Abstract
Background & aims
There are some studies indicating the effects of probiotic-containing foods or supplements on viral diseases. We aimed to conduct a rapid review of probiotics with specific emphasis on their potential for early administration in patients at greater risk of SARS-CoV-2 infection.
Methods
We searched on PubMed, EMBASE, Google Scholar, Science Direct, Scopus and Web of Science up to February 2021 to identify interventional and observational studies documenting the effects of probiotics strains on interleukins, virus titers, and antibody production with a focus on probiotic-containing foods (PROSPERO Registration ID. CRD42020181453)
Results
From a total of 163 records, 21 studies were classified into three domains based on the efficacy of probiotics on 1) the level of interleukins (n = 7), 2) virus titers (n = 2), and 3) interferon (IFN) and antibody production (n = 12). The suppuration of pro-inflammatory interleukins and type I INF production seemed to be the main anti-viral effect of probiotics. Nine studies also indicated the beneficial effects of probiotics and fermented foods on viral diseases.
Conclusion
Based on evidence, some probiotic strains may be useful in viral infections; randomized trials are needed to confirm these findings.
Keywords: Probiotics, COVID-19, SARS-CoV-2, Virus diseases
Graphical abstract
1. Introduction
Severe Acute Respiratory Coronavirus 2 (SARS-CoV-2) is a new public health crisis threatening the humanity [1,2]. SARS-CoV-2 attaches to Angiotensin Converting Enzyme 2 (ACE2) receptor with 10–20 folds higher affinity than SARS-CoV [3]. Practically, antibiotic therapy results in more susceptibility to subsequent infections in severe COVID-19 patients [4]; one of the most irritating complication is antibiotic-associated diarrhea [5].
Probiotics are live microorganisms which when administered in adequate amounts confer a health benefit to the host [6]. Clinical evidence shows that certain probiotics may help treat and prevent viral infections [7,8] due to the proven immunomodulatory activity and ability to stimulate interferon (INF) production [9]. Based on evidence, the gut–lung axis explains the possible relationship between respiratory disorders and gut microbiome [10]. It was indicated that some probiotic strains activate the innate and the acquired immune system, resulting in an anti-viral protection [[11], [12], [13]].
Recently, some COVID-19 patients showed microbial dysbiosis with decreased Lactobacillus and Bifidobacterium [14]. In addition, gut microbiota diversity is reduced in elder peoples, who are vulnerable to severe forms of COVID-19 [15]. There are limited data on the effects of probiotics in COVID-19. Several different probiotics, including Lactobacillus acidophilus, Bifidobacterium and Saccharomyces boulardii, along with minerals and vitamins were given to a COVID-19 case; this type of nutritional support lowered the complications of massive antibiotic therapy (antibiotic-associated diarrhea and recurrent Clostridioides Difficile infections) [16]. Furthermore, another author suggested the concurrent use of probiotics in COVID-19 patients to decrease the risk for Candida albicans (caused by prophylaxis with azithromycin) [17].
In another report, COVID-19-like symptoms disappeared after two days administration of oral probiotic in a 9 years-old boy [18]. Results of a case series - consists of 62 SARS-CoV2 infected patients in Zhejiang province - was interesting. In this research, probiotics were administered as adjunct and the authors reported that only two patients (3%) developed shortness of breath on admission. Moreover, only one was admitted to an intensive care unit [19]. Other reports showed a significant effect of probiotics; compared to patients with non-severe disease, patients with severe disease were more likely to be treated by probiotics therapy (87.5% vs 40.4%, p = 0.037) [20].
According to recent data, more than ten clinical trials have been registered regarding the probiotics supplementation in COVID-19 patients [21], however, more information is needed for designing future research protocols. We aimed to summarize the effects of probiotic supplementation on interleukins, viral titers, interferons, and antibody production in viral infections especially SARS-CoV-2.
2. Methods
2.1. Protocol registration
The protocol of this systematic review has been registered on PROSPERO website (www.crd.york.ac.uk/PROSPERO) developed based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement guidelines [22]. The selected studies did not provide sufficient data for quantitative analysis, therefore, all studies were just systematically reviewed.
2.2. Search strategy
A comprehensive search of the literature was conducted in the following databases up to June 30, 2020: PubMed, EMBASE, Google Scholar, Science Direct, Scopus, and Web of Science. All citations were imported into a bibliographic database (EndNote X8.1; Thomson Reuters) and duplicates were removed. The search strategy was handled independently by two authors.
The used search string - based on suitable MESH and non-MESH keywords - were: ((“COVID-19” OR “SARS-CoV-2” OR “Severe Acute Respiratory Coronavirus 2” OR “Coronavirus” OR “Virus Disease” OR “Viral Infection” OR “Virus”) AND (“Virus Titers” OR “Titers” OR “Interleukin” OR “IL-6” OR “IL-17” OR “Antibody” OR “IgG” OR “IgA” OR “Interferon” OR “INF”) AND (“Synbiotics” OR “Probiotics” OR “Prebiotics” OR “Probiotic Milk” OR “Probiotic Yoghurt” OR “Probiotic Honey” OR “Probiotic Food” OR “Fermented Foods”)). Moreover, bibliographies of all published reviews and studies were assessed for additional relevant papers.
2.3. Study selection criteria, data extraction and quality assessment
Title, abstract, and full-text of all articles were screened to select and extract studies that investigated the effect of probiotics on viral infections (with emphasis on SARS-CoV-2) in English language (all research types with both human and animal origin). We excluded HIV papers, due to not being directly related to respiratory viral infections. Data extraction from primary articles was performed by one author by using a standardized form. A second author checked the accuracy of the data extracted. Data collected from the studies included authors’ family name, type of study, probiotic strains and dose, sample size, and overall outcomes. The cochrane collaboration criteria for assessing risk of bias were applied to assess the quality of the studies included in the review [23].
2.4. Risk of bias assessment
The included studies were evaluated for bias by using the Cochrane Risk-Of-Bias. Each included study was evaluated for the following biases: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participant and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias) and other bias. The reviewers’ judgment was classified as “Low risk,” “High risk” or “Unclear risk” of bias.
3. Results
A diagram showed the details of included studies (Fig. 1 ). 163 records were identified initially from selected databases. After excluding duplicates and articles that did not meet the inclusion criteria, we obtained 58 papers with full-texts, which were read for further evaluation. Another 28 papers were excluded due to insufficient data and duplication. In general, we included 30 articles for qualitative analysis; 21 studies were classified into three domains based on the efficacy of probiotics on 1) the level of interleukins (n = 7), 2) virus titers (n = 2), and 3) interferon and antibody production (n = 12). Another nine papers evaluated the efficacy of probiotics-containing and fermented foods on viral diseases.
Fig. 1.
Flow diagram of the different phases of systematic review.
3.1. Risk of bias report
Risk of bias was low in the following domains: selection bias (random sequence generation and allocation concealment, eight and twelve studies, respectively), performance bias (nine studies), detection bias (thirteen studies), attrition bias (two studies), and reporting bias (four studies). In overall state, thirteen studies had low risk of bias, three studies seemed to be at a high risk of attrition bias. Moreover, five studies had unclear risk of bias.
3.2. Major effects of probiotics in viral infections
The effects of probiotics supplementation on interleukins, viral titers and antibody production were briefly displayed in Fig. 2 . Moreover, clinical evidence for the role of probiotics in COVID-19 - up to June 2021 - was summarized in Fig. 3 .
