Abstract
The present study aimed to review major depression, including its types, epidemiology, association with different diseases status and treatments, as well as its correlation with the current COVID-19 pandemic. Mental depression is a common disorder that affects most individuals at one time or another. During depression, there are changes in mood and behavior, accompanied by feelings of defeat, hopelessness, or even suicidal thoughts. Depression has a direct or indirect relation with a number of other diseases including Alzheimer's disease, stroke, epilepsy, diabetes, cardiovascular disease and cancer. In addition, antidepressant drugs have several side effects including sedation, increased weight, indigestion, sexual dysfunction, or a decrease in blood pressure. Stopping medication may cause a relapse of the symptoms of depression and pose a risk of attempted suicide. The pandemic of COVID-19 has affected the mental health of individuals, including patients, individuals contacting patients and medical staff with a number of mental disorders that may adversely affect the immune ability of their bodies. Some of the drugs currently included in the protocols for treating COVID-19 may negatively affect the mental health of patients. Evidence accumulated over the years indicates that serotonin (5HT) deficiencies and norepinephrine (NE) in the brain can lead to mental depression. Drugs that increase levels of NE and 5HT are commonly used in the treatment of depression. The common reason for mood disorders, including mania and bipolar disease are not clearly understood. It is assumed that hyperactivity in specific parts of the brain and excessive activity of neurotransmitters may be involved. Early diagnosis and developing new treatment strategies are essential for the prevention of the severe consequences of depression. In addition, extensive research should be directed towards the investigation of the mental health disturbances occurring during and/or after COVID-19 infection. This may lead to the incorporation of a suitable antidepressant into the current treatment protocols.
Keywords: major depression, neurotransmitters, molecular mechanism, COVID-19, latest treatments, mental health
1. Introduction
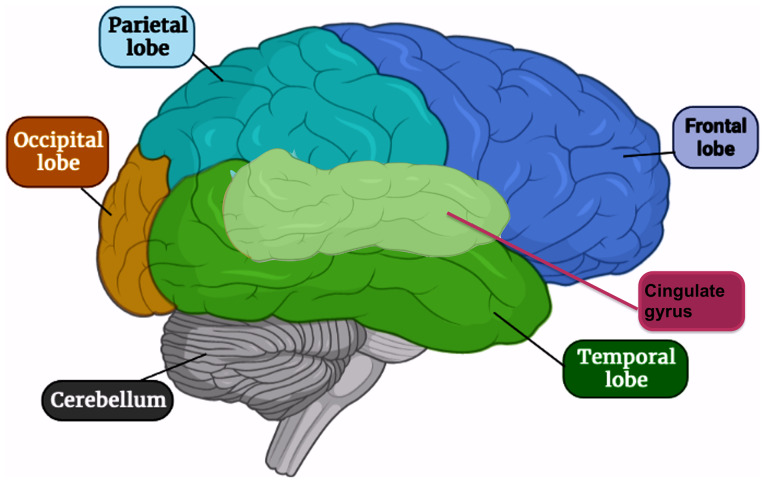
Mental depression is a common psychiatric disorder, where ~264 million individuals of different ages suffered from depressive behavior at January 2020 with ~800,000 annual suicides (World Health Organization, WHO) (1). Although depression has been extensively studied, its mechanisms remain to be elucidated. Norepinephrine and serotonin are two neurotransmitters considered to be the most efficient in mental depression (2). The areas in the brain supposed to be affected include the temporal and frontal lobes and the cingulate gyrus of the limbic system (Fig. 1) (3). The symptoms of depression are mostly mild and unpredicted either by patients or even physicians. The typical types of depression, whether major or minor, have only symptoms of depression (4). However, bipolar and cyclothymic types are accompanied by manic symptoms (5). According to the American Psychiatric Associations of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5), depressive disorders are classified into three common types based on a theoretical basis; the first type usually occurs due to stressful stimuli. The second type is genetically based and termed endogenous depression; it is demonstrated through patient difficulties in dealing with normal life (6). The third type is bipolar depression (or manic-depressive disorder).
Figure 1.
Areas affected in the brain during depression. The areas affected in the brain include the temporal and frontal lobes and the cingulate gyrus of the limbic system.
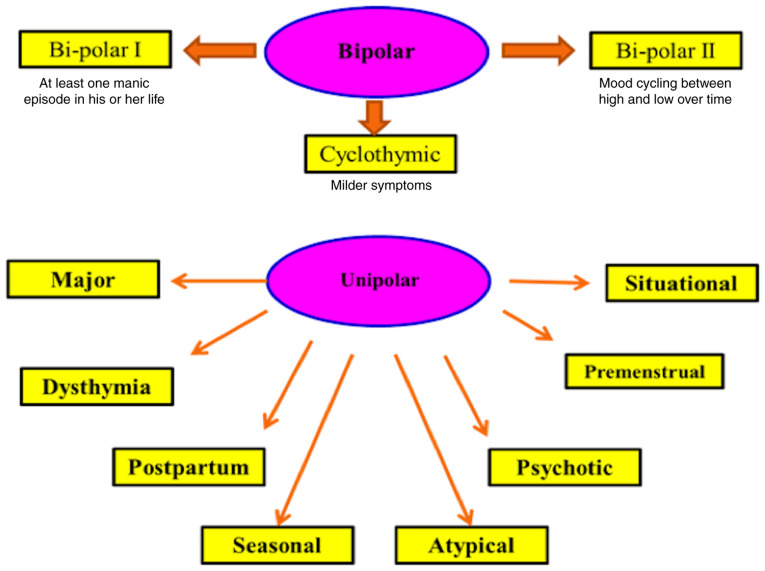
By contrast, the International Alliance on Mental Illness classifies depression into nine common types; Major Depression: patients with this type feel excessive sadness, lack of energy, irritability, hopelessness, physical pain, lower concentration, alterations in eating or sleep habits, feelings of guilt and attempted suicide. Only two weeks of suffering these symptoms are needed to officially diagnose major depression (7). Dysthymia is a type of depression that causes persistent low mood with changes in sleep habits, sadness, trouble in concentration, fatigue and appetite loss. Talk therapy, not medication, is usually suitable for this type of depression (8). Postpartum depression (PPD) usually happens after birth where the patient typically suffers from loneliness, hopelessness, deep sadness, fatigue, fear about baby disconnection and fears about harming her newborn (9). Seasonal Affective Disorder generally occurs in winter, probably due to a shortage of sunlight. Symptoms are generally mild to severe. This type usually lasts in spring and natural or artificial light is usually the suitable treatment (10).
Atypical Depression is commonly associated with physical symptoms, including a sense of heaviness in the legs or arms together with being overweight and problems with social communication (11). Psychotic Depression is characterized by psychological symptoms including delusions, and auditory and visual hallucinations (12). In Bipolar Disorder, patients have a cycle of depression and mania symptoms. It is classified into bipolar I (where patients have at least one manic episode) (13); bipolar II (where patients have hypomania as well as depression) (14) and cyclothymia, a cycle of depression followed by a cycle of mania (15). Premenstrual Dysphoric Disorder occurs in females during the last 2 weeks of the menstrual cycle (16). Situational Depression is related to certain stressful situations and the symptoms are unhappiness, anxiety and perhaps major depression (17) (Fig. 2).
Figure 2.
Common types of depression. Types of depression include unipolar (unipolar I, II and cyclothymia) and bipolar (major, dysthymia, postpartum, seasonal, atypical and psychotic).
Depression can be treated by non-drug or drug-containing therapy. None-drug treatment includes encouraging creativity, physical or alternative therapy including exposure to sunlight, St. John's Wort and acupuncture (18). On the other hand, well-known antidepressant drugs including selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants (TCAs), norepinephrine reuptake inhibitors (NRIs), serotonin antagonist and reuptake inhibitors (SARIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) are used (19). However, efforts have been made to find new antidepressants with high efficacy and faster response, fewer side effects, able the preserve sexual functions in patients, improved efficacy, rapid onset and improved safety profile. Mental health problems resulting from the COVID-19 pandemic comprise insomnia, anxiety, stress, fear and depressive behavior (20). So far, there have been no data about the changes in cognitive functioning or emotions from the direct effects of the virus on the brain. A proportion of the world's population are in health quarantine and isolated at home and have lost their jobs, family members, relatives, colleagues, or friends. Consequently, depression associated with COVID-19 can be included under a situational or emotional type of depression.
2. Epidemiology
According to the World Health Organization (WHO), 264 million individuals worldwide suffer from major depression. Almost 800,000 suicides are counted each year and it is the second leading cause of death among young individuals (21). Adolescents aged between 12 and 17 years exhibit the highest rate of major depressive episodes (11.3%) followed by young adults between 12 and 20 years old at 9.6% (22). It has been observed that adults >50 years of age are less likely to have major depressive episodes (4.5%) (23). The rate of moderate to severe depression increased from 23.2 to 41.1% from 2007 to 2018 (24). It is noted that the prevalence of depression among women is almost twice as high as that of men (25). Despite the availability of effective antidepressants with multiple mechanisms of action, it has been observed that between 76 and 85% of individuals who belong to developing countries do not receive any treatment for depression (25,26).
3. Biological markers and clinical presentation of depression
There are no specific biological markers for depression. Various medical and psychiatric disorders, as well as certain drugs, have been noted in patients with depressive illness (Table I) (27).
Table I.
Common medical disorders and drug therapies associated with depression (27).
| Type of disorder/therapy | Name of disorder/therapy |
|---|---|
| Medical disorders | |
| Deficiency situations | Wernicke's encephalopathy, severe anemia, pernicious anemia |
| Infections | Mononucleosis, AIDS, influenza, sexually transmitted diseases, encephalitis, tuberculosis |
| Collagen disorder | Systematic lupus, erythematosus |
| Endocrine diseases | Hypothyroidism, Addison's disease, Cushing's disease |
| Neurologic disorders | Epilepsy, Parkinson's disease, Poststroke, Huntingtons disease, Multiple sclerosis, Alzheimer's disease, pain |
| Metabolic disorders | Electrolyte imbalance, Hypokalemia, Hyponatremia, Hepatic encephalopathy |
| Cardiovascular disease | Coronary artery disease, congestive heart failure, myocardial infarction |
| Malignant disease | Breast cancer, colon cancer, lung cancer |
| Psychiatric disorders | Eating disorders, anxiety disorders, alcoholism, scizophrenia |
| Hormonal therapy | Steroids/adrenocoticotropic, hormone, oral contraceptives |
| Drug therapy | Methyldopa, clonidine, propranolol, antihypertensives, diuretics, guanethidine sulfate, hydralazine hydrochloride, reserpine |
| Acne therapy | Isotretinoin |
| Other | Interferon-beta-1a |
In all, 25% of patients with a chronic medical history, including cancer, diabetes, or cardiovascular diseases, develop depressive disorder without a clear and accurate diagnosis for depression (28). According to the DSM-5, the symptoms of major depression are sadness, anxiety or irritability, feelings of guilt and suicidal thoughts (29). The clinical presentation could be classified into emotional and physical symptoms. Emotional symptoms include stress, sadness, anxiety, or feeling guilty, whereas physical symptoms may include sleep disturbance, appetite changes, headache, pain, or fatigue (Table II).
Table II.
Physical and emotional symptoms in depression (163).
| Emotional symptoms | Physical symptoms |
|---|---|
| Stress | Lack of energy |
| Sadness and tearfulness | Tiredness/fatigue |
| Loss of interest | Body aches and pains |
| Anxiety/irritability | Sleep disturbances |
| Hopelessness | Headaches |
| Concentration difficulties | Psychomotor activity changes |
| Feelings of guilt | GI disturbances |
| Suicidal thoughts | Changes in appetite |
4. Depression and common medical diseases
Depression can cause several medical diseases, including Alzheimer's (by 2.0-fold) (30), cardiovascular diseases (1.5–2.0-fold) (31), stroke (1.8-fold) (31), epilepsy (4.0–6.0-fold) (32), diabetes (1.6-fold) (33) and cancer (1.3–1.8-fold) (34) (Fig. 3). Drugs used in controlling depressive illness have multiple unwanted effects, including increased weight, sedation, sexual dysfunction, indigestion, or decreased blood pressure. Incompliance or stopping medication may cause a relapse of the symptoms of depression and pose a risk of attempted suicide (35).
Figure 3.
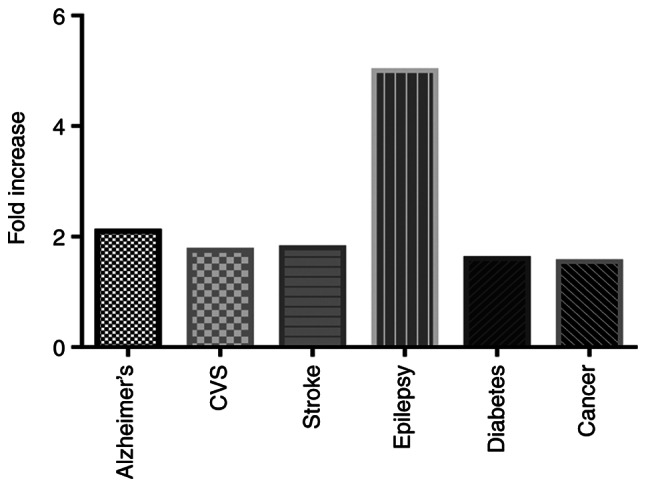
Fold increase in the risk of diseases resulting from depression. Depression can cause several medical diseases, including Alzheimer's (by 2.0-fold), CVS (1.5–2.0-fold), stroke (1.8-fold), epilepsy (4.0–6.0-fold), diabetes (1.6-fold) and cancer (1.3–1.8-fold) compared with control non-depressed individuals. CVS, cardiovascular disease.
