Abstract
We aimed to investigate association between mean platelet volume (MVP), platelet distribution width (PDW) and red cell distribution width (RDW) and mortality in patients with COVID-19 and find out in which patients the use of acetylsalicylic acid (ASA) affects the prognosis due to the effect of MPV on thromboxan A2. A total of 5142 patients were divided into those followed in the intensive care unit (ICU) and those followed in the ward. Patient medical records were examined retrospectively. ROC analysis showed that the area under curve (AUC) values were 0.714, 0.750, 0.843 for MPV, RDW and D-Dimer, the cutoff value was 10.45fl, 43.65fl, 500.2 ng/mL respectively. (all P < .001). Survival analysis showed that patients with MPV >10.45 f/l and D-Dimer >500.2 ng/mL, treatment with ASA had lower in-hospital and 180-day mortality than patients without ASA in ICU patients (HR = 0.773; 95% CI = 0.595-0.992; P = .048, HR = 0.763; 95% CI = 0.590-0.987; P = .036). Administration of low-dose ASA in addition to anti-coagulant according to MPV and D-dimer levels reduces mortality.
Keywords: mean platelet volume, platelet distribution width, red cell distribution width, COVID-19
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and the resulting coronavirus disease 2019 (Covid-19) is a rapidly evolving public health crisis worldwide. 1 In most patients infected with Covid-19, symptoms are mild or moderate, however, some patients experience severe illness, progressing rapidly to acute respiratory failure, coagulopathy, metabolic acidosis, acute respiratory distress syndrome (ARDS) and septic shock. 2
Laboratory testing suggests a coagulopathy with up to 30% of hospitalized Covid-19 patients developing thrombotic events. 3 Platelets are central role in both arterial and venous thrombosis, and platelet- virus interactions contribute to thrombotic risk by promoting inflammatory and procoagulant states during viral infection. Hematological parameters, such as white blood cell (WBCs) and their subpopulations, red cell distribution width (RDW), platelet distribution width (PDW) and mean platelet volume (MPV) are widely used for risk diagnosis, stratification and determination of prognosis in COVID-19. 2
MPV, the accurate measure of the platelet size, is considered a marker and determinant of platelet function. Larger platelets with higher MPV are hemostatically more reactive and produce higher amounts of the prothrombotic factor thromboxane A2 (TXA2). The increase in TXA2 generation results in thrombotic complications. 4 MPV counts are also used as markers of the inflammatory response. Previous studies have shown that thrombopoietin and inflammatory cytokines such as IL-1, IL-6, and TNFa regulate thrombopoiesis and MPV is a reflection of both proinflammatory and prothrombotic conditions. 5 Evidence has been suggested that increased MPV values are a marker of several thrombotic disorders, including acute coronary syndrome, stroke and venous thromboembolism.6,7
PDW reflects the variation in the size of platelets. PDW increases when platelet destruction increases and there are variations in the size of newly formed immature platelets. 8 Due to the added infections, the risk of sepsis increases in Covid-19. Platelet production increases alongside its destruction and new immature platelets enter circulation. Thus, PDW and MPV are also affected.
Red cell distribution width (RDW) refers to the degree of anisocytosis among red blood cells. Hypoxia causes disruption of erythropoiesis, alteration of erythropoiesis can cause heterogeneity of RBC size, which can provide evidence of ongoing pathological changes. 9
Therefore, we comprehensively analyzed and aimed to find out whether there is a relation between mortality in Covid-19 and levels of MPV,RDW and PDW, additionally 6-month consequences of aspirin use because of effect of MPV on TXA2.
Method
Study Settings and Population
A single-center retrospective study was conducted between March 21, 2020, and December, 2020, in Erzurum Bolge Education and Research Hospital, which is a tertiary level hospital in the east part of Turkey. The hospital was designated as the coronavirus pandemic hospital in the province by the Ministry of Health. The hospital has a total of 800 patient beds, 150 of which are intensive-care beds.We included all COVID-19 diagnosed and hospitalized patients in the study a total of 5578. Patients with malignancy, haematological disorders and receiving iron, folic acid and B12 replacement therapy were excluded. After exclusion 5412 patients were enrolled.
Complete blood count, C-reactive protein (CRP), and biochemistry tests are routinely performed on patients who attend the emergency department with complaints compatible with COVID-19 such as cough, fever, and shortness of breath. CBC was performed on Sysmex XN-3100™ Automated Hematology System (Sysmex). MPV, which is one of the platelet indices (MPV [fL] = plateletcrit/platelet count), was calculated obligatorily by the Sysmex XN system. D-dimer was measured by particle-enhanced, immunoturbidimetric assay (Innovance®D-dimer) on Siemens BCS®XP Systems automated coagulation analyzer (Siemens Healthcare GmbH). The reference range for MPV was between 7.8 to 12 fL, for PDW 10%-17.9% and for RDW-SD 40.0-55.0 fL. To reach a definitive diagnosis, oro-nasopharyngeal swab (ONS) samples are taken from patients for molecular analysis. Lung Computed Tomography (CT) is performed on patients with shortness of breath after being examined by the responsible physician. A second swab sample was taken from hospitalized patients when first sample was negative. When one of the two samples taken was positive, the patient was diagnosed with COVID-19, and if both were negative, COVID-19 was excluded.
