Abstract
Objective
Spain’s so-called Stroke Belt is an area with high prevalence of vascular disease. We aimed to determine the prevalence of undetected obstructive sleep apnea–hypopnea syndrome (OSAHS) among patients with acute ischemic stroke (AIS) in southern Spain.
Methods
We conducted a cross-sectional study at the Virgen Macarena University Hospital Stroke Unit during 2018 to 2019. We included patients <72 hours after AIS with a neuroimaging lesion and performed sleep tests.
Results
Seventy-two patients were included. The median participant age was 72 years. Mean body mass index was 27.07 kg/m2, and 40.28% were daily alcohol drinkers. Hypertension, atrial fibrillation, ischemic cardiomyopathy, and previous stroke were detected in 63.9%, 11.1%, 15.3%, and 17.6% of patients, respectively. Polygraphy was feasible in 91.38% of patients. The prevalence of OSAHS was 84.72% (apnea–hypopnea index ≥5). Patients with moderate and severe OSAHS were more likely to be obese and to have a larger neck circumference and facial palsy. The diagnostic criteria of central sleep apnea syndrome were met in only 1.38% of patients.
Conclusions
The high prevalence of OSAHS found in the Spanish Stroke Belt justifies further investigation and development of a screening program as a strategy to identify patients with undetected OSAHS.
Keywords: Spanish Stroke Belt, ischemic stroke, obstructive sleep apnea–hypopnea syndrome, polygraphy, polysomnography, screening
Introduction
Classical risk factors for acute ischemic stroke (AIS), such as hypertension, diabetes, or atrial fibrillation, have been widely established. 1 Obstructive sleep apnea–hypopnea syndrome (OSAHS) has been recently described as an emerging risk factor for the first and recurrent AIS. 2 Moreover, OSAHS has been associated with worse neurological prognosis and higher mortality in patients with AIS. 2 The relationship between OSAHS and stroke might be partially explained by the presence of common risk factors for both diseases. For example, OSAHS has been proposed as a risk factor for refractory hypertension and high nocturnal blood pressure, 3 both of which also increase the risk of AIS. Additionally, it is estimated that two-thirds of patients with atrial fibrillation, another risk factor for AIS, might also have OSAHS. 4 There are several pathogenic hypotheses relating OSAHS with vascular disease. The potential molecular signatures of OSAHS are oxidative stress mechanisms and activation of inflammatory pathways and the sympathetic system. 5
According to epidemiological studies, the estimated general prevalence of OSAHS among adults in Spain is approximately 27.1%, 6 with a significant increasing trend with older age; the prevalence of OSAHS in the 60- to 70-year age group is 46.9% to 52.1%. 6 However, in epidemiological terms, this prevalence only represents the “tip of the iceberg.” Data from the Wisconsin Sleep Cohort Study of patients estimated that 93% of women and 82% of men with moderate-to-severe sleep apnea are undiagnosed. 7 There is cross-sectional evidence suggesting that OSAHS is more common among patients with stroke. 8 The prevalence of OSAHS in Spain among patients with AIS ranges from 62% to 85%. 8 The so-called Spanish Stroke Belt is an area in southern Spain (Andalusia) with a high prevalence of vascular disease owing to some known and other unknown reasons. 9 OSAHS prevalence among patients with AIS in this region has not been widely studied. The gap regarding prevalence among previous epidemiological studies could be related to different target populations or diagnostic methods. Epidemiological studies in which the diagnosis of OSAHS has been made using clinical scales might have methodological limitations in regions with low health education, like some parts of Spain.
A study performed in a wealthy region of Spain found that more than 10% of the population was unaware of stroke and only 37% of the population had good knowledge of risk factors for stroke. 1 These results could be even worse in the Spanish Stroke Belt, where some less-developed economic areas and lower education rates could partly explain insufficient control of vascular risk factors and high mortality from stroke. 9
The main research question in this study was to investigate the magnitude of undetected OSAHS among patients with stroke in Andalusia. We hypothesized that a high prevalence of OSAHS exists in this population. The main aim of this study was to determine the prevalence of undetected OSAHS among patients with AIS in the Spanish Stroke Belt using sleep tests during hospitalization.
Methods
We conducted a cross-sectional study at the Virgen Macarena Stroke Unit during 2018 to 2019. We included consecutive patients with AIS admitted to the Stroke Unit within 72 hours of symptom onset during 2018. Exclusion criteria were baseline modified Rankin score >3, 10 home oxygen therapy for any cause, severe cognitive impairment, upper airway tumors, significant neuromuscular and chest wall disorders, previous diagnosis of OSAHS, clinical instability, and severe agitation or need for high-flow oxygen therapy owing to any disease during hospitalization. Screening failure was considered to be any of the following situations: stroke mimic, clinical instability or agitation onset during sleep testing, technical problems with polygraphy analysis software, or patient revocation of informed consent.
This study was approved by the ethics committee of Virgen Macarena University Hospital (ADM-SAS-2017/1773-N-17) and was performed in compliance with the principles laid down in the Declaration of Helsinki. The experiments were undertaken with the written informed consent of each patient to participate in the study and for publication. We de-identified patient details such that the identity of any person cannot be ascertained in any way. Baseline variables were recorded using a standardized protocol. Full data and materials are available upon request. The reporting of this study conforms to the STROBE guidelines. 11
Demographic and health characteristics
Our hospital serves a population of 511,000 inhabitants of metropolitan and rural areas in the province of Seville, Spain. The province has 1.94 million inhabitants and two stroke units at the two largest hospitals in the capital city of Seville. Our Stroke Unit admits all patients with stroke within 72 hours of onset, except those who are dependent or have any acute severe comorbidities.
