Abstract
Purpose of review
Here, we review recent progress made on the genetic characterization of Giardia duodenalis assemblages and their relationship with virulence. We also discuss the implications of virulence factors in the pathogenesis of giardiasis, and advances in the development of vaccines and drugs based on knowledge of virulence markers.
Recent findings
The use of transcriptomic and proteomic technologies as well as whole genome sequencing (WGS) from single cysts has allowed the assembly of the draft genome sequences for assemblages C and D of G. duodenalis. These findings, along with the published genomes for assemblages A, B, and E, have allowed comparative genomic investigations. In addition, the use of these methodologies for the characterization of the secretomes of trophozoite-epithelial cell interactions for assemblages A/B has led to the identification of virulence markers including energy metabolism enzymes, proteinases, high-cysteine membrane proteins (HCMPs), and variant surface proteins (VSPs). Recently, some drugs and vaccines, targeting virulence factors have been developed, offering possible alternatives to current treatment and prevention options against giardiasis.
Summary
Among the nine recognized species of Giardia, G. duodenalis stands out because of its broad spectrum of hosts and its socio-economic importance. This species comprises eight genetic assemblages (A to H), of which A and B are zoonotic, and the other assemblages have narrow host specificities. Assemblages A and B may be considered as the most virulent ones, but the existence of asymptomatic carriers and considerable genetic variability within and among these assemblages hampers the definition of common virulence factors. The attachment of Giardia trophozoites to epithelial cells and structural cytoskeleton components of the adhesive disk, such as giardins or tubulins, is proposed to play key roles, but toxins have not yet been precisely defined. However, recent transcriptomic and proteomic analyses of the secretomes of trophozoites representing assemblages A and B and interacting with particular epithelial cell lines have defined a series of virulence factors, including glycolytic (e.g., enolase) and arginolytic (e.g., arginine deiminase) enzymes, cysteine proteases (e.g., giardipain-1) and VSPs (e.g., VSP9B10A). Other factors, such as HCMPs and tenascins, have been consistently found to be excreted/secreted, but their role(s) in the pathogenesis of giardiasis has not yet been elucidated. Interestingly, recent investigations of single cysts representing assemblages C and D using advanced sequencing and informatic methods have suggested that the transcription/expression profiles of virulence factors vary both within and between assemblages, thus assemblage-specific molecules might allow adaptation to the microenvironment within the host. Importantly, some drugs active against cysteine-rich proteins of Giardia, including giardipain-1, VSPs and arginine deiminase, have been shown to be targeted by cysteine-modifying compounds as disulfiram, L-canavanin and allicin. On the other hand, VSPs are presently considered as key vaccine candidates because they induce protection against Giardia in rodents and dogs. Overall, this review reveals that much more work is needed to identify, characterize, and understand the roles of virulence factors in Giardia and to assess their validity as drug and vaccine targets. Clear, advanced omics and informatic tools should assist in this future endeavor, with a focus on targeting virulence factors that are common and/or unique to distinct assemblages to develop new and effective interventions against Giardia.
Keywords: Giardia, Virulence, Pathogenicity, Host–pathogen interaction, Apoptosis
Introduction
Giardiasis is a leading cause of intestinal infection in humans and vertebrates caused by members of the protistan genus Giardia. In humans, the clinical manifestation of giardiasis caused by Giardia duodenalis (syn. G. intestinalis, G. lamblia) varies from acute to chronic and from symptomatic to asymptomatic; current estimates indicate that ~ 184 million people are affected annually; ~ 1 billion people are at risk of disease; and that up to 30% of people are infected in disadvantaged countries [1, 2]. The genus Giardia includes early-diverging eukaryotes (~ 2.2 billion years ago; [3]) and highly evolved organisms among diplomonads with a parasitic life-style and marked genetic variation among species and assemblages/genotypes that reflects their adaptation to, and co-evolution with hosts. This diversity has progressively been unveiled from the 1990s through the use of PCR–RFLP-based sequencing of regions within the small subunit of the nuclear ribosomal RNA gene [SSU-rDNA], the internal transcribed spacer regions [ITS1-2]), and the genes encoding metabolic (glutamate dehydrogenase [GDH] and triose phosphate isomerase [TPI]) or cytoskeletal (β-giardin) proteins [4–7]. Currently, nine distinct species of Giardia are recognized, characterized based on morphogenetic criteria, of which four predominantly parasitize mammals with a restricted host range (G. muris in rodents, G. microti in rodents and voles, G. cricetidarum in hamsters, G. paramelis in quendas), two species infect birds (G. ardae in herons and G. psittaci in parrots, parakeets and cockatoos), whereas G. agilis is found in amphibians and G. varani in lizards.
Of these species, G. duodenalis stands out because of its ability to infect a broad range of vertebrates (mammals, birds, and reptiles); multi-locus ‘genotyping’ has identified eight recognized genetic lineages, designated “assemblages A to H”, within additional variants (subtypes or genotypes) that have been identified. Most assemblages have narrow host ranges, which contrasts the situation for G. duodenalis, which is now increasingly being considered as a complex of species [8•, 9]. Assemblages C and D (proposed to represent G. canis) are found in canines and related animals (dogs, foxes, coyotes and seals), whereas assemblage E (G. bovis) has been recorded in domestic artiodactyls (including sheep, goat, pig, cattle and water buffaloes), assemblage F (G. cati) in felids, and assemblage G (G. simondi) in rodents. The assemblage H has been characterized from grey/harbor seals and a gull by sequencing of the gdh gene, but, interestingly, it falls outside the phylogenetic cluster containing all other assemblages [10, 11]. By contrast, assemblages A (G. duodenalis, subtypes A1–A5) and B (G. enterica, subtypes BI-BIV) have most genetic diversity and host range, and, hence, are considered as zoonotic, although assemblage A is more frequently found in livestock, companion and wild animals than assemblage B. However, epidemiological studies of humans indicate a higher global prevalence of assemblage B, particularly in Eurasia, while assemblage A predominates in the Americas. This geographic pattern seems to be the same for wild non-human primates [11, 12]. Therefore, it is conceivable that assemblages A and B could parasitize primates, including gorillas and chimpanzees, which diverged (~ 7–8 million years ago), long before the emergence and migration of Homo sapiens (~ 0.2 million years ago). Thus, it is possible that these species disseminated assemblages A and B worldwide. The main, current controversy regarding the taxonomy G. duodenalis is whether these two assemblages represent sub-species or distinct species [9, 13]. Future research should tackle this issue.
Giardia Assemblages and Virulence
In recent years, an emphasis has been placed on the use of advanced technologies including whole genome sequencing (WGS), cytometry sorting-based single cell WGS and exome capture [14] for samples with few cysts or unavailable trophozoite cultures. These approaches have been used to identify and differentiate genotypes (subtypes or assemblages) of G. duodenalis, and explore assemblage-host relationships [9], infections caused by multiple assemblages in a single host [8•, 9–12, 14], transmission dynamics between humans and companion, domestic and wildlife animal species. Comparative genomics and proteomics have been used to define markers for parasite virulence and host susceptibility for Giardia assemblages [8•, 9, 11]. In this latter context, virulence refers to the ability of “lower” infectious agents to infect and cause a degree of damage to “higher” host animals; and pathogenicity refers to the ability of the infectious agent to cause loss of fitness (i.e. disease) in the host animal. Pathogenicity is determined by an agent´s virulence factors, i.e. structures, molecules and/or mechanisms that contribute to the colonization of a niche; such factors relate to pathogen attachment/invasion, immune evasion, immunosuppression, entry/exit from host cells or destruction of host molecules within tissues or fluids required for a pathogen´s nutrition and invasiveness.
