Abstract
Staphylococcusaureus is an important opportunistic pathogen that causes many infections in humans and animals. The inappropriate use of antibiotics has favored the diffusion of methicillin-resistant S. aureus (MRSA), nullifying the efforts undertaken in the discovery of antimicrobial agents. Oxadiazole heterocycles represent privileged scaffolds for the development of new drugs because of their unique bioisosteric properties, easy synthesis, and therapeutic potential. A vast number of oxadiazole-containing derivatives have been discovered as potent antibacterial agents against multidrug-resistant MRSA strains. Here, we investigate the ability of a new library of oxadiazoles to contrast the growth of Gram-positive and Gram-negative strains. The strongest antimicrobial activity was obtained with compounds 3 (4 µM) and 12 (2 µM). Compound 12, selected for further evaluation, was found to be noncytotoxic on the HaCaT cell line up to 25 µM, bactericidal, and was able to improve the activity of oxacillin against the MRSA. The highest synergistic interaction was obtained with the combination values of 0.78 μM for compound 12, and 0.06 μg/mL for oxacillin. The FIC index value of 0.396 confirms the synergistic effect of compound 12 and oxacillin. MRSA treatment with compound 12 reduced the expression of genes included in the mec operon. In conclusion, 12 inhibited the growth of the MRSA and restored the activity of oxacillin, thus resulting in a promising compound in the treatment of MRSA infection.
Keywords: 1,2,4-oxadiazole; Staphylococcus aureus; MRSA; antimicrobial activity; synergistic interaction; structure–activity relationship; drug discovery
1. Introduction
Antibiotics are a class of drugs traditionally used as the first strategy (weapon) against serious infections caused by bacteria. Among these, Staphylococcus aureus is an important opportunistic pathogen that causes many infections in humans and animals, such as skin and lung infections, endocarditis, toxic shock syndrome, and sepsis [1]. It is a member of the human microbiota (about 30% of the human population is colonized with S. aureus) with a pathogenic potential derived from the ability to express a wide variety of virulence factors used to overcome the host defense mechanism [2]. Antibiotics have generally shown efficacy in the treatment of bacterial infections, but their inappropriate and indiscriminate use has favored the diffusion of resistant bacteria. This is the case with β-lactams and the selection of methicillin-resistant S. aureus (MRSA), which has nullified the efforts undertaken in the discovery of antimicrobial agents [3]. S. aureus can also form biofilm on medical devices, and this represents a further complication in the management of infections because of the difficulties of antibiotics to penetrate the biofilm layer [4]. For all these reasons, S. aureus is considered one of the major causes of hospital-acquired infections worldwide [5].
The discovery of new targets or compounds that are potentially able to control diseases by resistant bacteria represents an urgent goal [6].
The 1,2,4-oxadiazole heterocyclic ring represents an important scaffold in organic and medicinal chemistry, with an elevated versatility that affords molecular diversity.
A vast number of 1,2,4-oxadiazole-bearing molecules have been found to possess diverse biological activities, such as anticancer, anti-inflammatory, anticonvulsant, antiviral, antibacterial, and anti-Alzheimer activities, as well as specific inhibitory properties against enzymes [7,8,9,10].
In 2014, using in silico screening, 1,2,4-oxadiazole-derived compounds were discovered by Chang and coworkers as a new class of non-β-lactam antibiotics [11].
On the basis of the previously disclosed lead compound 1, our study aims to further expand the structural diversity around the 1,2,4-oxadiazole-selected scaffold in order to identify novel and promising antibacterial chemical entities.
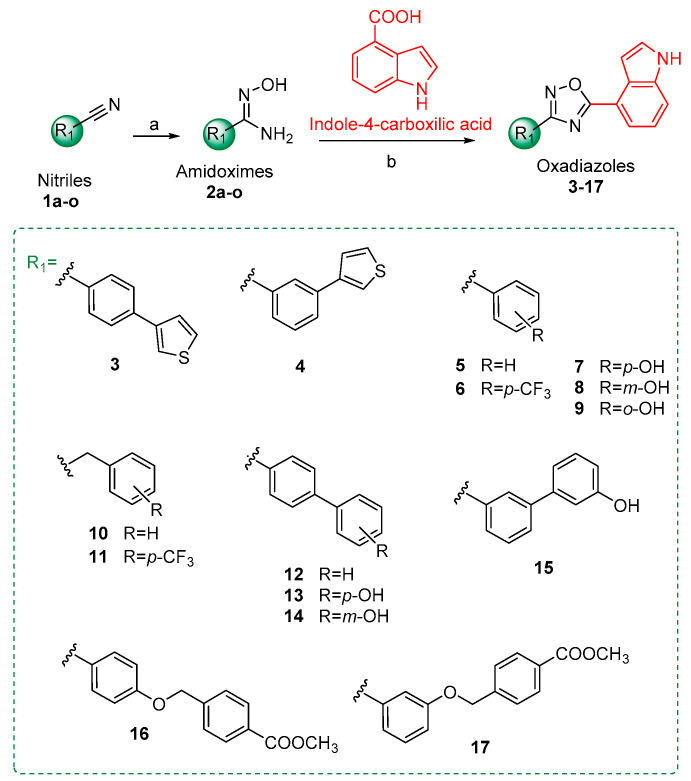
New 1,2,4-oxadiazoles (3–17) were designed by fixing a common 4-indole ring at C-5, and by varying the aromatic substituent in position 3. These compounds were synthesized based on amidoxime and carboxylic acid heterocyclization and were tested for their antimicrobial activity against S. aureus 29213 and 43300 (MRSA), P. aeruginosa, and K. pneumoniae.
2. Results
2.1. Compounds and Chemistry
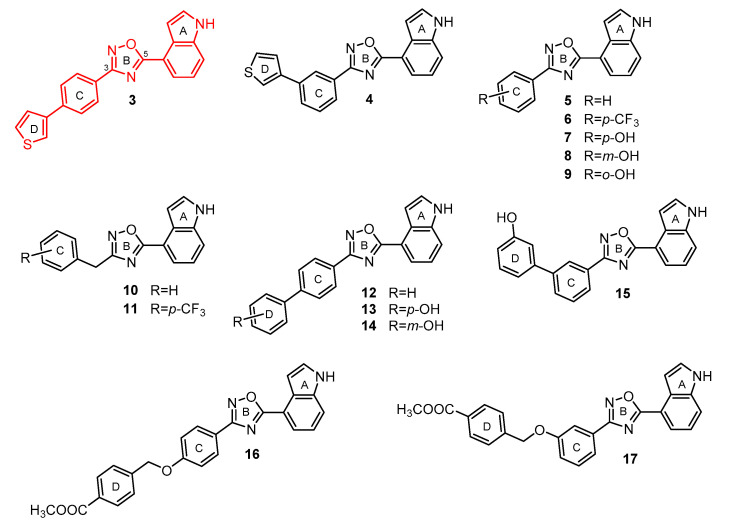
We report herein the synthesis and SAR results, against S. aureus, of a library of fifteen synthetic oxadiazoles (3–17), featuring a common 5-(4′ indolyl) substituent, and different aromatic substitutions at C-3 (Figure 1).
Figure 1.
1,2,4-oxadiazole derivatives generated in this study.
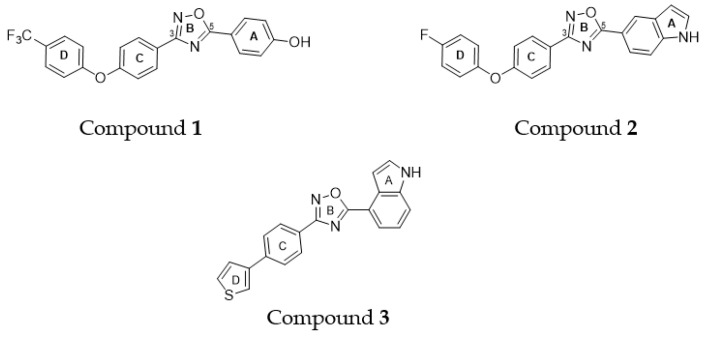
Starting from the lead structure 1 (Figure 2) discovered by Chang and coworkers [11], and featuring four interconnected aromatic rings named A-D, the same authors report an extensive structure–activity relationship that discloses compounds bearing a 5-indole ring A, for example, compound 2, (Figure 2), as the best candidates, in terms of activity, against MSRA strains, low toxicity, oral bioavailability, and in vivo activity in murine models of infection [12,13,14].
