Abstract
Transjugular intrahepatic portosystemic shun (TIPS) is an effective procedure for the treatment of portal hypertension complications such as esophagogastric variceal bleeding and refractory ascites. To date, its use by many experts and scholars at home and abroad has benefitted countless patients. In order to further promote the development and innovation of TIPS in China, this paper briefly reviews the development process of TIPS at home and abroad, objectively analyzes the bottlenecks and breakthrough achievements in the development of TIPS, and discusses the application status and research progress of TIPS technology for different indications. Finally, we provide an outlook about the development and application of TIPS in China.
Keywords: TIPS, Application situation, Research progress, Prospect
1. Development and progress of transjugular intrahepatic portosystemic shunt
Similar to radiography and percutaneous transluminal angioplasty, transjugular intrahepatic portosystemic shunt (TIPS) also originated from an “accidental technical operation.” In 1967, to reduce the incidence of biliary peritonitis caused by biliary puncture, Hanafee et al. attempted to perform cholangiography by puncturing the biliary tract through the jugular vein, during which the puncture needle accidently entered the portal vein, leading to visualization of the portal vein. This experiment aroused the attention of relevant medical experts about the transjugular portal vein pathway, and subsequently laid the foundation for the creation, application, and development of TIPS.1 In 1969, Rösch et al. first reported the experimental results of TIPS. In their study, the jugular vein of a dog was punctured, and a tube was placed after dilating the channel between the hepatic vein and portal vein. At the same time, a tube was placed in the splenic vein for angiographic follow-up. Four days later, angiography showed that the shunt was unobstructed.2 On the basis of these encouraging experimental results of TIPS, Colapinto et al., in 1982 successfully performed the first case of placement of a transjugular portosystemic shunt in the human body, in which only a balloon was used to dilate the shunt channel.3 Owing to the technical limitations at that time, a stent was not implanted in the shunt of the patient. With advances in medicine, the first human TIPS surgery with metal stent (Palmaz stent) implantation was reported in 1988. The patient had severe end-stage portal hypertension and Child–Pugh class C liver function. Through the jugular vein approach, a modified Brockenbrough needle was used to puncture the right branch of the portal vein through the right hepatic vein. Two Palmaz stents were implanted after the successful puncture and balloon dilation of the shunt channel to keep the puncture passage unobstructed. The pressure gradient of the portal system decreased from 38 to 18 mmHg. The patient’s clinical condition greatly improved during the next few days. However, the patient died 11 days after the surgery owing to a sudden attack of acute respiratory distress caused by acute nosocomial fungal and cytomegalovirus infection. The autopsy results showed that the shunt was completely unobstructed without superficial thrombosis. A thin layer of endothelial cells was found on the surface of the shunt.4 This milestone event placed TIPS, a minimally invasive intervention for portal hypertension, at the center stage of clinical medicine. With the application of bare metal stents, the treatment of portal hypertension has ushered in a vigorous upsurge in the use of TIPS. From the early 1990s to the mid-1990s, TIPS with bare metal stent implantation was widely used in clinical practice. In 1992, Laberge et al. reported the preliminary clinical results of 25 patients who underwent TIPS. The study showed that TIPS is a safe and effective treatment for patients with variceal bleeding, especially in those with severe liver disease waiting for transplantation.5 At that time, the self-expanding bare stent used was mainly Wallstent (Boston Scientific Corporation, USA).
With the international development of TIPS in full swing, the application and development of this treatment method also started in China. In June 1992, under the guidance of Professor Takahashi Yuanichiro of Hamamatsu Medical University, the team of Professor Ke Xu completed the first TIPS surgery in China, which took 12.5 h to complete (Fig. 1). In the same year, the results of the clinical application of TIPS in 15 cases were reported at the China-Japan Medical Conference. In 1993, the first article about the clinical application of TIPS was published in the Chinese Journal of Radiology,6 and the first clinical training class on TIPS was held at the same time. In April 1994, the team of Professor Zhang Jinshan of the People’s Liberation Army General Hospital held the first International Symposium on TIPS in Beijing. Thus, the clinical application of TIPS has opened a new era in its vigorous development in China. The promotion and application of TIPS have further stimulated international exchange and cooperation, as well as facilitated the development and innovation of TIPS technology in China.
