Abstract
Other than respiratory disease, patients with coronavirus disease 2019 (COVID-19) commonly have cardiovascular manifestations, which are recognized as significant risk factors for increased mortality. COVID-19 patients may present with a wide spectrum of clinical presentations ranging from asymptomatic heart disease detected incidentally by cardiac investigations (troponin, BNP, and imaging) to cardiogenic shock and sudden cardiac death. In this broad clinical course, advanced imaging plays an important role in the diagnosis of different patterns of myocardial injury, risk stratification of COVID-19 patients, and in detecting potential cardiac side effects of the current treatments and vaccines against the severe acute respiratory syndrome.
Keywords: COVID-19, Myocardial injury, Cardiovascular magnetic resonance (CMR), Myocarditis, Pulmonary embolism
Key points
-
•
Cardiovascular manifestations are common in COVID-19 patients with prognostic implications.
-
•
Most COVID-19–related cardiovascular complications are mainly the consequences of myocardial injury, although the pathophysiological mechanisms are still under investigation.
-
•
Common COVID-19-related cardiovascular manifestations include acute coronary syndromes, myo/pericarditis, pulmonary embolism, and heart failure.
-
•
Advanced cardiac imaging plays an essential role in the diagnosis of cardiac complications and the risk stratification of COVID-19 patients.
Introduction
At the end of 2019, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was identified as a novel cause of respiratory infection in Wuhan, which spread rapidly resulting in the global pandemic of coronavirus disease (COVID-19).1 It is widely recognized that patients with cardiovascular (CV) risk factors, or established CV disease (CVD), are at increased risk of developing severe COVID-19, and those with myocardial injury have a worse prognosis.2, 3, 4, 5 Studies have shown that COVID-19 can cause a broad spectrum of CV complications including myocarditis,6 myocardial infarction (MI),7 stress cardiomyopathy,8 heart failure (HF),9 arrhythmias,10 thromboembolic events,11 and cardiogenic shock.12 Advanced cardiac imaging is a reliable diagnostic tool for prompt diagnosis, risk stratification, monitoring, and management of patients with COVID-19–related CV manifestations. This review is focused on the role of multimodality imaging in identifying cardiac involvement in COVID-19 and the potential CV side effects of the available treatment and vaccines against SARS-CoV-2.
Pathogenesis of COVID-19–associated CV complications
Although the pathophysiological mechanisms of the myocardial injury caused by SARS-CoV-2 have not been entirely elucidated, multiple factors have been hypothesized to contribute directly or indirectly to the development of CV complications.
-
a.
Direct virus-mediated cytotoxicity. Although this theory is supported by an autopsy series, which confirmed the detection of viral genome detection within the myocardium,13 , 14 in clinical practice, the histologic evidence of myocardial injury in COVID-19 is limited. Low loads of viral genome were detected in histology specimens in 5 cases out of 104 patients undergoing endomyocardial biopsy (EMB) for myocarditis or unexplained HF during the COVID-19 pandemic.15 However, there was no evidence of myocardial injury detected in the autopsy of a patient with COVID-19 and acute respiratory distress syndrome, who died of sudden cardiac death, challenging the theory of a direct cardiotoxic effect of SARS-CoV-2.16
-
b.
Dysregulation of renin-angiotensin-aldosterone system. There is evidence that SARS-CoV-2 infection might cause downregulation of angiotensin-converting enzyme 2 (ACE2),17 which has a cardioprotective role as an antifibrotic, antioxidating, and anti-inflammatory factor.17 In addition, the connection of the viral protein S to human ACE2 can downregulate the degradation of angiotensin 2 to angiotensin 1-7.18 The accumulation of angiotensin 2 might activate the p38 Mitogen-Activated Protein Kinase (MAPK) pathway promoting thrombotic events19 and might also induce the production of reactive oxygen species (ROS) causing myocardial injury.20
-
c.
Endothelial cell damage and thromboinflammation. The direct invasion of the vascular endothelial cells via ACE2 receptors may result in inflammation and endothelial dysfunction contributing to thrombosis. There is early histologic evidence of direct toxic effects to endothelial cells caused by SARS-CoV-2.21 In this case series, there was evidence of lymphocytic endotheliitis in the lungs, heart, and kidneys in a patient who died from COVID-19 and multiorgan failure. This was also observed in the lungs, heart, kidneys, and liver in a patient who died with COVID-19, multisystem inflammatory response (MIS), and MI with ST elevation.
-
d.
Dysregulation of immune response. It is hypothesized that the SARS-CoV-2 infection can induce an excessive activation of immune cells and inflammatory response causing a cytokine storm. The overproduction of proinflammatory cytokines can lead to endothelial dysfunction and the activation of complement pathways, platelets, von Willebrand factor, and tissue factor; increasing the risk of thrombosis in the circulation including in the coronary system, and therefore increasing the risk of an acute coronary syndrome.22 , 23 SARS-CoV-2 infection can also promote a disproportionate production of factor VIII and neutrophil extracellular traps, which can facilitate the development of thrombotic events.22, 23, 24 Apart from type I MI, the exaggerated systemic inflammatory response increases the metabolic demand causing myocardial mismatch in oxygen demand and supply and, as a consequence, a type II MI.23, 24, 25
-
e.
Hypoxic injury. Hypoxia caused by SARS-CoV-2 infection induces intracellular acidosis and the release of ROS from mitochondria in cardiomyocytes, which destroys the cell membrane contributing to cardiomyocyte apoptosis.4 , 26
-
f.
Cardiovascular side effects of drugs and vaccines. It is widely known that antiretroviral therapy and other drugs used in the management of COVID-19 patients (azithromycin, tocilizumab, chloroquine, and hydroxychloroquine) can induce arrhythmias, or interact with some CV treatments.27 In addition, cases of thromboembolic events have been reported after ChAdOx1 nCov-19/AZD1222 (AstraZeneca COVID-19 vaccine) and Ad26.COV2.S (Janssen COVID-19 vaccine)28 , 29 vaccinations. More recently, there is a potential association of the mRNA vaccines, BNTb162b (Pfizer) and mRNA-1273 (Moderna) with myocarditis.30 It is currently believed that the thrombotic events have been associated with autoantibodies directed against the platelet factor 4 (PF4) antigen.31
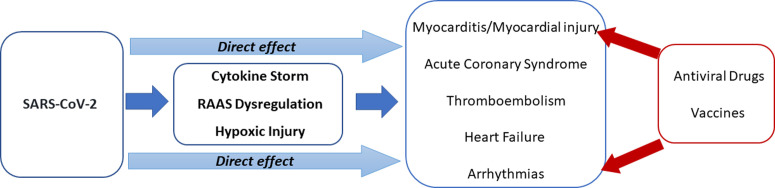
The underlying pathophysiological mechanisms causing COVID-19–related CV complications are presented in Fig. 1 .
