Abstract
As of July 31, 2021, SARS-CoV-2 had infected almost 200 million people worldwide. The growing burden of survivorship is substantial in terms of the complexity of long-term health effects and the number of people affected. Persistent symptoms have been reported in patients with both mild and severe acute COVID-19, including those admitted to the intensive care unit (ICU). Early reports on the post-acute sequelae of SARS-CoV-2 infection (PASC) indicate that fatigue, dyspnoea, cough, headache, loss of taste or smell, and cognitive or mental health impairments are among the most common symptoms. These complex, multifactorial impairments across the domains of physical, cognitive, and mental health require a coordinated, multidisciplinary approach to management. Decades of research on the multifaceted needs of and models of care for patients with post-intensive care syndrome provide a framework for the development of PASC clinics to address the immediate needs of both hospitalised and non-hospitalised survivors of COVID-19. Such clinics could also provide a platform for rigorous research into the natural history of PASC and the potential benefits of therapeutic interventions.
Introduction
The burden of COVID-19 survivorship is enormous. More than 190 million people had survived COVID-19 by the end of July 2021.1 Even among previously young, healthy people with COVID-19 who require only outpatient care, nearly 20% do not return to their usual state of health within 3 weeks of infection.2 In a study from China, approximately 75% of survivors who had been hospitalised with COVID-19 had at least one persistent symptom 6 months after acute illness.3 The most commonly reported post-acute symptoms include fatigue, dyspnoea, cough, headache, loss of taste or smell, and cognitive or mental health impairments (eg, anxiety or depression).4, 5, 6 Moreover, non-hospitalised patients with COVID-19 have a higher risk of death and health-care utilisation beyond the first 30 days of illness than do patients without COVID-19.7 In response to the rapidly growing population of survivors of COVID-19 who experience a wide array of complex, multifactorial symptoms—referred to collectively as the post-COVID-19 condition, post-acute sequelae of SARS-CoV-2 infection (PASC), or long COVID—the World Health Organization has advocated a multidisciplinary approach to support patients in their recovery.8
PASC might be specific to SARS-CoV-2 infection or represent a spectrum of symptoms that can also occur within the context of recovery from other acute infections or chronic conditions. For some patients, post-acute sequelae might arise as part of the post-intensive care syndrome (PICS), a collection of health impairments that are common in patients who survive critical illness and management in the intensive care unit (ICU). Although a clear definition of the post-COVID-19 condition is lacking at present, models of care are in development to address the long-term needs of patients with PASC. The overall goal of this Health-care Development paper is to inform the development of these multidisciplinary pathways of care, adapted from the PICS framework. The target audience includes patients, clinicians, educators, researchers, and policy makers. We describe the existing framework of care established to address the multifaceted needs of patients with PICS, and discuss the extension of ICU survivor clinics to meet the needs of patients after SARS-CoV-2 infection. We review key elements of a multidisciplinary programme—including physical medicine and rehabilitation, pharmacy, social work and welfare support, and primary care—and emphasise the importance of achieving equitable provision of care, including implementation in low-income and middle-income countries (LMICs). Finally, we consider the contribution that PASC clinics could make to research on the origins and natural history of PASC and the potential benefits of therapeutic interventions.
Key messages.
-
•
The burden of post-acute sequelae of SARS-CoV-2 infection (PASC) is substantial in terms of the clinical complexity of symptoms and the number of patients affected worldwide
-
•
Rigorous data characterising symptomology are limited at present, but physical, psychological, cognitive, and respiratory domains are key among those affected
-
•
Clinics developed for survivors of critical illness, who experience a wide array of multifactorial symptoms, offer a model to address PASC survivorship for both acute and non-hospitalised patients with COVID-19; understanding of the determinants, clustering, and course of PASC will help to refine care pathways
-
•
A multidisciplinary programme of care should include access to rehabilitation services, social work and welfare support, pharmacy, subspecialty care via direct inclusion or targeted referrals, and structured peer support programmes with trained moderators; coordination with primary care is essential
-
•
Ensuring equitable provision of care, including access to care for under-served populations and the development of care pathways in low-income and middle-income countries, will be integral to international pandemic management
-
•
PASC clinics offer research opportunities that should be harnessed to inform knowledge of survivorship trajectories after SARS-CoV-2 infection, and to improve service innovation and delivery
Post-acute sequelae of SARS-CoV-2 infection
Decades of research conducted prior to 2020 described the physical, cognitive, and mental health impairments experienced by survivors of critical illness, collectively termed PICS.9 Knowledge gained about PICS can be leveraged to understand, describe, diagnose, and treat PASC. Specifically, survivors of acute respiratory distress syndrome (ARDS) experience exercise intolerance (eg, achieve below the predicted distance on a 6-min walk test), and half will not have returned to work at a year after discharge from the ICU.10 Anxiety, depression, and post-traumatic stress disorder (PTSD) symptoms are common and persist for months to years after critical illness.11, 12, 13 Among critical illness survivors of all ages, median cognition scores are consistent with mild cognitive impairment; more than a third will have cognitive testing comparable to moderate traumatic brain injury a year later.14
There are few rigorous, prospective studies on the natural history of PASC, including prevalence and duration, with most studies following patients for fewer than 7 months (Table 1, Table 2 ).4, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 However, existing reports indicate that the burden of impairments is relatively high. An analysis of the US Department of Veterans Affairs electronic health database used a high-dimensional approach to evaluate 6-month outcomes for patients surviving the initial 30 days after COVID-19. Beyond the first 30 days, results showed a higher risk of death for non-hospitalised patients with COVID-19 compared with those without COVID-19 (hazard ratio 1·59, 95% CI 1·46–1·73), as well as for patients hospitalised with COVID-19 compared with seasonal influenza (1·51, 1·30–1·76).7
Table 1.
Systematic reviews of PASC studies
| Number of studies | Number of patients | Age of patients | Studies of hospitalised or non-hospitalised patients | Study locations | Follow-up period* | Proportion with ≥1 persistent symptom at follow-up | PRISMA guidelines24followed? | |
|---|---|---|---|---|---|---|---|---|
| Lopez-Leon et al4 | 15 | 47 910 | Range 17–87 years | 11 hospitalised; 4 mixed | 8 Europe; 4 North America; 1 Australia; 1 China; 1 Egypt | 14–110 days | 80% (95% CI 65–92)† | Yes |
| Michelen et al15‡ | 39 | 10 951 | Range <1 to 93 years (4 studies included children) | 26 hospitalised; 4 non-hospitalised; 9 mixed | 24 Europe; 9 Asia; 3 Middle East; 3 North America | Longest follow-up mean 222 (SD 11) days | NR | Yes |
| Nasserie et al17 | 45 | 9751 | 30 with mean or median <60 years; 14 with mean or median <50 years; 1 NR | 33 hospitalised; 2 non-hospitalised; 10 mixed | 31 Europe; 8 Asia; 5 North America; 1 Middle East | ≥2 months from symptom onset, diagnosis, or hospitalisation | Median 73% (IQR 55–80) | No§ |
| Domingo et al18‡¶ | 27‖ | 7162 | NR | 15 hospitalised; 5 non-hospitalised; 5 mixed; 2 unspecified | 16 Europe; 5 Asia; 3 North America; 3 others | 4–12 weeks; >12 weeks | 83% (95% CI 65–93)**; 56% (34–75)†† | Yes |
| Fernández-de-las-Peñas et al19 | 33 | 24 255 | Mean 48 (SD 17) years | 15 244 hospitalised; 9011 non-hospitalised‡‡ | 17 Europe; 8 North America; 5 Asia; 1 Egypt; 1 Russia; 1 multicountry | Median 21 days to 36 weeks | 63% (95% CI 44–79) at 30 days†; 72% (53–85) at 60 days**; 46% (28–65) at ≥90 days† | Yes |
| Iqbal et al20 | 43§§ (30 in meta-analysis) | 12 974 | NR | 20 hospitalised; 10 non-hospitalised; 13 mixed; 2 unspecified | 35 Europe; 4 Asia; 3 North America; 1 Australia; 1 Middle East; 1 South America | 28 days to 6 months | NR | Yes |
| Malik et al21¶ | 21 | 54 730 | Median 54 years | Mixed | 10 Europe; 4 North America; 2 Africa; 2 China; 2 Middle East; 1 Australia | >1 month in most studies | 68% | Yes |
| Cabrera Martimbianco et al22 | 25 | 5440 | NR | 18 hospitalised; 2 non-hospitalised; 4 mixed; 1 unspecified | 15 Europe; 5 China; 3 North America; 2 Middle East | NR | 5–80% | Yes |
| Sanchez-Ramirez et al23 | 24 | 5323 | Mean 55 (SD 8) years | Mixed | 11 Europe; 7 China; 5 North America; 1 Middle East | Mean 4 months | NR | Yes |
Systematic reviews of studies evaluating general post-COVID-19 symptoms in adult patients. Studies were excluded if they focused exclusively on a subset of symptoms (eg, neurological) or if they did not provide pooled estimates of symptom prevalence. NR=not reported. PASC=post-acute sequelae of SARS-CoV-2 infection.
Time zero differed between studies (eg, time from positive test vs symptom onset vs hospital discharge).
Based on seven studies.
Living systematic review.
Reported using the MOOSE (Meta-Analysis of Observational Studies in Epidemiology) guidelines.25
Preprint.
Studies including adults with laboratory-confirmed SARS-CoV-2 infection.
Based on three studies.
Based on four studies.
Number of hospitalised and non-hospitalised patients.
43 studies given in PRISMA diagram; 45 studies reported.
Table 2.