5. Depression and the metabolic diseases
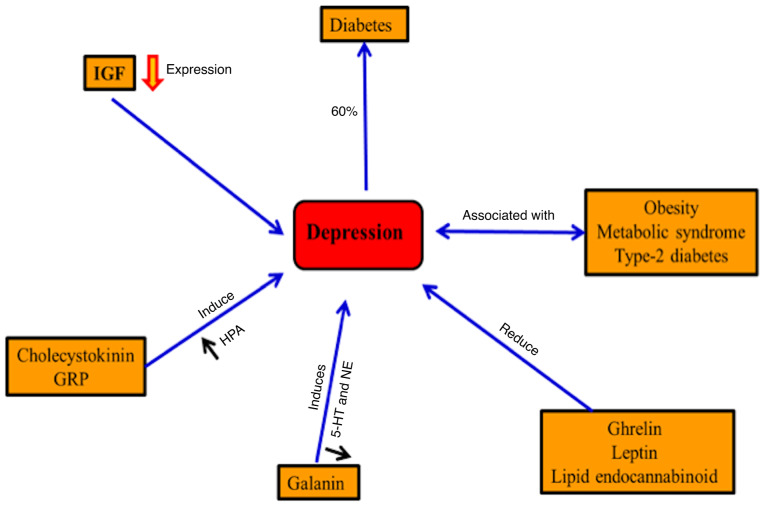
Depression is usually accompanied by metabolic syndrome, obesity and type-2 diabetes (36,37). In diabetes, ~20.9% of males and 27% of females exhibit major depression in western Kenya (38). By contrast, depression may increase the risk for diabetes (33). Gastrointestinal hormones, including ghrelin, leptin and the lipid endocannabinoid, may regulate mood in the case of depression (39). Improving the depression symptoms has been accompanied by inhibition of cholecystokinin receptors in mice and also inhibits the hyperactivity of the hypothalamic-pituitary-adrenal axis (40). Neuroendocrine, noradrenergic and serotonergic are controlled by the galanin neuropeptide systems in depression and anxiety-related behaviors (41). Furthermore, hippocampal neurogenesis is induced by an insulin-like growth factor (IGF) and the disturbance of neuronal IGF results in depression (42). Regulation of synaptic plasticity and improving depressive behavior results from releasing gastrin-releasing peptide in the hippocampus (43) (Fig. 4).
Figure 4.
Depression and metabolic disorders. Depression is accompanied by metabolic syndrome, obesity, T2DM and T1DM (30,31,38, respectively). By contrast, depression may increase the risk of diabetes. Gastrointestinal hormones, such as ghrelin, leptin and the lipid endocannabinoid, may regulate mood in the case of depression. Improving depression symptoms has been accompanied by inhibition of cholecystokinin receptors and the hyperactivity of the HPA axis. Hippocampal neurogenesis is induced by IGF and the disturbance of neuronal IGF results in depression. Regulation of synaptic plasticity and improving depressive behavior results from releasing gastrin-releasing peptide in the hippocampus. HPA, hypothalamic-pituitary-adrenal; IGF, insulin-like growth factor; GRP, gastrin-releasing peptide; 5-HT, 5-hydroxytryptamine; NE, norepinephrine.
6. Depression and endocrine disorders
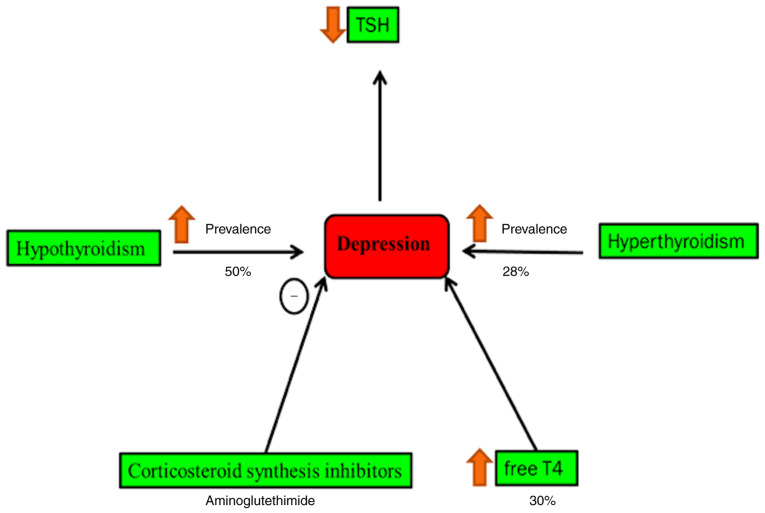
Cushing's syndrome and hypothyroidism are accompanied by a high rate of psychiatric morbidity (44). Secondary hypothyroidism is normally characterized by decreased blood level thyroid-stimulating hormone, which is concomitant with an increased risk of depressive episodes (45). It is found that 40% of patients with hypothyroidism suffer from clinical depression (46,47). Hyperthyroidism patients have a high risk of depression by 28% compared with normal individuals, whereas it occurs in 50% of all cases of patients with hypothyroidism (46,48). Blood and cerebrospinal levels of T4 are relatively increased during the depression (46) (Fig. 5). The raised concentration of corticotropin-releasing factor in cerebrospinal fluid in patients with depression is noted in a previous study (49).
Figure 5.
Depression and endocrine disorders. Cushing's syndrome and hypothyroidism are accompanied by a high rate of psychiatric morbidity. The raised concentration of thyrotrophin releasing hormone in cerebrospinal fluid in patients with depression is noted in a previous study. Secondary hypothyroidism is characterized by decreased blood level thyroid-stimulating hormone, which is concomitant with an increased risk of depressive episodes. It is found that 40% of patients with hypothyroidism suffer from clinical depression. Hyperthyroidism patients have a high risk of depression by 28% compared with normal individuals, whereas it occurs in 50% of all cases of patients with hypothyroidism. Blood and cerebrospinal levels of T4 are relatively increased during the depression. T4, thyroxin; TSH, thyroid stimulating hormone.
7. Depression and cardiovascular diseases
Decreased plasma volume and elevation in blood viscosity lead to a higher risk of depression (50,51). Patients with depression have a high risk for hypertension, congestive heart failure and myocardial infarction (52). Hypercoagulability, platelet activation and D-dimer proteins can be detected in patients with depression (50,53,54). In clinical trials, the risk of depression is significantly reduced in cardiovascular disease patients treated with statins (50,55). It has been demonstrated that vascular endothelial growth factor has neurogenic/neuroprotective effects (50,56,57). Furthermore, its level is notably increased in patients diagnosed with depression (58,59).
8. Depression and inflammatory status
Depression increases cytokines, IL-6, TNF-α and IL-1 (60). Furthermore, stress regulates prostaglandin-E2, lipid peroxidation IL-1β, cyclooxygenase-2 and Toll-like receptor-4 (Fig. 6). A previous study demonstrates that celecoxib can be used as a new strategy for major depression, especially bipolar disorder (61,62). In addition, Nery et al (63) describe the antidepressant action of celecoxib, which is characterized by its rapid-onset antidepressant activities. Sesamol, a potent antioxidant and cytokine inhibitor, has been shown to improve chronic stress-induced depression in an experimental mouse model (64).
Figure 6.
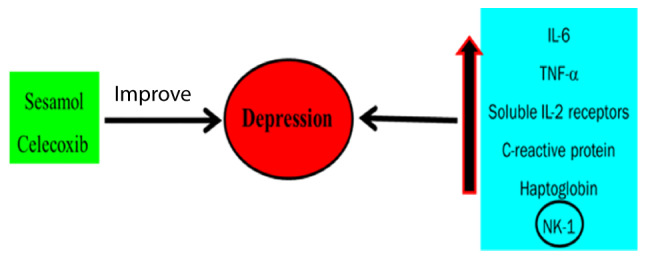
Depression and inflammatory disorders. Depression increases cytokines, including IL-6, TNF-α and IL-1. Furthermore, stress regulates prostaglandin-E2, lipid peroxidation IL-1β, cyclooxygenase-2 and Toll-like receptor-4. NK-1, neurokinin-1 receptor.
9. Depression and daylight
Deficiency of light has been shown to suppress neurogenesis and increase symptoms of depression (65). Nocturnal melatonin, vitamin D and platelet 5-HT values are induced by environmental light and improve depression symptoms (66,67). Melatonin has anti-oxidative effects, increases sleep propensity and has been known to decrease alertness and attenuate weight gain (68) (Fig. 7).
Figure 7.
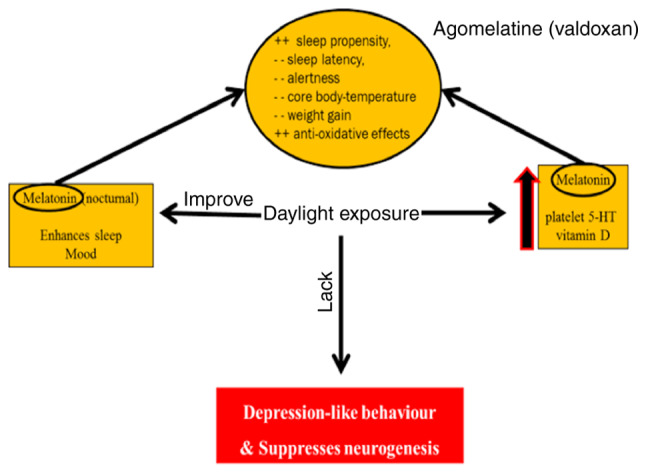
Depression and daylight. Deficiency of light has been shown to suppress neurogenesis and increase symptoms of depression. Nocturnal melatonin, vitamin D and platelet 5-HT values are induced by environmental light and improve depression symptoms. Melatonin has anti-oxidative effects, increases sleep propensity and has been known to decrease alertness and attenuate weight gain. 5-HT, 5-hydroxytryptamine.
10. Depression in the deficiency state
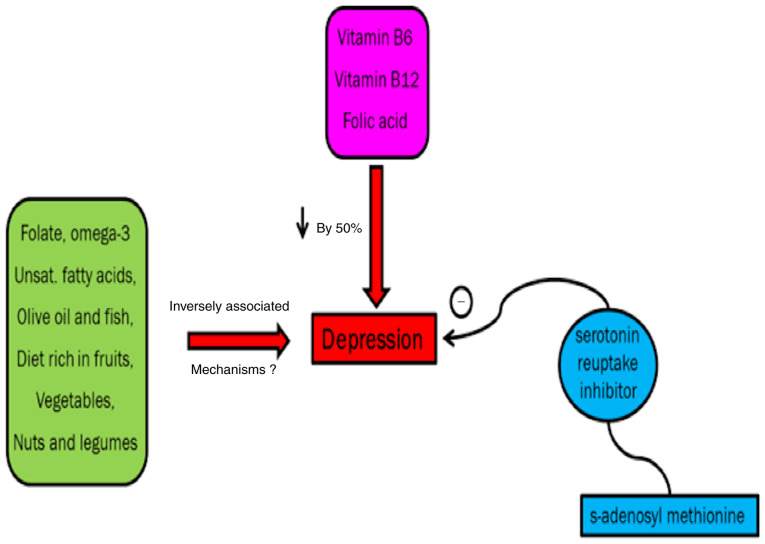
Intake of omega-3 fatty acids, fish, vitamin B6, vegetables, fruits, folate, olive oil, vitamin B12, nuts and legumes improves depressive symptoms although the exact mechanism remains to be elucidated (69,70).
A previous study demonstrates that unipolar depression can be effectively controlled by folic acid (71). A study conducted on >70 patients showed that major depressive disorder in non-responders to SSRIs could be improved by s-adenosyl methionine if taken concurrently with serotonin reuptake inhibitor (72) (Fig. 8).
Figure 8.
Depression and deficiency. Intake of omega-3 fatty acids, fish, vitamin B6, vegetables, fruits, folate, olive oil, vitamin B12, nuts and legumes improves depressive symptoms, although the exact mechanism remains to be elucidated. A previous study demonstrates that unipolar depression can be effectively controlled by folic acid. A study conducted on >70 patients showed that major depressive disorder in non-responders to selective serotonin reuptake inhibitors could be improved by s-adenosyl methionine if taken concurrently with serotonin reuptake inhibitor.
11. Depression and glutamate
The ionotropic glutamate receptors N-methyl-D-aspartate (NMDA), α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid and metabotropic glutamate receptors (mGluR1 to mGluR8) activation regulate synaptic plasticity and transmission. Glutamate is cleaned by high-affinity excitatory amino acid transporters (EAAT) 1 and 2 from extracellular space found in glial cells (73).
Following the conversion of the glial cells glutamate to glutamine, by glutamine synthetase, glutamine is hydrolyzed into glutamate via glutaminase (74). In experimental animal models of depression, drugs that increase glutamate clearance can improve stress and possess antidepressant effects (75,76).
