Abstract
Background
There are limited data regarding the factors associated with hepatocellular carcinoma (HCC) in non-alcoholic fatty liver disease (NAFLD) patients without cirrhosis. We sought to determine the prevalence and factors associated with HCC in NAFLD patients with or without cirrhosis.
Methods
Adults with NAFLD (June 2015 to May 2020) were identified using the electronic health record database (Explorys Inc, Cleveland, OH) from 26 major integrated US healthcare systems. The prevalence of HCC was calculated. Multivariable analyses adjusting for covariates were performed to evaluate the associated risk factors and the presence of HCC.
Results
A total of 392,800 NAFLD patients were identified. Among 1110 patients with HCC, 170 (15.3%) had no cirrhosis. The prevalence of HCC in non-cirrhotic and cirrhotic NAFLD patients was 4.6/10,000 persons (95% CI 3.9–5.3), and 374.4/10,000 persons (95% CI 350.9–398.8), respectively. Age > 65 years (adjusted OR; 3.37, 95% CI 2.47–4.59), ever had elevated alanine aminotransferase (2.69; 2.14–3.37), male gender (2.57; 1.88–3.49), smoker (1.75; 1.23–2.49), and diabetes (1.56; 1.15–2.11) were associated with HCC in non-cirrhotic NAFLD (all P < 0.05). The prevalence of HCC in the non-cirrhotic with all five risk factors was 45.5/10,000 persons (95% CI 17.4–73.6). The factors associated with HCC in cirrhotic NAFLD included clinical decompensation, age > 65 years, male gender, Hispanic race, elevated alanine aminotransferase, diabetes and smoker (all P < 0.05).
Conclusions
These data identified the major risk factors for the development of HCC in NAFLD patients. In the non-cirrhotics, older male patients with smoking history, diabetes and an elevated alanine aminotransferase had highest risk and may need increased judicious monitoring.
Keywords: Liver cirrhosis, Liver neoplasms, Fatty liver
Background
Nonalcoholic fatty liver disease (NAFLD) is a common complex, metabolic disease with an increasing incidence pari passu with the increase in obesity and diabetes worldwide [1]. An important liver-related complication in NAFLD is the development of hepatocellular carcinoma (HCC). In the United States, NAFLD became the second most common etiology of HCC in waitlisted liver transplant candidates in 2017 [2].
The development of HCC occurs in both cirrhotic and non-cirrhotic patients with NAFLD. Liver tumorigenesis in NAFLD is unique as it is related to interacting mechanisms including environmental factors, oxidative stress, chronic inflammation, and the immune response [3]. In the NAFLD population, there is significant higher proportion of HCC in non-cirrhotic to cirrhotic liver compared to other etiologies of liver diseases [4]. Previous data suggest that HCC occurrence in non-cirrhotic livers accounts for 20 to over 50% of all NAFLD-associated HCC cases [5–12]. Patients with non-cirrhotic NAFLD had a higher risk of HCC compared to the general population [10]. Despite the increased risk of HCC development in non-cirrhotic NAFLD patients, the risk was less than that in cirrhotics and did not exceed the annual incidence threshold for surveillance [10, 13]. Therefore, the current HCC screening recommendation is only offered to patients with cirrhosis [14] that can explain the larger tumor size in patients without cirrhosis than that in patients with cirrhosis [11, 15]. There are limited data in NAFLD patients without cirrhosis regarding the risk factors that alter the risk of HCC. This study was performed to estimate the prevalence of HCC in NAFLD patients with or without cirrhosis in the United States and to identify the factors associated with the presence of HCC in these patients, using a large electronic health record (EHR) database. Patient demographics and laboratory values were analyzed as potential risk factors that affect the prevalence of HCC in non-cirrhotic NAFLD patients.
Methods
A commercial database (Explorys Inc, Cleveland, OH), which includes EHR data from 26 major integrated US healthcare systems, was used for this study. The participating health care systems are distributed across all 50 states of the United States. The database uses de-identified inpatient and outpatient data from each participating health care organization. The data were collected from a variety of health information systems, including EHR, billing inquiries, and lab systems. The de-identified data are then standardized and normalized and are compliant with the Health Insurance Portability and Accountability Act (HIPAA) [16].
Explorys database categorizes the diagnoses, laboratory findings, and procedures by the Systematized Nomenclature of Medicine-Clinical Terms (SNOMED-CT) hierarchy, and the prescription drug orders are categorized by SNOMED terms for the pharmacologic class or RxNorm for each drug. SNOMED-CT is a comprehensive hierarchical code set, which provides more detailed information of a disease than ICD-10 code. It has been widely used in EHR for clinical data capture, storage, and research. The data are presented in number and proportion for each diagnosis, findings, procedures, and prescription drug. The database rounds cell counts to the nearest 10 for the identity protection of patients. It should be noted that all cell counts of less than five were treated as zero in this database.
