Abstract
Background
Cerebrospinal fluid (CSF) lactate levels can be used to differentiate between bacterial and viral meningitis. We measured CSF lactate in individuals with cryptococcal meningitis to determine its clinical significance.
Methods
We measured point-of-care CSF lactate at the bedside of 319 Ugandan adults living with human immunodeficiency virus at diagnosis of cryptococcal meningitis. We summarized demographic variables and clinical characteristics by CSF lactate tertiles. We evaluated the association of CSF lactate with clinical characteristics and survival.
Results
Individuals with high CSF lactate >5 mmol/L at cryptococcal diagnosis more likely presented with altered mental status (P < .0001), seizures (P = .0005), elevated intracranial opening pressure (P = .03), higher CSF white cells (P = .007), and lower CSF glucose (P = .0003) compared with those with mid-range (3.1 to 5 mmol/L) or low (≤3 mmol/L) CSF lactate levels. Two-week mortality was higher among individuals with high baseline CSF lactate >5 mmol/L (35%; 38 of 109) compared with individuals with mid-range (22%; 25 of 112) or low CSF lactate (9%; 9 of 97; P =<.0001). After multivariate adjustment, CSF lactate >5 mmol/L remained independently associated with excess mortality (adjusted hazard ratio = 3.41; 95% confidence interval, 1.55–7.51; P = .002). We found no correlation between baseline CSF lactate levels and blood capillary lactate levels.
Conclusions
Baseline point-of-care CSF lactate levels are a prognostic marker of disease severity and mortality in cryptococcal meningitis. Individuals with an elevated baseline CSF lactate level are more likely to present with altered mental status, seizures, and elevated CSF opening pressure and are at a greater risk of death. Future studies are needed to determine targeted therapeutic management strategies in persons with high CSF lactate.
Keywords: cryptococcal meningitis, cerebrospinal fluid, lactic acid, prognostic marker, mortality
In human immunodeficiency virus–associated cryptococcal meningitis, baseline cerebrospinal fluid (CSF) lactate levels are associated with increased intracranial pressures, seizures, and altered mental status. Elevated CSF lactate levels at baseline are associated with increased 2-week mortality.
The measurement of cerebrospinal fluid (CSF) lactate has been shown to have utility in the rapid differentiation of bacterial meningitis from viral meningitis [1, 2]. In cases of acute meningitis, CSF lactate concentrations that are >4.2 mmol/L have a sensitivity of 96% in the presumptive diagnosis of bacterial meningitis [1]. However, despite the high sensitivity, CSF lactate measurements are highly nonspecific and are also found to be elevated in several other infectious and noninfectious neurological conditions, including cerebral malaria, tuberculous meningitis, cryptococcal meningitis, cerebral injury, subarachnoid hemorrhage, seizures, and ischemia [3–9]. As a diagnostic tool, the current recommended clinical application to use CSF lactate, as dictated by the Infectious Diseases Society of America guidelines, is in the diagnostic workup of bacterial meningitis after postoperative neurosurgical procedures [5, 10].
As a prognostic marker in cases of bacterial meningitis, individuals with higher CSF lactate levels demonstrate a trend toward a lower Glasgow coma score and higher mortality [1, 2]. In cases of bacterial meningitis, serial measurements of CSF lactate predict future clinical outcomes. Specifically, a >50% decline in CSF lactate levels in the first 3 days of treatment resulted in an improved clinical course [1]. A similar trend has been reported in cerebral malaria and in cases of severe head injury, where the normalization of CSF lactate has favorable clinical outcomes, while persistently elevated or increasing CSF lactate levels result in overall poor clinical outcomes, including severe disability or death [3, 5].
