Abstract
Artemisia capillaris Thunb. (A. capillaris, Yin-Chen in Chinese) is a traditional medicinal herb with a wide spectrum of pharmacological properties ranging from effects against liver dysfunction to treatments of severe cirrhosis and cancer. We used relevant keywords to search electronic databases, including PubMed, Medline, and Google Scholar, for scientific contributions related to this medicinal herb and the pharmacokinetics of its components. The pharmaceutical effects of A. capillaris contribute to the treatment not only of viral hepatitis, cirrhosis, and hepatocellular hepatoma, but also metabolic syndrome, psoriasis, and enterovirus in the clinic. The bioactive compounds, including scoparone, capillarisin, scopoletin, and chlorogenic acid, exhibit antioxidant, anti-inflammatory, antisteatotic, antiviral, and antitumor properties, reflecting the pharmacological effects of A. capillaris. The pharmacokinetics of the main bioactive compounds in A. capillaris can achieve a maximum concentration within 1 hour, but only chlorogenic acid has a relatively long half-life. Regarding the use of the A. capillaris herb by health professionals to treat various diseases, the dosing schedule of this herb should be carefully considered to maximize therapeutic outcomes while lessening possible side effects.
Keywords: Artemisia, pharmacokinetics, pharmacology, herbal medicine, Yin-Chen, scoparone
1. Introduction
The original uses of Artemisia capillaris Thunb. (A. capillaris, Yin-Chen in Chinese) in traditional Chinese medicine included the treatment of pyrexia, jaundice, and dysuria. Current basic studies of traditional Chinese medicine aim to find molecular, cytological and pharmacological evidence supporting the use of traditional Chinese herbal medicines to confirm the nature of active ingredients and to explore the roles of these compounds in the treatment of various diseases. Studies of this traditional herb provide reliable empirical evidence for the development and progress of this traditional Chinese medicine, which has been widely used in the treatment of various diseases ranging from liver inflammation to severe cirrhosis and liver cancer [1,2,3]. Previous studies have shown that the whole plant exerts antioxidant, anti-inflammatory, antisteatotic, antiviral, and antitumor effects [4,5,6,7]. Most of the therapeutic effects can be attributed to the major or minor compounds found in medicinal herbs [8].
However, the bioactive components of an herb are influenced by the plant species and origin. For example, the clinical use of Artemisia in traditional Chinese medicine includes A. capillaris and Artemisia scoparia (A. scoparia) [9]. Though A. capillaris is the main species used in medicinal formulations, A. scoparia contains higher levels of the essential active compound scoparone, whereas chlorogenic acid is abundant in A. capillaris [9]. The harvested time and region of the plant also affect the chemical compositions of bioactive compounds. Both capillarisin and scoparone (6,7-dimethylesculetin) content reach peak levels in the leaf of A. capillaris at the end of July. However, the maximal level of capillarisin and scoparone is detected in the capitulum in early August and early September, respectively [10]. The appropriate time to harvest A. capillaris is an important consideration that affects pharmacological actions, including possible toxicity from scoparone. Thus, the therapeutic benefits of A. capillaris are influenced by various parts of the plant and harvest time, and should be balanced based on the contents of bioactive components.
The level of bioactive compounds determines an individual’s pharmacokinetic and pharmacodynamic reactions to herbal medicines. Recent studies have invested considerable efforts into exploring the multicomponent pharmacokinetics of herbal medicines and to elucidate relevant metabolic processes. For instance, an herbal formula containing A. capillaris was found to be disproportional to its administering dose in the area under the curve (AUC) by monitoring its bioactive components [11], which indicated pharmacokinetic saturation leading to excessive dosage or potential interaction with other drugs. The pharmacokinetic investigation of A. capillaris combined herbal formula upon dose appeared to interact with spironolactone, including the urinary sodium-to-potassium ratio [12]. In addition, a lack of information on the therapeutic window and pharmacodynamics that guide pharmacokinetics makes the efficacy and safety of herbal medicine questionable.
Previous studies reported pharmacokinetic properties or pharmacological activity of A. capillaris aims on one bioactive component or single therapeutic function. Few reviews summarize the pharmacokinetic properties of major bioactive compounds as well as currently proved pharmacological usage as suggestions for future potential applications of the plant. To the best of our knowledge, A. capillaris has been known for the treatment of liver disease but awareness is rare for the treatment for metabolic syndrome, psoriasis, or antifibrotic effects that indicate that this plant has an expanded range of therapeutic activities. Thus, the present review of pharmacological effects of A. capillaris and linking to the pharmacokinetics of extracted bioactive compounds aims to discover additional applications of this herbal medicine.
2. Research Methods
Scientific search engines, such as PubMed, Medline, and Google Scholar were used to collect all published articles on ethnomedicinal use, biological properties, and pharmacokinetics of A. capillaris. A synopsis of this search is presented in this review. Acquired manuscripts were assessed and identified based on the title and abstract. The following search terms were used as keywords: Artemisia, Artemisia capillaris, Yin-Chen (Chinese name), Yin-Chen-Hao-Tang (an ancient Chinese formula), and pharmacokinetics. The reference lists of retrieved publications were also examined to identify other relevant studies.
3. Pharmacological Effects of Artemisia capillaris
3.1. Viral Hepatitis B Infection
Hepatitis B virus (HBV) infection can progress to liver cirrhosis or liver cancer. At present, the therapeutic options for this disease are limited by agents used for eradication of HBV or of side effects of antiviral therapies. Compounds isolated from A. capillaris inhibit the secretion of HBsAg or HBeAg or replication of HBV DNA [13]. HBeAg secretion and HBV DNA replication in HepG cells are significantly inhibited by 90% ethanol extract of A. capillaris [14], and this activity is due to chlorogenic acid analogs and enynes found in A. capillaris [15].
3.2. Cirrhosis and Hepatoprotective Effects
Cirrhosis refers to the late stage of scarring caused by repeated pathological destruction and regeneration of the liver due to various forms of liver diseases. A strategy for the prevention of liver damage involves pretreatment with an aqueous extract of A. capillaris, which significantly reduces oxidative stress in the liver induced by 2,2′-azobis(2-amidinopropane) dihydrochloride (AAPH), as demonstrated by a decrease in the level of liver injury based on the enzyme markers aspartate transaminase (AST) and alanine transaminase (ALT) in rats [16]. Herbal formulations consisting of A. capillaris and Alisma canaliculatum (Saeng-kankunbi-tang, SKT) also prevent tert-butyl hydroperoxide (tBHP)-induced oxidative injury in HepG2 hepatocytes and acute oxidative hepatic damage caused by carbon tetrachloride (CCl4) in mice [17].
Antifibrotic effects of A. capillaris have also been reported. The β-sitosterol component derived from A. capillaris alleviates dimethylnitrosamine (DMN)-induced hepatofibrosis in mice [18]. In a model of bile duct ligation (BDL)-induced cholestatic fibrosis, aqueous extract of A. capillaris suppresses the expression of fibrogenic factors, including alpha-smooth muscle actin (α-SMA), platelet-derived growth factor (PDGF), and transforming growth factor-beta (TGF-β), and significantly reduces the levels of cholestatic markers malondialdehyde (MDA) in the serum and hydroxyproline in the liver after 2 weeks of treatment in rats [19]. An increase in the AST, ALT, and MDA levels induced by 30% alcohol plus pyrazole is also ameliorated by aqueous extract of A. capillaris in a rat model [20]. This hepatoprotective effect is attributed to an enhancement of antioxidant activity, including glutathione peroxidase (GSH-Px), glutathione reductase (GSH-Rd), catalase, and superoxide dismutase (SOD) [20]. However, another study demonstrated that 5-week treatment with aqueous A. capillaris extract did not alter liver enzymes, including ALT, AST, and alkaline phosphatase (ALP), in a rat model of CCl4-induced hepatic fibrosis, although different results were obtained in the case of Artemisia iwayomogi [21]. Another study reached a similar conclusion after administration of methanol extract of A. capillaris in rats with bile duct ligation; the results indicated that the serum levels of AST, ALT, and ALP and hepatic levels of hydroxyproline were significantly reduced in the Polygonum aviculare-treated group but not in the groups treated with A. capillaris and aqueous biphenyl dimethyl dicarboxylate [22]. Variability of hepatoprotective effects may be due to extraction or cultivation and should be confirmed by assessment of biological components.
3.3. Hepatocellular Carcinoma
Hepatocellular carcinoma (HCC) requires effective treatment due to low 10% 5-year survival rate of this disease [23]. Increasing evidence indicates that A. capillaris can efficiently suppress the proliferation of human hepatoma cells, and ethanol extract of A. capillaris exhibits dose-dependent antiproliferative effects against Huh7 and HepG2 human hepatoma cells mediated by inhibition of cancer cell migration via interleukin-6 (IL-6)-dependent regulation of the signal transducer and activator of transcription 3 (STAT3) pathway [24]. In HepG2 human hepatocarcinoma cells, aqueous extract of A. capillaris inhibits nuclear translocation of NF-κB and blocks the degradation of I-κB alpha, leading to inhibition of inflammatory proteins, such as inducible nitric oxide synthase (iNOS), cyclooxygenase-2 (COX-2), and tumor necrosis factor (TNF)-alpha [25]. Additionally, water-soluble macromolecular components of A. capillaris dose-dependently inhibit the proliferation of human hepatoma SMMC-7721 cells by inducing the cell-cycle arrest at the G0/G1 phase [26]. Ethyl acetate extract of A. capillaris can effectively inhibit the growth and induce apoptosis of hepatocellular carcinoma cells, and these effects are presumed to be mediated by inhibition of angiogenesis via the blockade of the PI3K/AKT/mTOR signaling pathway [27]. Dried leaves of A. capillaris use a similar mechanism to induce apoptosis in HepG2 and Huh7 cells and to suppress tumor growth in mouse xenograft models [28].
