Abstract
Evans syndrome presents as concurrent autoimmune hemolytic anemia (AIHA) and immune thrombocytopenia (ITP). Systemic lupus erythematosus (SLE) is the most frequent autoimmune disorder associated with Evans syndrome. We herein report a case of new-onset Evans syndrome associated with SLE after BNT162b2 mRNA coronavirus disease 2019 (COVID-19) vaccination in a 53-year-old woman. Blood examination at diagnosis showed hemolytic anemia with a positive Coombs test and thrombocytopenia. Hypocomplementemia and the presence of lupus anticoagulant indicated a strong association with SLE. Prednisolone administration rapidly restored hemoglobin level and platelet count. This case suggests that mRNA COVID-19 vaccination may cause an autoimmune disorder. Physicians should be aware of this adverse reaction by mRNA COVID-19 vaccination and should consider the benefits and risks of vaccination for each recipient.
Keywords: Evans syndrome, COVID-19 mRNA vaccine, ITP, AIHA, SLE
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has caused instability of medical and social systems worldwide. Vaccines are expected to prevent further spread and aggravation of COVID-19. BNT162b2 mRNA vaccine is a lipid nanoparticle-encapsulated mRNA-based vaccine that encodes the full-length S protein of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1, 2]. In a phase 3 trial, this vaccine showed 95% efficacy in preventing symptomatic SARS-CoV-2 infection [1]. On the other hands, recent studies have indicated that mRNA vaccination against COVID-19 is associated with onset and progression of autoimmune hematologic disorders, especially immune thrombocytopenia [3–5].
Evans syndrome is characterized by the coexistence of autoimmune hemolytic anemia (AIHA) and immune thrombocytopenia (ITP). Evans syndrome is a rare condition that presents to 7% of AIHA and around 2% of ITP [6, 7]. Secondary Evans syndrome has been shown to be associated with autoimmune disorders, hematological malignancies, infections, and pregnancy [8]. Previous studies showed that the incidence of autoimmune disorders in the patients with Evans syndrome were 18.2 and 20% and that systemic lupus erythematosus (SLE) was the most frequent autoimmune disorder [6, 8]. Here, we describe a case of new-onset Evans syndrome with SLE after BNT162b2 mRNA COVID-19 vaccination in a 53-year-old woman.
Case report
A 53-year-old Japanese woman presented with shortness of breath and was admitted to our hospital. She had medical histories of bronchial asthma, Vogt–Koyanagi–Harada disease, and Hashimoto disease. Anemia and thrombocytopenia were not observed in a routine checkup 3 months before admission. She had received two doses of BNT162b2 mRNA COVID-19 vaccine (Pfizer®) at five weeks and two weeks before admission. After the first dose of vaccination, purpura in her extremities and aggravation of wheezing had transiently appeared for a few days. After the second dose of vaccination, a routine checkup showed thrombocytopenia (platelet count of 7.6 × 104/µL) with mild anemia (hemoglobin level of 11.0 g/dL).
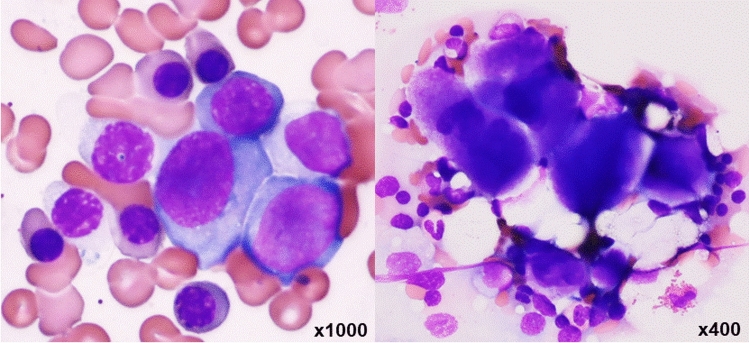
On examination, no fever or decrease in oxygen saturation was observed. Her skin and bulbar conjunctiva were yellowing. The palpebral conjunctiva was anemic and wheezing at inspiration was heard. There was no erythema, oral ulcer, arthritis, or lymphadenopathy. Laboratory test results are shown in Table 1. Complete blood count (CBC) revealed white blood cells (WBCs) at 5,330 /µL, red blood cells (RBCs) at 180 × 104/µL, hemoglobin (Hb) at 6.9 g/dL, platelets at 3.9 × 104/µL and number of reticulocytes at 36.54 × 104/µL. Serum tests revealed elevated indirect bilirubin (6.1 mg/dL), elevated lactate dehydrogenase (771 U/L), and low haptoglobin (3 mg/dL). Decline of renal function and proteinuria were not detected. Prolonged APTT (70.3 s) and positivity of lupus coagulant were observed. Anti-nuclear antibody was positive (160 × ) and its types were speckled and nucleolar. Both direct and indirect Coombs tests were positive, whereas the level of cold agglutinin was low. Schistocytes were not observed in a peripheral blood smear and activity of disintegrin-like and metalloproteinase with thrombospondin type 1 motifs 13 (ADAMTS13) was normal. Clone of paroxysmal nocturnal hemoglobinuria (PNH) was negative. Immunoglobulin G of COVID-19 measured by a chemiluminescent immunoassay was elevated to 27,201.7 AU/ml (assay cutoff < 50). In a bone marrow examination, M/E ratio was 0.2 and megakaryocyte count was 78/µL. Erythroid hyperplasia with megaloblastic change and megakaryocytes without dysplasia were observed (Fig. 1). The findings of enhanced computed tomography (CT) did not indicate a neoplasm and thrombosis.