Study populations
We identified a cohort of adults 18 years of age or older with NAFLD from June 2015 to May 2020. NAFLD patients included were those who had diagnoses based on SNOMED-CT of “nonalcoholic liver disease”, which is use as a synonyms for “fatty metamorphosis of the liver”, or “nonalcoholic steatohepatitis”. Patients with the diagnosis of “chronic viral hepatitis” and/or “autoimmune hepatitis” and/or “hemochromatosis” and/or “disorder caused by alcohol” were excluded. Cirrhosis patients were defined as those ever having a diagnosis of “cirrhosis” and/or “esophageal varices”, and HCC patients were defined as those having a diagnosis of “liver cell carcinoma” without the diagnosis of “secondary malignant neoplasm of the liver” and “bile duct neoplasm”.
Data collection
Patient demographic data and associated variables of interest were identified using relevant SNOMED-CT. The variables included age, gender, race, diabetes mellitus, dyslipidemia, hypertension, smoking status, obesity, morbid obesity, and elevated alanine aminotransferase (ALT). Clinical decompensation were identified in patients with cirrhosis, including the patients who had variceal bleeding, elevated bilirubin, hepatic encephalopathy or ascites. Medication data were identified by using SNOMED or RxNorm.
Statistical analysis
Patient demographic data were presented as numbers and proportions. Differences between groups of NAFLD patients with or without cirrhosis were tested using a chi-square test, or fisher exact test as appropriate. The prevalence of HCC in non-cirrhotic and cirrhotic NAFLD patients over the 5-year period was calculated. The prevalence in each subgroup was calculated by identifying patients with HCC in the subgroups of interest as a proportion of the total number of the patients at risk. The Wald method was used for the calculation of confidence intervals. To adjust for covariates, multiple searches were performed to account for every probability. Multivariable model adjusting for age, gender, and white race were performed to evaluate the associated factors and the presence of HCC in NAFLD patients with or without cirrhosis. Adjusted odds ratio (adjusted OR) and 95% confidence intervals were estimated for these associations. Stata version 15.1 (Stata Corp LCC, Texas) software was used, and a 2-sided P value to < 0.05 was considered statistically significant for all the analysis.
Results
Of the 72,563,480 individuals in the database from June 2015 to May 2020, we identified 392,800 adult patients with NAFLD. There were 25,110 patients (6.4%) with cirrhosis and 367,690 patients (93.6%) without cirrhosis.
In non-cirrhotic NAFLD patients, 58.3% were female, 34.3% were over 65 years of age, and 82.6% were Caucasian (Table 1). In the cirrhotic patients, 62.6% were female, 55.2% were over 65 years of age, and 87.3% were Caucasian. There was a higher proportion of cirrhotic patients compared to those without cirrhosis who had elevated ALT (52.8% vs. 45.4%), and a smoking history (21.1% vs. 14.9%). As expected, obesity and diabetes were more common in the cirrhotic than non-cirrhotic patients. In cirrhotic patients, 58.5% had clinical decompensation; 6.3% had variceal bleeding, 15.3% had hepatic encephalopathy, 29.6% had ascites, and 40.3% had elevated bilirubin.
Table 1.