Clearly, the virulence of Giardia varies depending on the genomic background of each genotype/assemblage and the host species it infects, and various parasite- and host-dependent factors need to be considered. If the zoonotic G. duodenalis assemblages A and B are the “most” virulent in humans, comparative omics between assemblages A, B, and E should reveal the presence of assemblage-specific proteins. However, higher similarity between amino acid sequences of assemblages A and E, along with a higher phylogenetic relation among assemblages A-E–F and B-C-D [11, 15, 16], have been reported, supporting the proposal that assemblages A and B are separate species. Transmission dynamics studies are needed to explore this proposal. Also in this context, distinct assemblages parasitizing the same host species may exhibit variable pathogenicity. For instance, in studies of human giardiasis [17, 18], different proportions of assemblages A and B have been observed in relation to the disease outcome (symptomatic vs. asymptomatic, diarrheal vs. non-diarrheal and acute vs. chronic). Further, in spite of the fact that assemblage B and subtype BIII have been associated with diarrheal episodes, a clear correlation between Giardia genotype and clinical spectrum of giardiasis is still lacking [19–21].
Understanding virulence in Giardia has implications for the treatment and/or control of giardiasis, and could aid vaccine and drug development. For protists other than Giardia (e.g., Blastocystis, Cryptosporidium), immunization with attenuated strains/lineages with reduced virulence can induce some protection against challenge infection (reviewed in [22]), but, to date, this has not been achieved for Giardia. In studies undertaken, trophozoites with an impaired fitness (i.e. low attachment ability) obtained by treatment with nitrosoguanidine-colchicine [23] or by induction of resistance to metronidazole (and other drugs) [24] have reduced ability to infect rodents. Supported by the completion of draft genomes for Giardia assemblages A–E [25••], structural and molecular factors that play a role in the pathogenicity of giardiasis have been identified at the RNA and protein levels. These virulence factors are discussed in the following section.
An Arsenal of Virulence Factors in Giardia Trophozoites
The ability of Giardia to perpetuate infection in a host relies on critical steps driven by host stimuli. Following ingestion, the ovoid infective cyst undergoes excystation in the gastric-duodenal milieu to generate four trophozoites. These adhere to the epithelial cells using their adhesive disk, allowing the colonization of the duodenum and jejunum by the parasite. In the distal small intestine, exposure to primary bile salts, fatty acids and an alkaline pH trigger, encystation followed by nuclear replication giving rise to mature infective cysts, which are excreted in the faeces and survive in stools for up to several weeks, contaminating water and food.
First, cysts of all Giardia assemblages excyst under the influence of hydrochloric acid secretion in the gastric fluid of all extant fishes, amphibians, reptiles, birds and mammals, likely because this process emerged ~ 350 million years ago [26]. Second, excysted trophozoites of each assemblage are adapted to and able to develop and replicate (“grow”) in the intestinal microenvironment within their susceptible host(s). This is readily inferred from: (a) the intrinsic growth ability of assemblages in standard TYI-S-33 medium (A > B > > E > C and D), where substitutions such as L-glutathione for L-cysteine allow the growth of cattle-derived assemblages and canine bile plus horse serum, instead of their bovine analogues, promote the growth of canine-derived trophozoites [27, 28]; (b) the low percentage of orthologous gene groups (3288/5508, 59.7%) shared by assemblages A through E, with the highest assemblage-specific orthologous groups (620/5508, 14.7%) being present in the zoonotic subtype AI (WB strain); and (c) the obvious presence/absence of several molecules in some assemblages — e.g., assemblages C and D lack flavohaemoglobin, an antioxidant response enzyme that counteract NO production by epithelial cells. [25••]. Third, the presence of similar types of bile salts (considered as cholesterol-sequestering agents that trigger encystation) among evolutionarily related vertebrates [29] supports the notion that parasite lineages have specifically adapted to the composition of host intestinal milieu to produce viable, infective cysts — as supported by in vitro studies [28]. Hence, the metabolic adaptation of Giardia lineages to their cognate hosts likely drives their virulence, and promotes the pathogenic potential of lumen-resident trophozoites against host intestinal epithelium through the expression and action of virulence factors.
Interactions between Giardia trophozoites and epithelial cells, including attachment enabled by cytoskeleton-rich structures and the expression and/or excretion/secretion of molecules that alter intestinal homeostasis, are key factors. On the one hand, the attachment of trophozoites to the microvillus border of intestinal epithelium is largely mediated by the ventral disk, a highly specialized structure composed of an internal spiral array of microtubules with polymerised αβ-tubulins, bridged by several types of annexin-like proteins called giardins, and a peripheral crest rich in other contractile proteins made up mainly of actin [30, 31]. Of the four pairs of flagella in a trophozoite, containing tubulin (9 + 2 arrays) and actin microtubules, the two ventrolateral pairs act to maintain hydrodynamic forces at the interface of the ventral disk and epithelial cell surface, allowing parasite attachment [30, 31]. On the other hand, parasites exposed to the host cell surface then excrete/secrete several soluble factors which are referred to as “major” virulence factors. The effect of these virulence factors on the host lead to the pathogenesis of disease, which link to the appearance of the clinical signs/symptoms of giardiasis.
Excreted/secreted virulence factors contribute to impair epithelial barrier function and to programmed cell death (apoptosis) in enterocytes. Several of these factors have been proposed [32••]. Toxin-like molecules were proposed to be secreted by G. duodenalis trophozoites of the WB strain (assemblage A1), of which CRP136 (VSP88-like, containing sarafotoxin-like repeats) has been proposed to interact with type A endothelial receptors to promote intestinal hypermotility [32••, 33]. In addition, the 58 kDa-sized glycoprotein gESP58 from strain P-1 of assemblage A has been reported to interact with the ganglioside M1, as does the subunit B of cholera toxin, triggering similar signal transduction pathways that induce the increase in Ca2+ and decreased Cl− levels in the enterocyte cytosol, leading to intestinal dehydration and hypermotility [34, 35] (Fig. 1.1). In spite of the cytotoxic and cytopathic effects of the latter two molecules, they have not been conclusively identified in the secretome of trophozoites interacting with epithelial cells.
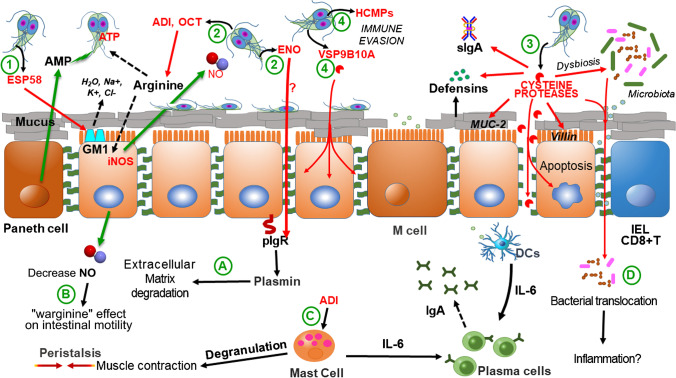
Fig. 1.
Role of Giardia duodenalis virulence factors in pathogenicity. After excystation, trophozoites attach to intestinal epithelium and they adapt to the host intestinal microenvironment where they release a series of molecules with different reported effects. ESP58, a toxin-like protein recognizing a surface receptor in enterocytes (GM1) promotes water and electrolyte leakage (1). Energy metabolism enzymes have been detected in secretomes, including arginolytic enzymes such as gADI and gOCT that compete for arginine with intestinal cells, and glycolytic enzymes such as gENO that bioinformatics approaches indicate that it interacts with human plasminogen (2). Cysteine proteinases such as GL50803_16160, GL50803_16779 and giardipain-1 which degrade MUC2, villin, defensins and immunoglobulins; in microbiota biofilms these proteinases cause its disaggregation, alters microbiota composition (dysbiosis) and facilitate bacterial translocation (3). High-cysteine surface proteins such as HCMPs that are involved in immune evasion and VSPs such as VSP9B10A that display cysteine proteinase activity and have a cytotoxic effect on epithelial cells (4). These virulence factors have some effects at the intraepithelial level which include gENO-mediated conversion of plasminogen into plasmin that promotes extracellular matrix degradation (A), a “warginine” effect on intestinal motility through decreased in NO production (B), muscle contraction involving IgE-independent degranulation of mast cells by gADI (C), and bacterial translocation facilitated by giardial cysteine proteases that could promote inflammatory responses as IBD (D). Modified from [22]
Recent studies using transcriptomic and proteomic approaches have shown that Giardia overexpresses and secretes molecules whose profiles during interaction with intestinal epithelial cells or murine intestinal loops differ among Giardia genotypes. For example, up to 196 and 155 proteins were detected, respectively, in the secretomes of the strains WB and GS interacting with mouse intestinal IEC cells by liquid chromatography-gas spectrometry (LC–MS) analyses [36]. Of these molecules, metabolic enzymes, proteinases, cysteine-rich surface proteins and tenascins have been reported.
