Abstract
Severe aortic stenosis patients with bicuspid anatomy have been excluded from the major transcatheter aortic valve replacement (TAVI) randomized clinical trials. As a result, there is no official recommendation on bicuspid TAVI. A panel of bicuspid experts was created to fill this gap. In this consensus statement, an algorithm is proposed to guide the choice of surgery or TAVI within this complex patient population, depending on aortic dilatation, age, surgical risk score, and anatomy. A step-by-step guide for sizing and positioning of the SAPIEN 3/Ultra TAVI bioprostheses is presented. Annular sizing remains the primary strategy in most bicuspid patients. However, some anatomies may require sizing at the supra-annular level, for which patients the panel recommends the circle method, a dedicated sizing and positioning approach for SAPIEN 3/Ultra. The consensus provides valuable pre-operative insights on the interactions between SAPIEN 3/Ultra and the bicuspid anatomy; understanding the valve–anatomy relationship is critical to avoid complications and to optimize outcomes for patients.
Keywords: Bicuspid aortic valve, TAVI, Bicuspid sizing, Bicuspid positioning, SAPIEN 3/Ultra
Key Summary Points
| Bicuspid aortic valve (BAV) patients were excluded from the major transcatheter aortic valve implantation (TAVI) trials. Consequently, there is no official recommendation for their management. |
| In the absence of dilated aorta (main indication for surgery), the Heart Team should consider the patient’s age (< 65 years, 65–80 years, > 80 years), anatomy (raphe, amount and location of calcifications), and ultimately surgical risk score to choose the best option for the patient: surgical aortic valve replacement or TAVI. |
| Annular sizing remains the primary strategy in most BAV patients. Some anatomies may require sizing at the supra-annular level: in these cases, the circle method is recommended when using SAPIEN 3/Ultra. |
| The circle method uses CT scans to project circles at 0, 3, 6, and 9 mm from the annulus to interrogate the anatomy for sizing, positioning, and predicting potential complications; these circles are equal in diameter to SAPIEN 3/Ultra. |
| Whereas conventional positioning of SAPIEN 3/Ultra leads to a final implant depth between 80%/20% and 90%/10% (aortic/ventricular), modified positioning in BAV (calcified raphe, severe leaflet calcification, smaller supra-annular space) leads to target a final position between 90%/10% and 100%/0%. |
Digital Features
This article is published with digital features, including a summary slide, to facilitate understanding of the article. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.14588565.
Introduction
Bicuspid aortic valve (BAV) is a common congenital abnormality, with a prevalence of 1–2%. It is a highly heterogeneous condition, with variable presentation, but is characterized by a risk of progressive aortic valve disease, predominantly aortic stenosis, and of progressive aortic dilatation with associated risk of aortic dissection or rupture. Although published data vary considerably, lifetime incidence of aortic valve surgery is approximately 25–50%, aortic surgery (for dilatation) 5–10%, and risk of aortic dissection 0.5–1.0%. Aortic stenosis tends to present at an earlier age, and in patients with lower surgical risk, than in the tricuspid population.
Patients with bicuspid aortic valve have been excluded from the major transcatheter aortic valve implantation (TAVI) trials, and therefore there are no official recommendations on management. Although multiple transcatheter aortic valve technologies, both self-expanding and balloon-expandable, have been reported in BAV, this document is focused on the use of the balloon-expandable Edwards SAPIEN 3/Ultra valve. A Bicuspid Expert Panel of interventional cardiologists and cardiac surgeons was established to design a step-by-step guide for the treatment of BAV with the SAPIEN 3/Ultra. A decision tree was defined outlining the BAV patients most suited for surgical aortic valve replacement (SAVR) or transcatheter aortic valve implantation (TAVI). Experts agreed on a methodology for optimal sizing and positioning of SAPIEN 3/Ultra in BAV. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Decision Tree: SAVR or TAVI?
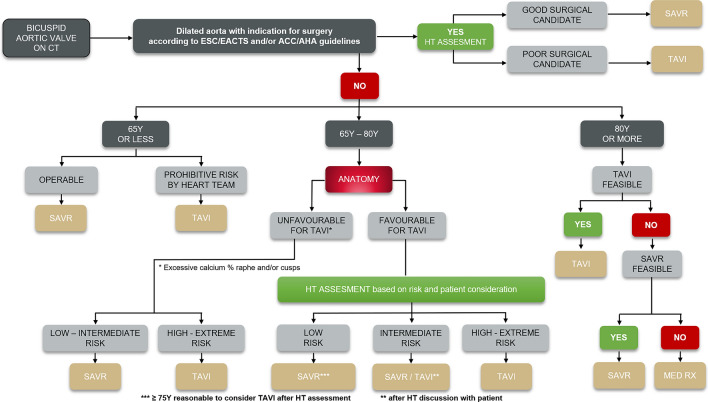
The consensus was reached on an algorithm defining the BAV patients most suited for SAVR or TAVI (Fig. 1). Throughout the decision tree, the Heart Team has a central role in assessing operative risk, while patient age and preference should also be considered.
