Abstract:
The novel coronavirus disease (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has rapidly evolved into a global pandemic. The substantial morbidity and mortality associated with the infection has prompted us to understand potential risk factors that can predict patient outcomes. Hypertension has been identified as the most prevalent cardiovascular comorbidity in patients infected with COVID-19 that demonstrably increases the risk of hospitalization and death. Initial studies implied that renin–angiotensin–aldosterone system inhibitors might increase the risk of viral infection and aggravate disease severity, thereby causing panic given the high global prevalence of hypertension. Nonetheless, subsequent evidence supported the administration of antihypertensive drugs and noted that they do not increase the severity of COVID-19 infection in patients with hypertension, rather may have a beneficial effect. To date, the precise mechanism by which hypertension predisposes to unfavorable outcomes in patients infected with COVID-19 remains unknown. In this mini review, we elaborate on the pathology of SARS-CoV-2 infection coexisting with hypertension and summarize potential mechanisms, focusing on the dual roles of angiotensin-converting enzyme 2 and the disorders of renin–angiotensin–aldosterone system in COVID-19 and hypertension. The effects of proinflammatory factors released because of immune response and gastrointestinal dysfunction in COVID-19 are also discussed.
Key Words: SARS-CoV-2, severe COVID-19, hypertension, angiotensin-converting enzyme 2, renin–angiotensin–aldosterone system, antihypertensive drugs
INTRODUCTION
Coronavirus disease (COVID-19), first discovered in Wuhan in December 2019, is an acute respiratory infectious disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).1 The resulting pandemic spread rapidly across China and the world2 and has affected over 200 countries and territories globally. As of March 22, 2021, the number of cases worldwide surpassed 120 million, including 2,711,071 deaths; the United States alone has an estimated 29,497,998 cases and 537,781 deaths.3 SARS-CoV-2 is highly transmissible among humans. Transmission routes include droplets, feces, and contact with surfaces on which the virus resides.4 Although a large proportion of patients with COVID-19 display mild-to-moderate illness, about 15% of older adults or those with chronic diseases may progress to severe pneumonia, develop acute respiratory distress syndrome (ARDS), septic shock, and/or multiple organ failure.5,6 Epidemiological studies have found that hypertension is the most frequent comorbidity in patients with COVID-19, and it has been identified as a major risk factor for the increased severity and mortality associated with COVID-19.7
The prevalence of hypertension in Chinese patients with COVID-19 was estimated to be between 15% and 25%.8 In the United States, meanwhile, 49.7% of COVID-19–hospitalized patients had hypertension.9 It was reported that a 2-fold to 3-fold higher prevalence of hypertension was a risk for progression to critical illness or even death in the population.10,11 Pranata et al demonstrated that hypertension increased the likelihood of bad outcomes, such as the severity of infection, ARDS, and mortality in patients infected with COVID-19.12
Presently, no specific drugs have been approved for the treatment of COVID-19. The current strategy for the management of COVID-19 involves symptomatic treatment alone, although drugs approved for other diseases, including antivirals, glucocorticoids, interferons, and traditional Chinese medicine, have been tested without much benefit.13 For patients with severe illness, the treatment focuses on controlling the infection, providing supportive care including ventilatory support if necessary, and treating COVID-19 sequelae and complications.13 Furthermore, the management of critically ill COVID-19 patients with hypertension is challenging. Previous animal studies suggested that renin–angiotensin–aldosterone system (RAAS) inhibitors increased angiotensin-converting enzyme 2 (ACE2) expression in the cardiac tissue,14 leading to concerns that hypertension might promote viral interaction with host cells and exacerbate COVID-19. Whether RAAS inhibitors can indeed adversely affect SARS-CoV-2 infection control or not has aroused great attention among clinicians and researchers alike.15 However, there is no doubt that hypertension increases the severity and mortality of COVID-19 and that a higher prevalence of hypertension is predictive of a worse prognosis in patients with COVID-19.7,16,17 We speculate that hypertension could play a potential causal role and that there is more than just a cause–effect association between these 2 diseases.
In this mini review, we highlight the pathogenesis of SARS-CoV-2 infection, summarize the potential mechanisms by which hypertension increases the severity of COVID-19, and review the effects of antihypertensive drugs, on the outcome of COVID-19 patients with pre-existing hypertension.