In sub-Saharan Africa, human immunodeficiency virus (HIV)–associated cryptococcal meningitis is the most common cause of meningitis, accounting for 15% of AIDS-related deaths globally [11–13]. In low-income countries where the burden of cryptococcal meningitis is greatest, 2-week mortality has ranged from 17% to 28% in clinical trials, with 1-year mortality as high as 70% in routine care [11]. In HIV-associated cryptococcal meningitis, CSF lactate levels have been found to be elevated (median, 3.0 mmol/L; interquartile range [IQR], 2.7–5.8) at the time of diagnosis [4]. However, the significance of elevated CSF lactate levels in cryptococcal meningitis is not well understood. We sought to understand the prognostic significance of elevated baseline CSF lactate levels in HIV-associated cryptococcal meningitis.
METHODS
Study Design and Participants
The study included Ugandan adults who presented with first-episode cryptococcal meningitis at Kiruddu General Hospital in Kampala and Mbarara Regional Referral Hospital from September 2016 to March 2020. Persons diagnosed with cryptococcal meningitis were enrolled into the Adjunctive Sertraline for the Treatment of HIV-Associated Cryptococcal Meningitis (ASTRO-CM) clinical trial (ClinicalTrials.gov identifier: NCT01802385) through May 2017. After conclusion of ASTRO-CM, participants were enrolled in an observational cohort focused on meningitis diagnostics [14–16]. All participants enrolled into the studies provided written informed consent. Participants were treated with standard antifungal therapy; those who were enrolled into ASTRO-CM were randomized to receive either adjunctive sertraline (400 mg/day for 14 days, followed by 200 mg/day for 12 weeks) or placebo. Standard antifungal therapy was amphotericin B (0.7–1.0 mg/kg/day) for up to 14 days and fluconazole 800 mg/day for approximately 4 weeks, followed by fluconazole 400 mg/day for 8 weeks of consolidation therapy, and fluconazole 200 mg/day for secondary prophylaxis. Participants in the randomized trial were followed up for 18 weeks, while participants in the observational study were followed up for 2 weeks or until hospital discharge.
Lactate Measurements
All participants had a baseline lumbar puncture performed at the time of cryptococcal meningitis diagnosis. CSF lactate measurements were performed bedside at the time of lumbar puncture (Lactate Plus Analyzer; Sports Resource Group Inc, Hawthorne, NY). In addition to CSF lactate measurements, a subset of individuals also had matched serum lactate measurements performed concurrently at baseline.
Statistical Methods
CSF lactate measurements were grouped into tertiles. We summarized baseline demographic variables and clinical characteristics by CSF lactate tertiles, presented as percentages and medians with IQR. We evaluated the relationship between CSF lactate level and 2-week mortality using log-rank tests and visually summarized by Kaplan-Meier curves. We used Cox proportional hazards models to estimate hazard ratios by CSF lactate level, adjusted for Glasgow coma score <15, baseline seizures, quantitative Cryptococcus CSF culture, and CSF opening pressure.
We conducted an area under the receiver operating characteristic (AUC-ROC) curve analysis of the continuous log2 transformed CSF lactate as a predictor of 2-week mortality to consider a threshold cut-point of lactate risk for mortality. As sensitivity analyses, we used Cox proportional hazards models to consider lactate cut at the threshold from the AUC result and as a continuous variable. We compared clinical characteristics of individuals who died with those who survived at 2 weeks using the Cox proportional hazards model. We used the Spearman correlation to estimate the correlation between CSF lactate and serum lactate. We performed analyses using SAS version 9.4 (SAS Institute, Cary, NC).
RESULTS
Of 622 participants with a first episode of cryptococcal meningitis, 51% (319 of 622) had a CSF lactate measurement performed at baseline. Demographic and clinical characteristics of participants with a CSF lactate measurement are summarized by CSF lactate tertiles in Table 1. CSF lactate measurements (overall median, 3.9; IQR, 2.8–5.7; range, 1.1–12.0) were grouped into low (≤3 mmol/L, n = 97), mid-range (3.1–5.0 mmol/L, n = 113), and high (>5.0 mmol/L, n = 109) tertiles, with distribution displayed in Figure 1.
Table 1.