Pooled estimates of proportion of patients with persistent symptoms from systematic reviews of PASC studies
| Lopez-Leon et al;4pooled prevalence (95% CI) | Michelen et al;15proportion (95% CI)* | Nasserie et al;17median proportion (IQR) | Domingo et al;18proportion (95% CI)† | Fernández-de-las-Peñas et al;19pooled prevalence (95% CI)† | Iqbal et al;20proportion (95% CI)† | Malik et al;21proportion | Cabrera Martimbianco et al;22proportion, range | Sanchez-Ramirez et al;23pooled prevalence (95% CI)‡ | |
|---|---|---|---|---|---|---|---|---|---|
| Systemic | |||||||||
| Fatigue | 58% (42–73) | 31% (24–39) | 40% (31–57) | 47% (27–68) | 35% (25–47) | 48% (23–73) | 54% | 7–64% | 38% (27–49) |
| Sweat or night sweats | 17% (6–30) | 24% (21–27) | .. | .. | .. | .. | .. | .. | .. |
| Weight loss | 12% (7–18) | 21% (8–45) | .. | .. | .. | .. | 8% | .. | .. |
| Fever | 11% (8–15) | 1% (0–5) | 1% (0–3) | 0% (0–1) | .. | .. | .. | 0–20% | .. |
| Pain | 11% (7–18) | .. | .. | .. | .. | .. | .. | 19–66% | .. |
| Decreased functional capacity | .. | .. | .. | .. | .. | .. | .. | 6–50% | 36% (22–49) |
| Neurological | |||||||||
| Headache | 44% (13–78) | 5% (2–10) | .. | 4% (2–10) | 6% (3–12) | 12% (0–44) | <1% | 2–39% | .. |
| Concentration impairment | .. | 26% (21–32) | 22–28%§ | .. | .. | .. | .. | .. | .. |
| Cognitive impairment | .. | 18% (0–98) | 18% (15–22) | .. | .. | .. | .. | 18–57%¶ | .. |
| Memory loss | 16% (0–55) | 18% (5–46) | 28% (19–36) | .. | .. | .. | 8% | .. | .. |
| Ageusia or taste disturbance | 23% (14–33) | 14% (9–20) | 16% (10–24) | 10% (7–15) | 10% (7–15) | 18% (10–28) | 10% | 1–22% | .. |
| Anosmia or smell disturbance | 21% (12–32) | 15% (11–21) | 24% (12–41)‖ | 14% (9–22) | 11% (8–15) | 17% (10–25) | 13% | 0–26% | .. |
| Hearing loss or tinnitus | 15% (10–20) | .. | .. | .. | .. | .. | .. | .. | .. |
| Dermatological | |||||||||
| Hair loss | 25% (17–34) | 14% (5–33) | .. | .. | .. | .. | 23% | 20–29% | .. |
| Skin manifestations, including rash | 12% (7–18) | 3% (1–8) | .. | 3% (2–3) | 3% (2–4) | .. | 4% | 2–20% | .. |
| Respiratory | |||||||||
| Dyspnoea or breathlessness | 24% (14–36) | 25% (18–34) | 36% (28–50) | 22% (12–35) | 26% (9–35) | 39% (16–64) | 24% | 6–61% | 32% (24–40) |
| Cough | 19% (7–34) | 8% (5–14) | 17% (14–25) | 6% (4–8) dry cough; 5% (3–7) productive cough | 9% (5–14) | 11% (7–17) | 17% | 2–59% | 13% (9–17) |
| Cardiac | |||||||||
| Chest pain or discomfort | 16% (10–22) | 6% (3–12) | 13% (11–18) | .. | 9% (7–13) | 17% (5–35) | 10% | 0–89% | 16% (12–21) |
| Palpitations | .. | 10% (6–15) | .. | .. | 10% (6–15) | .. | 9% | 9–62% | .. |
| Musculoskeletal | |||||||||
| Joint pain or arthralgia | 19% (7–34) | 9% (6–15) | .. | 9% (8–11) | 10% (7–15) | .. | 16% | 6–55% | .. |
| Muscle pain or myalgia | .. | 11% (6–20) | .. | 5% (2–12) | 11% (7–18) | .. | 6% | 2–51% | .. |
| Impaired mobility | .. | 14% (5–37) | .. | .. | .. | .. | .. | 7% | .. |
| Gastroenterological | |||||||||
| Nausea or vomiting | .. | 7% (2–24) | .. | .. | 5% (2–10) nausea; 1% (0–2) vomiting | .. | .. | .. | .. |
| Loss of appetite | .. | 18% (4–51) | .. | .. | .. | .. | .. | 6–8% | .. |
| Digestive disorders | .. | .. | .. | .. | .. | .. | 8% | 1–33% | .. |
| Sleep | |||||||||
| Sleep disorder | 11% (3–24) | 18% (10–32) | .. | 25% (24–29) | .. | 44% (8–85) | 26% | 22–53% | .. |
| Psychological | |||||||||
| Anxiety | 13% (3–26) | 19% (9–35) | 22% (10–30) | .. | .. | .. | 17% | 3–25%** | .. |
| Depression | 12% (3–23) | 8% (4–15) | 15% (11–18) | .. | .. | .. | 14% | 3–25%** | .. |
| Quality of life | |||||||||
| Reduced quality of life | .. | 37% (18–60) | .. | .. | .. | .. | .. | .. | 52% (33–71) |
Symptoms were included in the table if at least one systematic review reported a pooled proportion estimate of >10%. Estimates from systematic reviews were included if at least two studies in the review evaluated the symptom. PASC=post-acute sequelae of SARS-CoV-2 infection.
Michelen et al was the only systematic review to report on weakness (proportion 41%, 95% CI 25–59), malaise (33%, 15–57), and decreased sensation (11%, 7–17).
Includes estimates from studies reporting symptoms from approximately >12 weeks after acute illness.
Sanchez-Ramirez et al was the only systematic review to report pooled estimates for abnormal chest CT (59%, 44–73), impaired diffusing capacity (31%, 24–38), and lung restriction as measured by pulmonary function tests (12%, 8–17).
Median not reported; numbers represent range from four studies.
Includes composite of “cognitive/memory/concentration impairment”.
Proportion with persistent anosmia.
Anxiety and depression listed together.
As in PICS, cognitive impairment (so-called brain fog), fatigue, muscle weakness, and shortness of breath are among the commonly reported symptoms after COVID-19.3, 7, 26, 27, 28, 29, 30, 31 Using the 6-min walk test as a measure of physical function, nearly a quarter of survivors of COVID-19 perform below the lower limit of normal functioning 6 months after acute illness.3 In a large international survey of COVID-19 survivors,27 3203 of 3762 respondents (85%) self-reported cognitive impairment; both older and younger respondents reported that such impairment affected their daily lives and ability to return to work. Nearly one in four patients hospitalised with COVID-19 reports substantial depression or anxiety symptoms lasting at least 6 months after acute illness.3 In an observational study from Wuhan, China, patients also commonly self-reported symptoms of PTSD.32 Among hospitalised patients, the most common pulmonary impairment at 6 months after acute COVID-19 is a decreased diffusing capacity of the lung for carbon monoxide (DLCO; about 33% of patients), followed by reduced total lung capacity (approximately 17%).3 Persistent pulmonary impairments probably differ by initial disease severity. Patients with more severe initial COVID-19 pneumonia according to the WHO severity classification had significantly lower percentage of predicted DLCO at 4-month follow-up (mean 73% predicted [SD 18] vs 95% predicted [20]; p=0·003).33
Dyspnoea and fatigue are common among survivors of COVID-19.16, 30, 31, 33, 34, 35, 36 Importantly, there is no consistent association between dyspnoea and abnormalities on chest imaging, pulmonary function test, or initial disease severity.33, 34, 35, 36 Dyspnoea, like fatigue, is a complex, multifactorial symptom.34 Notably, fatigue is common in survivors of ARDS (approximately 70% of patients) at 1-year follow-up.37 Fatigue after acute respiratory failure is not consistently associated with severity of illness or ICU length of stay, but frequently co-occurs with impairments in cognition, mental health, and physical function.37 Addressing complex symptoms such as dyspnoea and fatigue, in both hospitalised and non-hospitalised COVID-19 survivors, requires multidisciplinary interventions, informed by an understanding of risk factors and co-occurring impairments.35
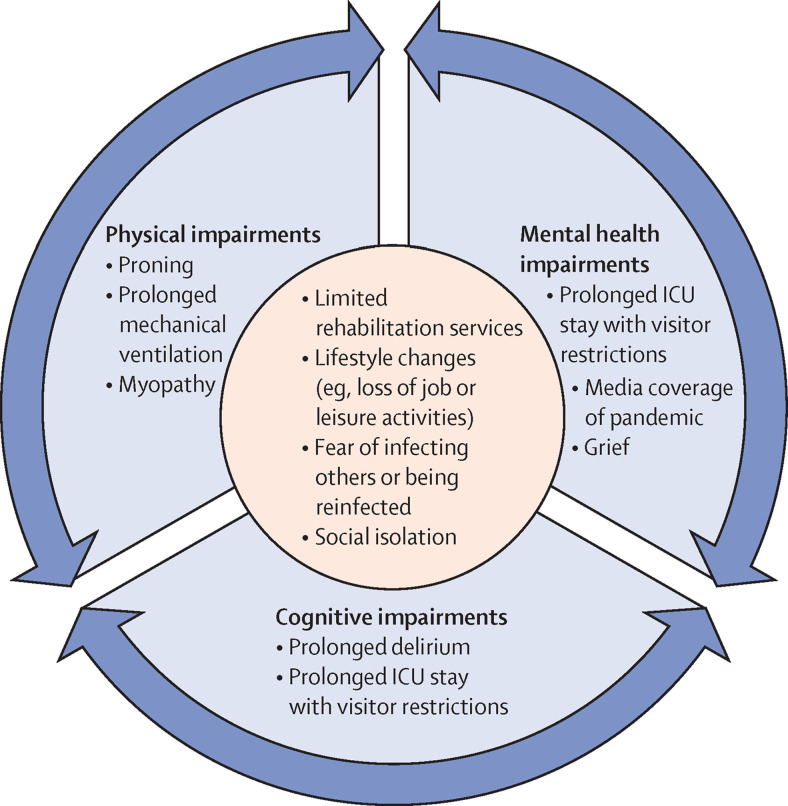
Infection prevention and control measures during the COVID-19 pandemic have the potential to further affect recovery after SARS-CoV-2 infection (figure 1 ). For example, visitor restrictions for hospitalised patients with COVID-19 might increase the risk of delirium and PTSD,38 and social isolation in the outpatient setting can contribute to mental health disturbances among non-hospitalised patients as well. Essential rehabilitation and non-infection-related medical services might be limited or eliminated for patients with COVID-19, both during and after hospitalisation.38, 39
Figure 1.