Fig. 1.
Decision tree: SAVR or TAVI for bicuspid severe aortic stenosis patients? This algorithm was created based on the consensus reached by the Bicuspid Expert Panel, including both interventional cardiologists and cardiac surgeons. CT computed tomography, HT Heart Team, SAVR surgical aortic valve replacement, TAVI transcatheter aortic valve replacement, MED RX standard medical therapy
In addition to routine echocardiographic assessment, all patients with suspected bicuspid anatomy should undergo a computed tomography (CT) scan to confirm the diagnosis and guide treatment. BAV patients are prone to dilatation of the ascending aorta, which may be an indication for surgery [1]. Thus, the first step is to classify patients by the presence or absence of aortic dilatation, according to definitions used in the European Society of Cardiology (ESC)/European Association for Cardio-Thoracic Surgery (EACTS) and American College of Cardiology (ACC)/American Heart Association (AHA) guidelines [2, 3].
Dilated Aorta
In patients with a dilated aorta, surgery enables repair/replacement of the aorta and the valve, reducing future risk of aortic rupture or dissection. Surgery is therefore recommended in those patients with a dilated aorta with indication for surgery according to ESC/EACTS and/or ACC/AHA guidelines. The Heart Team should assess patient suitability for surgery, taking into account age, EuroSCORE/Society of Thoracic Surgeons (STS) score, frailty, and comorbidities. Patients deemed good surgical candidates by the Heart Team should undergo SAVR. Poor surgical candidates should be offered TAVI, with no intervention to the ascending aorta.
Aorta Not Dilated
In the absence of aortic dilatation, the next gating point is age. While recognizing that any recommended age cut-off is inevitably arbitrary, and that each patient should be assessed on an individual basis by the Heart Team, it is broadly recommended that patients be divided into three categories: < 65 years, 65–80 years, and > 80 years.
< 65 years
Surgery is recommended in younger patients with BAV because of the greater evidence base on long-term durability of bioprostheses, as well as the option of a mechanical valve, which offers the prospect of life-long valve durability. Age under 65 years was selected as the cut off consistent with existing international guidelines which recommend: (1) that SAVR is favored over TAVI for the treatment of all patients with severe AS in this age group, and (2) that a mechanical valve may be considered below the age of 65 [4]. In this age category, patients deemed good surgical candidates by the Heart Team should undergo SAVR, while those considered poor surgical candidates should be selected for TAVI. Selection of both surgical and transcatheter heart valves for patients in this age group should also take into account the likely need for at least one further valve intervention in their lifetime.
-
b.
65–80 years
In patients aged 65–80 years, the decision for TAVI or SAVR depends on anatomy and surgical risk. Assessment of surgical risk by the Heart Team will include EuroSCORE/STS score, frailty, and comorbidities. Primary anatomical considerations include the presence, extent, and location of calcification on the leaflets and raphe, the risk of coronary occlusion by calcium, severe angulation of the aortic root and proximal ascending aorta, and fragile tissue, which carries a risk of dissection. Heart Teams should also consider the experience of their center and operators in the treatment of bicuspid anatomy.
Unfavorable anatomy for TAVI Patients with excessive calcium in the raphe and/or the cusps may be deemed unfavorable candidates for TAVI due to an increased risk of stroke, paravalvular (PV) leak, and pacemaker requirement. Factors which increase procedural risk in both tricuspid and bicuspid anatomy, including severe left ventricular outflow tract (LVOT) and annular calcification, should also be considered [5]. In patients with unfavorable anatomy and classified as low to intermediate surgical risk, SAVR is recommended. In those at high to extreme surgical risk, TAVI may be the preferred option.
Favorable anatomy for TAVI In patients with favorable anatomy for TAVI, without those adverse factors described above, choice of treatment should again be determined by age, and by Heart Team assessment of surgical risk. Patient’s preference should also be taken into account. In patients at low surgical risk, SAVR is advised in patients below 75 years old, with TAVI being recommended for those aged 75 or older.