Demographic data and clinical characteristics of patients with non-cirrhotic NAFLD and cirrhotic NAFLD
| Non-cirrhotic NAFLD n (%) |
Cirrhotic NAFLD n (%) |
P value | |
|---|---|---|---|
| Total | 367,690 (100) | 25,110 (100) | |
| Gender | < 0.001 | ||
| Male gender | 153,250 (41.7) | 9380 (37.4) | |
| Female gender | 214,440 (58.3) | 15,730 (62.6) | |
| Age group | < 0.001 | ||
| Age > 65 years | 126,100 (34.3) | 13,860 (55.2) | |
| Age 18–65 years | 241,590 (65.7) | 11,250 (44.8) | |
| Race | < 0.001 | ||
| Non-Hispanic white | 303,750 (82.6) | 21,910 (87.3) | |
| Non-Hispanic black | 31,510 (8.6) | 1240 (4.9) | |
| Hispanic | 6260 (1.7) | 560 (2.2) | |
| Others/unknown | 26,170 (7.1) | 1400 (5.6) | |
| Comorbidities | |||
| Obesity | 187,820 (51.1) | 13,810 (55.0) | < 0.001 |
| Morbid obesity | 91,790 (25.0) | 7560 (30.1) | < 0.001 |
| Diabetes mellitus | 154,710 (42.1) | 16,860 (67.1) | < 0.001 |
| Hypertension | 255,190 (69.4) | 20,120 (80.1) | < 0.001 |
| Hyperlipidemia | 256,880 (69.9) | 17,480 (69.6) | 0.405 |
| Smoker | 54,870 (14.9) | 5310 (21.1) | < 0.001 |
| Laboratory data | |||
| High ALT | 166,910 (45.4) | 13,250 (52.8) | < 0.001 |
| Medication use | |||
| Statin | 177,160 (48.2) | 13,060 (52.0) | < 0.001 |
| ACEI or ARBs | 197,440 (53.7) | 15,950 (63.5) | < 0.001 |
| Metformin | 113,480 (30.9) | 10,550 (42.0) | < 0.001 |
| Sulfonylurea | 50,940 (13.9) | 6420 (25.6) | < 0.001 |
| Thiazolidinedione | 16,170 (4.4) | 1890 (7.5) | < 0.001 |
| Insulin | 82,900 (22.6) | 12,530 (49.9) | < 0.001 |
| Liver decompensation | – | 14,690 (58.5) | |
| Variceal bleeding | – | 1590 (6.3) | |
| Hepatic encephalopathy | – | 3830 (15.3) | |
| Ascites | – | 7420 (29.6) | |
| Elevated bilirubin | – | 10,130 (40.3) | |
| HCC | 170 (0.000462) | 940 (0.037435) | < 0.001 |
ACEI angiotensin converting enzyme inhibitor, ALT alanine aminotransferases, ARBs angiotensin ii receptor blocker, HCC hepatocellular carcinoma, NAFLD non-alcoholic fatty liver disease
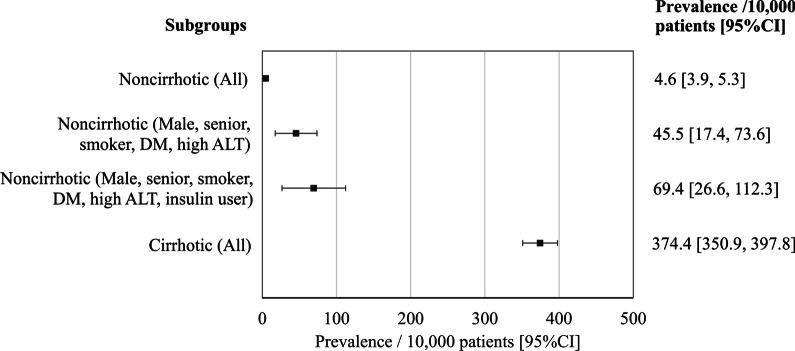
Of the 1110 HCC found in NAFLD patients, 15% occurred in non-cirrhotics. The prevalence of HCC in non-cirrhotic and cirrhotic NAFLD patients was 4.6/10,000 persons (95% CI 3.9–5.3/10,000 persons), and 374.4/10,000 persons (95% CI 350.9–397.8/10,000 persons), respectively (Fig. 1). The characteristics of HCC patients stratified by cirrhosis and no cirrhosis is shown in Table 2.
Fig. 1.
Prevalence (per 10,000 patients) of HCC among subgroups of patients with NAFLD
Table 2.
Demographic data and clinical characteristics of patients with HCC
| HCC in non-cirrhotic NAFLD n (%) |
HCC in cirrhotic NAFLD n (%) |
P value | |
|---|---|---|---|
| Total | 170 (100) | 940 (100) | |
| Gender | < 0.397 | ||
| Male gender | 100 (58.8) | 520 (55.3) | |
| Female gender | 70 (41.2) | 420 (44.7) | |
| Age group | 0.008 | ||
| Age > 65 years | 110 (64.7) | 700 (74.5) | |
| Age 18–65 years | 60 (35.3) | 240 (25.5) | |
| Race | < 0.001 | ||
| Non-Hispanic white | 130 (76.4) | 800 (85.1) | |
| Non-Hispanic black | 20 (11.8) | 20 (2.1) | |
| Hispanic | 20 (11.8) | 120 (12.8) | |
| Comorbidities | |||
| Obesity | 70 (41.2) | 490 (52.1) | 0.009 |
| Morbid obesity | 20 (11.8) | 240 (25.5) | < 0.001 |
