Abstract
Invasive fungal disease (IFD) is a common cause of morbidity and mortality in patients with hematologic malignancies, especially among those undergoing allogeneic hematopoietic stem cell transplantation (HSCT). The epidemiology of IFD in HSCT patients has been evolving over the last decades, mainly in relation to changes in HSCT therapies such as antifungal prophylaxis. A progressive decrease in Candida albicans infection has been documented, alongside a progressive increase in infections caused by non-albicans Candida species, filamentous fungi, and/or multidrug-resistant fungi. Currently, the most frequent IFD is invasive aspergillosis. In some parts of the world, especially in north Central Europe, a high percentage of Aspergillus fumigatus isolates are azole-resistant. New diagnostic techniques have documented the existence of cryptic Aspergillus species with specific characteristics. An increase in mucormycosis and fusariosis diagnoses, as well as diagnoses of other rare fungi, have also been described. IFD epidemiology is likely to continue changing further due to both an increased use of mold-active antifungals and a lengthened survival of patients with HSCT that may result in hosts with weaker immune systems. Improvements in microbiology laboratories and the widespread use of molecular diagnostic tools will facilitate more precise descriptions of current IFD epidemiology. Additionally, rising resistance to antifungal drugs poses a major threat. In this scenario, knowledge of current epidemiology and accurate IFD diagnoses are mandatory in order to establish correct prophylaxis guidelines and appropriate early treatments.
Keywords: antifungal, fungal infection, immunosuppression, invasive fungal disease, molds, mortality, pneumonia, prophylaxis, risk factors, yeasts
1. Introduction
Invasive fungal disease (IFD) is a common cause of morbidity and mortality in patients with hematologic malignancies, especially among those undergoing allogenic hematopoietic stem cell transplants (HSCT). The annual number of HSCT procedures has continuously been rising [1], even in lower income countries, since its inception in the late 1950s. Most importantly, older patients and those with more comorbidities are increasingly undergoing HSCT. This, together with the lengthened survival of these patients, is significantly raising the number of patients susceptible to opportunistic infections.
IFD epidemiology in HSCT is in continuous evolution due to changes in host and transplant characteristics, including antifungal pressure, and diagnostic improvements. The widespread use of antifungal agents as both prophylaxis and treatment, as well as in agriculture, has led to a dramatic increase in antifungal resistance [2]. Improvements in non-invasive diagnostic tests and microbiology laboratories have resulted in a higher likelihood of patients being diagnosed with IFD. Comprehensive knowledge of fungal infection epidemiology in patients undergoing HSCT is essential in order to decide optimal antifungal prophylaxis and initiate empirical antifungal therapy early in patients with suggestive clinical presentations. In this review, we focus on recent literature that concerns epidemiological data on IFD in hematologic HSCT recipients, conducting a comprehensive literature search in the PubMed/MEDLINE database of all English-written articles with the following Mesh terms: (“Stem Cell Transplantation” OR “Hematopoietic Stem Cell Transplantation” OR “Bone Marrow Transplantation”) AND (“Invasive Fungal Infections” OR “Mycoses”).
2. IFD Diagnosis and Consensus Criteria
In 2002, a consensus group from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group (EORTC) and the Mycoses Study Group (MSG) published standard definitions of IFD for clinical and epidemiological research [3]. These definitions assigned three different levels of probability of IFD (proven, probable, and possible) on the basis of host, clinical, and microbiological criteria. Since then, these definitions have been updated twice, of which one was rather recent, mainly due to growing evidence and advances made in microbiological techniques [4,5].
Culture-based detection remains the gold standard for the diagnosis of IFD. However, proven infections require the specimen to be obtained by a sterile procedure from a normally sterile site (apart from fungemia mostly caused by yeasts). This is often difficult to perform in patients with severe thrombocytopenia. Similarly, histopathological detection of fungi in a sterile sample is also a criterion for proven IFD. In the last update of the EORTC/MSG consensus, amplification of fungal DNA by polymerase chain reaction (PCR) combined with DNA sequencing when fungi are seen in formalin-fixed paraffin-embedded tissue also come to form part of the criteria for proven diagnosis [5].
Another molecular technique that has grown in use is Aspergillus PCR. Such an approach has been shown to perform well for screening and diagnosis confirmation of aspergillosis in blood and bronchoalveolar lavage (BAL) fluid [6]. Additionally, Candida PCR has good sensitivity and specificity [7], although its use has not become widespread. Regarding candidemia, the US Food and Drug Administration have recent approval of an innovative approach that combines targeted PCR with T2 magnetic resonance. Called T2Candida, it has shown high sensitivity and specificity in detecting Candida species directly in blood specimens [8].
Biomarkers are essential in the current scenario of IFD diagnosis. Serum, BAL, and even cerebrospinal fluid detection of aspergillus galactomannan antigen (GM) are helpful markers in diagnosing invasive aspergillosis [9]. The last EORT/MSG consensus has tried to standardize the diagnostic thresholds for the different specimens [5]. However, performance of GM remains clearly lower in non-neutropenic patients and/or those undergoing mold-active prophylaxis. β-D-glucan (BDG) can be detected in patients with different fungal infections, such as candidiasis, aspergillosis, pneumocystosis or fusariosis, whereas it is absent in zygomycosis. BDG displays an extremely high negative predictive value for these IFD, although a confirmatory positive result is recommended due to its lack of IFD specificity [5,10].
3. General Overview of IFD Epidemiology in HSCT and Epidemiological Changes within the Last Decades
3.1. The Most Common Fungi Causing IFD in HSCT Recipients
Some of the most important studies detailing the epidemiology of IFD in HSCT are described in Table 1. At the beginning of this millennium, incidence of IFD in different series of HSCT recipients ranged from 10% to as high as 50% [11,12]. In older series, Candida albicans was the most common causative agent of IFD (10–25%); associated mortality rates due to candidemia reached 39%, increasing to 90% when tissue invasion occurred [13]. At that time, Aspergillus spp. diagnosis occurred in less than 6% of patients, but mortality was almost 100%. Yet, a revolution in antifungal prophylaxis and IFD diagnosis has shifted the epidemiological landscape of IFD in patients undergoing HSCT, with a clear decrease in Candida spp. infections and an increase in invasive mold diseases (IMD), mainly caused by Aspergillus spp.
Table 1.
Important studies regarding invasive fungal disease in allogenic HSCT recipients.