3.4. Metabolic Syndrome and Diabetes
Metabolic syndrome is associated with a fivefold higher risk for type 2 diabetes (T2DM). Overabundance of circulating fatty acids is the major contributor to pathophysiology of metabolic syndrome. Fatty acids inhibit antilipolytic effect of insulin and induce lipolysis of stored triacylglycerol molecules in the adipose tissue, which leads to insulin resistance [29]. Glucosidase inhibitors delay the absorption of carbohydrates to decrease postprandial hyperglycemia. The inhibitory effect of A. capillaris on α-glucosidase is the most potent among that of 12 various Artemisia species, and this effect is even stronger than inhibition by acarbose [30]. Rats administered a high-fat diet (HFD) were treated with A. capillaris extracts for 7 weeks, and the body weight and levels of serum triglycerides (TG), total cholesterol (TC), and low-density lipoprotein cholesterol (LDL-c) of treated rats were measured and found to be lower than those in the HFD-induced obesity control group; however, the levels of high-density lipoprotein cholesterol (HDL-c) were not significantly different between the groups [31]. Similar results were obtained in another study using a traditional A. capillaris formula. The treatment abrogated an increase in liver enzymes and lipid parameters, such as TG, TC, and LDL-c, but not HDL-c, in HFD-fed rats. These effects were apparently mediated by miR-122-induced downregulation of fatty acid synthase genes in HepG2 cells [32].
Free fatty acids induce insulin resistance and inflammation in insulin-targeted organs, indicating a major link between obesity, insulin resistance, inflammation, and T2DM development [33]. Free fatty acid-induced steatosis was relieved and nonalcoholic steatohepatitis (NASH)-related mechanisms were inhibited in HepG2 cells treated with 30% ethanolic extract of A. capillaris, including activation of c-Jun NH2 terminal kinase (JNK) and p53-upregulated modulator of apoptosis (PUMA) [34]. An n-BuOH fraction of methanol extract of A. capillaris contains vicenin 2, which potently inhibits diabetes-targeting enzymes α-glucosidase, protein tyrosine phosphatase 1B (PTP1B), and rat lens aldose reductase (RLAR) [35]. However, A. capillaris extract combined with Alisma rhizome extract does not alter the levels of lipid metabolites, such as triacylglycerol and diacylglycerol, in diabetic mice [36]. In contrast, Hericium erinaceus cultivated with A. capillaris can elevate the HDL level and lower atherogenic index and cardiac risk factor values in hyperlipidemic rats; these effects were more pronounced than the effects of currently used drugs simvastatin and atorvastatin [37]. Thus, the regulatory effects of A. capillaris on dyslipidemia appear to be influenced by additions of other herbs.
3.5. Dermatitis, Psoriasis, and Skin Carcinogenesis
Atopic dermatitis is an inflammatory skin disease caused by an imbalance of Th cells, the overexpression of COX-2 and iNOS, which generate nitric oxide (NO) and prostaglandin E2, respectively, and stimulation of macrophages by lipopolysaccharide. Topical application of A. capillaris for 4 weeks reduced the atopic dermatitis scores and plasma levels of histamine and IgE in Dermatophagoides farinae-sensitized Nc/Nga mice [38]. Solid fermentation of Ganoderma lucidum on A. capillaris leaves reduced the expression of endothelial nitric oxide synthase (eNOS) in mice with 2,4-dinitrofluorobenzene (DNFB)-induced atopic dermatitis [39]. A. capillaris extract cream has been locally applied to skin lesions in a mouse model of imiquimod (IMQ)-induced psoriasis-like disease, and the level of intracellular adhesion molecule-1 (ICAM-1), modified psoriasis area, and severity index (PASI) scores of treated mice were significantly lower than those of mice in other experimental groups [40]. Moreover, the chloroform fraction of methanolic extract of A. capillaris markedly decreased the number and incidence of tumors in mice with 12-dimethylbenz(a)anthracene (DMBA)-induced epidermal carcinogenesis compared with the results obtained using other anticarcinogenic medicinal herbs, including Taxus cuspidata, Anthriscus sylvestris, and Curcuma longa [41].
3.6. Enterovirus 71 (EV71) and Helicobacter pylori (H. pylori)
Infectious diseases have affected humans for centuries and have civilization-altering consequences. Studies in human foreskin fibroblasts and rhabdomyosarcoma cells demonstrated that aqueous extract of A. capillaris dose-dependently protects against EV71 infection, mainly due to inhibition of viral internalization [42]. Acidic polysaccharides from A. capillaris are potent inhibitors of adhesion of H. pylori to erythrocytes; however, this effect was less potent than that of acidic polysaccharides from Panax ginseng (P. ginseng) [43]. Aqueous extract of A. capillaris has no acid-neutralizing activity and does not prevent histamine secretion from HMC-1 mast cells; however, pretreatment with this aqueous extract decreases HCl/ethanol-induced gastric mucosal lesions [44,45]. A summary of the therapeutic effects of A. capillaris is provided in Table 1.
Table 1.
Experimental therapeutic effects of Artemisia capillaris.
| Disease | Extract | Doses/Concentrations | Time Period | Results/Mechanism | Active Compounds | Reference |
|---|---|---|---|---|---|---|
| Hepatitis B | 90% ethanol | 76.1 μg/mL | NR | inhibition of HBeAg secretion and HBV DNA replication in HepG cells | chlorogenic acid | [14] |
| Cirrhosis | water | 10 mg/kg | NR | alleviation of hepatic fibrosis in a DMN-induced mouse model | β-sitosterol | [18] |
| water | 25 or 50 mg/kg | 2 weeks | suppression of fibrogenic factors, including hepatic α-SMA, PDGF, and TGF-β | NR | [19] | |
| water | 7.5 g/kg | 7 days | lowering of AST and ALT in a rat model of AAPH liver damage | NR | [16] | |
| water | 50 or 100 mg/kg | 10 days | amelioration of AST, ALT, and MDA in an alcohol-pyrazole-fed rat model; an increase in antioxidant activity due to GSH-Px, GSH-Rd, catalase, and SOD | NR | [20] | |
| water | 25 or 50 mg/kg | 6 weeks | unchanged ALT, AST, and ALP in a rat model of CCl4-induced liver fibrosis | NR | [21] | |
| Hepatocellular carcinoma | 70% ethanol | 100 and 250 μg/mL | NR | inhibition of IL-6 mediated STAT3 pathway in Huh7 and HepG2 human hepatoma cells | NR | [24] |
| water | 50, 150, and 300 mg/kg | NR | Inhibition of NF-kappaB translocation, inhibition of iNOS, COX-2, and TNF-alpha in HepG2 cells | NR | [25] | |
| water | 25–200 µg/mL | NR | cell cycle arrest at the G0/G1 phase in SMMC-7721 cells | NR | [26] | |
| 70% ethanol | 50 and 100 μg/ml | NR | Induction of apoptosis of HCC cells by blocking the PI3K/AKT/mTOR signaling pathway | NR | [27] | |
| 70% ethanol | 100 μg/mL | 15 days | induction of apoptosis in HepG2 and Huh7 cell; suppression of tumor growth in a mouse xenograft | NR | [28] | |
| Metabolic syndrome and diabetes | methanol | 0.4% and 0.8% extract | 7 weeks | lowering of TG, TC, and LDL-c levels in high-fat diet-fed rats; a decrease in fatty acid synthase expression via miR-122 | NR | [31,32] |
| 30% ethanol | 100 μg/mL | NR | inhibition of activation of JNK and PUMA in NASH | NR | [34] | |
| methanol | IC50 values: 270.5 μM (α-glucosidase); 7.85 μM (RLAR); 139.75 μM (PLAR) | NR | inhibition of α-glucosidase, PTP1B, and RLAR. | vicenin 2 | [35] | |
| Dermatitis, psoriasis, and skin carcinogenesis | 70% ethanol | 10 mg | 4 weeks | reduction in atopic dermatitis scores, histamine, and IgE in Dermatophagoides farinae-sensitized Nc/Nga mice (topical). | chlorogenic acid, caffeic acid, isochlorogenic acid A, hyperoside, isoquercitrin, and scoparone | [38] |
| 70% ethanol | 250 mg/kg | 5 days | lowering of ICAM-1 and modified PASI scores in a mouse model of induced psoriasis-like disease (topical) | chlorogenic acid, 3,5-dicaffeoylquinic acid, 4,5-dicaffeoyl-quinic acid, and scoparone | [40] | |
| methanol | 50 g sample in 500 mL of MeOH | 7 days | reduction in the tumor number and incidence in a mouse mode of DMBA-induced epidermal carcinogenesis | camphor, 1-borneol, coumarin, and achillin | [41] | |
| Enterovirus 71 and H. pylori l | water | 10 μg/mL | NR | effect against EV71 infection due to inhibition of viral internalization | chlorogenic acid | [42] |
| water | 0.2–2.8 mg/mL | NR | inhibition of H. pylori adhesion to erythrocytes | acidic polysaccharide | [43] | |
| water | 200 or 400 mg/kg | NR | inhibition of lipid peroxide formation due to SOD activation, a reduction in proinflammatory cytokines IL-6 and IL-1β mediated by NF-κB downregulation | NR | [44] | |
| ethanol | 30 or 100 mg/kg | 1 h prior | attenuation of ethanol-induced proinflammatory cytokines IL-1β and interferon-γ and ICAM-1 | NR | [45] |
α-SMA, alpha-smooth muscle actin; AAPH, 2,2′-azobis(2-amidinopropane) dihydrochloride; ALT, alanine transaminase; ALP, alkaline phosphatase; AST, aspartate transaminase; CCl4, carbon tetrachloride; COX-2, cyclooxygenase-2; GSH-Px, glutathione peroxidase; GSH-Rd, glutathione reductase; ICAM-1, intracellular adhesion molecule-1; IL-1β, interleukin-1 beta; IL-6, interleukin-6; iNOS, inducible nitric oxide synthase; LDL-c, low-density lipoprotein cholesterol; JNK, c-Jun NH2-terminal kinase; MDA, malondialdehyde; NASH, nonalcoholic steatohepatitis; NF-κB, nuclear factor kappa B; NR, not reported; PASI, psoriasis area and severity index; PDGF, platelet-derived growth factor; PTP1B, protein tyrosine phosphatase 1B; PUMA, p53-upregulated modulator of apoptosis; RLAR, rat lens aldose reductase; SOD, superoxide dismutase; STAT3, signal transducer and activator of transcription 3; TC, total cholesterol; TG, triglycerides; TGF-β, transforming growth factor beta; TNF-alpha, tumor necrosis factor alpha.