Table 1.
Laboratory data on admission
| Assay | Standard value | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| WBC | 5.330 | /µL | TP | 7.6 | g/dL | C3 | 38 | mg/dL | 80—140 |
| Neutro | 52 | % | Alb | 4.3 | g/dL | C4 | 3.1 | mg/dL | 11.0—34.0 |
| Lym | 34 | % | Total-Bil | 6.4 | mg/dL | CH50 | 7 | U/mL | 30—45 |
| Mono | 11 | % | Indirect-Bil | 6.1 | mg/dL | PA-IgG | 259.2 | ng/L | < 27.6 |
| Baso | 2 | % | AST | 50 | IU/L | COVID-19 IgG | 27,201.7 | AU/mL | < 50 |
| Eosino | 1 | % | ALT | 32 | IU/L | ANA | 160 | x | < 40 |
| RBC | 180 | × 104/µL | ALP | 346 | IU/L | Homogeneous | < 40 | x | < 40 |
| Hb | 6.9 | g/dL | LDH | 771 | IU/L | Speckled | 160 | x | < 40 |
| Ht | 22 | % | BUN | 9.1 | mg/dL | Nucleolar | 160 | x | < 40 |
| Plt | 3.9 | × 104/µL | Cr | 0.61 | mg/dL | Centromere | < 40 | x | < 40 |
| Reticulocyte | 36.54 | × 104/µL | Haptoglobin | 3 | mg/dL | Peripheral | < 40 | x | < 40 |
| CRP | 0.43 | mg/dL | LA | 2 | < 1.3 | ||||
| PT | 12.4 | sec | aCL-β2GP1 | < 1.3 | U/mL | < 3.5 | |||
| PT-INR | 1.07 | Coombs test | |||||||
| APTT | 70.3 | sec | Direct | positive | |||||
| Fibrinogen | 298 | mg/dL | Indirect | positive | |||||
| FDP | 6 | µg/dL | CA | 4 | x | < 256 | |||
PA-IgG platelet-associated Immunoglobulin G; ANA anti-nuclear antibody; CA cold agglutination; LA lupus anticoagulant; aCL-β2GP1 anticardiolipin β2-glycoprotein-1 complex antibody
Fig. 1.
Bone marrow smear showed erythroid hyperplasia with megaloblastic change and megakaryocytes without dysplasia
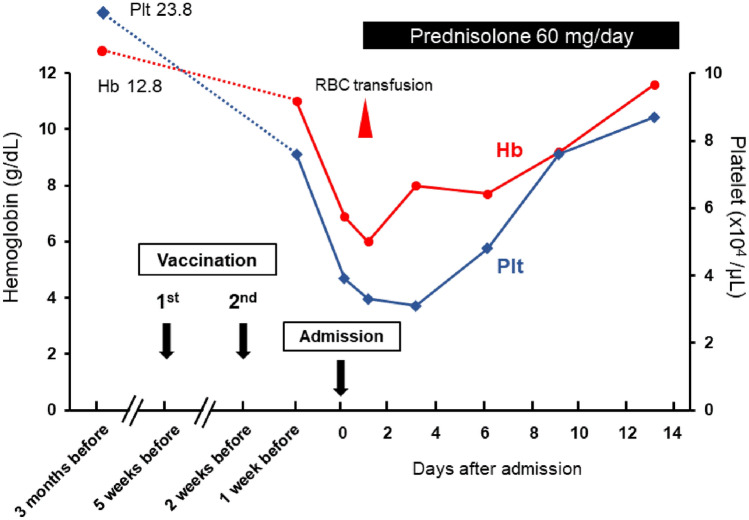
We assumed the development of Evans syndrome associated with SLE and exacerbation of bronchial asthma following mRNA COVID-19 vaccination. She met two classification criteria for SLE: hematological findings (hemolytic anemia and thrombocytopenia), positivity of lupus anticoagulant, and hypocomplementemia [9, 10]. Prednisolone at 60 mg (1 mg/kg) per day was administered two days after hospitalization. Wheezing immediately disappeared. Hemoglobin level and platelet count were restored day by day with only once RBC transfusion (Fig. 2). In parallel, lactate dehydrogenase, indirect bilirubin, and reticulocyte percentage gradually decreased. Antibodies of anti-Sm and anti-double-stranded DNA, which we tested after administration of prednisolone, were negative. Consequently, we plan to follow-up the patient while tapering the dose of prednisolone and consulting with a rheumatologist.