| Reference and Year of Publication & | Study Type and Period | n | Prophylaxis | IFD Incidence | IFD Epidemiology | Time from HSCT to IFD |
|---|---|---|---|---|---|---|
| Martino et al. [11] 2002 |
Retrospective study 1996–2000 |
395 allo-HSCT | 73% fluconazole, 17% itraconazole, 4% amphotericin B, 6% no prophylaxis | 14% | 64% aspergillosis, 20% candidiasis, 6% mucormycosis, 6% other | Median day post HSCT: +90 days (range +4 to +522) Post-HSCT periods: 19% <21 days, 32% 21–90 days, 49% >90 days |
| Pagano et al. [12] 2007 |
Retrospective study 1999–2003 |
1249 allo-HSCT | 39% fluconazole, 21% itraconazole | 8% | 81% aspergillosis, 14% candidiasis (50% non-albicans), 3% fusariosis, 2% other molds | 50% yeast infections and 36% mold disease occurring >100 days post HSCT |
| Garcia-Vidal et al. [21] 2008 |
Retrospective study 1998–2002 |
1248 allo-HSCT | Not reported | 13% invasive mold disease | 87% aspergillosis, 4% fusariosis, 3% mucormycosis | Post-HSCT periods: 22% <40 days, 40% 40–100 days, 38% >100 days |
| Neofytos et al. [20] 2009 |
Prospective study 2004–2007 |
161 IFD in allo-HSCT | Not reported | Not applicable | 57% aspergillosis, 25% candidiasis, 7% mucormycosis, 8% other molds | Median days post HSCT (range): 83 days (3–6542) for aspergillosis, 108 days (0–2219) for candidiasis, 162 days (7–932) for mucormycosis and other mold diseases |
| Kontoyiannis et al. [19] 2010 |
Prospective study 2001–2005 |
6666 allo-HSCT | Not reported | ≈8% | 43% aspergillosis, 28% candidiasis, 8% mucormycosis, 10% other molds | Median days post HSCT: candidiasis, 61 days; aspergillosis, 99 days; fusariosis, 123 days; mucormycosis, 135 days |
| Nucci et al. [22] 2013 |
Prospective study 2007–2009 |
378 allo-HSCT | 81% fluconazole, 1% itraconazole, 4% voriconazole, 4% amphotericin B | 11% | 35% fusariosis, 30% aspergillosis, 17% invasive candidiasis, and 12% hyalohyphomicosis | Median (IQR) days post HSCT: 53 (19–232) days |
| Girmenia et al. [23] 2014 |
Prospective study 2008–2010 |
1858 allo-HSCT | 75% fluconazole, 14% mold-active prophylaxis (NS), 5% secondary prophylaxis (NS), 6% no prophylaxis | 9% | 81% aspergillosis, 11% candidiasis, 4% mucormycosis, 2% fusariosis, 1% other molds, 1% rare yeasts | Post-HSCT periods: 57% <40 days, 24% 40–100 days, 19% >100 days |
| Sun et al. [24] 2015 |
Prospective study 2011 |
1053 allo-HSCT | 61% fluconazole, 22% itraconazole, 19% voriconazole | 9% | 33% aspergillosis, 13% candidiasis, 54% non-identified | Median (IQR) days post HSCT: 45 (16–93) days |
| Gomez et al. [25] 2018 |
Retrospective study Pediatric patients 1998–2016 |
143 allo-HSCT | Fluconazole or voriconazole (rates not reported) | 13% | 86% candidiasis, 17% aspergillosis | Not reported |
| Linke et al. [26] 2019 |
Retrospective study Pediatric patients 2005–2015 |
221 allo-HSCT | 52% fluconazole, 9% mold-active azole, 32% liposomal amphotericin B, 1% micafungin, 6% no prophylaxis | 7% | 73% aspergillosis, 27% candidiasis | Post-HSCT periods: 33% pre-engraftment, 13% engraftment-180 days, 53% >180 days |
| Souza et al. [27] 2020 |
Prospective study 2015–2016 |
71 allo-HSCT | 68% fluconazole, 17% micafungin, 11% mold-active azole (NS) | 11% | 50% aspergillosis, 38% candidiasis, 12% other molds | Not reported |
& Arranged chronologically. Abbreviations. IFD: invasive fungal disease; HSCT: hematopoietic stem cell transplantation; allo-HSCT: allogenic HSCT; IQR: interquartile range; NS: not specified.
Several factors have influenced this epidemiological development, including changes in conditioning regimens, graft-versus-host disease (GVHD) management, and intravascular catheter management. However, the widespread introduction of antifungal prophylaxis is the most likely leading factor in this change in epidemiology.
In the early 1990s, Goodman et al. conducted the first trial comparing prophylactic use of fluconazole vs. placebo in patients undergoing HSCT [14]. Investigators observed that fluconazole use lowered the incidence of systemic and superficial fungal infections and reduced infection-associated mortality. A separate trial performed by Slavin et al. confirmed the findings, with prophylactic use of fluconazole being associated with improved 110-day survival [15]. Both trials observed an incidence of fungal infection of 16% in the placebo arm, with an approximate 90% of IFD caused by Candida spp. Finally, another trial by Marr et al. demonstrated an association between fluconazole use and protection against Candida infections, alongside reduced GVHD and improved overall survival [16].
Following these trials, antifungal prophylaxis with fluconazole in HSCT recipients became the standard of care in most centers around the world and is a recommendation set by most international guidelines [17]. Lately, posaconazole has demonstrated superiority to fluconazole in preventing invasive aspergillosis and reducing mortality related to fungal infections in patients with GVHD [18]. For this reason, most centers have established its use for this indication too.
A few prospective studies have evaluated the composition of IFD in HSCT after the vast introduction of antifungal prophylaxis. In 2010, the Transplant-Associated Infections Surveillance Net (TRANSNET) reported 2001–2006 IFD epidemiology in HSCT recipients across a network of 23 transplant centers in the United States [19]. In this study, Kontoyiannis et al. documented a proven or probable IFD in 9.2% of allogenic HSCT recipients. Invasive aspergillosis was the most common IFD, causing 43% of cases, followed by invasive candidiasis (28%) and zygomycosis (8%).
The Prospective Antifungal Therapy (PATH) Alliance registry documented proven or probable IFD in HSCT recipients across 16 medical centers from North America between 2004 and 2007 [20]. Approximately 60% of IFD cases were due to Aspergillus spp., while 25% of IFD cases were due to candidiasis and 7% due to both zygomycosis and other molds. Investigators did not report rates of prior antifungal treatment in either the TRANSNET or PATH cohorts.
The Italian HSCT Cooperative Group (GITMO) evaluated 1858 patients undergoing an allogenic HSCT across 30 transplant centers between 2008 and 2010 [23]. In that study, 95% of patients received antifungal prophylaxis (75% primary fluconazole prophylaxis; 15% primary mold-active prophylaxis; and 5% secondary prophylaxis), and 1 year cumulative incidence of proven or probable IFD was 8.8%. Once again, invasive aspergillosis was the most common infection (81.1%), followed by invasive candidiasis (11.0%), zygomycosis (3.7%), and fusariosis (1.8%).
Nucci et al. reported data on a prospective cohort of eight transplant centers in Brazil between 2007 and 2009 [22]. In this cohort, 90% of allogenic HSCT recipients received antifungal prophylaxis, primarily with fluconazole (91%). One year cumulative incidence of IFD was 11.3%. Remarkably, the leading IFD was fusariosis (35%), followed by aspergillosis (30%), invasive candidiasis (17%), and hyalohyphomycosis (12%). These results highlight the importance of geographical and environmental context in fungal epidemiology and antifungal susceptibility. However, it should be noted that galactomannan testing was not routinely performed in this study, thereby possibly contributing to an underestimation of the real incidence of aspergillosis. A rather recent, prospective study by the same group (2015–2016) showed that invasive aspergillosis was, indeed, the most frequent IFD (56%), followed by candidemia (24%) and fusariosis (12%) [27].
Finally, one of the last prospective studies in this setting was that done across 31 HSCT centers in China [24]. Of the total number of allogenic HCST patients, 86% received prophylaxis (61%, fluconazole; 22%, itraconazole; and 19%, voriconazole). Despite these high rates of anti-mold prophylaxis, 6 month cumulative incidence of IFD was 9.2%. In this study, Aspergillus spp. and Candida caused 71% and 28% of identified cases, respectively.
3.2. Time since HSCT to IFD
Three different periods have been typically defined in the post-HSCT setting: (i) pre-engraftment; (ii) early post-engraftment; and (iii) late post-engraftment. The time interval between HSCT and the IFD appears to have increased, with a shift occurring from early- to late-onset infections. This may be attributable to the rising use of peripheral blood stem cells and/or reduced intensity conditioning procedures, which are often associated with a shorter duration of neutropenia and, thereby, lower risk of fungal infection in the early period. However, length of post-transplant periods is not well standardized; some variability exists among the different studies. In the Italian transplant cohort [23], 57% of IFD diagnoses took place during the early period (defined as within 40 days of HSCT); 24% during early post-engraftment (41 to 100 days after HSCT); and 19% during late post-engraftment (>100 days from transplant). In the TRANSNET cohort, median time after HSCT to IFD was 61 days for candidiasis, 99 days for aspergillosis, 123 days for fusariosis, and 135 days for zygomycosis [19]. Specifically, in the case of invasive aspergillosis, “only” 22% of episodes occurred within the first month, while almost half of all total episodes were diagnosed in the four-month period following HSCT. The PATH registry obtained similar results, with median time from transplant to IFD being 83 days for invasive aspergillosis and 108 days for invasive candidiasis [20]. IFI due to zygomycetes and other molds occurred later after HSCT, with a median time of 162 days since HSCT.
Although neutropenia has been classically identified as the main risk factor for IFD, the aforementioned shift to later IFD underlines the importance and relevance of other risk factors, such as GVHD and its associated severe immunosuppressive treatment, viral infections, lymphopenia, and cellular immunity dysfunction in IFD risk. However, both risk and timing of IFD after HSCT are highly conditioned by the length of antifungal prophylaxis and the drug used; variability among centers would be, in turn, high.
3.3. IFD Epidemiology in Pediatric HSCT Recipients
As with adults, improvements in medical care have resulted in an increased burden of IFD risk in children undergoing HSCT. Yet, despite the fact that mortality rates have been reported as high as 50–60%, limited data is available regarding epidemiology, treatment, and overall management of IFD in this population [25,26,28].
