Abstract
An increasing evidence suggests that vanadium compounds are novel potential drugs in the treatment of diabetes, atherosclerosis, and cancer. Vanadium has also demonstrated activities against RNA viruses and is a promising candidate for treating acute respiratory diseases. The antidiabetic, antihypertensive, lipid-lowering, cardioprotective, antineoplastic, antiviral, and other potential effects of vanadium are summarized here. Given the beneficial antihyperglycemic and antiinflammatory effects as well as the potential mechanistic link between the COVID-19 and diabetes, vanadium compounds could be considered as a complement to the prescribed treatment of COVID-19. Thus, further clinical trials are warranted to confirm these favorable effects of vanadium treatment in COVID-19 patients, which appear not to be studied yet.
Abbreviations: ACE2, angiotensin-converting enzyme; BEOV, bis(ethylmaltolato)oxovanadium(IV); BK, bradykinin; B1R, BK type 1 receptors; B2R, BK type 2 receptors; BMI, body mass index; BMOV, bis(maltolato)oxovanadium(IV); COVID-19, Coronavirus disease 2019; COX-2, cyclooxygenase-2; CREB, cyclic AMP response binding element; DPP-IV, dipeptidyl peptidase IV; EGFR, epidermal growth factor receptor; ERK, extracellular signal-regulated kinase; FFA, free fatty acid; GLUT4, glucose transporter 4; GS, glycogen synthase; HbA1c, hemoglobin A1c; HDL-C, high-density lipoprotein cholesterol; HIV, human immunodeficiency virus; HOMA-IR, homeostasis model assessment-insulin resistance; IBV, infectious bronchitis virus; IL-1, interleukin-1; IL-4, interleukin 4; IL-6, interleukin-6; IFN-I, type I interferons; IFN-γ, interferon-γ; JAKs, Janus kinases; MAPK, mitogen-activated protein kinase; Mpro, SARS-Cov-2 main protease; NF-kappa B, transcription factor nuclear factor kappa B; PI3K, phosphatidylinositol 3′-kinase; PIP3, phosphatidylinositol 3, 4, 5 triphosphate; PLC, phosphoinositide phospholipase C; PP1, protein phosphatase-1; PTP, protein tyrosine phosphatases; PTP1B, protein tyrosine phosphatase 1B; RAS, renin-angiotensin system; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; STATs, signal transducers and activators of transcription; STZ, streptozotocin; Th1, T‑helper 1; TNF-α, tumor necrosis factor-α; TYK2, tyrosine kinase 2
Keywords: Vanadium, COVID-19, SARS-CoV-2 coronavirus, Diabetes mellitus, Inflammation, Hyperglycemia, Insulin sensitivity
1. Introduction
As the number of confirmed cases of the Coronavirus disease 2019 (COVID-19) overpassed 230 million and almost 5 million people have lost their lives due to this devastating disease, significant efforts are being made to efficiently treat COVID-19 and prevent its progression to the serious stage of disease, particularly in the patients with the preexisting conditions. The most reported comorbidities in patients with severe COVID-19 include hypertension, diabetes, and obesity [[1], [2], [3]]. The diabetic patients are exposed to an enlarged risk of serious complications, such as cardiac and neurological complications [4,5], as well as to an increased severity of this disease [6]. Based on recent clinical findings, pathophysiological mechanisms associated with the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), such as cytokine storm and hypercoagulation, appear to make the diabetic patients more susceptible to these COVID-19 complications [6,7]. Furthermore, this multifaceted disease may also predispose the patients to difficult-to-treat hyperglycemia [8,9] as well as to lowered sensitivity to insulin and perturbed insulin secretion [10].
The potential role of the trace metals, such as zinc and selenium, in the prevention and treatment of COVID-19 has recently been suggested [[11], [12], [13], [14], [15]]. Supplementation with zinc and selenium was reported to be crucial for the resistance to viral disease, immune function, and alleviated inflammation, particularly if it was administered during the early phase of a viral infection [11]. A recent study also indicated that based on clinical evidence, the trace elements should be added to the treatment of the early stages of COVID-19 as an immunoprotection strategy and for an efficient elimination of the virus [16].
Vanadium is a trace, ubiquitous element with a potentially unique biological function. It is widely distributed in nature, it can be found in certain algae, sea squirts, fungi, and mammalian tissues [17,18], and it has been recognized to be essential for animal development [18]. Vanadium-dependent enzymes, such as vanadate-dependent haloperoxidases and vanadium nitrogenases, have been described in different microorganisms, including bacteria [19]. In humans, it is mainly present in liver, kidney, and bones, while its concentration in blood is reported to be around 200 nM [20]. Vanadium is present in a variety of foods, including vegetable oils, skim milk, grains, cereals, and vegetables such as beans, peas, and squash [21,22]. It has been reported that only about 1% of vanadium contained in the diet is absorbed [23] and that this percentage of absorbed vanadium, as well as its disposition, therapeutic and toxic effects, are affected by its biotransformation into different vanadium species upon oral digestion [20]. This led to the development of inorganic and organic vanadium compounds containing ligands to protect the compound from speciation, to increase its absorption, and to conserve its pharmacological properties [20].
The reported effects of both, inorganic and organic vanadium compounds, include antihyperglycemic and insulin-enhancing actions, lipid-lowering, antihypertensive, cardioprotective, and antineoplastic effects [18,24,25]. Furthermore, vanadium derivatives are being used as a nutritional supplement for the enhancement of performance by body builders, weight management, and the prevention of obesity [18,26]. These biomimetic functions and mechanisms of vanadium actions will be reviewed in this article, including its potential use against viruses, with a particular focus on SARS-CoV-2. Given the significance of the puzzling link between COVID-19 and diabetes as well as the observed antihyperglycemic, insulin-enhancing, and antiinflammatory effects of vanadium compounds, further clinical studies have a high potential to open additional avenues for the treatment of both of these pandemic diseases.
2. Vanadium treatment of diabetes and other diseases
2.1. Beneficial vanadium effects
A large amount of evidence demonstrated vanadium’s antidiabetic and insulin-mimicking effects, as well as its ability to counteract insulin resistance [18,20,[27], [28], [29]]. Heyliger et al. were the first to demonstrate vanadium effects in streptozotocin (STZ)-induced diabetic rats [30], which reduced blood glucose levels to normal and prevented the diabetes-induced deterioration of cardiac function [30,31], even three months after withdrawal from an oral vanadium treatment [32]. Furthermore, recently it was suggested that the antihyperglycaemic effects of vanadium may improve kidney dysfunction in diabetes [33]. In addition, a recent report showed that a peroxyvanadate compound activated insulin-signaling cascade and improved insulin sensitivity [34], while Naglah et al. (2020) reported antidiabetic effects of vanadyl(IV) folate-amino acid-complexes in animal models of diabetes [35]. A recent study demonstrated the capability of the oxidovanadium complexes to target the active site and inhibit dipeptidyl peptidase IV (DPP-IV), which is target of recently introduced antihyperglycaemic agents used to treat T2D [36]. Vanadium compounds were also reported to be effective in the prevention of Type 2 diabetes development in animal models of diabetes [37].
