Abstract
Background:
At-risk alcohol use is a common and costly form of substance misuse and is highly prevalent among people living with HIV (PLWH). The goal of the current analysis was to test the hypothesis that PLWH with at-risk alcohol use would be more likely to meet the clinical criteria for prediabetes/diabetes than PLWH with low-risk alcohol use.
Methods:
A cross-sectional analysis was performed on measures of alcohol and glycemic control in adult PLWH (n=105) enrolled in a prospective, interventional study investigating the effects of aerobic exercise on metabolic dysregulation in PLWH with at-risk alcohol use; the ALIVE-Ex Study (NCT03299205). The Alcohol Use Disorder Identification Test (AUDIT), Timeline Followback (TLFB), and phosphatidylethanol (PEth) determined alcohol use. Participants were stratified into low-risk (AUDIT < 5) and at-risk alcohol use (AUDIT ≥ 5). All participants underwent an oral glucose tolerance test (OGTT), and measures of glycemic control, HOMA-IR, and Matsuda Index were correlated with alcohol measures and compared by AUDIT score using mixed effects linear and logistic regression models, adjusting for age, sex, race, body mass index (BMI), and viral load.
Results:
In response to the glucose challenge, participants with at-risk alcohol use (n=46) had higher glucose levels and were five times more likely to meet criteria for prediabetes/diabetes (OR: 5.3, (1.8, 15.9)). 2-hour glucose values were positively associated with AUDIT score and PEth and a higher percentage of PLWH with at-risk alcohol use had glucose values ≥ 140mg/dL compared to those with low-risk alcohol use (34.8% vs. 10.2%, respectively).
Conclusion:
In this cohort of PLWH, at-risk alcohol use increased the likelihood for meeting the clinical criteria for prediabetes/diabetes (2-hour glucose ≥ 140mg/dL). Established determinants of metabolic dysfunction (e.g., BMI, waist-hip ratio) were not associated with increased alcohol use and dysglycemia, suggesting that other mechanisms may contribute to impaired glycemic control observed in this cohort.
Keywords: At-risk alcohol use, dysglycemia, glycemic control, HIV, OGTT
Introduction
At-risk alcohol use is a common and costly form of substance misuse in the United States (U.S.) and is highly prevalent among people living with HIV (PLWH) (Azar et al., 2010, Galvan et al., 2002, Lazo et al., 2007, Samet et al., 2004). Studies indicate that mortality rates associated with HIV infection have decreased with the use of antiretroviral therapy (ART) and HIV has emerged as a chronic disease (Kohli et al., 2006, Bhatia et al., 2012, Welsh et al., 2019). Enhanced survival rates with ART and subsequent aging, along with lifestyle choices (e.g. at-risk alcohol use) and living in an obesogenic environment, may increase the risk of metabolic comorbidities among PLWH (Bhatia et al., 2012, Bray and Popkin, 1999, Bray, 2004, Burgess et al., 2015, Carr, 2003, Falutz, 2011, Kohli et al., 2006, Martin et al., 2004, Worm et al., 2010, Wand et al., 2007, Willig and Overton, 2016). The development of insulin resistance (IR), which increases the risk for type 2 diabetes (T2D), is one of the most prevalent metabolic comorbidities in PLWH (Brener et al., 2016, Gutierrez and Balasubramanyam, 2012, Hadigan et al., 2000, Zigman et al., 2005, Lombo et al., 2015, Muyanja et al., 2016, Willig and Overton, 2016). The rate of diagnosed T2D in the U.S. population is approximately 10.2%, whereas PLWH have a T2D diagnosis rate as high as 14% (Brown et al., 2005, Hernandez-Romieu et al., 2017, Monroe et al., 2015, Prevention, 2020, Willig and Overton, 2016). Determining factors that contribute to higher rates of T2D in PLWH is imperative for improving their health, longevity, and quality of life.
Numerous studies, in preclinical and clinical models, suggest that alcohol use is a risk factor for the development of IR (Andersen et al., 1983, Kim et al., 2014, Lang et al., 2014, Nguyen et al., 2012, Shelmet et al., 1988, Wan et al., 2005). Surprisingly, even though PLWH consume higher rates of alcohol than individuals without HIV, studies investigating the effects of alcohol consumption on the development of IR and T2D in PLWH are limited. Preclinical research from our laboratory investigated the effects of alcohol on metabolic comorbidities in a simian model of HIV. Our results demonstrated that chronic binge alcohol (CBA) administration in simian immunodeficiency virus (SIV)-infected macaques leads to alterations in metabolic homeostasis (Ford et al., 2016). CBA/SIV animals had a decreased disposition index (indicator of IR), markedly reduced insulin release in response to a glucose load, and decreased insulin sensitivity, irrespective of ART. These metabolic alterations were not associated with fasting hyperglycemia, suggesting they likely preceded a phase of overt glucose intolerance developing with disease progression (Ford et al., 2016) and were associated with adverse metabolic effects in an organ-specific manner (Ford et al., 2018). These studies have led us to investigate the role of alcohol use on the development of metabolic comorbidities in PLWH.
Recently, we reported the prevalence of alcohol use and metabolic comorbidities in a cohort of PLWH from the Greater New Orleans Area (New Orleans Alcohol Use in HIV (NOAH) Study) (Welsh et al., 2019). In this cohort, 57% met criteria for at-risk alcohol use, 28.5% had a Body Mass Index (BMI) over 30, which is indicative of obesity, 36.4% met criteria for metabolic syndrome, 50.2% had IR (Homeostatic Model Assessment of Insulin Resistance (HOMA-IR) > 1.9) and 14.5% had received a T2D diagnosis (Simon et al., 2020). These results support a role for at-risk alcohol use in the development of IR and T2D in PLWH and provide the basis for the current study. The Alcohol & Metabolic Comorbidities in PLWH: Evidence Driven Interventions Study (ALIVE-Ex Study, NCT03299205), is a longitudinal prospective, interventional study investigating the effects of aerobic exercise in PLWH with at-risk alcohol use. Here, we present pre-intervention, cross-sectional analyses testing the hypothesis that based on OGTT, PLWH with at-risk alcohol use will be more likely to meet the clinical criteria for prediabetes/diabetes than PLWH with low-risk alcohol use.
Materials and Methods:
Study Design and Participants
In-care PLWH (aged ≥ 18 years) were recruited from the Comprehensive Alcohol HIV/AIDS Research Center (CARC) NOAH Study (Welsh et al., 2019) and from the Greater New Orleans, Louisiana metropolitan area. Participants meeting inclusion criteria were enrolled in this first phase of the ALIVE-Ex Study (n=105). Inclusion criteria for this study required a fasting blood glucose of 95–124 mg/dL (high normal to impaired), measured by a single-use auto-disabling finger stick device and glucose meter (True Metrix® Pro Blood Glucose Monitoring System, McKesson), at the Screening Visit. Exclusion criteria included a diagnosis of diabetes mellitus, pregnancy, acute illness, new antibiotic use or an unscheduled healthcare visit for illness, acute alcohol use (positive breathalyzer test at time of study), or impaired decision-making ability. Informed consent was obtained from all study participants. All procedures were reviewed and approved by the Louisiana State University Health Sciences Center Institutional Review Board (IRB#736) and were in accordance with the Declaration of Helsinki (World Medical, 2013).