OSAHS assessment
Each participant underwent a sleep study (polygraphy and/or polysomnography). Treatments that could interfere with sleep tests, such as use of benzodiazepines or opioids, were withdrawn after patients’ inclusion in the study.
Polygraphy
Polygraphy was performed in all patients at the Stroke Unit during hospitalization. Polygraphies were conducted by trained nursing staff of the Stroke Unit. Following the current guidelines for OSAHS management, 12 polygraphy included recordings of airflow with a nasal cannula and thermistor, body position, snoring, oxygen saturation, heart rate, and respiratory effort. Data were recorded using a validated multichannel digital polygraph (Sleep&Go, Sibelmed, Spain). Only polygraphs with a total recording time >4 hours were considered in this study. 12 Each polygraph was manually scored by a sleep pulmonologist. Feasibility of overnight cardiorespiratory polygraphy was defined as an analyzable polygraph in 90% of the patients studied.
Polysomnography
If polygraphy was inconclusive for a diagnosis of OSAHS because of artifacts, an outpatient polysomnography was performed in the sleep laboratory. Polysomnography was also carried out in some cases to confirm similar diagnostic power in both types of sleep study.
A validated polysomnography platform (Nihon Coden EEG-1200 System Program Version 01-91) was used to record eight electroencephalogram channels, two electrooculogram channels, one chin electromyogram channel, nasal airflow, chest and abdominal wall excursion, heart rate, oxygen saturation, and two anterior tibialis electromyogram channels.
Polysomnographies were performed by trained medical staff at the sleep laboratory during one night. Sleep-stage scoring was done visually according to standard criteria. 13 A sleep neurophysiologist manually analyzed each polysomnograph. Only polysomnographs with a total recording time >6.5 hours and a total sleep time >180 minutes were included in this study. 12
Sleep test data analysis
Apnea was defined as a ≥90% reduction in nasal airflow lasting ≥10 s. Hypopnea was defined as a ≥30% and <90% reduction in nasal airflow in association with oxygen desaturation ≥3% or with respiratory effort-related arousals. An arousal was recorded in polysomnography if there was an abrupt shift lasting 3 s or longer in electroencephalogram frequency to alpha or theta or >16 Hz, following at least 10 s of sleep, and an increase in electromyogram tone with arousal during rapid eye movement sleep. 12
The apnea–hypopnea index (AHI) is defined as the mean number of apneas and hypopneas per hour of study in polygraphy and per hour of sleep in polysomnography. 13 Apneas with thoracic motion, without thoracic motion, or with an initial lack of motion followed by respiratory effort were classified as obstructive, central, and mixed, respectively. 13 OSAHS is defined as AHI ≥5 per hour, according to the third edition of the International Classification of Sleep Disorders. 13 Severity of OSAHS was graded mild, moderate, and severe according to AHI ranges of 5 to 14.9, 15 to 29.9, and ≥30 per hour, respectively. 13 Comorbid central sleep apnea syndrome (CSAS) is defined as five or more central apneas or hypopneas per hour of sleep and a total number of central events >50% of the total number of apneas and hypopneas. 13
If the same patient underwent both polygraphy and polysomnography, the data of the latter, which is the gold standard test, were taken into account. Diagnostic agreement was defined as the ability of each test to yield the same OSAHS grade, according to previous guidelines. 14
Stroke assessment
Diagnosis of stroke was confirmed by a vascular neurologist on the basis of a history of sudden onset of neurological deficit lasting at least 1 hour and a brain lesion on neuroimaging. Severity of stroke was assessed using a validated Spanish version of the National Institute of Health Stroke Scale (NIHSS). 15 The TOAST system (Trial of ORG 10172 in Acute Stroke Treatment Subtype Classification) was used to categorize ischemic stroke subtypes. 16 The degree of previous disability was classified according to the modified Rankin score. 10 A face-to-face evaluation was carried out during hospitalization in which anthropometric and epidemiological data, such as vascular risk factors, smoking, alcohol consumption (standard drink units per day), previous treatments, and comorbidities were collected.
Statistical analysis
We used the Student t-test or Mann–Whitney U test to detect differences among the groups. Values were compared using Fisher’s exact test and the chi-square test in statistical analysis. A p-value of <0.05 was considered significant. All data were analyzed using IBM SPSS version 25 (IBM Corp., Armonk, NY, USA).