Ketamine N-methyl-D-aspartate receptor antagonist in subanesthetic dose (0.5 mg/kg) has a short onset antidepressant effect (77). Another rapid-acting antidepressant is dextromethorphan (NMDA receptor antagonist) (78). The antimuscarinic drug scopolamine has short onset with a potent antidepressant effect for bipolar and major depression (79,80). As with NMDA receptor antagonists, zinc and magnesium have an antidepressant effect in experimental animal models (81). It has been stated that magnesium has an antidepressant effect in type II diabetic patients, compared with imipramine (82).
12. Depression in neurodegenerative cases
Long-time stress may negatively affect the process of neurogenesis and cause neuronal loss in the hippocampus (83,84). In depressive behaviors, synaptic plasticity has been revealed to be controlled by brain-derived neurotrophic factor (BDNF) (85,86). The blood level of BDNF is decreased in patients with depression and its level is increased by antidepressant drugs (87,88).
In patients with depression, a decreased level of vascular growth factor (VGF) has been shown (89) and an antidepressant effect is produced by treatment with recombinant VGF in an experimental animal model (90). Neurotrophin-3 and nerve growth factor may contribute to the antidepressant responses by improving plasticity and neurogenesis in the hippocampus (91,92). Other factors such as glial cell line-derived neurotrophic factor and fibroblast growth factor-2 are modulated in depression (58,93) (Fig. 9).
Figure 9.
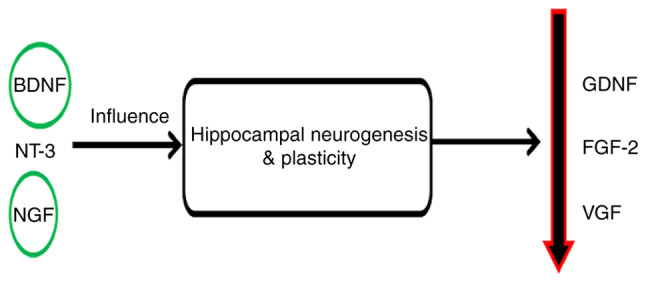
Depression and neurodegenerative cases. Long-time stress may negatively affect the process of neurogenesis and cause neuronal loss in the hippocampus. In depressive behaviors, synaptic plasticity has been revealed to be controlled by BDNF. The blood level of BDNF is decreased in patients with depression and its level is increased by antidepressant drugs. In patients with depression, a decreased level of VGF has been shown and an antidepressant effect is produced by treatment with recombinant VGF in an experimental animal model. NT-3 and NGF may contribute to the antidepressant responses by improving plasticity and neurogenesis in the hippocampus. Other factors, such as GDNF and FGF-2, are modulated in depression. BDNF, brain-derived neurotrophic factor; VGF, vascular growth factor; NT-3, neurotrophin-3; NGF, nerve growth factor; GDNF, glial cell line-derived neurotrophic factor; FGF-2, fibroblast growth factor-2.
13. Depression and oxidative stress
In patients with depression, numerous enzyme-mediated reactive oxygen species (ROS) production is increased (94). Superoxide and hydrogen peroxide are products of oxidation of xanthine mediated by xanthine oxidase enzyme that has been found to increase in the thalamus in postmortems of patients with depression (95). Metabolism and oxidative deamination of serotonin are exaggerated by monoamine oxidase, which can cause ROS production leading to mitochondrial dysfunction and neuronal apoptosis (96). In addition, COX-2 is involved in the inflammatory cascades and its inhibitors can lead to neuronal inflammation that may worsen depression (97). Decreased superoxide dismutase activity is shown in patients diagnosed with depression (98). Peroxides, such as hydrogen peroxide catalyzed by glutathione peroxidase, have lower activity in patients with depression compared to healthy individuals (99). Microglia are the main inflammatory neuronal cells that are activated by the increased levels of superoxide and other ROS (100). This activated cell releases a number of inflammatory cytokines, mostly TNF-α and IL-6 (101).
It has been demonstrated that an increased level of nitric oxide (NO) mediates the 3′,5′-cyclic guanosine monophosphate (cGMP) signaling pathway leading to depressive symptoms; however, the reduction of NO and/or cGMP results in improvement in depression-like symptoms in experimental animal models (102). The elevated level of NO may react with superoxide anions and produce highly reactive inflammogenic and neurotoxic peroxynitrite and consequently a local surge in inflammatory cytokines (103). The previous NO effects are involved in the pathogenesis of major depression (104), Parkinson's disease and Alzheimer's (103).
14. Gene expression in depression
There are differences in the cortical gene expression in patients with schizophrenia, bipolar disorder and autism including differentiation of the glial cell and metabolism of fatty acid (105,106). Notably, the pathophysiology of major depression (MD) involves inflammatory pathways and the hypothalamic-pituitary axis (107). Another study found low expression of two genes, ATP binding cassette subfamily G member 2 and prominin 1 in patients with MD (108). Similarly, a low level of mRNA encoding the gene Fli-1 proto-oncogene, ETS transcription factor has been found and it serves a role in the growth of the neuronal crest in patients with depression (109). Reduction in the expression of SLC1A2 and SLC1A3 genes has been found in the dorsolateral prefrontal cortex and anterior cingulate of patients with MD and bipolar disorder (110). A lower expression of NMDA receptor subunits was found in patients with MD including PSD-95, NR1, NR2A and NR2B in the anterior prefrontal cortex (111). GABA signaling-related genes such as ALDH9A1 and glutamate ammonia ligase, are changed in patients with depression (112). ALDH9A1, which catalyzes the conversion of γ-aminobutyraldehyde into GABA, was increased in depressed subjects, whereas glutamate ammonia ligase, which increases glutamate brain clearance by its conversion to glutamine, was decreased in depressed brain. Furthermore, decreased activation and functions in the gene expression of the extracellular signal-regulated kinase (ERK) cascade is recorded in MD (113). Several deviations in the gene expression in MD were found to be gender-specific, occurring mostly in females. For instance, hub gene Dusp6 (female-specific gene) is downregulated in female, but not male mice prefrontal cortex simulated stress by increasing pyramidal neuron excitability and ERK signaling. This gender-specific feature in MD patients could be due to changes in the specificity of ventromedial cortex content of phosphatase 6 in female patients and empty spiracles homeobox 1 in males (113).
15. Treatment of depression
None drug treatment
Encouraging creativity
Reading books or journals, gardening, painting and playing music can offer a passage to drive out the painful thoughts and bad moods. It is known that creative individuals are at high risk for depression than non-creative ones (114).
Physical therapy
In this, patients with depression are directed to perform physical exercise. The only challenge facing this strategy is that patients with depression often suffer from a lack of energy. However, starting a new physical activity can be influential in managing such symptoms. Performing physical exercise is known to produce euphoria by releasing endorphins (115).
Psychiatric therapy
Cognitive Behavioral Therapy is a common psychotherapy strategy used in the treatment of depression. This strategy directs patients to correct negative non-constructive thinking (116).
Alternative therapy
A 40 min exposure to sunlight by patients suffering from seasonal depression results in a good response (117). This type of therapy may be helpful in patients suffering from seasonal depression. St. John's Wort and acupuncture are used and can improve depressive behavioral symptoms (118).
Antidepressant drugs
Most antidepressants employ important activities on the metabolism of monoamine neurotransmitters and their receptors, mainly norepinephrine and serotonin (119). The well-known antidepressant classes include SSRIs, TCAs, NRIs, SARIs, SNRIs and noradrenaline and dopamine reuptake inhibitors (Table III,Table IV,Table V,Table VI,Table VII,Table VIII,Table IX) (120).
Table III.
Mono amino oxidase inhibitors (120).
| Drug | Selectivity | FDA approval | Common adverse effects >10% incidence | Administration in pregnancy | Interactions |
|---|---|---|---|---|---|
| Isocarboxazide | Non-selective MAOIs | 1959 | Weight gain, chills orthostatic hypotension, forgetful, headache, sexual dysfunction, lethargy, constipation, dizziness, serotonin syndrome | No data | Salmeterol, trazodone, alcohol, citalopram, zolpidem, duloxetine, amitriptyline, venlafaxine, escitalopram, phenelzine and lisdexamfetamine. |
| Phenelzine | 1961 | Dizziness, confusion, chills, tremors, drowsiness, weakness, insomnia and dry mouth | No data | Citalopram, duloxetine, dextromethorphan, meperidine, fentanyl, tryptophan, lisdexamfetamine and vilazodone. | |
| Selegiline | 1998 | Orthostasis, dizziness, nausea and abdominal pain | No | Duloxetine, escitalopram, meperidine, rasagiline, bupropion and amphetamine | |
| Tranylcypromine | Selective MAO-A inhibitors | 1961 | Anxiety, dizziness, weakness and constipation. | No data | Duloxetine, citalopram, selegiline, phenelzine, escitalopram, paroxetine, pseudoephedrine and lisdexamfetamine. |
| Brofaromine | 1995 | Tremor, sleep disorders, dry mouth and hypotension | No data | Warfarin, amantadine, amitriptyline, apomorphine, benzodiazepine, bupropion, dobutamine, duloxetine and ethanol. |
MAO, mono amino oxidase.
Table IV.
Selective serotonin reuptake inhibitors (164).
| Drug | FDA approval | Common adverse effects >10% incidence | Administration in pregnancy | Contraindications/interactions |
|---|---|---|---|---|
| Cetalopram | 1998 | Sexual dysfunction, Sedation, drowsiness, blurred vision, insomnia, hypernatremia, nausea, eye pain or swelling, vivid dreaming, pounding heartbeats, decreased sex drive, anorgasmia, dry mouth, trembling, diarrhea and slurred speech. | No | Tryptophan, St John's Wort, 5-HTP (serotonin syndrome), cimetidine, lithium and Omeprazole |
| Escitalopram | 2002 | Headache, nausea, decreased libido, delayed ejaculation, weight gain, dose-dependent QT interval prolongation on the ECG. | Has been linked a very small increased to risk of problems for your unborn baby. | Substrates that increase CYP2D6 such as codeine, aripiprazole, or risperidone. Acetaminophen, citalopram, clonazepam, levothyroxine or quetiapine |
| Paroxetine | 1992 | Loss of appetite, nausea, asthenia diarrhea, blurred vision, constipation, dry mouth, somnolence, insomnia, headache, hypomania, nervousness, paraesthesia, dizziness and sexual dysfunction. | No | MAOI, pimozide, thioridazine, tryptophan, or warfarin Substrates of CYP2B6, CYP3A4, CYP1A2, CYP2C9 and CYP2C19. |
| Fluoxetine | 1987 | Headache, nausea, agitation, insomnia, appetite loss, Anxiety, asthenia, diarrhea and somnolence | Major birth defects during the first trimester. | Inhibits a number of isozymes of the cytochrome P450 Avoided with serotonergic drugs such as methamphetamine, MAOI, buspirone, TCA, MDMA, triptans and serotonin-norepinephrine reuptake inhibitors. |
| Fluvoxamine | 1983 | Nausea, headache, anorexia, insomnia, agitation, dizziness, nervousness, anxiety, somnolence, tremor and palpitations. | No | Fluvoxamine the following cytochrome P450; metabolizes agomelatine, amitriptyline, caffeine, alprazolam and aripiprazole. |
| Sertraline | – | Psychiatric side effects, anxiety, agitation, insomnia, sexual arousal disorder and diarrhea | No | Moderate of CYP2D6 inhibitor and CYP2B6 in vitro? |
Table V.
Tricyclic antidepressants (165).
| Drug | FDA approval | Common adverse effects >10% incidence | Administration in pregnancy | Contraindications/interactions |
|---|---|---|---|---|
| Amitriptyline | 1961 | Anticholinergic side effects, dizziness, headache, somnolence, orthostatic hypotension, loss of libido, agitation and insomnia | No | Hypersensitivity, history of myocardial infarction and history of arrhythmias. Monoamine oxidase inhibitors, anticholinergic, antipsychotics, cimetidine and antithyroid medications. |
| Butriptyline | Not | Similar to amitriptyline | No | Similar to amitriptyline |
| Clomipramine | 1960 | Dizziness, blurred vision, tremor, sexual dysfunction, nausea, dry mouth, fatigue, constipation, weight gain and increased appetite. | No | MAOI, quinidine (metabolized by CYP2D6) hypersensitivity, myocardial infarction, severe liver disease and narrow angle glaucoma |
| Desipramine | 1964 | Cardiac dysrhythmias and increased risk of breast cancer in women | Has not been well studied | Overactive thyroid gland, schizophrenia, manic-depression, alcoholism, suicidal thoughts, closed angle glaucoma. Aripiprazole, aripiprazole, amphetamine duloxetine, duloxetine, and lamotrigine. |
| Dosulepin | Not | Paraesthesias, drowsiness, dizziness, sweating, extrapyramidal symptoms and tremor, hypotension, confusion, disorientation, ECG alterations, dry mouth and urinary retention. | No | Epilepsy, myocardial infarction and hepatic failure. Potentiate the effects of alcohol, barbiturates, CNS depressants and tranquillizers. |
| Doxepin | Approved? | CNS: fatigue, dizziness, drowsiness, confusion and nightmares. Anticholinergic, allergy and may increase or decrease liver function in some patients. | No | MAOI and potentiates sympathomimetics Hypersensitivities, glaucoma, poor metabolizers and respiratory impairment |
| Imipramine | 1950s | CNS: Dizziness, drowsiness, confusion, seizures, headache and anxiety. | – | – |
| CVS: Orthostatic hypotension, hypertension, tachycardia. | ||||
| GIT: anorexia, epigastric distress, diarrhea Hematological: bone marrow depression. | ||||
| Iprindole | – | Anticholinergic side effects and sedation | Inactivates cytochrome | Inactivates cytochrome P450 Should not be taken by alcoholics and those with liver disease. |
Table VI.