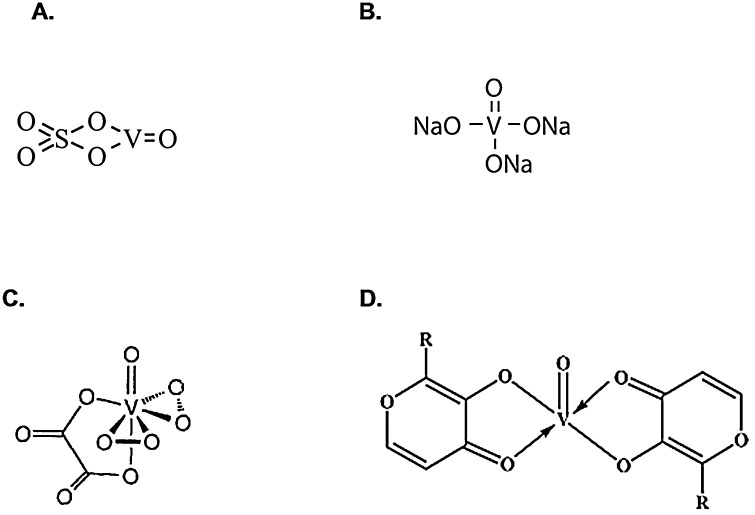
The effects of vanadium salts, such as vanadyl sulfate, sodium orthovanadate and peroxovanadium compounds, as well as organic vanadium complexes, including bis(maltolato)oxovanadium(IV) (BMOV), have been investigated in diabetes treatment (Fig. 1 ). These studies have found that organic vanadium compounds have improved the bioavailability and safety profile as compared to the inorganic salts [[38], [39], [40]]. The BMOV and its ethylmaltol analog, bis(ethylmaltolato)oxovanadium(IV) (BEOV), were shown to have similar effects on the diabetic traits in Zucker Fatty rats and STZ-diabetic Wistar rats [41,42]. BEOV demonstrated the improved characteristics for pro-drug use, so it was selected for phase 1 and phase 2 clinical trials [43]. It was demonstrated that its use in T2D subjects was associated with reduction in fasting glucose and hemoglobin A1c (HbA1c) levels, as well as with an improved response to the oral glucose tolerance test [43]. Similarly, other clinical trials also demonstrated that in both, Type 1 and Type 2 diabetic subjects, vanadium treatment decreased the need for exogenous insulin, suggesting an enhancement of insulin sensitivity [44,45] and its potential to treat diabetes [46]. Interestingly, an oral administration of vanadium compounds appears not to cause hypoglycemia, a serious potential side effect in insulin-treated diabetic patients [47]. Furthermore, vanadium treatment did not affect C-peptide levels in Type 1 diabetic patients, suggesting that restored insulin secretion was not responsible for the decreased need for insulin administration in these individuals [48]. Eriksson et al. [49] demonstrated that vanadium treatment significantly increased insulin binding capacity at the cell surface of normal and insulin-resistant cells.
Fig. 1.
Chemical structures of: A. Vanadyl sulfate; B. Sodium orthovanadate; C. Peroxovanadium complex; D. Bis(maltolato)oxovanadium(IV) (BMOV), R = CH3, and bis(ethylmaltolato)oxovanadium(IV) (BEOV), R = C2H5.
In addition to antihyperglycemic and insulin-enhancing effects, vanadium compounds also demonstrated antihyperlipidemic action. Recently, decavanadate showed insulin-mimicking effects in human adipocytes, including the inhibition of lipolysis and the activation of glucose transport, thus limiting lipotoxicity associated with obesity and insulin resistance [50]. The inhibition of lipolysis and activation of glucose uptake by different vanadium compounds was also demonstrated in other studies [26,[51], [52], [53]]. Nakai et al. [54] showed that vanadium was incorporated into most organs, including the adipose tissues of the STZ-induced diabetic rats, which was accompanied by the normalization of increased glucose and free fatty acid (FFA) levels. The gene-array study reported that oral administration of vanadyl sulfate, which alleviated diabetic hyperglycemia and hyperlipidemia, also corrected diabetes-altered gene expression in skeletal muscle of STZ-induced diabetic rats [47]. This study showed that diabetes altered the expression of about 130 genes, and the expression of 30 % of these genes was normalized by vanadyl sulfate [47]. Interestingly, these genes, whose expression appeared to be affected by vanadium treatment, include those related to the lipid metabolism, signal transduction, oxidative stress, and the complement system [47]. A recent study demonstrated that vanadium exposure appeared to be associated with increased high-density lipoprotein cholesterol (HDL-C) and apoA-I levels as well as decreased atherogenic indexes, indicating further the beneficial effects of vanadium in the treatment of atherosclerosis [55]. Interestingly, recent genome-wide CRISPR screens in SARS-CoV-2-infected cells have reported that inhibition of cholesterol homeostasis reduced the replication of coronavirus, which suggested the development of therapies targeting those intracellular pathways [56].
In addition to diabetes and atherosclerosis, vanadium compounds have been also investigated as potential medication for the treatment of other diseases, including its potential antirheumatic [57] and anticancer effects [18,58,59]. A recent novel use of vanadium compounds to stimulate viral oncolytic and systemic anticancer immunity was also demonstrated [60]. Furthermore, previous studies indicated an application of vanadium treatment in cardiac and neuronal diseases, parasitic, bacterial [61], and viral infections, such as influenza, HIV, and SARS [18].
2.2. Adverse effects of vanadium
Despite these beneficial outcomes, vanadium treatment resulted in adverse effects, including gastrointestinal symptoms, hepatotoxicity, nephrotoxicity [61,62], and neurotoxicity [63], which represent the major obstacle for more efficient transfer of vanadium compounds in the clinical use for long-term diabetes treatment, as reviewed by Domingo et al. [64,65]. Recently, different factors have been recognized to be associated with the vanadium toxicity, including the structure of the vanadium compound (inorganic/organic) and the characteristics of ligands attached to vanadium complexes, valence, dose, route of administration, duration of action/exposure, as well as an individual sensitivity to vanadium action [18]. A recent study that used highly dispersible and water-soluble graphene quantum dots as nanoplatform/delivery system for vanadium, demonstrated its improved pharmacokinetic characteristics, hypoglycemic effects, and β-cell protection in vivo [66]. Furthermore, another recent study demonstrated no toxic effects in animals after their sub-chronic treatment with vanadium nanoparticles [67]. In addition, the combinatory treatment of vanadium with several supplements also showed the protective effects against the adverse effects of vanadium [35,62,[68], [69], [70]]. Thus, further development of vanadium-based drugs, including the formation of nanoparticles and other drug-formulation strategies, should be employed to develop vanadium compounds with alleviated pharmacological properties and decreased adverse effects.
3. Rationales for potential use of vanadium compounds in COVID-19 treatment
3.1. Mechanisms of antiviral vanadium effects
As shown in the Table 1 , previous studies demonstrated that vanadium compounds affected the human immunodeficiency virus 1 (HIV-1) gene expression, thus suggesting their use in therapy of viral infections [71]. The mechanism of anti-HIV action of vanadium appears to include an effect on HIV binding to the cell membrane [72]. Ross et al. [73] reported the vanadium complexes docked into the human chemokine receptor CXCR4 model, thus affecting the function of these coreceptors essential for HIV-1 entry into CD4+ cells. This appears to prevent viral multiplication and to protect against the development of infection [18]. Vanadium complexes also inhibited the activity of viral reverse transcriptase [74], which are responsible for the integration of viral DNA into the host cell genome, thereby preventing viral replication [18]. Furthermore, physiologically stable vanadium porphyrins demonstrated a high potency in inhibiting HIV-1 replication in cultured cells, thus representing a new class of anti-HIV agents [75]. In addition, previous in vitro and in vivo studies demonstrated a broad spectrum and non-toxic anti-RNA virus activity of vanadium containing polyoxotungstate, which was suggested to be employed as a novel first-line treatment in acute respiratory diseases, including infections with the SARS coronavirus [76].
Table 1.
The rationales for potential use of vanadium compounds in prevention and treatment of COVID-19.
| General Effects of Vanadium | Effects Observed in Selected Studies | References |
|---|---|---|
| Antiviral effects | Affected HIV-1 gene expression and viral entry | [71,72,73,74] |
| Inhibited the activity of viral reverse transcriptase | [74] | |
| Prevented viral replication | [18,75] | |
| Showed a broad spectrum and non-toxic anti-RNA virus activity. | [76] | |
| Computational analysis indicated the favorable physicochemical properties of vanadium, including targeting of the SARS-Cov-2 main protease (Mpro) used by virus to enter the host cell. | [79,80] | |
| Antiinflammatory & anticoagulation effects | Affected the secretion/expression/function of inflammatory cytokines, including IFNγ, TNFα, IL-1α and IL-1β. | [57,117,118,119,120] |
| Demonstrated the inhibition of d-dimer formation, suggesting that the vanadium compounds could potentially relieve a hypercoagulative state in diabetic patients. | [100] | |
| Lowered levels of kallikrein which has a crucial role in the molecular mechanisms of vasodilation and blood coagulation. | [135,136] | |
| Antihyperglycemic & insulin-enhancing effects | Decreased high levels of glucose and insulin as well as body weight in animals. | [84] |
| Affected the activity of members of the insulin receptor signaling pathway, including but limited to TYK, PI3K, and PTP1B. | [95,96,97,98] | |
| Showed insulin-mimetic/enhancing action | [41,42,43,44,45,86,87,88,89,90,91] | |
| Demonstrated antidiabetic effects and counteracted insulin resistance. | [18,20,27,28,29] | |
| Corrected diabetes-altered gene expression in skeletal muscle of STZ-induced diabetic rats. | [47] | |
| Normalized blood glucose levels and prevented the diabetes-induced deterioration of cardiac function | [30,31,32] | |
| Showed antihyperglycaemic effects and improved kidney dysfunction in diabetic animals. | [33] | |
| Demonstrated the capability to inhibit DPP-IV, which is target of recently introduced antihyperglycaemic agents used to treat T2D. | [36] |
As described earlier, COVID-19 is caused by the RNA virus SARS-CoV-2, which employs angiotensin-converting enzyme 2 (ACE2) receptor, the transmembrane serine protease TMPRSS2, and the SARS-Cov-2 main protease (Mpro) to enter the host cell [77,78]. Viral binding to ACE2 and the other two proteases lead to the virus replication and spreading throughout the body. The recent computational studies emphasized the favorable physicochemical properties of vanadium compounds, including Mpro targeting, which could be potentially employed in treatment of SARS-CoV-2 infection [79,80].
