ABSTRACT
Effective disinfection technology to combat severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can help reduce viral transmission during the ongoing COVID-19 global pandemic and in the future. UV devices emitting UVC irradiation (200 to 280 nm) have proven to be effective for virus disinfection, but limited information is available for SARS-CoV-2 due to the safety requirements of testing, which is limited to biosafety level 3 (BSL3) laboratories. In this study, inactivation of SARS-CoV-2 in thin-film buffered aqueous solution (pH 7.4) was determined across UVC irradiation wavelengths of 222 to 282 nm from krypton chloride (KrCl*) excimers, a low-pressure mercury-vapor lamp, and two UVC light-emitting diodes. Our results show that all tested UVC devices can effectively inactivate SARS-CoV-2, among which the KrCl* excimer had the best disinfection performance (i.e., highest inactivation rate). The inactivation rate constants of SARS-CoV-2 across wavelengths are similar to those for murine hepatitis virus (MHV) from our previous investigation, suggesting that MHV can serve as a reliable surrogate of SARS-CoV-2 with a lower BSL requirement (BSL2) during UV disinfection tests. This study provides fundamental information on UVC’s action on SARS-CoV-2 and guidance for achieving reliable disinfection performance with UVC devices.
IMPORTANCE UV light is an effective tool to help stem the spread of respiratory viruses and protect public health in commercial, public, transportation, and health care settings. For effective use of UV, there is a need to determine the efficiency of different UV wavelengths in killing pathogens, specifically SARS-CoV-2, to support efforts to control the ongoing COVID-19 global pandemic and future coronavirus-caused respiratory virus pandemics. We found that SARS-CoV-2 can be inactivated effectively using a broad range of UVC wavelengths, and 222 nm provided the best disinfection performance. Interestingly, 222-nm irradiation has been found to be safe for human exposure up to thresholds that are beyond those effective for inactivating viruses. Therefore, applying UV light from KrCl* excimers in public spaces can effectively help reduce viral aerosol or surface-based transmissions.
KEYWORDS: UV disinfection, far UVC, COVID-19, surrogate, human coronavirus 229E, murine hepatitis virus, MHV, bacteriophage Phi6
INTRODUCTION
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is an enveloped, nonsegmented positive-sense RNA virus (1), which is causing the ongoing COVID-19 global pandemic. It is transmitted primarily via respiratory droplets produced while talking, coughing, and sneezing (2). Indirect routes, such as airborne and surface-mediated transmission, are also possible, especially considering SARS-CoV-2 can stay viable in aerosols and on surfaces for up to 72 h (3). Effective disinfection procedures can help reduce viral transmission, especially in high-risk places, such as hospitals, other health care facilities, and public transportation systems.
UV devices emitting UVC irradiation (200 to 280 nm), such as the low-pressure (LP) UV lamp and UV light-emitting diodes (LEDs), have been widely used for virus disinfection of water, air, and surfaces since the early 20th century (4–8). Compared to other disinfection methods (e.g., heating and using chemical oxidants), UVC disinfection has several advantages, including rapid effectiveness, no chemical residual, and limited to no material degradation (6). One limitation of conventional UVC devices is that they are not safe for human exposure due to adverse effects on human skin and eyes (9, 10). Emerging far-UVC devices (emitting UVC irradiation in the wavelength range of 200 to 225 nm) like the krypton chloride (KrCl*) excimer, however, have been proposed to disinfect occupied public spaces, as recent studies reported that far-UVC light exposure results in no adverse effects on skin or eyes in mouse studies due to its very limited penetration into biological materials (11–14).
There are only a few studies that document inactivation efficiencies of SARS-CoV-2 using UVC devices. An average UV fluence of 1.2 to over 60 mJ/cm2 was required for 1-log inactivation (90%) of SARS-CoV-2 in aqueous solutions using LP UV lamps, reported in previous investigations (15–18), whereas 1.6 mJ/cm2 of UVC irradiation from a KrCl* excimer with a 222-nm bandpass filter was needed to achieve the same virus reduction (19). A few other studies also investigated UV inactivation effectiveness against SARS-CoV-2 in virus droplets and on surfaces using LP UV lamps and KrCl* excimers (17, 20, 21). Despite these prior works, information on UVC inactivation of SARS-CoV-2 is still limited across UV wavelengths and compared to that of surrogate enveloped viruses, primarily due to the safety requirement of testing, which is limited to biosafety level 3 (BSL3) laboratories. Thus, comparative studies, including reliable and accessible surrogates of SARS-CoV-2 with a lower BSL requirement, are needed for extensive assessment of UVC devices, sources, and wavelength disinfection performance.
