Presentation of Case
Dr. Philippe-Antoine Bilodeau (Medicine): A 21-year-old man presented to this hospital with a sore throat, epistaxis, and petechiae of the oropharynx.
One week before the current presentation, blood-filled blisters developed inside the patient’s mouth, on the buccal mucosa. He had occasional bleeding from the mouth that resolved spontaneously. He consulted with his dentist, who was concerned about the possibility of impacted wisdom teeth. Two days before the current presentation, a sore throat developed. There was no difficulty with swallowing. On the day of the current presentation, epistaxis developed and did not resolve over a period of 2 hours.
The patient presented to another hospital for evaluation. On examination, the temperature was 37.1°C, the blood pressure 142/76 mm Hg, the pulse 98 beats per minute, the respiratory rate 16 breaths per minute, and the oxygen saturation 99% while he was breathing ambient air. He had blood in the nares and petechiae on the soft palate. There was a large ecchymosis on the left forearm that the patient attributed to carrying heavy boxes several days earlier. The remainder of the examination was normal. Laboratory testing revealed a hemoglobin level of 9.8 g per deciliter (reference range, 13.5 to 17.5), a platelet count of 1000 per microliter (reference range, 150,000 to 450,000), and a white-cell count of 670 per microliter (reference range, 4000 to 11,000). The absolute neutrophil count was 50 per microliter (reference range, 1800 to 7000). Blood levels of electrolytes and glucose were normal, as were results of tests for coagulation, renal function, and liver function. Other laboratory test results are shown in Table 1. The nose was packed to control the bleeding, and 1 unit of platelets was transfused. The patient was transferred to this hospital for additional evaluation and treatment.
Table 1. Laboratory Data.*.
| Variable | Reference Range, Other Hospital | On Arrival, Other Hospital | Reference Range, This Hospital† | Day 1, This Hospital | Day 5, This Hospital |
|---|---|---|---|---|---|
| Hemoglobin (g/dl) | 13.5–17.5 | 9.8 | 13.5–17.5 | 10.0 | 8.4 |
| Hematocrit (%) | 41.0–53.0 | 29.4 | 41.0–53.0 | 30.5 | 23.8 |
| Mean corpuscular volume (fl) | 80–100 | 71.9 | 80–100 | 72.3 | 69.6 |
| Platelet count (per μl) | 150,000–450,000 | 1000 | 150,000–400,000 | 22,000 | 7000 |
| White-cell count (per μl) | 4000–11,000 | 670 | 4500–11,000 | 990 | 800 |
| Differential count (per μl) | |||||
| Neutrophils | 1800–7000 | 50 | 1800–7700 | 50 | 10 |
| Lymphocytes | 1000–4800 | 590 | 1000–4800 | 930 | 790 |
| Monocytes | 200–1200 | 30 | 200–1200 | 0 | 10 |
| Plasma cells | — | — | 0 | 10 | 0 |
| Eosinophils | 50–250 | 0 | 50–250 | 0 | 0 |
| Basophils | 0–300 | 0 | 0–300 | 0 | 0 |
| Reticulocytes (%) | — | — | 0.5–2.5 | <0.5 | <0.5 |
| Sodium (mmol/liter) | 136–145 | 139 | 135–145 | 141 | 135 |
| Potassium (mmol/liter) | 3.6–5.1 | 4.3 | 3.5–5.0 | 4.4 | 4.8 |
| Calcium (mg/dl) | — | — | 8.5–10.5 | 9.7 | 9.4 |
| Phosphorus (mg/dl) | — | — | 2.6–4.5 | 3.1 | 3.7 |
| Urea nitrogen (mg/dl) | 6–20 | 14 | 8–25 | 15 | 13 |
| Creatinine (mg/dl) | 0.6–1.3 | 1.00 | 0.6–1.5 | 0.83 | 0.81 |
| Alanine aminotransferase (U/liter) | 10–50 | 83 | 10–55 | 77 | 44 |
| Aspartate aminotransferase (U/liter) | 15–41 | 49 | 10–40 | 42 | 21 |
| Alkaline phosphatase (U/liter) | 32–100 | 118 | 45–115 | 121 | 99 |
| Total bilirubin (mg/dl) | 0–1.2 | 0.4 | 0–1.0 | 0.6 | 0.5 |
| Albumin (g/dl) | 3.5–5.2 | 4.5 | 3.3–5.0 | 4.9 | 4.1 |
| Globulin (g/dl) | 1.9–4.1 | 3.1 | 1.9–4.1 | 3.2 | 3.6 |
| Lactate dehydrogenase (U/liter) | 98–192 | 282 | 110–210 | 270 | 163 |
| Uric acid (mg/dl) | — | — | 3.6–8.5 | 3.5 | 2.5 |
| Prothrombin time (sec) | 12.1–14.7 | 13.3 | 11.5–14.5 | 13.4 | 14.0 |
| International normalized ratio | 0.9–1.1 | 1.0 | 0.9–1.1 | 1.0 | 1.1 |
| Partial-thromboplastin time (sec) | — | — | 22.0–36.0 | 28.1 | 28.9 |
| d-dimer (ng/ml) | — | — | <500 | 571 | — |
| Fibrinogen (mg/dl) | 200–470 | 355 | 150–400 | 474 | — |
| Ferritin (μg/liter) | — | — | 20–300 | 439 | 528 |
| Iron (μg/dl) | — | — | 45–160 | 170 | — |
| Iron saturation (%) | — | — | 14–50 | 51 | — |
| Total iron-binding capacity (μg/dl) | — | — | 230–404 | 335 | — |
| Folate (ng/ml) | — | — | >4.7 | 10.5 | — |
| Vitamin B12 (pg/ml) | — | — | >231 | 505 | — |
| IgG (mg/dl) | — | — | 614–1295 | 1179 | — |
| IgA (mg/dl) | — | — | 69–309 | 237 | — |
| IgM (mg/dl) | — | — | 53–334 | 116 | — |
| Serum protein electrophoresis | — | — | Normal pattern | Normal pattern | — |
| Free kappa light chain (mg/liter) | — | — | 3.3–19.4 | 16.0 | — |
| Free lambda light chain (mg/liter) | — | — | 5.7–26.3 | 10.7 | — |
| Free kappa:lambda ratio | — | — | 0.30–1.70 | 1.50 | — |
| Hemoglobin electrophoresis (%) | |||||
| Hemoglobin A | — | — | 95.8–98.0 | 68.1 | — |
| Hemoglobin A2 | — | — | 2.0–3.3 | 3.8 | — |
| Hemoglobin F | — | — | 0–0.9 | 0 | — |
| Hemoglobin S | — | — | 0 | 28.1 | — |
To convert the values for calcium to millimoles per liter, multiply by 0.250. To convert the values for phosphorus to millimoles per liter, multiply by 0.3229. To convert the values for urea nitrogen to millimoles per liter, multiply by 0.357. To convert the values for creatinine to micromoles per liter, multiply by 88.4. To convert the values for bilirubin to micromoles per liter, multiply by 17.1. To convert the values for uric acid to micromoles per liter, multiply by 59.48. To convert the values for iron and iron-binding capacity to micromoles per liter, multiply by 0.1791. To convert the values for folate to nanomoles per liter, multiply by 2.266. To convert the values for vitamin B12 to picomoles per liter, multiply by 0.7378.
