Abstract
Background
Sutureless and rapid deployment valves for aortic valve replacement (AVR) were introduced in Korea in December 2016. This study evaluated changing trends in the prosthetic valves used for AVR in Korea after the introduction of sutureless and rapid deployment valves.
Methods
From December 2016 to December 2018, 4,899 patients underwent AVR in Korea. After applying the exclusion criteria, 4,872 patients were analyzed to determine changes in the type of prosthetic valve used for AVR. The study period was divided into 5 groups corresponding to 5-month intervals.
Results
The total number of AVR cases was 194.88±28.78 per month during the study period. Mechanical valves were used in approximately 27% to 33% of cases, and the proportion of mechanical valve use showed a tendency to decrease, with marginal significance overall (p=0.078) and significant decreases in patients less than 60 years of age and in men (p=0.013 and p=0.023, respectively). The use of sutureless valves increased from 13.4% to 25.8% of cases (p<0.001), especially in elderly patients (>70 years) and those requiring concomitant surgery. In a comparison between sutureless and rapid deployment valves, the use of Perceval S valves (a type of sutureless valve), gradually increased (p<0.001).
Conclusion
After the introduction of sutureless and rapid deployment valves in Korea, the rate of use of these new valves remarkably increased, especially in elderly patients and those requiring concomitant surgery. Further studies should investigate the clinical outcomes of these new prostheses.
Keywords: Aortic valve surgery, Korea, Rapid deployment valve, Sutureless valve, Trends
Introduction
Although surgical aortic valve replacement (AVR) is the standard treatment for aortic valve disease, there have been some limitations in applying conventional AVR in all patients. Recently, transcatheter aortic valve implantation has been used for intermediate- and high-risk patients [1,2], and sutureless (or rapid deployment) AVR (SuAVR) has also gained attention as a new alternative option for high-risk patients [3-5].
SuAVR involves a short aortic cross-clamp (ACC) time, since suture placement is not required, and enables straightforward implantation in limited surgical fields [6]. Currently, 2 types of sutureless aortic valves are available: The Edwards Intuity Elite valve system (Intuity valve; Edwards Lifesciences, Irvine, CA, USA) and the Perceval S valve (Perceval valve; LivaNova, London, UK). The Intuity valve system obtained the Conformité Européenne (CE) mark in 2012, and received Food and Drug Administration (FDA) approval in 2016 [7]. The Perceval valve obtained the CE mark and received FDA approval in 2011 and 2016, respectively [8]. In Korea, interest in sutureless valve systems has increased since December 2016, when the Korean National Health Insurance Service (NHIS) began covering these valves.
Several other studies have explored national trends in valve use for AVR; the frequency of using mechanical valves has decreased, and the use of bioprosthetic valves has increased [9-13]. However, there are few studies with relatively recent national data on valve use for AVR surgery. In particular, limited data have been reported on national trends in the types of prosthetic valves used for AVR since sutureless valves were introduced. Therefore, this study aimed to analyze the changing trends in the types of valves used in Korea after the introduction of the sutureless aortic valve.
Methods
Data source and patient characteristics
Data for the present study was obtained from the claims database of the Korean NHIS, which is a single insurer covering the entire Korean population that is managed by the government. The NHIS provides healthcare insurance coverage to the vast majority (>97%) of residents in Korea [14,15]. From December 2016, when SuAVR was approved for insurance coverage, to December 2018, 4,899 patients who underwent AVR using conventional prosthetic valves or sutureless aortic prostheses were identified based on the procedure codes of the Korean NHIS (O1793: aortic valve replacement, O1799: sutureless aortic valve replacement). Excluding 23 patients who were under the age of 19 and 4 patients for whom age data were not available, a total of 4,872 patients were enrolled in this study (Table 1).
Table 1.