Fig. 1.
The team of Professor Ke Xu completed the first TIPS operation of China.
However, shunt stenosis and occlusion are the main factors affecting the long-term efficacy of TIPS. The restenosis or shunt occlusion rates were reported to be as high as 50%–64%, 33%–70%, and 60%–85% at 1, 2, and 5 years after TIPS, respectively. The incidence of hepatic encephalopathy was demonstrated to be as high as 30%, which is much higher than that with endoscopic therapy (15%).7 Some authors have suggested that TIPS is only suitable as a bridge therapy before liver transplantation. Concerns about the poor long-term efficacy and the high incidence rate of postoperative complications hinder the wide clinical application of TIPS.
2. Breakthrough and progress in TIPS application research
To reduce the incidence of restenosis or occlusion of the shunt after TIPS, and to overcome the poor long-term curative effect, interventional radiologists have identified the causes and mechanisms of stenosis/occlusion of the TIPS shunt, and offered some solutions. One of the solutions was the development of a new type of TIPS shunt stent, the polytetrafluoroethylene (PTFE)-covered stent, which replaced the traditional bare stent. In the late 1990s, this new PTFE-covered stent showed encouraging results in preclinical trials. The stent can greatly improve the patency of the TIPS shunt.8,9 At the beginning of the 21st century, the PTFE-covered stent started being used in clinical practice. Compared with the bare metal stent, the PTFE-covered stent significantly improved the shunt patency rate and the survival rate without increasing the risk of hepatic encephalopathy.10 In 2004, a new type of modified covered stent, called the Viatorr stent (Gore Corporation, USA), was launched in the United States. Subsequently, this new type of PTFE-covered stent (TIPS special stent) started to become widely used in clinical practice, which brought about a second resurgence of TIPS and opened a new era in TIPS development.11 In 2005, the American Association for the Study of Liver Diseases (AASLD) published TIPS practice guidelines, which affirmed the role of TIPS in the management of portal hypertension.12 In 2009, the AASLD recommended the Budd–Chiari syndrome as another indication for TIPS, and placement of a PTFE-covered stent was set as the operation standard13 (Table 1).
Table 1.
Milestone events in the development history of international TIPS.
| Time | Milestone events |
|---|---|
| 1960s | Unexpected discovery of portal vein by transjugular cholangiography |
| 1969 | Rosch first reported the experimental study of portal vein shunt |
| 1982 | Colapinto et al. completed the first human balloon dilated TIPS |
| 1988 | The first case of human bare metal stent TIPS |
| Early 1990s | Self-expanding bare stent is widely used in TIPS |
| Late 1990s | Carry out the animal experiment of covered stent TIPS |
| 2001 | The treatment standard is portal pressure reduced to 12 mmHg |
| 2003–2004 | Covered stent is widely used in TIPS |
| 2005,2009 | AASLD formulated TIPS clinical application guidelines |
| Now | The clinical application of TIPS is fruitful |
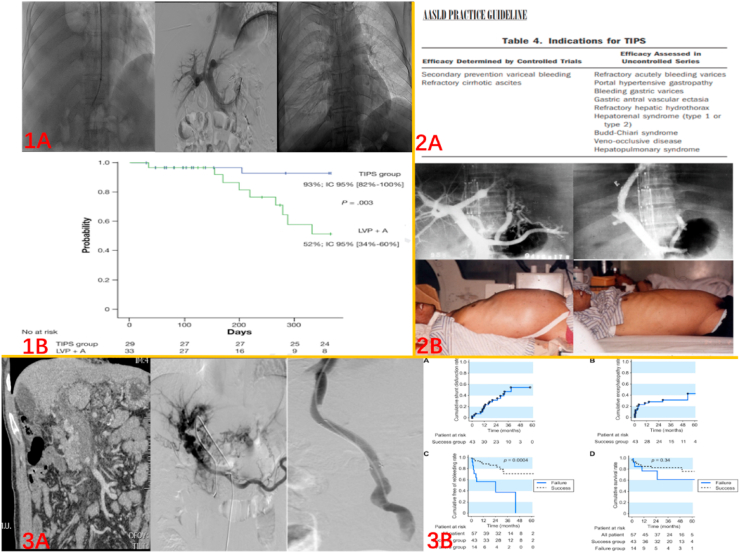
With the maturity of TIPS technology, its curative effect has been more widely recognized and its indications have considerably expanded. In 2010, a randomized controlled trial published in the New England Journal of Medicine showed the safety and efficacy of TIPS for the treatment of acute variceal bleeding. In that study, 63 patients with acute variceal bleeding due to cirrhosis were randomly divided into an early TIPS group and an endoscopic ligation group (standard treatment). After 16 months of follow-up, early TIPS treatment was found to significantly reduce the mortality rate of patients with acute variceal bleeding.14 An increasing number of studies have shown that TIPS can prevent and