Results
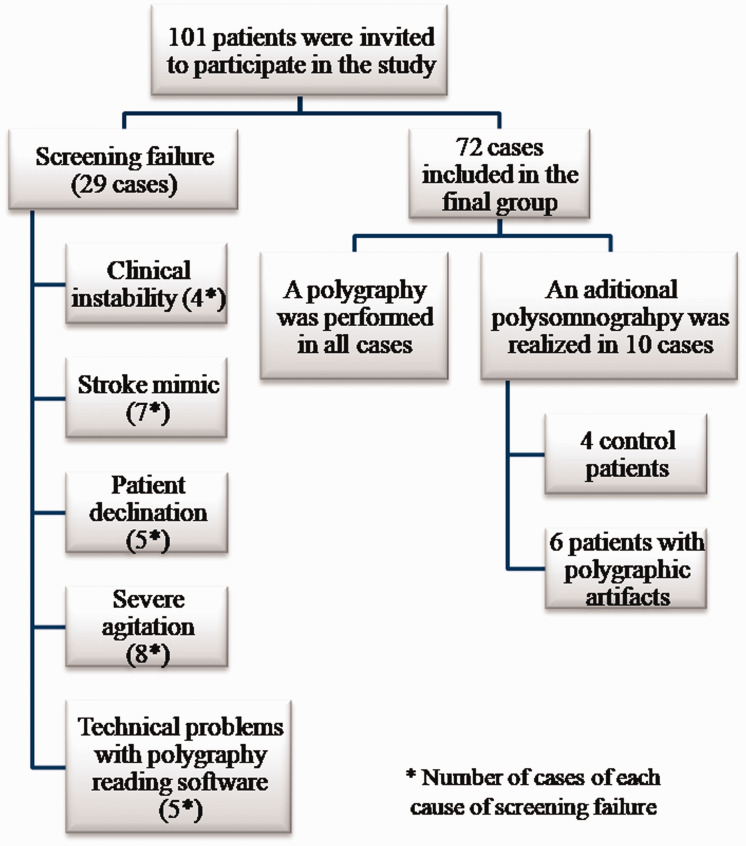
A total of 101 patients were invited to participate in the study (Figure 1). Screening failure occurred in 29 patients. The main reasons for screening failure were clinical instability after study inclusion in four patients, severe agitation in eight patients, seven stroke mimics, revocation of informed consent in five patients, and technical problems with polygraphy analysis software in five patients.
Figure 1.
Study design. Sample size, reasons for screening failure, and type of sleep test performed.
A sample of 72 patients with AIS was finally selected. Clinical baseline characteristics of patients are shown in Table 1. The mean patient age was 70.46 ± 10.83 years, and 30.6% of patients were women. The median baseline NIHSS was 4. Large vessel occlusion was detected in 29.16% of patients. In the group of patients with previous stroke, 69.23% had experienced only one cerebrovascular event. Patients with cognitive impairment had mild or moderate cases that were related to Alzheimer disease or vascular dementia. In univariate analyses, patients with moderate and severe OSAHS were more likely to be obese (p = 0.01) and have a larger neck circumference (p = 0.02) (Table 1). The presence of facial palsy because of stroke was significantly associated with moderate and severe OSAHS (p = 0.02). There was no association of stroke severity with baseline NIHSS and AHI (Table 1).
Table 1.
Baseline clinical data.
| All patients (n = 72) | AHI <15 (n = 27) | AHI ≥15 (n = 47) | p | |
|---|---|---|---|---|
| Mean age (years) ± SD | 70.46 ± 10.83 | 72.4 ± 11.2 | 69.3 ± 10.6 | 0.23‽0.21† |
| Female sex (%) | 30.60 | 28.90 | 33.30 | 0.692‡ |
| Diabetes (%) | 33.30 | 33.30 | 33.30 | 0.60‡ |
| Dyslipidemia (%) | 29.20 | 25.90 | 31.10 | 0.64‡ |
| Hypertension (%) | 63.9 | 55.6 | 68.9 | 0.25‡ |
| Basal atrial fibrillation (%) | 11.10 | 11.10 | 11.10 | 0.64‡ |
| Ischemic cardiomyopathy (%) | 15.30 | 11.10 | 17.80 | 0.45‡ |
| Benzodiazepine treatment in the past 3 months (%) | 25 | 25.9 | 24.4 | 0.89‡ |
| Opioid treatment in the past 3 months (%) | 11.10 | 11.10 | 11.10 | 0.64‡ |
| Mean body mass index ± SD | 27.07 ± 4.50 | 25.37 ± 4.37 | 28.09 ± 4.30 | 0.01‽† |
| Limb arteriopathy (%) | 9.70 | 7.40 | 11.10 | 0.61‡ |
| Previous stroke (%) | 17.60 | 14.80 | 17.80 | 0.74‡ |
| Stroke onset between 18:00 and 06:00 (%) | 32.40 | 20.00 | 39.50 | 0.09‡ |
| Facial palsy** (%) | 54.20 | 37.00 | 64.40 | 0.02‡ |
| Hypoglossal palsy** (%) | 4.20 | 3.70 | 4.40 | 0.87‡ |
| Dysphagia** (%) | 30.60 | 29.60 | 31.10 | 0.89‡ |
| Baseline NIHSS*** >4 (%) | 39.4 | 37.00 | 40.90 | 0.74‡ |
| Mean neck circumference (cm) ± SD | 42.13 ± 5.03 | 40.30 ± 4.89 | 43.14 ± 4.92 | 0.02‽0.04† |
| Hyperacute endovascular treatment (%) | 13.90 | 18.50 | 11.10 | 0.37‡ |
| Thrombolysis (%) | 26.4 | 37.00 | 20.00 | 0.11‡ |
AHI, apnea–hypopnea index; NIHSS, National Institute of Health Stroke Scale; SD, standard deviation.
‽Student t-test. Equal variances are assumed.
‡Chi-square or Fisher’s exact test.
†Mann–Whitney U test.