Patient characteristics
| Characteristic | Total (n=4,872) | Mechanical valve (n=1,493) | Bioprosthetic valve (n=3,379) | p-value |
|---|---|---|---|---|
| Age (yr) | ||||
| 19–59 | 1,114 (22.9) | 927 (62.1) | 187 (5.5) | <0.001 |
| 60–69 | 1,434 (29.4) | 505 (33.8) | 929 (27.5) | <0.001 |
| 70–79 | 1,774 (36.4) | 58 (3.9) | 1,716 (50.8) | <0.001 |
| >80 | 550 (11.3) | 3 (0.2) | 547 (16.2) | <0.001 |
| Male | 2,800 (57.4) | 936 (62.7) | 1,864 (55.2) | <0.001 |
| Combined operation | 2,250 (46.2) | 789 (52.8) | 1,461 (43.2) | <0.001 |
| Tricuspid valve | 417 (8.6) | 190 (12.7) | 227 (6.7) | <0.001 |
| Mitral valve | 847 (17.4) | 409 (27.4) | 438 (13.0) | <0.001 |
| Arrhythmia | 534 (11.0) | 229 (15.3) | 305 (9.0) | <0.001 |
| Aorta | 774 (15.9) | 260 (17.4) | 514 (15.2) | 0.052 |
| Coronary artery bypass grafting | 645 (13.2) | 108 (7.2) | 537 (15.9) | <0.001 |
| History of diseases | ||||
| Cancer | 537 (11.0) | 106 (7.1) | 431 (12.8) | <0.001 |
| Hypertension | 4,076 (83.7) | 1,138 (76.2) | 2,938 (86.9) | <0.001 |
| Dyslipidemia | 3,548 (72.8) | 959 (64.2) | 2,589 (76.6) | <0.001 |
| Chronic obstructive pulmonary disease | 2,358 (48.4) | 625 (41.9) | 1,733 (51.3) | <0.001 |
| Cerebrovascular disease | 1,054 (21.6) | 221 (14.8) | 833 (24.7) | <0.001 |
| Peripheral vascular disease | 1,587 (32.6) | 398 (26.7) | 1,189 (35.2) | <0.001 |
| Kidney disease | 946 (19.4) | 252 (16.9) | 694 (20.5) | 0.003 |
| Congestive heart failure | 2,633 (54.0) | 768 (51.4) | 1,865 (55.2) | 0.015 |
| Diabetes mellitus | 2,243 (46.0) | 601 (40.3) | 1,642 (48.6) | <0.001 |
| Liver disease | 1,388 (28.5) | 410 (27.5) | 978 (28.9) | 0.291 |
Values are presented as number (%).
The patients were divided into 4 groups according to age (19–59, 60–69, 70–79, and 80 years or older). There were 1,114 patients (22.9%) aged 19–59, 1,434 patients (29.4%) aged 60–69, 1,774 patients (36.4%) aged 70–79, and 550 patients (11.3%) aged 80 or older. Combined surgery was defined as the concomitant presence of the appropriate treatment code for tricuspid valve surgery, mitral valve surgery, arrhythmia surgery, aortic surgery, and coronary artery bypass grafting. A combined operation was performed in 2,250 patients (46.2%), including mitral valve surgery (n=847, 17.4%) and aorta surgery (n=774, 15.7%). To evaluate trends in the use of specific prosthetic valve types, the study period was divided into 5 groups corresponding to 5-month intervals.
Statistical analysis
Statistical analyses were performed using IBM SPSS ver. 26.0 (IBM Corp., Armonk, NY, USA). Data were expressed as the mean±standard deviation or proportions. The Kolmogorov-Smirnov test was used to confirm the normality of the test data. The chi-square test or Fisher exact test for categorical variables and the Student t-test for continuous variables were used to make comparisons between the 2 groups. The number of samples was too small to perform normality testing by period, so normality testing was conducted by month. The Cochran-Armitage test was used to analyze trends in the proportion of prosthetic valves used. A p-value <0.05 was used as the threshold for statistical significance, and a p-value ≥0.05 but <0.1 was considered to indicate marginal significance.
This study was reviewed and approved by the Institutional Review Board of the National Evidence-based Healthcare Collaborating Agency (approval no., NECA IRB 19-006). The requirement for informed consent from individual patients was omitted since it was to be conducted on existing anonymous data from previous years.