CLINICAL EVIDENCE OF THE EFFECT OF HYPERTENSION ON THE DEVELOPMENT OF SEVERE COVID-19
The first large-scale analysis of the data from 1590 laboratories found that hypertension was the most common comorbidity (16.9%), followed by diabetes (8.2%).7 Cox regression analysis adjusted for age and smoking status showed that hypertension was a significant risk factor for poor outcomes including admission to an intensive care unit, requirement for invasive ventilation, or death.7 Wu et al reported that the overall case-fatality rate was 2.3% (1023 deaths among 44,672 confirmed cases), and it was elevated in comorbid conditions—6.0% in patients with hypertension.16 Guan et al analyzed 1099 patients infected with COVID-19, including 173 patients with severe illness: The overall incidence of hypertension was 15% (165/1099), which rose to 23.7% (41/173) in patients with severe COVID-19 illness, and dropped to 13.4% (124/926) in those with nonsevere illness. Furthermore, severe COVID-19 patients with hypertension were more likely to reach adverse end points (35.8% vs. 13.7%).10 Wang et al conducted a single-center, retrospective study of 138 consecutive hospitalized patients with confirmed COVID-19 at the Zhongnan Hospital of Wuhan University in Wuhan, China.11 The results showed that the prevalence of hypertension was 31.2% among COVID-19 patients. Moreover, 58.3% of hypertensive patients were admitted to the intensive care unit, compared with 21.6% of normotensive patients.11 Furthermore, Leiva et al performed a multivariable logistic regression analysis based on the data from the above studies. Although hypertension was not included, the history of coronary artery disease, older age, elevated troponin I, a higher sequential organ failure assessment score, and D-dimer levels >1 μg/mL predicted COVID-19–related death.17 It is well known that hypertension and coronary disease usually accompany each other. Both COVID-19 case-fatality rate and the prevalence of hypertension increase with age. Long-term hypertension causes target organ damage, such as myocardial injury. On the other hand, COVID-19 can also exacerbate cardiac damage. This implies that the mechanisms by which hypertension increases the risk and severity of COVID-19 are complex and may be linked with comorbidities. However, an observational cohort study examining the association between blood pressure control and COVID-19 in 45,418 symptomatic patients with hypertension showed that better blood pressure control may be associated with worse COVID-19 outcomes, possibly because these patients have more advanced atherosclerosis.18 This finding is entirely different from previous views and may be an incidental finding. Thus, further studies should be performed to confirm this inverse relationship between blood pressure control and COVID-19–related deaths.18
THE PATHOGENESIS OF SARS-COV-2 INFECTION
There have been 2 coronavirus (CoV) pandemics before the current SARS-CoV-2 pandemic. One was the severe acute respiratory syndrome (SARS) pandemic in 2003 in Guangdong province, China,19 and the other was the Middle East respiratory syndrome (MERS) pandemic in Middle Eastern countries about 10 years later.20 SARS and MERS were caused by SARS-CoV and MERS-CoV, respectively. COVID-19, which broke out in Wuhan, Hubei province, China, was caused by SARS-CoV-2. These 3 CoVs are closely related to bat coronaviruses in species evolution, undergo interpersonal transmission, and cause pneumonia, ARDS, liver and kidney failures, and septic shock, resulting in significant mortality and morbidity.21 The genetic sequence alignment of the 3 CoVs showed that there was 79% shared identity between SARS-CoV and SARS-CoV-2 and 50% between SARS-CoV-2 and MERS-CoV.22 In addition, SARS-CoV-2 and SARS-CoV infect cells through the same cellular receptor, the zinc metallopeptidase ACE2 receptor, whereas MERS-CoV infects cells through the CD26 receptor.23,24 Compared with SARS-CoV, SARS-CoV-2 demonstrates a stronger binding to ACE2.
The entry of SARS-CoV-2 into cells is a complicated process that includes receptor binding and virus-cell fusion induced by proteolysis.21,25 CoV is composed of 4 structural proteins: spike (S), nucleocapsid (N), membrane (M), and envelope (E) proteins. SARS-CoV-2 binds its S protein to the ACE2 receptor in the S1 domain and mediates membrane fusion through the S2 subunit. After binding with ACE2, SARS-CoV-2 uses TMPRSS2, a proximal serine protease, to prime the spike protein and enter the cell through endocytosis. Viral RNA replicates in the cytosol and is further excreted by exocytosis to infect other cells.26 SARS-CoV-2–infected cells activate the innate and adaptive immune responses and release proinflammatory cytokines or chemokines, potentially leading to hyperinflammation or “cytokine storms.”27,28
POTENTIAL MECHANISMS OF THE EFFECT OF HYPERTENSION ON THE INCREASED SEVERITY OF COVID-19
It is well recognized that hypertension increases the severity of SARS-CoV-2–infected individuals. Hypertension is a high-risk factor for cardiovascular disease and affects millions of patients globally.29 Recent findings demonstrate that hypertension plays an important role in the regulation of RAAS, inflammation, immune responses, and the gastrointestinal tract, which partly explains the worse outcomes in COVID-19 patients. Thus, the coexistence of hypertension and SARS-CoV-2 infection can be a double blow to patients.
ACE2 and RAAS
ACE2 has been demonstrated to be a SARS-CoV-2 entry receptor. ACE2, the sole known human homolog of ACE, was first discovered in 2000, mainly in the heart, kidney, and testis. Subsequent studies showed that it exists in the lungs, blood vessels, small intestine, and brain.30 Current evidence shows that the basal ACE2 mRNA level in the respiratory system is lower than that in the other organs.31 How low ACE2 expression allows the infection and replication of SARS-CoV-2 requires further investigation. Notably, on one hand, the infection is not limited to ACE2-positive cells. Bioinformatics analysis based on human-virus protein interactions showed that human dipeptidyl peptidase 4 and the spike receptor–binding domain of SARS-CoV-2 exhibited high affinity for each other, suggesting that SARS-CoV-2 may use dipeptidyl peptidase 4 as a coreceptor to enter host cells.32 Another potential coreceptor, NRP1, has also been discovered. In vitro studies demonstrated that knockdown of NRP1 reduced the instance of SARS-CoV-2 infection and incubation of patient-derived SARS-CoV-2 with monoclonal anti-NRP1 decreased the infection efficiency of cells expressing ACE2.33 Other coreceptors that assist SARS-CoV-2 infecting cells, such as CD147 and GRP78, have been investigated.34 On the other hand, the endocytosis of SARS-CoV-2 along with ACE2 occurs after SARS-CoV-2 binds to the membrane receptor of ACE2.35 Thus, the virus replicates and is transmitted between cells.