Baseline Characteristics of Study Participants by Cerebrospinal Fluid Lactate Tertile
| Characteristic | Number With Data | CSF Lactate Tertile | P Value | ||
|---|---|---|---|---|---|
| Low ≤3.0 | Mid-Range 3.1–5.0 | High >5.0 | |||
| Number | 319 | 97 | 113 | 109 | |
| CSF lactate, mmol/L | 319 | 2.4 (2.1–2.7) | 3.8 (3.4–4.4) | 6.7 (5.6–8.1) | |
| Men | 319 | 51 (52.6%) | 75 (66.4%) | 79 (72.5%) | .01 |
| Age, years | 319 | 34 (29–40) | 35 (30–42) | 38 (31–45) | .02 |
| Glasgow coma score <15 | 314 | 30 (31.3%) | 39 (35.5%) | 71 (65.7%) | <.0001 |
| Seizure | 314 | 9 (9.4%) | 17 (15.5%) | 32 (29.6%) | .0006 |
| On human immunodeficiency virus therapy | 317 | 50 (52.1%) | 51 (45.1%) | 53 (49.1%) | .60 |
| Hemoglobin, g/dL | 239 | 11 (10–13) | 12 (10–14) | 11 (9–13) | .29 |
| CD4 cells/μL | 225 | 14 (7–38) | 15 (6–53) | 25 (8–58) | .18 |
| Creatinine, mg/dL | 236 | 0.7 (0.6–0.9) | 0.7 (0.6–0.9) | 0.8 (0.6–1.0) | .28 |
| CSF white cells/μL | 313 | <5 (<5–20) | <5 (<5–30) | <5 (<5–130) | .007 |
| CSF protein, mg/dL | 270 | 50 (24–102) | 59 (29–100) | 65 (28–122) | .25 |
| CSF glucose, mg/dL | 240 | 62 (42–86) | 59 (40–72) | 38 (22–68) | .0003 |
| CSF opening pressure, cm H2O | 280 | 22 (14–30) | 23 (16–33) | 27 (17–40) | .03 |
| CSF culture, log10 colony-forming units/mL | 306 | 4.0 (1.7–5.3) | 4.6 (2.5–5.6) | 4.6 (2.8–5.4) | .25 |
| CSF removed, mL | 290 | 10 (7–16) | 12 (9–18) | 12 (8–19) | .22 |
| Blood capillary lactate, mmol/L | 37 | 1.9 (1.9–5.3) | 2.9 (2.0–4.5) | 3.5 (1.8–5.0) | .72 |
Data are presented as median (interquartile range) or N (%). P value determined using the Kruskal-Wallis test for medians and the χ2 test for proportions.
Abbreviation: CSF, cerebrospinal fluid.
Figure 1.
Distribution of baseline CSF lactate measurements by 2-week survival. Distribution of baseline CSF lactate among persons who presented with cryptococcal meningitis. Persons who presented with a CSF lactate >5.0 mmol/L or greater were at a greater than 3 times the risk of mortality at 2 weeks (P < .0001). Abbreviation: CSF, cerebrospinal fluid.
Among persons diagnosed with HIV-associated cryptococcal meningitis, we found several demographic and clinical factors to be associated with elevated CSF lactate levels. Participants with high CSF lactate, >5.0 mmol/L, had significantly higher baseline CSF white cells (P = .007) and lower CSF glucose (P = .0003) than participants with low or mid-range CSF lactate at baseline. There were no differences in CSF quantitative Cryptococcus culture (P = .25) or CSF protein (P = .25) between lactate tertiles. However, participants with high baseline CSF lactate also had higher intracranial opening pressure (median, 27 cm H2O; IQR, 17–40) than participants with mid-range (median, 23 cm H2O; IQR, 16–33) or low baseline CSF lactate levels (median, 22 cm H2O; IQR, 14–30; P = .03). A significantly greater percentage of participants with high baseline CSF lactate presented with a Glasgow coma score <15 (65.7%) compared with participants with mid-range (35.5%) or low CSF lactate (31.3%; P < .0001). Baseline seizures were also more frequent in participants who presented with high baseline CSF lactate at the time of cryptococcal meningitis diagnosis (high 30% vs mid-range 16% and low 9%; P = .0006).