ACE2 exerts its major role in the renin-angiotensin system (RAS), consisting of the classical ACE-Ang-II-AT1R axis and non-classical, recently discovered ACE-2-(A1-7)-Mas axis [81]. It is suggested that the ACE2 expression is modified during diabetes and that this may affect glucose homeostasis and insulin sensitivity [82]. It is also proposed that the ACE-2-(A1-7)-Mas, which is already distressed in diabetes/insulin resistance, is additionally strained due to ACE2 use for the viral entry [83].
3.2. Mechanisms of antihyperglycemic and insulin-enhancing vanadium effects
The capability of vanadium to stimulate insulin signaling pathways appears to be an important mechanism responsible for its glucoregulatory effects and its insulin-mimicking characteristics. It was demonstrated that vanadium treatment of genetically diabetic mice decreased high levels of glucose and insulin, as well as their body weight [84]. In addition, vanadium appears to regulate appetite by promoting glucose uptake in the brain [85], thus preventing an increase of body weight and obesity.
As suggested by the previous studies, vanadium promotes insulin action by a direct insulin-mimetic action and by an enhancement of insulin sensitivity [[86], [87], [88]]. It was suggested that vanadium achieves its effects by enhancing insulin action [[89], [90], [91]] through the activation of the phosphatidylinositol 3'-kinase (PI3K), stimulated synthesis of phosphatidylinositol phosphates, such as phosphatidylinositol 3, 4, 5 triphosphate (PIP3), that mediates the phosphorylation of its downstream targets regulating glucose transport, glycogen synthesis, and gluconeogenesis [92]. Semiz et al. [42] showed that vanadium treatment improved an impaired insulin sensitivity in the Zucker fatty rats, resulting in an increased muscle glucose metabolism through enhanced glycogen synthase (GS) and insulin-stimulated protein phosphatase-1 (PP1) activity. Interestingly, recent genome-wide CRISPR screens in SARS-CoV-2 infected cells have recognized phosphatidylinositol phosphate biosynthesis as a critical pathway supporting the coronavirus infection and that the inhibition of phosphatidylinositol kinases decreased the coronavirus replication [56]. Furthermore, Kawabe et al. [93] indicated that the insulin-mimetic activity of vanadium compounds includes an inhibition of FFA release and that this effect is probably regulated by the inhibition of the glucose transporter 4 (GLUT4) and members of the insulin receptor-signaling pathway, such as tyrosine kinase and PI3K. The adipogenic potential of vanadium was also attributed to the activation of the cyclic AMP response binding element (CREB) transcriptional factor [94]. In addition, a recent review reported that the mechanisms of vanadium action include protein tyrosine phosphatases (PTP), mitogen-activated protein kinase (MAPK), epidermal growth factor receptor (EGFR), transcription factor nuclear factor kappa B (NF-kappa B), phosphoinositide phospholipase C (PLC) γ, and cyclooxygenase-2 (COX-2), which are associated with the insulin signaling network [95]. This is in line with the other studies, which also implicated the activation of NF-kappa B and MAPK2 in vanadium action [[96], [97], [98]]. In addition, the extracellular signal-regulated kinase (ERK) belonging to the MAPK family, has also been reported to be involved in pathogenesis of different viral infections [99]. A recent study reported that the inhibition of ERK1/2 signaling restricted the replication of the infectious bronchitis virus (IBV), which is the representative strain of gammacoronavirus, suggesting that the components of the ERK pathway may denote excellent targets for the novel antiviral drugs development [99].
Furthermore, previous studies also implicated the inhibition of the protein tyrosine phosphatase 1B (PTP1B) as potential mechanism of vanadium action [100,101]. Accordingly, additional studies confirmed the role of potent and selective PTP1B inhibitors in diabetes management [86,[102], [103], [104]]. It was suggested that these inhibitors could also potentially target a hypercoagulative state in diabetic patients [100] and that sensitivity of PTP inhibition and glucose transport to vanadate is probably mediated by the cellular redox state [96,97,105].
3.3. Mechanisms of antiinflammatory vanadium effects
Following viral infection by SARS-CoV-2, the levels of proinflammatory cytokines (interleukin-1 (IL-1), interleukin IL-1β, interleukin-6 (IL-6), interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α)), and chemokines are elevated, leading to the COVID-19-associated vascular inflammation, coagulopathy [106] and severe forms of this disease [107]. Recently, Smith et al. indicated that ACE2 is an interferon-stimulated gene upregulated by viral infections [108]. A recent GWAS reported the genome wide significant associations of interferon receptor IFNAR2 gene in seriously ill COVID-19 patients [109]. Furthermore, type I interferons (IFN-I) have been indicated as key players in an effective antiviral response [110]. Recent findings suggested that IFN-I signaling is affected by genetic variations of tyrosine kinase 2 (TYK2) [111], which was reported with the genome wide significant association in severe cases of COVID-19 [109]. This kinase is a Janus kinases (JAKs) family member, involved in the cytokine receptors’ downstream signaling, whose activation leads to phosphorylation of the signal transducers and activators of transcription (STATs) and increased production of inflammatory mediators, triggering the cytokine storm and inflammation in COVID-19 patients [112]. With an effort to alleviate excessive inflammation, JAK/STAT inhibitors, including the first-generation ruxolitinib, tofacitinib, and baricitinib, have been reported to act via suppression of cytokine signaling and protein tyrosine phosphatases activity [112]. The effects of vanadium on expression and activities of JAK/STAT pathway was investigated in the recent study, which appeared to demonstrate a trend of reduced STAT phosphorylation by vanadium [113].
An accumulating amount of evidence suggests that adipose tissue inflammation is an important risk factor for the development of insulin resistance and Type 2 diabetes in obese subjects [114,115], which may lead to increased proinflammatory cytokine levels [116]. As mentioned earlier, vanadium compounds acting as PTP inhibitors, inhibited the formation of D-dimer, a fibrin degradation product and a marker of coagulation activation [100]. This suggests a potential relieving effect of vanadium on a hypercoagulative state in diabetes [100] as well as in similar inflammatory and coagulative processes described above in COVID-19 patients. Recently, it was reported that vanadium modulated the effect of IFNγ on chemokine secretion [117]. It was also demonstrated that vanadium compounds were able to stimulate the secretion of T‑helper 1 (Th1) chemokines, synergistically increasing the effect of key Th1 cytokines, including IFNγ and TNFα [118]. Previous studies have also showed that vanadium affected the macrophage IFNγ-binding and -inducible responses [119], which were associated with lower DNA methylation of IFNγ and interleukin 4 (IL-4) [120]. Deregulated signaling of interleukin 1α (IL-1α) and IL-1β inflammatory cytokines has been reported to cause the devastating diseases associated with severe inflammation [121]. Recent systematic review/meta-analysis identified IL-1α as one of the novel therapeutic targets for COVID-19 treatment [122]. Interestingly, Oliver et al. [57] showed that BMOV therapy strongly reduced synovial mRNA expression of IL-1α and alleviated symptoms of clinical arthritis in BMOV-treated rats as compared to controls. Vanadium compounds may also affect the expression of the inducible form of cyclooxygenase COX-2 [98] and regulate its activity [123], which appears to be increased during acute and chronic inflammation contributing to the excessive cellular production of reactive oxygen species and tissue damage [124].