ALIVE-Ex Study participants recruited from the NOAH cohort (n=72) are currently adhering to an ART regimen (~98%) of primarily integrase inhibitors (~63%), report living with HIV for 21.0 ± 8.6 (mean ± SD) years and have a low incidence of Hepatitis C (~15%). As part of the ALIVE-Ex Study, all participants self-reported demographic information on biological sex (men or women), race (African American, Caucasian American, or other), age, marital status (married/living with partner or widowed/divorced/separated), housing (single family dwelling, HIV-specific group facility, or group/nursing home) and educational attainment (less than high school, high school diploma or GED, at least some college/trade school, or college graduate). Body mass index (BMI) was classified as: <18.5 as underweight, 18.5 to <25.0 as normal, 25.0 to <30.0 as overweight and ≥ 30.0 as obese. Smoking status was categorized as non-smoker, former smoker, or current smoker. Viral load was considered suppressed if < 200 copies/mL.
Oral Glucose Tolerance Test
Eligible participants underwent an oral glucose tolerance test (OGTT) to measure their response to a glucose challenge. Participants were required to fast overnight and height, weight, blood pressure, and waist and hip circumference were measured. Following baseline (fasted) blood collection, participants were instructed to consume a calibrated dose of glucose (75g Trutol®, Thermo Fisher Scientific) within 5 minutes. At 1-hour and 2-hours, blood was collected, processed by the CARC Analytical Core, and cryopreserved for further analyses. During the OGTT, participants completed additional questionnaires to assess demographics and alcohol use.
Blood collected at baseline, 1-hour and 2-hours was separated into serum and plasma and dried blood spots were used for phosphatidylethanol (PEth) determination. Plasma glucose was measured using a glucose Analox analyzer (GM7 Analox microstat, Analox Instruments, USA). Plasma insulin and C-peptide were measured at each time point using ELISAs (Millipore Human Insulin ELISA, cat.no. EZHI-14K; Alpco C-Peptide ELISA, cat.no. 80-CPTHU-E10.1) according to the manufacturer’s instructions. HOMA-IR was calculated (fasting glucose (mg/dL) x fasting insulin (μU/mL) divided by 405). A HOMA-IR ≥ 1.9 was indicative of IR (Isokuortti et al., 2017, Jing et al., 2017). HOMA-β was calculated (360 × fasting insulin (μU/mL)/ fasting glucose (mg/dL) – 63). Matsuda Index was calculated (10,000/square root of [fasting glucose x fasting insulin] x [mean glucose x mean insulin during OGTT]). A Matsuda Index < 3.0 was suggestive of IR (Matsuda and DeFronzo, 1999, DeFronzo and Matsuda, 2010). High molecular weight adiponectin was measured in the baseline samples using an ELISA (Alpco HMW Adiponectin ELISA, cat.no. 80-ADPHU-E01) according to the manufacturer’s instructions.
Measurement of Alcohol Use
Risky alcohol use was assessed using the Alcohol Use Disorder Identification Test (AUDIT). The AUDIT is a 10-item tool developed by the World Health Organization with an overall score ranging from 0–40 (Saunders et al., 1993). Participants were stratified based on their AUDIT score, into a low-risk alcohol use group (defined as AUDIT < 5), and an at-risk alcohol use group (AUDIT ≥ 5). A Timeline Followback (TLFB) Calendar was used to assess quantity and frequency of alcohol use and drinking patterns over the previous 30 days. For TLFB, participants were interviewed with prompts to report days in which alcoholic beverages were consumed. Total grams of alcohol consumed were tabulated by calculating the total standard alcohol drinks consumed by day and type (beer, wine, or liquor), which was then used to calculate total grams (g) of alcohol consumed each day; 14g of alcohol was equated to one standard drink. More than 7 drinks per week or more than 3 drinks per occasion for women, and more than 14 drinks per week or more than 4 drinks per occasion for men was considered hazardous alcohol use. Whole blood PEth, a highly reliable blood test for alcohol use, was used as a biomarker to assess alcohol use over the previous 3–4 weeks (Ulwelling and Smith, 2018). Dried blood spot samples were analyzed by the United States Drug Testing Laboratories, Inc. (Des Plains, IL). A measure of ≥ 8 ng/mL was considered a positive test and a measure of > 250 ng/mL was categorized as alcohol misuse.
Statistical Analysis
Descriptive statistics were used to characterize the population’s sociodemographic characteristics and alcohol use. Chi-square tests were conducted to compare demographic characteristics and at-risk alcohol use by AUDIT ≥ 5. Wilcoxon ranked sum tests were used due to the lack of normality to compare the means of the metabolic measures by at-risk alcohol use. Metabolic measures of glucose and insulin levels during the OGTT were compared by AUDIT using mixed effects to account for the repeated measures (repeated measures ANOVA). Spearman correlations were estimated for BMI, waist/hip ratio (WHR), adiponectin, glucose and insulin (baseline, 2-hours, area under the curve (AUC)), C-peptide (baseline, 2-hours), HOMA-IR, HOMA-β, and Matsuda Index with measures of alcohol use, including AUDIT, PEth and TLFB. Mixed effects linear regression was used to determine associations between measures of alcohol use and outcome measures of glycemic control. Logistic regression was used to model dichotomous outcomes of dysglycemia and alcohol use to determine the odds of meeting clinical criteria for prediabetes/diabetes. Linear and logistic regression models were adjusted by age, sex, race, BMI, and viral load. All analyses were completed using SAS version 9.4 (SAS Institute, Cary, NC). Statistical significance was set at p<.05.
Results
Participants
A total of 160 individuals were screened, 55 were excluded based on inclusion/exclusion criteria, and 105 were included in the current analyses. Demographics of participants in the low-risk (AUDIT < 5) and at-risk (AUDIT ≥ 5) alcohol use groups are shown in Table 1. The average age was 52.1 ± 9.9 (mean ± SD) years and the majority were male (69.5%) and African American (76%). The majority were never married (55.2%), 90.5% had an annual income of < $20,000, 38.1% had less than a high school education, 24.8% had a BMI ≥ 30, and 56.3% were current smokers. Overall, 97.1% had suppressed HIV viral loads. Most ALIVE-Ex participants reported recent alcohol consumption (57.9% consumed alcohol within the previous 30 days), and 43.8% had an AUDIT score ≥ 5, which was considered at-risk alcohol use in this study. A positive PEth was present in 55.8% of participants and 14.3% had a PEth ≥ 250ng/dL. PLWH with low-risk alcohol use had a higher education level than those with at-risk alcohol use (p=.03). Participants with at-risk alcohol use consumed more alcohol, as measured by TLFB (g), (p<.001), and had a higher incidence of a positive PEth test (p<.001) and PEth>250 (p=.002) (Table 1).
Table 1.