control gastrointestinal bleeding caused by portal hypertension more effectively than endoscopic therapy, without increasing the incidence of hepatic encephalopathy.15, 16, 17 This is particularly encouraging for making breakthroughs in expanding the scope of TIPS indications. Furthermore, Han et al. found that early TIPS improved survival in patients with severe cirrhosis with a Model for End-stage Liver Disease (MELD) score of ≥19 or Child–Pugh class C liver function. However, for patients with MELD score 12–18 or Child–Pugh class B liver function, further evaluations are necessary to determine optimal selection criteria for early TIPS.8 In addition, Dominique et al. demonstrated that severe cirrhosis limited the effect of early TIPS in patients with variceal bleeding.19 These results indicate that more multicenter clinical studies are needed to identify the optimal candidates for early TIPS.18 For the treatment of esophageal and gastric variceal rebleeding, the China Expert Consensus on TIPS proposed in 2017 that TIPS or external surgery can be the first-choice treatment after the failure of endoscopic hemostasis and drug treatment; however, for patients with poor liver function, TIPS is preferred.20 Intractable peritoneal effusion and hepatic pleural effusion are the most prominent clinical manifestations in the late stage of portal hypertension, which not only seriously reduce the quality of life but also portend a dismal 1-year survival rate of only 50%.20 The results of a randomized controlled trial by Bureau et al. showed that TIPS with a covered stent could significantly improve the survival time of patients with refractory ascites without increasing the risk of hepatic encephalopathy.21 Moreover, compared with abdominal puncture drainage, TIPS can significantly increase the transplant-free survival rate.22 In addition, TIPS is the only treatment for hepatic pleural effusion and refractory ascites caused by portal hypertension. The Budd–Chiari syndrome is a group of diseases caused by obstruction of the hepatic vein outflow tract, which can occur at any level from the hepatic vein to the inferior vena cava and right atrium. Most patients with Budd–Chiari syndrome are suitable for interventional treatment. However, TIPS is recommended in patients with the following conditions20,23: (1) extensive hepatic vein occlusion, (2) irreversible liver cirrhosis with portal hypertension, and (3) severe hepatic congestion with progressive liver function damage. The safety and effectiveness of TIPS in patients with Budd–Chiari syndrome have been confirmed. The 5-year survival rate can reach 90%, and the 10-year survival rate can reach 80% (Fig. 2).24, 25, 26 The use of TIPS for the treatment of portal vein thrombosis/cavernous transformation of the portal vein has been the most important technical breakthrough in the field of TIPS treatment in recent years. The incidence of portal vein thrombosis was reported to be 29%.27 In the past, portal hypertension syndrome with portal vein thrombosis/cavernous transformation was considered a relative contraindication to TIPS.28 However, with continuous progress in image-guided technology and the gradual accumulation of operative experience, this concept has now changed. A number of studies have shown that TIPS is effective for the treatment of cirrhotic portal vein thrombosis/cavernous transformation with portal hypertension complications. The success rate of surgery was reported to be 75%–100%, and the open rate of portal vein thrombosis was reported as 70%–100%.29,30 Although TIPS has made a breakthrough in its application to the treatment of portal vein thrombosis/cavernous transformation of the portal vein, some researchers believe that this operative technique has certain difficulties and risks, and thereby should be performed by medical centers with more experience in the procedure.31
Fig. 2.
