Abstract
Introduction
Bioelectrical impedance analysis (BIA) can be used to estimate Fat-Free Mass Index (FFMI). However, the use of directly measured BIA variables, such as phase angle (PhA), has gained attention. The frequency of low FFMI and PhA and its associations with exercise capacity and health-related quality of life (HRQL) in patients with idiopathic pulmonary fibrosis (IPF) have been scarcely studied.
Objectives
To investigate the frequency of low FFMI and PhA and their associations with exercise capacity and HRQL in patients with IPF.
Methods
Patients underwent assessment of lung function, body composition, exercise capacity by the 6 min walk distance (6MWD), and HRQL by the Medical Outcomes Study Short-Form 36-item Questionnaire (SF-36). Patients were classified as presenting normal or low PhA or FFMI, accordingly to the 10th percentiles of age-sex-body mass index (BMI)-specific reference values.
Results
98 patients (84 males, age: 68±8 years, forced vital capacity: 64%±18%predicted) were included. 24 patients presented low PhA. They were characterised by worse lung function, exercise capacity and HRQL compared with patients with normal PhA. 10 patients presented low FFMI, but despite differences in body composition, no differences were found between these patients and patients with normal FFMI. In a single regression analysis, age, lung function and body composition variables (except FFMI) were related to 6MWD and SF-36 Physical Summary Score (R²=0.06–0.36, p<0.05). None of the variables were related to SF-36 Mental Summary Score.
Conclusion
One-fourth of the patients with IPF with normal to obese BMI present abnormally low PhA. Patients classified as low PhA presented worse lung function, exercise capacity and HRQL.
Keywords: interstitial fibrosis, exercise, clinical epidemiology, equipment evaluations
Key messages.
What is the key question?
What is the frequency of abnormal low phase angle (PhA) and fat-free mass index in patients with idiopathic pulmonary fibrosis (IPF)? Are these body composition variables associated with lung function, exercise capacity and health-related quality of life (HRQL) in this population?
What is the bottom line?
One-fourth of the patients with IPF with normal to obese body mass index present abnormally low PhA. After adjustment for gender, age and lung function, the effect of being classified as low PhA on 6 min walk distance was −76.2 m.
Why read on?
We have demonstrated that body composition is an important factor associated with exercise capacity in patients with IPF and the assessment of PhA by bioelectrical impedance analysis can be used to discriminate patients with IPF with worse lung function, exercise capacity and HRQL.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a specific form of chronic, progressive, fibrosing interstitial pneumonia of unknown cause characterised by progressive worsening of dyspnoea and lung function.1 It is a rare disease with an unpredictable clinical course and high mortality. In Europe, the annual incidence ranges from 0.22 to 7.4 and the prevalence from 1.25 to 23.4 cases per 100 000 population.2 Patients with IPF suffer from exercise intolerance, physical inactivity and impaired health-related quality of life (HRQL).3–6 Schwebel et al found nearly 50% of patients with severe IPF with normal body weight had nutritional depletion.7 A prospective cohort with patients with interstitial lung disease (ILD), including 40 patients with IPF, found a significantly lower muscle mass and higher fat mass in subjects with more impaired lung function.8 In addition, low erector spine cross-sectional area and low Fat-Free Mass Index (FFMI) are related to increased mortality in this population, independent of body mass index (BMI).9–11 FFMI is frequently assessed by using bioelectrical impedance analysis (BIA). The estimation of FFMI by this method provides reliable information in subjects without significant fluid and electrolyte abnormalities when using appropriate population, age or pathology-specific BIA equations and established procedures.12 However, this commonly used method of estimating FFMI using BIA equations has been suggested to present disadvantages.12 On the other hand, the use of directly measured BIA variables, such as phase angle (PhA), have gained attention since they are not affected by some of these disadvantages, such as equation inherent errors and the necessary assumptions for BIA classification of body compartments (consequently for the estimation of FFMI).13
PhA is a measure of the relationship between reactance and resistance, two different electrical properties of tissues, obtained from BIA, that are affected in various ways by disease, nutrition and hydration status.12 PhA has been suggested to be an indicator of cellular health where higher values reflect higher cellularity, cell membrane integrity and better cell function.13 This variable has shown to be an independent predictor of muscle strength, more strongly associated to handgrip strength and respiratory muscle strength than BIA-based estimates of FFMI or anthropometric parameters in patients with chronic obstructive pulmonary disease (COPD).14 In addition, PhA is independently associated with measures of physical function, disease severity and early all-cause mortality in this population.15 16