Norepinephrine reuptake inhibitors (NRIs), (166).
| Drug | FDA approval >10% incidence | Common adverse effects | Administration in pregnancy | Contraindications/interactions |
|---|---|---|---|---|
| Atomoxetine | Approved to treat attention-deficit/hyperactivity disorder in pediatric and adult patients | Nausea, xerostomia (dry mouth), appetite loss, insomnia, fatigue, headache and cough | Use during pregnancy has not been well studied for | Hypersensitivity and Symptomatic cardiovascular disease. Treatment with CYP2D6 inhibitors or inducers, Antihypertensive and pressor agents and β-adrenoceptor agonists |
| Reboxetine | Still not fully approved | Insomnia, nausea, hyperhidrosis, dry mouth and constipation | Only if the potential benefits justify the potential risks to the fetus | Prostatic hypertrophy, narrow-angle glaucoma, epilepsy, CV disease, bipolar disorder, MAOIs and urinary retention. Markedly inhibited by papaverine and ketoconazole (due to inhibition of CYP3A4). |
| Viloxazine | Withdrawn in 2002 | Nausea, vomiting, insomnia, loss of appetite, increased ESR, EEG anomalies, vertigo, orthostatic hypotension, edema of the lower extremities, tremor and mental confusion. | Based on experimental studies; maternal harm when used during pregnancy | Increased plasma levels of phenytoin and increased plasma levels of theophylline. |
Table VII.
Serotonin antagonist and reuptake inhibitors (167).
| Drug | FDA approval | Common adverse effects >10% incidence | Administration in pregnancy | Contraindications/interactions |
|---|---|---|---|---|
| Etoperidone | Discontinued | – | – | – |
| Lubazodone | Discontinued | – | – | – |
| Nefazodone | 2004 | Dry mouth, sedation/somnolence, nausea, dizziness, blurred/abnormal vision, weakness, lightheadedness, confusion and orthostatic hypotension | Not allowed (category C) | Potent inhibitor for CYP3A4; interacts with compounds metabolized by CYP3A4 |
| Trazodone | 1981 | Sedation, orthostatic hypotension, Cardiac arrhythmia and Priapism (rare) | Sufficient data in humans are lacking | Increased serum phenytoin and digoxin levels. Enhance the sedative effects of barbiturates, alcohol and other CNS depressants |
Table VIII.
Serotonin-norepinephrine reuptake inhibitors (168).
| Drug | FDA approval | Common adverse effects >10% incidence | Administration in pregnancy | Contraindications/interactions |
|---|---|---|---|---|
| Desvenlafaxine | 2008 | Insomnia, nausea, headache, diarrhea, constipation, dry mouth, dizziness, fatigue and hyperhidrosis | Only if the potential benefits justify the potential risks to the fetus | Sertraline, aripiprazole, amphetamine, bupropion, dextroamphetamine, alprazolam, duloxetine, Fish Oil, lamotrigine, lurasidone, lisdexamfetamine, escitalopram, lithium, pregabalin, acetaminophen, ascorbic acid, quetiapine, cyanocobalamin, cholecalciferol and cetirizine. |
| Duloxetine | 2003 | Nausea, somnolence, insomnia and dizziness | Increases the risk of congenital malformations, preterm birth, or pre-eclampsia | Hypersensitivity, CNS acting drugs with similar mechanism and thioridazine |
| Levomilnacipran | 2013 | Blood pressure increases, nausea/vomiting, sweating, constipation, erectile dysfunction in men and increased heart rate | Only if the potential benefits justify the potential risks to the fetus | Aripiprazole, amphetamine, dextro amphetamine, clonazepam, duloxetine, gabapenti, clonazeamn, lamotrigine, pregabalin, metformin, omeprazole, quetiapine, topiramate, tramadol and trazodone |
| Milnacipran | 2009 | Tachycardia, palpitations, dry mouth, hypertension, nausea, vomiting, headache, insomnia, dizziness, constipation, hyperhidrosis and facial flushing | Not allowed (category C) | MAOIs (serotonin syndrome), angina pectoris, myocardial infarction, epinephrine, norepinephrine, clonidine, digitalis and triptans and alcohol |
| Venlafaxine | 2006 | Headache, nausea, dry mouth, somnolence, asthenia, dizziness, failure of ejaculation and sweating. | – | Hypersensitive and with MAOI |
Table IX.
Noradrenaline and dopamine reuptake inhibitors (169).
| Drug | FDA approval | Common adverse effects >10% incidence | Administration in pregnancy | Contraindications/interactions |
|---|---|---|---|---|
| Bupropion | 1985 | Anxiety, restlessness, dry mouth, hyperventilation, irritability, shaking, irregular heartbeats, trouble sleeping and ringing in the ears. | Only if the potential benefit outweighs the potential risk to the fetus | Epilepsy, conditions that lower the seizure threshold (anorexia nervosa, bulimia nervosa, benzodiazepine or alcohol withdrawal), MAOIs, liver damage, kidney disease, severe hypertension, in children and adolescents (risk of suicidal attempt). Drug-drug interactions: acetaminophen, diphenhydramine Drug-food/beverage interactions: grapefruit juice |
Other antidepressants
LY03005 (Ansofaxine hydrochloride)
Ansofaxine is developed by Luye Pharma Group for the treatment of major depression. The American food and drug administration (FDA) approved the serotonin-norepinephrine-dopamine triple reuptake inhibitor LY03005 in December 2020 under the name of Ansofaxine (121). It was developed under Luye pharma's new chemical and therapeutic entities research and development platform (122). Triple reuptake inhibitors encourage a class of antidepressants and are considered to have high efficacy and faster antidepressant response. They have relative advantages in preserving patients' sexual functions, improved efficacy, rapid onset and improved safety profile (122).
Duloxetine
Duloxetine hydrochloride (Cymbalta) is an oral selective serotonin and norepinephrine reuptake inhibitor. It has been accepted in the treatment of different mental disorders, including depressive behavioral illness (123). One important benefit that leads to the use of duloxetine in the treatment of depression is its effect in the prevention of suicidal thoughts and attempts (124). Wu et al (125) demonstrate that duloxetine may be effective in the treatment of epileptic patients with depression. A recent study demonstrated that a spray-dried complexation-based transdermal patch of duloxetine functions as a possible new drug delivery system in the treatment of depression (126).
Vilazodone (Viibryd)
Vilazodone was approved by the FDA in January 2011 and it is the first and only selective serotonin reuptake inhibitor and 5-HT1A receptor partial agonist (127). A large dose of vilazodone is accompanied by acute serotonin syndrome. Some serious uncommon adverse effects include suicidal thoughts, sexual dysfunction, manic behavior, serotonin syndrome and hyponatremia (128).
GLYX-13 (Rapastinel)
GLYX-13 finalized phase II in 2015 and on January 29, 2016 Allergan company reported that Rapastinel had received approval from the FDA for treatment of major depression (129). GLYX-13 functions by controlling the neuronal N-methyl-D-aspartate (NMDA) receptor. Although it is similar to ketamine receptor antagonists of NMDA, GLYX-13 does not cause severe adverse effects such as schizophrenic symptoms and hallucinations effects (130).
Vortioxetine (Brintellix)
Brintellix, used for the treatment of major depression, is recently available in the United States markets (131). The antidepressant mechanism of Brintellix remains to be elucidated. Vortioxetine is a 5-HT1D, 5-HT3 and 5-HT7 receptors antagonist, 5-HT1B partial receptors agonist and 5-HT1A receptor agonist (132). Plasma concentration of BDNF shows a significant increase and the concentration of platelet 5-HT displays significant reduction following Brintellix administration in patients with major depression (129).
Agomelatine (Valdoxan)
Valdoxan targets the melatonin system in the brain and is considered a melatonergic drug. It was approved for the treatment of major depression in Europe in 2009. It has been recently reported that Agomelatine is effective in PPD treatment and is safe during breastfeeding (133). Agomelatine works by targeting the neuronal effects of melatonin, which has an essential role as an antidepressant. Similar to serotonin, agomelatine appears to be important in controlling sleep and circadian rhythms (134,135).
Ketamine
It is an anesthetic drug that has an antidepressant effect but is not yet accepted as an antidepressant by the FDA. Ketamine acts on a number of central nervous system receptors responsible for learning and memory. These receptors work on the excitatory neurotransmitter, glutamate (136), which activates mTORC1 kinase via inactivation of eukaryotic initiation factor 4E-binding proteins (137). Intranasal spray of ketamine was FDA approved as an antidepressant. Finally, the safety and efficacy of ketamine in depression control requires further investigation (138).
Levomilnacipran
Levomilnacipran is a rapid onset drug used for treatment of depression that belongs to a class of SNRIs. There is no evidence to indicate any relative advantages of levomilnacipran compared with available SNRIs (139). A recent study demonstrated that levomilnacipran in older patients with depression has lower tolerability than in older adults with depression. Levomilnacipran requires to be more deeply studied to analyze its efficacy and tolerability in patients with depression (140).
Quetiapine (Seroquel XR)
Although it inhibits nerve communication between the brain areas, like other atypical anti-psychotics, the mechanism of quetiapine remains to be elucidated. A promising result of SNRI (Venlafaxine) and Quetiapine co-treatment in patients with depression has been reported (141). The benefits of Quetiapine may be due to inhibition of the serotonin type 2 and dopamine type 2 receptors (142). Hazards of mortality, suicidal thoughts and stroke in the elderly are common among patients treated with Quetiapine (143).
16. COVID-19 and depression
The outbreak of COVID-19 seriously influences mental health. The pandemic represents a stressful situation worldwide and at the time of writing this review, the confirmed cases are >177,108,695 including 3,840,223 mortalities (WHO; http://covid19.who.int). Mental health problems resulting from this pandemic include insomnia, anxiety, depressive behavior, stress and fear (144). In the aftermath of the COVID-19 pandemic, depression and anxiety are common psychological complaints (145). Lai et al (146) observed the mental health status during the COVID-19 pandemic in 1,257 doctors and other healthcare staff in China. It was found that 71.5% of the study sample claimed distress, 50.4% depression, 44.6% anxiety and 34.0% insomnia. In addition, a possible correlation between the symptoms in COVID-19 patients and potential neurological consequences has been found (147). At present, nothing is known about the changes in cognitive functioning or emotions from the direct effects of the virus on the brain. A number of individuals are in health quarantine and isolated at home and have lost their jobs, family members, relatives, colleagues, or friends. Accordingly, depression due to COVID-19 may be included under the situational or emotional type of depression.
There are a number of drugs under investigation in different research institutes, universities, or under clinical trials in hospitals. The investigational drugs include Baricitinib, interferon-β 1b, Bemcentinib, Bevacizumab, chloroquine phosphate, Colchicine, Dexamethasone, EIDD-2801, Favipiravir, Hydroxychloroquine, Azithromycin, anticoagulants, Lopinavir, Ritonavir and Remdesivir. The current use of some of these drugs in certain areas is marked with hopes and promises without attention being payed to their behavioral side effects and lethality. For example, Baricitinib (Olumiant R which is a Janus kinase inhibitor now in phase III clinical trials for COVID-19 treatment and has been used in the treatment of rheumatoid arthritis. Olumiant may occasionally lead to depression as a side effect (148).
Another drug that has been used recently in the treatment of COVID-19 is interferon-β 1b. Clinical studies on the efficacy of interferon I α and interferon β in SARS-CoV had differing results. Interferon-β 1b may increase the speed of COVID-19 viral clearance (149). The most serious side effects of interferon-β-1b in clinical trials are attempted suicide, depressive behavior and necrosis at the site of injection (150). Dexamethasone is a common and widely used synthetic steroid for a number of diseases, which can reduce mortality of seriously ill COVID-19 patients although with no improving effect in less serious cases (151). Fortunately, Dexamethasone level has previously been shown to possess a good prognosis for endocrine dysfunction in depression (152).
The United States FDA has approved the use of the antimalarial drugs chloroquine (N4-(7-chloro-4-quinolinyl)-N1,N1-diethyl-1,4-pentanediamine) and hydroxychloroquine sulfate (a derivative of chloroquine) in COVID-19 treatment regardless of a questionable clinical proof of their effectiveness (153). Behavioral changes, delirium and depression have been associated with chloroquine use (153). One of the most important points that must be taken into account is the older population, who are directed to self-isolate for a long time. This is to protect over-loaded health systems and to diminish the outbreak of COVID-19. This social isolation not only increases the risk for neurocognitive, cardiovascular and autoimmune diseases but also exposes elder populations to a greater risk of depression (154). Decision makers and leaders need to realize the resultant mental health changes. In addition, they must take the situational depression existing at present into their consideration and give it the care and attention it deserves.
