Abstract
Western and Eastern practices have traditionally differed in their approach to treating lateral lymph nodes in rectal cancer. While Western clinicians have primarily favored neoadjuvant (chemo)radiotherapy to sterilize lateral compartments, Eastern physicians have often opted for the surgical removed of lymphatic tissue with a lateral lymph node dissection without neoadjuvant treatment. The literature suggests similar oncological outcomes for these two separate techniques, while tangible differences exist. The combination of these paradigms may be beneficial in reducing overall morbidity while sustaining low recurrence rates. This article considers traditional Eastern and Western perspectives, discusses nodal features important for predicting malignancy and attempts to stimulate international, multidisciplinary consensus and collaboration.
Keywords: lateral lymph node dissection, lateral lymph nodes, neoadjuvant treatment
Western clinicians have primarily favoured neoadjuvant (chemo)radiotherapy to sterilize lateral compartments, while Eastern physicians have often opted for the surgical removed of lymphatic tissue with a lateral lymph node dissection (LLND) without neoadjuvant treatment. However, tangible differences exist between these two paradigms and a combined technique may be beneficial in reducing overall morbidity while sustaining low recurrence rates. This article considers traditional Eastern and Western perspectives, discusses nodal features important for predicting malignancy and attempts to stimulate international, multidisciplinary consensus and collaboration.
1. INTRODUCTION
Global research is increasingly presenting evidence for the significance of lateral lymph nodes (LLNs) in patients with low, locally advanced rectal cancer (LARC). 1 , 2 , 3 , 4 , 5 Low LARC tumors spread lymphatically towards the lateral pelvic compartments where LLNs are situated. 1 LLNs surround the internal iliac and obturator vessels and are not standardly removed during total mesorectal excision (TME) rectal surgery. 6 , 7 , 8 , 9 , 10 , 11
LARC has traditionally been treated differently in various parts of the world. 3 , 5 , 12 , 13 , 14 Eastern countries have customarily removed the lateral compartments prophylactically with a lateral lymph node dissection (LLND) for all tumors situated below the peritoneal reflection. 12 , 13 , 15 , 16 Retrospective Japanese studies indicate good overall survival and local control for those undergoing TME and LLND surgery. 15 , 17 Furthermore, while initially high, urinary and sexual dysfunction rates have decreased due to an increase in minimally invasive surgery with nerve‐sparing techniques. 18 , 19 , 20 In contrast, Western countries have favored neoadjuvant treatment with (chemo)radiotherapy (n(C)RT), believing irradiation to sterilize the lateral compartments. 8 , 9 , 14 Local recurrence (LR) rates have decreased to 5%–10% and morbidity and complications associated with the LLND are avoided. 20 , 21
2. EAST vs WEST
A traditional concept held by Western physicians is that lateral nodal disease represents metastatic disease. The primary focus has been to treat this by sterilizing the lateral compartments with n(C)RT + TME surgery without an LLND. 4 , 5 , 14 The Dutch TME trial investigated patients who underwent radiotherapy (RT) + TME vs TME alone and compared these to a matched cohort of Japanese patients undergoing TME + LLND. They found good rates of local control for both RT + TME and TME + LLND, suggesting that RT is also adequate in treating the lateral compartments. 22 Similar results were found in the randomized controlled trial by Nagawa et al and retrospective cohort by Watanabe et al with no significant differences between patients undergoing RT + TME compared to TME + LLND in the absence of enlarged LLNs. 23 , 24 A meta‐analysis of 6865 patients compared patients who did and did not undergo an LLND, concluding that additional LLND did not significantly improve prognosis. 25 However, other evidence suggests that n(C)RT and TME may not be sufficient in the presence of enlarged LLNs, with doubled 5‐y lateral LR (LLR) rates when LLNs with malignant features are present (20.9% vs 10.3% 3 ) and an increase in LLRs associated with LLN size; 87% of patients with an LLN ≥10 mm developed LLR within 5 y. 26 Such results indicate that CRT + TME surgery alone may be inadequate for certain cases.
Lymph nodes have customarily been treated surgically in Eastern countries, where prophylactic LLNDs are often performed for all low LARC patients because high LR rates were found for those treated only with TME surgery. 15 , 23 , 27 Fujita et al found that patients without enlarged LLNs (LLNs ≥10 mm were excluded) who underwent prophylactic LLND had a lower LR rate (7.4% vs 12.6%) and higher LR‐free survival rate (87.7% vs 82.4%) than TME surgery alone, 15 with similar urinary and sexual dysfunction rates. 28 , 29 Two additional studies found micro‐metastases present in histologically negative LLNs that had been removed during prophylactic LLNDs (20%–24%); both associated with an increased risk of LR (43% vs 11.5%). 30 , 31 The Japanese Society for Cancer of the Colon and Rectum (JSCCR) currently recommends a prophylactic LLND for patients with a tumor below the peritoneal reflection. 16
Unfortunately, many studies have excluded enlarged LLNs, which makes it difficult to accurately discuss the LR rates for enlarged LLNs after prophylactic LLND. Kanemitsu et al did not exclude enlarged LLNs and found, in a retrospective cohort of 1191 patients from two high‐volume Japanese centers, that enlarged LLNs treated with a prophylactic (often bilateral) LLND had LR rates up to 22%. 32 Even those with LLNs <10 mm, LR rates were around 14%. Fujita et al presented similar 5‐y LR rates of 12% for ME + LLND. 15 This is relatively high, considering the current overall LR rates are 5%–10% and it is expected that for larger LLNs this rate may be even higher. 21 , 33 This means that both approaches separately, TME + LLND and (C)RT + TME, may be insufficient for enlarged LLNs.