1A: one case of significantly reducing hepatic pleural effusion after TIPS; 1B: early TIPS prolongs survival time in patients with recurrent ascites. Reproduce from Ref. [19]. 2A: Budd-Chiari is considered as an indication of TIPS in AASLD. Reproduce from Ref. [13]; 2B: One Budd-Chiari patient with intractable ascites, and ascites decreased significantly after TIPS. 3A: One patient with portal vein thrombosis/cavernous transformation still have gastrointestinal bleeding after splenectomy and endoscopic ligation. After portal vein thrombolysis and TIPS treatment, gastrointestinal bleeding disappeared; 3B: Relevant research shows that TIPS should be considered a safe and feasible alternative therapy for patients with chronic portal vein thrombosis. Reproduce from Ref. [28].
With the expansion of indications, the technology of TIPS is also constantly evolving. In a randomized controlled trial, Chen et al. found that patients undergoing TIPS through the left portal vein had a lower incidence of hepatic encephalopathy. Luo et al. conducted a retrospective analysis of 1244 patients undergoing TIPS, and divided the patients into group A (targeting the left branch of the portal vein) and group B (targeting the right branch of the portal vein). The results showed that TIPS involving puncture of the left branch of the intrahepatic portal vein may reduce the risk of hepatic encephalopathy.32 A meta-analysis study revealed that TIPS performed by puncturing the left portal vein is associated with decreased rates of postoperative hepatic encephalopathy.33 Performing TIPS through the left portal vein could decrease the incidence of hepatic encephalopathy, which may be attributed to differences in blood ammonia levels between the right and left branches of the portal vein. Chu offered a theoretical basis for this claim by reporting that the ammonia content in the right branch of the portal vein is higher than that in the left branch.34 The experience of our center showed that regardless of whether the left or right branch of the portal vein is punctured, re-angiography after the standard TIPS procedure (with implantation of a 8-mm Viatorr stent) can only show that the portal vein flows with high pressure directly through the shunt channel from the portal vein, and it was difficult to distinguish the left and right branches of the portal vein. Therefore, further clinical randomized controlled studies are still highly warranted to determine whether performing TIPS through the left portal vein improves survival and reduces complications. Measurement of the hepatic venous pressure gradient is one of the gold-standard methods for assessing clinically relevant portal hypertension (hepatic venous pressure gradient ≥ 10 mmHg), which is an independent predictor of variceal rebleeding in patients with cirrhosis.35 Fan et al. suggested that hepatic venous pressure may help further identify patients who should undergo early TIPS, or those who should be maintained on pharmacological treatment and/or should undergo endoscopic hemostasis.36 In addition, Alberto et al. found that hepatic venous pressure is a determinant factor that influences the survival time of patients undergoing early TIPS.37 However, the invasiveness of traditional methods of hepatic venous pressure measurement has restricted their widespread use in clinical practice. Fortunately, the development of radiomics provides a new way to solve this problem.38 Qi et al. demonstrated that radiomics-based hepatic venous pressure gradient, a noninvasive radiomics model, could facilitate the rapid identification of portal hypertension.39 In addition, more attention should be paid to monitoring changes in hepatic venous pressure gradient in response to medical therapy, using a noninvasive method.
To date, the developmental history of TIPS has not been a smooth journey, but rather a path of overcoming many difficulties. TIPS creates a safe and effective outflow tract for “desperate” portal vein blood flow, and effectively relieves or eliminates the “barrier lake” of portal hypertension. TIPS, with the advantages of a producing good curative effect, causing small injury, and having wide adaptability, has become an irreplaceable and effective method for the clinical treatment of various complications of portal hypertension due to end-stage liver disease.
3. Confusion about and prospects of TIPS application in China
The developmental history of TIPS, from being an unexpected discovery to being recognized as an effective, minimally invasive, life-saving treatment method, consisted of both difficulties and excellent results. Nevertheless, the development of TIPS has also brought considerable confusion about the application of this interventional technique in China.
First, since 1992, China has expanded the application field of TIPS technology. However, it is not difficult to see that China’s contribution to the many milestone events in international TIPS development is not evident. In 2017, Han et al. conducted a randomized controlled study comparing the use of 10- and 8-mm covered stents, and reported that the use of an 8-mm covered stent for TIPS reduced the incidence of spontaneous hepatic encephalopathy and improved liver function in patients with variceal bleeding caused by portal hypertension.40 This finding was cited by the “European Association for the Study of the Liver clinical practice guidelines for the management of patients with decompensated cirrhosis (2018),” and it was the first time worldwide that a relatively small-diameter stent (8 mm) was selected for TIPS treatment. This also represents the developmental milestone labeled as “TIPS made in China.”