Recently, Rinaldi et al17 showed that in fibrotic ILD, low FFMI controlled for age and sex is significantly associated with exercise capacity independent of lung function. This same research group also investigated whether PhA is an appropriate surrogate marker of nutrition status as assessed using the subjective global assessment,11 however, a comparison of the frequency of patients with abnormal low FFMI and PhA, and which of these variables are strongly associated with exercise capacity and HRQL was not investigated. Based on previous findings in other populations, such as COPD14–16 and elderly patients with cancer,18 it seems reasonable to hypothesise that PhA is related to these outcomes and can offer information beyond BMI and FFMI in patients with IPF. Thus, the aim of this study was to investigate the frequency of abnormal low PhA and FFMI and their associations with exercise capacity and HRQL in patients with IPF. Preliminary results of this study have been previously reported in the form of abstract in the European Respiratory Society congress 2020.19
Materials and methods
Participants and study design
This study assessed for eligibility, all patients with IPF referred to the specialised rehabilitation centre (Schoen Klinik Berchtesgadener Land, Schoenau, Germany) from March 2012 to November 2017. The diagnosis of IPF has been previously confirmed according to the criteria of current guidelines.1 No patient presented clinical conditions that potentially influence fluid balance (eg, renal failure, cirrhosis, myocardial disease). All patients have signed an informed consent term at time of admission to authorise the use of data from all measures throughout the time of stay for further research. As all data included in this study were already collected an ethic approval was waived. During a prerehabilitation assessment demographic data (sex and age), lung function and smoking history, body composition, exercise capacity and HRQL were collected.
Assessments
Lung function was evaluated by body plethysmography (MasterScreen Body; Jaeger, Germany). The test procedures were performed according to ATS/ERS standardisation.20 Forced vital capacity (FVC), forced expiratory volume in the first second (FEV1), total lung capacity (TLC), residual volume and diffusion capacity for carbon monoxide (DLCO) were determined. Arterial oxygen tension (PaO2), arterial carbon dioxide tension from the hyperaemic earlobe and C reactive protein (CRP)) were also assessed.
Body weight was measured using a calibrated electronic scale to the nearest 0.1 kg and height was measured in an anthropometer to the nearest 0.5 cm. For both measures, patients were barefoot and in underwear. Body mass index (BMI) was calculated as the ratio between weight and height squared (kg/m2). Body composition was assessed by BIA using a multifrequency impedance analyser (Nutriguard-MS; Data Input, Germany). Assessments were performed between 7:00 and 7:45 hours, after an overnight fast and after a time of 10 min in the supine position. The procedure was performed according to manufacturer’s instructions. Fat-free mass was adjusted for differences in body surface by dividing by squared height, and FFMI was calculated.
FFMI values were compared with previously published age-sex-BMI-specific reference values obtained from the general population.21 PhA was also assessed and compared with previously published age-sex-BMI-specific reference values obtained from a German population.22 Values of FFMI or PhA lower than the 10th percentile of the reference values were considered low. Other variables such as fat mass index, body cell mass, extracellular mass, body water, intracellular and extracellular water were also assessed.
The 6 min walk test (6MWT) was used to assess exercise capacity and was performed according to the current international guidelines in a 30 m corridor.23 The predicted values for the 6 min walk distance (6MWD) were calculated according to the reference values of Troosters et al.24
HRQL was assessed using a validated German version of the Medical Outcomes Study Short-Form 36-item Questionnaire (SF-36). The questionnaire consisted of 36 questions covering eight health concepts: physical function, role-physical, bodily pain, general health, vitality, social functioning, role-emotional and mental health. These eight health components were combined in two summary dimensions: the Physical Summary and the Mental Summary Scores. The score ranges from 0 to 100, with higher scores indicating better HRQL.25
Statistical analysis
Data are reported as mean±SD or median (IQR 25%–75%). Variables were examined for normality with histograms and qq-plots. For continuous variables, comparisons between patients with normal and low PhA and normal and low FFMI were performed with Student’s t-test for independent samples or Mann-Whitney U test, according to normality in data distribution. For categorical variables, between-group comparisons were performed with the χ2 test. The analysis of covariance was used to compare 6MWD between patients with normal and low PhA and FFMI, while adjusting for gender and age. Simple linear regression analysis was used to assess the relationship between the variables of body composition and lung function with the variables of exercise capacity and HRQL. Multiple linear regression analyses were performed to compare 6MWD between patients with normal and low PhA and FFMI, while adjusting for gender, age and lung function. The χ2 test was used to compare the proportion of patients with normal and low PhA who present increased risk of mortality, by presenting 6MWD lower than 250 m.26 The software used for performing the statistical analyses was SPSS V.25.0 (IBMA). The significance level was set at p<0.05.