In addition to these interferon-regulated processes, it was also reported that ACE2 can affect the kinin-kallikrein system, which has a crucial role in the molecular mechanisms of vasodilation and blood coagulation [[125], [126], [127]]. ACE2 regulates both bradykinin (BK) receptors, the BK type 1 (B1R) and BK type 2 (B2R) receptors. Recently, Garvin et al. [128] performed the gene expression data analysis in bronchoalveolar lavage fluid cells from COVID-19 patients and showed increased levels of ACE2, RAS, renin, angiotensin, bradykinin receptors, kinogen and several kallikrein enzymes, as compared to the control subjects. Similarly, other recent studies showed that the virus binding to ACE2 increases bradykinin levels, leading to the systemic inflammation, coagulation, the complement system induction, and the cytokine storm in COVID-19 patients [7,78,129]. These bradykinin-driven pathological processes appear to be associated with many of the symptoms observed in the COVID-19 patients [128], with an extensive release of proinflammatory cytokines and with a decrease in T cell counts [78,129]. Recently, it was proposed that blocking the kallikrein-kinin system with lanadelumab, a monoclonal antibody against the plasmatic kallikrein, should prevent the inflammatory and coagulation storm in these patients and thus, should be used in parallel with antiviral therapy [78]. Another inhibitor of the kallikrein-kinin system is conestat alfa, which is a recombinant human C1 esterase inhibitor (C1INH). It also inhibits serine protease activity [130], which was reported to be elevated in T2D patients and to be positively associated with HbA1c levels, homeostasis model assessment-insulin resistance (HOMA-IR) and body mass index (BMI), thus supporting the potential role of serine protease in T2D development [116]. Previous studies demonstrated that vanadium compounds inhibited serine proteases [131,132]. Furthermore, a recent study demonstrated that treatment with the conestat alfa also resulted in reduced levels of complement activation products and decreased viral loads in nasopharyngeal swabs, leading to alleviation of the severe COVID-19 cases [133]. Furthermore, van de Veerdonk et al. [134] recently proposed that blocking the B2R and plasma kallikrein activity might produce beneficial effects in the early stages of disease caused by SARS-CoV-2 and prevent acute respiratory distress syndrome. Interestingly, it was reported that vanadate treatment lowered levels of urine kallikrein in animal models [135,136], suggesting its potential action on the coronavirus infection via regulation of kallikrein levels.
Therefore, vanadium treatment seems to modulate major risk factors for severe COVID-19, including its alleviating effect on hyperglycemia and dysregulated immune and inflammatory responses. Furthermore, as suggested by the recent computational studies, vanadium may also affect the SARS-CoV-2 viral entry. Thus, the potential of vanadium compounds as an adjuvant therapeutic agent in the treatment of COVID-19 should be explored in further studies, which appear to have not been investigated sufficiently.
4. Conclusions
In conclusion, an increasing evidence suggests that vanadium compounds may represent new potential medications in therapy of diabetes, cancer, atherosclerosis, and other diseases. Vanadium compounds have also demonstrated a broad spectrum and non-toxic activities against RNA viruses and are promising candidates for treating acute respiratory diseases. Given those benefits and the important relationship between COVID-19 and diabetes as well as the observed beneficial antiviral, antihyperglycemic, insulin-enhancing, and anti-inflammatory effects, vanadium compounds could be considered as an adjuvant therapy to the prescribed treatment of COVID-19. Furthermore, vanadium might be used to complement efforts in managing difficult-to-treat hyperglycemia in COVID-19 patients. Thus, a further clinical trials are warranted to confirm these beneficial effects of vanadium treatment of COVID-19, which seem to have not been studied yet.
Funding
No funding was received to carry out this research work.
Ethics approval and informed consent
Not applicable.
Availability of data
Not applicable.
Declaration of Competing Interest
I wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
Acknowledgements
Not applicable.
References
- 1.Erener S. Diabetes, infection risk and COVID-19. Mol. Metab. 2020;39(September):101044. doi: 10.1016/j.molmet.2020.101044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Guan W.J., et al. Comorbidity and its impact on 1590 patients with COVID-19 in China: a nationwide analysis. Eur. Respir. J. 2020;55(May (5)) doi: 10.1183/13993003.00547-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Richardson S., et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York city area. JAMA. 2020;323(May (20)):2052–2059. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Whittaker A., Anson M., Harky A. Neurological manifestations of COVID-19: a systematic review and current update. Acta Neurol. Scand. 2020;142(July (1)):14–22. doi: 10.1111/ane.13266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shao F., et al. In-hospital cardiac arrest outcomes among patients with COVID-19 pneumonia in Wuhan, China. Resuscitation. 2020;151(June):18–23. doi: 10.1016/j.resuscitation.2020.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lim S., Bae J.H., Kwon H.S., Nauck M.A. COVID-19 and diabetes mellitus: from pathophysiology to clinical management. Nat. Rev. Endocrinol. 2021;17(January (1)):11–30. doi: 10.1038/s41574-020-00435-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ye Q., Wang B., Mao J. The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19. J. Infect. 2020;80(June (6)):607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sachdeva S., Desai R., Gupta U., Prakash A., Jain A., Aggarwal A. Admission hyperglycemia in non-diabetics predicts mortality and disease severity in COVID-19: a pooled analysis and meta-summary of literature. SN Comp. Clin. Med. 2020;(October (12)):1–6. doi: 10.1007/s42399-020-00575-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ilias I., Zabuliene L. Hyperglycemia and the novel Covid-19 infection: possible pathophysiologic mechanisms. Med. Hypotheses. 2020;139(June):109699. doi: 10.1016/j.mehy.2020.109699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ilias I., et al. Glycemia, beta-cell function and sensitivity to insulin in mildly to critically ill Covid-19 patients. Medicina (Kaunas) 2021;57(January (1)) doi: 10.3390/medicina57010068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Alexander J., Tinkov A., Strand T.A., Alehagen U., Skalny A., Aaseth J. Early nutritional interventions with zinc, selenium and vitamin d for raising anti-viral resistance against progressive COVID-19. Nutrients. 2020;12(August (8)) doi: 10.3390/nu12082358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wessels I., Rolles B., Rink L. The potential impact of zinc supplementation on COVID-19 pathogenesis. Front. Immunol. 2020;11:1712. doi: 10.3389/fimmu.2020.01712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pal A., et al. Zinc and COVID-19: basis of current clinical trials. Biol. Trace Elem. Res. 2020;(October (22)) doi: 10.1007/s12011-020-02437-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hiffler L., Rakotoambinina B. Selenium and RNA virus interactions: potential implications for SARS-CoV-2 infection (COVID-19) Front. Nutr. 2020;7:164. doi: 10.3389/fnut.2020.00164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhang J., Saad R., Taylor E.W., Rayman M.P. Selenium and selenoproteins in viral infection with potential relevance to COVID-19. Redox Biol. 2020;37(October):101715. doi: 10.1016/j.redox.2020.101715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mingazova E.N., Gureev S.A. Deficient micronutrient status of the population of various countries as a risk factor in COVID-19. Probl. Sotsialnoi Gig. Istor. Med. 2021;29(June (Special Issue)):593–597. doi: 10.32687/0869-866X-2021-29-s1-593-597. [DOI] [PubMed] [Google Scholar]