17. Depression and cytokine storm in COVID-19
Alteration in consciousness is one of COVID-19 manifestations and accounts for 20% of patients (155). Furthermore, increased incidence of psychiatric disorders has been identified during and after the infection with COVID-19 (156). Most of these patients display cytokine storm or cytokine release syndrome and showed raised plasma levels of pro-inflammatory cytokines including IL-2R, IL-6, IL-8, IL-10 and TNF-α (157). Depression is known to be an inflammatory state in which the patients manifested high levels of inflammatory cytokines, normalized after anti-depressants treatment (158). In the central nervous system and the peripheral blood, elevated levels of IL-1β, IL-6, IL-8, CRP and TNF-α cytokines are identified in patients with depression (159). Blocking of cytokines by IL-1β receptor antagonist, Anakinra or IL-6 receptor antagonist Tocilizumab (160) has benefits on the inflammation-mediated respiratory failure in patients with depression. Based on the previous reports, the present study suggested the protective effect of cytokine blockers against depression symptoms in COVID-19 patients.
18. Summary
Treatment of depression remains a great public mental health problem. Depression comprises different types, some of which are related to events in a person's life while others are related to the chemical changes in the brain. In some cases, depression may be considered as a risk factor for certain diseases (e.g. type 1 or 2 diabetes), and in other cases, certain diseases (e.g. Alzheimer's and epilepsy) may lead to depressive episodes. Thus, the relationship between such diseases and depression was discussed in the present study. In addition, the newer treatments of depression, such as modulation of glutamatergic, neuronal NMDA and serotonergic systems and triple reuptake inhibitors, were also presented. The newer treatments result in a lower risk of suicidal attempts compared to older treatments. However, they still have some risks including the possibility of drug abuse. Cooperation between governmental organizations, researchers, clinicians, patients and family is crucial in future progress.
Until now, nothing has been known about the direct effect of COVID-19 on the brain. What health care staff can do is to treat depression resulting from COVID-19 infection and its complications, if there are any. The challenge facing health care staff is to treat the so-called defeat stress resulting from the consequences of COVID-19 infection. Defeat stress consequences may not be able to respond to the classical antidepressants which include maladaptive behaviors (161,162). COVID-19 patients need both pharmacological treatment and mental care. Mental health personnel have to be close to COVID-19 patients directly or indirectly through the internet video media or telephone to soothe or prevent waves depression or low moods. Decision-makers and officials all over the world need to know that emotional or situational depression has to be taken seriously.
Acknowledgements
Researchers would like to thank the Deanship of Scientific Research, Qassim University for funding the publication of this project.
Funding
Researchers would like to thank the Deanship of Scientific Research, Qassim University for funding the publication of this project.
Availability of data and materials
Not applicable.
Authors' contributions
MSAB conceived, designed, analyzed and presented diagrams, wrote and revised the review. AHA and EA constructed diagrams, wrote and revised the review. TMF wrote and revised the review. Data authentication is not applicable. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Averina OV, Zorkina YA, Yunes RA, Kovtun AS, Ushakova VM, Morozova AY, Kostyuk GP, Danilenko VN, Chekhonin VP. Bacterial metabolites of human gut microbiota correlating with depression. Int J Mol Sci. 2020;21:9234. doi: 10.3390/ijms21239234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Delgado PL, Moreno FA. Role of norepinephrine in depression. J Clin Psychiatry. 2000;61((Suppl 1)):S5–S12. [PubMed] [Google Scholar]
- 3.Zhang FF, Peng W, Sweeney JA, Jia ZY, Gong QY. Brain structure alterations in depression: Psychoradiological evidence. CNS Neurosci Ther. 2018;24:994–1003. doi: 10.1111/cns.12835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fox ME, Lobo MK. The molecular and cellular mechanisms of depression: A focus on reward circuitry. Mol Psychiatry. 2019;24:1798–1815. doi: 10.1038/s41380-019-0415-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Muller JK, Leweke FM. Bipolar disorder: Clinical overview. Med Monatsschr Pharm. 2016;39:363–369. [PubMed] [Google Scholar]
- 6.American Psychiatric Association A and Association AP, corp-author. American Psychiatric Association; Washington, DC: 2013. Diagnostic and Statistical Manual of Mental Dsorders (DSM-5) [Google Scholar]
- 7.Assari S. Chronic medical conditions and major depressive disorder: Differential role of positive religious coping among African Americans, Caribbean blacks and non-hispanic whites. Int J Prev Med. 2014;5:405–413. [PMC free article] [PubMed] [Google Scholar]
- 8.Devanand DP. Dysthymic disorder in the elderly population. Int Psychogeriatr. 2014;26:39–48. doi: 10.1017/S104161021300166X. [DOI] [PubMed] [Google Scholar]
- 9.Molyneaux E, Poston L, Ashurst-Williams S, Howard LM. Obesity and mental disorders during pregnancy and postpartum: A systematic review and meta-analysis. Obstet Gynecol. 2014;123:857–867. doi: 10.1097/AOG.0000000000000170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sanassi LA. Seasonal affective disorder: Is there light at the end of the tunnel? JAAPA. 2014;27:18–23. doi: 10.1097/01.JAA.0000442698.03223.f3. [DOI] [PubMed] [Google Scholar]
- 11.Gremaud-Heitz D, Riemenschneider A, Walter M, Sollberger D, Kuchenhoff J, Dammann G. Comorbid atypical depression in borderline personality disorder is common and correlated with anxiety-related psychopathology. Compr Psychiatry. 2014;55:650–656. doi: 10.1016/j.comppsych.2013.11.021. [DOI] [PubMed] [Google Scholar]
- 12.Østergaard SD, Meyers BS, Flint AJ, Mulsant BH, Whyte EM, Ulbricht CM, Bech P, Rothschild AJ, STOP-PD Study Group Measuring psychotic depression. Acta Psychiatr Scand. 2014;129:211–220. doi: 10.1111/acps.12165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Muralidharan K, Torres IJ, Silveira LE, Kozicky JM, Bücker J, Fernando N, Yatham LN. Impact of depressive episodes on cognitive deficits in early bipolar disorder: Data from the systematic treatment optimization programme for early mania (STOP-EM) Br J Psychiatry. 2014;205:36–43. doi: 10.1192/bjp.bp.113.135525. [DOI] [PubMed] [Google Scholar]
- 14.Benti L, Manicavasagar V, Proudfoot J, Parker G. Identifying early indicators in bipolar disorder: A qualitative study. Psychiatr Q. 2014;85:143–153. doi: 10.1007/s11126-013-9279-x. [DOI] [PubMed] [Google Scholar]
- 15.Harnic D, Pompili M, Innamorati M, Erbuto D, Lamis DA, Bria P, Girardi P, Janiri L. Affective temperament and attachment in adulthood in patients with bipolar disorder and cyclothymia. Compr Psychiatry. 2014;55:999–1006. doi: 10.1016/j.comppsych.2013.12.006. [DOI] [PubMed] [Google Scholar]
- 16.Craner JR, Sigmon ST, Martinson AA, McGillicuddy ML. Premenstrual disorders and rumination. J Clin Psychol. 2014;70:32–47. doi: 10.1002/jclp.22007. [DOI] [PubMed] [Google Scholar]
- 17.Simpson T, Ivey J. Pediatric management problems. Situational depression. Pediatr Nurs. 2005;31:312–313. [PubMed] [Google Scholar]
- 18.Jorm AF, Christensen H, Griffiths KM, Rodgers B. Effectiveness of complementary and self-help treatments for depression. Med J Aust. 2002;176:S84–S96. doi: 10.5694/j.1326-5377.2002.tb04508.x. [DOI] [PubMed] [Google Scholar]
- 19.Baldwin DS. Unmet needs in the pharmacological management of depression. Hum Psychopharmacol. 2001;16:S93–S99. doi: 10.1002/hup.337. [DOI] [PubMed] [Google Scholar]
- 20.Hossain MM, Tasnim S, Sultana A, Faizah F, Mazumder H, Zou L, McKyer ELJ, Ahmed HU, Ma P. Epidemiology of mental health problems in COVID-19: A review. F1000Res. 2020;9:636. doi: 10.12688/f1000research.24457.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pedersen CB, Mors O, Bertelsen A, Waltoft BL, Agerbo E, McGrath JJ, Mortensen PB, Eaton WW. A comprehensive nationwide study of the incidence rate and lifetime risk for treated mental disorders. JAMA Psychiatry. 2014;71:573–581. doi: 10.1001/jamapsychiatry.2014.16. [DOI] [PubMed] [Google Scholar]
- 22.Mojtabai R, Olfson M, Han B. National trends in the prevalence and treatment of depression in adolescents and young adults. Pediatrics. 2016;138:e20161878. doi: 10.1542/peds.2016-1878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Substance Abuse and Mental Health Services Association, corp-author. 2018 [Google Scholar]
- 24.Duffy ME, Twenge JM, Joiner TE. Trends in mood and anxiety symptoms and suicide-related outcomes among U.S. undergraduates, 2007–2018: Evidence from two national surveys. J Adolesc Health. 2019;65:590–598. doi: 10.1016/j.jadohealth.2019.04.033. [DOI] [PubMed] [Google Scholar]
- 25.Kuehner C. Why is depression more common among women than among men? Lancet Psychiatry. 2017;4:146–158. doi: 10.1016/S2215-0366(16)30263-2. [DOI] [PubMed] [Google Scholar]
- 26.Wang PS, Aguilar-Gaxiola S, Alonso J, Angermeyer MC, Borges G, Bromet EJ, Bruffaerts R, de Girolamo G, de Graaf R, Gureje O, et al. Use of mental health services for anxiety, mood, and substance disorders in 17 countries in the WHO world mental health surveys. Lancet. 2007;370:841–850. doi: 10.1016/S0140-6736(07)61414-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Katon W, Sullivan MD. Depression and chronic medical illness. J Clin Psychiatry. 1990;51((Suppl)):S3–S14. [PubMed] [Google Scholar]
- 28.Lebowitz BD, Pearson JL, Schneider LS, Reynolds CF, III, Alexopoulos GS, Bruce ML, Conwell Y, Katz IR, Meyers BS, Morrison MF, et al. Diagnosis and treatment of depression in late life. Consensus statement update. JAMA. 1997;278:1186–1190. doi: 10.1001/jama.278.14.1186. [DOI] [PubMed] [Google Scholar]
- 29.Slavich GM, Irwin MR. From stress to inflammation and major depressive disorder: A social signal transduction theory of depression. Psychol Bull. 2014;140:774–815. doi: 10.1037/a0035302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Green RC, Cupples LA, Kurz A, Auerbach S, Go R, Sadovnick D, Duara R, Kukull WA, Chui H, Edeki T, et al. Depression as a risk factor for Alzheimer disease: The MIRAGE study. Arch Neurol. 2003;60:753–759. doi: 10.1001/archneur.60.5.753. [DOI] [PubMed] [Google Scholar]
- 31.Ramasubbu R, Patten SB. Effect of depression on stroke morbidity and mortality. Can J Psychiatry. 2003;48:250–257. doi: 10.1177/070674370304800409. [DOI] [PubMed] [Google Scholar]
- 32.Hesdorffer DC, Hauser WA, Annegers JF, Cascino G. Major depression is a risk factor for seizures in older adults. Ann Neurol. 2000;47:246–249. doi: 10.1002/1531-8249(200002)47:2<246::AID-ANA17>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 33.Nouwen A, Lloyd CE, Pouwer F. Depression and type 2 diabetes over the lifespan: A meta-analysis. Diabetes Care. 2009;32:e56–e57. doi: 10.2337/dc09-0027. Response to Mezuk et al. [DOI] [PubMed] [Google Scholar]
- 34.Penninx BW, Guralnik JM, Pahor M, Ferrucci L, Cerhan JR, Wallace RB, Havlik RJ. Chronically depressed mood and cancer risk in older persons. J Natl Cancer Inst. 1998;90:1888–1893. doi: 10.1093/jnci/90.24.1888. [DOI] [PubMed] [Google Scholar]
- 35.Keller MB, Hirschfeld RM, Demyttenaere K, Baldwin DS. Optimizing outcomes in depression: Focus on antidepressant compliance. Int Clin Psychopharmacol. 2002;17:265–271. doi: 10.1097/00004850-200211000-00001. [DOI] [PubMed] [Google Scholar]