Patient and public involvement
There is no patient or public involvement to report in the design, conduction or dissemination of this retrospective observational study.
Results
Table 1 displays the main characteristics of the 98 patients with IPF analysed in the study. Overall, 86% of the patients were male, 56% were on long-term oxygen therapy (LTOT), and 22% were never smokers. As a group, patients demonstrated moderate restrictive lung function impairment with severe diffusion abnormality. In addition, patients presented moderate exercise intolerance and impaired HRQL. Finally, accordingly with BMI, most patients were classified as overweight or normal weight (41% and 37%, respectively) whereas 20% of the patients were classified as obese and 2% as underweight. The frequency of patients with low FFMI was 9%, whereas 26% of the patients presented low PhA.
Table 1.
Characteristics of the sample
| Variables | (n=98) |
| Sex (male), n (%) | 84 (85.7) |
| Age (years) | 68±8 |
| BMI (kg/m2) | 26.6±4.3 |
| Underweight, n (%) | 2 (2.0) |
| Normal weight, n (%) | 36 (36.7) |
| Overweight, n (%) | 40 (40.8) |
| Obese, n (%) | 20 (20.4) |
| FFMI (kg/m²) | 19.8±2.6 |
| FMI (kg/m²) | 6.7±2.7 |
| Low FFMI, n (%) | 9 (9.2) |
| Body cell mass (kg) | 29±6 |
| Extracellular mass (kg) | 32±6 |
| Body water (L) | 44±8 |
| Intracellular water (L) | 26±3 |
| Extracellular water (L) | 19±4 |
| Phase angle (°) | 5.2±0.9 |
| Low phase angle, n (%) | 25 (25.5) |
| Never smoker, n (%) | 22 (22.4) |
| Pack years | 20(10-40) |
| FVC (%predicted) | 64±18 |
| FEV1 (%predicted) | 72±19 |
| TLC (%predicted) | 71±14 |
| RV (%predicted) | 86±21 |
| DLCO (%predicted)* | 31±15 |
| PaO2 (mm Hg) | 67±12 |
| PaCO2 (mm Hg) | 37±4 |
| LTOT, n (%) | 55 (56.1) |
| CRP (mg/dL) | 6 (3–13) |
| 6MWD (m) | 383±114 |
| 6MWD (%predicted) | 68±20 |
| SF-36 Physical Summary Score | 37±10 |
| SF-36 Mental Summary Score | 43±14 |
Data expressed as frequency, mean±SD or median (IQR 25%–75%).
*N=78.
BMI, body mass index; CRP, C reactive protein; DLCO, diffusion capacity for carbon monoxide; FEV1, forced expiratory volume in 1 s; FFMI, Fat-Free Mass Index; FMI, Fat Mass Index; FVC, forced vital capacity; LTOT, long-term oxygen therapy; 6MWD, 6 min walk distance; PaCO2, arterial carbon dioxidetension; PaO2, arterial oxygen tension; RV, residual volume; SF-36, Short-Form 36-item Questionnaire; TLC, total lung capacity.
Table 2 displays the comparisons between patients with normal and low FFMI or PhA. Patients with low FFMI presented lower, BMI, FFMI, body cell mass, body water and PhA compared with patients with normal FFMI. On the other hand, patients with low PhA presented lower FVC, FEV1, PaO2, 6MWD and SF-36 Physical Summary score, but higher amount of extracellular mass, CRP levels and pack years compared with patients with normal PhA. In addition, the proportion of patients on LTOT where higher in patients with low PhA compared with patients with normal PhA. After controlling for gender and age, the mean difference in 6MWD between patients with normal and low PhA was −106 m (95% CI −154 to −58 m; p≤0.001), (figure 1).
Table 2.