Fig. 2.
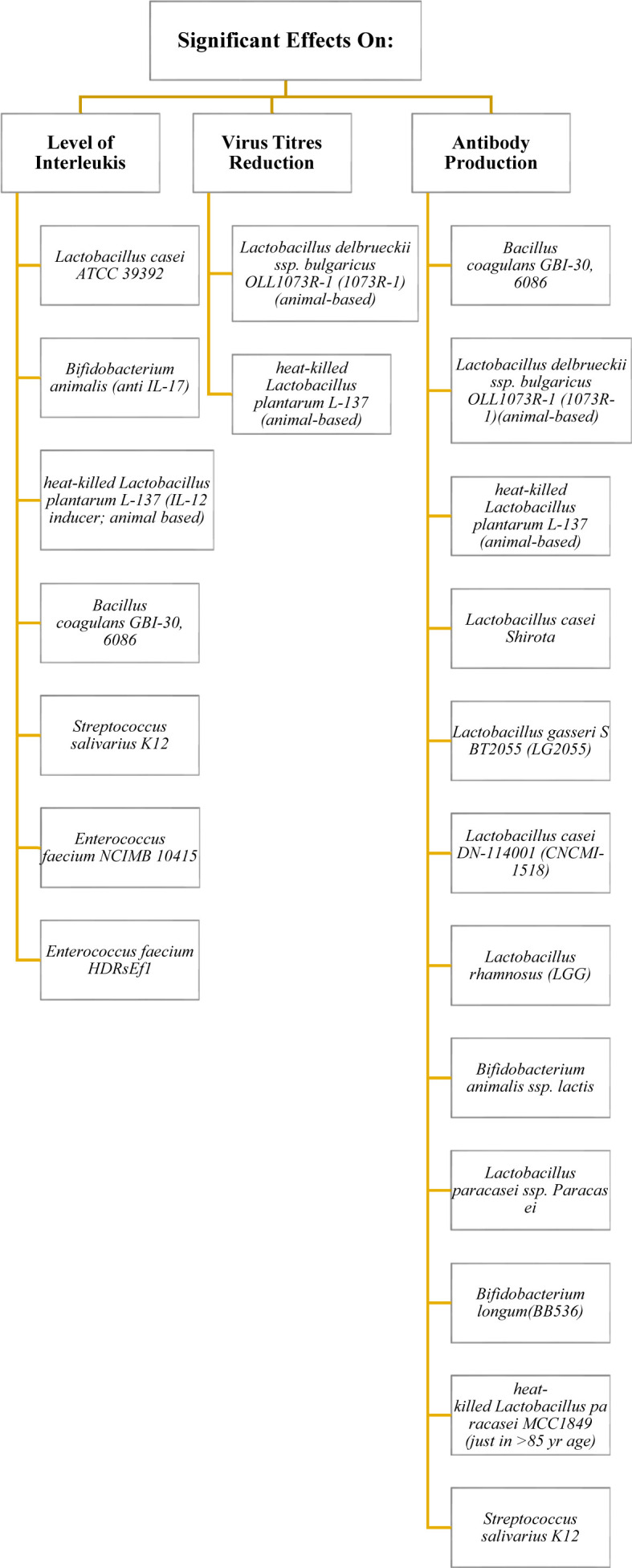
Evidence-based efficacy of some probiotic strains on interleukins, virus titers and antibody production in viral infections.
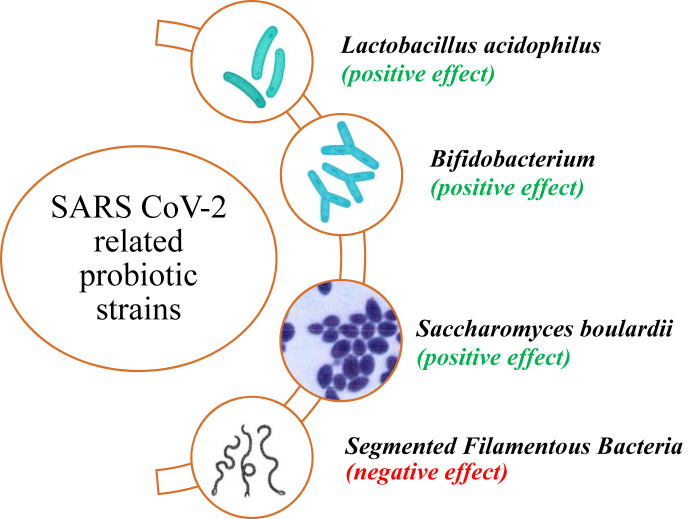
Fig. 3.
The probiotics strains that have been reported in the context of COVID-19.
3.2.1. Effects on levels of interleukins
Jiang et al. [24] administered an oral Lactobacillus casei ATCC 39392 vaccine against anti-transmissible gastroenteritis virus (TGEV) in piglets. This vaccine polarized Th2 cell immunity, stimulated Interleukin (IL)-17 expression, and induced an anti-TGEV infection Th17 pathway. However, Bozkurt et al. [25] reported that the Bifidobacterium animalis strain may be effective in preventing colorectal cancer through non-stimulatory effects on Th17 (and IL-17) cells, and a promoting effect on Th1 cells. In another study, the strain ‘heat-killed Lactobacillus plantarum L-137’ was introduced as an IL-12 inducer in mice [26].
In a trial conducted by Kimmel et al. [27], ten healthy Subjects consumed one capsule/day of Bacillus coagulans GBI-30 6086 (GanedenBC30) for 28 days and returned for post-treatment immunological markers evaluation; IL-6, IL-8, and Tumor Necrosis Factor-alpha (TNF-α) levels were increased after exposure to a strain of adenovirus (AdenoVI) and influenza A (H3N2 Texas strain; FluTex). IL-1beta levels also increased after exposure to AdenoVI but were reduced after ex vivo exposure to FluTex.
Abt et al. [28] described the antiviral activity of Streptococcus salivarius K12. Some hypotheses indicated that oral administration of the S. salivarius K12, through a still not perfectly clear molecular anti-inflammatory mechanism, reduced IL-8 plasma concentrations and increased salivary INF-γ. These modulations may also realistically account for anti-inflammatory and antiviral activity, which would then be added to the antibacterial action of the K12 strain [29]. However, it seems that not all S. salivarius strains elicit similar immune responses, since S. salivarius ATCC 25975 was reported to upregulate IL-6, IL-8 and TNF-α gene expression [30].
Coronaviruses may also be vulnerable to probiotics. Chi et al. [31] noted that pretreating swine testicular cells with Enterococcus faecium NCIMB 10415, which is approved as a feed additive in the European Union, led to protective effects against the enteropathogenic coronavirus transmissible gastroenteritis virus (TGEV). A stimulated cellular defense was suggested as the underlying mechanism because of increased cellular production of nitric oxide and IL-6 and IL-8 expression. Although E. faecium NCIMB 10415 reduced all viral structural proteins, this strain significantly increased the production of pro-inflammatory factors IL-6 and IL-8 in TGEV-infected ST cells. In contrast, Tian et al. [32] claimed that E. faecium HDRsEf1 can significantly downregulate the mRNA level of pro-inflammatory factors IL-1β, IL-6, IL-8, IL-12, IL-17, and TNF-α.