IFD incidence rates of approximately 7–16% have been reported in pediatric allogenic HSCT recipients. Most IFD cases (around 50%) occur during pre-engraftment [29], although some series have reported a predominance of cases during the late post-engraftment period [26]. Unlike in adult patients, yeasts and Candida spp. particularly remain the most frequent causative agents of IFD in pediatric HSCT recipients [25,26,28]. The second most common causative fungus is Aspergillus spp., which is responsible for most IMD. The epidemiology of IFD caused by other yeasts or molds in this population has not been well defined [29,30].
4. Risk Factors for IFD in HSCT Recipients
Risk factors for yeast and mold infections are significantly different in general. How-ever, some common risk factors, such as those related to the host, transplants or concomitant infections, influence the risk of IFD.
4.1. Host Issues
Older age has been linked with an increased risk of IFD. Diabetes mellitus and hyperglycemia impair innate immunity and have been particularly associated with an elevated risk of mucormycosis. Some prognostic scores in HSCT patients have been shown to correlate with the risk of IFD [31].
Glucocorticoids are a common risk factor for IFD due to neutrophil chemotaxis and oxidative burst inhibition, as well as hindrance of macrophages’ capacity to remove conidia [32]. Delayed engraftment and prolonged neutropenia continue to be some main risk factors for IFD and are associated with treatment failure and increased mortality [33,34].
The main risk factors for disseminated yeast infection (mainly candidemia) are as follows: (i) disruption of the cutaneous barrier, mostly due to intravascular access devices (which may lead to catheter-related fungemia); (ii) disruption of the mucosal barrier due to gastrointestinal tract surgery or GVHD; (iii) alteration of normal bacterial flora due to broad-spectrum antibiotics (leading to yeast overgrowth and predominance); and (iv) total parenteral nutrition (with lipid formulations favoring fungal invasion).
4.2. Transplant Issues
Active or refractory disease at the time of the transplant is a major risk determinant. Graft source and donor relatedness have an impact on transplant-related toxicity and the risk of IFD [35]. Compared to peripheral blood stem cells, bone marrow and umbilical cord transplants are associated with the highest risk of IFD due to delayed immune reconstitution. Similarly, haploidentical transplants and, to a lesser extent, stem cell donors who are not matched siblings are associated with longer immunosuppression and an increased risk of IFD [21,36]. GVHD poses an extremely high risk of IFD due to both its own immunosuppressive effect, and the required immunosuppressive treatment that often includes high-dose steroids [37].
4.3. Concomitant Infections
Cytomegalovirus is associated with an increased risk of IFD in the post-transplant setting. This association is perhaps due to the virus’ deleterious effect on innate immunity. Respiratory viruses, such as the influenza virus, respiratory syncytial virus, adenovirus, and parainfluenza, impair mucociliary activity, local innate immunity and systemic host defenses, and are thus associated with a higher risk of IFD [38,39]. This association is particularly relevant in patients with severe influenza and severe SARS-CoV-2 who require ICU admission [40,41].
5. The Most Common Causative Agents
5.1. Aspergillus spp.
Aspergillus spp. are ubiquitous, environmental molds, forming spores that enter the body via respiratory inhalation. As already mentioned, invasive aspergillosis is currently the most common IFD in HSCT recipients, mainly with pulmonary involvement. Aspergillus fumigatus is the most common causative species, probably due to the relatively small size that characterizes A. fumigatus conidia, which allows for its deep penetration into the alveolar space. Additionally, A. fumigatus can grow in high temperatures (37–50 °C), being more resistant and thermotolerant than other Aspergillus species [39]. In the TRANSNET and PATH studies, 44% and 37% of aspergillosis cases, respectively, were due to A. fumigatus, although in a considerable percentage of cases, identification at the species level was not achieved [19,20]. However, invasive aspergillosis caused by non-fumigatus species seems to be increasing. In a study performed at the Fred Hutchinson Cancer Research Center comparing two close periods (1993–1995 vs. 1996–1998), invasive aspergillosis caused by non-fumigatus species rose from 18% to 34% [42]. Nonetheless, these changes could just reflect improvements made in microbiological identification, which may increase with the advent of molecular techniques. Additionally, this seems to be dependent on whether or not it is a breakthrough episode [43]. The most commonly isolated non-fumigatus species are Aspergillus niger, Aspergillus terreus, and Aspergillus flavus.
Antifungal resistance in Aspergillus is a huge threat. Since the beginning of the century, different studies have reported an increasing prevalence of azole resistance in A. fumigatus [44,45]. Interestingly, this antifungal resistance is widely mediated by mutations in a specific gene (TR34/L98H) and could have occurred due to vast use of triazole fungicides in agriculture [2]. Besides this mutation, some Aspergillus species are known to have varying susceptibilities to different antifungal drugs [46]. For example, Aspergillus terreus is typically less susceptible to amphotericin B. Additionally, the use of molecular tools has permitted the description of new cryptic species among different Aspergillus species complexes. Remarkably, these species commonly exhibit innate, high-level resistance to multiple antifungal agents, including amphotericin B and the triazoles [47,48]. This observation underlines the importance of identifying to the species level and performing antifungal resistance testing, especially in those cases wherein a lack of response to initial therapy is present.
5.2. Candida spp.
Candida spp. forms part of the gastrointestinal tract and skin microbiota. Loss of mucosal and skin barrier integrity—as occurs in mucositis, GVHD, and endovascular catheters—may lead to invasive candidiasis in HSCT recipients. Invasive candidiasis is the second most common cause of IFD in HSCT recipients.
Candida albicans had classically been the most common IFD-causing pathogen. However, the widespread use of azole prophylaxis not only decreased the overall incidence of invasive candidiasis, but also increased the percentage of cases caused by non-albicans species. In the PATH cohort, 76% of invasive candidiasis episodes were caused by non-albicans species, with 44% and 11% of episodes caused by Nakaseomyces glabrata (formerly known as Candida glabrata) and Pichia kudriavzevii (formerly known as Candida krusei), respectively [20]. Similar results have been reported in other series [12,19]. Current candidiasis epidemiology is mainly determined by antifungal selection pressure [49,50]. This should be considered when invasive candidiasis is clinically suspected or diagnosed. In such a challenging scenario, implementing a bundle of measures, including initial treatment (adequate antifungal and source control within 72 h), identification of complicated candidemia (follow-up blood cultures, ophthalmoscopic evaluation, and echocardiography in at-risk patients), and final treatment adequacy (de-escalation when indicated and adequate length of antifungal treatment), has proven to reduce mortality [51].
5.3. Zygomycetes
Mucormycosis-producing agents are ubiquitous fungi, commonly found in decaying organic matter that cause infection in patients with hematologic malignancies and HSCT recipients, and are characterized by violent evolution with frequent angioinvasion, tissue infarction, and necrosis [52,53]. In different prospective cohorts of HSCT recipients with IFD, mucormycosis accounted for approximately 4–8% of cases [19,20,23]. By contrast, in the Chinese cohort, there was only a single case of mucormycosis diagnosed among the 1401 transplanted patients [24]. Potential explanations for these differences include environmental factors and factors related to the use of different diagnostic approaches.
Some groups from various countries have reported an increasing incidence in mucormycosis in patients with hematologic malignancies [54,55]. This rise in mucormycosis incidence could be partially due to selection pressure by voriconazole use. It could also be due to increased long-term immunosuppression of HSCT patients and decreased aspergillosis-related mortality, which results in the eventual emergence of rarer molds later after transplantation [20]. Furthermore, no specific antigenic diagnostic methods for Zygomycetes exist, and histological findings serve as the basis for most diagnoses. Therefore, mucormycosis cases may be assumed as being underdiagnosed currently.
5.4. Hyalohyphomycoses
Hyalohyphomycoses is a term referring to IFD caused by non-pigmented molds (other than the genera Aspergillus or Penicillium or the class Zygomycetes) that form hyphal elements with hyaline or clear walls in tissue. Most representative molds in these group include Fusarium, Scedosporium, Lomentospora, Acremonium, and Paecilomyces spp. Although IFD caused by hyalohyphomycetes are very uncommon, exceedingly high mortality rates of up to 90% have been reported [56,57]. The most frequently isolated mold in this group is Fusarium spp., accounting for 3% and 7% of all IFD in the TRANSNET and PATH studies, respectively [19,20]. However, hyalohyphomycoses are probably highly influenced by both the host’s degree of immunosuppression and the geographical context. In this regard, fusariosis represented 12–35% of all IFD cases in Brazilian cohorts [22,27].