Comparisons between patients with normal and low phase angle (PhA) or Fat-Free Mass Index (FFMI)
| Variables | PhA | FFMI | ||||
| Normal (n=73) | Low (n=25) | P value | Normal (n=89) | Low (n=9) | P value | |
| Sex (male), n (%) | 62 (85) | 22 (88) | 0.70 | 76 (85) | 8 (89) | 0.77 |
| Age (years) | 69±4 | 66±10 | 0.28 | 68±8 | 71±5 | 0.21 |
| BMI (kg/m2) | 26.3±4.3 | 27.4±4.3 | 0.31 | 27.0±4.1 | 23.3±5.0 | 0.01 |
| FFMI (kg/m²) | 19.8±2.7 | 19.9±2.5 | 0.87 | 20.1±2.5 | 16.7±2.2 | <0.01 |
| FMI (kg/m²) | 6.5±2.5 | 7.4±3.0 | 0.14 | 6.8±2.6 | 5.7±3.1 | 0.23 |
| Body cell mass (kg) | 29±7 | 27±6 | 0.10 | 29±6 | 23±6 | <0.01 |
| Extracellular mass (kg) | 30±5 | 36±6 | <0.01 | 32±6 | 29±7 | 0.14 |
| Body water (L) | 44±8 | 46±8 | 0.27 | 45±8 | 38±8 | 0.01 |
| Intracellular water (L) | 26±4 | 26±3 | 0.67 | 26±3 | 23±10 | 0.02 |
| Extracellular water (L) | 18±4 | 20±4 | 0.11 | 19±4 | 15±5 | 0.01 |
| Phase angle (°) | 5.5±0.7 | 4.4±0.7 | <0.01 | 5.3±0.8 | 4.7±1.0 | 0.05 |
| Pack years | 13(8-34) | 40(15-50) | 0.02 | 20(9-40) | 30(10-50) | 0.25 |
| FVC (%predicted) | 67±19 | 55±13 | <0.01 | 65±19 | 58±12 | 0.25 |
| FEV1 (%predicted) | 75±20 | 63±14 | <0.01 | 73±20 | 67±14 | 0.50 |
| TLC (%predicted) | 72±14 | 66±13 | 0.05 | 71±14 | 66±14 | 0.45 |
| RV (%predicted) | 85±20 | 89±24 | 0.44 | 86±21 | 86±21 | 0.97 |
| DLCO (%predicted) | 32±16 | 26±14 | 0.14 | 31±15 | 27±18 | 0.69 |
| PaO2 (mm Hg) | 69±11 | 60±12 | <0.01 | 67±11 | 61±13 | 0.15 |
| PaCO2 (mm Hg) | 37±4 | 36±5 | 0.80 | 37±4 | 37±4 | 0.99 |
| LTOT, n (%) | 36 (49) | 19 (76) | 0.02 | 49 (55) | 6 (67) | 0.50 |
| CRP (mg/dL) | 5 (3–10) | 8 (5–18) | 0.03 | 6 (3–13) | 6 (3–25) | 0.93 |
| 6MWD (m) | 407±109 | 312±103 | <0.01 | 387±114 | 344±117 | 0.28 |
| 6MWD (%predicted) | 73±19 | 54±17 | <0.01 | 69±20 | 61±21 | 0.23 |
| SF-36 Physical Summary Score | 39±9 | 33±8 | <0.01 | 37±10 | 38±7 | 0.80 |
| SF-36 Mental Summary Score | 44±14 | 41±14 | 0.47 | 43±14 | 44±14 | 0.95 |
Data expressed as frequency, mean±SD or median (IQR 25%–75%).
BMI, body mass index; CRP, C reactive protein; DLCO, diffusion capacity for carbon monoxide; FEV1, forced expiratory volume in the first second; FMI, fat mass index; FVC, forced vital capacity; LTOT, long-term oxygen therapy; 6MWD, 6 min walk distance; PaCO2, arterial carbon dioxide tension; PaO2, arterial oxygen tension; RV, residual volume; SF-36, Short-Form 36-item Questionnaire; TLC, total lung capacity.
Figure 1.
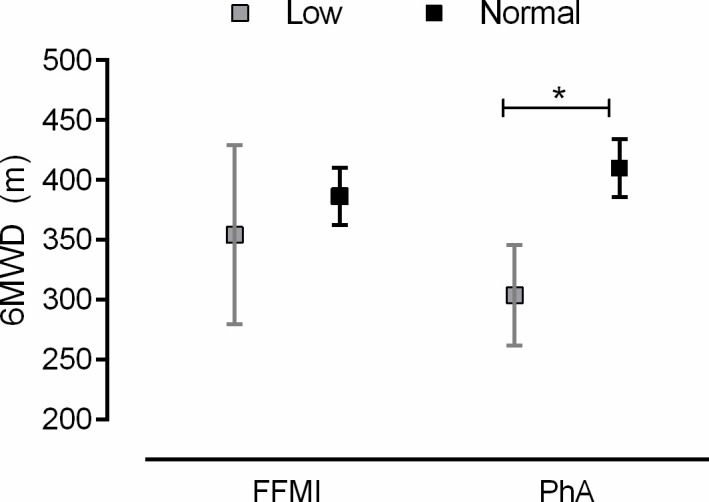
Comparisons of 6 min walk distance (6MWD) between patients with normal and low fat-freemass index (FFMI) and phase angle (PhA). Adjusted means and CIs reported from ANCOVA, after adjusting for gender and age. *P<0.05. ANCOVA, analysis of covariance.
