Abstract
Despite the rapid development of diagnostic and therapeutic modalities and techniques to manage LGIB patients from interventional radiology’s standpoint, a successful localization of the bleeding site that leads to an effective embolotherapy remains a significant technical challenge. The interventional radiologist’s decisions when managing patients with LGIB may significantly impact the clinical outcomes; therefore, management should be made based on careful and thorough considerations of factors such as etiology, locations, patient’s comorbidities, and potential post-procedure complications, among others. The purpose of this paper is to review the management of LGIB by interventional radiology, focusing on a few challenging and common clinical situations that require special consideration by interventional radiologists.
Keywords: Lower gastrointestinal bleeding, Angiography, Embolization, Anticoagulation related lower gastrointestinal bleeding
1. Introduction
Lower gastrointestinal bleeding (LGIB) refers to bleeding originating from the gastrointestinal tract distal to the ligament of Treitz, thus including the small intestine, colon, rectum or anus. LGIB accounts for approximately 20%–25% of all gastrointestinal hemorrhages, with a reported annual incidence of 21–27 per 100,000 populations, and a mortality rate of 2–4% in North America.1, 2, 3 Currently, conventional treatment options for LGIB include endoscopic therapy, transcatheter arterial embolization, and surgery. Colonoscopy is considered the best management tool for patients with chronic and intermittent LGIB but is less reliable when treating patients with severe, life-threatening hematochezia due to inadequate visualization caused by inadequate bowel preparation or active bleeding. Indications for emergent surgery include life-threatening bleeding in patients who fail endoscopy and embolization treatments, generally as a last resort treatment option given the much higher rates of morbidity and mortality.4,5
With the development of coaxial microcatheters, super-selective embolization has emerged as a viable treatment option for LGIB and has demonstrated many advantages compared to endoscopic or surgical treatments.6 By using appropriate embolic agents, hemostasis in the lower gastrointestinal tract can be achieved rapidly and safely by decreasing arterial perfusion pressure to the bleeding site without causing complete devascularization.6 Thus, it has been widely used to treat acute and life-threatening LGIB and has become the treatment of choice in many cases.
Despite the rapid development of diagnostic and therapeutic modalities and techniques to manage LGIB patients from interventional radiology’s standpoint, a successful localization of the bleeding site that leads to an effective embolotherapy remains a significant technical challenge. The interventional radiologist’s decisions when managing patient’s with LGIB may significantly impact the clinical outcomes, therefore, management should be made based on careful and thorough considerations of factors such as etiology, locations, patient’s comorbidities, and potential post-procedure complications, among others. The purpose of this paper is to review the management of LGIB by interventional radiology, focusing on a few challenging and common clinical situations that require special considerations by interventional radiologists.
2. Angiographically occult LGIB and prophylactic embolotherapy
One of the most significant clinical challenges for interventional radiologists to manage LGIB is angiographically occult bleeding. In past decades there has been a significant evolution in the diagnostic and treatment modalities used to manage these patients by gastroenterologists, radiologists, and surgeons. However, the primary challenge of LGIB management remains the identification of the source rather than the treatment of bleeding.7 Accurate detection of LGIB by angiography requires a reported bleeding rate of at least 1 ml/min8, thus a negative angiography is common in patients with LGIB given the intermittent nature of bleeding. The overall yield of angiography for the detection of LGIB source ranges widely with success rates reported from 40%-78%.9,10 For bleeding patients without an identifiable angiographic source, a super-selective embolization is generally performed, and treatment options become very limited and challenging. If the patient remains hemodynamically stable, observation with supportive treatment is usually selected, despite a high rebleeding rate in untreated LGIB, especially when bleeding from diverticular disease or angiodysplasia.11 Once the bleeding recurs, the mortality rate may approach 30%.6 If the patient becomes hemodynamically unstable, emergency surgery may be indicated, with mortality rates as high as 33–57%.4,5
Provocative angiography may improve the diagnostic yield for LGIB. Several studies have reported that following a nonlocalized angiography, bleeding can be induced by using systemic heparinization and selective injection of a vasodilator with a thrombolytic.12, 13, 14 However, this approach remains controversial due to concerns of inducing uncontrolled bleeding and is usually reserved for LGIB patients with repeatedly negative angiographies.15 In upper gastrointestinal bleeding (UGIB), prophylactic embolization has proven to be effective.16,17 For example, Lang et al.16 reported successful treatment of massive UGIB by prophylactic embolization of the left gastric artery. Theoretically, prophylactic embolization could be a feasible alternative to treat angiographically occult LGIB as well, if source localization by pre-angiographic imaging studies such as nuclear scintigraphy or CT angiogram is sufficiently accurate. Few studies have been conducted to evaluate the efficacy of prophylactic embolization in LGIB to date. There are multiple factors for this paucity of data. LGIB is only one-fifth to one-third as common as UGIB. It generally has a more indolent clinical presentation and spontaneously stops in 80%–85% cases.2 Second, the left gastric artery is reported to be responsible for approximately 85% of the UGIB cases.18 The extensive collateral blood supply to the upper GI tract also allows relatively safe embolization of suspected vessels with low risk of end-organ ischemia. The bowel distal to the ligament of Treitz (except for the rectum) does not have a dual blood supply.19 The left gastric artery may be empirically embolized in the correct clinical setting, and there is a high likelihood of treating the bleeding source.20 Conversely, in angiographically occult bleeding within the lower GI tract, the end organ arterial distribution renders prophylactic embolization hazardous by producing bowel ischemia.