| Diabetes mellitus | 100 (58.8) | 700 (74.5) | < 0.001 |
| Hypertension | 140 (82.4) | 770 (81.9) | 0.891 |
| Hyperlipidemia | 130 (76.5) | 590 (62.8) | 0.001 |
| Smoker | 30 (17.6) | 230 (24.5) | 0.053 |
| Laboratory data | |||
| High ALT | 110 (64.7) | 600 (63.8) | 0.827 |
| Medication use | |||
| Statin | 100 (58.8) | 450 (47.9) | 0.009 |
| ACEI or ARBs | 120 (70.6) | 610 (64.9) | 0.150 |
| Metformin | 60 (35.3) | 390 (41.5) | 0.130 |
| Sulfonylurea | 40 (23.5) | 280 (39.8) | 0.097 |
| Thiazolidinedione | 0 (0) | 70 (7.4) | < 0.001 |
| Insulin | 80 (47.0) | 620 (66.0) | < 0.001 |
| Liver decompensation | – | 675 (71.8) |
Factors associated with hepatocellular carcinoma in non-cirrhotic NAFLD
HCC was diagnosed in 170 patients with NAFLD without cirrhosis with a prevalence of 4.6/10,000 persons within a 5-year period. As shown in Table 3, in multivariable analyses, age > 65 years (adjusted OR 3.37; 95% CI 2.47–4.59), elevated ALT (adjusted OR 2.69; 95% CI 2.14–3.37), male gender (adjusted OR 2.57; 95% CI 1.88–3.49), a smoking history (adjusted OR 1.75; 95% CI 1.23–2.49), and diabetes mellitus (adjusted OR 1.56; 95% CI 1.15–2.11) were significant factors associated with HCC. The prevalence of HCC was 45.5/10,000 persons (95% CI 17.4–73.6/10,000 persons) in patients with all five risk factors (Fig. 1). When insulin use is added to the above five risk factors, the risk of developing HCC in non-cirrhotic NAFLD increases to 69.44/10,000 persons (95% CI 26.55–112.34/10,000 persons).
Table 3.
Multivariable analysis of the factors associated with HCC prevalence in non-cirrhotic NAFLD
| Noncirrhotic NAFLD | |||
|---|---|---|---|
| Adjusted ORa | 95% CI | P value | |
| Age > 65 years | 3.37 | 2.47–4.59 | < 0.001 |
| Male gender | 2.57 | 1.88–3.49 | < 0.001 |
| Non-Hispanic white | 0.67 | 0.47–0.97 | 0.036 |
| Non-Hispanic black | 0.74 | 0.45–1.23 | 0.245 |
| Hispanic | 1.32 | 0.91–1.91 | 0.150 |
| Diabetes | 1.56 | 1.15–2.11 | 0.004 |
| Hypertension | 1.24 | 0.84–1.83 | 0.278 |
| Hyperlipidemia | 0.95 | 0.66–1.37 | 0.793 |
| Obese | 0.84 | 0.62–1.13 | 0.252 |
| Morbid obese | 1.06 | 0.81–1.39 | 0.658 |
| Smoker | 1.75 | 1.23–2.49 | 0.002 |
| High ALT | 2.69 | 2.14–3.37 | < 0.001 |
ALT alanine aminotransferases, CI confidence interval, HCC hepatocellular carcinoma, NAFLD non-alcoholic fatty liver disease, OR odds ratio
aFactors adjusted with age, gender and white race
Factors associated with hepatocellular carcinoma in cirrhotic NAFLD
HCC was diagnosed in 940 cirrhotic NAFLD patients with the prevalence of 374.4/10,000 persons within a 5-year period. In multivariable analyses (Table 4), decompensated liver (adjusted OR 2.78; 95% CI 2.44–3.17), age > 65 years (adjusted OR 2.35; 95% CI 2.05–2.70), male gender (adjusted OR 2.24; 95% CI 1.98–2.53), elevated ALT (adjusted OR 1.97; 95% CI 1.74–2.24), Hispanic ethnicity (adjusted OR 1.66; 95% CI 1.39–1.99), diagnosed with diabetes (adjusted OR 1.27; 95% CI 1.10–1.46), and a smoking history (adjusted OR 1.20; 95% CI 1.05–1.38) were associated with HCC. Patients who were Caucasian, African–Americans or had hyperlipidemia were less likely to have HCC (adjusted OR 0.79; 95% CI 0.66–0.94, 0.40; 95% CI 0.26–0.61, and 0.68; 95% CI 0.60–0.77 respectively).
Table 4.