3.2.2. Effects on viral titers
In an animal study, the administration of yogurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1 (1073R-1) for 21 days significantly decreased influenza virus titers in infected mice [33].
Results of another animal study, conducted by Maeda et al. [34], showed that heat-killed Lactobacillus plantarum L-137 can lower the viral titers of influenza virus A/FM/1/47 (H1N1, a mouse-adapted strain) in infected mice, however, researchers demonstrated this effect on humans [35]. Contrary, the strain “heat-treated Enterococcus faecalis FK-23” during 6–36 months administration for 39 adult HCV-positive subjects could reduce Alanine Aminotransferase (ALT) and Aspartate Transaminase (AST) with no significant effect on viral load [36].
3.2.3. Effects on interferon and antibody production
IgA and type 1 Interferons (IFNs) production are direct mediators of protection against viral infections especially in SARS-CoV-2 [37]. Based on previous findings, the consumption of one capsule/day Bacillus coagulans GBI-30 6086 (GanedenBC30) for 28 days increased CD3+CD69+ cells and IFN-gamma levels [27]. Moreover, Nagai et al. [33] reported that Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1 (1073R-1) increases anti-influenza virus (A/PR/8/34 (H1N1)) antibodies (IgA, IgG1) in an animal model. Interestingly, heat-killed Lactobacillus plantarum L-137 can act as an IFN-β inducer [34].
Lactobacillus casei Shirota, another potential probiotic, was capable to reduce plasma antibody titers in cytomegalovirus and Epstein–Barr virus infected people through modulation the activity of Natural Killer (NK) cells [38]; but it did not have any therapeutic effect on norovirus-induced gastroenteritis [39].
Oral administration of yogurt containing Lactobacillus gasseri SBT2055 (LG2055) in healthy adult subjects potentiated vaccine-specific antibody production against A/H1N1 and B influenza viruses [40]. In another trial, titers against the influenza B strain increased significantly in group receiving Lactobacillus casei DN-114 001 (CNCMI-1518) for 13 weeks [41].
Another strain, Lactobacillus rhamnosus GG (LGG), when administered for 4 weeks to children with gastroenteritis, who were positive for either rotavirus or Cryptosporidium species in stool, caused a significant increase in serum Immunoglobulin (Ig) G levels [42]. According to evidence, the titers of vaccine-specific IgG, IgG1, and IgG3 in plasma as well as that of vaccine-specific secretory IgA in saliva were significantly greater in those who received both probiotics “B. animalis ssp. lactis and Lactobacillus paracasei ssp. Paracasei (109 CFU for 6 weeks)” [43]. Furthermore, 172 full-term infants who were fed probiotic formula 106 CFU/g B. animalis subspecies lactis (Bb12), had more anti-poliovirus-specific IgA in the feces sample, but not anti-rotavirus-specific IgA [44].
Akatsu et al. [45] highlighted a significant association between Bifidobacterium and increased immune function and intestinal microbiota in the elderly; administration of Bifidobacterium longum (BB536) for 5 weeks resulted in an increase of serum IgA. BB536 did not significantly affect hemagglutination inhibition in response to influenza vaccine. NK cell activity did not also decrease significantly in probiotic group.
Interestingly, Receiving a jelly containing 10 billion heat-killed Lactobacillus paracasei MCC189 cells for 6 weeks could improve the antibody responses to the A/H1N1 and B antigens in ≥85 years of age subgroup (n = 27), however, the overall results were not significant in comparison with placebo group [46].
3.3. Major effects of probiotic-containing and fermented foods on viral infections
Probiotic milk, yoghurt and honey are the most common probiotics-containing foods worldwide. There are some reports indicating the beneficial effects of these foods on viral diseases. Human milk is the main source of Lactic Acid Bacteria (LAB); LAB indicated a protective effect against rotavirus and TGEV [47]. Moreover, two strains Lacticaseibacillus rhamnosus LHL6 and LHL7 have significant probiotic property in breast milk [48]. Administration of L. rhamnosus showed a beneficial effects on the protection against enteric viruses in vivo [49].
Kimchi, a traditional Korean fermented vegetable food, has a variety of probiotic strains [50] which may induce the immune response against the viral infections. In 2018, researchers demonstrated that a new Lactobacillus plantarum strain (LRCC5310) - derived from kimchi-inhibits the adherence and proliferation of rotavirus in vivo and in vitro [51]. Shin et al. [52] also reported that LRCC5310 inhibit viral proliferation in human rotavirus-induced gastroenteritis.
Evidence showed that dietary intake of fermented dairy products reduces the risk of infectious diseases and enhances immune cell activity [[53], [54], [55]]. The administration of lactobacillus plantarum JLK0142 isolated from fermented dairy tofu significantly improved phagocytic activity of macrophages and the secretion of NO in cyclophosphamide-induced immunosuppressed mice [56]. Another study showed that oral administration of milk fermented with Lactococcus lactis subsp. cremoris FC protects mice against influenza viral infection [57]. In 2019, Rigo-Adrover et al. [58] reported that a fermented milk, made by Bifidobacterium breve C50 (BbC50), Streptococcus thermophilus 065 (St065), and prebiotic mixture short chain galactooligosaccharides/long chain fructooligosaccharides, prevents from rotavirus diarrhea in suckling rats. Although there are positive findings about the antiviral effects of fermented milk [33,59,60], future research is needed to make a robust conclusion.
In a trial, the dietary intake of yogurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1 did not show a significant preventive effect against influenza or a significant enhancement in NK cell activity. However, intake of this yogurt showed an increase in IFN-γ production [61]. Previous investigations also revealed that the consumption of yoghurt fermented with L. bulgaricus OLL1073R-1 increases NK cell activity and reduces the risk of common cold occurrence in elderly individuals [62].
However, it is notable that fermented food may interact with other probiotic-based products. Hishiki et al. [63] indicated that a probiotic strain of LAB, Pediococcus acidilactici K15, has a beneficial effect on the prevention of respiratory tract infections just for children who eat less than two fermented food or yogurt items per week. According to findings, probiotic-based and fermented foods are beneficial for the prevention and treatment of viral diseases, however, these effects should be investigated in SARS-CoV2 infection [64].
4. Discussion
Here we reviewed clinical trials on single probiotic or probiotic-containing foods, which potentially affected viral diseases. Probiotic strains like Lactobacillus plantarum, Lactobacillus casei, B. animalis, Bacillus coagulans, S. salivarius, and E. faecium had inhibitory effects on pro-inflammatory interleukins. Moreover, several conventional probiotics (Lactobacillus gasseri, Lactobacillus rhamnosus, B. longum) potentially increased the level of antibodies in viral infections.
According to recent evidence, gut dysbiosis occurs in COVID-19; gut–lung axis greatly affects the severity of the pulmonary complications [65,66]. D'Ettorre et al. [67] observed that the administration of probiotic mixture (L.brevis DSM27961, L. acidophilus DSM32241, L. helveticus DSM32242, L. paracasei DSM32243, L. plantarum DSM32244, S. thermophilus DSM32245, Bifidobacterium lactis DSM32246, and B. lactis DSM32247) for 2 weeks at 2.4 × 1012 CFU/day have beneficial effect on diarrhea and respiratory failure in COVID-19 patients, however, they did not conducted an initial gut microbiome screening before intervention. Altered intestinal microbiomes have been detected in COVID-19 patients [[68], [69], [70], [71]], therefore, nutritional interventions such as probiotics and prebiotics may be beneficial as a fundamental therapeutic technique [[72], [73]].