5.5. Pneumocystis jirovecii
Pneumocystis jirovecii, which was long thought to be a protozoan organism, is an ascomycetous fungi that classically causes pneumonia in immunosuppressed patients. Before the introduction of antibiotic prophylaxis, the risk of Pneumocystis pneumonia was around 5–15% in patients receiving a HSCT [58,59]. This has dramatically decreased since the widespread implementation of prophylactic strategies initiated in the 1980s; Pneumocystis jirovecii pneumonia represented less than 1.5% of all IFD in the current HSCT cohort studies [19,20,23]. Indeed, recent cohorts have shown that most pneumocystosis cases in this population occur late after HSCT in patients who were no longer receiving Pneumocystis prophylaxis [60,61].
5.6. Other
Cryptococci are basidiomycetous, encapsulated yeasts that typically cause disseminated and/or central nervous system infections in immunocompromised patients, e.g., Cryptococcus neoformans or C. gattii. Delayed reconstitution of CD4+ lymphocytes and B lymphocytes after allogenic HSCT places these patients at a theoretically high risk of cryptococcal infection. However, prevalence of cryptococcosis in HSCT is very low (<1%), probably due to effective antifungal prophylaxis [19,62].
Non-Candida opportunistic yeasts are emerging causes of bloodstream infections in immunosuppressed patients with hematologic malignancies. However, scarce information is available regarding this type of IFD in HSCT recipients. In a retrospective study conducted at the MD Anderson Cancer Center, the most commonly rare (non-Candida, non-Cryptococcus) yeasts to cause bloodstream infections in patients with cancer were Rhodotorula, Trichosporon, Saccharomyces, and Geotrichum [63]. However, in this study, only 29% of the patients had received a prior HSCT.
6. Breakthrough IFD: The New Reality of IFD in HSCT Recipients
Current IFD epidemiology in HSCT recipients is highly conditioned by the widespread introduction of antifungal prophylaxis. In this setting, there is an increasing number of centers using mold-active antifungals (mainly mold-active triazoles and/or echinocandins) as primary prophylaxis. Further, a prior IFD is no longer a contraindication of HSCT, and the number of patients receiving secondary prophylaxis (or treatment) for prior infections is on the rise [35]. In this context, breakthrough IFD is therefore likely associated with a further epidemiological change and poses a great challenge to treating physicians [64]. For example, in a retrospective study including HSCT recipients, Lamoth et al. described the epidemiology of IMD episodes occurring in a certain period, considering such episodes as breakthrough or not. An increased number of mucormycosis cases (from 15% to 31%) was reported in breakthrough IMD episodes to voriconazole or posaconazole.
Prior antifungal treatment highly influences breakthrough IFD epidemiology.
6.1. Breakthrough Infections to Posaconazole
Posaconazole is the most widely used mold-active azole, showing good activity against Aspergillus as well as against Zygomycetes. As previously stated, it has been incorporated in prophylaxis in patients receiving a HSCT. In the most important prophylaxis trial [18], 5.3% of patients in the posaconazole group presented a breakthrough IFD, of which 44% were caused by Aspergillus spp., 25% by Candida spp., and 31% by other molds. Some unicentric and retrospective studies later reported varying incidences ranging between 3% and 11% [65,66]. In these cohorts, rates of invasive aspergillosis among diagnosed IFD were relatively low, with a high proportion of breakthrough episodes caused by Zygomycetes and rare yeasts. However, at that time, posaconazole efficacy was limited by erratic absorption of posaconazole solution [67]. Data on breakthrough episodes to posaconazole tablets in HSCT is very scarce. A retrospective unicenter study evaluated over three hundred patients with hematologic malignancies receiving posaconazole prophylaxis, of whom 70 had undergone a HSCT [68]. The overall rate of breakthrough IFD was 2%, increasing to 4% (3 of 70) when considering HSCT recipients. These three cases were one aspergillosis, one rare mold (Penicillium), and one rare, unidentified yeasts infection. All three patients died within six weeks.
6.2. Breakthrough Infections to Voriconazole
Two randomized control trials have evaluated the use of voriconazole prophylaxis in the HSCT setting [69,70]. Wingard et al. reported a 1 year cumulative incidence of IFD of 13% (including proven, probable, and presumptive IFD) in the voriconazole arm. The most common IFD was aspergillosis (41%), but Aspergillus species were not identified [69]. In the second trial, Marks et al. only documented three breakthrough IFD episodes to voriconazole (one A. fumigatus, one P. kudriavzevii, and one C. parapsilosis). However, significantly different information has been documented in “real-life” retrospective cohort studies [71,72,73,74]. Most of these studies report breakthrough infection incidence of around 2–7%, with most episodes caused by Zygomycetes, remarkably. Candidemia due to P. kudriavzevii and N. glabrata were also frequent. These data seem expectable due to reduced susceptibility of these pathogens to voriconazole, even though it demonstrates excellent activity against A. fumigatus.
Voriconazole level has large interpersonal variability, given that it depends on several factors, i.e., the patient’s age, potential drug–drug interactions, and cytochrome P450 polymorphism [75]. In this setting, therapeutic drug monitoring is mandatory whenever there is clinical suspicion of breakthrough IFD.
6.3. Breakthrough Infections to Echinocandins and Other Antifungals
Four randomized clinical trials have evaluated echinocandins versus azoles as prophylaxis in patients receiving a HSCT, showing contradictory results [76,77,78,79]. Incidence of breakthrough infections in echinocandins ranged between 2–7%. Though some of these studies did not report on isolated fungi, invasive aspergillosis and invasive candidiasis seemed to be the most common entities. Some cohort studies on HSCT recipients presenting breakthrough episodes to echinocandins have reported similar incidence rates [80,81,82,83]. Remarkably, despite good in vitro activity by echinocandins against Aspergillus spp., most breakthrough episodes in these studies were invasive aspergillosis.
To our best knowledge, data are missing concerning breakthrough episodes to isavuconazole or amphotericin B in HSCT patients.
6.4. A Recommended Approach to HSCT Recipients with Suspicion of Breakthrough IFD
Breakthrough fungal infection is associated with an exceedingly high mortality [64]. In this context, clinicians should perform an early and aggressive diagnostic work-up in those patients with clinically suspicion of having a breakthrough IFD. Early CT imaging +/− bronchoscopy with BAL performance should take place. In cases wherein BAL results are negative, clinicians should perform a transthoracic or transbronchial biopsy when feasible. Additionally, whenever fungal isolation occurs, identification to the species level and assessment of antifungal susceptibility are highly recommended.
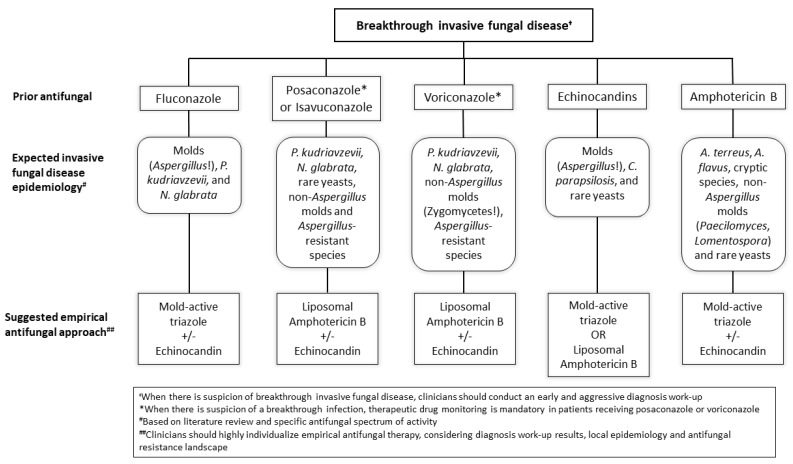
As previously mentioned, breakthrough IFD epidemiology is mainly determined by prior antifungal therapy. Figure 1 summarizes the “expected” epidemiology per prior antifungal therapy, as well as our personal recommendations on potentially empirical treatment in cases of suspected breakthrough IFD.
Figure 1.
Algorithm of expected breakthrough invasive fungal disease epidemiology per prior antifungal therapy and suggested empirical treatment.
7. Conclusions and Future Perspectives
IFD epidemiology in HSCT recipients has been changing in recent decades, mainly following the widespread use of antifungal prophylaxis. Currently, the most frequent IFD is invasive aspergillosis, mainly due to Aspergillus fumigatus. The second most common IFD is invasive candidiasis, with most current cases caused by non-albicans species. Mucormycosis and fusariosis follow thereafter, considering frequency, and are associated with very high mortality. IFD caused by rarer molds and yeasts are uncommon but appear to be increasing in patients undergoing a HSCT.