Multivariable analysis of the factors associated with HCC prevalence in cirrhotic NAFLD
| Cirrhotic NAFLD | |||
|---|---|---|---|
| Adjusted ORa | 95% CI | P value | |
| Age > 65 years | 2.35 | 2.05–2.70 | < 0.001 |
| Male gender | 2.24 | 1.98–2.53 | < 0.001 |
| Non-Hispanic white | 0.79 | 0.66–0.94 | 0.008 |
| Non-Hispanic black | 0.40 | 0.26–0.61 | < 0.001 |
| Hispanic | 1.66 | 1.39–1.99 | < 0.001 |
| Diabetes | 1.27 | 1.10–1.46 | 0.001 |
| Hypertension | 0.89 | 0.75–1.05 | 0.173 |
| Hyperlipidemia | 0.68 | 0.60–0.77 | < 0.001 |
| Obese | 1.12 | 0.98–1.28 | 0.085 |
| Morbid obese | 0.97 | 0.85–1.11 | 0.685 |
| Smoker | 1.20 | 1.05–1.38 | 0.007 |
| Decompensation | 2.78 | 2.44–3.17 | < 0.001 |
| High ALT | 1.97 | 1.74–2.24 | < 0.001 |
ALT alanine aminotransferases, CI confidence interval, HCC hepatocellular carcinoma, NAFLD non-alcoholic fatty liver disease, OR odds ratio
aFactors adjusted with age, gender and white race
Discussion
There are three major findings in this study. First and not surprisingly, cirrhosis is the major risk factor for the development of HCC in NAFLD patients. Second, although the risk of HCC in non-cirrhotic NAFLD is low overall, there is increased risk for HCC in the subgroup of male patients over 65 with a history of smoking, an elevated ALT, and diabetes mellitus. In non-cirrhotics with all five of these risk factors, there is a 9.9-fold increased risk for developing HCC compared to overall prevalence in non-cirrhotic NAFLD patients. Third, ethnicity may have a significant impact on the risk of HCC development in NAFLD population.
Cirrhosis is well-known to be a major risk factor for the development of HCC in all etiologies of liver diseases [17]. Our study confirmed that the presence of liver cirrhosis is the most potent risk factor for HCC in NAFLD patients. The ratio of HCC prevalence in the cirrhotic/non-cirrhotic in NAFLD patients was 81-fold. The proportion of HCC in non-cirrhotic NAFLD patients to HCC in all NAFLD was 15.3%, in our study, which is slightly lower than previously reported (20–63%) [5–12]. However, the proportion of non-cirrhotic NAFLD patients with HCC varied widely between studies and may be due to studies reported from tertiary centers and the disproportionate contribution of non-cirrhotic patients with HCC who undergo surgical resection.
Of the risk factors identified, age > 65 years was the highest risk factor for HCC development in non-cirrhotic NAFLD patients and was similar to reports by others [18]. Our observations of a higher association of HCC with male gender was also consistent with previous study [19]. The gender disparity might be explained by differences in hormonal functions, in which estrogen might be protective against HCC development [20] and the effect of sex hormones on visceral fat [21] Diabetes mellitus [22, 23] and smoking history [24, 25] are known risk factors for HCC as was observed in our cohort. Prior data on the predictive value of ALT levels for long-term liver-related outcomes in NAFLD remain limited. However, elevated ALT suggests histologic nonalcoholic steatohepatitis [26, 27] with fibrosis [27]. A study, using a hepatoprotectant as a surrogate marker of elevated ALT demonstrated a significant risk for the development of HCC in non-cirrhotic NAFLD patients [18]. It is important that future research investigate the correlation between elevated transaminases and the risk of HCC.
In the current study, clinical decompensation in cirrhotic patients was associated with a higher risk of HCC, while patients with hyperlipidemia had a lower risk of HCC. It is clear that more advanced liver disease had a higher risk of clinical decompensation and liver related complications. The protective effect of hyperlipidemia against the development of HCC could be related to better liver function or the likelihood that these patients were more likely to be on a statin.
The racial differences reported in the prevalence of NAFLD [28] may explain, in part, the variation in risk of HCC development in NAFLD patients in the present study and prior reports [10]. Our observation in this large cohort of NAFLD patients shows that a lower risk of HCC in cirrhotic African Americans and both cirrhotic and non-cirrhotic non-Hispanic whites, compared with Hispanics with cirrhosis. However, it is possible that ethnicity, health behavior, and socioeconomic status are correlated and prospective studies in large cohorts are required to dissect the contribution of ethnicity to the risk of HCC in NAFLD.
Despite the increasing risk of HCC observed in obese patients in the general population [29] and other chronic liver diseases [30, 31], obesity and morbid obesity were not independently associated with HCC in NAFLD patients in our study and was consistent with the data reported from a previous longitudinal study [10]. One potential explanation may be the association of obesity with both NAFLD and HCC.