Demographic parameters and participant distribution according to alcohol use disorders identification test (AUDIT), ALIVE-Ex Study (2018–2020).
| AUDIT | ||||
|---|---|---|---|---|
| All (n = 105) | < 5 (n = 59) | ≥ 5 (n = 46) | ||
| % (n) | % (n) | % (n) | p-value | |
|
| ||||
| Sex | 0.197 | |||
| Female | 30.5 (32) | 35.6 (21) | 23.9 (11) | |
| Male | 69.5 (73) | 64.4 (38) | 76.1 (35) | |
| Race | 0.554 | |||
| African American | 76.0 (79) | 74.6 (44) | 77.8 (35) | |
| White | 22.1 (23) | 22.0 (13) | 22.2 (10) | |
| Other | 1.9 (2) | 3.4 (2) | 0 | |
| Age, years | 0.250 | |||
| 20 to 29 | 1.9 (2) | 3.4 (2) | 0 | |
| 30 to 39 | 10.5 (11) | 15.2 (9) | 4.3 (2) | |
| 40 to 49 | 20 (21) | 20.3 (12) | 19.6 (9) | |
| 50 to 59 | 41.9 (44) | 35.6 (21) | 50 (23) | |
| 60+ | 25.7 (27) | 25.4 (15) | 26.1 (12) | |
| Marital Status | 0.809 | |||
| Married/Living with partner | 17.1 (18) | 18.6 (11) | 15.2 (7) | |
| Widowed/Divorced/Separated | 27.6 (29) | 25.4 (15) | 30.4 (14) | |
| Never married | 55.2 (58) | 55.9 (33) | 54.3 (25) | |
| Income | 0.071 | |||
| <$20,000 | 90.5 (95) | 84.7 (50) | 97.8 (45) | |
| $20,000 to $39,999 | 4.8 (5) | 8.5 (5) | 0 | |
| $40,000+ | 4.7 (5) | 6.8 (4) | 2.2 (1) | |
| Education | 0.032* | |||
| < High School | 38.1 (40) | 35.6 (21) | 41.3 (19) | |
| High School Graduate | 27.6 (29) | 28.8 (17) | 26.1 (12) | |
| Some College, Junior/Community College, Vocational/Trade School | 25.7 (27) | 20.3 (12) | 32.6 (15) | |
| 4-year College/Graduate/Professional School | 8.6 (9) | 15.3 (9) | 0 | |
| BMI | 0.790 | |||
| <18.5 | 2.9 (3) | 1.7 (1) | 4.3 (2) | |
| 18.5 to 24.9 | 36.2 (38) | 33.9 (20) | 39.1 (18) | |
| 25 to 29.9 | 36.2 (38) | 35.6 (21) | 37.0 (17) | |
| 30 to 34.9 | 10.5 (11) | 11.9 (7) | 8.7 (4) | |
| 35+ | 14.3 (15) | 16.9 (10) | 10.9 (5) | |
| Suppressed viral load (<200 copies/mL) | 97.1 (102) | 98.3 (58) | 95.7 (44) | 0.418 |
| Smoking status (n = 71) | ||||
| Nonsmoker | 23.9 (17) | 13.6 (8) | 19.6 (9) | 0.091 |
| Current smoker | 56.3 (40) | 30.5 (18) | 47.8 (22) | |
| Former smoker | 19.7 (14) | 18.6 (11) | 6.5 (3) | |
| Alcohol measures | ||||
| Positive Phosphatidylethanol (PEth) (≥ 8 ng/dL) | 55.8 (58) | 36.2 (21) | 80.4 (37) | <0.001* |
| Misuse PEth (≥250 ng/dL) | 14.3 (15) | 1.7 (1) | 30.4 (14) | 0.002* |
| 30-day TLFB (g) median (interquartile range) | 57.9 (392) | 11.2 (112) | 336.3 (883.7) | <0.001* |
Circulating Measures of Glycemic Control
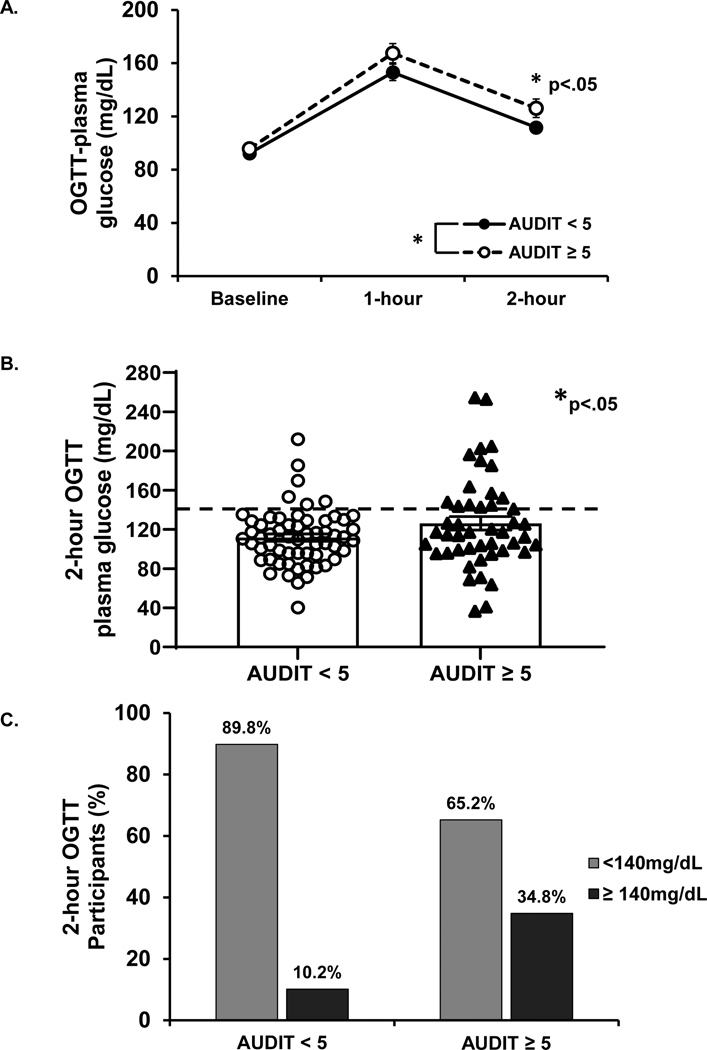
For OGTT, main effects of alcohol risk (F=4.19, p<.05) and OGTT time point (F=118.89, p<.0001) were detected for glucose values, suggesting that PLWH with at-risk alcohol use have an impaired response to a glucose challenge (Figure 1A). Further analysis revealed significantly higher 2-hour glucose levels in PLWH with at-risk alcohol use compared to those with low-risk alcohol use (p< .05; Figure 1B). In participants with low-risk alcohol use, 89.8% had a “normal” 2-hour glucose level (<140 mg/dL) and 10.2% had a glucose level that was ≥ 140mg/dL, which meets clinical criteria for prediabetes/diabetes. In PLWH with at-risk alcohol use, 65.2% exhibited a normal 2-hour glucose level and 34.8% had a glucose level ≥ 140mg/dL (Figure 1C). A main effect of OGTT time point on insulin (F=70.58, p<.0001) and C-peptide levels (F=246.39, p< .0001) w also detected, though insulin and C-peptide levels did not differ by alcohol risk. Area under the curve (AUC) for glucose and insulin did not differ between PLWH with low-risk and at-risk alcohol use. Adiponectin levels were measured at baseline and did not differ by alcohol risk. HOMA-β and HOMA-IR did not differ between groups, however 37% of PLWH with at-risk alcohol use had a HOMA-IR > 1.9, which is indicative of IR, compared to 29.3% among those with low-risk alcohol use (p>.05). The Matsuda Index did not differ by alcohol risk. The percentage of PLWH with a Matsuda Index < 3.0 (> 3.0 denotes insulin sensitivity) was similar between PLWH with low-risk and at-risk alcohol use (14.3 % vs. 11.9%, respectively).