- 17.Shaver A., Ng J.B., Hall D.A., Posner B.I. The chemistry of peroxovanadium compounds relevant to insulin mimesis. Mol. Cell. Biochem. 1995;153(December (1-2)):5–15. doi: 10.1007/BF01075913. [DOI] [PubMed] [Google Scholar]
- 18.Scibior A., Pietrzyk L., Plewa Z., Skiba A. Vanadium: risks and possible benefits in the light of a comprehensive overview of its pharmacotoxicological mechanisms and multi-applications with a summary of further research trends. J. Trace Elem. Med. Biol. 2020;61(September):126508. doi: 10.1016/j.jtemb.2020.126508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rehder D. The future of/for vanadium. Dalton Trans. 2013;42(September (33)):11749–11761. doi: 10.1039/c3dt50457c. [DOI] [PubMed] [Google Scholar]
- 20.Trevino S., Diaz A., Sanchez-Lara E., Sanchez-Gaytan B.L., Perez-Aguilar J.M., Gonzalez-Vergara E. Vanadium in biological action: chemical, pharmacological aspects, and metabolic implications in diabetes mellitus. Biol. Trace Elem. Res. 2019;188(March (1)):68–98. doi: 10.1007/s12011-018-1540-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Badmaev V., Prakash S., Majeed M. Vanadium: a review of its potential role in the fight against diabetes. J. Altern. Complement. Med. 1999;5(June (3)):273–291. doi: 10.1089/acm.1999.5.273. [DOI] [PubMed] [Google Scholar]
- 22.Vanadium - help for blood sugar problems? TreatmentUpdate. 1999;11(August (6)):5–7. https://www.ncbi.nlm.nih.gov/pubmed/11366936 [Online]. Available: [PubMed] [Google Scholar]
- 23.Ma J., Pan L.B., Wang Q., Lin C.Y., Duan X.L., Hou H. Estimation of the daily soil/dust (SD) ingestion rate of children from Gansu Province, China via hand-to-mouth contact using tracer elements. Environ. Geochem. Health. 2018;40(February (1)):295–301. doi: 10.1007/s10653-016-9906-1. [DOI] [PubMed] [Google Scholar]
- 24.Scrivens P.J., et al. Cdc25A-inhibitory properties and antineoplastic activity of bisperoxovanadium analogues. Mol. Cancer Ther. 2003;2(October (10)):1053–1059. https://www.ncbi.nlm.nih.gov/pubmed/14578470 [Online]. Available: [PubMed] [Google Scholar]
- 25.Verma S., Cam M.C., McNeill J.H. Nutritional factors that can favorably influence the glucose/insulin system: vanadium. J. Am. Coll. Nutr. 1998;17(February (1)):11–18. doi: 10.1080/07315724.1998.10718730. [DOI] [PubMed] [Google Scholar]
- 26.Adachi Y., et al. Improvement of diabetes, obesity and hypertension in type 2 diabetic KKAy mice by bis(allixinato)oxovanadium(IV) complex. Biochem. Biophys. Res. Commun. 2006;345(July (3)):945–950. doi: 10.1016/j.bbrc.2006.05.003. [DOI] [PubMed] [Google Scholar]
- 27.Shechter Y. Insulin-mimetic effects of vanadate. Possible implications for future treatment of diabetes. Diabetes. 1990;39(January (1)):1–5. doi: 10.2337/diacare.39.1.1. [DOI] [PubMed] [Google Scholar]
- 28.Brichard S.M., Pottier A.M., Henquin J.C. Long term improvement of glucose homeostasis by vanadate in obese hyperinsulinemic fa/fa rats. Endocrinology. 1989;125(November (5)):2510–2516. doi: 10.1210/endo-125-5-2510. [DOI] [PubMed] [Google Scholar]
- 29.Yuen V.G., Pederson R.A., Dai S., Orvig C., McNeill J.H. Effects of low and high dose administration of bis(maltolato)oxovanadium(IV) on fa/fa Zucker rats. Can. J. Physiol. Pharmacol. 1996;74(September (9)):1001–1009. https://www.ncbi.nlm.nih.gov/pubmed/8960391 [Online]. Available: [PubMed] [Google Scholar]
- 30.Heyliger C.E., Tahiliani A.G., McNeill J.H. Effect of vanadate on elevated blood glucose and depressed cardiac performance of diabetic rats. Science. 1985;227(March (4693)):1474–1477. doi: 10.1126/science.3156405. [DOI] [PubMed] [Google Scholar]
- 31.Poucheret P., Verma S., Grynpas M.D., McNeill J.H. Vanadium and diabetes. Mol. Cell. Biochem. 1998;188(November (1-2)):73–80. https://www.ncbi.nlm.nih.gov/pubmed/9823013 [Online]. Available: [PubMed] [Google Scholar]
- 32.Ramanadham S., Brownsey R.W., Cros G.H., Mongold J.J., McNeill J.H. Sustained prevention of myocardial and metabolic abnormalities in diabetic rats following withdrawal from oral vanadyl treatment. Metabolism. 1989;38(October (10)):1022–1028. doi: 10.1016/0026-0495(89)90016-4. [DOI] [PubMed] [Google Scholar]
- 33.Mbatha B., Khathi A., Sibiya N., Booysen I., Mangundu P., Ngubane P. Anti-hyperglycaemic effects of dioxidovanadium complex cis-[VO2(obz)py] avert kidney dysfunction in streptozotocin-induced diabetic male Sprague-Dawley rats. Can. J. Physiol. Pharmacol. 2021;99(April (4)):402–410. doi: 10.1139/cjpp-2020-0278. [DOI] [PubMed] [Google Scholar]
- 34.Mukherjee S., et al. A small insulinomimetic molecule also improves insulin sensitivity in diabetic mice. PLoS One. 2017;12(1):e0169809. doi: 10.1371/journal.pone.0169809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Naglah A.M., et al. Synthesis, characterization, and anti-diabetic activity of some novel vanadium-folate-amino acid materials. Biomolecules. 2020;10(May (5)) doi: 10.3390/biom10050781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Xie M.J., et al. Synthesis and characterization of oxidovanadium complexes as enzyme inhibitors targeting dipeptidyl peptidase IV. J. Inorg. Biochem. 2017;175(October):29–35. doi: 10.1016/j.jinorgbio.2017.06.014. [DOI] [PubMed] [Google Scholar]
- 37.Winter C.L., et al. A nonspecific phosphotyrosine phosphatase inhibitor, bis(maltolato)oxovanadium(IV), improves glucose tolerance and prevents diabetes in Zucker diabetic fatty rats. Exp. Biol. Med. (Maywood) 2005;230(March (3)):207–216. doi: 10.1177/153537020523000307. [DOI] [PubMed] [Google Scholar]
- 38.Fedorova E.V., Buriakina A.V., Vorob’eva N.M., Baranova N.I. The vanadium compounds: chemistry, synthesis, insulinomimetic properties. Biomed. Khim. 2014;60(July-August (4)):416–429. doi: 10.18097/pbmc20146004416. [DOI] [PubMed] [Google Scholar]
- 39.Rehder D. The potentiality of vanadium in medicinal applications. Future Med. Chem. 2012;4(September (14)):1823–1837. doi: 10.4155/fmc.12.103. [DOI] [PubMed] [Google Scholar]
- 40.Willsky G.R., et al. Effect of vanadium(IV) compounds in the treatment of diabetes: in vivo and in vitro studies with vanadyl sulfate and bis(maltolato)oxovandium(IV) J. Inorg. Biochem. 2001;85(May (1)):33–42. doi: 10.1016/s0162-0134(00)00226-9. [DOI] [PubMed] [Google Scholar]
- 41.Semiz S., Orvig C., McNeill J.H. Effects of diabetes, vanadium, and insulin on glycogen synthase activation in Wistar rats. Mol. Cell. Biochem. 2002;231(February (1-2)):23–35. doi: 10.1023/a:1014437019586. [DOI] [PubMed] [Google Scholar]
- 42.Semiz S., McNeill J.H. Oral treatment with vanadium of Zucker fatty rats activates muscle glycogen synthesis and insulin-stimulated protein phosphatase-1 activity. Mol. Cell. Biochem. 2002;236(July (1-2)):123–131. doi: 10.1023/a:1016116700632. [DOI] [PubMed] [Google Scholar]
- 43.Thompson K.H., Lichter J., LeBel C., Scaife M.C., McNeill J.H., Orvig C. Vanadium treatment of type 2 diabetes: a view to the future. J. Inorg. Biochem. 2009;103(April (4)):554–558. doi: 10.1016/j.jinorgbio.2008.12.003. [DOI] [PubMed] [Google Scholar]
- 44.Cohen N., Halberstam M., Shlimovich P., Chang C.J., Shamoon H., Rossetti L. Oral vanadyl sulfate improves hepatic and peripheral insulin sensitivity in patients with non-insulin-dependent diabetes mellitus. J. Clin. Invest. 1995;95(June (6)):2501–2509. doi: 10.1172/JCI117951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Goldfine A.B., et al. Metabolic effects of vanadyl sulfate in humans with non-insulin-dependent diabetes mellitus: in vivo and in vitro studies. Metabolism. 2000;49(March (3)):400–410. doi: 10.1016/s0026-0495(00)90418-9. [DOI] [PubMed] [Google Scholar]