Limitations of our study include the potential for the same patient to be included more than once despite Explorys using a robust patient matching algorithm to match the same patient across different participating healthcare institutions and combining the data. Some information could not be obtained from the database as all the data was de-identified and categorized. Therefore, manual individual chart review, an analysis of continuous variables, and the sensitivity analysis cannot be performed when using this database. Whether HCC was newly diagnosed or recurrence was unknown. Several variables associated with the development of HCC in NAFLD, for example, gene polymorphisms, and the NAFLD treatment, especially non-pharmacological treatment, were not available and these factors should be further investigated. The aforementioned limitation of this database precluded the investigator to retrieve the liver biopsy data and the method of HCC diagnosis. Hence, there was a potential risk that within the group classed as non-cirrhotics, there could be patients with asymptomatic cirrhosis which has not been reported. However, these limitations are overcome by the major strength of our study of the analysis of a large population-based data set that included both inpatients and outpatients from a large cross section in the United States and that our study provide hypothesis-generating for the future research. The utilization of basic clinical data to identify the non-cirrhotic NAFLD patients who have a high risk for HCC development in our study will help clinical practice and the design of future studies.
Conclusions
Cirrhosis in NAFLD patients poses the most significant risk for HCC occurrence. Among non-cirrhotic NAFLD patients, male patients, over 65 years old, with diabetes mellitus and a history of smoking with an elevated ALT were associated with the greatest risk of HCC. These patients could benefit from the offer of HCC surveillance and prospective study is warranted to further confirm these findings.
Acknowledgements
Not applicable.
Abbreviations
- ALT
Alanine aminotransferase
- HCC
Hepatocellular carcinoma
- HER
Electronic health record
- HIPAA
Health Insurance Portability and Accountability Act
- NAFLD
Non-alcoholic fatty liver disease
- OR
Odds ratio
- SNOMED-CT
Systematized Nomenclature of Medicine-Clinical Terms
Authors' contributions
KWP, AM, EM, MA, and KPP were involved in the study conception and the design. KWP and EM were involved in the acquisition of the data. KWP, GK, and CA analyzed and interpretated the data. KWP, KPP, and GK drafted the manuscript. All authors read and approved the final manuscript.
Funding
Supported in part by RO1 GM119174 (SD); RO1 DK113196 (SD); P50 AA024333 (AJM, SD); UO1 AA 021890 (AJM, SD); UO1 DK 061732 (AJM, SD).
Availability of data and materials
The data that support the findings of this study are available from a commercial database (Explorys Inc, Cleveland, OH) but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available.
Declarations
Ethics approval and consent to participate
Ethics and consent were not required as it was a secondary analysis of an anonymizes dataset. All methods were carried out in accordance with relevant guidelines and regulations.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no conflict of interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Allen AM, Therneau TM, Larson JJ, Coward A, Somers VK, Kamath PS. Nonalcoholic fatty liver disease incidence and impact on metabolic burden and death: a 20 year-community study. Hepatology. 2018;67(5):1726–1736. doi: 10.1002/hep.29546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Younossi Z, Stepanova M, Ong JP, Jacobson IM, Bugianesi E, Duseja A, Eguchi Y, Wong VW, Negro F, Yilmaz Y, et al. Nonalcoholic steatohepatitis is the fastest growing cause of hepatocellular carcinoma in liver transplant candidates. Clin Gastroenterol Hepatol. 2019;17(4):748–755.e743. doi: 10.1016/j.cgh.2018.05.057. [DOI] [PubMed] [Google Scholar]
- 3.Anstee QM, Reeves HL, Kotsiliti E, Govaere O, Heikenwalder M. From NASH to HCC: current concepts and future challenges. Nat Rev Gastroenterol Hepatol. 2019;16(7):411–428. doi: 10.1038/s41575-019-0145-7. [DOI] [PubMed] [Google Scholar]