In the coming years, IFD epidemiology is likely to keep changing due to an increased use of mold-active antifungals. At the same time, improvements in microbiology laboratories and techniques, as well as the generalization of molecular diagnoses will help characterize the real epidemiology of fungal infections more precisely in these patients. For a nuanced description, it is important to identify to the species level; it could have an impact on antifungal resistance. Finally, increasing antifungal resistance in Aspergillus, but also in overall IFD, poses a major threat. In this scenario, knowledge of current epidemiology and accurate diagnosis of IFD remain crucial to establishing correct prophylaxis guidelines and appropriate early treatments.
Acknowledgments
We would like to thank Anthony Armenta for providing medical editing assistance for the manuscript at hand.
Author Contributions
Conceptualization, P.P.-A. and C.G.-V.; methodology, P.P.-A. and C.G.-V.; software, not applicable; validation, P.P.-A. and C.G.-V.; formal analysis, not applicable; investigation, P.P.-A.; resources, C.G.-V.; data curation, P.P.-A.; writing—original draft preparation, P.P.-A.; writing—review and editing, P.P.-A. and C.G.-V.; visualization, P.P.-A.; supervision, C.G.-V.; project administration, not applicable; funding acquisition, P.P.-A. and C.G.-V. Both authors have read and agreed to the published version of the manuscript.
Funding
P.P.-A. [JR20/00012] and C.G.-V. [FIS PI18/01061] have received research grants from the Ministerio de Sanidad y Consumo, Instituto de Salud Carlos III. The funder had no role in the current manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
P.P.-A. has received honoraria for talks on behalf of Gilead Science, MSD, ViiV Healthcare and Lilly. C.G.-V. has received honoraria for talks on behalf of Gilead Science, MSD, Novartis, Pfizer, Janssen, Lilly, and GlaxoSmithKline as well as a grant from Gilead Science, Pfizer, and MSD.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Niederwieser D., Baldomero H., Atsuta Y., Aljurf M., Seber A., Greinix H.T., Koh M., Worel N., Galeano S., Jaimovich G., et al. One and Half Million Hematopoietic Stem Cell Transplants (HSCT). Dissemination, Trends and Potential to Improve Activity by Telemedicine from the Worldwide Network for Blood and Marrow Transplantation (WBMT) Blood. 2019;134:2035. doi: 10.1182/blood-2019-125232. [DOI] [Google Scholar]
- 2.Verweij P.E., Snelders E., Kema G.H., Mellado E., Melchers W.J. Azole resistance in Aspergillus fumigatus: A side-effect of environmental fungicide use? Lancet Infect. Dis. 2009;9:789–795. doi: 10.1016/S1473-3099(09)70265-8. [DOI] [PubMed] [Google Scholar]
- 3.Ascioglu S., Rex J.H., De Pauw B., Bennett J.E., Bille J., Crokaert F., Denning D.W., Donnelly J.P., Edwards J.E., Erjavec Z., et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: An international consensus. Clin. Infect. Dis. 2002;34:7–14. doi: 10.1086/323335. [DOI] [PubMed] [Google Scholar]
- 4.De Pauw B., Walsh T.J., Donnelly J.P., Stevens D.A., Edwards J.E., Calandra T., Pappas P.G., Maertens J., Lortholary O., Kauffman C.A., et al. Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) C. Clin. Infect. Dis. 2008;46:1813–1821. doi: 10.1086/588660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Donnelly J.P., Chen S.C., Kauffman C.A., Steinbach W.J., Baddley J.W., Verweij P.E., Clancy C.J., Wingard J.R., Lockhart S.R., Groll A.H., et al. Revision and update of the consensus definitions of invasive fungal disease from the european organization for research and treatment of cancer and the mycoses study group education and research consortium. Clin. Infect. Dis. 2020;71:1367–1376. doi: 10.1093/cid/ciz1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mengoli C., Cruciani M., Barnes R.A., Loeffler J., Donnelly J.P. Use of PCR for diagnosis of invasive aspergillosis: Systematic review and meta-analysis. Lancet Infect. Dis. 2009;9:89–96. doi: 10.1016/S1473-3099(09)70019-2. [DOI] [PubMed] [Google Scholar]
- 7.Avni T., Leibovici L., Paul M. PCR diagnosis of invasive candidiasis: Systematic review and meta-analysis. J. Clin. Microbiol. 2011;49:665–670. doi: 10.1128/JCM.01602-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Clancy C.J., Nguyen M.H. T2 magnetic resonance for the diagnosis of bloodstream infections: Charting a path forward. J. Antimicrob. Chemother. 2018;73:iv2–iv5. doi: 10.1093/jac/dky050. [DOI] [PubMed] [Google Scholar]
- 9.Ambasta A., Carson J., Church D.L. The use of biomarkers and molecular methods for the earlier diagnosis of invasive aspergillosis in immunocompromised patients. Med. Mycol. 2015;53:531–557. doi: 10.1093/mmy/myv026. [DOI] [PubMed] [Google Scholar]
- 10.Lamoth F., Cruciani M., Mengoli C., Castagnola E., Lortholary O., Richardson M., Marchetti O. β-glucan antigenemia assay for the diagnosis of invasive fungal infections in patients with hematological malignancies: A systematic review and meta-analysis of cohort studies from the third European Conference on Infections in Leukemia (ECIL-3) Clin. Infect. Dis. 2012;54:633–643. doi: 10.1093/cid/cir897. [DOI] [PubMed] [Google Scholar]
- 11.Martino R., Subirá M., Rovira M., Solano C., Vázquez L., Sanz G.F., Urbano-Ispizua A., Brunet S., De la Cámara R. Invasive fungal infections after allogeneic peripheral blood stem cell transplantation: Incidence and risk factors in 395 patients. Br. J. Haematol. 2002;116:475–482. doi: 10.1046/j.1365-2141.2002.03259.x. [DOI] [PubMed] [Google Scholar]
- 12.Pagano L., Caira M., Nosari A., Van Lint M.T., Candoni A., Offidani M., Aloisi T., Irrera G., Bonini A., Picardi M., et al. Fungal infections in recipients of hematopoietic stem cell transplants: Results of the SEIFEM B-2004 study—Sorveglianza Epidemiologica Infezioni Fungine nelle Emopatie Maligne. Clin. Infect. Dis. 2007;45:1161–1170. doi: 10.1086/522189. [DOI] [PubMed] [Google Scholar]
- 13.Larocco M.T., Burgert S.J. Infection in the bone marrow transplant recipient and role of the microbiology laboratory in clinical transplantation. Clin. Microbiol. Rev. 1997;10:277–297. doi: 10.1128/CMR.10.2.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Goodman J.L., Winston D.J., Greenfield R.A., Chandrasekar P.H., Fox B., Kaizer H., Shadduck R.K., Shea T.C., Stiff P., Friedman D.J., et al. A Controlled Trial of Fluconazole to Prevent Fungal Infections in Patients Undergoing Bone Marrow Transplantation. N. Engl. J. Med. 1992;326:845–851. doi: 10.1056/NEJM199203263261301. [DOI] [PubMed] [Google Scholar]
- 15.Slavin M.A., Osborne B., Adams R., Levenstein M.J., Schoch H.G., Feldman A.R., Meyers J.D., Bowden R.A. Efficacy and Safety of Fluconazole Prophylaxis for Fungal Infections after Marrow Transplantation—A Prospective, Randomized, Double-Blind Study. J. Infect. Dis. 1995;171:1545–1552. doi: 10.1093/infdis/171.6.1545. [DOI] [PubMed] [Google Scholar]
- 16.Marr K.A., Seidel K., Slavin M.A., Bowden R.A., Gary Schoch H., Flowers M.E.D., Corey L., Boeckh M. Prolonged fluconazole prophylaxis is associated with persistent protection against candidiasis-Related death in allogeneic marrow transplant recipients: Long-Term follow-up of a randomized, placebo-controlled trial. Blood. 2000;96:2055–2061. doi: 10.1182/blood.V96.6.2055. [DOI] [PubMed] [Google Scholar]