In those at intermediate surgical risk, both SAVR and TAVI can be considered by the Heart Team in discussion with the patient, recognizing that there are no studies evaluating the relative safety and efficacy of TAVI and SAVR in BAV patients at intermediate risk.
In patients categorized as high or extreme surgical risk, TAVI is recommended.
-
c.
80 years or above
TAVI is the recommended treatment for all patients aged 80 or older, unless anatomical or other factors mean that TAVI is not feasible. If TAVI is not possible, SAVR should be considered. Inoperable patients should receive medical therapy only, with aortic balloon valvuloplasty if considered appropriate.
Sizing the BAV
There is no internationally agreed sizing methodology for TAVI in BAV. A number of BAV classifications have been proposed to differentiate the anatomy, predict complications, and define annulus/orifice mismatch. Sievers and Schmidtke used the number of raphes to differentiate three major anatomical types: type 0 (no raphe), type 1 (one raphe), and type 2 (two raphes), followed by two supplementary characteristics, namely spatial position of cusps or raphes, and functional status of the valve [6]. It remains the most frequently used classification. Makkar and Yoon identified calcified raphe and excess leaflet calcification as independent predictors of procedural complications (e.g., aortic root injury, moderate-to-severe PV regurgitation) and mortality at 30 days and two years [7]. The Bicuspid Aortic Valve Anatomy and Relationship with Devices (BAVARD) registry used size of the aortic annulus and intercommissural distance (ICD) to predict sealing, position, and device size [8]. Petronio et al. described an algorithm to decide sizing in bicuspid anatomy by using a combination of raphe length, raphe calcium, and overall calcium to modify annular sizing, which was coined the Calcium Algorithm Sizing for bicusPid Evaluation with Raphe (CASPER) [9].
CT Scan for Sizing in Bicuspid Anatomy
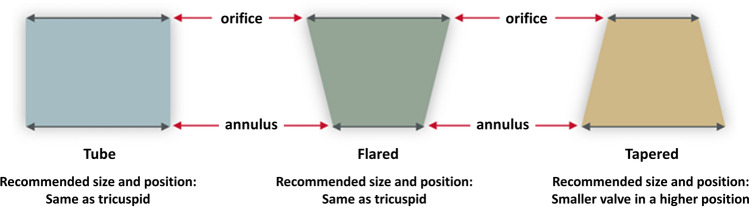
A high-quality CT scan is mandatory for assessment of valve structure and to guide sizing. Key factors in BAV include leaflet configuration, the location and extent of calcium, and the landing zone configuration (tubular, flared, or tapered, Fig. 2). Both annular and supra-annular dimensions may be used to select the size of SAPIEN 3/Ultra in BAV, and both should be assessed.
Fig. 2.
Aortic root configurations. The orifice is the perimeter created by the free edge of the leaflets, whereas the annulus is the virtual ring formed by linking the basal attachments of the aortic leaflets. As defined in the BAVARD Registry [8], bicuspid landing zone may present with three different configurations: tubular (both annulus and orifice have the same size), flared (annulus smaller than the orifice), or tapered (annulus larger than the orifice)
Calcium Assessment
Extensive calcification is common in bicuspid valves, and both the amount and location of calcium are important [7]. Calcium volume should be assessed with non-contrast CT. The location of calcium predicts complications: calcification of the raphe(s) affects device expansion, and increases the risks of regurgitation, misplacement, and pacemaker requirement; calcium at the commissures may cause perforation; calcium at the tip of a leaflet risks coronary obstruction (although in most BAV patients spacious sinuses often protect against coronary occlusion). In addition, severe calcification in BAV with a tapered configuration confers a risk of Valsalva rupture [10].
Modified Sizing in Bicuspid Anatomy
Bicuspid valve leaflets may not open in the same way as in tricuspid anatomy during deployment of the SAPIEN valve. In general, a smaller and more asymmetrical, or less circular, orifice will be created. As a result, conventional sizing according to annular measurements may result in over-sizing at the level of the leaflets. This is likely to lead to even more marked asymmetry and may also increase the risk of trauma related to calcium distribution in the aortic valvular complex.
These properties of some bicuspid valves may also allow effective anchoring and sealing of the transcatheter heart valve (THV) in the cusps, i.e., at the supra-annular level in tapered configuration, allowing the implanting physician to consider sizing according to the supra-annular space, rather than purely based on annular measurements.