Fig. 1.
The pathophysiological interaction between cardiovascular system and COVID-19 showing the potential underlying mechanisms in the development of cardiovascular complications. COVID-19, coronovirus disease 2019; RAAS, renin-angiotensin-aldosterone system; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
The role of imaging techniques in diagnosis, prognosis, and management of COVID-19–related CV complications
There is increasing evidence that patients with pre-existing CVD are at an increased risk of developing acute myocardial injury, with high troponin levels associated with a poorer prognosis.2 , 3 Studies show that 12% to 15% of hospitalized COVID-19 patients had an elevated troponin suggestive of myocardial damage, whereas up to 31% with severe COVID-19 presentation had cardiac involvement.32 , 33 Another study with 112 patients with COVID-19 showed that troponin levels were mostly normal at admission. Troponin increased during hospitalization in 37.5% of the patients, especially in those who died, whereas typical signs of myocarditis were not detected on echocardiography.34 These findings suggested that myocardial injury might be the consequence of the systemic inflammatory response rather than the direct invasion and damage by SARS-CoV-2.
Transthoracic Echocardiography
In a study of 305 hospitalized patients with COVID 19, myocardial injury (defined by elevated troponin levels) was observed in 190 patients.35 Echocardiographic abnormalities, including global or regional left ventricular (LV) wall motion abnormalities, LV diastolic dysfunction, right ventricular (RV) dysfunction, and pericardial effusions were noted in almost two-thirds of patients with myocardial injury. In the same study, the detection of structural abnormalities by transthoracic echocardiography (TTE) was an independent predictive factor of mortality in the subgroup with myocardial injury. An international prospective study of 1216 patients from 69 countries with clinical indication for TTE revealed the spectrum of echocardiographic abnormalities.36 Abnormal TTE was found in 55%, including LV and RV abnormalities at 39% and 33% respectively, whereas the prevalence of echocardiographic abnormalities was slightly lower (46%) in the subgroup with no pre-existing CVD. The LV abnormalities were predominantly nonspecific in nature and the mechanism of dysfunction was often not identified unless the TTE findings were suggestive of acute MI (3%), myocarditis (3%), and stress cardiomyopathy (2%). In the same study, severe LV, RV, or biventricular systolic dysfunction was observed in 14% and cardiac tamponade occurred in 1%. In contrast, another study showed that RV dilatation and systolic dysfunction were more common (39%) compared with LV systolic dysfunction (10%) in 100 hospitalized patients with COVID-19 37. The most common echocardiographic findings among those with subsequent clinical deterioration were worsening RV and LV function (12 and 5 patients respectively).
Myocardial infarction, myocarditis, or acute pulmonary hypertension might be the cause of isolated RV dysfunction in COVID-19 patients.38 Acute cor pulmonale precipitated by acute pulmonary embolism (PE) or adult respiratory distress syndrome has also been reported.39, 40, 41 In addition, the implementation of positive end-expiratory pressure in the respiratory support of critically ill COVID-19 patients can contribute to RV dysfunction.42 Interestingly, RV dysfunction has been shown to correlate with poor prognosis in COVID-19.38 , 42, 43, 44
Cases of pericardial effusion in the context of pericarditis detected on TTE, either in association with myocarditis or as an isolated presentation, along with rare cases of pericardial tamponade in patients with COVID-19 have also been reported.45, 46, 47, 48, 49 Although there are no studies currently investigating the prevalence of pericardial involvement in COVID-19, the diagnosis of pericarditis should be considered in COVID-19 patients presenting with chest pain, ST elevation in the electrocardiogram (ECG), and normal coronary arteries.
Speckle tracking echocardiography is a novel echocardiographic technique. It allows quantification of myocardial deformation, providing early detection of cardiac dysfunction and plays an important prognostic role in HF and patients with end-stage renal disease.50 , 51 In addition, impaired LV and RV longitudinal strain are suggestive of early ventricular dysfunction with prognostic value in COVID-19 patients.44 , 52 A specific pattern of prominent longitudinal strain reduction of the basal LV segments has been described in COVID-19 patients suggesting that these areas might be vulnerable to myocardial injury during SARS-CoV-2 infection.53
Cardiovascular Magnetic Resonance
Cardiovascular magnetic resonance (CMR) imaging is increasingly recognized as an essential tool in the assessment of COVID-19–induced myocardial injury because of the unique capability of CMR for noninvasive tissue characterization. Fig. 2 shows the specific CMR techniques used for the characterization of tissue damage in clinical practice. Guidelines have been published for safe CMR scanning during the COVID-19 pandemic54 , 55 and the Society for Cardiovascular Magnetic Resonance recommends a specific protocol to perform in patients with suspected COVID-19 cardiac involvement.56 The primary advantage of CMR is the ability to differentiate between ischemic and nonischemic pathologies.57 Although EMB remains the gold standard for the diagnosis of acute myocarditis, its use in routine clinical practice is limited because of low diagnostic accuracy and periprocedural risks. The Lake Louise Criteria (LLC) were initially established in 2009 using specific CMR techniques, including T2-weighted sequences, early and late gadolinium enhancement sequences (EGE and LGE, respectively), for the diagnosis of acute myocarditis58; expanding them to the use of parametric mapping in the updated version.59 Therefore, CMR represents a robust noninvasive technique for the diagnosis of COVID-19–related myocarditis. CMR typically shows diffuse myocardial edema, noninfarct patterns of LGE, and increased signal on T2-weighted imaging, T1- and T2-mapping sequences60 (Fig. 3 ). CMR can also confirm the involvement of pericardium revealing edematous and enhancing pericardial layers suggestive of pericardial inflammation. Moreover, it has incremental value in differentiating COVID-19–associated stress (Takotsubo) cardiomyopathy and MI in patients with SARS-CoV-2 infection presenting with chest pain, high troponin, and ECG abnormalities. The absence of LGE in dysfunctional LV segments with the evidence of edema allows the differentiation from MI when transmural or subendocardial LGE with a coronary distribution is detected (Fig. 4 ).61 There is also an increased incidence of stress cardiomyopathy in patients without COVID-19 during the pandemic compared with the prepandemic era indicating the intense emotional stress.62
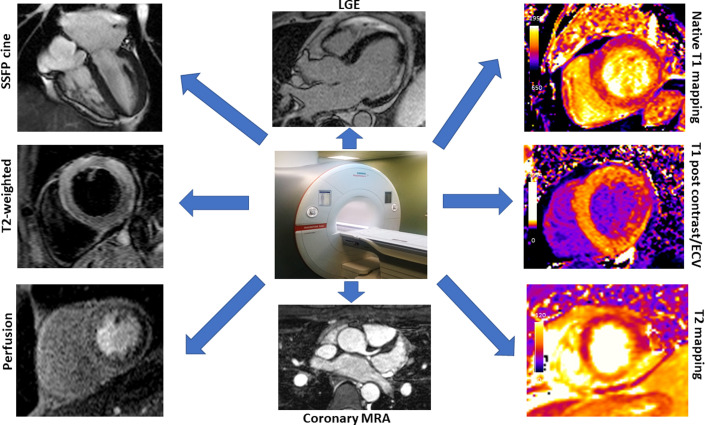
Fig. 2.