Typically, metabolic enzymes are proteins involved in intracellular energy metabolism in G. duodenalis. The main enzymes detected either in soluble or extracellular microvesicle (EMV)-bound forms [36] might have an extracellular role in competing for glucose/arginine with host epithelium and even over the hemostatic system. In the glycolytic cascade of Giardia, fructose bis-phosphate aldolase (gFBPA), glyceraldehyde-3-phosphate dehydrogenase (gG3PDH), phosphoglycerate kinase (gPGK), phosphoglucomutase (gPGM) and enolase (gENO) have been found to be either up-regulated or detected in the secretome of the human-infective assemblages A and B [37–39]. In particular, the secretion of gFBPA, gPGM and gENO is enhanced upon interaction of trophozoites with human colorectal CaCo2 monolayers [39]. In other parasitic protists and helminths, these enzymes display a role in immunomodulation, recognition and/or attachment [40–42], but their precise roles in one or more of these processes is still unknown for Giardia. Nevertheless, some insights have been gained through a study of gENO, suggesting that, in its dimer form, it could activate human plasminogen (HsPLS) to plasmin [43], thereby promoting cytotoxic and cytopathic effects through extracellular matrix degradation (Fig. 1.A). In fact, gENO has been found as the predominant glycolytic enzyme in secretomes during interaction of trophozoites with CaCo2 cells and murine intestinal loops [37].
Regarding the enzymes in the arginine dehydrolase pathway secreted by trophozoites interacting with model epithelial cell lines (e.g., HT29 and CaCo2), these may have a role in which has been referred as “warginine”, i.e. the competition for arginine between parasites and host intestinal cells. Arginine deiminase (gADI) and ornithine carbamoyl transferase (gOCT) were reported as major arginolytic components in secretomes of Giardia [36, 37] (Fig. 1.2). Of interest, these enzymes form the catabolic axis arginine → citrulline → ornithine [46] and gADI and an arginine/ornithine antiport transporter in trophozoite surface (GL50803_92223 in WB strain) may contribute to arginine consumption and arginine uptake, respectively. Thus avoiding NO production by epithelial cells at giardicidal levels which in turn promotes increased intestinal motility and parasite elimination [47–50] (Fig. 1.B). To date, although the extracellular role of gOCT is unclear, there is experimental evidence that gADI might promote TNFα production via immune cells. The evidence indicates that human-derived dendritic cells (DCs) stimulated with bacterial LPS/gADI overexpress TNFα, as do rat mast cells in the presence of gADI in an IgE-independent fashion that reinforces the effect of arginine and citrulline [51, 52] (Fig. 1.C) This would suggest that an uncontrolled production of TNFα produces damage to epithelial barrier function [51, 53].
Another dominant group of enzymes present in the secretome of G. duodenalis trophozoites are the proteinases, among which cysteine proteinases are relevant due to their role in the sulfur metabolism of Giardia and other parasitic protists including Entamoeba and Trichomonas [54]. Cathepsins, particularly of the B type, are major members encoded in the genome of assemblages A to E, with variant repertoires. Up to 85 cathepsin-B sequences have been identified and classified into nine clades. Of these, clades 1–6 contain members of each assemblage and orthologs display limited variation, while clades 7, 8 and 9 are more diverse. For example, clades 7 and 9 contain only orthologues of assemblages A and B, while clade 8 includes orthologues of dog assemblages, but lacked those of assemblages A and E [25••]. This suggests that selection and host specificity influence their expression. A restricted repertoire of these cathepsins has been found to be released by trophozoites interacting with epithelial cells [55, 56] (Fig. 1.3). In the assemblage A/B – CaCo2 interaction system, some cathepsin B-like proteinases were identified in the secretomes in two independent proteomic studies [36, 57]. Of these, cathepsins B from assemblage A1 (GL50803_16160, clade 4; _14019, clade 5; _16779, clade 6) exhibit some differences in substrate specificity and optimum pH [58]. This information indicates that these secreted paralogs, along with their catalytic competence reach the extracellular level by pro-domain excision. This is exemplified by GL50803_14019, formerly known as CP2 and designated giardipain-1 [59] that may serve to broaden the repertoire of host proteins targeted by these proteinases. Indeed, these kinds of enzymes are directly involved in virulence of Giardia and pathogenesis of giardiasis due to their effects at multiple levels on the intestinal homeostasis in the host.
A first barrier degraded by giardial cysteine proteases is the mucus layer covering the intestinal epithelial cell surface by targeting MUC2. This has been shown in mucus-producing cell lines and Muc2 − / − mice, together with other effects such as goblet cell enlargement, thinner mucus layer and bacterial translocation [60]. The microvilli protein Villin was shown to be degraded and redistributed in CaCo2 cells through the action of surface-exposed cysteine proteases of trophozoites from assemblages A or B [61]. Furthermore, proteins from intercellular, apical junctional complexes, particularly those in tight (claudins, occludins, ZO proteins) and adherent (E-cadherin, β-cathenin) junctions have been shown to be affected by giardial cysteine proteinases and by each of the three aforementioned proteinases [36, 58, 59]. In the case of giardipain-1, the purified enzyme promotes classical hallmarks of apoptosis in epithelial cell lines, including IEC6 and MDCK [59]. Beyond their cytotoxic effect, the same set of cysteine proteinases can affect immune responses, as they degrade immunoglobulins IgA1, IgA2 and IgG [62]. Human defensins αDf1, βDf5 and αDf6 are also degraded by these enzymes, with giardipain-1 showing the highest activity [62]. In the case of cytokines/chemokines, these are upregulated during trophozoite-epithelial cell interactions and include CXCL1-3, CXCL8, CCL2 and CCL20 [51]. Of these, CXCL3 was not degraded by giardipain-1 and GL50803_16779 [58]. Interestingly, during Giardia-microbiota interactions, a role for cysteine proteinases has been demonstrated in the helminth model of Caenorhabditis elegans, which dies by paralysis when exposed to human microbiota exposed to Giardia-derived excretory/secretory products (ESP) [63]. Also, cysteine proteinases in ESP cause biofilm disruption and dysbiosis in human-derived planktonic (commensal) bacteria when transferred to microbiota-free mice. Upon biofilm release these became a pathobiont, causing bacterial translocation, apoptosis and overexpression of pro-inflammatory receptors and cytokines in intestinal cells [64, 65] (Fig. 1.D). These observations seem to be related to the appearance of post-giardiasis sequelae, consistent with irritable bowel syndrome (PI-IBD) [65, 66].