Figure 2.
Structures of previously synthetized oxadiazoles (1–3).
Very recently, in the frame of our interest toward the development of 1,2,4-oxadiazole-based active molecules [15,16], we designed and synthesized a small library of oxadiazole-containing compounds endowed with a polypharmacological profile against the enzymes of the eicosanoid biosynthesis [17].
Since all of the compounds within the library were filtered through the detailed in silico assessment of their drug-like properties, we envisaged a repositioning of the molecules that were lacking in anti-inflammatory activity.
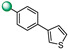
Compound 3, synthetized in our previous study [17], features some analogies with compounds 1 and 2, namely, the concatenation of four aromatic rings and a regioisomeric 4-indole group at the C-5 position of the 1,2,4-oxadiazole ring (Figure 2). On the basis of these observations, we analyzed the antimicrobial activity of compound 3 on two strains of S. aureus (ATCC 29213 and MRSA ATCC 43300). As reported in Table 1, it showed strong activity, with an MIC value of 4 µM, on both S. aureus strains.
Table 1.
In vitro antimicrobial activity of compounds 3–17, MIC values are expressed as µM.
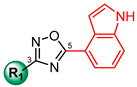
| Cmpds | R1 | S. aureus ATCC 29213 | S. aureus ATCC 43300 |
|---|---|---|---|
| 3 |
|
4 | 4 |
| 4 |
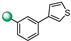
|
>100 | >100 |
| 5 |
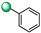
|
25 | 12.5 |
| 6 |
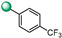
|
6.25 | 6.25 |
| 7 |
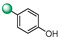
|
100 | 100 |
| 8 |
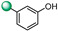
|
>100 | >100 |
| 9 |
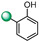
|
25 | 25 |
| 10 |
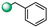
|
>100 | >100 |
| 11 |
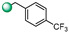
|
6.25 | 6.25 |
| 12 |
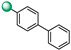
|
2 | 2 |
| 13 |
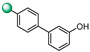
|
6.25 | 6.25 |
| 14 |
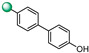
|
6.25 | 6.25 |
| 15 |
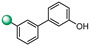
|
12.5 | 12.5 |
| 16 |
|
>100 | >100 |
| 17 |
|
>100 | >100 |
| Vancomycin a | 2 | 2 | |
| Oxacillin a | 2 | 10 |
a Antibiotic concentrations are expressed as µg/mL (2 μg/mL corresponds to 1.35 µM for vancomycin, and 4.72 µM for oxacillin; 10 µg/mL of oxacillin corresponds to 23.6 µM).
Thus, starting from this encouraging finding, we further expanded the chemodiversity around the 1,2,4-oxadiazole ring by fixing the 4-indolyl system as ring A, and by varying the structures of rings C and D. In particular, in some compounds, we removed ring D and added different substituents on ring C (see compounds 5–9), while in others, we replaced the diphenyl ether portion (rings C and D) with different benzyl, biphenyl, thienyl, or benzyloxyphenyl rings substituted at different positions (see compounds 3,4 and 10–17) (Figure 1). A key attractive feature of this class of compounds is its easy synthetic preparation, outlined in Scheme 1. In particular, indole-4-carboxylic acid was reacted, using HBTU as a coupling agent, with amidoximes 2a-o, in turn, prepared by the reaction of corresponding nitriles 1a-o with hydroxylamine hydrochloride to give compounds 3–17. While most of these nitriles are commercially available, nitriles 1n and 1o had to be synthesized following Williamson reactions between 4-cyanophenol and methyl 4-(bromomethyl)benzoate, or methyl 3-(bromomethyl)benzoate, respectively (procedures reported in Materials and Methods).
Scheme 1.
General synthetic routes to access the 1,2,4-oxadiazole derivatives 3–17. Reagents and conditions: (a) NH2OH HCl, K2CO3 in CH3OH, reflux; (b) indole-4-carboxylic acid, DIPEA, HBTU in dry DMF, 140 °C.
The structure of the synthetized compounds was confirmed by analysis of the NMR data (1H and13C NMR spectra) and ESI-MS (see Materials and Methods).
2.2. Antimicrobial Activity
The new synthetized compounds (4–17) were tested for their antimicrobial activity against S. aureus ATCC 29213 and S. aureus ATCC 43300, and two Gram-negative strains, P. aeruginosa ATCC 27853 and K. pneumoniae ATCC 13883. The results are reported in Table 1. Compounds 6, and 11–14 showed the strongest antimicrobial activity against the two Staphylococcal strains, reporting MIC values of 6.25 µM. However, by further lowering the concentration, a more accurate estimate of the MIC value was obtained for compound 12 (2 µM). Compound 7 resulted active at 100 µM, while compounds 4, 8, 10, 16 and 17 were not active. None of the compounds were active against P. aeruginosa ATCC 27853 and K. pneumoniae (data not shown).
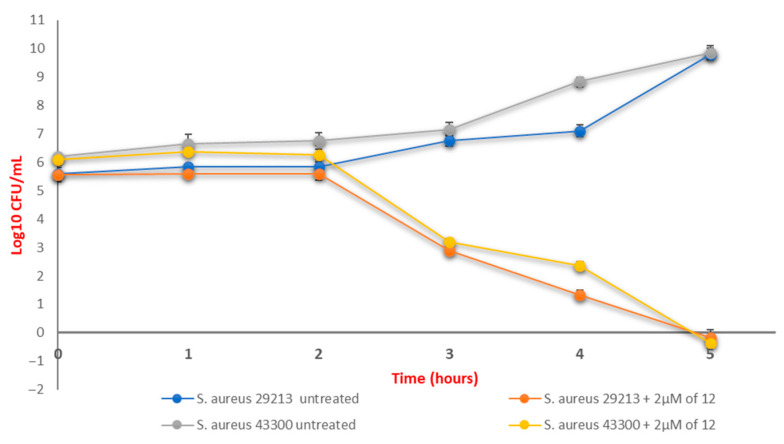
The minimum bactericidal concentration (MBC) resulted in 2 μM for compound 12, and 4 μM for compound 3. To confirm the bacteriostatic, or bactericidal activity, of 12, we performed the time kill assay on S. aureus ATCC 29213 and S. aureus ATCC 43300 at the MIC value (Figure 3) [18]. Compound 12 caused an over 3log10 -fold reduction in the bacterial count after 3 h, compared to initial inoculum, and completely abrogated the cell growth 24 h after.
Figure 3.
Time-kill assay of compound 12 against S. aureus ATCC 29213 and S. aureus ATCC 43300. Each experiment is the result of three independent experiments performed in triplicate.
2.3. Synergistic Study
The synergism between 12 and oxacillin against MRSA was determined using the checkerboard technique. The highest synergistic interaction was obtained with the combination values of 0.78 μM for compound 12, and 0.06 μg/mL for oxacillin. The FIC index value of 0.396 confirmed the synergistic effect of compound 12 and oxacillin.
2.4. Molecular Analysis
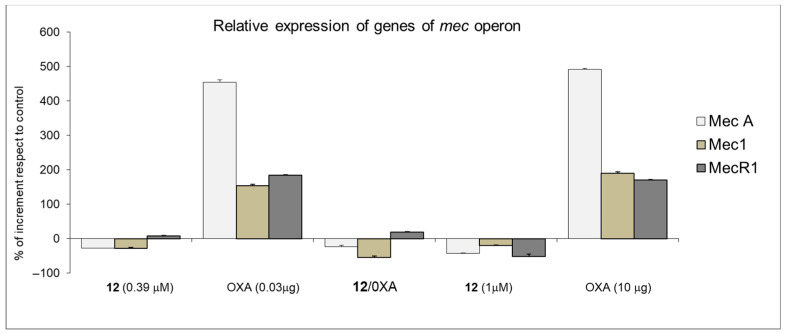
Here we investigated the effect of the compound 12 treatment on mecA, mec1, mecR1 gene expression modulation [19]. The treatment with the subinhibitory concentrations of 12 (1 µM and 0.39 μM), for 30 min, induced a reduced expression of all the mec operon genes, whereas oxacillin strongly induced them both at subinhibitory (0.03 µg/mL) and MIC values (10 µg/mL) (Figure 4). On the contrary, the synergic interaction between 12 and oxacillin, used at subinhibitory concentrations, counteracted the effect of oxacillin, restoring the levels of expression of all genes (mecA, mec1, and mecR1) to values comparable to 12-treated cells.