Coronaviruses (CoVs) involve a large family of surrounded, positive-stranded RNA viruses that infect a wide range of animal hosts as well as humans. The most recognized coronavirus, the Middle East respiratory syndrome virus, was found in Africa and Asia [74]. The currently available antiviral agents for management of CoV-induced infections does not take into consideration the effect of other factors on the treatment process including intestinal microbiota. Lactobacilli and Bifidobacteria are two conventional probiotics that can really tip the balance of a diverse gut ecosystem in combating COVID-19 [14].
There are some mechanisms explaining the antiviral impacts of probiotics. Some authors claimed that high-single dose endoscopic administration of Bifidobacterium Infantis (BB-12), as a potential probiotic, or bacterial lipopolysaccharide membrane in vaccine production can result in more therapeutic and preventive effects in CoV-infected patients especially in individuals with gastrointestinal symptoms (diarrhea, abdomen pain, vomiting) [[75], [76], [77]]. Moreover, B. animalis theoretically can inhibit the replication of CoVs by reducing endoplasmic reticulum stress-related autophagy, especially through the Inositol-Requiring Enzyme 1 (IRE1) pathway, over its anti interleukin-17 effect [25]. Apparently, the main pathogenic mechanism in the viral infection-induced pneumonia seems to be a “cytokine storm”; IL-6 is the main pro-inflammatory marker in the infectious diseases [78]. Coomes et al. [79] in a meta-analysis concluded that inhibition of IL-6 may be a novel target for therapeutic goals in patients with COVID-19. The current state of knowledge on the immunomodulatory effects of probiotics has recently advanced [80].
Some probiotics have potency to induce the IFN production pathways. L. lactis JCM5805 can activate human plasmacytoid Dendritic Cells (pDCs) in vitro; pDCs play a crucial role in antiviral immunity as proficient type I IFN producing cells (IPCs) [81,82] and mucosal T cell independent IgA production [83]. pDC-derived type I IFNs can indirectly inhibit viral replication and spread [84], and activation of NK cells [83]. Two distinct types of Toll-Like Receptors (TLR7 and TLR9) - expressed by pDCs - have some molecular role [85]; TLR7 recognizes the microbial RNA of virus [86,87], and TLR9 senses the ssDNA containing CpG motifs [88,89]. Upon sensing the viral nucleic acid, Interferon Regulatory Factor 7 (IRF7) is activated, phosphorylated, and translocated into the nucleus to begin the transcription of type I IFNs [90]. In conclusion, this strain had many important roles in both innate immunity and adaptive immunity. In addition, several human-based trials confirmed this observation (influenza virus as main target) [[91], [92], [93]]. Furthermore, some probiotic strains such as E. faecium HDRsEf1 could significantly downregulate the mRNA level of TLR4, TLR5, TLR7, and TLR8 [32].
As discussed earlier, there have also been negative reports against probiotics. According to scRNA-Seq analysis, Feng et al. found that the SARS-Cov-2 receptor, ACE2, could be elevated in the presence of both invasive bacteria Salmonella Enterica and its counterpart, Segmented Filamentous Bacteria as probiotics in the mouse small intestine [94] and human enterocytes [95]. in another study Lactobacillus acidophilus and Bacillus clausii also failed to decrease the coronavirus receptors expression in the murine small intestine compared to control and post Salmonella infection [96].
In total, the US Food and Drug Administration classifies probiotics as Generally Recognized As Safe (GRAS) organisms, meaning that the risks of probiotics administration seem to be low, however, the amount and type of probiotic strains are also considerable [[97], [98], [99]]. Notably, unconfirmed prescription of conventional probiotics for COVID-19 is not recommended until further investigations regarding the pathogenesis of SARS-CoV-2 and their effect on gut microbiota were published.
A few limitations of this review shall be highlighted. The systematic review table was not designed due to inconsistent data of relevant studies. Moreover, meta-analysis has not been performed due to heterogeneity of studies, especially in relation to published data for SARS-CoV-2. In addition, we excluded a large number of research on supplementation of different nutrients, which were related to immune function. However, the main strength of the current study was that we performed an exclusive investigation for diseases of the same origin i.e. viral diseases, especially COVID-19.
5. Conclusion
We described the efficacy of probiotics for the prevention or treatment of viral-based infectious diseases; more than twenty strains improved the anti-inflammatory interleukins and anti-body production against viruses. Moreover, virus titers were lowered after probiotics supplementation. Probiotic-containing foods and fermented food products also showed significant effect on prevention and treatment of viral diseases. The large number of viral species and their subtypes as well as the high mutation rate of viruses do not allow scientists to discover appropriate vaccines and antiviral drugs, therefore, the administration of pro/prebiotics - as immune function modulators and antibiotic-related side effects removers - is recommended. Although further detailed research is necessary, the authors recommend researchers/physicians/dietitians to use probiotics as more rational adjunctive option in COVID-19 pandemic, especially in mechanically ventilated patients.
Contributors
Shahrzad Mirashrafi: Writing-Original draft preparation and Software.
Amir Reza Moravejolahkami: Conceptualization, Writing-Original draft preparation and Software, Critical Reviewing and Editing the Final Version of the Manuscript, Assessing Study Eligibility and Conducting Quality Assessments, Supervision, Critical Reviewing and Editing the Final Version of the Manuscript.
Zakiyeh Balouch Zehi: Investigation, Visualization.
Mohammad Ali Hojjati Kermani, Marjan Ganjali Dashti: Assessing Study Eligibility and Conducting Quality.
Mohsen Haratian: Validation.
Meraj Pourhossein, Nimah Bahreini-Esfahani: Critical Reviewing and Editing the Final Version of the Manuscript.
Funding
The authors declare no support from any commercial organization for the submitted study.
Declaration of competing interest
None of the authors had a conflict of interest.
Acknowledgement
We thank all the nurses, doctors, clinicians and researchers during COVID-19 pandemic.
This study protocol was registered on International prospective register of systematic reviews (PROSPERO):
(https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=181453).
Registration ID. CRD42020181453.
Footnotes
This study has not been duplicate publication or submission elsewhere. The authors received no financial support for the research and/or authorship of this article. The authors declare that they have no conflict of interest to the publication of this article.