In a single regression analysis (table 3), age, body cell mass, PhA, FVC, FEV1, TLC and DLCO, but not FFMI, were significantly related to 6MWD, whereas the same variables, except age and body cell mass were significantly related with SF-36 Physical Summary Score (p<0.05, for all). None of the variables were significantly related with SF-36 Mental Summary Score. Table 4 displays the impact of low PhA and FFMI, after adjustment for gender, age, DLCO and FVC, the mean difference in 6MWD between patients with normal and low PhA was −76.2 m (95% CI −119.1 to −33.3 m; p=0.001), whereas the mean difference in 6MWD between patients with normal and low FFMI was −7.4 m (95% CI −75.6 to 60.7 m; p=0.83). The proportion of patients who presented a 6MWD lower than 250 m showed statistically significant difference between patients with normal and low PhA (7% vs 39%; p<0.01).
Table 3.
Relationship between exercise capacity and health related quality of life with body composition and lung function
| Variables | 6MWD(m) | SF-36 (physical summary) | SF-36 (mental summary) |
| Age (years) | 0.07** | NS | NS |
| FFMI (kg/m²) | NS | NS | NS |
| Body cell mass (kg) | 0.13* | NS | NS |
| Phase angle (°) | 0.29* | 0.06** | NS |
| FVC (%predicted) | 0.24* | 0.21* | NS |
| FEV1 (%predicted) | 0.17* | 0.20* | NS |
| TLC (%predicted) | 0.16* | 0.21* | NS |
| DLCO (%predicted) | 0.36* | 0.26* | NS |
Single regression analysis. R² values are shown.
*p<0.01.
**p<0.05.
DLCO, diffusion capacity for carbon monoxide; FEV1, forced expiratory volume in the first second; FFMI, fat-free mass index; FVC, forced vital capacity; 6MWD, 6 min walk distance; NS, not significant; SF-36, Short-Form 36-item Questionnaire; TLC, total lung capacity.
Table 4.
Multiple linear regression analyses to compare 6MWD between patients with normal and low PHA and FFMI, while adjusting for gender, age and lung function
| Model | Correlates | Beta | 95% CI | P value |
| 6MWD (m) Adjusted R²=0.54 P<0.001 |
(Constant) | 557.9 | 388.0 to 727.8 | <0.001 |
| Sex (male) | −33.6 | −17.7 to 84.8 | 0.19 | |
| Age (years) | −5.2 | −7.5 to −2.9 | <0.001 | |
| DLCO (%predicted) | 3.7 | 2.2 to 5.2 | <0.001 | |
| FVC (%predicted) | 0.9 | −0.5 to 2.2 | 0.20 | |
| Phase Angle (Low) | −76.2 | −119.1 to −33.3 | 0.001 | |
| 6MWD (m) Adjusted R²=0.46 P<0.001 |
(Constant) | 480.4 | 301.6 to 659.1 | <0.001 |
| Sex (male) | 28.6 | −26.9 to 84.2 | 0.31 | |
| Age (years) | −4.7 | −7.2 to −2.2 | <0.001 | |
| DLCO (%predicted) | 3.6 | 2.0 to 5.3 | <0.001 | |
| FVC (%predicted) | 1.4 | −0.02 to 2.8 | 0.05 | |
| FFMI (Low) | −7.4 | −75.5 to 60.7 | 0.83 |
DLCO, diffusion capacity for carbon monoxide; FFMI, Fat-Free Mass Index; FVC, forced vital capacity; 6MWD, 6 min walk distance.
Discussion
This is the first study to report (1) the frequency of low FFMI and PhA according to the 10th percentile of age-sex-BMI-specific reference values and (2) the clinical impact of presenting low FFMI and PhA in terms of lung function, exercise capacity and HRQL in patients with IPF. It was demonstrated that PhA is associated with exercise capacity and HRQL, whereas FFMI was not related to these outcomes. In addition, stratification of patients in normal and low PhA could better discriminate patients with worse lung function, exercise capacity and HRQL, compared with stratification in normal and low FFMI. After adjustment for gender, age and lung function, the effect of being classified as low PhA on 6MWD was −76.2 m. These findings are in accordance with a previous study, that included a large cohort of patients with COPD, and demonstrated that PhA is a valid functional and prognostic biomarker, offering information beyond FFMI, which did not identify patients with the greatest level of impairment or disease severity.15
The 6MWT has been demonstrated as a valid, reliable and responsive measure for the assessment of exercise capacity27 28 and as an independent predictor of mortality in patients with IPF.26 A previous study identified several determinants of the 6MWD, including cardiac, circulatory and pulmonary variables, suggesting a multifactorial nature of exercise limitation in this population.29 This study demonstrated that body composition is also an important factor associated with exercise capacity in patients with IPF and could be a factor limiting exercise capacity or a consequence of reduced exercise capacity and physical inactivity in this population.