Anatomical Factors Influencing Sizing in Bicuspid Anatomy
It is recommended that implanting physicians consider a number of specific anatomical variables when contemplating whether and how to modify sizing in individual patients.
Calcification of the raphe If the raphe is severely calcified, valve expansion is likely to be reduced or the THV may end up malpositioned, and relative under-sizing may be considered, relative to the aortic annulus.
Calcification of the cusps Severe calcification of the cusps will enhance anchoring in the supra-annular space and may favor relative under-sizing.
Valve configuration (Fig. 2) In patients with tapered, as opposed to tubular or flared, valve configuration, i.e., where the supra-annular space is smaller, relative under-sizing may be considered.
Large annuli A large annular area above the upper limit of the SAPIEN 3 sizing chart is more frequently encountered in BAV. The possibility of sizing to the supra-annular space to allow TAVI to be performed in these cases should be considered.
If the anatomy, based on the variables described above, indicates that sizing to the supra-annular space may be appropriate, then a robust method for sizing is required. Measurement of the inter-commissural distance (ICD) has been advocated but is considered by the panel to be insufficient. Instead, use of the circle method for supra-annular sizing is advocated in these cases.
Circle Method for Supra-Annular Sizing and Positioning
Evaluation of the supra-annular space is done by scrolling the CT images in the axial view to identify different bicuspid configurations (tube, flared, and tapered [8]) and the location of calcium that could affect valve deployment (size, sealing, circularity) and lead to complications.
The circle technique determines a projection of an ellipse circle equal in diameter to the SAPIEN valve platform.
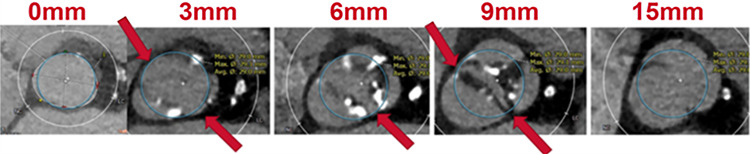
A projected circle of the identical diameter to the proposed SAPIEN valve is placed at 3-mm increments beginning at the annular plane, up to the sinotubular junction (STJ, Fig. 3). Each circle demonstrates the valve-to-anatomy interaction. The SAPIEN 3/Ultra transcatheter heart valve (THV) has shown a good index of circularity even in bicuspid valve [8]. For this reason, projecting a circle through the aortic complex is the ideal method, both for sizing and to evaluate the best placement.
Fig. 3.
The circle method. SAPIEN 3 visual simulation to provide pre-operative insights into the interaction between SAPIEN 3 and bicuspid anatomy. Circles are drawn at 0–3-6–9 mm from the annulus to understand sealing zone at the commissures to determine size and position. Coronary take-off and above levels are checked for left main/right coronary artery clearance
In patients where the anatomy supports supra-annular sizing, a circle, which is one size smaller than the annulus size, should be projected in addition to the circle that is equivalent to the conventional sizing method.
Circles projected at 3, 6, and 9 mm simulate the apposition of the SAPIEN valve and skirt’s height (Table 1) to the leaflets and commissures. Interrogation of the anatomy at the level of the SAPIEN’s outer sealing skirt is an important assessment for anchoring and sealing. Three situations can be encountered:
The circle is too large it extends beyond the commissures, with a potential risk of commissure rupture.
If the circle is large enough to touch the commissures, then sealing is expected.
If the circle is undersized, and does not touch the commissures, there is a risk of PVL or valve embolization.
Table 1.
SAPIEN nominal deployment: expanded and outer sealing skirt heights
| SAPIEN THV SIZE | 20 mm | 23 mm | 26 mm | 29 mm |
|---|---|---|---|---|
| Expanded height | 15.5 mm | 18 mm | 20 mm | 22.5 mm |
|
Outer sealing skirt height SAPIEN 3 THV |
5.2 mm | 6.6 mm | 7.0 mm | 8.1 mm |
|
Outer sealing skirt height SAPIEN 3 Ultra THVa |
7.3 mm | 9.0 mm | 9.7 mm | N/A |
aRounded to the nearest 0.5 mm
The circle technique can also determine how the deployed SAPIEN valve will interact with calcification in the leaflets and/or raphe, and consequently predict valve expansion, and the risk of complications including rupture and coronary obstruction.
-
d.
Valve expansion If there is a bulky calcium on one cusp, the SAPIEN valve will shift away from this cusp during balloon expansion. The projected circle should be positioned accordingly to simulate this effect.