References
- 1.Malik Y.S., Sircar S., Bhat S., Vinodhkumar O.R., Tiwari R., Sah R., et al. 2020. Emerging Coronavirus Disease (COVID-19), a pandemic public health emergency with animal linkages: current status update. [Google Scholar]
- 2.Lipsitch M., Swerdlow D.L., Finelli L. Defining the epidemiology of Covid-19—studies needed. N Engl J Med. 2020;382(13):1194–1196. doi: 10.1056/NEJMp2002125. [DOI] [PubMed] [Google Scholar]
- 3.Xu X., Chen P., Wang J., Feng J., Zhou H., Li X., et al. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci China Life Sci. 2020;63(3):457–460. doi: 10.1007/s11427-020-1637-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mak J.W., Chan F.K., Ng S.C. Probiotics and COVID-19: one size does not fit all. Lancet Gastroenterol & Hepatol. 2020;5(7):644–645. doi: 10.1016/S2468-1253(20)30122-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wei X.-S., Wang X., Niu Y.-R., Ye L.-L., Peng W.-B., Wang Z.-H., et al. Diarrhea is associated with prolonged symptoms and viral carriage in COVID-19. Clin Gastroenterol Hepatol. 2020;18(8):1753–1759. doi: 10.1016/j.cgh.2020.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bahreini-Esfahani N., Moravejolahkami A.R. Can synbiotic dietary pattern predict lactobacillales strains in breast milk? Breastfeed Med. 2020;15(6):387–393. doi: 10.1089/bfm.2019.0301. [DOI] [PubMed] [Google Scholar]
- 7.Mousa H.A.-L. Prevention and treatment of influenza, influenza-like illness, and common cold by herbal, complementary, and natural therapies. J Evidence-Based Complement & Alt Med. 2017;22(1):166–174. doi: 10.1177/2156587216641831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Paknahad Z., Moravejolahkami A.R. Probiotics against viruses; COVID-19 is a paper tiger: a Systematic Review. Endocr Metab Immune Disord - Drug Targets. 2020;21(7):1252–1260. doi: 10.2174/1871530320666200917114033. [DOI] [PubMed] [Google Scholar]
- 9.Bohlouli J., Namjoo I., Borzoo-Isfahani M., Kermani M.A.H., Zehi Z.B., Moravejolahkami A.R. Effect of probiotics on oxidative stress and inflammatory status in diabetic nephropathy: a systematic review and meta-analysis of clinical trials. Heliyon. 2021;7(1) doi: 10.1016/j.heliyon.2021.e05925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Marsland B.J., Trompette A., Gollwitzer E.S. The gut–lung axis in respiratory disease. Annals Am Thoracic Soc. 2015;12(Supplement 2):S150–S156. doi: 10.1513/AnnalsATS.201503-133AW. [DOI] [PubMed] [Google Scholar]
- 11.Asama T., Uematsu T., Kobayashi N., Tatefuji T., Hashimoto K. Oral administration of heat-killed Lactobacillus kunkeei YB38 improves murine influenza pneumonia by enhancing IgA production. Bioscience of Microbiota, Food and Health. 2017;36(1):1–9. doi: 10.12938/bmfh.16-010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kanauchi O., Andoh A., AbuBakar S., Yamamoto N. Probiotics and paraprobiotics in viral infection: clinical application and effects on the innate and acquired immune systems. Curr Pharmaceut Des. 2018;24(6):710–717. doi: 10.2174/1381612824666180116163411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Michael H., Paim F.C., Miyazaki A., Langel S.N., Fischer D.D., Chepngeno J., et al. Escherichia coli Nissle 1917 administered as a dextranomar microsphere biofilm enhances immune responses against human rotavirus in a neonatal malnourished pig model colonized with human infant fecal microbiota. PLoS One. 2021;16(2) doi: 10.1371/journal.pone.0246193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Xu K., Cai H., Shen Y., Ni Q., Chen Y., Hu S., et al. Management of corona virus disease-19 (COVID-19): the Zhejiang experience. J Zhejiang Univ. 2020;49(1) doi: 10.3785/j.issn.1008-9292.2020.02.02. 0-. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dhar D., Mohanty A. Gut microbiota and Covid-19-possible link and implications. Virus Res. 2020:198018. doi: 10.1016/j.virusres.2020.198018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Horowitz R.I., Freeman P.R., Bruzzese J. Efficacy of glutathione therapy in relieving dyspnea associated with COVID-19 pneumonia: a report of 2 cases. Respiratory Med Case Reports. 2020:101063. doi: 10.1016/j.rmcr.2020.101063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Block J. 2020. High risk COVID-19: potential intervention at multiple points in the COVID-19 disease process via prophylactic treatment with azithromycin or bee derived products. [Google Scholar]
- 18.Ji L.-N., Chao S., Wang Y.-J., Li X.-J., Mu X.-D., Lin M.-G., et al. Clinical features of pediatric patients with COVID-19: a report of two family cluster cases. World J Pediatr. 2020:1–4. doi: 10.1007/s12519-020-00356-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Xu X.-W., Wu X.-X., Jiang X.-G., Xu K.-J., Ying L.-J., Ma C.-L., et al. Clinical findings in a group of patients infected with the 2019 novel coronavirus (SARS-Cov-2) outside of Wuhan, China: retrospective case series. Br Med J. 2020:368. doi: 10.1136/bmj.m606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jiang X., Tao J., Wu H., Wang Y., Zhao W., Zhou M., et al. Clinical features and management of severe COVID-19: a retrospective study in Wuxi, Jiangsu Province, China. MedRxiv. 2020 [Google Scholar]
- 21.Hu J., Zhang L., Lin W., Tang W., Chan F.K., Ng S.C. Probiotics, prebiotics and dietary approaches during COVID-19 pandemic. Trends Food Sci Technol. 2020 doi: 10.1016/j.tifs.2020.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liberati A., Altman D.G., Tetzlaff J., Mulrow C., Gøtzsche P.C., Ioannidis J.P., et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann Intern Med. 2009;151(4) doi: 10.7326/0003-4819-151-4-200908180-00136. W-65-W-94. [DOI] [PubMed] [Google Scholar]
- 23.Higgins J.P., Green S. John Wiley & Sons; 2011. Cochrane handbook for systematic reviews of interventions. [Google Scholar]
- 24.Jiang X., Hou X., Tang L., Jiang Y., Ma G., Li Y. A phase trial of the oral Lactobacillus casei vaccine polarizes Th2 cell immunity against transmissible gastroenteritis coronavirus infection. Appl Microbiol Biotechnol. 2016;100(17):7457–7469. doi: 10.1007/s00253-016-7424-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bozkurt H.S., Quigley E.M. Bifidobacteria and mucosal-associated invariant T (mait) cells: a new approach to colorectal cancer prevention? Gastrointestinal Disorders. 2019;1(2):266–272. [Google Scholar]
- 26.Murosaki S., Yamamoto Y., Ito K., Inokuchi T., Kusaka H., Ikeda H., et al. Heat-killed Lactobacillus plantarum L-137 suppresses naturally fed antigen–specific IgE production by stimulation of IL-12 production in mice. J Allergy Clin Immunol. 1998;102(1):57–64. doi: 10.1016/s0091-6749(98)70055-7. [DOI] [PubMed] [Google Scholar]