- 4.Stine JG, Wentworth BJ, Zimmet A, Rinella ME, Loomba R, Caldwell SH, Argo CK. Systematic review with meta-analysis: risk of hepatocellular carcinoma in non-alcoholic steatohepatitis without cirrhosis compared to other liver diseases. Aliment Pharmacol Ther. 2018;48(7):696–703. doi: 10.1111/apt.14937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Piscaglia F, Svegliati-Baroni G, Barchetti A, Pecorelli A, Marinelli S, Tiribelli C, Bellentani S. Group H-NIS: clinical patterns of hepatocellular carcinoma in nonalcoholic fatty liver disease: a multicenter prospective study. Hepatology. 2016;63(3):827–838. doi: 10.1002/hep.28368. [DOI] [PubMed] [Google Scholar]
- 6.Ertle J, Dechene A, Sowa JP, Penndorf V, Herzer K, Kaiser G, Schlaak JF, Gerken G, Syn WK, Canbay A. Non-alcoholic fatty liver disease progresses to hepatocellular carcinoma in the absence of apparent cirrhosis. Int J Cancer. 2011;128(10):2436–2443. doi: 10.1002/ijc.25797. [DOI] [PubMed] [Google Scholar]
- 7.Yasui K, Hashimoto E, Komorizono Y, Koike K, Arii S, Imai Y, Shima T, Kanbara Y, Saibara T, Mori T, et al. Characteristics of patients with nonalcoholic steatohepatitis who develop hepatocellular carcinoma. Clin Gastroenterol Hepatol. 2011;9(5):428–433. doi: 10.1016/j.cgh.2011.01.023. [DOI] [PubMed] [Google Scholar]
- 8.Mohamad B, Shah V, Onyshchenko M, Elshamy M, Aucejo F, Lopez R, Hanouneh IA, Alhaddad R, Alkhouri N. Characterization of hepatocellular carcinoma (HCC) in non-alcoholic fatty liver disease (NAFLD) patients without cirrhosis. Hepatol Int. 2016;10(4):632–639. doi: 10.1007/s12072-015-9679-0. [DOI] [PubMed] [Google Scholar]
- 9.Mittal S, El-Serag HB, Sada YH, Kanwal F, Duan Z, Temple S, May SB, Kramer JR, Richardson PA, Davila JA. Hepatocellular carcinoma in the absence of cirrhosis in united states veterans is associated with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol. 2016;14(1):124–131.e121. doi: 10.1016/j.cgh.2015.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kanwal F, Kramer JR, Mapakshi S, Natarajan Y, Chayanupatkul M, Richardson PA, Li L, Desiderio R, Thrift AP, Asch SM, et al. Risk of hepatocellular cancer in patients with non-alcoholic fatty liver disease. Gastroenterology. 2018;155(6):1828–1837.e1822. doi: 10.1053/j.gastro.2018.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bengtsson B, Stal P, Wahlin S, Bjorkstrom NK, Hagstrom H. Characteristics and outcome of hepatocellular carcinoma in patients with NAFLD without cirrhosis. Liver Int. 2019;39(6):1098–1108. doi: 10.1111/liv.14087. [DOI] [PubMed] [Google Scholar]
- 12.Pais R, Fartoux L, Goumard C, Scatton O, Wendum D, Rosmorduc O, Ratziu V. Temporal trends, clinical patterns and outcomes of NAFLD-related HCC in patients undergoing liver resection over a 20-year period. Aliment Pharmacol Ther. 2017;46(9):856–863. doi: 10.1111/apt.14261. [DOI] [PubMed] [Google Scholar]
- 13.Kawamura Y, Arase Y, Ikeda K, Seko Y, Imai N, Hosaka T, Kobayashi M, Saitoh S, Sezaki H, Akuta N, et al. Large-scale long-term follow-up study of Japanese patients with non-alcoholic fatty liver disease for the onset of hepatocellular carcinoma. Am J Gastroenterol. 2012;107(2):253–261. doi: 10.1038/ajg.2011.327. [DOI] [PubMed] [Google Scholar]
- 14.Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, Harrison SA, Brunt EM, Sanyal AJ. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67(1):328–357. doi: 10.1002/hep.29367. [DOI] [PubMed] [Google Scholar]
- 15.Kodama K, Kawaguchi T, Hyogo H, Nakajima T, Ono M, Seike M, Takahashi H, Nozaki Y, Kawanaka M, Tanaka S, et al. Clinical features of hepatocellular carcinoma in nonalcoholic fatty liver disease patients without advanced fibrosis. J Gastroenterol Hepatol. 2019;34(9):1626–1632. doi: 10.1111/jgh.14608. [DOI] [PubMed] [Google Scholar]
- 16.Kaelber DC, Foster W, Gilder J, Love TE, Jain AK. Patient characteristics associated with venous thromboembolic events: a cohort study using pooled electronic health record data. J Am Med Inform Assoc. 2012;19(6):965–972. doi: 10.1136/amiajnl-2011-000782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tarao K, Nozaki A, Ikeda T, Sato A, Komatsu H, Komatsu T, Taguri M, Tanaka K. Real impact of liver cirrhosis on the development of hepatocellular carcinoma in various liver diseases-meta-analytic assessment. Cancer Med. 2019;8(3):1054–1065. doi: 10.1002/cam4.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee TY, Wu JC, Yu SH, Lin JT, Wu MS, Wu CY. The occurrence of hepatocellular carcinoma in different risk stratifications of clinically noncirrhotic nonalcoholic fatty liver disease. Int J Cancer. 2017;141(7):1307–1314. doi: 10.1002/ijc.30784. [DOI] [PubMed] [Google Scholar]
