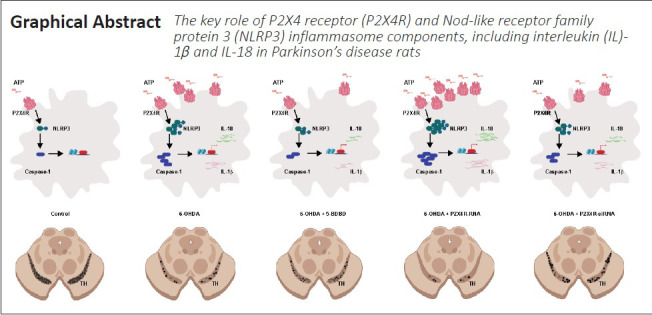
Key Words: ATP, neurodegenerative disorder, neuroinflammation, neuroinflammatory response, NLRP3, P2X4, Parkinson's disease
Abstract
Previous studies have shown that the ATP-P2X4 receptor signaling pathway mediates the activation of the Nod-like receptor family protein 3 (NLRP3) inflammasome. The NLRP3 inflammasome may promote renal interstitial inflammation in diabetic nephropathy. As inflammation also plays an important role in the pathogenesis of Parkinson’s disease, we hypothesized that the ATP-P2X4 receptor signaling pathway may activate the NLRP3 inflammasome in Parkinson’s disease. A male rat model of Parkinson’s disease was induced by stereotactic injection of 6-hydroxydopamine into the pars compacta of the substantia nigra. The P2X4 receptor and the NLRP3 inflammasome (interleukin-1β and interleukin-18) were activated. Intracerebroventricular injection of the selective P2X4 receptor antagonist 5-(3-bromophenyl)-1,3-dihydro-2H-benzofuro[3,2-e]-1,4-diazepin-2-one (5-BDBD) or knockdown of P2X4 receptor expression by siRNA inhibited the activation of the NLRP3 inflammasome and alleviated dopaminergic neurodegeneration and neuroinflammation. Our results suggest that the ATP-P2X4 receptor signaling pathway mediates NLRP3 inflammasome activation, dopaminergic neurodegeneration, and dopamine levels. These findings reveal a novel role of the ATP-P2X4 axis in the molecular mechanisms underlying Parkinson’s disease, thus providing a new target for treatment. This study was approved by the Animal Ethics Committee of Qingdao University, China, on March 5, 2015 (approval No. QYFYWZLL 26119).
Chinese Library Classification No. R456; R741; R318
Introduction
Parkinson’s disease (PD) is the second-most-common progressive neurodegenerative disorder (Moon and Paek, 2015). Although the exact mechanisms underlying PD remain unknown, inflammatory factors are critical determinants of PD onset (Forloni et al., 2021; Martin-Bastida et al., 2021). Accordingly, the role of inflammatory signaling pathways in the pathogenesis of PD has become a focus of recent research. Secretion of inflammatory molecules and degeneration of dopaminergic neurons are regulated by inflammatory pathways and may influence the development of PD (Lee et al., 2021). Therefore, a better understanding of the pathways underlying PD pathogenesis will facilitate the development of novel neuroprotective strategies.
The P2X4 receptor (P2X4R), a ligand-gated ion channel that is part of the ionotropic purinergic P2X receptor (P2XR) family, is sensitive to ATP and functions as a nonselective cation channel to permit Na+, K+, and Ca2+ fluxes (Burnstock, 2015; Zhang et al., 2020). Extracellular ATP is widely recognized as an exclusive neurotransmitter or an important co-neurotransmitter in most nerve types. P2X4R was the first P2X receptor identified in the central nervous system (CNS) (Antonioli et al., 2019). Along with P2X1R and P2X6R, it is among the most widely expressed ATP-gated purinergic receptor in most neurons and glial cells (Burnstock et al., 2011). P2X4 regulates microglial cell activation and migration at a site of injury (Trang et al., 2020). Microglia regulate cytokine production and inflammatory processes in the brain (Duveau et al., 2020). Elevated pro-inflammatory cytokines and chemokines typically define a state of neuroinflammation (González et al., 2014). Some neurodegenerative diseases are associated with neuroinflammation, in which microglia play a central role (Mehrabadi and Sadr, 2020; Rivers-Auty et al., 2021). In patients with PD, P2X4R may exacerbate inflammation in the CNS by regulating microglial pathways (Liu et al., 2013). The ATP-P2X4R signaling axis mediates activation of the NLRP3 inflammasome. Extracellular ATP mediates the inflammatory responses and activates the NLRP3 inflammasome via P2X7R and P2X4R (Chen et al., 2018).
The NLRP3 inflammasome is composed of NLRP3, procaspase-1, and apoptosis-associated speck-like protein containing a caspase recruitment domain-containing protein (Malhotra et al., 2020; Wani et al., 2021). It senses danger signals including lipopolysaccharides and high glucose levels and then triggers the inflammatory cascade, resulting in the cleavage of procaspase-1 and the activation of interleukin (IL)-1β, IL-18, and IL-33 (Piccini et al., 2008; Jiang et al., 2021).
The NLRP3 inflammasome plays an important role in the molecular mechanisms underlying many inflammatory diseases, including diabetes, Alzheimer’s disease, arteriosclerosis, and gout (Tan et al., 2013; Holbrook et al., 2021). The inflammasome may also contribute to renal interstitial inflammation in diabetic nephropathy (Chen et al., 2013). Dopamine may exert anti-inflammatory effects by inhibiting NLRP3 (Yan et al., 2015). Because inflammatory factors play a crucial role in the onset of PD, NLRP3 activation is very likely to participate in PD pathogenesis (Mao et al., 2017). Therefore, we hypothesized that the ATP-P2X4R axis mediates NLRP3 inflammasome activation in PD. The present study was performed to verify this hypothesis.
Materials and Methods
Experimental animals
Two hundred and forty adult male Wistar rats of 40–50 days of age (specific-pathogen-free, weighing 180–200 g) were obtained from the Qingdao Experimental Animal Center, Qingdao, China (license No. SCXK (Lu) 2014-0001). This study adhered to the principles of the Basel Declaration and to the recommendations of the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH publication No. 8023). The protocol was approved by the Animal Ethics Committee of Qingdao University, China (approval No. QYFYWZLL 26119) on March 5, 2015. Isoflurane (MilliporeSigma, St. Louis, MO, USA) was used to anesthetize animals via inhalation.
Experimental design and procedures
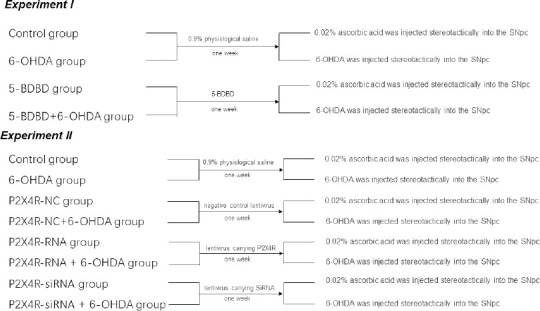
Experiment I
Rats were divided randomly into four treatment groups (n = 20 in each group; Figure 1). Control group: after 1 week of pretreatment with 0.9% physiological saline, we injected 0.02% ascorbic acid into the substantia nigra pars compacta (SNpc) stereotactically; 6-hydroxydopamine (6-OHDA) group: after 1 week of pretreatment with 0.9% physiological saline, we injected 6-OHDA into the SNpc stereotactically to establish PD; 5-(3-bromophenyl)-1,3-dihydro-2H-benzofuro3,2-e]-1,4-diazepin-2-one (5-BDBD) group: after 1 week of pretreatment with 5-BDBD (a selective P2X4R antagonist), we injected ascorbic acid into the SNpc stereotactically; and 5-BDBD + 6-OHDA group: after 1 week of pretreatment with 5-BDBD, we injected 6-OHDA into the SNpc stereotactically.