- 27.Kimmel M., Keller D., Farmer S., Warrino D. A controlled clinical trial to evaluate the effect of GanedenBC (30) on immunological markers. Methods Find Exp Clin Pharmacol. 2010;32(2):129–132. doi: 10.1358/mf.2010.32.2.1423881. [DOI] [PubMed] [Google Scholar]
- 28.Abt M.C., Osborne L.C., Monticelli L.A., Doering T.A., Alenghat T., Sonnenberg G.F., et al. Commensal bacteria calibrate the activation threshold of innate antiviral immunity. Immunity. 2012;37(1):158–170. doi: 10.1016/j.immuni.2012.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wescombe P.A., Hale J.D., Heng N.C., Tagg J.R. Developing oral probiotics from Streptococcus salivarius. Future Microbiol. 2012;7(12):1355–1371. doi: 10.2217/fmb.12.113. [DOI] [PubMed] [Google Scholar]
- 30.Mostefaoui Y., Bart C., Frenette M., Rouabhia M. Candida albicans and Streptococcus salivarius modulate IL-6, IL-8, and TNF-α expression and secretion by engineered human oral mucosa cells. Cell Microbiol. 2004;6(11):1085–1096. doi: 10.1111/j.1462-5822.2004.00420.x. [DOI] [PubMed] [Google Scholar]
- 31.Chai W., Burwinkel M., Wang Z., Palissa C., Esch B., Twardziok S., et al. Antiviral effects of a probiotic Enterococcus faecium strain against transmissible gastroenteritis coronavirus. Arch Virol. 2013;158(4):799–807. doi: 10.1007/s00705-012-1543-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tian Z., Yang L., Li P., Xiao Y., Peng J., Wang X., et al. The inflammation regulation effects of Enterococcus faecium HDRsEf1 on human enterocyte-like HT-29 cells. Anim Cell Syst. 2016;20(2):70–76. [Google Scholar]
- 33.Nagai T., Makino S., Ikegami S., Itoh H., Yamada H. Effects of oral administration of yogurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1 and its exopolysaccharides against influenza virus infection in mice. Int Immunopharm. 2011;11(12):2246–2250. doi: 10.1016/j.intimp.2011.09.012. [DOI] [PubMed] [Google Scholar]
- 34.Maeda N., Nakamura R., Hirose Y., Murosaki S., Yamamoto Y., Kase T., et al. Oral administration of heat-killed Lactobacillus plantarum L-137 enhances protection against influenza virus infection by stimulation of type I interferon production in mice. Int Immunopharm. 2009;9(9):1122–1125. doi: 10.1016/j.intimp.2009.04.015. [DOI] [PubMed] [Google Scholar]
- 35.Arimori Y., Nakamura R., Hirose Y., Murosaki S., Yamamoto Y., Shidara O., et al. Daily intake of heat-killed Lactobacillus plantarum L-137 enhances type I interferon production in healthy humans and pigs. Immunopharmacol Immunotoxicol. 2012;34(6):937–943. doi: 10.3109/08923973.2012.672425. [DOI] [PubMed] [Google Scholar]
- 36.Oo K.M., Ayelwin A., Kyaw Y.Y., Tun W.M., Fukada K., Goshima A., et al. Safety and long-term effect of the probiotic FK-23 in patients with hepatitis C virus infection. Bioscience Microbiota, Food and Health. 2016:2015–2024. doi: 10.12938/bmfh.2015-024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.King C., Sprent J. The dual nature of type-I interferons in SARS-CoV-2 induced inflammation. Trends Immunol. 2021;108:187–196. doi: 10.1016/j.it.2021.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kawai T., Akira S. Innate immune recognition of viral infection. Nat Immunol. 2006;7(2):131–137. doi: 10.1038/ni1303. [DOI] [PubMed] [Google Scholar]
- 39.Nagata S., Asahara T., Ohta T., Yamada T., Kondo S., Bian L., et al. Effect of the continuous intake of probiotic-fermented milk containing Lactobacillus casei strain Shirota on fever in a mass outbreak of norovirus gastroenteritis and the faecal microflora in a health service facility for the aged. Br J Nutr. 2011;106(4):549–556. doi: 10.1017/S000711451100064X. [DOI] [PubMed] [Google Scholar]
- 40.Nishihira J., Nishimura M., Moriya T., Sakai F., Kabuki T., Kawasaki Y. Lactobacillus gasseri potentiates immune response against influenza virus infection. Immunity and Inflammation in Health and Disease. 2018:249–255. Elsevier. [Google Scholar]
- 41.Boge T., Rémigy M., Vaudaine S., Tanguy J., Bourdet-Sicard R., Van Der Werf S. A probiotic fermented dairy drink improves antibody response to influenza vaccination in the elderly in two randomised controlled trials. Vaccine. 2009;27(41):5677–5684. doi: 10.1016/j.vaccine.2009.06.094. [DOI] [PubMed] [Google Scholar]
- 42.Sindhu K.N., Sowmyanarayanan T.V., Paul A., Babji S., Ajjampur S.S., Priyadarshini S., et al. Immune response and intestinal permeability in children with acute gastroenteritis treated with Lactobacillus rhamnosus GG: a randomized, double-blind, placebo-controlled trial. Clin Infect Dis. 2014;58(8):1107–1115. doi: 10.1093/cid/ciu065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rizzardini G., Eskesen D., Calder P.C., Capetti A., Jespersen L., Clerici M. Evaluation of the immune benefits of two probiotic strains Bifidobacterium animalis ssp. lactis, BB-12® and Lactobacillus paracasei ssp. paracasei, L. casei 431® in an influenza vaccination model: a randomised, double-blind, placebo-controlled study. Br J Nutr. 2012;107(6):876–884. doi: 10.1017/S000711451100420X. [DOI] [PubMed] [Google Scholar]
- 44.Holscher H.D., Czerkies L.A., Cekola P., Litov R., Benbow M., Santema S., et al. Bifidobacterium lactis Bb12 enhances intestinal antibody response in formula-fed infants: a randomized, double-blind, controlled trial. J Parenter Enteral Nutr. 2012;36 doi: 10.1177/0148607111430817. 106S-17S. [DOI] [PubMed] [Google Scholar]
- 45.Akatsu H., Iwabuchi N., Xiao Jz, Matsuyama Z., Kurihara R., Okuda K., et al. Clinical effects of probiotic Bifidobacterium longum BB536 on immune function and intestinal microbiota in elderly patients receiving enteral tube feeding. J Parenter Enteral Nutr. 2013;37(5):631–640. doi: 10.1177/0148607112467819. [DOI] [PubMed] [Google Scholar]
- 46.Maruyama M., Abe R., Shimono T., Iwabuchi N., Abe F., Xiao J.-Z. The effects of non-viable Lactobacillus on immune function in the elderly: a randomised, double-blind, placebo-controlled study. Int J Food Sci Nutr. 2016;67(1):67–73. doi: 10.3109/09637486.2015.1126564. [DOI] [PubMed] [Google Scholar]
- 47.Maragkoudakis P.A., Chingwaru W., Gradisnik L., Tsakalidou E., Cencic A. Lactic acid bacteria efficiently protect human and animal intestinal epithelial and immune cells from enteric virus infection. Int J Food Microbiol. 2010;141:S91–S97. doi: 10.1016/j.ijfoodmicro.2009.12.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kang W., Pan L., Peng C., Dong L., Cao S., Cheng H., et al. Isolation and characterization of lactic acid bacteria from human milk. J Dairy Sci. 2020;103(11):9980–9991. doi: 10.3168/jds.2020-18704. [DOI] [PubMed] [Google Scholar]