ACE2 exerts multiple physiological and pathological effects. ACE2 serves not only as a cellular receptor of SARS-CoV-2 infection but also as a master regulator of RAAS. Angiotensin II (Ang II), a vasoactive peptide with vasoconstrictive and inflammatory properties, is regarded as a potent hypertensive hormone. As shown in Figure 1, the ACE/Ang II/Ang II type 1 receptor (AT1R) axis plays a positive role in regulating RAAS.36 ACE converts Ang I into Ang II, which stimulates the release of aldosterone and increases blood pressure. Ang II also activates AT1R and induces vasoconstriction. By contrast, ACE2 counteracts the action of ACE. The ACE2/Ang (1-7)/AT2R axis negatively regulates RAAS.37 ACE2 metabolizes Ang I and Ang II into Ang (1-9) and Ang (1-7), respectively. Both Ang (1-7) and Ang (1-9) bind and activate the Ang II type 2 receptor (AT2R), causing vasodilation and decreasing blood pressure. In addition, Ang (1-7) binds to the MAS receptor and plays protective roles in a variety of human target organs, by reducing cardiac hypertrophy and pathological cardiac remodeling, preventing the occurrence of heart failure after myocardial infarction,38,39 reducing lung tissue damage and inflammation, and avoiding severe acute lung failure.40
FIGURE 1.
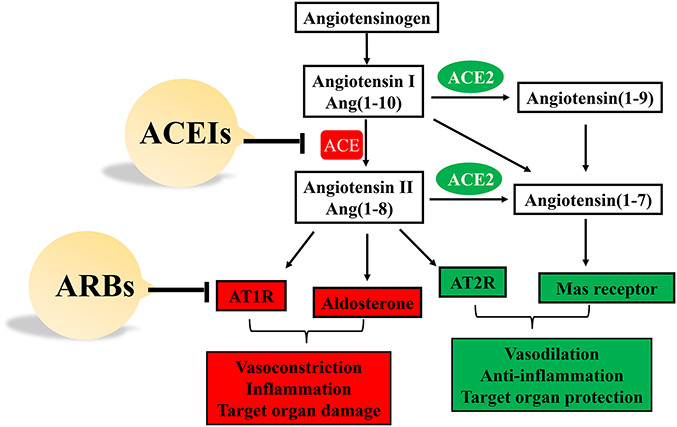
Role of ACE and ACE2 in the RAAS system. ACEIs and ARBs target RAAS to reduce blood pressure and exert protection on organs.
The ACE/Ang II/AT1R and ACE2/Ang (1-7)/AT2R axis are coexpressed in some tissues, including the lung, kidney, and heart.41 The balance between them is important for maintaining normal physiological functions. However, ACE/Ang II/AT1R activation or ACE2/Ang (1-7)/AT2R deactivation results in target organ damage. ACE inhibitors (ACEIs) and angiotensin II receptor blockers (ARBs) are antihypertensive drugs frequently used in clinical practice. ACEIs reduce the generation of Ang II by inhibiting ACE, whereas ARBs reduce blood pressure by blocking the binding of Ang II with AT1R (Fig. 1). Interestingly, both upregulate ACE2 levels.42 Considering the dual roles of ACE2, the question of whether ACEIs/ARBs affect the prognosis of hypertensive COVID-19 patients has become a hot debate. On the one hand, RAAS inhibitors have been demonstrated to play protective functions in heart failure, kidney impairment, and acute lung injury.37 By contrast, augmented ACE2 levels may increase the receptors for SARS-CoV-2 infection in the lungs and heart.43 Three studies using correlation analyses implied that ACEIs/ARBs did not increase the risk and mortality of COVID-19.15,44,45 Subsequently, consistent information was obtained from further observational studies.46,47 Thus, there is insufficient evidence that RAAS inhibitors should be discontinued for the treatment of hypertension with COVID-19. Therefore, as suggested by the European Society of Cardiology and other medical associations, it is advisable not to change the treatment regimen of ACEIs/ARBs for patients with hypertension during the COVID-19 pandemic, unless supported by definitive clinical evidence (Lopes R. BRACE CORONA: continuing vs suspending ACE inhibitors and ARBs in COVID-19. Oral presentation. European Society of Cardiology Congress 2020; Online; September 1, 2020).
Inflammation and Immune Activation
SARS-CoV-2 infection activates both innate and adaptive immune responses, triggers release of proinflammatory factors, and results in hyperinflammation or “cytokine storms.” Hadjadj et al found that the activity of type-I interferon (IFN), which is crucial for protection against viral infection, was impaired because of a significant reduction in IFN-stimulated genes in severe COVID-19 patients, and the inflammatory responses were accordingly exacerbated.48 Compared with mild COVID-19 disease, severe illness causes lymphopenia with a fall in immune cells such as CD4+ T cells, CD8+ T cells, natural killer cells, and B cells.49,50 Indeed, lymphocytopenia is negatively correlated with the severity of SARS-CoV-2 infection.51 The reduced functional diversity of immune cells induces proinflammatory cytokines. Most patients with severe COVID-19 show sharply elevated serum levels of cytokines, including interleukin-1 beta (IL-1β), IL-6, IL-17, and tumor necrosis factor–alpha, a phenomenon termed the “cytokine storm.”52 Thus, uncontrolled innate response and impaired adaptive immune response result in shock, tissue damage, or multiorgan failure. However, hypertension also activates the innate and adaptive immune systems, leading to cytokine release and enhanced inflammation.53,54 A growing body of evidence has shown that immune cell infiltration plays an important role in both blood pressure elevation and end-organ damage. Wenzel et al observed that Ang II increased the number of macrophages and induced them to infiltrate the vascular wall.55 Selective ablation of lysozyme M-positive monocytes reduces circulating monocytes and limits their infiltration. These results suggest that infiltrating monocytes are crucial for Ang II–induced arterial hypertension.56 Animal hypertension models have demonstrated that inflammation and immune cells accumulate in the kidney, arteries, and central nervous system.57 Cytokines released by immune cells promote vascular dysfunction and organ damage. In hypertensive patients, the amounts of circulating monocytes, macrophages, CD8+ T cells, and CD4+ T cells are increased in the inflammatory environment.58 High blood pressure promotes an acute cardiac inflammatory response and induces immune cell infiltration and activation in the myocardium. Ma et al demonstrated that CD8+ T cells initiated an immune response to high blood pressure, secreted IFN-γ, and activated macrophage infiltration in the myocardium.59 Although pathological reports of COVID-19 cases did not detect viral infection or replication in cardiac tissues,6 cardiac inflammation was observed. Thus, hypertension may drive cardiac or other organ damage in patients with severe COVID-19 by exacerbating inflammation or immune responses.
