Abstract

In this work, an alternative flame-retardant filler based on phosphate- and urea-grafted bamboo charcoal (BC-m) at 10–30 wt % addition was aimed at improving the flame retardancy of polylactic acid (PLA) composites. The filler caused only a small reduction in strength properties but a slight increase in the modulus of elasticity of PLA composites. BC-m significantly improved the flame-retardant performance compared with pure BC. The limiting oxygen index (LOI) was 28.0 vol % when 10 wt % of BC-m was added, and 32.1 vol % for 30 wt % addition, which was much greater than the value of 22.5 vol % for 30 wt % pure BC. Unlike pure BC, adding BC-m at 20 wt % or more gave a UL-94 vertical flame test rating of V-0 with significantly reduced melt dripping. The peak heat release rate (pHRR) and total heat release (THR) of BC-m/PLA composites decreased by more than 50% compared with pure PLA, and the values for 20% BC-m were significantly less than that for 25% BC addition. The grafted biochar-based system provides an effective flame retardancy effect by a condensed-phase protective barrier through the rapid formation of a dense, honeycomb-like cross-linked carbonized char layer. The results suggest a promising route to enhancing the flame-retardant properties of biodegradable polymer composites using nontoxic, more environmentally friendly grafted biochar.
1. Introduction
In the past few decades, hundreds of millions of tons of petroleum-based polymer plastics have been produced and used in a wide range of applications including packaging, with an annual growth rate of 20–30%.1 Most are not biodegradable and over time have become major land and marine pollutants with particularly serious negative impacts on marine life.2−5 Plastics used in disposable packaging need to transition to biobased and biodegradable polymers to reduce their environmental impact.6 Polylactic acid (PLA) is an established alternative biodegradable polymer with high processability, biocompatibility, and mechanical properties comparable to petroleum-based polymers and is extensively used in medical, food packaging, and other applications such as three-dimensional (3D) printing.7−10 However, its inherently biodegradable, fermented biomass-derived carbon–hydrogen chemical composition and structure give PLA lower heat and melt resistance when burned, which limits its use as a high-performance, long-lasting, durable polymer.11−14
There are several methods used to improve the flame retardancy of PLA, some of which use hazardous materials and/or reduce its biodegradability, making the development of greener, safer, effective flame retardants for PLA a research priority. Polymer-graft treatments form the basis of flame-retardant PLA, resulting in significant improvement of its flame-retardant performance.15−17 Other methods involve intumescent flame-retardant (IFR) additives that rely on an expansion catalyst (acid source), a carbon-rich agent, and an expansion agent.11 These reagents degrade with heating to form a stiff foamlike physical barrier of carbonized material or “char” (condensed-phase retardancy) during combustion that effectively insulates the inner material and blocks oxygen and heat transfer, hampering further combustion.18 In the IFR system, condensed-phase retardancy works in tandem with gas-phase flame retardancy, producing free radicals that help “scavenge” and dilute flammable gaseous products formed during polymer combustion.19 Several natural biomass materials have been developed for use as IFRs. For example, Casetta et al.18 developed an IFR composed of ammonium polyphosphate (APP), pentaerythritol (PER), lignin, and starch as a flame retardant for PLA. The flame retardancy of PLA was greatly improved at 40 wt % addition of this IFR.18 Wang et al.20 used β-cyclodextrin (β-CD) and poly(propylene glycol) (PPG) with APP and melamine (MA) to improve the flame retardancy of PLA. The limiting oxygen index (LOI) reached 34 vol %, and a UL-94 V-0 vertical flame test result was achieved.20 Starch has also been considered as a carbonizing agent in the IFR system and combined with phosphorous-containing flame retardants to improve the flame retardancy performance of foamed PLA.21 At 15–25 wt % addition of a starch-based IFR, the LOI of foamed PLA increased from 18.2 to 24.8–28.4 vol %.19
Many of these additive systems require high levels of addition for efficacy, often enough to significantly reduce the mechanical properties of the material.22 Certain flame retardants can reduce polymer properties, e.g., the hygroscopic behavior of APP.23 Others reduce the biodegradability of PLA, for example, PER, MA, and urea–formaldehyde resins, which are commonly used in expansion-type flame retardants.18,22 Newer synthetic polymeric flame retardants (PNFRs) and hyperbranched polyamine charring agents (HPCAs) can also affect the biodegradability of PLA.19,24−26 Further research efforts are needed to find the optimal balance between improved flame retardancy and the mechanical and biodegradation properties of PLA.27
Carbon-based agents such as carbon nanotubes (CNTs) and expanded graphite (EG) are considered to be excellent flame-retardant fillers by forming dense networks, but these are costly.28,29 Therefore, cheaper sources such as bamboo charcoal (BC) have also been investigated as flame-retardant additives for PLA.30 Functional flame retardants based on the chemical graft-modification of BC are in development,31,32 and BC is considered to have significant potential in flame retardancy.33,34 From previous research,35−37 BC has much better compatibility and compounding capacity with polymers than powdered bamboo (bamboo flour), which requires chemical modification (by alkali treatment or adding coupling agents) to prevent phase separation.37,38 Significant chemical functionalization is required to impart the flame-retardant capability to bamboo flour.33,38 Bamboo has tiny, complex pores, and the controlled pyrolysis and grinding process for BC give it greater specific surface area and a rich, irregular pore structure, potentially allowing it to become better integrated with graft products and the polymer matrix.39,40 Interaction of BC with the polymer interface has been shown to increase the crystallinity of the matrix by promoting heterogeneous nucleation and enhancing certain properties of the material, such as stiffness and rigidity.41,42 Using BC with a small particle size as a reinforcing agent in PLA, Ho et al.43 successfully created BC/PLA composites whose maximum tensile strength, flexural strength, and ductility index (DI) were increased by 43, 99, and 52%, respectively. Qian et al.44 found the maximum tensile strength and modulus of BC/PLA composites when 10% ultrafine, alkali-modified (15 wt % NaOH) BC was added. Tensile strength and modulus increased from 33.6 to 358.5 MPa of pure PLA to 65.4 and 1012.2 MPa, respectively.44
Although BC has a very low organic matter content and good thermal conductivity after high-temperature carbonization, its active contribution to flame retardancy is limited.35,45 Previous research35 found that the LOI of the BC/PLA (25/75 ratio) composite increased only slightly to 23.8 vol % from 22.0 vol % for pure PLA. After adding a combination of BC and AHP (25 and 30% of mix, respectively) to PLA, the total heat release (THR) of the BC/PLA/AHP composite decreased by 72.2%, and LOI reached 31 vol %. The flame retardancy of the material was significantly improved, indicating that coaddition of a purpose-designed flame retardant is necessary. A disadvantage of coadding AHP was that the total required amount of the filler and flame retardant significantly reduced the flexural properties of the PLA composite.35,45 Toxic gases such as PH3 were detected from the combustion of the composites containing AHP fire retardant,45 which can cause health and contamination concerns when the materials are exposed to fire.46,47
The objective of this follow-up study was, therefore, to develop and test a more sustainable BC-based flame-retardant grafted with nitrogen and phosphorus compounds. The hypothesis is phosphorous-grafted BC will impart better flame retardancy than pure BC based on our previous results with plain BC.35,45 Using grafted BC is also aimed at reducing the amounts of filler required so as to mitigate the negative effects on the mechanical properties of PLA. Furthermore, the formulation is designed to be nontoxic and not reduce the biodegradability of PLA.
2. Experimental Section
2.1. Materials
PLA powder was supplied by Cargill Dow Inc. (4032D, NatureWorks Co., Ltd., Blair, Nebraska) with the following key properties: density, 1.24 g/cm3; melting point, 160 °C; glass transition temperature, 57.8 °C; and crystallization peak temperature, 150–170 °C.
BC was supplied by Zhejiang Anji Huasen Bamboo Charcoal Products Co., Ltd. (Zhejiang, China). The BC was produced by pyrolyzing fresh Moso (Phyllostachys pubescens) bamboo in a brick kiln at a carbonization temperature of 700 to 800 °C, followed by grinding to a particle size of 100–300 mesh.
Chemical reagents used were as follows: hydrogen peroxide (H2O2) at 30% concn (Yonghua Chemical Reagent Co., Ltd., Jiangsu, China); phosphoric acid (PA) at 85% concn, urea at 99% purity, ethanol absolute at 99% purity with 0.789–0.791 g/mL density at 20 °C (all from Sinopharm Chemical Reagent Co., Ltd., Shanghai, China), and ammonium hydroxide solution at 3% concn and 0.91 g/mL density at 20 °C (Macklin Biochemical Co., Ltd., Zhejiang, China).
2.2. Preparation of the Fire-Retardant Mix
2.2.1. Modification and Grafting of BC
Approximately 50 g of BC powder was washed in deionized water for 4 h in an ultrasonic bath (DL720D, Wuxiang instrument Co., Ltd., Shanghai, China). After drying, two batches of 30% concn aqueous H2O2 solution were used to treat BC in succession. Using a BC/H2O2 ratio of 1:3, the BC was treated for 8 h. After filtration, H2O2 was added again, followed by 1 h of microwave irradiation at 200 W and oven drying at 90 °C overnight to produce a treated BC intermediate termed “BC-o”. The BC-o powder was reacted in a three-necked flask with PA at 85% concn using a 1:1 ratio to BC-o. The mixture was stirred at 80 °C for 30 min and then gradually heated further to 100 °C and maintained for 1 h. Urea dispersed in deionized water was added, giving a PA/urea ratio of 1:1.8, with 3% ammonia used to adjust the pH of the mixture to between 8 and 10. The mixture was heated slowly to 120 °C for 30 min and then gradually increased to 160 °C to generate a thick foam, which was then cured in an oven at 200 °C for 6 h. The hardened foam, termed “BC-m”, was ground to 100–300 mesh and washed several times with a hot ethanol solution and dried at 90 °C for 2 h.