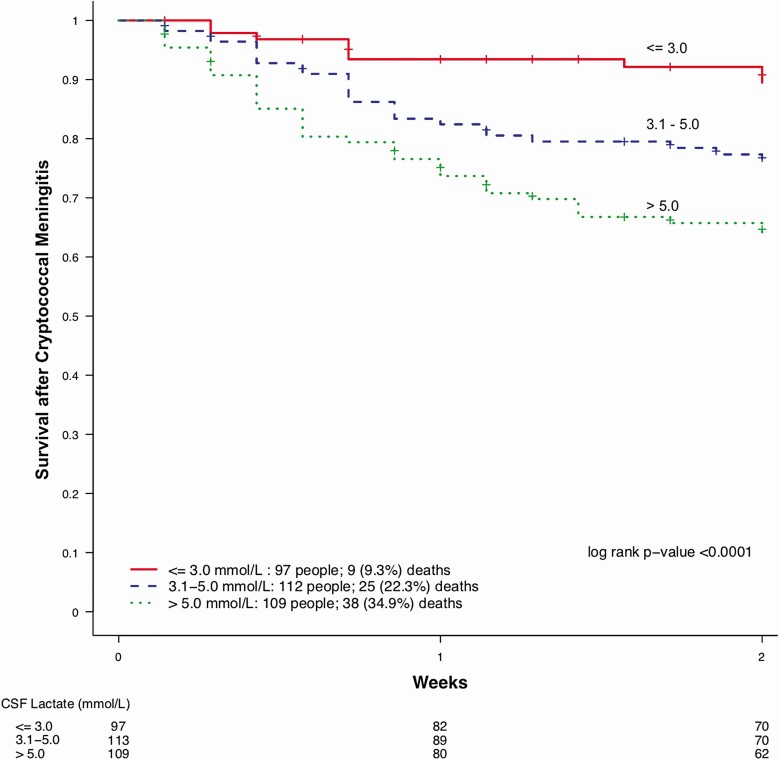
Two-week mortality was significantly higher among individuals with high baseline CSF lactate compared with individuals with mid-range and low CSF lactate levels (high 35% [38 of 109] vs mid-range 22% [25 of 112] and low 9% [9 of 97]; P < .0001; Figure 2). In multivariate analysis, after adjusting for Glasgow coma score, baseline seizures, baseline opening pressure, and quantitative Cryptococcus CSF culture, individuals with high baseline CSF lactate continued to be at a 3-fold higher risk of mortality at 2 weeks compared with those with CSF lactate ≤3.0 mmol/L (adjusted hazard ratio [aHR] = 3.41; 95% confidence interval [CI], 1.55–7.51; P = .0021; Table 2). The increased risk of mortality with higher lactate levels was also seen in sensitivity analysis when baseline CSF lactate was considered as a continuous variable (aHR = 1.13 for each 1 mmol/L increase in baseline CSF lactate; 95% CI, 1.02–1.24). An area under a ROC curve analysis found that a CSF lactate cutoff point of 4.3 mmol/L had the highest sensitivity and specificity for predicting 2-week mortality (AUC, 0.664; 95% CI, .60–.73). In sensitivity analysis, those with baseline CSF lactate of ≥4.3 mmol/L had a 2-fold higher risk of mortality at 2 weeks compared with those with baseline CSF lactate <4.3 mmol/L (aHR = 2.10; 95% CI, 1.22–3.62).
Figure 2.
Mortality at 2 weeks by baseline CSF lactate tertiles. Abbreviation: CSF, cerebrospinal fluid.
Table 2.