-
e.
Risk of rupture If there is severe calcification of the raphe between the right coronary cusp (RCC) and non-coronary cusp (NCC) in contact with the right ventricle (RV) in a shallow sinus, balloon-expansion of the valve risks pushing the calcified raphe into the RV, causing a ventricular septal defect (VSD). The circle method can predict this and suggest under-sizing or an alternative approach.
-
f.
Coronary obstruction Circles projected at the coronary ostia, above coronary take-off, and up to the STJ, with assessment of leaflet height relative to the coronary ostia, allow prediction of the risk of coronary occlusion as well as sinus sequestration that may affect future THV-in-THV.
Advantages of the circle method
Helps implanters understand the sealing zone at the commissures, enabling them to determine the size and position of the SAPIEN 3/Ultra valves.
Provides pre-operative insights into the interaction between SAPIEN 3/Ultra and the bicuspid anatomy, predicting sealing, anchoring, and the risk of complications such as coronary obstruction and rupture. This completes the ICD method, which only indicates the valve size.
Provides visual reassurance of the size and position—e.g., when downsizing.
May allow treatment of patients with large annuli above the usual recommended upper size limit.
Illustrates the optimal implantation height to achieve supra-annular sealing.
Fast and reproducible.
Easy to teach new implanters.
Disadvantages of the circle method
Qualitative method with no strict thresholds on how to size the valve. Operators must look at the circle and match it to the anatomy.
Balloon Pre-dilatation and Sizing
Balloon pre-dilatation is recommended to facilitate crossing and deployment in heavily calcified bicuspid valves (e.g., calcium score > 1000 HU), when fusion is between the non-coronary and right coronary cusps, and when the guide wire has not reached the greater curve of the ascending aorta.
Balloon pre-dilatation may also be used as an adjunct to sizing, in particular when sizing is borderline, and when sizing to the supra-annular space, with a high target position, is considered.
Pre-dilatation should be performed with a semi-compliant balloon, sized to match the smaller valve size under consideration (i.e., 23 mm if in doubt between 23/26 valve, 26 mm between 26/29 valve). Valvuloplasty with a simultaneous aortogram will show: (1) no leak around the balloon matching the smaller valve size; (2) the anatomy gripping the balloon confirming it will also grip and anchor the valve; (3) the level of the waist, indicating the location of the best sealing when the valve is deployed. Moreover, leaflets’, and attached calcium, movement towards the coronary ostia can be assessed during the aortogram.
Borderline Cases
When conventional annular measurements indicate borderline sizing between two valves, the smaller valve size should be selected. Oversizing must be avoided, particularly in calcified BAV, to prevent complications [11, 12]. Balloon sizing may be used to confirm the smaller valve size.
Overall Approach to Sizing in Bicuspid Anatomy
Although there are a number of factors which would argue for a modified approach to sizing in bicuspid anatomy, data to support this are limited. The BAVARD registry indicated that when implanting physicians incorporated other variables, sizing remained broadly the same as if it had been based on a conventional annular sizing approach, and outcomes were good. It also demonstrated that in 13% of the BAV, the undersized self-expandable or balloon-expandable valve showed the same results in long-term follow-up.
The panel therefore recommends that sizing in bicuspid anatomy should continue to be based primarily on measurement of the annulus, with sizing according to annulus area. However, relative under-sizing, targeting supra-annular sizing and deployment, may be considered, particularly in borderline cases. In these cases, the circle method is recommended. Table 2 details specific concerns in BAV and how to mitigate complications.
Table 2.
Specific concerns checklist for bicuspid valve when using balloon-expandable valve
| Scenario | Potential risks | What to check before proceeding | How to mitigate complications |
|---|---|---|---|
| Difficult coplanar view in BAV type 0 | Malposition | CT coplanar view | 2 pig tails (1 in each cusp) |
| Calcium in the raphe | Creates malposition (THV not centered in the annulus or implanted lower in the LVOT), SOV perforation/VSD | CT calcium location in axial view to identify excessive calcified raphe |
Screening out patient Undersize the valve is possible |
| Hook of calcium in the RCC | Fistula, VSD | ||
| Calcium in the center of the annulus | Malposition in the LVOT and PVL | CT evaluation of the movement of the leaflet with the raphe | Starting implant position very high |
| Long fibrotic leaflets | LMCA occlusion | 1-CT evaluation of distance between leaflets and STJ and coronary artery |
1-protection with wire, balloon, stent in the coronary 2-Valvuloplasty testing |
CT computed tomography, LMCA left main coronary artery, LVOT left ventricular outflow tract, PVL paravalvular leak, RCC right coronary cusp, SOV sinus of Valsalva, STJ sinotubular junction, VSD ventricular septal defect
Positioning of SAPIEN 3/Ultra
Conventional Positioning
For most bicuspid valves, the SAPIEN 3/Ultra valve should be implanted using the same strategy for sizing and positioning as implemented for tricuspid valves. The target final implant depth in these cases is between 80/20 and 90%/10% aortic/ventricular. The panel recommends this exact same approach is used in the majority of bicuspid cases, in which sizing will be based on the annulus, with anchoring and sealing at the annular level, and positioning based on the annulus, just as it is for tricuspid anatomy.