COVID-19 pandemic-related factors that could exacerbate physical, cognitive, or mental health impairments
The COVID-19 pandemic could further complicate recovery across key domains of mental, physical, and cognitive health, which are intimately linked and commonly impaired in the setting of critical illness. For example, rehabilitation services could be limited because of infection control and prevention measures, and social isolation in the community and due to restrictions on family presence in hospitals could affect the recovery of patients with post-acute sequelae of SARS-CoV-2 infection. Awareness of these factors could help in the provision of holistic post-acute care.
Although cohort studies of people who become critically ill with COVID-19 will be important in identifying any unique long-term sequelae of infection, outcomes are expected to mirror what we already know about ICU survivorship. Namely, survivors' experiences vary greatly, with many experiencing new and persistent impairments—including complex, multifactorial symptoms—for months to years. It remains unknown whether the experiences of patients with COVID-19 who require ICU care overlap substantially with those of hospitalised or non-hospitalised patients who are not admitted to the ICU, or whether these populations have distinct, multidisciplinary needs.
Survivorship clinics in the COVID-19 era
There are clinics in Europe, the Americas, and the Asia-Pacific that aim to address the multifaceted needs of survivors of critical illness and ICU care. These clinics offer key insights into potential barriers and facilitators in the establishment of PASC programmes, which have three key aims: (1) to meet survivors' immediate clinical needs; (2) to create a platform for research on the natural history of PASC; and (3) to expedite the rigorous evaluation of therapeutic options.40 Challenges to creating ICU survivor or PASC programmes fall into two broad domains: patient engagement and organisation of care pathways.
Most patients who are discharged from the ICU will not have access to a post-ICU clinic, with fewer than half of eligible survivors cared for by an ICU survivor clinic in their region.41, 42 Reasons for lack of access to care include the following: the clinic is unable to contact the survivor; the survivor does not have transportation to access the clinic; the survivor lacks insurance or the means to access services; or the survivor is unaware of the potential benefit of attending follow-up services.43 Similar barriers will affect access to PASC clinics for patients who have received community-based or hospital-based care for COVID-19. The COVID-19 pandemic is disproportionately affecting poorer communities of Black, Asian, and minority ethnic groups in high-income regions such as Europe and North America, and it is important that disparate provision of rehabilitation services does not exacerbate these inequalities further.44, 45 To overcome these challenges, clinicians should be flexible in their approach. While telemedicine, which was rapidly adopted during the pandemic, can overcome transportation barriers, digital poverty and illiteracy can hinder access to telemedicine appointments.46 Hence, where possible, a hybrid model that incorporates both virtual and in-person clinic options is optimal.
Acute care teams need to proactively communicate with hospitalised patients and their primary care providers, ideally before discharge from hospital, to counsel about the recovery trajectory, convey the potential benefit of post-acute clinic attendance, ensure access, and foster patient engagement. This communication can be achieved through direct patient education at the bedside by acute care physicians, advanced practice providers, and nurses, as well as by members of essential therapy services. The electronic medical record can also be used to share discharge summaries and follow-up recommendations with primary care teams, and to facilitate referrals to multidisciplinary clinics.
In some health-care systems, billing infrastructure—including inconsistent insurance reimbursement and billing codes—presents a significant barrier to the creation of follow-up services.43 Addressing this barrier requires changes in policy to reflect extension of support for acute COVID-19 care to cover PASC as well. A recent US Congressional hearing included advocacy for billing codes to capture PASC, allowing tracking of needs and services rendered, and incentivising the provision of services for this population.47 For the time being, existing billing structures can capture the type of care provided in the USA. For example, transitional care codes are designed specifically to capture the complexity and high need of the post-hospital period. The value of survivorship clinics to both patients and institutions must be demonstrated to ensure consistent staffing and support. Given the increased risk of health-care utilisation among survivors of COVID-19,7 programmes should prospectively evaluate the effect of multidisciplinary interventions on outcomes, including readmissions and health-care resource use.40 Moreover, working in survivorship clinics might improve staff wellbeing and reduce burnout by allowing staff to reconnect with patients and families they cared for in the inpatient setting.48 Importantly, the surge in COVID-19 survivors provides an opportunity to address gaps in knowledge regarding the management of frequently experienced symptoms after acute illness (panel ). This includes identifying unmet needs, especially during transitions of care, and targeting interventions to meet such needs.49
Panel. Research considerations to advance PASC knowledge and management.
Subjects for research
-
•
Prevalence of long-term symptoms, including stratification by key variables (eg, demographic factors such as age and ethnicity, initial severity of COVID-19)
-
•
Risk factors for development of persistent symptoms
-
•
Trajectory, changes over time, and clustering
-
•
Pathophysiological mechanisms of persistent symptoms
-
•
Therapeutic interventions
-
•
Economic, societal, and individual costs associated with persistent symptoms
Methodological considerations
-
•
Rigorous prospective studies that aim to reduce bias through thoughtful recruitment and retention strategies, and careful consideration of control groups
-
•
Development of core outcomes measurement sets to standardise assessments
-
•
Randomised controlled trials of therapeutic interventions
-
•
Epidemiological and interventional studies conducted in low-income and middle-income countries and areas with limited resources; participation of a diverse patient population in terms of factors such as age, sex, ethnicity, and socioeconomic status
PASC=post-acute sequelae of SARS-CoV-2 infection.
The PASC clinic model
PASC have not yet been fully defined. Hence, the development of a clear case definition of the post-COVID-19 condition—and understanding of the origins, nature, and course of persistent symptoms—is the focus of ongoing research. Existing PASC clinics must base criteria for referral and ongoing care on capacity and the availability of local resources. Primary care practitioners can provide initial screening to identify patients with ongoing symptoms beyond the initial acute illness, and a number of post-acute COVID-19 services use a screen-and-refer model to identify which specialists are most urgently needed for each individual patient's problems. Current guidelines recommend that patients with PASC be referred to multidisciplinary programmes with expertise in evaluating and managing commonly reported symptoms after COVID-19, including fatigue, dyspnoea, mental health disturbances, and cognitive impairments.50 Given overlapping symptoms and the need for multidisciplinary care, ICU recovery clinics and the PICS framework provide a foundation for the management of PASC for both ICU and non-ICU populations. It is not essential that ICU practitioners lead such clinics. Existing PASC clinics have adopted different approaches to leadership, including both primary and cooperative leadership by internal medicine, respiratory medicine, physiatry, endocrinology, and infectious disease. More importantly, a multidisciplinary approach with integrated referral pathways50 between PASC clinics, primary care practitioners, and core subspecialists is essential to success. Given the wide array of potential COVID-19 sequelae described over the weeks and months after acute infection,4, 5, 6, 7 a detailed history and physical examination is essential to identifying complications, evaluating contributing factors, and guiding referrals. Current guidelines recommend that follow-up begin 4–8 weeks after discharge, or from acute illness for those who were not hospitalised, and continue at 3-month intervals as needed.51, 52 Such guidelines might require adaptation both at the institution or site level based on available resources and to meet individual patient needs. Here, we describe the roles of specialists central to both ICU recovery services and comprehensive PASC programmes for all levels of acute illness (Figure 2, Figure 3 ).8, 50, 53, 54, 55
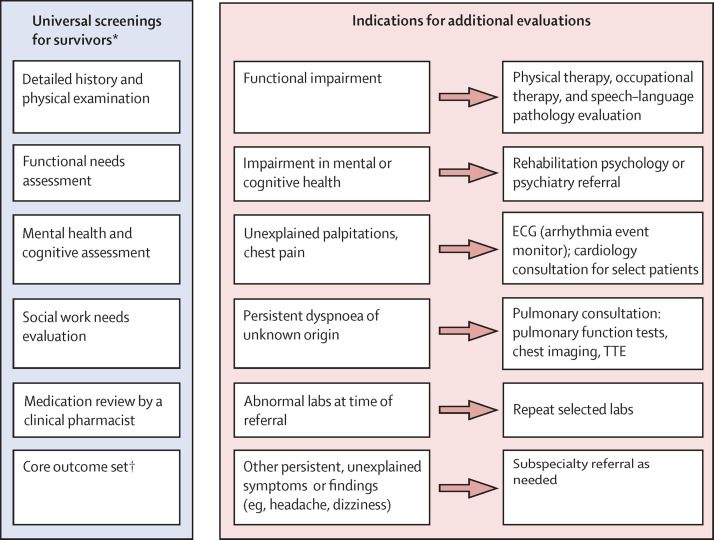
Figure 2.