Second, in recent years, the interventional devices used in TIPS have rapidly developed, leading to not only shorter operation time but also improved operation safety and postoperative efficacy. However, the cost of TIPS treatment is increasing every year. Imported consumables are one of the main reasons for the high treatment cost of TIPS. The nearly 10 kinds of different brands of equipment required for TIPS surgery are mostly imported products (Table 2). Domestic brands are still not available on the market, especially the core devices such as covered stents and portal vein puncture systems. To date, it is particularly worrisome that few national enterprises are committed to the research and development of TIPS-related devices. Therefore, we hope that, while welcoming the importation of equipment, state-owned products should also be independently and vigorously developed. Practitioners must overcome difficulties and master the core technology. As the saying goes, “own for yourself better than your parents did.” China should break through the technical barriers and gain the core technology of TIPS. Moreover, with the continuous expansion of interventional radiology teams and increasing number of talented professionals, young and middle-aged doctors can skillfully perform TIPS treatment, leading to the publication of many high-quality articles. Although most doctors can perform operations well, only a few can conduct research and make innovations. Most of them can publish papers, but only a few can conduct multicenter randomized controlled studies. These are the current problems that need to be overcome in the current TIPS field. In 2019, Han et al. conducted the first randomized controlled trial on early TIPS, with survival as the main observation end point.41 The findings from their study expanded the candidate population for early TIPS and indicated that early TIPS can improve the survival rate of patients.
Table 2.
List of imported TIPS instruments (Example).
| Operating Instrument | Brand |
|---|---|
| Viattor stent | GORE |
| RUPS-100 | COOK |
| InterLock | Boston Scienctific |
| High pressure balloon catheter | Boston Scienctific |
| Angiographic catheter | COOK |
| Merit analog inflation device | MERIT |
| Arterial sheaths | TERMO |
| Guide wire | TERMO |
| Guide wire (stiff type) | TERMO |
Finally, in recent years, an increasing number of clinical guidelines have been published to standardize and update the technical operations and application concepts of TIPS. The AASLD in the United States has released two editions, whereas the Baveno in Europe is updated once every 5 years. The majority of TIPS experts and scholars in China refer to these guidelines, but their contributions to clinical evidence are relatively few. In addition, some clinical guidelines have been published in related fields in China, such as the “Clinical technical guidelines for TIPS”,42 “Consensus opinions on the treatment of cirrhotic portal hypertension with TIPS”,43 and “Expert consensus on TIPS”.20 However, these “guidelines” are basically the results of different professional academic organizations from different viewpoints and at different stages. They are widely recognized and inherited, but rarely revised and reprinted. Moreover, there are relatively few treatment centers, which limits the ability to gain experience and improve skill according to the guidelines.
Therefore, the domestic situation needs to be assessed, and it should be recognized that the development of TIPS in China still has a long way to go. Preparations should be made in times when no problems exist, opportunities should be seized when possible, and the challenge of supply interruption caused by various factors should be met. Steady progress in clinical technology should be made, while also paying attention to scientific research, improving research awareness, converting clinical problems to scientific issues, and making breakthroughs one at a time (from small to large ones). In addition, a sense of transformation and cooperation with scientific research institutions or related enterprises are also needed to accomplish scientific and technological research activities. With the cooperation and joint efforts of industry, universities, research institutions, and medical facilities, the development of TIPS in China will further improve.
Declaration of competing interest
The authors declare that they have no conflicts of interests to this work. We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Acknowledgments
The authors acknowledge financial support from National Natural Science Foundation of China (Nos. 81630053, . 81873918).
Contributor Information
Peng Wang, Email: 18865383352@163.com.
Ke Xu, Email: kxu@cmu.edu.cn.