3. EAST MEETS WEST
Similar LR rates for prophylactic LLND vs n(C)RT, combined with often higher sexual and urinary dysfunction rates, prompted a search for alternative treatment schedules. While LLNDs performed by experienced surgeons reveal the lowest LR rates, it has been hypothesized that the combination of n(C)RT with selective LLND may be the future for enlarged LLNs. 34 Akiyoshi et al 13 studied 38 patients who were considered “high‐risk” due to primarily enlarged (≥7 mm short‐axis [SA]) LLNs and underwent selective LLND; these patients resulted in a 0% LR rate after 3 y. 13 They further found that primarily enlarged LLNs (≥8 mm, SA) that remained persistently enlarged after CRT (>5 mm, SA) had a significantly higher percentage of positive metastases (75% vs 20%, P < .0001). 12 Malakorn et al found that LLN size of >5 mm after neoadjuvant treatment was associated with pathological positivity. 2 No pathological positivity was found for LLNs <5 mm and for the patients who had pathologically positive LLNs and underwent a selective LLND; the LR rate was 0% after 39 months. 2
Various studies have considered such LLN features related to oncological outcomes; Shirouzu et al found that the number of LLNs was related to decreased 5‐y survival (<3 LLNs: 60%, ≥3 LLNs: 16.7% 35 ) and others found that low LARC tumors with positive mesorectal lymph nodes were highly associated with positive LLNs. 17 , 36 In a cohort of 1068 LARCs who underwent CRT, 67 patients had LLNs measuring ≥5 mm on the primary magnetic resonance imaging (MRI) and underwent LLNDs. Overall, 40% of this group (32/82) had pathologically positive LLNs and this proportion increased to 59% when considering only LLNs that were ≥10 mm. 37
This evidence suggests that neoadjuvant treatment can be useful, especially for the sterilization of smaller LLNs, 4 but that in situations where enlarged LLNs do not sufficiently downsize, n(C)RT can be inadequate and a selective LLND is necessary. The selective LLND combines Western and Eastern principles and could lead to an overall reduction in LLRs and morbidity due to the selection of high‐risk cases. Western perceptions of LLNs also appear to be shifting 3 , 14 , 38 with an increase in research. 3 , 4 , 7 , 8 , 9 A recent survey among 62 Dutch colorectal surgeons found that only 10% believe LLNs to represent metastatic disease (Hazen et al, submitted). Considering this shift in mentality, LLN features should be discussed.
4. LATERAL LYMPH NODES: SIZE
While advances in multidisciplinary treatment has resulted in overall LR rates of approximately 5%–10% for LARC patients, 5 , 21 , 39 , 40 the proportion of LLRs is increasing. LLRs currently account for almost 50% of all LRs. 33 This increase in LLR is most likely explained by the adequate treatment of LARC and mesorectal lymph nodes with TME surgery, but inadequate treatment of malignant LLNs. 33 It is important to ensure that there is awareness and appropriate treatment of suspicious LLNs, but what makes an LLN suspicious? Research provides two characteristics that increase the likelihood that an LLN is suspicious.
The first is size. Unlike mesorectal lymph nodes where size is just one of many factors associated with its malignancy, 39 , 41 LLR risks have been significantly associated with the size (SA) of an LLN. 11 , 26 , 42 , 43 Primarily enlarged LLNs (>10 mm) have LLR rates reaching 40% 16 , 17 , 18 and increase even further when remaining persistently enlarged after neoadjuvant treatment. 8 , 9 , 10 , 26 , 43 , 44 Ogura et al 9 found in a retrospective, international cohort of 1216 patients with re‐evaluation of all MRIs, that patients with LLNs measuring ≥7 mm on the primary MRI resulted in an LLR rate of 19.5% after 5 y. When considering the restaging size (SA) after neoadjuvant treatment, significant associations were found. A size of >4 mm (SA; for LLNs located in the internal iliac compartment) resulted in an LLR rate of 52.3% after 5 y, while LLNs located in the obturator compartment that were >6 mm on the restaging MRI, had a 17.8% LLR rate. 8 , 9 These results are primarily applicable to Western centers, as most Eastern clinics do not standardly provide neoadjuvant treatment (Table 1).
TABLE 1.