** Stroke sequelae during baseline neurological examination.
Polygraphy was performed in all patients, and polysomnography was also conducted in 10 patients. In six patients, the presence of polygraphic artifacts necessitated polysomnography. The remaining four patients were selected to undergo both sleep tests, to confirm their similar diagnostic power. In the four patients who underwent polygraphy and polysomnography, OSAHS grade diagnostic agreement was 100% between both sleep tests. In patients who underwent both types of sleep study, the mean variation in AHI was 4.8 (with higher values in polysomnography than in polygraphy). Sleep test recordings (polygraphy and polysomnography) were adequate in all patients, so there were no missing values in the study.
In the final group of enrolled patients, the feasibility of polygraphy was 91.38% because only 7 of 72 recordings were not able to be analyzed. In these cases, polysomnography was also conducted. The mean time from stroke onset to polygraphy was 35.9 hours, and the mean time from stroke onset to polysomnography was 101 days.
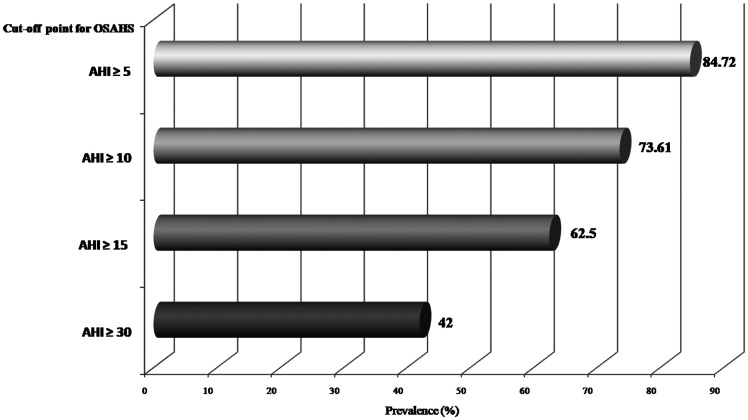
The prevalence of undetected OSAHS in our study was 84.72% (AHI cutoff ≥5 per hour, per the guidelines). OSAHS prevalence data according to different diagnostic cutoff points are presented in Figure 2. In patients with positive results, the most frequent OSAHS grade was severe, which was detected in 59.34% of patients. Among these patients, 20.33% had moderate OSAHS grade and 20.33% had mild grade. Sleep test results are summarized in Table 2. The mean for obstructive AHI was 18.46 ± 20.33 in patients with AHI ≥15 versus 4.93 ± 10.77 in patients with AHI <15 (p < 0.001). The mean for central AHI in the whole sample was 4.41 ± 9.12. The mean for central AHI was 6.57 ± 10.95 in patients with AHI ≥15 versus 0.81 ± 1.62 in those with AHI <15 (p = 0.002). The diagnostic criteria for CSAS were only met in two patients (1.38% of the total). Both of these patients had an OSAHS diagnosis with comorbid CSAS. In one of these patients, a Cheynes–Stokes pattern was confirmed.
Figure 2.
Prevalence of obstructive sleep apnea–hypopnea syndrome (OSAHS) according to apnea–hypopnea index (AHI) cutoff point.
Table 2.
Sleep study results.
| N = 72 | |
|---|---|
| Mean AHI ± SD | 26.70 ± 27.43 |
| Mean obstructive AHI ± SD | 13.39 ± 18.50 |
| Mean central AHI ± SD | 4.41 ± 9.12 |
| Mean mixed AHI ± SD | 5.75 ± 11.52 |
| Mean CT90 (%) ± SD | 19.42 ± 25.97 |
| Mean minimum oxygen saturation (%) ± SD | 75.61 ± 13.42 |
| Mean of mean oxygen saturation (%) ± SD (n = 72) | 92.03 ± 4.41 |
| Mean oxygen desaturation index (events per hour) ± SD | 29.22 ± 26.65 |
AHI, apnea–hypopnea index; CT90, percentage of study period during which patient had oxygen saturation <90%; SD, standard deviation.
Patients with normal polygraphy had no OSAHS symptoms, so no further sleep testing was necessary. A comparison among the baseline characteristics and results of published OSAHS prevalence studies with those of the present study is given in Table 3.
Table 3.
Comparison among the baseline characteristics and results of published studies on OSAHS prevalence with those of our study.
| n (% female sex) | Mean BMI | Mean age | % Hyper-tension | % Ischemic cardio-myopathy | % Previous stroke | AHI ≥5 | AHI≥10 | AHI≥20 | AHI≥30 | |
|---|---|---|---|---|---|---|---|---|---|---|
| Parra 2000 (Spain) | 161 (49.1) | 26.6 | 72 | – | – | 0 | 62 | 72 | 28 | |
| Iranzo 2002 (Spain) | 50 (40) | 26 | 67 | 68 | – | 0 | 62 | 46 | ||
| Turkington 2002 (UK) 38 | 120 (58) | 24 | 79 | 35.8 | – | 73.3 | 80 | 61 | 46 | 24 |
| Noradina 2006 (Malaysia) | 28 (29) | 23 | 60 | 78.6 | 3.6 | 10.7 | 92.8 | 78.5 | 37.7 | – |
| Dziewas 2008 (Germany) 39 | 55 (24) | 26 | 66 | 40.4 | – | – | – | 78 | – | – |
| Kepplinger 2013 (Germany) | 56 (64) | 27.1 | 66 | 88 | 7 | 36 | 91 | – | – | 29 |
| Camilo 2016 (Brazil) 40 | 69 (24.6) | 26.3 | 61 | 65.4 | 13 | – | 76.8 | 47.8 | ||
| Siarnik 2016 (Slovakia) 41 | 88 (36.4) | 28.5 | 65 | – | – | – | 70.5 | – | – | 29.6 |
| Domínguez-Mayoral 2021 (Spain)† | 72 (30.6) | 27.1 | 72 | 64.5 | 11.6 | 16.7 | 84.7 | 73.6 | 57 | 42 |
OSAHS, obstructive sleep apnea–hypopnea syndrome; BMI, body mass index; UK, United Kingdom; AHI, apnea–hypopnea index.