Core services and evaluations for survivors of COVID-19 in a PASC clinic
Elements of the initial screening evaluation, done in person or remotely, for survivors of COVID-19 are shown, based on current guidelines, with recommendations for additional evaluations. ECG=electrocardiogram. TTE=transthoracic echocardiogram. *Core services recommended by the UK National Institute for Health and Care Excellence guidelines.50 †Core outcome set for acute respiratory failure53 and COVID-19.54, 55
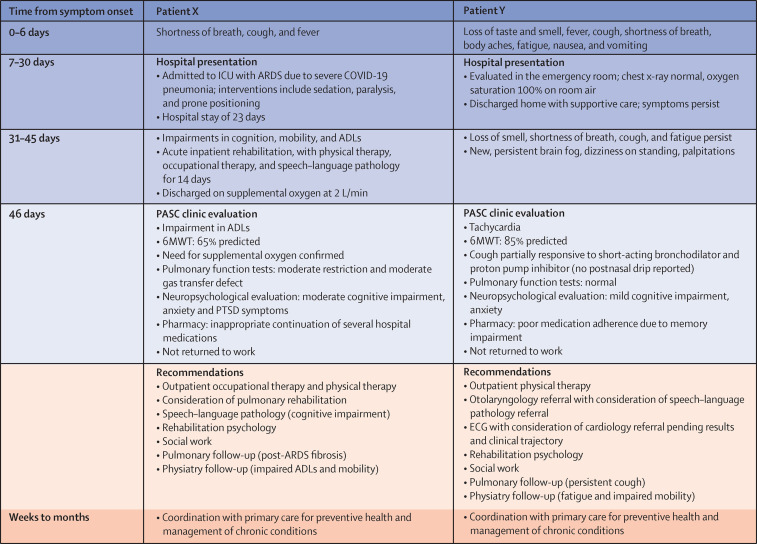
Figure 3.
Potential evaluation and management in a PASC clinic of patients with initial mild or severe COVID-19
Details for patients X and Y, both about 50 years of age, represent common clinical manifestations and evaluations for patients presenting to a PASC clinic after COVID-19. Patients receiving care for PASC might have had acute COVID-19 ranging from mild (patient Y) to severe (patient X). 6MWT=6-min walk test. ADLs=activities of daily living. ARDS=acute respiratory distress syndrome. ECG=electrocardiogram. ICU=intensive care unit. PTSD=post-traumatic stress disorder.
Physical medicine and rehabilitation
Physiatrists, rehabilitation psychologists, physical therapists, occupational therapists, and speech and language pathologists have a fundamental role in evaluating functional, cognitive, and mental health needs, and provide essential rehabilitation services in the post-acute setting. Rehabilitation in the post-acute phase falls under five general categories: (1) acute or subacute inpatient rehabilitation; (2) rehabilitation in skilled nursing facilities; (3) rehabilitation at home with home therapy services; (4) rehabilitation at home with outpatient therapy services; and (5) rehabilitation at home without any services. Patients might engage with one or all three therapy pathways during their recovery, and the provision of such services hinges on evaluations by key members of the rehabilitation team.
Physiatry
Physiatrists are integral members of the multidisciplinary team and coordinate rehabilitation care in acute and all five post-acute phases, focusing on biopsychosocial functioning and return to life with a new functional baseline.56, 57 This care involves not only cardiopulmonary rehabilitation after ARDS, but also a comprehensive rehabilitation plan to address physical, cognitive, psychosocial, and vocational needs given the complex and long-lasting nature of PASC. Physiatrists diagnose neurological and musculoskeletal complications, including brachial plexopathy, critical illness neuropathy and myopathy, stroke, or limb ischaemia due to thrombotic events. Autonomic dysfunction occurs in patients with PASC, so the timely completion of autonomic function tests (eg, the tilt table test), when indicated on the basis of initial screening, for proper diagnosis followed by appropriate medical and rehabilitation treatments is important to prevent further physical, mental, and cognitive impairments.58 Standardised, routine evaluations can determine the need for essential rehabilitation services, including physical therapy, occupational therapy, speech–language pathology, and rehabilitation psychology.
Physical therapy
ICU-acquired weakness (ICUAW), including critical illness polyneuropathy or myopathy, is associated with decreased strength and physical function that can persist for years after acute illness.49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61 Although the long-term effects of early intervention are uncertain, beginning early rehabilitation within 48 h of ICU admission is associated with improvement in strength and independent walking in the hospital.62, 63 Physical therapy services in the post-acute phase can be provided in the home as well as in the outpatient setting.64 Some standardised assessments of muscle strength and activity limitations (eg, exercise capacity and gait speed) in the post-acute setting can be performed both via telemedicine and in person (eg, use of the Activity Measure for Post-Acute Care assessment or sit-to-stand tests), whereas others require in-person visits (eg, the 6-min walk test and manual muscle testing score for strength).65 Early in the post-acute period, restorative interventions are of limited efficacy for patients recovering from critical illness. Therefore, physical therapy interventions in the post-acute setting initially focus on teaching both patients and caregivers compensatory strategies to adapt and improve functioning.65 Once functioning has been optimised, restorative exercises (eg, aerobic strength and endurance training) titrated to overload can be implemented while assessing cardiorespiratory demand.65
Occupational therapy
In the post-acute phase, practice guidelines specific to occupational therapy for survivors of COVID-19 include the following: considering the effects of oxygen weaning on activities of daily living; providing energy conservation and work simplification strategies; addressing the impact of cognitive impairments on occupational performance; addressing mental health, stress, and coping related to PTSD or anxiety disorders; and continuing the plan set in the early phases of the disease to return to participation in meaningful activities.66 Occupational therapists assist survivors with returning to work by facilitating a graded return—part time versus full time, working from home, and adjusted work tasks and responsibilities—and by providing assistive technology and vocational rehabilitation.67 Therapists also monitor for residual impairments in range of motion, sensation, fine motor coordination, or pain related to positioning such as prolonged proning. Occupational therapy interventions include remediation, such as strengthening or fine motor training, and compensation, such as writing aids or assistive technology.
Occupational therapists play an important part in the evaluation and management of cognitive impairment, encouraging active engagement in meaningful activities, resumption of a productive life, and a return to social participation and human connection.68 Therapists help patients to focus on engagement in meaningful occupations, including activities they need to, want to, and are expected to do.69 Cognitive rehabilitation includes retraining orientation, memory, attention, and executive functioning skills that are required to successfully complete tasks such as financial management and multitasking for return to work. In addition to retraining internal cognitive skills, occupational therapists also educate patients on the use of external strategies such as memory aids and assistive technology.70
Speech and language pathology
Speech–language pathologists are vital members of the multidisciplinary survivorship team.9, 71, 72 They evaluate and treat impairments of speech, language, and cognition, disorders of the voice and airway, and swallowing disorders. In the ICU, speech–language pathologists focus on delirium prevention, communication options for mechanically ventilated patients, and swallowing assessments. As patients transition to the wards, rehabilitation facilities, outpatient clinics, and home care, focus shifts to a more traditional rehabilitative approach.73 Patients who experienced delirium while critically ill appear to be particularly prone to cognitive-linguistic changes, including deficits of attention, memory, and other executive functions (eg, planning, organisation, and self-monitoring).14, 74, 75 Because behavioural treatments by speech–language pathologists also occur after ICU discharge, they are essential members of outpatient survivorship clinics.
Patients who were intubated during the acute phase of critical illness are at high risk of voice and airway disorders after extubation.76, 77, 78, 79 Speech–language pathologists are often the first clinicians to identify the presence and nature of voice and airway disorders while evaluating laryngeal and upper airway anatomy.80 Voice or breathing changes might reflect changes in vocal fold mobility and obstructions (eg, inflammation or tissue changes).76, 77 Intubation injuries extend from the voice and airway to dysphagia.77, 81, 82 Speech–language pathologists evaluate patients for dysphagia, either after a failed swallow screening or as a result of clinical referral.83 A videofluoroscopic swallow study or flexible endoscopic evaluation of swallowing is used to characterise disordered swallowing physiology and to plan treatment.84, 85 Although dysphagia can persist for many months after hospital discharge, most survivors will recover with proper care.86
Rehabilitation psychology and neuropsychology
Rehabilitation psychologists provide comprehensive evaluation, targeted psychotherapy, and health and behaviour interventions. Evaluations of current functioning follow a biopsychosocial model, which includes physical, emotional, cognitive, and social aspects of an individual's status in the context of medical and psychosocial history and current circumstances. A thorough assessment helps to identify barriers to optimal function as well as areas of strength and resilience. The following Patient-Reported Outcomes Measurement Information System (PROMIS)87 health-related quality-of-life measures have been used to evaluate symptoms and gauge treatment progress: Pain Interference; Fatigue; Anxiety; Depression; Self-Efficacy for Managing Symptoms; and Ability to Participate in Social Roles and Activities. Since the start of the COVID-19 pandemic, most evaluations have been conducted using telehealth.
Psychotherapy or behavioural interventions focus on helping survivors to develop coping and compensatory strategies. Evidence-based therapeutic interventions for mental health difficulties include the following: cognitive behavioural therapy (CBT) for depression, anxiety, and pain; cognitive processing therapy for PTSD; interventions to improve sleep (eg, CBT for insomnia or CBT-I); fatigue management; and mindfulness-based interventions to improve awareness and distress tolerance. Patients with pre-existing psychiatric comorbidities or those requiring medication management are also evaluated by psychiatrists.
In many cases, persistent fatigue, sleep disturbance, and emotional distress further aggravate cognitive difficulties. Consistent with reported patterns of cognitive difficulties in PICS,9, 14, 88 many patients with PASC present in the post-acute phase with generalised cognitive slowing and deficits in executive function, including impairments in divided and sustained attention, initiation, cognitive flexibility, and executive control over memory. Neuropsychological evaluation of cognitive function in the context of estimated optimal ability can help to identify current difficulties, aid in differential diagnosis, and develop treatment recommendations. A subset of survivors might benefit from referral to speech–language pathology services for cognitive rehabilitation. As a patient's disease process often is experienced closely by their family members, screening for psychological symptoms should be extended to this group.89
Neuropsychiatry
Neuropsychiatrists can provide specific input when the medical, biological, and psychological aspects of a patient's clinical presentation converge; for example, when knowledge about the antidepressant properties of anti-inflammatory treatments90 and the anti-inflammatory aspects of antidepressant treatments can work synergistically.91 Whereas rehabilitation psychologists approach a patient's problems from a mind-based perspective, with implications for brain function, neuropsychiatrists do the opposite, beginning with a brain-based perspective that has implications for psychological wellbeing. Together, rehabilitation psychologists and neuropsychiatrists can offer a complementary and collaborative approach to the care of patients, with the combined tools to address the many biopsychological manifestations of post-acute COVID-19.