2.2.2. Preparation of the Flame-Retardant PLA Composite
PLA resin, BC, and BC-m powders were oven-dried at 80 °C for 8 h and then compounded together using a twin-roll mixing mill (XK-160, Wuhan, China) at 180 °C. The rotation speed of the screw was set to 30 rpm. Three mix ratios of BC/PLA (controls) and BC-m/PLA composites were prepared, as shown in Table 1. A commercial PLA resin was also tested as the pure reference polymer.
Table 1. Formulations for PLA/BC-m Composites.
| composition (wt %) |
|||
|---|---|---|---|
| sample | PLA | BC | BC-m |
| PLA | 100.0 | 0 | 0 |
| BC/PLA1 | 90.0 | 10.0 | 0 |
| BC/PLA2 | 80.0 | 20.0 | 0 |
| BC/PLA3 | 70.0 | 30.0 | 0 |
| BC-m/PLA1 | 90.0 | 0 | 10.0 |
| BC-m/PLA2 | 80.0 | 0 | 20.0 |
| BC-m/PLA3 | 70.0 | 0 | 30.0 |
Compounded extruded mixes were cut into pellets and remixed in the mill until a visually good dispersion was achieved. The compounded pellets were hot-pressed at 180 °C and 10 MPa pressure for 10 min to form sheets (100 × 100 × 4 mm3) for CONE tests. The pellets were also injection molded (180 °C) into splines at a temperature of 170 °C and pressure of 6 MPa for mechanical tests of tensile and flexural properties. The appearance of all test specimens is shown in Figure 1. A schematic diagram of the preparation and test process is shown in Figure 2.
Figure 1.
Appearance of test specimens: (a) LOI/UL-94 vertical flame, (b) CONE, (c) flexural, and (d) tensile.
Figure 2.
Schematic diagram of composite preparation and testing.
2.3. Measurement and Characterization
2.3.1. Materials Characterization
Fourier transform infrared spectroscopy (FTIR) was performed to detect the changes in chemical groups and the structure of BC after its modification and graft synthesis using a Nicolet 6700FT-IR spectrophotometer with a KBr slice. The transmission mode was used, and the wavenumber range was set from 4000 to 400 cm–1.
13C NMR spectra were recorded on a Bruker 400M solid-state NMR spectrometer at a spin rate of 10 kHz with magic angle spinning (MAS) using a 4 mm rotor. The resonance frequency was set to 400 MHz. All samples used for solid-state NMR analyses were gently ground to ensure homogeneity. All spectra were acquired as the result of 1024 scans, and the prescan was delayed by 6.5 μs.
X-ray diffraction (XRD) patterns using Cu Kα radiation (λ = 1.542 Å) were recorded on an XRD-6000 machine (Shimadzu Co., Ltd., Kyoto, Japan) under a scanning step length of 2°/min in the range of 10–80°. All samples of 5–8 mg were prepared and analyzed under a tube voltage of 40 kV and a tube current of 30 mA.
A laser particle size distribution instrument (Mastersizer 2000, Malvern Instruments Ltd., Birmingham, U.K.) was used to measure the particle size of BC and BC-m under ethanol conditions.
The surface morphologies of BC and BC-m were observed by field emission scanning electron microscopy (FE-SEM, SU8010, Hitachi Co., Ltd., Tokyo, Japan) equipped with an energy-dispersive analysis X-ray (EDAX, OCTANE PLUS, Ametek Inc., California) microanalysis system. Specimens were sputter-coated with 24 ct gold for 90 s at a current of 15 mA.
2.3.2. Mechanical Properties
The mechanical properties for the composites were tested using a microcomputer-controlled electronic universal testing machine (CMT610, MTS Industrial Systems (China) Co., Ltd., Shanghai, China) according to the Chinese National Testing Standard GB/T 1040-2006 (for tensile properties) and GB/T 9341-2008 (for flexural properties). For the tensile test, the width and thickness of the narrow stress zone in specimens were 5 and 2 mm, respectively. The gauge length was set to 60 mm, and the loading speed was 2 mm/min. The flexural test specimen dimensions were 80 × 10 × 4 mm3 with a load span of 60 mm and a loading speed of 2 mm/min.
2.3.3. Thermal Stability
2.3.3.1. Thermal Gravimetric Analysis (TGA)
Thermal gravimetric analysis (TGA) was carried out using a thermogravimetric analyzer (F1, NETZSCH Co., Ltd., Selbu, Germany) under a nitrogen atmosphere. Approximately 5–10 mg of samples was assayed in an aluminum crucible and tested at temperatures ranging from 50 to 800 °C with a heating rate of 10 °C/min under a nitrogen flow of 20 mL/min. An empty crucible was used as a reference. The theoretical residue rate (TRR) was estimated according to eq 1 as follows:
| 1 |
where CPLA refers to the residue mass (RM) of PLA at 800 °C, b is the residue mass of fillers (BC/BC-m) at 800 °C, and β the weight ratio of PLA in the composite blends.
2.3.4. Combustion Performance Test
2.3.4.1. Limiting Oxygen Index
Limiting oxygen index (LOI), expressed as vol % minimum concentration of oxygen required to initiate polymer combustion, was measured using a JF-3 oxygen index meter (Jiangsu, Jiangning Analysis Instrument Company, China) according to ISO 4589-2:2017.48 The dimensions of the test specimens were 100 × 10 × 4 mm3.
The Underwriters Laboratories (UL-94) vertical flame test was carried out using a 5402-type vertical burn test instrument (Suzhou Yangyi Woerqi Detection Technology Co., Ltd., Jiangsu, China) with specimens measuring 130 × 13 × 3 mm3. The classifications are defined according to ATSM D3801-10.49
2.3.4.2. Cone Calorimeter Combustion (CONE) Tests
CONE tests were carried out using a cone calorimeter (FTT0007, Fire Testing Technology Co., Ltd., East Grinstead, U.K.) according to ISO 5660-1.50 Specimens measuring 100 × 100 × 4 mm3 sitting horizontally in a bed of aluminum foil were placed under the combustion chamber in which the predetermined test height was 25 cm. Using an external heat flux of 35 kW/m2, the material was ignited and the relevant combustion performance indices were recorded until combustion was complete. Three CONE tests were conducted per group.
2.3.5. Gas Decomposition Analysis
2.3.5.1. Thermogravimetric Analysis-Fourier Transform Infrared Spectrometry (TG-FTIR)
Thermogravimetric analysis-Fourier transform infrared spectrometry (TG-FTIR) was used to reveal the chemical structure and the nature of pyrolysis products from the composites using a TG analyzer (TG 209 F3, NETZSCH Co., Ltd., Selbu, Germany) coupled to an FTIR spectrophotometer (Tensor 27, BRUKER AXS GmbH, Karlsruhe, Germany) by a heated transfer line at 280 °C. Samples (10–15 mg) that had been dried overnight and ground into powder were placed in an aluminum crucible and heated from 50 to 800 °C at a heating rate of 10 °C/min in a nitrogen atmosphere with a flow rate of 70 mL/min. The FTIR spectrometer range was set to a scanning range of 4000–400 cm–1.
2.3.6. Residue Analysis
All BC and BC-m/PLA composites before and after combustion were collected and imaged under a SEM (XL30E, Philips, the Netherlands) under an accelerating voltage of 15 kV to further analyze the difference in the surface morphology. Specimens were sputter-coated with 24 ct gold for 90 s at a current of 15 mA.
X-ray photoelectron spectroscopy (XPS) was used to investigate the residual element contents of the samples. This was carried out using a Thermo Scientific K-Alpha spectrometer (Thermo Fisher Scientific Inc., Waltham, MA) equipped with a monochromatic Al Kα X-ray source (1486.6 eV) operating at 100 W. The samples were analyzed under vacuum (P < 10–8 mbar) with a pass energy of 150 eV (survey scans) or 25 eV (high-resolution scans). All peaks were calibrated with C 1s peak binding energy at 284.8 eV for adventitious carbon. The experimental peaks were fitted using Avantage software.