Reference values are affected by many variables, including the patient population and the laboratory methods used. The ranges used at Massachusetts General Hospital are for adults who are not pregnant and do not have medical conditions that could affect the results. They may therefore not be appropriate for all patients.
In the emergency department of this hospital, the patient reported a mild sore throat but no other symptoms; epistaxis had resolved with nasal packing. There was no fever, weight loss, rash, nausea, vomiting, or diarrhea. The patient had a history of sickle cell trait. He took no medications and had no known allergies. He had been living with his parents and younger brother since he had left his college campus at the start of the coronavirus disease 2019 (Covid-19) pandemic. He worked in a grocery store, and his only known sick contact was his mother, who had been infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) 2 months earlier. She had been isolated in a room in their home during her illness. The patient did not have any symptoms associated with Covid-19 and had never been tested for SARS-CoV-2 infection. His father and brother had been well. The family lived adjacent to a wooded region, and the patient had been hiking in northern New England 2 months before the current presentation. He had not noticed any tick bites. He had no animal contacts. The patient was born in Central America and had moved to a suburban region of New England at 3 years of age. He had previously traveled to Mexico and Canada. He was sexually active with female partners only. He did not smoke tobacco, drink alcohol, or use illicit drugs. His father had hypertension and hyperlipidemia, and paternal family members had sickle cell disease.
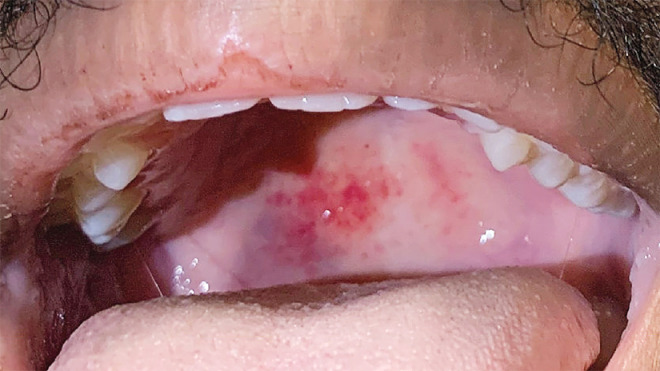
On examination, the patient appeared well. There were petechiae on the soft palate (Figure 1). There was no scleral icterus, lymphadenopathy, or hepatosplenomegaly. A large ecchymosis was present on the left forearm. The remainder of the examination was normal. Blood levels of folate and cobalamin were normal, as were results of serum protein electrophoresis and the ratio of free kappa to lambda light chains. Other laboratory test results are shown in Table 1. A radiograph of the chest and an ultrasound image of the upper abdomen were normal, without splenomegaly. Testing of a specimen obtained from the nasopharynx was positive for SARS-CoV-2 RNA; the patient was admitted to the hospital and placed in enhanced isolation. Examination of thick and thin peripheral-blood smears for babesia was negative.
Figure 1. Clinical Photograph.
A photograph obtained on the day of admission shows a palatal petechia.
A second unit of platelets was transfused, and a diagnostic test was performed.
Differential Diagnosis
Dr. Hanno Hock: I participated in the care of this patient, and I am aware of the diagnosis. This 21-year-old man had been in excellent health until the week of admission, when oropharyngeal and cutaneous bleeding developed. On presentation, the severely decreased platelet count explained the bleeding. The very low total white-cell count and absolute neutrophil count indicated that he was at risk of infection. The only moderately decreased hematocrit suggested that he did not have long-standing severe anemia, but the absence of reticulocytes showed that red-cell production was profoundly impaired. Because he tested positive for SARS-CoV-2 RNA, could this patient’s cytopenias be related to SARS-CoV-2 infection?
Patients with SARS-CoV-2 infection and an acute febrile illness often have blood abnormalities such as pancytopenia,1-3 which are sometimes triggered by hemophagocytic lymphohistiocytosis.4,5 However, this patient did not have a febrile illness. In the absence of illness, his positive SARS-CoV-2 test could be indicative of the presymptomatic incubation period of SARS-CoV-2 infection, asymptomatic active infection, or shedding of RNA after asymptomatic infection. However, none of these circumstances have been associated with major blood abnormalities. Therefore, we needed to consider the wide spectrum of causes of pancytopenia.
