To the Editor:
The underlying mechanisms leading to bronchiectasis symptoms and exacerbations are poorly understood. Recent data suggest that daily symptoms such as cough and sputum are linked to airway bacterial load and corresponding neutrophilic inflammation (1–3). In contrast, recent data have also suggested that bronchiectasis exacerbations arise because of changes in the interactions between pathogens in the airway microbiome that may be disrupted by viral infections (4). Respiratory viruses can be identified during exacerbations in up to 50% of patients with bronchiectasis (5). The coronavirus disease (COVID-19) pandemic resulted in the introduction of social distancing and mitigation measures that have reduced person-to-person interactions worldwide (6). This has reduced the circulation of respiratory viruses such as influenza and rhinovirus, which are commonly identified in exacerbations of bronchiectasis (5). The COVID-19 pandemic therefore represents a “natural experiment” to test the hypothesis that many bronchiectasis exacerbations are related to external exposures whereas daily chronic symptoms such as cough and sputum are more “intrinsic.” In this study, we therefore hypothesized that social distancing during 2020 would be associated with reduced reported exacerbations but no change in the chronic symptoms typically experienced during stable state.
We performed a prospective observational study embedded within the EMBARC (European Multicentre Bronchiectasis Audit and Research Collaboration) registry of patients with computed tomography–confirmed bronchiectasis who were enrolled at Ninewells Hospital in Dundee, United Kingdom. Patients were enrolled between June 2019 and February 2020 as part of a study to validate a novel patient-reported outcome measure (the Bronchiectasis Impact Measure [BIM]) that has recently been reported (Ethical Approval Number: 19/NW/03/64) (7).
An amendment to the protocol was made at the onset of the COVID-19 pandemic to administer additional BIM and “Quality of Life Questionnaire–Bronchiectasis” questionnaires during the UK’s first lockdown period as a measure of symptoms during a period of social distancing. COVID-19 data collection for symptoms took place from May to July 2020. Patient symptoms during lockdown were compared with those from their most recent stable prelockdown symptom questionnaire. All questionnaires were performed while patients were clinically stable and free from exacerbation. Exacerbation frequency from 23 March 2018 to 22 March 2019, from 23 March 2019 to 22 March 2020, and from 23 March 2020 to 22 March 2021 was recorded through the EMBARC registry (8). Dates were selected so that the 2020/2021 period started with the beginning of the first national lockdown in the UK. Exacerbations were reported by patients and verified by prescription for antibiotics. Symptom and exacerbation data were not normally distributed and so are compared using a Wilcoxon paired signed rank test. Negative binomial regression was used to model exacerbation rates over time.
We included 173 patients in the original study. Nineteen patients were lost to follow-up, and seven patients died, resulting in the 147 patients that are included in the present analysis. The median (interquartile range) age was 70 (64–75) years, and 84 (57.1%) patients were female. The mean baseline FEV1 value was 84.0% predicted (SD, 28.4). The median bronchiectasis severity index score was 6 (4–9). Sixty-four (43.5%) patients had Haemophilus influenzae chronic infection, and 25 (17.0%) patients had Pseudomonas aeruginosa chronic infection. Of the patients, 82.1% reported that they were “shielding” during the pandemic. “Shielding” was used to describe recommended additional protective measures encouraged in the UK for people who were at high risk and extremely vulnerable, including leaving their homes as seldom as possible and minimizing all person-to-person contact. Only two patients in the cohort had PCR-confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection.
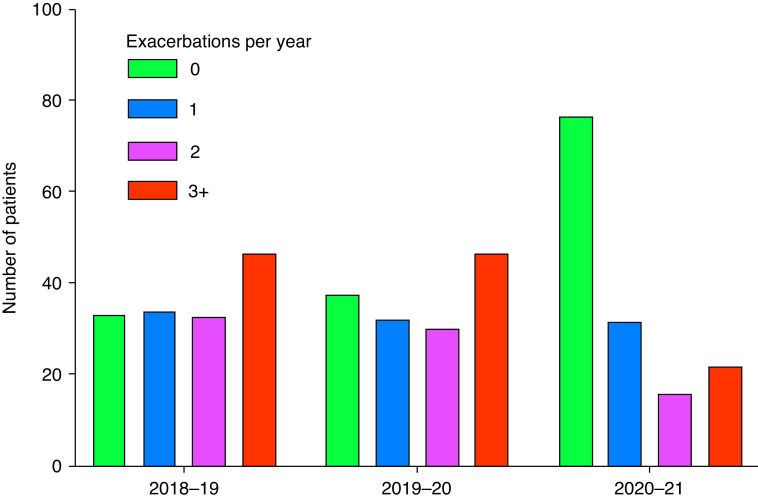
There was a statistically significant reduction in the frequency of reported exacerbations during the lockdown period. The number of exacerbations per patient per year was 2.08 in 2018/2019, 2.01 in 2019/2020, and 1.12 in 2020/2021. Figure 1 shows that the number of patients experiencing no exacerbations over a 12-month period increased from 22.4% in 2018/2019 and 25.6% in 2019/2020 to 52.3% in 2020/2021. Paired data between years was compared using the Wilcoxon matched pair signed rank test, demonstrating a significant reduction in exacerbations between 2020/2021 and both 2018/2019 and 2019/2020 (P < 0.0001 for both comparisons). The proportion of patients experiencing a hospitalization due to severe exacerbation was 8.8%, which was lower than 14.3% and 16.3% in the 2 previous years, respectively. We analyzed which clinical parameters were associated with continuing to have exacerbations during 2020/2021 in a negative binomial model, and significant associations were found for prior exacerbation frequency (rate ratio [RR], 1.20; 95% confidence interval [CI], 1.07–1.35; P = 0.002) and chronic P. aeruginosa infection (RR, 1.78; 95% CI, 1.01–3.14; P = 0.047).