3. Results and Discussion
3.1. Characterization of BC, BC-o, and BC-m
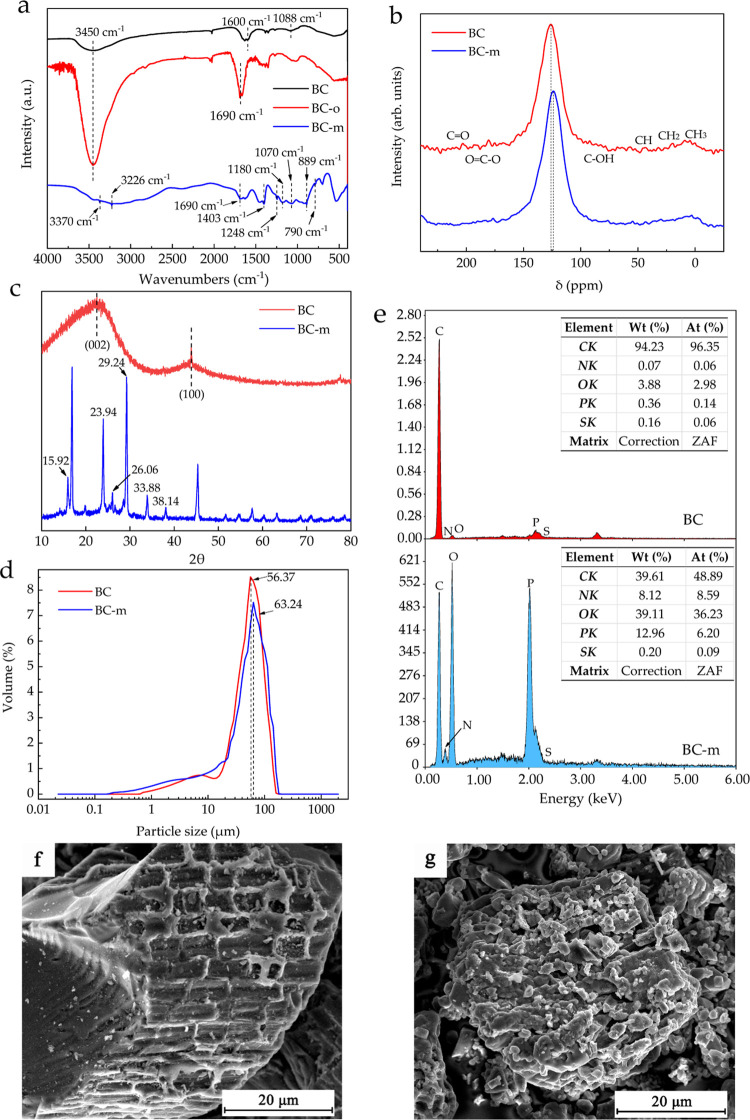
FTIR adsorption peaks are summarized in Table 2 and Figure 3a. As shown in Figure 3a, the typical peaks of BC are 3450 cm–1 (−OH), 1600 cm–1 (C=C), and 1088 cm–1 (C–O).37 After surface oxidation with H2O2, the absorption peaks of BC-o occurring at 3450 and 1690 cm–1 (C=O) were significantly enhanced in absolute value, corresponding to the formation of acidic phenolic and carboxylic groups, without the release of harmful gases.51,52 Characteristic absorption peaks for phenolic hydroxyl and carboxylic groups increased significantly with grafted BC-m. Typical peaks were observed at 3370 cm–1 (NH4+) and 3226 cm–1 (N–H). The peak at 1248 cm–1 was assigned to −P=O, and the other new absorption bands at 1180, 1070, 889, and 790 cm–1 correspond to C–N, symmetrical stretching of P–O, and asymmetrical stretching of P–O and P–O–P, respectively.53,54 The appearance of these characteristic peaks suggests the successful synthesis of phosphorous- and nitrogen-grafted BC-m.
Table 2. FTIR Characteristic Absorption of BC, BC-o, and BC-m.
| description | wavenumber (cm–1) | groups |
|---|---|---|
| peaks in BC | 3450 | –OH |
| 1600 | C=C | |
| 1088 | C–O | |
| new peak in BC-o | 1690 | C=O |
| new peaks in BC-m | 3370 | NH4+ |
| 3226 | N–H | |
| 1248 | –P=O | |
| 1180 | C–N | |
| 1070 | P–O (symmetrical stretching) | |
| 889 | P–O (asymmetrical stretching) | |
| 790 | P–O–P |
Figure 3.
Raw material assays: (a) FTIR spectra of BC, BC-o, and BC-m. (b) 13C MAS–NMR spectra of BC and BC-m. (c) XRD patterns of BC and BC-m. (d) Particle size distribution of BC and BC-m. (e) EDAX spectrum of BC and BC-m. (f) Surface micrograph of the outer surface of BC. (g) Surface micrograph of the outer surface of BC-m.
The structural differences between BC and BC-m analyzed by 13C NMR are shown in Figure 3b. Like many other biochars, BC is rich in complex, amorphous carbon structures. The spectra for biochars are dominated by the band for aromatic carbons around 126 ppm, which is characterized by the changing trend of 13C spectrum at high temperatures.55 Many other small signals such as C=O (210–183 ppm), O=C–O (183–165 ppm), C–OH (40–90 ppm), and alkyl groups (90–6 ppm) were also detected.56 After modification, the aromatic carbon spectrum of BC-m showed a slight shift to a higher field (δ = 124 ppm), while other small resonance peaks moved to high-field or disappeared to varying degrees, suggesting that the usual order of 13C may be altered by the chemical interaction between groups introduced through new chemical bonds.57 It gives further confirmation that the PA and urea reacted successfully with the BC during synthesis of BC-m.
XRD analysis was further used to verify differences between BC and BC-m. As can be seen from Figure 3c, the crystal peak of BC at 2θ = 20–30° implies the stacking structure of aromatic layers (graphite 002),58 and the peaks at 2θ ≈ 43.9° characterizing the in-plane symmetry along the graphene layers (graphite 100)59 were almost nonexistent. Many new crystalline structures were formed, such as the peaks at 2θ = 15.92, 23.94, 26.06, 33.88, and 38.14°, indicating that through reactions with PA and urea, BC-m has a crystalline structure similar to the conventional fire-retardant APP.60−62
To address the question of whether the particle size of BC changed significantly after grafting and regrinding, particle size analysis was performed. As shown in Figure 3d, most particles of BC were between 40 and 110 μm, and the majority of the distribution had a mean diameter of 56.37 μm. After the grafting and regrinding of BC-m, the mean particle size shifted upward very slightly (from 50.93 μm for pure BC to 54.20 μm of BC-m) and the distribution narrowed slightly.
SEM images used to examine differences in the surface morphology between BC and BC-m are shown in Figure 3f,g. Note that the original pore structure of the bamboo tissue can be clearly seen on the surface of BC particles, while the surface of BC-m and reground grains had become amorphous. EDAX elemental analysis (Figure 3e) further supports the successful conversion to BC-m. From the elemental analysis peaks, it can be seen that the N, O, and P contents of BC-m were increased significantly and may be attributed to the reaction of PA and urea with oxygen-containing functional groups on the BC surface. This corresponds to the new absorption peak and the new crystal peak in the FTIR and XRD analyses.
3.2. Mechanical Properties of PLA Composites
The tensile and flexural properties of pure PLA and the flame-retardant PLA are shown in Figure 4, with means and variation (standard deviation values) given in Table 3. Generally speaking, the tensile and bending strength decreased and the modulus of elasticity increased with the addition of BC (Figure 4a,b) and BC-m (Figure 4c,d). The tensile strength of the pure PLA was 58.1 MPa, decreasing by 3.8 and 14.5% with the addition of 10 and 30 wt % addition of BC-m, respectively (Figure 4b). The mean tensile modulus increased from 744.2 MPa for pure PLA to 995.7 MPa with 30 wt % BC-m. A similar phenomenon was reflected in the flexural properties: the mean flexural strength decreased with the increasing addition level of BC-m, but the flexural modulus increased slightly, i.e., the BC-m-filled PLA increased in stiffness and rigidity. Figure 4e,f displays typical tensile stress–strain relationships of the composite materials for PLA/BC and PLA/BC-m, respectively. Pure PLA appeared to behave in a more nonlinear and ductile manner than the composite materials. BC or BC-m is generally a harder material than PLA. Therefore, the resulting composite modulus should be higher depending on the quality of mixing and bonding between the constituents according to the rule of mixture. The dispersed BC-m in the PLA may limit the mobility of PLA molecules, thereby reducing its plasticity and increasing its rigidity.43 The composite materials also showed lower elongation at break, as can be seen in Figure 4e,f. Greater addition levels of filler lead to a greater probability of local stress concentration caused by agglomerations, which reduces strength properties.43,63
Figure 4.
Mechanical properties of PLA and composites (error bars represent standard deviation): (a, b) BC/PLA and (c, d) BC-m/PLA. Typical tensile stress–strain curves of selected samples: (e) BC/PLA and (f) BC-m/PLA.
Table 3. Mechanical Properties of PLA, BC/PLA, and PLA/BC-m Composites ± Stdev.
| sample | tensile strength (MPa) | tensile modulus (MPa) | flexural strength (MPa) | flexural modulus (GPa) |
|---|---|---|---|---|
| PLA | 58.12 ± 1.76 | 774.24 ± 33.43 | 95.76 ± 0.80 | 3.73 ± 0.17 |
| BC/PLA1 | 56.17 ± 2.76 | 692.54 ± 10.72 | 76.32 ± 3.66 | 3.29 ± 0.21 |
| BC/PLA2 | 45.29 ± 1.16 | 798.86 ± 20.73 | 67.21 ± 3.13 | 3.96 ± 0.13 |
| BC/PLA3 | 35.48 ± 3.42 | 808.37 ± 31.06 | 62.66 ± 3.14 | 4.82 ± 0.14 |
| BC-m/PLA1 | 55.91 ±2.23 | 917.06 ± 77.74 | 84.15 ± 2.18 | 3.75 ±0.08 |
| BC-m/PLA2 | 51.94 ± 1.77 | 941.15 ± 141.33 | 87.01 ± 1.90 | 3.81 ± 0.32 |
| BC-m/PLA3 | 49.71 ± 0.88 | 995.70 ± 112.20 | 81.77 ± 1.65 | 4.28 ± 0.34 |
The results indicate that the extent to which both tensile and bending strengths decreased with filler content was greater for the pure BC than BC-m. For example, tensile strength with 20 wt % addition was 45.29 MPa for pure BC and 51.94 MPa for BC-m. BC-m may either have improved chemical compatibility with PLA compared with pure BC and/or the same weight of material BC-m had fewer grains of BC than pure BC, leading to a reduced negative effect on composite strength. Better chemical compatibility means stresses may be dissipated by stronger particle–particle and particle–polymer interactions.35 For comparison, a study by Shukor et al.66 showed that with adding just 10 wt % APP, the flexural strength of PLA composites decreased from (a lower pure strength of) 34 to 18 MPa, an almost 50% reduction, much larger than the decrease of 16.1% found in this study for 10 wt % BC-m.
3.3. Thermal Stability
Thermogravimetry (TG) and derivative thermogravimetric (DTG) curves of BC, BC-m, PLA, BC/PLA, and BC-m/PLA composites in a nitrogen atmosphere are shown in Figure 5 with corresponding data in Table 4. The degradation onset temperature (T–5%) was considered to be the temperature at which the material has undergone 5% weight loss.