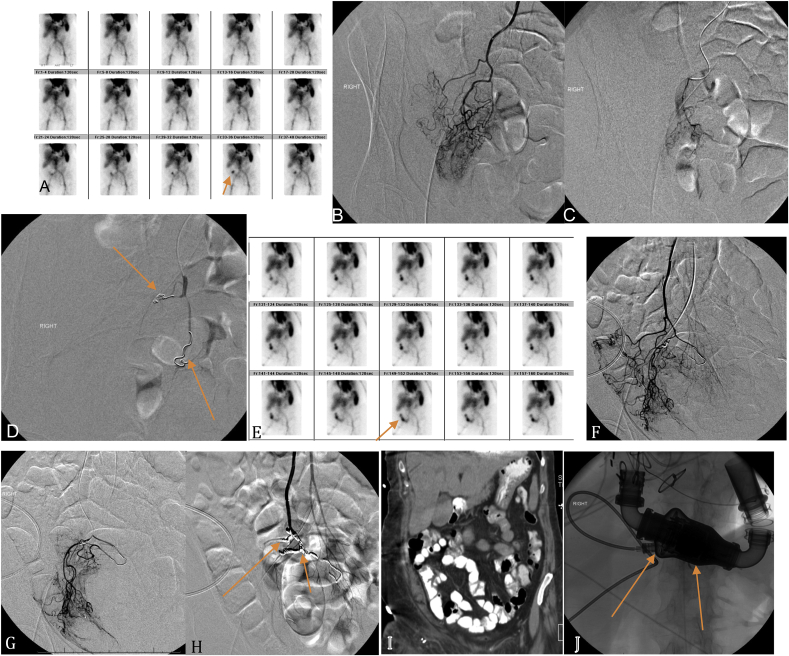
Our group recently conducted a retrospective study including 126 patients of LGIB for with mesenteric angiography performed during a 10 years period.21 Approximately one-third of the initial mesenteric angiographies demonstrated no identifiable bleeding source. Subsequently, a total of 18 patients underwent 24 prophylactic embolization procedures based on the pre-angiographic localization by nuclear scintigraphy. When compared to the angiographically occult LGIB patients who had no interventions, the patients who had prophylactic embolotherapy were found to have significantly decreased bleeding recurrence without increased risk of post-embolization bowel infarct, suggesting that prophylactic embolotherapy of LGIB may be valuable and safe in selected clinical populations. Future studies need to be performed to verify the safety and efficacy of prophylactic embolotherapy for angiographically occult LGIB, with focus on bleeding etiologies, anatomic location, comorbidities, and embolization techniques/agents (Fig. 1).
Fig. 1.
(A-E), 49 years old male has anticoagulation related lower GI bleeding. A. SMA arteriogram showed significant tissue stain/contrast extravasation, indicating active bleeding at the proximal jejunum (arrow). B. Sub-selective jejunal branch of SMA showed significant tissue stain, consistent with findings of angiodysplasia. C. Status post sub-selective micro-coils and Gel-foam slurry embolization of the jejunal branch of SMA with minimal residual tissue stain (arrow). D. CT angiogram one day post first embolization: active contrast extravasation again noted at proximal jejunum (arrow). E. Repeat mesenteric angiography: active bleeding localized from different segmental jejunal artery (arrow). F. Successful trans-catheter embolization with micro-coils (arrow) with no further bleeding and complications.