Figure 1.
The study groups and interventions.
5-BDBD: 5-(3-Bromophenyl)-1,3-dihydro-2H-benzofuro3,2-e]-1,4-diazepin-2-one; 6-OHDA: 6-hydroxydopamine; NC: negative control lentivirus; P2X4R: P2X4 receptor; siRNA: small-interfering RNA; SNpc: substantia nigra pars compacta.
Experiment II
Rats were divided randomly into eight treatment groups (n = 20 in each group; Figure 1). Control group: after 1 week of pretreatment with 0.9% physiological saline, we injected 0.02% ascorbic acid into the SNpc stereotactically; 6-OHDA group: after 1 week of pretreatment with 0.9% physiological saline, we injected 6-OHDA into the SNpc stereotactically to establish PD; P2X4R-NC group: after 1 week of pretreatment with a negative control lentivirus (NC), we injected 0.02% ascorbic acid into the SNpc stereotactically; P2X4R-NC + 6-OHDA group: after 1 week of pretreatment with the negative control lentivirus, we injected 6-OHDA into the SNpc stereotactically; P2X4R-RNA group: after 1 week of pretreatment with a lentivirus carrying P2X4R, we injected 0.02% ascorbic acid into the SNpc stereotactically; P2X4R-RNA + 6-OHDA group: after 1 week of pretreatment with a lentivirus carrying P2X4R, we injected 6-OHDA into the SNpc stereotactically; P2X4R-siRNA group: after 1 week of pretreatment with a lentivirus carrying small-interfering RNA (siRNA) targeting P2X4R, we injected 0.02% ascorbic acid into the SNpc stereotactically; P2X4R-siRNA + 6-OHDA group: after 1 week of pretreatment with the lentivirus carrying siRNA targeting P2X4R, we injected 6-OHDA into the SNpc stereotactically.
Intracerebroventricular injection of 5-BDBD
Each rat was slowly injected, over 4 minutes, with 10 µg of 5-BDBD (MilliporeSigma) dissolved in 2 µL of 0.9% physiological saline into the left lateral ventricle, determined according to the stereotactic coordinates calculated using the rat brain atlas, The Rat Brain in Stereotaxic Coordinates, 7th edition (Paxinos and Watson, 2013): posterior of anterior fontanel, –0.9 mm; mediolateral, –1.5 mm; dorsoventral, 3.5 mm. After daily injections for 1 week, the animals were injected every other day for 2 weeks. Rats in the control and 6-OHDA groups in Experiment I were stereotactically injected with 2 µL of 0.9% physiological saline.
Pretreatment with lentiviral vectors carrying P2X4R or siRNA targeting P2X4R
Rats received injections (2 µL) of a lentivirus carrying P2X4R, lentivirus carrying siRNA targeting P2X4R, or a negative control (Shanghai GeneChem Co., Ltd., Shanghai, China) into the SNpc according to the following stereotactic coordinates: posterior of anterior fontanel, –5 mm; dorsoventral, 7.7 mm; mediolateral, –2.1 mm. Rats in the control and 6-OHDA groups in Experiment II were stereotactically injected with 2 µL of 0.9% physiological saline.
6-OHDA lesions
One week after lentivirus pretreatment, 8 µg of 6-OHDA (MilliporeSigma) dissolved in 2 µL of 0.02% ascorbic acid (MilliporeSigma) was injected into the SNpc stereotactically to establish PD (Bigham et al., 2021). Rats in the control, 5-BDBD, and P2X4R-NC groups were injected with the same volume (2 µL) of 0.02% ascorbic acid.
Behavioral test
PD model rats were injected with apomorphine (MilliporeSigma; 0.5 mg/kg, intraperitoneally) on day 14 after surgery and placed in stainless steel bowls. The number of rotation cycles in 30 minutes was recorded. The rate for PD rats should be > 7 rotations/min (Liu et al., 2018) (Results are shown in Additional Table 1).
Additional Table 1.
Results of the rotation of Parkinson’s disease model rats
| Group | Rotations (times/min) |
|---|---|
| 6-OHDA | 9.11±0.92 |
| 5-BDBD+6-OHDA | 9.16±0.99 |
| P2X4R-NC+6-OHDA | 9.21±1.06 |
| P2X4R-RNA+6-OHDA | 9.47±1.28 |
| P2X4R-siRNA+ 6-OHDA | 9.26±0.99 |
Data are expressed as mean ± SEM (n = 20 mice/group). Control group: After 1 week of pretreatment with 0.9% physiological saline, ascorbic acid was injected into the SNpc stereotactically; 5-BDBD + 6-OHDA group: after 1 week of pretreatment with 5-BDBD, 6-OHDA was injected into the SNpc stereotactically; P2X4R-NC + 6-OHDA group: after 1 week of pretreatment with the negative control lentivirus, 6-OHDA was injected into the SNpc stereotactically; P2X4R-RNA + 6-OHDA group: after 1 week of pretreatment with the lentivirus carrying P2X4R, 6-OHDA was injected into the SNpc stereotactically; P2X4R-siRNA + 6-OHDA group: after 1 week of pretreatment with the lentivirus carrying siRNA targeting P2X4R, 6-OHDA was injected into the SNpc stereotactically. 5-BDBD: 5-(3-Bromophenyl)-1,3-dihydro-2H-benzofuro[3,2-e]-1,4-diazepin-2-one; 6-OHDA: 6-hydroxydopamine; NC: negative control lentivirus; P2X4R: P2X4 receptor; siRNA: small-interfering RNA; SNpc: substantia nigra pars compacta.
Immunofluorescence staining
Four weeks after surgery, five rats in each group were deeply anesthetized intraperitoneally with ethyl carbamate (Sinopharm Chemical Reagent Co. Ltd., Shanghai, China) and perfused transcardially with 4% paraformaldehyde. Each rat’s brain was removed and post-fixed in 4% paraformaldehyde for 24 hours. Next, we dehydrated the brain tissue with a sucrose gradient at 4°C. We created continuous coronal sections (20 µm) through the SNpc using a freezing cryostat (Leica Biosystems, Nussloch, Germany). The sections were kept free-floating in phosphate-buffered saline (PBS). We incubated the sections with an anti-tyrosine hydroxylase (TH) antibody (rabbit, 1:2000, Cat # 58844S; Cell Signaling Technology, Danvers, MA, USA). The sections were shaken at 4°C overnight and then washed with 0.05% Tween-20 in PBS thrice. The sections were incubated with a secondary antibody (rabbit, 1:1000, Cat # 4413; Cell Signaling Technology), shaken at room temperature for 2 hours, and washed thrice with 0.05% Tween-20 in PBS. The sections were placed on glass slides, sealed with 70 g/L glycerophosphate buffer, and rinsed with distilled water. We analyzed every sixth section for a total of 20 sections. We counted the TH-positive neurons in the SNpc of these slices under a fluorescence microscope (Axio Imager, LSM 800; Carl Zeiss Meditec AG, Jena, Germany).