Oncological outcomes for patients with lateral lymph nodes who received neoadjuvant chemoradiotherapy ((C)RT)
| Design | Patients (n) | Patient population | LLN size based on? | Oncological outcomes after CRT | |
|---|---|---|---|---|---|
| Kim et al 26 | Retrospective | 366 | cT3/T4 primary rectal cancer, <8 cm from anal verge | Restaging MRI |
5‐y LR: 7.9% (21% central pelvis, 82% lateral pelvis)
|
| Kim et al 42 | Retrospective | 443 | Stage 2/3 primary rectal cancer, <15 cm from anal verge | Restaging MRI |
5‐y LR: 12% (53 patients)
LLN >10 mm and 2 LLNs as increased risk factors (OR: 1.5, CI 95% 1.2–1.9) |
| Akiyoshi et al 13 | Retrospective | 127 | Stage 2/3 primary low rectal cancer | Primary MRI |
3‐y LR 3.4% TME vs 0% TME + LLND (LLN 7 mm or larger) LLN 7 mm or larger on MRI: 66% pathological positive |
| Kim et al 43 | Retrospective | 900 | Stage 2/3 primary rectal cancer, <10 cm from anal verge | Primary MRI |
5‐y LR 7.2% (65 patients)
LLNs <5 mm, 5–9.9 mm, >10 mm
|
| Malakorn et al 2 | Retrospective | 64 | Primary rectal cancer and suspicious LLN on primary MRI | Restaging MRI |
OS: 79% (LLN−) vs 61% (LLN+) DFS: 84% (LLN−) vs 66% (LLN+) LLN <5 mm post‐CRT = 0% positive LLN >5 mm post‐CRT = 64% positive |
| Ogura et al 8 | Retrospective | 1216 | cT3/4 primary rectal cancer, <8 cm anal verge | Primary MRI |
LLN >7 mm, HR for LLR = 2.06 5‐y LR: 108 (10%)
|
| Ogura et al 9 | Retrospective: a subset of patients from Ogura et al 8 | 741/1216 | cT3/4 primary rectal cancer, <8 cm anal verge, restaging MRI | Restaging MRI |
>7 mm primary MRI = 17.9% LLR
|
DFS, disease‐free survival; HR, hazard ratio; LLN, lateral lymph node; LLR, lateral local recurrence; LR, local recurrence; LRFS, lateral recurrence‐free survival; MRI, magnetic resonance imaging; OR, odds ratio; OS, overall survival; RFS, recurrence‐free survival.
5. LATERAL LYMPH NODES: LOCATION
The second feature indicative of increased LLR is the anatomical location. The lateral pelvis contains an abundance of lymphatic tissue, nerves, and vessels. The lateral compartments, which contain LLNs, are located between the bony pelvis and pelvic muscles laterally and the rectum with its mesorectum and mesorectal fascia medially. Various visual anatomical atlases exist 8 , 10 depicting individual lateral compartments. The internal iliac compartment (see Figure 1) contains lymphatic tissue located medially of the lateral border of the internal iliac artery. Lymphatic tissue located caudally of where the internal iliac artery exits the pelvis through the sciatic foramen in the form of the superior gluteal artery, is considered part of the obturator compartment. This compartment also includes all lymphatic tissue located laterally of the lateral border of the internal iliac artery (see Figure 1).
FIGURE 1.
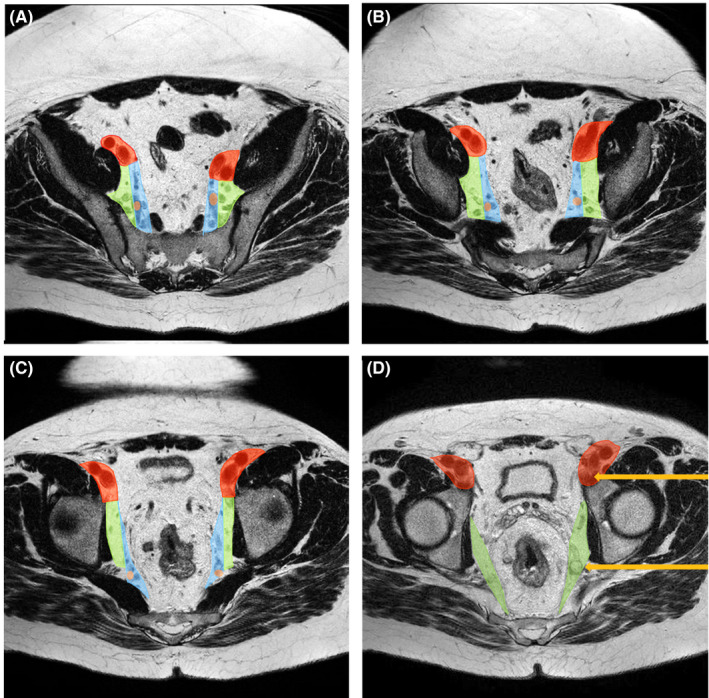
Lateral lymph node compartments. A–D: Caudal progression through a T2‐MRI. Red: external iliac compartment surrounding the externa iliac vessels. Green: obturator compartment located lateral of the lateral border of the internal iliac artery (brown spot) and caudal of when the internal iliac artery exits the pelvis. Blue: internal iliac compartment located medial of the lateral border of the internal iliac artery (brown spot). Orange arrows indicate lateral lymph nodes
In the Lateral Node Consortium study only 22% of internal iliac LLNs, compared to 63% of obturator LLNs, reduced significantly in size after neoadjuvant treatment and the LLR rate was significantly higher for persistently enlarged LLNs in the internal iliac compartment, 52% vs 17.8% after 5 y. 8 , 9 A recent study has attempted to explain why internal iliac and obturator LLNs behave differently. Schaap et al 11 found noticeable differences between different locations. First of all, while a higher cT‐stage and cN‐stage were significantly associated with predicting the occurrence of an enlarged (≥7 mm) LLN in the obturator compartment, there were no predicting features associated with the occurrence of an enlarged internal iliac LLN. Second, persistently enlarged internal iliac LLNs (≥7 mm before CRT and >4 mm after CRT) were predictive of developing LLR, but did not influence distant metastasis (DM) rates. Alternatively, persistently enlarged obturator LLNs (≥7 mm before CRT and >6 mm after CRT) had a significantly higher 5‐y DM rate and lower 5‐y cancer‐specific survival (CSS), compared to obturator LLNs <6 mm. These differences disappeared for patients with enlarged obturator LLNs who underwent an LLND. 11