- 46.Brichard S.M., Henquin J.C. The role of vanadium in the management of diabetes. Trends Pharmacol. Sci. 1995;16(August (8)):265–270. doi: 10.1016/s0165-6147(00)89043-4. [DOI] [PubMed] [Google Scholar]
- 47.Willsky G.R., Chi L.H., Liang Y., Gaile D.P., Hu Z., Crans D.C. Diabetes-altered gene expression in rat skeletal muscle corrected by oral administration of vanadyl sulfate. Physiol. Genomics. 2006;26(August (3)):192–201. doi: 10.1152/physiolgenomics.00196.2005. [DOI] [PubMed] [Google Scholar]
- 48.Goldfine A.B., Simonson D.C., Folli F., Patti M.E., Kahn C.R. Metabolic effects of sodium metavanadate in humans with insulin-dependent and noninsulin-dependent diabetes mellitus in vivo and in vitro studies. J. Clin. Endocrinol. Metab. 1995;80(November (11)):3311–3320. doi: 10.1210/jcem.80.11.7593444. [DOI] [PubMed] [Google Scholar]
- 49.Eriksson J.W., Lonnroth P., Smith U. Vanadate increases cell surface insulin binding and improves insulin sensitivity in both normal and insulin-resistant rat adipocytes. Diabetologia. 1992;35(June (6)):510–516. doi: 10.1007/BF00400477. [DOI] [PubMed] [Google Scholar]
- 50.Carpene C., et al. Insulin-mimetic compound hexaquis (benzylammonium) decavanadate is antilipolytic in human fat cells. World J. Diabetes. 2017;8(April (4)):143–153. doi: 10.4239/wjd.v8.i4.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Nilsson J., et al. Bis- and tris(pyridyl)amine-oxidovanadium complexes: characteristics and insulin-mimetic potential. Dalton Trans. 2009;38(October (14)):7902–7911. doi: 10.1039/b903456k. [DOI] [PubMed] [Google Scholar]
- 52.Sakurai H. Therapeutic potential of vanadium in treating diabetes mellitus. Clin. Calcium. 2005;15(January (1)):49–57. doi: CliCA05014957. [PubMed] [Google Scholar]
- 53.Gatjens J., et al. A new family of insulin-mimetic vanadium complexes derived from 5-carboalkoxypicolinates. Chemistry. 2003;9(October (20)):4924–4935. doi: 10.1002/chem.200305019. [DOI] [PubMed] [Google Scholar]
- 54.Nakai M., et al. Mechanism on insulin-like action of vanadyl sulfate: studies on interaction between rat adipocytes and vanadium compounds. Biol. Pharm. Bull. 1995;18(May (5)):719–725. doi: 10.1248/bpb.18.719. [DOI] [PubMed] [Google Scholar]
- 55.Zhang Y., et al. Influence of vanadium on serum lipid and lipoprotein profiles: a population-based study among vanadium exposed workers. Lipids Health Dis. 2014;13(February (24)):39. doi: 10.1186/1476-511X-13-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wang R., et al. Genetic screens identify host factors for SARS-CoV-2 and common cold coronaviruses. Cell. 2021;184(January (1)):106–119. doi: 10.1016/j.cell.2020.12.004. e14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Oliver S.J., et al. Vanadate, an inhibitor of stromelysin and collagenase expression, suppresses collagen induced arthritis. J. Rheumatol. 2007;34(September (9)):1802–1809. https://www.ncbi.nlm.nih.gov/pubmed/17696279 [Online]. Available: [PubMed] [Google Scholar]
- 58.Chakraborty T., Ghosh S., Datta S., Chakraborty P., Chatterjee M. Vanadium suppresses sister-chromatid exchange and DNA-protein crosslink formation and restores antioxidant status and hepatocellular architecture during 2-acetylaminofluorene-induced experimental rat hepatocarcinogenesis. J. Exp. Ther. Oncol. 2003;3(November-December (6)):346–362. doi: 10.1111/j.1533-869x.2003.01107.x. [DOI] [PubMed] [Google Scholar]
- 59.Ni L., et al. Synthesis, in vitro cytotoxicity, and structure-activity relationships (SAR) of multidentate oxidovanadium(iv) complexes as anticancer agents. Dalton Trans. 2018;47(July (30)):10035–10045. doi: 10.1039/c8dt01778f. [DOI] [PubMed] [Google Scholar]
- 60.Crans D.C., Henry L., Cardiff G., Posner B.I. Developing vanadium as an antidiabetic or anticancer drug: a clinical and historical perspective. Met. Ions Life Sci. 2019;19(January (14)) doi: 10.1515/9783110527872-014. [DOI] [PubMed] [Google Scholar]
- 61.Rehder D. Vanadium. Its role for humans. Met. Ions Life Sci. 2013;13:139–169. doi: 10.1007/978-94-007-7500-8_5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wilk A., Szypulska-Koziarska D., Wiszniewska B. The toxicity of vanadium on gastrointestinal, urinary and reproductive system, and its influence on fertility and fetuses malformations. Postepy Hig. Med. Dosw. Online (Online) 2017;71(September (25)):850–859. doi: 10.5604/01.3001.0010.4783. [DOI] [PubMed] [Google Scholar]
- 63.Jaiswal M.R., Kale P.P. Mini review-vanadium-induced neurotoxicity and possible targets. Neurol. Sci. 2020;41(April (4)):763–768. doi: 10.1007/s10072-019-04188-5. [DOI] [PubMed] [Google Scholar]
- 64.Domingo J.L. Vanadium and tungsten derivatives as antidiabetic agents: a review of their toxic effects. Biol. Trace Elem. Res. 2002;88(August (2)):97–112. doi: 10.1385/BTER:88:2:097. [DOI] [PubMed] [Google Scholar]
- 65.Domingo J.L., Gomez M. Vanadium compounds for the treatment of human diabetes mellitus: a scientific curiosity? A review of thirty years of research. Food Chem. Toxicol. 2016;95(September):137–141. doi: 10.1016/j.fct.2016.07.005. [DOI] [PubMed] [Google Scholar]
- 66.Du J., Feng B., Dong Y., Zhao M., Yang X. Vanadium coordination compounds loaded on graphene quantum dots (GQDs) exhibit improved pharmaceutical properties and enhanced anti-diabetic effects. Nanoscale. 2020;12(April (16)):9219–9230. doi: 10.1039/d0nr00810a. [DOI] [PubMed] [Google Scholar]
- 67.Das S., et al. Anti-angiogenic vanadium pentoxide nanoparticles for the treatment of melanoma and their in vivo toxicity study. Nanoscale. 2020;12(April (14)):7604–7621. doi: 10.1039/d0nr00631a. [DOI] [PubMed] [Google Scholar]
- 68.Olaolorun F.A., Obasa A.A., Balogun H.A., Aina O.O., Olopade J.O. Lactational Vitamin E Protects Against the Histotoxic Effects of Systemically Administered Vanadium in Neonatal Rats. Niger. J. Physiol. Sci. 2014;29(December (2)):125–129. https://www.ncbi.nlm.nih.gov/pubmed/26196578 [Online]. Available: [PubMed] [Google Scholar]
- 69.Shrivastava S., Joshi D., Bhadauria M., Shukla S., Mathur R. Cotherapy of Tiron and selenium against vanadium induced toxic effects in lactating rats. Iran. J. Reprod. Med. 2011;9(Summer (3)):229–238. https://www.ncbi.nlm.nih.gov/pubmed/26396569 [Online]. Available: [PMC free article] [PubMed] [Google Scholar]
- 70.Zwolak I. Protective effects of dietary antioxidants against vanadium-induced toxicity: a review. Oxid. Med. Cell. Longev. 2020;2020:1490316. doi: 10.1155/2020/1490316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ouellet M., Barbeau B., Tremblay M.J. Protein tyrosyl phosphatases in T cell activation: implication for human immunodeficiency virus transcriptional activity. Prog. Nucleic Acid Res. Mol. Biol. 2003;73:69–105. doi: 10.1016/s0079-6603(03)01003-1. [DOI] [PubMed] [Google Scholar]
- 72.Shigeta S., Mori S., Kodama E., Kodama J., Takahashi K., Yamase T. Broad spectrum anti-RNA virus activities of titanium and vanadium substituted polyoxotungstates. Antiviral Res. 2003;58(May (3)):265–271. doi: 10.1016/s0166-3542(03)00009-3. [DOI] [PubMed] [Google Scholar]
- 73.Ross A., et al. Oxovanadium(IV) cyclam and bicyclam complexes: potential CXCR4 receptor antagonists. Inorg. Chem. 2010;49(February (3)):1122–1132. doi: 10.1021/ic9020614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Pessoa J.C., Etcheverry S., Gambino D. Vanadium compounds in medicine. Coord. Chem. Rev. 2015;301(October (15)):24–48. doi: 10.1016/j.ccr.2014.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wong S.Y., Wai-Yin Sun R., Chung N.P., Lin C.L., Che C.M. Physiologically stable vanadium(IV) porphyrins as a new class of anti-HIV agents. Chem. Commun. (Camb.) 2005;28(July (28)):3544–3546. doi: 10.1039/b503535j. [DOI] [PubMed] [Google Scholar]