Western blot assay
Four weeks after surgery, eight rats in each group were deeply intraperitoneally anesthetized with ethyl carbamate, and the SNpc of the left brain was separated. Tissues were homogenized in 200 µL of lysis buffer containing protease and phosphatase inhibitors (Roche, Basel, Switzerland). The homogenates were centrifuged at 13,000 × g at 4°C for 5 minutes. A bicinchoninic acid colorimetric protein assay kit (Thermo Fisher Scientific, Waltham, MA, USA) was used to analyze protein concentrations in the supernatants. Protein samples were homogenized with 20% 5× loading buffer, boiled for 10 minutes, and stored at –80°C.
Total protein (20 µg) was separated on 10% sodium dodecyl sulfate-polyacrylamide gels and transferred electrophoretically to polyvinylidene difluoride membranes (Roche). The membranes were blocked in 5% non-fat dried milk at room temperature for 1 hour and incubated with primary antibodies against P2X4R (rabbit, 1:400, Cat# APR-002; Alomone Labs, Jerusalem, Israel), NLRP3 (rabbit, 1:1000, Cat# 13158; Cell Signaling Technology), caspase-1 (rabbit, 1:500, Cat# 3866; Cell Signaling Technology), IL-1β (rabbit, 1:2000, Cat# 12703; Cell Signaling Technology), IL-18 (rabbit, 1:1000, Cat# 54943; Cell Signaling Technology), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH; rabbit, 1:1000, Cat# 5174; Cell Signaling Technology) at 4°C overnight. The membranes were washed with 0.05% Tween-20 in PBS and incubated with horseradish peroxidase-conjugated IgG (rabbit, 1:2000, Cat# 7074; Cell Signaling Technology) at room temperature for 1 hour. Immunoreactive proteins were visualized using an enhanced luminescence reagent (Thermo Fisher Scientific). The net absorbance value for the target bands was analyzed using a gel image processing system (Liaoning Saias Technology Co. Ltd., Shenyang, China). Protein expression levels were calculated as the optical density ratio of target protein to GAPDH protein.
Quantitative reverse-transcription polymerase chain reaction
Four weeks after surgery, seven rats in each group were deeply intraperitoneally anesthetized with ethyl carbamate, and the SNpc of the left brain was separated. Tissues were homogenized and total RNA was extracted using TRIzol reagent (Takara Bio Inc., Shiga, Japan). Total RNA was reverse transcribed into first-strand cDNA using a reverse-transcription reagent kit (Takara Bio) in accordance with the manufacturer’s procedure. Quantitative reverse-transcription polymerase chain reaction was performed using the SYBR Green fluorescent dye (Takara Bio) methodology. The following primers (synthesized by the BGI Group, Beijing, China) specifically targeting rat genes were used. P2X4R forward, 5′-TTA CAA TGC TCA AAC GGA TCC C-3′; P2X4R reverse, 5′-GGA TAC CCA TGA TGC CTC CC-3′ (207 bp). NLRP3 forward, 5′-TTG GCT GCG GAT GGA ATT T-3′; NLRP3 reverse, 5′-TCT CGC AGT CCA CCT TCT-3′ (133 bp). Caspase-1 forward, 5′-CCA ATA ATG AAA ACA CCC ACT CGT-3′; Caspase-1 reverse, 5′-CAC AGT ATA CCC CAG ATC CTG CA-3′ (141 bp). IL-1β forward, 5′-TGA TGA CGA CCT GCT AGT GTG TG-3′; IL-1β reverse, 5′-TGT TGG CTT ATG TTC TGT CCA TTG-3′ (141 bp). IL-18 forward, 5′-TTC AGA AAC GTG TGC CAG GAC-3′; IL-18 reverse, 5′-ACG AGT TTG AAA GCA TCA TCT TCC-3′ (103 bp). Gapdh forward, 5′-TGG CCT TCC GTG TTC CTA C-3′; Gapdh reverse, 5′-GAG TTG CTG TTG AAG TCG CA-3′ (191 bp).
The quantitative reverse-transcription polymerase chain reaction mixture (20 µL) contained RNase-free water, 7.6 µL; SYBR Advantage qPCR Premix (Takara Bio), 10 µL; forward primer, 0.4 µL; reverse primer, 0.4 µL; cDNA, 1.6 µL. Thermal cycling conditions were pre-denaturation at 95°C for 10 minutes, followed by 45 cycles of denaturation at 95°C for 15 seconds, and annealing at 60°C for 1 minute. Target gene expression was expressed relative to Gapdh expression.
Statistical analysis
Differences between group means were evaluated by one-way analysis of variance followed by the Student-Newman-Keuls test using GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA). P < 0.05 was considered statistically significant.
Results
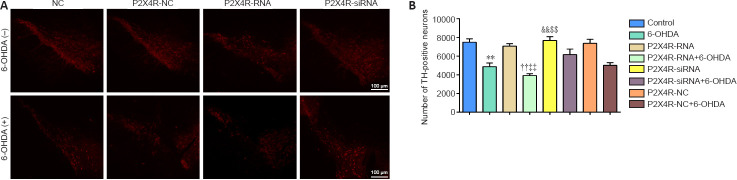
Effect of 5-BDBD on the number of TH-positive neurons in the SNpc
There were significantly fewer TH-positive neurons in the 6-OHDA and 5-BDBD + 6-OHDA groups compared with the 5-BDBD and the control groups (all P < 0.01). Compared with the 6-OHDA group, the quantity of TH-positive neurons increased significantly in the 5-BDBD + 6-OHDA group (P < 0.01). There were no significant differences in the number of TH-positive neurons in the 5-BDBD group compared with the control group (P > 0.05; Figure 2).
Figure 2.
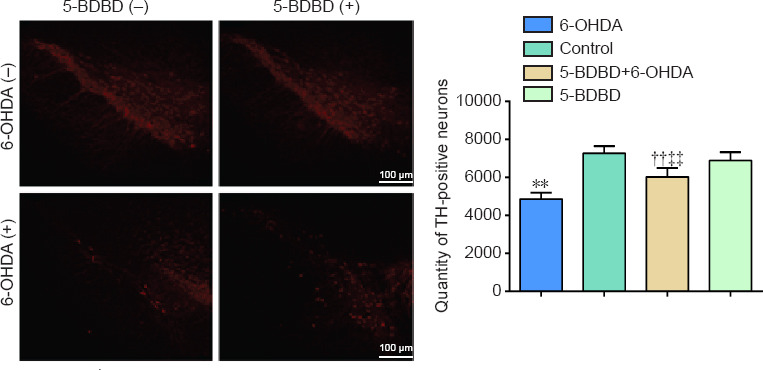
Effect of 5-BDBD on the number of tyrosine hydroxylase-positive neurons in the substantia nigra pars compacta.
(A) Immunofluorescence staining of TH-positive neurons (red, Alexa Fluor® 555). (B) Number of TH-positive neurons. Data are expressed as mean ± SEM (n = 5 mice/group). **P < 0.01, vs. control group; ††P < 0.01, vs. 5-BDBD group; ‡‡P < 0.01, vs. 6-OHDA group (one-way analysis of variance followed by the Student-Newman-Keuls test). Control group: After 1 week of pretreatment with 0.9% physiological saline, ascorbic acid was injected into the SNpc stereotactically; 6-OHDA group: after 1 week of pretreatment with 0.9% physiological saline, 6-OHDA was injected into the SNpc stereotactically; 5-BDBD group: after 1 week of pretreatment with 5-BDBD, ascorbic acid was injected into the SNpc stereotactically; 5-BDBD + 6-OHDA group: after 1 week of pretreatment with 5-BDBD, 6-OHDA was injected into the SNpc stereotactically. 5-BDBD: 5-(3-Bromophenyl)-1,3-dihydro-2H-benzofuro3,2-e]-1,4-diazepin-2-one; 6-OHDA: 6-hydroxydopamine; SNpc: substantia nigra pars compacta; TH: tyrosine hydroxylase.