Trump et al assessed whether the proinflammatory status of hypertensive patients could exacerbate COVID-19 severity. Data based on clinical characteristics (n = 144) and single-cell RNA sequencing transcriptome analyses on nasopharyngeal swabs (n = 48) demonstrated that COVID-19 patients with hypertension exhibited obvious inflammation in immune cells, which correlated with COVID-19 progression. Furthermore, the effect of antihypertensive drugs on COVID-19 severity was evaluated. ACEI treatment dampened hyperinflammation and increased intrinsic antiviral responses of the cell, whereas ARB-treated patients exhibited an exaggerated hyperinflammatory response and reduced antiviral response. The study suggested that ACEI may be more favorable for hypertensive patients during COVID-19.60
Immune dysregulation plays an important role in both SARS-CoV-2 infection and hypertension. Zeng et al analyzed the dynamic immunological characteristics of 51 COVID-19 patients with hypertension.61 T-cell lymphopenia in COVID-19 cases with hypertension was associated with COVID-19 severity and mortality. Furthermore, patients with fatal outcomes exhibited high CD38+ and HLA-DR+, both key markers of CD8+ T-cell activation, as well as PD-1+, an activation and exhaustion marker of CD8+ T cells, on CD8+ T cells during SARS-CoV-2 infection. The proportions of SARS-CoV-2-specific IFNγ+ CD8+ cells, IFNγ+ CD4+ T cells, and immunoglobulins such as immunoglobulin G, immunoglobulin M, and immunoglobulin A were significantly lower in fatal cases. However, a high SARS-CoV-2 viral load was persistent for 4 weeks in fatal cases. These findings suggest that T cells are critical for clinical outcomes in hypertensive patients with COVID-19.61
In short, the activated innate immune response and chronic inflammation in hypertensive patients weaken their initial immunity to fight SARS-CoV-2 infection. Uncontrolled viral replication induces an adaptive immune response, which is compounded in patients with hypertension, thereby releasing large amounts of cytokines that reach all the organs by circulation. Thus, it is critically important to fight viral infections and balance immune responses. Although the viral load in patients with severe illness is low, overactivated immune response damages organs; moderate immune suppression is therefore needed. As for hypertensive patients, improving immunity and controlling chronic inflammation may be helpful in fighting the virus.
Gut Dysfunction
ACE2 is also expressed in the gastrointestinal tract, which may be another site of SARS-CoV-2 infection.62 Based on the clinical features of COVID-19 patients, gastrointestinal discomfort appears early and viral RNA can be detected in feces.63 ACE2 sustains intestinal nutritional homeostasis, and the deficiency of ACE2 in murine models increases epithelial damage–induced intestinal inflammation.64 These studies suggest that ACE2 serves as a key regulator of gut microbial ecology and that the intestine is a secondary site of infection. In addition, comorbidities such as hypertension and diabetes exert adverse effects on the gut microbiome,65 which may be worsened by SARS-CoV-2 infection. An imbalance of gut microbiota was detected in spontaneously hypertensive and Ang II–infused rats, as well as in hypertensive patients.66 A reduction in short-chain fatty acids such as butyrate and acetate that possess anti-inflammatory properties and an increase in gut leakiness and wall pathology were noted, which may aggravate SARS-CoV-2 infection. COVID-19 shares many common pathophysiological features with hypertension, such as inflammation, gut dysfunction, and decreased availability of butyrate-producing bacteria.67 Butyrate is a histone deacetylase inhibitor that maintains the acetylation of histones, thereby aiding chromatin organization and gene expression. Li et al studied the role of butyrate in gut epithelial organoids.68 The results of high-throughput RNA sequencing showed that ACE2 and TMPRSS2, which facilitate SARS-CoV-2 entry into host cells, were both significantly downregulated. In addition, high-mobility group protein-1, which is critical for SARS-CoV-2 replication after entry, decreased with butyrate treatment. On the other hand, butyrate upregulated the toll-like receptor signaling pathway to enhance the innate immune system's attack on viruses and elevate interleukin-1-beta and interferon regulatory factor-7 levels. These results imply that the increased severity of COVID-19 in hypertension may be due to the cumulative depletion of favorable bacteria in the gut.68 There is also evidence that gastrointestinal SARS-CoV-2 might destroy the gut–blood barrier, affecting the host's response to inflammation and resulting in multiorgan dysfunction and septic shock.10 Thus, for hypertensive COVID-19 patients, the double blow of gastrointestinal dysfunction and inflammation induced by SARS-CoV-2 infection and high blood pressure should be considered (as shown in Fig. 2). Further investigations are required to explore the effects of the interactions between COVID-19 infection and hypertension on the gastrointestinal tract.
FIGURE 2.