4. Pharmacokinetics of Bioactive Compounds Found in Artemisia capillaris
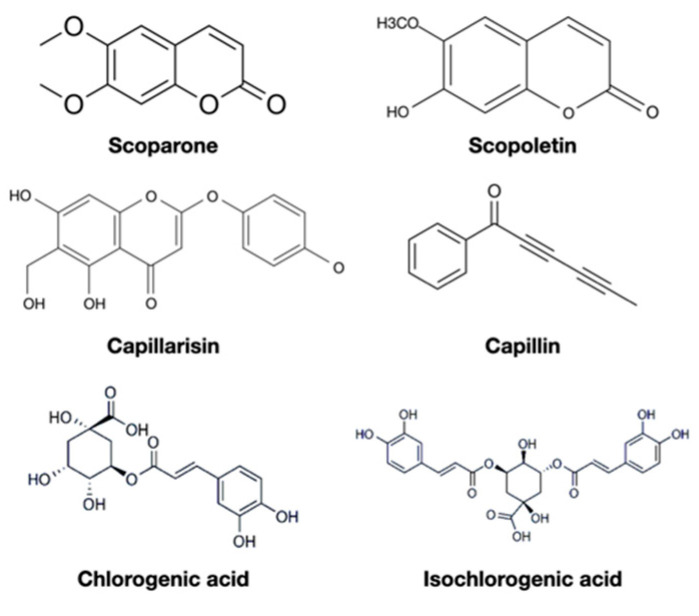
A. capillaris have been intensively studied to evaluate its effects on healthcare, demonstrating that A. capillaris has protective effects against hepatitis, cirrhosis, cancer, metabolic syndrome, dermatitis, and microbes, which triggered additional scientific inquiries. Numerous bioactive compounds were therefore extracted and found to have beneficial effects, including anti-inflammatory, antioxidant, antitumor, or even anti-HIV activities. The most commonly identified constituents include scoparone, capillarisin, and chlorogenic acid (Table 2) [46,47,48,49]. Their chemical structures are shown in Figure 1. From a practical perspective, these bioactive compounds composed of the pharmacology of A. capillaris need rigorous pharmacokinetics research to provide science-based dosage recommendations for various therapeutic properties of A. capillaris. Realizing the pharmacokinetics of bioactive compounds in A. capillaris could improve the administration schedule to achieve medicinal effects in an efficient manner. The pharmacokinetics of bioactive compounds in A. capillaris are discussed below.
Table 2.
Bioactive compounds of Artemisia capillaris extraction studies.
| Constituents | Extract Solution | Molecular Weight (g/mol) | LC-Mass Detection Ions | References | |
|---|---|---|---|---|---|
| [M − H]− | [M + H] + | ||||
| Scoparone 1 | supercritical carbon dioxide with ethyl acetate | 206.20 | 207 | [46,47,48,49] | |
| Capillarisin 1 | 316.27 | 315 (M−) | 317 (M+) | ||
| Chlorogenic acid 2 | 354.31 | 191 | 163 | [46,47,49] | |
| Scopoletin 2 | ethyl acetate and water | 192.17 | 104 | 234 3 | [47,48,49] |
| Capillin | 168.20 | 140 3 | [48,49] | ||
| Isoscopoletin | 192.17 | 176 4 | 133 | ||
| Capillene | 154.21 | 153 3 | |||
| Capillartemisin B | 316.40 | ||||
| Cirsimaritin | 314.29 | 315 | |||
| Capillarisin-7-methyl ether | 330.34 | [48] | |||
| artepillin A | 316.40 | ||||
| Artepillin C | 300.40 | ||||
| Capillarin | 198.22 | ||||
| Eugenol | sodium dodecyl sulfate and sodium borate buffer | 164.20 | 165 | [47] | |
| Caffeic acid | 180.16 | 135 | 163 | ||
| Phenol | 94.11 | 95 | |||
| o-cresol | 108.14 | 108 (M+) | |||
| m-cresol | 108.14 | 109 (M+) | |||
| p-cresol | 108.14 | 93.0 (M+) | |||
| 2-ethylphenol | 122.17 | 107 3 | |||
| 4-ethylphenol | 122.17 | 107 3 | |||
| Artemisidin A | methanol | 642.50 | [49] | ||
| Artemicapins A | 250.20 | ||||
| Artemicapins B | 236.18 | ||||
| Artemicapins C | 206.15 | ||||
| Artemicapins D | 440.4 | ||||
| other 70 known compounds | |||||
1 Compounds were found in all references. 2 Compounds were found in three references. 3 Ions were detected by GC-MS. 4 Ions were detected by MS–MS.
Figure 1.
The chemical structure of commonly identified constituents in Artemisia capillaris.
4.1. Scoparone
Scoparone is considered the main and most important active constituent of A. capillaris. Analysis of Soxhlet extracts of A. capillaris indicated that the plant contains higher levels of scoparone than the levels of capillarisin or chlorogenic acid [46]. This compound was considered of interest due to its preventive and therapeutic effects against liver disease, which prompted subsequent investigations [50,51]. Scoparone isolated from A. capillaris has been shown to have antioxidant properties demonstrated by a reduction in the MDA and ALT levels in cold-preserved rat hepatocytes [52]. Scoparone decreases the levels of interleukin (IL)-1-beta, IL-6, and TNF-alpha due to anti-inflammatory activities of the compound and suppresses the levels of iNOS and COX-2 in IFN-gamma- or LPS-stimulated cells [53]. Moreover, scoparone inhibits the transcriptional activity of peroxisome proliferator-activated receptor gamma (PPARγ) and downregulation of the target genes, and this effect leads to inhibition of triglyceride (TG) accumulation in mature adipocytes [54]. Choleretic effect of scoparone is indirectly potentiated by cytochrome P4501A2 via the bile salt export pump promoter [55]. Scoparone is considered a potential hepatoprotective candidate for hepatitis therapy based on published evidence [56]. Additionally, scoparone 60 mg/kg daily could alleviate angiotensin II infusion-induced cardiac hypertrophy and fibrosis in mice with maintaining cardiac output, left ventricular pressure, and left ventricular workload [57,58].
Analysis of pharmacokinetic parameters of scoparone could achieve a wide range of the Cmax values from 0.02 mg/L or up to 16.1 mg/L accompanying with standard compounds as Yin-Chen-Hao-Tang ingredients, and most Tmax values ranged from 6 to 54 min with the exception of one study that reported a Tmax value of 1.9 h after combined administration with Gardenia jasminoides Ellis [59]; the corresponding elimination half-life ranges from 25.8 min to 5.11 h [11,59,60,61,62,63]. The IC50 value for scoparone-associated significant inhibition of the proliferation of DU145 prostate cancer cells is 8.5 mg/L (41.3 μmol/L) [64]. High levels of scoparone in A. capillaris are apparently responsible for inhibition of prostate cancer proliferation. Consequently, two characteristics of scoparone have been noticed. First, a combination of geniposide delay absorption of scoparone and combined with geniposide and rhein increased the AUC [63]. Additionally, the herbal formula Yin-Chen-Hao-Tang has been used to illustrate nonlinear pharmacokinetic properties of scoparone, which represents a potential enhancement of pharmacological effects of this drug [11].
4.2. Scopoletin
Scopoletin isolated from A. capillaris contributes to bile secretion similarly to scoparone but has no effect on bile acid and cholesterol secretion [48]. These effects may be due to inhibition of lipid biosynthesis, which results in downregulation of gene expression related to cholesterol, triglyceride synthesis, and inflammation induced by steatosis [65]. This compound may assist in lowering postprandial hyperglycemia and improving antidiabetic treatments [66]. Scopoletin can enhance histone deacetylase expression to inactive p53 in human lung fibroblasts, which leads to autophagy-related antiaging effects [67]. Moreover, scopoletin is cytotoxic toward cancer cells, such as prostate cancer cells (PC-3) and acute lymphoblastic leukemia cells [68,69]. Although NF-κB activation by scopoletin implies a resistance mechanism of cancer cells, the main resistance mechanisms, such as ATP-binding cassette (ABC) transporters, EGFR, and TP53, do not affect cellular resistance to scopoletin [70].
Oral administration of pure scopoletin at the doses of 5, 10, or 20 mg/kg results in the Cmax values in the plasma of 49.8, 101.3, or 217.3 μg/L, respectively, reached within 0.4 h [71]. Another study of oral administration at a dose of 50 mg/kg resulted in the Cmax value of 0.4 μg/L within 14 min. However, treatment with a decoction prepared from 720 g of A. capillaris resulted in a relatively low plasma concentration of only 3.5 μg/L after feeding [61]. Similarly, administration of Radix angelicae pubescentis extract containing 0.055 mg/kg scopoletin did not result in detectable levels of the compound in rat plasma [72]. It has also been revealed with a short elimination half-life in dogs [73]; thus, scopoletin is not easily absorbed or rapidly metabolized when administered in an herbal formula. The IC50 value of scopoletin for human CCRF-CEM leukemia cells is 499.6 μg/L [69]. These findings indicate that the level of scopoletin in A. capillaris and Radix angelicae pubescentis is insufficient to detect pharmacological effects of pure scopoletin against leukemia cells in the clinic.
4.3. Capillarisin
Capillarisin is derived naturally from chromone. The compound has antioxidant, anti-inflammatory, and potential antitumor properties [74], and antioxidant and anti-inflammatory activities are regulated via the Nrf2/ARE-dependent pathway and activation of ERK, JNK, NF-κB, and MAPK [75,76,77]. Capillarisin also has inhibitory effects on prostate carcinoma cells, apparently mediated by suppression of the activation of androgen receptor, survivin, matrix metalloproteinase (MMP)-2, MMP-9, and STAT3 [78,79].
Treatment with Yin-Chen-Hao-Tang (YCHT) formula, which combines 18 g of A. capillaris with Gardenia jasminoides Ellis (9 or 12 g) and Rheum palmatum L. (6 g), reported two remarkable maximal concentrations of capillarisin of 196 μg/L or 490 μg/L [80,81]. The Tmax value for capillarisin administered as a component of the YCHT decoction ranges from 5 to 39 min, and the AUC is highly variable, with elimination half-lives ranging from 26 to 159 min. Capillarisin has notably higher affinity to human serum albumin than that of scoparone, indicating that capillarisin bioactivity has a stationary phase [82]. Capillarisin (95% (wt)) has inhibitory effects on human hepatoma Hep-G2 and HUH7 cells, with the IC50 values of 72 and 105 μg/mL, respectively [46], and the IC50 values for inhibition of the migration and proliferation of colon cancer cells are 92.1 and 76.7 µg/mL, respectively [83].