Figure 4.
Relative expression of the genes of mec operons. S. aureus ATCC 43300 was treated with subinhibitory concentrations of 12 (0.39 µM) and oxacillin (OXA) (0.03 µg/mL) for 30 min. Transcript levels were monitored by RT-PCR. The data are presented as the fold change in gene expression normalized to an endogenous reference gene (16 S) and relative to the untreated. Values represent the mean ± SD for three independent experiments.
2.5. Cytotoxicity Studies
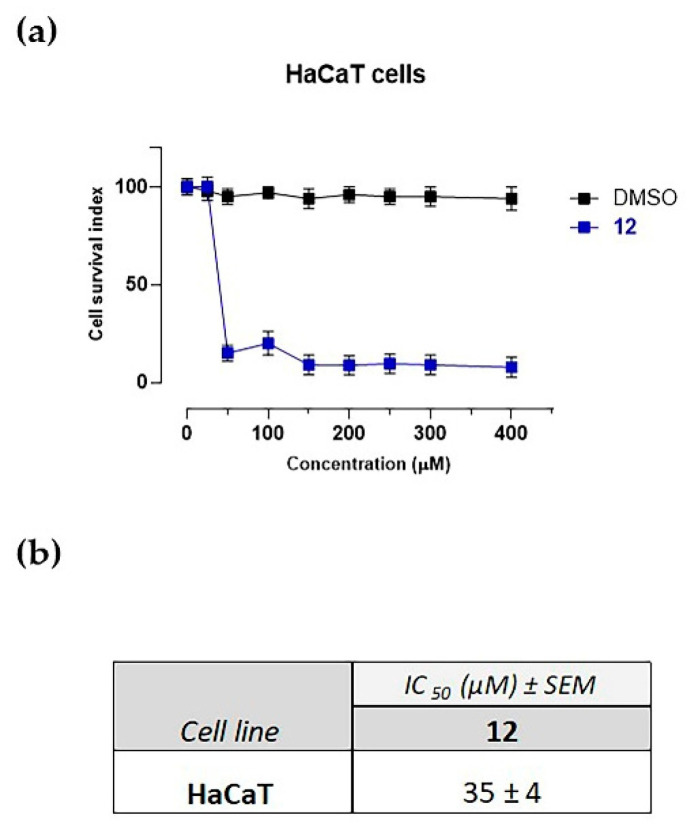
The cytotoxic properties of 12 were investigated using HaCaT keratinocytes as human model in vitro. The results show that compound 12 does not interfere with cell viability at lower concentrations (≤25 µM) after 48 h of incubation, while the cytotoxic activity of 12 on the HaCaT cell line at higher concentrations (≥35) was observed. The concentration–effect curve (Figure 5a)—here reported in terms of a “cell survival index” that combines the measurements of cell number and viability—shows a typical concentration-dependent sigmoid trend yielding IC50 values in the low micromolar range (Figure 5b). In order to exclude any effect given by the vehicle, DMSO was used as negative control. As reported in Figure 5a, there is no interfering with cell viability following incubation with DMSO, even at higher concentrations.
Figure 5.
(a) Cell survival index, evaluated by the MTT assay and monitoring of live/dead cell ratio relative for the compound 12 for the HaCaT cell line following 48 h of incubation with the indicated concentration (the range 1 → 400 μM), as indicated in the legend. Data are expressed as percentage of untreated control cells and are reported as mean of five independent experiments ± SEM (n = 30); (b) IC50 values (μM), relative to the compound 12 for the HaCaT cell line following 48 h of incubation. The IC50 values are reported as mean values ± SEM (n = 30).
3. Discussion
The oxadiazoles are a class of five-membered heterocycles that are of considerable interest in organic and medicinal chemistry [20]. In some bioactive compounds, they act as bioisosteres of ester and amide, with improved pharmacokinetic and pharmacodynamic profiles and enhanced selectivity; in other cases, the rigid heterocyclic ring acts as a flat aromatic spacer, assuring the appropriate substituent geometry. A vast number of oxadiazole-containing scaffolds are found to possess diverse biological activities, such as anticancer, anti-inflammatory, antiallergic, anticonvulsant, antiviral, antibacterial, anti-Alzheimer activities, as well as specific inhibitor properties against enzymes [21,22].
This study aims to further explore the chemical space around the oxadiazole ring. In particular, we have focused our attention on 1,2,4-oxadiazole-containing derivatives in order to find easily accessible chemical entities active against clinically relevant bacterial strains.
Starting from the lead compound 1, obtained by Chang and coworkers after an extensive SAR investigation of oxadiazole derivatives [11], and taking into account the promising MIC of the related structure 3, in the present study, we fixed the 4-indolyl system as ring A, and we varied the size and the functionalization of the aromatic right portion at the C-3 position of the oxadiazole ring. The SAR was established by an initial screening of the synthetic derivatives against two MSRA S. aureus strains. Among the fifteen synthesized compounds, two (3 and 12) displayed the best activity, and 12 was selected as the most promising for its ability to contrast the growth of S. aureus and the MRSA strain at an MIC value of 2 µM.
The MIC values reported were indicative of the marked influence of the substituents at C-3 of the oxadiazole ring on the antimicrobial activity. In particular, the most active compounds, 3 and 12, featured, respectively, 4-(3-thienyl)phenyl and 1,1′-biphenyl-4-yl substituents, namely, a para-biaryl unsubstituted system. No activity was observed for compound 4, with ring C meta-substituted with 3-thienyl ring. Moreover, the weaker activity of compound 3 (MIC 4 µM), as compared to compound 12 (MIC 2 µM), could be ascribed to the lesser degree of aromaticity of the heterocyclic thiophene in compound 3 vs. the phenyl moiety in compound 12.
The introduction of hydroxy groups on ring C, and the simultaneous absence of ring D in compounds 7 and 8, or the introduction of a benzyl group (10), dramatically decreased the antibacterial activity, resulting in MIC values ≥ 100 µM. The presence of the OH group at the orto position of ring C (compound 9) enhanced activity with an MIC of 25 µM. On the other hand, the strong electron-withdrawing group, CF3, enhanced the activity (6 vs. 5 and 11 vs. 10). The introduction of an oxymethylene ether spacer between rings C and D (compounds 16 and 17) caused the loss of antibacterial activity. Compounds 13 and 14, characterized by the introduction of hydroxy groups on the biphenyl ring, showed moderate activity, with an MIC of 6.25 µM, while compound 15 showed weaker activity (MIC 12.5 µM).
Of interest, previous studies have reported that this class of compounds shows antimicrobial activity against S. aureus at very low concentrations (values ranging from 1 to 2 µg/mL) [13,23]. However, compound 12 resulted in activity at a lower concentration (2 µM corresponds to 0.674 µg/mL), representing the most active compound so far developed within the oxadiazole class. Therefore, the conjugated para-biphenyl system in 12 appears more effective than the para-diphenyl ether moiety in compounds 1 and 2. Moreover, compound 12 was not toxic at concentrations less than 25 µM, and strongly contrasted the effect of oxacillin when used in combination. The FIC index demonstrated the synergic activity of 12 with oxacillin. The latter result is of interest since antibiotic research worldwide is focused on the discovery of new compounds that act synergically with the old class of unutilized antibiotic, because of the lack of activity against multidrug-resistant bacteria, but which are well-known for their pharmacokinetic and safety parameters [24]. Repurposing existing antibiotics would immediately respond to clinical requests, as these drugs have long been administered and are already approved for human use. In this context, programs and interventions aiming at optimizing the use of antimicrobial drugs, termed “antimicrobial stewardship”, have been welcome [25]. The clinical resistance to β-lactam antibiotics by MRSA occurs because of the acquisition of the mecA gene, contained in the staphylococcal cassette chromosome mec (SCCmec), which encodes a cell-wall DD-transpeptidase that is not inhibited by the latter antibiotics [26,27]. Bacteria are, thus, not deprived of the final stages of cell-wall biosynthesis, and they can grow and replicate undisturbed.