Conventional Positioning: Step-by-Step
Positioning of the THV utilizes the 3-mm Center Marker, a radio-opaque marker found on the balloon catheter of the Commander Delivery system. The conventional approach is to position the bottom of the Center Marker at the base of the cusps or slightly above [13].
Foreshortening of the SAPIEN 3/Ultra valve is expected, and the total amount of foreshortening is dependent on the annulus size and oversizing applied (Table 3).
Table 3.
SAPIEN foreshortening according to THV’s size
| SAPIEN THV SIZE | 20 mm | 23 mm | 26 mm | 29 mm |
|---|---|---|---|---|
| Foreshortening | 5.5 mm | 6.5 mm | 7 mm | 8.5 mm |
The target final deployment using this method is between 80/20 and 90%/10% aortic/ventricular.
Modified Positioning in Bicuspid Anatomy
In those bicuspid cases where supra-annular sealing and anchoring (rather than annular) is intended, a modified positioning strategy should be employed.
As we have already outlined in the sizing recommendations above, this would include patients with calcified raphe, severe leaflet calcification, a smaller supra-annular space (‘tapered’ shape), and those patients with very large annuli above the recommended range in whom a supra-annular sealing strategy is thought to be feasible.
In these cases, the final target implant depth will be between 90/10 and 100/0. The panel does not recommend positioning above the annulus, even when the anatomy indicates that anchoring and sealing in the leaflets may be possible. Figure 1 outlines the recommended position for different underlying morphologies. Figure 4 shows how to position the valve before opening, Table 4 outlines tricky scenarios to watch out for when positioning SAPIEN 3/Ultra in BAV and what to do in each case.
Fig. 4.
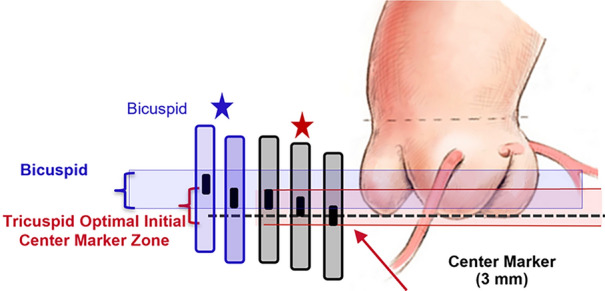
How to position the valve before opening
Table 4.
Tricky scenarios for positioning SAPIEN 3/Ultra in BAV
| Scenario | Potential result |
|---|---|
| Heavy localized calcification in the annulus/LVOT | Foreshortening in the LVOT may not occur as expected, and the position may end up too low |
| Heavy calcification above the annulus | Position can end up too high |
| Annulus bigger than 683 mm2 | Migration embolization in the LVOT |
BAV bicuspid aortic valve, LVOT left ventricular outflow tract
It should be noted that higher implantation of SAPIEN 3 in all anatomies has been shown to reduce conduction abnormalities and permanent pacemaker requirement after TAVI [14, 15].
Modified Positioning: Step-by-Step
Two techniques can be employed for modified positioning in bicuspid anatomy with a higher final target position:
Position of the center marker 3 mm above the coplanar view
This is a modification of the standard technique and has also been used in tricuspid anatomy to minimize contact with LVOT and reduce the incidence of permanent pacemaker. The bottom of the center marker is positioned at least 3 mm above the annulus (bottom of the cusps) in the coplanar projection. The final position will be 100/0.
-
2.
Radiolucent line at coplanar view
The radiotransparent line, which is visible on fluoroscopy, at the inflow of the crimped SAPIEN 3/Ultra, represents the separation of the last stent cell. If the crimped THV is perpendicular to the coplanar view, the line can be used for the implant, leading to a final position of 100/0 aortic/ventricular.