4.4. Capillin
Capillin extracted from Artemisia capillaris spica inhibits apoptosis induced by transforming growth factor-beta 1 (TGF-β1), which is observed in various inflammatory liver diseases [84]. Human leukemia HL-60 cells are induced to undergo apoptosis after treatment with capillin, and this effect is regulated by activation of the JNK pathway [85]. Antitumor effects of capillin have also been detected in colon carcinoma (HT29 cells), pancreatic carcinoma (MIA PaCa-2 cells), epidermoid carcinoma of the larynx (HEP-2 cells), and lung carcinoma (A549 cells) [84]. Moreover, capillin is a potent inhibitor of α-glucosidase, protein PTP1B, and RLAR for management of diabetes and related complications [86]. However, extraction of capillin from A. capillaris has been rarely reported; thus, the pharmacokinetics of capillin are unknown. Further pharmacokinetic studies based on the IC50 values should be considered in the investigation of antitumor properties of the compound.
4.5. Chlorogenic Acid
Chlorogenic acid is the most abundant compound (38.5 mg/g) in hydroethanolic extract of A. capillaris [87]. Chlorogenic acid is also extensively enriched in various foods, such as coffee, tea, cocoa, citrus fruits, berry fruits, apples, and pears [88]. The biological activities of chlorogenic acid have been reported against various diseases, including metabolic syndrome, hypertension, and diabetes [89,90,91], indicating a broad range of anti-inflammatory, antihyperglycemic, and antioxidant activities of the compound [92,93,94]. The mechanism of action of chlorogenic acid mainly involves uptake and synthesis of fatty acids in the liver by modulating hepatic peroxisome proliferator-activated receptor γ or liver X receptors-α (PPARαγ, LXRα) [95]. The compound suppresses inflammation induced by a high-fat diet due to scavenging of reactive oxygen species (ROS) [96]. Chlorogenic acid regulates apoptosis-related genes, and this effect may contribute to the anticancer effect in a lung cancer cell line [97].
Administration of YCHT results in a Cmax for chlorogenic acid of 33 μg/L or 78 μg/L within 27 min, but the half-life can be minutes to several days [80,81]. Administration of 60 mg/kg dose of chlorogenic acid in extracts Lonicerae japonicae Flos achieved a maximum concentration of 2.4 mg/L in the plasma of beagle dogs after 1.0 h [98]. Eucommia ulmoides extract containing 1.2, 2.6, and 5.1 mg/kg chlorogenic acid acquired Cmax of 22.5, 39.8, and 61.0 ng/mL, respectively, within 26 min for all three concentrations after its administration to rats [99]. Chlorogenic acid has been administered to humans by 10 commercially available products with an average content of 3.6 mg, which achieved a maximal concentration of 0.76 ng/mL after approximately 1 h [100]. Though chlorogenic acid was found to be not well-absorbed from the digestive tract [101], the currently available pharmacokinetics of chlorogenic acid in A. capillaris revealed rapid time to maximum concentration with up to a month of half-life. Future studies in pharmacology of A. capillaris should consider the prolonged elimination in the pharmacokinetics of chlorogenic acid.
4.6. Isochlorogenic Acid
Other constituents isolated from Artemisia capillaris include isoscopoletin, artepillin, esculetin, isochlorogenic acid, β-sitosterol, and quercetin. Natural isochlorogenic acid refers to a mixture of several isomers. Isochlorogenic acid A is the most or second-most abundant component found in ethanol extract of A. capillaris [38,87], and is also a major bioactive constituent in other medicinal plants such as Lonicerae japonicae Flos, Gynura divaricata, and Laggera alata [102,103,104]. Isochlorogenic acid A probably blocks HBV replication to provide for potent anti-HBV activity and induces heme oxygenase-1 (HO-1) expression, which leads to antioxidant effects [104]. The viral load of enterovirus 71 and cytokine secretion decreased after treatment with isochlorogenic acid C [105]. Antioxidant effects and suppression of profibrogenic factors by isochlorogenic acid B are mediated via the Nrf2 and miR-122/HIF-1α signaling pathways to protect against fibrosis in nonalcoholic steatohepatitis (NASH) [106].
The pharmacokinetics of isochlorogenic acid from A. capillaris extracts have not been reported. Analysis of pharmacokinetics of isochlorogenic acid after administration of 5.0 g/kg of Erigeron breviscapus ethanol extract, which contains 16.56 mg/kg isochlorogenic acid A and 10.64 mg/kg isochlorogenic acid C, revealed the Cmax values of 1.033 μg/mL and 0.230 μg/mL after 1.17 and 0.11 h, respectively [107]. Oral administration of isochlorogenic acid C at the doses of 5 mg/kg or 25 mg/kg resulted in the Tmax values ranging from 30 min to 1 h [108]. However, the bioavailability of isochlorogenic acid C is relatively low, ranging from 14.4% to 16.9%. Although the compound has a linear pharmacokinetics profile, poor gastrointestinal absorption and low potency of the compound in herbal medicines may diminish potential therapeutic applications of isochlorogenic acid C.
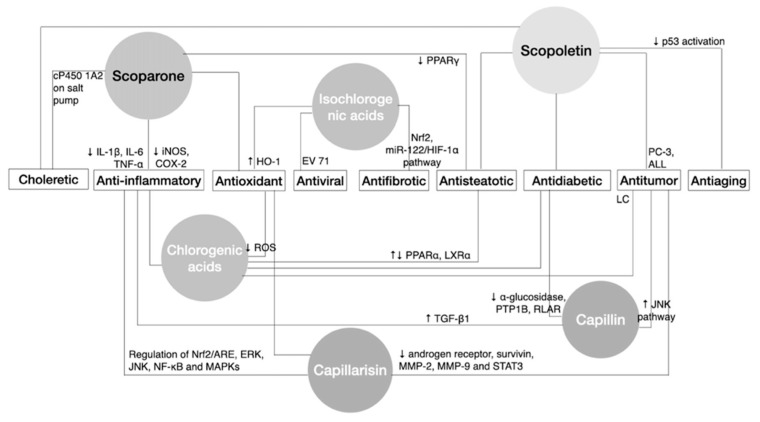
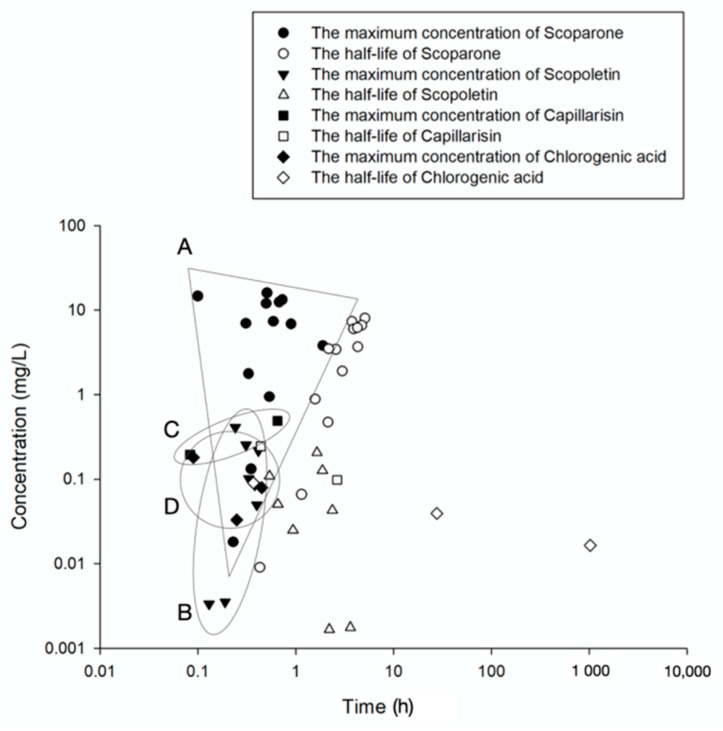
Analysis of the pharmacodynamic properties of bioactive components of A. capillaris, such as the choleretic, anti-inflammatory, and antioxidant effects, are mainly from scoparone, scopoletin, capillarisin, capillin, and chlorogenic acids, while antidiabetic, antisteatotic, and antitumor effects are from scopolectin, capillarisin, capillin, and chlorogenic acids (Figure 2). Considering the relatively low plasma concentration detected from scopoletin, the antitumor properties of A. capillaris consequently in part come from capillarisin, capillin, and chlorogenic acids. Despite the unknown pharmacokinetics of capillin, all compounds could reach the corresponding Cmax within an hour and diminished to half-concentration within 5 hours except for chlorogenic acids, which could last more than a month (Figure 3).
Figure 2.
Schematic illustration of the bioactive components of Artemisia capillaris contributing to its therapeutic effects. Bioactive compounds include scoparone, scopoletin, capillarisin, capillin, chlorogenic acid, and isochlorogenic acid, which induce pharmacological effects of A. capillaris in a synergistic manner. PC-3: prostate cancer cells; ALL: acute lymphoblastic leukemia cells; LC: lung cancer cells; ↓ decrease or inhibit effects; ↑ increase or enhance effects; ↓↑ modulate effects.
Figure 3.
The pharmacokinetic extent of bioactive compounds in Artemisia capillaris. The figure illustrates the absorption and elimination of scoparone, scopoletin, capillarisin, and chlorogenic acid in vivo after oral administration of Artemisia capillaris extracts. The black symbols represent the maximum concentration (Cmax) of the main bioactive compounds, including scoparone, scopoletin, capillarisin, and chlorogenic acid. The white symbols represent the half-life (t1/2) of these compounds. A, B, C, D indicate the area of the Cmax of scoparone, scopoletin, capillarisin, and chlorogenic acid, respectively.
5. Conclusions
A. capillaris has a wide spectrum of pharmacological properties. In addition to HBeAg inhibition, lowering of AST and ALT, and induction of apoptosis of HCC cells, it also lowers TG, TC, and LDL, ICAM-1, inhibits α-glucosidase, and causes viral internalization and H. pylori adhesion. Published studies have revealed these effects were attributed to verified bioactive compounds such as scoparone, scopoletin, capillarisin, capillin, and chlorogenic acids. These commonly extracted bioactive compounds induce pharmacological effects, including anti-inflammatory, antioxidant, choleretic, antisteatotic, antidiabetic, and antitumor activities, and synergistically contribute to the therapeutic effects of A. capillaris. Considering the pharmacokinetics of main bioactive compounds in A. capillaris, the choleretic, anti-inflammatory, and antioxidant effects that partly come from scoparone and scopoletin contributing to anti-hepatitis treatment could achieve a therapeutic window within a relatively short time. On the other hand, for antisteatotic, antidiabetic, and antitumor activities attributed to capillarisin, capillin, and chlorogenic acids with relatively low Cmax and a long half-life, the dosing may need to be adjusted to suit a particular therapeutic purpose. Pharmacokinetic parameters of some bioactive compounds in A. capillaris, including scoparone and capillarisin, were acquired from the herbal formula Yin-Chen-Hao-Tang, which significantly altered the pharmacokinetics of bioactive compounds. Regarding the use of this herb by health professionals for treatments of various diseases, the development of A. capillaris in composition or dosing schedule for more effective and efficient treatment clinically should be considered for future clinical studies in patients.