In this study, UVC inactivation of SARS-CoV-2 in thin-film aqueous solution was determined using five UVC devices with different emission spectra in a bench-scale collimated beam apparatus. The UV sensitivities of SARS-CoV-2 and its potential testing surrogates classified as BSL1 and BSL2 viruses, including human coronavirus (HCoV) 229E, murine hepatitis virus (MHV), and bacteriophage Phi6 (22), were compared, and recommendations for reliable UV testing surrogates of SARS-CoV-2 are made.
RESULTS AND DISCUSSION
All UVC devices tested in this study were very effective in inactivating SARS-CoV-2 in aqueous solution (see Fig. 2). Among all tested UVC devices, unfiltered and filtered KrCl* excimers exhibited the greatest performance, with inactivation rate constants (mean ± standard error [SE]) of 1.52 ± 0.17 and 1.42 ± 0.40 cm2/mJ, respectively. These values are much higher than the value reported by Robinson et al. (19) (0.64 cm2/mJ). One possible explanation for such difference is that sample absorbance at 222 nm was much higher in the Robinson study (>30 cm−1) than this study (0.05 cm−1) (Fig. 1B), and UV absorption by constituents in the sample matrix (i.e., proteins and other constituents from the cell culture extracts) may affect the virus sensitivity to UV irradiation. Greater performance of KrCl* excimers compared to other UVC devices was also observed for nonenveloped viruses (e.g., MS2 coliphage and adenovirus) (4, 5, 23), enveloped bacteriophage Phi6 (22), and coronaviruses (22) in previous studies, suggesting such superior performance may be universal across virus types. This is likely because KrCl* excimers were capable of inflicting greater viral protein and nucleic acid damage than the other UVC devices due to the higher protein absorbance at far-UVC wavelengths around the 222-nm wavelength emitted by these devices (Fig. 1B). The superior performance of the KrCl* excimer is particularly promising because far-UVC devices are safe to be applied in occupied public spaces—up to the daily allowable threshold limit value of 25 mJ/cm2 at 220 nm (24) or perhaps beyond—to disinfect viruses in respiratory secretions and airborne droplets, as well as on contaminated surfaces, to limit the presence and transmission of SARS-CoV-2 or other respiratory viruses. Previous studies on aerosol and surface UV disinfection (25, 26) suggested that viruses in airborne droplets and on surfaces tend to be more susceptible to UVC irradiation. Recent work with UV 222-nm inactivation of SARS-CoV-2 on surfaces (17, 20, 21) and of other coronaviruses in air (12), however, show very similar inactivation compared to this study, suggesting data for inactivation generated using thin-film aqueous suspensions can represent inactivation of coronaviruses across various media.
FIG 2.
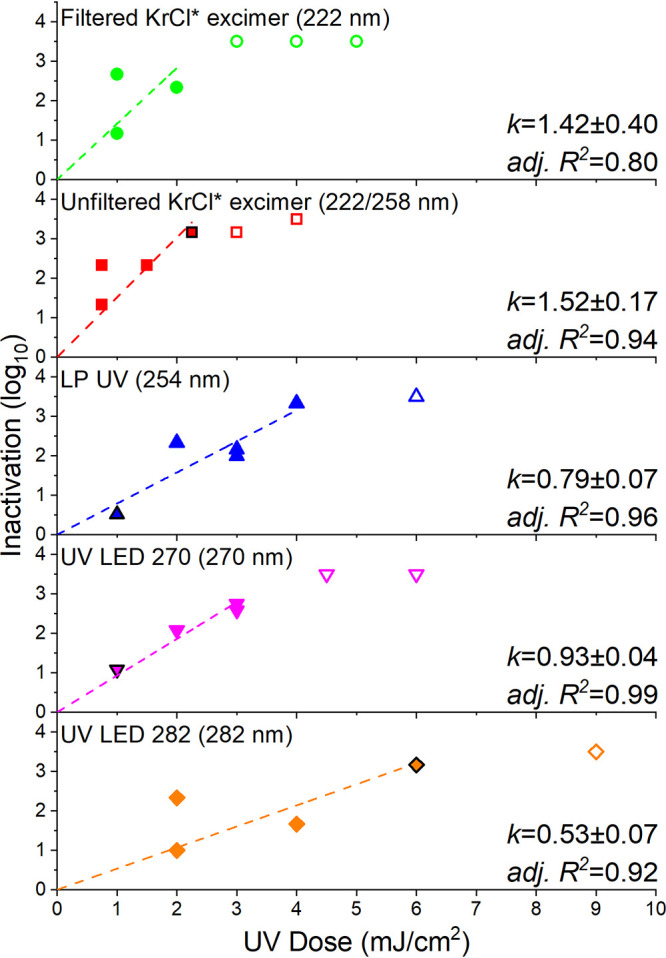
Dose response of SARS-CoV-2 to UV irradiation from all tested UVC devices. Dashed lines represent linear regression results computed from experimental data. The k values (mean ± standard error in cm2/mJ) and adjusted R2 values are listed. Open symbols represent samples with infectivity equal to or less than the detection limits. Solid symbols with a black edge represent two samples overlapping in the plot with the same UV dose response. Primary emission wavelengths for UVC devices are listed in each panel.