Figure 1:
Assessment of circulating glucose values following an Oral Glucose Tolerance Test in the ALIVE-Ex Study (2018–2020). A. Glucose levels in the OGTT were higher in PLWH with at-risk alcohol use compared to PLWH with low-risk alcohol use. B. 2-hour glucose levels were significantly higher in PLWH with an AUDIT ≥ 5. C. The percentage of participants with 2-hour glucose levels ≥ 140mg/dL, which is indicative of prediabetes/diabetes. Data shown as mean ± SEM, *p<.05.
Association of Alcohol Use and Measures of Glycemic Control
Spearman correlations between alcohol and measures of glycemic control were performed (Table 2). AUDIT was positively correlated with glucose AUC (r= 0.204, p<.05), indicating higher blood glucose in response to the glucose challenge in PLWH with higher AUDIT scores. PEth and alcohol consumed (g), measured by TLFB, were negatively correlated with BMI (r= −0.199 and −0.203, respectively; p<.05), suggesting that higher recent alcohol intake was associated with a lower BMI. TLFB (g) was negatively correlated with baseline levels of C-peptide (r= −0.277, p<.05), suggesting that higher recent alcohol use was associated with lower circulating C-peptide levels. AUDIT, PEth and TLFB were not significantly correlated with HOMA-IR, HOMA-β or Matsuda Index.
Table 2.
Spearman Correlation of Glucose and Insulin Measures and Alcohol Measures in People Living with HIV (PLWH), ALIVE-Ex Study (2018–2020).
| Body Mass Index | Glucose | Insulin | C-Peptide | HOMA-IR | HOMA-β | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||||
| 0 | 2 | AUC | 0 | 2 | AUC | 0 | 2 | ||||
|
| |||||||||||
| AUDIT | −0.111 | 0.106 | 0.156 | 0.204 * | −0.089 | 0.048 | 0.009 | −0.101 | 0.027 | −0.071 | −0.167 |
| PEth | −0.199* | −0.057 | 0.115 | 0.176 | 0.072 | 0.021 | 0.082 | 0.113 | 0.069 | −0.076 | −0.055 |
| TLFB (g) | −0.203* | −0.054 | 0.072 | 0.054 | −0.092 | −0.039 | −0.017 | −0.277* | −0.099 | −0.096 | −0.116 |
p-value <0.05
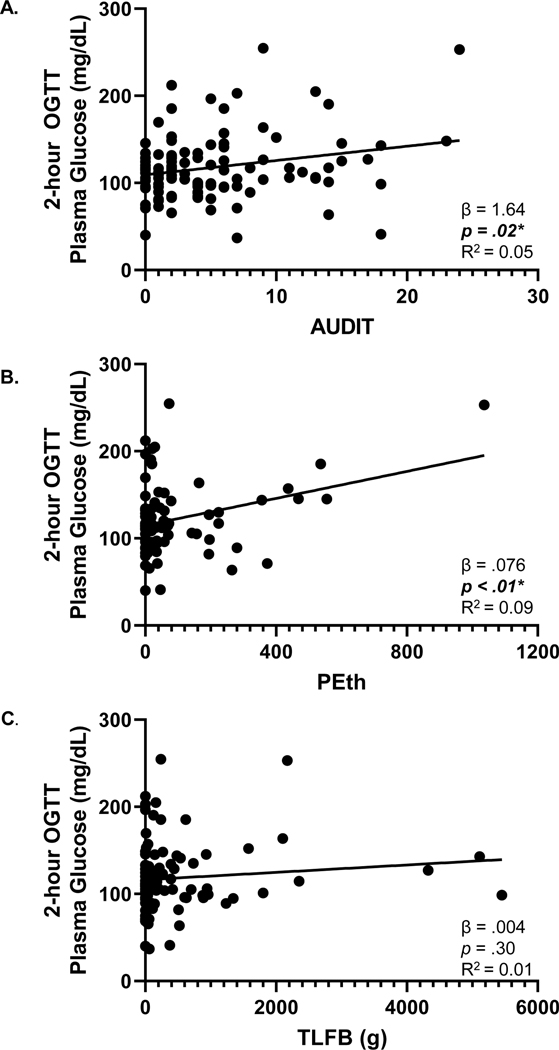
Linear regression adjusting for age, sex, race, BMI, and viral load were performed between outcome measures of alcohol and glycemic control (Table 3). 2-hour glucose levels were positively associated with AUDIT (p <.05; Figure 2A, unadjusted shown) and PEth values (p<.01, Figure 2B, unadjusted shown), though not associated with TLFB (g) (p>.05, Figure 2C, unadjusted shown). Glucose AUC was positively associated with AUDIT and PEth (p<.05), however, 2-hour insulin and C-peptide levels and HOMA-IR, HOMA-β and Matsuda Index were not associated with AUDIT, PEth or TLFB (g) (p>.05, Table 3).
Table 3.
Linear Regression Beta Coefficients (β) of Glucose and Insulin Measures and Alcohol Measures adjusted by age, sex, race, body mass index (BMI), and viral load in People Living with HIV (PLWH), ALIVE-Ex Study (2018–2020).
| Glucose | Insulin | C-Peptide | HOMA-IR | HOMA-β | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||
| 0 | 2 | AUC | 0 | 2 | AUC | 0 | 2 | |||
|
| ||||||||||
| AUDIT | 0.22 | 1.49 * | 2.34 * | −6.65e-2 | 0.72 | 0.98 | −1.85 | −0.75 | −5.27e-3 | −3.00 |
| PEth | 6.39e-3 | 6.68e-2 * | 0.13 * | −2.52e-3 | −2.76e-3 | 7.54e-2 | −6.19e-2 | 0.43 | −5.07e-4 | −6.12e-2 |
| TLFB (g) | −7.89e-4 | 4.19e-3 | 4.49e-3 | −2.46e-4 | 3.28e-3 | 1.99e-2 | −1.75e-2 | 2.03e-3 | −7.63e-5 | −1.75e-2 |
p-value < 0.05
Figure 2:
Scatter plots and unadjusted linear regression analyses of Alcohol Use Disorder Identification Test (AUDIT) scores and measures of glycemic control in PLWH in the ALIVE-Ex Study (2018–2020). A. Circulating glucose levels at 2-hours following oral glucose tolerance tests (OGTT) were positively associated with AUDIT score. B. Circulating 2-hour glucose levels were positively associated with PEth values. C. Circulating 2-hour glucose levels were not associated with TLFB (g). *p<.05, unadjusted.