Pre-angiographic localization of LGIB is crucial for an effective prophylactic embolization of LGIB. Mesenteric angiography with potential embolization typically follows a positive pre-angiography Tc-99M RBC scan or CT angiogram, and occasionally both.14,15 If the initial mesenteric angiogram is negative, the decision is usually to observe and continue conservative treatment (16). However, our study suggested that with a sufficiently precise pre-angiography localization of the bleeding source, a prophylactic embolization can be performed to prevent recurrent bleeding effectively and may not increase the risk of bowel infarct compared to super-selective embolization. Although the American College of Radiology Appropriateness Criteria rates Tc-99M red blood cell (RBC) scintigraphy and CT angiogram equally appropriate, it remains controversial that which one is superior for pre-angiography screening in LGIB patients. Tc-99M RBC scan has been reported as a more sensitive test compared to CT angiogram, detecting bleeding at rates of as low as 0.1–0.4 ml/min, whereas CT angiogram is reported to be sensitive for source detection with rates of at least 0.5 ml/min22. Importantly, Tc-99M RBC scintigraphy is helpful for intermittent bleeding because imaging is performed continuously during a 1–2 h period. As a result, scintigraphic screening increases the diagnostic yield of mesenteric angiography in patients who are not actively bleeding at the time of examination.23 However, a significant limitation for Tc-99M RBC scintigraphy is its poor ability to precisely localize the bleeding source, whereas a positive CT angiogram’s high spatial resolution may provide a clear roadmap to target angiography.19,22 Our institutional protocol for patients with clinical suspicion of LGIB, with or without a prior colonoscopy, is to perform a Tc-99M RBC scintigraphy before angiography. CT angiogram may be performed on the basis of an equivocal Tc-99M RBC scan to more precisely obtain pre-angiography localization of the bleeding source. Following a positive pre-angiography screening, it is critical to perform an angiography as quickly as reasonably possible, to increase the yield of angiography as suggested by literature.23 Future studies on prophylactic embolization treating LGIB should include evaluations of screening CT angiogram on its role of providing precise anatomic information of the bleeding source, which possibly results in safer and more effective prophylactic embolotherapy.
3. Management of LGIB in anticoagulation and LVAD recipients by interventional radiology
In the past decade, anticoagulation therapy has become a common therapy for variety patients, and also, a rapidly growing population of patients with end-stage heart disease have undergone left ventricular assist device (LVAD) implantation due to organ shortage. Currently, second generation LVAD - continuous-flow LVAD (CF LVAD) is widely used. Compared to first generation LVAD which were pulsatile volume-displacement devices, CF LVAD provides improved reliability and allows implantation in smaller patients,24 expanding the eligible patient population.
LGIB is a significant adverse event following LVAD implantation, its incidence in LVAD recipients is reported to range from 17.6%-40%.25,27 The higher risk does not appear entirely attributable to the aggressive anticoagulation therapy associated with LVAD use. A recent study showed a higher rate of GIB in LVAD recipients compared with patients receiving anticoagulation for cardiac valve replacement surgery.28 The underlying mechanism to account for increased bleeding risk beyond that of anticoagulation therapy remains unclear. Factors such as altered hemodynamics, reduced pulsatility, association with arteriovenous malformation (AVM), and acquired von Willebrand disease have been proposed as possible mechanisms.25,29,30
The most common etiology for LGIB in anticoagulation and LVAD recipients is intestinal angiodysplasia.31,32 Angiodysplasia is a type of AVM with ectatic mucosal capillaries in the intestine wall, communicating with dilated and tortuous submucosal veins. The abnormal vessels may be single or multiple, with 80% of these lesions located in the right colon and cecum.22,33 Angiodysplasia is uncommon in young patients. In patients older than 65, angiodysplasia is reported as an identifiable source of bleeding in 20% of LGIB cases. Approximately 15% of patients with angiodysplasia will suffer from massive bleeding.11,34,35 Angiodysplasia also has an increased prevalence in patients with certain predisposing conditions, such as end-stage renal disease,36 von Willebrand disease,37 and aortic