References
- 1.Hanafee W., Weiner M. Transjugular percutaneous cholangiography. Radiology. 1967;88:35–39. doi: 10.1148/88.1.35. [DOI] [PubMed] [Google Scholar]
- 2.Rösch J., Hanafee W.N., Snow H. Transjugular portal venography and radiologic portacaval shunt: an experimental study. Radiology. 1969;92:1112–1114. doi: 10.1148/92.5.1112. [DOI] [PubMed] [Google Scholar]
- 3.Colapinto R.F., Stronell R.D., Gildiner M. Formation of intrahepatic portosystemic shunts using a balloon dilatation catheter: preliminary clinical experience. AJR Am J Roentgenol. 1983;140:709–714. doi: 10.2214/ajr.140.4.709. [DOI] [PubMed] [Google Scholar]
- 4.Richter G.M., Palmaz J.C., Nöldge G. vol. 29. Radiologe; German: 1989. pp. 406–411. (Der Transjuguläre Intrahepatische Portosystemische Stent-Shunt (TIPSS). Eine Neue Nichtoperative, Perkutane Methode [The Transjugular Intrahepatic Portosystemic Stent-Shunt. A New Nonsurgical Percutaneous Method]). [PubMed] [Google Scholar]
- 5.LaBerge J.M., Ring E.J., Lake J.R. Transjugular intrahepatic portosystemic shunts: preliminary results in 25 patients. J Vasc Surg. 1992;16:258–267. doi: 10.1067/mva.1992.37161. [DOI] [PubMed] [Google Scholar]
- 6.Ke X., Hanguo Z., Fangxian H. Transjugular intrahepatic portal-caval stent shunt in the treatment of cirrhotic portal hypertension (analysis of 8 cases) Chin J Radiol. 1993;27:294–297. [Google Scholar]
- 7.Rössle M., Siegerstetter V., Huber M. The first decade of the transjugular intrahepatic portosystemic shunt (TIPS): state of the art. Liver. 1998;18:73–89. doi: 10.1111/j.1600-0676.1998.tb00132.x. [DOI] [PubMed] [Google Scholar]
- 8.Haskal Z.J., Davis A., McAllister A. PTFE-encapsulated endovascular stent-graft for transjugular intrahepatic portosystemic shunts: experimental evaluation. Radiology. 1997;205:682–688. doi: 10.1148/radiology.205.3.9393521. [DOI] [PubMed] [Google Scholar]
- 9.Nishimine K., Saxon R.R., Kichikawa K. Improved transjugular intrahepatic portosystemic shunt patency with PTFE-covered stent-grafts: experimental results in swine. Radiology. 1995;196:341–347. doi: 10.1148/radiology.196.2.7617843. [DOI] [PubMed] [Google Scholar]
- 10.Yang Z., Han G., Wu Q. Patency and clinical outcomes of transjugular intrahepatic portosystemic shunt with polytetrafluoroethylene-covered stents versus bare stents: a meta-analysis. J Gastroenterol Hepatol. 2010;25:1718–1725. doi: 10.1111/j.1440-1746.2010.06400.x. [DOI] [PubMed] [Google Scholar]
- 11.Bureau C., Garcia-Pagan J.C., Otal P. Improved clinical outcome using polytetrafluoroethylene-coated stents for TIPS: results of a randomized study. Gastroenterology. 2004;126:469–475. doi: 10.1053/j.gastro.2003.11.016. [DOI] [PubMed] [Google Scholar]
- 12.Boyer T.D., Haskal Z.J. The role of transjugular intrahepatic portosystemic shunt in the management of portal hypertension. Hepatology. 2005;41:386–400. doi: 10.1002/hep.20559. [DOI] [PubMed] [Google Scholar]
- 13.Runyon B.A. Management of adult patients with ascites due to cirrhosis: an update. Hepatology. 2009;49:2087–2107. doi: 10.1002/hep.22853. [DOI] [PubMed] [Google Scholar]
- 14.García-Pagán J.C., Caca K., Bureau C. Early use of TIPS in patients with cirrhosis and variceal bleeding[J] N Engl J Med. 2010;362(25):2370–2379. doi: 10.1056/NEJMoa0910102. [DOI] [PubMed] [Google Scholar]
- 15.de Franchis R. Revising consensus in portal hypertension: report of the Baveno V consensus workshop on methodology of diagnosis and therapy in portal hypertension. J Hepatol. 2010;53:762–768. doi: 10.1016/j.jhep.2010.06.004. [DOI] [PubMed] [Google Scholar]