Figure 5.
TG (a, c, e) and DTG (b, d, f) curves for BC, BC-m, PLA, and its blends under a N2 atmosphere.
Table 4. Thermogravimetry Test Data of PLA and BC-m/PLA Composites under a N2 Atmosphere.
| carbon
residue rate (%) |
||||||
|---|---|---|---|---|---|---|
| sample | T–5% (°C) | Rpeak/Tmax (%/°C) | 400 (°C) | 600 (°C) | 800 (°C) | TRR (%) |
| PLA | 343 | 5.72/382 | 1.17 | 0.60 | 0.58 | |
| BC | 484 | 0.32/295 | 96.37 | 93.75 | 93.18 | |
| BC-m | 373 | 2.66/382 | 83.78 | 82.49 | 82.24 | |
| BC/PLA1 | 341 | 5.56/347 | 10.10 | 9.31 | 9.27 | 9.84 |
| BC/PLA2 | 341 | 4.43/348 | 19.02 | 18.36 | 18.31 | 19.10 |
| BC/PLA3 | 339 | 3.67/348 | 28.03 | 27.58 | 27.51 | 28.36 |
| BC-m/PLA1 | 334 | 25.33/385 | 10.08 | 9.10 | 8.64 | 8.75 |
| BC-m/PLA2 | 331 | 20.94/383 | 26.73 | 22.23 | 21.81 | 16.91 |
| BC-m/PLA3 | 331 | 19.52/380 | 37.06 | 32.34 | 29.83 | 25.08 |
First, the rapid thermal degradation of the graft products on BC-m between 340 and 400 °C can be seen (Figure 5a), resulting in a mass loss of about 18%, whereas ungrafted BC undergoes a slower and more steady rate of mass loss of its residual combustible matter up to about 7% at 800 °C. Second, the PLA component rapidly degrades starting around 325 °C and losing mass completely at close to 400 °C. As shown in Table 4 and Figure 5c,d, pure BC caused an earlier onset of the PLA decomposition phase than when BC-m was added (Figure 5e,f). BC/PLA composites rapidly degraded to the residual mass corresponding to the addition level of BC (i.e., minimal protection of PLA), whereas with BC-m addition, especially 20 and 30 wt %, residual mass at 400 °C was higher than the mass of BC added, with continued gradual mass loss at higher temperatures. Note that in Table 4, the Tmax (temperature at the maximum rate of mass loss) was similar for PLA and the BC-m/PLA composites, which were 382, 385, 382, and 379 °C, respectively. With the greater addition of BC-m, Rpeak values were reduced. As shown in Table 4, there was little difference in the carbon residue of PLA from 600 to 800 °C, which is virtually completely pyrolyzed by 600 °C, but BC-m/PLA composites underwent continued mass loss, indicating retarded combustion in BC-m/PLA.
Note that the carbon residue rate of the composite containing 10 wt % unmodified BC was higher than those containing BC-m. The Tmax of BC/PLA composites was lower, and their Rpeak was slightly decreased, corresponding to previous results.45,64 As a carbonized material, BC undergoes relatively small mass loss during pyrolysis, as shown in Figure 5a,b, corresponding to an Rpeak of 0.32% at 295 °C and a final carbon residue rate of 93.18%. In contrast, the grafted additives being 10.94% of the mass of BC-m accounted for the obvious mass loss of BC-m at 382 °C, giving a final carbon residue of 82.24%. The grafted nitrogen and phosphorous compounds on BC-m decompose by heating and generate both gas-phase and condensed-phase flame retardant.65,66 These grafted additives reduced the relative mass of pure BC by 10.94% when BC-m was added to the composite, which helps account for a lower final carbon residue rate. As a result, the actual carbon residues of BC/PLA samples were close to the theoretical values of 9.84, 19.10, and 28.36% (Table 4). Actual residue rates of BC-m/PLA composites were 8.64, 21.81, and 29.83%; for BC-m/PLA2 and BC-m/PLA3, the rates were increased by 28.98 and 18.94%, respectively, compared to their theoretical residue rates, indicating that BC-m significantly improves the thermal stability of the composites. For pure BC or a low rate of BC-m addition, the actual residual rates of the composites were actually lower than the theoretical residual rates. This is mainly due to lack of or insufficient flame resistance and volatile loss of oxygen elements in BC in the form of CO2, CO, and H2O during heating.
3.4. Burning Behavior
3.4.1. LOI and UL-94 Vertical Flame Tests
Both LOI and UL-94 tests were used to evaluate the burning behavior of PLA and BC-m/PLA composites, with test results given in Table 5. Images of PLA and BC-m/PLA composites after LOI testing are shown in Figure 6. Pure PLA burned easily and exhibited significant melt dripping during the UL-94 test, and the registered LOI was just 20.7 vol %. At the addition of 30 wt % untreated BC, LOI reached 22.5 vol %, whereas when 20 or 30 wt % BC-m was added, both achieved a UL-94 V-0 rating and their LOI increased markedly to 29.2 and 32.1 vol %, respectively.
Table 5. LOI and UL-94 Results of PLA/BC-m Composites.
| UL-94 |
||||
|---|---|---|---|---|
| sample | LOI (vol %) | t1 (s)/t2 (s)a | dripping | rating |
| PLA | 20.7 | BCb | yes | NRc |
| BC/PLA1 | 21.2 | BC | yes | NR |
| BC/PLA2 | 21.8 | BC | yes | NR |
| BC/PLA3 | 22.5 | 10.3/7.8 | yes | V-2 |
| BC-m/PLA1 | 28.0 | 5.6/2.3 | yes | V-2 |
| BC-m/PLA2 | 29.2 | 2.1/1.1 | no | V-0 |
| BC-m/PLA3 | 32.1 | 1.2/1.4 | no | V-0 |
| BC/PLA/10AHPd | 26.5 | 3.1/1.6 | no | V-0 |
| BC/PLA/20AHPd | 29.3 | 1.0/1.1 | no | V-0 |
| BC/PLA/30AHPd | 31.0 | 1.2/1.1 | no | V-0 |
| 15APP/PLAe | 28.0 | 4.8/1.4 | yes | V-2 |
| 20APP/PLAf | 28.6 | 2.3/1.6 | V-2 | |
Figure 6.
Digital images of materials after LOI tests: (a) PLA; (b) BC/PLA1; (c) BC/PLA2; (d) BC/PLA3; (e) BC-m/PLA1; (f) BC-m/PLA2; and (g) BC-m/PLA3.
At 10 wt % BC-m, dripping was inhibited but only to a limited extent. With the addition of 20 and 30 wt % BC-m, dripping was completely suppressed and the burning intensity was less than that of pure PLA. LOI and UL-94 test data from this and previous studies are given in Table 5. When APP is used at 20 wt % addition,67 the LOI was 28.6 vol %, lower than the value here for 20 wt % BC-m (29.2 vol %). Note that the LOI with 15 wt % addition of APP68 was very similar to 10 wt % of BC-m added in this study. None of the APP-added composites reached a UL-94 V-0 rating. Compared with the AHP/BC/PLA composite tested in previous research,35 where the addition of AHP was 20 + 25 wt % untreated BC (a very high total filler percentage of 45%), the LOI was 29.3 vol %. This value was similar to the LOI of 29.2 vol % from adding just 20% BC-m (i.e., BC-m/PLA2). The UL-94 and LOI tests results suggest BC-m may be a viable substitute for AHP and APP flame retardants at lower required addition levels of filler.
3.4.2. Cone Calorimetry Testing
The results so far suggested no obvious, effective combustion retarding effects of pure BC compared with BC-m. To better understand the improved flame-retardant properties of BC-m in PLA under specific combustion conditions, cone calorimetry (CONE) tests were carried out as a standardized method for comparing the fire resistance of materials.69 Relevant CONE data for a 25% BC + PLA mix from previous research are shown for comparison.35 Key indices are the heat release rate (HRR), total heat release (THR), total smoke release (TSR), average effective heat of combustion (aEHC), and mass changes (Mass), as shown in Figure 7 and Table 6.
Figure 7.
Typical CONE testing curves: (a) HRR, (b) THR, (c) TSR, and (d) mass loss curves of PLA, 25BC/75PLA, and BC-m/PLA composites.
Table 6. Cone Calorimeter Results (Average of Three Samples) of PLA and BC-m/PLA Composites at 35 kW/m2a.
| sample | TTI (s) | THR (MJ/m2) | pHRR (kW/m2) | TSR (m2/m2) | pMLR (g/s) | aEHC (MJ/kg) | RM (%) | COY (×10–3) (kg/kg) | CO2Y (kg/kg) |
|---|---|---|---|---|---|---|---|---|---|
| PLA | 56 | 86.06 | 435.58 | 24.45 | 0.60 | 16.37 | 2.29 | 4.59 | 1.48 |
| 25BC/75PLAb | 33 | 59.94 | 320.32 | 34.22 | 0.57 | 15.43 | 19.46 | 1.33 × 10–3 | 1.82 |
| BC-m/PLA1 | 34 | 68.64 | 418.11 | 94.16 | 0.51 | 16.15 | 6.45 | 10.60 | 1.39 |
| BC-m/PLA2 | 33 | 53.48 | 201.02 | 80.01 | 0.48 | 15.08 | 18.99 | 15.04 | 1.25 |
| BC-m/PLA3 | 31 | 41.10 | 181.61 | 69.26 | 0.35 | 14.52 | 47.71 | 43.43 | 1.07 |
Notes: TTI, time to ignition, ±2 s; THR, total heat release, ±0.5 MJ/m2; pHRR, peak heat release rate, ±15 kW/m2; TSR, total smoke release, ±5 m2/m2; pMLR, peak mass loss rate, ±0.05 g/s; aEHC, average effective heat of combustion within 180 s, ±1 MJ/kg; COY, CO yield, ±2 × 10–3 kg/kg; CO2Y, CO2 yield, ±0.5 kg/kg.