† Current study.
Discussion
In the present study, we found among the highest prevalence of undetected OSAHS (84.72%, AHI≥5) reported in Spain and Europe in patients with AIS. It can be presumed that if all patients with OSAHS were included (those already diagnosed, in addition to undetected cases), the OSAHS prevalence would be much higher. Recently, a meta-analysis examined rigorous studies on OSAHS prevalence among patients with stroke worldwide. In that review, the mean OSAHS prevalence (AHI ≥5) was 71% in AIS. 17 In Spain, the most important studies on this topic have been conducted in northern Spain. Parra and colleagues described a prevalence of severe OSAHS (AHI ≥30) of 27.7% versus 42% in our study population. 18 Previous studies 19 have reported that 46% of patients with stroke had an AHI ≥20 versus 57% in our study. Our results revealed a higher prevalence than those of the above studies, highlighting the importance of our findings in the Spanish Stroke Belt. Martínez et al. observed an OSAHS prevalence of 85.6% in Spain (AHI ≥5). 20 However, that study was not included in the previously mentioned meta-analysis 17 owing to methodological issues. From an international standpoint, the highest OSAHS prevalence (93%, AHI ≥5) was reported by Noradina and colleagues in Malaysia.21,22 The target population was Asian, so anatomical factors could partly explain their finding. In Europe, the highest reported OSAHS prevalence is 91% (AHI ≥5) in a small-sample study among patients with AIS and transient ischemic attack. 23 As seen in Table 3, the high OSAHS prevalence found in our study among patients with AIS might be related to the high incidence of vascular risk factors in the study region, such as hypertension, ischemic cardiomyopathy, and obesity. For example, a higher prevalence of obesity in Andalusia (37%) in comparison with the rest of Spain (26%) has been revealed in previous studies. 24 In fact, we observed a mean body mass index of 27.07 in our study population. These factors, together with the poor socioeconomic status (education and income), could explain the higher prevalence of vascular diseases and OSAHS in the Spanish Stroke Belt. 25 Our study population (median baseline NIHSS of 4 and mean age 70.46 years) is representative of the Stroke Belt in Spain, according to our recent published findings. 26 Advanced age is also a risk factor for OSAHS. 7 However, we found the highest prevalence of undetected OSAHS in Spain, even though other studies have included older patients. 18 This suggests that the aforementioned conditions in the Spanish Stroke Belt are the main determinants of our findings regarding OSAHS prevalence. At the epidemiological level, it is interesting to note that there is another region with a high prevalence vascular diseases, namely, the southern United States, which is known as the American Stroke Belt. 27 In that region, Hispanic populations, such as Mexican Americans and their descendants, have worse poststroke outcomes and a higher OSAHS prevalence than non-Hispanic Whites. 27 All these findings support the hypothesis that there could be an ethnic predisposition among Hispanic populations to increasingly worsening vascular diseases.
Our study also revealed a novel finding, namely, the significant relationship (p < 0.02) between the presence of facial palsy because of stroke and a higher AHI. One possible explanation could be that orofacial dysfunction owing to seventh nerve palsy can exacerbate latent OSAHS. Although pharyngeal muscles are not directly controlled by the seventh nerve, its dysfunction can indirectly affect orofacial function. In patients with facial palsy, contralesional handgrip strength and tongue–palate contact during swallowing and chewing are significantly impaired. In fact, facial palsy is a risk factor for poststroke dysphagia (a quantifiable symptom of orofacial impairment), which could exacerbate the collapse of the pharyngeal tract in patients with OSAHS. 28 Furthermore, facial palsy is a cause of leakage under continuous positive airway pressure (CPAP), thereby limiting the response to therapy. 29 Following this hypothesis, a rehabilitation program for this sequela has been suggested to improve OSAHS evolution. 30 The improvement in orofacial function to relieve obstruction during sleep might explain these findings. Hypoglossal palsy was not related to AHI in our study, probably owing to the low frequency of this sequela and the limited statistical power of our study. The hypoglossal nerve controls the genioglossus, which is the main upper airway dilator; therefore, the relationship between palsy of this nerve and the length of sleep apnea events or oxygen desaturation is important to study in the future.
We observed that obese patients and those with larger neck circumference had more severe OSAHS. This is not a new finding; 6 however, these associations have not been widely verified in patients with stroke 8 and they could be important in terms of developing OSAHS diagnostic indexes in the future.
Accumulated evidence indicates that stroke can worsen preexisting OSAHS via different mechanisms, such as damage to brainstem respiratory drive centers, hypoglossal nerve dysfunction, disturbed coordination of the upper airway, reduction in voluntary chest movement on the paralyzed side, and prolonged supine position. 30 In line with this, a slight decrease in AHI from the acute to chronic stroke phase has been described in some studies. 31 However, these differences do not change substantially over time, thereby ruling out such changes as a therapeutic approach.