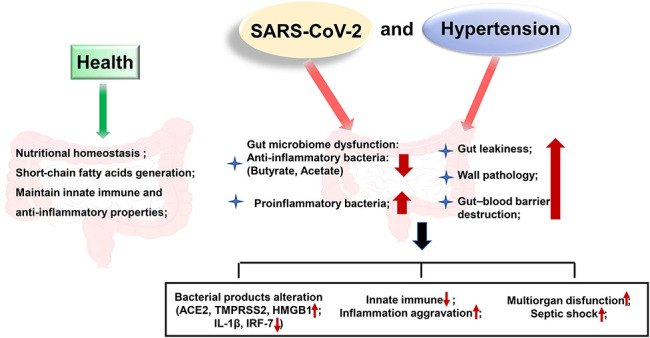
Effects of the interactions between SARS-CoV-2 infection and hypertension on the gastrointestinal tract. Gastrointestinal tract is another site of SARS-CoV-2 infection. In healthy individuals, ACE2 sustains nutritional homeostasis. The generation of short-chain fatty acids maintains innate immune and anti-inflammatory properties. However, hypertension induces gut microbiome dysfunction, leading to gut leakiness and wall pathology that aggravates SARS-CoV-2 infection. SARS-CoV-2 infection, in turn, worsens the gastrointestinal tract.
SARS-CoV-2 is undoubtedly a highly infectious virus which has caused dramatically bad effects on society. The management of this pandemic is challenging. To date, various models of artificial intelligence69–71 are widely used to screen, diagnose, and predict outcomes of COVID-19, providing assistances for the effective control of COVID-19 and saving lives. Thus, applying artificial intelligence to predict the risk of COVID-19 and the severity or mortality in hypertensive patients deserves further development.
EVALUATION OF ANTIHYPERTENSIVE DRUGS ON THE OUTCOMES OF COVID-19 PATIENTS WITH HYPERTENSION
Preliminary data have shown that RAAS inhibitors upregulate ACE2 receptor expression in animal models, raising concerns about whether these drugs increase the risk of SARS-CoV-2 infection and result in poor clinical prognosis. Recent evidence suggests a negative association between the administration of RAAS blockers and the risk of COVID-19. Jiang et al examined whether hypertension or RAS suppression could regulate ACE2 levels.72 The results showed that ACE2 expression was increased in both the lungs and kidneys of older adults. Neither hypertension nor antihypertensive treatment altered ACE2 expression in the human kidney. Kidney ACE2 is most likely nephroprotective in patients without SARS-CoV-2 infection.72 Li et al estimated whether RAAS inhibitors amplified the severity or risk of mortality in hospitalized COVID-19 patients with hypertension.15 The data showed that ACEIs/ARBs treatment increased neither the severity nor mortality of COVID-19–infected patients. A single-center retrospective study indicated that the rates of occurrence of critical illness and death did not differ significantly between ACEIs/ARBs and non-ACEIs/ARBs groups of hypertensive patients with COVID-19. Rather, ACEIs/ARBs significantly decreased the concentrations of inflammatory markers such as high-sensitivity C-reactive protein and procalcitonin.45 A nationwide study investigating the clinical outcomes of COVID-19 after the use of ACEI/ARB in patients with hypertension in South Korea confirmed this association and supported the continuation of ACEI/ARB use.73 In addition, ACEIs/ARBs increased CD8+ T-cell counts and decreased the peak viral load,74 supporting the continued use of RAAS inhibitors in patients with COVID-19. A mixed-effect Cox proportional hazards model adjusted for age, sex, comorbidities, and in-hospital medications in a multicenter retrospective study showed that ACEIs/ARBs decreased the all-cause mortality in patients compared with those who were not on ACEIs/ARBs. Further analysis consistently showed that ACEIs/ARBs decreased the mortality in COVID-19 patients with hypertension.44
Recently, Reynolds et al assessed the outcomes of COVID-19 in patients with hypertension previously treated with ACE inhibitors, angiotensin-receptor inhibitors, beta-blockers, calcium-channel blockers (CCBs), and thiazide diuretics.46 The results demonstrated no significant correlation between any medication class and the increased likelihood of infection or progression. Interestingly, a slightly more severe illness related to the previous use of calcium-channel blockers was found, whereas a modestly lower infection risk for COVID-19 was observed among patients on beta-blockers.46 A cohort study of 2 million hypertensive patients examined whether the COVID-19 risk differed among patients treated with ACEIs, ARBs, or CCBs. The subjects included in the study were limited to patients with uncomplicated hypertension. The use of ACEIs and ARBs was associated with a lower risk of COVID-19–related hospitalization than the use of CCBs [hazard ratio, 0.74 (95% confidence interval, 0.65–0.83) and 0.84 (0.76–0.93), respectively] and a lower risk of intubation/death. Moreover, risks were slightly lower for ACEI users than for ARB users.75
Therefore, clinical studies (Table 1) support the hypothesis that the administration of antihypertensive drugs does not enhance the severity of COVID-19 in patients with hypertension. Rather, RAAS inhibitors may benefit these patients.
TABLE 1.