Specific CMR sequences for the characterization of tissue damage in clinical practice. Steady-state free precession (SSFP) MRI cine: for the assessment of wall motion. T2-weighted imaging: for the assessment of edema. Perfusion imaging: for the assessment of inducible ischemia. Coronary MR angiography (MRA): for the evaluation of the origins of the coronary arteries. Late gadolinium enhancement (LGE): marker of acute myocardial injury, fibrosis, or infarction. Native T1 and T1 postcontrast/extracellular volume (ECV) mapping: for the evaluation of diffuse fibrosis, or infiltration. T2 mapping: a marker of myocardial edema.
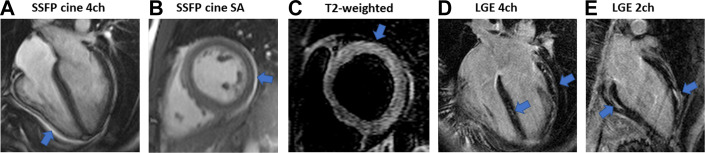
Fig. 3.
CMR findings in a 21-year-old COVID-19 patient with acute myopericarditis who presented with chest pain, high troponin levels (1143 ng/L, normal range <14 ng/L), and global mild left ventricular systolic dysfunction on the bedside transthoracic echocardiography (TTE). (A, B) Steady-state free precession (SSFP) MRI cine 4 chamber (4ch) and short axis (SA), the arrows show the small pericardial effusion. (C) T2-weighted imaging showing slightly increased signal in the basal anterior wall (arrow) extending to basal anterolateral wall suggestive of myocardial edema. (D, E) Extensive patchy subepicardial and midwall late gadolinium enhancement (LGE) in the basal inferior, midinferoseptal, anterolateral, and anterior walls (arrows).
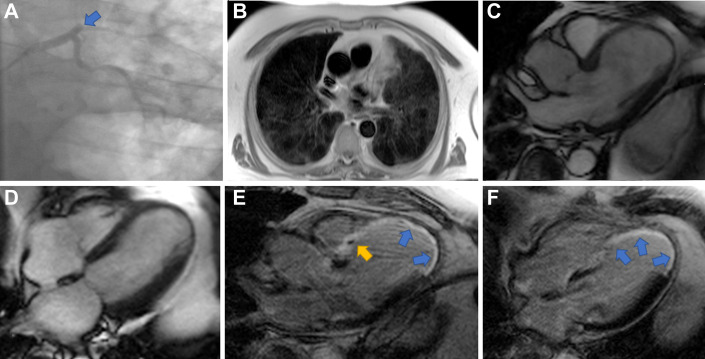
Fig. 4.
Coronary angiogram and CMR in a 69-year-old man who presented with anterior ST-elevation myocardial infarction (STEMI) in the context of COVID-19. CMR performed 25 days after the acute coronary syndrome. (A) Coronary angiogram showing proximal occlusion (arrow) of left anterior descending (LAD) artery with high burden of thrombus. (B) CMR axial Half-Fourier Acquisition Single-shot Turbo Spin Echo (HASTE) showing patchy bilateral lung changes of high signal in keeping with COVID-19. (C, D) Steady-state free precession (SSFP) MRI cine in endsystole showing thinning and akinesia of mid to apical septal walls extending to apical lateral walls and apical cap. (E, F) Late gadolinium enhancement (LGE) imaging showing transmural LGE in the mid to apical septal walls extending to the apical lateral walls and apical cap (blue arrows). In addition, there is partial extension of enhancement into the basal septal segments with evidence of microvascular obstruction in the basal anteroseptum (yellow arrow).
CMR abnormalities have been reported in patients who have recently recovered from COVID-19, though most of them have been nonspecific and it is unclear whether they were pre-existing findings and thus unrelated to COVID-19. A study of 100 patients investigated the outcomes of CMR in patients recently recovered from COVID-19, including 18 patients with asymptomatic SARS-CoV-2 infection, 49 patients with mild to moderate symptoms, and 33 patients with severe symptoms requiring hospitalization. Comparisons were made with healthy controls as well as with risk factor-matched controls.63 CMR was performed 2 to 3 months after the initial positive COVID-19 test. Significant high-sensitivity troponin I (hs-cTnI) elevation at the time of CMR was noted in 5% of the patients. Notably, high myocardial native T1 (73%), elevated myocardial native T2 (60%), myocardial LGE (32%), and pericardial LGE (22%) were detected in patients who recovered from COVID-19, whereas CMR abnormalities found in the risk-factor matched controls were less frequent including high native T1 and T2 (40% and 9%, respectively), myocardial LGE (17%), and pericardial LGE (15%). There was also mild LV and RV systolic dysfunction compared with healthy and risk-factor matched controls (LVEF 56% vs 60% and 61%, respectively; RVEF 56% vs 60% and 59% respectively). Similarly, a smaller study of 26 patients who remained symptomatic after recovery from COVID-19 found myocardial edema in 54%, LGE 31%, and decreased RV functional parameters in the subgroup with positive conventional CMR findings.64 A larger study included 148 patients with severe COVID-19 [all requiring hospital admission, 48 of whom (32%) requiring ventilatory support] and troponin elevation who underwent CMR at a median of 68 days after discharge.65 LV function was normal in 89%, myocarditis-like scar noted in 26%, infarction and/or ischemia in 22%, and dual pathology in 6%. However, whether these abnormalities represent de novo COVID-19–associated changes or pre-existed in the context of clinically silent disease remains unclear. Importantly, the extent of these abnormalities was quite limited and with minimal functional consequences.