Regarding CSAS, the low prevalence in our study population (1.38%) was similar to that in previous studies (1.4%). 32 There is some evidence suggesting that CSAS after stroke decreases from the acute phase compared with chronic stroke. 32 In cases of comorbid OSAHS and persistent central sleep apnea, CPAP titration is recommended. For these reasons, a second sleep study was not conducted for all patients in our study. Adequate polygraphy and polysomnography recordings were obtained in four of our patients.
Although in our study, polysomnography was conducted some months after polygraphy, the results were similar in the four patients who underwent both sleep studies. Diagnostic agreement between both sleep studies was not a primary objective of our study because the efficiency of polygraphy has been demonstrated previously.33,34 However, polysomnography remains the gold standard for OSAHS diagnosis, especially for sleep fragmentation caused by respiratory effort-related arousal. 35 Thus, in cases of a “normal” polygraph that does not reach the detection limit of OSAHS but with high clinical suspicion of OSAHS, polysomnography is needed for a correct diagnosis. 35 From the standpoint of diagnostic efficiency, most authors propose polygraphy as the initial screening and polysomnography as the gold standard test if confirmation is needed.33,34 In our study, patients with normal polygraphy had no OSAHS symptoms, so no further testing was necessary.
According to all the reasons above, OSAHS management does not appear to be influenced by the stroke phase, the type of sleep test, or the presence of comorbid central apneas. Recently, OSAHS screening for patients with ischemic stroke has been proposed in current guidelines. 36
Initiatives to carry out OSAHS screening in high-risk groups with a high prevalence, such as patients with stroke, have been successfully carried out. 37 Some studies highlight the important role of neuroscience nurses in OSAHS screening, 37 although more evidence is needed. In our opinion, the role of nurses at our Stroke Unit should be to conduct OSAHS screening as well as perform polygraphy. If OSAHS screening scores are based exclusively on patients’ responses, their sensitivity could be suboptimal. This might be related to the aforementioned low education rates, which could limit patients’ ability to express or explain their symptoms. Therefore, a good strategy would be the use of new OSAHS screening indexes on the basis of observation of patients’ sleep patterns to select those who should undergo a sleep study. Regarding sleep tests, polygraphy is the most efficient tool to verify a diagnosis of OSAHS. The feasibility of polygraphy (91.38%) has been verified in our study population.
The greatest strength of the present study is that we included a real-life population similar to that in our daily clinical practice. The use of a validated diagnostic tool like polygraphy confers robustness to our study methods. Of note, all tests were conducted by trained nurses and the recordings were reviewed by physicians specialized in sleep medicine to correct automatic scoring as needed and improve the sensitivity of the tests.
Our results should be interpreted in the context of the study design and there are some limitations. Patients with an OSAHS diagnosis before study enrollment were excluded; therefore the effect of preexisting sleep disorders cannot be explained by our findings. Benzodiazepine and opioid treatments were withdrawn after patient inclusion in the study, and the mean time from stroke onset to polygraphy was 35.9 hours. Therefore, interaction with the results for AHI could exist, but this would be minimal, taking into account the half-life of these drugs. The limited number of recording channels in polygraphy, which did not include electroencephalography to differentiate sleep from wakefulness, might lead to some level of underestimation of sleep apnea. Moreover, the OSAHS prevalence could be underestimated because some patients with respiratory effort-related arousals may have false negative results of polygraphy. Although polysomnography was not conducted for all patients in our study, the results can be expected to be similar. Given the relatively small sample size, our results might require further validation in other larger populations.
In conclusion, this was the first real-life study to investigate the prevalence of OSAHS among patients with stroke in southern Spain. Polygraphy is feasible among patients with AIS. The high prevalence of undetected OSAHS in the Spanish Stroke Belt (84.72% of patients with AIS) warrants further research into development of an OSAHS screening program among stable patients with stroke as an efficient strategy to identify those who should undergo a sleep study.
Acknowledgements
The authors wish to thank all staff who participated in the study for their invaluable help: María Ángeles Sánchez Armengol, Eloy Ferrete, José Sánchez-Miura, María Velarde, María Luz Jiménez, Manuel González, Teresa Reviriego, Manuela Bautista, Franscisco Álvarez, Asunción Herrero, Julia Franco, Myriam Montblanc, Myriam Cabezali, Carmen Albalá, Laura Ruiz, Montserrat Calado, Elena Martínez, Victoria Galiani, Leandro Gila, Teresa Rodríguez, Concepción Navarrete, Inmaculada García, Antonia Carrión, Amparo Núñez, Rocío Lara, Berta Gallardo, Asunción Herrero, Eva Zaya, Natalia Delgado, Rafael Gómez, Ana María Hérnandez, Josefa Jiménez, Carmen Piña, Rosa Muñoz, and Deborah Fernández.
Footnotes
Authors' contributions: DMA, SGJ, BJM, and MJ designed the study. DMA, SGJ, GP, FM, GC, and AM carried out data collection and data analysis and interpretation. DMA, FRN, and MJ drafted the manuscript. DMA, SGJ, GP, FM, GC, AM, FRN, BJM, PSS, GMA, DTR, BA, AP, RBL, and MJ. contributed to critical revision of the article. All authors approved the final version to be published.
Declaration of conflicting interest: The authors declare that there is no conflict of interest.
Funding: The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Neurovascular Research Group, part of the Cooperative Cerebrovascular Disease Research Network (INVICTUS+) (RD16/0019/0015).