- 16.Halabi S.A., Sawas T., Sadat B. Early TIPS versus endoscopic therapy for secondary prophylaxis after management of acute esophageal variceal bleeding in cirrhotic patients: a meta-analysis of randomized controlled trials. J Gastroenterol Hepatol. 2016;31:1519–1526. doi: 10.1111/jgh.13303. [DOI] [PubMed] [Google Scholar]
- 17.Bai M., Qi X.S., Yang Z.P. EVS vs TIPS shunt for gastric variceal bleeding in patients with cirrhosis: a meta-analysis. World J Gastrointest Pharmacol Therapeut. 2014;5:97–104. doi: 10.4292/wjgpt.v5.i2.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lv Y., Zuo L., Zhu X. Identifying optimal candidates for early TIPS among patients with cirrhosis and acute variceal bleeding: a multicentre observational study. Gut. 2019;68:1297–1310. doi: 10.1136/gutjnl-2018-317057. [DOI] [PubMed] [Google Scholar]
- 19.Thabut D., Pauwels A., Carbonell N. Cirrhotic patients with portal hypertension-related bleeding and an indication for early-TIPS: a large multicentre audit with real-life results. J Hepatol. 2017;68:73–81. doi: 10.1016/j.jhep.2017.09.002. [DOI] [PubMed] [Google Scholar]
- 20.Interventional group rb, Chinese Medical Association Expert consensus on transjugular intrahepatic portosystemic shunt. Chin J Radiol. 2017;51:324–333. [Google Scholar]
- 21.Bureau C., Thabut D., Oberti F. Transjugular intrahepatic portosystemic shunts with covered stents increase transplant-free survival of patients with cirrhosis and recurrent ascites. Gastroenterology. 2017;152:157–163. doi: 10.1053/j.gastro.2016.09.016. [DOI] [PubMed] [Google Scholar]
- 22.Salerno F., Cammà C., Enea M. Transjugular intrahepatic portosystemic shunt for refractory ascites: a meta-analysis of individual patient data. Gastroenterology. 2007;133:825–834. doi: 10.1053/j.gastro.2007.06.020. [DOI] [PubMed] [Google Scholar]
- 23.Association IpboCM Chinese clinical practice guidelines for transjugular intrahepatic portosystemic shunt in portal hypertension (2019 Edition) Journal of clinical hepatobiliary diseases. 2019;35:2694–2699. [Google Scholar]
- 24.Valla D.C. Budd-Chiari syndrome and veno-occlusive disease/sinusoidal obstruction syndrome. Gut. 2008;57:1469–1478. doi: 10.1136/gut.2007.133637. [DOI] [PubMed] [Google Scholar]
- 25.Garcia-Pagán J.C., Heydtmann M., Raffa S. TIPS for Budd-Chiari syndrome: long-term results and prognostics factors in 124 patients. Gastroenterology. 2008;135:808–815. doi: 10.1053/j.gastro.2008.05.051. [DOI] [PubMed] [Google Scholar]
- 26.Qi X., Guo W., He C. Transjugular intrahepatic portosystemic shunt for Budd-Chiari syndrome: techniques, indications and results on 51 Chinese patients from a single centre. Liver Int. 2014;34:1164–1175. doi: 10.1111/liv.12355. [DOI] [PubMed] [Google Scholar]
- 27.Qi X., Han G., Yin Z. Transjugular intrahepatic portosystemic shunt for portal cavernoma with symptomatic portal hypertension in non-cirrhotic patients. Dig Dis Sci. 2012;57:1072–1082. doi: 10.1007/s10620-011-1975-5. [DOI] [PubMed] [Google Scholar]
- 28.Boyer T.D., Haskal Z.J. The role of transjugular intrahepatic portosystemic shunt (TIPS) in the management of portal hypertension: update 2009. Hepatology. 2010;51:306. doi: 10.1002/hep.23383. [DOI] [PubMed] [Google Scholar]
- 29.Salem R., Vouche M., Baker T. Pretransplant portal vein recanalization-transjugular intrahepatic portosystemic shunt in patients with complete obliterative portal vein thrombosis. Transplantation. 2015;99:2347–2355. doi: 10.1097/TP.0000000000000729. [DOI] [PubMed] [Google Scholar]