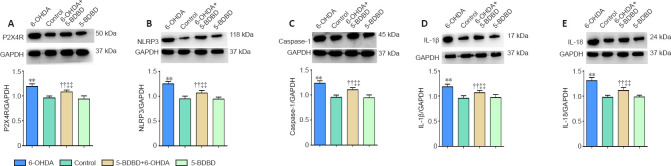
Effect of 5-BDBD on inflammatory- and apoptosis-related mRNA and protein expression in the SNpc
We investigated the effect of 5-BDBD on the mRNA and protein expression levels of P2X4, NLRP3, caspase-1, IL-1β, and IL-18 in the SNpc. The mRNA and protein levels of P2X4, NLRP3, caspase-1, IL-1β, and IL-18 were significantly higher in the 6-OHDA and 5-BDBD + 6-OHDA groups than the control and 5-BDBD groups (all P < 0.01). The mRNA and protein levels of P2X4, NLRP3, caspase-1, IL-1β, and IL-18 were significantly lower in the 5-BDBD + 6-OHDA group than the 6-OHDA group (P < 0.01). There were no significant statistical differences in P2X4 mRNA and protein expression between the control and 5-BDBD group (P > 0.05; Figure 3 and Table 1).
Figure 3.
Effect of 5-(3-bromophenyl)-1,3-dihydro-2H-benzofuro3,2-e]-1,4-diazepin-2-one (5-BDBD) on the protein expression of P2X4R (A), NLRP3 (B), caspase-1 (C), IL-1β (D), and IL-18 (E) in the substantia nigra pars compacta.
Data are expressed as mean ± SEM (n = 8 mice/group). **P < 0.01, vs. control group; ††P < 0.01, vs. 5-BDBD group; ‡‡P < 0.01, vs. 6-OHDA group (one-way analysis of variance followed by the Student-Newman-Keuls test). Control group: After 1 week of pretreatment with 0.9% physiological saline, ascorbic acid was injected into the SNpc stereotactically; 6-OHDA group: after 1 week of pretreatment with 0.9% physiological saline, 6-OHDA was injected into the SNpc stereotactically; 5-BDBD group: after 1 week of pretreatment with 5-BDBD, ascorbic acid was injected into the SNpc stereotactically; 5-BDBD + 6-OHDA group: after 1 week of pretreatment with 5-BDBD, 6-OHDA was injected into the SNpc stereotactically. 5-BDBD: 5-(3-Bromophenyl)-1,3-dihydro-2H-benzofuro3,2-e]-1,4-diazepin-2-one; 6-OHDA: 6-hydroxydopamine; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; IL: interleukin; NLRP3: Nod-like receptor family protein 3; P2X4R: P2X4 receptor; SNpc: substantia nigra pars compacta.
Table 1.
Effect of 5-BDBD on the mRNA levels of P2X4R, NLRP3, caspase-1, IL-1β, and IL-18 in the substantia nigra pars compacta
| Gene | Control | 6-OHDA | 5-BDBD | 5-BDBD+6-OHDA |
|---|---|---|---|---|
| P2X4R | 1.031±0.104 | 3.217±0.09** | 1.070±0.044 | 2.741±0.094††‡‡ |
| NLRP3 | 1.004±0.037 | 3.933±0.111** | 1.033±0.034 | 3.039±0.057††‡‡ |
| Caspase-1 | 1.011±0.048 | 3.961±0.132** | 1.056±0.053 | 3.007±0.096††‡‡ |
| IL-1β | 1.033±0.058 | 3.876±0.099** | 1.047±0.041 | 3.068±0.071††‡‡ |
| IL-18 | 1.007±0.047 | 3.437±0.123** | 1.029±0.045 | 2.625±0.117††‡‡ |
Control group: after 1 week of pretreatment with 0.9% physiological saline, ascorbic acid was injected into the SNpc stereotactically; 6-OHDA group: after 1 week of pretreatment with 0.9% physiological saline, 6-OHDA was injected into the SNpc stereotactically; 5-BDBD group: after 1 week of pretreatment with 5-BDBD, ascorbic acid was injected into the SNpc stereotactically; 5-BDBD + 6-OHDA group: after 1 week of pretreatment with 5-BDBD, 6-OHDA was injected into the SNpc stereotactically. Data are expressed as mean ± SEM (n = 7 mice/group). **P < 0.01, vs. control group; ††P < 0.01, vs. 5-BDBD group; ‡‡P < 0.01, vs. 6-OHDA group (one-way analysis of variance followed by the Student–Newman–Keuls test). 5-BDBD: 5-(3-Bromophenyl)-1,3-dihydro-2H-benzofuro[3,2-e]-1,4-diazepin-2-one; 6-OHDA: 6-hydroxydopamine; IL: interleukin; NLRP3: Nod-like receptor family protein 3; P2X4R: P2X4 receptor; SNpc: substantia nigra pars compacta.
Effect of P2X4R overexpression and silencing on the number of TH-positive neurons in the SNpc
The numbers of TH-positive neurons were significantly lower in the 6-OHDA, P2X4R-NC + 6-OHDA, P2X4R-RNA + 6-OHDA, and P2X4R-siRNA + 6-OHDA groups compared with the control, P2X4R-NC, P2X4R-RNA, and P2X4R siRNA groups (all P < 0.01). Compared with the P2X4R-NC + 6-OHDA group, there were significantly fewer TH-positive neurons in the P2X4R-RNA + 6-OHDA group (P < 0.01) and significantly more TH-positive neurons in the P2X4R-siRNA + 6-OHDA group (P < 0.01). There were no significant differences between the control and P2X4R-NC groups (P > 0.05; Figures 4 and 5).
Figure 4.
Effect of P2X4R overexpression and silencing on the number of tyrosine hydroxylase-positive neurons in the substantia nigra pars compacta.
(A) Immunofluorescence staining of TH-positive neurons (red, Alexa Fluor® 555). (B) Number of TH-positive neurons. Data are expressed as mean ± SEM (n = 5 mice/group). **P < 0.01, vs. control group; ††P < 0.01, vs. P2X4R-RNA group; ‡‡P < 0.01, vs. P2X4R-NC + 6-OHDA group; &&P < 0.01, vs. P2X4R-siRNA group; $$P < 0.01, vs. P2X4R-NC + 6-OHDA group (one-way analysis of variance followed by the Student-Newman-Keuls test). Control group: After 1 week of pretreatment with 0.9% physiological saline, ascorbic acid was injected into the SNpc stereotactically; 6-OHDA group: after 1 week of pretreatment with 0.9% physiological saline, 6-OHDA was injected into the SNpc stereotactically to establish PD; P2X4R-NC group: after 1 week of pretreatment with a negative control lentivirus (NC), ascorbic acid was injected into the SNpc stereotactically; P2X4R-NC + 6-OHDA group: after 1 week of pretreatment with the negative control lentivirus, 6-OHDA was injected into the SNpc stereotactically; P2X4R-RNA group: after 1 week of pretreatment with a lentivirus carrying P2X4R, ascorbic acid was injected into the SNpc stereotactically; P2X4R-RNA + 6-OHDA group: after 1 week of pretreatment with the lentivirus carrying P2X4R, 6-OHDA was injected into the SNpc stereotactically; P2X4R-siRNA group: after 1 week of pretreatment with a lentivirus carrying siRNA targeting P2X4R, ascorbic acid was injected into the SNpc stereotactically; P2X4R-siRNA + 6-OHDA group: after 1 week of pretreatment with the lentivirus carrying siRNA targeting P2X4R, 6-OHDA was injected into the SNpc stereotactically. 6-OHDA: 6-Hydroxydopamine; NC: negative control lentivirus; P2X4R: P2X4 receptor; PD: Parkinson’s disease; siRNA: small-interfering RNA; SNpc: substantia nigra pars compacta; TH: tyrosine hydroxylase.