Hazard Ratios for 2-Week Mortality
| CSF Lactate, mmol/L | Number of Deaths (%) | Hazard Ratio (95% CI) | P Value | Adjusteda Hazard Ratio (95% CI) | P Value |
|---|---|---|---|---|---|
| By CSF lactate tertiles | |||||
| ≤3.0 | 9 (9) | 1.0 | 1.0 | ||
| 3.1–5.0 | 25 (22) | 2.51 (1.17–5.38) | .02 | 1.99 (.87–4.52) | .10 |
| >5.0 | 38 (35) | 4.21 (2.04–8.71) | .0001 | 3.41 (1.55–7.51) | .002 |
| By 1 mmol/L increase in CSF lactate | |||||
| 1 unit increase | 1.16 (1.07–1.27) | .0005 | 1.13 (1.02–1.24) | .02 | |
| By area under the curve cutoff of CSF lactate | |||||
| <4.3 | 25 (14) | 1.0 | 1.0 | ||
| ≥4.3 | 47 (33) | 2.53 (1.56–4.12) | .0002 | 2.10 (1.22–3.62) | .008 |
Abbreviations: CI, confidence interval; CSF, cerebrospinal fluid.
aAdjusted for Glasgow coma scale score, baseline seizures, quantitative cryptococcal CSF culture, and CSF opening pressure.
We assessed for other possible confounders for association with 2-week mortality in those who presented with CSF lactate >5.0 mmol/L. There was a notable difference in the percentage of people with baseline Glasgow coma score <15 in those who died compared with those who survived (died 78% [29 of 38] vs survived 59% [42 of 71]; P = .05; Table 3). Among persons who died at 2 weeks, baseline hemoglobin was higher in those who survived (died, median 10.2 g/dL and IQR, 8.1–12.6 vs survived, median 11.6 g/dL and IQR, 9.7–13.1; P = .04). There was also a difference in baseline serum creatinine levels such that people who died at 2 weeks presented with higher baseline serum creatinine compared with those who survived (P = .001). There were no significant differences in baseline CSF opening pressure (P = .22), CSF glucose (P = .36), or quantitative Cryptococcus CSF culture (P = .24) in persons who survived compared with those who died at 2 weeks.
Table 3.
Baseline Characteristics of Patients With Cerebrospinal Fluid Lactate >5.0 mmol/L, Compared by 2-Week Survivor Status
| Characteristic | Died by 2 Weeks | Survived 2 Weeks | P Value |
|---|---|---|---|
| Number | 38 | 71 | |
| Men | 25 (66%) | 54 (76%) | .22 |
| Age, years | 40 (33–50) | 37 (30–42) | .06 |
| Glasgow coma score <15 | 29 (78%) | 42 (59%) | .05 |
| Seizure | 13 (35%) | 19 (27%) | .34 |
| On HIV therapy | 21 (55%) | 32 (46%) | .32 |
| Hemoglobin, g/dL | 10.2 (8.1–12.6) | 11.6 (9.7–13.1) | .04 |
| CD4 count, cells/μL | 20 (7–52) | 27 (10–70) | .33 |
| Creatinine, mg/dL | 0.8 (0.6–1.7) | 0.8 (0.6–0.9) | .001 |
| CSF lactate, mmol/L | 7.0 (5.8–7.7) | 6.7 (5.5–8.5) | .57 |
| CSF white cells, per μL | <5 (<5–130) | 5 (<5–130) | .64 |
| CSF glucose, mg/dL | 37 (20–76) | 39 (26–61) | .36 |
| CSF opening pressure, cm H2O | 27 (17–43) | 26 (18–37) | .22 |
| CSF culture, log10 colony-orming units/mL | 4.6 (2.6–5.5) | 4.6 (2.8–5.4) | .48 |
| CSF removed at baseline, mL | 13 (8–20) | 12 (8–18) | .63 |
Data are presented as median (interquartile range) or N (%). P values determined using Cox proportional hazards models.
Abbreviations: CSF, cerebrospinal fluid; HIV, human immunodeficiency virus.
Among persons with CSF lactate measurements at baseline, 37 had matched capillary blood lactate also measured. No correlation existed between CSF and capillary blood lactate levels (r2 = 0.02; P = .60; Supplementary Figure 1). Whole blood lactate levels did not differ among low, mid-range, and high CSF lactate tertiles (P = .72).