Akiyoshi et al compared internal iliac LLNs to mesorectal nodes and LLNs located beyond the internal iliac compartment. 45 They found that internal iliac LLNs and N2a mesorectal lymph nodes resulted in similar 5‐y overall survival rates (45% and 45%), while N2b mesorectal nodes and LLNs located beyond the internal iliac area were associated with worse survival outcomes (32% and 29%). This difference in survival stratified to anatomical location has led to the conclusion that LLNs may be a regional disease when considering internal iliac LLNs, but distant disease for LLNs beyond this area. 45 Anatomical location may also reflect differences in etiology and disease advancement. Considering the disappearance of these differences after LLND, the LLND may not only improve local control for internal iliac LLNs, but help control the chances of systemic spread caused by persistently enlarged obturator LLNs.
The available evidence advocates for the consideration of size (SA in mm) and anatomical location as primary factors when considering the malignancy of LLNs. It is important to note that these same studies found no LLRs for enlarged LLNs surrounding the external iliac vessels, which is why these lymph nodes are not mentioned further. Increased DM rates were found for external iliac lymph nodes (hazard ratio 2.5 [95% CI, 1.4‐4.4] 8 , 9 , 10 , 11 ) implying enlarged external iliac LLNs to be more indicative of systemic disease.
6. TOWARDS INTERNATIONAL COLLABORATION
An increase in research into LLNs represents a positive development, in which international perspectives appear to be nearing each other. With these steps, it is important to underline the need for international guidelines and consensus concerning the ideal treatment of lateral nodal disease. Small steps are being made in both directions, with Western clinicians beginning to perform LLND procedures in high‐risk cases.
Considering these developments, concise terminology is essential. The latest Tumour‐Node‐Metastasis classification of the American Joint Committee on Cancer describes internal iliac LLNs as a regional disease, while obturator, external iliac, and common iliac lymph nodes are defined as metastatic disease. This is in contrast to the description of LLNs to be a local disease, accepted by many Eastern clinicians and increasingly so by Western physicians. 6 , 27 , 46 , 47 This reflects the still insufficient awareness of LLNs in the West and exposes the lack of explicit recommendations for enlarged LLNs in Western guidelines. 48 , 49 The European Society for Gastrointestinal and Abdominal Radiology meeting in 2016 concluded that there was still insufficient evidence for a separate guideline for LLNs and the Royal College of Radiologists guidelines from 2014 state that only the presence or absence of malignant nodes should be reported. Exact definitions, whether this is for extra‐mesorectal lymph nodes and/or mesorectal lymph nodes and what makes a lymph node suspicious are not mentioned. 48 , 50
The evidence currently available demonstrates clinical implications related to the size and/or anatomical location of an LLN. 8 , 9 , 10 , 11 , 12 , 13 Sufficient awareness of LLNs and knowledge of their consequences is therefore necessary. One step towards awareness in Western clinics could be by introducing templates in radiology reporting. Brown et al 51 found that many aspects of radiology reports improved after introducing a template.