Figure 5.
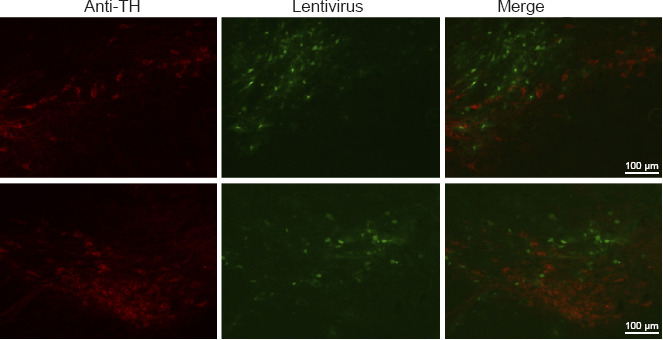
Distribution of the lentivirus in tyrosine hydroxylase-positive neurons in the substantia nigra pars compacta in P2X4R-RNA and P2X4R-siRNA groups.
P2X4R-RNA group: After 1 week of pretreatment with a lentivirus carrying P2X4R, ascorbic acid was injected into the SNpc stereotactically; P2X4R-siRNA group: after 1 week of pretreatment with a lentivirus carrying siRNA targeting P2X4R, ascorbic acid was injected into the SNpc stereotactically. Anti-TH: TH-positive neurons; lentivirus: lentivirus in the SNpc; merge: lentivirus in TH-positive neurons (red, Alexa Fluor® 555). P2X4R: P2X4 receptor; siRNA: small-interfering RNA; SNpc: substantia nigra pars compacta; TH: tyrosine hydroxylase.
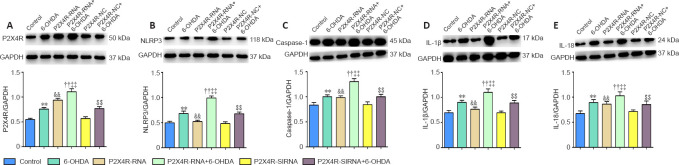
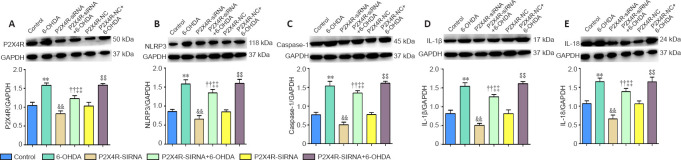
Effect of P2X4R overexpression and silencing on inflammatory- and apoptosis-related gene and protein expression levels in the SNpc
P2X4R, NLRP3, caspase-1, IL-1β, and IL-18 mRNA levels were significantly higher in the 6-OHDA, P2X4R-NC + 6-OHDA, P2X4R-RNA + 6-OHDA, and P2X4R-siRNA + 6-OHDA groups compared with the control, P2X4R-NC, P2X4R-RNA, and P2X4R siRNA groups (P < 0.01). Compared with the P2X4R-NC + 6-OHDA group, P2X4R, NLRP3, caspase-1, IL-1β, and IL-18 mRNA levels were significantly higher in the P2X4R-RNA + 6-OHDA group (P < 0.01) and significantly lower in the P2X4R-siRNA + 6-OHDA group (P < 0.01). There were no significant differences in P2X4R, NLRP3, caspase-1, IL-1β, and IL-18 mRNA expression between the control and P2X4R-NC groups (P > 0.05). Compared with the P2X4R-NC group, the mRNA levels of P2X4R, NLRP3, caspase-1, IL-1β, and IL-18 were significantly higher in the P2X4R-RNA group (P < 0.01) and significantly lower in the P2X4R-siRNA group (P < 0.01; Tables 2 and 3).
Table 2.
Effect of the lentivirus carrying P2X4R on the mRNA levels of P2X4R, NLRP3, Caspase-1, IL-1β, and IL-18 in the substantia nigra pars compacta
| Gene | Control | 6-OHDA | P2X4R-RNA | P2X4R-RNA+6-OHDA | P2X4R-NC | P2X4R-NC+6-OHDA |
|---|---|---|---|---|---|---|
| P2X4R | 0.471±0.024 | 1.268±0.050** | 0.845±0.021&& | 2.101±0.044††‡‡ | 0.436±0.025 | 1.280±0.025$$ |
| NLRP3 | 0.522±0.024 | 1.767±0.067** | 0.876±0.019&& | 2.410±0.041††‡‡ | 0.470±0.022 | 1.624±0.050$$ |
| Caspase-1 | 0.456±0.032 | 1.475±0.044** | 0.811±0.019&& | 2.409±0.037††‡‡ | 0.454±0.021 | 1.464±0.028$$ |
| IL-1β | 0.485±0.033 | 1.485±0.049** | 0.959±0.022&& | 2.502±0.059††‡‡ | 0.472±0.022 | 1.653±0.052$$ |
| IL-18 | 0.477±0.033 | 1.706±0.075** | 0.849±0.018&& | 2.486±0.042††‡‡ | 0.527±0.051 | 1.536±0.043$$ |
Control group: After 1 week of pretreatment with 0.9% physiological saline, ascorbic acid was injected into the SNpc stereotactically; 6-OHDA group: after 1 week of pretreatment with 0.9% physiological saline, 6-OHDA was injected into the SNpc stereotactically to establish PD; P2X4R-NC group: after 1 week of pretreatment with a negative control lentivirus (NC), ascorbic acid was injected into the SNpc stereotactically; P2X4R-NC + 6-OHDA group: after 1 week of pretreatment with the negative control lentivirus, 6-OHDA was injected into the SNpc stereotactically; P2X4R-RNA group: after 1 week of pretreatment with a lentivirus carrying P2X4R, ascorbic acid was injected into the SNpc stereotactically; P2X4R-RNA + 6-OHDA group: after 1 week of pretreatment with the lentivirus carrying P2X4R, 6-OHDA was injected into the SNpc stereotactically. Data are expressed as mean ± SEM (n = 7 mice/group). **P < 0.01, vs. control group; ††P < 0.01, vs. P2X4R-RNA group; ‡‡P < 0.01, vs. P2X4R-NC + 6-OHDA group; &&P < 0.01, vs. P2X4R-NC group; $$P < 0.01, vs. P2X4R-NC group (one-way analysis of variance followed by the Student-Newman-Keuls test). 6-OHDA: 6-Hydroxydopamine; IL: interleukin; NC: negative control lentivirus; NLRP3: Nod-like receptor family protein 3; P2X4R: P2X4 receptor; PD: Parkinson’s disease; SNpc: substantia nigra pars compacta.
Table 3.