Studies Investigating the Association Between ACEIs/ARBs and Severity or Mortality of Illness in Patients With Hypertension for COVID-19 Infection
| Study (References) | COVID-19 Patients With Hypertension, Taking ACEI/ARBs | Study Design | Taking ACEI/ARBs on the Severity or Mortality in COVID-19 Patients With Hypertension |
| Li et al15 | 362, 115 | Retrospective, single-center | Not associated with the severity or mortality of COVID-19 |
| Zhang et al44 | 1128, 188 | Retrospective, multicenter | Lower risk of all-cause mortality |
| Yang et al45 | 126, 43 | Retrospective, single-center | Lower critical patients and death rate |
| Reynolds et al46 | 2573, 1019 | Observational analysis | No association |
| Kim et al70 | 1290, 682 | Nationwide study | No association |
| Meng et al71 | 51, 17 | Retrospective, single-center | Lower rate of severity |
| Semenzato et al74 | 2338, 1779 | Cohort study | Lower risk of hospitalization and intubation/death |
CONCLUSIONS
Hypertension is strongly associated with elevated mortality in patients with COVID-19. Dysregulation of the RAAS, immune response, gastrointestinal tract, and inflammation may increase the severity of COVID-19 (Fig. 3). However, current clinical studies suggest that the administration of antihypertensive drugs is not associated with the outcomes of COVID-19 in patients with hypertension. By contrast, ACEIs/ARBs even attenuate mortality in some cases. Thus, specific mechanisms that enable an understanding of how hypertension accelerates the pathogenesis of COVID-19 and how ACEIs/ARBs affect the outcomes of COVID-19 patients with hypertension deserve further investigation. Improving autoimmunity and controlling chronic inflammation are critically important for patients with hypertension to win the fight against the SARS-CoV-2 virus.
FIGURE 3.
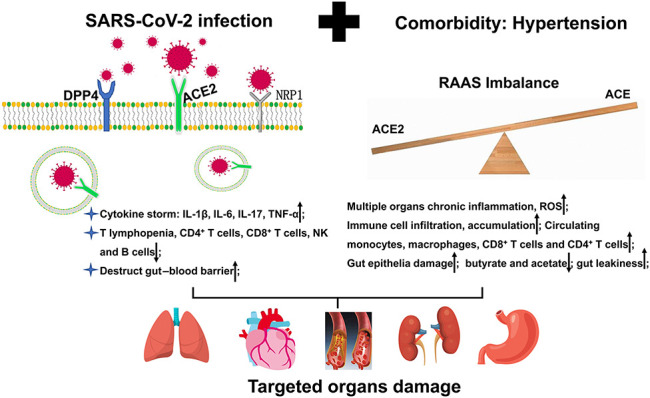
Hypertension exacerbates the severity of SARS-CoV-2-infected individuals, mainly through increasing inflammation, inducing excessive immune response, and disrupting gastrointestinal flora. Both hypertension and SARS-CoV-2 eventually do damages to multiple targeted organs.
In summary, this review emphasized the following points in the COVID-19 clinical setting. First, hypertension is the most common comorbidity. Second, hypertension increases the severity and mortality. Third, taking antihypertensive drugs such as ACEI and ARBs is not associated with the severity or mortality of patients with hypertension. By contrast, it brings benefits to the outcomes of COVID-19 patients with pre-existing hypertension.
ACKNOWLEDGMENTS
Authors thank Dr Xiaoping Yang, the director of Key Laboratory of Study and Discovery of Small Targeted Molecules of Hunan Province, for the review and modification of this article.
Footnotes
The authors report no conflicts of interest.
Concept and design: S. Liu and Z. Gong; Literatures review: M. Peng, J. He and Y. Xue; Writing manuscript: M. Peng and X. Yang.
Contributor Information
Jia He, Email: joeyhe92@126.com.
Ying Xue, Email: xueying091@126.com.
Xue Yang, Email: xyyangxue2016@163.com.
Shao Liu, Email: liushao999@csu.edu.cn.
Zhicheng Gong, Email: gongzhicheng@csu.edu.cn.
REFERENCES
- 1.Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hui DS, I Azhar E, Madani TA, et al. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health—the latest 2019 novel coronavirus outbreak in Wuhan, China. Int J Infect Dis. 2020;91:264–266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.World Health Organization. Coronavirus Disease (COVID-2019) Situation Reports [EB/OL]. Available at: https://covid19.who.int/. Accessed March 22, 2021. [Google Scholar]
- 4.Wiersinga WJ, Rhodes A, Cheng AC, et al. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;324:782–793. [DOI] [PubMed] [Google Scholar]
- 5.Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020;395:497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420–422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Guan WJ, Liang WH, Zhao Y, et al. Comorbidity and its impact on 1590 patients with covid-19 in China: a nationwide analysis. Eur Respir J. 2020;55:2000547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hu Y, Sun J, Dai Z, et al. Prevalence and severity of corona virus disease 2019 (COVID-19): a systematic review and meta-analysis. J Clin Virol. 2020;127:104371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Garg S, Kim L, Whitaker M, et al. Hospitalization rates and characteristics of patients hospitalized with laboratory-confirmed coronavirus disease 2019—COVID-NET, 14 states, March 1–30, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:458–464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;58:711–712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pranata R, Lim MA, Huang I, et al. Hypertension is associated with increased mortality and severity of disease in COVID-19 pneumonia: a systematic review, meta-analysis and meta-regression. J Renin Angiotensin Aldosterone Syst. 2020;21:1470320320926899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pascarella G, Strumia A, Piliego C, et al. COVID-19 diagnosis and management: a comprehensive review. J Intern Med. 2020;288:192–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Igase M, Strawn WB, Gallagher PE, et al. Angiotensin II AT1 receptors regulate ACE2 and angiotensin-(1-7) expression in the aorta of spontaneously hypertensive rats. Am J Physiol Heart Circ Physiol. 2005;289:H1013–H1019. [DOI] [PubMed] [Google Scholar]
- 15.Li J, Wang X, Chen J, et al. Association of renin-angiotensin system inhibitors with severity or risk of death in patients with hypertension hospitalized for coronavirus disease 2019 (COVID-19) infection in Wuhan, China. JAMA Cardiol. 2020;5:825–830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72,314 cases from the Chinese center for disease control and prevention. JAMA. 2020;323:1239–1242. [DOI] [PubMed] [Google Scholar]