CMR findings have also been reported among athletes who have recovered from COVID-19.66, 67, 68 In a study of 145 university student-athletes who were asymptomatic or experienced mild to moderate symptoms, CMR was performed at a median of 15 days after the positive COVID-19 tests.67 CMR findings consistent with updated LLC for myocarditis were found in only 2 patients (1.4%), whereas 40 patients (27.6%) had small nonspecific foci of LGE. A smaller study of 26 competitive collegiate athletes who underwent CMR examination 11 to 53 days after diagnosis of COVID-19 showed that 4 athletes (15%) met the updated LLC for clinically suspected myocarditis and 8 athletes (30%) had nonspecific LGE.66 In the same study, none of the athletes had elevated serum troponin I levels or diagnostic ST/T wave changes on ECG and biventricular size and function were both normal on TTE and CMR.
CMR abnormalities detected in convalescent COVID-19 raised concerns for long-term CV complications even in asymptomatic patients. However, the clinical significance of these findings remains uncertain and further studies are needed to confirm the clinical role of imaging in a long-term setting. The potential role of CMR in screening athletes is debated in the literature.69 The COVID-HEART trial is an ongoing multicentre UK trial, which will clarify the association of COVID-19–related cardiac involvement with comorbidity, genetics, patient-reported quality of life measures, and functional capacity.70
Table 1 summarizes the studies of TTE and CMR findings in patients with COVID-19.
Table 1.
Studies of imaging findings (TTE/CMR) in patients with COVID-19
| Authors | Study Design | Imaging Modality | Population | Results |
|---|---|---|---|---|
| Giustino et al,35 2020 | International, multicenter retrospective study Cardiac Injury Research in COVID-19 Registry (CRIC-19) |
TTE | N = 305 hospitalized patients with COVID-19 Age, range (y): 63 (53–73) Male/Female, n: 205/305 |
|
| Dweck et al,36 2020 | Prospective international survey (www.escardio.org/eacvi/surveys) | TTE | N = 1216 hospitalized patients with COVID-19, 69 countries Age, range (y): 62 (52–71) Male/Female, n: 844/365 |
|
| Szekely et al,37 2020 | Prospective observational single-center study | TTE | N = 100 hospitalized patients with COVID-19 Age, mean ± SD (y): 66.1 ± 17.3 Male/Female, n: 63/37 |
|
| Kim et al,43 2020 | Prospective Multicenter Registry | TTE | N = 510 hospitalized patients with COVID-19 Age, mean ± SD (y): 64 ± 14 Male/Female, n: 335/175 |
|
| Li et al,44 2020 | Prospective observational single-center study | TTE | N = 120 hospitalized patients with COVID-19 Age, mean ± SD (y): 61 ± 14 Male/Female, n: 57/63 N = 37 healthy volunteers |
RVLS was a powerful predictor of higher mortality in patients with COVID-19 (HR 1.33; 95% CI, 1.15–1.53; P < .001) The best cut-off value of RVLS for prediction of outcome was −23% (AUC: 0.87; P < .001; sensitivity, 94.4%; specificity, 64.7%). |
| Goerlich et al,53 2020 | Retrospective observational single-center study | TTE | N = 75 hospitalized patients with COVID-19 Cases (n = 39): basal LS <13.9% (absolute value) Controls (n = 36): basal LS >13.9% (absolute value) Age, mean ± SD (y): 61.9 ± 13.5 Male/Female, n: 44/31 |
52% had a reduced basal strain on STE (basal LS 10.0 ± 2.9% vs 16.9 ± 2.3%, P < .001) GLS was significantly lower in COVID-19 cases vs controls (13.9 ± 4.1% vs 18.8 ± 2.7%, P < .001) LVEF (%) was similar between groups (62.5 [55.0–64.4] vs 57.5 [47.5–62.5], P = .11 |
| Puntmann et al,63 2020 | Prospective observational single-center study | CMR | N = 100 patients recovered from COVID-19, CMR 71 (64–92) days from positive test Age, mean ± SD (y): 49 ± 14 Male/Female:53/47 N = 50 age and sex matched healthy controls N = 57 risk factor matched controls |
Patients recovered from COVID-19 had lower LVEF and RVEF, higher LVEDVi, and raised native T1 and T2 values compared with both control groups. Greater proportions of patients with ischemic (32% vs 17%) and nonischemic (20% vs 7%) LGE patterns than the risk factor matched control group. There was a greater proportion of cases with pericardial enhancement (22% vs 14%) and pericardial effusion (20% vs 7%) compared with the risk factor matched control group. |
| Huang et al,64 2020 | Retrospective observational single-center study | CMR | N = 26 patients recovered from moderate-severe COVID-19 Age, range (y): 38 (32–45) Male/Female: 10/16 N = 20 age and sex matched healthy controls |
|
| Kotecha et al,65 2021 | Prospective observational multicentre study | CMR | N = 148 recovered COVID-19 patients (moderate-severe COVID-19) Age mean ± SD (y): 64 ± 12 Male/Female: 104/44 N = 40 risk factor matched controls N = 40 healthy volunteers |
|
| Rajpal et al,66 2021 | Case Series (single centre) | TTE, CMR | N = 26 competitive college athletes recovered from COVID-19 (14 asymptomatic, 12 mild symptoms) Age, mean ± SD (y): 19.5 ± 1.5 Male/Female: 15/11 |
Normal biventricular size and function by TTE and CMR None had troponin elevation or diagnostic ST/T wave changes on ECG 4 athletes (15%) met the updated LLC for clinically suspected myocarditis 8 athletes (30%) had nonspecific LGE |
| Starekova et al,67 2021 | Case Series (single centre) | TTE, CMR | N = 145 competitive college athletes recovered from COVID-19 (17% asymptomatic, 49% mild, 28% moderate symptoms) Age, range (y): 20 (17–23) Male/Female: 108/37 |
TTE was unremarkable 2 athletes (1.4%) had myocarditis by LLC, troponin abnormal in the more severe case 40 patients (27.6%) had small nonspecific foci of LGE |
| Gorecka et al,70 2021 COVID-HEART Investigators | Prospective observational multicentre study (COVID-HEART study) | CMR |
Inclusion criteria: hospitalized patient population (age ≥ 18 y), or those recently discharged from hospital (within 28 d after discharge), with a diagnosis of COVID-19 Exclusion criteria: unable or unwilling to consent, contraindication to CMR, pregnancy or breast-feeding Risk factor matched controls: matched on age and CVD risk factors cohort |
Ongoing trial |
AUC, area under the receiver operating characteristic curve; CI, confidence interval; CMR, cardiac magnetic resonance; COVID-19, coronavirus disease 2019; DVT, deep vein thrombosis; ECG, electrocardiogram; ECV, extracellular volume fraction; GLS, global longitudinal strain; HR, hazard ratio; LGE, late gadolinium enhancement; LLC, Lake Louise criteria; LS, longitudinal strain; LV, left ventricular; LVEDVi, left ventricular end-diastolic volume index; LVEF, left ventricular ejection fraction; LVESVi, left ventricular end-systolic volume index; NPs, natriuretic peptides; OR, odds ratio; RV, right ventricular; RVCI, right ventricular cardiac index; RVCO, right ventricular cardiac output; RVEDVi, right ventricular end-diastolic volume index; RVEF, right ventricular ejection fraction; RVESVi, right ventricular end-systolic volume index; RVLS, right ventricular longitudinal strain; RVSV, right ventricular stroke volume; SD, standard deviation; STE, speckle tracking echocardiography; TTE, transthoracic echocardiogram.