The regulation of mecA expression is controlled by its own regulators, mecR1 and mecI, also carried on the SCCmec element. MecA transcription is so repressed by mec1, and only the presence of β-lactam antibiotics, detected by the sensory domains in MecR1, removes the repression of mecA transcription by MecI, leading to mecA transcription, PBP2a production, and the expression of methicillin resistance [28]. The results here demonstrate that the treatment of MRSA with compound 12 induces a decreased expression of the genes included in the mec operon. More interestingly, 12 contrasts the protective response of bacteria to oxacillin treatment, namely, the strong induction of the mecA gene and the related genes of the operon, resensitizing MRSA to beta-lactam antibiotics. The ability of oxadiazole to inhibit PBP2a is known [10]. We can speculate that compound 12 displays the same effect, inhibiting the activity of the enzyme. It can, perhaps, bind to the MecR1 domain competing with oxacillin binding, thus inhibiting the transcription of the mec operon genes. Further studies are necessary in order to ascertain the hypothesis here formulated.
Furthermore, cytotoxicity studies performed by bioscreens in vitro revealed an activity of 12 in inhibiting cell growth and proliferation only at higher concentrations.
4. Materials and Methods
4.1. Materials
All chemical reagents are commercially available from Sigma Aldrich® (Milan, Italy) and TCI (Tokyo, Japan). Solvents and reagents were used as supplied from commercial sources, with the exception of methanol, which was anhydrified from magnesium methoxide as follows: Magnesium turnings (5 g) and iodine (0.5 g) were refluxed in a small amount of methanol (50–100 mL), until all the magnesium was reacted. The mixture was diluted (up to 1 L) with methanol, refluxed for 2–3 h, and then distilled under argon. All reactions were carried out under argon atmosphere using flame-dried glassware.
The reaction progress was monitored via thin-layer chromatography (TLC) on Alugram silica gel G/UV254 plates. The C-18 Phenomenex Luna column was used as a chromatography column. The silica gel, MN Kiesel gel 60 (70–230 mesh) of Macherey-Nagel, was used for flash chromatography. HPLC was performed using a Waters Model 510 pump equipped with a Waters Rheodyne injector and a differential refractometer, model 401.The purities of the compounds were estimated to be greater than 95% by HPLC.
NMR spectra were recorded on Bruker Avance NEO 400 and 700 spectrometers equipped with an RT-DR-BF/1H-5 mm-OZ SmartProbe (1H at 400 MHz and 13C at 100 MHz; 1H at 700 MHz and 13C at 175 MHz).
Coupling constants (J) are given in Hertz (Hz), chemical shifts were reported in δ (ppm), and referred to the residual CH3OD and CHCl3 as internal standards (δH = 3.31 and δC = 49.0 ppm; δH = 7.26 and δC = 77.0 ppm). All the recorded signals were in accordance with the proposed structures. Spin multiplicities are given as s (singlet), br s (broad singlet), d (doublet), or m (multiplet). ESI-MS analysis was carried out on a mass spectrometer, LTQ-XL.
4.2. Synthesis
4.2.1. Synthetic Procedures for Nitriles 1n and 1o
Methyl 4-(bromomethyl)benzoate and methyl 3-(bromomethyl)benzoate (1.2 mol eq.) were added to a solution of 4-cyanophenol (1 mol eq.) and K2CO3 (2 mol eq.), respectively, in DMF dried at 100 °C. The reaction was stirred at 25 °C overnight, then fractionated in water and ethyl acetate three times. The organic layers were dried over anhydrous Na2SO4, and filtrated and concentrated under vacuum to obtain the correspondent nitriles, methyl 4-[(4-cyanobenzyl)oxy]benzoate (1n) and methyl 3-[(4-cyanobenzyl)oxy]benzoate (1o).
4.2.2. General Synthetic Procedures for Amidoximes 2a-o
Each nitrile (1 mol eq.) was dissolved in dry methanol and potassium carbonate (1.5 mol eq.), and hydroxylamine hydrochloride (2.5 mol eq.) was added to the solution. The mixture was refluxed for 8 h in inert atmosphere. The reaction was concentrated under vacuum to remove MeOH, diluted with water, and extracted three times with CH2Cl2. The organic phases were dried over anhydrous Na2SO4, filtered, and concentrated under vacuum to obtain the correspondent amidoximes (yield 70–90%). The product was subjected to the next steps without any purification.
4.2.3. Synthetic procedures for compounds 3–17
Indole-4-carboxylic acid (1.2 mol eq.) was dissolved in dry DMF and DIPEA (1.8 mol eq.), and the coupling reagent, HBTU (1.5 mol eq.), was added to the solution. Amidoximes 2a-o (1 mol eq.) were added ten minutes later in the corresponding reactions. The mixture was stirred at 140° C for 12 h, then fractionated in water and ethyl acetate three times. The organic layers were cooled and washed three times with a saturated solution of LiBr, and then with a saturated solution of NaHCO3 and distilled water. The organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure obtaining the oxadiazoles 3–17. The crude reactions were purified as follows:
5-(1H-indol-4-yl)-3-(4-(3-thienyl)phenyl)-1,2,4-oxadiazole (3). NMR data as previously reported [17].
5-(1H-indol-4-yl)-3-(3-(3-thienyl)phenyl)-1,2,4-oxadiazole (4). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (80:20) as eluent (flow rate 3.00 mL/min), affording compound 4 (tR = 9.0 min). 1H NMR (700 MHz, CD3OD): δH 8.20 (1H, s), 7.99 (1H, d, J = 7.4 Hz), 7.83 (1H, d, J = 7.8 Hz), 7.77 (1H, m), 7.75 (1H, d, J = 7.8 Hz), 7.69 (1H, d, J = 7.8 Hz), 7.58 (1H, dd, J = 5.0, 1.2 Hz), 7.51 (1H, d, J = 3.1 Hz), 7.45 (1H, d, J = 3.1 Hz), 7.25 (1H, t, J = 7.8 Hz) 7.13 (1H, d, J = 3.0 Hz); 13C NMR (175 MHz, CD3OD): δC 167.8, 160.0, 142.8, 138.7, 137.7, 133.6, 130.3, 129.8, 129.1, 128.4, 127.6, 127.3, 126.8, 126.4, 123.8, 122.2, 121.6, 121.1, 117.8, 103.5. ESI-MS m/z 344.2 [M + H]+.
5-(1H-indol-4-yl)-3-phenyl-1,2,4-oxadiazole (5). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (80:20) as eluent (flow rate 3.00 mL/min), affording compound 5 (tR = 21.0 min). 1H NMR (700 MHz, CD3OD): δH 8.20 (2H, d, J = 7.7 Hz), 8.01 (1H, d, J = 7.4 Hz), 7.71 (1H, d, J = 7.8 Hz), 7.56 (3H, ovl), 7.51 (1H, d, J = 3.1 Hz), 7.31 (1H, t, J = 7.8 Hz), 7.28 (1H, d, J = 3.1 Hz); 13C NMR (175 MHz, CD3OD): δC 178.7, 170.1, 138.7, 132.5, 130.4 (3C), 128.9, 128.6 (2C), 127.6, 122.4, 122.2, 117.4, 116.1, 103.6. ESI-MS m/z 262.1 [M + H]+.
5-(1H-indol-4-yl)-3-(4-(trifluoromethyl)phenyl)-1,2,4-oxadiazole (6). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (90:10) as eluent (flow rate 3.00 mL/min), affording compound 6 (tR = 17.5 min). 1H NMR (700 MHz, CD3OD): δH 8.29 (2H, d, J = 8.0 Hz), 7.95 (1H, d, J = 7.7 Hz), 7.79 (2H, d, J = 8.0 Hz), 7.67 (1H, d, J = 7.7 Hz), 7.48 (1H, d, J = 3.0 Hz), 7.26 (1H, t, J = 7.8 Hz) 7.24 (1H, d, J = 3.0 Hz); 13C NMR (175 MHz, CD3OD): δC 178.5, 165.7, 138.5, 133.5 (q, J = 32.7 Hz), 132.2, 128.9 (2C), 128.7, 127.4, 126.8 (2C, q, J= 3.5 Hz), 125.4 (q, J = 271.2 Hz), 122.2, 121.9, 117.6, 115.3, 103.3. ESI-MS m/z 330.1 [M + H]+.