stenosis.38 In LVAD patients the reduction of pulsatility of blood flow is thought to cause intestinal hypoperfusion and therefore lead to the development of angiodysplasia. Interestingly, most recipients of LVAD also developed acquired von Willebrand syndrome as demonstrated by reduced high-molecular-weight von Willebrand factor multimer levels.39 It has been hypothesized that patients with Heyde syndrome, which refers to an association between aortic valve stenosis and GIB, may share similar pathophysiology with those LVAD recipients who suffer GIB. The etiology of Heyde syndrome is thought to be related to intestinal angiodysplasia and acquired von Willebrand syndrome.40,41 In patients with Heyde syndrome, stenotic aortic valve causes decreased pulsatility with an associated risk of angiodysplasia as well as the destruction of von Willebrand factor multimers proteins due to mechanical shearing at the stiffened valve, leading to the development of acquired von Willebrand syndrome and subsequent increased risk of GIB.42,43 Patients with a LVAD likely share similar pathophysiology as the device may affect similar hearing forces by the impeller mechanism.44, 45, 46
Currently, the first-line treatment for angiodysplasia is endoscopic interventions. In some patients with LVAD, despite repeated endoscopic interventions, no definitive cure can be obtained.43 Clinical management of LGIB in LVAD recipients by interventional radiology has therefore remained an essential adjunctive therapy. Angiodysplasia presents with multiple lesions in more than 50% cases, most commonly in the cecum and right colon on the antimesenteric border, so it is a frequently identifiable bleeding source in LGIB.47 Bleeding from angiodysplasia stops spontaneously in the majority of the cases, however in approximately 15% of the patients it can be life-threatening, and requires immediate and appropriate management to achieve hemostasis. Recurrent angiodysplastic bleeding is also associated with an increased mortality rate. Particularly in patients with LVAD, a recurrent GIB may necessitate multiple blood transfusions, leading to the development of circulating antibodies, which subsequently reduces the probability of an organ donor match and simultaneously raises the risk of cellular rejection after heart transplantation and transfusion incompatibility.48
To our knowledge, few clinical studies have been performed by far to address the outcomes of patients with LVAD induced LGIB. Mehta et al. recently conducted a retrospective study including both upper and lower GIB patients in LVAD recipients, to evaluate clinical outcomes following mesenteric angiography.49 They observed a lower rate of a positive angiograms, decreased embolization efficacy, higher rates of repeat angiography procedures, and prolonged length of stay in GIB patients with LVADs. Further, they reported this patient population had higher rates of clinical failure following interventional radiology management, with a more frequent need for subsequent surgical intervention, compared to those without LVADs. More recently, a retrospective study was performed by our group to investigate the clinical outcome following superselective transarterial embolization as well as prophylactic embolotherapy of LGIB in LVAD recipients in comparison to patients in which no embolization was performed.50 Focusing only on LGIB, we found bleeding in LVAD recipients is more likely to be angiographically occult, therefore leading to more frequent prophylactic embolizations based on pre-procedural imaging compared to patients without LVADs. Our study also suggested that transarterial embolization has a lower success rate in treating LGIB in LVAD recipients, whereas prophylactic embolotherapy might be useful and safe when treating angiographically occult LGIB in this particular patient group (Fig. 2).
Fig. 2.
(A–J), 72 years old female, s/p LVAD, with anticoagulation related LGIB. A, nuclear scintigraphy: Bleeding at distal ileum (arrow); B-D Mesenteric angiography: no active bleeding localized. Prophylactic trans-catheter embolization of two branches of ileal artery with coils was done (arrows); E, s/p prophylactic coil embolization 1 week after. nuclear scintigraphy showed again bleeding at distal ileum (arrow); F–H, Mesenteric angiography again showed no signs of active bleeding. Additional prophylactic trans-catheter embolization of more branches of ileal artery with coils was performed (arrows); I, despite repeat prophylactic embolization, contrast enhanced CT scan showed no signs of bowel ischemia or infarction; J, abdominal scout picture showed left ventricular assisted device (arrow).