- 36.Bouwman V, Adriaanse MC, van't Riet E, Snoek FJ, Dekker JM, Nijpels G. Depression, anxiety and glucose metabolism in the general dutch population: The New Hoorn Study. PLoS One. 2010;5:e9971. doi: 10.1371/journal.pone.0009971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shelton RC, Miller AH. Eating ourselves to death (and despair): The contribution of adiposity and inflammation to depression. Prog Neurobiol. 2010;91:275–299. doi: 10.1016/j.pneurobio.2010.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shirey K, Manyara SM, Atwoli L, Tomlin R, Gakinya B, Cheng S, Kamano J, Laktabai J, Pastakia S. Symptoms of depression among patients attending a diabetes care clinic in rural western Kenya. J Clin Transl Endocrinol. 2015;2:51–54. doi: 10.1016/j.jcte.2015.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Häfner S, Baumert J, Emeny RT, Lacruz ME, Thorand B, Herder C, Koenig W, Rupprecht R, Ladwig KH. Sleep disturbances and depressed mood: A harmful combination associated with increased leptin levels in women with normal weight. Biol Psychol. 2012;89:163–169. doi: 10.1016/j.biopsycho.2011.10.005. [DOI] [PubMed] [Google Scholar]
- 40.Becker C, Zeau B, Rivat C, Blugeot A, Hamon M, Benoliel JJ. Repeated social defeat-induced depression-like behavioral and biological alterations in rats: Involvement of cholecystokinin. Mol Psychiatry. 2008;13:1079–1092. doi: 10.1038/sj.mp.4002097. [DOI] [PubMed] [Google Scholar]
- 41.Kozlovsky N, Matar MA, Kaplan Z, Zohar J, Cohen H. The role of the galaninergic system in modulating stress-related responses in an animal model of posttraumatic stress disorder. Biol Psychiatry. 2009;65:383–391. doi: 10.1016/j.biopsych.2008.10.034. [DOI] [PubMed] [Google Scholar]
- 42.Cline BH, Steinbusch HW, Malin D, Revishchin AV, Pavlova GV, Cespuglio R, Strekalova T. The neuronal insulin sensitizer dicholine succinate reduces stress-induced depressive traits and memory deficit: Possible role of insulin-like growth factor 2. BMC Neurosci. 2012;13:110. doi: 10.1186/1471-2202-13-110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Roesler R, Henriques JA, Schwartsmann G. Gastrin-releasing peptide receptor as a molecular target for psychiatric and neurological disorders. CNS Neurol Disord Drug Targets. 2006;5:197–204. doi: 10.2174/187152706776359673. [DOI] [PubMed] [Google Scholar]
- 44.Musselman DL, Nemeroff CB. Depression and endocrine disorders: Focus on the thyroid and adrenal system. Br J Psychiatry Suppl. 1996:123–128. doi: 10.1192/S0007125000298504. [DOI] [PubMed] [Google Scholar]
- 45.Frey A, Lampert A, Dietz K, Striebich S, Locher C, Fedorenko O, Möhle R, Gallinat J, Lang F, Lang UE. Thyrotropin serum concentrations in healthy volunteers are associated with depression-related personality traits. Neuropsychobiology. 2007;56:123–126. doi: 10.1159/000112954. [DOI] [PubMed] [Google Scholar]
- 46.Bahls SC, de Carvalho GA. The relation between thyroid function and depression: A review. Braz J Psychiatry. 2004;26:41–49. doi: 10.1590/S1516-44462004000100012. (In Portuguese) [DOI] [PubMed] [Google Scholar]
- 47.Cleare AJ, McGregor A, O'Keane V. Neuroendocrine evidence for an association between hypothyroidism, reduced central 5-HT activity and depression. Clin Endocrinol (Oxf) 1995;43:713–719. doi: 10.1111/j.1365-2265.1995.tb00540.x. [DOI] [PubMed] [Google Scholar]
- 48.Holtzheimer PE, III, Nemeroff CB. Future prospects in depression research. Dialogues Clin Neurosci. 2006;8:175–189. doi: 10.31887/DCNS.2006.8.2/pholtzheimer. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Banki CM, Bissette G, Arato M, O'Connor L, Nemeroff CB. CSF corticotropin-releasing factor-like immunoreactivity in depression and schizophrenia. Am J Psychiatry. 1987;144:873–877. doi: 10.1176/ajp.144.7.873. [DOI] [PubMed] [Google Scholar]
- 50.Lippi G, Montagnana M, Favaloro EJ, Franchini M. Mental depression and cardiovascular disease: A multifaceted, bidirectional association. Semin Thromb Hemost. 2009;35:325–336. doi: 10.1055/s-0029-1222611. [DOI] [PubMed] [Google Scholar]
- 51.Tiemeier H, van Dijck W, Hofman A, Witteman JC, Stijnen T, Breteler MM. Relationship between atherosclerosis and late-life depression: The rotterdam study. Arch Gen Psychiatry. 2004;61:369–376. doi: 10.1001/archpsyc.61.4.369. [DOI] [PubMed] [Google Scholar]
- 52.Nemeroff CB, Goldschmidt-Clermont PJ. Heartache and heartbreak-the link between depression and cardiovascular disease. Nat Rev Cardiol. 2012;9:526–539. doi: 10.1038/nrcardio.2012.91. [DOI] [PubMed] [Google Scholar]
- 53.Geiser F, Meier C, Wegener I, Imbierowicz K, Conrad R, Liedtke R, Oldenburg J, Harbrecht U. Association between anxiety and factors of coagulation and fibrinolysis. Psychother Psychosom. 2008;77:377–383. doi: 10.1159/000151518. [DOI] [PubMed] [Google Scholar]
- 54.Nemeroff CB, Musselman DL. Are platelets the link between depression and ischemic heart disease? Am Heart J. 2000;140((Suppl 4)):S57–S62. doi: 10.1067/mhj.2000.109978. [DOI] [PubMed] [Google Scholar]
- 55.Stafford L, Berk M. The use of statins after a cardiac intervention is associated with reduced risk of subsequent depression: Proof of concept for the inflammatory and oxidative hypotheses of depression? J Clin Psychiatry. 2011;72:1229–1235. doi: 10.4088/JCP.09m05825blu. [DOI] [PubMed] [Google Scholar]
- 56.Jin K, Zhu Y, Sun Y, Mao XO, Xie L, Greenberg DA. Vascular endothelial growth factor (VEGF) stimulates neurogenesis in vitro and in vivo. Proc Natl Acad Sci USA. 2002;99:11946–11950. doi: 10.1073/pnas.182296499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Warner-Schmidt JL, Duman RS. VEGF is an essential mediator of the neurogenic and behavioral actions of antidepressants. Proc Natl Acad Sci USA. 2007;104:4647–4652. doi: 10.1073/pnas.0610282104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kahl KG, Bens S, Ziegler K, Rudolf S, Kordon A, Dibbelt L, Schweiger U. Angiogenic factors in patients with current major depressive disorder comorbid with borderline personality disorder. Psychoneuroendocrinology. 2009;34:353–357. doi: 10.1016/j.psyneuen.2008.09.016. [DOI] [PubMed] [Google Scholar]
- 59.Nakataki M, Iga J, Numata S, Yoshimoto E, Kodera K, Watanabe SY, Song H, Ueno S, Ohmori T. Gene expression and association analysis of the epithelial membrane protein 1 gene in major depressive disorder in the Japanese population. Neurosci Lett. 2011;489:126–130. doi: 10.1016/j.neulet.2010.12.001. [DOI] [PubMed] [Google Scholar]
- 60.Dantzer R. Cytokine, sickness behavior, and depression. Immunol Allergy Clin North Am. 2009;29:247–264. doi: 10.1016/j.iac.2009.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Akhondzadeh S, Jafari S, Raisi F, Nasehi AA, Ghoreishi A, Salehi B, Mohebbi-Rasa S, Raznahan M, Kamalipour A. Clinical trial of adjunctive celecoxib treatment in patients with major depression: A double blind and placebo controlled trial. Depress Anxiety. 2009;26:607–611. doi: 10.1002/da.20589. [DOI] [PubMed] [Google Scholar]
- 62.Müller N, Schwarz MJ, Dehning S, Douhe A, Cerovecki A, Goldstein-Müller B, Spellmann I, Hetzel G, Maino K, Kleindienst N, et al. The cyclooxygenase-2 inhibitor celecoxib has therapeutic effects in major depression: Results of a double-blind, randomized, placebo controlled, add-on pilot study to reboxetine. Mol Psychiatry. 2006;11:680–684. doi: 10.1038/sj.mp.4001805. [DOI] [PubMed] [Google Scholar]
- 63.Nery FG, Monkul ES, Hatch JP, Fonseca M, Zunta-Soares GB, Frey BN, Bowden CL, Soares JC. Celecoxib as an adjunct in the treatment of depressive or mixed episodes of bipolar disorder: A double-blind, randomized, placebo-controlled study. Hum Psychopharmacol. 2008;23:87–94. doi: 10.1002/hup.912. [DOI] [PubMed] [Google Scholar]
- 64.Kumar B, Kuhad A, Chopra K. Neuropsychopharmacological effect of sesamol in unpredictable chronic mild stress model of depression: Behavioral and biochemical evidences. Psychopharmacology (Berl) 2011;214:819–828. doi: 10.1007/s00213-010-2094-2. [DOI] [PubMed] [Google Scholar]
- 65.Lau BW, Ren C, Yang J, Yan SW, Chang RC, Pu M, So KF. Light deprivation induces depression-like behavior and suppresses neurogenesis in diurnal mongolian gerbil (Meriones unguiculatus) Cell Transplant. 2011;20:871–881. doi: 10.3727/096368910X539065. [DOI] [PubMed] [Google Scholar]
- 66.Ljubicić D, Stipcević T, Pivac N, Jakovljević M, Mück-Seler D. The influence of daylight exposure on platelet 5-HT levels in patients with major depression and schizophrenia. J Photochem Photobiol B. 2007;89:63–69. doi: 10.1016/j.jphotobiol.2007.09.002. [DOI] [PubMed] [Google Scholar]
- 67.Ubbenhorst A, Striebich S, Lang F, Lang UE. Exploring the relationship between vitamin D and basic personality traits. Psychopharmacology (Berl) 2011;215:733–737. doi: 10.1007/s00213-011-2175-x. [DOI] [PubMed] [Google Scholar]
- 68.LeBourgeois MK, Carskadon MA, Akacem LD, Simpkin CT, Wright KP, Jr, Achermann P, Jenni OG. Circadian phase and its relationship to nighttime sleep in toddlers. J Biol Rhythms. 2013;28:322–331. doi: 10.1177/0748730413506543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Almeida OP, Marsh K, Alfonso H, Flicker L, Davis TM, Hankey GJ. B-vitamins reduce the long-term risk of depression after stroke: The VITATOPS-DEP trial. Ann Neurol. 2010;68:503–510. doi: 10.1002/ana.22189. [DOI] [PubMed] [Google Scholar]
- 70.Sanhueza C, Ryan L, Foxcroft DR. Diet and the risk of unipolar depression in adults: Systematic review of cohort studies. J Hum Nutr Diet. 2013;26:56–70. doi: 10.1111/j.1365-277X.2012.01283.x. [DOI] [PubMed] [Google Scholar]
- 71.Lazarou C, Kouta C. The role of nurses in the prevention and management of obesity. Br J Nurs. 2010;19:641–647. doi: 10.12968/bjon.2010.19.10.48203. [DOI] [PubMed] [Google Scholar]
- 72.Papakostas GI, Mischoulon D, Shyu I, Alpert JE, Fava M. S-adenosyl methionine (SAMe) augmentation of serotonin reuptake inhibitors for antidepressant nonresponders with major depressive disorder: A double-blind, randomized clinical trial. Am J Psychiatry. 2010;167:942–948. doi: 10.1176/appi.ajp.2009.09081198. [DOI] [PubMed] [Google Scholar]
- 73.O'Shea RD. Roles and regulation of glutamate transporters in the central nervous system. Clin Exp Pharmacol Physiol. 2002;29:1018–1023. doi: 10.1046/j.1440-1681.2002.03770.x. [DOI] [PubMed] [Google Scholar]
- 74.Erecińska M, Silver IA. Metabolism and role of glutamate in mammalian brain. Prog Neurobiol. 1990;35:245–296. doi: 10.1016/0301-0082(90)90013-7. [DOI] [PubMed] [Google Scholar]
- 75.Banasr M, Chowdhury GM, Terwilliger R, Newton SS, Duman RS, Behar KL, Sanacora G. Glial pathology in an animal model of depression: Reversal of stress-induced cellular, metabolic and behavioral deficits by the glutamate-modulating drug riluzole. Mol Psychiatry. 2010;15:501–511. doi: 10.1038/mp.2008.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Gourley SL, Espitia JW, Sanacora G, Taylor JR. Antidepressant-like properties of oral riluzole and utility of incentive disengagement models of depression in mice. Psychopharmacology (Berl) 2012;219:805–814. doi: 10.1007/s00213-011-2403-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Diazgranados N, Ibrahim L, Brutsche NE, Newberg A, Kronstein P, Khalife S, Kammerer WA, Quezado Z, Luckenbaugh DA, Salvadore G, et al. A randomized add-on trial of an N-methyl-D-aspartate antagonist in treatment-resistant bipolar depression. Arch Gen Psychiatry. 2010;67:793–802. doi: 10.1001/archgenpsychiatry.2010.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lauterbach EC. Dextromethorphan as a potential rapid-acting antidepressant. Med Hypotheses. 2011;76:717–719. doi: 10.1016/j.mehy.2011.02.003. [DOI] [PubMed] [Google Scholar]
- 79.Drevets WC, Furey ML. Replication of scopolamine's antidepressant efficacy in major depressive disorder: A randomized, placebo-controlled clinical trial. Biol Psychiatry. 2010;67:432–438. doi: 10.1016/j.biopsych.2009.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Furey ML, Drevets WC. Antidepressant efficacy of the antimuscarinic drug scopolamine: A randomized, placebo-controlled clinical trial. Arch Gen Psychiatry. 2006;63:1121–1129. doi: 10.1001/archpsyc.63.10.1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Sawada T, Yokoi K. Effect of zinc supplementation on mood states in young women: A pilot study. Eur J Clin Nutr. 2010;64:331–333. doi: 10.1038/ejcn.2009.158. [DOI] [PubMed] [Google Scholar]