Sickle Cell Trait
This patient did not use medications, illicit drugs, or alcohol and had normal levels of vitamin B6 and vitamin B12. These features rule out several common causes of cytopenia. Patients with sickle cell disease are at risk for aplastic crisis, which is an acute decrease in the red-cell count that often occurs after parvovirus B19 infection.6 This patient was a carrier of sickle cell trait but was not at risk for the major complications of sickle cell disease.7 Although sickle cell trait is associated with some health risks8 and should be considered as a potential factor contributing to poor outcomes among Black patients with Covid-19,9 it has not been linked to pancytopenia.7
Increased Cell Destruction
Pancytopenia can result from several conditions that cause increased cell destruction. Cells may pool in an enlarged spleen or be destroyed by the splenic reticuloendothelial system, but this patient did not have splenomegaly. Evans syndrome — a combination of immune thrombocytopenic purpura, autoimmune hemolytic anemia, and immune neutropenia — causes pancytopenia from peripheral destruction.10 Immune thrombocytopenic purpura has recently been reported in association with Covid-19.4 However, this patient did not have evidence of hemolysis or compensatory reticulocytosis, which would be expected in a patient with Evans syndrome. Hemophagocytic lymphohistiocytosis can be triggered by numerous infectious agents other than SARS-CoV-2, including Epstein–Barr virus and cytomegalovirus,5 but this patient did not have signs of infection or inflammation.
Decreased Cell Production
Infection can impair cell production through various mechanisms.11,12 Parvovirus B19,13 hepatitis C virus,14 human herpesvirus 6,15 human herpesvirus 7,16 cytomegalovirus,17 and the human immunodeficiency virus (HIV)18 can directly infect hematopoietic stem cells and progenitor cells. Cytomegalovirus, HIV, and coxsackievirus B can infect bone marrow stromal cells and suppress cell production,11 but this usually occurs in patients who have a chronic condition, take an immunosuppressive medication, or have overt signs of infection. Many pathogens suppress cell production indirectly, through cytokine release and inflammation.11 Respiratory viruses such as influenza A, influenza B, rhinovirus, and SARS-CoV-2 can cause transient cytopenias in ill patients, but this patient did not have a fever and was otherwise well.1,19
Cancer
Lymphoma, myeloma, leukemia, and myelodysplastic syndromes can cause pancytopenia by replacing normal hematopoietic stem cells with malignant cells. Leukemia can be aleukemic, without leukemic blasts in the blood. The presentation of myelodysplastic syndromes in children and young adults may be different from that in older adults; younger patients more often have hypocellular bone marrow, which makes it harder to differentiate the condition from aplastic anemia.20-23 In addition, paroxysmal nocturnal hemoglobinuria, a clonal disorder that initially manifests as hemolytic anemia because of complement-mediated destruction, may cause pancytopenia in the late stages.24 However, the absence of constitutional symptoms, sudden onset of bleeding, and nearly complete absence of cell production made cancer and paroxysmal nocturnal hemoglobinuria unlikely diagnoses in this case.
Aplastic Anemia
Aplastic anemia is characterized by pancytopenia and the loss of hematopoietic stem cells, progenitor cells, and precursor cells in the bone marrow.25,26 It was the most likely diagnosis in this previously healthy patient with pancytopenia in the absence of constitutional symptoms and abnormal cells. Aplastic anemia results from one of three main mechanisms25: damage by extrinsic factors, manifestations of familial genetic mutations, and autoimmune attack on hematopoietic stem cells and progenitor cells.
Extrinsic causes of aplastic anemia are usually obvious and include major accidental or therapeutic exposure to radiation, chemotherapy, or massive exposure to benzene25 or pesticides such as organochlorines and organophosphates.27 Several medications have been infrequently associated with aplastic anemia.27,28 However, this patient was not taking any medications and had no known chemical or radiation exposures.
The genetic disorder most commonly associated with aplastic anemia is Fanconi’s anemia, a DNA repair defect that results from a mutation in 1 of at least 15 known genes.29 Patients with Fanconi’s anemia typically have bone marrow failure in the first or second decade of life, as well as other congenital abnormalities, including thumb and facial deformities and short stature.29 The second most common genetic cause of bone marrow failure is dyskeratosis congenita, which is due to mutations in genes involved in telomere repair or protection.30 Dyskeratosis congenita typically manifests in childhood with skin pigmentation abnormalities, oral leukoplakia, and dystrophic nails. However, the disease may cause isolated bone marrow failure.31 Finally, GATA2 mutations have emerged as a potential genetic cause of aplastic anemia.32 These mutations are associated with other manifestations, including lymphedema, generalized warts from human papillomavirus infection, and mycobacterial infection.23 This patient’s pancytopenia was unlikely to be caused by a congenital genetic abnormality because he had no family history of such a condition and no associated signs or symptoms.
Up to 70% of cases of aplastic anemia occur sporadically, resulting from the sudden onset of T-cell–mediated destruction of hematopoietic stem cells and progenitor cells.28 Most cases of aplastic anemia are designated as idiopathic because the triggers for the immune attack on hematopoiesis are obscure. There are known associations with thymoma33 and eosinophilic fasciitis.34 However, both these diseases develop predominantly in the seventh decade of life, and this patient did not have the widened mediastinum on chest radiography that is suggestive of thymoma or the dimpled skin thickening (peau d’orange change) that is suggestive of eosinophilic fasciitis. 34
The most common known trigger for aplastic anemia is seronegative hepatitis, which precedes 5 to 10% of cases of aplastic anemia by approximately 2 to 3 months.35,36 The median age at the onset of hepatitis-associated aplastic anemia is 20 years, but this patient’s history did not suggest recent hepatitis. Aplastic anemia is also thought to occur after infection with common hepatitis viruses37 and other viruses, including HIV and parvovirus B19,12,38,39 but at a much lower frequency than with seronegative hepatitis.