Discussion
In this study, we demonstrate that a high baseline CSF lactate level is a reliable prognostic marker of disease severity in HIV-associated cryptococcal meningitis. Persons with HIV-associated cryptococcal meningitis who have elevated CSF lactate levels >5.0 mmol/L are also more likely to present with altered mental status (Glasgow coma score <15) and seizures at diagnosis. An elevated CSF lactate level >5.0 mmol/L is also accompanied by elevated intracranial pressure, higher CSF white cells, and lower CSF glucose. In contrast, CSF lactate levels are not associated with the burden of Cryptococcus based on a lack of association with quantitative CSF cultures. Persons with a baseline CSF lactate level >5.0 mmol/L were at a 3-fold increased risk of death at 2 weeks compared with those with levels ≤3.0 mmol/L. Even after controlling for Glasgow coma score, opening pressure, and CSF quantitative Cryptococcus cultures, high CSF lactate (>5.0 mmol/L) was found to be an independent predictor for increased 2-week mortality. Assessing for clinical risk factors among persons with a CSF lactate level >5.0 mmol/L who died vs those who survived at 2 weeks, persons who died were more likely to also present with a lower hemoglobin value and higher creatinine levels at baseline.
We did not find a correlation between CSF and capillary blood lactate measurements in cryptococcal meningitis [17]. In HIV-associated cryptococcal meningitis, the lack of correlation between CSF and capillary lactate levels supports a centrally occurring disease process that is not systemic and therefore requires compartment-specific interventions.
The exact mechanism behind CSF lactate elevations in cryptococcal meningitis and its association with mortality are unclear. We postulate that in HIV-associated cryptococcal meningitis, decreases in cerebral perfusion and cerebral hypoxia lead to the clinical presentation of altered mental status, seizures, and increased intracranial pressure at the time of presentation. The observed CSF profile, increase in white cell count, low glucose, and high lactate would support an inflammatory central nervous system (CNS) process that may be a result of or contribute to decreasing cerebral perfusion and cerebral hypoxia. Furthermore, when 2-week survival was compared among persons with cryptococcal meningitis who presented with a baseline CSF lactate >5.0 mmol/L, individuals who died had lower baseline hemoglobin levels. Our results would support the assumption that mortality may be driven by decreased cerebral perfusion and possibly cerebral ischemia.
In cryptococcal meningitis, percent regional cerebral tissue oxygenation (rSO2), as measured by cerebral oximetry, has been shown to be a predictor of 2-week mortality [18], such that individuals with low cerebral perfusion are more than 3 times more likely to die within 30 days. Similar to CSF lactate, cerebral oximetry is independently associated with mortality after having controlled for altered mental status, CSF quantitative Cryptococcus culture, CSF white cell count, and anemia. Low hemoglobin levels decrease oxygen-carrying capacity and are strongly correlated with measured regional cerebral tissue oxygenation and mortality. In fact, in previous cryptococcal meningitis cohorts, low hemoglobin levels have repeatedly been shown to be associated with increased mortality [19–21].
CSF lactate and glucose have long been recognized as highly variable in cryptococcal meningitis. The brain’s high energy demand creates a state of dependence on aerobic metabolism and a continuously sufficient supply of oxygen and glucose for cellular metabolism. A lack of adequate cerebral perfusion, by either a reduction in the oxygen-carrying capacity or cerebral ischemia secondary to increased intracranial pressure or seizures, may explain the rise in CSF lactate. The oxidation of pyruvate and gluconeogenesis require the presence of oxygen. When oxygen levels fall, glucose is primarily converted to lactate. Thus, decreased cerebral perfusion that lead to an increased risk for mortality would be supported by our observation of low CSF glucose levels in the presence of elevated CSF lactate.