Trophozoites interacting with epithelial cells also secrete cysteine-rich, surface-exposed proteins that are classified into two groups. These include high-cysteine membrane proteins (HCMPs, with at least 61 members classified into 9 groups) that are presumably secreted by recruitment to peripheral vesicles [67, 68] and variant surface proteins (VSPs, ~ 300 members classified into 3 clades) (Fig. 1.4). Interestingly, > 10 HCMPs may be concomitantly secreted, whereas VSP expression is tightly suppressed by RNA interference in an “all-but-one” fashion. Thus, a predominant VSP is usually secreted in a given population by excision of its CRGKA tail [69, 70]. Both groups have common and assemblage-specific members. Using proteomics approaches, up to 3 or 5 distinct HCMPs and one VSP (GL50581_3195, VSP160) have been detected in secretomes of WB or GS-CaCo2 cell interactions [36, 57], revealing a variable HCMP/VSP repertoire in parasite secretome, and this also occurs with isolates belonging to the same assemblage [58]. Further studies are needed to analyze individual members of these protein families to gain insights into their role at the host-parasite interface. An example is the VSP9B10A (also known as VSP88) which was detected in the proteolytic secretome in WB-IEC6 cocultures. Its activity was found to be related to the presence of a cathepsin B-like central region with the classical catalytic triad Cys-His-Asn similar to other member, such as VSP8, but with a substrate specificity likely distinct from the cysteine proteinases of clades 4, 5 and 6 — described above. However, VSP9B10A-transfected trophozoites were able to induce cell detachment in IEC6 monolayers, suggesting a degradation of tight/adherent junctions [71]. This information provides new perspectives on the functional plasticity of the VSPs. Based on the VSPs strong regulation of expression, the VSP9B10A protein can be defined as a conditional virulence factor.
A last group of proteins secreted by G. duodenalis trophozoites upon interaction with model intestinal cells are tenascins, which are involved in intestinal tissue repair processes. These polymeric proteins have four paralogous groups (C, R, X and W) and contain several types of repetitive elements including epidermal growth factor (EGF)-like, fibronectin III-like and an N-terminal fibrinogen-like domains [72]. The abundance of these proteins varies among assemblages: 10 in WB (assemblage A), 9 in GS (B) and 11 in P15 (E) strains. In Giardia secretomes, tenascin C-like (GL50803_8687) and tenascin X-like (GL50803_14573) have been detected [73]. In other studies, distinct tenascins identified have been designated as GL50803_10330/GL50581_4057 [36] and up to seven tenascins [57] have been identified using MS/MS methods. To date, it has been proposed that giardial tenascins may ligate EGF receptors which become accessible following mucus layer degradation in intestinal epithelial cells by activities of cysteine proteinases and an extracellular nuclease of Giardia. This interaction could induce epithelium disruption that would facilitate bacterial translocation and even pro-inflammatory responses. Clearly, further experimental studies are needed to rigorously test this hypothesis.
Virulence Factors as Targets for Giardiasis Intervention
As virulence factors of Giardia include structural and excreted/secreted elements involved in the colonization of the intestinal epithelium, in trophozoite-epithelial cell interactions, in the evasion of host immune responses and/or in the uptake of nutrients for growth, these factors might be targets for vaccine and drug development. Indeed, agents that block or inhibit these factors could interrupt parasite life-cycle and pathogenicity of Giardia.
Regarding drug development, structural proteins such as α-tubulin have been found as target of former anti-cancer agents, such as NBDHEX and oryzalin (a dinitroanilide herbicide) [74, 75]. The other component of giardial microtubules, β-tubulin, is inhibited by benzimidazole carbamates, of which albendazole and mebendazole are well known anti-giardial agents used in the clinical practice [76]. Importantly, such compounds cause ventral disk disassembly (albendazole) or flagella retraction (oryzalin) [75, 76]. In the case of cysteine-rich surface proteins (VSPs and HCMPs), only the acetaldehyde dehydrogenase inhibitor disulfiram (Antabuse™) has been proposed as a likely VSP inhibitor on the basis of the screening of selected Zinc finger-binding drugs [77]. Cysteine proteases have specific inhibitors. Amog these, the fungal epoxide E64 and its analogue CA-074 have been used to identify the multiple roles of Giardia cathepsins B in the pathogenesis of giardiasis. However, these compounds lack a defined therapeutic profile. Allicin, a diallyl thiosulfinate, extracted from garlic bulbs, is able to inhibit in vitro secreted proteinases, such as giardipain-1 and possibly other cysteine proteases, as well as VSPs and HCMPs. This is due to the thiol-disulfide conversion of cysteine residues in these biomolecules by this thiosulfinate, thereby blocking is cytophatogenic effects [59]. The potential therapeutic utility of Allicin has already been tested in Giardia-infected Mongolian gerbils, where this compound reduced and even eliminated trophozoites [78].
Regarding virulence factors related to micronutrient competition with intestinal cells, of which warginine is the most studied, the availability of solved crystal structures for gOCT (PDB ID: 3GRF) and gCK (PDB ID: #KZF) have been useful for studies of structure-based drug design. Notably, the differences between OCTs from human and Giardia have allowed the design of specific inhibitors [79]; of these, disulfiram has been shown to inhibit gCK at IC50 = 0.58 µM and to kill trophozoites in culture at LC50 = 0.90 µM [80]. In the case of the multifunctional gADI, cysteine-acting compounds, including arginine analogues such as as L-canavanine, display reversible inhibition, and others, such as S-nitroso-L-homocysteine, are irreversible inhibitors [81, 82]. Nevertheless, the potential toxicity of L-canavanine in mice and humans [83] needs to be addressed before further evaluation.
Regarding vaccine development against Giardia using virulence factors as immunogens, VSPs have been tested using genetically engineered trophozoites expressing multiple VSPs via inactivation of elements of the RNAi machinery. When gerbils were infected and then reinfected with such trophozoites animals shed reduced cyst numbers in the faeces [85]. The administration of Giardia expressing multiple VSPs in cats and dogs caused a self-limiting infection lasting ~ 40 days and appeared to induce resistance to infections with clonal populations expressing a single VSP that otherwise caused persistent infections. Furthermore, the use of an oral vaccine containing immunopurified VSPs from WB strain in infected dogs, under endemic conditions, caused not only reduction in cyst shedding, but also a decrease in the symptoms of giardiasis [86]. In gerbils, this protective effect related to increased levels of IL-4, IL-5, IL-6, IL-17, sIgA and IgG [87]. As reference, other potential vaccines using vectors, such as Salmonella typhimurium expressing gOCT or gENO, have shown a protective effect only for gENO [88].
Taken together, these experimental insights suggest that the cysteine-rich nature of several virulence factors in G. duodenalis (cysteine-proteinases, HCMPs, VSPs, ADI) may be exploited to find new or repurpose some existing drugs as antigiardial agents (e.g., disulfiram, allicin, S-nitroso-L-homocysteine), and to define immunogens (e.g. VSPs) to achieve high-level protective host immune responses. Developing new drugs and vaccines will have the advantage of circumventing existing problems with drug resistance in Giardia against commonly used compounds (e.g., metronidazole and albendazole) with cysteine-dependent mechanisms of action [84].
Concluding Remarks and Future Perspectives
In the ‘Giardia/giardiasis’ field, the terms virulence and pathogenicity need to be interpreted with some caution given the complexity and range of clinical outcomes associated with known genetic variation linked to eight known Giardia assemblages as well as the genetic background, nutritional status and immunological responses in host animals. However, the molecular characterization of these assemblages has been possible using advanced molecular and informatic technologies, including shotgun sequencing (assemblages A, B and E) and single cell-based sequencing approaches (assemblages D and E), which has allowed the characterization of genomes of select assemblages. These sequencing data reveal the presence of molecules that are unique to such assemblages or shared among them. An important observation is the fact that Giardia from each assemblage displays virulence in particular host species, possibly due to their ability to metabolically adapt to particular host microenvironments, guaranteeing the survival and replication of Giardia, irrespective of assemblage. Further, the pathogenic arsenal of each assemblage might retain differences, mainly in terms of protein structure, but having paralogues of similar function(s), reflecting co-evolution with host species. In particular, assemblages A and B seem to be two separate species, and their zoonotic potential is the broadest, possibly due to the often extensive migration of Homo sapiens and their interactions with most extant species around the world.