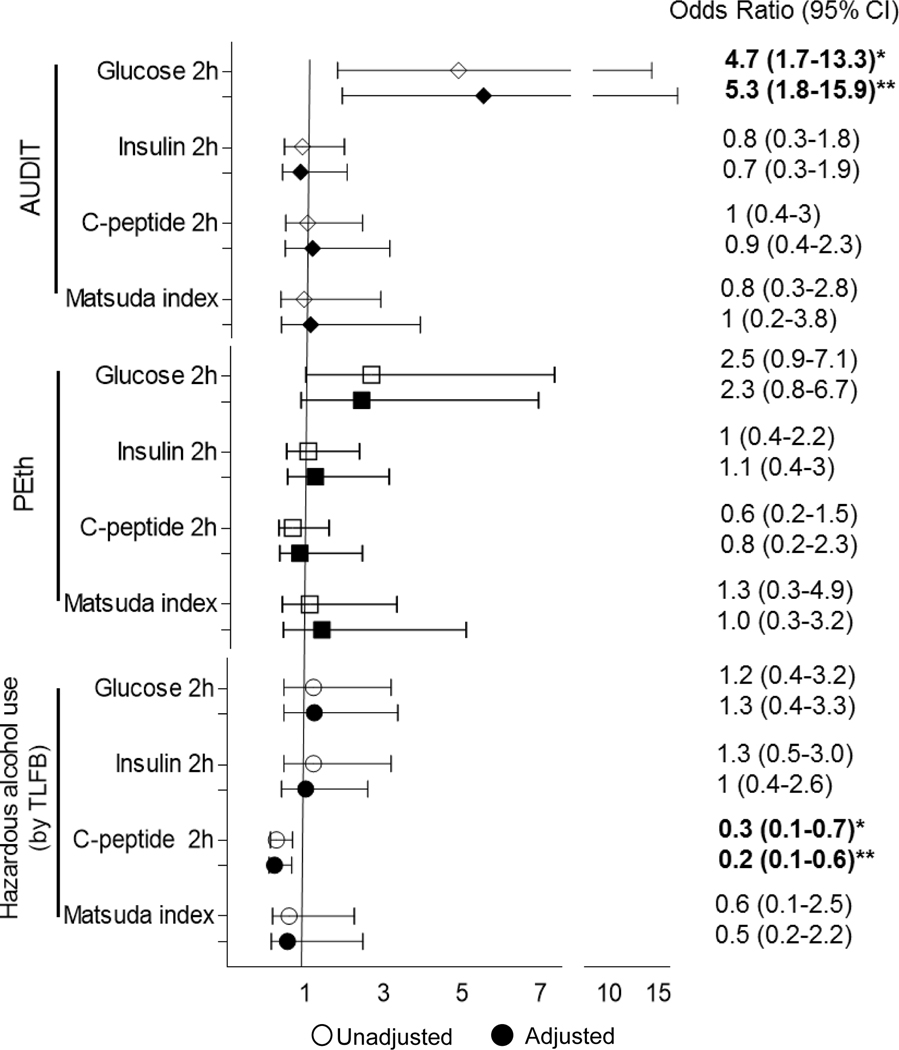
PLWH with at-risk alcohol use were five times more likely to meet criteria for prediabetes/diabetes (2-hour OGTT glucose ≥ 140mg/dL) when unadjusted or adjusted for age, sex, race, BMI, viral load (p<.05, adjusted Odds Ratio (OR) = 5.3; 95% confidence interval (1.8, 15.9); Figure 3). At-risk alcohol use did not affect the likelihood of elevated 2-hour insulin levels, impaired C-peptide levels, or insulin sensitivity (p>.05). Hazardous alcohol use was associated with impaired 2-hour C-peptide values, unadjusted or adjusted for age, sex, race, BMI and viral load (p<.05, adjusted OR = 0.2; (0.1, 0.6); Figure 3), but not an increased risk for meeting criteria for prediabetes/diabetes, elevated 2-hour insulin levels, or insulin sensitivity (p>.05). A positive PEth test was not associated with an increased risk for the assessed measures of glycemic control (p>.05).
Figure 3:
Odds ratios of glucose, insulin, C-peptide at 2-hours following Oral Glucose Tolerance Tests and Matsuda Index by alcohol measures in the ALIVE-Ex Study (2018–2020). Open symbols represent the unadjusted odds ratio and closed symbols represent odds ratio adjusted for age, sex, race, body mass index (BMI), and viral load. * p<.05, unadjusted, **p<.05 adjusted for age, sex, race, BMI, viral load.
Discussion
Increased life expectancy, ART use, living in an obesogenic environment, and at-risk alcohol use are factors which may contribute to the high rates of metabolic abnormalities in PLWH (Araujo et al., 2014, Calza et al., 2004, Kim et al., 2014, Lang et al., 2014, Nguyen et al., 2012, Rasouli and Kern, 2008, Samaras, 2009, Srinivasa and Grinspoon, 2014). We have recently shown that IR, metabolic syndrome, and T2D (50.2%, 36.4% and 14%, respectively) are prevalent in PLWH enrolled in the longitudinal, NOAH Study, in which 57% of enrolled subjects self-report at-risk alcohol use (Simon et al., 2020). For the current cross-sectional analyses, pre-exercise intervention data from the ALIVE-Ex Study were assessed to determine if at-risk alcohol use exacerbated the response to a glucose challenge in PLWH. Overall, our results indicate that in this cohort of PLWH, at-risk alcohol use increased the risk for dysglycemia and are 5 times more likely to meet clinical criteria for prediabetes/diabetes (2-hour glucose ≥ 140mg/dL) than PLWH with low-risk alcohol use.
Glucose levels, in response to a glucose challenge, were assessed at baseline (fasting), 1-hour and 2-hours following the challenge. Though fasting glucose did not differ between PLWH with low-risk and at-risk alcohol use, 2-hour glucose levels (≥ 140mg/dL), which are used clinically to diagnose prediabetes/diabetes, were higher in in this cohort of PLWH with at-risk alcohol use. In this cohort, 34.8% of PLWH with at-risk alcohol use had 2-hour glucose levels ≥ 140mg/dL, compared to only 10.2% of PLWH with low-risk alcohol use. Additionally, AUDIT score was positively associated with glucose AUC. Furthermore, when adjusted for BMI, age, sex, and race, 2-hour glucose levels and AUDIT were positively associated, suggesting that higher glucose levels in response to a glucose challenge are seen in PLWH with higher AUDIT scores. Furthermore, as mentioned, at-risk alcohol use was associated with a 5-fold increased risk for meeting criteria for prediabetes/diabetes.
Insulin levels were measured at baseline (fasting), 1-hour and 2-hours following the administration of glucose in the OGTT to determine the effects of at-risk alcohol use on the endocrine pancreas response following a glucose challenge. Insulin levels in this cohort were responsive to the glucose challenge and changed across the OGTT, while remaining within “normal” parameters. Fasting and 2-hour insulin levels following the glucose challenge did not differ between PLWH with low-risk and at-risk alcohol use and were not associated with AUDIT score, PEth, or TLFB. HOMA-IR, a measure of IR determined from fasting levels of insulin and glucose, HOMA-β, a measure of pancreatic β-cell function, and the Matsuda Index, a measure of whole-body insulin sensitivity derived from the OGTT, were determined to further assess the relationship between alcohol use and glycemic control. In this cohort, HOMA-IR, HOMA-β and Matsuda Index did not differ between PLWH with low-risk and at-risk alcohol use and were not associated with AUDIT score, PEth, or TLFB. We assessed the association between AUDIT-C, the first 3-questions of the AUDIT, and fasting insulin levels and HOMA-IR in the ALIVE-Ex Study cohort and did not find an association between AUDIT-C and the variables assessed. These results are somewhat discordant with the findings from our previous study using the NOAH cohort, which reported a negative association of AUDIT-C score with HOMA-IR and fasting insulin levels (Simon et al., 2020). We speculate that the inclusion criteria and the smaller number of participants for the ALIVE-Ex Study may contribute to these differential findings. For the ALIVE-Ex Study, eligibility criteria included a fasting glucose level of 95–124mg/dL. In contrast, in the NOAH Study, inclusion criteria was lack of a clinical diagnosis of diabetes, thereby increasing the range of fasting glucose and insulin values of enrolled participants.