High CSF lactate is independently associated with mortality, after controlling for increased intracranial pressure, altered mental status, and CSF quantitative Cryptococcus culture. This raises the possibility of irreversible CNS damage. In neurocritical care settings, cerebral microdialysis measurements of the lactate and pyruvate ratio help to distinguish between insufficient tissue oxygenation, which may be reversible, and mitochondrial damage, which may lead to neural cell death [22]. Further research is needed to understand the underlying pathophysiology of elevated CSF lactate levels in individuals with cryptococcal meningitis and to evaluate specific interventions targeted at improving mortality. At present, Our data would suggest that in persons with CSF lactate >5.0 mmol/L, interventions aimed at improving hemodynamics by addressing critical hemoglobin levels, controlling increased intracranial pressure, optimizing cerebral perfusion and oxygen delivery to the brain, as well as early detection and treatment of seizures are important aspects of supportive management in cryptococcal meningitis.
While our study is the largest thus far to evaluate CSF lactate levels in cryptococcal meningitis, we were limited in our ability to longitudinally evaluate the effects of CSF lactate on outcomes. Also, our sample size limited our ability to focus on risk factors in individuals with high baseline CSF lactate levels >5.0 mmol/L. Despite these limitations, we believe that our study has important clinical implications in the management of cryptococcal meningitis.
In conclusion, results of our study demonstrate that in cryptococcal meningitis, baseline CSF lactate levels may be used as a prognostic marker for disease severity and as a clinical tool to risk-stratify individuals who are at an increased risk of death. Our findings suggest that in individuals with cryptococcal meningitis, baseline CSF lactate levels >5.0 mmol/L should be used to identify those at increased risk of early, 2-week mortality.
Supplementary Data
Supplementary materials are available at Clinical Infectious Diseases online. Consisting of data provided by the authors to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the authors, so questions or comments should be addressed to the corresponding author.
Notes
Financial support. This work was supported by the US Fogarty International Center (K01TW010268, R25TW009345), National Institute of Neurologic Diseases and Stroke (R01NS086312), National Institute of Allergy and Infectious Diseases (T32AI055433), and the UK Medical Research Council/Wellcome Trust/Department for International Development (MRC MR/M007413/1). D. B. M. was also supported by DELTAS Africa Initiative (grant DEL-15–011) to THRiVE-2. The DELTAS Africa Initiative is an independent funding scheme of the African Academy of Sciences Alliance for Accelerating Excellence in Science in Africa and supported by the New Partnership for Africa’s Development Planning and Coordinating Agency with funding from the Wellcome Trust (grant 107742/Z/15/Z) and the UK government.
Potential conflicts of interest. All authors: No reported conflicts of interest. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1. Genton B, Berger JP. Cerebrospinal fluid lactate in 78 cases of adult meningitis. Intensive Care Med 1990; 16:196–200. [DOI] [PubMed] [Google Scholar]
- 2. Giulieri S, Chapuis-Taillard C, Jaton K, et al. CSF lactate for accurate diagnosis of community-acquired bacterial meningitis. Eur J Clin Microbiol Infect Dis 2015; 34:2049–55. [DOI] [PubMed] [Google Scholar]
- 3. White NJ, Warrell DA, Looareesuwan S, Chanthavanich P, Phillips RE, Pongpaew P. Pathophysiological and prognostic significance of cerebrospinal-fluid lactate in cerebral malaria. Lancet 1985; 1:776–8. [DOI] [PubMed] [Google Scholar]
- 4. de Almeida SM, Boritza K, Cogo LL, et al. Quantification of cerebrospinal fluid lactic acid in the differential diagnosis between HIV chronic meningitis and opportunistic meningitis. Clin Chem Lab Med 2011; 49:891–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Leib SL, Boscacci R, Gratzl O, Zimmerli W. Predictive value of cerebrospinal fluid (CSF) lactate level versus CSF/blood glucose ratio for the diagnosis of bacterial meningitis following neurosurgery. Clin Infect Dis 1999; 29:69–74. [DOI] [PubMed] [Google Scholar]