More future studies, ideally including prospective investigations with larger sample size on LGIB in LVAD recipients, should be performed to verify the findings from the limited numbers of prior studies, and to confirm that LGIB in LVAD recipients is a distinct entity from bleeding in patients receiving anticoagulation therapy. The incidence of bowel ischemia following embolotherapy in LVAD recipients is an important topic given LGIB in LVAD recipients tends to recur and therefore needs repetitive embolizations. Bowel infarct is the most feared complication following arterial embolization therapy in LGIB patients, and usually requires surgical bowel resection. Its incidence is estimated as 1.3%–5%.51,52 Risk of infarct is directly proportional to the extent of vascular territory embolized.15 Bowel ischemia occurs more often in the colon, because in contrast to the small intestine, the colon may have limited collateral flow through an incomplete marginal artery system on the colon wall.53 Within the large bowel, the cecum has the longest vasa recta with limited distal collaterals. These specific arterial anatomical arrangements may further predispose the cecum to ischemia.54 Future studies should specifically give attention to cecal bleeding in LVAD recipients, to address the potential higher risk of bowel infarct following embolotherapy. If confirmed, cecal branches should be approached with greater caution when embolization of cecal bleeding is considered. Given the increasing LVAD use and its strong association with LGIB, transcatheter arterial embolization and prophylactic embolotherapy by interventional radiologists may play an increasingly important role in this specific and challenging patient population.
4. Conclusion
Future studies need to be performed to verify the safety and efficacy of prophylactic embolotherapy for angiographically occult LGIB, with focus on bleeding etiologies, anatomic location, comorbidities, and embolization techniques/agents. More future studies, ideally including prospective investigations with larger sample size on LGIB in anticoagulation and LVAD recipients, should be performed to verify the findings from the limited numbers of prior studies, and to confirm that LGIB in LVAD recipients is a distinct entity from bleeding in patients receiving anticoagulation therapy. The incidence of bowel ischemia following embolotherapy in anticoagulation and LVAD recipients is an important topic given LGIB in LVAD recipients tends to recur and therefore need repetitive embolizations.
Contributor Information
Shihong Li, Email: Shihong.Li@emory.edu.
Shiliang Sun, Email: shiliang-sun@uiowa.edu.
References
- 1.Laine L., Yang H., Chang S.C. Trends for incidence of hospitalization and death due to GI complications in the United States from 2001 to 2009. Am J Gastroenterol. 2012;107:1190–1195. doi: 10.1038/ajg.2012.168. quiz 6. [DOI] [PubMed] [Google Scholar]
- 2.Farrell J.J., Friedman L.S. Review article: the management of lower gastrointestinal bleeding. Aliment Pharmacol Ther. 2005;21:1281–1298. doi: 10.1111/j.1365-2036.2005.02485.x. [DOI] [PubMed] [Google Scholar]
- 3.Longstreth G.F. Epidemiology and outcome of patients hospitalized with acute lower gastrointestinal hemorrhage: a population-based study. Am J Gastroenterol. 1997;92:419–424. [PubMed] [Google Scholar]
- 4.Chen C.Y., Wu C.C., Jao S.W. Colonic diverticular bleeding with comorbid diseases may need elective colectomy. J Gastrointest Surg. 2009;13:516–520. doi: 10.1007/s11605-008-0731-4. [DOI] [PubMed] [Google Scholar]
- 5.Parkes B.M., Obeid F.N., Sorensen V.J. The management of massive lower gastrointestinal bleeding. Am Surg. 1993;59:676–678. [PubMed] [Google Scholar]