5-(1H-indol-4-yl)-3-(4-(hydroxy)phenyl)-1,2,4-oxadiazole (7). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (80:20) as eluent (flow rate 3.00 mL/min), affording compound 7 (tR = 10.0 min). 1H NMR (400 MHz, CD3OD): δH 8.03 (2H, d, J = 8.7 Hz), 7.99 (1H, d, J = 7.8 Hz), 7.70 (1H, d, J = 7.8 Hz), 7.51 (1H, d, J = 3.1 Hz), 7.30 (1H, t, J = 7.8 Hz) 7.26 (1H, d, J = 3.1 Hz), 6.94 (2H, d, J = 8.7 Hz); 13C NMR (100 MHz, CD3OD): δC 178.0, 169.8, 161.7, 138.5, 130.2 (2C), 130.1, 128.5, 127.4, 122.0 (2C), 119.5, 117.3, 116.7, 116.0, 103.35. ESI-MS m/z 278.1 [M + H]+.
5-(1H-indol-4-yl)-3-(3-(hydroxy)phenyl)-1,2,4-oxadiazole (8). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (80:20) as eluent (flow rate 3.00 mL/min), affording compound 8 (tR = 11.5 min). 1H NMR (700 MHz, CD3OD): δH 8.00 (1H, d, J = 7.6 Hz), 7.71 (1H, d, J = 8.0 Hz), 7.67 (1H, d, J = 7.6 Hz), 7.64 (1H, d, J = 1.6 Hz), 7.51 (1H, d, J = 3.0 Hz), 7.37 (1H, t, J = 8.0 Hz), 7.31 (1H, t, J = 7.6 Hz), 7.28 (1H, d, J = 3.0 Hz), 6.97 (1H, dd, J = 8.0, 1.6 Hz); 13C NMR (175 MHz, CD3OD): δC 178.3, 169.9, 159.2, 138.5, 131.1, 129.7, 128.6, 127.4, 122.1, 122.0, 119.6, 119.3, 117.4, 115.9, 115.1, 103.3. ESI-MS m/z 278.1 [M + H]+.
5-(1H-indol-4-yl)-3-(2-(hydroxy)phenyl)-1,2,4-oxadiazole (9). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (80:20) as eluent (flow rate 3.00 mL/min), affording compound 9 (tR = 19.0 min). 1H NMR (400 MHz, CD3OD): δH 8.14 (1H, d, J = 7.7 Hz), 8.05 (1H, d, J = 7.4 Hz), 7.76 (1H, d, J = 8.0 Hz), 7.56 (1H, d, J = 3.0 Hz), 7.45 (1H, t, J = 8.0 Hz), 7.35 (1H, t, J = 7.7 Hz), 7.22 (1H, d, J = 3.0 Hz), 7.07 (1H, d, J = 8.0 Hz) 7.05 (1H, d, J = 7.7 Hz); 13C NMR (100 MHz, CD3OD): δC 177.1, 168.6, 158.4, 138.6, 134.1, 129.6, 129.0, 127.3, 122.6, 122.0, 120.9, 118.4, 118.0, 115.1, 112.6, 103.0. ESI-MS m/z 278.1 [M + H]+.
3-benzyl-5-(1H-indol-4-yl)-1,2,4-oxadiazole (10). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (80:20) as eluent (flow rate 3.00 mL/min), affording compound 10 (tR = 13.5 min). 1H NMR (400 MHz, CD3OD): δH 7.92 (1H, dd, J = 7.5, 0.8 Hz), 7.70 (1H, d, J = 8.0 Hz), 7.49 (1H, d, J = 3.2 Hz), 7.43 (2H, d, J = 7.5 Hz), 7.36 (2H, t, J = 7.5 Hz), 7.33 (2H, ovl), 7.15 (1H, dd, J = 3.2, 0.8 Hz); 13C NMR (175 MHz, CD3OD): δC 178.5, 171.3, 138.6, 137.5, 130.2 (2C), 128.9 (2C), 128.7, 128.1, 127.4, 122.2, 122.0, 117.5, 115.9, 103.2, 33.0. ESI-MS m/z 276.1 [M + H]+.
5-(1H-indol-4-yl)-3-(4-(trifluoromethyl)benzyl)-1,2,4-oxadiazole (11). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (80:20) as eluent (flow rate 3.00 mL/min), affording compound 11 (tR = 18.0 min). 1H NMR (700 MHz, CD3OD): δH 7.90 (1H, d, J = 7.5 Hz), 7.68 (2H, d, J = 8.0 Hz), 7.65 (2H, d, J = 8.0 Hz), 7.61 (2H, d, J = 8.0 Hz), 7.47 (1H, d, J = 3.1 Hz), 7.26 (1H, t, J = 7.8 Hz) 7.13 (1H, d, J = 3.1 Hz), 4.28 (2H, s); 13C NMR (175 MHz, CD3OD): δC 178.6, 170.6, 142.0, 138.5, 130.8 (2C), 130.3 (q, J = 32.2 Hz), 128.7, 127.3, 127.0 (2C, q, J = 3.7 Hz), 126.1 (q, J = 271 Hz), 122.1, 121.9, 117.5, 115.7, 103.1, 32.9. ESI-MS m/z 344.1 [M + H]+.
3-([1,1′-biphenyl]-4-yl)-5-(1H-indol-4-yl)-1,2,4-oxadiazole (12). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (80:20) as eluent (flow rate 3.00 mL/min), affording compound 12 (tR = 10.0 min). 1H NMR (700 MHz, CD3OD): δH 8.27 (2H, d, J = 8.3 Hz), 8.03 (1H, d, J = 7.4 Hz), 7.82 (2H, d, J = 8.3 Hz), 7.71 (3H, ovl), 7.53 (1H, d, J = 3.0 Hz), 7.48 (2H, t, J = 7.4 Hz), 7.39 (1H, t, J = 7.4 Hz), 7.32 (1H, d, J = 7.4 Hz), 7.30 (1H, d, J = 3.1 Hz); 13C NMR (175 MHz, CD3OD): δC 178.4, 169.7, 145.3, 141.3, 138.4, 130.0 (2C), 129.0, 128.9 (2C), 128.5, 128.4 (2C), 128.0 (2C), 127.3 (2C), 122.1, 121.9,117.4, 115.8, 103.3. ESI-MS m/z 338.2 [M + H]+.
3-(4′-hydroxy-[1,1′-biphenyl]-4-yl)-5-(1H-indol-4-yl)-1,2,4-oxadiazole (13). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (85:15) as eluent (flow rate 3.00 mL/min), affording compound 13 (tR = 11.5 min). 1H NMR (400 MHz, CD3OD): δH 8.23 (2H, d, J = 8.5 Hz), 8.03 (1H, d, J = 7.5 Hz), 7.77 (2H, d, J = 8.5 Hz), 7.72 (1H, d, J = 8.0 Hz), 7.57 (2H, d, J = 8.5 Hz), 7.53 (1H, d, J = 3.1 Hz), 7.33 (1H, t, J = 7.8 Hz), 7.30 (1H, d, J = 3.1 Hz), 6.90 (2H, dd, J = 8.5 Hz); 13C NMR (100 MHz, CD3OD): δC 178.8, 170.2, 159.4, 145.7, 139.0, 133.0, 129.6 (2C), 129.3 (2C), 129.1, 128.2 (2C), 127.8, 126.8, 122.5, 122.4, 117.8, 117.2 (2C), 116.4, 103.6. ESI-MS m/z 354.1 [M + H]+.