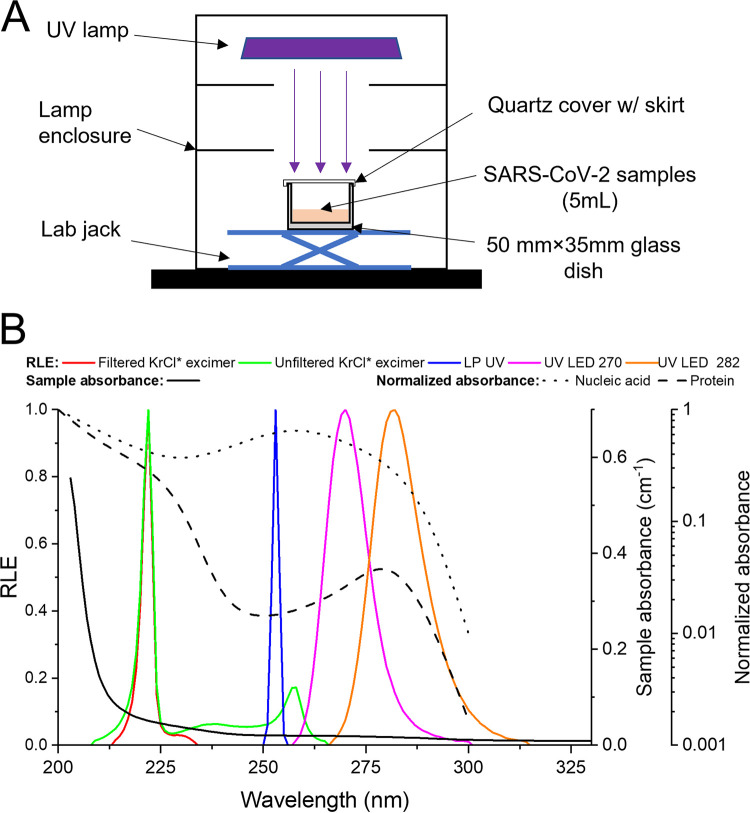
FIG 1.
Schematic diagram of bench-scale collimated beam apparatus (A) and relative lamp emission (RLE) for the UV devices used in this investigation, absorbance of samples used in UV exposure tests, and normalized absorbance (normalized to maximum value at 200 nm) of nucleic acid (DNA/RNA) and protein (B). The nucleic acid and protein absorbance data were reproduced from Ma et al. (22).
An average UV dose of 1.3 mJ/cm2 was required for 1-log inactivation of SARS-CoV-2 using the LP UV lamp, which is similar to the results from several previous studies (1.2 to 5.0 mJ/cm2 for 1-log inactivation) (15–17). Another study by Heilingloh et al. (18), however, suggested 1-log inactivation would require more than 60 mJ/cm2 using a LP UV source. This divergence from the UV doses reported in numerous other studies is likely due to the significant difference in the experimental setup for UV exposures and calculation for UV fluences. The inactivation tests reported by Heilingloh et al. were performed in cell culture media in 24-well plates with the UV source placed only 3 cm above the bottom of the plate, which could lead to differences in UV intensity between each well. Also, no information was given on how the UV irradiance was measured, there was no report of the absorbance of the suspending media, and standardized procedures for UV fluence calculation (e.g., corrections for sample UV absorbance, depth of sample, UV beam reflection and divergence, and petri factor) were not followed. Based on the data presented herein, no statistically significant difference in UV inactivation performance was observed between the LP UV lamp and the UV LED 270 (i.e., LED with a peak emission wavelength of 270 nm) (P = 0.16) (Fig. 2). Viral genome damage is likely to be the primary inactivation mechanism for these UVC devices (23), and SARS-CoV-2 should have similar sensitivities to UV irradiation from these devices due to similar levels of nucleic acid absorbance at their peak emission wavelengths (i.e., 254 and 270 nm, respectively) (Fig. 1B). UV LED 282 provided the lowest inactivation rate constant among all tested UVC devices. Viral genomes tend to absorb less UV irradiation in the wavelength range emitted from UV LED 282 (4, 27) (Fig. 1B), which leads to less genome damage. While viral proteins should be slightly more sensitive to UV irradiation from around the 282-nm wavelength (28), this previous observation did not appear to enhance the effectiveness of the 282-nm LED in the current study.