It is intriguing that this patient’s exposure to SARS-CoV-2 and probable asymptomatic infection occurred 2 months before his presentation, an interval similar to that between the onset of seronegative hepatitis and the development of aplastic anemia; therefore, it is tempting to speculate that SARS-CoV-2 infection may have been a trigger. However, his presentation and age also fit with idiopathic aplastic anemia, which has an incidence of 2 to 3 cases per 1 million per year.27 Given the scale of the Covid-19 pandemic, a potential association may eventually be confirmed if the incidence of aplastic anemia rises. Regardless of the trigger, this patient’s presentation and blood abnormalities were highly suggestive of aplastic anemia, a diagnosis that must be confirmed with evidence of markedly reduced hematopoiesis in a bone marrow–biopsy specimen. The severity of aplastic anemia is determined by the degree of the associated peripheral-blood cytopenias.28,40,41 The near absence of platelets, neutrophils, and reticulocytes in this case was associated with a high risk of death and a very low chance of recovery without urgent treatment and consideration of bone marrow transplantation.
Dr. Hanno Hock’s Diagnosis
Severe aplastic anemia in the presence of infection with severe acute respiratory syndrome coronavirus 2.
Diagnostic Testing
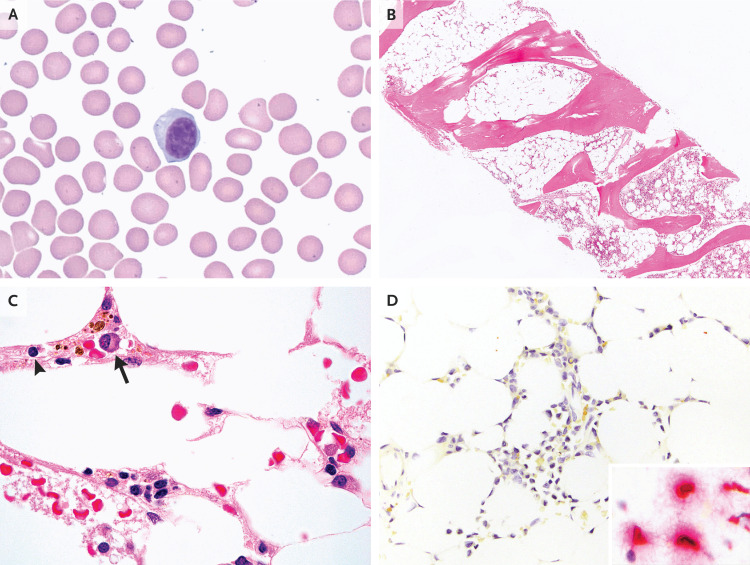
Dr. Lucas R. Massoth: Examination of a peripheral-blood smear confirmed the presence of severe leukopenia and thrombocytopenia. Most of the white cells were lymphocytes, including plasmacytoid forms, and there were rare plasma cells (Figure 2A). The neutrophils were morphologically normal. Results of flow cytometric studies of the peripheral blood, including tests for paroxysmal nocturnal hemoglobinuria and telomere length analysis, were unremarkable.
Figure 2. Peripheral-Blood and Bone Marrow Specimens.
Wright–Giemsa staining of a peripheral-blood smear (Panel A) shows a circulating plasmacytoid lymphocyte; platelets are notably absent, a finding consistent with thrombocytopenia. Hematoxylin and eosin staining of bone marrow (Panel B) shows markedly hypocellular marrow. At higher magnification (Panel C), the residual cellularity is composed mainly of lymphocytes (arrowhead) and plasma cells (arrow). In situ hybridization of bone marrow to detect severe acute respiratory syndrome coronavirus 2 (Panel D) is negative, without the red chromogen staining that indicates the presence of viral RNA; the inset shows an example of positive cellular staining in human lung tissue.
An aspirate and a core biopsy specimen of the bone marrow were obtained. The aspirate was paucicellular and composed of peripheral-blood elements. The core biopsy specimen (Figure 2B and 2C) showed markedly hypocellular marrow (<5% cellularity) for the patient’s age and was composed mainly of lymphocytes and plasma cells. Maturing hematopoietic elements were rare, and reticulin fibrosis was absent. Immunohistochemical stains used to detect proteins expressed by herpes simplex virus types 1 and 2, cytomegalovirus, and parvovirus B19 were negative. Chromogenic in situ hybridization studies used to detect RNA of Epstein–Barr virus and SARS-CoV-242 (Figure 2D) were also negative.
Flow cytometric studies of the bone marrow aspirate showed no abnormal lymphoid or myeloblast population. Cytogenetic studies failed to obtain metaphases for analysis, most likely because of low specimen cellularity. Targeted next-generation sequencing was negative for mutations commonly associated with hematologic cancer, including predisposing germline variants.
The overall findings of pancytopenia and severe trilineage bone marrow hypoplasia were most consistent with a diagnosis of acquired aplastic anemia. Although circulating plasmacytoid lymphocytes and plasma cells are not characteristic of aplastic anemia, their presence has been documented after various infections, including SARS-CoV-2 infection.43 The absence of detectable SARS-CoV-2 RNA on in situ hybridization argues against the possibility that intramedullary infection had led to bone marrow injury.
Pathological Diagnosis
Severe acquired aplastic anemia.
Hospital Course
Dr. Bilodeau: While the results of the bone marrow biopsy were pending, filgrastim and eltrombopag were administered. On hospital day 5, the sore throat worsened and fever developed, with a temperature of 38.2°C. An oral examination showed persistent petechiae and leftward deviation of the uvula. There was mild tenderness of the anterior aspect of the neck on the right side. Laboratory test results are shown in Table 1. Imaging studies were obtained, and intravenous cefepime and vancomycin were administered.