Study Design
The patients were divided into two groups. Severe disease was defined as any level of respiratory support, invasive/non-invasive ventilation or high flow oxygen devices or illness requiring intensive care unit (ICU) admission, while patients with mild-moderate symptoms or need supplemental oxygen by mask or nasal cannula were followed up in the hospital wards. All patients in ICU were given anticoagulation at a therapeutic dose, as long as no contraindication. Acetylsalicylic acid, which patients use for causes such as coronary artery disease, cerebrovascular disease, peripheral arter disease etc, was continued to be administered in intensive care addition to anti-coagulant. The use of anticoagulants and antiaggregant in ward follow-up was left to the physician's choice and a not following standart procedure. We also noted the hospitalisation unit (clinic/intensive care unit), hospitalisation length and mortality in-hospital. Survivor patients were followed for 6 months. The primary outcome of this study was in-hospital and total mortality in 6 month. The safety endpoint was major bleeding. Major bleeding was defined if one of the following holds: fatal bleeding, intracranial bleeding, intraocular bleeding, gastrointestinal bleeding, retroperitoneal bleeding, bleeding with hemodynamic compromise, >5 cm hematoma and hemoglobin drop (g/dL) ≥4.
This study was performed according to the principles laid out in the Declaration of Helsinki, and all procedures were approved by the Ethics Committee of the Erzurum Bolge Education and Research Hospital.
Statictical Analysis
All continuous variables were expressed as mean ± standard deviation (SD) or median (interquartile range [IQR]). Differences between patients with ICU and service patients were assessed using the independent sample t test, the Mann-Whitney U test, or the chi-squared test, as appropriate. Correlations between variables were examined using Spearman's rank correlation analysis. Univariate and multivariate analyses of predictors for mortality were performed using the regression analyse. All variables with a P value of less than.05 were included in the multivariate analysis, and the results of this regression analyses were reported as adjusted odds ratio with a 95% confidence interval. After reciprocal transformation of the mean platelet volume, red cell distribution width and D-dimer, significant predictors for mortality due to COVID-19, a receiver operating characteristic (ROC) curve was generated, and the area under the curve (AUC) was calculated to assess diagnostic value. ROC curve plots the true-positive rate (sensitivity) against the false-positive rate (1- specificity) for all possible cut off values (Youden's index). All statistical analyses were performed using SPSS version 25.0 (IBM Corp., Armonk, NY, USA). Values of P < .05 were considered statistically significant.
Results
The study population comprised a total of 5412 patients. 871 patients had severe COVID-19 infection and followed in ICU. 4541 patients admitted to ward. The study population's mean age was 61.7 ± 16.2years. Of the total participants, 49.1% were males, and 50.9% were females. On admission day, ICU patients had signifcantly higher WBC, MPV, RDW, PDW, platelets, neutrophil, and CRP values than ward COVID-19 patients (P < .001). Demographic and hematological parameters in both groups are shown in Table 1.
