes and its relation to angiographic extent of coronary arterial narrowing. Angiology. 2010;61:567–573. doi: 10.1177/0003319709356785. [DOI] [PubMed] [Google Scholar]
- 25.Lukasik M, Dworacki G, Kufel-Grabowska J, Watala C, Kozubski W. Upregulation of CD40 ligand and enhanced monocyte-platelet aggregate formation are associated with worse clinical outcome after ischaemic stroke. Thromb Haemost. 2012;107:346–355. doi: 10.1160/TH11-05-0345. [DOI] [PubMed] [Google Scholar]
- 26.Blake GJ, Ostfeld RJ, Yucel EK, Varo N, Schönbeck U, Blake MA, et al. Soluble CD40 ligand levels indicate lipid accumulation in carotid atheroma: an in vivo study with high-resolution MRI. Arterioscler Thromb Vasc Biol. 2003;23:e11–e14. doi: 10.1161/01.atv.0000050143.22910.62. [DOI] [PubMed] [Google Scholar]
- 27.Balla J, Magyar MT, Bereczki D, Valikovics A, Nagy E, Barna E, et al. Serum levels of platelet released CD40 ligand are increased in early onset occlusive carotid artery disease. Dis Markers. 2006;22:133–140. doi: 10.1155/2006/427174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Contin C, Pitard V, Delmas Y, Pelletier N, Defrance T, Moreau JF, et al. Potential role of soluble CD40 in the humoral immune response impairment of uraemic patients. Immunology. 2003;110:131–140. doi: 10.1046/j.1365-2567.2003.01716.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bae SC, Lee YH. Association between CD40 polymorphisms and systemic lupus erythematosus and correlation between soluble CD40 and CD40 ligand levels in the disease: a meta-analysis. Lupus. 2019;28:1452–1459. doi: 10.1177/0961203319878822. [DOI] [PubMed] [Google Scholar]
- 30.Durakoğlugil ME, Kocaman SA, Çetin M, Kirbas A, Canga A, Erdoğan T, et al. Increased circulating soluble CD40 levels in patients with slow coronary flow phenomenon: an observational study. Anadolu Kardiyol Derg. 2013;13:39–44. doi: 10.5152/akd.2013.005. [DOI] [PubMed] [Google Scholar]
- 31.Buchhave P, Janciauskiene S, Zetterberg H, Blennow K, Minthon L, Hansson O. Elevated plasma levels of soluble CD40 in incipient Alzheimer’s disease. Neurosci Lett. 2009;450:56–59. doi: 10.1016/j.neulet.2008.10.091. [DOI] [PubMed] [Google Scholar]
- 32.Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377:1119–1131. doi: 10.1056/NEJMoa1707914. [DOI] [PubMed] [Google Scholar]
- 33.Wirka RC, Wagh D, Paik DT, Pjanic M, Nguyen T, Miller CL, et al. Atheroprotective roles of smooth muscle cell phenotypic modulation and the TCF21 disease gene as revealed by single-cell analysis. Nat Med. 2019;25:1280–1289. doi: 10.1038/s41591-019-0512-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fernandez DM, Rahman AH, Fernandez NF, Chudnovskiy A, Amir ED, Amadori L, et al. Single-cell immune landscape of human atherosclerotic plaques. Nat Med. 2019;25:1576–1588. doi: 10.1038/s41591-019-0590-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cha JK, Jeong MH, Jang JY, Bae HR, Lim YJ, Kim JS, et al. Serial measurement of surface expressions of CD63, P-selectin and CD40 ligand on platelets in atherosclerotic ischemic stroke. A possible role of CD40 ligand on platelets in atherosclerotic ischemic stroke. Cerebrovasc Dis. 2003;16:376–382. doi: 10.1159/000072560. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Patient demographics in the CPIP and SUMMIT study cohorts
Spearman correlations between sCD40 and sCD40L in plasma (arbitrary unit) and the subjects’ general clinical characteristics (SUMMIT cohort)
Median with IQR values of sCD40 and sCD40L in plasma (A.U., SUMMIT cohort) when comparing between two groups (MannWhitney U test)
Spearman correlations between the circulating levels of sCD40 and sCD40L (A.U., SUMMIT cohort) and change in IMT in the CCA and the carotid bulb region assessed by vascular ultrasound (at the 3-year follow-up point compared to study inclusion)
Sensitivities, specificities and their 95% CI at various cut-offs of predicted values from LR in predicting atherosclerotic vascular events
Spearman correlations of CD40 and CD40L (A.U., CPIP cohort) measured in plaque homogenate versus plaque elements