Once there is an appropriate level of awareness, suitable treatment decisions can follow. Some Western clinicians believe that an irradiation boost may provide the extra enhancement required to treat LLNs without additional LLND surgery. 52 , 53 , 54 Just two studies have evaluated a boost in LLNs, with mixed results. Chen et al 53 studied 12 patients with suspicious LLNs, all of which had received a “boost” as treatment (3 × 5.4 Gy). They were compared to 41 patients without LLNs who received standard CRT (25 × 1.8 Gy). They found no significant differences in overall survival outcomes. The second study, also with only 12 patients, determined that a boost did not result in an increased risk of toxicity or perioperative complications. 54 However, more research is warranted before a boost should be considered part of a treatment schedule. Furthermore, with the potentiality that according to Ogura et al, 8 only 22% of internal iliac LLNs adequately respond to neoadjuvant treatment, the desired benefit of a boost may be limited. 8
Many studies point to additional surgery where persistently enlarged LLNs are surgically removed during an LLND. This is contrast to “node‐picking,” the removal of individual LLNs without removing the entire lateral compartments. Based on two studies with very limited patient numbers, there is currently insufficient evidence for “node‐picking” compared to a formal LLND. One group of 12 patients undergoing node‐picking resulted in a 51% recurrence rate, with all recurrences located in the lateral compartments. 8 Another, with just 30 patients, found that in five cases no lymph node or tumor cells were found by the pathologist. 55 In comparison, a formal LLND has been proven to significantly decrease LLR rates. Patients with primarily enlarged LLNs (≥7 mm) who underwent TME and LLND surgery had a significantly lower 5‐y LLR rate of 5.7% compared to 19.5%. Those with persistently enlarged LLNs in the internal iliac compartment (>4 mm on the restaging MRI) who underwent TME and LLND surgery, had a 5‐y LLR rate of 8.7% instead of 52.3% for those receiving only TME surgery. 8 , 9 These outcomes demonstrate the ability of an LLND to decrease LLR rates for certain patients. With increasing evidence for the benefits of the selective LLND, it is essential that the procedure should be internationally standardized to ensure a broad foundation of surgical consistency. Expert surgeons should perform minimally invasive LLNDs to decrease the chances of complications and care should be taken to remove all lymphatic tissue from both the obturator and internal iliac compartments in a nerve‐sparing manner. This can be done with or without additional resection of all side branches of the internal iliac artery, depending on the extent of lateral nodal disease.
Randomized trials investigating enlarged LLNs are technically challenging and low accrual is, for example, seen in the current Chinese trial randomizing between TME and TME + LLND for preoperatively enlarged LLNs. 56 Alternatively, the LaNoReC is an international prospective registration study currently including rectal cancer patients with at least one enlarged LLN (≥7 mm). Eligible patients undergo standardized neoadjuvant treatment with expert review of all MRI images and delineation plans. Patients with persistently enlarged LLNs on the restaging MRI (>4 mm internal iliac or >6 mm obturator compartment) are advised to undergo a selective LLND. This study aims to reveal whether standardization and centralization, with quality‐control measures for the multidisciplinary approach to enlarged LLNs, can significantly decrease the LLR rates. Important secondary outcomes are the quality of life and functional outcomes after minimally invasive, nerve‐sparing LLND procedures.
7. CONCLUSION
Lateral nodal disease warrants a broad understanding of LLNs. Evidence presented here suggests that size (SA) and anatomical location of LLNs are crucial features related to LLR risks and should be reviewed and reported for all patients. Western concepts still support the application of neoadjuvant treatment, but are beginning to understand the necessity of a selective LLND for high‐risk cases. The application of CRT to sterilize smaller nodes, followed by the selective application of the LLND procedure for persistently enlarged LLNs, may be the ideal treatment paradigm. The evidence presented here suggests that international consensus regarding LLNs is possible and that international collaboration, including the global agreement on terminology and treatment guidelines, should be pursued.
DISCLOSURE
Funding: No funding was received.
Conflict of interest: The authors declare no conflicts of interests for this article.
Author Contribution: All authors contributed substantially to the conception, creation and interpretation of this work and were involved in drafting and revising it critically. Each author approved the final version to be published and agree to be accountable for all aspects of the work.