Results
Overall trends of aortic valve replacement by prosthesis valve type
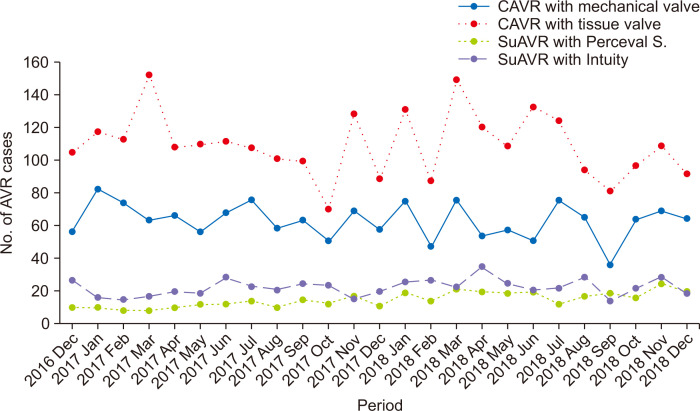
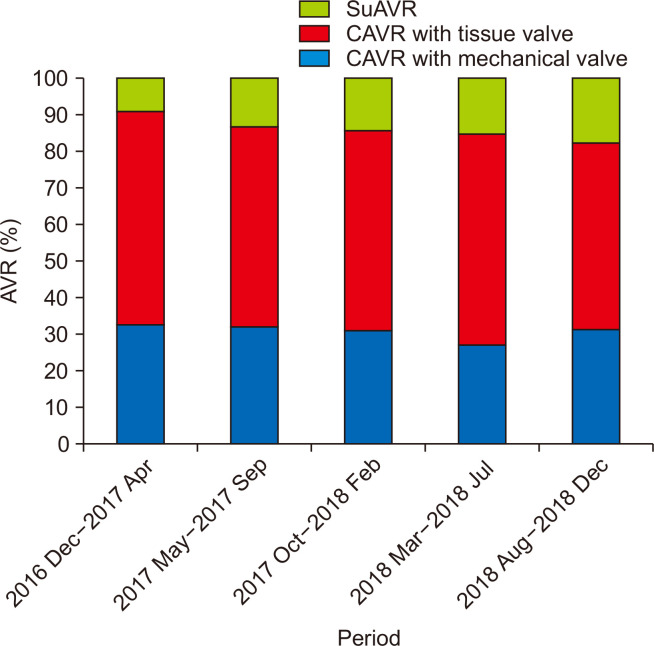
Fig. 1 shows the monthly trends in the types of prosthesis valves used for AVR for 25 months. Among the 4,872 patients, 1,493 patients (30.6%) underwent AVR with a mechanical valve and 3,379 patients (69.4%) underwent AVR with a bioprosthetic valve. The number of total AVR cases per month was 194.88±28.78. The numbers of bioprosthetic and mechanical AVR cases were 135.16±22.19 and 59.72±11.16, respectively. In groups divided into 5-month period, although the number of AVR procedures decreased from 1,006 cases (20.6%) in the first period to 901 cases (18.5%) in the last period, there was no significant change in the total number of AVR cases (p=0.20). Mechanical valves were used in 27% to 33% of cases, and the proportion of mechanical valves showed a tendency to decrease with marginal significance (p=0.078) (Table 2, Fig. 2).
Fig. 1.
Number of aortic valve replacement (AVR) procedures according to valve type. CAVR, conventional aortic valve replacement; SuAVR, sutureless (or rapid deployment) aortic valve replacement.
Table 2.
Trends in prosthetic valve types in aortic valve replacement
| Variable | Total | Dec 2016–Apr 2017 | May 2017–Sep 2017 | Oct 2017–Feb 2018 | Mar 2018–Jul 2018 | Aug 2018–Dec 2018 | p-value |
|---|---|---|---|---|---|---|---|
| Total (n=4,872) | |||||||
| Overall | 4,872 (100.0) | 1,006 (20.6) | 956 (19.6) | 912 (18.7) | 1,097 (22.5) | 901 (18.5) | 0.200a) |
| Mechanical valve | 1,493 (30.6) | 327 (32.5) | 306 (32.0) | 282 (30.9) | 297 (27.1) | 281 (31.2) | 0.078 |
| Bioprosthetic valve | 679 (67.5) | 650 (68.0) | 630 (69.1) | 800 (72.9) | 620 (68.8) | ||
| Male (n=2,800) | 3,379 (69.4) | 0.023 | |||||
| Mechanical valve | 936 (33.4) | 206 (35.2) | 206 (37.4) | 172 (33.9) | 184 (28.8) | 168 (32.6) | |
| Bioprosthetic valve | 380 (64.8) | 345 (62.6) | 336 (66.1) | 456 (71.3) | 347 (67.4) | ||
| Age 19–59 yr (n=1,114) | 1,864 (66.6) | 0.013 | |||||
| Mechanical valve | 927 (83.2) | 193 (86.2) | 214 (87.7) | 174 (82.1) | 178 (78.4) | 168 (81.2) | |
| Bioprosthetic valve | 31 (13.8) | 30 (12.3) | 38 (17.9) | 49 (21.6) | 39 (18.8) | ||
| Age 60–69 yr (n=1,434) | 187 (16.8) | 0.975 | |||||
| Mechanical valve | 505 (35.2) | 119 (40.6) | 81 (28.5) | 97 (35.1) | 105 (33.1) | 103 (39.0) | |
| Bioprosthetic valve | 174 (59.4) | 203 (71.5) | 179 (64.9) | 212 (66.9) | 161 (61.0) | ||
| Age 70–79 yr (n=1,774) | 929 (64.8) | 0.513 | |||||
| Mechanical valve | 58 (3.3) | 14 (3.6) | 11 (3.3) | 11 (3.6) | 12 (2.9) | 10 (2.9) | |
| Bioprosthetic valve | 373 (96.4) | 319 (96.7) | 291 (96.4) | 402 (97.1) | 331 (97.1) | ||
| Age >80 yr (n=550) | 1,716 (96.7) | 0.972 | |||||
| Mechanical valve | 3 (0.5) | 1 (1.0) | 0 | 0 | 2 (1.4) | 0 | |
| Bioprosthetic valve | 547 (99.5) | 101 (99.0) | 98 (100.0) | 122 (100.0) | 137 (98.6) | 89 (100.0) |
Values are presented as number (%).