Table 1.
Baseline Characteristics of the Patients.
| All patients n = 5412 | Intensive Care n = 871 | Ward n = 4541 | P value | |
|---|---|---|---|---|
| Age, mean ± SD | 61.7 ± 16.2 | 69.8 ± 13.7 | 60.5 ± 16.1 | <.001 |
| Gender (Male), n(%) | 2657 (49.1%) | 517 (59.5%) | 2142 (47.8) | <.001 |
| White Blood Count (K/ul), median(IQR) | 7.2 (5.5-9.5) | 9.9 (7.4-13.1) | 6.9 (5.4-8.9) | <.001 |
| C-reactive protein (mg/l), median (IQR) | 37 (13-76) | 82.4 (43-139.5) | 31 (10.3-63) | <.001 |
| Platelet (K/ul), median (IQR) | 232.1 (184.7-289.7) | 205.8 (154.6-269.5) | 236.8 (189.0-293.7) | <.001 |
| Mean platelet Volume (f/l), median (IQR) | 10.4 (9.9-11.1) | 10.8 (10.2-11.5) | 10.4 (9.8-11) | <.001 |
| Platelet Distribution Width (%), median (IQR) | 12.0 (10.8-13.5) | 12.8 (11.5-14.5) | 11.9 (10.7-13.3) | <.001 |
| Red Cell Distribution Width (f/l), median (IQR) | 42.5 (40.0-46.2) | 44.7 (41.7-49.3) | 41.8 (39.6-44.9) | <.001 |
| Hemoglobin (g/dL), mean ± SD | 13.2 ± 1.9 | 12.4 ± 2.5 | 13.4 ± 1.7 | <.001 |
| Procalcitonin (ng/mL), median (IQR) | 0.11 (0.02-0.57) | 0.85 (0.25-2.73) | 0.08 (0.02 to 0.39 | <.001 |
| Creatinine [mg/dL], median [IQR] | 0.88 [0.74 to 1.13] | 1.14 [0.82 to 1.75] | 0.86 [0.72 to 1.07] | <.001 |
| Sodium [mmol/L], median [IQR] | 137 [135 to 139] | 138 [135 to 142] | 137 [134.6 to 139] | .324 |
| Potassium [mmol/L], mean ± SD | 4.31 ± 0.49 | 4.36 ± 0.61 | 4.27 ± 0.46 | .254 |
| Glucose [mg/dL], median [IQR] | 131[106 to 179] | 169[129 to 205] | 126[104 to 172] | <.001 |
| ALT [U/L], median [IQR] | 32 [21 to 50] | 39 [24 to 71] | 31 [21 to 47] | <.001 |
| Hypertension, n[%] | 2704 [50.0%] | 533 [61.2%] | 2171 [47.8] | <.001 |
| Cerebrovascular disease, n [%] | 123 [2.3%] | 28 [3.2%] | 95 [2.1%] | .042 |
| Diabetes Mellitus, n[%] | 1473 [27.2%] | 261 [30.0%] | 1212 [26.7%] | .126 |
| Thyroid disease, n[%] | 118 [2.2%] | 26 [3.0%] | 92 [2.0%] | .129 |
| Hyperlipidemia, n[%] | 672 [12.4%] | 129 [14.8%] | 543 [12.0%] | .012 |
| COPD, n [%] | 736 [13.6%] | 168 [19.3%] | 568 [12.5%] | <.001 |
| History of Pulmonary embolism, n [%] | 132 [2.4%] | 29 [3.3%] | 103 [2.3%] | .177 |
| Previous Asa Exposure, n [%] | 1191 [22.1%] | 230 [26.5%] | 961 [21.2%] | .001 |
| Hospitalization day, median [IQR] | 8 [5 to 13] | 14 [8 to 22] | 8 [5 to 12] | <.001 |
The data without normal distribution is presented as median [interquartile range-IQR]. COPD: Chronic obstructive pulmonary disease ALT: Alanine Transaminase
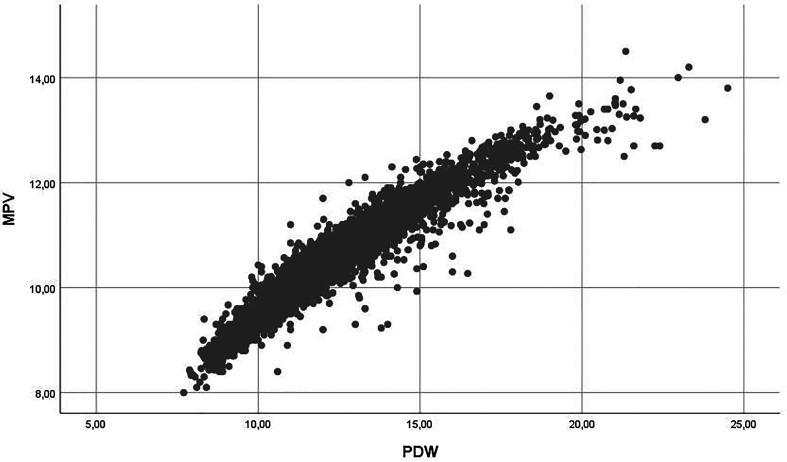
The average differance values of MPV 0.51f/l (95% CI = 0.44-0.58f/l), PDW 1.22% (95% CI = 1.04-1.40%), D-dimer 2098 ng/mL (95% CI = 1065-3132 ng/mL), and RDW 5.60fl (95% CI = 3.78-7.41fl) were statistically higher in patients with survivor and non-survivor. (P values are <.001, <.001, <.001, <.001. There was a positive correlation between MPV and PDW values [P <.001, r = 0.96, R2 = 0.91] [Figure-1]. No significant correlation was observed between other components.
Figure 1.
Relationship between MPV and PDW.