Sluckin TC, Hazen S‐MJA, Kusters M. From “East vs West” towards international multidisciplinary collaboration: An appraisal of lateral lymph nodes in rectal cancer. Ann Gastroenterol Surg. 2021;5:731–737. 10.1002/ags3.12490
REFERENCES
- 1. Steup WH, Moriya Y, van de Velde CJ. Patterns of lymphatic spread in rectal cancer. A topographical analysis on lymph node metastases. Eur J Cancer. 2002;38(7):911–8. [DOI] [PubMed] [Google Scholar]
- 2. Malakorn S, Yang Y, Bednarski BK, Kaur H, You YN, Holliday EB, et al. Who should get lateral pelvic lymph node dissection after neoadjuvant chemoradiation? Dis Colon Rectum. 2019;62(10):1158–66. [DOI] [PubMed] [Google Scholar]
- 3. Kusters M, Slater A, Muirhead R, Hompes R, Guy RJ, Jones OM, et al. What to do with lateral nodal disease in low locally advanced rectal cancer? A call for further reflection and research. Dis Colon Rectum. 2017;60(6):577–85. [DOI] [PubMed] [Google Scholar]
- 4. Kusters M, Uehara K, Velde C, Moriya Y. Is there any reason to still consider lateral lymph node dissection in rectal cancer? Rationale and technique. Clin Colon Rectal Surg. 2017;30(5):346–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Christou N, Meyer J, Toso C, Ris F, Buchs NC. Lateral lymph node dissection for low rectal cancer: is it necessary? World J Gastroenterol. 2019;25(31):4294–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Nakamura T, Watanabe M. Lateral lymph node dissection for lower rectal cancer. World J Surg. 2013;37(8):1808–13. [DOI] [PubMed] [Google Scholar]
- 7. Kusters M, Marijnen CA, van de Velde CJ, Rutten HJ, Lahaye MJ, Kim JH, et al. Patterns of local recurrence in rectal cancer; a study of the Dutch TME trial. Eur J Surg Oncol. 2010;36(5):470–6. [DOI] [PubMed] [Google Scholar]
- 8. Ogura A, Konishi T, Beets GL, Cunningham C, Garcia‐Aguilar J, Iversen H, et al. Lateral nodal features on restaging magnetic resonance imaging associated with lateral local recurrence in low rectal cancer after neoadjuvant chemoradiotherapy or radiotherapy. JAMA Surg. 2019;154(9):e192172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Ogura A, Konishi T, Cunningham C, Garcia‐Aguilar J, Iversen H, Toda S, et al. Neoadjuvant (Chemo)radiotherapy with total mesorectal excision only is not sufficient to prevent lateral local recurrence in enlarged nodes: results of the multicenter lateral node study of patients with low cT3/4 rectal cancer. J Clin Oncol. 2019;37(1):33–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Schaap DP, Boogerd LSF, Konishi T, Cunningham C, Ogura A, Garcia‐Aguilar J, et al. Rectal cancer lateral lymph nodes: multicentre study of the impact of obturator and internal iliac nodes on oncological outcomes. Br J Surg. 2021;108(2):205–13. [DOI] [PubMed] [Google Scholar]
- 11. Schaap DP, Ogura A, Nederend J, Maas M, Cnossen JS, Creemers GJ, et al. Prognostic implications of MRI‐detected lateral nodal disease and extramural vascular invasion in rectal cancer. Br J Surg. 2018;105(13):1844–52. [DOI] [PubMed] [Google Scholar]
- 12. Akiyoshi T, Matsueda K, Hiratsuka M, Unno T, Nagata J, Nagasaki T, et al. Indications for lateral pelvic lymph node dissection based on magnetic resonance imaging before and after preoperative chemoradiotherapy in patients with advanced low‐rectal cancer. Ann Surg Oncol. 2015;22(Suppl 3):S614–20. [DOI] [PubMed] [Google Scholar]
- 13. Akiyoshi T, Ueno M, Matsueda K, Konishi T, Fujimoto Y, Nagayama S, et al. Selective lateral pelvic lymph node dissection in patients with advanced low rectal cancer treated with preoperative chemoradiotherapy based on pretreatment imaging. Ann Surg Oncol. 2014;21(1):189–96. [DOI] [PubMed] [Google Scholar]
- 14. Williamson JS, Quyn AJ, Sagar PM. Rectal cancer lateral pelvic sidewall lymph nodes: a review of controversies and management. Br J Surg. 2020;107(12):1562–9. [DOI] [PubMed] [Google Scholar]
- 15. Fujita S, Mizusawa J, Kanemitsu Y, Ito M, Kinugasa Y, Komori K, et al. Mesorectal excision with or without lateral lymph node dissection for clinical stage II/III lower rectal cancer (JCOG0212): a multicenter, randomized controlled, noninferiority trial. Ann Surg. 2017;266(2):201–7. [DOI] [PubMed] [Google Scholar]
- 16. Watanabe T, Muro K, Ajioka Y, Hashiguchi Y, Ito Y, Saito Y, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2016 for the treatment of colorectal cancer. Int J Clin Oncol. 2018;23(1):1–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Ueno M, Oya M, Azekura K, Yamaguchi T, Muto T. Incidence and prognostic significance of lateral lymph node metastasis in patients with advanced low rectal cancer. Br J Surg. 2005;92(6):756–63. [DOI] [PubMed] [Google Scholar]
- 18. Moriya Y, Sugihara K, Akasu T, Fujita S. Importance of extended lymphadenectomy with lateral node dissection for advanced lower rectal cancer. World J Surg. 1997;21(7):728–32. [DOI] [PubMed] [Google Scholar]