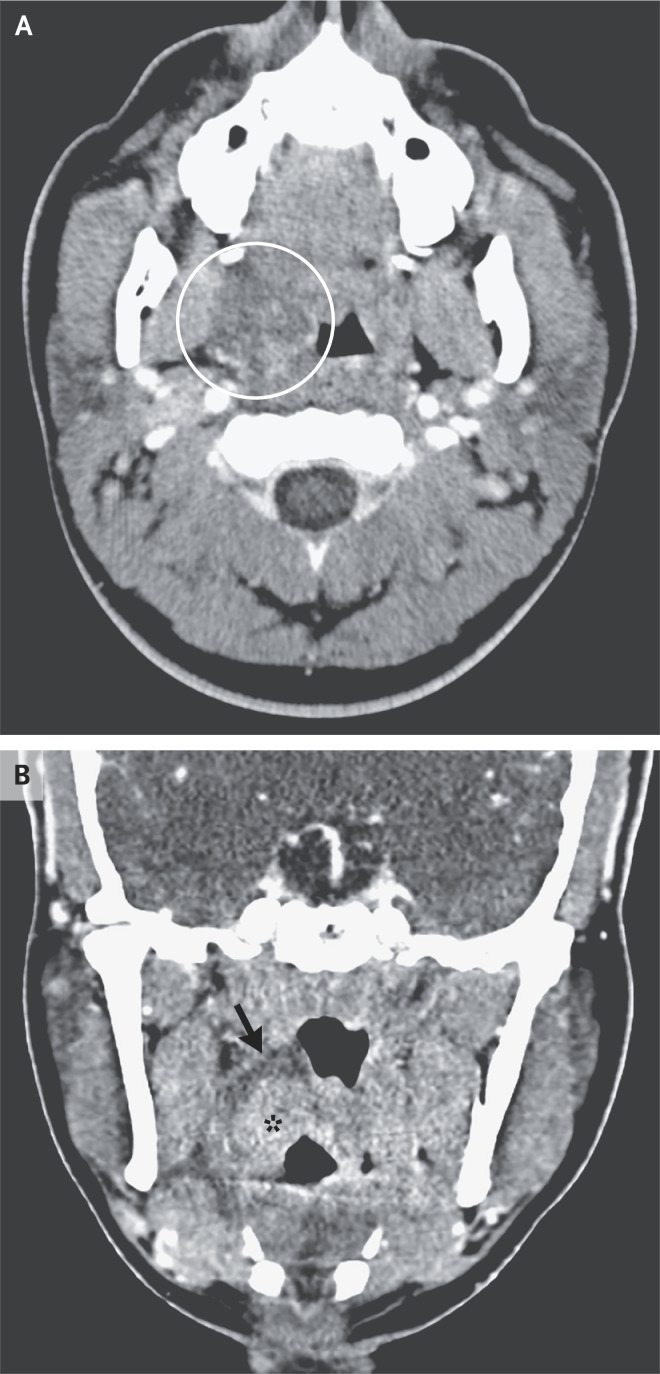
Dr. Hillary R. Kelly: Computed tomography of the neck (Figure 3), performed after the administration of intravenous contrast material, revealed marked enlargement of the right palatine tonsil. There was a surrounding hypoattenuating abnormality in the right peritonsillar space that was most consistent with phlegmon or edema. These changes were causing partial airway effacement. There was no evidence of a well-defined rim-enhancing peritonsillar abscess.
Figure 3. CT Scan of the Neck.
CT of the neck was performed after the administration of intravenous contrast material in a soft-tissue algorithm. An axial image (Panel A) shows hypoattenuating peritonsillar phlegmon (circled), which is causing partial effacement of the oropharyngeal airway. A coronal image (Panel B) shows the rim of the hypoattenuating phlegmon in the peritonsillar space (arrow) adjacent to the enlarged right palatine tonsil (asterisk). There is no evidence of a well-defined rim-enhancing abscess.
Dr. Bilodeau: The patient’s sore throat improved 48 hours after the administration of intravenous antibiotic agents, and bone marrow transplantation was considered.
Discussion of Management
Dr. Eric A. Meyerowitz: It was necessary to determine whether this patient had active SARS-CoV-2 infection. The presence of active infection would have major implications regarding the safety of bone marrow transplantation.
SARS-CoV-2 infection can be either symptomatic or asymptomatic. Asymptomatic infection occurs when a person has none of the many symptoms attributable to SARS-CoV-2 throughout the course of infection.44,45 This patient had a sore throat and fever on hospital day 5, which could have been consistent with the onset of symptoms. However, he had focal tenderness on the right side of the neck and phlegmon on imaging that responded well to the prompt administration of intravenous antibiotics. This response was most consistent with a pharyngeal bacterial infection in the presence of neutropenia.
Since the household is the most common site of viral transmission, we suspected that the patient had been infected with SARS-CoV-2 approximately 2 months earlier, when his mother had a confirmed infection; community transmission was uncontrolled at that time.46 In immunocompetent adults, viable SARS-CoV-2 can be recovered until approximately 10 days after the onset of mild disease and 20 days after the onset of severe disease.47 Viral replication occurs in the respiratory tract of infected persons; after an incubation period, the viral load increases rapidly, peaking within 3 days after it starts to rise, and then begins to decline.48 Although active viral replication occurs over a relatively short period, viral RNA is often detected well after this period, with a median duration of nasopharyngeal RNA shedding of 22 days in immunocompetent hosts with mild disease.49 Therefore, a positive nucleic acid test may indicate either the presence of replicating virus (a finding diagnostic of active infection) or RNA shedding (a finding consistent with either very early infection or late, resolving infection) but cannot be used to distinguish between these clinical scenarios.