Data from ref (35).
THR and HRR are important factors affecting fire propagation risk.70 As shown in Figure 7a, HRR was quickly suppressed in PLA containing 20 or 30 wt % BC-m. The peak heat release rate (pHRR) of PLA with 10% BC-m (418.11 kW/m2) was similar to that of pure PLA (435.58 kW/m2), only reducing by 4%. However, the pHRR values for 20 and 30 wt % BC-m were 201.02 and 181.61 kW/m2, respectively, with a corresponding decrease of 53.9 and 58.3%, respectively, compared to pure PLA. The enhanced flame-retardant effect of BC-m compared with BC at a higher dosage can be seen from CONE indices; THR and pHRR of 25% BC in PLA was 59.94 MJ/m2 and 320.32 kW/m2, respectively, which are higher than the 53.48 MJ/m2 and 201 kW/m2 for BC-m/PLA2 containing 20% BC-m.
The lowest addition (10 wt %) of BC-m appears to be insufficient as the PLA matrix continued to release energy in continuous thermal degradation. The flame-retardant level must be sufficient to allow the formation of a protective carbonized layer at the initial stage of combustion.71 At BC-m levels of 20% or more, the heat release fluctuated but was effectively inhibited. Compared with pure PLA (Figure 7b), the THR of BC-m/PLA3 was 41.10 MJ/m2, a decrease of 52.2%. The corresponding aEHC value decreased by 11.3% within 180 s of combustion from 16.37 to 14.52 MJ/kg.
The addition of BC-m increased TSR, compared with PLA and PLA + BC, and surprisingly high at low-level (10 wt %) addition. The TSR of BC-m/PLA1 was 94.16 m2/m2, quadruple that of PLA. Due to its high nitrogen and phosphorus content, BC-m was formulated to act as an intumescent flame retardant, i.e., it undergoes rapid thermal degradation at the early stages of heating, which produces nonflammable oxygen-diluting gases and forms a foamlike expanded layer of char on the surface of the composite.19,72 The expansion mechanism of an IFR involves the release of inert gases and increased smoke emissions.73 TSR curves for BC-m/PLA rose rapidly (Figure 7c), with significantly increased residue mass (RM) as a result of the protective effect. At 10 wt % BC-m, the char was limited and less effective, allowing external heat and oxygen ingress to the internal matrix. This helps explain the limited difference in peak mass loss rate (pMLR) and RM between pure PLA and BC-m/PLA1, as shown in Figure 7d. Examination of the post-CONE test carbon residue found more voluminous, continuous, and complete layers of expanded char with 20 and 30 wt % addition of BC-m (Figure 8c1–2,d1–2). The yield of CO2 was lower and that of CO was higher at 30 wt % addition (Table 6), indicating incomplete combustion caused by the char layer.
Figure 8.
Images of carbon residues from PLA and BC-m/PLA composites after CONE testing: (a1–2) PLA; (b1–2) BC-m/PLA1; (c1–2) BC-m/PLA2; and (d1–2) BC-m/PLA3.
The addition of BC-m shifted forward the time to ignition (TTI). The TTI of pure PLA was 56 s, reducing to 31 s for BC-m/PLA3. Likely reasons for this include the greater heat absorption rate of BC-m, leading to earlier combustion and char layer formation.54 BC-m degrades to release acidic substances (such as phosphoric acid and pyrophosphoric acid, which undergo hydrolysis to form phosphoric acid),12,13 which can prematurely degrade the PLA, leading to its accelerated ignition. The results from the CONE tests showed that 20 and 30 wt % BC-m addition were able to significantly inhibit the heat release rate from the combustion of the composite, indicating enhanced protection. The theoretical RM values for BC-m/PLA2 and BC-m/PLA3 (CONE samples) are 16.91 and 25.08%, respectively. From the CONE tests, the actual residual mass of BC-m/PLA2 and BC-m/PLA3 were 18.99 and 47.71%, respectively. Note that for BC-m/PLA3, the RM of 47.71% is almost twice the theoretical value, indicating good material protection from complete pyrolysis. CONE test residues can be seen in Figure 8c,d, showing that pure PLA was completely pyrolyzed (Figure 8a1,2). Different from our previous research in which only BC was added,35 BC-m made the residual carbon show a significant expansion and continuous effect, while the BC/PLA composite presents a flat shape and the char layer is more prone to damage.
3.5. Gas Decomposition Products
To further investigate the thermal decomposition and combustion gas release for pure PLA and BC-m/PLA composites, TG-FTIR tests were carried out. The absorbance spectra of different pyrolysis products for PLA and BC-m/PLA composites vs time are shown in Figure 9. CO2 (2360 cm–1), C–O (1242 cm–1), carbonyl (1750 cm–1), and hydrocarbons (3000 cm–1) were detected with their absorbance highest in pure PLA. Other absorption peaks identified by characteristic FTIR signals were for NH3 (931 cm–1), P–O (1084 cm–1), and P=O (1276 cm–1) detected in the pyrolysis products of BC-m/PLA composites.45,54,65 This is an important mechanism of the flame suppression mechanism in intumescents, as discussed later. Inert gases such as CO2 and NH3 that further contribute to diluting combustible gases also help to retard the combustion process.74 These phenomena help explain the reduction in organic volatiles from PLA combustion, as well as the associated significant reduction in combustion heat release and the increased carbon residue in the CONE and TG tests of the BC-m/PLA composites.
Figure 9.
FTIR spectra of gaseous products for the thermal decomposition of PLA and BC-m/PLA composites vs time: (a) CO2; (b) CO; (c) carbonyl compound; (d) hydrocarbons; (e) C–O bond; (f) NH3; (g) P–O bond; and (h) P=O bond.
To further elucidate the gas phase during combustion, FTIR curves for volatile products produced from BC-m/PLA3 under different heating temperatures are shown in Figure 10. The selection of heating temperatures was based on TG results. The absorption peak at 1750 cm–1 at 380 °C corresponds to carbonyl compounds produced during PLA pyrolysis. The peaks for NH3 (at 931 and 3056 cm–1), CO2 (2360 cm–1), P–O (1084 cm–1), and P=O (1276 cm–1)54,65 appear at the maximum thermal decomposition temperature (about 380 °C). Note that these also appear at 700 °C, suggesting some continued production of flame-retardant gases at much higher temperatures above the degradation point for pure PLA.
Figure 10.

FTIR spectra of the volatiles from BC-m/PLA3 heated at different temperatures.
3.6. Char Residue Analysis
3.6.1. SEM Images of BC-m/PLA before Combustion
SEM was used to examine the surfaces of unburnt BC-m/PLA composites (Figure 11). The vast majority of BC-m particles were dispersed uniformly and well-integrated with the PLA; however, composite surface roughness increased with the increasing addition of BC-m. With increasing BC-m addition, some flaws and debonding owing to particle agglomeration between BC-m and PLA are inevitable.43
Figure 11.
SEM images of fractured surface of BC-m/PLA composites before combustion: (a1) BC-m/PLA1; (a2) BC-m/PLA2; and (a3) BC-m/PLA3.
3.6.2. SEM Images of Residue Surfaces and Inner Layers
The carbonized residues on the outer surface and interior of BC-m/PLA composites after CONE testing were examined under SEM, shown in Figure 12. At a low level of BC-m (10 wt %), there were many fine fissures on the residue surface (Figure 12a1), indicating an incomplete and discontinuous char layer. Increasing the addition level of BC-m eliminated the fissures (Figure 12b1–c1). Images from the inner layers of char show an increased consolidation of the char layer with an increasing level of BC-m addition, as seen in Figure 12a2 (lowest) to c2 (highest). BC-m addition of 30 wt % formed a dense and uniform char layer, providing a superior condensed-phase flame-retardant effect (Figure 12b3–c3). At 10 wt % BC-m, shown in Figure 12a2, a large range of pore sizes in the foam can be seen. With 30 wt % BC-m addition, the char layer became denser with a honeycomb-like pore structure and the larger pores disappearing (Figure 12c2,c3), leaving a few smaller pores that allow gas exchange. The gases produced by early BC-m degradation create bubbles on the surface that are filled in by pyrophosphate products formed by acid catalysis, shifting the protection more to a physical, condensed-phase mechanism as combustion progresses. Filled pores are especially evident in Figure 12b2,3,c2,3.
Figure 12.
SEM images of residues on the outer surface (a1: BC-m/PLA1; b1: BC-m/PLA2; c1: BC-m/PLA3) and the internal surface (a2–a3: BC-m/PLA1; b2–b3: BC-m/PLA2; c2–c3: BC-m/PLA3) from BC-m/PLA composites after cone tests with different magnifications.
3.6.3. Structure and Composition
The chemical components of the char residue after CONE testing were investigated by XPS, with the corresponding spectra shown in Figure 13. Elements C and O constitute the majority of the residue, followed by P, whereas the N fraction was low. It was observed that with greater BC-m addition, the content of P and N increased, as expected.
Figure 13.