- 49.Chiba E., Villena J., Hosoya S., Takanashi N., Shimazu T., Aso H., et al. A newly established bovine intestinal epithelial cell line is effective for in vitro screening of potential antiviral immunobiotic microorganisms for cattle. Res Vet Sci. 2012;93(2):688–694. doi: 10.1016/j.rvsc.2011.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lee J.-E., Lee N.-K., Paik H.-D. Antimicrobial and anti-biofilm effects of probiotic Lactobacillus plantarum KU200656 isolated from kimchi. Food Sci Biotechnol. 2021;30(1):97–106. doi: 10.1007/s10068-020-00837-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kim K., Lee G., Thanh H.D., Kim J.-H., Konkit M., Yoon S., et al. Exopolysaccharide from Lactobacillus plantarum LRCC5310 offers protection against rotavirus-induced diarrhea and regulates inflammatory response. J Dairy Sci. 2018;101(7):5702–5712. doi: 10.3168/jds.2017-14151. [DOI] [PubMed] [Google Scholar]
- 52.Yi D.Y., Jo S., Lee Y.M., Kim J.-H., Kim W., ri Park M., et al. Effect of a new Lactobacillus plantarum product, LRCC5310, on clinical symptoms and virus reduction in children with rotaviral enteritis. Medicine. 2020;99(38) doi: 10.1097/MD.0000000000022192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Malagón-Rojas J.N., Mantziari A., Salminen S., Szajewska H. Postbiotics for preventing and treating common infectious diseases in children: a systematic review. Nutrients. 2020;12(2):389. doi: 10.3390/nu12020389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yan S., Yang B., Ross R.P., Stanton C., Zhang H., Zhao J., et al. Bifidobacterium longum subsp. longum YS108R fermented milk alleviates DSS induced colitis via anti-inflammation, mucosal barrier maintenance and gut microbiota modulation. J Functional Foods. 2020;73:104153. [Google Scholar]
- 55.Utz V.M., Visñuk D.P., Perdigón G., de LeBlanc AdM. Milk fermented by Lactobacillus casei CRL431 administered as an immune adjuvant in models of breast cancer and metastasis under chemotherapy. Appl Microbiol Biotechnol. 2021;105(1):327–340. doi: 10.1007/s00253-020-11007-x. [DOI] [PubMed] [Google Scholar]
- 56.Wang J., Wu T., Fang X., Min W., Yang Z. Characterization and immunomodulatory activity of an exopolysaccharide produced by Lactobacillus plantarum JLK0142 isolated from fermented dairy tofu. Int J Biol Macromol. 2018;115:985–993. doi: 10.1016/j.ijbiomac.2018.04.099. [DOI] [PubMed] [Google Scholar]
- 57.Maruo T., Gotoh Y., Nishimura H., Ohashi S., Toda T., Takahashi K. Oral administration of milk fermented with Lactococcus lactis subsp. cremoris FC protects mice against influenza virus infection. Lett Appl Microbiol. 2012;55(2):135–140. doi: 10.1111/j.1472-765X.2012.03270.x. [DOI] [PubMed] [Google Scholar]
- 58.Rigo-Adrover MdM., Knipping K., Garssen J., van Limpt K., Knol J., Franch À., et al. Prevention of rotavirus diarrhea in suckling rats by a specific fermented milk concentrate with prebiotic mixture. Nutrients. 2019;11(1):189. doi: 10.3390/nu11010189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Matsuzaki T., Saito M., Usuku K., Nose H., Izumo S., Arimura K., et al. A prospective uncontrolled trial of fermented milk drink containing viable Lactobacillus casei strain Shirota in the treatment of HTLV-1 associated myelopathy/tropical spastic paraparesis. J Neurol Sci. 2005;237(1–2):75–81. doi: 10.1016/j.jns.2005.05.011. [DOI] [PubMed] [Google Scholar]
- 60.Shida K., Sato T., Iizuka R., Hoshi R., Watanabe O., Igarashi T., et al. Daily intake of fermented milk with Lactobacillus casei strain Shirota reduces the incidence and duration of upper respiratory tract infections in healthy middle-aged office workers. Eur J Nutr. 2017;56(1):45–53. doi: 10.1007/s00394-015-1056-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kinoshita T., Maruyama K., Suyama K., Nishijima M., Akamatsu K., Jogamoto A., et al. The effects of OLL1073R-1 yogurt intake on influenza incidence and immunological markers among women healthcare workers: a randomized controlled trial. Food & Function. 2019;10(12):8129–8136. doi: 10.1039/c9fo02128k. [DOI] [PubMed] [Google Scholar]
- 62.Makino S., Ikegami S., Kume A., Horiuchi H., Sasaki H., Orii N. Reducing the risk of infection in the elderly by dietary intake of yoghurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1. Br J Nutr. 2010;104(7):998–1006. doi: 10.1017/S000711451000173X. [DOI] [PubMed] [Google Scholar]
- 63.Hishiki H., Kawashima T., Tsuji N.M., Ikari N., Takemura R., Kido H., et al. A double-blind, randomized, placebo-controlled trial of heat-killed Pediococcus acidilactici K15 for prevention of respiratory tract infections among preschool children. Nutrients. 2020;12(7):1989. doi: 10.3390/nu12071989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Bohlouli J., Moravejolahkami A.R., Ganjali Dashti M., Balouch Zehi Z., Hojjati Kermani M.A., Borzoo-Isfahani M., et al. COVID-19 and fast foods consumption: a review. Int J Food Prop. 2021;24(1):203–209. [Google Scholar]
- 65.Aktaş B., Aslim B. Gut-lung axis and dysbiosis in COVID-19. Turkish J Biol. 2020;44(SI-1):265–272. doi: 10.3906/biy-2005-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bahreini-Esfahani N, Moravejolahkami AR. Can synbiotic dietary pattern predict lactobacillales strains in breast milk? Breastfeed Med. 2020;15(6):387–393. doi: 10.1089/bfm.2019.0301. [DOI] [PubMed] [Google Scholar]
- 67.d'Ettorre G., Ceccarelli G., Marazzato M., Campagna G., Pinacchio C., Alessandri F., et al. Challenges in the management of SARS-CoV2 infection: the role of oral bacteriotherapy as complementary therapeutic strategy to avoid the progression of COVID-19. Front Med. 2020;7:389. doi: 10.3389/fmed.2020.00389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gill H.S., Rutherfurd K.J., Cross M.L., Gopal P.K. Enhancement of immunity in the elderly by dietary supplementation with the probiotic Bifidobacterium lactis HN019. Am J Clin Nutr. 2001;74(6):833–839. doi: 10.1093/ajcn/74.6.833. [DOI] [PubMed] [Google Scholar]
- 69.Kassir R. Risk of COVID-19 for patients with obesity. Obes Rev. 2020;21(6) doi: 10.1111/obr.13034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Misumi I., Starmer J., Uchimura T., Beck M.A., Magnuson T., Whitmire J.K. Obesity expands a distinct population of T cells in adipose tissue and increases vulnerability to infection. Cell Rep. 2019;27(2):514–524. e5. doi: 10.1016/j.celrep.2019.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kageyama Y., Akiyama T., Nakamura T. Intestinal dysbiosis and probiotics in COVID-19. J Clin Trials. 2020;10(421) 2167-0870.20. [Google Scholar]
- 72.Sen T., Cawthon C.R., Ihde B.T., Hajnal A., DiLorenzo P.M., Claire B., et al. Diet-driven microbiota dysbiosis is associated with vagal remodeling and obesity. Physiol Behav. 2017;173:305–317. doi: 10.1016/j.physbeh.2017.02.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Abenavoli L., Scarpellini E., Colica C., Boccuto L., Salehi B., Sharifi-Rad J., et al. Gut microbiota and obesity: a role for probiotics. Nutrients. 2019;11(11):2690. doi: 10.3390/nu11112690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Siddell S., Anderson R., Cavanagh D., Fujiwara K., Klenk H., Macnaughton M., et al. Coronaviridae Intervirology. 1983;20(4):181–189. doi: 10.1159/000149390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bozkurt H.S. 2020. Probiotic bacteria against the COVID-19. [Google Scholar]