- 17.Leiva Sisnieguez CE, Espeche WG, Salazar MR, et al. Arterial hypertension and the risk of severity and mortality of COVID-19. Eur Respir J. 2020;55:2001148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sheppard JP, Nicholson BD, Lee J, et al. Association between blood pressure control and coronavirus disease 2019 outcomes in 45 418 symptomatic patients with hypertension: an observational cohort study. Hypertension. 2021;77:846–855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liang G, Chen Q, Xu J, et al. Laboratory diagnosis of four recent sporadic cases of community-acquired SARS, Guangdong Province, China. Emerg Infect Dis. 2004;10:1774–1781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Khan MU, Shah S, Ahmad A, et al. Knowledge and attitude of healthcare workers about Middle East Respiratory Syndrome in multispecialty hospitals of Qassim, Saudi Arabia. BMC Public Health. 2014;14:1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rockx B, Kuiken T, Herfst S, et al. Comparative pathogenesis of COVID-19, MERS, and SARS in a nonhuman primate model. Science. 2020;368:1012–1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Andersen KG, Rambaut A, Lipkin WI, et al. The proximal origin of SARS-CoV-2. Nat Med. 2020;26:450–452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li W, Moore MJ, Vasilieva N, et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426:450–454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Raj VS, Mou H, Smits SL, et al. Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus-EMC. Nature. 2013;495:251–254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Walls AC, Park YJ, Tortorici MA, et al. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;181:281.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cao X. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol. 2020;20:269–270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kronbichler A, Effenberger M, Eisenhut M, et al. Seven recommendations to rescue the patients and reduce the mortality from COVID-19 infection: an immunological point of view. Autoimmun Rev. 2020;19:102570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mills KT, Stefanescu A, He J. The global epidemiology of hypertension. Nat Rev Nephrol. 2020;16:223–237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Harmer D, Gilbert M, Borman R, et al. Quantitative mRNA expression profiling of ACE 2, a novel homologue of angiotensin converting enzyme. FEBS Lett. 2002;532:107–110. [DOI] [PubMed] [Google Scholar]
- 31.Hikmet F, Méar L, Edvinsson Å, et al. The protein expression profile of ACE2 in human tissues. Mol Syst Biol. 2020;16:e9610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vankadari N, Wilce JA. Emerging WuHan (COVID-19) coronavirus: glycan shield and structure prediction of spike glycoprotein and its interaction with human CD26. Emerg Microbes Infect. 2020;9:601–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cantuti-Castelvetri L, Ojha R, Pedro LD, et al. Neuropilin-1 facilitates SARS-CoV-2 cell entry and provides a possible pathway into the central nervous system. bioRxiv. 2020;370:856–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zamorano Cuervo N, Grandvaux N. ACE2: evidence of role as entry receptor for SARS-CoV-2 and implications in comorbidities. Elife. 2020;9:e61390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rahman MM, Hasan M, Ahmed A. Potential detrimental role of soluble ACE2 in severe COVID-19 comorbid patients. Rev Med Virol. 2021. doi: 10.1002/rmv.2213. [epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vasudeva K, Balyan R, Munshi A. ACE-triggered hypertension incites stroke: genetic, molecular, and therapeutic aspects. Neuromolecular Med. 2020;22:194–209. [DOI] [PubMed] [Google Scholar]
- 37.Patel VB, Zhong JC, Grant MB, et al. Role of the ACE2/angiotensin 1-7 axis of the renin-angiotensin system in heart failure. Circ Res. 2016;118:1313–1326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gironacci MM, Cerniello FM, Longo Carbajosa NA, et al. Protective axis of the renin-angiotensin system in the brain. Clin Sci (Lond). 2014;127:295–306. [DOI] [PubMed] [Google Scholar]
- 39.Etelvino GM, Peluso AA, Santos RA. New components of the renin-angiotensin system: alamandine and the MAS-related G protein-coupled receptor D. Curr Hypertens Rep. 2014;16:433. [DOI] [PubMed] [Google Scholar]
- 40.Santos RA, Simões E Silva AC, Maric C, et al. Angiotensin-(1-7) is an endogenous ligand for the G protein-coupled receptor Mas. Proc Natl Acad Sci U S A. 2003;100:8258–8263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rivière G, Michaud A, Breton C, et al. Angiotensin-converting enzyme 2 (ACE2) and ACE activities display tissue-specific sensitivity to undernutrition-programmed hypertension in the adult rat. Hypertension. 2005;46:1169–1174. [DOI] [PubMed] [Google Scholar]
- 42.Keidar S, Gamliel-Lazarovich A, Kaplan M, et al. Mineralocorticoid receptor blocker increases angiotensin-converting enzyme 2 activity in congestive heart failure patients. Circ Res. 2005;97:946–953. [DOI] [PubMed] [Google Scholar]
- 43.Hanff TC, Harhay MO, Brown TS, et al. Is there an association between COVID-19 mortality and the renin-angiotensin system—a call for epidemiologic investigations. Clin Infect Dis. 2020;71:870–874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhang P, Zhu L, Cai J, et al. Association of inpatient use of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers with mortality among patients with hypertension hospitalized with COVID-19. Circ Res. 2020;126:1671–1681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yang G, Tan Z, Zhou L, et al. Effects of angiotensin II receptor blockers and ACE (angiotensin-converting enzyme) inhibitors on virus infection, inflammatory status, and clinical outcomes in patients with COVID-19 and hypertension: a single-center retrospective study. Hypertension. 2020;76:51–58. [DOI] [PubMed] [Google Scholar]