a)p-values of normality testing for the overall number of aortic valve replacement procedures were calculated monthly, not periodically.
Fig. 2.
Proportion of aortic valve replacements (AVRs) in 5-month periods according to valve type. CAVR, conventional aortic valve replacement; SuAVR, sutureless (or rapid deployment) aortic valve replacement.
For the groups divided by age, the use of mechanical valves significantly decreased in patients <60 years of age (p=0.013), whereas there were no significant changes in the proportion of cases that used mechanical or bioprosthetic valves in patients who were ≥60 years of age (Table 2). The proportion of mechanical valves used in men ranged from 29% to 37%, and the proportion significantly decreased throughout the study period (p=0.023) (Table 2).
Trends of sutureless valve utilization
Among the 3,379 patients who underwent AVR with a bioprosthetic valve, 686 patients (20.3%) underwent sutureless AVR. Intuity valves were used in 439 patients (64.0%), while Perceval valves were used in 247 patients (36.0%). Conventional bioprosthetic valves were frequently used in patients aged 60–69 years, patients with liver disease, and patients requiring concomitant surgery. Sutureless valves were preferred in patients over 80 years old (Table 3).
Table 3.
Characteristics of patients who received bioprosthetic valves
| Variable | Conventional bioprostheses (n=2,693) | Sutureless or RD valves (n=686) | Intuity (n=439) | Perceval (n=247) | p-valuea) | p-valueb) |
|---|---|---|---|---|---|---|
| Age (yr) | ||||||
| 19–59 | 152 (5.6) | 35 (5.1) | 29 (6.6) | 6 (2.4) | 0.579 | 0.017 |
| 60–69 | 766 (28.4) | 163 (23.8) | 118 (26.9) | 45 (18.2) | 0.014 | 0.011 |
| 70–79 | 1,378 (51.2) | 338 (49.3) | 201 (45.8) | 137 (55.5) | 0.375 | 0.015 |
| >80 | 390 (14.5) | 150 (21.9) | 91 (20.7) | 59 (23.9) | <0.001 | 0.337 |
| Male | 1,500 (55.7) | 364 (53.1) | 246 (56.0) | 118 (47.8) | 0.215 | 0.037 |
| Combined operation | 1,221 (45.3) | 240 (35) | 165 (37.6) | 75 (30.4) | <0.001 | 0.057 |
| Tricuspid valve | 188 (7.0) | 39 (5.7) | 17 (3.9) | 22 (8.9) | <0.001 | 0.006 |
| Mitral valve | 382 (14.2) | 56 (8.2) | 39 (8.9) | 17 (6.9) | <0.001 | 0.358 |
| Arrhythmia | 260 (9.7) | 45 (6.6) | 33 (7.5) | 12 (4.9) | 0.012 | 0.177 |
| Aorta | 452 (16.8) | 62 (9.0) | 59 (13.4) | 3 (1.2) | <0.001 | <0.001 |
| CABG | 440 (16.3) | 97 (14.1) | 55 (12.5) | 42 (17.0) | 0.016 | 0.106 |
| History of diseases | ||||||
| Cancer | 350 (13.0) | 81 (11.8) | 52 (11.8) | 29 (11.7) | 0.405 | 0.968 |
| Hypertension | 2,336 (86.7) | 602 (87.8) | 386 (87.9) | 216 (87.4) | 0.483 | 0.855 |
| Dyslipidemia | 2,056 (76.3) | 533 (77.7) | 346 (78.8) | 187 (75.7) | 0.232 | 0.348 |
| COPD | 1,385 (51.4) | 348 (50.7) | 216 (49.2) | 132 (53.4) | 0.743 | 0.286 |
| Cerebrovascular disease | 651 (24.2) | 182 (26.5) | 120 (27.3) | 62 (25.1) | 0.201 | 0.525 |
| Peripheral vascular disease | 965 (35.8) | 224 (32.7) | 147 (33.5) | 77 (31.2) | 0.119 | 0.536 |
| Kidney disease | 558 (20.7) | 136 (19.8) | 73 (16.6) | 63 (25.5) | 0.604 | 0.005 |
| Congestive heart failure | 1,479 (54.9) | 386 (56.3) | 236 (53.8) | 150 (60.7) | 0.526 | 0.077 |
| Diabetes mellitus | 1,304 (48.4) | 338 (49.3) | 204 (46.5) | 134 (54.3) | 0.691 | 0.050 |
| Liver disease | 808 (30.0) | 170 (24.8) | 112 (25.5) | 58 (23.5) | 0.007 | 0.554 |
Values are presented as number (%).