- 17.Maertens J.A., Girmenia C., Brüggemann R.J., Duarte R.F., Kibbler C.C., Ljungman P., Racil Z., Ribaud P., Slavin M.A., Cornely O.A., et al. European guidelines for primary antifungal prophylaxis in adult haematology patients: Summary of the updated recommendations from the European Conference on Infections in Leukaemia. J. Antimicrob. Chemother. 2018;73:3221–3230. doi: 10.1093/jac/dky286. [DOI] [PubMed] [Google Scholar]
- 18.Ullmann A.J., Lipton J.H., Vesole D.H., Chandrasekar P., Langston A., Tarantolo S.R., Greinix H., Morais de Azevedo W., Reddy V., Boparai N., et al. Posaconazole or Fluconazole for Prophylaxis in Severe Graft-versus-Host Disease. N. Engl. J. Med. 2007;356:335–347. doi: 10.1056/NEJMoa061098. [DOI] [PubMed] [Google Scholar]
- 19.Kontoyiannis D.P., Marr K.A., Park B.J., Alexander B.D., Anaissie E.J., Walsh T.J., Ito J., Andes D.R., Baddley J.W., Brown J.M., et al. Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001–2006: Overview of the transplant- associated infection surveillance network (TRANSNET) database. Clin. Infect. Dis. 2010;50:1091–1100. doi: 10.1086/651263. [DOI] [PubMed] [Google Scholar]
- 20.Neofytos D., Horn D., Anaissie E., Steinbach W., Olyaei A., Fishman J., Pfaller M., Chang C., Webster K., Marr K. Epidemiology and outcome of invasive fungal infection in adult hematopoietic stem cell transplant recipients: Analysis of multicenter prospective antifungal therapy (PATH) alliance registry. Clin. Infect. Dis. 2009;48:265–273. doi: 10.1086/595846. [DOI] [PubMed] [Google Scholar]
- 21.Garcia-Vidal C., Upton A., Kirby K.A., Marr K.A. Epidemiology of Invasive Mold Infections in Allogeneic Stem Cell Transplant Recipients: Biological Risk Factors for Infection According to Time after Transplantation. Clin. Infect. Dis. 2008;47:1041–1050. doi: 10.1086/591969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nucci M., Garnica M., Gloria A.B., Lehugeur D.S., Dias V.C.H., Palma L.C., Cappellano P., Fertrin K.Y., Carlesse F., Simões B., et al. Invasive fungal diseases in haematopoietic cell transplant recipients and in patients with acute myeloid leukaemia or myelodysplasia in Brazil. Clin. Microbiol. Infect. 2013;19:745–751. doi: 10.1111/1469-0691.12002. [DOI] [PubMed] [Google Scholar]
- 23.Girmenia C., Raiola A.M., Piciocchi A., Algarotti A., Stanzani M., Cudillo L., Pecoraro C., Guidi S., Iori A.P., Montante B., et al. Incidence and outcome of invasive fungal diseases after allogeneic stem cell transplantation: A prospective study of the gruppo italiano trapianto midollo osseo (GITMO) Biol. Blood Marrow Transplant. 2014;20:872–880. doi: 10.1016/j.bbmt.2014.03.004. [DOI] [PubMed] [Google Scholar]
- 24.Sun Y., Meng F., Han M., Zhang X., Yu L., Huang H., Wu D., Ren H., Wang C., Shen Z., et al. Epidemiology, Management, and Outcome of Invasive Fungal Disease in Patients Undergoing Hematopoietic Stem Cell Transplantation in China: A Multicenter Prospective Observational Study. Biol. Blood Marrow Transplant. 2015;21:1117–1126. doi: 10.1016/j.bbmt.2015.03.018. [DOI] [PubMed] [Google Scholar]
- 25.Gomez S.M., Caniza M., Fynn A., Vescina C., Ruiz C., Iglesias D., Sosa F., Sung L. Fungal infections in hematopoietic stem cell transplantation in children at a pediatric children’s hospital in Argentina. Transpl. Infect. Dis. 2018;20:e12913. doi: 10.1111/tid.12913. [DOI] [PubMed] [Google Scholar]
- 26.Linke C., Ehlert K., Ahlmann M., Fröhlich B., Mohring D., Burkhardt B., Rössig C., Groll A.H. Epidemiology, utilisation of healthcare resources and outcome of invasive fungal diseases following paediatric allogeneic haematopoietic stem cell transplantation. Mycoses. 2020;63:172–180. doi: 10.1111/myc.13029. [DOI] [PubMed] [Google Scholar]
- 27.Souza L., Nouér S.A., Morales H., Simões B., Solza C., Queiroz-Telles F., Nucci M. Epidemiology of invasive fungal disease in haematologic patients. Mycoses. 2021;64:252–256. doi: 10.1111/myc.13205. [DOI] [PubMed] [Google Scholar]
- 28.Simms-Waldrip T., Rosen G., Nielsen-Saines K., Ikeda A., Brown B., Moore T. Invasive fungal infections in pediatric hematopoietic stem cell transplant patients. Infect. Dis. 2015;47:218–224. doi: 10.3109/00365548.2014.985709. [DOI] [PubMed] [Google Scholar]
- 29.Otto W.R., Green A.M. Fungal infections in children with haematologic malignancies and stem cell transplant recipients. Br. J. Haematol. 2020;189:607–624. doi: 10.1111/bjh.16452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Otto W.R., Pahud B.A., Yin D.E. Pediatric Mucormycosis: A 10-Year Systematic Review of Reported Cases and Review of the Literature. J. Pediatric Infect. Dis. Soc. 2019;8:342–350. doi: 10.1093/jpids/piz007. [DOI] [PubMed] [Google Scholar]
- 31.Busca A., Passera R., Maffini E., Festuccia M., Brunello L., Dellacasa C.M., Aydin S., Frairia C., Manetta S., Butera S., et al. Hematopoietic cell transplantation comorbidity index and risk of developing invasive fungal infections after allografting. Bone Marrow Transplant. 2018;53:1304–1310. doi: 10.1038/s41409-018-0161-1. [DOI] [PubMed] [Google Scholar]
- 32.Stuehler C., Kuenzli E., Jaeger V.K., Baettig V., Ferracin F., Rajacic Z., Kaiser D., Bernardini C., Forrer P., Weisser M., et al. Immune Reconstitution After Allogeneic Hematopoietic Stem Cell Transplantation and Association With Occurrence and Outcome of Invasive Aspergillosis. J. Infect. Dis. 2015;212:959–967. doi: 10.1093/infdis/jiv143. [DOI] [PubMed] [Google Scholar]
- 33.Kontoyiannis D.P., Selleslag D., Mullane K., Cornely O.A., Hope W., Lortholary O., Croos-Dabrera R., Lademacher C., Engelhardt M., Patterson T.F. Impact of unresolved neutropenia in patients with neutropenia and invasive aspergillosis: A post hoc analysis of the SECURE trial. J. Antimicrob. Chemother. 2018;73:757–763. doi: 10.1093/jac/dkx423. [DOI] [PubMed] [Google Scholar]
- 34.Nucci M., Anaissie E.J., Queiroz-Telles F., Martins C.A., Trabasso P., Solza C., Mangini C., Simões B.P., Colombo A.L., Vaz J., et al. Outcome predictors of 84 patients with hematologic malignancies and Fusarium infection. Cancer. 2003;98:315–319. doi: 10.1002/cncr.11510. [DOI] [PubMed] [Google Scholar]
- 35.Puerta-Alcalde P., Champlin R.E., Kontoyiannis D.P. How I perform hematopoietic stem cell transplantation on patients with a history of invasive fungal disease. Blood. 2020;136:2741–2753. doi: 10.1182/blood.2020005884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Marr K.A., Carter R.A., Boeckh M., Martin P., Corey L. Invasive aspergillosis in allogeneic stem cell transplant recipients: Changes in epidemiology and risk factors. Blood. 2002;100:4358–4366. doi: 10.1182/blood-2002-05-1496. [DOI] [PubMed] [Google Scholar]
- 37.Teshima T., Reddy P., Zeiser R. Acute Graft-versus-Host Disease: Novel Biological Insights. Biol. Blood Marrow Transplant. 2016;22:11–16. doi: 10.1016/j.bbmt.2015.10.001. [DOI] [PubMed] [Google Scholar]
- 38.Garcia-Vidal C., Royo-Cebrecos C., Peghin M., Moreno A., Ruiz-Camps I., Cervera C., Belmonte J., Gudiol C., Labori M., Roselló E., et al. Environmental variables associated with an increased risk of invasive aspergillosis. Clin. Microbiol. Infect. 2014;20:O939–O945. doi: 10.1111/1469-0691.12650. [DOI] [PubMed] [Google Scholar]