- 6.Funaki B. Microcatheter embolization of lower gastrointestinal hemorrhage: an old idea whose time has come. Cardiovasc Interv Radiol. 2004;27:591–599. doi: 10.1007/s00270-004-0243-x. [DOI] [PubMed] [Google Scholar]
- 7.Marion Y., Lebreton G., Le Pennec V. The management of lower gastrointestinal bleeding. J Vis Surg. 2014;151:191–201. doi: 10.1016/j.jviscsurg.2014.03.008. [DOI] [PubMed] [Google Scholar]
- 8.Zuckerman D.A., Bocchini T.P., Birnbaum E.H. Massive hemorrhage in the lower gastrointestinal tract in adults: diagnostic imaging and intervention. AJR Am J Roentgenol. 1993;161:703–711. doi: 10.2214/ajr.161.4.8372742. [DOI] [PubMed] [Google Scholar]
- 9.Koval G., Benner K.G., Rosch J. Aggressive angiographic diagnosis in acute lower gastrointestinal hemorrhage. Dig Dis Sci. 1987;32:248–253. doi: 10.1007/BF01297049. [DOI] [PubMed] [Google Scholar]
- 10.Gomes A.S., Lois J.F., McCoy R.D. Angiographic treatment of gastrointestinal hemorrhage: comparison of vasopressin infusion and embolization. AJR Am J Roentgenol. 1986;146:1031–1037. doi: 10.2214/ajr.146.5.1031. [DOI] [PubMed] [Google Scholar]
- 11.Weldon D.T., Burke S.J., Sun S. Interventional management of lower gastrointestinal bleeding. Eur Radiol. 2008;18:857–867. doi: 10.1007/s00330-007-0844-2. [DOI] [PubMed] [Google Scholar]
- 12.Kim C.Y., Suhocki P.V., Miller M.J., Jr. Provocative mesenteric angiography for lower gastrointestinal hemorrhage: results from a single-institution study. J Vasc Interv Radiol. 2010;21:477–483. doi: 10.1016/j.jvir.2009.11.021. [DOI] [PubMed] [Google Scholar]
- 13.Bloomfeld R.S., Smith T.P., Schneider A.M. Provocative angiography in patients with gastrointestinal hemorrhage of obscure origin. Am J Gastroenterol. 2000;95:2807–2812. doi: 10.1111/j.1572-0241.2000.03191.x. [DOI] [PubMed] [Google Scholar]
- 14.Ryan J.M., Key S.M., Dumbleton S.A. Nonlocalized lower gastrointestinal bleeding: provocative bleeding studies with intraarterial tPA, heparin, and tolazoline. J Vasc Interv Radiol. 2001;12:1273–1277. doi: 10.1016/s1051-0443(07)61551-6. [DOI] [PubMed] [Google Scholar]
- 15.Speir E.J., Ermentrout R.M., Martin J.G. Management of acute lower gastrointestinal bleeding. Tech Vasc Interv Radiol. 2017;20:258–262. doi: 10.1053/j.tvir.2017.10.005. [DOI] [PubMed] [Google Scholar]
- 16.Lang E.V., Picus D., Marx M.V. Massive upper gastrointestinal hemorrhage with normal findings on arteriography: value of prophylactic embolization of the left gastric artery. AJR Am J Roentgenol. 1992;158:547–549. doi: 10.2214/ajr.158.3.1738991. [DOI] [PubMed] [Google Scholar]
- 17.Schenker M.P., Duszak R., Jr., Soulen M.C. Upper gastrointestinal hemorrhage and transcatheter embolotherapy: clinical and technical factors impacting success and survival. J Vasc Interv Radiol. 2001;12:1263–1271. doi: 10.1016/s1051-0443(07)61549-8. [DOI] [PubMed] [Google Scholar]
- 18.Kelemouridis V., Athanasoulis C.A., Waltman A.C. Gastric bleeding sites: an angiographic study. Radiology. 1983;149:643–648. doi: 10.1148/radiology.149.3.6606186. [DOI] [PubMed] [Google Scholar]
- 19.Funaki B. On-call treatment of acute gastrointestinal hemorrhage. Semin Interv Radiol. 2006;23:215–222. doi: 10.1055/s-2006-948758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lieberman D.A., Keller F.S., Katon R.M. Arterial embolization for massive upper gastrointestinal tract bleeding in poor surgical candidates. Gastroenterology. 1984;86:876–885. [PubMed] [Google Scholar]
- 21.Li S., Sun S., Wang S. Safety and outcome of prophylactic embolotherapy for angiographic negative lower gastrointestinal bleeding. J Vasc Interv Radiol. 2016;27:S11. [Google Scholar]
- 22.Filippone A., Cianci R., Milano A. Obscure and occult gastrointestinal bleeding: comparison of different imaging modalities. Abdom Imag. 2012;37:41–52. doi: 10.1007/s00261-011-9802-1. [DOI] [PubMed] [Google Scholar]