Pharmacy
A comprehensive medication review is the core service provided by a pharmacist in a PASC clinic.92, 93 This multifaceted review encompasses medication de-prescribing, initiation or restarting of drug regimens, and therapy optimisation. Whereas stress ulcer prophylaxis in the ICU is classically associated with inappropriate continuation, any medication initiated for an inpatient indication has the potential to be inappropriately continued in the outpatient setting. In the post-COVID-19 phase, emphasis is placed on determining the duration of antithrombotic therapy, which might have been initiated in the setting of an unconfirmed diagnosis. A full understanding of the new and dynamic impairments facing ICU survivors is needed to properly adapt and adjust drug therapy regimens. Improving physical function can affect blood pressure or glucose levels and their associated medications. Close monitoring and frequent medication dose adjustments are particularly important for survivors experiencing recovery of kidney or liver function.
A pharmacist who understands the challenges of COVID-19 survivorship can also help to ensure that prescribed medications are accessible and taken at the right time. For example, new cognitive impairment could limit a survivor's understanding of newly prescribed medications and their side-effects. Memory impairments can decrease medication compliance. Restrictions on in-person primary care in 2020 might have resulted in missed immunisations, and changes in employment status or medication insurance coverage might affect the continuation of previous drug therapy.92, 93 A critical care clinical pharmacist is well positioned to address these challenges as part of an interdisciplinary clinic team.94 Pharmacists work with a medication access team to ensure affordability and appropriate access to medications. They supply pillboxes or discuss strategies for medication reminders to improve medication adherence. Depending on local practice laws, pharmacists are often able to obtain a vaccination history and administer immunisations.
Social work and welfare support
Social workers address several important aspects of recovery after COVID-19. About one-third of patients in recovery report that persistent symptoms affect their ability to return to work up to 7 months after acute illness.27 Social workers can connect patients with job resources and provide support to address financial needs. Social workers and community health workers can intervene to improve access to care for underserved populations, and enhance communication between patients, their families, and the health-care team.95, 96 They also conduct screening for mental health impairments in both patients and their family members or caregivers, and provide psychoeducation and referrals as needed.95 Addressing the needs of family caregivers is particularly important during large-scale disasters such as the COVID-19 pandemic, because social distancing and restricted family presence in hospitals threaten family integrity and make it difficult for family members to feel calm, connected, and useful.97
Primary care
In the long term, primary care practitioners will provide both aftercare and care coordination for the majority of survivors of COVID-19.98, 99 Their knowledge of patients' long-term medical history and expertise in care coordination mean that they are well placed to have a key role in the management of PASC, especially in regions where PASC clinics will not be available. However, the rapid growth of information relating to the diverse manifestations after SARS-CoV-2 infection is not yet readily available in a comprehensive format, making it challenging for primary care practitioners to stay up to date. Thus, effective information transfer from both hospital and the PASC clinic should be mandatory—regarding all dimensions, as detailed above, and including communication of evidence-based treatment recommendations. This process could occur through elaborated discharge letters handed to the patient directly with instruction to take to follow-up primary care appointments, direct communication between the hospital or PASC clinic and primary care providers, and further development of virtual services.98
Equitable provision of care
Under-served populations and implementation in LMICs
Ensuring equity of access to post-COVID-19 health-care services to optimise patient recovery is essential, and particular attention should be paid to under-served populations. Examples of under-served populations in Europe and North America include Black, Asian, and minority ethnic groups—which have an increased risk of SARS-CoV-2 infection and poorer outcomes, including higher mortality, from COVID-19100, 101—those at educational or socioeconomic disadvantage, religious minorities, people living in remote areas, those with caregiving responsibilities or in full-time employment, and those with factors associated with health status such as cognitive impairment, learning disability, or multiple comorbidities, among many others.102, 103 Strategies to help vulnerable communities to access health-care services have been reported, most notably the following four: (1) addressing the social determinants of health through screening and information provision, signposting health-care users to appropriate community services, and encouraging community stakeholders to support individualised service delivery according to the needs of patients; (2) adopting new and innovative virtual care strategies (eg, through various forms of telehealth); (3) designing global budget payments to support hospitals located in areas delivering services to vulnerable communities; and (4) using an inpatient–outpatient transformation strategy that more closely integrates hospital-based and community-based services, and adapts each, to meet the needs of individual communities.103
Implementation of multidisciplinary PASC clinics might be challenging in LMICs. Existing multidisciplinary models serve as a guide to addressing potential barriers.104 Educational programmes that target health-care workers and community and public health staff, and focus on providing information on screening for commonly experienced symptoms, are essential.104 Community health workers provide education and raise awareness in local communities.105 A hub-and-spoke model, in which larger academic centres provide specialised care and consultation to surrounding providers and communities, is effective.105 Finally, the provision of telehealth infrastructure to facilitate self-assessment and self-management can support patients in remote communities.105
Language services for non-native speakers
Language support services are vital for survivors of COVID-19 in immigrant communities that are experiencing disproportionately high rates of infection and severe disease (eg, Hispanic and Latino populations in the USA).106, 107 Multilingual staff are highly valued within PASC clinics, particularly when responding to phone calls from non-native speaking patients. Pre-translation of written informational documents is crucial to care delivery for these survivors. Interpreter services have adapted to telemedicine models, allowing interpreters to be present on both phone calls and video platforms, which can also be used during in-person clinic visits. In addition to being convenient to schedule, remote interpreter services reduce the need to accommodate additional people in the clinic space under infection control procedures.
Research collaboration
The chief goal of PASC clinics must be to support the recovery and wellbeing of people who survive infection. However, clinics can help to estimate the frequency of patient outcomes and identify survivor phenotypes without compromising their primary mission, by optimising the data they record in electronic health records.108, 109 Therefore, an international task force comprising 93 experts, including representatives of the European Respiratory Society (ERS) and the American Thoracic Society (ATS), that was convened in 2020 suggested that follow-up assessments of hospitalised COVID-19 survivors occur 6–8 weeks after discharge and include the core outcome set for survivors of acute respiratory failure.54, 55 The core outcome set comprises the EuroQol-5D (EQ-5D; a measure of health-related quality of life), the Hospital Anxiety and Depression Scale (designed to detect states of depression and anxiety in a general medical population of patients), and the Impact of Event Scale – Revised (a measure of PTSD symptoms), and can be administered entirely over the phone, making it feasible to implement in clinics using telehealth.53 Importantly, the core outcome set represents a minimum number of assessments to facilitate research and comparisons, and most clinics will choose to incorporate additional assessments determined by resources and interest. In addition to assessment at 6–8 weeks post-discharge, a coordinated approach to COVID-19 care should plan for serial assessments triggered by life events. For example, assessment before hospital discharge will determine the need for post-acute care services, whereas the end of employment-based medical disability benefits, plateaus in recovery, or significant changes in symptoms or disability should prompt reassessment.110
PASC clinics provide a unique opportunity to advance the field of ICU survivorship clinics, given the unprecedented surge of critical illness survivors presenting for care. A Cochrane review in 2018 found insufficient evidence to conclude that ICU survivorship clinics improve outcomes by addressing unmet needs of patients after discharge from the ICU.111 The review highlighted heterogeneity in the services delivered and in the design of studies evaluating efficacy as limitations. Prospective evaluation of the efficacy of PASC clinics, especially those including ICU survivors, and ideally with a randomised controlled design, could answer important questions about how such clinics might improve outcomes.
Conclusions
COVID-19 represents an unprecedented public health emergency. Multidisciplinary post-acute care programmes, established using a PICS framework, can deliver essential support for both hospitalised and non-hospitalised patients during their recovery. These programmes also provide an infrastructure for rigorous research, conducted alongside clinical care, to better understand the prevalence, natural history, and risk factors of PASC and to evaluate therapeutic interventions to improve outcomes. Given the disproportionate impact of COVID-19 on under-served populations, special attention should be given to ensuring the equitable delivery of care. The PICS–PASC model has the potential to endure beyond the COVID-19 pandemic as a means of providing essential multidisciplinary care for patients experiencing complex, multifactorial symptoms from a variety of acute and chronic illnesses. In parallel with clinical pathways of care, structured peer support groups, with trained moderators, provide an innovative opportunity for survivors of COVID-19 and their caregivers to support one another in their recovery.112 The current paucity of data on the effectiveness of specific interventions to treat PASC means that PASC clinics will need to maintain the ability to adapt to evolving evidence (panel). Such evidence should be informed by the inclusion in follow-up and research of a diverse population of patients, and include evaluation of innovative approaches to care, such as telemedicine and new electronic applications.
Search strategy and selection criteria
We searched PubMed and Embase for papers published in English between May 1, 2020, and Aug 1, 2021, using the terms “post-acute COVID-19 syndrome”, “post-intensive care syndrome”, “long COVID”, “long haul” and “COVID”, “post-acute sequelae” and “SARS-CoV-2 infection”, “chronic COVID syndrome”, and “post-COVID condition” or “post-COVID-19 condition”. We selected peer-reviewed papers and high-quality preprints that pertained to the main aim of this Health-care Development paper: to inform the development of multidisciplinary pathways of care for the post-acute sequelae of SARS-CoV-2 infection. Details of preprints were updated with final publication details as this paper was prepared for publication. We referred to the reference lists of selected papers and to papers in our own files to provide the reader with a review of relevant evidence from before the COVID-19 pandemic.