Computed Tomography
For patients with active COVID-19 and non-ST-elevation MI, noninvasive diagnostic testing before catheterization was recommended during the pandemic.71 In hemodynamically stable cases with low or intermediate CV risk, computed tomography coronary angiography (CTCa) could be considered as an alternative to invasive coronary angiogram.72 CTCa also provides information on lung parenchyma and pulmonary vessels, while excluding significant coronary artery disease with almost 100% negative predictive value.73
A retrospective study of 100 COVID-19 patients who underwent computed tomography pulmonary angiography (CTPA) showed that 23% had acute pulmonary embolism (PE).74 Those with PE had more severe disease requiring more intensive care admissions and mechanical ventilation (PE vs non-PE: 74% vs 22%, and 65% vs 25%, respectively, P = .001 for both comparisons).74 These findings were confirmed by several studies22 , 40 , 75 highlighting the important diagnostic and prognostic role of CTPA in COVID-19 patients. Moreover, the detection of subepicardial hyperattenuation in contrast-enhanced computed tomography combined with cardiac gating can be useful in the evaluation of COVID-19 patients with suspected myocarditis.76
Cardiovascular effects of drugs and vaccines against SARS-CoV-2: is there a role for cardiac imaging?
Chloroquine and hydroxychloroquine are well-established treatments in non-CV conditions and have now been used in the treatment of COVID-19.77 , 78 Although there are inconsistent data regarding their efficacy, and large, randomized controlled trials are needed to establish their use in clinical practice, they are widely known to cause arrhythmias.79 , 80 Azithromycin used in combination with hydroxychloroquine is known to prolong QT interval.81 A retrospective cohort study of 1438 patients hospitalized with COVID-19 showed that cardiac arrest was more likely in patients receiving both hydroxychloroquine and azithromycin than in patients receiving neither drug.82 In the clinical scenarios of arrhythmia, CMR plays an important role in risk stratification identifying possible pathologic substrates (myocardial edema, ischemia, fibrosis) in patients with new-onset arrhythmias.
Cases of thromboembolic events associated with thrombocytopenia have been reported after vaccination with both ChadOx1 nCoV-19/AZD1222 (AstraZeneca COVID-19 vaccine) and Ad26.COV2.S (Janssen COVID-19 vaccine).28 , 29
Cases of myocarditis and pericarditis have also been reported after mRNA vaccines, BNTb162b (Pfizer) and mRNA-1273 (Moderna),30 , 83, 84, 85 but a similar pattern of these complications have not been reported after Ad26.COV2.S (Janssen COVID-19 vaccine). However, presentations were mild, none with evidence of acute SARS-CoV-2 infection, nor met criteria for MIS. The CMR findings were consistent with myocarditis in all cases highlighting the role of CMR in detecting pathology even in less aggressive diseases. Fig. 5 shows the CMR findings of a patient admitted with acute myocarditis 1 week after mRNA-1273 vaccination.
Fig. 5.
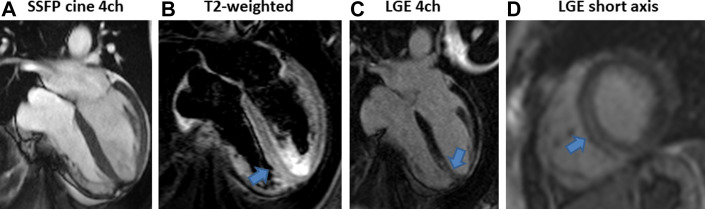
CMR findings in a 46-year-old lady with acute myocarditis 1 week after the second dose of mRNA-1273 (Moderna vaccine), who was admitted with chest pain, high troponin T levels (113 ng/L, cut off 14 ng/L), and inferolateral T-wave inversion on electrocardiogram (ECG). Computed tomography pulmonary angiogram (CTPA) was negative for pulmonary embolism. (A) Steady-state free precession (SSFP) MRI cine 4 chamber (4ch) showing mild impairment of left ventricular systolic function. (B) T2-weighted imaging showing high signal suggestive of myocardial edema (arrow) in the apical septum. (C, D) Late gadolinium enhancement (LGE) imaging 4 chanber (4ch) and short axis showing midwall LGE in the apical septum matching the relevant myocardial edema.
Summary
Advanced cardiac imaging plays an essential role in the diagnosis, prognosis, and management of COVID-19 patients. It provides noninvasive tools for the recognition of the broad spectrum of CV complications in COVID-19, along with the potential CV side effects of the available vaccinations against SARS-CoV-2. However, the clinical significance of the imaging findings after COVID-19 recovery remains unclear and needs further study in large, randomized clinical trials.
Clinics care points
-
•
Cardiovascular (CV) disease increases the risk of severe COVID-19 presentation.
-
•
CV manifestations may occur during the acute phase of COVID-19, and cardiac involvement appears to be correlated with COVID-19 severity.
-
•
Evidence suggests sustained CV involvement is uncommon. Even if patients have scar, structural and functional abnormalities, there is no difference to carefully matched control groups in the entire COVID-19 severity spectrum.
-
•
CV involvement in athletes after COVID-19 is rare.
-
•
There may be a link between mRNA vaccines and myocarditis, but the clinical course seems benign. More evidence is needed.
Acknowledgments
Disclosures
E. Nakou, E. De Garate, K. Liang, and M. Williams have nothing to disclose. C. Bucciarelli-Ducci is the chief executive officer (CEO; part-time) of the Society for Cardiovascular Magnetic Resonance (SCMR); she received speakers fees from Circle Cardiovascular Imaging. D.J. Pennell receives research support from Siemens, and speakers fees from Circle, Bayer, and Chiesi.
References
- 1.World Health Organization. Coronavirus Disease 2019 (COVID-19) Situation Report-51. 2020. Available at: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200311-sitrep-51-covid-19.pdf. Accessed 28 January, 2021.