ORCID iDs: Natalia Fouz-Rosón https://orcid.org/0000-0003-2725-7167
Soledad Pérez-Sánchez https://orcid.org/0000-0002-8940-9763
References
- 1.Montaner J, Vidal C, Molina C, et al. Selecting the target and the message for a stroke public education campaign: A local survey conducted by neurologists. Eur J Epidemiol 2001; 17: 581–586. [DOI] [PubMed] [Google Scholar]
- 2.Fulk GD, Boyne P, Hauger M, et al. The Impact of Sleep Disorders on Functional Recovery and Participation Following Stroke: A Systematic Review and Meta-Analysis. Neurorehabil Neural Repair 2020; 34: 1050–1061. [DOI] [PubMed] [Google Scholar]
- 3.Loke YK, Brown JWL, Kwok CS, et al. Association of Obstructive Sleep Apnea With Risk of Serious Cardiovascular Events. Circ Cardiovasc Qual Outcomes 2012; 5: 720–728. [DOI] [PubMed] [Google Scholar]
- 4.Arias MA, García-Río F, Alonso-Fernández A, et al. Sleep Apnea-Hypopnea Syndromes and Heart Failure. Rev Esp Cardiol 2007; 60: 415–427. [PubMed] [Google Scholar]
- 5.Lanfranchi P andSomers VK.. Obstructive sleep apnea and vascular disease. Respir Res 2001; 2: 315–319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Durán J, Esnaola S, Rubio R, et al. Obstructive sleep apnea-hypopnea and related clinical features in a population-based sample of subjects aged 30 to 70 yr. Am J Respir Crit Care Med 2001; 163: 685–689. [DOI] [PubMed] [Google Scholar]
- 7.Young T, Evans L, Finn L, et al. Estimation of the clinically diagnosed proportion of sleep apnea syndrome in middle-aged men and women. Sleep 1997; 20: 705–706. [DOI] [PubMed] [Google Scholar]
- 8.Casado-Naranjo I andSegura Martín T.. Breathing disorders during sleep and cerebrovascular disease. A presentation. Rev Neurol 2005; 41: S1. DOI: 10.33588/rn.41s03.2005419. [PubMed] [Google Scholar]
- 9.Cayuela A, Cayuela L, Rodríguez-Domínguez S, et al. Analysis of cerebrovascular disease mortality trends in Andalusia (1980–2014). Neurologia (Engl Ed) 2019; 34: 309–317. [DOI] [PubMed] [Google Scholar]
- 10.Banks JL andMarotta CA.. Outcomes Validity and Reliability of the Modified Rankin Scale: Implications for Stroke Clinical Trials. Stroke 2007; 38: 1091–1096. [DOI] [PubMed] [Google Scholar]
- 11.Von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Ann Intern Med 2007; 147: 573–577. [DOI] [PubMed] [Google Scholar]
- 12.Berry RB, Brooks R, Gamaldo C, et al. AASM scoring manual updates for 2017 (version 2.4). J Clin Sleep Med 2017; 13: 665–666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sateia MJ. International classification of sleep disorders-third edition highlights and modifications. Chest 2014; 146: 1387–1394. [DOI] [PubMed] [Google Scholar]
- 14.Alcázar Ramírez JD, Bujalance Zafra J, Campos Rodríguez F, et al. Documento de consenso sobre el síndrome de apneas-hipopneas del sueño en Andalucía. Rev Esp Pat Torac 2012; 24: 214–254. [Google Scholar]
- 15.Montaner J andÁlvarez-Sabín J. [ NIH stroke scale and its adaptation to Spanish]. Neurologia 2006; 21: 192–202. [PubMed] [Google Scholar]
- 16.Adams HP, Jr, Bendixen BH, Kappelle LJ, et al. Classification of Subtype of Acute Ischemic Stroke. Definitions for Use in a Multicenter Clinical Trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 1993; 24: 35–41. [DOI] [PubMed] [Google Scholar]
- 17.Seiler A, Camilo M, Korostovtseva L, et al. Prevalence of sleep-disordered breathing after stroke and TIA: A meta-analysis. Neurology 2019; 92: E648–E654. [DOI] [PubMed] [Google Scholar]
- 18.Parra O, Arboix A, Bechich S, et al. Time course of sleep-related breathing disorders in first-ever stroke or transient ischemic attack. Am J Respir Crit Care Med 2000; 161: 375–380. [DOI] [PubMed] [Google Scholar]
- 19.Iranzo A, Santamaría J, Berenguer J, et al. Prevalence and clinical importance of sleep apnea in the first night after cerebral infarction. Neurology 2002; 58: 911–916. [DOI] [PubMed] [Google Scholar]
- 20.Martínez García MA, Galiano Blancart R, Cabero Salt L, et al. Prevalence of Sleep-Disordered Breathing in Patients With Acute Ischemic Stroke: Influence of Onset Time of Stroke. Arch Bronconeumol 2004; 40: 196–202. [DOI] [PubMed] [Google Scholar]
- 21.Noradina AT, Hamidon BB, Roslan H, et al . Risk Factors for Developing Sleep-Disordered Breathing in Patients With Recent Ischaemic Stroke. Singapore Med J 2006; 47: 392–399. [PubMed] [Google Scholar]
- 22.Menon D, Sukumaran S, Varma R, et al. Impact of obstructive sleep apnea on neurological recovery after ischemic stroke: A prospective study. Acta Neurol Scand 2017; 136: 419–426. [DOI] [PubMed] [Google Scholar]