Giardia possesses several virulence markers, of which toxins have yet to be conclusively identified and characterized. However, structural elements of the ventral disk and flagellar cytoskeleton, in particular α- and β-tubulins, have been partially exploited for drug development. Indeed, the benzimidazole drug albendazole is known as one of the more effective antigiardial agents in the clinical practice. Among secreted virulent factors are some glycolytic and arginolytic enzymes that still need to be evaluated in greater detail, since a limited number of compounds has been proposed to inactivate gENO, gOCT, gCK or gADI, and the value of immunogens needs to be critically assessed. On the other hand, proteins associated with sulfur- and cysteine-rich metabolism in Giardia and that are surface-anchored or secreted — namely HCMPs and more noticeably cysteine proteinases and VSPs — have been studied as drug targets using compounds including disulfiram or allicin and, more recently, auranofin as promising new antigiardial agents. In addition, the use of Giardia expressing multiple VSPs (live vaccine) and purified VSPs (‘cocktail’ vaccine) may induce protection, not only in animals, but also in humans when evaluated in future clinical trials.
In conclusion, G. duodenalis “virulence” and “pathogenicity” are beginning to be understood. However, much more work needs to be undertaken to gain a deep understanding of assemblages (F, G and H). This will be achieved using advanced molecular, genomic and functional tools as well as in vitro and in vivo assays/models, in order to define particular patterns of metabolic adaptations to particular host species and to identify virulence markers that are unique to or shared by particular assemblages. It will also be important to define variability in the expression and regulation of such markers within individual assemblages, and explore how these aspects relate to pathogenicity. “To be, or not to be virulent “…. that´s still a big question for Giardia! Clearly, much more detailed work using advanced tools and resources is required to fully elucidate the processes and mechanisms linked to virulence and pathogenicity in this fascinating and complex protistan group.
Acknowledgements
We are immensely grateful to Robin B Gasser for his comments, technical and language editing of the manuscript. We are grateful to Arturo Pérez-Taylor for the art work. We would like to acknowledge support from Fondo SEP-Cinvestav Mexico Grant number FIDSC2018/95 and from Consejo Nacional de Ciencia y Tecnología (SEP-CONACYT) Mexico, Grant number A1-S-39422.
Compliance with Ethical Standards
Conflict of interest
Raúl Argüello-García and M. Guadalupe Ortega-Pierres declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Footnotes
This article is part of the Topical Collection on Giardia/Crypto
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
- 1.Kirk MD, Pires SM, Black RE, Caipo M, Crump JA, Devleesschauwer B, Dopfer D, Fazil A, Fischer-Walker CL., Hald T, Hall AJ., Keddy KH, Lake RJ., Lanata CF., Torgerson PR, Havelaar AH, and Angulo FJ. (2010) World Health Organization estimates of the global and regional disease burden of 22 foodborne bacterial, protozoal, and viral diseases: a data synthesis. PLoS Med. 2015 Dec;12(12):e1001921. [DOI] [PMC free article] [PubMed]
- 2.Einarsson E, Ma’ayeh S, Svard SG. (2016) An up-date on Giardia and giardiasis. Curr Opin Microbiol. Dec;34:47–52. [DOI] [PubMed]
- 3.Hedges SB, Chen H, Kumar S, Wang DY, Thompson AS, Watanabe H. A genomic timescale for the origin of eukaryotes. BMC Evol Biol. 2001;1:4. doi: 10.1186/1471-2148-1-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hopkins RM, Meloni BP, Groth DM, Wetherall JD, Reynoldson JA, Thompson RC.J Ribosomal RNA sequencing reveals differences between the genotypes of Giardia isolates recovered from humans and dogs living in the same locality. Parasitol. 1997 Feb;83(1):44–51 [PubMed]
- 5.Cacciò SM, De Giacomo M, Pozio E. Sequence analysis of the beta-giardin gene and development of a polymerase chain reaction-restriction fragment length polymorphism assay to genotype Giardia duodenalis cysts from human faecal samples. Int J Parasitol. 2002;32(8):1023–1030. doi: 10.1016/s0020-7519(02)00068-1. [DOI] [PubMed] [Google Scholar]
- 6.Sulaiman IM, Fayer R, Bern C, Gilman RH, Trout JM, Schantz PM, Das P, Lal AA, Xiao L. Triosephosphate isomerase gene characterization and potential zoonotic transmission of Giardia duodenalis. Emerg Infect Dis. 2003;9(11):1444–1452. doi: 10.3201/eid0911.030084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Read CM, Monis PT, Thompson RC. Discrimination of all genotypes of Giardia duodenalis at the glutamate dehydrogenase locus using PCR-RFLP. Infect Genet Evol. 2004;4(2):125–130. doi: 10.1016/j.meegid.2004.02.001. [DOI] [PubMed] [Google Scholar]
- 8.Cacciò SM, Lalle M, Svärd SG. Host specificity in the Giardia duodenalis species complex. Infect Genet Evol. 2018;66:335–345. doi: 10.1016/j.meegid.2017.12.001. [DOI] [PubMed] [Google Scholar]
- 9.Thompson RCA, Ash A. Molecular epidemiology of Giardia and Cryptosporidium infections — what's new? Infect Genet Evol. 2019;75:103951. [DOI] [PubMed]
- 10.Lasek-Nesselquist E, Welch DM, Sogin ML. The identification of a new Giardia duodenalis assemblage in marine vertebrates and a preliminary analysis of G. duodenalis population biology in marine systems. Int J Parasitol. 2010;40:1063–1074. doi: 10.1016/j.ijpara.2010.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Feng Y, Xiao L. Zoonotic potential and molecular epidemiology of Giardia species and giardiasis. Clin Microbiol Rev. 2011;24(1):110–140. doi: 10.1128/CMR.00033-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cacciò SM, Ryan U. Molecular epidemiology of giardiasis. Mol Biochem Parasitol. 2008;160(2):75–80. doi: 10.1016/j.molbiopara.2008.04.006. [DOI] [PubMed] [Google Scholar]
- 13.Emery-Corbin SJ, Vuong D, Lacey E, Svärd SG, Ansell BRE, Jex AR. Proteomic diversity in a prevalent human-infective Giardia duodenalis sub-species. Int J Parasitol. 2018;48(11):817–823. doi: 10.1016/j.ijpara.2018.05.003. [DOI] [PubMed] [Google Scholar]
- 14.Capewell P, Krumrie S, Katzer F, Alexander CL, Weir W. Molecular epidemiology of Giardia infections in the genomic era. Trends Parasitol. 2020;S1471–4922(20):30256–30257. doi: 10.1016/j.pt.2020.09.013. [DOI] [PubMed] [Google Scholar]
- 15.Thompson RC, Monis P. Giardia—from genome to proteome. Adv Parasitol. 2012;78:57–95. doi: 10.1016/B978-0-12-394303-3.00003-7. [DOI] [PubMed] [Google Scholar]
- 16.Jerlström-Hultqvist J, Ankarklev J, Svärd SG. Is human giardiasis caused by two different Giardia species? Gut Microbes. 2010;1(6):379–82. [DOI] [PMC free article] [PubMed]
- 17.Read C, Walters J, Robertson ID, Thompson RC. Correlation between genotype of Giardia duodenalis and diarrhoea. Int J Parasitol. 2002;32(2):229–231. doi: 10.1016/s0020-7519(01)00340-x. [DOI] [PubMed] [Google Scholar]