Our previous preclinical studies in SIV-infected rhesus macaques demonstrated that CBA administration reduced plasma insulin and C-peptide values and the acute insulin response to glucose following a frequently sampled intravenous glucose tolerance test in asymptomatic ART-treated macaques (Ford et al., 2016). C-peptide, produced in the pancreas along with insulin, is an indicator of beta cell function and can be used to determine the ability of the pancreas to adequately produce insulin when stimulated (Breda and Cobelli, 2001, Cretti et al., 2001, Kjems et al., 2000, Mari et al., 2002, Tura et al., 2006). Clinical studies have shown that individuals with at-risk alcohol use and decreased exocrine pancreatic function had significantly lower C-peptide and insulin values following OGTT, compared to individuals with normal exocrine pancreatic function (Andersen et al., 1983). In the ALIVE-Ex cohort, fasting C-peptide levels were negatively correlated with recent alcohol consumption, as measured by the TLFB (g), indicating that higher levels of recent alcohol intake were associated with lower fasting C-peptide levels. This finding suggests an impairment of the endocrine pancreatic response that could indicate lower basal insulin production. Additionally, the odds of having 2-hour C-peptide levels > 200 pmol/L, indicative of a normal endocrine pancreatic response, was reduced in this cohort of PLWH with hazardous alcohol use, further suggesting impairment of the endocrine pancreatic response. Fasting, 1-hour and 2-hour C-peptide levels did not differ between PLWH with low-risk and at-risk alcohol indicating that recent alcohol use and not AUDIT, which assesses alcohol use over the past year, may be a better predictor of endocrine pancreas impairment. These finding underscore the importance of using a comprehensive battery of alcohol measures to better identify the contribution of alcohol to tissue injury.
BMI and adiposity are often used to determine rates of overweight and obesity and individuals who are considered overweight/obese are at a higher risk for the development of IR. In the current study, 61% of participants had a BMI ≥ 25 and met criteria for overweight/obese. BMI was negatively associated with PEth and TLFB, suggesting that in this cohort, lower BMIs were associated with higher recent alcohol use. Increased adiposity is also considered a risk factor for the development of metabolic dysfunctions, including IR. Abdominal or visceral obesity is associated with increased inflammation and increased risk for developing obesity-related comorbidities (Fox et al., 2012, Michaud et al., 2012, Neeland et al., 2012, Poirier et al., 2006, Poret et al., 2018). Our results indicate that waist/hip ratio (WHR), an indicator of abdominal obesity, was not associated with any of the measures of alcohol use assessed in this study. Additionally, an interaction between WHR and race was not detected in this cohort. Furthermore, levels of the adipokine, adiponectin (Rogers et al., 2008), did not differ in PLWH with low-risk and at-risk alcohol use and no statistically significant association between adiponectin levels and any of the other measures of alcohol use assessed were detected. It is important to note that the potential of confounding factors that regulate adiponectin expression, including body adiposity, sex, as well as alcohol type, amount, and timing of consumption, and sex and age differences in adipose accumulation and deposition need to be addressed in future assessments of this cohort.
Overall, data from this cohort of PLWH (n=105) with at-risk alcohol use suggest that these individuals are at an increased risk for meeting criteria for impaired glucose tolerance and this increased risk of dysglycemia may be due to factors distinct from established determinants of metabolic dysfunction (i.e. obesity and visceral adiposity) that have yet to be elucidated. There are several potential mechanisms that may be contributing to glucose dysregulation in this cohort, including skeletal muscle insulin resistance and impaired endocrine pancreas function. Studies to investigate the role of skeletal muscle on glucose regulation are the focus of current investigations in our ongoing preclinical studies and a goal of the ALIVE-Ex Study. Collectively our current findings that fasting C-peptide levels were negatively associated with TLFB; reports that hazardous alcohol use increased the odds of poor beta cell reserve (C-peptide < 200pmol/L) (Leighton et al., 2017); and with our previous findings that HOMA-β is negatively associated with at-risk alcohol use (Simon et al., 2020), suggest an impairment of endocrine pancreas function in this cohort of PLWH with at-risk alcohol use. Several limitations of the current study; including the relatively small sample size (n=105), higher percentage of PLWH with low-risk alcohol use (56.2%), compared to those with at-risk alcohol use (43.8%) and the low percentage of participants with glucose ≥ 140mg/dL following the OGTT (20.1%), may limit generalizability of the results to broader populations of PLWH. Nevertheless, our current findings highlight the fact that mechanisms for dysglycemia among PLWH remain poorly understood (Gutierrez and Balasubramanyam, 2012) and the need for future studies to elucidate the mechanistic relationship between alcohol use and dysglycemia in PLWH is evident. A further goal of the ALIVE-Ex Study is to evaluate the impact of an aerobic exercise intervention among this cohort of PLWH to improve glucose regulation. The continual development of intervention strategies to manage the dual burden of metabolic disease and HIV infection has the potential to improve quality of life and overall health in this population.
Acknowledgments
The authors would like to thank everyone who has contributed to the success of the ALIVE-Ex study: all study participants, the ALIVE-Ex Study clinical investigators and staff, research staff affiliated with the LSUHSC Comprehensive Alcohol-HIV/AIDS Research Center for their technical assistance and the research staff with the LSUHSC Clinical Translational Research Center. The ALIVE-Ex Study is funded by the National Institute on Alcohol Abuse and Alcoholism of the National Institutes of Health under award numbers UH2AA026198 and P60AA009803, to PEM.
This study was supported by the National Institute on Alcohol and Alcohol Abuse of the National Institutes of Health under award numbers UH2AA026198 and P60AA009803, to PEM.
Footnotes
The authors declare no conflicts of interest.