- 6. Jordan GW, Statland B, Halsted C. CSF lactate in diseases of the CNS. Arch Intern Med 1983; 143:85–7. [PubMed] [Google Scholar]
- 7. Froman C, Smith AC. Metabolic acidosis of the cerebrospinal fluid associated with subarachnoid haemorrhage. Lancet 1967; 1:965–7. [DOI] [PubMed] [Google Scholar]
- 8. Lu CH, Chang WN, Chang HW, Chuang YC. The prognostic factors of cryptococcal meningitis in HIV-negative patients. J Hosp Infect 1999; 42:313–20. [DOI] [PubMed] [Google Scholar]
- 9. Tsai WC, Lien CY, Lee JJ, et al. The clinical characteristics of adult cryptococcal meningitis patients who died within one year of treatment with a focus on those with early mortality. J Clin Neurosci 2019; 67:80–4. [DOI] [PubMed] [Google Scholar]
- 10. Maskin LP, Capparelli F, Mora A, et al. Cerebrospinal fluid lactate in post-neurosurgical bacterial meningitis diagnosis. Clin Neurol Neurosurg 2013; 115:1820–5. [DOI] [PubMed] [Google Scholar]
- 11. Rajasingham R, Smith RM, Park BJ, et al. Global burden of disease of HIV-associated cryptococcal meningitis: an updated analysis. Lancet Infect Dis 2017; 17:873–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Ellis J, Bangdiwala AS, Cresswell FV, et al. The changing epidemiology of HIV-associated adult meningitis, Uganda 2015–2017. Open Forum Infect Dis 2019; 6:ofz419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Durski KN, Kuntz KM, Yasukawa K, Virnig BA, Meya DB, Boulware DR. Cost-effective diagnostic checklists for meningitis in resource-limited settings. J Acquir Immune Defic Syndr 2013; 63:e101–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Rhein J, Huppler Hullsiek K, Tugume L, et al. ; ASTRO-CM Team . Adjunctive sertraline for HIV-associated cryptococcal meningitis: a randomised, placebo-controlled, double-blind phase 3 trial. Lancet Infect Dis 2019; 19:843–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Cresswell FV, Tugume L, Bahr NC, et al. ; ASTRO-CM Team . Xpert MTB/RIF Ultra for the diagnosis of HIV-associated tuberculous meningitis: a prospective validation study. Lancet Infect Dis 2020; 20:308–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Cresswell FV, Ellis J, Kagimu E, et al. Standardized urine-based tuberculosis (TB) screening with TB-lipoarabinomannan and Xpert MTB/RIF Ultra in Ugandan adults with advanced human immunodeficiency virus disease and suspected meningitis. Open Forum Infect Dis 2020; 7:ofaa100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Posner JB, Plum F. Independence of blood and cerebrospinal fluid lactate. Arch Neurol 1967; 16:492–6. [DOI] [PubMed] [Google Scholar]
- 18. Diehl JW, Hullsiek KH, Okirwoth M, et al. Cerebral oximetry for detecting high-mortality risk patients with cryptococcal meningitis. Open Forum Infect Dis 2018; 5:ofy105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Tugume L, Morawski BM, Abassi M, et al. Prognostic implications of baseline anaemia and changes in haemoglobin concentrations with amphotericin B therapy for cryptococcal meningitis. HIV Med 2017; 18:13–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Jarvis JN, Bicanic T, Loyse A, et al. Determinants of mortality in a combined cohort of 501 patients with HIV-associated cryptococcal meningitis: implications for improving outcomes. Clin Infect Dis 2014; 58:736–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Bicanic T, Bottomley C, Loyse A, et al. Toxicity of amphotericin B deoxycholate-based induction therapy in patients with HIV-associated cryptococcal meningitis. Antimicrob Agents Chemother 2015; 59:7224–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Dienel GA, Rothman DL, Nordström CH. Microdialysate concentration changes do not provide sufficient information to evaluate metabolic effects of lactate supplementation in brain-injured patients. J Cereb Blood Flow Metab 2016; 36:1844–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.