- 18.Viesy S, Abdi J, Haghani K, Valizadeh R, Mirzaei A. Giardia lamblia subtypes and their relationship with clinical symptoms in patients with giardiasis. Infect Disord Drug Targets. 2020;20(3):396–400. doi: 10.2174/1871526519666190314094437. [DOI] [PubMed] [Google Scholar]
- 19.Minetti C, Lamden K, Durband C, Cheesbrough J, Fox A, Wastling JM. Determination of Giardia duodenalis assemblages and multi-locus genotypes in patients with sporadic giardiasis from England. Parasit Vectors. 2015;4(8):444. doi: 10.1186/s13071-015-1059-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hussein EM, Ismail OA, Mokhtar AB, Mohamed SE, Saad RM. Nested PCR targeting intergenic spacer (IGS) in genotyping of Giardia duodenalis isolated from symptomatic and asymptomatic infected Egyptian school children.Parasitol Res. 2017 Feb;116(2):763–771. [DOI] [PubMed]
- 21.Ma DW, Lee MR, Hong SH, Cho SH, Lee SE. Molecular prevalence and genotypes of Cryptosporidium parvum and Giardia duodenalis in patients with acute diarrhea in Korea, 2013–2016. Korean J Parasitol. 2019;57(5):531–536. doi: 10.3347/kjp.2019.57.5.531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Argüello-García R, Carrero J, Ortega-Pierres G. Host immune responses against intestinal unicellular parasites and their role in pathogenesis and protection. Encyclopedia of Infection and Immunity: Elsevier; 2020. [Google Scholar]
- 23.Hernández-Sánchez J, Liñan RF, Salinas-Tobón Mdel R, Ortega-Pierres G. Giardia duodenalis: adhesion-deficient clones have reduced ability to establish infection in Mongolian gerbils. Exp Parasitol. 2008;119(3):364–372. doi: 10.1016/j.exppara.2008.03.010. [DOI] [PubMed] [Google Scholar]
- 24.Tejman-Yarden N, Millman M, Lauwaet T, Davids BJ, Gillin FD, Dunn L, Upcroft JA, Miyamoto Y, Eckmann L. Impaired parasite attachment as fitness cost of metronidazole resistance in Giardia lamblia. Antimicrob Agents Chemother. 2011;55(10):4643–4651. doi: 10.1128/AAC.00384-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.•• Kooyman FNJ, Wagenaar JA, Zomer A. Whole-genome sequencing of dog-specific assemblages C and D of Giardia duodenalis from single and pooled cysts indicates host-associated genes. Microb Genom. 2019;5 (12):e000302. This work reports the use of whole genome sequencing (WGS) in samples from canine origin in which very few cysts were used for genome sequencing that was done with a coverage of >95%. The information described in this report not only broaden the genome drafts of Giardia assemblages but provides a platform to perform further studies on comparative genomics aimed to identify assemblage-common and asemblage-specific proteins with a focus in pathogenesis and virulence. [DOI] [PMC free article] [PubMed]
- 26.Koelz HR. Gastric acid in vertebrates. Scand J Gastroenterol Suppl. 1992;193:2–6. doi: 10.3109/00365529209095998. [DOI] [PubMed] [Google Scholar]
- 27.Bénéré E, Geurden T, Robertson L, Van Assche T, Cos P, Maes L. Infectivity of Giardia duodenalis assemblages A and E for the gerbil and axenisation of duodenal trophozoites. Parasitol Int. 2010;59(4):634–637. doi: 10.1016/j.parint.2010.08.001. [DOI] [PubMed] [Google Scholar]
- 28.Tysnes KR, Robertson LJ. Establishment of canine-derived Giardia duodenalis isolates in culture. J Parasitol. 2016;102(3):342–348. doi: 10.1645/15-910. [DOI] [PubMed] [Google Scholar]
- 29.Hofmann AF, Hagey LR, Krasowski MD. Bile salts of vertebrates: structural variation and possible evolutionary significance. J Lipid Res. 2010;51(2):226–246. doi: 10.1194/jlr.R000042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Elmendorf HG, Dawson SC, McCaffery JM. The cytoskeleton of Giardia lamblia. Int J Parasitol. 2003;33(1):3–28. doi: 10.1016/s0020-7519(02)00228-x. [DOI] [PubMed] [Google Scholar]
- 31.Nosala C, Dawson SC. The critical role of the cytoskeleton in the pathogenesis of Giardia. Curr Clin Microbiol Rep. 2015;2(4):155–162. doi: 10.1007/s40588-015-0026-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ortega-Pierres MG, Argüello-García R. Giardia duodenalis: role of secreted molecules as virulent factors in the cytotoxic effect on epithelial cells. Adv Parasitol. 2019;106:129–169. doi: 10.1016/bs.apar.2019.07.003. [DOI] [PubMed] [Google Scholar]
- 33.Shahbazian A, Holzer P. Regulation of guinea pig intestinal peristalsis by endogenous endothelin acting at ET(B) receptors. Gastroenterology. 2000;119(1):80–88. doi: 10.1053/gast.2000.8549. [DOI] [PubMed] [Google Scholar]
- 34.Shant J, Ghosh S, Bhattacharyya S, Ganguly NK, Majumdar S. The alteration in signal transduction parameters induced by the excretory-secretory product from Giardia lamblia. Parasitology. 2004;129(Pt 4):421–430. doi: 10.1017/s0031182004005852. [DOI] [PubMed] [Google Scholar]
- 35.Shant J, Ghosh S, Bhattacharyya S, Ganguly NK, Majumdar S. Mode of action of a potentially important excretory–secretory product from Giardia lamblia in mice enterocytes. Parasitology. 2005;131(Pt 1):57–69. doi: 10.1017/s0031182005007262. [DOI] [PubMed] [Google Scholar]
- 36.Ma’ayeh SY, Liu J, Peirasmaki D, Hörnaeus K, Bergström Lind S, Grabherr M, Bergquist J, Svärd SG. Characterization of the Giardia intestinalis secretome during interaction with human intestinal epithelial cells: the impact on host cells. PLoS Negl Trop Dis. 2017 Dec 11;11(12):e0006120. [DOI] [PMC free article] [PubMed]
- 37.Ringqvist E, Palm JE, Skarin H, Hehl AB, Weiland M, Davids BJ, Reiner DS, Griffiths WJ, Eckmann L, Gillin FD, Svärd SG. Release of metabolic enzymes by Giardia in response to interaction with intestinal epithelial cells. Mol Biochem Parasitol. 2008;159(2):85–91. doi: 10.1016/j.molbiopara.2008.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ringqvist E, Avesson L, Söderbom F, Svärd SG. Transcriptional changes in Giardia during host-parasite interactions. Int J Parasitol. 2011;41(3–4):277–285. doi: 10.1016/j.ijpara.2010.09.011. [DOI] [PubMed] [Google Scholar]
- 39.Ma’ayeh SY, Brook-Carter PT. Representational difference analysis identifies specific genes in the interaction of Giardia duodenalis with the murine intestinal epithelial cell line, IEC-6. Int J Parasitol. 2012;42(5):501–509. doi: 10.1016/j.ijpara.2012.04.004. [DOI] [PubMed] [Google Scholar]
- 40.Collingridge PW, Brown RW, Ginger ML. Moonlighting enzymes in parasitic protozoa. Parasitology. 2010;137(9):1467–1475. doi: 10.1017/S0031182010000259. [DOI] [PubMed] [Google Scholar]
- 41.Gomez-Arreaza A, Acosta H, Quinones W, Concepcion JL, Michels PA, Avilan L. Extracellular functions of glycolytic enzymes of parasites: unpredicted use of ancient proteins. Mol Biochem Parasitol. 2014;193(2):75–81. doi: 10.1016/j.molbiopara.2014.02.005. [DOI] [PubMed] [Google Scholar]