References
- Andersen BN, Hagen C, Faber OK, Lindholm J, Boisen P. & Worning H. 1983. Glucose tolerance and B cell function in chronic alcoholism: its relation to hepatic histology and exocrine pancreatic function. Metabolism, 32, 1029–32. [DOI] [PubMed] [Google Scholar]
- Araujo S, Bañón S, Machuca I, Moreno A, Pérez-Elías MJ & Casado JL 2014. Prevalence of insulin resistance and risk of diabetes mellitus in HIV-infected patients receiving current antiretroviral drugs. Eur J Endocrinol, 171, 545–54. [DOI] [PubMed] [Google Scholar]
- Azar MM, Springer SA, Meyer JP & Altice FL 2010. A systematic review of the impact of alcohol use disorders on HIV treatment outcomes, adherence to antiretroviral therapy and health care utilization. Drug Alcohol Depend, 112, 178–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhatia R, Ryscavage P. & Taiwo B. 2012. Accelerated aging and human immunodeficiency virus infection: emerging challenges of growing older in the era of successful antiretroviral therapy. J Neurovirol, 18, 247–55. [DOI] [PubMed] [Google Scholar]
- Bray GA 2004. Medical consequences of obesity. J Clin Endocrinol Metab, 89, 2583–9. [DOI] [PubMed] [Google Scholar]
- Bray GA & Popkin BM 1999. Dietary fat affects obesity rate. Am J Clin Nutr, 70, 572–573. [DOI] [PubMed] [Google Scholar]
- Breda E. & Cobelli C. 2001. Insulin secretion rate during glucose stimuli: alternative analyses of C-peptide data. Ann Biomed Eng, 29, 692–700. [DOI] [PubMed] [Google Scholar]
- Brener MI, Post WS, Haberlen SA, Zhang L, Palella FJ, Jacobson LP, Dobs AS, George RT, Witt MD, Budoff M, Kingsley LA & Brown TT 2016. Comparison of Insulin Resistance to Coronary Atherosclerosis in Human Immunodeficiency Virus Infected and Uninfected Men (from the Multicenter AIDS Cohort Study). Am J Cardiol, 117, 993–1000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown TT, Cole SR, Li X, Kingsley LA, Palella FJ, Riddler SA, Visscher BR, Margolick JB & Dobs AS 2005. Antiretroviral therapy and the prevalence and incidence of diabetes mellitus in the multicenter AIDS cohort study. Arch Intern Med, 165, 1179–84. [DOI] [PubMed] [Google Scholar]
- Burgess MJ, Zeuli JD & Kasten MJ 2015. Management of HIV/AIDS in older patients-drug/drug interactions and adherence to antiretroviral therapy. HIV AIDS (Auckl), 7, 251–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calza L, Manfredi R. & Chiodo F. 2004. Insulin Resistance and Diabetes Mellitus in HIV-Infected Patients Receiving Antiretroviral Therapy. Metab Syndr Relat Disord, 2, 241–50. [DOI] [PubMed] [Google Scholar]
- Carr MC 2003. The emergence of the metabolic syndrome with menopause. J Clin Endocrinol Metab, 88, 2404–11. [DOI] [PubMed] [Google Scholar]
- Cretti A, Lehtovirta M, Bonora E, Brunato B, Zenti MG, Tosi F, Caputo M, Caruso B, Groop LC, Muggeo M. & Bonadonna RC 2001. Assessment of beta-cell function during the oral glucose tolerance test by a minimal model of insulin secretion. Eur J Clin Invest, 31, 405–16. [DOI] [PubMed] [Google Scholar]
- Defronzo RA & Matsuda M. 2010. Reduced time points to calculate the composite index. Diabetes Care, 33, e93. [DOI] [PubMed] [Google Scholar]
- Falutz J. 2011. HIV infection, body composition changes and related metabolic complications: contributing factors and evolving management strategies. Curr Opin Clin Nutr Metab Care, 14, 255–60. [DOI] [PubMed] [Google Scholar]
- Ford SM Jr., Simon Peter L, Berner P, Cook G, Vande Stouwe C, Dufour J, Bagby G, Nelson S. & Molina PE 2018. Differential contribution of chronic binge alcohol and antiretroviral therapy to metabolic dysregulation in SIV-infected male macaques. Am J Physiol Endocrinol Metab, 315, E892–E903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ford SM, Simon L, Vande Stouwe C, Allerton T, Mercante DE, Byerley LO, Dufour JP, Bagby GJ, Nelson S. & Molina PE 2016. Chronic binge alcohol administration impairs glucose-insulin dynamics and decreases adiponectin in asymptomatic simian immunodeficiency virus-infected macaques. Am J Physiol Regul Integr Comp Physiol, 311, R888–R897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox CS, Liu Y, White CC, Feitosa M, Smith AV, Heard-Costa N, Lohman K, Johnson AD, Foster MC, Greenawalt DM, Griffin P, Ding J, Newman AB, Tylavsky F, Miljkovic I, Kritchevsky SB, Launer L, Garcia M, Eiriksdottir G, Carr JJ, Gudnason V, Harris TB, Cupples LA, Borecki IB, Consortium G, Consortium M. & Consortium G. 2012. Genome-wide association for abdominal subcutaneous and visceral adipose reveals a novel locus for visceral fat in women. PLoS Genet, 8, e1002695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galvan FH, Bing EG, Fleishman JA, London AS, Caetano R. & Burnam MA 2002. The prevalence of alcohol consumption and heavy drinking among people with HIV in the United States: Results form HIV Cost and Service Utilization Study. J Stud Alcohol, 63, 179–186. [DOI] [PubMed] [Google Scholar]
- Gutierrez AD & Balasubramanyam A. 2012. Dysregulation of glucose metabolism in HIV patients: epidemiology, mechanisms, and management. Endocrine, 41, 1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadigan C, Corcoran C, Stanley T, Piecuch S, Klibanski A. & Grinspoon S. 2000. Fasting hyperinsulinemia in human immunodeficiency virus-infected men: relationship to body composition, gonadal function, and protease inhibitor use. J Clin Endocrinol Metab, 85, 35–41. [DOI] [PubMed] [Google Scholar]
- Hernandez-Romieu AC, Garg S, Rosenberg ES, Thompson-Paul AM & Skarbinski J. 2017. Is diabetes prevalence higher among HIV-infected individuals compared with the general population? Evidence from MMP and NHANES 2009–2010. BMJ Open Diabetes Res Care, 5, e000304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isokuortti E, Zhou Y, Peltonen M, Bugianesi E, Clement K, Bonnefont-Rousselot D, Lacorte JM, Gastaldelli A, Schuppan D, Schattenberg JM, Hakkarainen A, Lundbom N, Jousilahti P, Männistö S, Keinänen-Kiukaanniemi S, Saltevo J, Anstee QM & Yki-Järvinen H. 2017. Use of HOMA-IR to diagnose non-alcoholic fatty liver disease: a population-based and inter-laboratory study. Diabetologia, 60, 1873–1882. [DOI] [PubMed] [Google Scholar]
- Jing J, Pan Y, Zhao X, Zheng H, Jia Q, Mi D, Chen W, Li H, Liu L, Wang C, He Y, Wang D, Wang Y. & Across-China IF 2017. Insulin Resistance and Prognosis of Nondiabetic Patients With Ischemic Stroke: The ACROSS-China Study (Abnormal Glucose Regulation in Patients With Acute Stroke Across China). Stroke, 48, 887–893. [DOI] [PubMed] [Google Scholar]
- Kim JY, Hwang JY, Lee DY, Song EH, Park KJ, Kim GH, Jeong EA, Lee YJ, Go MJ, Kim DJ, Lee SS, Kim BJ, Song J, Roh GS, Gao B. & Kim WH 2014. Chronic ethanol consumption inhibits glucokinase transcriptional activity by Atf3 and triggers metabolic syndrome in vivo. J Biol Chem, 289, 27065–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kjems LL, Christiansen E, Vølund A, Bergman RN & Madsbad S. 2000. Validation of methods for measurement of insulin secretion in humans in vivo. Diabetes, 49, 580–8. [DOI] [PubMed] [Google Scholar]