- 23.Gunderman R., Leef J., Ong K. Scintigraphic screening prior to visceral arteriography in acute lower gastrointestinal bleeding. J Nucl Med. 1998;39:1081–1083. [PubMed] [Google Scholar]
- 24.Lalonde S.D., Alba A.C., Rigobon A. Clinical differences between continuous flow ventricular assist devices: a comparison between HeartMate II and HeartWare HVAD. J Card Surg. 2013;28:604–610. doi: 10.1111/jocs.12158. [DOI] [PubMed] [Google Scholar]
- 25.Morgan J.A., Paone G., Nemeh H.W. Gastrointestinal bleeding with the HeartMate II left ventricular assist device. J Heart Lung Transplant. 2012;31:715–718. doi: 10.1016/j.healun.2012.02.015. [DOI] [PubMed] [Google Scholar]
- 27.Aggarwal A., Pant R., Kumar S. Incidence and management of gastrointestinal bleeding with continuous flow assist devices. Ann Thorac Surg. 2012;93:1534–1540. doi: 10.1016/j.athoracsur.2012.02.035. [DOI] [PubMed] [Google Scholar]
- 28.Shrode C.W., Draper K.V., Huang R.J. Significantly higher rates of gastrointestinal bleeding and thromboembolic events with left ventricular assist devices. Clin Gastroenterol Hepatol. 2014;12:1461–1467. doi: 10.1016/j.cgh.2014.01.027. [DOI] [PubMed] [Google Scholar]
- 29.Stulak J.M., Lee D., Haft J.W. Gastrointestinal bleeding and subsequent risk of thromboembolic events during support with a left ventricular assist device. J Heart Lung Transplant. 2014;33:60–64. doi: 10.1016/j.healun.2013.07.020. [DOI] [PubMed] [Google Scholar]
- 30.Schaffer J.M., Arnaoutakis G.J., Allen J.G. Bleeding complications and blood product utilization with left ventricular assist device implantation. Ann Thorac Surg. 2011;91:740–747. doi: 10.1016/j.athoracsur.2010.11.007. discussion 7-9. [DOI] [PubMed] [Google Scholar]
- 31.Singh G., Albeldawi M., Kalra S.S. Features of patients with gastrointestinal bleeding after implantation of ventricular assist devices. Clin Gastroenterol Hepatol. 2015;13 doi: 10.1016/j.cgh.2014.05.012. 107-14.e1. [DOI] [PubMed] [Google Scholar]
- 32.Draper K.V., Huang R.J., Gerson L.B. GI bleeding in patients with continuous-flow left ventricular assist devices: a systematic review and meta-analysis. Gastrointest Endosc. 2014;80:435–446.e1. doi: 10.1016/j.gie.2014.03.040. [DOI] [PubMed] [Google Scholar]
- 33.Dray X., Camus M., Coelho J. Treatment of gastrointestinal angiodysplasia and unmet needs. Dig Liver Dis. 2011;43:515–522. doi: 10.1016/j.dld.2010.12.007. [DOI] [PubMed] [Google Scholar]
- 34.Stern D.R., Kazam J., Edwards P. Increased incidence of gastrointestinal bleeding following implantation of the HeartMate II LVAD. J Card Surg. 2010;25:352–356. doi: 10.1111/j.1540-8191.2010.01025.x. [DOI] [PubMed] [Google Scholar]
- 35.Sharma R., Gorbien M.J. Angiodysplasia and lower gastrointestinal tract bleeding in elderly patients. Arch Intern Med. 1995;155:807–812. [PubMed] [Google Scholar]
- 36.Zuckerman G.R., Cornette G.L., Clouse R.E. Upper gastrointestinal bleeding in patients with chronic renal failure. Ann Intern Med. 1985;102:588–592. doi: 10.7326/0003-4819-102-5-588. [DOI] [PubMed] [Google Scholar]
- 37.Duray P.H., Marcal J.M., Jr., LiVolsi V.A. Gastrointestinal angiodysplasia: a possible component of von Willebrand’s disease. Hum Pathol. 1984;15:539–544. doi: 10.1016/s0046-8177(84)80007-6. [DOI] [PubMed] [Google Scholar]
- 38.Pate G.E., Mulligan A. An epidemiological study of Heyde’s syndrome: an association between aortic stenosis and gastrointestinal bleeding. J Heart Valve Dis. 2004;13:713–716. [PubMed] [Google Scholar]
- 39.Crow S., Chen D., Milano C. Acquired von Willebrand syndrome in continuous-flow ventricular assist device recipients. Ann Thorac Surg. 2010;90:1263–1269. doi: 10.1016/j.athoracsur.2010.04.099. discussion 7-9. [DOI] [PubMed] [Google Scholar]