Conclusions
The Heart Team remains fundamental to select the appropriate treatment for bicuspid aortic valves patients. Bicuspid patients are a heterogenous population with significant variation in particular in configuration of the root, and in the extent and location of calcification.
When contemplating TAVI, conventional sizing and positioning remain the most appropriate strategy in the majority of bicuspid patients. However, in some anatomies supra-annular sizing, utilizing the circle method, combined with a higher final target position, is recommended. Further research is needed to confirm the optimal method for the treatment of bicuspid anatomy with the SAPIEN 3/Ultra valve. This expert consensus statement should assist TAVI operators in optimizing outcomes for their patients based on current knowledge.
Acknowledgements
Funding
The journal’s Rapid Service Fee was supported by Edwards Lifesciences.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All authors contributed equally. In addition, Dr. Daniel Blackman designed the structure of the article, and proofread the manuscript.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Disclosures
Daniel Blackman, Davide Gabbieri, Bruno García Del Blanco, Jörg Kempfert, Mika Laine, Julia Mascherbauer, Radoslaw Parma and Didier Tchétché were part of an advisory panel at Edwards for which they got paid. Furthermore, Davide Gabbieri, Jörg Kempfert, and Radoslaw Parma are proctors for Edwards Lifesciences.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Contributor Information
Daniel Blackman, Email: daniel.blackman1@nhs.net.
Davide Gabbieri, Email: dgabbieri@yahoo.it.
Bruno García Del Blanco, Email: brunogb51@gmail.com.
Jörg Kempfert, Email: kempfert@dhzb.de.
Mika Laine, Email: mika.laine@hus.fi.
Julia Mascherbauer, Email: julia.mascherbauer@meduniwien.ac.at.
Radoslaw Parma, Email: radoslaw.parma@gmail.com.
Didier Tchétché, Email: dtchetche@clinique-pasteur.com.
References
- 1.Baumgartner H, De Backer J. The ESC clinical practice guidelines for the management of adult congenital heart disease 2020. Eur Heart J. 2020;41(43):4153–4154. doi: 10.1093/eurheartj/ehaa701. [DOI] [PubMed] [Google Scholar]
- 2.Baumgartner H, Falk V, Bax JJ, De Bonis M, Hamm C, Holm PJ, Iung B, Lancellotti P, Lansac E, Rodriguez Munoz D, Rosenhek R, Sjogren J, Tornos Mas P, Vahanian A, Walther T, Wendler O, Windecker S, Zamorano JL, Group ESCSD 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2017;38(36):2739–2791. doi: 10.1093/eurheartj/ehx391. [DOI] [PubMed] [Google Scholar]
- 3.Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP, 3rd, Gentile F, Jneid H, Krieger EV, Mack M, McLeod C, O'Gara PT, Rigolin VH, Sundt TM, III, Thompson A, Toly C. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2020;2:3. doi: 10.1161/CIR.0000000000000923. [DOI] [PubMed] [Google Scholar]
- 4.Head SJ, Celik M, Kappetein AP. Mechanical versus bioprosthetic aortic valve replacement. Eur Heart J. 2017;38(28):2183–2191. doi: 10.1093/eurheartj/ehx141. [DOI] [PubMed] [Google Scholar]
- 5.Pollari F, Hitzl W, Vogt F, Cuomo M, Schwab J, Sohn C, Kalisnik JM, Langhammer C, Bertsch T, Fischlein T, Pfeiffer S. Aortic valve calcification as a risk factor for major complications and reduced survival after transcatheter replacement. J Cardiovasc Comput Tomogr. 2020;14(4):307–313. doi: 10.1016/j.jcct.2019.12.001. [DOI] [PubMed] [Google Scholar]
- 6.Sievers HH, Schmidtke C. A classification system for the bicuspid aortic valve from 304 surgical specimens. J Thorac Cardiovasc Surg. 2007;133(5):1226–1233. doi: 10.1016/j.jtcvs.2007.01.039. [DOI] [PubMed] [Google Scholar]