HRQL is a component of the broader concept of quality of life and is defined as satisfaction with health.30 Many different instruments have been used to assess HRQL in patients with IPF, one of the most used is the SF-36,3 31 which have been demonstrated as a valid questionnaire.32 Patients with IPF have significantly impaired HRQL in both Physical and Mental Summary Scores,3 32 however, recently Cox et al31 showed that domain scores reflecting physical wellness (activity and symptoms) were generally worse than those reflecting emotional wellness (impact), which agrees with the findings of this study. No factor was associated with the Mental Summary Score of the SF-36. This could be explained due to the fact that the Mental Summary Score includes questions that measure mainly individual’s perception, and these measures tend to be more highly correlated with other perception-based measures such as reported dyspnoea, in other words, patients with objectively equal physiological parameters can present different self-reported quality of life.3
This study adds to the current literature information regarding the clinical applications of BIA in patients with IPF. We found that stratification of patients with IPF into normal and low PhA or FFMI discriminates patients with clearly different characteristics. While stratification into low FFMI identified patients with significantly lower weight due to tissue depletion, including not only lower FFMI, but also lower body cell mass and body water, the stratification into low PhA performed better to discriminate patients with worse lung function, exercise capacity and HRQL, despite no differences in BMI and other body composition variables (except for higher amount of extracellular). Extracellular mass includes all metabolically inactive tissues of the body, and a higher extracellular mass/body cell mass ratio is an early warning sign of worsening nutritional status.33 34 These findings support the ability of PhA as a proxy of cellular health (higher number of cells with better membrane integrity and function).13 A limitation of the study is the use of only a generic instrument to measure HRQL, it could be valuable to compare whether the results would be similar when using a disease-specific instrument, such as the IPF-specific version of the St George’s Respiratory Questionnaire and the King’s Brief Interstitial Lung Disease questionnaire, which are able to capture unique aspects of the disease.
It is already known that a 12-week supervised exercise training programme is clinically beneficial to enhance exercise capacity, quality of life, physical activity and body composition outcomes in patients with IPF35 36; however, there are no studies showing if improvement in these outcomes are associated. Thus, future studies should investigate whether PhA is a stronger prognostic factor than FFMI and can be improved after interventions, such as pulmonary rehabilitation and nutritional support/counselling. Finally, it would be interesting to understand the associations between body composition, exercise capacity and HRQL in a prospective study design to better explore cause-consequence understanding.
Conclusion
The findings of this study indicate that the frequency of abnormal low PhA (26%) is higher than expected, according to the use of the 10th percentile of the reference values for the general population. While the use of BMI and FFMI would identify only 2% and 9% of the patients with low weight and muscle mass, the use of PhA revealed two groups of patients with clearly distinct characteristics, it should be noted that patients with low PhA present worse lung function, exercise capacity and HRQL.
Acknowledgments
The authors would like to acknowledge the participants of the study and the assistance of colleagues, which somehow contributed for this study. FVCM is financially supported by ZonMW (ERACoSysMed grant #90030355).
Footnotes
Contributors: TS, IJ, RG, SW, ARK and KK designed the research. FM performed statistical analysis. FM, AEMB and TS produced a first draft of the manuscript. FF, FP, MAS and KK reviewed/edited the article and contributed with intellectual input. All authors read and approved the final manuscript. KK is responsible for the overall content as guarantor.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient and public involvement: Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Ethics statements
Patient consent for publication
Not applicable.