- 76.Schiavi E., Gleinser M., Molloy E., Groeger D., Frei R., Ferstl R., et al. The surface-associated exopolysaccharide of Bifidobacterium longum 35624 plays an essential role in dampening host proinflammatory responses and repressing local TH17 responses. Appl Environ Microbiol. 2016;82(24):7185–7196. doi: 10.1128/AEM.02238-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Bozkurt K., Denktaş C., Özdemir O., Altındal A., Avdan Z., Bozkurt H. 2019. Charge Transport in Bifidobacterium animalis subsp. lactis BB-12 under the various Atmosphere. arXiv preprint arXiv:190110765. [Google Scholar]
- 78.Angeletti S., Benvenuto D., Bianchi M., Giovanetti M., Pascarella S., Ciccozzi M. COVID-2019: the role of the nsp 2 and nsp 3 in its pathogenesis. J Med Virol. 2020;92(6):584–588. doi: 10.1002/jmv.25719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Coomes E.A., Haghbayan H. Interleukin-6 in COVID-19: a systematic review and meta-analysis. Rev Med Virol. 2020;30(6):1–9. doi: 10.1002/rmv.2141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Askari G., Ghavami A., Shahdadian F., Moravejolahkami A.R. Effect of synbiotics and probiotics supplementation on autoimmune diseases: a systematic review and meta-analysis of clinical trials. Clin Nutr. 2021;40(5):3221–3234. doi: 10.1016/j.clnu.2021.02.015. [DOI] [PubMed] [Google Scholar]
- 81.Trinchieri G., Santoli D. Anti-viral activity induced by culturing lymphocytes with tumor-derived or virus-transformed cells. Enhancement of human natural killer cell activity by interferon and antagonistic inhibition of susceptibility of target cells to lysis. J Exp Med. 1978;147(5):1314–1333. doi: 10.1084/jem.147.5.1314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Siegal F.P., Kadowaki N., Shodell M., Fitzgerald-Bocarsly P.A., Shah K., Ho S., et al. The nature of the principal type 1 interferon-producing cells in human blood. Science. 1999;284(5421):1835–1837. doi: 10.1126/science.284.5421.1835. [DOI] [PubMed] [Google Scholar]
- 83.Tezuka H., Abe Y., Asano J., Sato T., Liu J., Iwata M., et al. Prominent role for plasmacytoid dendritic cells in mucosal T cell-independent IgA induction. Immunity. 2011;34(2):247–257. doi: 10.1016/j.immuni.2011.02.002. [DOI] [PubMed] [Google Scholar]
- 84.Theofilopoulos A.N., Baccala R., Beutler B., Kono D.H. Type I interferons (α/β) in immunity and autoimmunity. Annu Rev Immunol. 2005;23:307–335. doi: 10.1146/annurev.immunol.23.021704.115843. [DOI] [PubMed] [Google Scholar]
- 85.Gilliet M., Cao W., Liu Y.-J. Plasmacytoid dendritic cells: sensing nucleic acids in viral infection and autoimmune diseases. Nat Rev Immunol. 2008;8(8):594–606. doi: 10.1038/nri2358. [DOI] [PubMed] [Google Scholar]
- 86.Lund J.M., Alexopoulou L., Sato A., Karow M., Adams N.C., Gale N.W., et al. Recognition of single-stranded RNA viruses by Toll-like receptor 7. Proc Natl Acad Sci Unit States Am. 2004;101(15):5598–5603. doi: 10.1073/pnas.0400937101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lee H.K., Lund J.M., Ramanathan B., Mizushima N., Iwasaki A. Autophagy-dependent viral recognition by plasmacytoid dendritic cells. Science. 2007;315(5817):1398–1401. doi: 10.1126/science.1136880. [DOI] [PubMed] [Google Scholar]
- 88.Bauer S., Kirschning C.J., Häcker H., Redecke V., Hausmann S., Akira S., et al. Human TLR9 confers responsiveness to bacterial DNA via species-specific CpG motif recognition. Proc Natl Acad Sci Unit States Am. 2001;98(16):9237–9242. doi: 10.1073/pnas.161293498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Latz E., Verma A., Visintin A., Gong M., Sirois C.M., Klein D.C., et al. Ligand-induced conformational changes allosterically activate Toll-like receptor 9. Nat Immunol. 2007;8(7):772–779. doi: 10.1038/ni1479. [DOI] [PubMed] [Google Scholar]
- 90.Kawai T., Sato S., Ishii K.J., Coban C., Hemmi H., Yamamoto M., et al. Interferon-α induction through Toll-like receptors involves a direct interaction of IRF7 with MyD88 and TRAF6. Nat Immunol. 2004;5(10):1061–1068. doi: 10.1038/ni1118. [DOI] [PubMed] [Google Scholar]
- 91.Shibata T., Kanayama M., Haida M., Fujimoto S., Oroguchi T., Sata K., et al. Lactococcus lactis JCM5805 activates anti-viral immunity and reduces symptoms of common cold and influenza in healthy adults in a randomized controlled trial. J Functional Foods. 2016;24:492–500. [Google Scholar]
- 92.Jounai K., Sugimura T., Ohshio K., Fujiwara D. Oral administration of Lactococcus lactis subsp. lactis JCM5805 enhances lung immune response resulting in protection from murine parainfluenza virus infection. PLoS One. 2015;10(3) doi: 10.1371/journal.pone.0119055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Fujii T., Jounai K., Horie A., Takahashi H., Suzuki H., Ohshio K., et al. Effects of heat-killed Lactococcus lactis subsp. lactis JCM 5805 on mucosal and systemic immune parameters, and antiviral reactions to influenza virus in healthy adults; a randomized controlled double-blind study. J Functional Foods. 2017;35:513–521. [Google Scholar]
- 94.Liang W., Feng Z., Rao S., Xiao C., Xue X., Lin Z., et al. Diarrhoea may be underestimated: a missing link in 2019 novel coronavirus. Gut. 2020;69(6):1141–1143. doi: 10.1136/gutjnl-2020-320832. [DOI] [PubMed] [Google Scholar]
- 95.Feng Z., Wang Y., Qi W. The small intestine, an underestimated site of SARS-CoV-2 infection: from red queen effect to probiotics. Preprints. 2020 [Google Scholar]
- 96.Pradhan B., Guha D., Naik A.K., Banerjee A., Tambat S., Chawla S., et al. Probiotics L. acidophilus and B. clausii modulate gut microbiota in Th1-and Th2-biased mice to ameliorate salmonella typhimurium-induced diarrhea. Probiotics and Antimicrobial Proteins. 2019;11(3):887–904. doi: 10.1007/s12602-018-9436-5. [DOI] [PubMed] [Google Scholar]
- 97.Askari G., Moravejolahkami A.R. Synbiotic supplementation may relieve anterior uveitis, an ocular manifestation in behcet's syndrome. American J Case Reports. 2019;20:548. doi: 10.12659/AJCR.912023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Moravejolahkami A., Chitsaz A. Mediterranean-style diet Co-supplemented with synbiotics improved quality of life, fatigue and disease activity in five secondary progressive multiple sclerosis patients. Ann Med & Surg Case Rep: AMSCR. 2019;2019(2) [Google Scholar]
- 99.Moravejolahkami A.R., Paknahad Z., Chitsaz A. Dietary intake of energy and fiber in MS patients; an approach to prebiotics role. Nutr Food Sci. 2019;49(6):1039–1050. [Google Scholar]