- 7.Yoon SH, Kim WK, Dhoble A, Milhorini Pio S, Babaliaros V, Jilaihawi H, Pilgrim T, De Backer O, Bleiziffer S, Vincent F, Shmidt T, Butter C, Kamioka N, Eschenbach L, Renker M, Asami M, Lazkani M, Fujita B, Birs A, Barbanti M, Pershad A, Landes U, Oldemeyer B, Kitamura M, Oakley L, Ochiai T, Chakravarty T, Nakamura M, Ruile P, Deuschl F, Berman D, Modine T, Ensminger S, Kornowski R, Lange R, McCabe JM, Williams MR, Whisenant B, Delgado V, Windecker S, Van Belle E, Sondergaard L, Chevalier B, Mack M, Bax JJ, Leon MB, Makkar RR, Bicuspid Aortic Valve Stenosis Transcatheter Aortic Valve Replacement Registry I Bicuspid aortic valve morphology and outcomes after transcatheter aortic valve replacement. J Am Coll Cardiol. 2020;76(9):1018–1030. doi: 10.1016/j.jacc.2020.07.005. [DOI] [PubMed] [Google Scholar]
- 8.Tchetche D, de Biase C, van Gils L, Parma R, Ochala A, Lefevre T, Hovasse T, De Backer O, Sondergaard L, Bleiziffer S, Lange R, Kornowski R, Landes U, Norgaard BL, Biasco L, Philippart R, Molina-Martin de Nicolas J, Mylotte D, Lemee C, Dumonteil N, Van Mieghem NM. Bicuspid aortic valve anatomy and relationship with devices: the BAVARD Multicenter Registry. Circ Cardiovasc Interv. 2019;12(1):e007107. doi: 10.1161/CIRCINTERVENTIONS.118.007107. [DOI] [PubMed] [Google Scholar]
- 9.Petronio AS, Angelillis M, De Backer O, Giannini C, Costa G, Fiorina C, Castriota F, Bedogni F, Laborde JC, Sondergaard L. Bicuspid aortic valve sizing for transcatheter aortic valve implantation: development and validation of an algorithm based on multi-slice computed tomography. J Cardiovasc Comput Tomogr. 2020;14(5):452–461. doi: 10.1016/j.jcct.2020.01.007. [DOI] [PubMed] [Google Scholar]
- 10.Milhorini Pio S, Bax J, Delgado V. How valvular calcification can affect the outcomes of transcatheter aortic valve implantation. Expert Rev Med Devices. 2020;17(8):773–784. doi: 10.1080/17434440.2020.1789456. [DOI] [PubMed] [Google Scholar]
- 11.Arai T, Lefevre T, Hovasse T, Morice MC, Romano M, Benamer H, Garot P, Hayashida K, Bouvier E, Chevalier B. The feasibility of transcatheter aortic valve implantation using the Edwards SAPIEN 3 for patients with severe bicuspid aortic stenosis. J Cardiol. 2017;70(3):220–224. doi: 10.1016/j.jjcc.2016.12.009. [DOI] [PubMed] [Google Scholar]
- 12.Xiong TY, Li YJ, Feng Y, Liao YB, Zhao ZG, Mylotte D, Wei X, Xu YN, Peng Y, Wei JF, Zheng MX, Zhou X, Meng W, Piazza N, Chen M. Understanding the interaction between transcatheter aortic valve prostheses and supra-annular structures from post-implant stent geometry. JACC Cardiovasc Interv. 2019;12(12):1164–1171. doi: 10.1016/j.jcin.2019.02.051. [DOI] [PubMed] [Google Scholar]
- 13.Dvir D, Walther T, Wood DA, Santos MS, Cocchieri R, Baan J, Kim W, Webb J. TCT-718 large multicenter assessment of SAPIEN 3 transcatheter aortic valve implantation: optimization of clinical outcomes with precise device positioning. J Am Coll Cardiol. 2014;64(11):B211. doi: 10.1016/j.jacc.2014.07.790. [DOI] [Google Scholar]
- 14.Schwerg M, Fulde F, Dreger H, Poller WC, Stangl K, Laule M. Optimized implantation height of the Edwards SAPIEN 3 valve to minimize pacemaker implantation after TAVI. J Interv Cardiol. 2016;29(4):370–374. doi: 10.1111/joic.12302. [DOI] [PubMed] [Google Scholar]
- 15.Sammour Y, Banerjee K, Kumar A, Lak H, Chawla S, Incognito C, Patel J, Kaur M, Abdelfattah O, Svensson LG, Tuzcu EM, Reed GW, Puri R, Yun J, Krishnaswamy A, Kapadia S. Systematic approach to high implantation of SAPIEN-3 valve achieves a lower rate of conduction abnormalities including pacemaker implantation. Circ Cardiovasc Interv. 2021;14(1):e009407. doi: 10.1161/CIRCINTERVENTIONS.120.009407. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.