- 2.Bonow R.O., Fonarow G.C., O’Gara P.T., et al. Association of coronovirus disease 2019 (COVID-19) with myocardial injury and mortality. JAMA Cardiol. 2020;5:751–753. doi: 10.1001/jamacardio.2020.1105. [DOI] [PubMed] [Google Scholar]
- 3.Driggin E., Madhavan M.V., Bikdeli B., et al. Cardiovascular considerations for patients, health care workers, and health systems during the COVID-19 pandemic. J Am Coll Cardiol. 2020;75:2352–2371. doi: 10.1016/j.jacc.2020.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li B., Yang J., Zhao F., et al. Prevalence and impact of cardiovascular metabolic disease on COVID-19 in China. Clin Res Cardiol. 2020;109:531–538. doi: 10.1007/s00392-020-01626-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhou F., Yu T., Du R., et al. Clinical course and risk factor for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hu H., Ma F., Wei X., et al. Coronavirus fulminant myocarditis saved with glucocorticoid and human immunoglobulin. Eur Heart J. 2021;42:206. doi: 10.1093/eurheartj/ehaa190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bangalore S., Sharma A., Slotwiner A., et al. ST-segment elevation in patients with covid-19- A case series. N Engl J Med. 2020;382:2478–2480. doi: 10.1056/NEJMc2009020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tsao C.W., Strom J.B., Chang J.D., et al. COVID-19-Associated stress (Takotsubo) cardiomyopathy. Circ Cardiovasc Imaging. 2020;13:e011222. doi: 10.1161/CIRCIMAGING.120.011222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen T., Wu D., Chen H., et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. 2020;368 doi: 10.1136/bmj.m1091. m1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chang W.T., Toh H.S., Liao C.T., et al. Cardiac involvement of COVID-19: a comprehensive review. Am J Med Sci. 2020;361:14–22. doi: 10.1016/j.amjms.2020.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lax S.F., Skok K., Zechner P., et al. Pulmonary arterial thrombosis in COVID-19 with fatal outcome: results from a prospective, single-center, clinicopathologic case series. Ann Intern Med. 2020;173:350–361. doi: 10.7326/M20-2566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tavazzi G., Pellegrini C., Maurelli M., et al. Myocardial localization of coronavirus in COVID-19 cardiogenic shock. Eur J Heart Fail. 2020;22:911–915. doi: 10.1002/ejhf.1828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lindner D., Fitzek A., Brauninger H., et al. Association of cardiac infection with SARS-CoV-2 in confirmed COVID-19 autopsy cases. JAMA Cardiol. 2020;5:1281–1285. doi: 10.1001/jamacardio.2020.3551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Puelles V.G., Lütgehetmann M., Lindenmeyer M.T., et al. Multiorgan and renal tropism of SARS-CoV-2. N Engl J Med. 2020;383:590–592. doi: 10.1056/NEJMc2011400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Escher F., Pietsch H., Aleshcheva G., et al. Detection of viral SARS-CoV-2 genomes and histopathological changes in endomyocardial biopsies. ESC Heart Fail. 2020;7:2440–2447. doi: 10.1002/ehf2.12805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xu Z., Shi L., Wang Y., et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Oudit G.Y., Kassiri Z., Jiang C., et al. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur J Clin Invest. 2009;39:618–625. doi: 10.1111/j.1365-2362.2009.02153.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vaduganathan M., Vardeny O., Michel T., et al. Renin-angiotensin-aldosterone system inhibitors in patients with covid-19. N Engl J Med. 2020;382:1653–1659. doi: 10.1056/NEJMsr2005760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Grimes J.M., Grimes K.V. p38 MAPK inhibition: a promising therapeutic approach for COVID-19. J Mol Cell Cardiol. 2020;144:63–65. doi: 10.1016/j.yjmcc.2020.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Violi F., Pastori D., Pignatelli P., et al. SARS-CoV-2 and myocardial injury: a role for Nox2? Intern Emerg Med. 2020;15:755–758. doi: 10.1007/s11739-020-02348-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Varga Z., Flammer A.J., Steiger P., et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Helms J., Tacquard C., Severac F., et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Med. 2020;46:1089–1098. doi: 10.1007/s00134-020-06062-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee C.C.E., Ali K., Connell D., et al. COVID-19-Associated cardiovascular complications. Diseases. 2021;9:47. doi: 10.3390/diseases9030047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Panigada M., Bottino N., Tagliabue P., et al. Hypercoagulability of COVID-19 patients in intensive care unit: a report of thromboelastography findings and other parameters of hemostasis. J Thromb Haemost. 2020;18:1738–1742. doi: 10.1111/jth.14850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lala A., Johnson K., Januzzi J.L., et al. Prevalence and impact of myocardial injury in patients hospitalized with COVID-19 infection. J Am Coll Cardiol. 2020;76:533–546. doi: 10.1016/j.jacc.2020.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Catapano F., Marchitelli L., Cundari G., et al. Role of advanced imaging in COVID-19 cardiovascular complications. Insights Imaging. 2021;12:28. doi: 10.1186/s13244-021-00973-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nishiga M., Wang D.W., Han Y., et al. COVID-19 and cardiovascular disease: from basic mechanisms to clinical perspectives. Nat Rev Cardiol. 2020;17:543–558. doi: 10.1038/s41569-020-0413-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schultz N.H., Sørvoll I.H., Michelsen A.E., et al. Thrombosis and thrombocytopenia after ChAdOx1 nCoV-19 vaccination. N Engl J Med. 2021;384:2124–2130. doi: 10.1056/NEJMoa2104882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.See I., Su J.R., Lale A., et al. Case reports of cerebral venous sinus thrombosis with thrombocytopenia after Ad26.COV2.S vaccination, March 2 to April 21, 2021. JAMA. 2021;325:2448–2456. doi: 10.1001/jama.2021.7517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.EMA. Meeting highlights from the Pharmacovigilance Risk Assessment Committee (PRAC) 3-6 May 2021 Internet Document : 7 May 2021. https://www.ema.europa.eu/en/news/meeting-highlights-pharmacovigilance-risk-assessment-committee-prac-3-6-may-2021.
- 31.Greinacher A., Thiele T., Warkentin T.E., et al. Thrombotic thrombocytopenia after ChAdOx1 nCov-19 vaccination. N Engl J Med. 2021;384:2092–2101. doi: 10.1056/NEJMoa2104840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.EMA. Meeting highlights from the Pharmacovigilance Risk Assessment Committee (PRAC) 3-6 May 2021 Internet Document : 7 May 2021. Available at: https://www.ema.europa.eu/en/news/meeting-highlights-pharmacovigilance-risk-assessment-committee-prac-3-6-may-2021.