The inactivation rate constants of SARS-CoV-2 were compared with the values of potential enveloped virus surrogates: HCoV 229E, MHV, and bacteriophage Phi6 (Fig. 3). These three viruses were selected as candidates of SARS-CoV-2 surrogates for UV inactivation tests due to their molecular similarities (i.e., all are enveloped RNA viruses) and lower biosafety requirements (BSL1 for Phi6 and BSL2 for HCoV 229E and MHV). All virus surrogates were previously tested in the identical collimated beam apparatus, except that the quartz lid was not applied for non-BSL3 organisms. The inactivation rate constants were also calculated following the same data analysis method (22). Among the three candidates, MHV exhibited the greatest similarities in inactivation rate constants across UVC devices compared to SARS-CoV-2. No statistically significant differences in the rate constant values (P > 0.05) were observed for all tested UVC devices, except for the unfiltered KrCl* excimer (P = 0.008), for which the inactivation rate of MHV was only 26% lower than the value for SARS-CoV-2 (Fig. 3). These results suggest that MHV can serve as a reliable UV surrogate of SARS-CoV-2 testing across UVC wavelengths when a lower biosafety requirement is needed. HCoV 229E could also serve as a viable surrogate of SARS-CoV-2, especially for testing unfiltered KrCl* excimer (Fig. 3). Considering SARS-CoV-2, MHV, and HCoV 229E are all coronaviruses, evidence suggests that coronaviruses in general have similar sensitivities to UVC irradiation across wavelengths due to their similar molecular structures. This is further supported by comparing the UV inactivation rate constants of other coronaviruses, such as HCoV-OC43. UV inactivation rate constants of 0.77, 0.64, and 0.43 cm2/mJ were reported by Gerchman et al. (29) using UV LEDs with peak emission at 267, 279, and 286 nm, respectively, which are similar to the values we observed using UV LED 270 and UV LED 282 (0.93 and 0.53 cm2/mJ) (Fig. 2). Although significantly lower inactivation rate constants were observed for bacteriophage Phi6 (P < 0.05) (Fig. 3), it can still serve as a conservative virus surrogate where use of coronaviruses is not feasible (e.g., lack of mammalian cell culture facilities). Compared to nonenveloped viruses, use of enveloped viruses like bacteriophage Phi6 is particularly desirable in surface and aerosol disinfection tests to best represent any interactions between the viral envelope and its surrounding environment that may affect viral sensitivity to UVC irradiation (30–32). Other bacteriophages, such as T1 and T7, although nonenveloped double-stranded DNA phages, exhibit similar sensitivities to SARS-CoV-2 across UVC wavelengths (27) and could also serve as UV disinfection surrogates.
FIG 3.
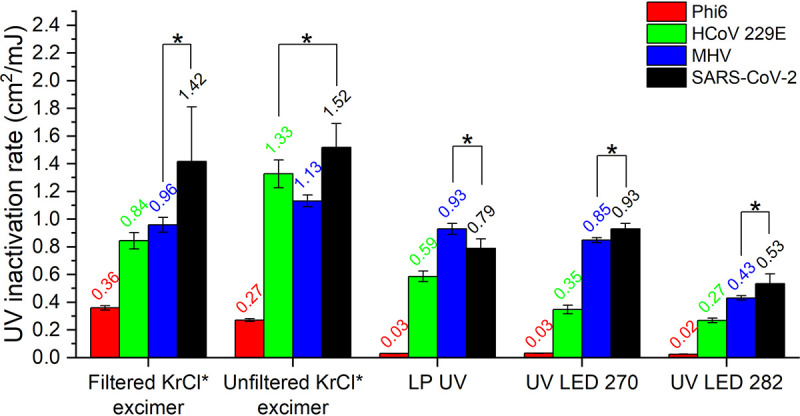
UV inactivation rate constants of SARS-CoV-2, two coronaviruses (HCoV 229E and MHV), and enveloped bacteriophage Phi6 for all tested UVC devices. The mean inactivation rate values are labeled. The values for Phi6, HCoV 229E, and MHV were published by Ma et al. (22). Asterisk brackets represent two inactivation rate values that were not statistically significantly different at the 95% confidence level (P > 0.05).