- 42.Hewitson, J.P., Ruckerl, D., Harcus, Y., Murray, J., Webb, L.M., Babayan, S.A., Allen, J.E., Kurniawan, A., Maizels, R.M., 2014. The secreted triose phosphate isomerase of Brugia malayi is required to sustain microfilaria production in vivo. PLoS Pathog. 10 (2), e1003930. [DOI] [PMC free article] [PubMed]
- 43.Aguayo-Ortiz R, Meza-Cervantez P, Castillo R, Hernández-Campos A, Dominguez L, Yépez-Mulia L. Insights into the Giardia intestinalis enolase and human plasminogen interaction. Mol Biosyst. 2017;13(10):2015–2023. doi: 10.1039/c7mb00252a. [DOI] [PubMed] [Google Scholar]
- 44.Halliez MC, Buret AG. Extra-intestinal and long term consequences of Giardia duodenalis infections. World J Gastroenterol. 2013;19(47):8974–8985. doi: 10.3748/wjg.v19.i47.8974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Allain, T., Amat, C.B., Motta, J.P., Manko, A., Buret, A.G., 2017. Interactions of Giardia sp. with the intestinal barrier: epithelium, mucus, and microbiota. Tissue Barriers 5 (1), e1274354. [DOI] [PMC free article] [PubMed]
- 46.Schofield PJ, Costello M, Edwards MR, O’Sullivan WJ. The arginine dihydrolase pathway is present in Giardia intestinalis. Int J Parasitol. 1990;20(5):697–699. doi: 10.1016/0020-7519(90)90133-8. [DOI] [PubMed] [Google Scholar]
- 47.Banik S, Renner Viveros P, Seeber F, Klotz C, Ignatius R, Aebischer T. Giardia duodenalis arginine deiminase modulates the phenotype and cytokine secretion of human dendritic cells by depletion of arginine and formation of ammonia. Infect Immun. 2013;81(7):2309–2317. doi: 10.1128/IAI.00004-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Eckmann L, Laurent F, Langford TD, Hetsko ML, Smith JR, Kagnoff MF, Gillin FD. Nitric oxide production by human intestinal epithelial cells and competition for arginine as potential determinants of host defense against the lumen-dwelling pathogen Giardia lamblia. J Immunol. 2000;164(3):1478–1487. doi: 10.4049/jimmunol.164.3.1478. [DOI] [PubMed] [Google Scholar]
- 49.Stadelmann, B., Merino, M.C., Persson, L., Svard, S.G., 2012. Arginine consumption by the intestinal parasite Giardia intestinalis reduces proliferation of intestinal epithelial cells. PLoS One 7 (9), e45325. [DOI] [PMC free article] [PubMed]
- 50.Stadelmann B, Hanevik K, Andersson MK, Bruserud O, Svard SG. The role of arginine and arginine-metabolizing enzymes during Giardia—host cell interaction in vitro. BMC Microbiol. 2013;13:256. doi: 10.1186/1471-2180-13-256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Roxström-Lindquist K, Ringqvist E, Palm D, Svärd S. Giardia lamblia-induced changes in gene expression in differentiated Caco-2 human intestinal epithelial cells. Infect Immun. 2005;73(12):8204–8208. doi: 10.1128/IAI.73.12.8204-8208.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Muñoz-Cruz S, Gomez-García A, Matadamas-Martínez F, Alvarado-Torres JA, Meza-Cervantez P, Arriaga-Pizano L, Yépez-Mulia L. Giardia lamblia: identification of molecules that contribute to direct mast cell activation.Parasitol Res. 2018 Aug;117(8):2555–2567. [DOI] [PubMed]
- 53.Troeger H, Epple HJ, Schneider T, Wahnschaffe U, Ullrich R, Burchard GD, Jelinek T, Zeitz M, Fromm M, Schulzke JD. Effect of chronic Giardia lamblia infection on epithelial transport and barrier function in human duodenum. Gut. 2007;56(3):328–335. doi: 10.1136/gut.2006.100198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ali V, Nozaki T. Current therapeutics, their problems, and sulfur-containing-amino-acid metabolism as a novel target against infections by “amitochondriate” protozoan parasites. Clin Microbiol Rev. 2007;20(1):164–187. doi: 10.1128/CMR.00019-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jiménez JC, Uzcanga G, Zambrano A, Di Prisco MC, Lynch NR. Identification and partial characterization of excretory/secretory products with proteolytic activity in Giardia intestinalis. J Parasitol. 2000;86(4):859–862. doi: 10.1645/0022-3395(2000)086[0859:IAPCOE]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 56.Rodríguez-Fuentes GB, Cedillo-Rivera R, Fonseca-Liñán R, Argüello-García R, Muñoz O, Ortega-Pierres G, Yépez-Mulia L. Giardia duodenalis: analysis of secreted proteases upon trophozoite-epithelial cell interaction in vitro. Mem Inst Oswaldo Cruz. 2006;101(6):693–696. doi: 10.1590/s0074-02762006000600020. [DOI] [PubMed] [Google Scholar]
- 57.Dubourg A, Xia D, Winpenny JP, Al Naimi S, Bouzid M, Sexton DW, Wastling JM, Hunter PR, Tyler KM. Giardia secretome highlights secreted tenascins as a key component of pathogenesis. Gigascience. 2018;7(3):1–13. doi: 10.1093/gigascience/giy003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Liu J, Ma’ayeh S, Peirasmaki D, Lundström-Stadelmann B, Hellman L, et al. Secreted Giardia intestinalis cysteine proteases disrupt intestinal epithelial cell junctional complexes and degrade chemokines. Virulence. 2018;9:879–894. doi: 10.1080/21505594.2018.1451284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ortega-Pierres G, Argüello-García R, Laredo-Cisneros MS, Fonseca-Linán R, Gómez-Mondragón M, Inzunza-Arroyo R, Flores-Benítez D, Raya-Sandino A, Chavez-Munguía B, Ventura-Gallegos JL, Zentella-Dehesa A, Bermúdez-Cruz RM, González-Mariscal L. Giardipain-1, a protease secreted by Giardia duodenalis trophozoites, causes junctional, barrier and apoptotic damage in epithelial cell monolayers.Int J Parasitol. 2018 Jul;48(8):621–639. [DOI] [PubMed]
- 60.Amat CB, Motta JP, Fekete E, Moreau F, Chadee K, Buret AG. Cysteine protease-dependent mucous disruptions and differential mucin gene expression in Giardia duodenalis Infection. Am J Pathol. 2017;187(11):2486–2498. doi: 10.1016/j.ajpath.2017.07.009. [DOI] [PubMed] [Google Scholar]
- 61.Bhargava A, Cotton JA, Dixon BR, Gedamu L, Yates RM, Buret AG. Giardia duodenalis surface cysteine proteases induce cleavage of the intestinal epithelial cytoskeletal protein villin via myosin light chain kinase. PLoS One. 2015 Sep 3;10(9):e0136102. [DOI] [PMC free article] [PubMed]
- 62.Liu J, Fu Z, Hellman L, Svärd SG. Cleavage specificity of recombinant Giardia intestinalis cysteine proteases: degradation of immunoglobulins and defensins. Mol Biochem Parasitol. 2019;227:29–38. doi: 10.1016/j.molbiopara.2018.10.004. [DOI] [PubMed] [Google Scholar]
- 63.Gerbaba TK, Green-Harrison L, Buret AG. Modeling host-microbiome interactions in Caenorhabditis elegans. J Nematol. 2017;49(4):348–356. [PMC free article] [PubMed] [Google Scholar]
- 64.Barash NR, Maloney JG, Singer SM, Dawson SC. Giardia alters commensal microbial diversity throughout the murine gut. Infect Immun. 2017;85(6):e00948–e1016. doi: 10.1128/IAI.00948-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Beatty JK, Akierman SV, Motta JP, Muise S, Workentine ML, Harrison JJ, Bhargava A, Beck PL, Rioux KP, McKnight GW, Wallace JL, Buret AG. Giardia duodenalis induces pathogenic dysbiosis of human intestinal microbiota biofilms.Int J Parasitol. 2017 May;47(6):311–326. [DOI] [PubMed]