Effect of the lentivirus carrying small interfering RNA for P2X4R on the mRNA levels of P2X4R, NLRP3, Caspase-1, IL-1β, and IL-18 in the substantia nigra pars compacta
| Gene | Control | 6-OHDA | P2X4R-siRNA | P2X4R-siRNA+6-OHDA | P2X4R-NC | P2X4R-NC+6-OHDA |
|---|---|---|---|---|---|---|
| P2X4R | 0.694±0.267 | 1.798±0.056** | 0.494±0.017&& | 1.284±0.089††‡‡ | 0.655±0.030 | 1.848±0.036$$ |
| NLRP3 | 0.730±0.033 | 1.883±0.034** | 0.504±0.013&& | 1.357±0.029††‡‡ | 0.729±0.038 | 1.898±0.050$$ |
| Caspase-1 | 0.727±0.026 | 1.852±0.044** | 0.511±0.016&& | 1.443±0.041††‡‡ | 0.699±0.025 | 1.872±0.026$$ |
| IL-1β | 0.737±0.036 | 1.842±0.046** | 0.505±0.013&& | 1.344±0.034††‡‡ | 0.696±0.022 | 1.857±0.027$$ |
| IL-18 | 0.676±0.026 | 1.845±0.035** | 0.486±0.013&& | 1.349±0.027††‡‡ | 0.640±0.026 | 1.861±0.047$$ |
Control group: After 1 week of pretreatment with 0.9% physiological saline, ascorbic acid was injected into the SNpc stereotactically; 6-OHDA group: after 1 week of pretreatment with 0.9% physiological saline, 6-OHDA was injected into the SNpc stereotactically to establish PD; P2X4R-NC group: after 1 week of pretreatment with a negative control lentivirus (NC), ascorbic acid was injected into the SNpc stereotactically; P2X4R-NC + 6-OHDA group: after 1 week of pretreatment with the negative control lentivirus, 6-OHDA was injected into the SNpc stereotactically; P2X4R-siRNA group: after 1 week of pretreatment with a lentivirus carrying siRNA targeting P2X4R, ascorbic acid was injected into the SNpc stereotactically; P2X4R-siRNA + 6-OHDA group: after 1 week of pretreatment with the lentivirus carrying siRNA targeting P2X4R, 6-OHDA was injected into the SNpc stereotactically. Data are expressed as mean ± SEM (n = 7 mice/group). **P < 0.01, vs. control group; ††P < 0.01, vs. P2X4R-RNA group; ‡‡P < 0.01, vs. P2X4R-NC + 6-OHDA group; &&P < 0.01, vs. P2X4R-NC group; $$P < 0.01, vs. P2X4R-NC group (one-way analysis of variance followed by the Student-Newman-Keuls test). 6-OHDA: 6-Hydroxydopamine; IL: interleukin; NC: negative control lentivirus; NLRP3: Nod-like receptor family protein 3; P2X4R: P2X4 receptor; PD: Parkinson’s disease; SNpc: substantia nigra pars compacta.
NLRP3, caspase-1, IL-1β, and IL-18 protein levels were significantly higher in the 6-OHDA, P2X4R-NC + 6-OHDA, P2X4R-RNA + 6-OHDA, and P2X4R-siRNA + 6-OHDA groups compared with the control, P2X4R-NC, P2X4R-RNA, and P2X4R siRNA groups (P < 0.01). NLRP3, caspase-1, IL-1β, and IL-18 protein levels were significantly higher in the P2X4R-RNA + 6-OHDA group (P < 0.01) and significantly lower in the P2X4R-siRNA + 6-OHDA group (P < 0.01). There were no significant differences in the expression of NLRP3, caspase-1, IL-1β, and IL-18 proteins between the control and P2X4R-NC groups (P > 0.05; Figures 6 and 7).
Figure 6.
Effect of a lentivirus carrying P2X4R on the protein expression levels of P2X4R (A), NLRP3 (B), caspase-1 (C), IL-1β (D), and IL-18 (E) in the substantia nigra pars compacta.
Data are expressed as mean ± SEM (n = 8 mice/group). **P < 0.01, vs. control group; ††P < 0.01, vs. P2X4R-RNA group; ‡‡P < 0.01, vs. P2X4R-NC + 6-OHDA group; &&P < 0.01, vs. P2X4R-NC group; $$P < 0.01, vs. P2X4R-NC group (one-way analysis of variance followed by the Student-Newman-Keuls test). Control group: After 1 week of pretreatment with 0.9% physiological saline, ascorbic acid was injected into the SNpc stereotactically; 6-OHDA group: after 1 week of pretreatment with 0.9% physiological saline, 6-OHDA was injected into the SNpc stereotactically to establish PD; P2X4R-NC group: after 1 week of pretreatment with a negative control lentivirus (NC), ascorbic acid was injected into the SNpc stereotactically; P2X4R-NC + 6-OHDA group: after 1 week of pretreatment with the negative control lentivirus, 6-OHDA was injected into the SNpc stereotactically; P2X4R-RNA group: after 1 week of pretreatment with a lentivirus carrying P2X4R, ascorbic acid was injected into the SNpc stereotactically; P2X4R-RNA + 6-OHDA group: after 1 week of pretreatment with the lentivirus carrying P2X4R, 6-OHDA was injected into the SNpc stereotactically. 6-OHDA: 6-Hydroxydopamine; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; IL: interleukin; NC: negative control lentivirus; NLRP3: Nod-like receptor family protein 3; P2X4R: P2X4 receptor; PD: Parkinson’s disease; SNpc: substantia nigra pars compacta.
Figure 7.
Effect of the lentivirus carrying siRNA for P2X4R on the protein expression levels of P2X4R (A), NLRP3 (B), caspase-1 (C), IL-1β (D), and IL-18 (E) in the substantia nigra pars compacta.
Data are expressed as mean ± SEM (n = 8 mice/group). **P < 0.01, vs. control group; ††P < 0.01, vs. P2X4R-RNA group; ‡‡P < 0.01, vs. P2X4R-NC + 6-OHDA group; &&P < 0.01, vs. P2X4R-NC group; $$P < 0.01, vs. P2X4R-NC group (one-way analysis of variance followed by the Student-Newman-Keuls test). Control group: After 1 week of pretreatment with 0.9% physiological saline, ascorbic acid was injected into the SNpc stereotactically; 6-OHDA group: after 1 week of pretreatment with 0.9% physiological saline, 6-OHDA was injected into the SNpc stereotactically to establish PD; P2X4R-NC group: after 1 week of pretreatment with a negative control lentivirus (NC), ascorbic acid was injected into the SNpc stereotactically; P2X4R-NC + 6-OHDA group: after 1 week of pretreatment with the negative control lentivirus, 6-OHDA was injected into the SNpc stereotactically; P2X4R-siRNA group: after 1 week of pretreatment with a lentivirus carrying siRNA targeting P2X4R, ascorbic acid was injected into the SNpc stereotactically; P2X4R-siRNA + 6-OHDA group: after 1 week of pretreatment with the lentivirus carrying siRNA targeting P2X4R, 6-OHDA was injected into the SNpc stereotactically. 6-OHDA: 6-Hydroxydopamine; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; IL: interleukin; NC: negative control lentivirus; NLRP3: Nod-like receptor family protein 3; P2X4R: P2X4 receptor; PD: Parkinson’s disease; siRNA: small-interfering RNA; SNpc: substantia nigra pars compacta.
Discussion
Neuroinflammation is critical for the onset of PD (Pradhan and Andreasson, 2013). Microglial cells are central mediators of inflammatory effects in the brain and are regarded as stationary macrophages in the CNS; they derive from the yolk sac and are associated with myeloid immune cells (González et al., 2014). Extracellular ATP, an important signaling molecule, is mainly released by injured neurons or glial cells. ATP initiates a purinergic signaling pathway by binding to purinergic receptors and participates in the physiological or pathological processes of the nervous system (Bours et al., 2011). Purinergic signaling plays an important role in neuroimmune-related inflammation (Di Virgilio et al., 2009). The purinergic receptor family can be divided into two classes: P1 and P2 nucleotide receptors. P2 is further divided into P2X and P2Y. P2X receptors are mainly involved in pathological processes such as CNS injury, immune inflammation, metabolic disorders, and neurodegeneration (Köles et al., 2011).