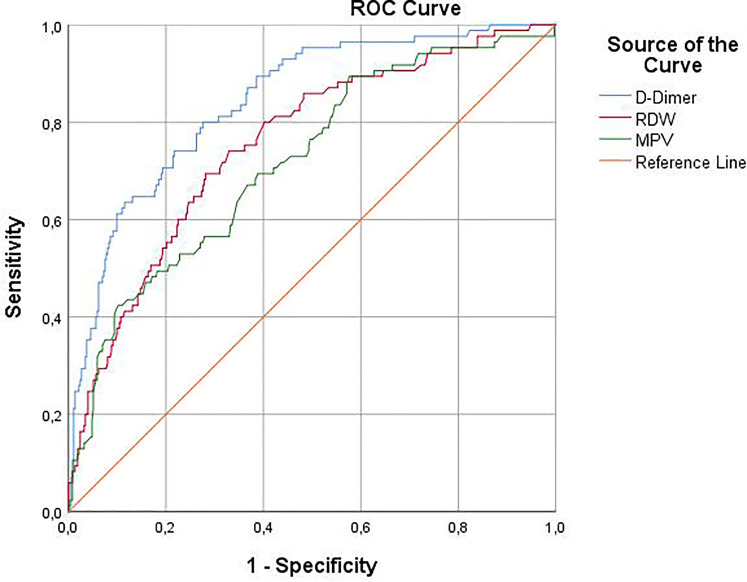
In the univariate analysis, increase in age (OR = 1,08; P < 0001), MPV (OR = 1,85; P < 0001), RDW (OR = 1,16; P < 0001) and D-dimer level (OR = 1,62; P < 001), presence of hypertension (OR = 2.03; P < 0001) and COPD (OR = 2.21; P < 0001) were determined as possible risk factors for mortality. In the multivariable regression model, in which possible risk factors were included, increasing age (OR = 1,06; P < 0001), MPV (OR = 2.40; P < 0001), RDW (OR = 1,14; P < 0001) and D-Dimer (OR = 1.93; P < 0001) levels were found to be independent risk factors for mortality.(-2 Log Likelihood: 235, 546 Nagelkerke R2 :0.58) (Table 2) We used ROC analysis to examine the ability of hematological markers to discriminate mortality. The AUC of MPV for mortality prediction was 0.714 (95% CI = 0.653-0.775; P < .001), the cutoff value was 10.45fl, the sensitivity and specificity were 66.3% and 62.8%. The AUC values was 0.750 (95% CI = 0.693-0.807; P < .001) the sensitivity and specificity were 70.9% and 68% for RDW and the cutoff value was 43.65fl. When evaluated together with other parameters, D-dimer showed the highest AUC value for predicting mortality. The AUC of D-dimer was 0.843, the cutoff value was 500.2 ng/mL, and the sensitivity and specificity were 72.5% and 78% (P < .001). PDW was not included in the multivariate analysis and ROC analysis because it was strongly correlated with MPV. The AUC values were shown in Figure 2 and Table 3.
Table 2.
Logistic Regression Analysis to Identify Risk Factors Associated with Mortality in Patients with COVID-19.
| Variables | Univariate analysis | Multivariate analysis | ||
|---|---|---|---|---|
| OR (95 CI%) | P value | OR (95% CI) | P value | |
| Age, years | 1.08 (1.07-1.09) | <.001 | 1.06 (1.02-1.09) | <.001 |
| Hemoglobin (g/dL) | 0.77 (0.74-0.80) | <.001 | 0.83 (0.71-0.97) | .019 |
| White Blood Count (K/ul) | 1.25 (1.22-1.27) | <.001 | 1.19 (1.02-1.22) | .017 |
| Lymphocyte (K/ul) | 1.02 (1.0-1.04) | .11 | ||
| Mean platelet Volume (f/l) | 1.85 (1.70-2.02) | <.001 | 2.40 (1.66-3.56) | <.001 |
| Red Cell Distribution Width (f/l) | 1.16 (1.11-1.21) | <.001 | 1.14 (1.08-1.21) | <.001 |
| D-Dimer (ng/dl) | 1.62 (1.56-1.68) | <.001 | 1.93 (1.52-2.43) | <.001 |
| Chronic obstructive pulmonary disease | 2.21 (1.82-2.68) | <.001 | 0.59 (0.42-1.75) | .21 |
| Hypertension | 2.03 (1.72-2.38) | <.001 | 0.80 (0.38-1.66) | .55 |
| Diabetes Mellitus | 1.13 (0.95-1.34) | .164 | ||
Figure 2.
ROC curve comparing the diagnostic validity of RDW, MPV and D-Dimer in the prediction of mortality among COVID-19 patients.
Table 3.
ROC Analysis Results to Predict Mortality.
| Cut-off value | Sensitivity (%) | Specificity (%) | AUC (95% CI) | SE | P |
|---|---|---|---|---|---|
| MPV >10.45fl | 66.3 | 62.8 | 0.718 (0.657-0.779) | 0.031 | <.001 |
| RDW > 43.65fl | 70.9 | 68 | 0.755 (0.698-0.811) | 0.029 | <.001 |
| D-Dimer >500.2 ng/dl | 73.5 | 78 | 0.843 (0.798-0.888) | 0.023 | <.001 |
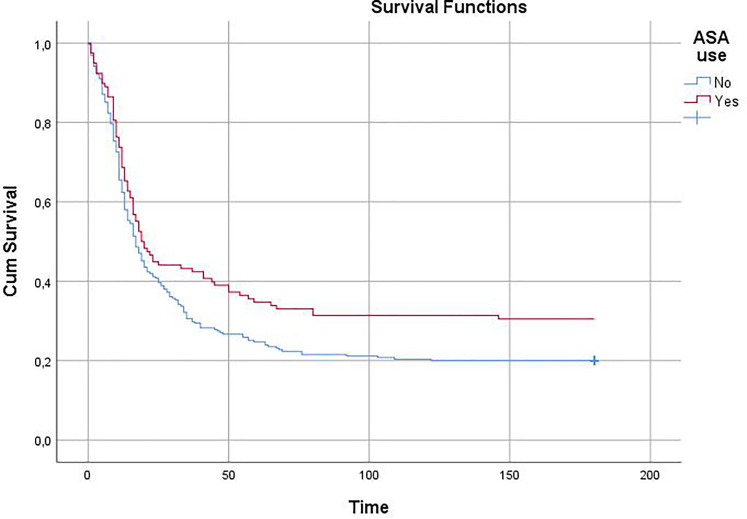
373 patients with MPV > 10.45 f/l and D-Dimer >500.2 were considered as a separate group. Demographic/clinical data of patients in this group who use and do not use aspirin are shown in Table-4. Patients using aspirin had higher rates of diabetes, hypertension, and male gender. There was no significant difference in other parameters. ASA use was found to be an independent predictor of mortality in this population in a regression analysis. (HR = 0.466; 95% CI = 0.264-0.823; P = .009, B = −,764) Survival analysis showed that patients with MPV > 10.45 f/l and D-Dimer >500.2 ng/mL, treatment with ASA had lower in-hospital and 6-month mortality than patients without ASA in ICU patients. (n = 373, Hazard ratio = 0.773; 95% CI = 0.595-0.992; P = .048, HR = 0.763; 95% CI = 0.590-0.987; P = .036, respectively.) Figure 3. Major bleeding was detected in 9.3% of ASA users (n = 11) and 6.7% of non-users (n = 17) (P = .365).