References
- 1.Raghu G, Collard HR, Egan JJ, et al. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med 2011;183:788–824. 10.1164/rccm.2009-040GL [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nalysnyk L, Cid-Ruzafa J, Rotella P, et al. Incidence and prevalence of idiopathic pulmonary fibrosis: review of the literature. Eur Respir Rev 2012;21:355–61. 10.1183/09059180.00002512 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Swigris JJ, Kuschner WG, Jacobs SS, et al. Health-Related quality of life in patients with idiopathic pulmonary fibrosis: a systematic review. Thorax 2005;60:588–94. 10.1136/thx.2004.035220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tomioka H, Imanaka K, Hashimoto K, et al. Health-related quality of life in patients with idiopathic pulmonary fibrosis--cross-sectional and longitudinal study. Intern Med 2007;46:1533–42. 10.2169/internalmedicine.46.6218 [DOI] [PubMed] [Google Scholar]
- 5.Blackwell TS, Tager AM, Borok Z, et al. Future directions in idiopathic pulmonary fibrosis research. An NHLBI workshop report. Am J Respir Crit Care Med 2014;189:214–22. 10.1164/rccm.201306-1141WS [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bahmer T, Kirsten A-M, Waschki B, et al. Prognosis and longitudinal changes of physical activity in idiopathic pulmonary fibrosis. BMC Pulm Med 2017;17:1–8. 10.1186/s12890-017-0444-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schwebel C, Pin I, Barnoud D, et al. Prevalence and consequences of nutritional depletion in lung transplant candidates. Eur Respir J 2000;16:1050–5. 10.1034/j.1399-3003.2000.16f05.x [DOI] [PubMed] [Google Scholar]
- 8.Guler SA, Hur SA, Lear SA, et al. Body composition, muscle function, and physical performance in fibrotic interstitial lung disease: a prospective cohort study. Respir Res 2019;20:56. 10.1186/s12931-019-1019-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nishiyama O, Yamazaki R, Sano H, et al. Fat-Free mass index predicts survival in patients with idiopathic pulmonary fibrosis. Respirology 2017;22:480–5. 10.1111/resp.12941 [DOI] [PubMed] [Google Scholar]
- 10.Suzuki Y, Yoshimura K, Enomoto Y, et al. Distinct profile and prognostic impact of body composition changes in idiopathic pulmonary fibrosis and idiopathic pleuroparenchymal fibroelastosis. Sci Rep 2018;8:14074. 10.1038/s41598-018-32478-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rinaldi S, Gilliland J, O'Connor C, et al. Fat-Free mass index controlled for age and sex and malnutrition are predictors of survival in interstitial lung disease. Respiration 2021;100:379–86. 10.1159/000512732 [DOI] [PubMed] [Google Scholar]
- 12.Kyle UG, Bosaeus I, De Lorenzo AD, et al. Bioelectrical impedance analysis--part I: review of principles and methods. Clin Nutr 2004;23:1226–43. 10.1016/j.clnu.2004.06.004 [DOI] [PubMed] [Google Scholar]
- 13.Norman K, Stobäus N, Pirlich M, et al. Bioelectrical phase angle and impedance vector analysis--clinical relevance and applicability of impedance parameters. Clin Nutr 2012;31:854–61. 10.1016/j.clnu.2012.05.008 [DOI] [PubMed] [Google Scholar]
- 14.de Blasio F, Santaniello MG, de Blasio F, et al. Raw BIA variables are predictors of muscle strength in patients with chronic obstructive pulmonary disease. Eur J Clin Nutr 2017;71:1336–40. 10.1038/ejcn.2017.147 [DOI] [PubMed] [Google Scholar]
- 15.Maddocks M, Kon SSC, Jones SE, et al. Bioelectrical impedance phase angle relates to function, disease severity and prognosis in stable chronic obstructive pulmonary disease. Clin Nutr 2015;34:1245–50. 10.1016/j.clnu.2014.12.020 [DOI] [PubMed] [Google Scholar]
- 16.de Blasio F, Scalfi L, Di Gregorio A, et al. Raw bioelectrical impedance analysis variables are independent predictors of early all-cause mortality in patients with COPD. Chest 2019;155:1148–57. 10.1016/j.chest.2019.01.001 [DOI] [PubMed] [Google Scholar]
- 17.Rinaldi S, Gilliland J, O'Connor C, et al. Exercise capacity and its relationship with body composition and nutrition status in patients with interstitial lung disease. Nutr Clin Pract 2021;36:891–8. 10.1002/ncp.10651 [DOI] [PubMed] [Google Scholar]
- 18.Norman K, Wirth R, Neubauer M, et al. The bioimpedance phase angle predicts low muscle strength, impaired quality of life, and increased mortality in old patients with cancer. J Am Med Dir Assoc 2015;16:173.e17–173.e22. 10.1016/j.jamda.2014.10.024 [DOI] [PubMed] [Google Scholar]