3-(3′-hydroxy-[1,1′-biphenyl]-4-yl)-5-(1H-indol-4-yl)-1,2,4-oxadiazole (14). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (85:15) as eluent (flow rate 3.00 mL/min), affording compound 14 (tR = 12.0 min). 1H NMR (700 MHz, CD3OD): δH 8.22 (2H, d, J = 8.2 Hz), 8.01 (1H, d, J = 7.3 Hz), 7.75 (2H, d, J = 8.2 Hz), 7.70 (1H, d, J = 8.0 Hz), 7.51 (1H, d, J = 3.0 Hz), 7.28 (3H, ovl), 7.13 (1H, d, J = 3.0 Hz), 7.12 (1H, d, J = 1.8 Hz), 6.82 (1H, dd, J = 8.0, 1.8 Hz); 13C NMR (175 MHz, CD3OD): δC 178.3, 169.6, 159.5, 145.3, 142.8, 138.5, 131.0, 128.8 (2C), 128.6, 128.4 (2C), 127.4, 127.3, 122.1, 122.0, 119.1, 117.4, 116.2, 115.9, 115.0, 103.4. ESI-MS m/z 354.1 [M + H]+.
3-(3′-hydroxy-[1,1′-biphenyl]-3-yl)-5-(1H-indol-4-yl)-1,2,4-oxadiazole (15). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (85:15) as eluent (flow rate 3.00 mL/min), affording compound 15 (tR = 12.0 min). 1H NMR (700 MHz, CD3OD): δH 8.39 (1H, s), 8.12 (1H, d, J = 7.7 Hz), 8.02 (1H, d, J = 7.5 Hz), 7.77 (1H, d, J = 7.9 Hz), 7.70 (1H, d, J = 7.9 Hz), 7.59 (1H, t, J = 7.7 Hz), 7.51 (1H, d, J = 3.0 Hz), 7.30 (1H, t, J = 7.7 Hz), 7.29 (1H, d, J = 3.0 Hz), 7.24 (1H, d, J = 7.9 Hz), 7.09 (1H, d, J = 1.8 Hz), 7.04 (1H, d, J = 7.5 Hz), 6.78 (1H, dd, J = 8.0, 1.8 Hz); 13C NMR (175 MHz, CD3OD): δC 178.1, 169.7, 161.9, 143.6, 142.4, 138.3, 130.6, 130.5, 130.1, 128.6, 128.4, 127.1, 126.6, 126.5, 121.9, 121.7, 117.3, 117.1, 116.8, 116.0, 115.6, 103.1. ESI-MS m/z 354.1 [M + H]+.
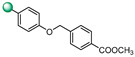
5-(1H-indol-4-yl)-3-(4-((4-methoxycarbonylbenzyl)oxy)phenyl)-1,2,4-oxadiazole (16). An analytic sample of crude reaction was purified by HPLC using a Luna Column C-18 (10 µm, 250 mm × 10 mm), with MeOH/H2O (88:12) as eluent (flow rate 3.00 mL/min), affording compound 16 (tR = 14.0 min). 1H NMR (400 MHz, CDCl3): δH 8.47 (1H, br s, NH), 8.21 (2H, d, J = 8.8 Hz), 8.10 (3H, ovl), 7.66 (1H, d, J = 8.0 Hz), 7.55 (2H, d, J = 8.1 Hz), 7.46 (2H, d, J = 2.2 Hz), 7.36 (1H, t, J = 8.0 Hz), 7.12 (2H, d, J = 8.8 Hz), 5.23 (2H, s), 3.95 (3H, s); 13C NMR (175 MHz, CDCl3): δC 176.1, 168.3, 166.8, 160.6, 141.7, 136.5, 130.0 (2C), 129.8, 129.2 (2C), 127.0 (2C), 126.6, 126.0, 121.9, 121.8, 120.4, 115.8, 115.6, 115.1 (2C), 103.6, 69.6, 52.4. ESI-MS m/z 426.1 [M + H]+.
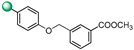
5-(1H-indol-4-yl)-3-(4-((3-methoxycarbonylbenzyl)oxy)phenyl)-1,2,4-oxadiazole (17). The crude reaction was purified by flash chromatography using a gradient from 9:1 to 8:2 Hexane/Ethyl Acetate affording 30 mg of compound 17 (54% yield). 1H NMR (400 MHz, CDCl3): δH 8.54 (1H, s, NH), 8.22 (2H, d, J = 8.7 Hz), 8.17 (1H, s), 8.12 (1H, d, J = 7.6 Hz), 8.06 (1H, d, J = 7.6 Hz), 7.70 (2H, ovl), 7.52 (2H, ovl), 7.38 (1H, t, J = 7.6 Hz), 7.30 (1H, s), 7.13 (2H, d, J = 8.7 Hz), 5.22 (2H, s), 3.96 (3H, s); 13C NMR (100 MHz, CDCl3): δC 175.5, 168.4, 166.3, 160.5, 137.0, 136.5, 131.9, 130.6, 129.4, 129.2 (2C), 128.7, 128.6, 126.6, 126.0, 121.8 (2C), 120.4, 115.8, 115.5, 115.1 (2C), 103.8, 69.3, 52.1. ESI-MS m/z 426.1 [M + H]+.
4.3. Antibiotics and Strains
Vancomycin and oxacillin were purchased from Sigma-Aldrich (Milan, Italy). Staphylococcus aureus ATCC 43300 (a methicillin-resistant strain characterized by the presence of the mec operon), S. aureus ATCC 29213, P. aeruginosa ATCC 27853, and K. pneumoniae ATCC 13883 were obtained from the American Type Culture Collection (Rockville, MD, USA).
4.4. Antimicrobial Susceptibility Testing
The minimal inhibitory concentrations (MIC) of all the compounds were determined in Mueller–Hinton medium (MH) by the broth microdilution assay, following the procedure already described [29]. The compounds were added to bacterial suspension in each well, yielding a final cell concentration of 1 × 106 CFU/mL, and a final compound concentration ranging from 1.56 to 100 μM. Compounds 3 and 12 were further tested at the final compound concentrations of 1, 2, 4, and 8 μM. Negative control wells were set to contain bacteria in Mueller–Hinton broth plus the amount of vehicle (DMSO) used to dilute each compound. Positive controls included vancomycin (2 μg/mL) and oxacillin (2 and 10 μg/mL). The MIC was defined as the lowest concentration of the drug that caused a total inhibition of microbial growth after a 24 h incubation time at 37 °C. Medium turbidity was measured by a microtiter plate reader (Biorad mod 680, Milan, Italy) at 595 nm. Minimum bactericidal concentration (MBC) was defined as the lowest test concentration that kills the organism (exhibited no growth on agar plates) after 24 h of incubation at 37 °C.
4.5. Killing Rate
Bacterial suspension (105 CFU/mL) was added to the microplates along with compound 12 at the MIC value. The plates were incubated at 37 °C on an orbital shaker at 120 rpm. Viability assessments were performed at 0, 2, 4, 6, and 24 h by plating 0.01 mL undiluted, and 10-fold serially diluted, samples onto Mueller–Hinton plates in triplicate. After the overnight incubation at 37 °C, the bacterial colonies were counted and compared with the counts from the control cultures [30].
4.6. Checkerboard Method
The interaction between compound 12 and oxacillin against MRSA was evaluated by the checkerboard method in 96-well microtiter plates containing Mueller–Hinton broth. Briefly, compound 12 and oxacillin were serially diluted along the y and x axes, respectively. The final concentration ranged from 0.03 to 10 µg/mL for oxacillin, and from 0.5 to 3.12 µM (0.5, 0.78, 1, 1.56, 2, 3.12 µM) for 12. The checkerboard plates were inoculated with bacteria at an approximate concentration of 105 × CFU/mL, and incubated at 37 °C for 24 h, following which the bacterial growth was assessed visually and the turbidity measured by microplate reader at 595 nm. The FIC index for each combination was calculated as follows: FIC index = FIC of 12 + FIC of oxacillin, where FIC of 12 (or oxacillin) was defined as the ratio of the MIC of 12 (or oxacillin) in combination, and the MIC of 12 (or oxacillin) alone. The FIC index values were interpreted as follows: ≤0.5, synergistic; >0.5 to <2.0, additive; >1.0 to ≤2.0, indifferent; and >2.0, antagonistic effects [31].