RD, rapid deployment; CABG, coronary artery bypass grafting; COPD, chronic obstructive pulmonary disease.
a)Comparisons between conventional bioprostheses and sutureless and RD valves. b)Comparisons between Intuity and Perceval.
The proportion of sutureless AVR to total bioprosthetic AVR was around 13.4%–25.8% in the groups divided by 5-month period. The proportion of sutureless AVR dramatically increased from 13.4% in the first period to 25.8% in the last period (p<0.001) (Table 4, Fig. 2). The use of Perceval valves significantly increased from 23.1% to 45.0%, while the use of Intuity valves significantly decreased from 76.9% to 55.0% during the study period (p<0.001).
Table 4.
Trends in bioprosthetic valves by patient characteristics
| Variable | Total | Dec 2016–Apr 2017 | May 2017–Sep 2017 | Oct 2017–Feb 2018 | Mar 2018–Jul 2018 | Aug 2018–Dec 2018 | p-value |
|---|---|---|---|---|---|---|---|
| Total (n=3,379) | <0.001 | ||||||
| Conventional bioprostheses | 2,693 (79.7) | 588 (86.6) | 520 (80.0) | 495 (78.6) | 630 (78.8) | 460 (74.2) | |
| Sutureless or rapid deployment valves | 686 (20.3) | 91 (13.4) | 130 (20.0) | 135 (21.4) | 170 (21.3) | 160 (25.8) | |
| Perceval S | 247 (36.0) | 21 (23.1) | 38 (29.2) | 48 (35.6) | 68 (40.0) | 72 (45.0) | <0.001 |
| Intuity | 439 (64.0) | 70 (76.9) | 92 (70.8) | 87 (64.4) | 102 (60.0) | 88 (55.0) | |
| Combined procedure (n=1,461) | <0.001 | ||||||
| Conventional bioprostheses | 1,221 (83.6) | 253 (89.1) | 236 (85.5) | 229 (81.5) | 309 (83.1) | 194 (78.2) | |
| Sutureless or rapid deployment valves | 240 (16.4) | 31 (10.9) | 40 (14.5) | 52 (18.5) | 63 (16.9) | 54 (21.8) | |
| Age 19–59 yr (n=1,114) | 0.250 | ||||||
| Conventional bioprostheses | 152 (81.3) | 26 (83.9) | 25 (83.3) | 32 (84.2) | 41 (83.7) | 28 (71.8) | |
| Sutureless or rapid deployment valves | 35 (18.7) | 5 (16.1) | 5 (16.7) | 6 (15.8) | 8 (16.3) | 11 (28.2) | |
| Age 60–69 yr (n=1,434) | 0.287 | ||||||
| Conventional bioprostheses | 766 (82.5) | 151 (86.8) | 164 (80.8) | 148 (82.7) | 171 (80.7) | 132 (82.0) | |
| Sutureless or rapid deployment valves | 163 (17.5) | 23 (13.2) | 39 (19.2) | 31 (17.3) | 41 (19.3) | 29 (18.0) | |
| Age 70–79 yr (n=1,774) | <0.001 | ||||||
| Conventional bioprostheses | 1,378 (80.3) | 329 (88.2) | 258 (80.9) | 226 (77.7) | 322 (80.1) | 243 (73.4) | |
| Sutureless or rapid deployment valves | 338 (19.7) | 44 (11.8) | 61 (19.1) | 65 (22.3) | 80 (19.9) | 88 (26.6) | |
| Age >80 yr (n=550) | 0.007 | ||||||
| Conventional bioprostheses | 397 (72.6) | 82 (81.2) | 73 (74.5) | 89 (73.0) | 96 (70.1) | 57 (64.0) | |
| Sutureless or rapid deployment valves | 150 (27.4) | 19 (18.8) | 25 (25.5) | 33 (27.0) | 41 (29.9) | 32 (36.0) |
Values are presented as number (%).