- 39.Garcia-Vidal C., Viasus D., Carratalà J. Pathogenesis of invasive fungal infections. Curr. Opin. Infect. Dis. 2013;26:270–276. doi: 10.1097/QCO.0b013e32835fb920. [DOI] [PubMed] [Google Scholar]
- 40.Marr K.A., Platt A., Tornheim J.A., Zhang S.X., Datta K., Cardozo C., Garcia-Vidal C. Aspergillosis Complicating Severe Coronavirus Disease. Emerg. Infect. Dis. 2020;27 doi: 10.3201/eid2701.202896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Garcia-Vidal C., Barba P., Arnan M., Moreno A., Ruiz-Camps I., Gudiol C., Ayats J., Ortí G., Carratal J. Invasive aspergillosis complicating pandemic influenza A (H1N1) infection in severely immunocompromised patients. Clin. Infect. Dis. 2011;53 doi: 10.1093/cid/cir485. [DOI] [PubMed] [Google Scholar]
- 42.Marr K.A., Carter R.A., Crippa F., Wald A., Corey L. Epidemiology and outcome of mould infections in hematopoietic stem cell transplant recipients. Clin. Infect. Dis. 2002;34:909–917. doi: 10.1086/339202. [DOI] [PubMed] [Google Scholar]
- 43.Kuster S., Stampf S., Gerber B., Baettig V., Weisser M., Gerull S., Medinger M., Passweg J., Schanz U., Garzoni C., et al. Incidence and outcome of invasive fungal diseases after allogeneic hematopoietic stem cell transplantation: A Swiss transplant cohort study. Transpl. Infect. Dis. 2018;20:e12981. doi: 10.1111/tid.12981. [DOI] [PubMed] [Google Scholar]
- 44.Bueid A., Howard S.J., Moore C.B., Richardson M.D., Harrison E., Bowyer P., Denning D.W. Azole antifungal resistance in Aspergillus fumigatus: 2008 and 2009. J. Antimicrob. Chemother. 2010;65:2116–2118. doi: 10.1093/jac/dkq279. [DOI] [PubMed] [Google Scholar]
- 45.Jensen R.H., Hagen F., Astvad K.M.T., Tyron A., Meis J.F., Arendrup M.C. Azole-resistant Aspergillus fumigatus in Denmark: A laboratory-based study on resistance mechanisms and genotypes. Clin. Microbiol. Infect. 2016;22:570.e1–570.e9. doi: 10.1016/j.cmi.2016.04.001. [DOI] [PubMed] [Google Scholar]
- 46.Lamoth F., Lewis R.E., Kontoyiannis D.P. Role and interpretation of antifungal susceptibility testing for the management of invasive fungal infections. J. Fungi. 2021;7:17. doi: 10.3390/jof7010017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Alastruey-Izquierdo A., Alcazar-Fuoli L., Cuenca-Estrella M. Antifungal susceptibility profile of cryptic species of aspergillus. Mycopathologia. 2014;178:427–433. doi: 10.1007/s11046-014-9775-z. [DOI] [PubMed] [Google Scholar]
- 48.Escribano P., Rodríguez-Sánchez B., Díaz-García J., Martín-Gómez M.T., Ibáñez E., Rodríguez-Mayo M., Peláez T., García-Gómez de la Pedrosa E., Tejero-García R., Marimón J.M., et al. Azole resistance survey on clinical Aspergillus fumigatus isolates in Spain. Clin. Microbiol. Infect. 2020 doi: 10.1016/j.cmi.2020.09.042. [DOI] [PubMed] [Google Scholar]
- 49.Marr K.A., Seidel K., White T.C., Bowden R.A. Candidemia in allogeneic blood and marrow transplant recipients: Evolution of risk factors after the adoption of prophylactic fluconazole. J. Infect. Dis. 2000;181:309–316. doi: 10.1086/315193. [DOI] [PubMed] [Google Scholar]
- 50.Lortholary O., Desnos-Ollivier M., Sitbon K., Fontanet A., Bretagne S., Dromer F., Bouges-Michel C., Poilane I., Dunan J., Galeazzi G., et al. Recent exposure to caspofungin or fluconazole influences the epidemiology of candidemia: A prospective multicenter study involving 2441 patients. Antimicrob. Agents Chemother. 2011;55:532–538. doi: 10.1128/AAC.01128-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cardozo C., Cuervo G., Salavert M., Merino P., Gioia F., Fernández-Ruiz M., López-Cortés L.E.L.E., Escolá-Vergé L., Montejo M., Muñoz P., et al. An evidence-based bundle improves the quality of care and outcomes of patients with candidaemia. J. Antimicrob. Chemother. 2020;75:730–737. doi: 10.1093/jac/dkz491. [DOI] [PubMed] [Google Scholar]
- 52.Kontoyiannis D.P., Wessel V.C., Bodey G.P., Rolston K.V.I. Zygomycosis in the 1990s in a tertiary-care cancer center. Clin. Infect. Dis. 2000;30:851–856. doi: 10.1086/313803. [DOI] [PubMed] [Google Scholar]
- 53.Chamilos G., Lewis R.E., Kontoyiannis D.P. Delaying Amphotericin B-Based Frontline Therapy Significantly Increases Mortality among Patients with Hematologic Malignancy Who Have Zygomycosis. Clin. Infect. Dis. 2008;47:503–509. doi: 10.1086/590004. [DOI] [PubMed] [Google Scholar]
- 54.Bitar D., Van Cauteren D., Lanternier F., Dannaoui E., Che D., Dromer F., Desenclos J.C., Lortholary O. Increasing incidence of zygomycosis (mucormycosis), France, 1997–2006. Emerg. Infect. Dis. 2009;15:1395–1401. doi: 10.3201/eid1509.090334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Petrikkos G., Skiada A., Lortholary O., Roilides E., Walsh T.J., Kontoyiannis D.P. Epidemiology and clinical manifestations of mucormycosis. Clin. Infect. Dis. 2012;54 doi: 10.1093/cid/cir866. [DOI] [PubMed] [Google Scholar]
- 56.Lamaris G.A., Chamilos G., Lewis R.E., Safdar A., Raad I.I., Kontoyiannis D.P. Scedosporium infection in a tertiary care cancer center: A review of 25 cases from 1989–2006. Clin. Infect. Dis. 2006;43:1580–1584. doi: 10.1086/509579. [DOI] [PubMed] [Google Scholar]
- 57.Riches M.L., Trifilio S., Chen M., Ahn K.W., Langston A., Lazarus H.M., Marks D.I., Martino R., Maziarz R.T., Papanicolou G.A., et al. Risk factors and impact of non-Aspergillus mold infections following allogeneic HCT: A CIBMTR infection and immune reconstitution analysis. Bone Marrow Transplant. 2016;51:277–282. doi: 10.1038/bmt.2015.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Meyers J.D., Flournoy N., Thomas E.D. Nonbacterial Pneumonia After Allogeneic Marrow Transplantation: A Review of Ten Years’ Experience. Rev. Infect. Dis. 1982;4:1119–1132. doi: 10.1093/clinids/4.6.1119. [DOI] [PubMed] [Google Scholar]
- 59.Hughes W.T., Kuhn S., Chaudhary S., Feldman S., Verzosa M., Aur R.J.A., Pratt C., George S.L. Successful Chemoprophylaxis for Pneumocystis carinii Pneumonitis. N. Engl. J. Med. 1977;297:1419–1426. doi: 10.1056/NEJM197712292972602. [DOI] [PubMed] [Google Scholar]
- 60.Torres H.A., Chemaly R.F., Storey R., Aguilera E.A., Nogueras G.M., Safdar A., Rolston K.V.I., Raad I.I., Kontoyiannis D.P. Influence of type of cancer and hematopoietic stem cell transplantation on clinical presentation of Pneumocystis jiroveci pneumonia in cancer patients. Eur. J. Clin. Microbiol. Infect. Dis. 2006;25:382–388. doi: 10.1007/s10096-006-0149-4. [DOI] [PubMed] [Google Scholar]
- 61.De Castro N., Neuville S., Sarfati C., Ribaud P., Derouin F., Gluckman E., Socié G., Molina J.M. Occurrence of Pneumocystis jiroveci pneumonia after allogeneic stem cell transplantation: A 6-year retrospective study. Bone Marrow Transplant. 2005;36:879–883. doi: 10.1038/sj.bmt.1705149. [DOI] [PubMed] [Google Scholar]
- 62.Nematollahi S., Dioverti-Prono V. Cryptococcal infection in haematologic malignancies and haematopoietic stem cell transplantation. Mycoses. 2020;63:1033–1046. doi: 10.1111/myc.13153. [DOI] [PubMed] [Google Scholar]