Table 4.
Demographic and Clinical Features of Patients with MPV >10.45fl and D-dimer >500.2 ng/dL in ICU.
| ASA use ( + ) n = 118 | ASA use (–) n = 255 | P value | |
|---|---|---|---|
| Age, mean ± SD | 73.9 ± 0.9 | 69.1 ± 1.9 | .265 |
| Gender (Male), n(%) | 86(72.9%) | 148 (58%) | .006 |
| White Blood Count (K/uL), median(IQR | 9.69 [7.13 to 12.80] | 11.03 [6.89 to 14.00] | .077 |
| C-reactive protein [mg/L], median [IQR] | 97.0 [57.4 to 152.2] | 88.4 [72.7 to 137.0] | .764 |
| Platelet [K/uL], median [IQR] | 193.3 [143.8 to 233.3] | 177.6 [131.9 to 206.1] | .139 |
| D-Dimer [ng/dL], median [IQR] | 4710 [2157 to 8000] | 5349 [2719 to 8000] | .100 |
| Mean platelet Volume [f/l], median [IQR] | 11.37 [10.75 to 11.63] | 11.31 [10.75 to 11.79] | .505 |
| Platelet Distribution Width [%], median [IQR] | 14.40 [13.20 to 15.08] | 14.42 [12.53 to 15.44] | .415 |
| Red Cell Distribution Width [f/l], median [IQR] | 43.43 [42.03 to 48.65] | 46.60 [42.77 to 49.38] | .339 |
| Hemoglobin [g/dL], median [IQR] | 12.3 [10.3 to 14.2] | 11.42 [9.46 to 12.38] | .729 |
| Creatinine [mg/dL], median [IQR] | 1.32 [1.07 to 2.23] | 1.12 [.73 to 2.20] | .150 |
| Sodium [mmol/L], median [IQR] | 140.0 [137.1 to 149.6] | 140.5 [137.6 to 145.7] | .182 |
| Troponin [ng/mL], median [IQR], [normal range 0 to 0.06 ng/mL] | 0.46 [0.05 to 2.33] | 0.30 [0.06 to 1.80] | .355 |
| Potassium [mmol/L], median [IQR] | 4.52 [4.09 to 4.80] | 4.35 [3.87 to 4.99] | .869 |
| Hypertension, n[%] | 98 [83.1%] | 150 [58.8%] | <.001 |
| Diabetes Mellitus, n[%] | 54 [45.8%] | 65 [25.1%] | <.001 |
| COPD, n [%] | 29[24.6%] | 61 [23.9%] | .897 |
| Hyperlipidemia, n[%] | 27 [22.9%] | 26 [10.2%] | .005 |
| Cerebrovascular disease, n [%] | 5[4.2%] | 8 [3.1%] | .559 |
| ICU Hospitalization day, median [IQR] | 9 [3 to 15] | 8 [4 to 17] | .992 |
Figure 3.
Kaplan-Meier showed showed that patients with MPV > 10.45 f/l and D-Dimer >500.2 ng/mL, treatment with ASA had lower 6-month mortality in ICU.
Discussion
This study was designed because studies on this subject were generally conducted with low sample numbers.10-12 In this large study population analysis we demostrated that MPV,PDW and RDW levels at hospital admission were signifcant parameters in COVID-19 patients for predicting mortality. Additionally, for ICU patiens who had above cutoff MPV and D-dimer levels, use of low-dose asetylsaliclic asit in additon to anti-coagulant was reduced in-hospital and total mortality.