In the 1,461 patients (43.2%) who underwent concomitant procedures, the use of sutureless valves significantly increased from 10.9% in the first period to 21.8% in the last period (p<0.001). There were no significant changes in the proportion of the use of sutureless aortic valves in patients <70 years of age, but the use of sutureless valves increased significantly in patients ≥70 years of age (Table 4). In particular, the use of sutureless valves increased from 18.8% to 27.4% in patients who were older than 80 years of age. When comparing the use of the Perceval and Intuity valves, the use of the Perceval valve in patients aged 70–79 years (p=0.007) and ≥80 years (p=0.009) showed significant increases.
Discussion
There were 3 main findings of this study. First, the frequency of using mechanical valves in the aortic position declined. Second, the frequency of using sutureless or rapid deployment aortic valves significantly increased in patients aged >70 years or patients who required a concomitant procedure. Third, the frequency of using Perceval valves steadily increased, whereas the proportion of cases using Intuity valves decreased.
During the study period, the frequency of using mechanical valves decreased with marginal significance. In particular, the use of mechanical valves in patients under 60 years of age significantly decreased from 86.2% to 81.2%. The age criteria for valve selection between mechanical and tissue valves depend on patients’ lifestyle and choices, and the indications for using bioprosthetic valves have been expanded [16-21]. The latest guidelines published in 2020 suggested that for patients between 50 and 65 years of age who require AVR, individualizing the choice of a mechanical or bioprosthetic valve prosthesis based on patient factors and after informed shared decision-making would be rational [21]. In addition, improvements in the technology of valve-in-valve transcatheter AVR could increase the number of patients who choose a bioprosthetic valve [22].
This study confirmed that the use of sutureless and rapid deployment valves has gradually increased after these new types of prostheses were introduced in Korea. In particular, the use of sutureless and rapid deployment valves increased remarkably in patients over 70 years of age and those who required concomitant surgery. According to a previous meta-analysis, using a sutureless or rapid deployment valve can reduce the ACC time and cardiopulmonary bypass (CPB) time by approximately 25 minutes [6]. Because prolonged ACC and CPB times are associated with increased morbidity and mortality [23,24], surgeons may prefer to use a sutureless or rapid deployment valve for high-risk patients with other cardiac diseases. Considering the growth of the aging population and increasing demand for minimally invasive surgery, the use of sutureless or rapid deployment valves is expected to increase in the future. In addition, many recent studies have reported good mid- and long-term clinical outcomes of sutureless and rapid deployment valves in terms of safety, efficacy, hemodynamic performance, and durability [4,5,25,26].
When comparing sutureless (Perceval) valves with rapid deployment (Intuity) valves during the study period, the use of sutureless valves was seen to increase, whereas the use of rapid deployment valves decreased. In the first period, the use of rapid deployment valves was 3.3 times more common than the use of sutureless valves. However, the use of sutureless valves gradually increased and reached a similar level as that of rapid deployment valves in the most recent period. These trends are related to the characteristics of the 2 valves. At first, the rapid deployment valve may seem more familiar to surgeons because the valve is combined with a preexisting valve (Carpentier-Edwards Perimount Magna Ease) with a sub-annular frame and can be inserted through aortotomy, which is usually used for conventional AVR. In the same context, sutureless valves require higher aortotomy, which could be a barrier to inexperienced surgeons. However, the additional benefit of the sutureless valve in high-risk patients who have small sinotubular junctions or aortic root calcification might have contributed to the increased use of this valve.
The present study has several limitations. First, although the surgical approach used depends on the choice of the prosthetic valve type, data on the surgical approach were not analyzed. Second, this study was designed to collect insurance claims data after sutureless valves were introduced to clinical practice. Since data were not collected on the use of prostheses before the introduction of sutureless valves, a comparison of the use of prostheses before and after the introduction of sutureless valves could not be made. Third, although the pathophysiology of the aortic valve (regurgitation or stenosis) and some anatomical factors, such as leaflet status or dilated sinus of Valsalva, are commonly considered in valve selection, this claim data were impossible to obtain; likewise, it was not possible to analyze data related to echocardiography or computed tomography. Therefore, the specific indications of each valve were not considered when analyzing trends in prosthesis use in this study.