- 33.Wang D., Hu B., Hu C., et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1601–1669. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Deng O., Hu B., Zhang Y., et al. Suspected myocardial injury in patients with COVID-19: evidence from front-line clinical observation in Wuhan, China. Int J Cardiol. 2020;311:116–121. doi: 10.1016/j.ijcard.2020.03.087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Giustino G., Croft L.B., Stefanini G.G., et al. Characterization of myocardial injury in patients with COVID-19. J Am Coll Cardiol. 2020;76:2043–2055. doi: 10.1016/j.jacc.2020.08.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dweck M.R., Bularga A., Hahn R.T., et al. Global evaluation of echocardiography in patients with COVID-19. Eur Heart J Cardiovasc Imaging. 2020;21:949–958. doi: 10.1093/ehjci/jeaa178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Szekely Y., Lichter Y., Taieb P., et al. Spectrum of cardiac manifestations in COVID-19: a systematic echocardiographic study. Circulation. 2020;142:342–353. doi: 10.1161/CIRCULATIONAHA.120.047971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fayssoil A., Mustafic H., Mansencal N. The right ventricle in COVID-19 patients. Am J Cardiol. 2020;130:166–167. doi: 10.1016/j.amjcard.2020.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Creel-Bulos C., Hockstein M., Amin N., et al. Acute cor pulmonale in critically ill patients with Covid-19. N Engl J Med. 2020;382:e70. doi: 10.1056/NEJMc2010459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Poissy J., Goutay J., Caplan M., et al. Pulmonary embolism in patients with COVID-19: Awareness of an increased prevalence. Circulation. 2020;142:184–186. doi: 10.1161/CIRCULATIONAHA.120.047430. [DOI] [PubMed] [Google Scholar]
- 41.Ullah W., Saeed R., Sarwar U., et al. COVID-19 complicated by acute pulmonary embolism and right-Sided heart failure. JACC Case Rep. 2020;2:1379–1382. doi: 10.1016/j.jaccas.2020.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Himebauch A.S., Yehya N., Wang Y., et al. New or persistent right ventricular systolic dysfunction is associated with worse outcomes in pediatric acute respiratory distress syndrome. Pediatr Crit Care Med. 2020;21:e121–e128. doi: 10.1097/PCC.0000000000002206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kim J., Volodarskiy A., Sultana R., et al. Prognostic utility of right ventricular remodelling over conventional risk stratification in patients with COVID-19. J Am Coll Cardiol. 2020;76:1965–1977. doi: 10.1016/j.jacc.2020.08.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Li Y., Li H., Zhu S., et al. Prognostic value of right ventricular longitudinal strain in patients with COVID-19. JACC Cardiovasc Imaging. 2020;13:2287–2299. doi: 10.1016/j.jcmg.2020.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sauer F., Dagrenat C., Couppie P., et al. Pericardial effusion in patients with COVID-19: case series. Eur Hear J Case Rep. 2020;4(FI1):1–7. doi: 10.1093/ehjcr/ytaa287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Blagojevic N.R., Bosnjakovic D., Vukomanovic V., et al. Acute pericarditis and SARS-CoV-2: case report. Int J Infect Dis. 2020;101:180–182. doi: 10.1016/j.ijid.2020.09.1440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Purohit R., Kanwal A., Pandit A., et al. Acute myopericarditis with pericardial effusion and cardiac tamponade in a patient with COVID-19. Am J Case Rep. 2020;21:e925554. doi: 10.12659/AJCR.925554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Dabbagh M.F., Aurora L., D’Souza P., et al. Cardiac tamponade secondary to COVID-19. JACC Case Rep. 2020;2:1326–1330. doi: 10.1016/j.jaccas.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hua A., O'Gallagher K., Sado D., et al. Life-threatening cardiac tamponade complicating myo-pericarditis in COVID-19. Eur Heart J. 2020;41:2130. doi: 10.1093/eurheartj/ehaa253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pastore M.C., De Carli G., Mandoli G.E., et al. The prognostic role of speckle tracking echocardiography in clinical practice: evidence and reference values from the literature. Heart Fail Rev. 2020 doi: 10.1007/s10741-020-09945-9. Online ahead of print. [DOI] [PubMed] [Google Scholar]
- 51.Jahn L., Kramann R., Marx N., et al. Speckle tracking echocardiography and all-cause and cardiovascular mortality risk in Chronic kidney disease patients. Kidney Blood Press Res. 2019;44:690–703. doi: 10.1159/000501225. [DOI] [PubMed] [Google Scholar]
- 52.Janus S.E., Hajjari J., Karnib M., et al. Prognostic value of left ventricular global longitudinal strain in COVID-19. Am J Cardiol. 2020;131:134–136. doi: 10.1016/j.amjcard.2020.06.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Goerlich E., Gilotra N.A., Minhas A.S., et al. Prominent longitudinal strain reduction of basal left ventricular segments in patients with coronavirus disease-19. J Card Fail. 2020;27:100–104. doi: 10.1016/j.cardfail.2020.09.469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Allen B.D., Wong T.C., Bucciarelli-Ducci C., et al. Society for Cardiovascular Magnetic Resonance (SCMR) guidance for re-activation of cardiovascular magnetic resonance practice after peak phase of the COVID-19 pandemic. J Cardiovasc Magn Reson. 2020;22:58. doi: 10.1186/s12968-020-00654-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Petersen S, Friedrich MG, Leiner T, et al. Cardiovascular magnetic resonance for patients with coronavirus disease 2019 (COVID-19). JACC CVI 2021 in press.