- 76.Shigeta S., Mori S., Yamase T., Yamamoto N., Yamamoto N. Anti-RNA virus activity of polyoxometalates. Biomed. Pharmacother. 2006;60(5) doi: 10.1016/j.biopha.2006.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Hoffmann M., et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(April (2)):271–280. doi: 10.1016/j.cell.2020.02.052. e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Colarusso C., Terlizzi M., Pinto A., Sorrentino R. A lesson from a saboteur: High-MW kininogen impact in coronavirus-induced disease 2019. Br. J. Pharmacol. 2020;177(November (21)):4866–4872. doi: 10.1111/bph.15154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Scior T., Abdallah H.H., Mustafa S.F.Z., Guevara-Garcia J.A., Rehder D. Are vanadium complexes druggable against the main protease M(pro) of SARS-CoV-2? - A computational approach. Inorganica Chim. Acta. 2021;519(May (1)):120287. doi: 10.1016/j.ica.2021.120287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Vlasiou M.C., Pafti K.S. Screening possible drug molecules for Covid-19. The example of vanadium (III/IV/V) complex molecules with computational chemistry and molecular docking. Comput Toxicol. 2021;18(May):100157. doi: 10.1016/j.comtox.2021.100157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Gheblawi M., et al. Angiotensin-converting enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system: celebrating the 20th anniversary of the discovery of ACE2. Circ. Res. 2020;126(May (10)):1456–1474. doi: 10.1161/CIRCRESAHA.120.317015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Bindom S.M., Lazartigues E. The sweeter side of ACE2: physiological evidence for a role in diabetes. Mol. Cell. Endocrinol. 2009;302(April (2)):193–202. doi: 10.1016/j.mce.2008.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Dalan R., et al. The ACE-2 in COVID-19: Foe or Friend? Horm. Metab. Res. 2020;52(May (5)):257–263. doi: 10.1055/a-1155-0501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Pugazhenthi S., Angel J.F., Khandelwal R.L. Long-term effects of vanadate treatment on glycogen metabolizing and lipogenic enzymes of liver in genetically diabetic (db/db) mice. Metabolism. 1991;40(September (9)):941–946. doi: 10.1016/0026-0495(91)90070-d. [DOI] [PubMed] [Google Scholar]
- 85.Meyerovitch J., Shechter Y., Amir S. Vanadate stimulates in vivo glucose uptake in brain and arrests food intake and body weight gain in rats. Physiol. Behav. 1989;45(June (6)):1113–1116. doi: 10.1016/0031-9384(89)90096-6. [DOI] [PubMed] [Google Scholar]
- 86.Fantus I.G., Deragon G., Lai R., Tang S. Modulation of insulin action by vanadate: evidence of a role for phosphotyrosine phosphatase activity to alter cellular signaling. Mol. Cell. Biochem. 1995;153(December (1-2)):103–112. doi: 10.1007/BF01075924. [DOI] [PubMed] [Google Scholar]
- 87.Fantus I.G., Tsiani E. Multifunctional actions of vanadium compounds on insulin signaling pathways: evidence for preferential enhancement of metabolic versus mitogenic effects. Mol. Cell. Biochem. 1998;182(May (1-2)):109–119. https://www.ncbi.nlm.nih.gov/pubmed/9609120 [Online]. Available: [PubMed] [Google Scholar]
- 88.Sprietsma J.E., Schuitemaker G.E. Diabetes can be prevented by reducing insulin production. Med. Hypotheses. 1994;42(January (1)):15–23. doi: 10.1016/0306-9877(94)90029-9. [DOI] [PubMed] [Google Scholar]
- 89.Gherzi R., et al. Direct modulation of insulin receptor protein tyrosine kinase by vanadate and anti-insulin receptor monoclonal antibodies. Biochem. Biophys. Res. Commun. 1988;152(May (3)):1474–1480. doi: 10.1016/s0006-291x(88)80452-2. [DOI] [PubMed] [Google Scholar]
- 90.Fantus I.G., Kadota S., Deragon G., Foster B., Posner B.I. Pervanadate [peroxide(s) of vanadate] mimics insulin action in rat adipocytes via activation of the insulin receptor tyrosine kinase. Biochemistry. 1989;28(October (22)):8864–8871. doi: 10.1021/bi00448a027. [DOI] [PubMed] [Google Scholar]
- 91.Zick Y., Sagi-Eisenberg R. A combination of H2O2 and vanadate concomitantly stimulates protein tyrosine phosphorylation and polyphosphoinositide breakdown in different cell lines. Biochemistry. 1990;29(November (44)):10240–10245. doi: 10.1021/bi00496a013. [DOI] [PubMed] [Google Scholar]
- 92.Vardatsikos G., Mehdi M.Z., Srivastava A.K. Bis(maltolato)-oxovanadium (IV)-induced phosphorylation of PKB, GSK-3 and FOXO1 contributes to its glucoregulatory responses (review) Int. J. Mol. Med. 2009;24(September (3)):303–309. doi: 10.3892/ijmm_00000233. [DOI] [PubMed] [Google Scholar]
- 93.Kawabe K., Yoshikawa Y., Adachi Y., Sakurai H. Possible mode of action for insulinomimetic activity of vanadyl(IV) compounds in adipocytes. Life Sci. 2006;78(May (24)):2860–2866. doi: 10.1016/j.lfs.2005.11.008. [DOI] [PubMed] [Google Scholar]
- 94.Shukla R., Bhonde R.R. Adipogenic action of vanadium: a new dimension in treating diabetes. Biometals. 2008;21(April (2)):205–210. doi: 10.1007/s10534-007-9109-4. [DOI] [PubMed] [Google Scholar]
- 95.Korbecki J., Baranowska-Bosiacka I., Gutowska I., Chlubek D. Vanadium compounds as pro-inflammatory agents: effects on cyclooxygenases. Int. J. Mol. Sci. 2015;16(June (6)):12648–12668. doi: 10.3390/ijms160612648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Krejsa C.M., Schieven G.L. Impact of oxidative stress on signal transduction control by phosphotyrosine phosphatases. Environ. Health Perspect. 1998;106(Suppl 5):1179–1184. doi: 10.1289/ehp.98106s51179. Oct. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Krejsa C.M., Nadler S.G., Esselstyn J.M., Kavanagh T.J., Ledbetter J.A., Schieven G.L. Role of oxidative stress in the action of vanadium phosphotyrosine phosphatase inhibitors. Redox independent activation of NF-kappaB. J. Biol. Chem. 1997;272(April (17)):11541–11549. doi: 10.1074/jbc.272.17.11541. [DOI] [PubMed] [Google Scholar]
- 98.Korbecki J., Baranowska-Bosiacka I., Gutowska I., Chlubek D. Biochemical and medical importance of vanadium compounds. Acta Biochim. Pol. 2012;59(2):195–200. https://www.ncbi.nlm.nih.gov/pubmed/22693688 [Online]. Available: [PubMed] [Google Scholar]
- 99.Wang H., et al. Upregulation of DUSP6 impairs infectious bronchitis virus replication by negatively regulating ERK pathway and promoting apoptosis. Vet. Res. 2021;52(January (1)):7. doi: 10.1186/s13567-020-00866-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Gundhla I.Z., et al. pH-metric chemical speciation modeling and studies of in vitro antidiabetic effects of bis[(imidazolyl)carboxylato]oxidovanadium(IV) complexes. J. Inorg. Biochem. 2015;145(April):11–18. doi: 10.1016/j.jinorgbio.2014.12.019. [DOI] [PubMed] [Google Scholar]
- 101.Irving E., Stoker A.W. Vanadium compounds as PTP inhibitors. Molecules. 2017;22(December (12)) doi: 10.3390/molecules22122269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Thareja S., Aggarwal S., Bhardwaj T.R., Kumar M. Protein tyrosine phosphatase 1B inhibitors: a molecular level legitimate approach for the management of diabetes mellitus. Med. Res. Rev. 2012;32(May (3)):459–517. doi: 10.1002/med.20219. [DOI] [PubMed] [Google Scholar]
- 103.Scior T., Mack H.G., Garcia J.A., Koch W. Antidiabetic Bis-Maltolato-OxoVanadium(IV): conversion of inactive trans- to bioactive cis-BMOV for possible binding to target PTP-1B. Drug Des. Devel. Ther. 2009;2(February (6)):221–231. https://www.ncbi.nlm.nih.gov/pubmed/19920909 [Online]. Available: [PMC free article] [PubMed] [Google Scholar]
- 104.Han H., et al. Synthesis and evaluation of oxovanadium(IV) complexes of Schiff-base condensates from 5-substituted-2-hydroxybenzaldehyde and 2-substituted-benzenamine as selective inhibitors of protein tyrosine phosphatase 1B. Dalton Trans. 2012;41(August (36)):11116–11124. doi: 10.1039/c2dt30198a. [DOI] [PubMed] [Google Scholar]
- 105.Lu B., et al. Enhanced sensitivity of insulin-resistant adipocytes to vanadate is associated with oxidative stress and decreased reduction of vanadate (+5) to vanadyl (+4) J. Biol. Chem. 2001;276(September (38)):35589–35598. doi: 10.1074/jbc.M106783200. [DOI] [PubMed] [Google Scholar]