- 40.Sucker C. The Heyde syndrome: proposal for a unifying concept explaining the association of aortic valve stenosis, gastrointestinal angiodysplasia and bleeding. Int J Cardiol. 2007;115:77–78. doi: 10.1016/j.ijcard.2006.01.014. [DOI] [PubMed] [Google Scholar]
- 41.Thompson J.L., 3rd, Schaff H.V., Dearani J.A. Risk of recurrent gastrointestinal bleeding after aortic valve replacement in patients with Heyde syndrome. J Thorac Cardiovasc Surg. 2012;144:112–116. doi: 10.1016/j.jtcvs.2011.05.034. [DOI] [PubMed] [Google Scholar]
- 42.Pareti F.I., Lattuada A., Bressi C. Proteolysis of von Willebrand factor and shear stress-induced platelet aggregation in patients with aortic valve stenosis. Circulation. 2000;102:1290–1295. doi: 10.1161/01.cir.102.11.1290. [DOI] [PubMed] [Google Scholar]
- 43.Joy P.S., Kumar G., Guddati A.K. Risk factors and outcomes of gastrointestinal bleeding in left ventricular assist device recipients. Am J Cardiol. 2016;117:240–244. doi: 10.1016/j.amjcard.2015.10.041. [DOI] [PubMed] [Google Scholar]
- 44.Demirozu Z.T., Radovancevic R., Hochman L.F. Arteriovenous malformation and gastrointestinal bleeding in patients with the HeartMate II left ventricular assist device. J Heart Lung Transplant. 2011;30:849–853. doi: 10.1016/j.healun.2011.03.008. [DOI] [PubMed] [Google Scholar]
- 45.Islam S., Cevik C., Madonna R. Left ventricular assist devices and gastrointestinal bleeding: a narrative review of case reports and case series. Clin Cardiol. 2013;36:190–200. doi: 10.1002/clc.22096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kurien S., Hughes K.A. Anticoagulation and bleeding in patients with ventricular assist devices: walking the tightrope. AACN Adv Crit Care. 2012;23:91–98. doi: 10.1097/NCI.0b013e31824124d0. [DOI] [PubMed] [Google Scholar]
- 47.Jackson C.S., Gerson L.B. Management of gastrointestinal angiodysplastic lesions (GIADs): a systematic review and meta-analysis. Am J Gastroenterol. 2014;109:474–483. doi: 10.1038/ajg.2014.19. quiz 84. [DOI] [PubMed] [Google Scholar]
- 48.John R., Lietz K., Schuster M. Immunologic sensitization in recipients of left ventricular assist devices. J Thorac Cardiovasc Surg. 2003;125:578–591. doi: 10.1067/mtc.2003.30. [DOI] [PubMed] [Google Scholar]
- 49.Mehta A., Kim S., Ahmed O. Outcomes of patients with left ventricular assist devices undergoing mesenteric angiography for gastrointestinal bleeding. J Vasc Interv Radiol. 2015;26:1710–1717. doi: 10.1016/j.jvir.2015.07.026. [DOI] [PubMed] [Google Scholar]
- 50.Li S, Sun S. Clinical Significance of Prophylactic Embolotherapy of Lower Gastrointestinal Bleeding in Left Ventricular Assist Device Recipients. 77th Annual Meeting of the Japan Radiological Society; April 12-15, 2018; Yokohama, Japan.
- 51.Kickuth R., Rattunde H., Gschossmann J. Acute lower gastrointestinal hemorrhage: minimally invasive management with microcatheter embolization. J Vasc Interv Radiol. 2008;19:1289–1296.e2. doi: 10.1016/j.jvir.2008.06.003. [DOI] [PubMed] [Google Scholar]
- 52.Lipof T., Sardella W.V., Bartus C.M. The efficacy and durability of super-selective embolization in the treatment of lower gastrointestinal bleeding. Dis Colon Rectum. 2008;51:301–305. doi: 10.1007/s10350-007-9149-4. [DOI] [PubMed] [Google Scholar]
- 53.Ross J.A. Vascular patterns of small and large intestine compared. Br J Surg. 1952;39:330–333. doi: 10.1002/bjs.18003915612. [DOI] [PubMed] [Google Scholar]
- 54.Simon A.M., Birnbaum B.A., Jacobs J.E. Isolated infarction of the cecum: CT findings in two patients. Radiology. 2000;214:513–516. doi: 10.1148/radiology.214.2.r00fe15513. [DOI] [PubMed] [Google Scholar]