Footnotes
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Funding
This work was supported by the National Evidence-based Healthcare Collaborating Agency (grant no., NR19-002).
References
- 1.Singh K, Bhalla AS, Qutub MA, Carson K, Labinaz M. Systematic review and meta-analysis to compare outcomes between intermediate- and high-risk patients undergoing transcatheter aortic valve implantation. Eur Heart J Qual Care Clin Outcomes. 2017;3:289–95. doi: 10.1093/ehjqcco/qcx014. [DOI] [PubMed] [Google Scholar]
- 2.Reardon MJ, van Mieghem NM, Popma JJ, et al. Surgical or transcatheter aortic-valve replacement in intermediate-risk patients. N Engl J Med. 2017;376:1321–31. doi: 10.1056/NEJMoa1700456. [DOI] [PubMed] [Google Scholar]
- 3.Folliguet TA, Laborde F, Zannis K, Ghorayeb G, Haverich A, Shrestha M. Sutureless perceval aortic valve replacement: results of two European centers. Ann Thorac Surg. 2012;93:1483–8. doi: 10.1016/j.athoracsur.2012.01.071. [DOI] [PubMed] [Google Scholar]
- 4.Szecel D, Eurlings R, Rega F, Verbrugghe P, Meuris B. Perceval sutureless aortic valve implantation: midterm outcomes. Ann Thorac Surg. 2021;111:1331–7. doi: 10.1016/j.athoracsur.2020.06.064. [DOI] [PubMed] [Google Scholar]
- 5.Andreas M, Coti I, Rosenhek R, et al. Intermediate-term outcome of 500 consecutive rapid-deployment surgical aortic valve procedures. Eur J Cardiothorac Surg. 2019;55:527–33. doi: 10.1093/ejcts/ezy273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hurley ET, O'Sullivan KE, Segurado R, Hurley JP. A meta-analysis examining differences in short-term outcomes between sutureless and conventional aortic valve prostheses. Innovations (Phila) 2015;10:375–82. doi: 10.1097/imi.0000000000000221. [DOI] [PubMed] [Google Scholar]
- 7.D'Onofrio A, Tessari C, Filippini C, et al. Early and mid-term results of rapid deployment valves: the Intuity Italian Registry (INTU-ITA) Ann Thorac Surg. 2018;106:1742–9. doi: 10.1016/j.athoracsur.2018.07.002. [DOI] [PubMed] [Google Scholar]
- 8.Concistre G, Chiaramonti F, Bianchi G, et al. Aortic valve replacement with perceval bioprosthesis: single-center experience with 617 implants. Ann Thorac Surg. 2018;105:40–6. doi: 10.1016/j.athoracsur.2017.05.080. [DOI] [PubMed] [Google Scholar]
- 9.Abe T, Kumamaru H, Nakano K, Motomura N, Miyata H, Takamoto S. Status of cardiovascular surgery in Japan between 2017 and 2018: a report based on the Japan Cardiovascular Surgery Database: 3. valvular heart surgery. Asian Cardiovasc Thorac Ann. 2021;29:300–9. doi: 10.1177/0218492320981459. [DOI] [PubMed] [Google Scholar]
- 10.Dunning J, Gao H, Chambers J, et al. Aortic valve surgery: marked increases in volume and significant decreases in mechanical valve use: an analysis of 41,227 patients over 5 years from the Society for Cardiothoracic Surgery in Great Britain and Ireland National Database. J Thorac Cardiovasc Surg. 2011;142:776–82. doi: 10.1016/j.jtcvs.2011.04.048. [DOI] [PubMed] [Google Scholar]
- 11.Siregar S, de Heer F, Groenwold RH, et al. Trends and outcomes of valve surgery: 16-year results of Netherlands Cardiac Surgery National Database. Eur J Cardiothorac Surg. 2014;46:386–97. doi: 10.1093/ejcts/ezu017. [DOI] [PubMed] [Google Scholar]
- 12.Isaacs AJ, Shuhaiber J, Salemi A, Isom OW, Sedrakyan A. National trends in utilization and in-hospital outcomes of mechanical versus bioprosthetic aortic valve replacements. J Thorac Cardiovasc Surg. 2015;149:1262–9. doi: 10.1016/j.jtcvs.2015.01.052. [DOI] [PubMed] [Google Scholar]
- 13.Carnero-Alcazar M, Maroto-Castellanos LC, Hernandez-Vaquero D, et al. Isolated aortic valve replacement in Spain: national trends in risks, valve types, and mortality from 1998 to 2017. Rev Esp Cardiol (Engl Ed) 2021;74:700–7. doi: 10.1016/j.rec.2020.06.008. [DOI] [PubMed] [Google Scholar]