A worldwide outbreak of respiratory infection beginning in December 2019 has challenged scientists to find antiviral agents for the treatment of the disease. Researchers proposed the development of Artemisia annua for the treatment of COVID-19 due to its antiviral and antioxidant activity in pulmonary fibrosis [109]. A. capillaris belongs to the same genus as A. annua, has antioxidant, antiviral, and antifibrotic activities, and contains the same bioactive compounds, including scoparone and scopoletin [110,111]. Moreover, A. capillaris has been successfully applied in a formula as a complementary treatment for patients with pulmonary fibrosis at critical illness and recovery stages in Taiwan [112]. The present review describes the pharmaceutical properties and pharmacokinetics of bioactive compounds of A. capillaris. Future studies of A. capillaris, including verifying the pharmacokinetics of capillin and isochlorogenic acid, which have antioxidant, antiviral, and antitumor effects, alongside considerations of harvest time and different parts of the plant, are needed to inform the effective doses of A. capillaris for clinical applications.
Author Contributions
Conceptualization, T.-P.H. and T.-H.T.; methodology, T.-P.H. and T.-H.T.; investigation, T.-P.H. and W.-L.L.; data curation, T.-P.H. and W.-L.L.; writing—original draft preparation, T.-P.H.; writing—review and editing, T.-P.H., J.W.D., and T.-H.T.; supervision, T.-H.T.; project administration, T.-H.T.; funding acquisition, T.-H.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded in part by research grants from the Ministry of Science and Technology of Taiwan (MOST 110-2981-I-239-001; MOST 110-2113-M-A49A-503).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Tsai T.Y., Livneh H., Hung T.H., Lin I.H., Lu M.C., Yeh C.C. Associations between prescribed chinese herbal medicine and risk of hepatocellular carcinoma in patients with chronic hepatitis b: A nationwide population-based cohort study. BMJ Open. 2017;7:e014571. doi: 10.1136/bmjopen-2016-014571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ting C.T., Kuo C.J., Hu H.Y., Lee Y.L., Tsai T.H. Prescription frequency and patterns of chinese herbal medicine for liver cancer patients in taiwan: A cross-sectional analysis of the national health insurance research database. BMC Complement. Altern. Med. 2017;17:118. doi: 10.1186/s12906-017-1628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Liu C.Y., Chu J.Y., Chiang J.H., Yen H.R., Hsu C.H. Utilization and prescription patterns of traditional chinese medicine for patients with hepatitis c in taiwan: A population-based study. BMC Complement. Altern. Med. 2016;16:397. doi: 10.1186/s12906-016-1379-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lee T.Y., Chang H.H., Lo W.C., Lin H.C. Alleviation of hepatic oxidative stress by chinese herbal medicine yin-chen-hao-tang in obese mice with steatosis. Int. J. Mol. Med. 2010;25:837–844. doi: 10.3892/ijmm_00000412. [DOI] [PubMed] [Google Scholar]
- 5.Lee T.Y., Chang H.H., Kuo J.J., Shen J.J. Changes of hepatic proteome in bile duct ligated rats with hepatic fibrosis following treatment with yin-chen-hao-tang. Int. J. Mol. Med. 2009;23:477–484. doi: 10.3892/ijmm_00000154. [DOI] [PubMed] [Google Scholar]
- 6.Lee T.Y., Chang H.H., Wu M.Y., Lin H.C. Yin-chen-hao-tang ameliorates obstruction-induced hepatic apoptosis in rats. J. Pharm. Pharmacol. 2007;59:583–590. doi: 10.1211/jpp.59.4.0014. [DOI] [PubMed] [Google Scholar]
- 7.Lee T.Y., Chang H.H., Chen J.H., Hsueh M.L., Kuo J.J. Herb medicine yin-chen-hao-tang ameliorates hepatic fibrosis in bile duct ligation rats. J. Ethnopharmacol. 2007;109:318–324. doi: 10.1016/j.jep.2006.07.042. [DOI] [PubMed] [Google Scholar]
- 8.Wang X., Sun W., Sun H., Lv H., Wu Z., Wang P., Liu L., Cao H. Analysis of the constituents in the rat plasma after oral administration of yin chen hao tang by uplc/q-tof-ms/ms. J. Pharm. Biomed. Anal. 2008;46:477–490. doi: 10.1016/j.jpba.2007.11.014. [DOI] [PubMed] [Google Scholar]
- 9.Tang W., Eisenbrand G. Chinese Drugs of Plant Origin. Springer; Berlin/Heidelberg, Germany: 1992. Artemisia scoparia waldst. et kit. and A. capillaris thunb; pp. 179–183. [Google Scholar]
- 10.Ju I.-O., You D.-H., Song Y.-E., Jang I., Ryu J., Choi S.-R. Changes of major components and growth characteristics according to harvesting times of artemisia capillaris thunberg. Korean J. Med. Crop. Sci. 2007;15:189–193. [Google Scholar]
- 11.Hsueh T.-P., Tsai T.-H. Preclinical pharmacokinetics of scoparone, geniposide and rhein in an herbal medicine using a validated lc-ms/ms method. Molecules. 2018;23:2716. doi: 10.3390/molecules23102716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hsueh T.P., Tsai T.H. Preclinical study of simultaneous pharmacokinetic and pharmacodynamic herb-drug interactions between yin-chen-hao-tang and spironolactone. BMC Complement. Med. Ther. 2020;20:253. doi: 10.1186/s12906-020-03042-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhao Y., Geng C.A., Sun C.L., Ma Y.B., Huang X.Y., Cao T.W., He K., Wang H., Zhang X.M., Chen J.J. Polyacetylenes and anti-hepatitis b virus active constituents from artemisia capillaris. Fitoterapia. 2014;95:187–193. doi: 10.1016/j.fitote.2014.03.017. [DOI] [PubMed] [Google Scholar]
- 14.Zhao Y., Geng C.A., Ma Y.B., Huang X.Y., Chen H., Cao T.W., He K., Wang H., Zhang X.M., Chen J.J. Uflc/ms-it-tof guided isolation of anti-hbv active chlorogenic acid analogues from artemisia capillaris as a traditional chinese herb for the treatment of hepatitis. J. Ethnopharmacol. 2014;156:147–154. doi: 10.1016/j.jep.2014.08.043. [DOI] [PubMed] [Google Scholar]
- 15.Geng C.A., Yang T.H., Huang X.Y., Yang J., Ma Y.B., Li T.Z., Zhang X.M., Chen J.J. Anti-hepatitis b virus effects of the traditional chinese herb artemisia capillaris and its active enynes. J. Ethnopharmacol. 2018;224:283–289. doi: 10.1016/j.jep.2018.06.005. [DOI] [PubMed] [Google Scholar]
- 16.Han K.H., Jeon Y.J., Athukorala Y., Choi K.D., Kim C.J., Cho J.K., Sekikawa M., Fukushima M., Lee C.H. A water extract of artemisia capillaris prevents 2,2′-azobis(2-amidinopropane) dihydrochloride-induced liver damage in rats. J. Med. Food. 2006;9:342–347. doi: 10.1089/jmf.2006.9.342. [DOI] [PubMed] [Google Scholar]
- 17.Lee I.W., Choi H.Y., Lee J.H., Park S.D., Kim S.M., Ku S.K., Zhao R.J., Kim S.C., Kim Y.W., Choi H.S. Saeng-kankunbi-tang protects liver against oxidative damage through activation of erk/nrf2 pathway. Chin. J. Integr. Med. 2016;22:619–628. doi: 10.1007/s11655-016-2466-5. [DOI] [PubMed] [Google Scholar]
- 18.Kim K.S., Yang H.J., Lee J.Y., Na Y.C., Kwon S.Y., Kim Y.C., Lee J.H., Jang H.J. Effects of beta-sitosterol derived from artemisia capillaris on the activated human hepatic stellate cells and dimethylnitrosamine-induced mouse liver fibrosis. BMC Complement. Altern. Med. 2014;14:363. doi: 10.1186/1472-6882-14-363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Han J.M., Kim H.G., Choi M.K., Lee J.S., Lee J.S., Wang J.H., Park H.J., Son S.W., Hwang S.Y., Son C.G. Artemisia capillaris extract protects against bile duct ligation-induced liver fibrosis in rats. Exp. Toxicol. Pathol. 2013;65:837–844. doi: 10.1016/j.etp.2012.12.002. [DOI] [PubMed] [Google Scholar]
- 20.Choi M.K., Han J.M., Kim H.G., Lee J.S., Lee J.S., Wang J.H., Son S.W., Park H.J., Son C.G. Aqueous extract of artemisia capillaris exerts hepatoprotective action in alcohol-pyrazole-fed rat model. J. Ethnopharmacol. 2013;147:662–670. doi: 10.1016/j.jep.2013.03.065. [DOI] [PubMed] [Google Scholar]
- 21.Wang J.H., Choi M.K., Shin J.W., Hwang S.Y., Son C.G. Antifibrotic effects of artemisia capillaris and artemisia iwayomogi in a carbon tetrachloride-induced chronic hepatic fibrosis animal model. J. Ethnopharmacol. 2012;140:179–185. doi: 10.1016/j.jep.2012.01.007. [DOI] [PubMed] [Google Scholar]
- 22.Nan J.X., Park E.J., Kim H.J., Ko G., Sohn D.H. Antifibrotic effects of the methanol extract of polygonum aviculare in fibrotic rats induced by bile duct ligation and scission. Biol. Pharm. Bull. 2000;23:240–243. doi: 10.1248/bpb.23.240. [DOI] [PubMed] [Google Scholar]
- 23.Altekruse S.F., McGlynn K.A., Reichman M.E. Hepatocellular carcinoma incidence, mortality, and survival trends in the United States from 1975 to 2005. J. Clin. Oncol. 2009;27:1485–1491. doi: 10.1200/JCO.2008.20.7753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jang E., Kim S.Y., Lee N.R., Yi C.M., Hong D.R., Lee W.S., Kim J.H., Lee K.T., Kim B.J., Lee J.H., et al. Evaluation of antitumor activity of artemisia capillaris extract against hepatocellular carcinoma through the inhibition of il-6/stat3 signaling axis. Oncol. Rep. 2017;37:526–532. doi: 10.3892/or.2016.5283. [DOI] [PubMed] [Google Scholar]