My approach to interpreting the positive SARS-CoV-2 test in this patient involved estimating where he was in the course of infection. Tracking the cycle threshold (Ct) value is an indirect but evidence-based method to assess for viable virus. The Ct value is the number of cycles required to amplify viral RNA to a level at which it can be detected by the assay. It is inversely correlated with the viral load. Many SARS-CoV-2 nucleic acid tests use a Ct value as a cutoff; if the patient’s Ct value is below the cutoff, the test is considered to be positive. Most commercially available assays that use Ct values typically undergo approximately 40 cycles. For each patient, serial testing should be performed at a single laboratory. A first test with a Ct value of less than 30 is a strong predictor of acute infection.48 Two tests separated by at least 2 days with Ct values of 30 or more suggest that early infection, whether primary infection or reinfection, is unlikely.48,50-53 This patient had a positive SARS-CoV-2 test with a Ct value of 38 on admission and had another positive test with a Ct value of 35 on hospital day 6; on both tests, only one of two targets was amplified, which highlighted the profoundly low level of viral RNA present. These Ct values have not been associated with active viral replication in the extensive literature published to date.
In putting this information together, our assessment was that this patient was a previously immunocompetent young man who had a history of persistent asymptomatic SARS-CoV-2 infection, which was currently in a state of prolonged RNA shedding, without evidence of replication-competent virus. On the basis of these findings, I thought that the patient could safely undergo bone marrow transplantation and was considered to have a very low risk of additional complications from SARS-CoV-2 infection.
Dr. Matthew J. Frigault: Management of aplastic anemia depends on the severity of illness, the patient’s age, the availability of an appropriate stem-cell donor, and the presence of coexisting conditions that could limit the patient’s ability to undergo allogeneic stem-cell transplantation.26 This patient met the criteria for severe aplastic anemia, which are a bone marrow cellularity of less than 25% and at least two of the following features: an absolute neutrophil count in peripheral blood of less than 500 per microliter, a platelet count of less than 20,000 per microliter, or a reticulocyte count of less than 20,000 per microliter. This patient also met the criteria for very severe aplastic anemia, because his absolute neutrophil count was less than 200 per microliter.
Other than stem-cell transplantation, standard intensive approaches include combination immunosuppressive therapy. Although this approach is promising, rates of relapse and clonal evolution to myelodysplastic syndrome or acute myeloid leukemia are often higher in patients treated with immunosuppressive therapy alone than in those treated with stem-cell transplantation, with rates of failure-free survival beyond 10 years of less than 50% in some series.54 For this reason, the use of allogeneic stem-cell transplantation in medically fit patients with appropriate stem-cell donors has increased.55 Traditional risks associated with allogeneic stem-cell transplantation include graft failure and graft-versus-host disease; the risk of graft-versus-host disease depends on the degree of HLA disparity, the sex of the donor, the intensity of transplantation conditioning, and the graft source.56
This patient had a 16-year-old brother who was a complete match, and because the brother had robust fitness and did not have any precluding conditions, we elected to pursue stem-cell transplantation with the brother as the donor. The risks of transplantation have been substantially reduced through advances in the use of alternative donor sources and in strategies for the prevention of graft-versus-host disease. In two recent series that evaluated the use of nonmyeloablative conditioning with a post-transplantation regimen of cyclophosphamide in combination with tacrolimus and mycophenolate mofetil in patients with severe aplastic anemia who received stem-cell transplants from bone marrow grafts of matched related and unrelated donors, overall survival at 2 years was nearly 100% and graft-versus-host disease–free survival was more than 84%.57,58 For this patient, we proceeded with upfront allogeneic stem-cell transplantation.
Despite the challenges of a recent SARS-CoV-2 infection, with close collaboration from our infectious disease colleagues, we were able to begin transplantation in the patient within 28 days after the initial presentation. Now, 1 year after transplantation, he has full donor chimerism and a complete hematologic response. He has had no evidence of acute or chronic graft-versus-host disease. The patient is completing college coursework and working part time, and he went skydiving to celebrate his 1-year anniversary of completing transplantation.
Final Diagnosis
Severe acquired aplastic anemia after infection with severe acute respiratory syndrome coronavirus 2.
Acknowledgments
We thank Dr. Valentina Nardi for her review of the pathological discussion and Dr. Niyati Desai for the performance of SARS-CoV-2 in situ hybridization.
Disclosure Forms
Footnotes
This case was presented at the Medical Case Conference.
Disclosure forms provided by the authors are available with the full text of this article at NEJM.org.