- 63.Chitasombat M.N., Kofteridis D.P., Jiang Y., Tarrand J., Lewis R.E., Kontoyiannis D.P. Rare opportunistic (non-Candida, non-Cryptococcus) yeast bloodstream infections in patients with cancer. J. Infect. 2012;64:68–75. doi: 10.1016/j.jinf.2011.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lionakis M.S., Lewis R.E., Kontoyiannis D.P. Breakthrough invasive mold infections in the hematology patient: Current concepts and future directions. Clin. Infect. Dis. 2018;67:1621–1630. doi: 10.1093/cid/ciy473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lerolle N., Raffoux E., Socie G., Touratier S., Sauvageon H., Porcher R., Bretagne S., Bergeron A., Azoulay E., Molina J.M., et al. Breakthrough invasive fungal disease in patients receiving posaconazole primary prophylaxis: A 4-year study. Clin. Microbiol. Infect. 2014;20:O952–O959. doi: 10.1111/1469-0691.12688. [DOI] [PubMed] [Google Scholar]
- 66.Auberger J., Lass-Flörl C., Aigner M., Clausen J., Gastl G., Nachbaur D. Invasive fungal breakthrough infections, fungal colonization and emergence of resistant strains in high-risk patients receiving antifungal prophylaxis with posaconazole: Real-life data from a single-centre institutional retrospective observational study. J. Antimicrob. Chemother. 2012;67:2268–2273. doi: 10.1093/jac/dks189. [DOI] [PubMed] [Google Scholar]
- 67.Hoenigl M., Raggam R.B., Salzer H.J.F., Valentin T., Valentin A., Zollner-Schwetz I., Strohmeier A.T., Seeber K., Wölfler A., Sill H., et al. Posaconazole plasma concentrations and invasive mould infections in patients with haematological malignancies. Int. J. Antimicrob. Agents. 2012;39:510–513. doi: 10.1016/j.ijantimicag.2012.02.002. [DOI] [PubMed] [Google Scholar]
- 68.Tverdek F.P., Heo S.T., Aitken S.L., Granwehr B., Kontoyiannis D.P. Real-life assessment of the safety and effectiveness of the new tablet and intravenous formulations of posaconazole in the prophylaxis of invasive fungal infections via analysis of 343 courses. Antimicrob. Agents Chemother. 2017;61:e00188-17. doi: 10.1128/AAC.00188-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wingard J.R., Carter S.L., Walsh T.J., Kurtzberg J., Small T.N., Baden L.R., Gersten I.D., Mendizabal A.M., Leather H.L., Confer D.L., et al. Randomized, double-blind trial of fluconazole versus voriconazole for prevention of invasive fungal infection after allogeneic hematopoietic cell transplantation. Blood. 2010;116:5111–5118. doi: 10.1182/blood-2010-02-268151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Marks D.I., Pagliuca A., Kibbler C.C., Glasmacher A., Heussel C.P., Kantecki M., Miller P.J.S., Ribaud P., Schlamm H.T., Solano C., et al. Voriconazole versus itraconazole for antifungal prophylaxis following allogeneic haematopoietic stem-cell transplantation. Br. J. Haematol. 2011;155:318–327. doi: 10.1111/j.1365-2141.2011.08838.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Marty F.M., Cosimi L.A., Baden L.R. Breakthrough Zygomycosis after Voriconazole Treatment in Recipients of Hematopoietic Stem-Cell Transplants. N. Engl. J. Med. 2004;350:950–952. doi: 10.1056/NEJM200402263500923. [DOI] [PubMed] [Google Scholar]
- 72.Kim S.B., Cho S.Y., Lee D.G., Choi J.K., Lee H.J., Kim S.H., Park S.H., Choi S.M., Choi J.H., Yoo J.H., et al. Breakthrough invasive fungal diseases during voriconazole treatment for aspergillosis: A 5-year retrospective cohort study. Med. Mycol. 2017;55:237–245. doi: 10.1093/mmy/myw067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Imhof A., Balajee S.A., Fredricks D.N., Englund J.A., Marr K.A. Breakthrough fungal infections in stem cell transplant recipients receiving voriconazole. Clin. Infect. Dis. 2004;39:743–746. doi: 10.1086/423274. [DOI] [PubMed] [Google Scholar]
- 74.Trifilio S., Singhal S., Williams S., Frankfurt O., Gordon L., Evens A., Winter J., Tallman M., Pi J., Mehta J. Breakthrough fungal infections after allogeneic hematopoietic stem cell transplantation in patients on prophylactic voriconazole. Bone Marrow Transplant. 2007;40:451–456. doi: 10.1038/sj.bmt.1705754. [DOI] [PubMed] [Google Scholar]
- 75.Racil Z., Winterova J., Kouba M., Zak P., Malaskova L., Buresova L., Toskova M., Lengerova M., Kocmanova I., Weinbergerova B., et al. Monitoring trough voriconazole plasma concentrations in haematological patients: Real life multicentre experience. Mycoses. 2012;55:483–492. doi: 10.1111/j.1439-0507.2012.02186.x. [DOI] [PubMed] [Google Scholar]
- 76.Van Burik J.A.H., Ratanatharathorn V., Stepan D.E., Miller C.B., Lipton J.H., Vesole D.H., Bunin N., Wall D.A., Hiemenz J.W., Satoi Y., et al. Micafungin versus fluconazole for prophylaxis against invasive fungal infections during neutropenia in patients undergoing hematopoietic stem cell transplantation. Clin. Infect. Dis. 2004;39:1407–1416. doi: 10.1086/422312. [DOI] [PubMed] [Google Scholar]
- 77.Park S., Kim K., Jang J.H., Kim S.J., Kim W.S., Chung D.R., Kang C.I., Peck K.R., Jung C.W. Randomized trial of micafungin versus fluconazole as prophylaxis against invasive fungal infections in hematopoietic stem cell transplant recipients. J. Infect. 2016;73:496–505. doi: 10.1016/j.jinf.2016.06.011. [DOI] [PubMed] [Google Scholar]
- 78.Huang X., Chen H., Han M., Zou P., Wu D., Lai Y., Huang H., Chen X., Liu T., Zhu H., et al. Multicenter, Randomized, Open-Label Study Comparing the Efficacy and Safety of Micafungin versus Itraconazole for Prophylaxis of Invasive Fungal Infections in Patients undergoing Hematopoietic Stem Cell Transplant. Biol. Blood Marrow Transplant. 2012;18:1509–1516. doi: 10.1016/j.bbmt.2012.03.014. [DOI] [PubMed] [Google Scholar]
- 79.Dvorak C.C., Fisher B.T., Esbenshade A.J., Nieder M.L., Alexander S., Steinbach W.J., Dang H., Villaluna D., Chen L., Skeens M., et al. A Randomized Trial of Caspofungin vs Triazoles Prophylaxis for Invasive Fungal Disease in Pediatric Allogeneic Hematopoietic Cell Transplant. J. Pediatric Infect. Dis. Soc. 2021;10:417–425. doi: 10.1093/jpids/piaa119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pang K.A.P., Godet C., Fekkar A., Scholler J., Nivoix Y., Letscher-Bru V., Massias L., Kauffmann-Lacroix C., Elsendoorn A., Uzunov M., et al. Breakthrough invasive mould infections in patients treated with caspofungin. J. Infect. 2012;64:424–429. doi: 10.1016/j.jinf.2011.12.015. [DOI] [PubMed] [Google Scholar]
- 81.Neofytos D., Huang Y.T., Cheng K., Cohen N., Perales M.A., Barker J., Giralt S., Jakubowski A., Papanicolaou G. Safety and Efficacy of Intermittent Intravenous Administration of High-Dose Micafungin. Clin. Infect. Dis. 2015;61:S652–S661. doi: 10.1093/cid/civ818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Chou L.S., Lewis R.E., Ippoliti C., Champlin R.E., Kontoyiannis D.P. Caspofungin as primary antifungal prophylaxis in stem cell transplant recipients. Pharmacotherapy. 2007;27:1644–1650. doi: 10.1592/phco.27.12.1644. [DOI] [PubMed] [Google Scholar]
- 83.Yáñez L., Insunza A., Ibarrondo P., de Miguel C., Bermúdez A., Colorado M., López-Duarte M., Richard C., Conde E. Experience with anidulafungin in patients with allogeneic hematopoietic stem cell transplantation and graft-versus-host disease. Transpl. Infect. Dis. 2015;17:761–767. doi: 10.1111/tid.12429. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.