- 106.Zhang J., Tecson K.M., McCullough P.A. Endothelial dysfunction contributes to COVID-19-associated vascular inflammation and coagulopathy. Rev. Cardiovasc. Med. 2020;21(September (3)):315–319. doi: 10.31083/j.rcm.2020.03.126. [DOI] [PubMed] [Google Scholar]
- 107.Costela-Ruiz V.J., Illescas-Montes R., Puerta-Puerta J.M., Ruiz C., Melguizo-Rodriguez L. SARS-CoV-2 infection: the role of cytokines in COVID-19 disease. Cytokine Growth Factor Rev. 2020;54(August):62–75. doi: 10.1016/j.cytogfr.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Smith J.C., et al. Cigarette smoke exposure and inflammatory signaling increase the expression of the SARS-CoV-2 receptor ACE2 in the respiratory tract. Dev. Cell. 2020;53(June (5)):514–529. doi: 10.1016/j.devcel.2020.05.012. e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Pairo-Castineira E., et al. Genetic mechanisms of critical illness in Covid-19. Nature. 2020;(December (11)) doi: 10.1038/s41586-020-03065-y. [DOI] [PubMed] [Google Scholar]
- 110.Murira A., Lamarre A. Type-I Interferon Responses: From Friend to Foe in the Battle against Chronic Viral Infection. Front. Immunol. 2016;7:609. doi: 10.3389/fimmu.2016.00609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Gorman J.A., et al. The TYK2-P1104A autoimmune protective variant limits coordinate signals required to generate specialized t cell subsets. Front. Immunol. 2019;10:44. doi: 10.3389/fimmu.2019.00044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Satarker S., Tom A.A., Shaji R.A., Alosious A., Luvis M., Nampoothiri M. JAK-STAT pathway inhibition and their implications in COVID-19 therapy. Postgrad. Med. 2020;(December (16)):1–19. doi: 10.1080/00325481.2020.1855921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Gallardo-Vera F., et al. Vanadium pentoxide prevents NK-92MI cell proliferation and IFNgamma secretion through sustained JAK3 phosphorylation. J. Immunotoxicol. 2016;13(1):27–37. doi: 10.3109/1547691X.2014.996681. [DOI] [PubMed] [Google Scholar]
- 114.Zatterale F., et al. Chronic adipose tissue inflammation linking obesity to insulin resistance and type 2 diabetes. Front. Physiol. 2019;10:1607. doi: 10.3389/fphys.2019.01607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Burhans M.S., Hagman D.K., Kuzma J.N., Schmidt K.A., Kratz M. Contribution of adipose tissue inflammation to the development of type 2 diabetes mellitus. Compr. Physiol. 2018;9(December (1)):1–58. doi: 10.1002/cphy.c170040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Kuo C.S., et al. Inhibition of serine protease activity protects against high fat diet-induced inflammation and insulin resistance. Sci. Rep. 2020;10(February (1)):1725. doi: 10.1038/s41598-020-58361-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Fallahi P., et al. CXCL8 and CXCL11 chemokine secretion in dermal fibroblasts is differentially modulated by vanadium pentoxide. Mol. Med. Rep. 2018;18(August (2)):1798–1803. doi: 10.3892/mmr.2018.9121. [DOI] [PubMed] [Google Scholar]
- 118.Fallahi P., et al. Induction of Th1 chemokine secretion in dermal fibroblasts by vanadium pentoxide. Mol. Med. Rep. 2018;17(May (5)):6914–6918. doi: 10.3892/mmr.2018.8712. [DOI] [PubMed] [Google Scholar]
- 119.Cohen M.D., McManus T.P., Yang Z., Qu Q., Schlesinger R.B., Zelikoff J.T. Vanadium affects macrophage interferon-gamma-binding and -inducible responses. Toxicol. Appl. Pharmacol. 1996;138(May (1)):110–120. doi: 10.1006/taap.1996.0104. [DOI] [PubMed] [Google Scholar]
- 120.Jung K.H., et al. Short-term exposure to PM2.5 and vanadium and changes in asthma gene DNA methylation and lung function decrements among urban children. Respir. Res. 2017;18(April (1)):63. doi: 10.1186/s12931-017-0550-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Di Paolo N.C., Shayakhmetov D.M. Interleukin 1alpha and the inflammatory process. Nat. Immunol. 2016;17(July (8)):906–913. doi: 10.1038/ni.3503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Parkinson N., et al. Dynamic data-driven meta-analysis for prioritisation of host genes implicated in COVID-19. Sci. Rep. 2020;10(December (1)):22303. doi: 10.1038/s41598-020-79033-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Thompson K.H. Vanadium and diabetes. Biofactors. 1999;10(1):43–51. doi: 10.1002/biof.5520100105. [DOI] [PubMed] [Google Scholar]
- 124.Onodera Y., Teramura T., Takehara T., Shigi K., Fukuda K. Reactive oxygen species induce Cox-2 expression via TAK1 activation in synovial fibroblast cells. FEBS Open Bio. 2015;5:492–501. doi: 10.1016/j.fob.2015.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Scialo F., et al. ACE2: the major cell entry receptor for SARS-CoV-2. Lung. 2020;198(December (6)):867–877. doi: 10.1007/s00408-020-00408-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Bryant J.W., Shariat-Madar Z. Human plasma kallikrein-kinin system: physiological and biochemical parameters. Cardiovasc. Hematol. Agents Med. Chem. 2009;7(July (3)):234–250. doi: 10.2174/187152509789105444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Meini S., et al. Understanding the pathophysiology of COVID-19: could the contact system Be the key? Front. Immunol. 2020;11:2014. doi: 10.3389/fimmu.2020.02014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Garvin M.R., et al. A mechanistic model and therapeutic interventions for COVID-19 involving a RAS-mediated bradykinin storm. Elife. 2020;9(July (7)) doi: 10.7554/eLife.59177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Polidoro R.B., Hagan R.S., de Santis Santiago R., Schmidt N.W. Overview: systemic inflammatory response derived from lung injury caused by SARS-CoV-2 infection explains severe outcomes in COVID-19. Front. Immunol. 2020;11:1626. doi: 10.3389/fimmu.2020.01626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Karnaukhova E. C1-inhibitor: structure, functional diversity and therapeutic development. Curr. Med. Chem. 2021;(August (3)) doi: 10.2174/0929867328666210804085636. [DOI] [PubMed] [Google Scholar]
- 131.Guerrieri N., Cerletti P., De Vincentiis M., Salvati A., Scippa S. Vanadium inhibition of serine and cysteine proteases. Comp Biochem Physiol A Mol Integr Physiol. 1999;122(March (3)):331–336. doi: 10.1016/s1095-6433(99)00014-8. [DOI] [PubMed] [Google Scholar]
- 132.Moulin A., Bell J.H., Pratt R.F., Ringe D. Inhibition of chymotrypsin by a complex of ortho-vanadate and benzohydroxamic acid: structure of the inert complex and its mechanistic interpretation. Biochemistry. 2007;46(May (20)):5982–5990. doi: 10.1021/bi6025209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Urwyler P., et al. Treatment of COVID-19 with Conestat Alfa, a regulator of the complement, contact activation and kallikrein-kinin system. Front. Immunol. 2020;11:2072. doi: 10.3389/fimmu.2020.02072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.van de Veerdonk F.L., et al. Kallikrein-kinin blockade in patients with COVID-19 to prevent acute respiratory distress syndrome. Elife. 2020;9(April (27)) doi: 10.7554/eLife.57555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Carmignani M., Volpe A.R., Grilli A., Boscolo P., Felaco M. Neurohumoral, autacoid and transductional mechanisms in the cardiovascular effects of vanadate: histochemical correlations. G. Ital. Med. Lav. Ergon. 1997;19(January–March (1)):20–22. [Online]. Available: https://www.ncbi.nlm.nih.gov/pubmed/9377736. Meccanismi neuroumorali, autacoidali e trasduzionali negli effetti cardiovasali di vanadato: correlati istochimici. [PubMed] [Google Scholar]
- 136.Boscolo P., et al. Renal toxicity and arterial hypertension in rats chronically exposed to vanadate. Occup. Environ. Med. 1994;51(July (7)):500–503. doi: 10.1136/oem.51.7.500. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.