- 25.Hong S.H., Seo S.H., Lee J.H., Choi B.T. The aqueous extract from artemisia capillaris thunb. Inhibits lipopolysaccharide-induced inflammatory response through preventing nf-kappab activation in human hepatoma cell line and rat liver. Int. J. Mol. Med. 2004;13:717–720. [PubMed] [Google Scholar]
- 26.Hu Y.Q., Tan R.X., Chu M.Y., Zhou J. Apoptosis in human hepatoma cell line smmc-7721 induced by water-soluble macromolecular components of artemisia capillaris thunberg. Jpn. J. Cancer Res. 2000;91:113–117. doi: 10.1111/j.1349-7006.2000.tb00867.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jung K.H., Rumman M., Yan H., Cheon M.J., Choi J.G., Jin X., Park S., Oh M.S., Hong S.S. An ethyl acetate fraction of artemisia capillaris (ace-63) induced apoptosis and anti-angiogenesis via inhibition of pi3k/akt signaling in hepatocellular carcinoma. Phytother. Res. 2018;32:2034–2046. doi: 10.1002/ptr.6135. [DOI] [PubMed] [Google Scholar]
- 28.Kim J., Jung K.H., Yan H.H., Cheon M.J., Kang S., Jin X., Park S., Oh M.S., Hong S.S. Artemisia capillaris leaves inhibit cell proliferation and induce apoptosis in hepatocellular carcinoma. BMC Complement. Altern. Med. 2018;18:147. doi: 10.1186/s12906-018-2217-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Eckel R.H., Grundy S.M., Zimmet P.Z. The metabolic syndrome. Lancet. 2005;365:1415–1428. doi: 10.1016/S0140-6736(05)66378-7. [DOI] [PubMed] [Google Scholar]
- 30.Nurul Islam M., Jung H.A., Sohn H.S., Kim H.M., Choi J.S. Potent alpha-glucosidase and protein tyrosine phosphatase 1b inhibitors from artemisia capillaris. Arch. Pharm. Res. 2013;36:542–552. doi: 10.1007/s12272-013-0069-7. [DOI] [PubMed] [Google Scholar]
- 31.Lim D.W., Kim Y.T., Jang Y.J., Kim Y.E., Han D. Anti-obesity effect of artemisia capillaris extracts in high-fat diet-induced obese rats. Molecules. 2013;18:9241–9252. doi: 10.3390/molecules18089241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liu L., Zhao J., Li Y., Wan Y., Lin J., Shen A., Xu W., Li H., Zhang Y., Xu J., et al. Artemisia capillaris formula inhibits hepatic steatosis via an mir122 induced decrease in fatty acid synthase expression in vivo and in vitro. Mol. Med. Rep. 2016;13:4751–4758. doi: 10.3892/mmr.2016.5131. [DOI] [PubMed] [Google Scholar]
- 33.Boden G. Obesity and free fatty acids. Endocrinol. Metab. Clin. N. Am. 2008;37:635. doi: 10.1016/j.ecl.2008.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jang E., Shin M.H., Kim K.S., Kim Y., Na Y.C., Woo H.J., Kim Y., Lee J.H., Jang H.J. Anti-lipoapoptotic effect of artemisia capillaris extract on free fatty acids-induced hepg2 cells. BMC Complement. Altern. Med. 2014;14:253. doi: 10.1186/1472-6882-14-253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Islam M.N., Ishita I.J., Jung H.A., Choi J.S. Vicenin 2 isolated from artemisia capillaris exhibited potent anti-glycation properties. Food Chem. Toxicol. 2014;69:55–62. doi: 10.1016/j.fct.2014.03.042. [DOI] [PubMed] [Google Scholar]
- 36.Kim Y., Lee I.S., Kim K.H., Park J., Lee J.H., Bang E., Jang H.J., Na Y.C. Metabolic profiling of liver tissue in diabetic mice treated with artemisia capillaris and alisma rhizome using lc-ms and ce-ms. Am. J. Chin. Med. 2016;44:1639–1661. doi: 10.1142/S0192415X16500920. [DOI] [PubMed] [Google Scholar]
- 37.Choi W.S., Kim Y.S., Park B.S., Kim J.E., Lee S.E. Hypolipidaemic effect of hericium erinaceum grown in artemisia capillaris on obese rats. Mycobiology. 2013;41:94–99. doi: 10.5941/MYCO.2013.41.2.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ha H., Lee H., Seo C.S., Lim H.-S., Lee J.K., Lee M.-Y., Shin H. Artemisia capillaris inhibits atopic dermatitis-like skin lesions in dermatophagoides farinae-sensitized nc/nga mice. BMC Complement. Altern. Med. 2014;14:100. doi: 10.1186/1472-6882-14-100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Son H.U., Lee S., Heo J.C., Lee S.H. The solid-state fermentation of artemisia capillaris leaves with ganoderma lucidum enhances the anti-inflammatory effects in a model of atopic dermatitis. Int. J. Mol. Med. 2017;39:1233–1241. doi: 10.3892/ijmm.2017.2945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lee S.Y., Nam S., Kim S., Koo J.S., Hong I.K., Kim H., Han S., Kang M., Yang H., Cho H.J. Therapeutic efficacies of artemisia capillaris extract cream formulation in imiquimod-induced psoriasis models. Evid. Based Complement. Alternat. Med. 2018;2018:3610494. doi: 10.1155/2018/3610494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kim Y.S., Bahn K.N., Hah C.K., Gang H.I., Ha Y.L. Inhibition of 7,12-dimethylbenz[a]anthracene induced mouse skin carcinogenesis by artemisia capillaris. J. Food Sci. 2008;73:T16–T20. doi: 10.1111/j.1750-3841.2007.00585.x. [DOI] [PubMed] [Google Scholar]
- 42.Yen M.H., Huang C.I., Lee M.S., Cheng Y.P., Hsieh C.J., Chiang L.C., Chang J.S. Artemisia capillaris inhibited enterovirus 71-induced cell injury by preventing viral internalization. Kaohsiung J. Med. Sci. 2018;34:150–159. doi: 10.1016/j.kjms.2017.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lee J.H., Park E.K., Uhm C.S., Chung M.S., Kim K.H. Inhibition of helicobacter pylori adhesion to human gastric adenocarcinoma epithelial cells by acidic polysaccharides from artemisia capillaris and panax ginseng. Planta Med. 2004;70:615–619. doi: 10.1055/s-2004-827183. [DOI] [PubMed] [Google Scholar]
- 44.Yeo D., Hwang S.J., Kim W.J., Youn H.-J., Lee H.-J. The aqueous extract from artemisia capillaris inhibits acute gastric mucosal injury by inhibition of ros and nf-kb. Biomed. Pharmacother. 2018;99:681–687. doi: 10.1016/j.biopha.2018.01.118. [DOI] [PubMed] [Google Scholar]
- 45.Park S.W., Oh T.Y., Kim Y.S., Sim H., Park S.J., Jang E.J., Park J.S., Baik H.W., Hahm K.B. Artemisia asiatica extracts protect against ethanol-induced injury in gastric mucosa of rats. J. Gastroenterol. Hepatol. 2008;23:976–984. doi: 10.1111/j.1440-1746.2008.05333.x. [DOI] [PubMed] [Google Scholar]
- 46.Yang C.-C., Lee M.-R., Hsu S.-L., Chang C.-M.J. Supercritical fluids extraction of capillarisin from artemisia capillaris and its inhibition of in vitro growth of hepatoma cells. J. Supercrit. Fluids. 2007;42:96–103. doi: 10.1016/j.supflu.2006.12.022. [DOI] [Google Scholar]
- 47.Sheu S.J., Chieh C.L., Weng W.C. Capillary electrophoretic determination of the constituents of artemisiae capillaris herba. J. Chromatogr. A. 2001;911:285–293. doi: 10.1016/S0021-9673(01)00513-1. [DOI] [PubMed] [Google Scholar]
- 48.Okuno I., Uchida K., Nakamura M., Sakurai K. Studies on choleretic consituents in artemisia capillaris thunb. Chem. Pharm. Bull. 1988;36:769–775. doi: 10.1248/cpb.36.769. [DOI] [PubMed] [Google Scholar]
- 49.Wu T.-S., Tsang Z.-J., Wu P.-L., Lin F.-W., Li C.-Y., Teng C.-M., Lee K.-H. New constituents and antiplatelet aggregation and anti-hiv principles of artemisia capillaris. Bioorg. Med. Chem. 2001;9:77–83. doi: 10.1016/S0968-0896(00)00225-X. [DOI] [PubMed] [Google Scholar]
- 50.Geng C.A., Huang X.Y., Chen X.L., Ma Y.B., Rong G.Q., Zhao Y., Zhang X.M., Chen J.J. Three new anti-hbv active constituents from the traditional chinese herb of yin-chen (artemisia scoparia) J. Ethnopharmacol. 2015;176:109–117. doi: 10.1016/j.jep.2015.10.032. [DOI] [PubMed] [Google Scholar]
- 51.Cai H., Song Y.H., Xia W.J., Jin M.W. Aqueous extract of yin-chen-hao decoction, a traditional chinese prescription, exerts protective effects on concanavalin a-induced hepatitis in mice through inhibition of nf-kappab. J. Pharm. Pharmacol. 2006;58:677–684. doi: 10.1211/jpp.58.5.0013. [DOI] [PubMed] [Google Scholar]
- 52.Cho H.R., Choi D.H., Ko B.K., Nam C.W., Park K.M., Lee Y.J., Lee S.G., Lee J.S., Lee K.A., Lee E.A., et al. Cold preservation of rat cultured hepatocytes: The scoparone effect. Transplant. Proc. 2000;32:2325–2327. doi: 10.1016/S0041-1345(00)01684-5. [DOI] [PubMed] [Google Scholar]
- 53.Jang S.I., Kim Y.J., Lee W.Y., Kwak K.C., Baek S.H., Kwak G.B., Yun Y.G., Kwon T.O., Chung H.T., Chai K.Y. Scoparone from artemisia capillaris inhibits the release of inflammatory mediators in raw 264.7 cells upon stimulation cells by interferon-gamma plus lps. Arch. Pharm. Res. 2005;28:203–208. doi: 10.1007/BF02977716. [DOI] [PubMed] [Google Scholar]
- 54.Noh J.R., Kim Y.H., Hwang J.H., Gang G.T., Yeo S.H., Kim K.S., Oh W.K., Ly S.Y., Lee I.K., Lee C.H. Scoparone inhibits adipocyte differentiation through down-regulation of peroxisome proliferators-activated receptor gamma in 3t3-l1 preadipocytes. Food Chem. 2013;141:723–730. doi: 10.1016/j.foodchem.2013.04.036. [DOI] [PubMed] [Google Scholar]