- 82.Barragán-Rodríguez L, Rodríguez-Morán M, Guerrero-Romero F. Efficacy and safety of oral magnesium supplementation in the treatment of depression in the elderly with type 2 diabetes: A randomized, equivalent trial. Magnes Res. 2008;21:218–223. [PubMed] [Google Scholar]
- 83.Schmidt HD, Duman RS. The role of neurotrophic factors in adult hippocampal neurogenesis, antidepressant treatments and animal models of depressive-like behavior. Behav Pharmacol. 2007;18:391–418. doi: 10.1097/FBP.0b013e3282ee2aa8. [DOI] [PubMed] [Google Scholar]
- 84.Schmidt HD, Shelton RC, Duman RS. Functional biomarkers of depression: Diagnosis, treatment, and pathophysiology. Neuropsychopharmacology. 2011;36:2375–2394. doi: 10.1038/npp.2011.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Pittenger C, Duman RS. Stress, depression, and neuroplasticity: A convergence of mechanisms. Neuropsychopharmacology. 2008;33:88–109. doi: 10.1038/sj.npp.1301574. [DOI] [PubMed] [Google Scholar]
- 86.Schinder AF, Poo M. The neurotrophin hypothesis for synaptic plasticity. Trends Neurosci. 2000;23:639–645. doi: 10.1016/S0166-2236(00)01672-6. [DOI] [PubMed] [Google Scholar]
- 87.Aydemir O, Deveci A, Taneli F. The effect of chronic antidepressant treatment on serum brain-derived neurotrophic factor levels in depressed patients: A preliminary study. Prog Neuropsychopharmacol Biol Psychiatry. 2005;29:261–265. doi: 10.1016/j.pnpbp.2004.11.009. [DOI] [PubMed] [Google Scholar]
- 88.Sen S, Duman R, Sanacora G. Serum brain-derived neurotrophic factor, depression, and antidepressant medications: Meta-analyses and implications. Biol Psychiatry. 2008;64:527–532. doi: 10.1016/j.biopsych.2008.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Cattaneo A, Sesta A, Calabrese F, Nielsen G, Riva MA, Gennarelli M. The expression of VGF is reduced in leukocytes of depressed patients and it is restored by effective antidepressant treatment. Neuropsychopharmacology. 2010;35:1423–1428. doi: 10.1038/npp.2010.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Hunsberger JG, Newton SS, Bennett AH, Duman CH, Russell DS, Salton SR, Duman RS. Antidepressant actions of the exercise-regulated gene VGF. Nat Med. 2007;13:1476–1482. doi: 10.1038/nm1669. [DOI] [PubMed] [Google Scholar]
- 91.Hellweg R, Lang UE, Nagel M, Baumgartner A. Subchronic treatment with lithium increases nerve growth factor content in distinct brain regions of adult rats. Mol Psychiatry. 2002;7:604–608. doi: 10.1038/sj.mp.4001042. [DOI] [PubMed] [Google Scholar]
- 92.von Richthofen S, Lang UE, Hellweg R. Effects of different kinds of acute stress on nerve growth factor content in rat brain. Brain Res. 2003;987:207–213. doi: 10.1016/S0006-8993(03)03338-9. [DOI] [PubMed] [Google Scholar]
- 93.Rosa AR, Frey BN, Andreazza AC, Ceresér KM, Cunha AB, Quevedo J, Santin A, Gottfried C, Gonçalves CA, Vieta E, Kapczinski F. Increased serum glial cell line-derived neurotrophic factor immunocontent during manic and depressive episodes in individuals with bipolar disorder. Neurosci Lett. 2006;407:146–150. doi: 10.1016/j.neulet.2006.08.026. [DOI] [PubMed] [Google Scholar]
- 94.Scapagnini G, Davinelli S, Drago F, De Lorenzo A, Oriani G. Antioxidants as antidepressants: Fact or fiction? CNS Drugs. 2012;26:477–490. doi: 10.2165/11633190-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 95.Leonard B, Maes M. Mechanistic explanations how cell-mediated immune activation, inflammation and oxidative and nitrosative stress pathways and their sequels and concomitants play a role in the pathophysiology of unipolar depression. Neurosci Biobehav Rev. 2012;36:764–785. doi: 10.1016/j.neubiorev.2011.12.005. [DOI] [PubMed] [Google Scholar]
- 96.Lopresti AL, Hood SD, Drummond PD. A review of lifestyle factors that contribute to important pathways associated with major depression: Diet, sleep and exercise. J Affect Disord. 2013;148:12–27. doi: 10.1016/j.jad.2013.01.014. [DOI] [PubMed] [Google Scholar]
- 97.Galecki P. Oxidative stress in depression. In: Laher I, editor. Systems Biology of Free Radicals and Antioxidants. Springer; Berlin: 2014. pp. 2369–2395. [DOI] [Google Scholar]
- 98.Lukic I, Mitic M, Djordjevic J, Tatalovic N, Bozovic N, Soldatovic I, Mihaljevic M, Pavlovic Z, Radojcic MB, Maric NP, Adzic M. Lymphocyte levels of redox-sensitive transcription factors and antioxidative enzymes as indicators of pro-oxidative state in depressive patients. Neuropsychobiology. 2014;70:1–9. doi: 10.1159/000362841. [DOI] [PubMed] [Google Scholar]
- 99.Maes M, Galecki P, Chang YS, Berk M. A review on the oxidative and nitrosative stress (O&NS) pathways in major depression and their possible contribution to the (neuro)degenerative processes in that illness. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35:676–692. doi: 10.1016/j.pnpbp.2010.05.004. [DOI] [PubMed] [Google Scholar]
- 100.Halliwell B. Free radicals and antioxidants-quo vadis? Trends Pharmacol Sci. 2011;32:125–130. doi: 10.1016/j.tips.2010.12.002. [DOI] [PubMed] [Google Scholar]
- 101.Sperner-Unterweger B, Kohl C, Fuchs D. Immune changes and neurotransmitters: Possible interactions in depression? Prog Neuropsychopharmacol Biol Psychiatry. 2014;48:268–276. doi: 10.1016/j.pnpbp.2012.10.006. [DOI] [PubMed] [Google Scholar]
- 102.Kudlow P, Cha DS, Carvalho AF, McIntyre RS. Nitric oxide and major depressive disorder: Pathophysiology and treatment implications. Curr Mol Med. 2016;16:206–215. doi: 10.2174/1566524016666160126144722. [DOI] [PubMed] [Google Scholar]
- 103.Steinert JR, Chernova T, Forsythe ID. Nitric oxide signaling in brain function, dysfunction, and dementia. Neuroscientist. 2010;16:435–452. doi: 10.1177/1073858410366481. [DOI] [PubMed] [Google Scholar]
- 104.Tobe EH. Mitochondrial dysfunction, oxidative stress, and major depressive disorder. Neuropsychiatr Dis Treat. 2013;9:567–573. doi: 10.2147/NDT.S44282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Gandal MJ, Haney JR, Parikshak NN, Leppa V, Ramaswami G, Hartl C, Schork AJ, Appadurai V, Buil A, Werge TM, et al. Shared molecular neuropathology across major psychiatric disorders parallels polygenic overlap. Focus (Am Psychiatr Publ) 2019;17:66–72. doi: 10.1176/appi.focus.17103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Gandal MJ, Haney JR, Parikshak NN, Leppa V, Ramaswami G, Hartl C, Schork AJ, Appadurai V, Buil A, Werge TM, et al. Shared molecular neuropathology across major psychiatric disorders parallels polygenic overlap. Science. 2018;359:693–697. doi: 10.1126/science.aad6469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Dean B. Understanding the role of inflammatory-related pathways in the pathophysiology and treatment of psychiatric disorders: Evidence from human peripheral studies and CNS studies. Int J Neuropsychopharmacol. 2011;14:997–1012. doi: 10.1017/S1461145710001410. [DOI] [PubMed] [Google Scholar]
- 108.Akula N, Barb J, Jiang X, Wendland JR, Choi KH, Sen SK, Hou L, Chen DT, Laje G, Johnson K, et al. RNA-sequencing of the brain transcriptome implicates dysregulation of neuroplasticity, circadian rhythms and GTPase binding in bipolar disorder. Mol Psychiatry. 2014;19:1179–1185. doi: 10.1038/mp.2013.170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Zhao Z, Xu J, Chen J, Kim S, Reimers M, Bacanu SA, Yu H, Liu C, Sun J, Wang Q, et al. Transcriptome sequencing and genome-wide association analyses reveal lysosomal function and actin cytoskeleton remodeling in schizophrenia and bipolar disorder. Mol Psychiatry. 2015;20:563–572. doi: 10.1038/mp.2014.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Bernard R, Kerman IA, Thompson RC, Jones EG, Bunney WE, Barchas JD, Schatzberg AF, Myers RM, Akil H, Watson SJ. Altered expression of glutamate signaling, growth factor, and glia genes in the locus coeruleus of patients with major depression. Mol Psychiatry. 2011;16:634–646. doi: 10.1038/mp.2010.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Feyissa AM, Chandran A, Stockmeier CA, Karolewicz B. Reduced levels of NR2A and NR2B subunits of NMDA receptor and PSD-95 in the prefrontal cortex in major depression. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33:70–75. doi: 10.1016/j.pnpbp.2008.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Kang HJ, Adams DH, Simen A, Simen BB, Rajkowska G, Stockmeier CA, Overholser JC, Meltzer HY, Jurjus GJ, Konick LC, et al. Gene expression profiling in postmortem prefrontal cortex of major depressive disorder. J Neurosci. 2007;27:13329–13340. doi: 10.1523/JNEUROSCI.4083-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Labonté B, Engmann O, Purushothaman I, Menard C, Wang J, Tan C, Scarpa JR, Moy G, Loh YE, Cahill M, et al. Sex-specific transcriptional signatures in human depression. Nat Med. 2017;23:1102–1111. doi: 10.1038/nm.4386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Ando V, Claridge G, Clark K. Psychotic traits in comedians. Br J Psychiatry. 2014;204:341–345. doi: 10.1192/bjp.bp.113.134569. [DOI] [PubMed] [Google Scholar]
- 115.Heyman E, Gamelin FX, Goekint M, Piscitelli F, Roelands B, Leclair E, Di Marzo V, Meeusen R. Intense exercise increases circulating endocannabinoid and BDNF levels in humans-possible implications for reward and depression. Psychoneuroendocrinology. 2012;37:844–851. doi: 10.1016/j.psyneuen.2011.09.017. [DOI] [PubMed] [Google Scholar]
- 116.Blanke U, te Wildt B. Depressive disorders in the context of biographical and historical changes-experiences within a psychiatric outpatient service. Psychiatr Prax. 2007;34((Suppl 3)):S269–S272. doi: 10.1055/s-2007-970965. (In German) [DOI] [PubMed] [Google Scholar]
- 117.Wang SJ, Chen MY. The effects of sunlight exposure therapy on the improvement of depression and quality of life in post-stroke patients: A RCT study. Heliyon. 2020;6:e04379. doi: 10.1016/j.heliyon.2020.e04379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Bouron A, Lorrain E. Cellular and molecular effects of the antidepressant hyperforin on brain cells: Review of the literature. Encephale. 2014;40:108–113. doi: 10.1016/j.encep.2013.03.004. (In French) [DOI] [PubMed] [Google Scholar]
- 119.Owens MJ, Morgan WN, Plott SJ, Nemeroff CB. Neurotransmitter receptor and transporter binding profile of antidepressants and their metabolites. J Pharmacol Exp Ther. 1997;283:1305–1322. [PubMed] [Google Scholar]
- 120.Menkes D, Bosanac P, Castle D. MAOIs-does the evidence warrant their resurrection? Australas Psychiatry. 2016;24:371–373. doi: 10.1177/1039856216634824. [DOI] [PubMed] [Google Scholar]
- 121.Dale E, Bang-Andersen B, Sánchez C. Emerging mechanisms and treatments for depression beyond SSRIs and SNRIs. Biochem Pharmacol. 2015;95:81–97. doi: 10.1016/j.bcp.2015.03.011. [DOI] [PubMed] [Google Scholar]
- 122.Sharma H, Santra S, Dutta A. Triple reuptake inhibitors as potential next-generation antidepressants: A new hope? Future Med Chem. 2015;7:2385–2406. doi: 10.4155/fmc.15.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Cipriani A, Koesters M, Furukawa TA, Nosè M, Purgato M, Omori IM, Trespidi C, Barbui C. Duloxetine versus other anti-depressive agents for depression. Cochrane Database Syst Rev. 2012;10:CD006533. doi: 10.1002/14651858.CD006533.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Parker L, Huelin R, Khankhel Z, Wasiak R, Mould J. A systematic review of pharmacoeconomic studies for pregabalin. Pain Pract. 2015;15:82–94. doi: 10.1111/papr.12193. [DOI] [PubMed] [Google Scholar]
- 125.Wu Y, Huang Y, Song M, Zhang Z, Liang Z, Deng X. Anticonvulsive activity of duloxetine: A new choice for the epileptic patients with depression. Pak J Pharm Sci. 2019;32:997–1003. [PubMed] [Google Scholar]