- 19.Machado F, Bloem A, Schneeberger T. Relationship between body composition, exercise capacity and health related quality of life in idiopathic pulmonary fibrosis. European Respiratory Journal 2020;56:272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Miller MR, Crapo R, Hankinson J, et al. General considerations for lung function testing. Eur Respir J 2005;26:153–61. 10.1183/09031936.05.00034505 [DOI] [PubMed] [Google Scholar]
- 21.Franssen FME, Rutten EPA, Groenen MTJ, et al. New reference values for body composition by bioelectrical impedance analysis in the general population: results from the UK Biobank. J Am Med Dir Assoc 2014;15:448.e1–448.e6. 10.1016/j.jamda.2014.03.012 [DOI] [PubMed] [Google Scholar]
- 22.Bosy-Westphal A, Danielzik S, Dörhöfer R-P, et al. Phase angle from bioelectrical impedance analysis: population reference values by age, sex, and body mass index. JPEN J Parenter Enteral Nutr 2006;30:309–16. 10.1177/0148607106030004309 [DOI] [PubMed] [Google Scholar]
- 23.ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories . ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med 2002;166:111–7. 10.1164/ajrccm.166.1.at1102 [DOI] [PubMed] [Google Scholar]
- 24.Troosters T, Gosselink R, Decramer M. Six minute walking distance in healthy elderly subjects. Eur Respir J 1999;14:270–4. 10.1034/j.1399-3003.1999.14b06.x [DOI] [PubMed] [Google Scholar]
- 25.Lyons RA, Perry HM, Littlepage BN. Evidence for the validity of the short-form 36 questionnaire (SF-36) in an elderly population. Age Ageing 1994;23:182–4. 10.1093/ageing/23.3.182 [DOI] [PubMed] [Google Scholar]
- 26.du Bois RM, Albera C, Bradford WZ, et al. 6-Minute walk distance is an independent predictor of mortality in patients with idiopathic pulmonary fibrosis. Eur Respir J 2014;43:1421–9. 10.1183/09031936.00131813 [DOI] [PubMed] [Google Scholar]
- 27.du Bois RM, Weycker D, Albera C, et al. Six-minute-walk test in idiopathic pulmonary fibrosis: test validation and minimal clinically important difference. Am J Respir Crit Care Med 2011;183:1231–7. 10.1164/rccm.201007-1179OC [DOI] [PubMed] [Google Scholar]
- 28.Nathan SD, du Bois RM, Albera C, et al. Validation of test performance characteristics and minimal clinically important difference of the 6-minute walk test in patients with idiopathic pulmonary fibrosis. Respir Med 2015;109:914–22. 10.1016/j.rmed.2015.04.008 [DOI] [PubMed] [Google Scholar]
- 29.Porteous MK, Rivera-Lebron BN, Kreider M, et al. Determinants of 6-minute walk distance in patients with idiopathic pulmonary fibrosis undergoing lung transplant evaluation. Pulm Circ 2016;6:30–6. 10.1086/685022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Spruit MA, Singh SJ, Garvey C, et al. An official American thoracic Society/European respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med 2013;188:e13–64. 10.1164/rccm.201309-1634ST [DOI] [PubMed] [Google Scholar]
- 31.Cox IA, Borchers Arriagada N, de Graaff B, et al. Health-Related quality of life of patients with idiopathic pulmonary fibrosis: a systematic review and meta-analysis. Eur Respir Rev 2020;29:200154–22. 10.1183/16000617.0154-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Martinez TY, Pereira CA, dos Santos ML, et al. Evaluation of the short-form 36-item questionnaire to measure health-related quality of life in patients with idiopathic pulmonary fibrosis. Chest 2000;117:1627–32. 10.1378/chest.117.6.1627 [DOI] [PubMed] [Google Scholar]
- 33.Talluri T, Lietdke RJ, Evangelisti A, et al. Fat-Free mass qualitative assessment with bioelectric impedance analysis (BIA). Ann N Y Acad Sci 1999;873:94–8. 10.1111/j.1749-6632.1999.tb09454.x [DOI] [PubMed] [Google Scholar]
- 34.Pelzer U, Arnold D, Gövercin M, et al. Parenteral nutrition support for patients with pancreatic cancer. Results of a phase II study. BMC Cancer 2010;10:86. 10.1186/1471-2407-10-86 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Vainshelboim B, Oliveira J, Yehoshua L, et al. Exercise training-based pulmonary rehabilitation program is clinically beneficial for idiopathic pulmonary fibrosis. Respiration 2014;88:378–88. 10.1159/000367899 [DOI] [PubMed] [Google Scholar]
- 36.Vainshelboim B, Fox BD, Kramer MR, et al. Short-Term improvement in physical activity and body composition after supervised exercise training program in idiopathic pulmonary fibrosis. Arch Phys Med Rehabil 2016;97:788–97. 10.1016/j.apmr.2016.01.018 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.