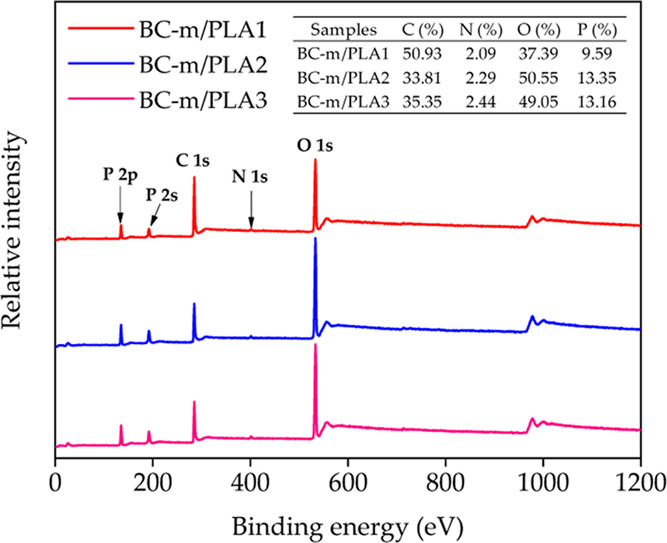
XPS spectra of the char residue of BC-m/PLA composites after the cone calorimeter test.
Corresponding spectra are shown in Figure 14 for elements on the surface of the carbon residue of BC-m/PLA3. As shown in Figure 14a, the binding energy at 533.2 eV is assigned to −O– in C–O–C and/or C–O–P groups. Two peaks were observed from the C 1s spectra in Figure 14b: the peak at 284.7 eV is attributed to C–H and C–C bonds in aliphatic and aromatic species and the smaller peak at 286.0 eV is assigned to C–O. The peak at 134.7 eV of the P 2p spectra in Figure 14c and 401.2 eV of N 1s spectra in Figure 14d correspond to the P–O–P bond and the N in NH4+.29,45,75 The results show that the BC-m dispersed in the PLA matrix led to the formation of a more stable expanded char layer structure containing P and N reflected in the detection of C–O–P, P–O–P, and N in NH4+. The char appears not to be rapidly and completely decomposed even at higher temperatures, thus continuing to provide a gas phase flame-retardant effect throughout the combustion process.
Figure 14.
Spectra of the char residue of BC-m/PLA3 for (a) O 1s, (b) C 1s, (c) P 2p, and (d) N 1s.
3.6.4. Proposed Flame-Retardant Mechanism
A schematic representation of the possible flame-retardant mechanism of BC-m/PLA composites is presented in Figure 15. Being carbonized, BC itself has excellent thermal stability.76 However, without the grafting of intumescent nitrogen and phosphorous compounds, it may only be considered as a noncombustible filler that displaces the flammable polymer fraction in the composite. BC on its own may simply delay combustion by absorbing heat early and promoting the formation of char of the surrounding matrix, but from the indices compared here, its effect is limited even at 25% addition. In contrast, BC-m undergoes rapid thermal degradation as the temperature increases to around 382 °C, whereby the grafted flame-retardant compounds release noncombustible gases, such as NH3 and CO2, which dilute combustible gases.13 This follows the typical gas phase mechanism of IFRs early in the process.
Figure 15.
Schematic representation of the possible flame-retardant mechanism for BC-m/PLA composites.
These gases also create an expanded foamy char layer, and at higher temperatures, phosphorus-based IFRs degrade to produce ultraphosphate and phosphoric acid that catalyze the formation of solid char residues by promoting the dehydration reaction of the terminal alcohols in the polymer to which they are added.13 In the case of BC-m here, the bubbles formed by early gas generation are consolidated by filling with pyrophosphate products, contributing to the effective formation of a more continuous, dense, and uniform expanded char layer. The minimum effective addition level of BC-m is about 20%, as the formation of continuous, stable char is central to the mechanism by which BC-m helps retard further combustion. As might be expected of a carbonized material, the BC component of BC-m does not undergo complete pyrolysis and, therefore, contributes a greater mass and volume to the protective char layer than might be expected from adding the same mass of a functionalized bamboo powder and contribute less-flammable gases to the system as it combusts. Using BC as the graft substrate is, therefore, more likely to continue to provide a flame-retardant effect at higher temperatures; however, a direct comparison of these two graft substrates is planned and will be reported in a future paper.
4. Conclusions
In this work, a novel flame-retardant filler based on phosphate- and urea-grafted bamboo charcoal (BC) was formulated and tested for enhancing flame retardancy in PLA. The dried, ground BC-m was able to be evenly and successfully compounded with PLA at addition levels of 10, 20, and 30 wt %. The main conclusions are drawn as follows:
-
1.
BC-m/PLA displayed improved mechanical properties compared with pure BC/PLA due to better constituent compatibility. UL-94 and Cone calorimetry tests showed that the flame-retardant properties of the BC-m/PLA composites were significantly better than the pure BC/PLA mix.
-
2.
Analyses of TG-FTIR, SEM, and XPS suggested that BC-m produces inert gases such as CO2 and NH3, diluting combustible gases and phosphoric acid and promoting the early degradation and carbonization of PLA to form a foamy protective char layer. For the grafted BC fire-retardant system, the foam bubbles caused by gas are quickly filled and solidified with pyrophosphate products from the degradation of BC-m, producing a dense, continuous, and compact char layer that provides an excellent and stable flame-retardant effect.
-
3.
Biodegradable BC-m does not contain any toxic or harmful chemical elements, and the BC-m/PLA composite materials are likely to compost easily. The results suggest a promising route to enhancing the flame-retardant properties of biodegradable polymer composites using more environmentally friendly grafted biochar. Further research will also examine the biodegradability and effects on compost of waste PLA packaging containing BC-m flame-retardant filler.
Acknowledgments
The authors would like to acknowledge the Zhejiang Natural Science Foundation for the financial support of this research and the Zhejiang Academy of Forestry for the technical assistance. Financial support from the Natural Sciences and Engineering Research Council of Canada (NSERC) through a Discovery Grant is also gratefully acknowledged.
Author Contributions
§ L.Z. and W.C. contributed equally to this work.
Author Contributions
Initial conceptualization and methodology: W.Z. and W.L., providing resources as well; experiments: L.Z., W.C., and W.L.; data curation and management: L.Z. and W.C.; original draft writing: L.Z. and W.C. with help from K.S. and N.Y.; manuscript review and editing: W.L., W.Z., K.S. and C.D.; project supervision and administration: W.Z. and L.Z.; and funding acquisition: W.Z. and C.D.
This research was supported by the National Natural Science Foundation of China (32171878), Science and Technology Plan Project of Zhejiang Province (2018C02008), and the Natural Sciences and Engineering Research Council of Canada (NSERC) through a Discovery Grant (RGPIN-2020-06097).
The authors declare no competing financial interest.
References
- Nampoothiri K. M.; Nair N. R.; John R. P. An overview of the recent developments in polylactide (PLA) research. Bioresour. Technol. 2010, 101, 8493–8501. 10.1016/j.biortech.2010.05.092. [DOI] [PubMed] [Google Scholar]
- Conner D. K.; O’Dell R.. The Tightening Net of Marine Plastics Pollution. In Environment: Science and Policy for Sustainable Development; Taylor & Francis, 1988; Vol. 30, pp 16–36. [Google Scholar]
- Müller C.; Townsend K.; Matschullat J. Experimental degradation of polymer shopping bags (standard and degradable plastic, and biodegradable) in the gastrointestinal fluids of sea turtles. Sci. Total Environ. 2012, 416, 464–467. 10.1016/j.scitotenv.2011.10.069. [DOI] [PubMed] [Google Scholar]
- Gu L.; Qiu J.; Qiu C.; Yao Y.; Sakai E.; Yang L. Mechanical properties and degrading behaviors of aluminum hypophosphite-poly (Lactic Acid) (PLA) nanocomposites. Polym.-Plast. Technol. Mater. 2019, 58, 126–138. 10.1080/03602559.2018.1466169. [DOI] [Google Scholar]
- Zhou X.; Li J.; Wu Y. Synergistic effect of aluminum hypophosphite and intumescent flame-retardants in polylactide. Polym. Adv. Technol. 2015, 26, 255–265. 10.1002/pat.3451. [DOI] [Google Scholar]
- Battegazzore D.; Frache A.; Carosio F. Sustainable and high performing biocomposites with chitosan/sepiolite layer-by-layer nanoengineered interphases. ACS Sustainable Chem. Eng. 2018, 6, 9601–9605. 10.1021/acssuschemeng.8b02907. [DOI] [Google Scholar]
- Ou X.; Cakmak M. Comparative study on development of structural hierarchy in constrained annealed simultaneous and sequential biaxially stretched polylactic acid films. Polymer 2010, 51, 783–792. 10.1016/j.polymer.2009.11.058. [DOI] [Google Scholar]
- Hwang S. W.; Shim J. K.; Selke S. E.; Soto-Valdez H.; Matuana L.; Rubino M.; Auras R. Poly (L-lactic acid) with added α-tocopherol and resveratrol: optical, physical, thermal and mechanical properties. Polym. Int. 2012, 61, 418–425. 10.1002/pi.3232. [DOI] [Google Scholar]