- 19. Sugihara K, Moriya Y, Akasu T, Fujita S. Pelvic autonomic nerve preservation for patients with rectal carcinoma. Oncologic and functional outcome. Cancer. 1996;78(9):1871–80. [PubMed] [Google Scholar]
- 20. Konishi T, Watanabe T, Nagawa H, Oya M, Ueno M, Kuroyanagi H, et al. Preoperative chemoradiation and extended pelvic lymphadenectomy for rectal cancer: Two distinct principles. World J Gastrointest Surg. 2010;2(4):95–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Kapiteijn E, Marijnen CA, Nagtegaal ID, Putter H, Steup WH, Wiggers T, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med. 2001;345(9):638–46. [DOI] [PubMed] [Google Scholar]
- 22. Kusters M, Beets GL, van de Velde CJ, Beets‐Tan RG, Marijnen CA, Rutten HJ, et al. A comparison between the treatment of low rectal cancer in Japan and the Netherlands, focusing on the patterns of local recurrence. Ann Surg. 2009;249(2):229–35. [DOI] [PubMed] [Google Scholar]
- 23. Watanabe T, Matsuda K, Nozawa K, Kobunai T. Lateral pelvic lymph node dissection or chemoradiotherapy: which is the procedure of choice to reduce local recurrence rate in lower rectal cancer? Ann Surg. 2008;248(2):342–3. author reply 3. [DOI] [PubMed] [Google Scholar]
- 24. Nagawa H, Muto T, Sunouchi K, Higuchi Y, Tsurita G, Watanabe T, et al. Randomized, controlled trial of lateral node dissection vs. nerve‐preserving resection in patients with rectal cancer after preoperative radiotherapy. Dis Colon Rectum. 2001;44(9):1274–80. [DOI] [PubMed] [Google Scholar]
- 25. Ma P, Yuan Y, Yan P, Chen G, Ma S, Niu X, et al. The efficacy and safety of lateral lymph node dissection for patients with rectal cancer: a systematic review and meta‐analysis. Asian J Surg. 2020;43(9):891–901. [DOI] [PubMed] [Google Scholar]
- 26. Kim TH, Jeong SY, Choi DH, Kim DY, Jung KH, Moon SH, et al. Lateral lymph node metastasis is a major cause of locoregional recurrence in rectal cancer treated with preoperative chemoradiotherapy and curative resection. Ann Surg Oncol. 2008;15(3):729–37. [DOI] [PubMed] [Google Scholar]
- 27. Tamura H, Shimada Y, Kameyama H, Yagi R, Tajima Y, Okamura T, et al. Prophylactic lateral pelvic lymph node dissection in stage IV low rectal cancer. World J Clin Oncol. 2017;8(5):412–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Saito S, Fujita S, Mizusawa J, Kanemitsu Y, Saito N, Kinugasa Y, et al. Male sexual dysfunction after rectal cancer surgery: results of a randomized trial comparing mesorectal excision with and without lateral lymph node dissection for patients with lower rectal cancer: Japan Clinical Oncology Group Study JCOG0212. Eur J Surg Oncol. 2016;42(12):1851–8. [DOI] [PubMed] [Google Scholar]
- 29. Ito M, Kobayashi A, Fujita S, Mizusawa J, Kanemitsu Y, Kinugasa Y, et al. Urinary dysfunction after rectal cancer surgery: results from a randomized trial comparing mesorectal excision with and without lateral lymph node dissection for clinical stage II or III lower rectal cancer (Japan Clinical Oncology Group Study, JCOG0212). Eur J Surg Oncol. 2018;44(4):463–8. [DOI] [PubMed] [Google Scholar]
- 30. Shimoyama M, Yamazaki T, Suda T, Hatakeyama K. Prognostic significance of lateral lymph node micrometastases in lower rectal cancer: an immunohistochemical study with CAM5.2. Dis Colon Rectum. 2003;46(3):333–9. [DOI] [PubMed] [Google Scholar]
- 31. Yang X, Hu T, Gu C, Yang S, Jiang D, Deng X, et al. The prognostic significance of isolated tumor cells detected within lateral lymph nodes in rectal cancer patients after laparoscopic lateral lymph node dissection. J Laparoendosc Adv Surg Tech A. 2019;29(11):1462–8. [DOI] [PubMed] [Google Scholar]
- 32. Kanemitsu Y, Komori K, Shida D, Ochiai H, Tsukamoto S, Kinoshita T, et al. Potential impact of lateral lymph node dissection (LLND) for low rectal cancer on prognoses and local control: a comparison of 2 high‐volume centers in Japan that employ different policies concerning LLND. Surgery. 2017;162(2):303–14. [DOI] [PubMed] [Google Scholar]
- 33. Iversen H, Martling A, Johansson H, Nilsson PJ, Holm T. Pelvic local recurrence from colorectal cancer: surgical challenge with changing preconditions. Colorectal Dis. 2018;20(5):399–406. [DOI] [PubMed] [Google Scholar]
- 34. Konishi T. Management of lateral pelvic lymph node in the East: time to learn from the Western viewpoints. Ann Gastroenterol Surg. 2018;2(5):330–1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Shirouzu K, Ogata Y, Araki Y, Sasatomi T, Nozoe Y, Nakagawa M, et al. Total mesorectal excision, lateral lymphadenectomy and autonomic nerve preservation for lower rectal cancer: significance in the long‐term follow‐up study. Kurume Med J. 2001;48(4):307–19. [DOI] [PubMed] [Google Scholar]
- 36. Sugihara K, Kobayashi H, Kato T, Mori T, Mochizuki H, Kameoka S, et al. Indication and benefit of pelvic sidewall dissection for rectal cancer. Dis Colon Rectum. 2006;49(11):1663–72. [DOI] [PubMed] [Google Scholar]