Figure 1.
Absolute number of patients experiencing 0, 1, 2, and 3+ exacerbations per year during the 3 years of observation (n = 147 total patients for each year).
After adjusting for prior exacerbation history, patients with more severe symptoms were more likely to experience exacerbations during 2020/2021, including sputum production (RR, 1.14; 95% CI, 1.05–1.24; P = 0.002), dyspnea (RR, 1.11; 95% CI, 1.02–1.21; P = 0.018), tiredness (RR, 1.14; 95% CI, 1.04–1.24; P = 0.004), activity (RR, 1.13; 95% CI, 1.03–1.23; P = 0.007), overall health (RR, 1.11; 95% CI, 1.02–1.21; P = 0.022), and control (RR, 1.12; 95% CI, 1.03–1.22; P = 0.006). The only domain that was not significantly associated with exacerbations was cough (RR, 1.08; 95% CI, 0.99–1.18; P = 0.07).
Comparing patients’ symptoms using the BIM before and during lockdown, we found no significant differences in the impact of cough, sputum, dyspnea, tiredness, activity, overall health, control, and exacerbations on patient quality of life (Table 1). This was also noted in the “Quality of Life Questionnaire–Bronchiectasis” questionnaire, in which no significant change was seen in the respiratory symptoms score that was completed alongside the BIM at each time point. It should be noted that the BIM questionnaire asks about the impact of exacerbations on quality of life, not the frequency of exacerbations.
Table 1.
Change in Disease Impact during the Prepandemic Period and the Pandemic Period
| Questionnaire | Symptom | Before Lockdown | Lockdown | P Value |
|---|---|---|---|---|
| BIM | Cough | 4.0 (1.1–7.0) | 3.1 (1.0–7.0) | 0.19 |
| Sputum | 3.7 (1.0–7.0) | 3.8 (1.0–7.0) | 0.89 | |
| Dyspnea | 5.0 (1.5–75) | 4.9 (2.0–7.5) | 0.19 | |
| Tiredness | 6.0 (2.0–8.0) | 5.0 (2.6–8.0) | 0.79 | |
| Activity | 4.0 (1.0–8.0) | 4.0 (1.0–7.0) | 0.67 | |
| Overall health | 3.0 (1.0–7.0) | 4.0 (1.0–7.0) | 0.31 | |
| Control | 3.0 (1.0–7.0) | 3.0 (1.0–7.0) | 0.95 | |
| Impact of exacerbations | 3.0 (1.0–8.0) | 3.9 (1.0–7.0) | 0.82 | |
| Quality of Life Bronchiectasiis Questionnaire | Respiratory symptoms score | 70.4 (55.6–81.5) | 70.4 (51.9–81.5) | 0.56 |
Definition of abbreviation: BIM = Bronchiectasis Impact Measure.
Patients’ most recent stable BIM questionnaire and their “lockdown” questionnaire performed during June/July 2020 are compared. Symptom data are presented as median (interquartile range). P values were calculated by Wilcoxon paired signed-rank test.
In summary, bronchiectasis exacerbation frequency had a markedly greater reduction between March 2020 and March 2021 than it did during the same time period in the prior 2 years. Respiratory symptoms were unchanged from the prepandemic period to the pandemic period. Our data support a key role for external environmental factors in the pathogenesis of bronchiectasis exacerbations. Although a reduction in circulating viruses is the most likely reason for reduced exacerbation frequency (5), there are other potential contributors to our findings, including reductions in traffic-related air pollution, which were documented during the lockdown periods (9). Although reduced access to health care—or avoidance of healthcare contacts by patients—is a potential alternative explanation for our findings, this seems less likely because access to primary and secondary care were largely maintained in the region via virtual appointments during the study period. We might also expect that, if patients had exacerbations that were not treated, we would see a deterioration in symptoms or an increase in severe exacerbations requiring hospitalization, neither of which was observed. Results of our study are consistent with observations in other diseases, such as chronic obstructive pulmonary disease, for which exacerbation frequency has decreased during lockdown periods (10). Our study has limitations, including a single-center design, its relatively small sample size, and the lack of data on viruses at exacerbation to confirm the mechanism of exacerbation reduction. Our study also has unique strengths in that we could perform symptom and exacerbation assessments in a standardized fashion within a cohort established before the pandemic.