Declaration of interests
JM reports payment from Springer for the book Improving Critical Care Survivorship. CMS also reports royalties from Springer for the book Improving Critical Care Survivorship. MBB reports payment for educational materials from MedBridge, outside of the submitted work. AET reports payment for consulting from Medical Science Affiliates, outside of the submitted work. AMP, EB, BC, AVA, MTK, KC, CR, KFRS, SYK, and AK declare no competing interests.
Acknowledgments
Acknowledgments
AMP is funded by a US National Institutes of Health (NIH)–National Heart, Lung, and Blood Institute award (K23HL138206). EB (K23ES029105) and AET (K01HL141637) are each funded by an NIH early career development award. JM receives a fellowship from University of Cambridge, UK (THIS Institute, PD-2019-02-16). CMS receives grant support from the US Department of Defense and the Society of Critical Care Medicine. MBB receives grant support from the NIH (5R01NR017433).
Contributors
AMP and AET compiled the initial full draft of the manuscript, made essential content edits, and coordinated editing and revisions. Each co-author made direct contributions to key sections of the manuscript. All authors contributed to the literature search and review, and reviewed and edited the final draft of this Health-care Development paper.
References
- 1.Johns Hopkins University & Medicine COVID-19 dashboard by the Center for Systems Science and Engineering (CSS) at Johns Hopkins University (JHU) https://coronavirus.jhu.edu/map.html
- 2.Tenforde MW, Kim SS, Lindsell CJ, et al. Symptom duration and risk factors for delayed return to usual health among outpatients with COVID-19 in a multistate health care systems network — United States, March–June 2020. MMWR Morb Mortal Wkly Rep. 2020;2020:993–998. doi: 10.15585/mmwr.mm6930e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Huang C, Huang L, Wang Y, et al. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. 2021;397:220–232. doi: 10.1016/S0140-6736(20)32656-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lopez-Leon S, Wegman-Ostrosky T, Perelman C, et al. More than 50 long-term effects of COVID-19: a systematic review and meta-analysis. Sci Rep. 2021;11:16144. doi: 10.1038/s41598-021-95565-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nalbandian A, Sehgal K, Gupta A, et al. Post-acute COVID-19 syndrome. Nat Med. 2021;27:601–615. doi: 10.1038/s41591-021-01283-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Al-Aly Z, Xie Y, Bowe B. High-dimensional characterization of post-acute sequelae of COVID-19. Nature. 2021;594:259–264. doi: 10.1038/s41586-021-03553-9. [DOI] [PubMed] [Google Scholar]
- 8.Rajan S, Khunti K, Alwan N, et al. Policy Brief 39: In the wake of the pandemic: preparing for long COVID. World Health Organization, 2021. https://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/publications-and-technical-guidance/2021/in-the-wake-of-the-pandemic-preparing-for-long-covid-2021 [PubMed]
- 9.Needham DM, Davidson J, Cohen H, et al. Improving long-term outcomes after discharge from intensive care unit: report from a stakeholders’ conference. Crit Care Med. 2012;40:502–509. doi: 10.1097/CCM.0b013e318232da75. [DOI] [PubMed] [Google Scholar]
- 10.Herridge MS, Cheung AM, Tansey CM, et al. One-year outcomes in survivors of the acute respiratory distress syndrome. N Engl J Med. 2003;348:683–693. doi: 10.1056/NEJMoa022450. [DOI] [PubMed] [Google Scholar]
- 11.Nikayin S, Rabiee A, Hashem MD, et al. Anxiety symptoms in survivors of critical illness: a systematic review and meta-analysis. Gen Hosp Psychiatry. 2016;43:23–29. doi: 10.1016/j.genhosppsych.2016.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rabiee A, Nikayin S, Hashem MD, et al. Depressive symptoms after critical illness: a systematic review and meta-analysis. Crit Care Med. 2016;44:1744–1753. doi: 10.1097/CCM.0000000000001811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Parker AM, Sricharoenchai T, Raparla S, Schneck KW, Bienvenu OJ, Needham DM. Posttraumatic stress disorder in critical illness survivors: a metaanalysis. Crit Care Med. 2015;43:1121–1129. doi: 10.1097/CCM.0000000000000882. [DOI] [PubMed] [Google Scholar]
- 14.Pandharipande PP, Girard TD, Jackson JC, et al. Long-term cognitive impairment after critical illness. N Engl J Med. 2013;369:1306–1316. doi: 10.1056/NEJMoa1301372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Michelen M, Manoharan L, Elkheir N, et al. Characterising long COVID: a living systematic review. BMJ Glob Health. 2021;6:e005427. doi: 10.1136/bmjgh-2021-005427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Venturelli S, Benatti SV, Casati M, et al. Surviving COVID-19 in Bergamo province: a post-acute outpatient re-evaluation. Epidemiol Infect. 2021;149:e32. doi: 10.1017/S0950268821000145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nasserie T, Hittle M, Goodman SN. Assessment of the frequency and variety of persistent symptoms among patients with COVID-19: a systematic review. JAMA Netw Open. 2021;4:e2111417. doi: 10.1001/jamanetworkopen.2021.11417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Domingo FR, Waddell LA, Cheung AM, et al. Prevalence of long-term effects in individuals diagnosed with COVID-19: a living systematic review. medRxiv. 2021 https://doi.org/10.1101/2021.06.03.21258317 (preprint) published online June 6. [Google Scholar]
- 19.Fernández-de-las-Peñas C, Palacios-Ceña D, Gómez-Mayordomo V, et al. Prevalence of post-COVID-19 symptoms in hospitalized and non-hospitalized COVID-19 survivors: a systematic review and meta-analysis. Eur J Intern Med. 2021 doi: 10.1016/j.ejim.2021.06.009. published online June 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Iqbal FM, Lam K, Sounderajah V, Clarke JM, Ashrafian H, Darzi A. Characteristics and predictors of acute and chronic post-COVID syndrome: a systematic review and meta-analysis. EClinicalMedicine. 2021;36:100899. doi: 10.1016/j.eclinm.2021.100899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Malik J, Zaidi SMJ, Iqbal R, et al. Post-acute COVID-19 syndrome and its prolonged effects: an updated systematic review. medRxiv. 2021 doi: 10.1016/j.amsu.2022.103995. https://doi.org/10.1101/2021.05.09.21256911 (preprint) published online May 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cabrera Martimbianco AL, Pacheco RL, Bagattini ÂM, Riera R. Frequency, signs and symptoms, and criteria adopted for long COVID-19: a systematic review. Int J Clin Pract. 2021 doi: 10.1111/ijcp.14357. published online May 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sanchez-Ramirez DC, Normand K, Zhaoyun Y, Torres-Castro R. Long-term impact of COVID-19: a systematic review of the literature and meta-analysis. Biomedicines. 2021;9:900. doi: 10.3390/biomedicines9080900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Page MJ, Moher D, Bossuyt PM, et al. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ. 2021;372:n160. doi: 10.1136/bmj.n160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stroup DF, Berlin JA, Morton SC, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. JAMA. 2000;283:2008–2012. doi: 10.1001/jama.283.15.2008. [DOI] [PubMed] [Google Scholar]
- 26.Baker HA, Safavynia SA, Evered LA. The ‘third wave’: impending cognitive and functional decline in COVID-19 survivors. Br J Anaesth. 2021;126:44–47. doi: 10.1016/j.bja.2020.09.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Davis HE, Assaf GS, McCorkell L, et al. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. EClinicalMedicine. 2021;38:101019. doi: 10.1016/j.eclinm.2021.101019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hampshire A, Trender W, Chamberlain SR, et al. Cognitive deficits in people who have recovered from COVID-19. EClinicalMedicine. 2021 doi: 10.1016/j.eclinm.2021.101044. published online July 23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Heneka MT, Golenbock D, Latz E, Morgan D, Brown R. Immediate and long-term consequences of COVID-19 infections for the development of neurological disease. Alzheimers Res Ther. 2020;12:69. doi: 10.1186/s13195-020-00640-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Carfì A, Bernabei R, Landi F. Persistent symptoms in patients after acute COVID-19. JAMA. 2020;324:603–605. doi: 10.1001/jama.2020.12603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Goërtz YMJ, Van Herck M, Delbressine JM, et al. Persistent symptoms 3 months after a SARS-CoV-2 infection: the post-COVID-19 syndrome? ERJ Open Res. 2020;6:00542–02020. doi: 10.1183/23120541.00542-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bo H-X, Li W, Yang Y, et al. Posttraumatic stress symptoms and attitude toward crisis mental health services among clinically stable patients with COVID-19 in China. Psychol Med. 2021;51:1052–1053. doi: 10.1017/S0033291720000999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Guler SA, Ebner L, Aubry-Beigelman C, et al. Pulmonary function and radiological features 4 months after COVID-19: first results from the national prospective observational Swiss COVID-19 lung study. Eur Respir J. 2021;57:2003690. doi: 10.1183/13993003.03690-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lerum TV, Aaløkken TM, Brønstad E, et al. Dyspnoea, lung function and CT findings three months after hospital admission for COVID-19. Eur Respir J. 2021;57:2003448. doi: 10.1183/13993003.03448-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Townsend L, Dowds J, O’Brien K, et al. Persistent poor health post-COVID-19 is not associated with respiratory complications or initial disease severity. Ann Am Thorac Soc. 2021;18:997–1003. doi: 10.1513/AnnalsATS.202009-1175OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Raman B, Cassar MP, Tunnicliffe EM, et al. Medium-term effects of SARS-CoV-2 infection on multiple vital organs, exercise capacity, cognition, quality of life and mental health, post-hospital discharge. EClinicalMedicine. 