Fig. 2.
Clinical course. Anemia and thrombocytopenia were not observed in a routine checkup 3 months before admission. Hemoglobin level and platelet count rapidly decreased after vaccination and were gradually restored by administration of prednisolone
Discussion
COVID-19, which is caused by SARS-CoV-2, has spread worldwide. While mRNA COVID-19 vaccination is absolutely essential for preventing COVID-19 [1, 11], autoimmune cytopenia following COVID-19 vaccination has been increasingly reported. Most studies demonstrated the occurrence of thrombocytopenia or progression of ITP [3–5]. Two recent studies have presented cases of AIHA two days and three weeks after receiving the second dose of the mRNA COVID-19 vaccine [12, 13]. In addition, hemolytic attack was observed in 6 patients with PNH after vaccination [14].
Evans syndrome, which is a rare condition characterized by the coexistence of AIHA and ITP, was proposed early in 1951 [15]. It is diagnosed by ruling out other causative disorders [16], whereas it is associated with systemic lupus erythematosus (SLE), lymphoproliferative disorders, or primary immune deficiencies. A previous study including 34 patients with secondary Evans syndrome showed that 14 patients had autoimmune disorders and 10 of those patients had SLE [8]. In this case, immunological findings including hypocomplementemia and the presence of lupus anticoagulant indicated a strong association with SLE. The patient’s past histories (bronchial asthma, Vogt–Koyanagi–Harada disease, and Hashimoto disease) are generally categorized as autoimmune diseases. An overactive immune response after mRNA COVID-19 vaccination may be related to the onset of Evans syndrome associated with SLE. New-onset SLE after COVID-19 vaccination has already been reported [17]. However, another study showed that autoimmune disease flare occurred in only one patient in 280 patients [18]. A case of Evans syndrome following influenza vaccination was reported [19]. Some cases of onset or progression of Evans syndrome in patients with COVID-19 were reported [20–23]. Although the exact mechanism underlying the relationship between Evans syndrome and vaccination is unclear at present, vaccination for patients with Evans syndrome is should be positively considered for prevention of COVID-19.
This case suggests that mRNA COVID-19 vaccine can cause an autoimmune disorder. For patients with autoimmune hematological conditions, close monitoring is recommended 1–4 days after each dose of the double-dose COVID-19 vaccine series [24]. Physicians need to carefully determine the indication for vaccination and inform patients with autoimmune hematologic disorders about the possibility of disease progression. Further studies are required to clarify the mechanism of immune reaction and improve the safety of mRNA COVID-19 vaccination.
Declarations
Conflict of interest
The authors have no conflicts of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383:2603–2615. doi: 10.1056/NEJMoa2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Roest S, Hoek RAS, Manintveld OC. BNT162b2 mRNA Covid-19 vaccine in a nationwide mass vaccination setting. N Engl J Med. 2021;384:1968–1970. doi: 10.1056/NEJMc2104281. [DOI] [PubMed] [Google Scholar]
- 3.Ganzel C, Ben-Chetrit E. Immune thrombocytopenia following the Pfizer-BioNTech BNT162b2 mRNA COVID-19 vaccine. Isr Med Assoc J. 2021;23:341. [PubMed] [Google Scholar]
- 4.Lee EJ, Cines DB, Gernsheimer T, Kessler C, Michel M, Tarantino MD, et al. Thrombocytopenia following Pfizer and Moderna SARS-CoV-2 vaccination. Am J Hematol. 2021;96:534–537. doi: 10.1002/ajh.26132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wise J. Covid-19: AstraZeneca vaccine linked with small risk of ITP, real world data show. BMJ. 2021;373:n1489. doi: 10.1136/bmj.n1489. [DOI] [PubMed] [Google Scholar]
- 6.Hansen DL, Moller S, Andersen K, Gaist D, Frederiksen H. Evans syndrome in adults: incidence, prevalence, and survival in a nationwide cohort. Am J Hematol. 2019;94:1081–1090. doi: 10.1002/ajh.25574. [DOI] [PubMed] [Google Scholar]
- 7.Barcellini W, Fattizzo B, Zaninoni A, Radice T, Nichele I, Di Bona E, et al. Clinical heterogeneity and predictors of outcome in primary autoimmune hemolytic anemia: a GIMEMA study of 308 patients. Blood. 2014;124:2930–2936. doi: 10.1182/blood-2014-06-583021. [DOI] [PubMed] [Google Scholar]