This research defines the fundamental inactivation rate constants of SARS-CoV-2 for UVC devices with peak emission wavelengths of 222 to 282 nm. These devices can be used to effectively inactivate SARS-CoV-2, among which far-UVC devices like the KrCl* excimer provided the best disinfection performance, with the added benefit of limited safety requirements when applied in occupied spaces. MHV is recommended as a reliable UV testing surrogate of SARS-CoV-2 due to its similar UV sensitivities across UVC wavelengths, but other T phages could also serve as surrogates. While these inactivation data align well with those from previous studies of UV disinfection of coronaviruses in aerosols and dried on surfaces, future work should continue to evaluate UV inactivation of SARS-CoV-2 in aqueous and other media relative to surrogates such as MHV or bacteriophage and expand these comparisons to other disinfectants important to minimizing the transmission of respiratory viruses.
MATERIALS AND METHODS
Virus preparation and enumeration.
SARS-CoV-2 (isolate USA WA1 2020), an enveloped respiratory virus, was propagated and assayed in the monkey kidney Vero E6 cell line (ATCC CRL-1586). Vero E6 cells were grown in Dulbecco’s modified Eagle’s medium (DMEM) with 10% fetal bovine serum (FBS), 100 μg/ml kanamycin sulfate, 200 U/ml penicillin, 200 μg/ml streptomycin, and 0.5 μg/ml amphotericin B and incubated for 3 days at 37°C in a 5% CO2 atmosphere. The viral stock was then added into Vero E6 cells with fresh medium and incubated for 2 days at the same conditions, when cytopathogenic effects (CPE) were observed in the monolayer. Infected cells were subjected to three freeze-thaw cycles to release the viruses, and the cell lysates were centrifuged at 1,000 × g for 20 min to pellet the cell debris for removal, and the pellet was discarded. The viruses in the supernatant then underwent a polyethylene glycol (12% [wt/vol], molecular weight [MW] of 8,000) precipitation with 0.5 M sodium chloride, and slow mixing overnight at 4°C. After centrifugation at 10,000 × g for 60 min at 4°C, the pelleted virus was resuspended in 0.01 M phosphate-buffered saline (PBS [pH 7.4]) to a volume of approximately 10 ml. The SARS-CoV-2 stocks were then aliquoted and stored at −80°C.
The viral stocks were enumerated on Vero E6 cells seeded into 96-well cell culture trays using the TCID50 (tissue culture infectious dose at the 50% endpoint) technique as described by Payment and Trudel (33). This technique determines the dilution at which 50% of the wells show CPE. Serial 10-fold dilutions of SARS-CoV-2 samples were prepared in DMEM without FBS, followed by plating onto Vero E6 monolayers prepared in 96-well trays in replicates of 6 per dilution with 50 μl per well. DMEM containing 2% FBS was then added to bring the volume in each well up to 180 μl. After the plates were further incubated for 7 days at 37°C in a 5% CO2 atmosphere, TCID50 values were then calculated using the Spearman and Kärber algorithm (detection limit of 6 TCID50/ml) (34).
UV exposure experiments.
The UV lamps were set up in a bench-scale collimated beam apparatus (Fig. 1A) as described by Bolton and Linden (35). Normalized emission spectra for these UV lamps as used in the experiments (Fig. 1B) were measured using a calibrated Maya 2000 Pro spectrometer (Ocean Insight, Dunedin, FL). Five UV sources were used in this investigation: an unfiltered KrCl* excimer lamp emitting primarily at 222 nm with additional radiation up to 270 nm (USHIO, Cypress, CA, USA), a filtered KrCl* excimer lamp with a 200-230-nm bandpass filter preinstalled (USHIO, Cypress, CA, USA), a conventional LP mercury lamp emitting at 254 nm, and two benchtop UV LED systems with peak emission wavelengths of 270 and 282 nm (AquiSense Technologies, Earlanger, KY, USA).