- 23.Kepplinger J, Barlinn K, Albright KC, et al. Early sleep apnea screening on a stroke unit is feasible in patients with acute cerebral ischemia. J Neurol 2013; 260: 1343–1350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Valdés S, García-Torres F, Maldonado-Araque C, et al. Prevalence of Obesity, Diabetes and Other Cardiovascular Risk Factors in Andalusia (Southern Spain). Comparison With National Prevalence Data. The Di@bet.es Study. Rev Esp Cardiol (Engl Ed) 2014; 67: 442–448. [DOI] [PubMed] [Google Scholar]
- 25.Castilla Guerra L Del Carmen Fernández-Moreno M andMarín-Martín J.. Factors explaining excess stroke prevalence: the Spanish stroke belt. Stroke 2010; 41: e114. [DOI] [PubMed] [Google Scholar]
- 26.Montaner J, Barragán-Prieto A, Pérez-Sánchez S, et al. Break in the Stroke Chain of Survival due to COVID-19. Stroke 2020; 51: 2307–2314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lisabeth LD, Sánchez BN, Chervin RD, et al. High prevalence of poststroke sleep-disordered breathing in Mexican Americans. Sleep Med 2017; 33: 97–102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schimmel M, Ono T, Lam O, et al. Oro-facial impairment in stroke patients. J Oral Rehabil 2017; 44: 313–326. [DOI] [PubMed] [Google Scholar]
- 29.Ye D, Chen C, Song D, et al. Oropharyngeal muscle exercise therapy improves signs and symptoms of post-stroke moderate obstructive sleep apnea syndrome. Front Neurol 2018; 9: 912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Parra O, Sánchez-Armengol Á, Bonnin M, et al. Early treatment of obstructive apnoea and stroke outcome: A randomised controlled trial. Eur Respir J 2011; 37: 1128–1136. [DOI] [PubMed] [Google Scholar]
- 31.Hasan F, Gordon C, Wu D, et al. Dynamic Prevalence of Sleep Disorders Following Stroke or Transient Ischemic Attack: Systematic Review and Meta-Analysis. Stroke 2021; 52: 655–663. [DOI] [PubMed] [Google Scholar]
- 32.Schütz SG, Lisabeth LD, Hsu CW, et al. Central sleep apnea is uncommon after stroke. Sleep Med 2021; 77: 304–306. DOI: 10.1016/j.sleep.2020.08.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Corral J, Sánchez-Quiroga MÁ, Carmona-Bernal C, et al. Conventional Polysomnography Is Not Necessary for the Management of Most Patients with Suspected Obstructive Sleep Apnea. Noninferiority, Randomized Controlled Trial. Am J Respir Crit Care Med 2017; 196: 1181–1190. [DOI] [PubMed] [Google Scholar]
- 34.Chai-Coetzer CL andMcEvoy RD.. The Debate Should Now Be Over: Simplified Cardiorespiratory Sleep Tests Are a Reliable, Cost-Saving Option for Diagnosing Obstructive Sleep Apnea. Am J Respir Crit Care Med 2017; 196: 1096–1098. [DOI] [PubMed] [Google Scholar]
- 35.Nerfeldt P Aoki F andFriberg D.. Polygraphy vs. polysomnography: missing OSAS in symptomatic snorers–a reminder for clinicians. Sleep Breath 2014; 18: 297–303. [DOI] [PubMed] [Google Scholar]
- 36.Kleindorfer DO, Towfighi A, Chaturvedi S, et al. 2021 Guideline for the Prevention of Stroke in Patients With Stroke and Transient Ischemic Attack: A Guideline From the American Heart Association/American Stroke Association. Stroke 2021; 52: E364–E467. [DOI] [PubMed] [Google Scholar]
- 37.Kuhen L, Woidtke R, Wynd C, et al. Systematic Identification and Management of Sleep Apnea (SA) in Hospitalized Patients: Implementation of an Interprofessional Care Guideline in a Community Hospital. Chest 2014; 146: 520A. [Google Scholar]
- 38.Turkington PM, Bamford J, Wanklyn P, Elliott MW. Prevalence and Predictors of Upper Airway Obstruction in the First 24 Hours After Acute Stroke. Stroke 2002; 33: 2037–2042. DOI: 10.1161/01.STR.0000023576.94311.27. [DOI] [PubMed] [Google Scholar]
- 39.Dziewas R, Hopmann B, Humpert M, et al. Positional sleep apnea in patients with ischemic stroke. Neurol Res 2008; 30: 645–648. DOI: 10.1179/174313208X289598. [DOI] [PubMed] [Google Scholar]
- 40.Camilo MR, Schnitman SV, Sander HH, et al. Sleep-disordered breathing among acute ischemic stroke patients in Brazil. Sleep Med 2016; 19: 8–12. DOI: 10.1016/j.sleep.2015.11.008. [DOI] [PubMed] [Google Scholar]
- 41.Šiarnik P, Kollár B, Čarnická Z, et al. Association of Sleep Disordered Breathing with Wake-Up Acute Ischemic Stroke: A Full Polysomnographic Study. J Clin Sleep Med 2016; 12: 549–554. DOI: 10.5664/jcsm.5688. [DOI] [PMC free article] [PubMed] [Google Scholar]