- 46.Reynolds H, Adhikari S, Pulgarin C, et al. Renin–angiotensin–aldosterone system inhibitors and risk of covid-19. N Engl J Med. 2020;382:2441–2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mancia G, Rea F, Ludergnani M, et al. Renin–angiotensin–aldosterone system inhibitors and risk of covid-19. N Engl J Med. 2020;382:2431–2440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hadjadj J, Yatim N, Barnabei L, et al. Impaired type I interferon activity and exacerbated inflammatory responses in severe Covid-19 patients. MedRxiv. 2020;369:718–724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Qin C, Zhou L, Hu Z, et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin Infect Dis. 2020;71:762–768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Shi Y, Tan M, Chen X, et al. Immunopathological characteristics of coronavirus disease 2019 cases in Guangzhou, China. Immunology. 2020;160:261–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhang B, Zhou X, Zhu C, et al. Immune phenotyping based on neutrophil- to-lymphocyte ratio and IgG predicts disease severity and outcome for patients with COVID-19. Front Mol Biosci. 2020;7:157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tamimi F, Abusamak M, Akkanti B, et al. The case for chronotherapy in COVID-19 induced acute respiratory distress syndrome (ARDS). Br J Pharmacol. 2020;177:4845–4850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rucker AJ, Rudemiller NP, Crowley SD. Salt, hypertension, and immunity. Annu Rev Physiol. 2018;80:283–307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Rodríguez-Iturbe B, Pons H, Quiroz Y, et al. Autoimmunity in the pathogenesis of hypertension. Nat Rev Nephrol. 2014;10:56–62. [DOI] [PubMed] [Google Scholar]
- 55.Wenzel P, Knorr M, Kossmann S, et al. Lysozyme M-positive monocytes mediate angiotensin II-induced arterial hypertension and vascular dysfunction. Circulation. 2011;124:1370–1381. [DOI] [PubMed] [Google Scholar]
- 56.Svendsen UG. Evidence for an initial, thymus independent and a chronic, thymus dependent phase of DOCA and salt hypertension in mice. Acta Pathol Microbiol Scand A. 1976;84:523–528. [DOI] [PubMed] [Google Scholar]
- 57.Li Y, Wei B, Liu X, et al. Microglia, autonomic nervous system, immunity and hypertension: is there a link? Pharmacol Res. 2020;155:104451. [DOI] [PubMed] [Google Scholar]
- 58.Norlander AE, Madhur MS, Harrison DG. The immunology of hypertension. J Exp Med. 2018;215:21–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ma F, Feng J, Zhang C, et al. The requirement of CD8+ T cells to initiate and augment acute cardiac inflammatory response to high blood pressure. J Immunol. 2014;192:3365–3373. [DOI] [PubMed] [Google Scholar]
- 60.Trump S, Lukassen S, Anker MS, et al. Hypertension delays viral clearance and exacerbates airway hyperinflammation in patients with COVID-19. Nat Biotechnol. 2020;39:705–716. [DOI] [PubMed] [Google Scholar]
- 61.Zeng Q, Li YZ, Dong SY, et al. Dynamic SARS-CoV-2-specific immunity in critically ill patients with hypertension. Front Immunol. 2020;11:596684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zuo T, Zhang F, Lui GC, et al. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology 2020;159:944–955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wu Y, Guo C, Tang L, et al. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol Hepatol. 2020;19:434–435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hashimoto T, Perlot T, Rehman A, et al. Ace2 links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature. 2012;487:477–481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Woting A, Blaut M. The intestinal microbiota in metabolic disease. Nutrients. 2016;8:202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yang T, Santisteban MM, Rodriguez V, et al. Gut dysbiosis is linked to hypertension. Hypertension. 2015;65:1331–1340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Sharma RK, Stevens BR, Obukhov AG, et al. ACE2 (angiotensin-converting enzyme 2) in cardiopulmonary diseases: ramifications for the control of SARS-CoV-2. Hypertension. 2020;76:651–661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Li J, Richards EM, Handberg EM, et al. Butyrate regulates COVID-19-relevant genes in gut epithelial organoids from normotensive rats. Hypertension. 2021;77:e13–e16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Vadyala SR, Betgeri SN, Sherer EA, et al. Prediction of the number of Covid-19 confirmed cases based on k-means-lstm. arXiv. 2006;10:14752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Adamidi ES, Mitsis K, Nikita KS. Artificial intelligence in clinical care amidst COVID-19 pandemic: a systematic review. Comput Struct Biotechnol J. 2021;19:2833–2850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Amritphale A, Chatterjee R, Chatterjee S, et al. Predictors of 30-day unplanned readmission after carotid artery stenting using artificial intelligence. Adv Ther. 2021;38:2954–2972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Jiang X, Eales JM, Scannali D, et al. Hypertension and renin-angiotensin system blockers are not associated with expression of angiotensin-converting enzyme 2 (ACE2) in the kidney. Eur Heart J. 2020;41:4580–4588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kim JH, Baek YH, Lee H, et al. Clinical outcomes of COVID-19 following the use of angiotensin-converting enzyme inhibitors or angiotensin-receptor blockers among patients with hypertension in Korea: a nationwide study. Epidemiol Health. 2021;43:e2021004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Meng J, Xiao G, Zhang J, et al. Renin-angiotensin system inhibitors improve the clinical outcomes of COVID-19 patients with hypertension. Emerg Microbes Infect. 2020;9:757–760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Semenzato L, Botton J, Drouin J, et al. Antihypertensive drugs and COVID-19 risk: a cohort study of 2 million hypertensive patients. Hypertension. 2021;77:833–842. [DOI] [PMC free article] [PubMed] [Google Scholar]