- 8.Michel M, Chanet V, Dechartres A, Morin AS, Piette JC, Cirasino L, et al. The spectrum of Evans syndrome in adults: new insight into the disease based on the analysis of 68 cases. Blood. 2009;114:3167–3172. doi: 10.1182/blood-2009-04-215368. [DOI] [PubMed] [Google Scholar]
- 9.Aringer M, Costenbader K, Daikh D, Brinks R, Mosca M, Ramsey-Goldman R, et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Ann Rheum Dis. 2019;78:1151–1159. doi: 10.1136/annrheumdis-2018-214819. [DOI] [PubMed] [Google Scholar]
- 10.Petri M, Orbai AM, Alarcon GS, Gordon C, Merrill JT, Fortin PR, et al. Derivation and validation of the systemic lupus international collaborating clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012;64:2677–2686. doi: 10.1002/art.34473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Baden LR, El Sahly HM, Essink B, Kotloff K, Frey S, Novak R, et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med. 2021;384:403–416. doi: 10.1056/NEJMoa2035389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brito S, Ferreira N, Mateus S, Bernardo M, Pinto B, Lourenco A, et al. A case of autoimmune hemolytic anemia following COVID-19 messenger ribonucleic acid vaccination. Cureus. 2021;13:e15035. doi: 10.7759/cureus.15035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Murdych TM. A case of severe autoimmune hemolytic anemia after a receipt of a first dose of SARS-CoV-2 vaccine. Int J Lab Hematol. 2021 doi: 10.1111/ijlh.13653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gerber GF, Yuan X, Yu J, Cher BAY, Braunstein EM, Chaturvedi S, et al. COVID-19 vaccines induce severe hemolysis in paroxysmal nocturnal hemoglobinuria. Blood. 2021;137:3670–3673. doi: 10.1182/blood.2021011548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Evans RS, Takahashi K, Duane RT, Payne R, Liu C. Primary thrombocytopenic purpura and acquired hemolytic anemia; evidence for a common etiology. AMA Arch Intern Med. 1951;87:48–65. doi: 10.1001/archinte.1951.03810010058005. [DOI] [PubMed] [Google Scholar]
- 16.Norton A, Roberts I. Management of Evans syndrome. Br J Haematol. 2006;132:125–137. doi: 10.1111/j.1365-2141.2005.05809.x. [DOI] [PubMed] [Google Scholar]
- 17.Nune A, Iyengar KP, Ish P, Varupula B, Musat CA, Sapkota HR. Emergence of new onset SLE following SARS-CoV-2 vaccination. QJM. 2021 doi: 10.1093/qjmed/hcab229. [DOI] [PubMed] [Google Scholar]
- 18.Gaur P, Agrawat H, Shukla A. COVID-19 vaccine hesitancy in patients with systemic autoimmune rheumatic disease: an interview-based survey. Rheumatol Int. 2021;41:1601–1605. doi: 10.1007/s00296-021-04938-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shlamovitz GZ, Johar S. A case of Evans’ syndrome following influenza vaccine. J Emerg Med. 2013;44:e149–e151. doi: 10.1016/j.jemermed.2012.01.060. [DOI] [PubMed] [Google Scholar]
- 20.Vadlamudi G, Hong L, Keerthy M. Evans syndrome associated with pregnancy and COVID-19 infection. Case Rep Obstet Gynecol. 2020;2020:8862545. doi: 10.1155/2020/8862545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zarza J, Von Horoch J, Aguayo N, Baez E. Evans syndrome associated with antiphospholipid antibodies in a patient with SARS-COV-2 infection. Hematol Transfus Cell Ther. 2020;42:309–312. doi: 10.1016/j.htct.2020.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Demir NA, Basturk A, Ural O, Sumer S, Erdogdu B, Kiratli HE, et al. A case of Evans syndrome secondary to COVID-19. Blood Transfus. 2021;19:85–88. doi: 10.2450/2020.0221-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Georgy JT, Jayakaran JAJ, Jacob AS, Gunasekaran K, Korula PJ, Devasia AJ, et al. Evans syndrome and immune thrombocytopenia in two patients with COVID-19. J Med Virol. 2021;93:2642–2644. doi: 10.1002/jmv.26906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Portuguese AJ, Sunga C, Kruse-Jarres R, Gernsheimer T, Abkowitz J. Autoimmune- and complement-mediated hematologic condition recrudescence following SARS-CoV-2 vaccination. Blood Adv. 2021;5:2794–2798. doi: 10.1182/bloodadvances.2021004957. [DOI] [PMC free article] [PubMed] [Google Scholar]