- Vink E. T. H.; Rábago K. R.; Glassner D. A.; Gruber P. R. Applications of life cycle assessment to Nature Works polylactide (PLA) production. Polym. Degrad. Stab. 2003, 80, 403–419. 10.1016/s0141-3910(02)00372-5. [DOI] [Google Scholar]
- Södergård A.; Stolt M. Properties of lactic acid based polymers and their correlation with composition. Prog. Polym. Sci. 2002, 27, 1123–1163. 10.1016/s0079-6700(02)00012-6. [DOI] [Google Scholar]
- Li S.; Yuan H.; Yu T.; Yuan W.; Ren J. Flame-retardancy and anti-dripping effects of intumescent flame-retardant incorporating montmorillonite on poly(lactic acid). Polym. Adv. Technol. 2009, 20, 1114–1120. 10.1002/pat.1372. [DOI] [Google Scholar]
- Tang G.; Wang X.; Xing W.; Zhang P.; Wang B.; Hong N.; Yang W.; Hu Y.; Song L. Thermal Degradation and Flame-retardance of biobased polylactide composites based on Aluminum Hypophosphite. Ind. Eng. Chem. Res. 2012, 51, 12009–12016. 10.1021/ie3008133. [DOI] [Google Scholar]
- Laoutid F.; Bonnaud L.; Alexandre M.; Lopez-Cuesta J. M.; Dubois P. New prospects in flame-retardant polymer materials: from fundamentals to nanocomposites. Mater. Sci. Eng., R 2009, 63, 100–125. 10.1016/j.mser.2008.09.002. [DOI] [Google Scholar]
- Nurul Fazita M. R.; Jayaraman K.; Bhattacharyya D.; Mohamad Haafiz M. K.; Saurabh C. K.; Hussin M. H.; HPS A. K. Green composites made of bamboo fabric and polylactic acid for packaging applications—A review. Materials 2016, 9, 435 10.3390/ma9060435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D. Y.; Song Y. P.; Lin L.; Wang X. L.; Wang Y. Z. A novel phosphorus-containing poly(lactic acid) toward its flame-retardation. Polymer 2011, 52, 233–238. 10.1016/j.polymer.2010.11.023. [DOI] [Google Scholar]
- Yuan X. Y.; Wang D. Y.; Chen L.; Wang X. L.; Wang Y. Z. Inherent flame-retardation of bio-based poly(lactic acid) by incorporating phosphorus linked pendent group into the backbone. Polym. Degrad. Stab. 2011, 96, 1669–1675. 10.1016/j.polymdegradstab.2011.06.012. [DOI] [Google Scholar]
- Li Y.; Sun X. S. Preparation and characterization of polymer–inorganic nanocomposites by in situ melt polycondensation of l-lactic acid and surface-hydroxylated MgO. Biomacromolecules 2010, 11, 1847–1855. 10.1021/bm100320q. [DOI] [PubMed] [Google Scholar]
- Réti C.; Casetta M.; Duquesne S.; Bourbigot S.; Delobel R. Flammability properties of intumescent PLA including starch and lignin. Polym. Adv. Technol. 2008, 19, 628–635. 10.1002/pat.1130. [DOI] [Google Scholar]
- Bourbigot S.; Le Bras M.; Duquesne S.; Rochery M. Recent advances for intumescent polymers. Macromol. Mater. Eng. 2004, 289, 499–511. 10.1002/mame.200400007. [DOI] [Google Scholar]
- Wang X.; Xing W.; Wang B.; Wen P.; Song L.; Hu Y.; Zhang P. Comparative study on the effect of beta-cyclodextrin and polypseudorotaxane as carbon sources on the thermal stability and flame-retardance of polylactic acid. Ind. Eng. Chem. Res. 2013, 52, 3287–3294. 10.1021/ie303002b. [DOI] [Google Scholar]
- Wang J.; Ren Q.; Zheng W.; Zhai W. Improved flame-retardant properties of poly(lactic acid) foams using starch as a natural charring agent. Ind. Eng. Chem. Res. 2014, 53, 1422–1430. 10.1021/ie403041h. [DOI] [Google Scholar]
- Zhang R.; Xiao X.; Tai Q.; Huang H.; Hu Y. Modification of lignin and its application as char agent in intumescent flame-retardant poly(lactic acid). Polym. Eng. Sci. 2012, 52, 2620–2626. 10.1002/pen.23214. [DOI] [Google Scholar]
- Liao F.; Zhou L.; Ju Y.; Yang Y.; Wang X. Synthesis of a novel phosphorus–nitrogen-silicon polymeric flame-retardant and its application in poly(lactic acid). Ind. Eng. Chem. Res. 2014, 53, 10015–10023. 10.1021/ie5008745. [DOI] [Google Scholar]
- Feng J. X.; Su S. P.; Zhu J. An intumescent flame-retardant system using β-cyclodextrin as a carbon source in polylactic acid (PLA). Polym. Adv. Technol. 2011, 22, 1115–1122. 10.1002/pat.1954. [DOI] [Google Scholar]
- Liao F.; Ju Y.; Dai X.; Cao Y.; Li J.; Wang X. A novel efficient polymeric flame-retardant for poly (lactic acid)(PLA): Synthesis and its effects on flame-retardancy and crystallization of PLA. Polym. Degrad. Stab. 2015, 120, 251–261. 10.1016/j.polymdegradstab.2015.07.012. [DOI] [Google Scholar]
- Ke C. H.; Li J.; Fang K. Y.; Zhu Q. L.; Zhu J.; Yan Q.; Wang Y. Z. Synergistic effect between a novel hyperbranched charring agent and ammonium polyphosphate on the flame-retardant and anti-dripping properties of polylactide. Polym. Degrad. Stab. 2010, 95, 763–770. 10.1016/j.polymdegradstab.2010.02.011. [DOI] [Google Scholar]
- Battegazzore D.; Frache A.; Carosio F. Layer-by-Layer nanostructured interphase produces mechanically strong and flame retardant bio-composites. Composites, Part B 2020, 200, 108310 10.1016/j.compositesb.2020.108310. [DOI] [Google Scholar]
- Kong Q.; Wu T.; Tang Y.; Xiong L.; Liu H.; Zhang J.; Guo R.; Zhang F. Improving thermal and flame-retardant properties of epoxy resin with organic NiFe-layered double hydroxide-carbon nanotubes hybrids. Chin. J. Chem. 2017, 35, 1875–1880. 10.1002/cjoc.201700313. [DOI] [Google Scholar]
- Tang G.; Zhang R.; Wang X.; Wang B.; Song L.; Hu Y.; Gong X. Enhancement of flame-retardant performance of bio-based polylactic acid composites with the incorporation of aluminum hypophosphite and expanded graphite. J. Macromol. Sci., Part A 2013, 50, 255–269. 10.1080/10601325.2013.742835. [DOI] [Google Scholar]
- Srisuk R.; Techawinyutham L.; Mavinkere Rangappa S.; Siengchin S.; Dangtungee R. Development of masterbatch for composites using bamboo charcoal powders in polylactic acid. Polym. Compos. 2020, 41, 5082–5095. 10.1002/pc.25776. [DOI] [Google Scholar]
- Chen K. S.; Chen W. Y.; Liao S. C.; Haung Y. T.; Chen S. C.; Lin H. R.; Lin F. H. Surface graft polymerization acrylic acid onto bamboo charcoal and to improve ammonia adsorption. Desalin. Water Treat. 2010, 17, 168–175. 10.5004/dwt.2010.1714. [DOI] [Google Scholar]
- Chen K. S.; Chou C. Y.; Liao S. C.; Wu H. M.; Tsai H. T. Temperature sensitivity of composite hydrogel prepared by surface graft polymer of NIPAAm and bamboo charcoal powder. Biomed. Eng. 2012, 24, 171–177. 10.4015/S1016237212500172. [DOI] [Google Scholar]
- Li X.; Wu Y.; Zheng X. Effect of nano anhydrous magnesium carbonateon fire-retardant performance of polylactic acid/bamboo fibers composites. J. Nanosci. Nanotechnol. 2011, 11, 10620–10623. 10.1166/jnn.2011.4103. [DOI] [PubMed] [Google Scholar]
- Liu S. J.; Han Y. M.; Zhu R. X.; Chu F. X.; Yu W. J. Effect of flame retardant on combustion and mechanical properties of bamboo-fiber based composites. Adv. Mater. Res. 2012, 488, 597–601. 10.4028/www.scientific.net/AMR.488-489.597. [DOI] [Google Scholar]
- Wang S.; Zhang L.; Semple K.; Zhang M.; Zhang W.; Dai C. Development of biodegradable flame-retardant bamboo charcoal composites, Part I: thermal and elemental analyses. Polymers 2020, 12, 2217 10.3390/polym12102217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X.; Lei B.; Lin Z.; Huang L.; Tan S.; Cai X. The utilization of bamboo charcoal enhances wood plastic composites with excellent mechanical and thermal properties. Mater. Des. 2014, 53, 419–424. 10.1016/j.matdes.2013.07.028. [DOI] [Google Scholar]
- Song X. Y.; Wang M.; Weng Y. X.; Huang Z. G. Effect of bamboo flour grafted lactide on the interfacial compatibility of polylactic acid/bamboo flour composites. Polymers 2017, 9, 323 10.3390/polym9080323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan Z.; Yousif B. F.; Islam M. Fracture behaviour of bamboo fiber reinforced epoxy composites. Composites, Part B 2017, 116, 186–199. 10.1016/j.compositesb.2017.02.015. [DOI] [Google Scholar]
- Li S.; Li X.; Deng Q.; Li D. Three kinds of charcoal powder reinforced ultra-high molecular weight polyethylene composites with excellent mechanical and electrical properties. Mater. Des. 2015, 85, 54–59. 10.1016/j.matdes.2015.06.163. [DOI] [Google Scholar]
- Zhang H.; Yao W.; Qian S.; Sheng K. Fabrication and reinforcement of ternary composites based on polypropylene matrix with bamboo particle/ultrafine bamboo-char. Polym. Compos. 2018, 39, 4364–4371. 10.1002/pc.24521. [DOI] [Google Scholar]
- Simis K. S.; Bistolfi A.; Bellare A.; Pruitt L. A. The combined effects of crosslinking and high crystallinity on the microstructural and mechanical properties of ultra high molecular weight polyethylene. Biomaterials 2006, 27, 1688–1694. 10.1016/j.biomaterials.2005.09.033. [DOI] [PubMed] [Google Scholar]
- Brown M. E.Introduction to Thermal Analysis: Techniques and Applications; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1988. [Google Scholar]