- 55.Yang D., Yang J., Shi D., Deng R., Yan B. Scoparone potentiates transactivation of the bile salt export pump gene and this effect is enhanced by cytochrome p450 metabolism but abolished by a pkc inhibitor. Br. J. Pharmacol. 2011;164:1547–1557. doi: 10.1111/j.1476-5381.2011.01522.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zhang A., Sun H., Dou S., Sun W., Wu X., Wang P., Wang X. Metabolomics study on the hepatoprotective effect of scoparone using ultra-performance liquid chromatography/electrospray ionization quadruple time-of-flight mass spectrometry. Analyst. 2013;138:353–361. doi: 10.1039/C2AN36382H. [DOI] [PubMed] [Google Scholar]
- 57.Lyu L., Chen J., Wang W., Yan T., Lin J., Gao H., Li H., Lv R., Xu F., Fang L. Scoparone alleviates ang ii-induced pathological myocardial hypertrophy in mice by inhibiting oxidative stress. J. Cell. Mol. Med. 2021;25:3136–3148. doi: 10.1111/jcmm.16304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Fu B., Su Y., Ma X., Mu C., Yu F. Scoparone attenuates angiotensin ii-induced extracellular matrix remodeling in cardiac fibroblasts. J. Pharmacol. Sci. 2018;137:110–115. doi: 10.1016/j.jphs.2018.05.006. [DOI] [PubMed] [Google Scholar]
- 59.Lv H., Sun H., Sun W., Liu L., Wang P., Wang X., Cao H. Pharmacokinetic studies of a chinese triple herbal drug formula. Phytomedicine. 2008;15:993–1001. doi: 10.1016/j.phymed.2008.01.006. [DOI] [PubMed] [Google Scholar]
- 60.Yin Q., Sun H., Zhang A., Wang X. Pharmacokinetics and tissue distribution study of scoparone in rats by ultraperformance liquid-chromatography with tandem high-definition mass spectrometry. Fitoterapia. 2012;83:795–800. doi: 10.1016/j.fitote.2012.03.010. [DOI] [PubMed] [Google Scholar]
- 61.Wang Y., Xing X., Cao Y., Zhao L., Sun S., Chen Y., Chai Y., Chen S., Zhu Z. Development and application of an uhplc-ms/ms method for comparative pharmacokinetic study of eight major bioactive components from yin chen hao tang in normal and acute liver injured rats. Evid. Based Complement. Alternat. Med. 2018;2018:1–12. doi: 10.1155/2018/3239785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zhang A., Sun H., Wang X., Jiao G., Yuan Y., Sun W. Simultaneous in vivo rp-hplc-dad quantification of multiple-component and drug-drug interaction by pharmacokinetics, using 6,7-dimethylesculetin, geniposide and rhein as examples. Biomed. Chromatogr. 2012;26:844–850. doi: 10.1002/bmc.1739. [DOI] [PubMed] [Google Scholar]
- 63.Wang X., Lv H., Sun H., Sun W., Liu L., Wang P., Cao H. Simultaneous determination of 6,7-dimethylesculetin and geniposide in rat plasma and its application to pharmacokinetic studies of yin chen hao tang preparation. Arzneimittelforschung. 2008;58:336–341. doi: 10.1055/s-0031-1296516. [DOI] [PubMed] [Google Scholar]
- 64.Kim J.K., Kim J.Y., Kim H.J., Park K.G., Harris R.A., Cho W.J., Lee J.T., Lee I.K. Scoparone exerts anti-tumor activity against du145 prostate cancer cells via inhibition of stat3 activity. PLoS ONE. 2013;8:e80391. doi: 10.1371/journal.pone.0080391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Choi R.Y., Ham J.R., Lee H.I., Cho H.W., Choi M.S., Park S.K., Lee J., Kim M.J., Seo K.I., Lee M.K. Scopoletin supplementation ameliorates steatosis and inflammation in diabetic mice. Phytother. Res. 2017;31:1795–1804. doi: 10.1002/ptr.5925. [DOI] [PubMed] [Google Scholar]
- 66.Jang J.H., Park J.E., Han J.S. Scopoletin inhibits alpha-glucosidase in vitro and alleviates postprandial hyperglycemia in mice with diabetes. Eur. J. Pharmacol. 2018;834:152–156. doi: 10.1016/j.ejphar.2018.07.032. [DOI] [PubMed] [Google Scholar]
- 67.Nam H., Kim M.M. Scopoletin has a potential activity for anti-aging via autophagy in human lung fibroblasts. Phytomedicine. 2015;22:362–368. doi: 10.1016/j.phymed.2015.01.004. [DOI] [PubMed] [Google Scholar]
- 68.Liu X.L., Zhang L., Fu X.L., Chen K., Qian B.C. Effect of scopoletin on pc3 cell proliferation and apoptosis. Acta Pharmacol. Sin. 2001;22:929–933. [PubMed] [Google Scholar]
- 69.Adams M., Efferth T., Bauer R. Activity-guided isolation of scopoletin and isoscopoletin, the inhibitory active principles towards ccrf-cem leukaemia cells and multi-drug resistant cem/adr5000 cells, from artemisia argyi. Planta Med. 2006;72:862–864. doi: 10.1055/s-2006-947165. [DOI] [PubMed] [Google Scholar]
- 70.Seo E.J., Saeed M., Law B.Y., Wu A.G., Kadioglu O., Greten H.J., Efferth T. Pharmacogenomics of scopoletin in tumor cells. Molecules. 2016;21:496. doi: 10.3390/molecules21040496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zeng Y., Li S., Wang X., Gong T., Sun X., Zhang Z. Validated lc-ms/ms method for the determination of scopoletin in rat plasma and its application to pharmacokinetic studies. Molecules. 2015;20:18988–19001. doi: 10.3390/molecules201018988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Chang Y., Zhang Q.-H., Li J., Zhang L., Guo X., He J., Zhang P., Ma L., Deng Y., Zhang B., et al. Simultaneous determination of scopoletin, psoralen, bergapten, xanthotoxin, columbianetin acetate, imperatorin, osthole and isoimperatorin in rat plasma by lc–ms/ms for pharmacokinetic studies following oral administration of radix angelicae pubescentis extract. J. Pharm. Biomed. Anal. 2013;77:71–75. doi: 10.1016/j.jpba.2012.12.031. [DOI] [PubMed] [Google Scholar]
- 73.Zhao Y.X., Wang M., Dong W.T., Li Y. Pharmacokinetics, bioavailability and metabolism of scopoletin in dog by ultra-high-performance liquid chromatography combined with linear ion trap-orbitrap tandem mass spectrometry. Biomed. Chromatogr. 2019;33:e4436. doi: 10.1002/bmc.4436. [DOI] [PubMed] [Google Scholar]
- 74.Kim J., Lim J., Kang B.Y., Jung K., Choi H.J. Capillarisin augments anti-oxidative and anti-inflammatory responses by activating nrf2/ho-1 signaling. Neurochem. Int. 2017;105:11–20. doi: 10.1016/j.neuint.2017.01.018. [DOI] [PubMed] [Google Scholar]
- 75.Yu Z., Tang L., Chen L., Li J., Wu W., Hu C. Capillarisin suppresses lipopolysaccharide-induced inflammatory mediators in bv2 microglial cells by suppressing tlr4-mediated nf-kappab and mapks signaling pathway. Neurochem. Res. 2015;40:1095–1101. doi: 10.1007/s11064-015-1567-4. [DOI] [PubMed] [Google Scholar]
- 76.Han S., Lee J.H., Kim C., Nam D., Chung W.S., Lee S.G., Ahn K.S., Cho S.K., Cho M., Ahn K.S. Capillarisin inhibits inos, cox-2 expression, and proinflammatory cytokines in lps-induced raw 264.7 macrophages via the suppression of erk, jnk, and nf-kappab activation. Immunopharmacol. Immunotoxicol. 2013;35:34–42. doi: 10.3109/08923973.2012.736522. [DOI] [PubMed] [Google Scholar]
- 77.Kim M., Chun J., Jung H.A., Choi J.S., Kim Y.S. Capillarisin attenuates exercise-induced muscle damage through mapk and nf-kappab signaling. Phytomedicine. 2017;32:30–36. doi: 10.1016/j.phymed.2017.04.007. [DOI] [PubMed] [Google Scholar]
- 78.Tsui K.H., Chang Y.L., Yang P.S., Hou C.P., Lin Y.H., Lin B.W., Feng T.H., Juang H.H. The inhibitory effects of capillarisin on cell proliferation and invasion of prostate carcinoma cells. Cell Prolif. 2018;51:e12429. doi: 10.1111/cpr.12429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Tsui K.H., Chang Y.L., Feng T.H., Hou C.P., Lin Y.H., Yang P.S., Lee B.W., Juang H.H. Capillarisin blocks prostate-specific antigen expression on activation of androgen receptor in prostate carcinoma cells. Prostate. 2018;78:242–249. doi: 10.1002/pros.23463. [DOI] [PubMed] [Google Scholar]
- 80.Yi Y.-X., Ding Y., Zhang Y., Ma N.-H., Shi F., Kang P., Cai Z.-Z., Zhang T. Yinchenhao decoction ameliorates alpha-naphthylisothiocyanate induced intrahepatic cholestasis in rats by regulating phase ii metabolic enzymes and transporters. Front. Pharmacol. 2018;9:510. doi: 10.3389/fphar.2018.00510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wang X., Sun H., Zhang A., Jiao G., Sun W., Yuan Y. Pharmacokinetics screening for multi-components absorbed in the rat plasma after oral administration traditional chinese medicine formula yin-chen-hao-tang by ultra performance liquid chromatography-electrospray ionization/quadrupole-time-of-flight mass spectrometry combined with pattern recognition methods. Analyst. 2011;136:5068–5076. doi: 10.1039/c1an15752c. [DOI] [PubMed] [Google Scholar]
- 82.Wang H., Zou H., Ni J., Kong L., Gao S., Guo B. Fractionation and analysis of artemisia capillaris thunb. By affinity chromatography with human serum albumin as stationary phase. J. Chromatogr. A. 2000;870:501–510. doi: 10.1016/S0021-9673(99)01062-6. [DOI] [PubMed] [Google Scholar]