- 126.Kumar R, Sinha VR, Dahiya L, Sarwal A. Transdermal delivery of duloxetine-sulfobutylether-β-cyclodextrin complex for effective management of depression. Int J Pharm. 2021;594:120129. doi: 10.1016/j.ijpharm.2020.120129. [DOI] [PubMed] [Google Scholar]
- 127.Ashby CR, Jr, Kehne JH, Bartoszyk GD, Renda MJ, Athanasiou M, Pierz KA, Seyfried CA. Electrophysiological evidence for rapid 5-HT1A autoreceptor inhibition by vilazodone, a 5-HT1A receptor partial agonist and 5-HT reuptake inhibitor. Eur J Pharmacol. 2013;714:359–365. doi: 10.1016/j.ejphar.2013.07.014. [DOI] [PubMed] [Google Scholar]
- 128.Bathla M, Anjum S. A 12-week prospective randomized controlled comparative trial of vilazodone and sertraline in Indian patients with depression. Indian J Pharmacol. 2020;52:10–15. doi: 10.4103/ijp.IJP_618_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Hashimoto K, Malchow B, Falkai P, Schmitt A. Glutamate modulators as potential therapeutic drugs in schizophrenia and affective disorders. Eur Arch Psychiatry Clin Neurosci. 2013;263:367–377. doi: 10.1007/s00406-013-0399-y. [DOI] [PubMed] [Google Scholar]
- 130.Burgdorf J, Zhang XL, Weiss C, Matthews E, Disterhoft JF, Stanton PK, Moskal JR. The N-methyl-D-aspartate receptor modulator GLYX-13 enhances learning and memory, in young adult and learning impaired aging rats. Neurobiol Aging. 2011;32:698–706. doi: 10.1016/j.neurobiolaging.2009.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Mørk A, Montezinho LP, Miller S, Trippodi-Murphy C, Plath N, Li Y, Gulinello M, Sanchez C. Vortioxetine (Lu AA21004), a novel multimodal antidepressant, enhances memory in rats. Pharmacol Biochem Behav. 2013;105:41–50. doi: 10.1016/j.pbb.2013.01.019. [DOI] [PubMed] [Google Scholar]
- 132.Katona C, Hansen T, Olsen CK. A randomized, double-blind, placebo-controlled, duloxetine-referenced, fixed-dose study comparing the efficacy and safety of Lu AA21004 in elderly patients with major depressive disorder. Int Clin Psychopharmacol. 2012;27:215–223. doi: 10.1097/YIC.0b013e3283542457. [DOI] [PubMed] [Google Scholar]
- 133.Xiao L. Agomelatine for postpartum depression and breastfeeding. Ther Adv Psychopharmacol. 2021;11:20451253211022172. doi: 10.1177/20451253211022172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.de Bodinat C, Guardiola-Lemaitre B, Mocaër E, Renard P, Muñoz C, Millan MJ. Agomelatine, the first melatonergic antidepressant: Discovery, characterization and development. Nat Rev Drug Discov. 2010;9:628–642. doi: 10.1038/nrd3274. [DOI] [PubMed] [Google Scholar]
- 135.Gumuslu E, Mutlu O, Sunnetci D, Ulak G, Celikyurt IK, Cine N, Akar F, Savlı H, Erden F. The antidepressant agomelatine improves memory deterioration and upregulates CREB and BDNF gene expression levels in unpredictable chronic mild stress (UCMS)-exposed mice. Drug Target Insights. 2014;8:11–21. doi: 10.4137/DTI.S13870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Diamond PR, Farmery AD, Atkinson S, Haldar J, Williams N, Cowen PJ, Geddes JR, McShane R. Ketamine infusions for treatment resistant depression: A series of 28 patients treated weekly or twice weekly in an ECT clinic. J Psychopharmacol. 2014;28:536–544. doi: 10.1177/0269881114527361. [DOI] [PubMed] [Google Scholar]
- 137.Aguilar-Valles A, De Gregorio D, Matta-Camacho E, Eslamizade MJ, Khlaifia A, Skaleka A, Lopez-Canul M, Torres-Berrio A, Bermudez S, Rurak GM, et al. Antidepressant actions of ketamine engage cell-specific translation via eIF4E. Nature. 2021;590:315–319. doi: 10.1038/s41586-020-03047-0. [DOI] [PubMed] [Google Scholar]
- 138.Corriger A, Pickering G. Ketamine and depression: A narrative review. Drug Des Devel Ther. 2019;13:3051–3067. doi: 10.2147/DDDT.S221437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Palmer EC, Binns LN, Carey H. Levomilnacipran: A new serotonin-norepinephrine reuptake inhibitor for the treatment of major depressive disorder. Ann Pharmacother. 2014;48:1030–1039. doi: 10.1177/1060028014535074. [DOI] [PubMed] [Google Scholar]
- 140.Krause-Sorio B, Kilpatrick L, Siddarth P, Ercoli L, Laird KT, Aguilar-Faustino Y, Milillo MM, Narr KL, Lavretsky H. Cortical thickness increases with levomilnacipran treatment in a pilot randomised double-blind placebo-controlled trial in late-life depression. Psychogeriatrics. 2020;20:140–148. doi: 10.1111/psyg.12475. [DOI] [PubMed] [Google Scholar]
- 141.Anthonis E, Sienaert P. Farmacotherapeutic treatment of psychotic depression: A review. Tijdschr Psychiatr. 2021;63:358–365. (In Dutch) [PubMed] [Google Scholar]
- 142.Greer TL, Grannemann BD, Chansard M, Karim AI, Trivedi MH. Dose-dependent changes in cognitive function with exercise augmentation for major depression: Results from the TREAD study. Eur Neuropsychopharmacol. 2015;25:248–256. doi: 10.1016/j.euroneuro.2014.10.001. [DOI] [PubMed] [Google Scholar]
- 143.Reutfors J, Brenner P, Brody B, Wray H, Andersen M, Brandt L. A post-authorization safety study of quetiapine as antidepressant treatment in Sweden: Nested case-control analyses of select outcomes. Drug Saf. 2020;43:135–145. doi: 10.1007/s40264-019-00889-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Torales J, O'Higgins M, Castaldelli-Maia JM, Ventriglio A. The outbreak of COVID-19 coronavirus and its impact on global mental health. Int J Soc Psychiatry. 2020;66:317–320. doi: 10.1177/0020764020915212. [DOI] [PubMed] [Google Scholar]
- 145.Sensoy B, Gunes A, Ari S. Anxiety and depression levels in Covid-19 disease and their relation to hypertension. Clin Exp Hypertens. 2021;43:237–241. doi: 10.1080/10641963.2020.1847132. [DOI] [PubMed] [Google Scholar]
- 146.Lai J, Ma S, Wang Y, Cai Z, Hu J, Wei N, Wu J, Du H, Chen T, Li R, et al. Factors associated with mental health outcomes among health care workers exposed to coronavirus disease 2019. JAMA Netw Open. 2020;3:e203976. doi: 10.1001/jamanetworkopen.2020.3976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Mahalakshmi AM, Ray B, Tuladhar S, Bhat A, Paneyala S, Patteswari D, Sakharkar MK, Hamdan H, Ojcius DM, Bolla SR, et al. Does COVID-19 contribute to development of neurological disease? Immun Inflamm Dis. 2021;9:48–58. doi: 10.1002/iid3.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Combe B, Balsa A, Sarzi-Puttini P, Tony HP, de la Torre I, Rogai V, Durand F, Witt S, Zhong J, Dougados M. Efficacy and safety data based on historical or pre-existing conditions at baseline for patients with active rheumatoid arthritis who were treated with baricitinib. Ann Rheum Dis. 2019;78:1135–1138. doi: 10.1136/annrheumdis-2018-214261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Lipworth B, Kuo CR, Chan R. Emerging pharmacotherapy for COVID-19. J R Coll Physicians Edinb. 2020;50:133–137. doi: 10.4997/JRCPE.2020.210. [DOI] [PubMed] [Google Scholar]
- 150.Reder AT, Oger JF, Kappos L, O'Connor P, Rametta M. Short-term and long-term safety and tolerability of interferon β-1b in multiple sclerosis. Mult Scler Relat Disord. 2014;3:294–302. doi: 10.1016/j.msard.2013.11.005. [DOI] [PubMed] [Google Scholar]
- 151.Theoharides TC, Conti P. Dexamethasone for COVID-19? Not so fast. J Biol Regul Homeost Agents. 2020;34:1241–1243. doi: 10.23812/20-EDITORIAL_1-5. [DOI] [PubMed] [Google Scholar]
- 152.Menke A, Arloth J, Best J, Namendorf C, Gerlach T, Czamara D, Lucae S, Dunlop BW, Crowe TM, Garlow SJ, et al. Time-dependent effects of dexamethasone plasma concentrations on glucocorticoid receptor challenge tests. Psychoneuroendocrinology. 2016;69:161–171. doi: 10.1016/j.psyneuen.2016.04.003. [DOI] [PubMed] [Google Scholar]
- 153.Shader RI. COVID-19 and depression. Clin Ther. 2020;42:962–963. doi: 10.1016/j.clinthera.2020.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Armitage R, Nellums LB. COVID-19 and the consequences of isolating the elderly. Lancet Public Health. 2020;5:e256. doi: 10.1016/S2468-2667(20)30061-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Kumar S, Veldhuis A, Malhotra T. Neuropsychiatric and cognitive sequelae of COVID-19. Front Psychol. 2021;12:577529. doi: 10.3389/fpsyg.2021.577529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Rogers JP, Chesney E, Oliver D, Pollak TA, McGuire P, Fusar-Poli P, Zandi MS, Lewis G, David AS. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: A systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry. 2020;7:611–627. doi: 10.1016/S2215-0366(20)30203-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Liu J, Li S, Liu J, Liang B, Wang X, Wang H, Li W, Tong Q, Yi J, Zhao L, et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. doi: 10.1016/j.ebiom.2020.102763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Tsamakis K, Mueller C, Tsirigotis P, Tsiptsios D, Tsamakis C, Charakopoulos E, Charalampous C, Spandidos DA, Douzenis A, Papageorgiou C, et al. Depression following graft-versus-host disease in a patient with acute lymphoblastic leukaemia: A case report. Mol Clin Oncol. 2020;12:208–211. doi: 10.3892/mco.2019.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Cavalli G, De Luca G, Campochiaro C, Della-Torre E, Ripa M, Canetti D, Oltolini C, Castiglioni B, Tassan Din C, Boffini N, et al. Interleukin-1 blockade with high-dose anakinra in patients with COVID-19, acute respiratory distress syndrome, and hyperinflammation: A retrospective cohort study. Lancet Rheumatol. 2020;2:e325–e331. doi: 10.1016/S2665-9913(20)30127-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Campochiaro C, Della-Torre E, Cavalli G, De Luca G, Ripa M, Boffini N, Tomelleri A, Baldissera E, Rovere-Querini P, Ruggeri A, et al. Efficacy and safety of tocilizumab in severe COVID-19 patients: A single-centre retrospective cohort study. Eur J Intern Med. 2020;76:43–49. doi: 10.1016/j.ejim.2020.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Gonda X, Pompili M, Serafini G, Carvalho AF, Rihmer Z, Dome P. The role of cognitive dysfunction in the symptoms and remission from depression. Ann Gen Psychiatry. 2015;14:27. doi: 10.1186/s12991-015-0068-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Martin V, Allaïli N, Euvrard M, Marday T, Riffaud A, Franc B, Mocaër E, Gabriel C, Fossati P, Lehericy S, Lanfumey L. Effect of agomelatine on memory deficits and hippocampal gene expression induced by chronic social defeat stress in mice. Sci Rep. 2017;8:45907. doi: 10.1038/srep45907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Yamada M, Yasuhara H. Clinical pharmacology of MAO inhibitors: Safety and future. Neurotoxicology. 2004;25:215–221. doi: 10.1016/S0161-813X(03)00097-4. [DOI] [PubMed] [Google Scholar]
- 164.Andrade C. Selective serotonin reuptake inhibitor drug interactions in patients receiving statins. J Clin Psychiatry. 2014;75:e95–e99. doi: 10.4088/JCP.13f08941. [DOI] [PubMed] [Google Scholar]
- 165.Voican CS, Corruble E, Naveau S, Perlemuter G. Antidepressant-induced liver injury: A review for clinicians. Am J Psychiatry. 2014;171:404–415. doi: 10.1176/appi.ajp.2013.13050709. [DOI] [PubMed] [Google Scholar]
- 166.Nelson JC. A review of the efficacy of serotonergic and noradrenergic reuptake inhibitors for treatment of major depression. Biol Psychiatry. 1999;46:1301–1308. doi: 10.1016/S0006-3223(99)00173-0. [DOI] [PubMed] [Google Scholar]
- 167.Fagiolini A, Comandini A, Catena Dell'Osso M, Kasper S. Rediscovering trazodone for the treatment of major depressive disorder. CNS Drugs. 2012;26:1033–1049. doi: 10.1007/s40263-012-0010-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Jensen NH, Rodriguiz RM, Caron MG, Wetsel WC, Rothman RB, Roth BL. N-desalkylquetiapine, a potent norepinephrine reuptake inhibitor and partial 5-HT1A agonist, as a putative mediator of quetiapine's antidepressant activity. Neuropsychopharmacology. 2008;33:2303–2312. doi: 10.1038/sj.npp.1301646. [DOI] [PubMed] [Google Scholar]
- 169.Ketter TA, Jenkins JB, Schroeder DH, Pazzaglia PJ, Marangell LB, George MS, Callahan AM, Hinton ML, Chao J, Post RM. Carbamazepine but not valproate induces bupropion metabolism. J Clin Psychopharmacol. 1995;15:327–333. doi: 10.1097/00004714-199510000-00004. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.