References
- 1.Fan BE, Chong VCL, Chan SSW, et al. Hematologic parameters in patients with COVID-19 infection. Am J Hematol 2020;95(6):E131-E134. [DOI] [PubMed] [Google Scholar]
- 2.Zulfiqar A-A, Lorenzo-Villalba N, Hassler P, Andrès E. Immune thrombocytopenic purpura in a patient with Covid-19. N Engl J Med 2020;382(18):e43-e43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Issa N, Lacassin F, Camou F. First case of persistent pancytopenia associated with SARS-CoV-2 bone marrow infiltration in an immunocompromised patient. Ann Oncol 2020;31:1418-1419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dimopoulos G, de Mast Q, Markou N, et al. Favorable Anakinra responses in severe Covid-19 patients with secondary hemophagocytic lymphohistiocytosis. Cell Host Microbe 2020;28(1):117-123.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ramos-Casals M, Brito-Zerón P, López-Guillermo A, Khamashta MA, Bosch X. Adult haemophagocytic syndrome. Lancet 2014;383:1503-1516. [DOI] [PubMed] [Google Scholar]
- 6.Serjeant GR, Topley JM, Mason K, et al. Outbreak of aplastic crises in sickle cell anaemia associated with parvovirus-like agent. Lancet 1981;2:595-597. [DOI] [PubMed] [Google Scholar]
- 7.Naik RP, Smith-Whitley K, Hassell KL, et al. Clinical outcomes associated with sickle cell trait: a systematic review. Ann Intern Med 2018;169:619-627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Xu JZ, Thein SL. The carrier state for sickle cell disease is not completely harmless. Haematologica 2019;104:1106-1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kehinde TA, Osundiji MA. Sickle cell trait and the potential risk of severe coronavirus disease 2019 — a mini-review. Eur J Haematol 2020;105:519-523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jaime-Pérez JC, Aguilar-Calderón PE, Salazar-Cavazos L, Gómez-Almaguer D. Evans syndrome: clinical perspectives, biological insights and treatment modalities. J Blood Med 2018;9:171-184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pascutti MF, Erkelens MN, Nolte MA. Impact of viral infections on hematopoiesis: from beneficial to detrimental effects on bone marrow output. Front Immunol 2016;7:364-364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Morinet F, Leruez-Ville M, Pillet S, Fichelson S. Concise review: anemia caused by viruses. Stem Cells 2011;29:1656-1660. [DOI] [PubMed] [Google Scholar]
- 13.Young NS, Brown KE. Parvovirus B19. N Engl J Med 2004;350:586-597. [DOI] [PubMed] [Google Scholar]
- 14.Sansonno D, Lotesoriere C, Cornacchiulo V, et al. Hepatitis C virus infection involves CD34(+) hematopoietic progenitor cells in hepatitis C virus chronic carriers. Blood 1998;92:3328-3337. [PubMed] [Google Scholar]
- 15.Isomura H, Yamada M, Yoshida M, et al. Suppressive effects of human herpesvirus 6 on in vitro colony formation of hematopoietic progenitor cells. J Med Virol 1997;52:406-412. [DOI] [PubMed] [Google Scholar]
- 16.Mirandola P, Secchiero P, Pierpaoli S, et al. Infection of CD34(+) hematopoietic progenitor cells by human herpesvirus 7 (HHV-7). Blood 2000;96:126-131. [PubMed] [Google Scholar]
- 17.Maciejewski JP, Bruening EE, Donahue RE, Mocarski ES, Young NS, St Jeor SC. Infection of hematopoietic progenitor cells by human cytomegalovirus. Blood 1992;80:170-178. [PubMed] [Google Scholar]
- 18.Prost S, Le Dantec M, Augé S, et al. Human and simian immunodeficiency viruses deregulate early hematopoiesis through a Nef/PPARgamma/STAT5 signaling pathway in macaques. J Clin Invest 2008;118:1765-1775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Alexandropoulou O, Kossiva L, Giannaki M, Panagiotou J, Tsolia M, Karavanaki K. The epidemiology, clinical course and outcome of febrile cytopenia in children. Acta Paediatr 2015;104(3):e112-e118. [DOI] [PubMed] [Google Scholar]
- 20.Nakano TA, Lau BW, Dickerson KE, et al. Diagnosis and treatment of pediatric myelodysplastic syndromes: a survey of the North American Pediatric Aplastic Anemia Consortium. Pediatr Blood Cancer 2020;67(10):e28652-e28652. [DOI] [PubMed] [Google Scholar]
- 21.Glaubach T, Robinson LJ, Corey SJ. Pediatric myelodysplastic syndromes: they do exist! J Pediatr Hematol Oncol 2014;36:1-7. [DOI] [PubMed] [Google Scholar]
- 22.Wlodarski MW, Hirabayashi S, Pastor V, et al. Prevalence, clinical characteristics, and prognosis of GATA2-related myelodysplastic syndromes in children and adolescents. Blood 2016;127:1387-1397. [DOI] [PubMed] [Google Scholar]
- 23.Kallen ME, Dulau-Florea A, Wang W, Calvo KR. Acquired and germline predisposition to bone marrow failure: diagnostic features and clinical implications. Semin Hematol 2019;56:69-82. [DOI] [PubMed] [Google Scholar]
- 24.Socié G, Mary JY, de Gramont A, et al. Paroxysmal nocturnal haemoglobinuria: long-term follow-up and prognostic factors. Lancet 1996;348:573-577. [DOI] [PubMed] [Google Scholar]
- 25.Young NS. Aplastic anemia. N Engl J Med 2018;379:1643-1656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Killick SB, Bown N, Cavenagh J, et al. Guidelines for the diagnosis and management of adult aplastic anaemia. Br J Haematol 2016;172:187-207. [DOI] [PubMed] [Google Scholar]
- 27.Shallis RM, Ahmad R, Zeidan AM. Aplastic anemia: etiology, molecular pathogenesis, and emerging concepts. Eur J Haematol 2018;101:711-720. [DOI] [PubMed] [Google Scholar]
- 28.Scheinberg P. Novel therapeutic choices in immune aplastic anemia. F1000Res 2020;9:F1000 Faculty Rev-1118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Soulier J. Fanconi anemia. Hematology Am Soc Hematol Educ Program 2011;2011:492-497. [DOI] [PubMed] [Google Scholar]
- 30.Dokal I. Dyskeratosis congenita. Hematology Am Soc Hematol Educ Program 2011;2011:480-486. [DOI] [PubMed] [Google Scholar]