- 14.Moon TJ. Light and shadows of the Korean healthcare system. J Korean Med Sci. 2012;27(Suppl):S3–6. doi: 10.3346/jkms.2012.27.S.S3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Song SO, Jung CH, Song YD, et al. Background and data configuration process of a nationwide population-based study using the Korean national health insurance system. Diabetes Metab J. 2014;38:395–403. doi: 10.4093/dmj.2014.38.5.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bartus K, Sadowski J, Litwinowicz R, et al. Changing trends in aortic valve procedures over the past ten years-from mechanical prosthesis via stented bioprosthesis to TAVI procedures-analysis of 50,846 aortic valve cases based on a Polish National Cardiac Surgery Database. J Thorac Dis. 2019;11:2340–9. doi: 10.21037/jtd.2019.06.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.American College of Cardiology/American Heart Association Task Force on Practice Guidelines, author; Society of Cardiovascular Anesthesiologists, author; Society for Cardiovascular Angiography and Interventions, et al, author. ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing committee to revise the 1998 guidelines for the management of patients with valvular heart disease): developed in collaboration with the Society of Cardiovascular Anesthesiologists: endorsed by the Society for Cardiovascular Angiography and Interventions and the Society of Thoracic Surgeons. Circulation. 2006;114:e84–231. doi: 10.1161/CIRCULATIONAHA.106.177303. [DOI] [PubMed] [Google Scholar]
- 18.Nishimura RA, Otto CM, Bonow RO, et al. 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Thorac Cardiovasc Surg. 2014;148:e1–132. doi: 10.1161/CIR.0000000000000031. [DOI] [PubMed] [Google Scholar]
- 19.Nishimura RA, Otto CM, Bonow RO, et al. 2017 AHA/ACC focused update of the 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2017;135:e1159–95. doi: 10.1161/CIR.0000000000000503. [DOI] [PubMed] [Google Scholar]
- 20.Falk V, Baumgartner H, Bax JJ, et al. 2017 ESC/EACTS guidelines for the management of valvular heart disease. Eur J Cardiothorac Surg. 2017;52:616–64. doi: 10.1093/ejcts/ezx324. [DOI] [PubMed] [Google Scholar]
- 21.Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021;143:e72–227. doi: 10.1161/CIR.0000000000000932. [DOI] [PubMed] [Google Scholar]
- 22.Takagi H, Mitta S, Ando T. Meta-analysis of valve-in-valve transcatheter versus redo surgical aortic valve replacement. Thorac Cardiovasc Surg. 2019;67:243–50. doi: 10.1055/s-0038-1668135. [DOI] [PubMed] [Google Scholar]
- 23.Al-Sarraf N, Thalib L, Hughes A, et al. Cross-clamp time is an independent predictor of mortality and morbidity in low- and high-risk cardiac patients. Int J Surg. 2011;9:104–9. doi: 10.1016/j.ijsu.2010.10.007. [DOI] [PubMed] [Google Scholar]
- 24.Iino K, Miyata H, Motomura N, et al. Prolonged cross-clamping during aortic valve replacement is an independent predictor of postoperative morbidity and mortality: analysis of the Japan Cardiovascular Surgery Database. Ann Thorac Surg. 2017;103:602–9. doi: 10.1016/j.athoracsur.2016.06.060. [DOI] [PubMed] [Google Scholar]
- 25.Filip G, Litwinowicz R, Kapelak B, et al. Mid-term follow-up after suture-less aortic heart valve implantation. J Thorac Dis. 2018;10:6128–36. doi: 10.21037/jtd.2018.10.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Williams ML, Flynn CD, Mamo AA, et al. Long-term outcomes of sutureless and rapid-deployment aortic valve replacement: a systematic review and meta-analysis. Ann Cardiothorac Surg. 2020;9:265–79. doi: 10.21037/acs-2020-surd-25. [DOI] [PMC free article] [PubMed] [Google Scholar]