- 66.Hanevik K, Wensaas KA, Rortveit G, Eide GE, Mørch K, Langeland N. Irritable bowel syndrome and chronic fatigue 6 years after giardia infection: a controlled prospective cohort study. Clin Infect Dis. 2014;59(10):1394–1400. doi: 10.1093/cid/ciu629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Davids BJ, Reiner DS, Birkeland SR, Preheim SP, Cipriano MJ, McArthur AG, Gillin FD.A new family of giardial cysteine-rich non-VSP protein genes and a novel cyst protein. PLoS One. 2006 Dec 20;1(1):e44. [DOI] [PMC free article] [PubMed]
- 68.Peirasmaki D, Ma’ayeh SY, Xu F, Ferella M, Campos S, Liu J, Svärd SG. High Cysteine Membrane Proteins (HCMPs) are up-regulated during Giardia-Host Cell Interactions. Front Genet. 2020;18(11):913. doi: 10.3389/fgene.2020.00913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Prucca CG, Lujan HD. Antigenic variation in Giardia lamblia. Cell Microbiol. 2009;11(12):1706–1715. doi: 10.1111/j.1462-5822.2009.01367.x. [DOI] [PubMed] [Google Scholar]
- 70.Emery SJ, van Sluyter S, Haynes PA. Proteomic analysis in Giardia duodenalis yields insights into strain virulence and antigenic variation. Proteomics. 2014;14(21–22):2523–2534. doi: 10.1002/pmic.201400144. [DOI] [PubMed] [Google Scholar]
- 71.Cabrera-Licona A, Solano-González E, Fonseca-Liñán R, Bazán-Tejeda ML, Raúl Argüello-García, Bermúdez-Cruz RM, Ortega-Pierres G. Expression and secretion of the Giardia duodenalis variant surface protein 9B10A by transfected trophozoites causes damage to epithelial cell monolayers mediated by protease activity. Exp Parasitol. 2017 Aug;179:49–64. [DOI] [PubMed]
- 72.Hsia HC, Schwarzbauer JE. Meet the tenascins: multifunctional and mysterious. J Biol Chem. 2005;280(29):26641–26644. doi: 10.1074/jbc.R500005200. [DOI] [PubMed] [Google Scholar]
- 73.Emery SJ, Mirzaei M, Vuong D, Pascovici D, Chick JM, Lacey E, Haynes PA. Induction of virulence factors in Giardia duodenalis independent of host attachment. Sci Rep. 2016;12(6):20765. doi: 10.1038/srep20765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Camerini S, Bocedi A, Cecchetti S, Casella M, Carbo M, Morea V, Pozio E, Ricci G, Lalle M. Proteomic and functional analyses reveal pleiotropic action of the anti-tumoral compound NBDHEX in Giardia duodenalis. Int J Parasitol Drugs Drug Resist. 2017;7(2):147–158. doi: 10.1016/j.ijpddr.2017.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lyons-Abbott S, Sackett DL, Wloga D, Gaertig J, Morgan RE, Werbovetz KA. Naomi S. Morrissette α-tubulin mutations alter oryzalin affinity and microtubule assembly properties to confer dinitroaniline resistance Eukaryot Cell. 2010;9(12):1825–1834. doi: 10.1128/EC.00140-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.U M Morgan, J A Reynoldson, R C Thompson Activities of several benzimidazoles and tubulin inhibitors against Giardia spp. in vitro. Antimicrob Agents Chemother. 1993 Feb; 37(2): 328–331. [DOI] [PMC free article] [PubMed]
- 77.Nash T, Rice WG. Efficacies of zinc-finger-active drugs against Giardia lamblia. Antimicrob Agents Chemother. 1998;42(6):1488–1492. doi: 10.1128/aac.42.6.1488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Raúl Argüello-García, Mariana de la Vega-Arnaud, Iraís J. Loredo-Rodríguez, Adriana M. Mejía-Corona, Elizabeth Melgarejo-Trejo, Eulogia A. Espinoza-Contreras, Rocío Fonseca-Liñán, Arturo González-Robles, Nury Pérez-Hernández, M. Guadalupe Ortega-Pierres. Activity of thioallyl compounds from garlic against Giardia duodenalis trophozoites and in experimental giardiasis. Front Cell Infect Microbiol. 2018; 8: 353. Published online 2018 Oct 15. [DOI] [PMC free article] [PubMed]
- 79.Andrey Galkin, Liudmila Kulakova, Rui Wu, Maozhen Gong, Debra Dunaway-Mariano, Osnat Herzberg. X-ray structure and kinetic properties of ornithine transcarbamoylase from the human parasite Giardia lamblia. Proteins. Author manuscript; available in PMC 2010 Sep 1. Published in final edited form as: Proteins. 2009 Sep; 76(4): 1049–1053. Andrey Galkin, Liudmila Kulakova, Kap Lim, Catherine Z. Chen, Wei Zheng, Illarion V. Turko, Osnat Herzberg [DOI] [PMC free article] [PubMed]
- 80.Galkin A, Kulakova L, Lim K, Chen CZ, Zheng W, Turko IV, Herzberg O. Structural basis for inactivation of Giardia lamblia carbamate kinase by disulfiram. J Biol Chem. 2014;289(15):10502–10509. doi: 10.1074/jbc.M114.553123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Zhimin Li, Liudmila Kulakova, Ling Li, Andrey Galkin, Zhiming Zhao, Markus Knipp, Theodore E. Nash, Patrick S. Mariano, Osnat Herzberg, Debra Dunaway-Mariano. Mechanisms of catalysis and inhibition operative in the arginine deiminase from the human pathogen Giardia lamblia. Bioorg Chem. Author manuscript; available in PMC 2015 Oct 1.Published in final edited form as: Bioorg Chem. 2009 Oct; 37(5): [DOI] [PMC free article] [PubMed]
- 82.Justin E. Jones, Christina J. Dreyton, Heather Flick, Corey P. Causey, Paul R. Thompson. Mechanistic studies of Agmatine Deiminase from multiple bacterial species. Biochemistry. Author manuscript; available in PMC 2011 Nov 2.Published in final edited form as: Biochemistry. 2010 Nov 2; 49(43): 9413–9423. [DOI] [PMC free article] [PubMed]
- 83.Siyabulela S. B. N. Ntuli, Wentzel C. A. Gelderblom, David R. Katerere. The mutagenic and antimutagenic activity of Sutherlandia frutescens extracts and marker compounds. BMC Complement Altern Med. 2018; 18: 93. [DOI] [PMC free article] [PubMed]
- 84.Argüello-García R, Leitsch D, Skinner-Adams T, Ortega-Pierres MG. Drug resistance in Giardia: mechanisms and alternative treatments for Giardiasis. Adv Parasitol. 2020;107:201–282. doi: 10.1016/bs.apar.2019.11.003. [DOI] [PubMed] [Google Scholar]
- 85.Rivero FD, Saura A, Prucca CG, Carranza PG, Torri A, Lujan HD. Disruption of antigenic variation is crucial for effective parasite vaccine. Nat Med. 2010 May;16(5):551–7, 1p following 557. [DOI] [PubMed]
- 86.Serradell MC, Saura A, Rupil LL, Gargantini PR, Faya MI, Furlan PJ, Lujan HD. Vaccination of domestic animals with a novel oral vaccine prevents Giardia infections, alleviates signs of giardiasis and reduces transmission to humans. NPJ Vaccines. 2016;15(1):16018. doi: 10.1038/npjvaccines.2016.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Serradell MC, Gargantini PR, Saura A, Oms SR, Rupil LL, Berod L, Sparwasser T, Luján HD. Cytokines, antibodies, and histopathological profiles during Giardia infection and variant-specific surface protein-based vaccination. Infect Immun. 2018;86(6):e00773–e817. doi: 10.1128/IAI.00773-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Jenikova G, Hruz P, Andersson MK, Tejman-Yarden N, Ferreira PC, Andersen YS, Davids BJ, Gillin FD, Svärd SG, Curtiss R 3rd, Eckmann L.Α1-giardin based live heterologous vaccine protects against Giardia lamblia infection in a murine model. Vaccine. 2011 Nov 28;29(51):9529–37. doi: 10.1016/j.vaccine.2011.09.126. [DOI] [PMC free article] [PubMed]