- 31.Townsley DM, Dumitriu B, Liu D, et al. Danazol treatment for telomere diseases. N Engl J Med 2016;374:1922-1931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wlodarski MW, Collin M, Horwitz MS. GATA2 deficiency and related myeloid neoplasms. Semin Hematol 2017;54:81-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bernard C, Frih H, Pasquet F, et al. Thymoma associated with autoimmune diseases: 85 cases and literature review. Autoimmun Rev 2016;15:82-92. [DOI] [PubMed] [Google Scholar]
- 34.de Masson A, Bouaziz J-D, de Latour RP, et al. Severe aplastic anemia associated with eosinophilic fasciitis: report of 4 cases and review of the literature. Medicine (Baltimore) 2013;92:69-81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Brown KE, Tisdale J, Barrett AJ, Dunbar CE, Young NS. Hepatitis-associated aplastic anemia. N Engl J Med 1997;336:1059-1064. [DOI] [PubMed] [Google Scholar]
- 36.Camitta BM, Nathan DG, Forman EN, Parkman R, Rappeport JM, Orellana TD. Posthepatitic severe aplastic anemia — an indication for early bone marrow transplantation. Blood 1974;43:473-483. [PubMed] [Google Scholar]
- 37.Rauff B, Idrees M, Shah SAR, et al. Hepatitis associated aplastic anemia: a review. Virol J 2011;8:87-87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Brown KE, Young NS. Parvoviruses and bone marrow failure. Stem Cells 1996;14:151-163. [DOI] [PubMed] [Google Scholar]
- 39.Mishra B, Malhotra P, Ratho RK, Singh MP, Varma S, Varma N. Human parvovirus B19 in patients with aplastic anemia. Am J Hematol 2005;79:166-167. [DOI] [PubMed] [Google Scholar]
- 40.Camitta BM, Rappeport JM, Parkman R, Nathan DG. Selection of patients for bone marrow transplantation in severe aplastic anemia. Blood 1975;45:355-363. [PubMed] [Google Scholar]
- 41.Bacigalupo A, Hows J, Gluckman E, et al. Bone marrow transplantation (BMT) versus immunosuppression for the treatment of severe aplastic anaemia (SAA): a report of the EBMT SAA working party. Br J Haematol 1988;70:177-182. [DOI] [PubMed] [Google Scholar]
- 42.Massoth LR, Desai N, Szabolcs A, et al. Comparison of RNA in situ hybridization and immunohistochemistry techniques for the detection and localization of SARS-CoV-2 in human tissues. Am J Surg Pathol 2021;45:14-24. [DOI] [PubMed] [Google Scholar]
- 43.Sadigh S, Massoth LR, Christensen BB, Stefely JA, Keefe J, Sohani AR. Peripheral blood morphologic findings in patients with COVID-19. Int J Lab Hematol 2020;42(6):e248-e251. [DOI] [PubMed] [Google Scholar]
- 44.Meyerowitz EA, Richterman A, Bogoch II, Low N, Cevik M. Towards an accurate and systematic characterisation of persistently asymptomatic infection with SARS-CoV-2. Lancet Infect Dis 2021;21(6):e163-e169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Buitrago-Garcia D, Egli-Gany D, Counotte MJ, et al. Occurrence and transmission potential of asymptomatic and presymptomatic SARS-CoV-2 infections: a living systematic review and meta-analysis. PLoS Med 2020;17(9):e1003346-e1003346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Adam DC, Wu P, Wong JY, et al. Clustering and superspreading potential of SARS-CoV-2 infections in Hong Kong. Nat Med 2020;26:1714-1719. [DOI] [PubMed] [Google Scholar]
- 47.Meyerowitz EA, Richterman A, Gandhi RT, Sax PE. Transmission of SARS-CoV-2: a review of viral, host, and environmental factors. Ann Intern Med 2021;174:69-79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kissler SM, Fauver JR, Mack C, et al. Viral dynamics of acute SARS-CoV-2 infection. June 6, 2021. (https://www.medrxiv.org/content/10.1101/2020.10.21.20217042v3). preprint. [DOI] [PMC free article] [PubMed]
- 49.Sun J, Xiao J, Sun R, et al. Prolonged persistence of SARS-CoV-2 RNA in body fluids. Emerg Infect Dis 2020;26:1834-1838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bullard J, Dust K, Funk D, et al. Predicting infectious severe acute respiratory syndrome coronavirus 2 from diagnostic samples. Clin Infect Dis 2020;71:2663-2666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wölfel R, Corman VM, Guggemos W, et al. Virological assessment of hospitalized patients with COVID-2019. Nature 2020;581:465-469. [DOI] [PubMed] [Google Scholar]
- 52.Arons MM, Hatfield KM, Reddy SC, et al. Presymptomatic SARS-CoV-2 infections and transmission in a skilled nursing facility. N Engl J Med 2020;382:2081-2090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kim AY, Gandhi RT. Re-infection with SARS-CoV-2: what goes around may come back around. Clin Infect Dis 2020. October 9 (Epub ahead of print).33035308 [Google Scholar]
- 54.Drexler B, Zurbriggen F, Diesch T, et al. Very long-term follow-up of aplastic anemia treated with immunosuppressive therapy or allogeneic hematopoietic cell transplantation. Ann Hematol 2020;99:2529-2538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Armand P, Antin JH. Allogeneic stem cell transplantation for aplastic anemia. Biol Blood Marrow Transplant 2007;13:505-516. [DOI] [PubMed] [Google Scholar]
- 56.Flowers MED, Inamoto Y, Carpenter PA, et al. Comparative analysis of risk factors for acute graft-versus-host disease and for chronic graft-versus-host disease according to National Institutes of Health consensus criteria. Blood 2011;117:3214-3219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.DeZern AE, Zahurak ML, Symons HJ, et al. Haploidentical BMT for severe aplastic anemia with intensive GVHD prophylaxis including posttransplant cyclophosphamide. Blood Adv 2020;4:1770-1779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Leick M, Hunter B, DeFilipp Z, et al. Posttransplant cyclophosphamide in allogeneic bone marrow transplantation for the treatment of nonmalignant hematological diseases. Bone Marrow Transplant 2020;55:758-762. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.