- Ho M. P.; Lau K. T.; Wang H.; Hui D. Improvement on the properties of polylactic acid (PLA) using bamboo charcoal particles. Composites, Part B 2015, 81, 14–25. 10.1016/j.compositesb.2015.05.048. [DOI] [Google Scholar]
- Qian S.; Yan W.; Zhu S.; Fontanillo Lopez C. A.; Sheng K. Surface modification of bamboo-char and its reinforcement in PLA biocomposites. Polym. Compos. 2018, 39, E633–E639. 10.1002/pc.24800. [DOI] [Google Scholar]
- Wang S.; Zhang L.; Semple K.; Zhang M.; Zhang W.; Dai C. Development of biodegradable flame-retardant bamboo charcoal composites, Part II: thermal degradation, gas phase, and elemental analyses. Polymers 2020, 12, 2238 10.3390/polym12102238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ge H.; Tang G.; Hu W. Z.; Wang B. B.; Pan Y.; Song L.; Hu Y. Aluminum hypophosphite microencapsulated to improve its safety and application to flame retardant polyamide 6. J. Hazard. Mater. 2015, 294, 186–194. 10.1016/j.jhazmat.2015.04.002. [DOI] [PubMed] [Google Scholar]
- Li Y.; Li X.; Pan Y. T.; Xu X.; Song Y.; Yang R. Mitigation the release of toxic PH3 and the fire hazard of PA6/AHP composite by MOFs. J. Hazard. Mater. 2020, 395, 122604 10.1016/j.jhazmat.2020.122604. [DOI] [PubMed] [Google Scholar]
- ISO 4589-2 . Standard Test Method for Plastics-Determination of Burning Behaviour by Oxygen Index; ISO: Switzerland, 2017. [Google Scholar]
- ATSM D3801-10 . Standard Test Method for Measuring the Comparative Burning Characteristics of Solid Plastics in a Vertical Position; ASTM: Philadelphia, 2020. [Google Scholar]
- ISO 5660-1 . Reaction to Fire Tests-Heat Release, Smoke Production and Mass Loss Rate; ISO: Switzerland, 2015. [Google Scholar]
- Qiao W.; Korai Y.; Mochida I.; Hori Y.; Maeda T. Preparation of an activated carbon artifact: oxidative modification of coconut shell-based carbon to improve the strength. Carbon 2002, 40, 351–358. 10.1016/s0008-6223(01)00110-5. [DOI] [Google Scholar]
- Domingo-García M.; Lopez-Garzon F. J.; Perez-Mendoza M. Effect of some oxidation treatments on the textural characteristics and surface chemical nature of an activated carbon. J. Colloids Interface Sci. 2000, 222, 233–240. 10.1006/jcis.1999.6619. [DOI] [PubMed] [Google Scholar]
- Jiang D.; Pan M.; Cai X.; Zhao Y. Flame-retardancy of rice straw-polyethylene composites affected by in situ polymerization of ammonium polyphosphate/silica. Composites, Part A 2018, 109, 1–9. 10.1016/j.compositesa.2018.02.023. [DOI] [Google Scholar]
- Ran G.; Liu X.; Guo J.; Sun J.; Li H.; Gu X.; Zhang S. Improving the flame-retardancy and water resistance of polylactic acid by introducing polyborosiloxane microencapsulated ammonium polyphosphate. Composites, Part B 2019, 173, 106772 10.1016/j.compositesb.2019.04.033. [DOI] [Google Scholar]
- McBeath A. V.; Smernik R. J.; Krull E. S.; Lehmann J. The influence of feedstock and production temperature on biochar carbon chemistry: a solid-state 13C NMR study. Biomass Bioenergy 2014, 60, 121–129. 10.1016/j.biombioe.2013.11.002. [DOI] [Google Scholar]
- Brewer C. E.Biochar Characterization and Engineering. Graduate Theses and Dissertations, Iowa State University Paper, 2012; p12284. [Google Scholar]
- Yang F. C.; Wu K. H.; Lin W. P.; Hu M. K. Preparation and antibacterial efficacy of bamboo charcoal/polyoxometalate biological protective material. Microporous Mesoporous Mater. 2009, 118, 467–472. 10.1016/j.micromeso.2008.09.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan Y.; Wang B.; Yuan S.; Wu X.; Chen J.; Wang L. Adsorptive removal of chloramphenicol from wastewater by NaOH modified bamboo charcoal. Bioresour. Technol. 2010, 101, 7661–7664. 10.1016/j.biortech.2010.04.046. [DOI] [PubMed] [Google Scholar]
- Tang C. M.; Tian Y. H.; Hsu S. H. Poly (vinyl alcohol) nanocomposites reinforced with bamboo charcoal nanoparticles: mineralization behavior and characterization. Materials 2015, 8, 4895–4911. 10.3390/ma8084895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu B. R.; Deng C.; Li Y.; Lu P.; Zhao P. P.; Wang Y. Z. Novel amino glycerin decorated ammonium polyphosphate for the highly-efficient intumescent flame-retardance of wood flour/polypropylene composite via simultaneous interfacial and bulk charring. Composites, Part B 2019, 172, 636–648. 10.1016/j.compositesb.2019.05.099. [DOI] [Google Scholar]
- Liu G.; Liu X.; Yu J. Ammonium polyphosphate with crystalline form V by ammonium dihydrogen phosphate process. Ind. Eng. Chem. Res. 2010, 49, 5523–5529. 10.1021/ie100453a. [DOI] [Google Scholar]
- Pan D.; Zhu J.; Wu Y.; Chen K.; Wu B.; Ji L. Study on the crystal transformation of ammonium polyphosphate crystalline form V. Phosphorus, Sulfur Silicon Relat. Elem. 2016, 191, 1306–1312. 10.1080/10426507.2016.1192621. [DOI] [Google Scholar]
- Ho M. P.; Lau K. T. Enhancement of impact resistance of biodegradable polymer using bamboo charcoal particles. Mater. Lett. 2014, 136, 122–125. 10.1016/j.matlet.2014.07.165. [DOI] [Google Scholar]
- Qian S.; Sheng K.; Yao W.; Yu H. Poly (lactic acid) biocomposites reinforced with ultrafine bamboo-char: Morphology, mechanical, thermal, and water absorption properties. J. Appl. Polym. Sci. 2016, 133, 43425 10.1002/app.43425. [DOI] [Google Scholar]
- Jing J.; Zhang Y.; Tang X.; Zhou Y.; Li X.; Kandola B. K.; Fang Z. Layer by layer deposition of polyethylenimine and bio-based polyphosphate on ammonium polyphosphate: A novel hybrid for simultaneously improving the flame-retardancy and toughness of polylactic acid. Polymer 2017, 108, 361–371. 10.1016/j.polymer.2016.12.008. [DOI] [Google Scholar]
- Shukor F.; Hassan A.; Islam M. S.; Mokhtar M.; Hasan M. Effect of ammonium polyphosphate on flame-retardancy, thermal stability and mechanical properties of alkali treated kenaf fiber filled PLA biocomposites. Mater. Des. 2014, 54, 425–429. 10.1016/j.matdes.2013.07.095. [DOI] [Google Scholar]
- Cao H.; Zhang Y.; Li X.; Wang F.; Zhang X. Effects of treated waste silicon rubber on properties of poly (lactic acid)/ammonium polyphosphate composites. J. Appl. Polym. Sci. 2017, 134, 45231 10.1002/app.45231. [DOI] [Google Scholar]
- Decsov K.; Bocz K.; Szolnoki B.; Bourbigot S.; Fontaine G.; Vadas D.; Marosi G. Development of bioepoxy resin microencapsulated ammonium-polyphosphate for flame-retardancy of polylactic acid. Molecules 2019, 24, 4123 10.3390/molecules24224123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schartel B.; Hull T. R. Development of fire-retarded materials—interpretation of cone calorimeter data. Fire Mater. 2007, 31, 327–354. 10.1002/fam.949. [DOI] [Google Scholar]
- Kim H.; Kim D. W.; Vasagar V.; Ha H.; Nazarenko S.; Ellison C. J. Polydopamine-graphene oxide flame-retardant nanocoatings applied via an aqueous liquid crystalline scaffold. Adv. Funct. Mater. 2018, 28, 1803172 10.1002/adfm.201803172. [DOI] [Google Scholar]
- Zhang Y.; Han P.; Fang Z. Synthesis of phospholipidated β-cyclodextrin and its application for flame-retardant poly (lactic acid) with ammonium polyphosphate. J. Appl. Polym. Sci. 2018, 135, 46054 10.1002/app.46054. [DOI] [Google Scholar]
- Stark N. M.; White R. H.; Mueller S. A.; Osswald T. A. Evaluation of various fire retardants for use in wood flour–polyethylene composites. Polym. Degrad. Stab. 2010, 95, 1903–1910. 10.1016/j.polymdegradstab.2010.04.014. [DOI] [Google Scholar]
- Chen Y.; Wang W.; Qiu Y.; Li L.; Qian L.; Xin F. Terminal group effects of phosphazene-triazine bi-group flame retardant additives in flame retardant polylactic acid composites. Polym. Degrad. Stab. 2017, 140, 166–175. 10.1016/j.polymdegradstab.2017.04.024. [DOI] [Google Scholar]
- Schartel B. Phosphorus-based flame-retardancy mechanisms—old hat or a starting point for future development?. Materials 2010, 3, 4710–4745. 10.3390/ma3104710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prabhakar M. N.; Raghavendra G. M.; Vijaykumar B. V. D.; Patil K.; Seo J.; Jung-il S. Synthesis of a novel compound based on chitosan and ammonium polyphosphate for flame-retardancy applications. Cellulose 2019, 26, 8801–8812. 10.1007/s10570-019-02671-y. [DOI] [Google Scholar]
- Zawawi E. E.; Amirrudin M.; Kamarun D.; Bonnia N. N.; Rozana M. D. Characterizations of Polylactic Acid/Polypropylene Filled with Bamboo Charcoal Powder. J. Phys.: Conf. Ser. 2019, 1349, 012122 10.1088/1742-6596/1349/1/012122. [DOI] [Google Scholar]