MPV counts are used as a marker of the inflammatory response. Inflammatory cytokines have been shown to reflect both proinflammatory and prothrombotic states by regulating thrombopoiesis and MPV. Previous studies have shown a correlation between MPV and platelet function and activation.13,14 Large and small sized platelets in circulation are associated with the intensity of systemic inflammation. 5 Platelet production increases as platelet count decreases. Larger and young platelets are metabolically and enzymatically more active and have higher homeostasis than smaller platelets. 15 Three hypotheses related to platelet count and structure are proposed in COVID-19. Firstly, thrombocytopenia is likely due to infection of the bone marrow as other coronavıruses. Secondly, platelet destruction by the immune system. Thirdly, platelet consumption due to aggregation in the lungs. 16 These changes may explain the increase in MPV and PDW. In a study, high PDW shows a significant association with increased mortality a cut-off value of 17%. (X2 = 6.31, P = .012). 12 Our findings concordant with Güçlü et al. and Yun et al.17,18
In comparison to smaller ones, larger platelets have more granules, aggregate more rapidly with collagen, have higher TXA2 level.19,20 TXA2 is responsible for vasoconstriction, induction of vascular remodeling, and increasing platelet aggregation and adhesion. COX-1 is the only form of COX in platelets and is related to the synthesis of TXA2. 21 Aspirin causes platelet dysfunction and effectively inhibits platelet release by COX-1. Irreversible inactivation of COX-1 is induced, which inhibits platelet aggregation. Matthew L. Meizlish et al. showed that in-hospital aspirin compared to no antiplatelet therapy was associated with a significantly lower cumulative incidence of in-hospital death in COVID-19 patients (n = 638, HR = 0.522 [0.336-0.812]). 22 Although in some study there was no difference in platelet activation in COVID and non-COVID ICU patients due to their differences in baseline characteristics, critically ill COVID-19 patients showed higher circulating values of several inflammation and thrombosis markers, such as P-selectin and sCD40L. 23 Furtermore, in critically ill patients, increased MAPK signaling increased cytosolic phospholipase A2 activation and subsequently increased thromboxane formation. All these parameters significantly affect the platelet aggregation. Pretreatment of platelets from SARS-CoV-2 infected patients with aspirin abolishes this hyperactivity. 24 The preliminary result of RECOVERY trial, in which established as a randomised clinical trial to test aspirin efficiency for patients hospitalised with COVID-19 showed that administration of aspirin to all hospitalized patients did not affect outcomes. Therefore, it is critically important to initiate aspirin therapy in cases of increased platelet aggregation taking into account the profit-loss balance. As a result, we tried to find the answer to this question in our study with a large patient population.
It is known that D-dimer is associated with an increased risk of arterial and venous thrombotic events, irrespective of baseline vascular disease, therefore, it is necessary to consider not only MPV but also D-dimer levels in order to give antiaggregant in addition to anticoagulant therapy. D-dimer may be used to increase the sensitivity and specificity in the analysis of the hypercoagulable state in COVID-19 patients. 25 In our study, it was determined that aspirin use decreased mortality in intensive care patients according to MPV and D-dimer levels, but no significant difference was found in ward patients. Aspirin may not have provided additional benefit because of lower age, less comorbid conditions and less severity of disease, and lower prothrombotic markers such as MPV, CRP and D-dimer in ward patients. Hottz et al. demonstrated that platelet activation, degranulasyon and TXB2 syntesis, a metabolite from platelet TXA2, was increased from platelet from severe COVID but not mild subject. 26 This may be the reason for the decreased efficacy of aspirin in low-risk patients. In our study too, the positive effect of aspirin could not be demonstrated in low-risk patients. The clinical usefulness of ASA was demonstrated in improving overall survival in patients especially critically ill patients. 27 Because it has antiviral activity related to its ability to inhibit the coronavirus-induced nuclear factor kappa B pathway based on in silico docking analysis and in vitro cell culture study. 28 This efficacy of ASA has also been demonstrated in non-COVID patients. So that; A more recent meta-analysis of ten cohort studies enrolling 689 897 patients with sepsis revealed that ASA, administered either before or after sepsis, reduced ICU or hospital mortality. 29
As a result of its widespread use around the world and in the light of the latest scientific data, it has been determined that therapeutic anticoagulation decreased need for life support and improved outcome in hospitalized COVID-19 patients. 30 The newly published Therapeutic Anticoagulation with Heparin in Noncritically Ill Patients with Covid-19 study showed that therapeutic anticoagulation improves outcomes in non-critical patients. 31 Contrary to expectations, no significant benefit was observed in the effectiveness of therapeutic anticoagulation in high-risk patients of the same study. 32 However, the open label nature of the study is may have introduced bias due to the clinician's tendency to prefer therapeutic doses, especially in patients with high risk criteria in laboratory parameters, pre-existing cardiac disease, immobile patients. Likewise, patients who might benefit from anti-coagulant therapy may have preferred a lower dose because of old age or frailty. Such reasons may have led to reduced clinical benefit.