2021;31:100683. doi: 10.1016/j.eclinm.2020.100683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Neufeld KJ, Leoutsakos JS, Yan H, et al. Fatigue symptoms during the first year following ARDS. Chest. 2020;158:999–1007. doi: 10.1016/j.chest.2020.03.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hosey MM, Needham DM. Survivorship after COVID-19 ICU stay. Nat Rev Dis Primers. 2020;6:60. doi: 10.1038/s41572-020-0201-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kandori K, Okada Y, Ishii W, Narumiya H, Maebayashi Y, Iizuka R. Association between visitation restriction during the COVID-19 pandemic and delirium incidence among emergency admission patients: a single-center retrospective observational cohort study in Japan. J Intensive Care. 2020;8:90. doi: 10.1186/s40560-020-00511-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lasiter S, Oles SK, Mundell J, London S, Khan B. Critical care follow-up clinics: a scoping review of interventions and outcomes. Clin Nurse Spec. 2016;30:227–237. doi: 10.1097/NUR.0000000000000219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sevin CM, Bloom SL, Jackson JC, Wang L, Ely EW, Stollings JL. Comprehensive care of ICU survivors: development and implementation of an ICU recovery center. J Crit Care. 2018;46:141–148. doi: 10.1016/j.jcrc.2018.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.MacTavish P, Quasim T, Purdie C, et al. Medication-related problems in intensive care unit survivors: learning from a multicenter program. Ann Am Thorac Soc. 2020;17:1326–1329. doi: 10.1513/AnnalsATS.202005-444RL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Haines KJ, McPeake J, Hibbert E, et al. Enablers and barriers to implementing ICU follow-up clinics and peer support groups following critical illness: the Thrive collaboratives. Crit Care Med. 2019;47:1194–1200. doi: 10.1097/CCM.0000000000003818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lone NI, McPeake J, Stewart NI, et al. Influence of socioeconomic deprivation on interventions and outcomes for patients admitted with COVID-19 to critical care units in Scotland: a national cohort study. Lancet Reg Health Eur. 2021;1:100005. doi: 10.1016/j.lanepe.2020.100005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sze S, Pan D, Nevill CR, et al. Ethnicity and clinical outcomes in COVID-19: a systematic review and meta-analysis. EClinicalMedicine. 2020;29:100630. doi: 10.1016/j.eclinm.2020.100630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Seah KM. COVID-19: Exposing digital poverty in a pandemic. Int J Surg. 2020;79:127–128. doi: 10.1016/j.ijsu.2020.05.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hearing on “The Long Haul: Forging a Path through the Lingering Effects of COVID-19” before the Subcommittee on Health of the Committee on Energy and Commerce, US House of Representatives, 117th Congress. April 28, 2021. https://energycommerce.house.gov/committee-activity/hearings/hearing-on-the-long-haul-forging-a-path-through-the-lingering-effects-of?fbclid=IwAR1ZcmtOIRv_hmS9mRhRhADq-jOXvpTgO3NdBzYubuf-I0QWyLm2t3hkqts
- 48.Haines KJ, Sevin CM, Hibbert E, et al. Key mechanisms by which post-ICU activities can improve in-ICU care: results of the international THRIVE collaboratives. Intensive Care Med. 2019;45:939–947. doi: 10.1007/s00134-019-05647-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Brown SM, Bose S, Banner-Goodspeed V, et al. Approaches to addressing post-intensive care syndrome among intensive care unit survivors. A narrative review. Ann Am Thorac Soc. 2019;16:947–956. doi: 10.1513/AnnalsATS.201812-913FR. [DOI] [PubMed] [Google Scholar]
- 50.National Institute for Health and Care Excellence COVID-19 rapid guideline: managing the long-term effects of COVID-19. Dec 18, 2020. https://www.nice.org.uk/guidance/NG188 [PubMed]
- 51.Global COVID-19 clinical platform case report form (CRF) for post COVID condition (Post COVID-19 CRF) World Health Organization, 2021. https://www.who.int/teams/health-care-readiness-clinical-unit/covid-19/data-platform
- 52.Tan T, Brett SJ, Stokes T. Rehabilitation after critical illness: summary of NICE guidance. BMJ. 2009;338:b822. doi: 10.1136/bmj.b822. [DOI] [PubMed] [Google Scholar]
- 53.Needham DM, Sepulveda KA, Dinglas VD, et al. Core outcome measures for clinical research in acute respiratory failure survivors. an international modified Delphi consensus study. Am J Respir Crit Care Med. 2017;196:1122–1130. doi: 10.1164/rccm.201702-0372OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Spruit MA, Holland AE, Singh SJ, Tonia T, Wilson KC, Troosters T. COVID-19: interim guidance on rehabilitation in the hospital and post-hospital phase from a European Respiratory Society- and American Thoracic Society-coordinated international task force. Eur Respir J. 2020;56:2002197. doi: 10.1183/13993003.02197-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Tong A, Elliott JH, Azevedo LC, et al. Core outcomes set for trials in people with coronavirus disease 2019. Crit Care Med. 2020;48:1622–1635. doi: 10.1097/CCM.0000000000004585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kim SY, Kumble S, Patel B, et al. Managing the rehabilitation wave: rehabilitation services for COVID-19 survivors. Arch Phys Med Rehabil. 2020;101:2243–2249. doi: 10.1016/j.apmr.2020.09.372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Simpson R, Robinson L. Rehabilitation after critical illness in people with COVID-19 infection. Am J Phys Med Rehabil. 2020;99:470–474. doi: 10.1097/PHM.0000000000001443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Dani M, Dirksen A, Taraborrelli P, et al. Autonomic dysfunction in ‘long COVID’: rationale, physiology and management strategies. Clin Med (Lond) 2021;21:e63–e67. doi: 10.7861/clinmed.2020-0896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Fan E, Dowdy DW, Colantuoni E, et al. Physical complications in acute lung injury survivors: a two-year longitudinal prospective study. Crit Care Med. 2014;42:849–859. doi: 10.1097/CCM.0000000000000040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hermans G, De Jonghe B, Bruyninckx F, Van den Berghe G. Critical illness polyneuropathy and myopathy. Crit Care. 2008;12:238. doi: 10.1186/cc7100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kress JP, Hall JB. ICU-acquired weakness and recovery from critical illness. N Engl J Med. 2014;370:1626–1635. doi: 10.1056/NEJMra1209390. [DOI] [PubMed] [Google Scholar]
- 62.Schweickert WD, Pohlman MC, Pohlman AS, et al. Early physical and occupational therapy in mechanically ventilated, critically ill patients: a randomised controlled trial. Lancet. 2009;373:1874–1882. doi: 10.1016/S0140-6736(09)60658-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ding N, Zhang Z, Zhang C, et al. What is the optimum time for initiation of early mobilization in mechanically ventilated patients? A network meta-analysis. PLoS One. 2019;14:e0223151. doi: 10.1371/journal.pone.0223151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ohtake PJ, Lee AC, Scott JC, et al. Physical impairments associated with post-intensive care syndrome: systematic review based on the World Health Organization's international classification of functioning, disability and health framework. Phys Ther. 2018;98:631–645. doi: 10.1093/ptj/pzy059. [DOI] [PubMed] [Google Scholar]
- 65.Smith JM, Lee AC, Zeleznik H, et al. Home and community-based physical therapist management of adults with post-intensive care syndrome. Phys Ther. 2020;100:1062–1073. doi: 10.1093/ptj/pzaa059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Robinson MR, Koverman B, Becker C, Ciancio KE, Fisher G, Saake S. Lessons learned from the COVID-19 pandemic: occupational therapy on the front line. Am J Occup Ther. 2021;75:7502090010. doi: 10.5014/ajot.2021.047654. [DOI] [PubMed] [Google Scholar]
- 67.Frank A. Rehabilitation after COVID-19: supporting those in employment back to work. Clin Med (Lond) 2020;20:e280–e281. doi: 10.7861/clinmed.Let.20.6.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kotfis K, Williams Roberson S, Wilson J, et al. COVID-19: What do we need to know about ICU delirium during the SARS-CoV-2 pandemic? Anaesthesiol Intensive Ther. 2020;52:132–138. doi: 10.5114/ait.2020.95164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.World Federation of Occupational Therapists Occupational therapy response to the COVID-19 pandemic. 2020. https://www.wfot.org/resources/occupational-therapy-response-to-the-covid-19-pandemic
- 70.Royal College of Occupational Therapists A quick guide for occupational therapists: rehabilitation for people recovering from COVID-19. 2020. https://www.rcot.co.uk/files/guidance-quick-guide-occupational-therapists-rehabilitation-people-recovering-covid-19-2020
- 71.Stam HJ, Stucki G, Bickenbach J. Covid-19 and post intensive care syndrome: a call for action. J Rehabil Med. 2020;52:jrm00044. doi: 10.2340/16501977-2677. [DOI] [PubMed] [Google Scholar]
- 72.Stierli S, Buss I, Redecker H, et al. Insights from an interprofessional post-COVID-19 rehabilitation unit: a speech and language therapy and respiratory medicine perspective. J Rehabil Med. 2020;52:jrm00100. doi: 10.2340/16501977-2735. [DOI] [PubMed] [Google Scholar]
- 73.Ramage AE. Potential for cognitive communication impairment in COVID-19 survivors: a call to action for speech-language pathologists. Am J Speech Lang Pathol. 2020;29:1821–1832. doi: 10.1044/2020_AJSLP-20-00147. [DOI] [PubMed] [Google Scholar]