P2X4R was the first P2XR identified in the CNS and is expressed frequently on neurons and glial cells; it plays an important part in the modulation of synaptic transmission and exchange between CNS neurons and neighboring glial cells (Burnstock, 2015). The molecular mechanism of P2X4R in microglial cells has had increasing attention over the last ten years, and it may regulate microglial cell activation and migration. Activated microglia play an important role in the development of neurodegenerative diseases such as PD. Therefore, P2X4R may contribute to the molecular mechanisms of these diseases by exacerbating inflammation in the CNS (Hirsch et al., 2012). We found that the mRNA and protein levels of P2X4R expression were upregulated in the rat model of PD, which we induced by 6-OHDA stereotactic injection. In line with our findings, P2X4R was also upregulated in the microglial cells of the CNS in an animal model of multiple sclerosis, and genes encoding P2X4R were upregulated in PC12 cells treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine for PD induction (Hracskó et al., 2011).
The NLRP3 inflammasome is an innate immune system complex activated by a variety of danger signals and is mainly expressed by myeloid cells. It consists of three main effectors: NLRP3, procaspase-1, and the apoptosis speck-like protein (Zahid et al., 2019). Activation of the NLRP3 inflammasome pathway is increasingly considered to be the basis of a variety of many inflammatory diseases. The mechanism underlying its activation is not fully understood, but the purinergic receptors P2X4 and P2X7 and damage-associated molecular patterns, such as ATP release, are involved (Chen et al., 2013). The NLRP3 inflammasome mediates the aggregation of caspase-1, which converts IL-1β into a mature form with biological activity (Juliana et al., 2012). Furthermore, IL-18 is significantly upregulated after NLRP3 inflammasome activation (Juliana et al., 2012). Therefore, we hypothesized that upregulation of the downstream signaling components of the NLRP3 inflammasome contributes to the pathogenesis of PD. Our data demonstrate that NLRP3 inflammasome components, including IL-1β and IL-18, are activated in PD-induced rats. The levels of P2X4R, NLRP3, caspase-1, IL-1β, and IL-18 were significantly increased in PD-induced rats overexpressing P2X4R and were significantly decreased in PD-induced rats treated with the selective P2X4R antagonist 5-BDBD and in P2X4R-knockdown rats.
Our study has some limitations. First, inflammasome activation in rat models of PD is not exactly equivalent to human brains, and more experiments are needed to investigate inflammasome activation in humans. Second, our current understanding of the potential beneficial effects of antagonizing P2X4R signaling is limited. We will continue to study the potential beneficial effects of antagonizing P2X4R signaling with the aim of attenuating neuropathological and behavioral impairment of PD-induced rats.
Based on our results, we conclude that ATP-P2X4R signaling mediates NLRP3 inflammasome activation, resulting in CNS inflammation in a 6-OHDA-induced rat model of PD. In a wider perspective, we found that the ATP-P2X4R axis mediates NLRP3 inflammasome activation to regulate glial cell activation, dopaminergic neurodegeneration, and dopamine levels. These findings reveal novel roles for the ATP-P2X4R axis in the pathogenesis of PD and provide new therapeutic targets for the disease.
Acknowledgments: We are grateful to the Central Laboratory of the Affiliated Hospital of Qingdao University for providing experimental support and technical guidance.
Additional files:
Additional file 1: Open peer review reports 1 (92.6KB, pdf) and 2 (87.8KB, pdf) .
Additional Table 1: Results of the rotation of Parkinson’s disease model rats.
Footnotes
P-Reviewers: Schweitzer J, Haining RL; C-Editor: Zhao M; S-Editors: Yu J, Li CH; L-Editors: Crow E, Yu J, Song LP; T-Editor: Jia Y
Conflicts of interest: The authors declare no conflicts of interest.
Financial support: This study was supported by the National Natural Science Foundation of China, No. 81971192 (to AMX). The funding source had no role in study conception and design, data analysis or interpretation, paper writing or deciding to submit this paper for publication.
Institutional review board statement: The study was approved by the Animal Ethics Committee of Qingdao University, China (approval No. QYFYWZLL 26119) on March 5, 2015.
Copyright license agreement: The Copyright License Agreement has been signed by all authors before publication.
Data sharing statement: Datasets analyzed during the current study are available from the corresponding author on reasonable request.
Plagiarism check: Checked twice by iThenticate.
Peer review: Externally peer reviewed.
Open peer reviewers: Jeffrey Schweitzer, Massachusetts General Hospital, USA; Robert L. Haining, Georgia Gwinnett College, USA.
Funding: This study was supported by the National Natural Science Foundation of China, No. 81971192 (to AMX).
References
- 1.Antonioli L, Blandizzi C, Fornai M, Pacher P, Lee HT, Haskó G. P2X4 receptors, immunity, and sepsis. Curr Opin Pharmacol. 2019;47:65–74. doi: 10.1016/j.coph.2019.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bours MJ, Dagnelie PC, Giuliani AL, Wesselius A, Di Virgilio F. P2 receptors and extracellular ATP: a novel homeostatic pathway in inflammation. Front Biosci (Schol Ed) 2011;3:1443–1456. doi: 10.2741/235. [DOI] [PubMed] [Google Scholar]
- 3.Burnstock G. Physiopathological roles of P2X receptors in the central nervous system. Curr Med Chem. 2015;22:819–844. doi: 10.2174/0929867321666140706130415. [DOI] [PubMed] [Google Scholar]
- 4.Burnstock G, Krügel U, Abbracchio MP, Illes P. Purinergic signalling: from normal behaviour to pathological brain function. Prog Neurobiol. 2011;95:229–274. doi: 10.1016/j.pneurobio.2011.08.006. [DOI] [PubMed] [Google Scholar]
- 5.Chen F, Wei G, Xu J, Ma X, Wang Q. Naringin ameliorates the high glucose-induced rat mesangial cell inflammatory reaction by modulating the NLRP3 inflammasome. BMC Complement Altern Med. 2018;18:192. doi: 10.1186/s12906-018-2257-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen K, Zhang J, Zhang W, Zhang J, Yang J, Li K, He Y. ATP-P2X4 signaling mediates NLRP3 inflammasome activation: a novel pathway of diabetic nephropathy. Int J Biochem Cell Biol. 2013;45:932–943. doi: 10.1016/j.biocel.2013.02.009. [DOI] [PubMed] [Google Scholar]
- 7.Di Virgilio F, Ceruti S, Bramanti P, Abbracchio MP. Purinergic signalling in inflammation of the central nervous system. Trends Neurosci. 2009;32:79–87. doi: 10.1016/j.tins.2008.11.003. [DOI] [PubMed] [Google Scholar]