However, since the effectiveness of anticoagulation in arterial thrombophylaxis is controversial, the addition of anti-platelet to anticoagulant therapy has also come to the fore especially high risk and clinical ill patients. In a review, given anticoagulation when used alone and results showing that combined anticoagulant and antiplatelet therapy in the treatment of COVID-19, implied a better clinical outcome when compared with the use of anticoagulation alone without the occurrence of major bleeding. 33 In addition to its ability to prevent arterial thrombo-embolic events, there is some evidence that ASA may also reduce venous thromboembolism. 34
The RDW has played an important role due to its predictability the risk of death in the general population, 35 sepsis, 36 pneumonia, and other respiratory tract infections. 37 It is not surprising that our results demonstrate RDW as a significant predictor of severity in patients with COVID-19 infection. Mechanism of anisocytosis includes indirect erythrocyte damage due to intravascular coagulopathy, direct cytopathic damage, and disruption of iron metabolism due to inflammatory response. These conditions contribute to impaired erythrocyte formation and explain circulating size differences. 38 The results of this study suggest that RDW should be part of routine laboratory assessment of COVID-19.
This study has some limitations. Firstly it is a retrospective and single-center study. Treatment procedures had to change from time to time throughout the pandemic process, as the Ministry of Health was constantly updating the guidelines. Since the administration of antiaggregant especially for ward patients is determined according to the clinician's decision, the antiaggregant effect could not be clearly evaluated in mild-moderate symptom patients. It was not possible to determine the platelet function and proportion of young platelets. While severe bleeding can be detected, we do not have data on minor bleeding.
Conclusion
In this pandemia, which is spreading rapidly throughout the world and causing a major crisis, a simple and inexpensive method, the evaluation of hemogram parameters, can predict severity and mortality. Additionally in eligible patients, low-dose acetylsalicylic acid in addition to anticoagulant improves clinic outcomes without significantly increasing the risk of major bleeding.
Footnotes
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship and/or publication of this article.
ORCID iD: Faruk Aydınyılmaz https://orcid.org/0000-0003-1088-3559
References
- 1.Guan W-j, Ni Z-y, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708-1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lin S, Mao W, Zou Q, et al. Associations between hematological parameters and disease severity in patients with SARS-CoV-2 infection. J Clin Lab Anal. 2021;35(1):e23604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bilaloglu S, Aphinyanaphongs Y, Jones S, et al. Thrombosis in hospitalized patients with COVID-19 in a New York city health system. Jama. 2020;324(8):799-801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shimodaira M, Niwa T, Nakajima K, et al. Correlation between mean platelet volume and fasting plasma glucose levels in prediabetic and normoglycemic individuals. Cardiovasc Diabetol. 2013;12(1):1-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yolcu S, Beceren GN, Tomruk Ö, et al. Can mean platelet volume levels of trauma patients predict severity of trauma? Platelets. 2014;25(4):279-284. [DOI] [PubMed] [Google Scholar]
- 6.Lippi G, Sanchis-Gomar F, Favaloro EJ. Mean platelet volume in arterial and venous thrombotic disorders. Journal of Laboratory Medicine. 2020;44(5):305-312. [Google Scholar]
- 7.Sarkar RN, Das CK, Bhattacharjee U, et al. Platelet indices as a marker of severity in Non-diabetic NonHypertensive acute ischemic stroke patients. J Assoc Physicians India. 2018;66(7):40-42. [PubMed] [Google Scholar]
- 8.Gao Y, Li Y, Yu X, et al. The impact of various platelet indices as prognostic markers of septic shock. PLoS One. 2014;9(8):e103761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ramachandran P, Gajendran M, Perisetti A, et al. Red blood cell distribution width (RDW) in hospitalized COVID-19 patients. medRxiv. 2020. 10.1101/2020.06.29.20143081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Foy BH, Carlson JC, Reinertsen E, et al. Association of red blood cell distribution width with mortality risk in hospitalized adults with SARS-CoV-2 infection. JAMA Network Open. 2020;3(9):e2022058-e2022058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lorente L, Martín MM, Argueso M, et al. Association between red blood cell distribution width and mortality of COVID-19 patients. Anaesthesia Critical Care & Pain Medicine. 2021;40(1):100777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gowda SB, Gosavi S, Rao AA, et al. Prognosis of COVID-19: red cell distribution width, platelet distribution width, and C-reactive protein. Cureus. 2021;13(2):e13078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sansanayudh N, Muntham D, Yamwong S, et al. The association between mean platelet volume and cardiovascular risk factors. Eur J Intern Med. 2016;30:37-42. 10.1016/j.ejim.2015.11.028 [DOI] [PubMed] [Google Scholar]