4.7. Molecular Analysis
RNA extraction was performed by using the GenUp Total RNA kit (BiotechRabbit), according to the manufacturer’s instructions. Five hundred nanograms of total cellular RNA were reverse-transcribed (RevertUP II Reverse Transcriptase, BiotechRabbit) into cDNA, using random hexamer primers (Random hexamer, Roche Diagnostics, Germany), at 48 °C for 60 min, according to the manufacturer’s instructions. RT-PCR was carried out using: 2 μL of cDNA amplified in a reaction mixture containing 10 mM Tris–HCl (pH 8.3); 1.5 mM MgCl2; 50 mM KCl; 10 µM dNTP; 10 µM forward and reverse primers (mecA, mec1, mecR1), or 1 µM forward and reverse 16S rRNA primers; and 2.5 U of Taq DNA polymerase (BiotechRabbit) in a final volume of 25 µL. The cycling conditions are reported in Table 2. The reaction was carried out in a DNA thermal cycler (MyCycler, Biorad, USA). The PCR products were analyzed by electrophoresis on 1.8% agarose gel in TBE, and analyzed on a Gel Doc EZ System (BioRad). Quantification data were normalized to the reference gene for the 16S rRNA gene and analyzed by Image Lab software 5.2.1 (BioRad).
Table 2.
Staphylococcal sense and antisense primer sequences and expected PCR products (bp: base pairs).
| Gene | Sense and Antisense Sequences | Conditions | bp |
|---|---|---|---|
| mecA | 5′–TCCACCCTCAAACAGGTGAA-3′ 5′-TGGAACTTGTTGAGCAGAGGT-3′ |
95 °C for 5′ 94 °C for 30′’, 55 °C for 30′’, 72 °C for 30′’ for 33 cycles 72 °C for 7′ |
139 |
| mecI | 5′-TCATCTGCAGAATGGGAAGTT-3′ 5′-TTGGACTCCAGTCCTTTTGC-3′ |
103 | |
| mecR1 | 5′-AGCACCGTTACTATCTGCACA-3′ 5′-AGAATAAGCTTGCTCCCGTTCA-3′ |
142 | |
| rRNA16S | 5′-CGGTCCAGACTCCTACGGGAGGCAGCA-3′ 5′-GCGTGGACTACCAGGGTATCTAATCC-3′ |
450 |
4.8. Bioscreens In Vitro for Cytotoxicity Studies
For the cytotoxicity studies, human HaCaT keratinocytes were grown in DMEM (Invitrogen, Paisley, UK), supplemented with 10% fetal bovine serum (FBS, Cambrex, Verviers, Belgium), L-glutamine (2 mM, Sigma, Milan, Italy), penicillin (100 units/mL, Sigma), and streptomycin (100 μ g/mL, Sigma), and cultured in a humidified 5% carbon dioxide atmosphere at 37 °C, according to ATCC recommendations. The cells were inoculated and allowed to grow for 24 h in 96-microwell culture plates at a density of 104 cells/well. The medium was then replaced with fresh medium and the cells were treated for a further 48 h with a range of concentrations (1 → 400 μM) of 12. Using the same experimental procedure, the cell cultures were also incubated with vehicle DMSO as negative control. The cytotoxic activity of compound 12 was investigated through the estimation of a “cell survival index”, arising from the combination of the cell viability evaluation with cell counting [32]. Cell viability was evaluated using the MTT assay procedure, which measures the level of mitochondrial dehydrogenase activity using yellow 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT, Sigma) as substrate. Briefly, after the treatments, the medium was removed, and the cells were incubated with 20 μL/well of an MTT solution (5 mg/mL) for 1 h in a humidified 5% CO2 incubator at 37 °C. The incubation was stopped by removing the MTT solution, and by adding 100 μL/well of DMSO to solubilize the obtained formazan. Finally, the absorbance was monitored at 550 nm using a microplate reader (iMark microplate reader, Bio-Rad, Milan, Italy). The cell number was determined by a TC20 automated cell counter (Bio-Rad, Milan, Italy), providing an accurate and reproducible total count of the cells, and a live/dead ratio in one step by a specific dye (trypan blue) exclusion assay. Bio-Rad’s TC20 automated cell counter uses disposable slides and TC20 trypan blue dye (0.4% trypan blue dye w/v in 0.81% sodium chloride and 0.06% potassium phosphate dibasic solution). Once the loaded slide is inserted into the slide port, the TC20 automatically focuses on the cells and detects the presence of the trypan blue dye, providing the count. The calculation of the concentration required to inhibit the net increase in the cell number and viability by 50% (IC50) is based on plots of data (n = 6 for each experiment) and repeated five times (total n = 30). IC50 values were obtained by means of a concentration response curve by nonlinear regression using a curve fitting program, GraphPad Prism 8.0, and are expressed as the mean values ± SEM (n = 30) of the five independent experiments.
5. Conclusions
In summary, we have described the discovery of new oxadiazole derivatives as antibiotics active against methicillin-resistant Staphylococcus aureus. This study has resulted in the identification of compounds 3 and 12 as the most active of the series. Moreover, we demonstrated that compound 12 works like an antibiotic resistance breaker (ARB), restoring the activity of oxacillin inducing the expression of the genes included in the mec operon.
This compound can increase the effectiveness of β-lactam antibiotics by combatting the resistance mechanisms employed against them.
Acknowledgments
The authors would like to acknowledge Adriana De Rosa for her technical assistance.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/antibiotics10101258/s1.
Author Contributions
E.B., C.F., S.D.M., and M.V.D. designed the study, and drafted and wrote the manuscript; M.S. and C.F. performed the synthesis; S.D.M. and C.F. performed the NMR experiments; E.B. performed antimicrobial analysis; M.P. conducted the cytotoxicity studies. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by MIUR Italy PRIN 2017 project (2017A95NCJ) “Stolen molecules—Stealing natural products from the depot and reselling them as new drug candidate”.
Data Availability Statement
All data are included in the article or uploaded as supplementary data.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Tong S.Y., Davis J.S., Eichenberger E., Holland T.L., Fowler V.G., Jr. Staphylococcus aureus infections: Epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 2015;28:603–661. doi: 10.1128/CMR.00134-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wu S.C., Liu F., Zhu K., Shen J.Z. Natural products that target virulence factors in antibiotic-resistant Staphylococcus aureus. J. Agric. Food Chem. 2019;67:13195–13211. doi: 10.1021/acs.jafc.9b05595. [DOI] [PubMed] [Google Scholar]
- 3.Gajdács M. The continuing threat of methicillin-resistant Staphylococcus aureus. Antibiotics. 2019;8:52. doi: 10.3390/antibiotics8020052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Guzzo F., Scognamiglio M., Fiorentino A., Buommino E., D’Abrosca B. Plant derived natural products against Pseudomonas aeruginosa and Staphylococcus aureus: Antibiofilm activity and molecular mechanisms. Molecules. 2020;25:5024. doi: 10.3390/molecules25215024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Laupland K.B., Lyytikainen O., Sogaard M., Kennedy K.J., Knudsen J.D., Ostergaard C., Galbraith J.C., Valiquette L., Jacobsson G., Collignon P., et al. The changing epidemiology of Staphylococcus aureus bloodstream infection: A multinational population-based surveillance study. Clin. Microbiol. Infect. 2013;19:465–471. doi: 10.1111/j.1469-0691.2012.03903.x. [DOI] [PubMed] [Google Scholar]
- 6.Chambers H.F., Deleo F.R. Waves of resistance: Staphylococcus aureus in the antibiotic era. Nat. Rev. Microbiol. 2009;7:629–641. doi: 10.1038/nrmicro2200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dalvie D.K., Kalgutkar A.S., Khojasteh-Bakht S.C., Obach R.S., O’Donnell J.P. Biotransformation reactions of five-membered aromatic heterocyclic rings. Chem. Res. Toxicol. 2002;15:269–299. doi: 10.1021/tx015574b. [DOI] [PubMed] [Google Scholar]