- 8.Duveau A, Bertin E, Boué-Grabot E. Implication of neuronal versus microglial P2X4 receptors in central nervous system disorders. Neurosci Bull. 2020;36:1327–1343. doi: 10.1007/s12264-020-00570-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Forloni G, La Vitola P, Cerovic M, Balducci C. Inflammation and Parkinson’s disease pathogenesis: Mechanisms and therapeutic insight. Prog Mol Biol Transl Sci. 2021;177:175–202. doi: 10.1016/bs.pmbts.2020.11.001. [DOI] [PubMed] [Google Scholar]
- 10.González H, Elgueta D, Montoya A, Pacheco R. Neuroimmune regulation of microglial activity involved in neuroinflammation and neurodegenerative diseases. J Neuroimmunol. 2014;274:1–13. doi: 10.1016/j.jneuroim.2014.07.012. [DOI] [PubMed] [Google Scholar]
- 11.Hirsch EC, Vyas S, Hunot S. Neuroinflammation in Parkinson’s disease. Parkinsonism Relat Disord. 2012;18(Suppl 1):S210–212. doi: 10.1016/S1353-8020(11)70065-7. [DOI] [PubMed] [Google Scholar]
- 12.Holbrook JA, Jarosz-Griffiths HH, Caseley E, Lara-Reyna S, Poulter JA, Williams-Gray CH, Peckham D, McDermott MF. Neurodegenerative disease and the NLRP3 inflammasome. Front Pharmacol. 2021;12:643254. doi: 10.3389/fphar.2021.643254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hracskó Z, Baranyi M, Csölle C, Gölöncsér F, Madarász E, Kittel A, Sperlágh B. Lack of neuroprotection in the absence of P2X7 receptors in toxin-induced animal models of Parkinson’s disease. Mol Neurodegener. 2011;6:28. doi: 10.1186/1750-1326-6-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jiang J, Shi Y, Cao J, Lu Y, Sun G, Yang J. Role of ASM/Cer/TXNIP signaling module in the NLRP3 inflammasome activation. Lipids Health Dis. 2021;20:19. doi: 10.1186/s12944-021-01446-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Juliana C, Fernandes-Alnemri T, Kang S, Farias A, Qin F, Alnemri ES. Non-transcriptional priming and deubiquitination regulate NLRP3 inflammasome activation. J Biol Chem. 2012;287:36617–36622. doi: 10.1074/jbc.M112.407130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Köles L, Leichsenring A, Rubini P, Illes P. P2 receptor signaling in neurons and glial cells of the central nervous system. Adv Pharmacol. 2011;61:441–493. doi: 10.1016/B978-0-12-385526-8.00014-X. [DOI] [PubMed] [Google Scholar]
- 17.Lee SYH, Yates NJ, Tye SJ. Inflammatory mechanisms in Parkinson’s disease: from pathogenesis to targeted therapies. Neuroscientist. 2021 doi: 10.1177/1073858421992265. doi: 10.1177/1073858421992265. [DOI] [PubMed] [Google Scholar]
- 18.Liu H, Han X, Li Y, Zou H, Xie A. Association of P2X7 receptor gene polymorphisms with sporadic Parkinson’s disease in a Han Chinese population. Neurosci Lett. 2013;546:42–45. doi: 10.1016/j.neulet.2013.04.049. [DOI] [PubMed] [Google Scholar]
- 19.Liu H, Jia L, Chen X, Shi L, Xie J. The Kv7/KCNQ channel blocker XE991 protects nigral dopaminergic neurons in the 6-hydroxydopamine rat model of Parkinson’s disease. Brain Res Bull. 2018;137:132–139. doi: 10.1016/j.brainresbull.2017.11.011. [DOI] [PubMed] [Google Scholar]
- 20.Malhotra S, Costa C, Eixarch H, Keller CW, Amman L, Martínez-Banaclocha H, Midaglia L, Sarró E, Machín-Díaz I, Villar LM, Triviño JC, Oliver-Martos B, Parladé LN, Calvo-Barreiro L, Matesanz F, Vandenbroeck K, Urcelay E, Martínez-Ginés ML, Tejeda-Velarde A, Fissolo N, et al. NLRP3 inflammasome as prognostic factor and therapeutic target in primary progressive multiple sclerosis patients. Brain. 2020;143:1414–1430. doi: 10.1093/brain/awaa084. [DOI] [PubMed] [Google Scholar]
- 21.Mao Z, Liu C, Ji S, Yang Q, Ye H, Han H, Xue Z. The NLRP3 inflammasome is involved in the pathogenesis of Parkinson’s disease in rats. Neurochem Res. 2017;42:1104–1115. doi: 10.1007/s11064-017-2185-0. [DOI] [PubMed] [Google Scholar]
- 22.Martin-Bastida A, Tilley BS, Bansal S, Gentleman SM, Dexter DT, Ward RJ. Iron and inflammation: in vivo and post-mortem studies in Parkinson’s disease. J Neural Transm (Vienna) 2021;128:15–25. doi: 10.1007/s00702-020-02271-2. [DOI] [PubMed] [Google Scholar]
- 23.Mehrabadi S, Sadr SS. Assessment of probiotics mixture on memory function, inflammation markers, and oxidative stress in an Alzheimer’s disease model of rats. Iran Biomed J. 2020;24:220–228. doi: 10.29252/ibj.24.4.220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Moon HE, Paek SH. Mitochondrial dysfunction in Parkinson’s disease. Exp Neurobiol. 2015;24:103–116. doi: 10.5607/en.2015.24.2.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Piccini A, Carta S, Tassi S, Lasiglié D, Fossati G, Rubartelli A. ATP is released by monocytes stimulated with pathogen-sensing receptor ligands and induces IL-1beta and IL-18 secretion in an autocrine way. Proc Natl Acad Sci U S A. 2008;105:8067–8072. doi: 10.1073/pnas.0709684105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pradhan S, Andreasson K. Commentary: Progressive inflammation as a contributing factor to early development of Parkinson’s disease. Exp Neurol. 2013;241:148–155. doi: 10.1016/j.expneurol.2012.12.008. [DOI] [PubMed] [Google Scholar]
- 27.Rivers-Auty J, Tapia VS, White CS, Daniels MJD, Drinkall S, Kennedy PT, Spence HG, Yu S, Green JP, Hoyle C, Cook J, Bradley A, Mather AE, Peters R, Tzeng TC, Gordon MJ, Beattie JH, Brough D, Lawrence CB. Zinc status alters Alzheimer’s disease progression through NLRP3-dependent inflammation. J Neurosci. 2021;41:3025–3038. doi: 10.1523/JNEUROSCI.1980-20.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tan MS, Yu JT, Jiang T, Zhu XC, Wang HF, Zhang W, Wang YL, Jiang W, Tan L. NLRP3 polymorphisms are associated with late-onset Alzheimer’s disease in Han Chinese. J Neuroimmunol. 2013;265:91–95. doi: 10.1016/j.jneuroim.2013.10.002. [DOI] [PubMed] [Google Scholar]
- 29.Trang M, Schmalzing G, Müller CE, Markwardt F. Dissection of P2X4 and P2X7 receptor current components in BV-2 microglia. Int J Mol Sci. 2020;21:8489. doi: 10.3390/ijms21228489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wani K, AlHarthi H, Alghamdi A, Sabico S, Al-Daghri NM. Role of NLRP3 inflammasome activation in obesity-mediated metabolic disorders. Int J Environ Res Public Health. 2021;18:511. doi: 10.3390/ijerph18020511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yan Y, Jiang W, Liu L, Wang X, Ding C, Tian Z, Zhou R. Dopamine controls systemic inflammation through inhibition of NLRP3 inflammasome. Cell. 2015;160:62–73. doi: 10.1016/j.cell.2014.11.047. [DOI] [PubMed] [Google Scholar]
- 32.Zahid A, Li B, Kombe AJK, Jin T, Tao J. Pharmacological inhibitors of the NLRP3 inflammasome. Front Immunol. 2019;10:2538. doi: 10.3389/fimmu.2019.02538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhang WJ, Luo C, Pu FQ, Zhu JF, Zhu Z. The role and pharmacological characteristics of ATP-gated ionotropic receptor P2X in cancer pain. Pharmacol Res. 2020;161:105106. doi: 10.1016/j.phrs.2020.105106. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.