UV exposure experiments were performed according to a standard protocol by Bolton and Linden (35). Virus samples (5 ml each) were made by diluting virus stocks 100-fold into sterile PBS (pH 7.4) and placed in 50- by 35-mm (diameter × height) sterile glass dishes (0.38 cm in depth) with a customized quartz lid (Corning 7980; Corning, NY, USA) (Fig. 1A) with UV transmittance (UVT) greater than 90% at 200 to 400 nm, according to the information provided by the manufacturer and confirmed by measuring the UV absorbance of the lid using a UV-visible (UV-Vis) spectrophotometer (DR-6000; Hach Company, Loveland, CO, USA). Absorbance for the samples was measured using a UV-Vis spectrophotometer. The UV incident irradiance at the center of sample surface was measured using a calibrated radiometer (ILT-2400; International Light Technologies, Inc., Peabody, MA, USA) set at the respective peak emission wavelength for a UVC device. (The wavelength 222 nm was used for the unfiltered KrCl* excimer.) The radiometer detector was placed directly under the quartz lid during irradiance measurements at the liquid surface to include any effects from absorption and reflection of the quartz plate. The UV exposure time for each sample was calculated using target UV fluences for unweighted emissions between 200 and 300 nm according to a protocol by Linden and Darby (36) and Bolton and Linden (35), where average UV fluence calculation included corrections for radiometer detector sensitivity correction across lamp emission spectra (i.e., lamp correction factor), sample absorbance from 200 to 300 nm, and path length (i.e., Beers law), divergence of light through the sample, reflection factor, and nonuniformity of incident irradiance across the sample surface (i.e., petri factor). Duplicate control samples (no UV exposure) were collected at the beginning and the end of UV exposure tests for each UVC device. Eight virus samples were exposed with five different UV fluences for each UVC device, in which three fluences were tested with duplicate samples and the other two fluences were tested with one sample. All virus samples after UV exposures were collected sacrificially, so no subsampling was performed in the UV exposure tests. The infectivities of virus samples without and after UV exposure were measured as described above, and the infectivity reduction in log10 scale was determined.
Statistical analysis.
The UV dose responses using different UVC devices were evaluated based on a pseudo-first-order inactivation kinetics model in log10 scale:
Log10 I is infectivity reduction in log10 scale, N0 and N are the virus sample infectivities before and after UV exposure, respectively, D is UV fluence in mJ/cm2, and k is the pseudo-first-order inactivation rate constant in cm2/mJ computed from a log10-scale kinetic model. The log10-scale inactivation rate constant was used, which made it easy to calculate the log inactivation from the rate constant.
The mean and standard error (SE) of inactivation rate constants were calculated using the “linear regression” function in OriginPro 2021 (with the intercept fixed at zero). Samples with infectivity equal to or less than the detection limits were excluded from the regression analyses. Analysis of covariance (ANCOVA) was used to determine if there was a statistically significant difference in the inactivation rate constants.
ACKNOWLEDGMENTS
Financial support for this work was provided by the National Science Foundation (grant CBET 2029695).
We declare no competing interests.
Contributor Information
Karl G. Linden, Email: karl.linden@colorado.edu.
Edward G. Dudley, The Pennsylvania State University
REFERENCES
- 1.World Health Organization. 2020. Transmission of SARS-CoV-2: implications for infection prevention precautions. World Health Organization, Geneva, Switzerland. [Google Scholar]
- 2.Pitol AK, Julian TR. 2021. Community transmission of SARS-CoV-2 by fomites: risks and risk reduction strategies. Environ Sci Technol Lett 8:263–269. 10.1021/acs.estlett.0c00966. [DOI] [PubMed] [Google Scholar]