- 8.Biernacki K., Daśko M., Ciupak O., Kubiński K., Rachon J., Demkowicz S. Novel 1,2,4-oxadiazole derivatives in drug discovery. Pharmaceuticals. 2020;13:111. doi: 10.3390/ph13060111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chawla G. 1,2,4-Oxadiazole as a privileged scaffold for anti-inflammatory and analgesic activities: A review. Mini Rev. Med. Chem. 2018;18:1536–1547. doi: 10.2174/1389557518666180524112050. [DOI] [PubMed] [Google Scholar]
- 10.Ceballos S., Kim C., Ding D., Mobashery S., Chang M., Torresa C. Activities of oxadiazole antibacterials against Staphylococcus aureus and other gram-positive bacteria. Antimicrob. Agents Chemother. 2018;62:e00453-18. doi: 10.1128/AAC.00453-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.O’Daniel P.I., Pi Z.P.H., Testero S.A., Ding D., Spink E., Leemans E., Boudreau M.A., Yamaguchi T., Schroeder V.A., Wolter W.R., et al. Discovery of a new class of non-β-lactam inhibitors of penicillin binding proteins with Gram-Positive antibacterial activity. J. Am. Chem. Soc. 2014;136:3664–3672. doi: 10.1021/ja500053x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Spink E., Ding D., Peng Z., Boudreau M.A., Leemans E., Lastochkin E., Song W., Lichtenwalter K., O’Daniel P.I., Testero S.A., et al. Structure−activity relationship for the oxadiazole class of antibiotics. J. Med. Chem. 2015;58:1380–1389. doi: 10.1021/jm501661f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Boudreau M.A., Ding D., Meisel J.E., Janardhanan J., Spink E., Peng Z., Qian Y., Yamaguchi T., Testero S.A., O’Daniel P.I., et al. Structure–Activity relationship for the oxadiazole class of antibacterials. ACS Med. Chem. Lett. 2020;11:322–326. doi: 10.1021/acsmedchemlett.9b00379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ding D., Boudreau M.A., Leemans E., Spink E., Yamaguchi T., Testero S.A., O’Daniel P.I., Lastochkin E., Chang M. Mobashery, S. Exploration of the structure-activity relationship of 1,2,4-oxadiazole antibiotics. Bioorg. Med. Chem. Lett. 2015;25:4854–4857. doi: 10.1016/j.bmcl.2015.06.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Di Leva F.S., Festa C., Carino A., De Marino S., Marchianò S., Di Marino D., Finamore C., Monti M.C., Zampella A., Fiorucci S., et al. Discovery of ((1,2,4-oxadiazol-5-yl) pyrrolidin-3-yl)ureidyl derivatives as selective non-steroidal agonists of the G-protein coupled bile acid receptor-1. Sci. Rep. 2019;9:2504. doi: 10.1038/s41598-019-38840-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Festa C., Finamore C., Marchiano S., Di Leva F.S., Carino A., Monti M.C., del Gaudio F., Ceccacci S., Limongelli V., Zampella A., et al. Investigation around the oxadiazole core in the discovery of a new chemotype of potent and selective FXR antagonists. ACS Med. Chem. Lett. 2019;10:504–510. doi: 10.1021/acsmedchemlett.8b00534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Potenza M., Sciarretta M., Chini M.G., Saviano A., Maione F., D’Auria M.V., De Marino S., Giordano A., Hofstetter R.K., Festa C., et al. Structure-based screening for the discovery of 1,2,4-oxadiazoles as promising hits for the development of new anti-inflammatory agents interfering with eicosanoid biosynthesis pathways. Eur. J. Med. Chem. 2021;224:113693. doi: 10.1016/j.ejmech.2021.113693. [DOI] [PubMed] [Google Scholar]
- 18.Sharma K.K., Maurya I.K., Khan S.I., Jacob M.R., Kumar V., Tikoo K., Jain R. Discovery of a membrane-active, ring-modified histidine containing ultrashort amphiphilic peptide that exhibits potent inhibition of Cryptococcus neoformans. J. Med. Chem. 2017;60:6607–6621. doi: 10.1021/acs.jmedchem.7b00481. [DOI] [PubMed] [Google Scholar]
- 19.Bellavita R., Vollaro A., Catania M.R., Merlino F., De Martino L., Nocera F.P., Della Greca M., Lembo F., Grieco P., Buommino E. Novel antimicrobial peptide from temporin L in the treatment of Staphylococcus pseudintermedius and Malassezia pachydermatis in polymicrobial inter-kingdom infection. Antibiotics. 2020;9:530. doi: 10.3390/antibiotics9090530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zarghi A., Hajimahdi Z. Substituted oxadiazoles: A patent review (2010−2012) Expert Opin. Ther. Patents. 2013;23:1209–1232. doi: 10.1517/13543776.2013.797409. [DOI] [PubMed] [Google Scholar]
- 21.Pidugu V.R., Yarla N.S., Pedada S.R., Kalle A.M., Satya A.K. Design and synthesis of novel HDAC8 inhibitory 2,5-disubstituted-1,3,4-oxadiazoles containing glycine and alanine hybrids with anti-cancer activity. Bioorg. Med. Chem. 2016;24:5611–5617. doi: 10.1016/j.bmc.2016.09.022. [DOI] [PubMed] [Google Scholar]
- 22.Guda D.R., Park S.-J., Lee M.-W., Kim T.-J., Lee M.E. Syntheses and antiallergic activity of 2-((bis(trimethylsilyl)methylthio/methylsulfonyl) methyl)-5-aryl-1,3,4-oxadiazoles. Eur. J. Med. Chem. 2013;62:84–88. doi: 10.1016/j.ejmech.2012.12.035. [DOI] [PubMed] [Google Scholar]
- 23.Janardhanan J., Chang M., Mobashery S. The oxadiazole antibacterials. Curr. Opin. Microbiol. 2016;33:13–17. doi: 10.1016/j.mib.2016.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Laws M., Shaaban A., Rahman K.M. Antibiotic resistance breakers: Current approaches and future directions. FEMS Microbiol. Rev. 2019;43:490–516. doi: 10.1093/femsre/fuz014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ha D.R., Haste N.M., Gluckstein D.P. The role of antibiotic stewardship in promoting appropriate antibiotic use. Am. J. Lifestyle Med. 2017;13:376–383. doi: 10.1177/1559827617700824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Katayama Y., Ito T., Hiramatsu K. A new class of genetic element, staphylococcus cassette chromosome mec, encodes methicillin resistance in Staphylococcus aureus. Antimicrob. Agents Chemother. 2000;44:1549–1555. doi: 10.1128/AAC.44.6.1549-1555.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Liu J., Chen D., Peters B.M., Li L., Li B., Xu Z., Shirliff M.E. Staphylococcal chromosomal cassettes mec (SCCmec): A mobile genetic element in methicillin-resistant Staphylococcus aureus. Microb. Pathog. 2016;101:56–67. doi: 10.1016/j.micpath.2016.10.028. [DOI] [PubMed] [Google Scholar]
- 28.Rudkin J.K., Laabei M., Edwards A.M., Joo H.S., Otto M., Lennon K.L., O’Gara J.P., Waterfield N.R., Massey R.C. Oxacillin alters the toxin expression profile of community-associated methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 2014;58:1100–1107. doi: 10.1128/AAC.01618-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bellavita R., Falanga A., Buommino E., Merlino F., Casciaro B., Cappiello F., Mangoni M.L., Novellino E., Catania M.R., Paolillo R., et al. Novel temporin L antimicrobial peptides: Promoting self-assembling by lipidic tags to tackle superbugs. J. Enzyme Inhib. Med. Chem. 2020;35:1751–1764. doi: 10.1080/14756366.2020.1819258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Olajuyigbe O.O., Afolayan A.J. In vitro antibacterial and time- kill evaluation of the Erythrina caffra thunb. extract against bacteria associated with diarrhea. Sci. World J. 2012;2012:738314. doi: 10.1100/2012/738314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yap J.K.Y., Tan S.Y.Y., Tang S.Q., Thien V.K., Chan E.W.L. Synergistic antibacterial activity between 1,4-naphthoquinone and β-lactam antibiotics against methicillin-resistant Staphylococcus aureus. Microb. Drug Resist. 2021;27:234–240. doi: 10.1089/mdr.2020.0178. [DOI] [PubMed] [Google Scholar]
- 32.Irace C., Misso G., Capuozzo A., Piccolo M., Riccardi C., Luchini A., Caraglia M., Paduano L., Montesarchio D., Santamaria R. Antiproliferative effects of ruthenium-based nucleolipidic nanoaggregates in human models of breast cancer in vitro: Insights into their mode of action. Sci. Rep. 2017;7:45236. doi: 10.1038/srep45236. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data are included in the article or uploaded as supplementary data.