- Kohli R, Klein RS, Schoenbaum EE, Anastos K, Minkoff H. & Sacks HS 2006. Aging and HIV infection. J Urban Health, 83, 31–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lang CH, Derdak Z. & Wands JR 2014. Strain-dependent differences for suppression of insulin-stimulated glucose uptake in skeletal and cardiac muscle by ethanol. Alcohol Clin Exp Res, 38, 897–910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazo M, Gange SJ, Wilson TE, Anastos K, Ostrow DG, Witt MD & Jacobson LP 2007. Patterns and predictors of changes in adherence to highly active antiretroviral therapy: longitudinal study of men and women. Clin Infect Dis, 45, 1377–85. [DOI] [PubMed] [Google Scholar]
- Leighton E, Sainsbury CA & Jones GC 2017. A Practical Review of C-Peptide Testing in Diabetes. Diabetes Ther, 8, 475–487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lombo B, Alkhalil I, Golden MP, Fotjadhi I, Ravi S, Virata M, Lievano M, Diez J, Ghantous A. & Donohue T. 2015. Prevalence of Metabolic Syndrome in Patients with HIV in the Era of Highly Active Antiretroviral Therapy. Conn Med, 79, 277–81. [PubMed] [Google Scholar]
- Mari A, Schmitz O, Gastaldelli A, Oestergaard T, Nyholm B. & Ferrannini E. 2002. Meal and oral glucose tests for assessment of beta -cell function: modeling analysis in normal subjects. Am J Physiol Endocrinol Metab, 283, E1159–66. [DOI] [PubMed] [Google Scholar]
- Martin A, Smith D, Carr A, Hoy J, Chuah J, Mallal S, Law M, Clements M, Cooper DA & Group PS 2004. Progression of lipodystrophy (LD) with continued thymidine analogue usage: long-term follow-up from a randomized clinical trial (the PIILR study). HIV Clin Trials, 5, 192–200. [DOI] [PubMed] [Google Scholar]
- Matsuda M. & Defronzo RA 1999. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care, 22, 1462–70. [DOI] [PubMed] [Google Scholar]
- Michaud A, Drolet R, Noël S, Paris G. & Tchernof A. 2012. Visceral fat accumulation is an indicator of adipose tissue macrophage infiltration in women. Metabolism, 61, 689–98. [DOI] [PubMed] [Google Scholar]
- Monroe AK, Glesby MJ & Brown TT 2015. Diagnosing and managing diabetes in HIV-infected patients: current concepts. Clin Infect Dis, 60, 453–62. [DOI] [PubMed] [Google Scholar]
- Muyanja D, Muzoora C, Muyingo A, Muyindike W. & Siedner MJ 2016. High Prevalence of Metabolic Syndrome and Cardiovascular Disease Risk Among People with HIV on Stable ART in Southwestern Uganda. AIDS Patient Care STDS, 30, 4–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neeland IJ, Turer AT, Ayers CR, Powell-Wiley TM, Vega GL, Farzaneh-Far R, Grundy SM, Khera A, Mcguire DK & De Lemos JA 2012. Dysfunctional adiposity and the risk of prediabetes and type 2 diabetes in obese adults. JAMA, 308, 1150–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen VA, Le T, Tong M, Silbermann E, Gundogan F. & De La Monte SM 2012. Impaired insulin/IGF signaling in experimental alcohol-related myopathy. Nutrients, 4, 1058–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poirier P, Giles TD, Bray GA, Hong Y, Stern JS, Pi-Sunyer FX & Eckel RH 2006. Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss. Arterioscler Thromb Vasc Biol, 26, 968–76. [DOI] [PubMed] [Google Scholar]
- Poret JM, Souza-Smith F, Marcell SJ, Gaudet DA, Tzeng TH, Braymer HD, Harrison-Bernard LM & Primeaux SD 2018. High fat diet consumption differentially affects adipose tissue inflammation and adipocyte size in obesity-prone and obesity-resistant rats. Int J Obes (Lond), 42, 535–541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prevention CFDCA 2020. National Diabetes Statistics Report, 2020. In: Services, a. H. USDOH (ed.). [Google Scholar]
- Rasouli N. & Kern PA 2008. Adipocytokines and the metabolic complications of obesity. J Clin Endocrinol Metab, 93, S64–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers CQ, Ajmo JM & You M. 2008. Adiponectin and alcoholic fatty liver disease. IUBMB Life, 60, 790–7. [DOI] [PubMed] [Google Scholar]
- Samaras K. 2009. Prevalence and pathogenesis of diabetes mellitus in HIV-1 infection treated with combined antiretroviral therapy. J Acquir Immune Defic Syndr, 50, 499–505. [DOI] [PubMed] [Google Scholar]
- Samet JH, Horton NJ, Meli S, Freedberg KA & Palepu A. 2004. Alcohol consumption and antiretroviral adherence among HIV-infected persons with alcohol problems. Alcohol Clin Exp Res, 28, 572–577. [DOI] [PubMed] [Google Scholar]
- Saunders JB, Aasland OG, Babor TF, De La Fuente JR & Grant M. 1993. Development of the Alcohol Use Disorders Identification Test (AUDIT): WHO Collaborative Project on Early Detection of Persons with Harmful Alcohol Consumption--II. Addiction, 88, 791–804. [DOI] [PubMed] [Google Scholar]
- Shelmet JJ, Reichard GA, Skutches CL, Hoeldtke RD, Owen OE & Boden G. 1988. Ethanol causes acute inhibition of carbohydrate, fat, and protein oxidation and insulin resistance. J Clin Invest, 81, 1137–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon L, Ferguson TF, Vande Stouwe C, Brashear MM, Primeaux SD, Theall KP, Welsh DA & Molina PE 2020. Prevalence of Insulin Resistance in Adults Living with HIV: Implications of Alcohol Use. AIDS Res Hum Retroviruses, 36, 742–752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srinivasa S. & Grinspoon SK 2014. Metabolic and body composition effects of newer antiretrovirals in HIV-infected patients. Eur J Endocrinol, 170, R185–202. [DOI] [PubMed] [Google Scholar]
- Tura A, Kautzky-Willer A. & Pacini G. 2006. Insulinogenic indices from insulin and C-peptide: comparison of beta-cell function from OGTT and IVGTT. Diabetes Res Clin Pract, 72, 298–301. [DOI] [PubMed] [Google Scholar]
- Ulwelling W. & Smith K. 2018. The PEth Blood Test in the Security Environment: What it is; Why it is Important; and Interpretative Guidelines. J Forensic Sci, 63, 1634–1640. [DOI] [PubMed] [Google Scholar]
- Wan Q, Liu Y, Guan Q, Gao L, Lee KO & Zhao J. 2005. Ethanol feeding impairs insulin-stimulated glucose uptake in isolated rat skeletal muscle: role of Gs alpha and cAMP. Alcohol Clin Exp Res, 29, 1450–6. [DOI] [PubMed] [Google Scholar]
- Wand H, Calmy A, Carey DL, Samaras K, Carr A, Law MG, Cooper DA, Emery S. & Committee ITIC 2007. Metabolic syndrome, cardiovascular disease and type 2 diabetes mellitus after initiation of antiretroviral therapy in HIV infection. AIDS, 21, 2445–53. [DOI] [PubMed] [Google Scholar]
- Welsh DA, Ferguson T, Theall KP, Simon L, Amedee A, Siggins RW, Nelson S, Brashear M, Mercante D. & Molina PE 2019. The New Orleans Alcohol Use in HIV Study: Launching a Translational Investigation of the Interaction of Alcohol Use with Biological and Socioenvironmental Risk Factors for Multimorbidity in People Living with HIV. Alcohol Clin Exp Res, 43, 704–709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willig AL & Overton ET 2016. Metabolic Complications and Glucose Metabolism in HIV Infection: A Review of the Evidence. Curr HIV/AIDS Rep, 13, 289–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Medical A. 2013. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA, 310, 2191–2194. [DOI] [PubMed] [Google Scholar]
- Worm SW, Friis-Møller N, Bruyand M, D’arminio Monforte A, Rickenbach M, Reiss P, El-Sadr W, Phillips A, Lundgren J, Sabin C. & Group D. a. D. S. 2010. High prevalence of the metabolic syndrome in HIV-infected patients: impact of different definitions of the metabolic syndrome. AIDS, 24, 427–35. [DOI] [PubMed] [Google Scholar]
- Zigman JM, Nakano Y, Coppari R, Balthasar N, Marcus JN, Lee CE, Jones JF, Deysher AE, Waxman AR, White RD, Williams TD, Lachey JL, Seeley RJ, Lowell BB & Elmquist JK 2005. Mice lacking ghrelin receptors resist the development of diet-induced obesity. [DOI] [PMC free article] [PubMed] [Google Scholar]