- 83.Ogasawara M., Matsubara T., Suzuki H. Screening of natural compounds for inhibitory activity on colon cancer cell migration. Biol. Pharm. Bull. 2001;24:720–723. doi: 10.1248/bpb.24.720. [DOI] [PubMed] [Google Scholar]
- 84.Whelan L.C., Ryan M.F. Effects of the polyacetylene capillin on human tumour cell lines. Anticancer Res. 2004;24:2281–2286. [PubMed] [Google Scholar]
- 85.Masuda Y., Asada K., Satoh R., Takada K., Kitajima J. Capillin, a major constituent of artemisia capillaris thunb. Flower essential oil, induces apoptosis through the mitochondrial pathway in human leukemia hl-60 cells. Phytomedicine. 2015;22:545–552. doi: 10.1016/j.phymed.2015.03.008. [DOI] [PubMed] [Google Scholar]
- 86.Islam M.N., Choi R.J., Jung H.A., Oh S.H., Choi J.S. Promising anti-diabetic potential of capillin and capillinol isolated from artemisia capillaris. Arch. Pharm. Res. 2016;39:340–349. doi: 10.1007/s12272-016-0715-y. [DOI] [PubMed] [Google Scholar]
- 87.Lee S.H., Lee J.Y., Kwon Y.I., Jang H.D. Anti-osteoclastic activity of artemisia capillaris thunb. Extract depends upon attenuation of osteoclast differentiation and bone resorption-associated acidification due to chlorogenic acid, hyperoside, and scoparone. Int. J. Mol. Sci. 2017;18:322. doi: 10.3390/ijms18020322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Budryn G., Zaczyńska D., Oracz J. Effect of addition of green coffee extract and nanoencapsulated chlorogenic acids on aroma of different food products. LWT. 2016;73:197–204. doi: 10.1016/j.lwt.2016.06.019. [DOI] [Google Scholar]
- 89.Patti A.M., Al-Rasadi K., Katsiki N., Banerjee Y., Nikolic D., Vanella L., Giglio R.V., Giannone V.A., Montalto G., Rizzo M. Effect of a natural supplement containing curcuma longa, guggul, and chlorogenic acid in patients with metabolic syndrome. Angiology. 2015;66:856–861. doi: 10.1177/0003319714568792. [DOI] [PubMed] [Google Scholar]
- 90.Suzuki A., Yamamoto N., Jokura H., Yamamoto M., Fujii A., Tokimitsu I., Saito I. Chlorogenic acid attenuates hypertension and improves endothelial function in spontaneously hypertensive rats. J. Hypertens. 2006;24:1065–1073. doi: 10.1097/01.hjh.0000226196.67052.c0. [DOI] [PubMed] [Google Scholar]
- 91.Ahrens M.J., Thompson D.L. Effect of emulin on blood glucose in type 2 diabetics. J. Med. Food. 2013;16:211–215. doi: 10.1089/jmf.2012.0069. [DOI] [PubMed] [Google Scholar]
- 92.Lee K.-W., Im J.-Y., Woo J.-M., Grosso H., Kim Y.-S., Cristovao A.C., Sonsalla P.K., Schuster D.S., Jalbut M.M., Fernandez J.R. Neuroprotective and anti-inflammatory properties of a coffee component in the mptp model of parkinson’s disease. Neurotherapeutics. 2013;10:143–153. doi: 10.1007/s13311-012-0165-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Shi H., Dong L., Dang X., Liu Y., Jiang J., Wang Y., Lu X., Guo X. Effect of chlorogenic acid on lps-induced proinflammatory signaling in hepatic stellate cells. Inflamm. Res. 2013;62:581–587. doi: 10.1007/s00011-013-0610-7. [DOI] [PubMed] [Google Scholar]
- 94.Silva B.A., Ferreres F., Malva J.O., Dias A.C. Phytochemical and antioxidant characterization of hypericum perforatum alcoholic extracts. Food Chem. 2005;90:157–167. doi: 10.1016/j.foodchem.2004.03.049. [DOI] [Google Scholar]
- 95.Huang K., Liang X.C., Zhong Y.L., He W.Y., Wang Z. 5-caffeoylquinic acid decreases diet-induced obesity in rats by modulating pparalpha and lxralpha transcription. J. Sci. Food Agric. 2015;95:1903–1910. doi: 10.1002/jsfa.6896. [DOI] [PubMed] [Google Scholar]
- 96.Liang N., Kitts D.D. Role of chlorogenic acids in controlling oxidative and inflammatory stress conditions. Nutrients. 2015;8:16. doi: 10.3390/nu8010016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Yamagata K., Izawa Y., Onodera D., Tagami M. Chlorogenic acid regulates apoptosis and stem cell marker-related gene expression in a549 human lung cancer cells. Mol. Cell. Biochem. 2018;441:9–19. doi: 10.1007/s11010-017-3171-1. [DOI] [PubMed] [Google Scholar]
- 98.Liu R., Lai K., Xiao Y., Ren J. Comparative pharmacokinetics of chlorogenic acid in beagles after oral administrations of single compound, the extracts of lonicera japanica, and the mixture of chlorogenic acid, baicalin, and forsythia suspense. Pharm. Biol. 2017;55:1234–1238. doi: 10.1080/13880209.2017.1296002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Gong X., Luan Q., Zhou X., Zhao Y., Zhao C. Uhplc-esi-ms/ms determination and pharmacokinetics of pinoresinol glucoside and chlorogenic acid in rat plasma after oral administration of eucommia ulmoides oliv extract. Biomed. Chromatogr. 2017;31:e4008. doi: 10.1002/bmc.4008. [DOI] [PubMed] [Google Scholar]
- 100.Gu P., Liu R.-J., Cheng M.-L., Wu Y., Zheng L., Liu Y.-J., Ma P.-C., Ding L. Simultaneous quantification of chlorogenic acid and taurocholic acid in human plasma by lc-ms/ms and its application to a pharmacokinetic study after oral administration of shuanghua baihe tablets. Chin. J. Nat. Med. 2016;14:313–320. doi: 10.1016/S1875-5364(16)30034-6. [DOI] [PubMed] [Google Scholar]
- 101.Gonthier M.-P., Verny M.-A., Besson C., Rémésy C., Scalbert A. Chlorogenic acid bioavailability largely depends on its metabolism by the gut microflora in rats. J. Nutr. 2003;133:1853–1859. doi: 10.1093/jn/133.6.1853. [DOI] [PubMed] [Google Scholar]
- 102.Tang D., Li H.J., Chen J., Guo C.W., Li P. Rapid and simple method for screening of natural antioxidants from chinese herb flos lonicerae japonicae by dpph-hplc-dad-tof/ms. J. Sep. Sci. 2008;31:3519–3526. doi: 10.1002/jssc.200800173. [DOI] [PubMed] [Google Scholar]
- 103.Chen J., Mangelinckx S., Ma L., Wang Z., Li W., De Kimpe N. Caffeoylquinic acid derivatives isolated from the aerial parts of gynura divaricata and their yeast alpha-glucosidase and ptp1b inhibitory activity. Fitoterapia. 2014;99:1–6. doi: 10.1016/j.fitote.2014.08.015. [DOI] [PubMed] [Google Scholar]
- 104.Hao B.J., Wu Y.H., Wang J.G., Hu S.Q., Keil D.J., Hu H.J., Lou J.D., Zhao Y. Hepatoprotective and antiviral properties of isochlorogenic acid a from laggera alata against hepatitis b virus infection. J. Ethnopharmacol. 2012;144:190–194. doi: 10.1016/j.jep.2012.09.003. [DOI] [PubMed] [Google Scholar]
- 105.Cao Z., Ding Y., Cao L., Ding G., Wang Z., Xiao W. Isochlorogenic acid c prevents enterovirus 71 infection via modulating redox homeostasis of glutathione. Sci. Rep. 2017;7:16278. doi: 10.1038/s41598-017-16446-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Liu X., Huang K., Niu Z., Mei D., Zhang B. Protective effect of isochlorogenic acid b on liver fibrosis in non-alcoholic steatohepatitis of mice. Basic Clin. Pharmacol. Toxicol. 2019;124:144–153. doi: 10.1111/bcpt.13122. [DOI] [PubMed] [Google Scholar]
- 107.Tian Y., Li Q., Zhou X., Pang Q., Xu Y. A uhplc-ms/ms method for simultaneous determination of twelve constituents from erigeron breviscapus extract in rat plasma: Application to a pharmacokinetic study. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2017;1046:1–12. doi: 10.1016/j.jchromb.2017.01.020. [DOI] [PubMed] [Google Scholar]
- 108.Huang L.H., Xiong X.H., Zhong Y.M., Cen M.F., Cheng X.G., Wang G.X., Zang L.Q., Wang S.J. Pharmacokinetics of isochlorgenic acid c in rats by hplc-ms: Absolute bioavailability and dose proportionality. J. Ethnopharmacol. 2016;185:105–109. doi: 10.1016/j.jep.2016.03.019. [DOI] [PubMed] [Google Scholar]
- 109.Haq F.U., Roman M., Ahmad K., Rahman S.U., Shah S.M.A., Suleman N., Ullah S., Ahmad I., Ullah W. Artemisia annua: Trials are needed for covid-19. Phytother. Res. 2020;34:2423–2424. doi: 10.1002/ptr.6733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Zhang X., Zhao Y., Guo L., Qiu Z., Huang L., Qu X. Differences in chemical constituents of artemisia annua l from different geographical regions in china. PLoS ONE. 2017;12:e0183047. doi: 10.1371/journal.pone.0183047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Lang S.J., Schmiech M., Hafner S., Paetz C., Steinborn C., Huber R., Gaafary M.E., Werner K., Schmidt C.Q., Syrovets T., et al. Antitumor activity of an artemisia annua herbal preparation and identification of active ingredients. Phytomedicine. 2019;62:152962. doi: 10.1016/j.phymed.2019.152962. [DOI] [PubMed] [Google Scholar]
- 112.Tsai K.C., Huang Y.C., Liaw C.C., Tsai C.I., Chiou C.T., Lin C.J., Wei W.C., Lin S.J., Tseng Y.H., Yeh K.M., et al. A traditional chinese medicine formula nricm101 to target covid-19 through multiple pathways: A bedside-to-bench study. Biomed. Pharmacother. 2021;133:111037. doi: 10.1016/j.biopha.2020.111037. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.