- 19.Yao KF, Ma M, Ding GY, Li ZM, Chen HL, Han B, Chen Q, Jiang XQ, Wang LS. Meta-analysis reveals gender difference in the association of liver cancer incidence and excess BMI. Oncotarget. 2017;8(42):72959–72971. doi: 10.18632/oncotarget.20127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yang W, Lu Y, Xu Y, Xu L, Zheng W, Wu Y, Li L, Shen P. Estrogen represses hepatocellular carcinoma (HCC) growth via inhibiting alternative activation of tumor-associated macrophages (TAMs) J Biol Chem. 2012;287(48):40140–40149. doi: 10.1074/jbc.M112.348763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cheung OK, Cheng AS. Gender differences in adipocyte metabolism and liver cancer progression. Front Genet. 2016;7:168. doi: 10.3389/fgene.2016.00168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Alexander M, Loomis AK, van der Lei J, Duarte-Salles T, Prieto-Alhambra D, Ansell D, Pasqua A, Lapi F, Rijnbeek P, Mosseveld M, et al. Risks and clinical predictors of cirrhosis and hepatocellular carcinoma diagnoses in adults with diagnosed NAFLD: real-world study of 18 million patients in four European cohorts. BMC Med. 2019;17(1):95. doi: 10.1186/s12916-019-1321-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang C, Wang X, Gong G, Ben Q, Qiu W, Chen Y, Li G, Wang L. Increased risk of hepatocellular carcinoma in patients with diabetes mellitus: a systematic review and meta-analysis of cohort studies. Int J Cancer. 2012;130(7):1639–1648. doi: 10.1002/ijc.26165. [DOI] [PubMed] [Google Scholar]
- 24.Petrick JL, Campbell PT, Koshiol J, Thistle JE, Andreotti G, Beane-Freeman LE, Buring JE, Chan AT, Chong DQ, Doody MM, et al. Tobacco, alcohol use and risk of hepatocellular carcinoma and intrahepatic cholangiocarcinoma: the Liver Cancer Pooling Project. Br J Cancer. 2018;118(7):1005–1012. doi: 10.1038/s41416-018-0007-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Abdel-Rahman O, Helbling D, Schob O, Eltobgy M, Mohamed H, Schmidt J, Giryes A, Mehrabi A, Iype S, John H, et al. Cigarette smoking as a risk factor for the development of and mortality from hepatocellular carcinoma: an updated systematic review of 81 epidemiological studies. J Evid Based Med. 2017;10(4):245–254. doi: 10.1111/jebm.12270. [DOI] [PubMed] [Google Scholar]
- 26.Verma S, Jensen D, Hart J, Mohanty SR. Predictive value of ALT levels for non-alcoholic steatohepatitis (NASH) and advanced fibrosis in non-alcoholic fatty liver disease (NAFLD) Liver Int. 2013;33(9):1398–1405. doi: 10.1111/liv.12226. [DOI] [PubMed] [Google Scholar]
- 27.Ballestri S, Nascimbeni F, Romagnoli D, Lonardo A. The independent predictors of non-alcoholic steatohepatitis and its individual histological features: insulin resistance, serum uric acid, metabolic syndrome, alanine aminotransferase and serum total cholesterol are a clue to pathogenesis and candidate targets for treatment. Hepatol Res. 2016;46(11):1074–1087. doi: 10.1111/hepr.12656. [DOI] [PubMed] [Google Scholar]
- 28.Rich NE, Oji S, Mufti AR, Browning JD, Parikh ND, Odewole M, Mayo H, Singal AG. Racial and ethnic disparities in nonalcoholic fatty liver disease prevalence, severity, and outcomes in the United States: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2018;16(2):198–210.e192. doi: 10.1016/j.cgh.2017.09.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Welzel TM, Graubard BI, Zeuzem S, El-Serag HB, Davila JA, McGlynn KA. Metabolic syndrome increases the risk of primary liver cancer in the United States: a study in the SEER-Medicare database. Hepatology. 2011;54(2):463–471. doi: 10.1002/hep.24397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ohki T, Tateishi R, Sato T, Masuzaki R, Imamura J, Goto T, Yamashiki N, Yoshida H, Kanai F, Kato N, et al. Obesity is an independent risk factor for hepatocellular carcinoma development in chronic hepatitis C patients. Clin Gastroenterol Hepatol. 2008;6(4):459–464. doi: 10.1016/j.cgh.2008.02.012. [DOI] [PubMed] [Google Scholar]
- 31.Chen CL, Yang HI, Yang WS, Liu CJ, Chen PJ, You SL, Wang LY, Sun CA, Lu SN, Chen DS, et al. Metabolic factors and risk of hepatocellular carcinoma by chronic hepatitis B/C infection: a follow-up study in Taiwan. Gastroenterology. 2008;135(1):111–121. doi: 10.1053/j.gastro.2008.03.073. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from a commercial database (Explorys Inc, Cleveland, OH) but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available.