- 30.Han G., Qi X., He C. Transjugular intrahepatic portosystemic shunt for portal vein thrombosis with symptomatic portal hypertension in liver cirrhosis. J Hepatol. 2011;54:78–88. doi: 10.1016/j.jhep.2010.06.029. [DOI] [PubMed] [Google Scholar]
- 31.Krajina A., Hulek P., Fejfar T. Quality improvement guidelines for transjugular intrahepatic portosystemic shunt (TIPS) Cardiovasc Intervent Radiol. 2012;35:1295–1300. doi: 10.1007/s00270-012-0493-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Luo S.H., Chu J.G., Huang H. Targeted puncture of left branch of intrahepatic portal vein in transjugular intrahepatic portosystemic shunt to reduce hepatic encephalopathy. World J Gastroenterol. 2019;25:1088–1099. doi: 10.3748/wjg.v25.i9.1088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zuo K., Wang C., Wang J. Transjugular intrahepatic portosystemic shunt through left branch versus right branch of portal vein: a meta-analysis. Abdom Radiol (NY) 2021;46:1718–1725. doi: 10.1007/s00261-020-02789-9. [DOI] [PubMed] [Google Scholar]
- 34.Chu J., Sun X., Piao L. [Portosystemic shunt via the left branch of portal vein for the prevention of encephalopathy following transjugular intrahepatic portosystemic shunt] Zhonghua Gan Zang Bing Za Zhi. 2002;10:437–440. [PubMed] [Google Scholar]
- 35.Li L., Liu C., Qi X. Effects of early placement of transjugular portosystemic shunts in patients with high-risk acute variceal bleeding: a meta-analysis of individual patient data. Gastroenterology. 2021 Jan 12;S0016–5085(21) doi: 10.1053/j.gastro.2021.01.006. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 36.Qi X.S., Fan D.M. Hepatic venous pressure gradient measurement before TIPS for acute variceal bleeding. World J Gastroenterol. 2014;20:7523–7524. doi: 10.3748/wjg.v20.i23.7523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Monescillo A., Martínez-Lagares F., Ruiz-del-Arbol L. Influence of portal hypertension and its early decompression by TIPS placement on the outcome of variceal bleeding. Hepatology. 2004;40:793–801. doi: 10.1002/hep.20386. [DOI] [PubMed] [Google Scholar]
- 38.Qi X., Berzigotti A., Cardenas A. Emerging non-invasive approaches for diagnosis and monitoring of portal hypertension. Lancet Gastroenterol Hepatol. 2018;3:708–719. doi: 10.1016/S2468-1253(18)30232-2. [DOI] [PubMed] [Google Scholar]
- 39.Liu F., Ning Z., Liu Y. Development and validation of a radiomics signature for clinically significant portal hypertension in cirrhosis (CHESS1701): a prospective multicenter study. EBioMedicine. 2018;36:151–158. doi: 10.1016/j.ebiom.2018.09.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang Q., Lv Y., Bai M. Eight millimetre covered TIPS does not compromise shunt function but reduces hepatic encephalopathy in preventing variceal rebleeding. J Hepatol. 2017;67:508–516. doi: 10.1016/j.jhep.2017.05.006. [DOI] [PubMed] [Google Scholar]
- 41.Lv Y., Yang Z., Liu L. Early TIPS with covered stents versus standard treatment for acute variceal bleeding in patients with advanced cirrhosis: a randomised controlled trial. Lancet Gastroenterol Hepatol. 2019;4:587–598. doi: 10.1016/S2468-1253(19)30090-1. [DOI] [PubMed] [Google Scholar]
- 42.Radiology IgoeboCJo Clinical technical guidelines for transjugular intrahepatic portosystemic shunt. Chin J Radiol. 2004;38:1329–1332. [Google Scholar]
- 43.Digestive intervention group dsoCMA Consensus opinion on transjugular intrahepatic portosystemic shunt for cirrhotic portal hypertension. Chinese Journal of Digestion. 2014;34:3–6. [Google Scholar]