- 56.Kelle S., Bucciarelli-Ducci C., Judd R.M., et al. Society for Cardiovascular Magnetic Resonance (SCMR) recommended CMR protocols for scanning patients with active or convalescent phase COVID-19 infection. J Cardiovasc Magn Reson. 2020;22:61. doi: 10.1186/s12968-020-00656-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mahrholdt H., Wagner A., Judd R.M., et al. Delayed enhancement cardiovascular magnetic resonance assessment of non-ischaemic cardiomyopathies. Eur Heart. 2005;26:1461–1474. doi: 10.1093/eurheartj/ehi258. [DOI] [PubMed] [Google Scholar]
- 58.Friedrich M.G., Sechtem U., Schulz-Menger J., et al. Cardiovascular magnetic resonance in myocarditis: a JACC white paper. JACC. 2009;53:1475–1487. doi: 10.1016/j.jacc.2009.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ferreira V.M., Schulz-Menger J., Holmvang G., et al. Cardiovascular magnetic resonance in nonischemic myocardial inflammation: expert recommendations. J Am Coll Cardiol. 2018;72:3158–3176. doi: 10.1016/j.jacc.2018.09.072. [DOI] [PubMed] [Google Scholar]
- 60.Demirkiran A., Everaars H., Amier R.P., et al. Cardiovascular magnetic resonance techniques for tissue characterization after acute myocardial injury. Eur Heart J Cardiovasc Imaging. 2020;20:723–734. doi: 10.1093/ehjci/jez094. [DOI] [PubMed] [Google Scholar]
- 61.Ghadri J.R., Wittstein I.S., Prasad A., et al. International expert consensus document on takotsubo syndrome (part i): clinical characteristics, diagnostic criteria, and pathophysiology. Eur Heart J. 2018;39:2032–2046. doi: 10.1093/eurheartj/ehy076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jabri A., Kalra A., Kumar A., et al. Incidence of stress cardiomyopathy during the coronavirus disease 2019 pandemic. JAMA Netw Open. 2020;3:e2014780. doi: 10.1001/jamanetworkopen.2020.14780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Puntmann V.O., Carerj M.L., Wieters I., et al. Outcomes of cardiovascular magnetic resonance imaging in patients recently recovered from coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5:1265–1273. doi: 10.1001/jamacardio.2020.3557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Huang L., Zhao P., Tang D., et al. Cardiac involvement in patients recovered from COVID-2019 identified using magnetic resonance imaging. JACC Cardiovasc Imaging. 2020;13:2330–2339. doi: 10.1016/j.jcmg.2020.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kotecha T., Knight D.S., Razvi Y., et al. Patterns of myocardial injury in recovered troponin-positive COVID-19 patients assessed by cardiovascular magnetic resonance. Eur Heart J. 2021;42:1866–1878. doi: 10.1093/eurheartj/ehab075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Rajpal S., Tong M.S., Borchers J., et al. Cardiovascular magnetic resonance findings in competitive athletes recovering from COVID-19 infection. JAMA Cardiol. 2021;6:116–118. doi: 10.1001/jamacardio.2020.4916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Starekova J., Bluemke D.A., Bradham W.S., et al. Evaluation for myocarditis in competitive student athletes recovering from coronavirus disease 2019 with cardiac magnetic resonance imaging. JAMA Cardiol. 2021;6:945–950. doi: 10.1001/jamacardio.2020.7444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Martinez M.W., Tucker A.M., Bloom O.J., et al. Prevalence of inflammatory heart disease among professional athletes with prior COVID-19 infection who received systematic return-to-play cardiac screening. JAMA Cardiol. 2021;6:745–752. doi: 10.1001/jamacardio.2021.0565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Phelan D., Kim J.H., Elliott M.D., et al. Screening of potential cardiac involvement in competitive athletes recovering from COVID-19: an expert consensus statement. JACC Cardiovasc Imaging. 2020;13:2635–2652. doi: 10.1016/j.jcmg.2020.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Gorecka M., McCann G.P., Berry C., et al. Demographic, multi-morbidity and genetic impact on myocardial involvement and its recovery from COVID-19: protocol design of COVID-HEART-a UK, multicentre, observational study. J Cardiovasc Magn Reson. 2021;23:77. doi: 10.1186/s12968-021-00752-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Long B., Brady W.J., Koyfman A., et al. Cardiovascular complications in COVID-19. Am J Emerg Med. 2020;38:1504–1507. doi: 10.1016/j.ajem.2020.04.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Pontone G., Baggiano A., Conte E., et al. “Quadruple rule out” with cardiac computed tomography in COVID-19 patient with equivocal acute coronary syndrome presentation. JACC Cardiovasc Imaging. 2020;13:1854–1856. doi: 10.1016/j.jcmg.2020.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Agricola E., Beneduce A., Esposito A., et al. Heart and lung multimodality imaging in COVID-19. JACC Cardiovasc Imaging. 2020;13:1792–1808. doi: 10.1016/j.jcmg.2020.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Grillet F., Behr J., Calame P., et al. Acute pulmonary embolism associated with COVID-19 pneumonia detected by pulmonary CT angiography. Radiology. 2020;296:E186–E188. doi: 10.1148/radiol.2020201544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Leonard-Lorant I., Delabranche X., Severac F., et al. Acute pulmonary embolism in COVID-19 patients on CT angiography and relationship to D-dimer levels. Radiology. 2020;296:E189–E191. doi: 10.1148/radiol.2020201561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Singh V., Choi A.D., Leipsic J., et al. Use of cardiac CT amidst the COVID-19 pandemic and beyond: North American perspective. J Cardiovasc Comput Tomogr. 2020;15:16–26. doi: 10.1016/j.jcct.2020.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Gautret P., Lagier J.C., Parola P., et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents. 2020;56:105949. doi: 10.1016/j.ijantimicag.2020.105949. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 78.Wang M., Cao R., Zhang L., et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30:269–271. doi: 10.1038/s41422-020-0282-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Roden D.M., Harrington R.A., Poppas A., et al. Considerations for drug interactions on QTc in exploratory COVID-19 treatment. Circulation. 2020;141:e906–e907. doi: 10.1161/CIRCULATIONAHA.120.047521. [DOI] [PubMed] [Google Scholar]
- 80.Mercuro N.J., Yen C.F., Shim D.J., et al. Risk of QT interval prolongation associated with use of hydroxychloroquine with or without concomitant azithromycin among hospitalized patients testing positive for coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5:1036–1041. doi: 10.1001/jamacardio.2020.1834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hancox J.C., Hasnain M., Vieweg W.V., et al. Azithromycin, cardiovascular risks, QTc interval prolongation, Torsade de Pointes, and regulatory issues: a narrative review based on the study of case reports. Ther Adv Infect Dis. 2013;1:155–165. doi: 10.1177/2049936113501816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Rosenberg E.S., Dufort E.M., Udo T., et al. Association of treatment with hydroxychloroquine or azithromycin with in- hospital mortality in patients with COVID-19 in New York state. JAMA. 2020;323:2493–2502. doi: 10.1001/jama.2020.8630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Gargano J.W., Wallace M., Hadler S.C., et al. Use of mRNA COVID-19 vaccine after reports of myocarditis among vaccine recipients: update from the advisory committee on immunization practices - United States, June 2021. MMWR Morb Mortal Wkly Rep. 2021;70:977–982. doi: 10.15585/mmwr.mm7027e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Dickey J.B., Albert E., Badr M., et al. A series of patients with myocarditis following SARS-CoV-2 vaccination with mRNA-1279 and BNT162b2. JACC Cardiovasc Imaging. 2021;14:1862–1863. doi: 10.1016/j.jcmg.2021.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Shaw K.E., Cavalcante J.L., Han B.K., et al. Possible association between COVID-19 vaccine and myocarditis: clinical and CMR findings. JACC Cardiovasc Imaging. 2021;14:1856–1861. doi: 10.1016/j.jcmg.2021.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]