- 74.Girard TD, Thompson JL, Pandharipande PP, et al. Clinical phenotypes of delirium during critical illness and severity of subsequent long-term cognitive impairment: a prospective cohort study. Lancet Respir Med. 2018;6:213–222. doi: 10.1016/S2213-2600(18)30062-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Marra A, Pandharipande PP, Girard TD, et al. Co-occurrence of post-intensive care syndrome problems among 406 survivors of critical illness. Crit Care Med. 2018;46:1393–1401. doi: 10.1097/CCM.0000000000003218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Benjamin B. Prolonged intubation injuries of the larynx: endoscopic diagnosis, classification, and treatment. Ann Otol Rhinol Laryngol Suppl. 1993;160:1–15. doi: 10.1177/00034894931020s401. [DOI] [PubMed] [Google Scholar]
- 77.Brodsky MB, Levy MJ, Jedlanek E, et al. Laryngeal injury and upper airway symptoms after oral endotracheal intubation with mechanical ventilation during critical care: a systematic review. Crit Care Med. 2018;46:2010–2017. doi: 10.1097/CCM.0000000000003368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Shinn JR, Kimura KS, Campbell BR, et al. Incidence and outcomes of acute laryngeal injury after prolonged mechanical ventilation. Crit Care Med. 2019;47:1699–1706. doi: 10.1097/CCM.0000000000004015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Fiacchini G, Tricò D, Ribechini A, et al. Evaluation of the incidence and potential mechanisms of tracheal complications in patients with COVID-19. JAMA Otolaryngol Head Neck Surg. 2021;147:70–76. doi: 10.1001/jamaoto.2020.4148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Langmore SE. History of fiberoptic endoscopic evaluation of swallowing for evaluation and management of pharyngeal dysphagia: changes over the years. Dysphagia. 2017;32:27–38. doi: 10.1007/s00455-016-9775-x. [DOI] [PubMed] [Google Scholar]
- 81.Moss M, White SD, Warner H, et al. Development of an accurate bedside swallowing evaluation decision tree algorithm for detecting aspiration in acute respiratory failure survivors. Chest. 2020;158:1923–1933. doi: 10.1016/j.chest.2020.07.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Krisciunas GP, Langmore SE, Gomez-Taborda S, et al. The association between endotracheal tube size and aspiration (during flexible endoscopic evaluation of swallowing) in acute respiratory failure survivors. Crit Care Med. 2020;48:1604–1611. doi: 10.1097/CCM.0000000000004554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Brodsky MB, González-Fernández M, Mendez-Tellez PA, Shanholtz C, Palmer JB, Needham DM. Factors associated with swallowing assessment after oral endotracheal intubation and mechanical ventilation for acute lung injury. Ann Am Thorac Soc. 2014;11:1545–1552. doi: 10.1513/AnnalsATS.201406-274OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Macht M, Wimbish T, Clark BJ, et al. Diagnosis and treatment of post-extubation dysphagia: results from a national survey. J Crit Care. 2012;27:578–586. doi: 10.1016/j.jcrc.2012.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Brodsky MB, Mayfield EB, Gross RD. Clinical decision making in the ICU: dysphagia screening, assessment, and treatment. Semin Speech Lang. 2019;40:170–187. doi: 10.1055/s-0039-1688980. [DOI] [PubMed] [Google Scholar]
- 86.Brodsky MB, Huang M, Shanholtz C, et al. Recovery from dysphagia symptoms after oral endotracheal intubation in acute respiratory distress syndrome survivors. A 5-year longitudinal study. Ann Am Thorac Soc. 2017;14:376–383. doi: 10.1513/AnnalsATS.201606-455OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Cella D, Yount S, Rothrock N, et al. The Patient-Reported Outcomes Measurement Information System (PROMIS): progress of an NIH Roadmap cooperative group during its first two years. Med Care. 2007;45(suppl 1):S3–11. doi: 10.1097/01.mlr.0000258615.42478.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Rengel KF, Hayhurst CJ, Pandharipande PP, Hughes CG. Long-term cognitive and functional impairments after critical illness. Anesth Analg. 2019;128:772–780. doi: 10.1213/ANE.0000000000004066. [DOI] [PubMed] [Google Scholar]
- 89.Schmidt M, Azoulay E. Having a loved one in the ICU: the forgotten family. Curr Opin Crit Care. 2012;18:540–547. doi: 10.1097/MCC.0b013e328357f141. [DOI] [PubMed] [Google Scholar]
- 90.Wittenberg GM, Stylianou A, Zhang Y, et al. Effects of immunomodulatory drugs on depressive symptoms: a mega-analysis of randomized, placebo-controlled clinical trials in inflammatory disorders. Mol Psychiatry. 2020;25:1275–1285. doi: 10.1038/s41380-019-0471-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Jansen van Vuren E, Steyn SF, Brink CB, Möller M, Viljoen FP, Harvey BH. The neuropsychiatric manifestations of COVID-19: Interactions with psychiatric illness and pharmacological treatment. Biomed Pharmacother. 2021;135:111200. doi: 10.1016/j.biopha.2020.111200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Coe AB, Bookstaver RE, Fritschle AC, et al. Pharmacists’ perceptions on their role, activities, facilitators, and barriers to practicing in a post-intensive care recovery clinic. Hosp Pharm. 2020;55:119–125. doi: 10.1177/0018578718823740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Mohammad RA, Betthauser KD, Korona RB, et al. Clinical pharmacist services within intensive care unit recovery clinics: an opinion of the critical care practice and research network of the American College of Clinical Pharmacy. J Am Coll Clin Pharm. 2020;3:1369–1379. [Google Scholar]
- 94.Stollings JL, Bloom SL, Sevin CM. Reply: critical care pharmacists and medication management in an ICU recovery center. Ann Pharmacother. 2019;53:106. doi: 10.1177/1060028018799501. [DOI] [PubMed] [Google Scholar]
- 95.Khan BA, Lasiter S, Boustani MA. Critical care recovery center: an innovative collaborative care model for ICU survivors. Am J Nurs. 2015;115:24–31. doi: 10.1097/01.NAJ.0000461807.42226.3e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Flash MJE, Johnson SF, Nguemeni Tiako MJ, et al. Disparities in post-intensive care syndrome during the COVID-19 pandemic: challenges and solutions. NEJM Catal. 2020 https://catalyst.nejm.org/doi/pdf/10.1056/CAT.20.0568 published online Nov 13. [Google Scholar]
- 97.Hart JL, Turnbull AE, Oppenheim IM, Courtright KR. Family-centered care during the COVID-19 era. J Pain Symptom Manage. 2020;60:e93–e97. doi: 10.1016/j.jpainsymman.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Schmidt K, Gensichen J, Gehrke-Beck S, et al. Management of COVID-19 ICU-survivors in primary care: a narrative review. BMC Fam Pract. 2021;22:160. doi: 10.1186/s12875-021-01464-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Schmidt K, Gehrke-Beck S. In: Improving critical care survivorship: a guide for prevention, recovery, and reintegration. Haines KJ, McPeake J, Sevin CM, editors. Springer Verlag; 2021. Transitions to primary care; pp. 207–227. [Google Scholar]
- 100.Pan D, Sze S, Minhas JS, et al. The impact of ethnicity on clinical outcomes in COVID-19: a systematic review. EClinicalMedicine. 2020;2:04. doi: 10.1016/j.eclinm.2020.100404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Ferrando-Vivas P, Doidge J, Thomas K, et al. Prognostic factors for 30-day mortality in critically ill patients with coronavirus disease 2019: an observational cohort study. Crit Care Med. 2021;49:102–111. doi: 10.1097/CCM.0000000000004740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.National Institute for Health Research Improving inclusion of under-served groups in clinical research: guidance from the NIINCLUDE project. Aug 8, 2020. https://www.nihr.ac.uk/documents/improving-inclusion-of-under-served-groups-in-clinical-research-guidance-from-include-project/25435
- 103.Bhatt J, Bathija P. Ensuring access to quality health care in vulnerable communities. Acad Med. 2018;93:1271–1275. doi: 10.1097/ACM.0000000000002254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Pantoja T, Opiyo N, Lewin S, et al. Implementation strategies for health systems in low-income countries: an overview of systematic reviews. Cochrane Database Syst Rev. 2017;9:CD011086. doi: 10.1002/14651858.CD011086.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Pandian JD, Kalkonde Y, Sebastian IA, Felix C, Urimubenshi G, Bosch J. Stroke systems of care in low-income and middle-income countries: challenges and opportunities. Lancet. 2020;396:1443–1451. doi: 10.1016/S0140-6736(20)31374-X. [DOI] [PubMed] [Google Scholar]
- 106.Page KR, Flores-Miller A. Lessons we’ve learned — covid-19 and the undocumented Latinx community. N Engl J Med. 2021;384:5–7. doi: 10.1056/NEJMp2024897. [DOI] [PubMed] [Google Scholar]
- 107.Stokes EK, Zambrano LD, Anderson KN, et al. Coronavirus disease 2019 case surveillance — United States, January 22–May 30, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:759–765. doi: 10.15585/mmwr.mm6924e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Semler MW, Bernard GR, Aaron SD, et al. Identifying clinical research priorities in adult pulmonary and critical care: NHLBI Working Group Report. Am J Respir Crit Care Med. 2020;202:511–523. doi: 10.1164/rccm.201908-1595WS. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Prescott HC, Girard TD. Recovery from severe COVID-19: leveraging the lessons of survival from sepsis. JAMA. 2020;324:739–740. doi: 10.1001/jama.2020.14103. [DOI] [PubMed] [Google Scholar]
- 110.Mikkelsen ME, Still M, Anderson BJ, et al. Society of Critical Care Medicine's international consensus conference on prediction and identification of long-term impairments after critical illness. Crit Care Med. 2020;48:1670–1679. doi: 10.1097/CCM.0000000000004586. [DOI] [PubMed] [Google Scholar]
- 111.Schofield-Robinson OJ, Lewis SR, Smith AF, McPeake J, Alderson P. Follow-up services for improving long-term outcomes in intensive care unit (ICU) survivors. Cochrane Database Syst Rev. 2018;11:CD012701. doi: 10.1002/14651858.CD012701.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Hope AA, Johnson AA, McPeake J, et al. Establishing a peer support program for survivors of COVID-19: a report from the Critical and Acute Illness Recovery Organization. Am J Crit Care. 2021 doi: 10.4037/ajcc2021675. published online Feb 15. [DOI] [PMC free article] [PubMed] [Google Scholar]