- 3.van Doremalen N, Bushmaker T, Morris DH, Holbrook MG, Gamble A, Williamson BN, Tamin A, Harcourt JL, Thornburg NJ, Gerber SI, Lloyd-Smith JO, de Wit E, Munster VJ. 2020. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med 382:1564–1567. 10.1056/NEJMc2004973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Beck SE, Rodriguez RA, Linden KG, Hargy TM, Larason TC, Wright HB. 2014. Wavelength dependent UV inactivation and DNA damage of adenovirus as measured by cell culture infectivity and long range quantitative PCR. Environ Sci Technol 48:591–598. 10.1021/es403850b. [DOI] [PubMed] [Google Scholar]
- 5.Hull NM, Linden KG. 2018. Synergy of MS2 disinfection by sequential exposure to tailored UV wavelengths. Water Res 143:292–300. 10.1016/j.watres.2018.06.017. [DOI] [PubMed] [Google Scholar]
- 6.Linden KG, Hull N, Speight V. 2019. Thinking outside the treatment plant: UV for water distribution system disinfection. Acc Chem Res 52:1226–1233. 10.1021/acs.accounts.9b00060. [DOI] [PubMed] [Google Scholar]
- 7.Beck SE, Ryu H, Boczek LA, Cashdollar JL, Jeanis KM, Rosenblum JS, Lawal OR, Linden KG. 2017. Evaluating UV-C LED disinfection performance and investigating potential dual-wavelength synergy. Water Res 109:207–216. 10.1016/j.watres.2016.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kowalski W, Bahnfleth W, Hernandez M. 2009. A genomic model for predicting the ultraviolet susceptibility of viruses. IUVA News 11:15–28. [Google Scholar]
- 9.Tenkate TD. 1998. Ultraviolet radiation: human exposure and health risks. J Environ Health 61:9–15. [Google Scholar]
- 10.Zaffina S, Camisa V, Lembo M, Vinci MR, Tucci MG, Borra M, Napolitano A, Cannatã V. 2012. Accidental exposure to UV radiation produced by germicidal lamp: case report and risk assessment. Photochem Photobiol 88:1001–1004. 10.1111/j.1751-1097.2012.01151.x. [DOI] [PubMed] [Google Scholar]
- 11.Buonanno M, Ponnaiya B, Welch D, Stanislauskas M, Randers-Pehrson G, Smilenov L, Lowy FD, Owens DM, Brenner DJ. 2017. Germicidal efficacy and mammalian skin safety of 222-nm UV light. Radiat Res 187:483–491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Buonanno M, Welch D, Shuryak I, Brenner DJ. 2020. Far-UVC light (222 nm) efficiently and safely inactivates airborne human coronaviruses. Sci Rep 10:10285–10288. 10.1038/s41598-020-67211-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Narita K, Asano K, Morimoto Y, Igarashi T, Nakane A. 2018. Chronic irradiation with 222-nm UVC light induces neither DNA damage nor epidermal lesions in mouse skin, even at high doses. PLoS One 13:e0201259. 10.1371/journal.pone.0201259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kaidzu S, Sugihara K, Sasaki M, Nishiaki A, Ohashi H, Igarashi T, Tanito M. 2021. Re-evaluation of rat corneal damage by short-wavelength UV revealed extremely less hazardous property of far-UV-C. Photochem Photobiol 97:505–516. 10.1111/php.13419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Biasin M, Bianco A, Pareschi G, Cavalleri A, Cavatorta C, Fenizia C, Galli P, Lessio L, Lualdi M, Tombetti E, Ambrosi A, Redaelli EMA, Saulle I, Trabattoni D, Zanutta A, Clerici M. 2021. UV-C irradiation is highly effective in inactivating SARS-CoV-2 replication. Sci Rep 11:6260–6267. 10.1038/s41598-021-85425-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Patterson EI, Prince T, Anderson ER, Casas-Sanchez A, Smith SL, Cansado-Utrilla C, Solomon T, Griffiths MJ, Acosta-Serrano Á, Turtle L, Hughes GL. 2020. Methods of inactivation of SARS-CoV-2 for downstream biological assays. J Infect Dis 222:1462–1467. 10.1093/infdis/jiaa507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Storm N, McKay LGA, Downs SN, Johnson RI, Birru D, de Samber M, Willaert W, Cennini G, Griffiths A. 2020. Rapid and complete inactivation of SARS-CoV-2 by ultraviolet-C irradiation. Sci Rep 10:22421–22425. 10.1038/s41598-020-79600-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Heilingloh CS, Aufderhorst UW, Schipper L, Dittmer U, Witzke O, Yang D, Zheng X, Sutter K, Trilling M, Alt M, Steinmann E, Krawczyk A. 2020. Susceptibility of SARS-CoV-2 to UV irradiation. Am J Infect Control 48:1273–1275. 10.1016/j.ajic.2020.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Robinson RT, Mahfooz N, Rosas-Mejia O, Liu Y, Hull NM. 2021. SARS-CoV-2 disinfection in aqueous solution by UV 222 from a krypton chlorine excilamp. medRxiv 10.1101/2021.02.19.21252101. [DOI]