- 37. Lim SB, Yu CS, Kim CW, Yoon YS, Park SH, Kim TW, et al. Clinical implication of additional selective lateral lymph node excision in patients with locally advanced rectal cancer who underwent preoperative chemoradiotherapy. Int J Colorectal Dis. 2013;28(12):1667–74. [DOI] [PubMed] [Google Scholar]
- 38. Peacock O, Chang GJ. The landmark series: management of lateral lymph nodes in locally advanced rectal cancer. Ann Surg Oncol. 2020;27(8):2723–31. [DOI] [PubMed] [Google Scholar]
- 39. Beets‐Tan RG, Beets GL. Rectal cancer: review with emphasis on MR imaging. Radiology. 2004;232(2):335–46. [DOI] [PubMed] [Google Scholar]
- 40. Heald RJ, Ryall RD. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet (London, England). 1986;1(8496):1479–82. [DOI] [PubMed] [Google Scholar]
- 41. Kim JH, Beets GL, Kim MJ, Kessels AG, Beets‐Tan RG. High‐resolution MR imaging for nodal staging in rectal cancer: are there any criteria in addition to the size? Eur J Radiol. 2004;52(1):78–83. [DOI] [PubMed] [Google Scholar]
- 42. Kim DJ, Chung JJ, Yu JS, Cho ES, Kim JH. Evaluation of lateral pelvic nodes in patients with advanced rectal cancer. AJR Am J Roentgenol. 2014;202(6):1245–55. [DOI] [PubMed] [Google Scholar]
- 43. Kim MJ, Kim TH, Kim DY, Kim SY, Baek JY, Chang HJ, et al. Can chemoradiation allow for omission of lateral pelvic node dissection for locally advanced rectal cancer? J Surg Oncol. 2015;111(4):459–64. [DOI] [PubMed] [Google Scholar]
- 44. Kim TG, Park W, Choi DH, Park HC, Kim SH, Cho YB, et al. Factors associated with lateral pelvic recurrence after curative resection following neoadjuvant chemoradiotherapy in rectal cancer patients. Int J Colorectal Dis. 2014;29(2):193–200. [DOI] [PubMed] [Google Scholar]
- 45. Akiyoshi T, Watanabe T, Miyata S, Kotake K, Muto T, Sugihara K. Results of a Japanese nationwide multi‐institutional study on lateral pelvic lymph node metastasis in low rectal cancer: is it regional or distant disease? Ann Surg. 2012;255(6):1129–34. [DOI] [PubMed] [Google Scholar]
- 46. Obara S, Koyama F, Nakagawa T, Nakamura S, Ueda T, Nishigori N, et al. Laparoscopic lateral pelvic lymph node dissection for lower rectal cancer: initial clinical experiences with prophylactic dissection. Gan To Kagaku Ryoho. 2012;39(12):2173–5. [PubMed] [Google Scholar]
- 47. Weiser MR. AJCC 8th edition: colorectal cancer. Ann Surg Oncol. 2018;25(6):1454–5. [DOI] [PubMed] [Google Scholar]
- 48. Beets‐Tan RGH, Lambregts DMJ, Maas M, Bipat S, Barbaro B, Curvo‐Semedo L, et al. Magnetic resonance imaging for clinical management of rectal cancer: updated recommendations from the 2016 European Society of Gastrointestinal and Abdominal Radiology (ESGAR) consensus meeting. Eur Radiol. 2018;28(4):1465–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Glynne‐Jones R, Wyrwicz L, Tiret E, Brown G, Rödel C, Cervantes A, et al. Rectal cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow‐up. Ann Oncol. 2017;28:iv22–40. [DOI] [PubMed] [Google Scholar]
- 50. Brown G, Rimmer M, Williams S. Colon, rectum and anal cancer. In: Nicholson T, editor. Recommendations for Cross‐sectional Imaging in Cancer Management, 2nd edn. London: The Royal College of Radiologists; 2014. [Google Scholar]
- 51. Brown PJ, Rossington H, Taylor J, Lambregts DMJ, Morris E, West NP, et al. Standardised reports with a template format are superior to free text reports: the case for rectal cancer reporting in clinical practice. Eur Radiol. 2019;29(9):5121–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Burbach JP, Verkooijen HM, Intven M, Kleijnen JP, Bosman ME, Raaymakers BW, et al. RandomizEd controlled trial for pre‐operAtive dose‐escaLation BOOST in locally advanced rectal cancer (RECTAL BOOST study): study protocol for a randomized controlled trial. Trials. 2015;16:58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Chen H, Nguyen KNB, Huang H, Feng C, Zhao X, Daly ME, et al. Effect and safety of radiation therapy boost to extramesorectal lymph nodes in rectal cancer. Pract Radiat Oncol. 2020;10(5):e372–e7. [DOI] [PubMed] [Google Scholar]
- 54. Hartvigson PE, Apisarnthanarax S, Schaub S, Cohen S, Bernier G, Koh WJ, et al. Radiation therapy dose escalation to clinically involved pelvic sidewall lymph nodes in locally advanced rectal cancer. Adv Radiat Oncol. 2019;4(3):478–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Kim YI, Jang JK, Park IJ, Park SH, Kim JB, Park JH, et al. Lateral lymph node and its association with distant recurrence in rectal cancer: a clue of systemic disease. Surg Oncol. 2020;35:174–81. [DOI] [PubMed] [Google Scholar]
- 56. Wei M, Wu Q, Fan C, Li Y, Chen X, Zhou Z, et al. Lateral pelvic lymph node dissection after neoadjuvant chemo‐radiation for preoperative enlarged lateral nodes in advanced low rectal cancer: study protocol for a randomized controlled trial. Trials. 2016;17(1):561. [DOI] [PMC free article] [PubMed] [Google Scholar]