In summary, social distancing measures during the first 12 months of the COVID-19 pandemic were associated with a marked reduction in bronchiectasis exacerbations but no change in individual chronic respiratory symptoms.
Footnotes
Supported by the Innovative Medicines Initiative and The European Federation of Pharmaceutical Industries and Associations companies under the European Commission–funded Horizon 2020 Framework Program and by Inhaled Antibiotic for Bronchiectasis and Cystic Fibrosis (grant 115721), the European Respiratory Society through the EMBARC2 (European Multicentre Bronchiectasis Audit and Research Collaboration) consortium, and the United States Bronchiectasis Research Registry/COPD Foundation. EMBARC2 is supported by project partners AstraZeneca, Chiesi, Grifols, Janssen, Insmed, Novartis, and Zambon. J.D.C. is supported by the GlaxoSmithKline/British Lung Foundation through his role as Chair of Respiratory Research.
Author Contributions: Conception and design: M.L.C., A.S., and J.D.C.; analysis and interpretation: M.L.C., A.S., and J.D.C.; drafting the manuscript for important intellectual content: M.L.C., A.S., and J.D.C.
Originally Published in Press as DOI: 10.1164/rccm.202105-1137LE on July 15, 2021
Author disclosures are available with the text of this letter at www.atsjournals.org.
References
- 1. Crichton ML, Lonergan M, Barker AF, Sibila O, Goeminne P, Shoemark A, et al. Inhaled aztreonam improves symptoms of cough and sputum production in patients with bronchiectasis: a post hoc analysis of the AIR-BX studies. Eur Respir J. 2020;56:2000608. doi: 10.1183/13993003.00608-2020. [DOI] [PubMed] [Google Scholar]
- 2. Sibila O, Laserna E, Shoemark A, Keir HR, Finch S, Rodrigo-Troyano A, et al. Airway bacterial load and inhaled antibiotic response in bronchiectasis. Am J Respir Crit Care Med. 2019;200:33–41. doi: 10.1164/rccm.201809-1651OC. [DOI] [PubMed] [Google Scholar]
- 3.Gao Y-H, Abo Leyah H, Finch S, Lonergan M, Aliberti S, De Soyza A, et al. Relationship between symptoms, exacerbations, and treatment response in bronchiectasis. Am J Respir Crit Care Med. 2020;201:1499–1507. doi: 10.1164/rccm.201910-1972OC. [DOI] [PubMed] [Google Scholar]
- 4. Mac Aogáin M, Narayana JK, Tiew PY, Ali NABM, Yong VFL, Jaggi TK, et al. Integrative microbiomics in bronchiectasis exacerbations. Nat Med. 2021;27:688–699. doi: 10.1038/s41591-021-01289-7. [DOI] [PubMed] [Google Scholar]
- 5. Gao Y-H, Guan W-J, Xu G, Lin ZY, Tang Y, Lin ZM, et al. The role of viral infection in pulmonary exacerbations of bronchiectasis in adults: a prospective study. Chest. 2015;147:1635–1643. doi: 10.1378/chest.14-1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Prem K, Liu Y, Russell TW, Kucharski AJ, Eggo RM, Davies N, et al. Centre for the Mathematical Modelling of Infectious Diseases COVID-19 Working Group. The effect of control strategies to reduce social mixing on outcomes of the COVID-19 epidemic in Wuhan, China: a modelling study. Lancet Public Health. 2020;5:e261–e270. doi: 10.1016/S2468-2667(20)30073-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Crichton ML, Dudgeon EK, Shoemark A, Chalmers JD. Validation of the Bronchiectasis Impact Measure (BIM): a novel patient-reported outcome measure. Eur Respir J. 2021;57:2003156. doi: 10.1183/13993003.03156-2020. [DOI] [PubMed] [Google Scholar]
- 8. Chalmers JD, Aliberti S, Polverino E, Vendrell M, Crichton M, Loebinger M, et al. The EMBARC European Bronchiectasis Registry: protocol for an international observational study. ERJ Open Res. 2016;2:00081-2015. doi: 10.1183/23120541.00081-2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Goeminne PC, Cox B, Finch S, Loebinger MR, Bedi P, Hill AT, et al. The impact of acute air pollution fluctuations on bronchiectasis pulmonary exacerbation: a case-crossover analysis. Eur Respir J. 2018;52:1702557. doi: 10.1183/13993003.02557-2017. [DOI] [PubMed] [Google Scholar]
- 10. Tan JY, Conceicao EP, Wee LE, Sim XYJ, Venkatachalam I. COVID-19 public health measures: a reduction in hospital admissions for COPD exacerbations. Thorax. 2021;76:512–513. doi: 10.1136/thoraxjnl-2020-216083. [DOI] [PubMed] [Google Scholar]