- 20.Kitagawa H, Nomura T, Nazmul T, Omori K, Shigemoto N, Sakaguchi T, Ohge H. 2021. Effectiveness of 222-nm ultraviolet light on disinfecting SARS-CoV-2 surface contamination. Am J Infect Control 49:299–301. 10.1016/j.ajic.2020.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kitagawa H, Nomura T, Nazmul T, Kawano R, Omori K, Shigemoto N, Sakaguchi T, Ohge H. 2021. Effect of intermittent irradiation and fluence-response of 222 nm ultraviolet light on SARS-CoV-2 contamination. Photodiagnosis Photodyn Ther 33:102184. 10.1016/j.pdpdt.2021.102184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ma B, Linden YS, Gundy PM, Gerba CP, Sobsey MD, Linden KG. 2021. Inactivation of coronaviruses and phage Phi6 from irradiation across UVC wavelengths. Environ Sci Technol Lett 8:425–430. 10.1021/acs.estlett.1c00178. [DOI] [PubMed] [Google Scholar]
- 23.Beck SE, Rodriguez RA, Hawkins MA, Hargy TM, Larason TC, Linden KG. 2015. Comparison of UV-induced inactivation and RNA damage in MS2 phage across the germicidal UV spectrum. Appl Environ Microbiol 82:1468–1474. 10.1128/AEM.02773-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.American Conference of Governmental Industrial Hygienists. 2021. 2021 threshold limit values (TLVs) and biological exposure indices (BEIs). American Conference of Governmental Industrial Hygienists, Washington, DC. [Google Scholar]
- 25.Chun-Chieh T, Li C-S. 2007. Inactivation of viruses on surfaces by ultraviolet germicidal irradiation. J Occup Environ Hyg 4:400–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Walker CM, Ko G. 2007. Effect of ultraviolet germicidal irradiation on viral aerosols. Environ Sci Technol 41:5460–5465. 10.1021/es070056u. [DOI] [PubMed] [Google Scholar]
- 27.Beck SE, Wright HB, Hargy TM, Larason TC, Linden KG. 2015. Action spectra for validation of pathogen disinfection in medium-pressure ultraviolet (UV) systems. Water Res 70:27–37. 10.1016/j.watres.2014.11.028. [DOI] [PubMed] [Google Scholar]
- 28.Beck SE, Hull NM, Poepping C, Linden KG. 2018. Wavelength-dependent damage to adenoviral proteins across the germicidal UV spectrum. Environ Sci Technol 52:223–229. 10.1021/acs.est.7b04602. [DOI] [PubMed] [Google Scholar]
- 29.Gerchman Y, Mamane H, Friedman N, Mandelboim M. 2020. UV-LED disinfection of coronavirus: wavelength effect. J Photochem Photobiol B Biol 212:112044. 10.1016/j.jphotobiol.2020.112044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kim K, Jothikumar N, Sen A, Murphy JL, Chellam S. 2021. Removal and inactivation of an enveloped virus surrogate by iron conventional coagulation and electrocoagulation. Environ Sci Technol 55:2674–2683. 10.1021/acs.est.0c07697. [DOI] [PubMed] [Google Scholar]
- 31.Silverman AI, Boehm AB. 2020. Systematic review and meta-analysis of the persistence and disinfection of human coronaviruses and their viral surrogates in water and wastewater. Environ Sci Technol Lett 7:544–553. 10.1021/acs.estlett.0c00313. [DOI] [PubMed] [Google Scholar]
- 32.Aquino De Carvalho N, Stachler EN, Cimabue N, Bibby K. 2017. Evaluation of Phi6 persistence and suitability as an enveloped virus surrogate. Environ Sci Technol 51:8692–8700. 10.1021/acs.est.7b01296. [DOI] [PubMed] [Google Scholar]
- 33.Payment P, Trudel M (ed). 1994. Methods and techniques in virology. Marcel Dekker, Inc, New York, NY. [Google Scholar]
- 34.Ramakrishnan MA. 2016. Determination of 50% endpoint titer using a simple formula. World J Virol 5:85–86. 10.5501/wjv.v5.i2.85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bolton JR, Linden KG. 2003. Standardization of methods for fluence (UV dose) determination in bench-scale UV experiments. J Environ Eng 129:209–215. 10.1061/(ASCE)0733-9372(2003)129:3(209). [DOI] [Google Scholar]
- 36.Linden KG, Darby JL. 1997. Estimating effective germicidal dose from medium pressure UV lamps. J Environ Eng 123:1142–1149. 10.1061/(ASCE)0733-9372(1997)123:11(1142). [DOI] [Google Scholar]