- 14.Frelinger AL, III, Grace RF, Gerrits AJ, et al. Platelet function tests, independent of platelet count, are associated with bleeding severity in ITP. Blood. The Journal of the American Society of Hematology. 2015;126(7):873-879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Martin J, Trowbridge E, Salmon G, et al. The biological significance of platelet volume: its relationship to bleeding time, platelet thromboxane B2 production and megakaryocyte nuclear DNA concentration. Thromb Res. 1983;32(5):443-460. [DOI] [PubMed] [Google Scholar]
- 16.Xu P, Zhou Q, Xu J. Mechanism of thrombocytopenia in COVID-19 patients. Ann Hematol. 2020;99(6):1205-1208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Güçlü E, Kocayiğit H, Okan HD, et al. Effect of COVID-19 on platelet count and its indices. Revista da Associação Médica Brasileira. 2020;66(8):1122-1127. [DOI] [PubMed] [Google Scholar]
- 18.Yun H, Sun Z, Wu J, et al. Laboratory data analysis of novel coronavirus (COVID-19) screening in 2510 patients. Clin Chim Acta. 2020;507:94-97. 10.1016/j.cca.2020.04.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vizioli L, Muscari S, Muscari A. The relationship of mean platelet volume with the risk and prognosis of cardiovascular diseases. Int J Clin Pract. 2009;63(10):1509-1515. [DOI] [PubMed] [Google Scholar]
- 20.Park Y, Schoene N, Harris W. Mean platelet volume as an indicator of platelet activation: methodological issues. Platelets. 2002;13(5-6):301-306. [DOI] [PubMed] [Google Scholar]
- 21.Bezugla Y, Kolada A, Kamionka S, et al. COX-1 and COX-2 contribute differentially to the LPS-induced release of PGE2 and TxA2 in liver macrophages. Prostaglandins Other Lipid Mediators. 2006;79(1-2):93-100. [DOI] [PubMed] [Google Scholar]
- 22.Meizlish ML, Goshua G, Liu Y, et al. Intermediate-dose anticoagulation, aspirin, and in-hospital mortality in COVID-19: a propensity score-matched analysis. Am J Hematol. 2021;96(4):471-479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Campo G, Contoli M, Fogagnolo A, et al. Over time relationship between platelet reactivity, myocardial injury and mortality in patients with SARS-CoV-2-associated respiratory failure. Platelets. 2020;32(4):1-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Manne BK, Denorme F, Middleton EA, et al. Platelet gene expression and function in patients with COVID-19. Blood. 2020;136(11):1317-1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Salamanna F, Maglio M, Landini MP, et al. Platelet functions and activities as potential hematologic parameters related to coronavirus disease 2019 (covid-19). Platelets. 2020;31(5):627-632. [DOI] [PubMed] [Google Scholar]
- 26.Hottz ED, Azevedo-Quintanilha IG, Palhinha L, et al. Platelet activation and platelet-monocyte aggregate formation trigger tissue factor expression in patients with severe COVID-19. Blood. 2020;136(11):1330-1341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bianconi V, Violi F, Fallarino F, et al. Is acetylsalicylic acid a safe and potentially useful choice for adult patients with COVID-19? Drugs. 2020;80(14):1-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Müller C, Karl N, Ziebuhr J, et al. L-lysine acetylsalicylate+glycine impairs coronavirus replication. J Antivir Antiretrovir. 2016;8(4):142-150. [Google Scholar]
- 29.Ouyang Y, Wang Y, Liu B, et al. Effects of antiplatelet therapy on the mortality rate of patients with sepsis: a meta-analysis. J Crit Care. 2019;50:162-168. 10.1016/j.jcrc.2018.12.004 [DOI] [PubMed] [Google Scholar]
- 30.Houston BL, Lawler PR, Goligher EC, et al. Anti-Thrombotic therapy to ameliorate complications of COVID-19 (ATTACC): study design and methodology for an international, adaptive Bayesian randomized controlled trial. Clinical Trials. 2020;17(5):491-500. [DOI] [PubMed] [Google Scholar]
- 31.A-a ATTACC, Investigators R-C. Therapeutic anticoagulation with heparin in noncritically ill patients with covid-19. N Engl J Med. 2021;385(9):790–802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.A-a REMAP-CAP, Investigators A. Therapeutic anticoagulation with heparin in critically ill patients with covid-19. N Engl J Med. 2021;385(9):777–789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Matli K, Farah R, Maalouf M, et al. Role of combining anticoagulant and antiplatelet agents in COVID-19 treatment: a rapid review. Open Heart. 2021;8(1):e001628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Simes J, Becattini C, Agnelli G, et al. Aspirin for the prevention of recurrent venous thromboembolism: the INSPIRE collaboration. Circulation. 2014;130(13):1062-1071. [DOI] [PubMed] [Google Scholar]
- 35.Henry BM, De Oliveira MHS, Benoit S, et al. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): a meta-analysis. Clinical Chemistry and Laboratory Medicine (CCLM. 2020;58(7):1021-1028. [DOI] [PubMed] [Google Scholar]
- 36.Hu Z-D, Lippi G, Montagnana M. Diagnostic and prognostic value of red blood cell distribution width in sepsis: a narrative review. Clin Biochem. 2020;77:1-6. 10.1016/j.clinbiochem.2020.01.001 [DOI] [PubMed] [Google Scholar]
- 37.Lee JH, Chung HJ, Kim K, et al. Red cell distribution width as a prognostic marker in patients with community-acquired pneumonia. Am J Emerg Med. 2013;31(1):72-79. [DOI] [PubMed] [Google Scholar]
- 38.Henry BM, Benoit JL, Benoit S, et al. Red blood cell distribution width (RDW) predicts COVID-19 severity: a prospective, observational study from the cincinnati SARS-CoV-2 emergency department cohort. Diagnostics. 2020;10(9):618. [DOI] [PMC free article] [PubMed] [Google Scholar]